Abstract
We observed multiple fatal intracranial hemorrhages shortly after initiating therapeutic anticoagulation for treatment of venous thromboembolism (VTE) in COVID-19 patients suggesting increased anticoagulation risk associated with COVID-19. The objective of this study is to quantify risk of major hemorrhage in hospitalized COVID-19 patients on therapeutic anticoagulation for deep venous thrombosis (DVT) or pulmonary embolism (PE). Hospitalized patients with COVID-19 receiving therapeutic anticoagulation for DVT, PE or both at four New York City hospitals were evaluated for hemorrhagic complications. These were categorized as major (including fatal) or clinically relevant non-major according to the criteria of the International Society of Thrombosis and Haemostasis. Hemorrhagic complications were correlated with clinical and laboratory data, ICD-10 code diagnoses and type of anticoagulation treatment. Minor hemorrhages were excluded. Major/clinically relevant hemorrhages occurred in 36 of 170 (21%) hospitalized COVID-19 patients being treated with therapeutic anticoagulation for VTE including 4 (2.4%) fatal hemorrhages. Hemorrhage was 3.4 times more likely with unfractionated heparin 27/76 (36%) compared to 8/81 (10%) with low molecular weight heparin (p = 0.002). Multivariate analysis showed that major hemorrhage was associated with intubation (p = 0.04) and elevated serum LDH (p < 0.001) and low fibrinogen (p = 0.05). Increased risk of hemorrhagic complications in treating VTE in hospitalized COVID-19 patients should be considered especially when using unfractionated heparin, in intubated patients, with low fibrinogen and/or elevated LDH. Checking serum fibrinogen and LDH before initiating therapeutic anticoagulation and monitoring coagulation parameters frequently may reduce bleeding complications.
Keywords: Complication, COVID-19, Deep venous thrombosis, Pulmonary embolism, Therapeutic anticoagulation
Highlights
Risk of hemorrhage from therapeutic anticoagulation in COVID-19 is unknown.
We identified hemorrhages in COVID-19 patients treated for venous thrombo-embolic disease.
Hemorrhages occurred in 36 of 170 (21%) COVID-19 patients on therapeutic anticoagulation including 4 deaths (2.4%).
Hemorrhage was 3.4 times more likely when treated with unfractionated compared to low molecular weight heparin and also more likely with low fibrinogen (p = 0.05) and elevated LDH (p < 0.001).
Treating VTE in COVID-19 patients with therapeutic anticoagulation should be carefully considered given the increased risk in using unfractionated heparin, as well as treating patients who are intubated, with low serum fibrinogen, or with high serum lactate dehydrogenase levels.
Introduction
Venous thromboembolism (VTE) in patients hospitalized with Coronavirus-19 Disease (COVID-19) is primarily treated with therapeutic anticoagulation presuming similar anticoagulation risks as for other hospitalized patients with VTE. However, several reports have suggested that the complication rates for anticoagulation may be increased in COVID-19 [1–4]. In addition, we observed several fatal intracranial hemorrhages (ICH) shortly after initiating therapeutic anticoagulation for treatment of VTE further supporting a hypothesis of increased anticoagulation risk in COVID-19. The purpose of this study was to quantify risk of clinically relevant and major hemorrhage in hospitalized COVID-19 patients on therapeutic anticoagulation for deep venous thrombosis (DVT) or pulmonary embolism (PE) and to identify associations that might help with risk mitigation.
Methods
Patients diagnosed with COVID-19, hospitalized from March 3 to June 5, 2020 and receiving therapeutic anticoagulation for DVT, PE, or both at four New York City hospitals were evaluated for hemorrhagic complications. To meet the inclusion criteria, patients had to (1) be ≥ 18 years-old, (2) test positive for SARS-CoV-2 on reverse transcription polymerase chain reaction (RT-PCR) test via nasopharyngeal swab, and (3) diagnosed with DVT, PE, or both using lower extremity US and/or Chest CT pulmonary angiography (CTPA) and (4) be treated with therapeutic anticoagulation during their COVID hospitalization.
Electronic medical records were searched to identify patients meeting inclusion criteria and their demographic, clinical, and laboratory data on the date of VTE diagnosis were recorded. ICD-10 diagnoses and details of anticoagulation, antiplatelet agents, placement of an inferior vena cava (IVC) filter were also recorded. Patient encounter notes were manually reviewed by three board certified physicians to identify which patients had hemorrhagic complications after receiving anticoagulation.
Hemorrhagic complications were categorized as major (including fatal) or clinically relevant non-major according to the criteria of the International Society of Thrombosis and Haemostasis [5]. Minor hemorrhages were excluded.
Laboratory and clinical parameters in patients without compared to with hemorrhagic complications were compared with Students t-test for continuous variables if normally distributed or Mann–Whitney if not normally distributed, and Chi Square or Fisher’s Exact for categorical variables. A multivariate regression model was created incorporating age, sex and all variables found to be significant on univariate analysis that had data for at least 90% of subjects. A second multivariate model was created also incorporating variables found to be significant on univariate analysis but where data was available in less than 90% of subjects.
Results
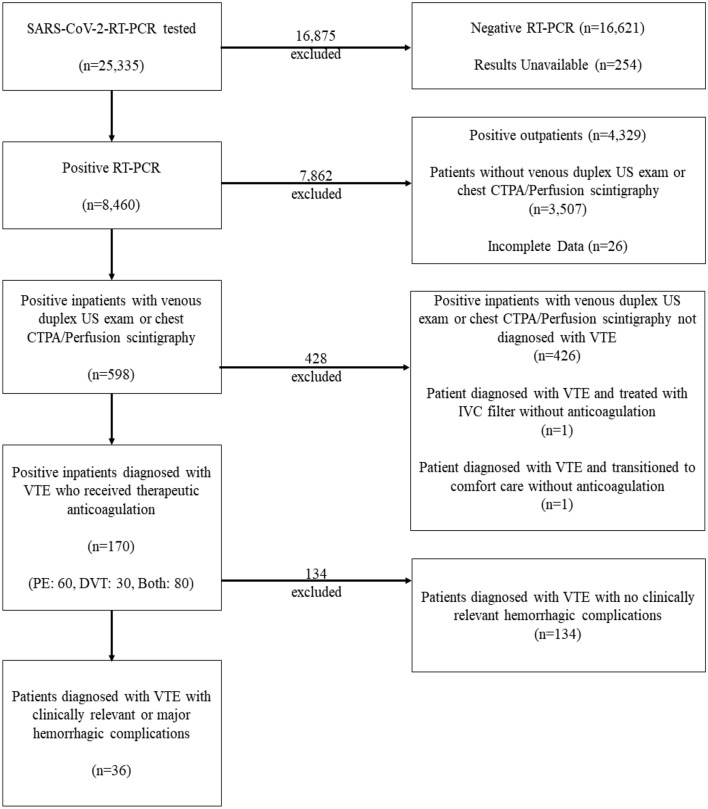
Among the SARS-CoV-2 RT-PCR tests performed from March 3 to June 5, 2020 (n = 25,335) at four hospitals, 8460 cases were positive, Fig. 1. From this cohort, 4131 were hospitalized with COVID-19 of which 172 were diagnosed with PE (n = 60), DVT (n = 31), or both (n = 81). One COVID-19 patient, 81 years old with cognitive impairment, multiple falls and a subdural hematoma, was diagnosed with VTE but treated with IVC filter and an antiplatelet agent, but without therapeutic anticoagulation, was excluded. A second excluded patient, 86 years old, was diagnosed with PE on the day of admission but had an acute on chronic left frontal subdural hematoma and a probable chronic right frontal subdural hematoma. He was transitioned to comfort care and died the next day without receiving anticoagulation or IVC filter. This resulted in 170 patients meeting the inclusion criteria.
Fig. 1.
Patient flowchart
The anticoagulation medications initiated for VTE treatment, including doses indexed to patient weight, are listed in Table 2. The hospital duration of anticoagulation ranged from 1 to 127 (mean = 28) days. Data was not available for the extent that anticoagulation was continued after discharge.
Table 2.
Hospital days on anticoagulation treatment and dose for COVID-19 patients with and without hemorrhagic complications
*Corresponds to the dose at the time of hemorrhage
Hemorrhagic complications following anticoagulation were reported in 36 of the 170 patients (21%), see Table 1. Major hemorrhages (n = 24, 14%) included 4 (2.4%) fatal hemorrhages. Three additional patients with altered mental status after initiating therapeutic anticoagulation were possible additional fatal intracranial hemorrhages but those patients were transitioned to comfort care and died without performing head CT. In this analysis they were not counted as hemorrhagic complications.
Table 1.
Risk factors for hemorrhagic complications in COVID-19
| Hemorrhagic complications | p value | ||||
|---|---|---|---|---|---|
| Present (n = 36) | Absent (n = 134) | Uni-variate | Multi-variate model 1, n = 157 w/o fibrinogen |
Multi-variate model 2, n = 118 w/fibrinogen |
|
| Age | 61 ± 14 | 63 ± 15 | 0.33 | 0.18 | 0.29 |
| Male:female (ratio) | 26:10 (2.6) | 88:46 (1.9) | 0.46 | 108:49 (2.2) | 49:39 (1.26) |
| Body mass index | 30 ± 8 | 28 ± 6 | 0.21 | ||
| History of hypercoagulability | 2 (6%) | 22 (16%) | 0.1 | 0.07 | 0.22 |
| Acute respiratory distress syndrome | 27 (75%) | 41 (31%) | < 0.001 | 0.14 | 0.07 |
| Acute kidney injury | 18 (50%) | 52 (39%) | 0.13 | ||
| Intensive care unit | 29 (81%) | 60 (45%) | < 0.001 | 0.72 | 0.69 |
| Intubated | 27 (75%) | 41 (31%) | < 0.001 | 0.04 | 0.16 |
| Initial treatment | |||||
| Unfractionated heparin | 27 (75%) | 49 (37%) | < 0.001 | 0.04** | 0.3** |
| Low molecular weight heparin | 8 (22%) | 73 (54%) | < 0.001 | ||
| Oral anticoagulation | 1 (3%) | 12 (9%) | 0.37 | ||
| Comfort care | 0 | 1**** | 1 | ||
| IVC filter only | 0 | 1**** | 1 | ||
| Combined treatment | |||||
| IVC filter + anticoagulation | 10 (28%)* | 8 (6%) | |||
| Anticoagulation + antiplatelet agent | 19 (53%) | 44 (33%) | 0.05 | 0.23 | 0.12 |
| Anticoagulation w/o antiplatelet agent | 17 (47%) | 90 (67%) | |||
| Laboratory (on VTE diagnosis date) | |||||
| Creatinine (mg/dL) | 2.2 ± 2.7 | 1.3 ± 1.4 | 0.009 | 0.98 | 0.77 |
| AST (U/L) | 269 ± 1002 | 49 ± 38 | 0.01 | 0.28 | 0.35 |
| ALT (U/L) | 230 ± 886 | 69 ± 134 | 0.08 | ||
| Albumin (g/dL) | 2.3 ± 0.9 | 2.2 ± 1.4 | 0.78 | ||
| Lactate dehydrogenase (U/L) | 871 ± 946 | 356 ± 279 | < 0.001 | < 0.001 | 0.005 |
| Troponin I (ng/mL) | 0.61 ± 1.25 | 0.39 ± 0.93 | 0.43 | ||
| Lymphocytes (× 103/μL) | 9.3 ± 26.6 | 4.6 ± 8.9 | 0.09 | ||
| D-dimer (ng/mL) | 10,606 ± 16,348 | 6390 ± 10,561 | 0.07 | ||
| Fibrinogen (mg/dL)*** | 473 ± 273 | 1028 ± 1325 | 0.02 | 0.05 | |
| Platelet count (/μL) | 268 ± 134 | 310 ± 148 | 0.13 | ||
| PT (s) | 16 ± 5 | 15 ± 4 | 0.13 | ||
| aPTT (just before hemorrhage or 2 days after AC initiation) (s) | 73 ± 40 | 50 ± 32 | < 0.001 | 0.25 | 0.20 |
P ≤ 0.05 are shown in bold
*All inferior vena cava (IVC) filters in patients with hemorrhage were placed after the hemorrhage
**Unfractionated heparin compared to all other agents
***Missing data for 52 (31%) patients, therefore excluded from multivariate analysis model 1
****These patients with VTE but not on anticoagulation were not included in the total
The time between starting therapeutic anticoagulation and documented hemorrhagic complications ranged from 1 to 60 days (mean = 14 days) with a shorter time interval to the intracranial hemorrhages (9 ± 10 days) and fatal hemorrhages (mean = 4 days, p = 0.02 compared to nonfatal hemorrhages).
Univariate analysis (Table 1) revealed that COVID-19 patients with hemorrhagic complications had more severe disease, including higher rates of acute respiratory distress syndrome, intubation, intensive care unit admission, elevated LDH, AST, fibrinogen and serum creatinine. A history of hypercoagulability (based upon ICD-10 coding) showed a trend toward being protective with only 2 of 36 (6%) with hypercoagulability having a hemorrhagic complication compared to 22 of 134 (16%, p = 0.1) hemorrhagic complications in those without hypercoagulability.
Antiplatelet agents, including aspirin (n = 58) and clopidogrel (n = 5), were used in addition to anticoagulation in 63 of the 170 (37%) COVID-19 patients being treated for VTE. Univariate analysis showed that hemorrhagic complications were more common in those taking antiplatelet agents in addition to anticoagulation (p = 0.05). However, this was not confirmed with either multivariate model.
Most patients were treated with either unfractionated heparin (UFH) (n = 76, 45%) or low molecular weight heparin (LMWH) (n = 81, 48%). Hemorrhage was 3.4 times more likely with UFH 27/76 (36%) compared to 8/81 (10%) with LMWH (p < 0.001). All fatal intracranial hemorrhages occurred following treatment with UFH and all had supratherapeutic activated partial thromboplastin time (aPTT) at the time of fatal intracranial hemorrhage. This higher risk of hemorrhage on UFH was confirmed on a multivariate analysis (Table 2—model 1) based upon age, sex, history of hypercoagulability and all the parameters found to be significant on univariate analysis (ARDS, ICU admission, intubation, antiplatelet agent supplementing anticoagulation, serum creatinine, AST, LDH, aPTT) except fibrinogen which was not available in 52 (31%) patients.
Serum fibrinogen was evaluated in a separate multivariate analysis, model 2, with fewer patients (n = 118) due to missing fibrinogen data on > 10% of patients, but still including all the parameters of model 1. Model 1 maximized statistical power with an overall R = 0.61 (p < 0.00001) while model 2 including fibrinogen had an overall R = 0.66 (p < 0.00001). LDH was highly significantly correlated with hemorrhagic complications for both models (p < 0.001, < 0.005). Serum fibrinogen was significantly inversely associated with hemorrhage in the model in which it was included (p = 0.05). Use of unfractionated heparin and intubation were significantly associated with hemorrhagic complications only in model 1 with the larger number of subjects. Model 1 also showed a trend toward hypercoagulability being protective against hemorrhage (p = 0.07).
Discussion
This 14% rate of major hemorrhage, all within 2 months of starting anticoagulation, in these COVID-19 patients hospitalized with VTE, is more than double the major hemorrhage rate reported for patients on anticoagulation for atrial fibrillation (6.2% for warfarin and 5.3% for non-oral anticoagulation [6]), where patients are anticoagulated for years. It is also greater than reports of a 1.8% to 4% rate of major hemorrhages for routine anticoagulation treatment of VTE [7–9]. The 2.4% rate of fatal hemorrhage observed in COVID-19 patients with VTE on therapeutic anticoagulation is an order-of-magnitude greater than the 0.1% to 0.3% fatal bleeding rate reported for therapeutic anticoagulation treatment of VTE [8, 9]. Our 2.4% fatality rate may underestimate risk since three additional patients with mental status changes after initiating anticoagulation were not investigated for ICH due to their transition to comfort-measures-only status.
Our data are similar to Godier et al. [1] who reported an 18% rate of major bleeding in COVID-19 ICU patients on anticoagulation. Those bleeding events occurred after fibrinogen levels decreased over the preceding 3 to 5 days which is consistent with our experience showing that low fibrinogen is associated with increased the risk of bleeding. Fibrinogen tends to be elevated in COVID-19 and is believed to be one source of hypercoagulability in these patients as well as a marker of excessive inflammation and disease severity [10]. But the initially high fibrinogen levels early in the disease may decrease later, possibly reflecting reduced inflammatory stimulation of fibrinogen production, depletion from excessive thrombosis or decreased production from hepatocyte injury. This creates the dangerous situation of insufficient fibrinogen to form effective clots. Thus, high serum fibrinogen is protective against hemorrhagic complications and when fibrinogen is low, the benefit to risk ratio for therapeutic anticoagulation should be reassessed.
Elevated LDH was highly significantly related to hemorrhage in both of our multivariate models. This may reflect liver injury especially since AST and ALT were also elevated in the univariate analyses. Liver injury could contribute to reduced production of fibrinogen and other clotting factors which were not measured in this study.
Given our incomplete understanding of the thrombotic profile of COVID-19 and its variable evolution over the course of the disease, periodic measurement of multiple biomarkers of thrombotic risk may improve the benefits and reduce hemorrhagic complications from anticoagulation treatment of VTE [11]. Bedside, point-of-care, tests of the entire coagulation system which can be performed frequently with immediate results such as thromboelastography (TEG6) or rotational thomboelastometry (ROTEM) may be helpful for guiding anticoagulation therapy [12, 13]). The higher risk of bleeding complications from UFH compared to LMWH observed here is consistent with thromboelastography measurements on COVID-19 patients showing UFH had greater suppression of coagulation with longer reaction times, delta R [12]. Point-of-care TEG6 measurements of fibrinogen levels or high fibrinogen clot strength performed frequently at the bedside may help optimize anticoagulation and prevent hemorrhages in VTE patients.
The supratherapeutic aPTT just prior to all four fatal hemorrhages suggests that patients on UFH may have been over-anticoagulated. This may result from difficulties in sufficiently frequently monitoring aPTT in the setting of a pandemic straining medical resources. It also reflects the clinical enthusiasm for anticoagulation in COVID-19 because of the high death rate from acute pulmonary embolism estimated to be the cause of death for 20% in a meta-analysis of autopsy studies [14]. These fatal complications may explain why some studies do not observe reduced mortality in COVID-19 treated with therapeutic anticoagulation [15]. Notably, a mortality benefit has been observed with anticoagulation administered at prophylactic doses [15, 16] which are less likely to have complications. Lower fibrinogen levels in patients with hemorrhages raises the possibility of depleted clotting factors also contributing to increased bleeding risk. One might hypothesize that antiplatelet agents in combination with therapeutic anticoagulation may also have contributed to increased bleeding risk although the association on univariate analysis was only a trend on multivariate analysis (p = 0.07).
One limitation is that three patients with mental status changes shortly following initiation of therapeutic anticoagulation were not assessed for intracranial hemorrhage. Those patients were transitioned to comfort care measure only, so they died without performing head CT. We did not count these as anticoagulation complications so our 2.4% fatal complication rate may underestimate the true mortality. The retrospective nature of this study is another limitation which meant that data was not systematically collected on all patients. Serum fibrinogen was missing for 52 patients so we ran two multivariate models: one without fibrinogen with more subjects and more statistical power and one with fibrinogen data but fewer subjects. There was insufficient information from the ICD10 codes identifying hypercoagulability to differentiate genetic from acquired causes. Although we recruited patients from 4 hospitals, the total number, 170, with 36 hemorrhagic events is still relatively small. Since all 4 hospitals were in New York City with cases from the initial pandemic wave, generalizability to other locations and to the new variants is uncertain. Furthermore, we only studied patients who had VTE, even though anticoagulation at various doses is commonly administered to COVID-19 patients without VTE. However, we expect that COVID-19 patients without VTE may also be at higher risk of hemorrhagic events when intubated with low fibrinogen, elevated LDH and especially when using UFH.
The decision to begin therapeutic anticoagulation to treat VTE complicating COVID-19 should consider these elevated risks especially when using unfractionated heparin or when treating intubated, ICU patients with low serum fibrinogen or elevated serum LDH.
Author contributions
All authors substantially contributed to data analysis, manuscript writing and agreed to submit the final version to JTT™. MRP, EL and HD also contributed to data collection.
Data availability
Data can be made available upon request with a standard institutional data use agreement.
Declarations
Conflict of interest
None of the authors has an actual or potential conflict of interest to declare. No funds, grants, or other support was received.
Ethical approval
The study was approved by the institutional review board and has been conducted in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
The Institutional Review Board of Weill Cornell Medicine approved this HIPAA compliant, retrospective study waiving the requirement for written informed consent.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Godier A, Clausse D, Meslin S, Bazine M, Lang E, Huche F, Cholley B, Hamada SR. Major bleeding complications in critically ill patients with COVID-19 pneumonia. J Thromb Thrombolysis. 2021;52(1):18–21. doi: 10.1007/s11239-021-02403-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Melmed KR, Cao M, Dogra S, Zhang R, Yaghi S, Lewis A, Jain R, Bilaloglu S, Chen J, Czeisler BM, Raz E, Lord A, Berger JS, Frontera JA. Risk factors for intracerebral hemorrhage in patients with COVID-19. J Thromb Thrombolysis. 2021;51(4):953–960. doi: 10.1007/s11239-020-02288-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Boira I, Esteban V, Vañes S, Castelló C, Celis C, Chiner E. Major bleeding complications in COVID-19 patients. Cureus. 2021;13(8):e16816. doi: 10.7759/cureus.16816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Abate V, Casoria A, Rendina D, Muscariello R, Nuzzo V, Vargas M, Servillo G, Venetucci P, Conca P, Tufano A, Galletti F, Di Minno G. Spontaneous muscle hematoma in patients with COVID-19: a systematic literature review with description of an additional case series. Semin Thromb Hemost. 2022;48(1):100–108. doi: 10.1055/s-0041-1732370. [DOI] [PubMed] [Google Scholar]
- 5.Kaatz S, Ahmad D, Spyropoulos AC, Schulman S, Subcommittee on Control of Anticoagulation Definition of clinically relevant non-major bleeding in studies of anticoagulants in atrial fibrillation and venous thromboembolic disease in non-surgical patients: communication from the SSC of the ISTH. J Thromb Haemost: JTH. 2015;13(11):2119–2126. doi: 10.1111/jth.13140. [DOI] [PubMed] [Google Scholar]
- 6.Ruff CT, Giugliano RP, Braunwald E, Hoffman EB, Deenadayalu N, Ezekowitz MD, Camm AJ, Weitz JI, Lewis BS, Parkhomenko A, Yamashita T, Antman EM. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet (London, England) 2014;383(9921):955–962. doi: 10.1016/S0140-6736(13)62343-0. [DOI] [PubMed] [Google Scholar]
- 7.Zidane M, Schram MT, Planken EW, Molendijk WH, Rosendaal FR, van der Meer FJ, Huisman MV. Frequency of major hemorrhage in patients treated with unfractionated intravenous heparin for deep venous thrombosis or pulmonary embolism: a study in routine clinical practice. Arch Intern Med. 2000;160(15):2369–2373. doi: 10.1001/archinte.160.15.2369. [DOI] [PubMed] [Google Scholar]
- 8.Wu C, Alotaibi GS, Alsaleh K, Sean McMurtry M. Case fatality of bleeding and recurrent venous thromboembolism during, initial therapy with direct oral anticoagulants: a systematic review. Thromb Res. 2014;134(3):627–632. doi: 10.1016/j.thromres.2014.07.001. [DOI] [PubMed] [Google Scholar]
- 9.Gómez-Outes A, Lecumberri R, Suárez-Gea ML, Terleira-Fernández AI, Monreal M, Vargas-Castrillón E. Case fatality rates of recurrent thromboembolism and bleeding in patients receiving direct oral anticoagulants for the initial and extended treatment of venous thromboembolism: a systematic review. J Cardiovasc Pharmacol Ther. 2015;20(5):490–500. doi: 10.1177/1074248415575154. [DOI] [PubMed] [Google Scholar]
- 10.Sui J, Noubouossie DF, Gandotra S, Cao L. Elevated plasma fibrinogen is associated with excessive inflammation and disease severity in COVID-19 patients. Front Cell Infect Microbiol. 2021;11:734005. doi: 10.3389/fcimb.2021.734005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gorog DA, Storey RF, Gurbel PA, Tantry US, Berger JS, Chan MY, Duerschmied D, Smyth SS, Parker W, Ajjan RA, Vilahur G, Badimon L, Berg J, Cate HT, Peyvandi F, Wang TT, Becker RC. Current and novel biomarkers of thrombotic risk in COVID-19: a Consensus Statement from the International COVID-19 Thrombosis Biomarkers Colloquium. Nat Rev Cardiol. 2022 doi: 10.1038/s41569-021-00665-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gurbel PA, Bliden KP, Rout A, Rapista N, Walia N, Chaudhary R, Ens G, Traianova M, Barnes JS, Tantry US. Bedside thromboelastography to rapidly assess the pharmacodynamic response of anticoagulants and aspirin in COVID-19: evidence of inadequate therapy in a predominantly minority population. J Thromb Thrombolysis. 2021;51(4):902–904. doi: 10.1007/s11239-021-02435-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gurbel PA, Tantry US, Storey RF. International COVID-19 thrombosis biomarkers colloquium: COVID-19 diagnostic tests. J Thromb Thrombolysis. 2021;52(4):992–998. doi: 10.1007/s11239-021-02465-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zuin M, Engelen MM, Bilato C, Vanassche T, Rigatelli G, Verhamme P, Vandenbriele C, Zuliani G, Roncon L. Prevalence of acute pulmonary embolism at autopsy in patients with COVID-19. Am J Cardiol. 2022;171:159–164. doi: 10.1016/j.amjcard.2022.01.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pesavento R, Ceccato D, Pasquetto G, Monticelli J, Leone L, Frigo A, Gorgi D, Postal A, Marchese GM, Cipriani A, Saller A, Sarais C, Criveller P, Gemelli M, Capone F, Fioretto P, Pagano C, Rossato M, Avogaro A, Simioni P, Prandoni P, Vettor R. The hazard of (sub)therapeutic doses of anticoagulants in non-critically ill patients with Covid-19: The Padua province experience. J Thromb Haemost: JTH. 2020;18(10):2629–2635. doi: 10.1111/jth.15022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tang N, Bai H, Chen X, Gong J, Li D, Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost: JTH. 2020;18(5):1094–1099. doi: 10.1111/jth.14817. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data can be made available upon request with a standard institutional data use agreement.