Abstract
Extracorporeal membrane oxygenation (ECMO) provides advanced cardiopulmonary life support for patients in cardiac and/or respiratory failure. Echocardiography provides essential diagnostic and anatomic information prior to ECMO initiation, allows for safe and efficient ECMO cannula positioning, guides optimization of flow, provides a modality for rapid troubleshooting and patient evaluation, and facilitates decision-making for eventual weaning of ECMO support.
Currently, guidelines for echocardiographic assessment in this clinical context are lacking. In this review, we provide an overview of echocardiographic considerations for advanced imagers involved in the care of these complex patients. We focus predominately on new cannulas and complex cannulation techniques including a special focus on double lumen cannulas and a section discussing indirect left ventricular venting. Echocardiography is tremendously valuable in providing optimal care in these challenging clinical situations. It is imperative for imaging physicians to understand the pertinent anatomic considerations, the often complicated physiological and hemodynamic context, and the limitations of the imaging modality.
Keywords: Echocardiography, extracorporeal membrane oxygenation, VA-ECMO, VV-ECMO, dual-lumen cannulation, LAVA-ECMO
Introduction
Extracorporeal membrane oxygenation (ECMO) provides advanced cardiopulmonary life support for patients in cardiac and/or respiratory failure. ECMO is increasingly utilized for a variety of indications. Expanding clinical applications are accompanied by the adoption of novel configuration strategies and new cannula designs. Echocardiography provides essential diagnostic and anatomic information prior to ECMO initiation, allows for safe and efficient ECMO cannula positioning, guides optimization of flow, provides a modality for rapid troubleshooting and patient evaluation, and facilitates decision-making for eventual weaning of ECMO support.
In this review, we provide an overview of echocardiographic considerations for advanced imagers involved in the care of these complex patients with a primary emphasis on transesophageal echocardiography (TEE). We focus predominately on new cannulas and complex cannulation techniques including options for indirect left ventricular venting. Currently, guidelines for echocardiographic assessment in this clinical context are lacking. We review the existing literature and provide a supportive framework to help facilitate the future development of guidelines and standards by focusing our discussion on; 1) pre-ECMO assessment, 2) cannulation and initiation of ECMO, 3) troubleshooting and ECMO emergencies, 4) weaning and assessment of recovery. First, we will briefly cover veno-venous ECMO, followed by veno-arterial ECMO with a special focus on left ventricular venting strategies, and concluding with a discussion of future avenues for research and guideline development. Echocardiography is tremendously valuable in providing optimal care in these challenging clinical situations. It is critical for the echocardiographer to understand the pertinent anatomic considerations for their patient and the proposed cannulation strategy, the often complicated physiological and hemodynamic context, and to understand the limitations of this imaging modality.
Veno-venous ECMO
Veno-venous (VV)-ECMO removes deoxygenated and hypercarbic blood as it returns to the heart, routing it through venous cannula(s) connected to a membrane oxygenator. The blood is oxygenated, CO2 is removed, and the blood returned to the patient through a venous return cannula(s). Therefore, the primary indication for VV-ECMO is to provide an extracorporeal “lung” for patients with severe respiratory failure. Because blood is returned to the right heart by a venous cannula (to the right atrium (RA) or venae cavae), the patient’s heart must be capable of providing the cardiac output necessary for both pulmonary and systemic circulation. Although traditional VV-ECMO does not provide any direct cardiac support, in practice, oxygenation and CO2 removal in a patient with severe respiratory failure is beneficial to the right ventricle (RV). VV-ECMO decreases the incidence of RV failure in patients by reducing pulmonary artery (PA) pressures, improving cardiac index, reducing central venous pressure, and reducing ventilatory settings1. The near universal adoption of lung protective ventilatory strategies has reduced the prevalence of right ventricular dysfunction in patients with respiratory failure, however, correction of acidosis and hypoxia using VV-ECMO can further improve RV function while concurrently reducing peak airway pressures2. A summary of focused considerations for echocardiographic assessment during VV-ECMO can be found in Table 1.
Table 1.
Focused echocardiography for ECMO.
| Focused ECHO for ECMO | ||||
|---|---|---|---|---|
| Pre-initiation TEE evaluation | ECMO Cannulation and Flow initiation | Trouble shooting | Weaning and post-decannulation | |
| VV ECMO | Right heart evaluation | Guide wire(s) confirmation | Ensure proper orientation of outflow/inflow port by CFD | Right heart evaluation |
| RA/RV dilatation | Beware of artifacts | Assess for pericardial and pleural effusions | RA/RV dilatation | |
| Eccentricity index | RA/RV size | Evaluate volume status | Eccentricity index | |
| TAPSE, S’, FAC | RV function | Evaluate for thrombus | TAPSE, S’, FAC | |
| 3D SV:ESV | TV and evolution of TR | 3D SV:ESV | ||
| TV evaluation | Screen for pericardial and pleural effusions | TV evaluation | ||
| TV annular dilatation | Assess intraventricular volume status | TV annular dilatation | ||
| TR presence/severity | TR presence/severity | |||
| PA evaluation | Correlate with PaO2 and PaCO2 | |||
| PA acceleration time | ||||
| RVSP | ||||
| RV-PA coupling | ||||
| Evaluate IVS and IAS | ||||
| Evaluation for anatomical variants | ||||
| Screen for pericardial/pleural effusions | ||||
| VA ECMO | Left heart evaluation | Venous guide wire confirmation | Ensure proper orientation of venous inflow port by CFD | Left heart evaluation |
| LA/LV dilation | Arterial guide wire confirmation | Assess for pericardial and pleural effusions | LA/LV dilation | |
| LVEF by 2D and 3D | Monitor for LV distention | Monitor LA size and assess for invagination | LVEF by 2D and 3D | |
| AV and MV | Dilated and impaired LV | Evaluate volume status | Right heart evaluation | |
| Note severity of AI and/or MR | Spontaneous echo contrast within LV and LA | Evaluate for thrombus | RA/RV dilatation | |
| Note presence/absence: | Lack of AV opening | Evaluate for LV distention | Eccentricity index | |
| Aortic dissection | Worsening AI | Opening of AV | TAPSE, S’, FAC | |
| Mobile atherosclerotic plaques | Worsening MR | Presence of spontaneous echo contrast | 3D SV:ESV | |
| Pericardial tamponade | Correlate clinically with arterial line pulsatility | Thrombus formation | IAS evaluation | |
| LV apical thrombus | Assess for pericardial and pleural effusions | Arrhythmia | Evaluation AV and MV | |
| Inotropic and/or mechanical support | Assess intraventricular volume status | Management options for LV distention | AV VTI>10cm | |
| IAS defect | Evaluate position and color inflow for venous cannula | Decrease afterload | increased AV opening | |
| Pericardial/pleural effusions | Reduce ECMO flow | Lateral MV S’>8cm/s | ||
| Systemic anticoagulation | Correlate with arterial line pulsatility | |||
| Consider LV venting | ||||
| Management of differential hypoxia | ||||
| CFD of descending aorta | ||||
| Correlate with right upper extremity arterial line waveform and PaO 2 | ||||
A comprehensive TEE exam should be performed for all patients if time and clinical scenario allows.
TTE may provide substantial information in the pre-ECMO assessment and during weaning from ECMO and should be considered as an alternative and non-invasive modality
AI: Aortic insufficiency, AV: Aortic valve, CFD: Color flow Doppler, ESV: End-systolic volume, FAC: Fractional area change, IAS: Interatrial septum, IVS: Interventricular septum, LVEF: Left ventricular ejection fraction, MV: Mitral valve, PA: Pulmonary artery, RA: Right atrium, RV: Right ventricle, RVSP: Right ventricular systolic pressure, SV: Stroke volume, TAPSE: Tricuspid annular plane systolic excursion, TEE: Transesophageal echocardiography, TTE: Transthoracic echocardiography, TR: Tricuspid regurgitation, TV: Tricuspid valve, VTI: Velocity time integral
VV-ECMO: initial pre-ECMO echocardiographic assessment
We advocate for the performance of a comprehensive echocardiographic examination in all patients considered candidates for ECMO cannulation when TEE is not contraindicated and timing and the patients’ clinical status allows. A thorough discussion of a comprehensive TEE examination is beyond the scope of this review and the reader is referred to the American Society of Echocardiography Guidelines3. Alternatively, transthoracic echocardiography (TTE) also provides considerable useful information to the clinician, can readily assess the RV, is non-invasive, and is often more rapidly available at the bedside. Much of the pre-ECMO assessment is particularly amendable to TTE, especially apical imaging to assess for LV or RV thrombus as well as taking measurements of structures at the base of the heart such as the LA or RA. TEE has many advantages over TTE, particularly in the assessment of valve structure and function, the imaging of wires and cannulas, and in providing consistent imaging windows irrespective of patient body habitus or recent sternotomy that may limit the obtainable TTE views. While we will refer predominately to TEE in this review, for most of the discussion herein, analogous TTE views are available and may provide useful information.
The downstream consequences of respiratory failure are readily apparent on TEE. A thorough evaluation of the right heart is essential. Key findings to look for include right atrial (RA) or RV dilation, tricuspid valve (TV) annular dilation, the presence and severity of tricuspid regurgitation (TR) (Movies 1 and 2), flattening of the interventricular septum (carefully noting the phase in the cardiac cycle where it predominates), an eccentricity index greater than 1, or McConnell’s sign (severe hypokinesis of the basal and mid RV wall with sparing of the apex).
Notably, assessing the TV is of particular importance prior to initiation of VV-ECMO. Once ECMO flow is established, accurate evaluation of TR is inhibited due to the drainage and reinjection of blood from and into the RA. Semi-quantitative measures of RV function include tricuspid annular plane systolic excursion (TAPSE), tricuspid annular systolic peak velocity (S’), and fractional area change (FAC)4. Due to the complex geometry of the RV, 3D assessment of size and function may prove valuable in the future, but their utility in this context at present remains unclear5. 3D assessment can differentiate RV longitudinal motion (shortening of the longitudinal axis of tricuspid annulus toward the apex), radial motion (inward movement of the RV free wall), and anteroposterior motion (traction of the RV free wall by LV myocardium circumferential deformation)6. Though longitudinal and anteroposterior motion of the RV are the most prominent contributors to RV performance, there is an age-dependent increase in radial motion contribution7. FAC, especially when derived from a 3D dataset, may provide more useful information than a subjective visual estimate of RV function or measurement of TAPSE. A key point for physicians is to understand both the geometric and mechanistic complexities of the RV and recognize the potential limitations of common indices used to assess RV function that are limited to a single plane (TAPSE for example). Elevated pulmonary pressures can be estimated by the simplified Bernoulli equation with the TR jet peak velocity. A PA acceleration time shorter than 100ms and a biphasic waveform can also indicate elevated pulmonary pressures even in the absence of TR8. Crucially, in the context of acute RV failure, the RV power output drops and may barely overcome the increased afterload of the pulmonary vasculature. Consequently, the RV systolic pressure (RVSP) may be low but is now reflective of cardiac failure as opposed to “normal” pulmonary pressures. It is imperative the physician conducting the exam convey this clearly in the diagnostic report, as a cursory glance at the automatically-generated number for RVSP may provide a false sense of security if not accompanied by a clear assessment of RV function for appropriate context. Furthermore, novel methods seek to assess the coupling between the RV and the PA, thus contextualizing the function of the RV with the afterload the chamber is facing. RV-PA coupling can predict RV failure in patients with pulmonary hypertension (pHTN) using cardiac magnetic resonance (CMR) imaging9. 3D echocardiography-derived stroke volume to end-systolic volume ratio (measuring RV-PA coupling) predicts adverse outcomes in pediatric pHTN and correlates with free-wall RV longitudinal strain10. Importantly, 3D echocardiography correlates well with CMR and may provide an alternative method for assessing this novel index in the ECMO population11.
Establishing a baseline assessment of TR and the interatrial septum (IAS) can serve as a reference for future weaning and monitoring of VV ECMO. Evaluation of the IAS and presence of patent foramen ovale (PFO) (Movie 3) or atrial septal defect (ASD) can sometimes be critically important, especially during initiation or weaning of ECMO. As the RA receives blood from the VV-ECMO circuit at a relatively high-velocity, it is possible for right to left shunting to occur in the presence of a PFO. Additionally, a PFO with significant shunt may cause persistent hypoxia while weaning. Other anatomical variants of embryologic structures should be noted as a prominent Chiari network or Eustachian valve could ensnare wires and hinder venous cannula placement and positioning. A dilated coronary sinus (>1.5cm) warrants caution and close evaluation for the presence of a persistent left superior vena cava (SVC)12. Injection of agitated saline through a left upper extremity intravenous line can identify the presence of a persistent left SVC by early opacification of the coronary sinus prior to the right atrium and ventricle (Movie 4). Incidental cannulation of a dilated coronary sinus could result in perforation, rupture, pericardial effusion, and hemodynamic compromise. If a newer dual-lumen cannula (discussed in detail later) is accidentally positioned within the coronary sinus orifice, one may note excellent superior drainage but poor IVC drainage with resultant venous congestion. Cannulation of a coronary sinus in the presence of a persistent left SVC with a support device like the Protek Duo may lead to outflow from the ECMO circuit recirculating though the coronary sinus into the left SVC and into the venous circulation of the left upper extremity.
Presence of pre-existing pacemaker wires, central venous lines, peripherally inserted central catheters, and PA catheters can also hinder with both the identification of guidewires and the insertion and positioning of venous cannulas13.
VV-ECMO: Guiding cannulation and initiation of ECMO
As discussed above, guidewire confirmation is often one of the first steps in the venous cannulation process and TEE can visualize wires within the venae cavae and RA. Clear communication is essential when confirming guidewires and must be emphasized. The imager should meticulously assess for the presence and location of guidewires, particularly when additional catheters and wires are present. The proceduralist should wait for explicit confirmation from the imager prior to proceeding as mistaking a preexisting intracardiac device lead for a guidewire may lead to catastrophic complications. Movie 5 illustrates confirmation of the “J-tip” of a guidewire adjacent to a preexisting pacing lead. Movie 6 demonstrates the use of biplane imaging to help confirm the presence of a guidewire in addition to two preexisting pacing leads.
With two separate single lumen cannulas, it is imperative the echocardiographer confirm two distinct venous guidewires as the guidewire can generate a number of strong echocardiographic artifacts. Reverberation artifacts can make identification of wires at greater echocardiographic depth more difficult as well as potentially creating the appearance of duplicate catheters or wires within the chamber (Movie 7). Mirror image and refraction artifacts can result in false positive identification of two separate guidewires. Beam width artifacts can be avoided by adjusting the focal zone in the area of interest to enhance lateral resolution. Side lobe and beam width artifacts can cause blurring of a guidewire14. Use of a 3D matrix-array TEE probe can provide simultaneous bi-plane imaging (also called x-plane) that may be beneficial in these situations15. (Movie 8). Figure 1a illustrates representative traditional two-cannula ECMO (Vf-Vj-ECMO) (Movies 9 and 10). and Figure 1b depicts traditional femoral access dual lumen cannulation [(ca)Vf-V-ECMO]. Availability of TEE and the capability to transport an unstable patient to a fluoroscopy suite may limit the application of these modalities in the acute setting. Bedside TTE however has been shown to be comparable to fluoroscopy guidance in placement of VA and VV cannulas and may provide a good option if neither TEE nor fluoroscopy is readily available16.
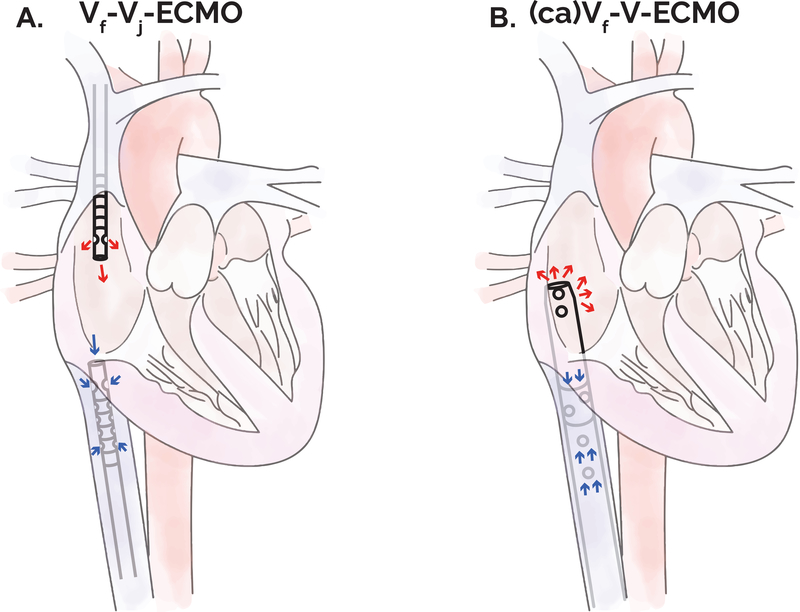
Figure 1. Schematic of common VV-ECMO cannulation strategies.
The Maastricht consensus nomenclature is used to describe cannula location. “Traditional” two-cannula approach to VV-ECMO. In Vf-Vj-ECMO the femoral venous drainage cannula tip is positioned within the IVC and an internal jugular venous return cannula tip is positioned within the right atrium (A). In (ca)Vf-V-ECMO a single, double-lumen (cavoatrial) cannula is placed with venous return achieved from the IVC and return flow into the RA. One example is the ProtekDuo RD cannula (CardiacAssist) (B). A similar strategy may be achieved with two separate femoral venous cannulas. ca: cavoatrial, ECMO: extracorporeal membrane oxygenation, f: femoral, IVC: inferior vena cava, j: jugular, RA: right atrium, SVC: superior vena cava, VV: veno-venous.
Dual lumen cannulas such as the Avalon Elite (Maquet, Rastatt, Germany) or Crescent MC3 (Cardiopulmonary, Dexter, MI) and the ProtekDuo Dual Lumen Cannula (LivaNova, England, UK) offer many potential advantages over traditional VV-ECMO including; a single-site for percutaneous insertion, typical deployment in the right internal jugular vein allowing avoidance of femoral cannulation, and a decreased incidence of recirculation17. With avoidance of femoral cannula placement, patients have improved mobilization, ambulation, and participation in physical therapy. Importantly, ECMO is not a curative therapy, but rather buys time for correction of underlying pathophysiologic disturbances. Thus, a cannulation strategy that improves patient mobilization may lead to swifter recovery and ECMO weaning18.
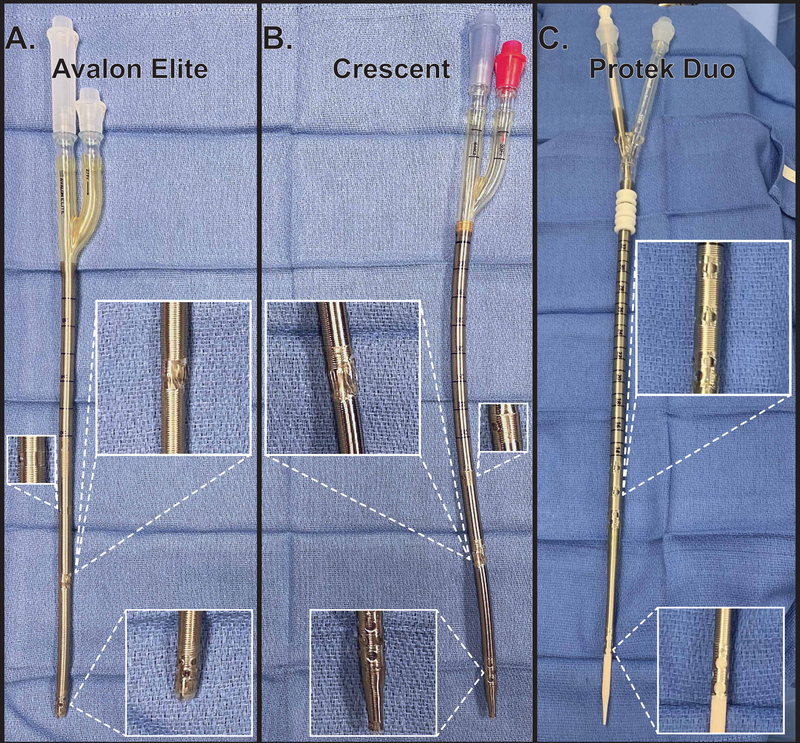
TEE provides an ideal imaging modality to guide placement of these single insertion site, dual lumen cannulas13. A summary of dual lumen cannulas and echocardiographic assessment can be found in Table 2 and Figure 2 provides images of the Avalon Elite (Fig. 2a), Crescent (Fig. 2b) and Protek Duo RD (Fig. 2c) with detailed insets depicting the inflow and outflow ports.
Table 2.
Dual lumen devices.
|
Dual Lumen Devices
| ||||
|---|---|---|---|---|
| Size | Site | Position | Utility | |
|
| ||||
| cannula tip in the IVC | ||||
| Avalon | 13–31 Fr dual lumen | RIJ | inflow from IVC and SVC | oxygenatation and clearing of CO2, returns blood to RA |
| outflow directed toward TV | ||||
|
| ||||
| cannula tip in the IVC | ||||
| Crescent MC3 | 24–32 Fr dual lumen | RIJ | inflow from IVC and SVC | oxygenatation and clearing of CO2, returns blood to RA |
| outflow directed toward TV | ||||
|
| ||||
| cannula tip in the PA | oxygenation and clearing of CO2, RV support (delivers blood directly into PA) | |||
| Protek Duo | 29–31 Fr dual lumen | RIJ | inflow from cavoatrial junction | |
| outflow directed toward main PA | ||||
Fr: French, IVC: Inferior vena cava, PA: Pulmonary artery, RA: Right atrium, RIJ: Right internal jugular, SVC: Superior vena cava, TV: Tricuspid valve
Figure 2. Examples of double lumen ECMO cannulas.
The Avalon Elite (Getinge AB) (A) and Crescent (MC3) (B) are used for bicaval cannulation with two inflow ports designed to be positioned within the SVC and IVC and a central outflow port that is oriented to direct flow towards the center of the tricuspid valve. The Protek Duo (CardiacAssist) has proximal inflow ports with outflow ports at the distal tip of the cannula (C). These cannulas come in various lengths and can be placed from the internal jugular vein and positioned with the proximal inflow port within the RA and the distal outflow port within the PA. Alternatively, the femoral vein can be cannulated, the proximal inflow ports positioned within the IVC, and the distal outflow port oriented within the RA. A shorter option, the Protek Duo RD cannula is pictured. ECMO: extracorporeal membrane oxygenation, IVC: inferior vena cava, RA: right atrium, SVC: superior vena cava, PA: pulmonary artery.
The Avalon Elite is a 13 to 31Fr dual lumen cannula introduced through the right internal jugular with the cannula tip positioned in the inferior vena cava (IVC)19. The Crescent MC3 is a 24 to 32Fr dual lumen cannula positioned and utilized in the same method as the Avalon Elite20. Differences between the two cannulas include the French diameter where the Avalon Elite includes sizes 13Fr, 16Fr, 19Fr, 20Fr, 23Fr, 27Fr, and 31Fr where the Crescent MC3 includes sizes 24Fr, 26Fr, 28Fr, 30Fr, and 32Fr. As internal lumen diameter increases, higher flow rates are achievable without drastic increases in pressure gradients. The appropriately sized cannula should be selected individually for each patient.
Vascular irregularities such as stenosis of the jugular vein or presence of thrombus may preclude vascular access. Alternatively, placement of these dual lumen cannulas in the left internal jugular vein and left subclavian vein have been reported21. With left-sided cannulation, there may be significant interaction between guidewires and cannulae with intracardiac device leads if present.
Using bedside TEE, dual lumen cannulas can be placed in the intensive care unit, preventing the need to transport an unstable patient in respiratory distress to a procedure room with fluoroscopy. Echocardiography also allows visualization of cannula outflow ports and their relationship to key soft tissue structures invisible on fluoroscopy (ie. tricuspid valve leaflets, the IAS, a Eustachian valve). In addition, the orientation and direction of cannula outflow can be assessed to ensure appropriate cannula positioning. Finally, complications may be more rapidly detected as echocardiography can identify a pericardial effusion or injury to intracardiac structures whereas fluoroscopy cannot. In one small study, TEE was found to be superior to fluoroscopy in positioning the Avalon Elite cannula13. Care must be taken to ensure proper orientation of the outflow jet. Improper positioning (for example, with flow directed against the IAS or into a hepatic vein) may lead to pressure alarms, structure damage or perforation, hepatic congestion, or recirculation. Using 2D, 3D, and color flow Doppler, the echocardiographer can identify distinct components of these dual lumen cannulas. The cannula has distal inflow ports designed for IVC drainage with proximal inflow ports for SVC drainage. The outflow port is located centrally on the cannula and should be positioned to direct outflow across the TV. The spacing of the ports in these dual lumen cannulas is designed to decrease the incidence of recirculation when compared to two separate inflow and outflow cannulas17. The midesophageal modified bicaval view allows visualization of the SVC, IVC, RA, left atrium (LA), IAS, and TV and is useful for appropriately positioning these dual lumen cannulas. Color flow Doppler should be used to visualize flow emanating from the central outflow port directed toward the TV. Additionally, color flow Doppler should be used to visualize and document blood inflow through the SVC and IVC ports. Figure 3 illustrates placement of an IJ approach double-lumen cannula for VV-ECMO with an anatomic diagram (Fig. 3a), an idealized TEE image diagram (Fig. 3b), and an actual TEE still frame depicting a Crescent double-lumen cannula with color Doppler flow directed towards the tricuspid valve annulus (Fig. 3c). Movies 11 and 12 also demonstrate Crescent cannula positioning using TEE. Figure 4 demonstrates the radiographic appearance of a Crescent cannula with radiopaque markers for the SVC and IVC drainage ports, the infusion site (within the RA), and the catheter tip readily visualized.
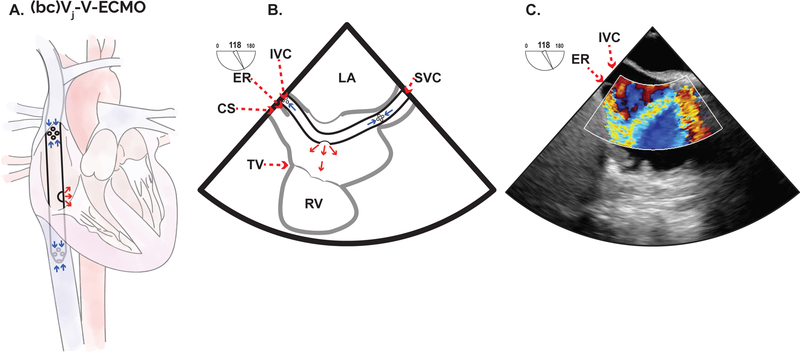
Figure 3. Double lumen bicaval cannulation for VV-ECMO.
A common method for conducting VV-ECMO is through placement of a double lumen cannula via the right internal jugular vein [(bc)Vj-V-ECMO]. Inflow ports are then positioned within the IVC and the SVC and a central outflow port is then oriented within the RA to direct blood flow across the tricuspid valve (A). Examples include the Avalon Elite (Getinge AB) and the Crescent (MC3). TEE readily confirms appropriate outflow port positioning and outflow jet orientation towards the tricuspid valve using color Doppler imaging. The mid-esophageal modified bicaval view is useful during positioning (B) and is often found by increasing the omniplane angle and making a slight clockwise turn of the probe from the mid-esophageal bicaval view. A modified bicaval view of a Crescent dual-lumen cannula with color flow Doppler demonstrating an outflow jet appropriately directed towards the center of the TV (C).
bc: bicaval, CS: coronary sinus, ER: Eustachian ridge, IVC: inferior vena cava, LA: left atrium, RA: right atrium, SVC: superior vena cava, TV: tricuspid valve, VV-ECMO: veno-venous extracorporeal membrane oxygenation.
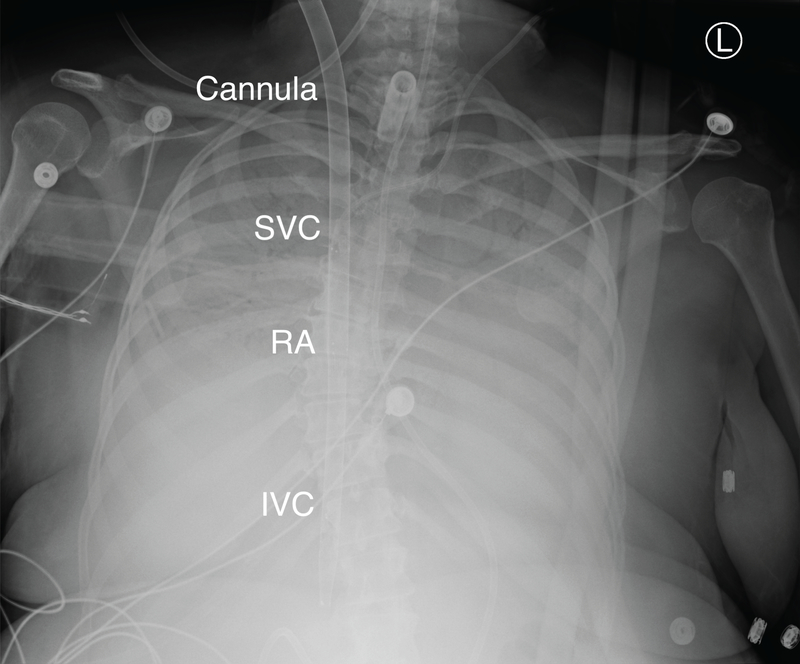
Figure 4. Radiographic appearance of dual lumen veno-venous ECMO cannula.
A Crescent MC3 (Cardiopulmonary, Dexter, MI) bicaval dual lumen cannula in a patient on (bc)Vj-V-ECMO support. The radiopaque markers at the site of SVC drainage, the infusion site (RA), the site of IVC drainage, and the catheter tip are readily apparent. (bc): bicaval, ECMO: extracorporeal membrane oxygenation, IVC: inferior vena cava, Vj: venous (jugular), RA: right atrium, SVC: superior vena cava.
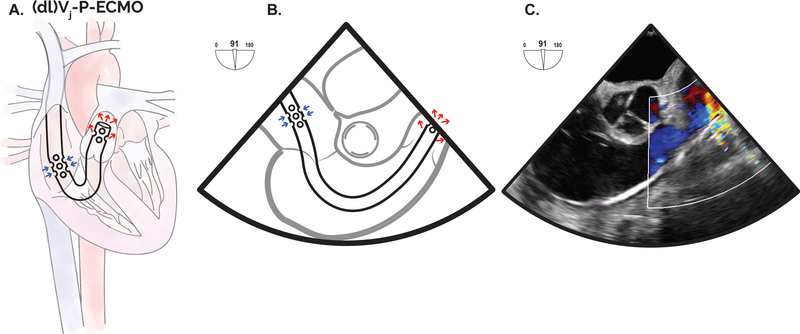
The ProtekDuo is also inserted through the right internal jugular vein, however, unlike the Avalon Elite, outflow of the VV ECMO circuit is delivered through the distal port (typically within the main PA) with venous inflow drained proximally (ideally at the superior cavoatrial junction)22. 2D echocardiography can guide placement of the ProtekDuo, similar to positioning a PA catheter, as the ProtekDuo traverses from right internal jugular vein through SVC into the RA, across the TV, through the RV and pulmonic valve into the main PA. The midesophageal modified bicaval view allows for visualization of the wire and subsequent cannula to pass through the TV and shifting to the midesophageal RV inflow-outflow view can allow for imaging of the entire ProtekDuo cannula. Color flow Doppler should confirm the cannula inflow port proximally at the cavoatrial junction and the outflow port in the PA. If unable to confirm the outflow port beyond the PV in the midesophageal RV inflow-outflow view, an aortic arch short axis view can be used. Unlike the Avalon Elite where blood is purely being oxygenated, cleared of carbon dioxide, and delivered back to the RA, the Protek Duo can assist a failing right ventricle by supporting the cardiac output and transporting blood from the RA, oxygenating it, and delivering it directly into the PA. These capabilities blur the delineation between VV ECMO and RV assist device (RVAD)18. Figure 5 illustrates the anatomic positioning of a Protek Duo cannula (Fig. 5a), an idealized TEE view demonstrating final positioning (Fig. 5b), and a TEE still-frame demonstrating color flow Doppler outflow from a Protek Duo within the main PA (Fig. 5c). Movies 13–15 illustrate 2D, color Doppler, and 3D imaging of a Protek Duo cannula in a patient undergoing (dl)Vj-P-ECMO.
Figure 5. Protek Duo cannulation for temporary RV support.
Double lumen cannulas are available for mechanical support of the RV including the Protek Duo (CardiacAssist) and the Impella RP (Abiomed). The Protek Duo is placed via the IJ with the inflow ports positioned within the RA and the distal outflow in the main PA [(dl)Vj-P-ECMO] allowing for offloading of the RV (A). The midesophageal RV inflow-outflow can demonstrate the Protek Duo cannula traversing the TV and the tip and outflow port location distal to the PV (B). A TEE view with color flow Doppler demonstrating outflow from the distal port within the main PA (C). dl: double lumen, ECMO: extracorporeal membrane oxygenation, j:jugular, P: Pulmonary artery, PV: Pulmonic valve, RV: right ventricle.
Once cannulas are appropriately positioned, TEE provides an excellent monitor during initiation of VV-ECMO and establishment of flow. Immediate re-assessment should include reevaluation of biventricular and biatrial size and function, re-assessing pre-existing pathology, monitoring cannula position for stability, screening for new pericardial or pleural effusions, and assessing intraventricular volume status. Under direct visualization of TEE, VV-ECMO flows can be adjusted by evaluation of RA and RV size, RV function, and evolution, either worsening or improvement, of TR.
VV-ECMO: Troubleshooting and ECMO emergencies
TEE is a useful tool for troubleshooting issues that arise during VV-ECMO and in evaluating patients who acutely decompensate while supported by VV-ECMO.
Cannula displacement or rotation with consequent mispositioning of ports is relatively common and can lead to hypoxemia, reduced flow, and high-pressure alarms23. TEE can readily identify cannula malposition and color flow Doppler is integral to ensure outflow jets are appropriately oriented. Reduced ECMO flow can also result from insufficient intravascular volume. Relatively restrictive intravenous fluid administration is common in patients with severe lung pathology, potentially predisposing these patients to intravascular depletion. Patient positioning can impact inflow drainage by altering cannula location and RA size. Evaluation of TR to estimate PA pressure is no longer reliable during ECMO given the alterations of flow in the RA from the inflow and outflow cannulas. Lastly, it is important to evaluate for thrombus adherent or adjacent to the cannula(s). Intravascular or intracardiac thrombus may impair ECMO flow, lead to high pressure alarms, embolize distally, and may contribute to or presage oxygenator thrombosis. Color flow Doppler, 2D, and 3D TEE can readily identify thrombotic material involving the cannula(s) and intracardiac or caval structures24.
VV-ECMO: Weaning, assessment of recovery, and post-decannulation evaluation
Currently, there are no well-accepted and globally recognized clinical guidelines, let alone echocardiography protocols, to guide weaning of VV ECMO25,26.There are three components of weaning from VV ECMO: the ECMO flow rate, the fraction of delivered oxygen in the sweep gas flow (FdO2), and the sweep gas flow rate. In addition, there is management of the patient’s ventilator settings in concert with the VV-ECMO wean as the patient transitions from extracorporeal support to intrinsic pulmonary support. Decision on which of the three VV-ECMO parameters to wean can be institution or physician specific and monitoring of PaO2, PaCO2, FiO2, ventilatory peak, plateau, driving, positive end-expiratory pressures (PEEP), and sedation all should be considered during an attempted VV-ECMO wean25.
TEE evaluation during weaning of VV-ECMO must consider RV function. As VV-ECMO flow decreases, evaluation of TR becomes more accurate and reflective of RVSP. RA, RV, TV annular dilation, flattening of the interventricular septum, along with the semi-quantitative measures including TAPSE, FAC, or tricuspid lateral S’ can be trended as alterations in flow and changes in PaO2 and PaCO2 may affect RV function4. As in the preoperative assessment, 3D analysis of the RV can be acquired, however, the utility of this evaluation is not known at this time5. Factors of successful RV weaning have been found to include a smaller RV size, increased RVEF, higher RV FAC, and higher magnitudes of RV strain. TAPSE and TR were not found to be associated with successful RV weaning27. TTE often provides superior alignment for assessment of TAPSE and given the non-invasive nature of TTE imaging may be preferred for VV-ECMO weaning, particularly in non-intubated patients.
VV-ECMO: Special considerations for SARS-CoV-2 infection
One year into the SARS-CoV-2 (COVID-19) pandemic, the role of VV ECMO for COVID-19 positive patients with acute respiratory distress syndrome (ARDS) is continuing to evolve28. Initial outcomes from VV-ECMO initiation in COVID-19 ARDS is comparable to patients supported with VV-ECMO for ARDS prior to the pandemic29.
TEE during the COVID-19 pandemic and specifically in known COVID-19 positive patients should include proper selection of patient population and protection of the personnel involved in image acquisition as recommended by The American Society of Echocardiography30. TEE should only be performed if the results are expected to provide clinical benefit and guide patient management. Given the potential for aerosol generation during the conduct of a TEE, TTE may be preferable. Minimizing exposure to known COVID-19 positive patients during TEE includes use of personal protective equipment (PPE) including, but not limited to proper handwashing, gloves, surgical face mask, gown, eye shields, and either an N-95 respirator or powered air purifying respirator (PAPR) system in accordance with institutional guidelines. TEE should be performed by the most experienced provider to minimize aerosolization and chance for accidental exposure31. We would also advocate a focused TEE examination in this unique circumstance to minimize operator and bystander exposure. With the continuing evolution of the COVID-19 pandemic and the introduction of vaccines, providers should continue to follow the recommendations of their respective societies.
Veno-arterial ECMO
VA-ECMO combines the pulmonary support of VV-ECMO (a membrane oxygenator) with an extracorporeal pump(s), providing the capability to support the failing ventricle(s) for patients in cardiogenic shock or with combined respiratory and cardiac failure. Labor intensive and technically challenging, VA-ECMO has a variety of clinical indications and a myriad of potential configurations that can be tailored to meet the needs of the patient and the clinical scenario. VA-ECMO may be indicated for such diverse situations as: post-cardiotomy cardiogenic shock, massive pulmonary embolism, circulatory support during lung transplantation, amniotic fluid embolism in parturients, “prophylactic” circulatory support for mediastinal mass resection, cardiogenic shock during acute myocardial infarction, or even out-of-hospital cardiac arrest (extracorporeal cardiopulmonary resuscitation [ECPR]). A full discussion of the indications for and specific configurations of VA-ECMO is beyond the scope of this review. We focus on anatomic, physiologic, and hemodynamic considerations of specific relevance to the echocardiographer. A summary of focused considerations for echocardiographic assessment during VA-ECMO can be found in Table 1.
VA-ECMO: initial pre-ECMO echocardiographic assessment
As with VV-ECMO, if no contraindications are present and time and clinical situation allows, a comprehensive TEE examination is valuable for all patients considered candidates for ECMO cannulation. The decision whether to institute VA-ECMO is complicated and adding to the complexity are the multiple configurations and cannulas available. TEE can aid in decision-making regarding the appropriateness of VA-ECMO as well as the optimal configuration strategy. A baseline exam establishes ventricular size and function, identifies anatomic defects, and provides a detailed assessment of valvular morphology and competency. TEE can quickly detect contraindications for cannulation or discover underlying disease necessitating surgical intervention. For example, detection of an aortic dissection flap could prompt emergent surgery, and aortic dissection is a relative contraindication for VA-ECMO (Movie 16). Severe, mobile atherosclerotic plaques within the aorta could prompt a decision to opt for central over peripheral (and surgical vs percutaneous) cannulation. Presence of pericardial effusion and tamponade physiology also could prompt a specific, targeted intervention that if successful, may obviate the need for extracorporeal support. A thorough baseline exam should capture and note the presence or absence of each of these pertinent findings as complications of VA-ECMO cannulation may include aortic dissection, stroke/embolic events, and pericardial tamponade. After initiation of VA-ECMO it is difficult to interpret the clinical relevance and etiology of a pericardial effusion if no baseline exam is available32. As with screening echocardiography for VV-ECMO, TTE may play an important role here as TTE can readily identify the presence of a pericardial effusion (Movie 17) as well as may provide superior apical imaging to screen for ventricular thrombus (Movie 18). In addition, TTE provides excellent non-invasive imaging of the IVC and in conjunction with a ventricular assessment can help estimate overall volume status (Movie 19), right atrial pressure, RVSP, and left atrial pressure.
Objective measurements of LV function should be obtained prior to VA-ECMO. It is our preference to use both 3D and 2D methods to calculate LVEF based upon EDV and ESV. We concur with the ASE guidance that when available and feasible 3D measurements should be used and believe this is particularly important in assessment prior to ECMO33. 3D or biplane imaging is also particularly helpful to ensure that the ventricular chamber is not foreshortened. This has implications not only for accurate assessment of global and regional function, but also in identifying the presence of an apical thrombus. As mentioned, if an apical thrombus is suspected, TTE may provide better apical imaging in some instances and can be used for quick verification. In low flow states, common when considering VA-ECMO, blood stasis can lead to intracardiac thrombus and this is important to identify prior to initiation of ECMO and potential distal embolization of the thrombotic material34. Notable aspects such as a dilated LA and dilated LV with thin ventricular walls correlate with persistently elevated LA and LV diastolic pressures indicating a chronic process35. TEE evaluation of the RV is conducted in a similar manner as described prior to VV-ECMO.
The echocardiographer should incorporate clinical context into their assessment and the final written interpretation should note the presence of inotropic agents, the use of mechanical circulatory support (eg. IABP), the patient’s ventilatory status (eg. positive pressure ventilation), and coexisting medical conditions such as sepsis15. A low cardiac output in the context of high-dose inotropic support and an IABP with 1:1 augmentation is markedly different than a low cardiac output in a spontaneously ventilating patient on their ambulatory dose of milrinone for example.
VA-ECMO increases the afterload on the LV (in many cases a failing LV), therefore pre-existing aortic and mitral valve insufficiency must be identified and quantified. Worsening of aortic insufficiency with VA-ECMO initiation can lead to LV distention and consequent increased wall stress, myocardial oxygen consumption, subendocardial ischemia, arrhythmia risk, worsened pulmonary edema, stasis, and thrombus formation36. Pre-existing mitral regurgitation (MR) should be evaluated. MR also may be exacerbated by a VA-ECMO-induced increase in afterload leading to LA distention, arrhythmia, and pulmonary edema.
VA-ECMO: Guiding cannulation and initiation of ECMO
Venous cannula guidewires are visualized as discussed above for VV-ECMO. Arterial cannulation can be performed peripherally via femoral or axillary cannulation or centrally via cannulation of the ascending aorta37. Visualization of the arterial guidewire within either the descending thoracic aorta or ascending aorta serves as additional confirmation of correct arterial vascular access. This may be especially helpful in cases of profoundly impaired cardiac output and/or oxygenation where identification of the femoral artery can be challenging, especially in the emergent setting.
After initiation of VA-ECMO, TEE provides real-time assessment of ventricular unloading, interventricular septal motion, and estimation of intravascular volume status and preload. Venous drainage decreases RV preload and peripheral venous congestion while providing an increase in systemic cardiac output via the ECMO arterial cannula38. Though removal of blood from the right heart may decrease LV preload from the RV system, increased arterial flow from ECMO increases LV preload from the residual pulmonary circuit, Thebesian coronary blood, bronchial blood, and aortic regurgitation if present39. Movies 20 and 21 demonstrate an increase in the severity of aortic insufficiency after initiation of peripheral VA-ECMO. The failing LV must be able to surmount the increase in afterload provided by the ECMO circuit to eject this increased blood return out of the aortic valve. If the aortic valve remains closed, blood will gradually accumulate within the LV leading to increased distention, increased wall stress, decreased myocardial perfusion by reduction of the transcoronary perfusion gradient, venous stasis, and thrombus formation (Movie 22)4.
TEE provides the ability to monitor LV distention and function. As VA-ECMO flow is increased, aortic pressure increases and LV stroke volume may decrease leading to increased LV volume, LV end-diastolic and end-systolic pressure, LA pressure, and PA pressure40. TEE can demonstrate increased LV distention and failure of the aortic valve to open potentially correlating to a decreased pulse pressure and decrease in pulsatility on the arterial line waveform38. However, the LV end-diastolic pressure versus volume relationship is nonlinear and the LV is contained within the constraints of an intact pericardium. Therefore, LV chamber size as determined by TEE may be an insensitive indicator of ventricular distention, end-diastolic pressure, and left atrial pressure and this limitation should be recognized by the echocariographer41.
VA-ECMO: Left ventricular venting
With commencement of peripheral VA-ECMO, the retrograde flow into the aorta from the femoral artery cannula causes an increase in afterload while the venous drainage from the RA will cause a decrease in preload. Patients with severe LV dysfunction placed on VA-ECMO may generate inadequate intraventricular pressures to open the AV in the face of this increased afterload. With no native cardiac ejection, the LV distends. Blood stasis within the heart can progress to thrombosis within the aortic root, LV, LA, or pulmonary veins. Key TEE findings of LV distention include lack of AV opening, a dilated and impaired LV, severe MR during systole and diastole, and spontaneous echo contrast throughout the LV and LA32. LV distention leads to LA pressure overload and contributes to the development of pulmonary edema presenting as declining respiratory function, difficulty in ventilator management, pink frothy secretions and presence of pleural effusions, peribronchial cuffing, air space opacification, and thickening of interlobar fissures on chest x-ray. Medical management of LV distention includes ensuring adequate anticoagulation, decreasing afterload with inodilators or vasodilators, and reducing ECMO flow. Changing the ECMO circuit can also be considered. Should medical management fail, alternative LV venting strategies may be employed42. A summary of common LV venting strategies and focused echocardiographic considerations is contained in Table 3.
Table 3.
Echocardiographic considerations for left ventricular venting strategies.
| LV Venting Strategies | |
|---|---|
| TEE Considerations | |
| Direct | |
| IABP | Screen for aortic dissection |
| Screen for severe AI | |
| Distal tip 2–3cm distal to left subclavian | |
| Impella | Visualize device positioning across AV into the LV |
| Inflow ports 3.5–5cm from the aortic annulus | |
| 45-degree bend in cannula at level of the aortic annulus | |
| Ensure no disruption of mitral valve, papillary muscles, or subvalvular apparatus | |
| CFD through the device from LV into the ascending aorta | |
| Visualize extent of LV decompression and note reduction in LV size | |
| Surgical venting | Visualize extent of LV decompression and note reduction in LV size |
| Indirect | |
| Atrial septostomy | TEE guidance of transseptal puncture, guidewire insertion, ballon atrial septostomy |
| Screen for pericardial effusion pre and post-intervention | |
| LAVA ECMO | TEE guidance of transseptal puncture, guidewire insertion, balloon atrial septostomy, cannula positioning |
| Transeptal puncture generally in “mid-mid” position | |
| Avoid posterior atrial wall perforation, damage to CS, TV, MV, pulmonary veins, aortic root | |
| If single, multiorifice cannula, distal port transseptal to LA, proximal port at inferior cavoatrial junction | |
| If multiple cannulas, confirm multiple guidewires | |
| CFD through cannula at respective inflow ports | |
| Assess AV opening with each cycle | |
| Assess pulmonary vein flow waveforms | |
| Assess LA size and monitor for invagination |
AI: Aortic insufficiency, AV: Aortic valve, CFD: Color flow Doppler, CS: Coronary sinus, LA: Left atrium, LV: Left ventricle, MV: Mitral valve, TV: Tricuspid valve
If interventions to reduce afterload (inodilators/vasodilators) and a decrease in VA-ECMO flows fail to relieve LV distention, percutaneous and surgical strategies may be indicated. In a meta-analysis of 7581 patients, LV venting strategies were associated with 35% higher probability of weaning and 12% lower risk of mortality43. TEE identification of risk factors may help predict which patients might require LV venting strategies and early initiation of these strategies may have a beneficial impact on patient outcome. A systemic review of 7995 patients showed early initiation (<12hr) after the start of VA ECMO was shown to have a reduction in 30-day mortality and an increase in successful weaning44. Therefore, while data is limited, echocardiographic screening with a goal of identifying patients who may benefit from early LV venting is an important area worthy of additional research.
The intra-aortic balloon pump (IABP) is one of the most commonly employed techniques, although use does not improve survival in patients on ECMO45. Inflation of the IABP during diastole increases coronary perfusion pressure and the decrease in afterload with the Venturi effect can lower mean arterial pressure, decrease the pressure gradient allowing for AV opening, and decompress the left ventricle46. Relative contraindications to IABP placement are similar to VA-ECMO including severe aortic insufficiency and aortic dissection. Though IABP can be placed under fluoroscopic guidance, TEE visualization of the descending aorta can help verify correct positioning. The distal tip of the IABP should be two to three centimeters below the take-off of the left subclavian artery at the level of the aortic arch. If the patient has a left sided upper extremity arterial line, dampening of the arterial line could indicate proximal migration of the distal tip of the IABP. If the IABP is positioned too far distally in the aorta (can be visualized by TEE) perfusion to the kidneys and bowel could be compromised.
The Impella (Abiomed, Danvers, MA) is a ventricular assist device capable of direct left ventricular unloading in addition to providing cardiac output for the failing LV47,48. The Impella is a family of catheters that utilize a transaortic axial flow pump that can provide up to 5.5L of blood flow per minute from the LV to the ascending aorta, directly reducing left ventricular distention from VA ECMO. In addition, it should be noted that the Impella RP is available for RV support and will be discussed later in this review. By directly decompressing the LV, the Impella has been shown to improve RV function and reduce PA pressures. A retrospective single-center trial including 66 patients showed combination of Impella and VA-ECMO significantly reduced mortality from 78% to 57%49. An international, multicenter study of 510 patients showed a reduction of 30-day mortality by 21%50. Impella placement and use carries similar risks as other mechanical circulatory support options including bleeding, hemolysis, limb ischemia, and need for renal replacement therapy50. After percutaneous or surgical insertion from the femoral, axillary, or subclavian artery, TEE can be utilized in placement of the Impella device and positioning it appropriately across the AV and into the LV51. The entire Impella should be visualized throughout the LVOT with the inflow port position 3.5 to 5.0 cm from the aortic annulus depending on type Impella deployed. The Impella has a 45-degree bend in the cannula designed to be positioned at the level of the aortic annulus51. Care must be taken that the device does not disturb the mitral valve, papillary muscles, or the subvalvular apparatus. After initiation of the Impella, a midesophageal long axis view should be obtained to confirm color flow through the device from the LV to the ascending aorta. Subsequent direct LV decompression and reduction in LV size should be visualized as well48.
Indirect LV venting can be obtained via atrial septostomy which can reduce distention of the LV by removing preload from the LA. Studies have shown improvement in return of LV function with early left ventricular decompression40, however, there has been no demonstration of improved survival52. Recently, limited balloon atrial septostomy via a percutaneous approach was demonstrated as a feasible technique for indirect LV venting in a small, single-center case series53. The authors found no immediate complications and noted a decrease in mean LA pressure in patients with available pre-and-post measurements. TandemHeart, a left atrial to femoral artery centrifugal pump employs a multi-holed cannula inserted via a transeptal puncture to directly drain the LA54. This procedure takes the older practice of atrial septostomy a step further by directly draining the LA after intentional creation of an interatrial connection.
Both the TandemHeart and the Impella, through either indirect left ventricular venting (positioning within the LA) or direct left ventricular venting, can effectively reduce LV stroke work by reducing left ventricular end diastolic volume and pressure55.
Several instances of direct percutaneous cannulation of the LA (left atrial veno-arterial [LAVA] ECMO) with a venous cannula as a method of indirect left ventricular venting are now reported in the literature56–58. As opposed to balloon septostomy, the addition of a drainage cannula allows for adjustment of flow to facilitate more or less unloading of the chamber.
In LAVA ECMO, TEE is essential for multiple aspects of the procedure. As with any patient who requires initiation of VA ECMO, a thorough pre-exam should be done. Key echocardiographic findings that could lead the clinician in considering LAVA ECMO as opposed to standard VA ECMO would include a LV likely to distend in the presence of the increased afterload of VA ECMO. Aortic insufficiency, severely decreased LV ejection fraction (EF), dilated LV chamber size, and MR are risk factors for increased LV distension, arrhythmia, pulmonary edema, venous stasis, and thrombus formation59.
TEE is used to guide transseptal puncture, guidewire insertion, balloon atrial septostomy, cannula positioning, and for flow titration. Careful imaging is integral to identify the optimal location for transseptal puncture and guide the performance of the puncture. Perforation of the RA, LA, coronary sinus, or damage to TV, mitral valve, pulmonary veins, or aortic root can occur but these risks are drastically mitigated through meticulous imaging and communication between the echocardiographer and the proceduralist.
The TandemHeart ProtekSolo Transeptal (LivaNova, London, UK) is a 21 French cannula specifically designed to be inserted via the femoral vein and placed in the left atrium by transseptal puncture22. Blood from the LA is returned via a ProtekSolo Arterial cannula into the femoral artery enabling indirect left ventricular decompression via the left atrium while supporting cardiac function60. This LA cannula may also be directed into a “Y” configuration with the arterial or venous limb of a pre-existing VA ECMO circuit adding indirect LV venting to the hemodynamic and gas exchange support of the basic VA circuit. This can be preplanned with initial VA ECMO institution for those deemed at risk for LV distension or may serve as a rescue technique, with one cannula draining the RA and the other draining the LA57. Two venous cannulas creates the ability to de-escalate biventricular venting support gradually with the removal of one cannula should the heart begin to recover.
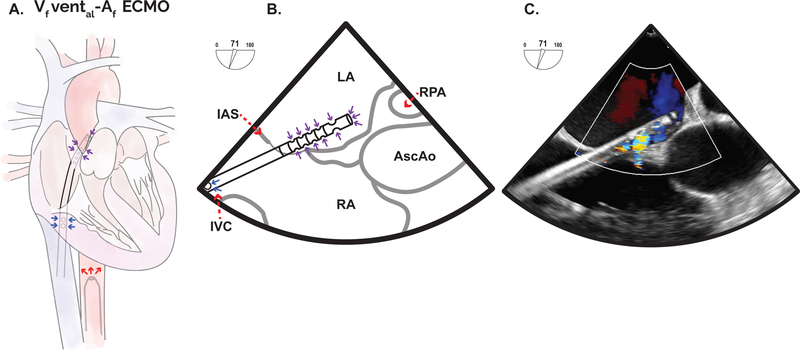
Alternatively, a single, multistage venous cannula can be used proactively when indirect LV venting is deemed necessary at initiation of support. To achieve both venous drainage as well as LA decompression, a transseptal puncture is performed and the distal drainage ports are positioned under TEE guidance within the LA (Figure 6) (Movies 23 and 24). A midesophageal bicaval view allows identification of the optimal transseptal puncture site in the superior-inferior plane and a midesophageal aortic valve short axis view provides anterior-inferior orientation with the desired puncture site in the “mid-mid” position. Biplane imaging and 3D supplementation may help facilitate optimal transseptal access. Once a wire is across, taking the time to orient the imaging plane to capture a view of the full length of the wire in long-axis (often obtained by starting with an aortic valve short axis view and increase to around 70 degrees on the Omniplane) will put the imager in the optimal position for visualizing the dilator and cannula as they enter the RA and move to cross the septum. The proximal ports then remain in the RA, at the inferior cavoatrial junction, or both depending on the cannula selected, to provide concurrent drainage of venous blood and simultaneous biatrial drainage. Color flow Doppler and 3D echocardiography can be used to visualize the flow and the ports in respect to their location in the RA and LA. 3D imaging allows for real time visualization of the cannula and its side ports, the IAS, the LA wall, the RA wall, and the aortic root (Movie 25)56.
Figure 6. Percutaneous, transseptal indirect left ventricular venting.
To address LV distention during VA-ECMO and indirect LV venting strategy may be employed. One approach is placement of a dedicated transseptal cannula across the IAS to provide LA drainage and consequent indirect LV venting. Alternatively, a single, multistage cannula may be employed for percutaneous transseptal cannulation with the distal inflow ports positioned within the LA and the proximal inflow ports within the inferior cavoatrial junction (A). As depicted (A) this represents both femoral venous drainage and left atrial venting in addition to the arterial return via the femoral artery. Purple arrows represent return of blood with variable oxygenation contingent upon the clinical scenario. A non-standard mid or upper esophageal window can demonstrate the position of a transseptal cannula and the inflow ports and can identify key intracardiac structures including the IAS, the RPA, and the AscAo (B). A TEE view demonstrating color Doppler inflow of blood into a multistage venous cannula positioned across the IAS (C).
al: left atrium, AscAo: Ascending aorta, ECMO: Extracorporeal membrane oxygenation, f: Femoral, IAS: Interatrial septum, IVC: Inferior vena cava, LA: Left atrium, LV: Left ventricle, RA: Right atrium, RPA: Right pulmonary artery.
Upon initiation of LAVA-ECMO, flows can be adjusted with TEE assistance with a goal of AV opening with each cardiac cycle (Movie 26)56. In the case of severe MR, initiation of LAVA-ECMO has been shown to improve pulmonary vein flow reversal as visualized by PWD to at least blunted systolic pulmonary vein flow58. Ventricular function is evaluated by TEE in the context of direct measurement of biventricular filling pressures to provide a more comprehensive picture of cardiac function after LAVA ECMO initiation. With LV decompression, a reduction in inotrope dosage, reduction in spontaneous echocardiography contrast, increased opening of the AV, resolution of arrhythmias, and improvement in pulmonary edema should be seen61. Alhussein et al. found significant improvement in pulmonary edema as viewed on chest x-ray in 7 patients with atrial septostomy with placement of a left atrial drainage cannula62.
As LAVA ECMO involves introduction of an iatrogenic atrial septal defect and placement of temporary cannulas for cardiopulmonary support, it is important to consider the implications of this new intracardiac shunt after resolution of ECMO. If cardiac function improves and the patient is successfully decannulated, the atrial septal defect can be repaired either percutaneously or surgically if clinically indicated based on size and direction of shunt, RV function and degree of pulmonary hypertension. In the case of severe persistent heart failure, ECMO would ultimately be switched for either a more durable device or the patient would be eligible and listed for transplant. Unfortunately, the mortality rate in this patient population receiving ECMO in need of left ventricular decompression is high and many patients do not survive due to multiorgan failure63.
Case reports have also presented percutaneous LV venting via pigtail catheter at the apex of the left ventricle, placed under ultrasound guidance or directly into the PA64. Should all minimally invasive and percutaneous interventions fail, sternotomy or thoracotomy and direct cannulation of the PA, pulmonary vein, LA, or LV and initiation of central ECMO can provide benefit by using larger cannulas and centrally bypassing the heart65. With an open surgical procedure, the patient must be under general anesthesia and mechanical ventilation, but this can be used as a last resort as a bridge to a more durable option.
VADs and ECMO
Ventricular assist devices (VADs) are traditionally considered durable implants and devices to aid a failing left or right ventricle. However, as numerous temporary percutaneous devices have been introduced (i.e., Protek Duo, Impella), enabling percutaneous transfer of the end-ventricular preload past the semilunar valves, the definition between “ventricular assist device” and “veno-arterial ECMO” has blurred. We first briefly discuss durable LVAD considerations and then focus our discussion predominately on echocardiography for temporary VADs.
As patients are bridged with VA-ECMO to placement of a durable left ventricular assist device (LVAD), some may develop subsequent RV failure due to changes in RV preload and afterload, altered RV chamber geometry, RV ischemia, and fluctuations in fluid balance66. TEE evaluation pre LVAD implantation should include thorough assessment of LV function and size, RV function and size, presence and/or severity of aortic insufficiency, mitral stenosis, thrombus, and interatrial communication. A full discussion of echocardiography for durable LVADs is beyond the scope of this review and the reader is referred to the recent and comprehensive ASE Guidelines for this specific patient population51. After LVAD implantation, the right ventricle is frequently supported with inotropic agents, pulmonary vasodilators (e.g. inhaled nitric oxide), judicious fluid management, and avoidance or minimization of situations leading to increased RV afterload (eg. adrenergic vasoconstrictors, acidosis, hypoxia, hypercapnia). In many cases however, mechanical support of the RV is necessary.
A temporary surgical implantation of a RVAD involves percutaneous RA drainage via either femoral or internal jugular venous access with outflow into the PA trunk, typically via a prosthetic graft anastomosed to the PA. Placement of the venous drainage cannula into the RA can be facilitated by TEE with care to ensure the tip of the cannula is not placed into the hepatic vein or perforates the atrial wall during positioning. Outflow into the PA trunk can be visualized with color flow Doppler and the PV should be evaluated to ensure no significant valvular disruption occurs after cannulation and flow initiation. Further evaluation of the RV with TEE and adjustment of the RVAD flows can be done simultaneously to optimize RV decompression.
The ProtekDuo Dual Lumen Catheter, as discussed above, is a percutaneous dual lumen, single cannula, ventricular support device designed to be inserted in the right internal jugular vein with RA drainage, compatibility with a centrifugal pump with or without an oxygenator based on gas exchange status, and outflow blood directed into the PA22. TEE guidance confirms correct positioning of the cannula with the distal tip in the PA and monitors RV function and decompression upon initiation of support. With percutaneous RVAD support patients can be extubated, mobilized, and ambulated. RV support can then be weaned as ventricular function recovers followed by percutaneous removal of the ProtekDuo cannula without necessitating a return to the operating room for removal of a PA graft66. The Impella RP (Abiomed) is another device designed specifically to support a failing RV and can provide flows > 4.0 lpm. An Impella RP is designed for femoral insertion and consists of a proximal inflow port positioned within the IVC and a distal outflow port positioned in the main PA. As discussed above, the ease of mobilization often may drive the ECMO team to prefer a jugular-SVC approach when feasible. An additional consideration is the potential of infection. Although the authors are not aware of specific literature in this population with respect comparing these devices, it is reasonable to consider femoral access sites as potentially at higher risk of infection.
VA-ECMO: Troubleshooting and ECMO emergencies
As VA ECMO partially bypasses the heart, especially with LV venting strategies, pericardial effusions may not lead to tamponade physiology until weaning is attempted. Injury to structures in the heart such as the TV or RA leading to pericardial effusion and tamponade are not uncommon and can be discovered by TEE67. The Protek Duo is placed in the main PA and consequently RV or PA perforation can occur while advancing the cannula. Cannula malposition is also a possibility as the distal cannula tip may migrates back into the RVOT68.
Low flow alarms are not uncommon with VA ECMO and TEE can confirm cannula positioning, suction events from the venous cannula, or presence of a thrombus at the cannula inflow. Depending on venous cannula positioning, RA or LA invagination can be seen due to high negative inflow cannula pressures (Movie 26)69. This invagination can easily be misidentified as a pericardial effusion or cannular malpositioning and should undergo thorough TEE evaluation and be interpreted in clinical context. In cases of thrombus formation at the distal cannula, TEE plays an imperative monitoring step in ensuring no thrombus remains in the body should the cannula be removed and replaced. It is not uncommon for thrombus to embolize into the lungs or systemically upon manipulation of these large bore cannulas32.
TEE allows for visualization of the opening and closure of the AV. As mentioned, failure of the AV to open is an indication the LV cannot generate enough power to overcome the higher afterload seen on peripheral VA-ECMO. This manifests as loss of pulsatility on the arterial line tracing and is diagnosed using TEE by noting dilation of the LV, spontaneous echo contrast present in the LV, and no or infrequent AV opening. In this scenario, the aforementioned medical, percutaneous, and surgical interventions of indirect and direct LV venting should be considered. With stasis noted in the aortic root, anticoagulation goals should be reevaluated to ensure prevention of thrombus formation. TEE can help guide anticoagulation management by identifying spontaneous echo contrast in the LV or aorta70.
If intrinsic cardiac function is maintained or begins to improve, differential hypoxemia can occur, in particular with femoral arterial cannulation71. Patients requiring ECMO universally undergo lung protective ventilation, however, if lung function is poor and there is sufficient native left ventricular function present, blood ejected from the left ventricle across the aortic valve can be poorly oxygenated72. This poorly oxygenated blood will eventually encounter the well oxygenated arterial ECMO blood from the femoral cannula and result in hypoxemic blood delivery to the aortic root and arch vessels73.
Color flow doppler in the descending aorta can determine mixing of VA ECMO support and native cardiac output, and the level at which mixing occurs can be indicative of cardiac recovery and weaning progress74. Use of a right radial arterial line can detect upper body hypoxemia by blood gas analysis. A narrow pulse-pressure indicates the watershed area may be closer to the aortic root, where a wider pressure suggests a more distal area of mixing73. Should differential hypoxia be determined to be a valid problem, ECMO flow can be increased and titrated by color flow doppler, visualization of the opening of the aortic valve, and serial blood gases to improve oxygenation of the upper extremity and aortic root74.
When cannulating for ECMO or during ECMO reconfiguring efforts, TEE plays in important role in actively monitoring for air entrainment. Although great care is taken by proceduralists to avoid air entrainment, and flows are reduced (or temporarily stopped) when reconfiguring, the risk of air embolism remains ever-present and of high potential consequence. Continuous imaging of the intracardiac chambers during high-risk portions of the procedure will readily identify the presence of even a small amount of intracardiac air and the proceduralist and perfusionist should be promptly alerted.
VA-ECMO: Weaning and Post-ECMO exam
Currently, there are no recognized echocardiography protocols for weaning of VA-ECMO. In the opinion of these authors, comprehensive, interdisciplinary societal consensus guidelines in this area are needed to develop consistency across institutions and provide a basis for additional research and investigation. In the interim, TEE evaluation for improvement of cardiac function as well as guidelines pertaining to evaluation of patients with VADs can assist in clinical decision making.
As the LV begins to recover, invasive monitors such as an arterial line may show improvement and increased pulsatility. Cardiac output monitoring seen on PA catheter will not give an accurate assessment of cardiac function as a blood is bypassing the PA. According to Extracorporeal Life Support Organization (ELSO) guidelines, hepatic function should be recovered before an attempt to wean ECMO, however, there is no recommendation for return of renal function75. In addition, pulmonary oxygenation of blood must be intact with PaO2/FiO2 ratio greater than 200 with 0.21 FiO2 delivered on the ECMO circuit and 0.60 FiO2 with the ventilator.
TEE findings such as an improvement of EF, an LV outflow tract velocity-time integral (VTI) greater than 10cm, absence of LV dilation, and increased AV opening could indicate recovery of LV function. Aissaoui et al explored alternative measurements by changing ECMO loading parameters and viewing blood pressure, transmitral E velocity, tissue Doppler of the lateral annulus (e’ and S’), E/e’, VTI of the AV, and strain76. Though currently unvalidated, these quantitative measures can offer guidance as a patient’s progress toward weaning begins77. Ortuno et al found in a systemic review (including the study described above), factors more predictive of successful LV weaning include an AV VTI greater than 10cm, an LVEF greater than 20%, and a lateral mitral annulus velocity S’ greater than 8cm/s27. Improvement of EF after cannulation of VA ECMO of at least 25% EF at time of ECMO weaning was found to be indicative of successful discontinuation of ECMO in 129 patients78. Consistent with these findings is recent data from Kim et al. demonstrating that lateral e’ velocity as well as tricuspid annular S’ velocity both improved in patients who were then successfully weaned from VA-ECMO79. Importantly, in this same cohort the authors also looked at previously published criteria for weaning including an LVEF >20–25%, LV VTI >/= 10cm, and a mitral annular S’ >/= 6cm/sec. They demonstrated that 30% of patients who were unable to be weaned off support met all three criteria for weaning while only 16% of patients successfully weaned met all three79.
As discussed in the context of VV-ECMO weaning, factors indicative of potential successful RV function after weaning included a smaller RV size, increased RVEF, higher RV FAC, and higher magnitudes of RV strain. TAPSE and TR were not found to be associated with successful RV weaning27. If more intracardiac volume and preload gets delivered to the RV due to decreased venous drainage and the RV cannot tolerate the volume in combination with possible increased PA pressures, TR and elevation in RA pressures could open a PFO to precipitate hypoxemia with a right-to-left shunt.4
As ECMO flows are weaned 0.5 to 1L at a time, heart rate, blood pressure and arterial oxygen saturation (preferably from a right sided arterial line), central venous pressure, and PA pressures can all be monitored to direct weaning. TEE can provide additional information including stroke volume, LV and RV size and function, and AV efficacy80. During weaning, LV and RV interdependence is present and should one ventricle struggle with the decreased support, it can lead to failure of the other ventricle81. Both LV and RV size and function should be evaluated during weaning of extracorporeal support and if distension and impending failure of either ventricle is noted it may warrant discontinuation of the weaning trial.
Future avenues for study
Currently there are no echocardiography guidelines for VA-ECMO and physicians rely on prior clinical experience, expert opinion, limited peer-reviewed literature, and extrapolations from existing echocardiography guidelines (including VAD guidelines)82. Guidelines for a complete and comprehensive pre cannulation examination, positioning of the variety of cannulas present and available, monitoring during ECMO support, weaning, and post cannulation examination need to be developed to facilitate clinical care, education, and research in this high risk and complex patient population.
Suggested echocardiography recommendations for institutions with an ECMO program should include a pre-ECMO assessment meeting diagnostic standards for assessment of valve function and pathology, evaluation and troubleshooting common problems on VA ECMO, and standard proposals for ECMO weaning with echocardiographic evaluation of cardiopulmonary recovery83.
Conclusion
Echocardiography is a powerful and significant asset in the assessment of patients undergoing extracorporeal support. Echocardiography can facilitate pre-procedure screening and assessment, guidance of cannulation and ECMO initiation, troubleshooting of problems or emergencies that may arise during ECMO, and facilitate weaning and assessment of recovery. Physicians performing echocardiography for these complex and critically ill patients need to understand the relevant anatomical considerations, cannula design, function, and positioning, the relevant hemodynamic context, and the limitations of the imaging modality. Additional research is warranted is this rapidly expanding field. Interdisciplinary consensus guidelines for echocardiography in patients undergoing ECMO are needed to facilitate improved clinical care, echocardiographic education, and to support future research in this area.
Supplementary Material
Movie 1. RV dysfunction and dilation with severe TR and increased right atrial pressure. Midesophageal 4-chamber view focused on the tricuspid valve demonstrating severe, eccentric tricuspid regurgitation directed towards the IAS in a patient with a dilated RV with reduced systolic function. The IAS bows significantly to the left, consistent with pressure overload of the right atrium. IAS: interatrial septum, RV: right ventricle, TR: tricuspid regurgitation.
Movie 2. RV dysfunction and dilation with severe TR and increased right atrial pressure. Midesophageal RV inflow-outflow view with color flow Doppler demonstrating severe tricuspid regurgitation, a dilated RV, and hypokinesis of the RV freewall. The IAS bows significantly to the left, consistent with pressure overload of the right atrium. IAS: interatrial septum, RV: right ventricle, TR: tricuspid regurgitation.
Movie 3. PFO with left to right flow. A modified midesophageal bicaval view with color flow Doppler demonstrating left to right flow across a PFO. A pulmonary artery catheter is visualized within the right atrium. PFO: patent foramen ovale.
Movie 5. Confirmation of a guidewire within the RA adjacent to a preexisting pacing lead. A modified midesophageal bicaval view focused on the SVC demonstrates the “J-tip” of a guidewire within the RA and adjacent to a preexisting pacemaker lead. Care is required to correctly identify intracardiac wires and not confuse indwelling leads or catheters for guidewires. LA: left atrium, RA: right atrium, SVC: superior vena cava.
Movie 6. Biplane imaging to confirm presence of a guidewire within the SVC in a patient with two preexisting pacemaker leads. A midesophageal bicaval view with biplane imaging through the SVC (image on the right, in short axis) demonstrates three distinct wires within the SVC in a patient with two preexisting pacemaker leads undergoing guidewire placement from the right IJ approach. In a single, 2D long axis view, a lead or wire may be out-of-plane. Biplane imaging may facilitate easier identification of a guidewire as it is advanced into position. 2D: two-dimensional, IJ: internal jugular vein, SVC: superior vena cava.
Movie 4. A persistent left SVC identified by agitated saline injection. A deep esophageal 4-chamber view demonstrates a dilated RV with a flattened IVS. A dilated coronary sinus orifice is noted and injection of agitated saline through a left upper extremity reveals early opacification of the coronary sinus prior to the right atrium consistent with presence of a persistent left SVC. CS: coronary sinus, IVS: interventricular septum, LV: left ventricle, RV: right ventricle, TV: tricuspid valve.
Movie 8. Reverberation artifact from guidewire within the RA using biplane imaging. A biplane image obtained from a midesophageal modified bicaval view demonstrates a short-axis view of the guidewire. Here it is readily apparent that the reverberation artifact is located directly “below” the guidewire consistent with artifact rather than a second guidewire. RA: right atrium.
Movie 9. Vf-Vj-ECMO cannulas visualized near the superior and inferior cavoatrial junctions. A midesophageal bicaval view demonstrates a femoral venous drainage cannula noted within the RA just past the inferior cavoatrial junction and a jugular venous return cannula with the tip within the RA near the superior cavoatrial junction in a patient on Vf-Vj-ECMO. f: femoral, j: jugular, LA: left atrium, RA: right atrium.
Movie 7. Reverberation artifact creating appearance of two guidewires within the right atrium. A midesophageal modified bicaval view illustrating how reverberation artifact due to a strong reflector (such as a guidewire) can create the illusion of a second guidewire within the right atrium. LA: left atrium, RA: right atrium.
Movie 10. Vf-Vj-ECMO cannulas with color Doppler imaging demonstrating blood flow. A midesophageal bicaval view demonstrates a femoral venous drainage cannula noted within the RA just past the inferior cavoatrial junction and a jugular venous return cannula with the tip within the RA near the superior cavoatrial junction in a patient on Vf-Vj-ECMO. Color Doppler imaging demonstrates flow (drainage) into to femoral cannula and flow emanating from the jugular cannula (return). f: femoral, j: jugular, LA: left atrium, RA: right atrium.
Movie 12. Color Doppler imaging used to assess orientation and position of the central outflow port in a patient undergoing (bc)Vj-V-ECMO. A color Doppler midesophageal bicaval view focus on the central outflow port of a Crescent MC3 (Cardiopulmonary, Dexter, MI) bicaval ECMO cannula. The outflow port should be oriented above the tricuspid valve such that flow will be directed across the valve orifice. bc: bicaval, IVC: inferior vena cava, j: jugular, LA: left atrium, RA: right atrium.
Movie 13. A Protek Duo cannula visualized traversing the RV in a patient undergoing (dl)Vj-P-ECMO. This midesophageal RV inflow-outflow view demonstrates a Protek Duo traversing the RV with the tip (outflow port) located within the main pulmonary artery. The inflow port is located within the RA and is not visualized in this image. AV: aortic valve, dl: double lumen, j: jugular, LA: left atrium, PV: pulmonic valve, RA: right atrium, RV: right ventricle.
Movie 11. A bicaval cannula is visualized to confirm appropriate positioning of the central outflow port in a patient undergoing (bc)Vj-V-ECMO. A midesophageal bicaval view focus on the central outflow port of a Crescent MC3 (Cardiopulmonary, Dexter, MI) bicaval ECMO cannula. The outflow port should be oriented above the tricuspid valve such that flow will be directed across the valve orifice. bc: bicaval, IVC: inferior vena cava, j: jugular, LA: left atrium, RA: right atrium.
Movie 15. 3D image demonstrating a Protek Duo cannula in a patient undergoing (dl)Vj-P-ECMO. A Protek Duo cannula is visualized within the RV with the tip located just distal to the pulmonic valve. AV: aortic valve, dl: double lumen, j: jugular, LA: left atrium, RV: right ventricle.
Movie 14. Color Doppler demonstrating outflow into the main PA from a Protek Duo cannula in a patient undergoing (dl)Vj-P-ECMO. A midesophageal RV inflow-outflow view focused on the pulmonic valve demonstrates color Doppler flow emanating from the tip (outflow port) located within the main pulmonary artery. AV: aortic valve, dl: double lumen, j: jugular, LA: left atrium, RV: right ventricle.
Movie 17. Transthoracic echocardiography. Parasternal long axis view demonstrates pericardial effusion (PE). The arrow points to diastolic collapse of the right ventricle (RV) suggestive of cardiac tamponade. AO=Aorta; DA= Descending thoracic aorta; LA=Left atrium; LV= left ventricle. Reproduced with permission from Nanda NC (Ed) Interesting Cases in Echocardiography (2017), Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India
Movie 16. A TEE clip of a patient with an acute aortic dissection. A color Doppler midesophageal aortic valve long-axis view in a patient with an acute aortic dissection demonstrating a dissection flap in the aortic root, a dilated aortic root with effacement of the sinuses of Valsalva, a flail right coronary cusp, severe eccentric holodiastolic aortic regurgitation, and a pericardial effusion. LA: left atrium, LV: left ventricle, RV: right ventricle, TEE: transesophageal echocardiography
Movie 18. Transthoracic echocardiography. The arrow in the apical 4-chamber view points to a large, mobile thrombus in the apex of the left ventricle (LV) with depressed function. LA=Left atrium; RA= Right atrium; RV= Right ventricle. Reproduced with permission from Nanda NC (Ed) Interesting Cases in Echocardiography (2017), Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India
Movie 19. Transthoracic echocardiography. Subcostal view shows a dilated, non-collapsing inferior vena cava (IVC) consistent with increased right atrial pressure. HV=hepatic vein; L=Liver. Reproduced with permission from Nanda NC (Ed) Interesting Cases in Echocardiography (2017), Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India
Movie 20. Aortic insufficiency noted prior to initiation of peripheral VA-ECMO. A midesophageal aortic valve long-axis view with color Doppler illustrates trace, early diastolic aortic insufficiency prior to initiation of peripheral VA-ECMO. The aortic valve is heavily calcified with poor cusp mobility. Spontaneous echo contrast (“smoke”) is visualized within the LA consistent with a low-flow state. LA: left atrium, LV: left ventricle.
Movie 21. Increased aortic insufficiency after initiation of peripheral VA-ECMO. A midesophageal aortic valve long-axis view with color Doppler demonstrates mild, central, holodiastolic aortic insufficiency after institution of peripheral VA-ECMO. The previously noted spontaneous echo contrast is no longer visualized. The calcific aortic valve opens with every cardiac cycle. LA: left atrium, LV: left ventricle.
Movie 22. An aortic valve that does not open during systole in a patient on peripheral VA-ECMO. A midesophageal long-axis view with color Doppler demonstrates an aortic valve that does not open at any point during the cardiac cycle in a patient on peripheral VA-ECMO as a bridge to successful durable LVAD implantation. AV: aortic valve, LA: left atrium, LV: left ventricle, LVAD: left ventricular assist device.
Movie 24. LAVA-ECMO. A non-standard midesophageal view demonstrating a multistage venous cannula with the tip and distal inflow ports positioned within the LA to facilitate indirect left ventricular venting in a patient on peripheral VA-ECMO. AV: aortic valve, IAS: interatrial septum, LA: left atrium, LAVA: left atrial veno-arterial, RA: right atrium, RV: right ventricle.
Movie 25. 3D mitral en face view demonstrating transseptal cannula positioning during LAVA-ECMO. A 3D en face view of the mitral valve with the AV at the 12 o’clock position demonstrates a multistage venous cannula positioned across the IAS to achieve indirect LV venting in a patient undergoing LAVA-ECMO. The distal inflow ports appear echolucent in this image. 3D: three-dimensional, AV: aortic valve, IAS: interatrial septum, LAVA: left atrial veno-arterial, MV: mitral valve, RA: right atrium.
Movie 23. Transseptal access obtained for institution of LAVA-ECMO. A non-standard midesophageal view demonstrating a dilator advanced across the IAS prior to placement of a multistage venous cannula within the LA to facilitate indirect left ventricular venting in a patient on peripheral VA-ECMO. AV: aortic valve, IAS: interatrial septum, LA: left atrium, LAVA: left atrial veno-arterial, RA: right atrium, RV: right ventricle.
Movie 26. Left atrial decompression with initiation of LAVA-ECMO. A midesophageal long axis view demonstrates left atrial collapse at the moment when inflow from the left atrium is established. The multistage venous cannula is visualized within the LA. Titration of flow is necessary to prevent excessive left atrial collapse and find a balance point where chamber decompression is facilitated but left atrial collapse does not occur. LA: left atrium, LAVA: left atrial veno-arterial, LV: left ventricle, RA: right atrium, RV: right ventricle.
Funding Statement:
Dr. Addis is supported by the National Institutes of Health (NIH) grant number T32HL129948
Footnotes
Financial Disclosures: Drs. Addis, Hussey, Nanda, have no financial disclosures to declare. Dr. Von Mering serves on the peripheral vascular medical advisory board for Boston Scientific Corporation and is a speaker regarding left atrial appendage closure. Dr. Ahmed serves as a consultant and proctor for Abbott, Edwards, and Medtronic.
Clinical trial number and registry URL: Not applicable
Prior Presentations: None
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
References
- 1.Grant C Jr., Richards JB, Frakes M, Cohen J, Wilcox SR. ECMO and Right Ventricular Failure: Review of the Literature. J Intensive Care Med. 2021;36(3):352–360. [DOI] [PubMed] [Google Scholar]
- 2.Bouferrache K, Vieillard-Baron A. Acute respiratory distress syndrome, mechanical ventilation, and right ventricular function. Curr Opin Crit Care. 2011;17(1):30–35. [DOI] [PubMed] [Google Scholar]
- 3.Hahn RT, Abraham T, Adams MS, et al. Guidelines for performing a comprehensive transesophageal echocardiographic examination: recommendations from the American Society of Echocardiography and the Society of Cardiovascular Anesthesiologists. J Am Soc Echocardiogr. 2013;26(9):921–964. [DOI] [PubMed] [Google Scholar]
- 4.Doufle G, Roscoe A, Billia F, Fan E. Echocardiography for adult patients supported with extracorporeal membrane oxygenation. Crit Care. 2015;19:326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rudski LG, Lai WW, Afilalo J, et al. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr. 2010;23(7):685–713; quiz 786–688. [DOI] [PubMed] [Google Scholar]
- 6.Addetia K, Muraru D, Badano LP, Lang RM. New Directions in Right Ventricular Assessment Using 3-Dimensional Echocardiography. JAMA Cardiol. 2019;1;4(9):936–944. [DOI] [PubMed] [Google Scholar]
- 7.Lakatos BK, Nabeshima Y, Tokodi M, et al. Importance of Nonlongitudinal Motion Components in Right Ventricular Function: Three-Dimensional Echocardiographic Study in Healthy Volunteers. J Am Soc Echocardiogr. 2020;33(8):995–1005.e1. [DOI] [PubMed] [Google Scholar]
- 8.Arkles JS, Opotowsky AR, Ojeda J, et al. Shape of the right ventricular Doppler envelope predicts hemodynamics and right heart function in pulmonary hypertension. Am J Respir Crit Care Med. 2011;183(2):268–276. [DOI] [PubMed] [Google Scholar]
- 9.Tello K, Dalmer A, Axmann J, et al. Reserve of Right Ventricular-Arterial Coupling in the Setting of Chronic Overload. Circ Heart Fail. 2019;12(1):e005512. [DOI] [PubMed] [Google Scholar]
- 10.Jone PN, Schafer M, Pan Z, Ivy DD. Right Ventricular-Arterial Coupling Ratio Derived From 3-Dimensional Echocardiography Predicts Outcomes in Pediatric Pulmonary Hypertension. Circ Cardiovasc Imaging. 2019;12(1):e008176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Aubert R, Venner C, Huttin O, et al. Three-Dimensional Echocardiography for the Assessment of Right Ventriculo-Arterial Coupling. J Am Soc Echocardiogr. 2018;31(8):905–915. [DOI] [PubMed] [Google Scholar]
- 12.Kronzon I, Tunick PA, Jortner R, et al. Echocardiographic evaluation of the coronary sinus. J Am Soc Echocardiogr. 1995;8(4):518–526. [DOI] [PubMed] [Google Scholar]
- 13.Hemamalini P, Dutta P, Attawar S. Transesophageal Echocardiography Compared to Fluoroscopy for Avalon Bicaval Dual-Lumen Cannula Positioning for Venovenous ECMO. Ann Card Anaesth. 2020;23(3):283–287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Le HT, Hangiandreou N, Timmerman R, et al. Imaging Artifacts in Echocardiography. Anesth Analg. 2016;122(3):633–646. [DOI] [PubMed] [Google Scholar]
- 15.Peris A, Lazzeri C, Cianchi G, et al. Clinical significance of echocardiography in patients supported by venous-venous extracorporeal membrane oxygenation. J Artif Organs. 2015;18(2):99–105. [DOI] [PubMed] [Google Scholar]
- 16.Ahn HJ, Lee JW, Joo KH, You YH, Ryu S, Lee JW, Kim SW. Point-of-Care Ultrasound-Guided Percutaneous Cannulation of Extracorporeal Membrane Oxygenation: Make it Simple. J Emerg Med. 2018;54(4):507–513. [DOI] [PubMed] [Google Scholar]
- 17.Tipograf Y, Gannon WD, Foley NM, et al. A Dual-Lumen Bicaval Cannula for Venovenous Extracorporeal Membrane Oxygenation. Ann Thorac Surg. 2020;109(4):1047–1053. [DOI] [PubMed] [Google Scholar]
- 18.Ravichandran AK, Baran DA, Stelling K, Cowger JA, Salerno CT. Outcomes with the Tandem Protek Duo Dual-Lumen Percutaneous Right Ventricular Assist Device. ASAIO J. 2018;64(4):570–572. [DOI] [PubMed] [Google Scholar]
- 19.Group MG. Instructions for Use Avalon Elite® Bi-Caval Dual Lumen Catheter. Rastatt, Germany: Maquet Cardiopulmonary GmbH; 2017. [Google Scholar]
- 20.Cardiopulmonary M Crescent™ Jugular Dual Lumen Catheter Instructions for Use. Dexter, MI: MC3 Cardiopulmonary; 2016. [Google Scholar]
- 21.Abrams D, Brodie D, Javidfar J, et al. Insertion of bicaval dual-lumen cannula via the left internal jugular vein for extracorporeal membrane oxygenation. ASAIO J. 2012;58(6):636–637. [DOI] [PubMed] [Google Scholar]
- 22.LivaNova. Procedure Guide. Pittsburgh, PA: CardiacAssist, Inc; 2019. [Google Scholar]
- 23.Bermudez CA, Rocha RV, Sappington PL, Toyoda Y, Murray HN, Boujoukos AJ. Initial experience with single cannulation for venovenous extracorporeal oxygenation in adults. Ann Thorac Surg. 2010;90(3):991–995. [DOI] [PubMed] [Google Scholar]
- 24.Rubino A, Vuylsteke A, Jenkins DP, Fowles JA, Hockings L, Valchanov K. Direct complications of the Avalon bicaval dual-lumen cannula in respiratory extracorporeal membrane oxygenation (ECMO): Single-center experience. Int J Artif Organs. 2014;37(10):741–747. [DOI] [PubMed] [Google Scholar]
- 25.Shekar K, Buscher H, Brodie D. Protocol-driven daily optimisation of venovenous extracorporeal membrane oxygenation blood flows: an alternate paradigm? J Thorac Dis. 2020;12(11):6854–6860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Grant AA, Hart VJ, Lineen EB, et al. A Weaning Protocol for Venovenous Extracorporeal Membrane Oxygenation With a Review of the Literature. Artif Organs. 2018;42(6):605–610. [DOI] [PubMed] [Google Scholar]
- 27.Ortuno S, Delmas C, Diehl JL, et al. Weaning from veno-arterial extra-corporeal membrane oxygenation: which strategy to use? Ann Cardiothorac Surg. 2019;8(1):E1–E8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Badulak J, Antonini MV, Stead CM, et al. ECMO for COVID-19: Updated 2021 Guidelines from the Extracorporeal Life Support Organization (ELSO). ASAIO J. 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Full COVID-19 Registry Dashboard. 2021. https://www.elso.org/Registry/FullCOVID19RegistryDashboard.aspx. Accessed March 9, 2021.
- 30.Kirkpatrick JN, Mitchell C, Taub C, Kort S, Hung J, Swaminathan M. ASE Statement on Protection of Patients and Echocardiography Service Providers During the 2019 Novel Coronavirus Outbreak: Endorsed by the American College of Cardiology. J Am Soc Echocardiogr. 2020;33(6):648–653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kirkpatrick JN, Mitchell C, Taub C, Kort S, Hung J, Swaminathan M. ASE Statement on Protection of Patients and Echocardiography Service Providers During the 2019 Novel Coronavirus Outbreak: Endorsed by the American College of Cardiology. J Am Coll Cardiol. 2020;75(24):3078–3084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Platts DG, Sedgwick JF, Burstow DJ, Mullany DV, Fraser JF. The role of echocardiography in the management of patients supported by extracorporeal membrane oxygenation. J Am Soc Echocardiogr. 2012;25(2):131–141. [DOI] [PubMed] [Google Scholar]
- 33.Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2015;28(1):1–39 e14. [DOI] [PubMed] [Google Scholar]
- 34.Ki KK, Passmore MR, Chan CHH, et al. Low flow rate alters haemostatic parameters in an ex-vivo extracorporeal membrane oxygenation circuit. Intensive Care Med Exp. 2019;7(1):51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2015;16(3):233–270. [DOI] [PubMed] [Google Scholar]
- 36.Lucas SK, Schaff HV, Flaherty JT, Gott VL, Gardner TJ. The harmful effects of ventricular distention during postischemic reperfusion. Ann Thorac Surg. 1981;32(5):486–494. [DOI] [PubMed] [Google Scholar]
- 37.Radakovic D, Hamouda K, Penov K, et al. Central Versus Peripheral Arterial Cannulation for Veno-Arterial Extracorporeal Membrane Oxygenation in Post-Cardiotomy Patients. ASAIO J. 2021;67(1):67–73. [DOI] [PubMed] [Google Scholar]
- 38.Michels G, Wengenmayer T, Hagl C, et al. Recommendations for extracorporeal cardiopulmonary resuscitation (eCPR): consensus statement of DGIIN, DGK, DGTHG, DGfK, DGNI, DGAI, DIVI and GRC. Clin Res Cardiol. 2019;108(5):455–464. [DOI] [PubMed] [Google Scholar]
- 39.Chocron S, Perrotti A, Durst C, Aupecle B. Left ventricular venting through the right subclavian artery access during peripheral extracorporeal life support. Interact Cardiovasc Thorac Surg. 2013;17(1):187–189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Aiyagari RM, Rocchini AP, Remenapp RT, Graziano JN. Decompression of the left atrium during extracorporeal membrane oxygenation using a transseptal cannula incorporated into the circuit. Crit Care Med. 2006;34(10):2603–2606. [DOI] [PubMed] [Google Scholar]
- 41.Rao P, Khalpey Z, Smith R, Burkhoff D, Kociol RD. Venoarterial Extracorporeal Membrane Oxygenation for Cardiogenic Shock and Cardiac Arrest. Circ Heart Fail. 2018;11(9):e004905. [DOI] [PubMed] [Google Scholar]
- 42.Xie A, Forrest P, Loforte A. Left ventricular decompression in veno-arterial extracorporeal membrane oxygenation. Ann Cardiothorac Surg. 2019;8(1):9–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kowalewski M, Malvindi PG, Zielinski K, et al. Left Ventricle Unloading with Veno-Arterial Extracorporeal Membrane Oxygenation for Cardiogenic Shock. Systematic Review and Meta-Analysis. J Clin Med. 2020;9(4). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Al-Fares AA, Randhawa VK, Englesakis M, et al. Optimal Strategy and Timing of Left Ventricular Venting During Veno-Arterial Extracorporeal Life Support for Adults in Cardiogenic Shock: A Systematic Review and Meta-Analysis. Circ Heart Fail. 2019;12(11):e006486. [DOI] [PubMed] [Google Scholar]
- 45.Cheng R, Hachamovitch R, Makkar R, et al. Lack of Survival Benefit Found With Use of Intraaortic Balloon Pump in Extracorporeal Membrane Oxygenation: A Pooled Experience of 1517 Patients. J Invasive Cardiol. 2015;27(10):453–458. [PubMed] [Google Scholar]
- 46.Petroni T, Harrois A, Amour J, et al. Intra-aortic balloon pump effects on macrocirculation and microcirculation in cardiogenic shock patients supported by venoarterial extracorporeal membrane oxygenation*. Crit Care Med. 2014;42(9):2075–2082. [DOI] [PubMed] [Google Scholar]
- 47.Cheng A, Swartz MF, Massey HT. Impella to unload the left ventricle during peripheral extracorporeal membrane oxygenation. ASAIO J. 2013;59(5):533–536. [DOI] [PubMed] [Google Scholar]
- 48.Fiedler AG, Dalia A, Axtell AL, et al. Impella Placement Guided by Echocardiography Can Be Used as a Strategy to Unload the Left Ventricle During Peripheral Venoarterial Extracorporeal Membrane Oxygenation. J Cardiothorac Vasc Anesth. 2018;32(6):2585–2591. [DOI] [PubMed] [Google Scholar]
- 49.Patel SM, Lipinski J, Al-Kindi SG, et al. Simultaneous Venoarterial Extracorporeal Membrane Oxygenation and Percutaneous Left Ventricular Decompression Therapy with Impella Is Associated with Improved Outcomes in Refractory Cardiogenic Shock. ASAIO J. 2019;65(1):21–28. [DOI] [PubMed] [Google Scholar]
- 50.Schrage B, Becher PM, Bernhardt A, et al. Left Ventricular Unloading Is Associated With Lower Mortality in Patients With Cardiogenic Shock Treated With Venoarterial Extracorporeal Membrane Oxygenation: Results From an International, Multicenter Cohort Study. Circulation. 2020;142(22):2095–2106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Stainback RF, Estep JD, Agler DA, et al. Echocardiography in the Management of Patients with Left Ventricular Assist Devices: Recommendations from the American Society of Echocardiography. J Am Soc Echocardiogr. 2015;28(8):853–909. [DOI] [PubMed] [Google Scholar]
- 52.Hacking DF, Best D, d’Udekem Y, et al. Elective decompression of the left ventricle in pediatric patients may reduce the duration of venoarterial extracorporeal membrane oxygenation. Artif Organs. 2015;39(4):319–326. [DOI] [PubMed] [Google Scholar]
- 53.Amancherla K, Menachem JN, Shah AS, Lindenfeld J, O’Leary J. Limited Balloon Atrial Septostomy for Left Ventricular Unloading in Peripheral Extracorporeal Membrane Oxygenation. J Card Fail. 2021;27(4):501–504. [DOI] [PubMed] [Google Scholar]
- 54.Bernhardt AM, Hillebrand M, Yildirim Y, et al. Percutaneous left atrial unloading to prevent pulmonary oedema and to facilitate ventricular recovery under extracorporeal membrane oxygenation therapy. Interact Cardiovasc Thorac Surg. 2018;26(1):4–7. [DOI] [PubMed] [Google Scholar]
- 55.Kapur NK, Esposito M. Hemodynamic support with percutaneous devices in patients with heart failure. Heart Fail Clin. 2015;11(2):215–230. [DOI] [PubMed] [Google Scholar]
- 56.Addis DR, Wang B, Prejean SP, Watts TE 3rd, Nanda NC, Ahmed MI. Utility of three-dimensional transesophageal echocardiography to guide transseptal positioning of a single multistage venous cannula to provide both venous drainage and indirect left ventricular venting in veno-arterial extracorporeal membrane oxygenation. Echocardiography. 2020;37(11):1860–1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jumean M, Pham DT, Kapur NK. Percutaneous bi-atrial extracorporeal membrane oxygenation for acute circulatory support in advanced heart failure. Catheter Cardiovasc Interv. 2015;85(6):1097–1099. [DOI] [PubMed] [Google Scholar]
- 58.Meers JB, Nanda NC, Watts TE 3rd, et al. Utility of transesophageal echocardiography to assess real time left atrial pressure changes and dynamic mitral regurgitation following placement of transseptal multistage venous cannula for systemic venous drainage and indirect left ventricular venting in venoarterial extracorporeal membrane oxygenation. Echocardiography. 2021. [DOI] [PubMed] [Google Scholar]
- 59.Weber C, Deppe AC, Sabashnikov A, et al. Left ventricular thrombus formation in patients undergoing femoral veno-arterial extracorporeal membrane oxygenation. Perfusion. 2018;33(4):283–288. [DOI] [PubMed] [Google Scholar]
- 60.Weil BR, Konecny F, Suzuki G, Iyer V, Canty JM Jr. Comparative Hemodynamic Effects of Contemporary Percutaneous Mechanical Circulatory Support Devices in a Porcine Model of Acute Myocardial Infarction. JACC Cardiovasc Interv. 2016;9(22):2292–2303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Burkhoff D, Naidu SS. The science behind percutaneous hemodynamic support: a review and comparison of support strategies. Catheter Cardiovasc Interv. 2012;80(5):816–829. [DOI] [PubMed] [Google Scholar]
- 62.Alhussein M, Osten M, Horlick E, et al. Percutaneous left atrial decompression in adults with refractory cardiogenic shock supported with veno-arterial extracorporeal membrane oxygenation. J Card Surg. 2017;32(6):396–401. [DOI] [PubMed] [Google Scholar]
- 63.ECLS Registry Report. 2021. https://www.elso.org/Registry/Statistics.aspx. Accessed March 9, 2021.
- 64.Hong TH, Byun JH, Yoo BH, Hwang SW, Kim HY, Park JH. Successful Left-Heart Decompression during Extracorporeal Membrane Oxygenation in an Adult Patient by Percutaneous Transaortic Catheter Venting. Korean J Thorac Cardiovasc Surg. 2015;48(3):210–213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Weymann A, Schmack B, Sabashnikov A, et al. Central extracorporeal life support with left ventricular decompression for the treatment of refractory cardiogenic shock and lung failure. J Cardiothorac Surg. 2014;9:60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Schmack B, Weymann A, Popov AF, et al. Concurrent Left Ventricular Assist Device (LVAD) Implantation and Percutaneous Temporary RVAD Support via CardiacAssist Protek-Duo TandemHeart to Preempt Right Heart Failure. Med Sci Monit Basic Res. 2016;22:53–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Chimot L, Marque S, Gros A, et al. Avalon(c) bicaval dual-lumen cannula for venovenous extracorporeal membrane oxygenation: survey of cannula use in France. ASAIO J. 2013;59(2):157–161. [DOI] [PubMed] [Google Scholar]
- 68.Hirose H, Yamane K, Marhefka G, Cavarocchi N. Right ventricular rupture and tamponade caused by malposition of the Avalon cannula for venovenous extracorporeal membrane oxygenation. J Cardiothorac Surg. 2012;7:36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Adams AJ, Guck AN, Shillcutt SK. Right Atrial Inversion Mimicking Right Atrial Mass in the Setting of Cardiac Tamponade. J Cardiothorac Vasc Anesth. 2019;33(8):2351–2355. [DOI] [PubMed] [Google Scholar]
- 70.Cardiac ultrasound and extracorporeal life support: the two go together. J Am Soc Echocardiogr. 2015;28(4):A18–19. [DOI] [PubMed] [Google Scholar]
- 71.Alexis-Ruiz A, Ghadimi K, Raiten J, et al. Hypoxia and Complications of Oxygenation in Extracorporeal Membrane Oxygenation. J Cardiothorac Vasc Anesth. 2019;33(5):1375–1381. [DOI] [PubMed] [Google Scholar]
- 72.Juan IC, Poorsattar SP, Thistlethwaite PA, et al. The Anterior Mediastinal Mass – the challenging management of hypoxemia despite peripheral cannulation for venoarterial ECMO. 2021. [DOI] [PubMed] [Google Scholar]
- 73.Antoniucci ME, De Paulis S, Bevilacqua F, et al. Unconventional Cannulation Strategy in Peripheral Extracorporeal Membrane Oxygenation to Achieve Central Perfusion and Prevent Differential Hypoxia. J Cardiothorac Vasc Anesth. 2019;33(5):1367–1369. [DOI] [PubMed] [Google Scholar]
- 74.Konishi H, Misawa Y, Nakagawa Y, Fuse K. Doppler aortic flow pattern in the recovering heart treated by cardiac extracorporeal membrane oxygenation International Society for Artificial Organs. Artif Organs. 1999;23(4):367–369. [DOI] [PubMed] [Google Scholar]
- 75.Organization ELS. Guidelines for Adult Respiratory Failure. 2017; https://www.elso.org/Portals/0/ELSO%20Guidelines%20For%20Adult%20Respiratory%20Failure%201_4.pdf. Accessed March 9, 2021, 2021.
- 76.Aissaoui N, Guerot E, Combes A, et al. Two-dimensional strain rate and Doppler tissue myocardial velocities: analysis by echocardiography of hemodynamic and functional changes of the failed left ventricle during different degrees of extracorporeal life support. J Am Soc Echocardiogr. 2012;25(6):632–640. [DOI] [PubMed] [Google Scholar]
- 77.Firstenberg MS, Orsinelli DA. ECMO and ECHO: the evolving role of quantitative echocardiography in the management of patients requiring extracorporeal membrane oxygenation. J Am Soc Echocardiogr. 2012;25(6):641–643. [DOI] [PubMed] [Google Scholar]
- 78.Pappalardo F, Pieri M, Arnaez Corada B, et al. Timing and Strategy for Weaning From Venoarterial ECMO are Complex Issues. J Cardiothorac Vasc Anesth. 2015;29(4):906–911. [DOI] [PubMed] [Google Scholar]
- 79.Kim D, Jang WJ, Park TK, et al. Echocardiographic Predictors of Successful Extracorporeal Membrane Oxygenation Weaning After Refractory Cardiogenic Shock. J Am Soc Echocardiogr. 2021;34(4):414–422 e414. [DOI] [PubMed] [Google Scholar]
- 80.Marasco SF, Lukas G, McDonald M, McMillan J, Ihle B. Review of ECMO (extra corporeal membrane oxygenation) support in critically ill adult patients. Heart Lung Circ. 2008;17 Suppl 4:S41–47. [DOI] [PubMed] [Google Scholar]
- 81.Aissaoui N, Caudron J, Leprince P, et al. Right-left ventricular interdependence: a promising predictor of successful extracorporeal membrane oxygenation (ECMO) weaning after assistance for refractory cardiogenic shock. Intensive Care Med. 2017;43(4):592–594. [DOI] [PubMed] [Google Scholar]
- 82.Osaki S, Sweitzer NK, Rahko PS, et al. To explant or not to explant: an invasive and noninvasive monitoring protocol to determine the need of continued ventricular assist device support. Congest Heart Fail. 2009;15(2):58–62. [DOI] [PubMed] [Google Scholar]
- 83.Gutsche JT, Ortoleva J. Echocardiography: Do We Need New Standards for ECMO Patients? J Cardiothorac Vasc Anesth. 2019;33(8):2356–2357. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Movie 1. RV dysfunction and dilation with severe TR and increased right atrial pressure. Midesophageal 4-chamber view focused on the tricuspid valve demonstrating severe, eccentric tricuspid regurgitation directed towards the IAS in a patient with a dilated RV with reduced systolic function. The IAS bows significantly to the left, consistent with pressure overload of the right atrium. IAS: interatrial septum, RV: right ventricle, TR: tricuspid regurgitation.
Movie 2. RV dysfunction and dilation with severe TR and increased right atrial pressure. Midesophageal RV inflow-outflow view with color flow Doppler demonstrating severe tricuspid regurgitation, a dilated RV, and hypokinesis of the RV freewall. The IAS bows significantly to the left, consistent with pressure overload of the right atrium. IAS: interatrial septum, RV: right ventricle, TR: tricuspid regurgitation.
Movie 3. PFO with left to right flow. A modified midesophageal bicaval view with color flow Doppler demonstrating left to right flow across a PFO. A pulmonary artery catheter is visualized within the right atrium. PFO: patent foramen ovale.
Movie 5. Confirmation of a guidewire within the RA adjacent to a preexisting pacing lead. A modified midesophageal bicaval view focused on the SVC demonstrates the “J-tip” of a guidewire within the RA and adjacent to a preexisting pacemaker lead. Care is required to correctly identify intracardiac wires and not confuse indwelling leads or catheters for guidewires. LA: left atrium, RA: right atrium, SVC: superior vena cava.
Movie 6. Biplane imaging to confirm presence of a guidewire within the SVC in a patient with two preexisting pacemaker leads. A midesophageal bicaval view with biplane imaging through the SVC (image on the right, in short axis) demonstrates three distinct wires within the SVC in a patient with two preexisting pacemaker leads undergoing guidewire placement from the right IJ approach. In a single, 2D long axis view, a lead or wire may be out-of-plane. Biplane imaging may facilitate easier identification of a guidewire as it is advanced into position. 2D: two-dimensional, IJ: internal jugular vein, SVC: superior vena cava.
Movie 4. A persistent left SVC identified by agitated saline injection. A deep esophageal 4-chamber view demonstrates a dilated RV with a flattened IVS. A dilated coronary sinus orifice is noted and injection of agitated saline through a left upper extremity reveals early opacification of the coronary sinus prior to the right atrium consistent with presence of a persistent left SVC. CS: coronary sinus, IVS: interventricular septum, LV: left ventricle, RV: right ventricle, TV: tricuspid valve.
Movie 8. Reverberation artifact from guidewire within the RA using biplane imaging. A biplane image obtained from a midesophageal modified bicaval view demonstrates a short-axis view of the guidewire. Here it is readily apparent that the reverberation artifact is located directly “below” the guidewire consistent with artifact rather than a second guidewire. RA: right atrium.
Movie 9. Vf-Vj-ECMO cannulas visualized near the superior and inferior cavoatrial junctions. A midesophageal bicaval view demonstrates a femoral venous drainage cannula noted within the RA just past the inferior cavoatrial junction and a jugular venous return cannula with the tip within the RA near the superior cavoatrial junction in a patient on Vf-Vj-ECMO. f: femoral, j: jugular, LA: left atrium, RA: right atrium.
Movie 7. Reverberation artifact creating appearance of two guidewires within the right atrium. A midesophageal modified bicaval view illustrating how reverberation artifact due to a strong reflector (such as a guidewire) can create the illusion of a second guidewire within the right atrium. LA: left atrium, RA: right atrium.
Movie 10. Vf-Vj-ECMO cannulas with color Doppler imaging demonstrating blood flow. A midesophageal bicaval view demonstrates a femoral venous drainage cannula noted within the RA just past the inferior cavoatrial junction and a jugular venous return cannula with the tip within the RA near the superior cavoatrial junction in a patient on Vf-Vj-ECMO. Color Doppler imaging demonstrates flow (drainage) into to femoral cannula and flow emanating from the jugular cannula (return). f: femoral, j: jugular, LA: left atrium, RA: right atrium.
Movie 12. Color Doppler imaging used to assess orientation and position of the central outflow port in a patient undergoing (bc)Vj-V-ECMO. A color Doppler midesophageal bicaval view focus on the central outflow port of a Crescent MC3 (Cardiopulmonary, Dexter, MI) bicaval ECMO cannula. The outflow port should be oriented above the tricuspid valve such that flow will be directed across the valve orifice. bc: bicaval, IVC: inferior vena cava, j: jugular, LA: left atrium, RA: right atrium.
Movie 13. A Protek Duo cannula visualized traversing the RV in a patient undergoing (dl)Vj-P-ECMO. This midesophageal RV inflow-outflow view demonstrates a Protek Duo traversing the RV with the tip (outflow port) located within the main pulmonary artery. The inflow port is located within the RA and is not visualized in this image. AV: aortic valve, dl: double lumen, j: jugular, LA: left atrium, PV: pulmonic valve, RA: right atrium, RV: right ventricle.
Movie 11. A bicaval cannula is visualized to confirm appropriate positioning of the central outflow port in a patient undergoing (bc)Vj-V-ECMO. A midesophageal bicaval view focus on the central outflow port of a Crescent MC3 (Cardiopulmonary, Dexter, MI) bicaval ECMO cannula. The outflow port should be oriented above the tricuspid valve such that flow will be directed across the valve orifice. bc: bicaval, IVC: inferior vena cava, j: jugular, LA: left atrium, RA: right atrium.
Movie 15. 3D image demonstrating a Protek Duo cannula in a patient undergoing (dl)Vj-P-ECMO. A Protek Duo cannula is visualized within the RV with the tip located just distal to the pulmonic valve. AV: aortic valve, dl: double lumen, j: jugular, LA: left atrium, RV: right ventricle.
Movie 14. Color Doppler demonstrating outflow into the main PA from a Protek Duo cannula in a patient undergoing (dl)Vj-P-ECMO. A midesophageal RV inflow-outflow view focused on the pulmonic valve demonstrates color Doppler flow emanating from the tip (outflow port) located within the main pulmonary artery. AV: aortic valve, dl: double lumen, j: jugular, LA: left atrium, RV: right ventricle.
Movie 17. Transthoracic echocardiography. Parasternal long axis view demonstrates pericardial effusion (PE). The arrow points to diastolic collapse of the right ventricle (RV) suggestive of cardiac tamponade. AO=Aorta; DA= Descending thoracic aorta; LA=Left atrium; LV= left ventricle. Reproduced with permission from Nanda NC (Ed) Interesting Cases in Echocardiography (2017), Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India
Movie 16. A TEE clip of a patient with an acute aortic dissection. A color Doppler midesophageal aortic valve long-axis view in a patient with an acute aortic dissection demonstrating a dissection flap in the aortic root, a dilated aortic root with effacement of the sinuses of Valsalva, a flail right coronary cusp, severe eccentric holodiastolic aortic regurgitation, and a pericardial effusion. LA: left atrium, LV: left ventricle, RV: right ventricle, TEE: transesophageal echocardiography
Movie 18. Transthoracic echocardiography. The arrow in the apical 4-chamber view points to a large, mobile thrombus in the apex of the left ventricle (LV) with depressed function. LA=Left atrium; RA= Right atrium; RV= Right ventricle. Reproduced with permission from Nanda NC (Ed) Interesting Cases in Echocardiography (2017), Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India
Movie 19. Transthoracic echocardiography. Subcostal view shows a dilated, non-collapsing inferior vena cava (IVC) consistent with increased right atrial pressure. HV=hepatic vein; L=Liver. Reproduced with permission from Nanda NC (Ed) Interesting Cases in Echocardiography (2017), Jaypee Brothers Medical Publishers (P) Ltd, New Delhi, India
Movie 20. Aortic insufficiency noted prior to initiation of peripheral VA-ECMO. A midesophageal aortic valve long-axis view with color Doppler illustrates trace, early diastolic aortic insufficiency prior to initiation of peripheral VA-ECMO. The aortic valve is heavily calcified with poor cusp mobility. Spontaneous echo contrast (“smoke”) is visualized within the LA consistent with a low-flow state. LA: left atrium, LV: left ventricle.
Movie 21. Increased aortic insufficiency after initiation of peripheral VA-ECMO. A midesophageal aortic valve long-axis view with color Doppler demonstrates mild, central, holodiastolic aortic insufficiency after institution of peripheral VA-ECMO. The previously noted spontaneous echo contrast is no longer visualized. The calcific aortic valve opens with every cardiac cycle. LA: left atrium, LV: left ventricle.
Movie 22. An aortic valve that does not open during systole in a patient on peripheral VA-ECMO. A midesophageal long-axis view with color Doppler demonstrates an aortic valve that does not open at any point during the cardiac cycle in a patient on peripheral VA-ECMO as a bridge to successful durable LVAD implantation. AV: aortic valve, LA: left atrium, LV: left ventricle, LVAD: left ventricular assist device.
Movie 24. LAVA-ECMO. A non-standard midesophageal view demonstrating a multistage venous cannula with the tip and distal inflow ports positioned within the LA to facilitate indirect left ventricular venting in a patient on peripheral VA-ECMO. AV: aortic valve, IAS: interatrial septum, LA: left atrium, LAVA: left atrial veno-arterial, RA: right atrium, RV: right ventricle.
Movie 25. 3D mitral en face view demonstrating transseptal cannula positioning during LAVA-ECMO. A 3D en face view of the mitral valve with the AV at the 12 o’clock position demonstrates a multistage venous cannula positioned across the IAS to achieve indirect LV venting in a patient undergoing LAVA-ECMO. The distal inflow ports appear echolucent in this image. 3D: three-dimensional, AV: aortic valve, IAS: interatrial septum, LAVA: left atrial veno-arterial, MV: mitral valve, RA: right atrium.
Movie 23. Transseptal access obtained for institution of LAVA-ECMO. A non-standard midesophageal view demonstrating a dilator advanced across the IAS prior to placement of a multistage venous cannula within the LA to facilitate indirect left ventricular venting in a patient on peripheral VA-ECMO. AV: aortic valve, IAS: interatrial septum, LA: left atrium, LAVA: left atrial veno-arterial, RA: right atrium, RV: right ventricle.
Movie 26. Left atrial decompression with initiation of LAVA-ECMO. A midesophageal long axis view demonstrates left atrial collapse at the moment when inflow from the left atrium is established. The multistage venous cannula is visualized within the LA. Titration of flow is necessary to prevent excessive left atrial collapse and find a balance point where chamber decompression is facilitated but left atrial collapse does not occur. LA: left atrium, LAVA: left atrial veno-arterial, LV: left ventricle, RA: right atrium, RV: right ventricle.