Abstract
Lactobacillus helveticus is a homofermentative thermophilic lactic acid bacterium that is used in the manufacture of Swiss type and long-ripened Italian cheeses, such as Emmental, Grana, and Provolone cheeses. Substantial differences in several technologically important characteristics are found among L. helveticus strains isolated from natural dairy starter cultures. In the present study we investigated the genotypic diversity of 74 strains isolated from different dairy cultures used for manufacturing Grana and Provolone cheeses and six collection strains. A restriction fragment length polymorphism analysis of both total genomic DNA and the 16S rRNA gene (ribotyping) was used as genotypic fingerprinting. A multivariate statistical analysis of the data enabled us to identify significant genotypic heterogeneity in L. helveticus. We found that genotypic fingerprinting could be used to distinguish strains; in particular, it was possible to associate the presence of specific strain genotypes with dairy ecosystem sources (e.g., Grana or Provolone cheese). Our data contribute to the description of microbial heterogeneity in L. helveticus and provide a more solid basis for understanding the functional and ecological significance of the presence of different L. helveticus biotypes in natural dairy starter cultures.
Lactobacillus helveticus is a homofermentative thermophilic lactic acid bacterium (LAB) that is used in the manufacture of Swiss type and long-ripened Italian cheeses, such as Emmental, Grana, and Provolone cheeses. In particular, L. helveticus is the most prevalent species recovered from natural lactic starter cultures used to produce typical Italian cheeses. Such cultures represent complex microbial associations of LAB that are characterized by the presence of various species and subspecies, such as L. helveticus, Lactobacillus delbrueckii subsp. bulgaricus, Lactobacillus delbrueckii subsp. lactis, and Lactobacillus fermentum, and many biotypes (13, 27) of these species. Substantial differences in several technologically important characteristics have been found among L. helveticus strains isolated from natural dairy starter cultures (1, 2, 8, 9, 11, 16, 18, 19, 21, 28, 31).
Because of the widespread use of L. helveticus in cheese technology, information concerning genotypic heterogeneity in this species is accumulating rapidly (6, 7, 14, 17). A variety of genotypic and phenotypic methods have been used to show that the dominant L. helveticus communities in various dairy starters are composed of different biotypes, which sometimes may be associated with the source of isolation (2, 6, 9, 12, 14, 20, 22).
In a previous study, 74 L. helveticus strains that were isolated from Grana and Provolone cheese natural whey starters were distinguished according to their origins by using both cell wall protein profiles and chemometric evaluation of some phenotypic parameters (10). The cell wall protein patterns allowed the researchers to distinguish L. helveticus strains according to their sources of isolation. Good discrimination between the two groups of L. helveticus strains was obtained by using multivariate statistical techniques, which allowed the workers to obtain all of the discriminating information related to phenotypic activities. The researchers concluded that the differences in the predominant biotypes of L. helveticus in various natural starters were related to the original habitats of the organisms and that the “ecological niches” from which the organisms had been isolated played a role in the selection of biotypes that had technologically significant physiological properties.
The aim of this study was to investigate whether the previous findings concerning phenotypic discrimination of L. helveticus strains were consistent with the genotypes of the organisms. To do this, strains were characterized by performing a restriction fragment length polymorphism (RFLP) analysis of both total genomic DNA and the 16S rRNA gene (ribotyping).
MATERIALS AND METHODS
Strains.
A total of 80 L. helveticus strains were used. Seventy-four of these strains had been isolated from seven different Grana cheese natural whey starters (33 strains) and 10 different Provolone cheese natural whey starters (41 strains) at different times and in different dairy plants during the last 10 years. One of the Grana cheese starter isolates (strain TH456) was obtained from the Veneto Agricoltura Collection (Thiene, Italy). The remaining six L. helveticus strains were strains ATCC 15009T, ATCC 10386, and ATCC 15807, which were obtained from the American Type Culture Collection (Rockville, Md.); strain CNRZ 493, which was isolated from an Emmental cheese whey starter in Finland and was obtained from the stock collection of Institut National de la Recherche Agronomique (Jouy-en-Josas, France); strain Lh79, which was isolated from a commercial dairy starter; and strain NCK 228, which was a helveticin J producer that was kindly provided by Todd Klaenhammer (North Carolina State University, Raleigh). The strains had been classified previously by using biochemical tests (15) and a specific oligonucleotide probe as described by Tailliez et al. (26).
Stock cultures and strain revitalization.
The bacterial strains were maintained as frozen stocks in MRS broth (Difco Laboratories, Detroit, Mich.) at −80°C in the presence of 15% of glycerol as a cryoprotective agent. Working cultures were prepared by making three overnight transfers at 42°C in MRS medium under anaerobic conditions.
Extraction of total DNA.
Total DNA were extracted from 5-ml samples of fresh overnight MRS broth cultures of different lactobacilli by using an alkaline lysis method described by de los Reyes-Gavilàn et al. (5). The quantity and purity of DNA were assessed by determining the optical density at 260 and 280 nm, as described by Sambrook et al. (24).
DNA hybridization with the L. helveticus probe.
An RFLP analysis of L. helveticus isolates was performed by using a species-specific DNA probe in Southern blot (24) hybridization experiments. Total DNA was cleaved by EcoRI (Life Technologies Italia, Milan, Italy). Restriction digestion was performed for 2 h at 37°C in 20-μl portions of incubation buffer (Life Technologies) containing 10 U of restriction enzyme EcoRI and 0.25 μg of total DNA.
DNA restriction fragments were separated electrophoretically in agarose (1%, wt/vol) gels and were blotted on a Hybond N+ membrane (Amersham Pharmacia Biotech Italia, Milan, Italy) under alkaline conditions (0.4 N NaOH). Subsequently, DNA-DNA hybridization was performed by using the enhanced chemiluminescence-direct nucleic acid labelling and detection systems (Amersham Pharmacia Biotech Italia) as recommended by the supplier. Overnight hybridization was carried out at 42°C with an internal PCR-amplified 388-bp fragment of IS1201 as a DNA probe. IS1201 is a 1,387-bp insertion sequence that was isolated from L. helveticus (5, 26) and was kindly provided by P. Tailliez (Unité de Recherches Laitières et Genetique Appliquée, Jouy-en-Josas, France). IS1201 was obtained from a BssHII-digested pBluescript plasmid which had been cloned in Escherichia coli CNRZ 1814 as described by Tailliez et al. (26). The 388-bp internal fragment was amplified from plasmid DNA of strain CNRZ 1814 by using the following primers: 5′ GCTGAGCGATAAGTTCTT 3′ and 5′ ATTGGCTTGCTGGTGAAT 3′. The two primers were designed to amplify region 594 to 981 of the previously described IS1201 DNA sequence (26). After signal generation and detection, autoradiographic films (Hyperfilm-ECL; Amersham Pharmacia Biotech Italia) were exposed to generate light as recommended by the manufacturer. The approximate molecular sizes (in base pairs) of the restriction fragments on the Southern blots were calculated by comparing their migration distances with those of HindIII-digested lambda DNA size markers (Life Technologies).
rRNA gene hybridization (ribotyping).
After extraction of total DNA, restriction, and blotting as described above, DNA restriction fragments from lactobacilli were hybridized with a probe complementary to 16S ribosomal DNA (rDNA). A PCR was used to amplify the 16S rRNA gene from the total DNA of L. helveticus ATCC 15009T. DNA labelling and hybridization were performed as described above. An approximately 1,500-bp fragment of the 16S rDNA was amplified by using the following primers: 5′ CCGAGCTCAACAGAGTTTGATCCTGGCTCAG 3′ and 5′ GGTCGACCGTTAATACGACTCACTATAGGGATACCTTGTTACGACTT 3′. The fragment amplified by these primers has been used previously for ribotyping Lactobacillus strains (23).
PCR amplification conditions.
The 388-bp internal DNA fragment of IS1201 and the 16S rRNA gene used for RFLP analysis and ribotyping analysis, respectively, were amplified by using a 100-μl mixture containing 50 mM Tris-HCl (pH 8.3), 1.5 mM MgCl2, 200 μM dATP, 200 μM dTTP, 200 μM dGTP, 200 μM dCTP, each primer (Celbio, Milan, Italy) at a concentration of 0.4 to 0.5 μM, 0.1 μg of genomic DNA (or plasmid DNA for IS1201), and 2.5 U of AmpliTaq DNA polymerase (Perkin-Elmer Italia, Monza, Italy). DNA amplification was performed with a Perkin-Elmer model 2400 thermoblock (Perkin-Elmer Italia). The amplification conditions used were as follows: one cycle consisting of 2 min at 94°C, followed by 30 cycles consisting of 30 s at 94°C, 30 s at 60°C, and 30 s at 72°C and finally one cycle consisting of 7 min of chain elongation at 72°C. The presence of specific PCR products was controlled by agarose (1%, wt/vol) gel electrophoresis. Amplified products were cleaned with a Geneclean III spin kit (Bio 101, La Jolla, Calif.) and were used as DNA probes.
Analysis and comparison of the DNA-DNA hybridization fingerprints.
Exposed autoradiographic film containing Southern-blotted fingerprint profiles obtained from RFLP and ribotyping hybridization experiments was scanned (model Scanjet 6100 C/T; Hewlett-Packard Italia, Milan, Italy), and the resulting densitometric traces of band profiles were analyzed with the pattern analysis software package GelCompar, version 4.1 (Applied Maths, Kortrjik, Belgium). Levels of similarity of the band profiles were calculated based on the Pearson (or Dice) similarity coefficients. A dendrogram was deduced from the similarity matrix by using the unweighted pair group method with arithmetic average (UPGMA) clustering algorithm (30). The quality of the cluster analysis was verified by calculating the cophenetic correlation value (expressed as a percentage) for each dendrogram by using the GelCompar software. The level of repeatability for DNA fingerprinting was calculated by including on every gel multiple samples of L. helveticus ATCC 15009T DNA, as well as L. helveticus ATCC 10386 and ATCC 15807 DNA (as reference profiles), and performing a numerical analysis of all of the profiles obtained.
Amplification and restriction analysis of rDNA.
16S rDNA were amplified from the total DNA of lactobacilli as described above. The presence of specific PCR products in each sample was controlled by agarose (1%, wt/vol) gel electrophoresis before a restriction enzyme analysis was performed with EcoRI (Life Technologies). Restriction digestion was carried out as described above. The restriction fragment patterns were analyzed by agarose (2%, wt/vol) gel electrophoresis.
16S rRNA gene sequence determination and analysis.
Amplified 16S rDNA fragments from L. helveticus ATCC 15009T, Lh8, Lh40, and Lh45 were sequenced directly by using a Taq dRhodamine terminator cycle sequencing kit (MicroSeq 16S rRNA gene kit; Perkin-Elmer Applied Biosystems, Foster City, Calif.) as recommended by the manufacturer. PCRs for cycle sequencing were performed with a Perkin-Elmer model 9700 thermoblock (Perkin-Elmer Applied Biosystems). The thermal cycling conditions used for cycle sequencing included 11 steps, as follows: six cycles consisting of 10 s at 96°C, followed by 1 min each at 65, 64, 63, 62, 61, 60, 59, 58, 57, 56, and 55°C. Excess dye terminators were removed by purifying PCR products with Centri-Sep spin columns (Perkin-Elmer Applied Biosystems) as recommended by the manufacturer. The sequences which we determined were aligned with 16S rRNA gene sequences of L. helveticus NCDO 2712 and Escherichia coli ATCC 11775T retrieved from the GenBank database (accession no. X61141 and X80725, respectively) by using the software Multalin, version 5.3.3 (4).
RESULTS
Hybridization experiments.
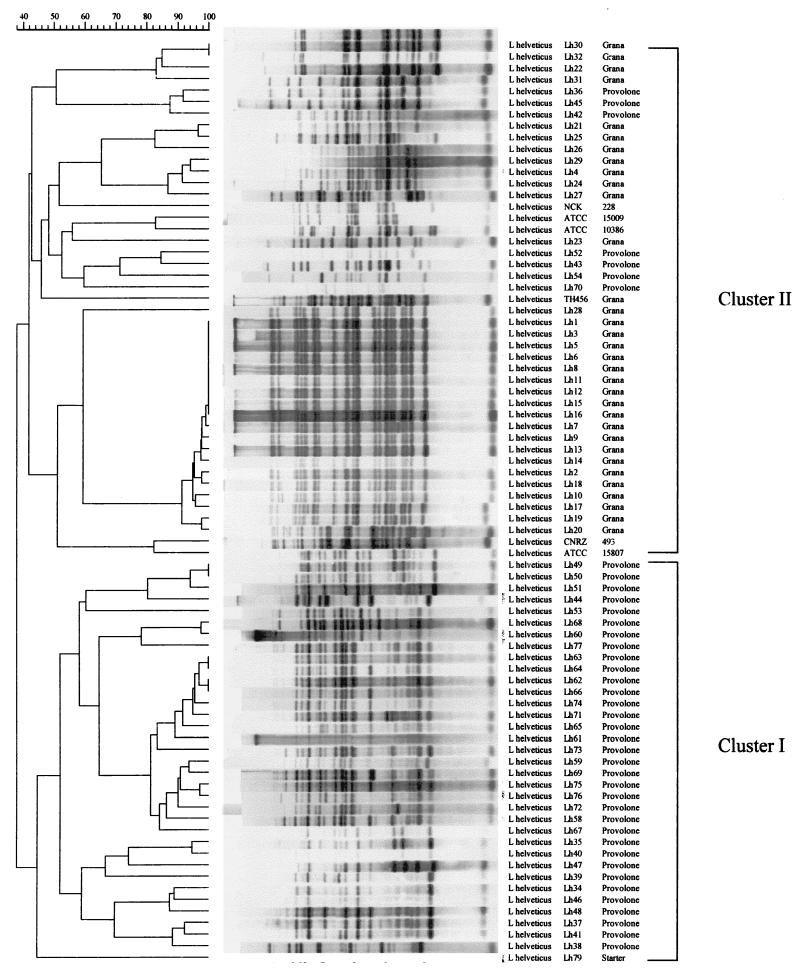
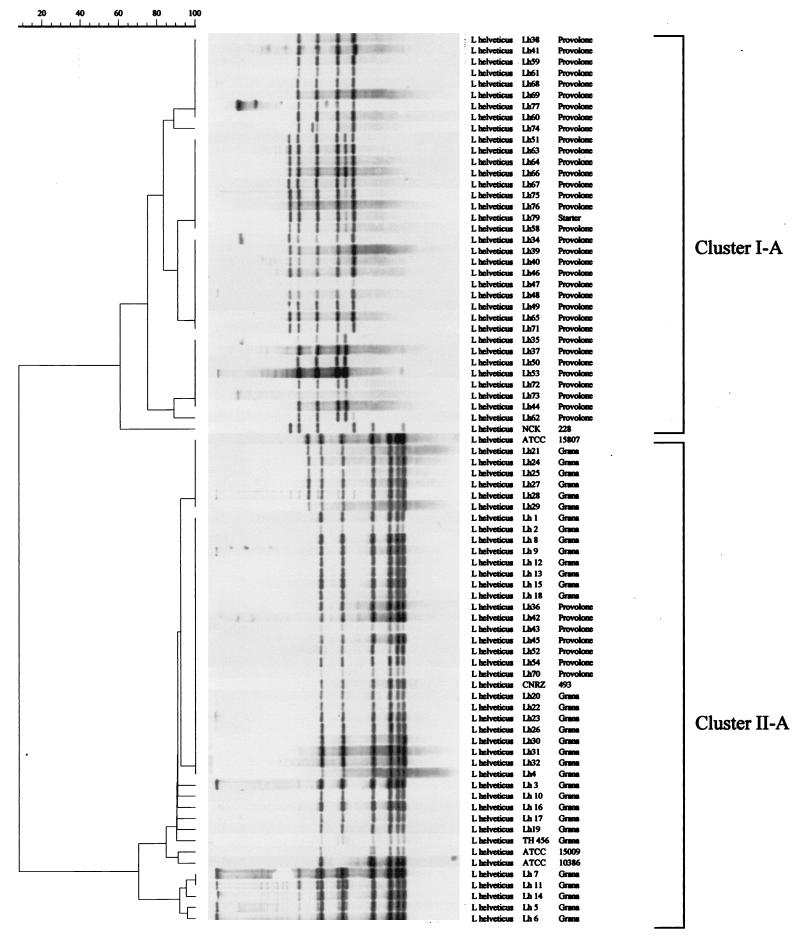
Hybridization of the internal PCR-amplified 388-bp fragment of IS1201 with total genomic DNA from all 80 L. helveticus strains following restriction with EcoRI yielded a variety of reproducible DNA restriction patterns, which resulted in good strain differentiation (Fig. 1). Hybridization of the 16S rRNA gene resulted in nine distinct ribopatterns; the number of EcoRI-generated bands ranged from four to seven (Fig. 2).
FIG. 1.
Dendrogram based on UPGMA clustering of Dice association coefficients for normalized RFLP patterns obtained after hybridization of the internal PCR-amplified 388-bp fragment of IS1201 with total genomic DNA from 80 L. helveticus strains following restriction with EcoRI.
FIG. 2.
Dendrogram based on UPGMA clustering of Dice association coefficients for normalized ribotyping patterns obtained after hybridization of the 16S rRNA gene with total genomic DNA from 80 L. helveticus strains following restriction with EcoRI.
Cluster analysis.
In preliminary experiments, a UPGMA cluster analysis was performed by using either the Pearson product-moment correlation coefficient or the Dice association coefficient as the similarity coefficient. The latter allowed us to obtain higher-quality cluster analysis results because the cophenetic correlation values for each dendrogram ranged from 91 to 96% when the Dice coefficient was used and from 74 to 95% when the Pearson coefficient was used (data not shown). When a cluster analysis was performed with reference strains L. helveticus ATCC 15009T, ATCC 10386, and ATCC 15807 and both the Pearson and Dice similarity coefficients were used, the levels of repeatability for our hybridization experiments were very high (around 90%) (data not shown). Hence, we deduced that (i) a band-matching technique in which the Dice association coefficient was used was more suitable for our RFLP patterns and (ii) only clusters with similarity coefficients of less than 90% were different.
The numerical analysis of the hybridization patterns obtained when the internal PCR-amplified 388-bp fragment of IS1201 was used as a DNA probe (shown as a dendrogram in Fig. 1) revealed that there were two distinct L. helveticus clusters at a level of similarity of approximately 40%. Cluster I contained most (34 of 41) of the Provolone cheese isolates; strain Lh79, which was isolated from a commercial starter, was located at a separate position in this cluster. All of the Grana cheese isolates, the seven remaining Provolone cheese isolates, strain NCK 228, and reference strains ATCC 15009T, ATCC 10386, ATCC 15807, and CNRZ 493 belonged to cluster II. Strain Lh28 was located at a separate position in this cluster (Fig. 1). The cophenetic correlation value for the dendrogram was 91.1%.
The numerical analysis of the hybridization patterns obtained from the 16S rDNA ribotyping analysis (shown as a dendrogram in Fig. 2) allowed us to identify two main L. helveticus clusters (clusters I-A and II-A) at a level of similarity of 60%. A total of 34 of 41 Provolone cheese isolates and commercial starter strain Lh79 belonged to cluster I-A, for which four (or five) EcoRI-generated bands were usually observed. Moreover, strain NCK 228 produced a quite peculiar ribotyping profile and therefore occupied a separate position in this cluster (Fig. 2). The seven remaining Provolone cheese isolates, all of the Grana cheese isolates, and reference strains ATCC 15009T, ATCC 10386, ATCC 15807, and CNRZ 493 were grouped in cluster II-A, for which six (or seven) EcoRI-generated bands were observed. The cophenetic correlation value for the dendrogram was 96.5%.
Studies of the 16S rRNA gene.
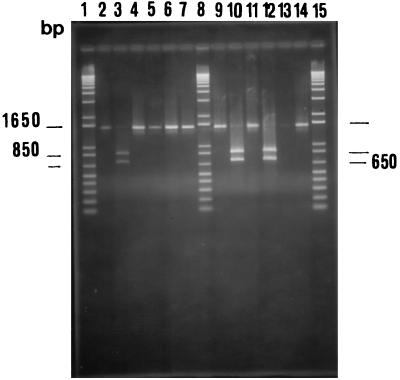
16S rDNAs were amplified from all of the L. helveticus isolates, and fragments that were approximately 1,500 bp long and were consistent with the 16S rRNA genes were obtained. After the restriction enzyme analysis, clear RFLP patterns were observed after digestion with EcoRI (Fig. 3). On the basis of the difference which was observed in repeated experiments, we identified two groups of strains. For one group of strains, which was composed of L. helveticus ATCC 15009T, ATCC 15807, and CNRZ 493, the 33 Grana cheese isolates, and 8 of the 41 Provolone cheese isolates, two fragments that were approximately 700 and 800 bp long were observed (Fig. 3, lanes 3, 10, and 12). For another group of strains, which was composed of L. helveticus ATCC 10386, strain NCK 228, commercial strain Lh79, and 33 of the 41 Provolone cheese isolates, the 16S rRNA genes were not digested by EcoRI (Fig. 3, lanes 5, 7, and 14).
FIG. 3.
Agarose gel electrophoresis of amplified 16S rDNA of L. helveticus strains digested with restriction endonuclease EcoRI. Lane 2, undigested 16S rDNA of L. helveticus ATCC 15009T; lane 3, EcoRI-digested 16S rDNA of L. helveticus ATCC 15009T; lane 4, ATCC 10386 undigested 16S rDNA; lane 5, ATCC 10386 digested 16S rDNA; lane 6, Lh33 undigested 16S rDNA; lane 7, Lh33 digested 16S rDNA; lane 9, Lh8 undigested 16S rDNA; lane 10, Lh8 digested 16S rDNA; lane 11, Lh45 undigested 16S rDNA; lane 12, Lh45 digested 16S rDNA; lane 13, Lh40 undigested 16S rDNA; lane 14, Lh40 digested 16S rDNA; lanes 1, 8, and 15, DNA molecular size markers (1-kb plus DNA ladder; Life Technologies Italia).
The 16S rDNA sequences of L. helveticus ATCC 15009T and strains Lh8, Lh40, and Lh45 were determined by directly sequencing PCR-amplified rRNA gene fragments. Almost complete gene sequences (>1,400 nucleotides) were determined and aligned with the sequences of L. helveticus NCDO 2712 and E. coli ATCC 11775T, which were retrieved from the GenBank database. The levels of sequence similarity between the L. helveticus strains were more than 99.9%, and there were one or two (depending on the strain) base differences between the 16S rRNA gene sequences of L. helveticus NCDO 2712 and the L. helveticus genes which we sequenced (data not shown). In particular, strain Lh40 contained a C (instead of T) in the EcoRI recognition sequence at position 705 of the gene (Fig. 4).
FIG. 4.
Alignment of partial DNA sequences of the 16S rRNA genes from L. helveticus ATCC 15009T, Lh8, Lh40, Lh45, and NCDO 2712 (accession no. X61141) and E. coli ATCC 11775T (accession no. X80725). The EcoRI recognition sequences are indicated by boldface type, and the mismatch at position 705 of the Lh40 gene is underlined.
DISCUSSION
In a previous study, strains of L. helveticus isolated from Grana and Provolone cheese natural whey starters were distinguished according to their origins by using both cell wall protein profiles and chemometric evaluation of some phenotypic parameters (10). In the present investigation our goal was to determine whether these two phenotypically distinct L. helveticus microbial communities were molecularly diverse or genetically homogeneous. Therefore, hybridization performed with an L. helveticus species-specific DNA probe and ribotyping were used in order to differentiate strains isolated from Grana and Provolone cheese whey starter cultures.
The substantial strain heterogeneity revealed by hybridization with IS1201, a 1,387-bp insertion sequence isolated from L. helveticus, and by ribotyping confirmed the ability of these approaches to identify RFLP in this species (5, 14, 26). The use of EcoRI was suggested by the RFLP of EcoRI digests of L. helveticus observed previously when a species-specific DNA probe was used (5). Our cluster analysis revealed the following two major hybridization groups: one group containing the Grana cheese isolates and another group containing most of the Provolone cheese isolates. This result is consistent with the previous differentiation of the same strains based on phenotypic data (10). The high cophenetic correlation values obtained for clusters when both RFLP techniques were used indicate that the quality of the UPGMA cluster analysis was excellent when the Dice similarity coefficient was used. Indeed, the cophenetic correlation value is the product-moment correlation value for all original levels of matrix similarity and all of the corresponding similarity values derived from the dendrogram. If a UPGMA dendrogram faithfully represents the similarity matrix, this value is high (e.g., more than 90%). Moreover, the similarity coefficients for the two main clusters were always less than the level of repeatability regardless of the method of hybridization used, which confirmed that the clusters were significantly different.
Although the clusters based on IS1201 and ribotyping hybridization profiles had similar basic structures, some important conclusions can be drawn. In the IS1201 clusters, strains grouped at lower similarity values than they grouped in the ribotyping clusters. This was because with the former approach, which allowed us to examine the RFLP in the whole genome, it was possible to observe more marked strain heterogeneity. This confirmed that the genetic differences between Grana and Provolone cheese isolates not only depend on the differences in the conserved 16S rDNA region or in the plasmid profiles observed previously (14) but also are more widely distributed along the genome. This result is very interesting because it confirms at a genetic level the extensive phenotypic differences between the two groups of L. helveticus isolates reported previously (9, 10). Sequencing data for L. helveticus isolates did not reveal extensive differences between the 16S rRNA gene sequences and the sequences of the species included in the GenBank database.
It is worth noting that the seven Provolone cheese isolates which clustered with the Grana cheese isolates when ribotyping was used were the same isolates that were identified in the RFLP analysis when IS1201 was used. The cluster structure was obviously similar to the cluster structure obtained when IS1201 was used because the same restriction enzyme (EcoRI) was used to digest the DNAs that were probed with both IS1201 and 16S rDNA. In the ribotyping profiles the number of EcoRI-generated bands was smaller with Provolone cheese isolates than with Grana cheese isolates. For a group of isolates, the EcoRI digestion profile of 16S rRNA amplified genes corresponded to the profile expected after a restriction analysis based on the previously described 16S rRNA gene sequence of L. helveticus NCDO 2712 (3), which revealed that there was a unique EcoRI recognition site at position 580 of the gene. For another group of isolates, EcoRI restriction revealed that the difference in bands between the two groups of isolates identified by ribotyping corresponded to an RFLP of the EcoRI restriction site located on the gene because the expected restriction, which was observed with L. helveticus ATCC 15009T and the Grana cheese isolates, was not observed for most of the Provolone cheese isolates. Sequence data indicated that this RFLP was based on a single nucleotide substitution located at the EcoRI site of the 16S rRNA gene. Sequence data for the conserved 16S rRNA gene did not reveal any other significant difference between strains.
Interestingly, six of seven Provolone cheese isolates which were cut by EcoRI after 16S rRNA gene restriction were the same isolates that clustered with Grana cheese isolates after RFLP and ribotyping hybridization experiments. Confirmatory sequence data showed that Lh45, a Provolone cheese isolate included in the group mentioned above, lacked the nucleotide substitution on the 16S rRNA gene. Therefore, the following two phenotypically and genotypically different L. helveticus biotypes were isolated from Grana and Provolone cheeses: the “Grana biotype,” which is typical of the Grana cheese L. helveticus population; and the “Provolone biotype,” which is typical of the Provolone cheese L. helveticus population and also is present in Grana cheese. The prevalence of a biotype may be related to the different conditions in the two cheeses, which, according to the differences in phenotypic traits described previously for the two groups of L. helveticus strains (9, 10), lead to selection of dominant strain populations.
Microbial communities differ both qualitatively and quantitatively. The relative proportions of the community members are subject to physicochemical changes in the environment, as well as changes caused by the physiological and metabolic activities of the organisms. Organisms that are abundant and/or cultivable under certain conditions may change into dormant and possibly less-cultivable forms (32).
The compositions of thermophilic LAB populations in natural whey starters may vary depending on the production zone, the concentration of microbes in milk, the technological processing parameters used, and the different locations in the cheese (12, 13). Often, wild strains isolated from natural habitats exhibit interstrain phenotypic variability due to different environmental pressures. This leads to identification of several biovars or biotypes that are adapted to specialized niches and have evolved with minor or major changes in phenotype and genotype. The observation that selection of L. helveticus biotypes depends on environmental and ecological factors is consistent with previous findings (10) concerning the presence of different protein patterns on the cell walls of L. helveticus isolates obtained from Provolone and Grana cheese starters. It is known that bacterial cell walls may evolve by selection in response to specific environmental and ecological pressures (25).
If the two L. helveticus populations in Grana and Provolone cheese starters are examined, it seems that the selective factors that lead to biotype selection are mainly technological. This is because natural cultures in which strains are selected come from cheeses (e.g., Grana and Provolone cheeses) that are produced in overlapping cheese-producing areas and, therefore, from similar ecosystems.
The fact that certain L. helveticus biotypes are competitive in the ecological niches represented by certain cheese types may not be directly related to a single strain activity or phenotypic characteristic but may be related to a number of activities or characteristics. In fact, in a previous study (10) in which different phenotypes of strains were compared, it was not possible to find one specific activity by which strains could be differentiated with respect to their sources. In contrast, good discrimination between the two groups of L. helveticus isolates was obtained when multivariate statistical techniques were used, which permitted the discriminating information present in the phenotypic characteristics to be identified.
The approach used in this study may contribute to the description of microbial heterogeneity in L. helveticus, as well as in other LAB species. Molecular techniques allow a better understanding of the genetic relationships of food-associated LAB and contribute to defining the ecological role of these organisms in foods. Molecular characterization and typing, both genetic and phenotypic, of LAB are developing to a point at which they are of increasing value to manufacturers of starter cultures and to food microbiologists. Application of typing techniques to a wider range of species and environments, as well as studies tracing the sources of food colonization by LAB, is helping to characterize food-associated or probiotic ecosystems (7). Identification of potential strain-specific markers is also providing powerful tools which can be used to distinguish members of microbial populations that belong to very similar taxonomic groups (29).
Interestingly, although the principal-component analysis method used to group phenotypic data (10) and the hierarchical clustering methods used in the present work for genotypic data are completely independent approaches, very similar groups were obtained with both methods. Therefore, polyphasic strain characterization provides a more solid basis for understanding the functional and ecological significance of the presence of different biotypes in natural dairy starter cultures. The ability to distinguish strains from two ecological niches at the genotypic level by studying both conserved and nonconserved DNA regions can be useful technologically and ecologically. This technique may be used to select strains that are more appropriate for a given cheese process.
REFERENCES
- 1.Bosi F, Bottazzi V, Vescovo M, Scolari G L, Battistotti B, Dellaglio F. Batteri lattici per la produzione di formaggio grana. I. Caratterizzazione tecnologica di ceppi di lattobacilli termofili. Sci Tecn Lattiero Casearia. 1990;41:105–136. [Google Scholar]
- 2.Carminati D, Mazzuccotelli L, Giraffa G, Neviani E. Incidence of inducible bacteriophage in Lactobacillus helveticus strains isolated from natural whey starter cultures. J Dairy Sci. 1997;80:1505–1511. [Google Scholar]
- 3.Collins M D, Martinez-Murcia A J. Phylogenetic analysis of the genus Lactobacillus and related lactic acid bacteria as determined by RT sequencing of 16S rRNA. FEMS Microbiol Lett. 1991;77:5–12. [Google Scholar]
- 4.Corpet F. Multiple sequence alignment with hierarchical clustering. Nucleic Acids Res. 1988;16:10881–10890. doi: 10.1093/nar/16.22.10881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.de los Reyes-Gavilàn C, Limsowtin G K Y, Tailliez P, Séchaud L, Accolas J P. A Lactobacillus helveticus-specific DNA probe detects restriction fragment length polymorphism in this species. Appl Environ Microbiol. 1992;58:3429–3432. doi: 10.1128/aem.58.10.3429-3432.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Drake M A, Small C L, Spence K D, Swanson B G. Differentiation of Lactobacillus helveticus strains using molecular typing methods. Food Res Int. 1996;29:451–455. [Google Scholar]
- 7.Dykes G A, von Holy A. Strain typing in the genus Lactobacillus. Lett Appl Microbiol. 1994;19:63–66. doi: 10.1111/j.1472-765x.1994.tb00906.x. [DOI] [PubMed] [Google Scholar]
- 8.Fortina M G, Rossi P, Mora D, Parini C, Neviani E. Slow milk coagulating variants of Lactobacillus helveticus. Folia Microbiol. 1996;41:33–38. [Google Scholar]
- 9.Fortina M G, Nicastro G, Carminati D, Neviani E, Manachini P L. Lactobacillus helveticus heterogeneity in natural cheese starters: the diversity in phenotypic characteristics. J Appl Microbiol. 1998;84:72–80. doi: 10.1046/j.1365-2672.1997.00312.x. [DOI] [PubMed] [Google Scholar]
- 10.Gatti M, Contarini G, Neviani E. Effectiveness of chemometric techniques in discrimination of Lactobacillus helveticus biotypes from natural dairy starter cultures on the basis of phenotypic characteristics. Appl Environ Microbiol. 1999;65:1450–1454. doi: 10.1128/aem.65.4.1450-1454.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gatti M, Fornasari M E, Perrone A, Neviani E. Relationship between acidification capacity in milk, proteolytic and peptidase activities and presence of yeast extract in Lactobacillus helveticus species. Microbiol Aliments Nutr. 1999;17:23–31. [Google Scholar]
- 12.Giraffa G, Neviani E. Different Lactobacillus helveticus strain populations dominate during Grana Padano cheese-making. Food Microbiol. 1999;16:205–210. [Google Scholar]
- 13.Giraffa G, Mucchetti G, Addeo F, Neviani E. Evolution of lactic acid microflora during Grana cheese-making and ripening. Microbiol Aliments Nutr. 1997;15:115–122. [Google Scholar]
- 14.Giraffa G, De Vecchi P, Rossi P, Nicastro G, Fortina M G. Genotypic heterogeneity among Lactobacillus helveticus strains isolated from natural cheese starters. J Appl Microbiol. 1998;85:411–416. doi: 10.1046/j.1365-2672.1998.853464.x. [DOI] [PubMed] [Google Scholar]
- 15.Kandler O, Weiss N. Regular, nonsporing Gram-positive rods. In: Sneath P H A, Mair N S, Sharpe M E, Holt J G, editors. Bergey's manual of systematic bacteriology. Vol. 2. Baltimore, Md: Williams & Wilkins; 1986. pp. 1208–1260. [Google Scholar]
- 16.Long M V, Bizzarro R, Gatti M, Neviani E. The influence of temperature on the autolytic activity of thermophilic lactobacilli. Microbiol Aliments Nutr. 1999;17:33–38. [Google Scholar]
- 17.Lortal S, Rouault A, Guezenec S, Gautier M. Lactobacillus helveticus: strain typing and genome size estimation by pulsed field gel electrophoresis. Curr Microbiol. 1997;34:180–185. doi: 10.1007/s002849900165. [DOI] [PubMed] [Google Scholar]
- 18.Neviani E, Carini S. Microbiology of Parmesan cheese. Microbiol Aliments Nutr. 1994;12:1–8. [Google Scholar]
- 19.Neviani E, Divizia R, Abbiati E, Gatti M. Acidification activity of thermophilic lactobacilli under the temperature gradient of Grana cheese making. J Dairy Sci. 1995;78:1248–1252. [Google Scholar]
- 20.Quiberoni A, Tailliez P, Quénée P, Suarez V, Reinheimer J. Genetic (RAPD-PCR) and technological diversities among wild Lactobacillus helveticus strains. J Appl Microbiol. 1998;85:591–596. [Google Scholar]
- 21.Reinheimer J A, Morelli L, Bottazzi V, Suarez V. Phenotypic variability among cells of Lactobacillus helveticus ATCC 15807. Int Dairy J. 1995;5:97–103. [Google Scholar]
- 22.Reinheimer J A, Quiberoni A, Tailliez P, Binetti A G, Suarez V B. The lactic acid microflora of natural whey starters used in Argentina for hard cheese production. Int Dairy J. 1996;6:869–879. [Google Scholar]
- 23.Rodtong S, Tannock G W. Differentiation of Lactobacillus strains by ribotyping. Appl Environ Microbiol. 1993;59:3480–3484. doi: 10.1128/aem.59.10.3480-3484.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sambrook J, Fritsch E F, Maniatis T. Molecular cloning: a laboratory manual. 2nd ed. Vol. 3. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory; 1989. [Google Scholar]
- 25.Sleytr U B, Beveridge T J. Bacterial S-layers. Trends Microbiol. 1999;7:253–260. doi: 10.1016/s0966-842x(99)01513-9. [DOI] [PubMed] [Google Scholar]
- 26.Tailliez P, Ehrlich S D, Chopin M C. Characterization of IS1201, an insertion sequence isolated from Lactobacillus helveticus. Gene. 1994;145:75–79. doi: 10.1016/0378-1119(94)90325-5. [DOI] [PubMed] [Google Scholar]
- 27.Torriani S, Vescovo M, Scolari G. An overview on Lactobacillus helveticus. Ann Microbiol Enzimol. 1994;44:163–191. [Google Scholar]
- 28.Valence F, Lortal S. Zymogram and preliminary characterization of L. helveticus autolysin. Appl Environ Microbiol. 1995;61:3391–3399. doi: 10.1128/aem.61.9.3391-3399.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Vandamme P, Pot B, Gillis M, De Vos P, Kersters K, Swings J. Polyphasic taxonomy, a consensus approach to bacterial systematics. Microbiol Rev. 1996;60:407–438. doi: 10.1128/mr.60.2.407-438.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vauterin L, Vauterin P. Computer aided objective comparison of electrophoresis patterns for grouping and identification of microorganisms. Eur Microbiol. 1992;1:37–41. [Google Scholar]
- 31.Veaux M, Neviani E, Giraffa G, Hermier J. Evidence for variability in the phenotypic expression of lysozyme resistance in Lactobacillus helveticus. Lait. 1991;71:75–85. [Google Scholar]
- 32.Wintzingerode F, Gobel U B, Stackebrandt E. Determination of microbial diversity in environmental samples: pitfalls of PCR-based rRNA analysis. FEMS Microbiol Rev. 1997;21:213–229. doi: 10.1111/j.1574-6976.1997.tb00351.x. [DOI] [PubMed] [Google Scholar]