A 42-year-old man was brought to the emergency department with 24 hours of fever to 102 Fahrenheit and altered mental status. Overnight, this had rapidly progressed to worsening global weakness, and decreasing level of consciousness with associated involuntary spasmodic movements of limbs.
Encephalopathy featuring involuntary spasmodic movements prioritizes evaluation for seizure, with status epilepticus a primary concern due to the progressive disturbance in sensorium.
However, involuntary movements are not specific to seizure. Decerebrate posturing and other brainstem lesions can mimic seizure and should be considered in any patient presenting with acute encephalopathy and abnormal movements. Further attention to the character of these movements on exam might clarify them as other movement abnormalities such as myoclonus. In myoclonus there are quick, involuntary muscle jerks due to muscle contractions (as seen in palatal myoclonus for example due to brainstem or cerebellar lesion) or muscle inhibition (as seen in asterixis due to hepatic encephalopathy).
Many insults to the brain—from primary brain processes such as stroke and meningitis to metabolic derangements, systemic inflammation, and toxins—can lead to disturbance in sensorium. Investigating the other two features of this presentation, fever and generalized weakness, may provide further clarity. The initial evaluation of fever is centered on infection, with secondary consideration given to medications, toxins, autoimmune diseases, malignancies, and endocrinopathy. Distinguishing between asthenia and true muscle weakness is a necessary step as the pattern and presence of true muscle weakness will help identify the syndrome.
Once family arrived, they provided additional history that two weeks prior to admission the patient had returned from a family trip to China. On this trip, he ate raw fish and visited a zoo but had no direct animal contact. During his travels, his daughter developed an upper respiratory infection though he was asymptomatic. 48 hours prior to presentation, he developed a headache, photophobia, nausea, and emesis. On the night prior to presentation, he fell while walking to the bathroom without loss of consciousness. His family noted that he was febrile, weak, requiring assistance ambulating, had difficulty swallowing water and that his “eyes were fixed straight forward.” The patient was previously healthy and did not use medications, alcohol, tobacco, or recreational drugs.
Subacute headache, photophobia, and fever is concerning for a progressive febrile central nervous system (CNS) syndrome with dysphagia, ophthalmoplegia, and weakness. In this context, dysphagia as a manifestation of bulbar weakness with concurrent ophthalmoplegia prioritizes concern for brainstem involvement. The differential also includes peripheral nervous system (PNS) involvement with inflammatory myopathies and neuromuscular junction (NMJ) disorders such as botulism and myasthenia gravis. Although fever is not a classic feature of these two disorders, an aspiration event as a complication of his dysphagia is plausible. A thorough exam will add clarity to these symptoms and neuroanatomic localization.
Raw fish consumption can lead to infection with viruses such as enterovirus family, bacterial pathogens such as Salmonella, Listeria, and Vibrio spp and parasites like Anisakis. Although these often present as acute gastroenteritis, more systemic illness including meningoencephalitis and prolonged course is possible. Listeria can lead to meningoencephalitis and rhombencephalitis. Infection with parasite Gnathostoma spp which is endemic to Southeast Asia can be complicated by headache, paralysis, and eosinophilic meningoencephalitis.
In the ED, his altered mental status necessitated urgent intubation. Immediately prior to intubation his temperature was 100 degrees Fahrenheit, heart rate 94 bpm, blood pressure 135/78, respiratory rate 18 breaths per minute and O2 saturation 98% on 21% FiO2. His cardiac, lung, abdominal and skin exams were normal. A full neurologic exam was performed after intubation and with sedation paused. He did not open his eyes to voice or noxious stimulation and did not respond to commands. He did not track or regard. No spontaneous movements of his eyes were apparent. Both pupils were sluggishly reactive to light. He had absent corneal reflex response, absent vestibulo-ocular reflex response, and no nystagmus in primary gaze. He had a weak gag with intact cough and absence of facial grimace. On motor examination, he had flaccid tone in his limbs with no spontaneous movement. With noxious stimulation, there was extensor posturing movement in his arms and flexion of the thigh, leg and dorsiflexion of the foot to distal and proximal stimuli. His deep tendon reflexes were absent throughout. He had upgoing hallux bilaterally with plantar stimulation.
Low-grade fever further supports the concern for progressive inflammatory syndrome. The profound encephalopathy suggests diffuse damage to bilateral cerebral hemispheres or pathology involving the ascending reticular activating system. Extensive bilateral involvement of cranial nerves prioritizes conditions affecting the brainstem. The Babinski sign and triple reflex point to upper motor neuron (UMN) involvement and the decerebrate posturing localizes to the brainstem, particularly the midbrain. The flaccid tone and absent reflexes are concerning for a lower motor neuron process; however early upper motor neuron processes can present similarly. Brainstem infarction, hemorrhage, demyelinating syndromes, and brainstem encephalitis should be further investigated with brain and cervical spine magnetic resonance imaging (MRI) scan and lumbar puncture.
The high morbidity and mortality associated with untreated acute bacterial meningitis most commonly caused by Streptococcus pneumoniae and Neisseria meningitidis warrants empiric treatment with vancomycin and ceftriaxone until a lumbar puncture can be safely performed. Given base rate of disease, addition of acyclovir to provide coverage for HSV meningoencephalitis is recommended.
He was started on empiric meningeal dosed intravenous (IV) acyclovir, ceftriaxone, vancomycin and IV thiamine. His white blood cell count was 15,600/L (reference 3,400–10,000/L), sodium was 133 mEq/L, serum glucose was 174 mmol/L, and renal function was normal. A head computed tomography (CT) scan without contrast showed no evidence of acute pathology or cerebral edema. Cerebrospinal fluid (CSF) analysis revealed 13 white blood cells/mm3 (40% lymphocytes, 27% polymorphonuclear cells, 32% monocytes, 1% basophils), 7 red blood cells, glucose of 67 mg/dL, and protein of 42 mg/dL. Opening pressure was 24.5 cm H2O. A brain and cervical spine MRI with and without contrast showed no abnormal enhancement, restricted diffusion, extra-axial collection, hydrocephalus, masses, or blood products.
This is a 42-year-old man who developed subacute fever, headaches, and photophobia that progressed to acute onset altered mental status and weakness after recent international travel with presentation concerning for progressive CNS inflammatory syndrome with exam localizing to the brainstem.
The most striking feature is the discrepancy between the unremarkable CSF and imaging findings and the degree of neurological deficits seen on exam that localizes to CNS. The CSF studies are significant for mild lymphocytic predominance with mild protein elevation. In general, lymphocytic pleocytosis is nonspecific as infections etiologies (including viral, fungal bacterial and parasitic), autoimmune processes, and malignancy can present with lymphocytic predominance.
In the context of concern for inflammatory CNS syndrome, the normal MRI despite pathologic brain involvement can either point to conditions like antibody-mediated encephalitides or vasculitides that are silent on imaging, or the need for a different imaging protocol. Often, a specific MR protocol such as Fast Imaging Employing Steady-State Acquisition (FIESTA), is needed to visualize brainstem abnormalities.
At this time, continuing the current antimicrobial therapy while CSF cultures and testing are pending is recommended. If the patient does not improve, addition of doxycycline to cover tickborne infections should be considered. CSF testing should include rare infectious etiologies such as West Nile virus given the neurological deficits patient is experiencing. Furthermore, Serum HIV testing is also warranted as a positive result would expand the differential diagnosis to include diseases such as progressive multifocal leukoencephalopathy.
A broad infectious work-up was initiated with early results including negative CSF testing for HSV polymerase chain reaction (PCR), VZV PCR and VZV IgM/IgG, West Nile virus IgM and IgG, and Enterovirus PCR. CSF gram stain and cultures were negative. Respiratory viral panel testing for influenza and parainfluenza, serum HIV and treponemal antibody testing were negative. After five days on antibiotic and antiviral treatment, his fever resolved without change in his neurologic examination. Broader viral, atypical bacterial, and fungal testing was ordered. Given lack of improvement, doxycycline was added to cover for atypical bacterial infections.
The CSF cell count in addition to the negative CSF cultures makes acute bacterial meningitis less likely. The resolution of fever could be due to either of the e processes: 1) There was partial response of underlying pathology to antimicrobial therapy, 2) The fever was due to a complication of the underlying disease (e.g., aspiration event) that the antimicrobial therapy treated, or 3) The natural history of underlying pathology unrelated to the therapies provided.
Given the tempo of this inflammatory CNS syndrome and suspicion for infectious etiology, a broader work-up for rare diseases is warranted. Improvement to doxycycline would be informative as it could represent infection by tickborne organisms. Tickborne infections are characterized by headache, fever, and myalgias caused by variety of animal (Leptospira, Brucella, Coxiella) and Tick (Rickettsia, Ehrlichia, Borrelia) exposures. Viral diseases such as rabies and arboviruses other than West Nile virus, and bacterial infections such as Burkholderia pseudomallei that can lead to encephalomyelitis with similar presentations to this patient. CSF serologies for these infections should be sent.
While the infectious work up is pursued and response to antimicrobial therapies closely monitored, autoimmune etiologies warrant consideration with CSF testing and serum serological work up at this time. Among these, special attention should be given to antibody-mediated encephalitis such as post-infectious complications (e.g., Bickerstaff’s brainstem encephalitis) and paraneoplastic syndromes as these diagnoses will influence treatment with discontinuation of antimicrobial therapies and initiation of therapies such as intravenous immune globulin (IVIg), plasma exchange and immunosuppression.
Additional testing, including nuchal skin biopsy for Rhabdovirus, serum testing for St. Louis, Eastern, Western, and California Equine Encephalitis, Japanese Encephalitis, and Zika, failed to identify an infectious cause.
A CT of abdomen and pelvis with IV contrast to rule out malignancy revealed a pancreatic mass, which was drained via fine need aspiration and was most consistent with a benign cyst.
Rheumatologic and autoimmune labs were also unrevealing including antinuclear antibody and rheumatoid factor testing. There were no unique CSF oligoclonal bands and normal IgG index. An electroencephalogram (EEG) showed normal state changes, sleep stages, and background reactivity but did have mild slowing. There were no seizures.
Given lack of improvement a week after presentation, he was treated empirically with methylprednisolone 1 gram intravenously daily for five days. Antibiotics and antiviral therapies were discontinued as testing for atypical bacterial infection including Rickettsia and Borrelia returned negative. A repeat MRI with contrast of the brain, again showed no parenchymal or intra-axial abnormalities, but now revealed subtle evidence of bilateral cranial III, IV, V, VI, and VII enhancement.
The EEG findings suggestive of relatively intact cortical function continues to keep the focus on brainstem pathology, in particular brainstem encephalitis. At this point, the thorough work up makes infectious etiology unlikely.
Regarding malignancy, there are limitations to CT scans, however the lack of non-neurological signs and symptoms based on the history and unremarkable serum studies provided, makes metastatic cancer less likely. Although, neurolymphomatosis which is characterized by direct infiltration of lymphoma into cranial nerves and peripheral nerves remains a possibility in this case.
In a majority of patients with paraneoplastic syndromes, neurological symptoms precede a cancer diagnosis. In addition to elevated protein in CSF, these tend to have unique oligoclonal bands and elevated IgG index which are not present here. An autoimmune disease to consider is neurosarcoidosis as neurological involvement without clear evidence of systemic disease has been reported.
The subacute nature, bulbar symptoms coupled with both UMN (Babinski sign and triple reflex) and LMN signs (flaccid paralysis and deep tendon areflexia) on exam, in additional to mild lymphocytic pleocytosis and subtle enhancement of upper cranial nerves on imaging are most consistent with Bickerstaff’s brainstem encephalitis (BBE). Like other Guillain-Barre Syndrome (GBS) spectrum diseases, the majority of BBE cases are preceded by infection which raises the possibility that the daughter’s illness caused a subclinical illness for the patient. Within the GBS spectrum diseases, BBE is considered a variant of Miller-Fisher syndrome (MFS), characterized by altered sensorium in addition to ophthalmoplegia and ataxia that is seen in MFS. Sending serum testing for BBE associated anti-GQ1b antibodies and empiric treatment with IVIg or plasma exchange would be the recommended next step.
Despite methylprednisolone treatment, the patient’s neurologic examination remained unchanged with persistent lack of eye movements and deep tendon reflexes and positive Babinski sign. Two weeks after presentation, he had an electromyography and nerve conduction study (EMG/NCS) which demonstrated an acute sensorimotor axonal polyneuropathy.
Given the results of EMG/NCS, serum testing for anti-GM1, GD1a, and GQ1b antibodies were sent. The patient was treated empirically with IVIg 2 grams/kg over three days. One week later, he started to follow one step axial commands and displayed volitional movement of arms and then legs. He was extubated and transferred to a rehabilitation hospital. There he gradually improved such that he was able to feed himself, communicate and walk. One month after IVIg treatment, his serum anti-GQ1b IgG antibody returned positive at titer of 1:800. Five months later, he was able to return to work while using a cane to ambulate, with residual mild right eye ophthalmoplegia, but no cognitive difficulties with intact memory and executive function.
Commentary
This case describes a 42-year-old man with a rapid decline in mental status. The patient underwent a comprehensive but initially unrevealing workup, leaving a striking dichotomy between the profound physical exam findings and paucity of abnormal test results.
Key features of this patient’s presentation included a neurological exam that localized to the brainstem, a subacute tempo of symptom onset, and multiple cranial nerve enhancement on brain MRI. This constellation of features led the discussants to the correct diagnosis of the GBS variant, Bickerstaff brainstem encephalitis.
Although GBS classically presents with ascending areflexic paralysis and CSF albuminocytologic dissociation, it is important to recognize that GBS encompasses a spectrum of disorders that span multiple phenotypic presentations with varying clinical severity (1). BBE is a particularly unique and devastating form of GBS defined by alterations in consciousness. Long tract signs, such as clonus, spasticity and hyper-reflexia (2) as was seen in this case with extensor posturing and clonus are also characteristic of BBE in addition to the triad of areflexia, ataxia, and ophthalmoplegia. This triad is also seen in the more commonly encountered GBS variant Miller Fisher syndrome (5–10% of GBS cases) where altered sensorium is absent. Notably, BBE encompasses 10% of this subset of MFS cases (3). As variants of GBS, MFS and BBE are autoimmune diseases of the peripheral nerves—and in the case of BBE also the brainstem. The pathogenesis is attributed to molecular mimicry initiating an autoimmune response typically triggered by antecedent infection (4).
The most vital part of the work-up for these patients is the history and physical exam. This is because when diagnosing GBS subtypes, imaging and lab testing results are highly variable. For example, the classic feature of albuminocytologic dissociation is only seen in 25% of BBE cases and 37% of MFS cases and was not present in our patient (4). With MR imaging as well, there are wide variations in findings in GBS variants—11 % of BBE patients have high intensity abnormalities on T2-weighted MRI of the brainstem, thalamus, cerebellum and cerebrum, whereas only 1% of MFS patients have abnormalities in the midbrain, cerebellum, or middle cerebellar peduncle (4). Although MR imaging is often normal, this patient did have delayed development of subtle cranial nerve enhancement on post-contrast sequences. Similarly, while the patient had evidence of an acute axonal sensorimotor neuropathy on EMG/NCS, routine motor and sensory nerve conduction study results are often normal for both groups.
When there is a clinical suspicion for BBE, the most consistently reliable evaluation is serum autoantibody testing. Anti-GQ1b antibodies are present in 83% of MFS and 68% of BBE patients (4), although the test turnaround time is often on the order of weeks. Autoantibodies in these syndromes are targeted against gangliosides which are chemical modifications found on the surfaces of cell membranes in peripheral nerves. The specific antigen targeted by the autoantibody in BBE and MFS (GQ1b) is strongly expressed in the oculomotor, trochlear, and abducens nerves, as well as muscle spindles in the limbs (5,6)—all locations with direct tie to the clinical features. One clinical sign in particular that can point to MFS or BBE and is tied to this pathophysiology is ophthalmoplegia. This exam finding has a more limited differential, including Wernicke’s encephalopathy, botulism, brainstem stroke, diphtheria, tick paralysis or infiltrative subarachnoid space process.
Thus, a reliance on the unique clinical findings—antecedent infection, ophthalmoplegia, a combination of upper and lower motor neuron signs—and strategic testing can lead to diagnosis of BBE. Empiric treatment is typically initiated without GQ1b antibody confirmation. Because of the rarity of MFS and BBE, there are no clinical trials testing therapies for these syndromes. However, given their close pathophysiological overlap with GBS, the mainstay of treatment includes plasma exchange and IVIg (7). Despite the striking presentation, patients with Bickerstaff typically make a full recovery by 6 months although diplopia, ataxia, and dysesthesia may persist in some patients (8).
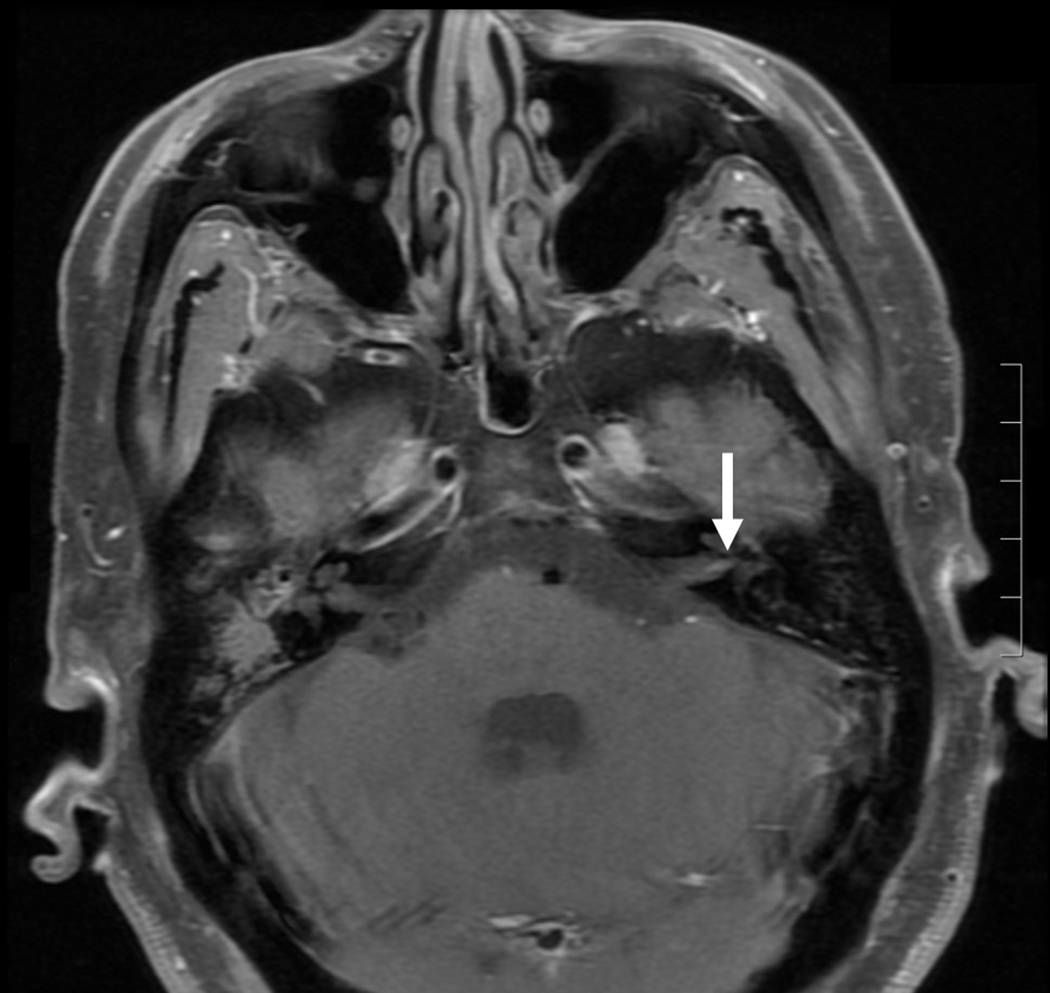
Figure 1:
T1 weighted Flex Brain MRI with white arrow indicating left cranial nerve V enhancement
Key teaching points:
Diagnosing BBE requires a thorough clinical assessment to localize to the brainstem and peripheral nerves with secondary supporting evidence from focused testing (MRI, CSF, EEG, EMG/NCS, antibodies).
The key clinical feature for diagnosing BBE or MFS is ophthalmoplegia with antecedent infection adding to the index of suspicion.
Anti-GQ1b testing is the most reliable testing for diagnosis of BBE but should not delay empiric treatment with IVIg or plasma exchange which can expedite recovery.
Acknowledgments
Brandon B Holmes: Funding source NIH 2R25NS070680
Footnotes
Nicholas J Thomas: None
Sharmin Shekarchian: None
Meredith A Bock: None
Rabih M Geha: None
Maulik P Shah: None
Conflict of interest disclosure and sources of support:
References
- 1.Yuki Nobuhiro, and Hartung Hans-Peter. “Guillain-Barré syndrome.” NEJM vol. 366,24 (2012): 2294–304. doi: 10.1056/NEJMra1114525 [DOI] [PubMed] [Google Scholar]
- 2.Horton E, Krishnamoorthy S, Reynolds L. Bickerstaff’s encephalitis. BMJ Case Rep. 2014;2014:bcr2014205336. 10.1136/bcr-2014-205336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dimachkie MM, Barohn RJ. Guillain-Barré syndrome and variants. Neurol Clin. 2013;31(2):491–510. 10.1016/j.ncl.2013.01.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ito M, Kuwabara S, Odaka M, Misawa S, Koga M, Hirata K, & Yuki N. Bickerstaff’s brainstem encephalitis and Fisher syndrome form a continuous spectrum: clinical analysis of 581 cases. J Neurol. 2008;255(5), 674–682. 10.1007/s00415-008-0775-0 [DOI] [PubMed] [Google Scholar]
- 5.Chiba A, Kusunoki S, Obata H, Machinami R, Kanazawa I. Serum anti-GQ1b IgG antibody is associated with ophthalmoplegia in Miller Fisher syndrome and Guillain-Barré syndrome: clinical and immunohistochemical studies. Neurology 1993;43:1911–7. 10.1212/wnl.43.10.1911 [DOI] [PubMed] [Google Scholar]
- 6.Liu JX, Willison HJ, Pedrosa-Domellof F. Immunolocalization of GQ1b and related gangliosides in human extraocular neuromuscular junctions and muscle spindles. Invest Ophthalmol Vis Sci 2009; 50:3226–32. 10.1167/iovs.08-3333 [DOI] [PubMed] [Google Scholar]
- 7.Overell JR, Hsieh ST, Odaka M, Yuki N, Willison HJ. Treatment for Fisher syndrome, Bickerstaff’s brainstem encephalitis and related disorders. Cochrane Database Syst Rev. 2007. Jan 24;(1):CD004761. 10.1002/14651858.CD004761.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Odaka M, Yuki N, Yamada M, et al. Bickerstaff’s brainstem encephalitis: clinical features of 62 cases and a subgroup associated with Guillain Barre syndrome. Brain. 2003; 126:2279–2290. 10.1093/brain/awg233 [DOI] [PubMed] [Google Scholar]