Abstract
Transcranial magnetic stimulation (TMS) is used to probe inhibitory intracortical neurotransmission and has been used to infer the neurobiological dysfunction that may underly several neurological disorders. One technique, short-interval intracortical inhibition (SICI), indexes gamma-aminobutyric acid (GABA) mediated inhibitory activity and is a promising biomarker. However emerging evidence suggests SICI does not exclusively represent GABAergic activity because it may be influenced by inter-individual differences in the specific excitatory neural populations activated by TMS. Here we used the latency of TMS motor evoked potentials (MEPs) to index these inter-individual differences, and found that a significant proportion of the observed variability in SICI magnitude was accounted for by MEP latency, r = − 0.57, r2 = 0.33, p = .014. We conclude that SICI is influenced by inter-individual differences in the excitatory neural populations activated by TMS, reducing the precision of this GABAergic probe. Interpreting SICI measures in the context of MEP latency may facilitate a more precise assessment of GABAergic intracortical inhibition. The reduced cortical inhibition observed in some neuropathologies could be influenced by reduced activity in specific excitatory neural populations. Including MEP latency assessment in research investigating SICI in clinical groups could assist in differentiating the cortical circuits impacted by neurological disorders.
Subject terms: Neural circuits, Synaptic transmission
Introduction
Short-interval intracortical inhibition (SICI) is a paired-pulse transcranial magnetic stimulation (TMS) technique that provides a non-invasive indication of intracortical inhibitory activity mediated by gamma-aminobutyric acid (GABA) A receptors1. SICI is abnormally reduced in several neurological disorders2–4, has recently informed the differentiation of dementia subtypes5, and has been found to predict response to TMS neuromodulation6. However, SICI assessment is influenced by inter-individual differences in the neural populations activated by TMS7,8, and this relationship may explain up to half of the inter-individual variability observed in SICI7. Indeed, it has been suggested that levels of SICI may depend on individual patterns of TMS neural recruitment as much as they do on the inhibitory activity that SICI attempts to assess9. Although SICI provides a valuable non-invasive marker of GABAergic activity, our understanding of how SICI assessment is affected by individual differences in the neural populations recruited by TMS remains incomplete.
Careful modification of TMS intensity, and cortical current direction (i.e., via changes in TMS coil orientation) can reveal inter-individual variations in the neural populations activated by TMS10. Invasive epidural recordings reveal that TMS delivered over the primary motor cortex (M1) elicits a descending cortico-spinal volley (CSV) of distinct waves, and individual waves are currently thought to represent activity in non-identical neural populations11,12. The CSV can produce a motor evoked potential (MEP) in peripheral muscle, which is quantifiable with electromyography (EMG)13,14. The onset latency of these MEPs can be used to indicate the likely neural populations activated in an individual11. Higher intensity latero-medially (LM) directed stimulation can directly activate axons of cortico-spinal neurons. Here the CSV commences with its earliest possible component, known as a direct (D) wave, and the later components, termed indirect (I) waves (I1, I2, I3 etc.), are generated by trans-synaptic activation of cortico-spinal neurons15. The presence of D waves in a CSV evokes MEPs of the shortest latency, which can be used to control for individual differences in the cortex to muscle pathway16. Lower intensity TMS delivered using posterior-anterior (PA) directed current tends to evoke a CSV commencing with an earlier I1-wave. Here, the latency of the CSV and resulting MEP is 1–1.5 ms longer than when D waves are present. Using an anterior–posterior (AP) directed current tends to evoke CSVs commencing with a later I wave (often corresponding to the I3 wave), and the resultant MEPs are approximately 3–7 ms later than D wave latency10,17. (See supplementary material Fig. S1 for illustration.) In summary, PA and AP MEP latencies, with LM latency subtracted, provide a non-invasive indication of the earliest component of the CSV that is recruited following TMS to M116.
Crucially it is only the later I waves (I3 and later) that appear to be inhibited by SICI, any early I waves present during the test are not reduced in amplitude18,19. (See supplementary material Fig. S2 for illustration.) SICI uses a subthreshold (i.e., below the stimulation intensity required to elicit an MEP) conditioning stimulus (CS) to activate inhibitory interneurons that suppress the amplitude of the MEP elicited by a test stimulus (TS) delivered 1–6 ms later20. At a 1 ms inter-stimulus interval, the suppression is thought to be substantially influenced by the neuronal refractory period1. While at a 2.5–3 ms interval the extent of this reduction in MEP amplitude, compared to MEP amplitude elicited by the TS alone, indicates levels of GABAergic inhibition1 but may also reflect individual differences in the composition of the TMS-evoked CSV9. The observations that the TS inhibition stems only from inhibition of later I waves18,21 provide a theoretical basis for the reports7,8 of a relationship between MEP latency difference and SICI. Given that early I waves are not inhibited by the CS, their presence would likely contribute to TS MEP amplitude and therefore reduce the assessed SICI. Interestingly, in individuals with longer MEP latency difference, and hence no unaffected early I waves in their CSV, SICI may provide a more accurate measure of GABAergic inhibitory activity.
Two recent reports have begun to describe the practical nature of the relationship between SICI and MEP latency difference7,8. One reported an association between AP latency and SICI assessed in the PA direction8 and another reported a strong association between AP latency and AP SICI7. In both cases, longer MEP latency differences were associated with greater assessed inhibition, in line with what would be expected given the specific interaction each measure has with the CSV. Both of these studies utilized threshold tracked (tt) SICI where the TS intensity is increased until it overcomes the inhibitory influence of the CS22. Given that adjustment to stimulus intensity is known to alter CSV composition17,23, and that the composition of the CSV appears to drive the relationship under investigation, we therefore investigated the untested relationship between conventional amplitude-ratio SICI (where the TS intensity is held constant) measured in the PA and AP current directions and MEP latency difference. We expected that greater SICI would be associated with longer latency difference.
Materials and methods
Participants
Twenty-eight (9 male) healthy right-handed participants aged 18–42 (M = 25.22; SD = 5.37) were recruited from an Australian university and surrounding area via online and poster advertisements. Standard TMS exclusion criteria and pre/post safety screening procedures were applied24,25, including exclusion of potential participants with any self-reported history of neurological or psychological disorder, or current medical or recreational use of psychoactive drugs. The study was approved by the Deakin University Human Research Ethics Committee and all participants provided written informed consent in accordance with the Declaration of Helsinki.
Experimental protocol
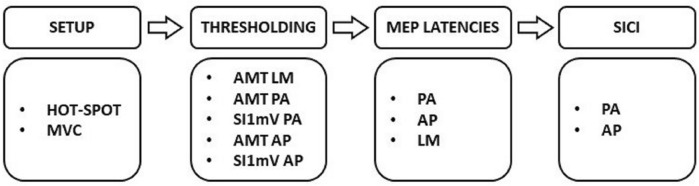
Participants were seated in an adjustable chair with their right arm resting on a table positioned just above their lap. See Fig. 1 for the experimental procedure (described in detail below). Briefly, the experiment began with locating the stimulation site, then the relevant coil orientations were used to determine motor thresholds, apply single pulse TMS to assess MEP latency, and administer paired pulse stimulation to assess intracortical inhibition. Experiments were well tolerated by participants and no significant adverse effects were reported.
Figure 1.
Experimental procedure. MVC maximum voluntary contraction, AMT active motor threshold, SI stimulus intensity, SICI short-interval intracortical inhibition, PA posterior–anterior, AP anterior–posterior, LM latero-medial.
Electromyographic recording
Surface EMG was recorded from the first dorsal interosseous (FDI) muscle of the right hand. Wet gel 10 mm Ag/AgCl electrodes were applied in a belly-tendon montage, grounded on the ulnar styloid process. EMG signals were amplified (× 1000; Bio Amp–ADInstruments New Zealand), bandpass filtered (1 Hz–2 kHz), digitized (10 kHz; PowerLab 4/35; ADInstruments), and recorded (LabChart 8.0—ADInstruments) from 200 ms before to 300 ms after TMS pulses26.
Transcranial magnetic stimulation
Monophasic TMS pulses of 100 µs rise time and 1 ms duration were generated with a Magstim Bistim2 system (Magstim United Kingdom) and delivered with a 70 mm Magstim figure-8 coil via the Bistim connecting module. Hotspot and thresholds were assessed via single pulses delivered in Bistim mode, latency and SICI blocks were assessed in independent Bistim triggering mode27. The left M1 FDI hot spot for the contralateral right hand was located in accordance with previous guidelines26 using PA current (note, all current directions refer to cortical current). The hot spot location was marked in a neuro-navigation system (BrainSight, Rogue Research Inc. Canada) and used for all subsequent TMS. Coil position was continually monitored throughout the experiment using the neuro-navigation system. Coil position errors were low with an average (and SD) distance from the hot spot of 1.15 (0.80) mm, tilt away from the target of 2.06 (1.59) degrees and a mean absolute twist error of 1.59 (1.50) degrees. Motor thresholds were assessed with 30 TMS pulses using maximum-likelihood parameter estimation by sequential testing, implemented in the TMS Motor Assessment Tool 2.028, to establish a stimulus intensity with a 50% probability of eliciting an MEP meeting the target criteria. For active motor threshold (AMT) which was assessed in PA, AP and LM current directions, the MEPs meeting criteria had amplitudes greater than 100 µV29, were distinguishable from background EMG, and occurred within a physiologically possible latency range (15–35 ms). During active thresholding, and subsequent latency assessments in active muscle, participants maintained a slight voluntary contraction of the FDI muscle (10% of maximum as measured with a force transducer and guided by visual feedback). Stimulus intensity with a target amplitude of 1 mV (SI1mV) was assessed in resting muscle in PA and AP current directions.
MEP latency assessment
MEP onset was assessed as per Hamada et.al16 using PA, AP, and LM current directions, with intensities determined by reference to the thresholds established in each direction. For PA and AP latencies in active muscle, 20 trials were assessed in each direction using a stimulus intensity set at 110% AMT. LM latencies were assessed with 10 trials in active muscle, and a stimulus intensity of 150% AMT in order to increase the likelihood of evoking an MEP commencing with a D wave30. Here, as previously16, 10 trials at this higher stimulus intensity provided sufficiently reliable latency estimates31. MEP onset was also assessed in the unconditioned test pulses used to calculate SICI. These comprised 20 trials delivered at SI1mV in resting muscle, using both PA and AP current.
SICI assessment
SICI was assessed in resting muscle in the PA and AP current directions. The CS was delivered at 90% AMT, followed 2.5 ms later by the TS delivered at SI1mV32,33. The intensities were determined with reference to the relevant thresholds established in each direction. Twenty SICI conditioned trials and 20 single pulse SI1mV trials were interleaved and jittered with 5, 6, and 7 s inter-stimulus intervals in pseudo-randomized order.
Data analysis
Because voluntary contraction can substantially alter the composition of the CSV34, and also alter SICI35, any trials with EMG amplitude greater than 20 µV in the 100 ms prior to the stimulus (24%, 545 of 2240 trials) were excluded from SICI calculations. Because SICI calculation relies on averaged amplitudes, we made a pragmatic decision that any average amplitudes calculated from less than 5 MEPs were likely unreliable36 and therefore were not calculated. This meant that in any block of 20 SICI trials (conditioned or unconditioned) the median number of trials averaged was 18 (min 5, max20). We ultimately calculated SICIPA magnitudes for 23 participants and SICIAP magnitudes for 25 participants. MEP latencies were calculated for each participant and each group of latency trials using a custom Matlab script. EMG signals for the block were averaged8 and the latency was identified as the first timepoint following 15 ms post stimulus where the averaged signal exceeded the mean plus 5 standard deviations of the averaged signal in the 100 ms prior to the stimulus. Any pulses that did not reach their individual 5 standard deviation threshold (14%, 350 of 2520 trials) were excluded from the averaging. Because latency can be altered by voluntary contraction23 any resting latency trials with EMG amplitude greater than 20 µV in the 100 ms prior to the stimulus (24%, 545 of 1120 trials) were also excluded from the averaging of EMG signals for resting PA and AP latency detection. Because MEP latency is more reliable than MEP amplitude37 we made a pragmatic decision that latency should only be calculated where at least 3 trials were available to create the averaged EMG signal. This meant that in any block of 20 latency trials the median number of trials averaged was 16 (min 3, max 20), and for LM latency all blocks were averaged from 10 trials. We ultimately obtained PA and LM active latencies for all (28) participants, AP active and AP resting for 27 participants, and PA resting for 26 participants. The averaged traces and detected latencies were plotted for visual inspection revealing 7 clear failures (of the 136 plots), due to noise or dc drift in the signal. These latencies were corrected by manually adjusting the onset to the first subsequent point clearly associated with the MEP response where the EMG signal exceeded the mean plus 5 SD of the pre-pulse EMG7,16. The mean plus 5 SD threshold was chosen to keep the failed detections, and hence the manual adjustments, to a minimum in order to maximize the objectivity of the extracted latency metric. We also made a further check on our automatically detected latencies (detailed and reported in the “Supplementary material”) and confirmed that they were similar to the onsets observed via visual inspection of overlay plots of all trials for each block.
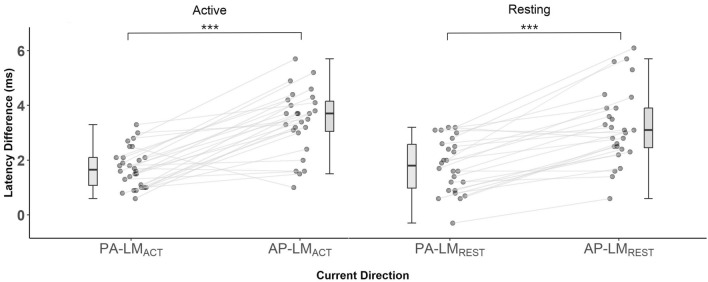
For each participant, active LM latency (the indicator of D wave latency) was subtracted from both resting and active PA and AP latencies to create latency difference metrics16. This provided an indication of whether the CSVs evoked in each direction (PA and AP) and condition (resting or active) tended to commence with earlier (i.e. smaller values, closer to D wave latency) or later (i.e., larger values, further from D wave latency) I waves. The resulting latency difference metrics were PA-LMACT, AP-LMACT, PA-LMREST, and AP-LMREST.
For each participant and current direction, (excluding trials containing EMG activity as explained above) SICI was calculated by expressing the average of the conditioned MEP amplitudes as a percentage of the average unconditioned amplitudes (i.e. (conditioned amplitude/unconditioned amplitude) × 100, 100 = no inhibition, below 100 = inhibition). The resulting metrics were SICIPA and SICIAP.
Statistical analysis
Assumption testing, analyses, post-hoc testing, and adjustments for multiple comparison are detailed here in order to simplify the presentation of the results section that follows. Firstly, three separate preliminary comparisons were conducted to confirm our measurements were in accordance with values previously reported16,23. These comparisons were of the motor thresholds (AMTPA, AMTAP, AMTLM, SI1mVPA, and SI1mVAP), raw latencies (PAACTIVE, APACTIVE, LMACTIVE, PARESTING, and APRESTING), and latency difference scores (PA-LMACT, AP-LMACT, PA-LMREST, and AP-LMREST). Quantile—standardized residual plots suggested that motor thresholds, raw latencies, and latency difference scores were approximately normally distributed. However none in this series met the assumption of sphericity assessed via Mauchley’s test therefore repeated-measures ANOVA with Greenhouse–Geisser correction applied was used for comparisons. Post-hoc pair-wise comparisons with Tukey’s adjustment were then conducted (see Supplementary Material Tables S1–S3). SICIPA and SICIAP difference scores were normally distributed, as assessed via a Shapiro–Wilk test and density plot, therefore a paired-samples T-test was used to compare them. For these preliminary analyses effect sizes are reported using Hedge’s gav to account for the inherent correlation between these intra-individual effects, and to facilitate any future use of these results38.
Scatterplots were assessed prior to the main correlational analyses and indicated no outliers or non-linear relationships were present. Linear correlations (Pearson’s) were used to test for a relationship between SICIPA, SICIAP and each of the four latency difference metrics. We note some positive skew (a possible floor effect) was present in SICIAP. This may suggest a degree of caution in the interpretation of the SICIAP Pearson’s coefficients. Family-wise error was controlled separately for the SICIPA and SICIAP comparisons with Holm-Bonferroni adjusted p values. Statistical analyses were performed using RStudio Version 1.4.1106.
Results
Descriptive statistics for SICI magnitudes, MEP latencies, and motor thresholds are reported in Table 1.
Table 1.
Descriptive statistics.
| Variable | M | SD | Min | Max | N |
|---|---|---|---|---|---|
| SICI (%) | |||||
| SICIPA | 44.10 | 25.78 | 2.55 | 102.90 | 24 |
| SICIAP | 26.67 | 23.47 | 1.57 | 69.25 | 26 |
| Raw latency (ms) | |||||
| PAACTIVE | 22.46 | 1.55 | 19.10 | 26.30 | 28 |
| APACTIVE | 24.24 | 1.92 | 19.50 | 27.30 | 27 |
| LMACTIVE | 20.73 | 1.53 | 17.50 | 23.00 | 28 |
| PARESTING | 22.66 | 1.54 | 19.20 | 26.20 | 26 |
| APRESTING | 24.00 | 1.82 | 20.20 | 29.10 | 27 |
| Latency difference (ms) | |||||
| PA-LMACT | 1.73 | 0.72 | 0.60 | 3.30 | 28 |
| AP-LMACT | 3.44 | 1.16 | 1.00 | 5.70 | 27 |
| PA-LMREST | 1.81 | 0.98 | − 0.30 | 3.20 | 26 |
| AP-LMREST | 3.24 | 1.35 | 0.60 | 6.1 | 27 |
| AMT (% MSO) | |||||
| AMTPA | 37.82 | 6.69 | 27.00 | 56.00 | 28 |
| AMTAP | 49.75 | 7.37 | 36.00 | 61.00 | 28 |
| AMTLM | 43.46 | 7.77 | 32.00 | 63.00 | 28 |
| SI1mV (% MSO) | |||||
| SI1mVPA | 58.57 | 10.89 | 43.00 | 81.00 | 28 |
| SI1mVAP | 71.39 | 11.67 | 50.00 | 94.00 | 28 |
SICI % Short-Interval Intracortical Inhibition, 100% no inhibition, < 100% inhibition, PA posterior–anterior, AP anterior–posterior, LM latero-medial; Raw Latency, ms, latencies in indicated condition; Latency Difference, ms, difference from LM latency; AMT, active motor threshold; SI1mV, stimulus intensity 1 mV; MSO, maximal stimulator output (MSO).
Cortico-spinal excitability—the effect of current direction and stimulus intensity
Repeated-measures ANOVA indicated that there were significant differences in motor thresholds (AMTPA, AMTAP, AMTLM, SI1mVPA, and SI1mVAP), F(2.48, 67.05) = 148.82, p < 0.001, ω2 = 0.84. Post-hoc comparisons with Tukey’s adjustment indicated there were significant differences between all thresholds (see table S1 in “Supplementary Material”). The lowest was AMTPA followed in increasing order by AMTLM, AMTAP, SI1mVPA, and SI1mVAP.
Short-interval intracortical inhibition—the effect of current direction
Participants demonstrated significantly greater SICI (i.e., greater MEP suppression) when assessed with AP relative to PA current, t(23) = 3.02, p = 0.006, Hedge’s gav = 0.68. See Fig. 2. for PA and AP SICI plot and “Supplementary Material” for discussion.
Figure 2.
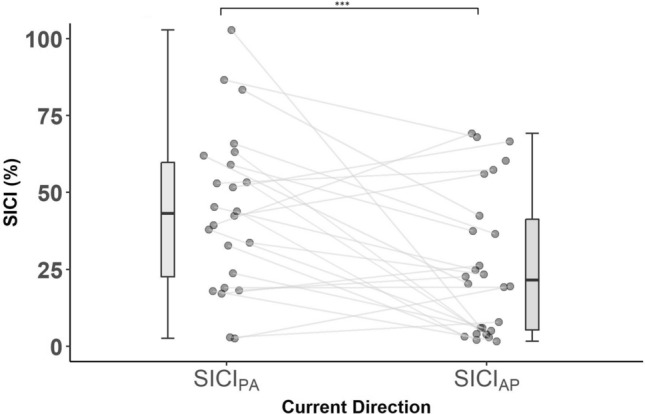
SICI in PA and AP current directions. SICI (%) Short-Interval Intracortical Inhibition, 100% no inhibition, < 100% inhibition; Current Direction, PA posterior–anterior, AP anterior–posterior. Boxplots showing medians and IQR; ***p < 0.001.
MEP latency—indicator of I wave recruitment
Repeated-measures ANOVA indicated significant differences in raw latencies (PAACTIVE, APACTIVE, LMACTIVE, PARESTING, APRESTING), F(2.54, 59.22) = 69.61, p < 0.001, ω2 = 0.74. Tukey’s post-hoc comparisons (see supplementary Table S2) indicated raw latencies were significantly different across all current directions, being shortest with LM current and longest with AP current. There was no significant difference between resting and active PA, or between resting and active AP latencies, however we note these were obtained under different conditions (see “Supplementary Material” for discussion).
There were significant differences in latency difference metrics (PA-LMACT, AP-LMACT, PA-LMREST, AP-LMREST) F(2.04, 47.02) = 27.69, p < 0.001, ω2 = 0.52. Tukey’s post-hoc comparisons (see supplementary material table S3) indicated all PA measures were significantly different to all AP measures (all p < 0.001), but no significant differences existed between PA-LMACT and PA-LMREST or AP-LMACT and AP-LMREST. See Fig. 3 for individual PA-LM to AP-LM latency difference plots.
Figure 3.
PA and AP Latency differences. Latency difference (ms), Individual PA and AP latencies subtract active LM latency; PA, posterior-anterior; AP, anterior–posterior; LM, latero-medial; Boxplots showing medians and IQR; ***pTukey < 0.001.
Relationship between SICI and MEP latency difference metrics
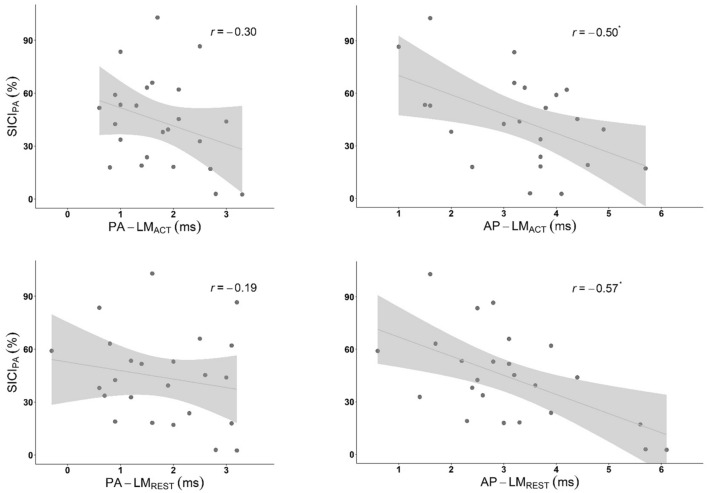
Greater SICIPA was associated with longer AP latency differences assessed in the active, r = − 0.50, r2 = 0.25, N = 23, padj = 0.048 praw = 0.015 (two-tailed), and resting FDI, r = − 0.57, r2 = 0.33, N = 24, padj = 0.014, praw = 0.004, (two-tailed). The associations between SICIPA and PA latency differences were weak and not statistically significant (active, r = − 0.30, r2 = 0.09, N = 24, padj = 0.303 praw = 0.151 (two-tailed), resting, r = − 0.19, r2 = 0.05, N = 24, padj = 0.379, praw = 0.379, (two-tailed). Scatterplots are presented in Fig. 4.
Figure 4.
SICIPA and MEP latency. SICI (%), Short-Interval Intracortical Inhibition 100% = no inhibition, < 100% = inhibition; Latency Difference (ms), Individual PA and AP latencies subtract active LM latency; PA, posterior-anterior; AP, anterior–posterior; LM, latero-medial; r, Pearson’s correlation coefficient; Shaded area, 95% CI; *padj < 0.05.
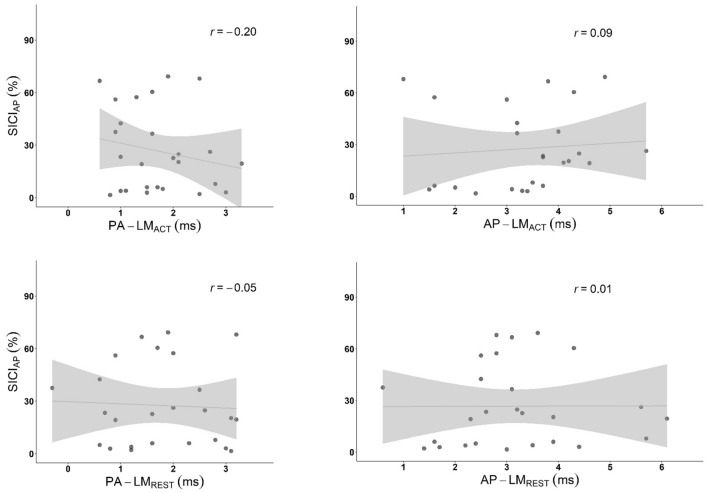
SICIAP was not significantly associated with any latency difference: PA-LMACT, r = − 0.20, r2 = 0.04, N = 26, padj = 1, praw = 0.331; PA-LMREST, r = − 0.051, r2 = 0.002, N = 25, padj = 1, praw = 0.807; AP-LMACT, r = − 0.09, r2 = 0.008, N = 25, padj = 1, praw = 0.663; and AP-LMREST, r = − 0.005, r2 = 0.00003, N = 26, padj = 1, praw = 0.980. See scatterplots in Fig. 5.
Figure 5.
SICIAP and MEP Latency. SICI (%), Short-Interval Intracortical Inhibition 100% = no inhibition, < 100% = inhibition; Latency Difference (ms), Individual PA and AP latencies subtract active LM latency; PA, posterior-anterior; AP, anterior–posterior; LM, latero-medial; r, Pearson’s correlation coefficient.
Discussion
The current study sought to further characterize the nature of the relationship between SICI and inter-individual variations in the neural populations activated by TMS (as assessed using MEP latency difference metrics). The relationship was examined, using both PA and AP current directions, by assessing MEP latency in active and resting muscle, and assessing SICI magnitude in resting muscle. Here we used conventional SICI where, because the test stimulus intensity is held constant, the composition of the test CSV is likely less variable than in the ttSICI examined previously. Our preliminary analyses indicated that mean PA-LMACT and AP-LMACT latency differences (1.7 ms and 3.4 ms respectively, see “Supplementary Material” for further discussion) were consistent with values previously used to index differences in I wave recruitment13,16. In line with our expectations, our results show that greater SICIPA is associated with longer AP-LM MEP latency difference, explaining up to 33% of the inter-individual variability in conventional SICI. The relationship between SICI and latency difference seen here, and in previous work, has significant implications (detailed below) for the interpretation of each measure. Contrary to our expectations, SICIAP was not associated with MEP latency difference.
We found that 33% of the observed individual variability in intracortical inhibition assessed with PA current was explained by AP-LM latency difference. As expected, longer AP-LM latencies were associated with greater SICIPA, supporting the idea that individual levels of SICI depend on individual tendency toward earlier or later I wave recruitment. Both resting and active AP-LM latencies were closely associated with SICIPA. To our knowledge, this is the first report of an association between AP-LMREST latency and SICIPA, suggesting that, for our current purpose, probing MEP latency at SI1mV in resting muscle may be equally as useful as the more common 110% AMT probe used in active muscle. Our findings also support the previous account of a correlation between AP-LMACT latency and ttSICIPA8, and demonstrate here that the correlation is also apparent when the TS is held constant. While AP-LM latencies do not directly reflect the PA TS used in SICIPA, they are used to infer the extent of an individual’s later I wave recruitment. It seems logical that SICI’s selective inhibition of I3 and later waves, combined with SICI’s lack of impact on early I waves, could be driving the relationships we observed. We did not detect statistically significant relationships between SICIPA and PA-LM latencies. Perhaps PA-LM latency is not sufficiently sensitive to the late I waves inhibited by SICI, however we note the scatterplots and coefficients appear to be in agreement with the direction of the relationship we found for AP-LM latencies.
Unlike with SICIPA, we observed no linear relationships between SICIAP and any latency difference measure. This was in contrast to a previous report of an association between active AP-LM latency and ttSICIAP7. We acknowledge that our study may be limited by the possibility of a floor effect being present in our conventional SICIAP, as evidenced in the scatterplots and Fig. 4, which may have obscured any relationship between SICIAP and latency difference. Alternatively, the absence of an association between SICIAP and latency difference measures could be because assessing SICI with AP current avoids early I wave recruitment that would otherwise mask assessed inhibition.
Our findings of an association between conventional SICIPA and AP-LM latencies represent the third report of a significant, likely neurophysiologically-driven, and arguably underappreciated relationship between SICI and MEP latency difference. Here we demonstrate for the first time that this association is present for conventional SICI, where test stimulus CSV composition is held relatively constant. Accounting for this relationship could contribute to a more individualized understanding of both GABAergic inhibitory activity and TMS preferential recruitment of distinct neural populations. The major implication here is that interpreting SICI in the context of latency difference could increase the accuracy and utility of the measure. For example, in individuals with short latency difference, who therefore have early I waves present in the CSV, the absence of SICI, i.e. no inhibition of the SICI test pulse, could mean that no later I waves were present to be inhibited, or alternatively that GABAergic activity was not apparent. In these individuals, a test pulse that reveals inhibition indicates the presence of later I waves, but the assessed SICI may only provide a diluted measure of GABAergic activity due to the presence of unaffected early I waves. However, in individuals with longer latency difference who therefore do not have early I waves present in their CSV, the extent of inhibition of the test pulse may reflect a more accurate index of GABAergic activity.
Greater precision in SICI assessment may be important in clinical investigations that report reduced SICI in a range of neurological disorders2–4,39. It is possible that the use of long latency difference subgroups may allow for more accurate comparisons of SICI function in clinical and healthy subjects. We also note that differences in I wave recruitment could be contributing to the SICI dysfunction identified in clinical populations. The inclusion of latency difference assessment in future SICI research could speak to this contribution, potentially increasing our understanding of the mechanisms underlying the reduced SICI observed in some clinical groups. A further implication of our findings is that conventional SICIPA may provide a diluted index of GABAergic inhibitory activity. As suggested previously40, SICIAP may deliver a more accurate assessment of intracortical inhibition by avoiding the unaffected early I wave recruitment that might mask assessed inhibition.
There are also implications for interpreting MEP latency. A growing body of research suggests that later I wave recruitment (assessed via MEP latency difference) is associated with TMS-induced neuromodulation outcomes, and with learning16,41–44, but because latency can only reflect the first component of the CSV, short latencies cannot speak to the presence of later I waves. In individuals with short latency difference the presence of SICI could indicate that later I waves are also present in their CSVs. We suggest that including SICI assessment in future research could, at the individual level, facilitate a more detailed understanding of how preferential TMS recruitment of distinct neural populations impacts TMS neuromodulation outcomes.
Our study used single and paired pulse TMS to examine the relationship between MEP latency difference and SICI assessed using PA and AP current. Latency difference was used to indicate whether individual motor response to TMS tended to commence with earlier or later I waves. We found that a significant proportion of the observed variability in PA SICI magnitude can be accounted for by MEP latency difference, reflecting individual differences in the neural populations preferentially activated by TMS. However, MEP latency difference did not account for the variability we observed in AP SICI. We suggest that interpreting SICI measures in the context of individual I wave recruitment patterns will contribute to more precise assessment of GABAergic intracortical inhibition, that AP SICI could more accurately reflect inhibitory processes, and that accounting for SICI could enhance our understanding of the relationship between MEP latency difference and TMS neuromodulation outcomes.
Supplementary Information
Author contributions
A.C. conceived the study, designed the experiments, performed the experiments, performed the statistical analyses, and wrote the manuscript. D.C., M.D., P.E., and C.H. designed the experiments, performed the experiments, and reviewed and edited the manuscript. G.O. and J.H. designed the experiments, and reviewed and edited the manuscript. B.S., P.B., and I.F. performed the experiments, and reviewed and edited the manuscript. All authors approved the final manuscript.
Funding
Andris Cerins was supported by an Australian Government Research Training Program (RTP) Scholarship.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-022-14271-1.
References
- 1.Ziemann U, et al. TMS and drugs revisited 2014. Clin. Neurophysiol. 2015;126:1847–1868. doi: 10.1016/j.clinph.2014.08.028. [DOI] [PubMed] [Google Scholar]
- 2.Hanajima R, et al. Difference in intracortical inhibition of the motor cortex between cortical myoclonus and focal hand dystonia. Clin. Neurophysiol. 2008;119:1400–1407. doi: 10.1016/j.clinph.2008.02.009. [DOI] [PubMed] [Google Scholar]
- 3.Ziemann U, et al. Impaired motor cortex inhibition in patients with amyotrophic lateral sclerosis: Evidence from paired transcranial magnetic stimulation. Neurology. 1997;49:1292–1298. doi: 10.1212/WNL.49.5.1292. [DOI] [PubMed] [Google Scholar]
- 4.Morin-Parent F, Champigny C, Lacroix A, Corbin F, Lepage J-F. Hyperexcitability and impaired intracortical inhibition in patients with fragile-X syndrome. Transl. Psychiatry. 2019;9:312. doi: 10.1038/s41398-019-0650-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Benussi A, et al. Classification accuracy of TMS for the diagnosis of mild cognitive impairment. Brain Stimul. 2021;14:241–249. doi: 10.1016/j.brs.2021.01.004. [DOI] [PubMed] [Google Scholar]
- 6.Murase N, Cengiz B, Rothwell JC. Inter-individual variation in the after-effect of paired associative stimulation can be predicted from short-interval intracortical inhibition with the threshold tracking method. Brain Stimul. 2015;8:105–113. doi: 10.1016/j.brs.2014.09.010. [DOI] [PubMed] [Google Scholar]
- 7.Cirillo J, Byblow WD. Threshold tracking primary motor cortex inhibition: The influence of current direction. Eur. J. Neurosci. 2016;44:2614–2621. doi: 10.1111/ejn.13369. [DOI] [PubMed] [Google Scholar]
- 8.Higashihara, M., Van den Bos, M. A., Menon, P., Kiernan, M. C. & Vucic, S. Interneuronal networks mediate cortical inhibition and facilitation. Clin. Neurophysiol. (2020). [DOI] [PubMed]
- 9.Di Lazzaro V, Rothwell JC. Corticospinal activity evoked and modulated by non-invasive stimulation of the intact human motor cortex. J. Physiol. 2014;592:4115–4128. doi: 10.1113/jphysiol.2014.274316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kaneko K, Kawai S, Fuchigami Y, Morita H, Ofuji A. The effect of current direction induced by transcranial magnetic stimulation on the corticospinal excitability in human brain. Electroencephalogr. Clin. Neurophysiol./Electromyogr. Motor Control. 1996;101:478–482. doi: 10.1016/s0013-4694(96)96021-x. [DOI] [PubMed] [Google Scholar]
- 11.Ziemann U. I-waves in motor cortex revisited. Exp. Brain Res. 2020 doi: 10.1007/s00221-020-05764-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Opie, G. M. & Semmler, J. G. Preferential activation of unique motor cortical networks with transcranial magnetic stimulation: A review of the physiological, functional, and clinical evidence. Neuromodulat. Technol. Neural Interface (2020). [DOI] [PubMed]
- 13.Day B, et al. Electric and magnetic stimulation of human motor cortex: Surface EMG and single motor unit responses. J. Physiol. 1989;412:449–473. doi: 10.1113/jphysiol.1989.sp017626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kaneko K, Kawai S, Fuchigami Y, Shiraishi G, Ito T. Effect of stimulus intensity and voluntary contraction on corticospinal potentials following transcranial magnetic stimulation. J. Neurol. Sci. 1996;139:131–136. doi: 10.1016/0022-510X(96)00050-0. [DOI] [PubMed] [Google Scholar]
- 15.Patton HD, Amassian VE. Single- and multiple-unit analysis of cortical stage of pyramidal tract activation. J. Neurophysiol. 1954;17:345–363. doi: 10.1152/jn.1954.17.4.345. [DOI] [PubMed] [Google Scholar]
- 16.Hamada M, Murase N, Hasan A, Balaratnam M, Rothwell JC. The role of interneuron networks in driving human motor cortical plasticity. Cereb. Cortex. 2012;23:1593–1605. doi: 10.1093/cercor/bhs147. [DOI] [PubMed] [Google Scholar]
- 17.Di Lazzaro V, et al. Comparison of descending volleys evoked by transcranial magnetic and electric stimulation in conscious humans. Electroencephalogr. Clin. Neurophysiol./Electromyogr. Motor Control. 1998;109:397–401. doi: 10.1016/S0924-980X(98)00038-1. [DOI] [PubMed] [Google Scholar]
- 18.Di Lazzaro V, et al. Magnetic transcranial stimulation at intensities below active motor threshold activates intracortical inhibitory circuits. Exp. Brain Res. 1998;119:265–268. doi: 10.1007/s002210050341. [DOI] [PubMed] [Google Scholar]
- 19.Nakamura H, Kitagawa H, Kawaguchi Y, Tsuji H. Intracortical facilitation and inhibition after transcranial magnetic stimulation in conscious humans. J. Physiol. 1997;498:817–823. doi: 10.1113/jphysiol.1997.sp021905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kujirai T, et al. Corticocortical inhibition in human motor cortex. J. Physiol. 1993;471:501–519. doi: 10.1113/jphysiol.1993.sp019912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hanajima R, et al. Paired-pulse magnetic stimulation of the human motor cortex: differences among I waves. J. Physiol. 1998;509:607–618. doi: 10.1111/j.1469-7793.1998.607bn.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fisher R, Nakamura Y, Bestmann S, Rothwell J, Bostock H. Two phases of intracortical inhibition revealed by transcranial magnetic threshold tracking. Exp. Brain Res. 2002;143:240–248. doi: 10.1007/s00221-001-0988-2. [DOI] [PubMed] [Google Scholar]
- 23.Day B, et al. Motor cortex stimulation in intact man: 2. Multiple descending volleys. Brain. 1987;110:1191–1209. doi: 10.1093/brain/110.5.1191. [DOI] [PubMed] [Google Scholar]
- 24.Rossi S, Hallett M, Rossini PM, Pascual-Leone A. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin. Neurophysiol. 2009;120:2008–2039. doi: 10.1016/j.clinph.2009.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rossi, S. et al. Safety and recommendations for TMS use in healthy subjects and patient populations, with updates on training, ethical and regulatory issues: Expert guidelines. (2020). [DOI] [PMC free article] [PubMed]
- 26.Groppa S, et al. A practical guide to diagnostic transcranial magnetic stimulation: Report of an IFCN committee. Clin. Neurophysiol. 2012;123:858–882. doi: 10.1016/j.clinph.2012.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Do M, et al. Magstim 2002 and Bistim Mode maximum stimulus output values are not equivalent: Configuration selection is critical. Brain Stimulat. Basic Transl. Clin. Res. Neuromodulat. 2020;13:444–446. doi: 10.1016/j.brs.2019.12.009. [DOI] [PubMed] [Google Scholar]
- 28.Awiszus, F. & Borckardt, J. TMS motor threshold assessment tool (MTAT 2.0). Brain Stimulation Laboratory, Medical University of South Carolina, USA (2011).
- 29.Rossini PM, et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: Basic principles and procedures for routine clinical and research application. An updated report from an IFCN Committee. Clin. Neurophysiol. 2015;126:1071–1107. doi: 10.1016/j.clinph.2015.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Werhahn K, et al. The effect of magnetic coil orientation on the latency of surface EMG and single motor unit responses in the first dorsal interosseous muscle. Electroencephalogr. Clin. Neurophysiol./Evoked Potentials Sect. 1994;93:138–146. doi: 10.1016/0168-5597(94)90077-9. [DOI] [PubMed] [Google Scholar]
- 31.Brown KE, et al. The reliability of commonly used electrophysiology measures. Brain Stimul. 2017;10:1102–1111. doi: 10.1016/j.brs.2017.07.011. [DOI] [PubMed] [Google Scholar]
- 32.Ziemann U, Rothwell JC, Ridding MC. Interaction between intracortical inhibition and facilitation in human motor cortex. J. Physiol. 1996;496:873–881. doi: 10.1113/jphysiol.1996.sp021734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Roshan L, Paradiso GO, Chen R. Two phases of short-interval intracortical inhibition. Exp. Brain Res. 2003;151:330–337. doi: 10.1007/s00221-003-1502-9. [DOI] [PubMed] [Google Scholar]
- 34.Di Lazzaro V, et al. The physiological basis of transcranial motor cortex stimulation in conscious humans. Clin. Neurophysiol. 2004;115:255–266. doi: 10.1016/j.clinph.2003.10.009. [DOI] [PubMed] [Google Scholar]
- 35.Ridding M, Rothwell J, Inzelberg R. Changes in excitability of motor cortical circuitry in patients with Parkinson's disease. Ann. Neurol. 1995;37:181–188. doi: 10.1002/ana.410370208. [DOI] [PubMed] [Google Scholar]
- 36.Biabani M, Farrell M, Zoghi M, Egan G, Jaberzadeh S. The minimal number of TMS trials required for the reliable assessment of corticospinal excitability, short interval intracortical inhibition, and intracortical facilitation. Neurosci. Lett. 2018;674:94–100. doi: 10.1016/j.neulet.2018.03.026. [DOI] [PubMed] [Google Scholar]
- 37.Bastani A, Jaberzadeh S. A higher number of TMS-elicited MEP from a combined hotspot improves intra-and inter-session reliability of the upper limb muscles in healthy individuals. PLoS ONE. 2012;7:e47582. doi: 10.1371/journal.pone.0047582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lakens D. Calculating and reporting effect sizes to facilitate cumulative science: A practical primer for t-tests and ANOVAs. Front. Psychol. 2013;4:863. doi: 10.3389/fpsyg.2013.00863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Silbert BI, et al. Evidence for an excitatory GABAA response in human motor cortex in idiopathic generalised epilepsy. Seizure. 2015;26:36–42. doi: 10.1016/j.seizure.2015.01.014. [DOI] [PubMed] [Google Scholar]
- 40.Zoghi M, Pearce SL, Nordstrom MA. Differential modulation of intracortical inhibition in human motor cortex during selective activation of an intrinsic hand muscle. J. Physiol. 2003;550:933–946. doi: 10.1113/jphysiol.2003.042606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Volz, L. et al. Modulation of I‐wave generating pathways by TBS: A model of plasticity induction. J. Physiol. (2019). [DOI] [PubMed]
- 42.Hamada M, et al. Two distinct interneuron circuits in human motor cortex are linked to different subsets of physiological and behavioral plasticity. J. Neurosci. 2014;34:12837–12849. doi: 10.1523/JNEUROSCI.1960-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hordacre B, et al. Variability in neural excitability and plasticity induction in the human cortex: A brain stimulation study. Brain Stimulat. Basic Transl. Clin. Res. Neuromodulat. 2017;10:588–595. doi: 10.1016/j.brs.2016.12.001. [DOI] [PubMed] [Google Scholar]
- 44.Dam JMV, Goldsworthy MR, Hague WM, Coat S, Pitcher JB. Cortical plasticity and interneuron recruitment in adolescents born to women with gestational diabetes mellitus. Brain Sci. 2021;11:388. doi: 10.3390/brainsci11030388. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.