Abstract
Cholesterol is an essential component of plasma membrane and precursor of biological active compounds, including hydroxycholesterols (HCs). HCs regulate cellular homeostasis of cholesterol; they can pass across the membrane and vascular barriers and act distantly as para- and endocrine agents. A small amount of 25-hydroxycholesterol (25-HC) is produced in the endoplasmic reticulum of most cells, where it serves as a potent regulator of the synthesis, intracellular transport, and storage of cholesterol. Production of 25-HC is strongly increased in the macrophages, dendrite cells, and microglia at the inflammatory response. The synthesis of 25-HC can be also upregulated in some neurological disorders, such as Alzheimer’s disease, amyotrophic lateral sclerosis, spastic paraplegia type 5, and X-linked adrenoleukodystrophy. However, it is unclear whether 25-HC aggravates these pathologies or has the protective properties. The molecular targets for 25-HC are transcriptional factors (LX receptors, SREBP2, ROR), G protein-coupled receptor (GPR183), ion channels (NMDA receptors, SLO1), adhesive molecules (α5β1 and ανβ3 integrins), and oxysterol-binding proteins. The diversity of 25-HC-binding proteins points to the ability of HC to affect many physiological and pathological processes. In this review, we focused on the regulation of 25-HC production and its universal role in the control of cellular cholesterol homeostasis, as well as the effects of 25-HC as a signaling molecule mediating the influence of inflammation on the processes in the neuromuscular system and brain. Based on the evidence collected, it can be suggested that 25-HC prevents accumulation of cellular cholesterol and serves as a potent modulator of neuroinflammation, synaptic transmission, and myelinization. An increased production of 25-HC in response to a various type of damage can have a protective role and reduce neuronal loss. At the same time, an excess of 25-HC may exert the neurotoxic effects.
Keywords: cholesterol, 25-hydroxycholesterol, liver X receptors, NMDA receptors, synaptic transmission, neuroinflammation, microglia
INTRODUCTION
Cholesterol is a main structural component of the plasma membrane that controls its fluidity and thickness. In the membrane, cholesterol organizes microdomains – lipid rafts – containing numerous receptors, ion channels, and signaling enzymes. Cholesterol is a precursor of steroid hormones, bile acids, and neurosteroids, as well as its numerous oxidized forms (oxysterols), some of which perform important signaling functions and control cholesterol synthesis, in particular, in the brain and retina – organs “disconnected” from the systemic cholesterol homeostasis [1].
Cholesterol can be oxidized to various oxysterol species whose concentrations vary in a wide range in different tissues. One of the oxysterols, 25-hydroxycholesterol (25-HC), is of particular interest. Firstly, it is produced by a specific enzyme of the endoplasmic reticulum (ER), cholesterol-25-hydroxylase (CH25H) [2]. Expression of the CH25H gene is low under physiological conditions and increases tenfold during inflammatory response [3], which, among other things, accompanies neurodegenerative diseases [4, 5]. Secondly, the main producers of 25-HC are macrophages (especially, alveolar macrophages [6]), dendritic cells, and microglia [3, 4]. Thirdly, the targets of 25-HC include G protein-coupled receptors, transcription factors, adhesive molecules, and ion channels [3, 7-10]. As a hydroxycholesterol (HC), with a hydroxyl group in the side chain, 25-HC, can freely penetrate through plasma membranes and vascular barriers [11] and, therefore, act remotely as a para- and/or endocrine factor [12].
REGULATION OF 25-HYDROXYCHOLESTEROL PRODUCTION
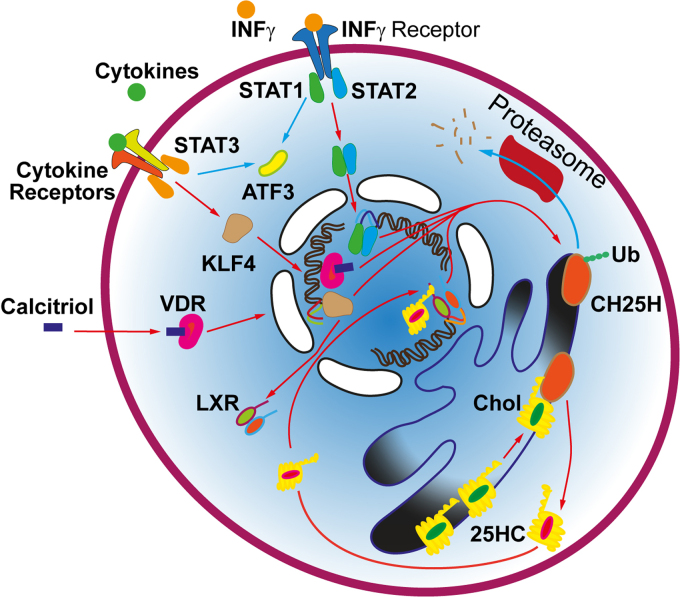
The synthesis of 25-HC is determined, to a large extent, by the content of CH25H in the cells (Fig. 1). Numerous studies have shown that pro-inflammatory cytokines significantly upregulate expression of the CH25H gene [3], which belongs to the family of interferon (IFN)-stimulated genes. Activation of IFN receptors leads to the assembly of a complex of the transcription factors STAT1 and STAT2 that promotes the expression of CH25H in the macrophages ~by 30 times and in dendritic cells by ~10 times [13]. Interestingly, 25-HC suppresses the translocation of IFNγ receptors to the lipid rafts in the microglia and, hence, blunts the IFNγ-induced pro-inflammatory activation of the microglia [14]. Therefore, IFN stimulates the synthesis of 25-HC, which, in turn, suppresses the signaling through the IFN receptors, i.e., there is a negative feedback that limits the action of IFN. Noteworthy, an increased expression of genes associated with cholesterol metabolism (including CH25H) is typical for the Trem2+ microglia, which has protective properties in neurodegenerative and demyelinating diseases [15].
Fig. 1.
Regulation of CH25H expression. IFNγ (or INFγ) and a number of cytokines upregulate CH25H expression via stimulation of the STAT and KLF4 transcription factors [13, 16] or suppress it through the activation of ATF3 [17]. CH25H expression is activated by the stimulation of vitamin D receptors (VDRs) and liver X receptors (LXRs) [18, 19]. 25-HC is a potent ligand of the latter receptors), which creates a positive feedback between the expression of CH25H and production of 25-HC. The synthesis of 25-HC increases with increasing cholesterol levels in the ER. On the contrary, CH25H ubiquitination directs it to proteasomal degradation, resulting in the decrease in the 25-HC synthesis [20]. Blue and red arrows show negative and positive regulation of 25-HC levels, respectively.
The plasma levels of 25-HC increase during viral and bacterial infections. By stimulating the Toll-like receptor 4 (TLR4), bacterial wall lipopolysaccharide (LPS) increases expression of CH25H in the macrophages [21] and microglia [4] by ~35 and 65 times, respectively. Similarly, 8-12 h after injection of the selective TLR4 agonist, the content of CH25H mRNA increased in the liver (by ~250 times), heart (by ~50 times), brain (by ~30 times), lungs (by ~20 times), muscles, kidneys, thymus (by 8-12 times), skin, and intestine (by 3-5 times), thus indicating a response of resident macrophages in many tissues [22]. CH25H expression is enhanced by the transcription factor KLF4, which regulates inflammation and apoptosis and is activated upon stimulation of cytokine receptors [16]. LPS injection increases the level of KLF4 in microglial cells, where KLF4 interacts with the transcription factor NF-κB, one of the key regulators of pro-inflammatory genes [23]. KLF4 promotes a change in the phenotype of microglia and macrophages from the pro-inflammatory M1 type to the anti-inflammatory M2 type [24, 25]. KLF4 also upregulates expression of liver X receptors (LXRs) in the macrophages [16]. The latter are important regulators of the inflammatory response and metabolism of lipids, including cholesterol. Moreover, 25-HC is a ligand of LXRs: by activating them, 25-HC promotes CH25H production in the macrophages and, consequently, activates the synthesis of 25-HC [18], thus creating a positive feedback aimed at the explosive increase in the 25-HC levels. Moreover, stimulation of LXRs by 25-HC is important for the transition of the macrophages to the M2 state [26].
Upregulated CH25H activity is observed in a number of diseases accompanied by chronic inflammation, including neuroinflammation. For example, an increase in the CH25H expression in the microglial cells has been found in the models of Alzheimer’s disease [27], X-linked adrenoleukodystrophy [28], and adult leukoencephalopathy with axonal spheroids and pigmented glia [29]. The concentrations of 25-HC can significantly increase in amyotrophic lateral sclerosis [5, 30] and spastic paraplegia type 5 [31] – pathologies that affect motor neurons.
There is also a negative regulation of CH25H content during inflammatory reactions. The expression of CH25H is suppressed by the transcription factor ATF3 [17] that is activated by a number of cytokines, including IFNs. The activation of ATF3 in the microglia occurs upon the nerve injury and is important for the nerve regeneration [32]. Ubiquitination of CH25H directs this enzyme to proteasomal degradation. Porcine reproductive and respiratory syndrome virus inhibits the synthesis of 25-HC (which has the antiviral properties) via promoting CH25H ubiquitination and subsequent proteolysis [20]. The levels of 25-HC are reduced in the blood plasma of patients with COVID-19, which is accompanied by a cytokine storm; while delivery of 25-HC in nanovesicles alleviates the cytokine storm [33].
Diet affects the content of CH25H and production of 25-HC. The synthesis of 25-HC increases rapidly after consumption of cholesterol-rich food [34]. Both CH25H expression and 25-HC production increase in the adipose tissue macrophages with the increase in the body mass index and fat intake [35]. Consumption of excess of arachidonic acid may elevate CH25H levels in the liver and brain [36]. It has been shown that the blood plasma concentration of 25-HC in patients with the vitamin D deficiency increases significantly after diet supplementation with vitamin D (50,000 IU per week for eight weeks) in combination with phototherapy [37]. This is probably due to the fact that vitamin D increases CH25H expression by acting on the nuclear receptors. Furthermore, the anti-proliferative effect of vitamin D depends on CH25H and is mediated by 25-HC [19]. Activation of vitamin D receptors suppresses both neuroinflammation and disruption of the blood-brain barrier, as well promotes myelination [38]. Besides, microglial vitamin D receptors contribute to the cell polarization towards the M2 phenotype [39]. It can be assumed that 25-HC plays a role in these neuroprotective effects of vitamin D.
It is worth noting that although CH25H is a key enzyme in the generation of 25-HC, some cytochromes (CYPs) can also produce 25-HC as a by-product of their activity. In particular, mitochondrial CYP27A1 and microsomal CYP46A1 and CYP3A can synthesize 25-HC [40].
HOMEOSTATIC ROLE OF 25-HYDROXYCHOLESTEROL AS AN INTRACELLULAR REGULATOR
In recent years, a number of detailed reviews have been published on the cellular cholesterol homeostasis [41, 42]. In this section, we have focused on the importance of endogenous production of 25-HC in the control of cellular cholesterol. Evidently, many oxysterols can influence cholesterol homeostasis; however, 25-HC has the most pronounced effect, even if the cells express CH25H at low levels under physiological conditions [2]. This is due to the properties of 25-HC and location of CH25H in the ER, the key region for cholesterol synthesis (about 20 enzymes of cholesterol biosynthetic pathway localize to the ER).
The main components of the mechanism controlling cholesterol homeostasis reside in the ER, and a constant “outflow” of cholesterol (due to the transport to other membranes and deposition in the ester form within lipid drops) ensures its low content (3-6%) in the ER membranes compared to other membranes [43].
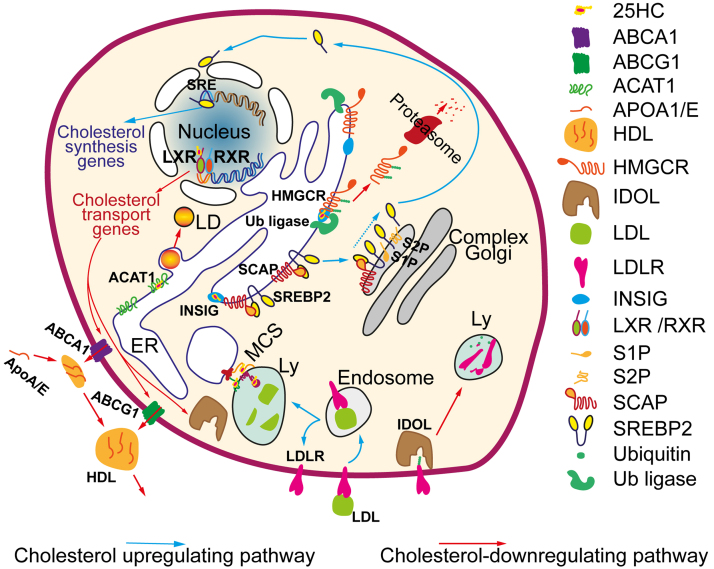
An increase in the cholesterol level in the ER (due to excessive synthesis and/or decrease in its efflux) leads to cholesterol 25-hydroxylation. Consequently, accumulation of 25-HC reflects an excess of cholesterol in cells, which has detrimental consequences, in particular, for neurons and microglia [1, 15]. Therefore, it is not surprising that 25-HC triggers the mechanisms directed to lowering cellular cholesterol levels (Fig. 2). First, 25-HC stimulates the binding of INSIG1/2 protein to 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGCR), the rate-limiting enzyme of cholesterol synthesis. As a result, HMGCR is ubiquitinated and transported from the ER to proteasomes for degradation [44]. By binding to INSIG, 25-HC promotes the retention of the SCAP-SREBP2-INSIG complex in the ER, thereby preventing an activation of the transcription factor SREBP2 responsible for the enhanced expression of cholesterol biosynthesis enzymes [45, 46]. Thus, an increase in the 25-HC level in the ER reduces the content of enzymes involved in cholesterol synthesis due to both their increased degradation and reduced expression. The submicromolar concentrations of 25-HC prevent SREBP2 activation in the neuronal culture by inhibiting expression of cholesterol synthesis enzymes [47]. The 25-HC-mediated suppression of the processing of the transcription factor SREBP inhibits expression of pro-inflammatory cytokines [48].
Fig. 2.
25-HC-dependent regulation of cellular cholesterol homeostasis. Excessive cholesterol is converted to 25-HC in the ER, where 25-HC binds to INSIG. (i) INSIG-25-HC promotes the retention of the SCAP-SREBP2 complex in the ER. In the absence of 25-HC, the SCAP-SREBP2 complex relocates to the Golgi apparatus, where SREBP2 undergoes proteolytic cleavage by the S1P and S2P proteases, releasing the active transcription factor SREBP2. SREBP2 binds to the regulatory regions (sterol regulatory elements, or SREs) of cholesterol biosynthesis genes and activates their expression [45, 46]. (ii) In the presence of 25-HC, INSIG binds to ubiquitin ligases, which promote ubiquitination of HMGCR, the rate-limiting enzyme of cholesterol synthesis. The ubiquitinated enzyme is then directed to proteasomal degradation [44]. 25-HC stimulates LXRs [3, 42], which activate expression of genes of ABC transporters (ABCA1, ABCG1) and ubiquitin ligase IDOL [49, 50]. (iii) ABC transporters efflux cellular cholesterol to the APO-A/E proteins, thus mediating formation and maturation of lipoprotein particles [42]. (iv) IDOL ubiquitinates LDL receptors and promotes their translocation to the lysosomes for proteolysis; as a result, the LDL receptor-mediated uptake of extracellular cholesterol decreases [49]. (v) 25-HC interacts with the proteins that organize membrane contact sites (MCSs), thus reducing the transport of cholesterol between organelles [41, 51, 52]. (vi) 25-HC directly activates ACAT1-dependent esterification of cholesterol in the ER, facilitating the deposition of excessive cholesterol in the lipid droplets (LDs) [47, 53]. Blue and red arrows show positive and negative regulation of cholesterol levels, respectively.
Another mechanism for reducing the level of cellular cholesterol by 25-HC is determined by its ability to activate nuclear LXRs which control the genes of lipid transport [3, 42]. Stimulation of LXRs increases the expression of ABC transporters (ABCA1, ABCG1, ABCG5, and ABCG8) responsible for the reverse transport of cholesterol from the cells to the extracellular acceptors (e.g., APOA-I or APOE). LXRs also upregulate ubiquitin ligase IDOL, which ubiquitinates the low-density lipoprotein (LDL) receptors, contributing to their degradation [49, 50]. As a result, the uptake of extracellular cholesterol via the receptor-mediated endocytosis of lipoprotein particles is reduced. Therefore, an increase in the 25-HC levels in the cytoplasm and nucleus (i.e., outside of the sites of its synthesis) through the stimulation of LXRs promotes the release of cholesterol from the cells and inhibits the uptake of extracellular cholesterol. Indeed, 25-HC at submicromolar concentrations increased ABCA1 expression through the activation of LXRs in neurons and oligodendrocytes [47, 54]. Interestingly, in hypothalamic neurons, IDOL controls the levels of very-low-density lipoprotein (VLDL) receptor, thus influencing food intake and thermogenesis [55]. This points to a possible role of 25-HC in the neuronal control of metabolic processes.
25-HC affects the intracellular transport of cholesterol and its deposition in the ester form. 25-HC directly enhances the activity of acyl-coenzyme A:cholesterol acyltransferase 1 (ACAT1) and, therefore, accelerates cholesterol esterification in the ER [53]. For example, 25-HC (2.5 and 5 µM) increased the storage of cholesterol esters in cultured neurons ~by 11 and 21%, respectively [47]. The delivery of cholesterol from lysosomes to other organelles (ER, peroxisomes, trans-Golgi apparatus, and mitochondria) occurs to a large extent via a non-vesicular pathway through the membrane contact sites (MCSs) [41]. Some of the main components of MCSs are oxysterol-binding proteins (OSBP, ORP1L, ORP2, ORP5, ORP6) and START [steroidogenic acute regulatory protein (StAR)-related lipid transfer]-domain containing proteins. 25-HC competes with cholesterol for binding to these proteins, thereby interfering with the delivery of cholesterol from the lysosomes [51, 52].
Normally, 25-HC at low concentrations is produced in all cells, where it prevents an appearance of excessive cholesterol. However, during inflammatory response, 25-HC from the macrophages and microglia can enter neighboring cells and disrupt cholesterol homeostasis in the latter, preventing cell growth and proliferation and inducing apoptosis. This mechanism partially underlies the antiviral, anticancer, and senolytic effects of 25-HC. It should be noted that neurons, the functioning of which in many aspects closely depends on cholesterol, generate 24-HC in the ER along with 25-HC due to the activity of the neuron-specific enzyme CYP46A1 [1, 56, 57]. Such parallel participation of two ER enzymes, CH25H and CYP46A1, in the conversion of cholesterol to HCs is probably necessary to prevent cholesterol accumulation in the neurons. This is especially essential for neurons, which, in addition to the in situ synthesis, receive most of cholesterol from astrocytes as a component of lipoprotein particles [1].
25-HYDROXYCHOLESTEROL AS A MOLECULE OF INTERCELLULAR INTERACTIONS IN THE NERVOUS SYSTEM
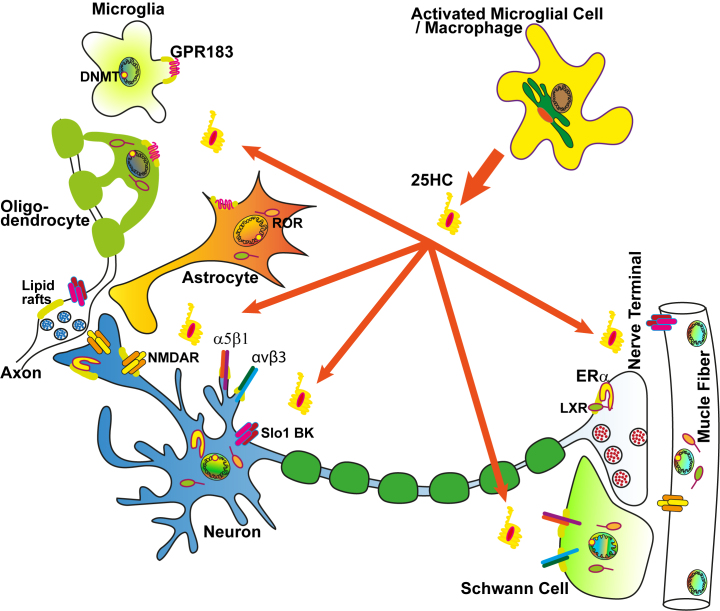
The central nervous system is “fenced off” by the blood-brain barrier from the systemic circulation. This barrier is impermeable for many molecules, including cholesterol. However, cholesterol derivatives with hydroxyl groups in the side chain can penetrate the vascular barriers [1, 57]. Therefore, 25-HC synthesized at the periphery (e.g., during systemic inflammation) can enter the brain. On the other hand, 25-HC, produced in the CNS in situ, is released into the bloodstream. The main producer of 25-HC in the brain is activated microglia [4, 58]. The peripheral sections of the nervous system are directly affected by 25-HC when its content increases both locally in tissues and systemically in response to a strong inflammatory response or consumption of cholesterol-enriched foods. Moreover, 25-HC-producing macrophages come into close contact with damaged peripheral nerves, and this interaction promotes regeneration [59]. Therefore, 25-HC may act as a para- and endocrine molecule that sends signals about inflammation/damage/cholesterol excess (Fig. 3).
Fig. 3.
25-HC targets in the nervous system. The main producers of 25-HC are activated macrophages and microglial cells [3, 4, 58]. Released 25-HC can affect glial cells (microglia, oligodendrocytes, astrocytes, and Schwann cells) and neurons. 25-HC influences synaptic transmission by acting on presynaptic and postsynaptic receptors (see the text for detailed explanation). Designations: DNMT, DNA methyltransferases; ERα, estrogen receptor α; Slo1 BK, large-conductance calcium-activated potassium channels.
EBI2 and CXCR2 receptors. It has been shown that a number of transmembrane proteins act as high-affinity receptors for 25-HC. Migration of immune and glial cells along the gradient of 25-HC and its derivative 7α,25-diHC occurs with the participation of GPR183 (EBI2), a receptor coupled to Gαi protein. The expression of GPR183 in glial cells can increase under pathological conditions, and the activation of GPR183 has protective effects. For instance, expression of GPR183 in the microglia increases in the focus of epileptic activity in the brain [60]. The stimulation of GPR183 in oligodendrocytes is involved in remyelination in a model of multiple sclerosis [61], and GRP183 activation in astrocytes inhibits the release of pro-inflammatory cytokines by these glial cells [62].
Oxysterols can activate another Gi protein-coupled receptor, CXCR2, which is important for cell migration to the site of inflammation. In the brain, CXCR2 expression in the microglia, oligodendrocytes, and neurons is upregulated upon injury, and the activation of CXCR2 can negatively control myelination, as well as disrupt excitatory neurotransmission [63, 64]. However, 25-HC (unlike 22R-HC) is a weak agonist of CXCR2 even at high concentrations [65].
Integrins. The migration and status of immune cells can be regulated by direct binding of 25-HC to the adhesion molecules, α5β1 and αvβ3 integrins. This activates focal adhesion kinase (FAK) and promotes production of cytokines, in particular tumor necrosis factor (TNF) and interleukin 6, by the macrophages [8]. It should be noted that activation of α5β1 and αvβ3 integrins accelerates the growth of neuronal processes [66, 67], as well as proliferation and migration of Schwann cells that envelop peripheral nerves and neuromuscular junctions [68, 69]. The 25-HC/integrin pathway may be involved in the positive effect of macrophages on the repair of damaged neuromuscular junctions [59, 70]. However, it should be kept in mind that excessive production of 25-HC can reduce the myelination of peripheral nerves by suppressing the synthesis of cholesterol in Schwann cells [71] and downregulating expression of myelin genes [72].
NMDA receptors. Another target of 25-HC is glutamate NMDA receptors (NMDARs), whose activation in the CNS is the key event in synaptic plasticity and memory. These receptors also modulate neurotransmission in the periphery [73]. 25-HC directly, although weakly, enhances the glutamate-induced current through NMDARs, but completely and noncompetitively blocks the powerful potentiating effect of 24(S)-HC on the Ca2+ currents via NMDARs [7] mainly composed of GluN2B subunits [74]. As a result, 25-HC is able to attenuate the 24(S)-HC-induced increase in the nerve cell loss due to the NMDAR hyperactivation, i.e., excitotoxicity. In addition, 25-HC significantly reduces neuronal death caused by the deprivation of glucose and oxygen [75].
25-HC at micromolar concentrations, as well as its excessive production by the microglia during neuroinflammation, disrupt the NMDAR-dependent long-term potentiation in the hippocampus [58]. However, this impairment of synaptic plasticity may be reflection of the general protective function of 25-HC secreted by the microglia. Indeed, 25-HC significantly reduced apoptotic loss of neurons caused by the overexpression of the pro-inflammatory regulator STING (stimulator of interferon genes) and autophagy following cerebral artery occlusion [76]. 25-HC rapidly suppresses translocation of the IFNγ receptors into lipid rafts and subsequent activation of these receptors; as a result, 25-HC causes a decrease in the IFNγ-mediated production of pro-inflammatory cytokines by the microglia [14].
Thus, although the production of 25-HC is stimulated under neuroinflammation conditions, it can limit the inflammatory reactions by acting as a key element of the negative feedback loop [48]. At the same time, it can prevent excessive synaptic activity in glutamatergic synapses and disrupt NMDAR-dependent synaptic plasticity. Undoubtedly, extremely high concentrations of 25-HC, which are frequently used in cell studies [77, 78], or probably present in the cases of chronic brain pathologies, can also be detrimental. Brain expression of CH25H is elevated in Alzheimer’s disease patients, and 25-HC promotes interleukin-1β-induced neuroinflammation in the APPPS1-21 mouse model [4]. In the ARPE-19 cell culture, amyloid β increased the levels of 25-HC, which activated P2X7 receptors, promoting apoptosis [79]. Similarly, overproduction of 25-HC may contribute to the neuroinflammation (through the activation of NLRP3 inflammasome and increased production of interleukin-1β) in the X-linked adrenoleukodystrophy, a progressive neurodegenerative disease associated with the accumulation of long-chain fatty acids [28].
BK (slo1) channels. 25-HC also affects the activity of potassium ion channels. At micromolar concentrations, 25-HC reduces the currents via large-conductance calcium-activated potassium channels (slo1 BK). However, this effect is not specific to 25-HC, since other oxysterols with hydroxylated side chain exhibit the same effect [80]. Therefore, oxysterols can potentially enhance calcium-dependent processes by limiting the activity of BK channels. In addition, 25-HC can increase the excitability of neurons by weakening the inhibitory effect of GABA-B receptors. However, this effect was observed upon prolonged action of 25-HC (for hours) on brain sections and only in some neurons [81].
Liver X receptors. 25-HC is a strong natural activator of LXRs [3] present in the plasma membranes (lipid rafts) of some cells [82, 83]. Recently, these receptors were found in the nerve terminals of motor neuron axons, where their distribution was depended on the integrity of lipid rafts [10]. The activation of synaptic LXRs by 25-HC increased the mobilization of synaptic vesicles stimulated by the release of Ca2+ from the ER. It was found that membrane LXRs activate phospholipase C (through the activation of Gi protein and its βγ-dimer) resulting in the elevation in cytosolic Ca2+ dependent on the inositol triphosphate receptors of the ER. Subsequent activation of Ca2+- and lipid-dependent protein kinase C promoted involvement of synaptic vesicles in exocytosis. In addition, 25-HC causes Ca2+-dependent increase in the intra- and extracellular levels of reactive oxygen species in the neuromuscular junctions. However, reactive oxygen species did not have a lipid-damaging effect, but contributed to the facilitation of neurotransmission [10]. It is interesting to note that despite the structural similarity to 25-HC, 24(S)-HC at a short-term application modulated neuromuscular transmission via an NMDAR-dependent but LXR-independent pathway [73, 84].
Interestingly, the absence of LXRβ leads to a disease resembling amyotrophic lateral sclerosis (ALS) with a loss of motor neurons and denervation of skeletal muscles [85]. Deletion of the LXR α and β genes causes locomotion defects accompanied by oxidative stress and protein carbonylation in the motor nerves [86]. This also disrupts the functioning of Schwann cells and makes myelin thinner in peripheral nerves [72]. On the contrary, stimulation of LXRs by 25-HC promotes expression of myelin genes (e.g., PLP, MBP) in oligodendrocytes [54]. Interestingly, the plasma levels of 25-HC were elevated in patients with ALS (during the 1st year of the disease manifestation), while SOD1G93A mice showed an increase in the CH25H expression in the brain at the presymptomatic stage followed by the later increase in the 25-HC content in the spinal cord [5, 30]. However, at the late stages, CH25H expression decreased in the brain of SOD1G93A mice and the concentration of 7α, 25-diHC (25-HC metabolite) reduced in the cerebrospinal fluid of ALS patients [5, 87].
Overall, the significance of increased 25-HC production in ALS is unclear. On one hand, this may be a reflection of inflammatory response and exacerbate the pathology; on the other hand, an increase in the 25-HC content may be a compensatory response aimed at slowing down both the loss of motor neurons and denervation in skeletal muscles. High concentrations of 25-HC (≥5-30 µM) reduce survival and induce apoptosis of cultured motor neurons [5, 30], but lower submicromolar (≤1 µM) concentrations, which are observed in the inflammatory response in vivo, have the opposite effects [30]. Acute exposure to 1 µM 25-HC is able to partially restore the impairments of neuromuscular junction membranes (disruption of lipid rafts, increased membrane fluidity) in SOD1G93A mice. At the same time, chronic administration of low doses of 25-HC (0.4 mg/kg every 4 days for a month) to the mice with ALS prevented fragmentation of neuromuscular junctions, lipid peroxidation of their membranes, accumulation of ceramide, and increase in the non-quantum secretion of acetylcholine in the diaphragm (main respiratory muscle) [88]. This indicates the therapeutic effect of 25-HC, since fragmentation of neuromuscular junctions, excessive release of acetylcholine, and ceramide build-up occur during aging, myasthenia gravis, and can also accelerate denervation and, consequently, atrophic alternations in skeletal muscles [89-93]. Interestingly, hypercholesterolemia is a protective factor in ALS, while treatment with statins, the drugs which suppress cholesterol synthesis, aggravates this pathology [94, 95]. This correlates with the levels of 25-HC in the plasma, as the content of 25-HC increases in hypercholesterolemia and decreases due to statin therapy [34, 96]. It is possible that 25-HC partially mediates the effects of hypercholesterolemia and statins on the development of ALS, and neuromuscular junctions are an important target for the action of 25-HC. Indeed, the labeled analog of 25-HC demonstrated a high affinity for the neuromuscular junction membranes, especially in the ALS model animals [88]. Macrophages, main producers of 25-HC, directly contact the neuromuscular junctions in the case of nerve damage or pathologies accompanied by denervation; and these interactions facilitate reinnervation [59, 70]. Macrophages also contribute to the maintenance of sympathetic (adrenergic) innervation of peripheral tissues [97], and β-adrenergic transmission can be modulated by HC [12].
Recently, it has been shown that 25-HC has the senolytic properties in skeletal muscle, preventing the appearance of signs of aging (e.g., CRYAB aggregation) and inflammation [98]. Increased production of 25-HC (through the activation of LXRs) may improve the conditions in a model of non-alcoholic fatty liver disease [99], which is accompanied by neuromuscular disorders and neurodegeneration. In addition, HCs activate nuclear LXRs in the postsynaptic regions of muscle fibers, leading to the upregulation of both expression of NO synthases (endothelial and neuronal isoforms) and NO synthesis during neuromuscular transmission [100]. In turn, NO can prevent muscle atrophy in response to muscle unloading [101, 102], which is accompanied by early synaptic alterations, including lipid raft disruption, increased membrane fluidity, and ceramide accumulation in the synaptic membranes [91, 92, 103]. Similar events also occur at the early stages of ALS and can be corrected by the administration of 25-HC [88].
Estrogen receptors (EsRs). The membrane-bound LXRs can directly interact with EsRα [82]. Moreover, EsRα is a potential target for 25-HC, and by stimulating it, 25-HC exhibits the estrogen-like activity in a number of cell types [9]. An increased mobilization of synaptic vesicles in the nerve terminals of motor neuron axons under the influence of 25-HC (see “Liver X receptors” section) also depends on EsRα [10]. Interestingly, in the central synapses, EsRα activation (with 17β-estradiol) can rapidly induce the long-term synaptic plasticity due to incorporation of additional glutamate AMPA receptors into the postsynaptic membrane [104]. On the other hand, hypercholesterolemia-induced increase in 27-HC, which can block EsRs, reduces expression of the postsynaptic marker PSD-95 in the hippocampus [105].
Retinoid-related orphan receptor (RORs). In addition to LXRs, 25-HC, along with other sterols, can bind to nuclear RORs, acting as their agonist, neutral antagonist, or even inverse agonist depending on the ROR type [106, 107]. ROR activation is important for the cortex organization, neuronal development, regulation of circadian rhythms, and can also suppress neuroinflammation by inhibiting the activity of transcriptional factor NF-κB and production of pro-inflammatory cytokines by glial cells [108, 109].
Direct effect of 25-HC on membrane properties. Oxysterols interacting with plasma membranes can affect their biophysical properties and formation of lipid microdomains [88, 110-112]. Moreover, structurally similar oxysterols can produce different effects on plasma membranes, including synaptic ones [110]. The studies of the properties of raft-forming lipids in Langmuir monolayers show that 25-HC interacts more strongly than cholesterol with sphingomyelin and slightly less strongly with ganglioside GM1. Therefore, 25-HC can participate in the formation of lipid rafts and even displace cholesterol from them [113]. Indeed, the addition of 25-HC to the artificial bilayer membrane composed of a mixture of cholesterol and 1-palmitoyl-2-oleoylphosphatidylcholine leads to a shift of cholesterol towards the water surface and increase in its accessibility to external acceptors; which reduces the ability of cholesterol to condense the lipid bilayer [114]. Moreover, the ability of 25-HC to increase the availability of membrane cholesterol depends on the lipid composition and is more pronounced in the membranes with a higher content of unsaturated lipids [115]. 25-HC promotes the formation of microdomains enriched in saturated lipids in the artificial membranes [116], as well as increases lipid ordering and decreases fluidity of synaptic membranes in the neuromuscular junctions [88]. On the other hand, 25-HC, having an inclined orientation (with respect to the phospholipid tails in the bilayer), is able to laterally expand and thin out the artificial bilayer membranes [117], especially those consisting of unsaturated lipids [118]. In addition, 25-HC inhibits the coalescence of lipid rafts into larger microdomains in the microglia [14]. The antiviral properties of 25-HC are partly associated with its direct effects on the plasma membranes; as a result, 25-HC suppresses the fusion of viruses, including neurotropic ones, with cells [119]. Considering the importance of membrane properties for many processes in the nervous system [14, 56, 120], the direct effects of 25-HC on the stability of lipid rafts, membrane fluidity and thickness, and cholesterol availability may contribute to the modulation of neuronal activity during inflammation. It should be noted that these effects of 25-HC depend on the membrane lipid composition and, therefore, may differ in neurons and glia, as well as in different cell compartments.
CONCLUSIONS
25-HC is an important regulator of cholesterol homeostasis in individual cells; on the other hand, it mediates the effects of inflammation, acting as a paracrine agent or even systemically. The high penetrating ability of 25-HC, as well as the existence of high-affinity receptors for 25-HC in neurons and glial cells (i) make this oxysterol an important modulator in the nervous system and (ii) make CH25H a potential target for pharmacological interventions. The production of 25-HC in response to injury may play a neuroprotective role by optimizing synaptic transmission and myelination, as well as by limiting neuroinflammation and neuronal loss. At the same time, very high concentrations of 25-HC, often used in cellular studies, can have negative consequences for the functioning of the nervous system. Therefore, additional studies are required to elucidate the direction of 25-HC action in vivo under normal and pathological conditions, in particular, in neurodegenerative diseases.
Acknowledgments
We are grateful to our colleagues G. F. Zakiryanova and prof. E. A. Bukharaeva for discussing this review.
Abbreviations
- CH25H
cholesterol 25-hydroxylase
- ER
endoplasmic reticulum
- EsR
estrogen receptor
- HC
hydroxycholesterol
- HMGCR
3-hydroxy-3-methylglutaryl coenzyme A reductase
- IFN
interferon
- LXR
liver X receptor
Funding
This review was funded by the Russian Foundation for Basic Research (project no. 20-04-00077, “25-Hydroxycholesterol as a molecule of intercellular interactions in the nervous system” section) and partly by the Russian Science Foundation (project no. 22-25-00396; “Regulation of 25-hydroxycholesterol production” and “Homeostatic role of 25-hydroxycholesterol as an intracellular regulator” sections; project no. 21-14-00044, “Direct effect of 25-HC on membrane properties” subsection). The work was partly supported by the government assignment for the Federal Research Center “Kazan Scientific Center of Russian Academy of Sciences”.
Ethics declarations
The authors declare no conflicts of interest. This article does not contain a description of studies involving humans or animals performed by any of the authors.
References
- 1.Petrov A. M., Kasimov M. R., Zefirov A. L. Brain cholesterol metabolism and its defects: Linkage to neurodegenerative diseases and synaptic dysfunction. Acta Naturae. 2016;8:58–73. doi: 10.32607/20758251-2016-8-1-58-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lund E. G., Kerr T. A., Sakai J., Li W. P., Russell D. W. cDNA cloning of mouse and human cholesterol 25-hydroxylases, polytopic membrane proteins that synthesize a potent oxysterol regulator of lipid metabolism. J. Biol. Chem. 1998;273:34316–34327. doi: 10.1074/jbc.273.51.34316. [DOI] [PubMed] [Google Scholar]
- 3.Cyster J. G., Dang E. V., Reboldi A., Yi T. 25-Hydroxycholesterols in innate and adaptive immunity. Nat. Rev. Immunol. 2014;14:731–743. doi: 10.1038/nri3755. [DOI] [PubMed] [Google Scholar]
- 4.Wong M. Y., Lewis M., Doherty J. J., Shi Y., Cashikar A. G., et al. 25-Hydroxycholesterol amplifies microglial IL-1beta production in an apoE isoform-dependent manner. J. Neuroinflamm. 2020;17:192. doi: 10.1186/s12974-020-01869-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kim S. M., Noh M. Y., Kim H., Cheon S. Y., Lee K. M., et al. 25-Hydroxycholesterol is involved in the pathogenesis of amyotrophic lateral sclerosis. Oncotarget. 2017;8:11855–11867. doi: 10.18632/oncotarget.14416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Madenspacher J. H., Morrell E. D., Gowdy K. M., McDonald J. G., Thompson B. M., et al. Cholesterol 25-hydroxylase promotes efferocytosis and resolution of lung inflammation. JCI Insight. 2020;5:e137189. doi: 10.1172/jci.insight.137189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Linsenbardt A. J., Taylor A., Emnett C. M., Doherty J. J., Krishnan K., et al. Different oxysterols have opposing actions at N-methyl-D-aspartate receptors. Neuropharmacology. 2014;85:232–242. doi: 10.1016/j.neuropharm.2014.05.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pokharel S. M., Shil N. K., Gc J. B., Colburn Z. T., Tsai S. Y., et al. Integrin activation by the lipid molecule 25-hydroxycholesterol induces a proinflammatory response. Nat. Commun. 2019;10:1482. doi: 10.1038/s41467-019-09453-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lappano R., Recchia A. G., De Francesco E. M., Angelone T., Cerra M. C., et al. The cholesterol metabolite 25-hydroxycholesterol activates estrogen receptor alpha-mediated signaling in cancer cells and in cardiomyocytes. PLoS One. 2011;6:e16631. doi: 10.1371/journal.pone.0016631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zakyrjanova G. F., Tsentsevitsky A. N., Kuznetsova E. A., Petrov A. M. Immune-related oxysterol modulates neuromuscular transmission via non-genomic liver X receptor-dependent mechanism. Free Radic Biol. Med. 2021;174:121–134. doi: 10.1016/j.freeradbiomed.2021.08.013. [DOI] [PubMed] [Google Scholar]
- 11.Leoni V., Masterman T., Patel P., Meaney S., Diczfalusy U., et al. Side chain oxidized oxysterols in cerebrospinal fluid and the integrity of blood-brain and blood-cerebrospinal fluid barriers. J. Lipid Res. 2003;44:793–799. doi: 10.1194/jlr.M200434-JLR200. [DOI] [PubMed] [Google Scholar]
- 12.Odnoshivkina U. G., Sytchev V. I., Starostin O., Petrov A. M. Brain cholesterol metabolite 24-hydroxycholesterol modulates inotropic responses to beta-adrenoceptor stimulation: The role of NO and phosphodiesterase. Life Sci. 2019;220:117–126. doi: 10.1016/j.lfs.2019.01.054. [DOI] [PubMed] [Google Scholar]
- 13.Liu S. Y., Aliyari R., Chikere K., Li G., Marsden M. D., et al. Interferon-inducible cholesterol-25-hydroxylase broadly inhibits viral entry by production of 25-hydroxycholesterol. Immunity. 2013;38:92–105. doi: 10.1016/j.immuni.2012.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lee J. H., Han J. H., Woo J. H., Jou I. 25-Hydroxycholesterol suppress IFN-gamma-induced inflammation in microglia by disrupting lipid raft formation and caveolin-mediated signaling endosomes. Free Radic Biol. Med. 2022;179:252–265. doi: 10.1016/j.freeradbiomed.2021.11.017. [DOI] [PubMed] [Google Scholar]
- 15.Nugent A. A., Lin K., van Lengerich B., Lianoglou S., Przybyla L., et al. TREM2 regulates microglial cholesterol metabolism upon chronic phagocytic challenge. Neuron. 2020;105:837–854.e9. doi: 10.1016/j.neuron.2019.12.007. [DOI] [PubMed] [Google Scholar]
- 16.Li Z., Martin M., Zhang J., Huang H. Y., Bai L., et al. Kruppel-like factor 4 regulation of cholesterol-25-hydroxylase and liver X receptor mitigates atherosclerosis susceptibility. Circulation. 2017;136:1315–1330. doi: 10.1161/CIRCULATIONAHA.117.027462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gold E. S., Ramsey S. A., Sartain M. J., Selinummi J., Podolsky I., et al. ATF3 protects against atherosclerosis by suppressing 25-hydroxycholesterol-induced lipid body formation. J. Exp. Med. 2012;209:807–817. doi: 10.1084/jem.20111202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liu Y., Wei Z., Ma X., Yang X., Chen Y., et al. 25-Hydroxycholesterol activates the expression of cholesterol 25-hydroxylase in an LXR-dependent mechanism. J. Lipid Res. 2018;59:439–451. doi: 10.1194/jlr.M080440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang J. H., Tuohimaa P. Regulation of cholesterol 25-hydroxylase expression by vitamin D3 metabolites in human prostate stromal cells. Biochem. Biophys. Res. Commun. 2006;345:720–725. doi: 10.1016/j.bbrc.2006.04.156. [DOI] [PubMed] [Google Scholar]
- 20.Ke W., Fang L., Tao R., Li Y., Jing H., et al. Porcine reproductive and respiratory syndrome virus E protein degrades porcine cholesterol 25-hydroxylase via the ubiquitin-proteasome pathway. J. Virol. 2019;93:e00767-19. doi: 10.1128/JVI.00767-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Diczfalusy U., Olofsson K. E., Carlsson A. M., Gong M., Golenbock D. T., et al. Marked upregulation of cholesterol 25-hydroxylase expression by lipopolysaccharide. J. Lipid Res. 2009;50:2258–2264. doi: 10.1194/jlr.M900107-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bauman D. R., Bitmansour A. D., McDonald J. G., Thompson B. M., Liang G., et al. 25-Hydroxycholesterol secreted by macrophages in response to Toll-like receptor activation suppresses immunoglobulin A production. Proc. Natl. Acad. Sci. USA. 2009;106:16764–16769. doi: 10.1073/pnas.0909142106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kaushik D. K., Gupta M., Das S., Basu A. Kruppel-like factor 4, a novel transcription factor regulates microglial activation and subsequent neuroinflammation. J. Neuroinflamm. 2010;7:68. doi: 10.1186/1742-2094-7-68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liao X., Sharma N., Kapadia F., Zhou G., Lu Y., et al. Kruppel-like factor 4 regulates macrophage polarization. J. Clin. Invest. 2011;121:2736–2749. doi: 10.1172/JCI45444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wen M., Ye J., Han Y., Huang L., Yang H., et al. Hypertonic saline regulates microglial M2 polarization via miR-200b/KLF4 in cerebral edema treatment. Biochem. Biophys. Res. Commun. 2018;499:345–353. doi: 10.1016/j.bbrc.2018.03.161. [DOI] [PubMed] [Google Scholar]
- 26.Kimura T., Nada S., Takegahara N., Okuno T., Nojima S., et al. Polarization of M2 macrophages requires Lamtor1 that integrates cytokine and amino-acid signals. Nat. Commun. 2016;7:13130. doi: 10.1038/ncomms13130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ofengeim D., Mazzitelli S., Ito Y., DeWitt J. P., Mifflin L., et al. RIPK1 mediates a disease-associated microglial response in Alzheimer’s disease. Proc. Natl. Acad. Sci. USA. 2017;114:E8788–E8797. doi: 10.1073/pnas.1714175114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jang J., Park S., Jin Hur H., Cho H. J., Hwang I., et al. 25-hydroxycholesterol contributes to cerebral inflammation of X-linked adrenoleukodystrophy through activation of the NLRP3 inflammasome. Nat. Commun. 2016;7:13129. doi: 10.1038/ncomms13129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chitu V., Biundo F., Shlager G. G. L., Park E. S., Wang P., et al. Microglial homeostasis requires balanced CSF-1/CSF-2 receptor signaling. Cell Rep. 2020;30:3004–3019.e3005. doi: 10.1016/j.celrep.2020.02.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dodge J. C., Yu J., Sardi S. P., Shihabuddin L. S. Sterol auto-oxidation adversely affects human motor neuron viability and is a neuropathological feature of amyotrophic lateral sclerosis. Sci. Rep. 2021;11:803. doi: 10.1038/s41598-020-80378-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Marelli C., Lamari F., Rainteau D., Lafourcade A., Banneau G., et al. Plasma oxysterols: Biomarkers for diagnosis and treatment in spastic paraplegia type 5. Brain. 2018;141:72–84. doi: 10.1093/brain/awx297. [DOI] [PubMed] [Google Scholar]
- 32.Tanaka T., Murakami K., Bando Y., Nomura T., Isonishi A., et al. Microglia support ATF3-positive neurons following hypoglossal nerve axotomy. Neurochem. Int. 2017;108:332–342. doi: 10.1016/j.neuint.2017.05.007. [DOI] [PubMed] [Google Scholar]
- 33.Kim H., Lee H. S., Ahn J. H., Hong K. S., Jang J. G., et al. Lung-selective 25-hydroxycholesterol nanotherapeutics as a suppressor of COVID-19-associated cytokine storm. Nano Today. 2021;38:101149. doi: 10.1016/j.nantod.2021.101149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Johnson K. A., Morrow C. J., Knight G. D., Scallen T. J. In vivo formation of 25-hydroxycholesterol from endogenous cholesterol after a single meal, dietary cholesterol challenge. J. Lipid Res. 1994;35:2241–2253. doi: 10.1016/S0022-2275(20)39930-2. [DOI] [PubMed] [Google Scholar]
- 35.Russo L., Muir L., Geletka L., Delproposto J., Baker N., et al. Cholesterol 25-hydroxylase (CH25H) as a promoter of adipose tissue inflammation in obesity and diabetes. Mol. Metab. 2020;39:100983. doi: 10.1016/j.molmet.2020.100983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Xu H., Sun B., Jia L., Wei Y., Liao Z., Liang M. Cloning and characterization of cholesterol 25-hydroxylase (ch25h) from a Marine Teleost, Chinese Tongue Sole (Cynoglossus semilaevis), and its gene expressions in response to dietary arachidonic acid. Front. Marine Sci. 2020;6:800. doi: 10.3389/fmars.2019.00800. [DOI] [Google Scholar]
- 37.Ponda M. P., Liang Y., Kim J., Hutt R., Dowd K., et al. A randomized clinical trial in vitamin D-deficient adults comparing replenishment with oral vitamin D3 with narrow-band UV type B light: Effects on cholesterol and the transcriptional profiles of skin and blood. Am. J. Clin. Nutr. 2017;105:1230–1238. doi: 10.3945/ajcn.116.150367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.De Oliveira L. R. C., Mimura L. A. N., Fraga-Silva T. F. C., Ishikawa L. L. W., Fernandes A. A. H., et al. Calcitriol prevents neuroinflammation and reduces blood-brain barrier disruption and local macrophage/microglia activation. Front. Pharmacol. 2020;11:161. doi: 10.3389/fphar.2020.00161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Cui C., Xu P., Li G., Qiao Y., Han W., et al. Vitamin D receptor activation regulates microglia polarization and oxidative stress in spontaneously hypertensive rats and angiotensin II-exposed microglial cells: Role of renin-angiotensin system. Redox Biol. 2019;26:101295. doi: 10.1016/j.redox.2019.101295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Honda A., Miyazaki T., Ikegami T., Iwamoto J., Maeda T., et al. Cholesterol 25-hydroxylation activity of CYP3A. J. Lipid Res. 2011;52:1509–1516. doi: 10.1194/jlr.M014084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Meng Y., Heybrock S., Neculai D., Saftig P. Cholesterol handling in lysosomes and beyond. Trends Cell Biol. 2020;30:452–466. doi: 10.1016/j.tcb.2020.02.007. [DOI] [PubMed] [Google Scholar]
- 42.Luo J., Yang H., Song B. L. Mechanisms and regulation of cholesterol homeostasis. Nat. Rev. Mol. Cell Biol. 2020;21:225–245. doi: 10.1038/s41580-019-0190-7. [DOI] [PubMed] [Google Scholar]
- 43.Ridsdale A., Denis M., Gougeon P. Y., Ngsee J. K., Presley J. F., et al. Cholesterol is required for efficient endoplasmic reticulum-to-Golgi transport of secretory membrane proteins. Mol. Biol. Cell. 2006;17:1593–1605. doi: 10.1091/mbc.e05-02-0100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sever N., Yang T., Brown M. S., Goldstein J. L., DeBose-Boyd R. A. Accelerated degradation of HMG CoA reductase mediated by binding of insig-1 to its sterol-sensing domain. Mol. Cell. 2003;11:25–33. doi: 10.1016/s1097-2765(02)00822-5. [DOI] [PubMed] [Google Scholar]
- 45.Brown M. S., Goldstein J. L. Cholesterol feedback: from Schoenheimer’s bottle to Scap’s MELADL. J. Lipid Res. 2009;50 Suppl:S15–27. doi: 10.1194/jlr.R800054-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Radhakrishnan A., Ikeda Y., Kwon H. J., Brown M. S., Goldstein J. L. Sterol-regulated transport of SREBPs from endoplasmic reticulum to Golgi: Oxysterols block transport by binding to Insig. Proc. Natl. Acad. Sci. USA. 2007;104:6511–6518. doi: 10.1073/pnas.0700899104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Waltl S., Patankar J. V., Fauler G., Nusshold C., Ullen A., et al. 25-Hydroxycholesterol regulates cholesterol homeostasis in the murine CATH. a neuronal cell line. Neurosci. Lett. 2013;539:16–21. doi: 10.1016/j.neulet.2013.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Reboldi A., Dang E. V., McDonald J. G., Liang G., Russell D. W., et al. Inflammation. 25-Hydroxycholesterol suppresses interleukin-1-driven inflammation downstream of type I interferon. Science. 2014;345:679–684. doi: 10.1126/science.1254790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zelcer N., Hong C., Boyadjian R., Tontonoz P. LXR regulates cholesterol uptake through Idol-dependent ubiquitination of the LDL receptor. Science. 2009;325:100–104. doi: 10.1126/science.1168974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Baldan A., Bojanic D. D., Edwards P. A. The ABCs of sterol transport. J. Lipid Res. 2009;50 Suppl:S80–85. doi: 10.1194/jlr.R800044-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Suchanek M., Hynynen R., Wohlfahrt G., Lehto M., Johansson M., et al. The mammalian oxysterol-binding protein-related proteins (ORPs) bind 25-hydroxycholesterol in an evolutionarily conserved pocket. Biochem. J. 2007;405:473–480. doi: 10.1042/BJ20070176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Alpy F., Tomasetto C. Give lipids a START: The StAR-related lipid transfer (START) domain in mammals. J. Cell Sci. 2005;118:2791–2801. doi: 10.1242/jcs.02485. [DOI] [PubMed] [Google Scholar]
- 53.Abrams M. E., Johnson K. A., Perelman S. S., Zhang L. S., Endapally S., et al. Oxysterols provide innate immunity to bacterial infection by mobilizing cell surface accessible cholesterol. Nat. Microbiol. 2020;5:929–942. doi: 10.1038/s41564-020-0701-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Meffre D., Shackleford G., Hichor M., Gorgievski V., Tzavara E. T., et al. Liver X receptors alpha and beta promote myelination and remyelination in the cerebellum. Proc. Natl. Acad. Sci. USA. 2015;112:7587–7592. doi: 10.1073/pnas.1424951112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lee S. D., Priest C., Bjursell M., Gao J., Arneson D. V., et al. IDOL regulates systemic energy balance through control of neuronal VLDLR expression. Nat. Metab. 2019;1:1089–1100. doi: 10.1038/s42255-019-0127-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Krivoi I. I., Petrov A. M. Cholesterol and the safety factor for neuromuscular transmission. Int. J. Mol. Sci. 2019;20:1046. doi: 10.3390/ijms20051046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Petrov A. M., Pikuleva I. A. Cholesterol 24-hydroxylation by CYP46A1: Benefits of modulation for brain diseases. Neurotherapeutics. 2019;16:635–648. doi: 10.1007/s13311-019-00731-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Izumi Y., Cashikar A. G., Krishnan K., Paul S. M., Covey D. F., et al. A proinflammatory stimulus disrupts hippocampal plasticity and learning via microglial activation and 25-hydroxycholesterol. J. Neurosci. 2021;41:10054–10064. doi: 10.1523/JNEUROSCI.1502-21.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Rios R., Jablonka-Shariff A., Broberg C., Snyder-Warwick A. K. Macrophage roles in peripheral nervous system injury and pathology: Allies in neuromuscular junction recovery. Mol. Cell. Neurosci. 2021;111:103590. doi: 10.1016/j.mcn.2021.103590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Cao J., Gan H., Xiao H., Chen H., Jian D., et al. Key protein-coding genes related to microglia in immune regulation and inflammatory response induced by epilepsy. Math. Biosci. Eng. 2021;18:9563–9578. doi: 10.3934/mbe.2021469. [DOI] [PubMed] [Google Scholar]
- 61.Klejbor I., Shimshek D. R., Klimaszewska-Lata J., Velasco-Estevez M., Morys J., et al. EBI2 is expressed in glial cells in multiple sclerosis lesions, and its knock-out modulates remyelination in the cuprizone model. Eur. J. Neurosci. 2021;54:5173–5188. doi: 10.1111/ejn.15359. [DOI] [PubMed] [Google Scholar]
- 62.Rutkowska A., Shimshek D. R., Sailer A. W., Dev K. K. EBI2 regulates pro-inflammatory signalling and cytokine release in astrocytes. Neuropharmacology. 2018;133:121–128. doi: 10.1016/j.neuropharm.2018.01.029. [DOI] [PubMed] [Google Scholar]
- 63.Zhang Z. J., Jiang B. C., Gao Y. J. Chemokines in neuron-glial cell interaction and pathogenesis of neuropathic pain. Cell. Mol. Life Sci. 2017;74:3275–3291. doi: 10.1007/s00018-017-2513-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Skinner D. D., Lane T. E. CXCR2 signaling and remyelination in preclinical models of demyelination. DNA Cell Biol. 2020;39:3–7. doi: 10.1089/dna.2019.5182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Raccosta L., Fontana R., Maggioni D., Lanterna C., Villablanca E. J., et al. The oxysterol-CXCR2 axis plays a key role in the recruitment of tumor-promoting neutrophils. J. Exp. Med. 2013;210:1711–1728. doi: 10.1084/jem.20130440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Shida M., Mikami T., Tamura J. I., Kitagawa H. Chondroitin sulfate-D promotes neurite outgrowth by acting as an extracellular ligand for neuronal integrin alphaVbeta3. Biochim. Biophys. Acta Gen. Subj. 2019;1863:1319–1331. doi: 10.1016/j.bbagen.2019.06.004. [DOI] [PubMed] [Google Scholar]
- 67.Gardiner N. J., Moffatt S., Fernyhough P., Humphries M. J., Streuli C. H., et al. Preconditioning injury-induced neurite outgrowth of adult rat sensory neurons on fibronectin is mediated by mobilisation of axonal alpha5 integrin. Mol. Cell Neurosci. 2007;35:249–260. doi: 10.1016/j.mcn.2007.02.020. [DOI] [PubMed] [Google Scholar]
- 68.Wakatsuki S., Araki T., Sehara-Fujisawa A. Neuregulin-1/glial growth factor stimulates Schwann cell migration by inducing alpha5 beta1 integrin-ErbB2-focal adhesion kinase complex formation. Genes Cells. 2014;19:66–77. doi: 10.1111/gtc.12108. [DOI] [PubMed] [Google Scholar]
- 69.Cheng Z., Zhang Y., Tian Y., Chen Y., Ding F., et al. Cyr61 promotes Schwann cell proliferation and migration via alphavbeta3 integrin. BMC Mol. Cell Biol. 2021;22:21. doi: 10.1186/s12860-021-00360-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lu C. Y., Santosa K. B., Jablonka-Shariff A., Vannucci B., Fuchs A., et al. Macrophage-derived vascular endothelial growth factor-A is integral to neuromuscular junction reinnervation after nerve injury. J. Neurosci. 2020;40:9602–9616. doi: 10.1523/JNEUROSCI.1736-20.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Fu Q., Goodrum J. F., Hayes C., Hostettler J. D., Toews A. D., et al. Control of cholesterol biosynthesis in Schwann cells. J. Neurochem. 1998;71:549–555. doi: 10.1046/j.1471-4159.1998.71020549.x. [DOI] [PubMed] [Google Scholar]
- 72.Makoukji J., Shackleford G., Meffre D., Grenier J., Liere P., et al. Interplay between LXR and Wnt/beta-catenin signaling in the negative regulation of peripheral myelin genes by oxysterols. J. Neurosci. 2011;31:9620–9629. doi: 10.1523/JNEUROSCI.0761-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kasimov M. R., Fatkhrakhmanova M. R., Mukhutdinova K. A., Petrov A. M. 24S-Hydroxycholesterol enhances synaptic vesicle cycling in the mouse neuromuscular junction: Implication of glutamate NMDA receptors and nitric oxide. Neuropharmacology. 2017;117:61–73. doi: 10.1016/j.neuropharm.2017.01.030. [DOI] [PubMed] [Google Scholar]
- 74.Wei X., Nishi T., Kondou S., Kimura H., Mody I. Preferential enhancement of GluN2B-containing native NMDA receptors by the endogenous modulator 24S-hydroxycholesterol in hippocampal neurons. Neuropharmacology. 2019;148:11–20. doi: 10.1016/j.neuropharm.2018.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sun M. Y., Taylor A., Zorumski C. F., Mennerick S. 24S-hydroxycholesterol and 25-hydroxycholesterol differentially impact hippocampal neuronal survival following oxygen-glucose deprivation. PLoS One. 2017;12:e0174416. doi: 10.1371/journal.pone.0174416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Lin F., Yao X., Kong C., Liu X., Zhao Z., et al. 25-Hydroxycholesterol protecting from cerebral ischemia-reperfusion injury through the inhibition of STING activity. Aging (Albany NY) 2021;13:20149–20163. doi: 10.18632/aging.203337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ullrich C., Pirchl M., Humpel C. Effects of cholesterol and its 24S-OH and 25-OH oxysterols on choline acetyltransferase-positive neurons in brain slices. Pharmacology. 2010;86:15–21. doi: 10.1159/000314333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Chang J. Y., Liu L. Z. 25-Hydroxycholesterol causes death but does not prevent nerve growth factor-induced neurite outgrowth in PC12 cells. Neurochem. Int. 1997;31:517–523. doi: 10.1016/s0197-0186(97)00020-x. [DOI] [PubMed] [Google Scholar]
- 79.Olivier E., Dutot M., Regazzetti A., Leguillier T., Dargere D., et al. P2X7-pannexin-1 and amyloid beta-induced oxysterol input in human retinal cell: Role in age-related macular degeneration? Biochimie. 2016;127:70–78. doi: 10.1016/j.biochi.2016.04.014. [DOI] [PubMed] [Google Scholar]
- 80.Tajima N., Xiaoyan L., Taniguchi M., Kato N. 24S-hydroxycholesterol alters activity of large-conductance Ca2+-dependent K+ (slo1 BK) channel through intercalation into plasma membrane. Biochim Biophys Acta Mol. Cell Biol. Lipids. 2019;1864:1525–1535. doi: 10.1016/j.bbalip.2019.05.010. [DOI] [PubMed] [Google Scholar]
- 81.Phelan K. D., Mahler H. R. Acute exposure to 25-hydroxy-cholesterol selectively reduces GABAb and not GABAa receptor-mediated synaptic inhibition. Biochem. Biophys. Res. Commun. 1997;237:68–73. doi: 10.1006/bbrc.1997.7070. [DOI] [PubMed] [Google Scholar]
- 82.Ishikawa T., Yuhanna I. S., Umetani J., Lee W. R., Korach K. S., et al. LXRbeta/estrogen receptor-alpha signaling in lipid rafts preserves endothelial integrity. J. Clin. Invest. 2013;123:3488–3497. doi: 10.1172/JCI66533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Unsworth A. J., Flora G. D., Gibbins J. M. Non-genomic effects of nuclear receptors: Insights from the anucleate platelet. Cardiovasc. Res. 2018;114:645–655. doi: 10.1093/cvr/cvy044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Mukhutdinova K. A., Kasimov M. R., Giniatullin A. R., Zakyrjanova G. F., Petrov A. M. 24S-hydroxycholesterol suppresses neuromuscular transmission in SOD1(G93A) mice: A possible role of NO and lipid rafts. Mol. Cell Neurosci. 2018;88:308–318. doi: 10.1016/j.mcn.2018.03.006. [DOI] [PubMed] [Google Scholar]
- 85.Bigini P., Steffensen K. R., Ferrario A., Diomede L., Ferrara G., et al. Neuropathologic and biochemical changes during disease progression in liver X receptor beta–/– mice, a model of adult neuron disease. J. Neuropathol. Exp. Neurol. 2010;69:593–605. doi: 10.1097/NEN.0b013e3181df20e1. [DOI] [PubMed] [Google Scholar]
- 86.Hichor M., Sundaram V. K., Eid S. A., Abdel-Rassoul R., Petit P. X., et al. Liver X Receptor exerts a protective effect against the oxidative stress in the peripheral nerve. Sci. Rep. 2018;8:2524. doi: 10.1038/s41598-018-20980-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Abdel-Khalik J., Yutuc E., Crick P. J., Gustafsson J. A., Warner M., et al. Defective cholesterol metabolism in amyotrophic lateral sclerosis. J. Lipid Res. 2017;58:267–278. doi: 10.1194/jlr.P071639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zakyrjanova G. F., Giniatullin A. R., Mukhutdinova K. A., Kuznetsova E. A., Petrov A. M. Early differences in membrane properties at the neuromuscular junctions of ALS model mice: Effects of 25-hydroxycholesterol. Life Sci. 2021;273:119300. doi: 10.1016/j.lfs.2021.119300. [DOI] [PubMed] [Google Scholar]
- 89.Henriques A., Croixmarie V., Priestman D. A., Rosenbohm A., Dirrig-Grosch S., et al. Amyotrophic lateral sclerosis and denervation alter sphingolipids and up-regulate glucosylceramide synthase. Hum. Mol. Genet. 2015;24:7390–7405. doi: 10.1093/hmg/ddv439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Sugita S., Fleming L. L., Wood C., Vaughan S. K., Gomes M. P., et al. VAChT overexpression increases acetylcholine at the synaptic cleft and accelerates aging of neuromuscular junctions. Skelet. Muscle. 2016;6:31. doi: 10.1186/s13395-016-0105-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Bryndina I. G., Shalagina M. N., Sekunov A. V., Zefirov A. L., Petrov A. M. Clomipramine counteracts lipid raft disturbance due to short-term muscle disuse. Neurosci. Lett. 2018;664:1–6. doi: 10.1016/j.neulet.2017.11.009. [DOI] [PubMed] [Google Scholar]
- 92.Petrov A. M., Shalagina M. N., Protopopov V. A., Sergeev V. G., Ovechkin S. V., et al. Changes in membrane ceramide pools in rat soleus muscle in response to short-term disuse. Int. J. Mol. Sci. 2019;20:4860. doi: 10.3390/ijms20194860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Slater C. R. “Fragmentation” of NMJs: A sign of degeneration or regeneration? A long journey with many junctions. Neuroscience. 2020;439:28–40. doi: 10.1016/j.neuroscience.2019.05.017. [DOI] [PubMed] [Google Scholar]
- 94.Dupuis L., Corcia P., Fergani A., Gonzalez De Aguilar J. L., Bonnefont-Rousselot D., et al. Dyslipidemia is a protective factor in amyotrophic lateral sclerosis. Neurology. 2008;70:1004–1009. doi: 10.1212/01.wnl.0000285080.70324.27. [DOI] [PubMed] [Google Scholar]
- 95.Su X. W., Nandar W., Neely E. B., Simmons Z., Connor J. R. Statins accelerate disease progression and shorten survival in SOD1(G93A) mice. Muscle Nerve. 2016;54:284–291. doi: 10.1002/mus.25048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Dias I. H. K., Milic I., Lip G. Y. H., Devitt A., Polidori M. C., et al. Simvastatin reduces circulating oxysterol levels in men with hypercholesterolaemia. Redox Biol. 2018;16:139–145. doi: 10.1016/j.redox.2018.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Wolf Y., Boura-Halfon S., Cortese N., Haimon Z., Sar Shalom H., et al. Brown-adipose-tissue macrophages control tissue innervation and homeostatic energy expenditure. Nat. Immunol. 2017;18:665–674. doi: 10.1038/ni.3746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Limbad C., Doi R., McGirr J., Ciotlos S., Perez K., et al. Senolysis induced by 25-hydroxycholesterol targets CRYAB in multiple cell types. iScience. 2022;25:103848. doi: 10.1016/j.isci.2022.103848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Dong Z., He F., Yan X., Xing Y., Lei Y., et al. Hepatic reduction in cholesterol 25-hydroxylase aggravates diet-induced steatosis. Cell. Mol. Gastroenterol. Hepatol. 2022;13:1161–1179. doi: 10.1016/j.jcmgh.2021.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Mukhutdinova K. A., Kasimov M. R., Zakyrjanova G. F., Gumerova M. R., Petrov A. M. Oxysterol modulates neurotransmission via liver-X receptor/NO synthase-dependent pathway at the mouse neuromuscular junctions. Neuropharmacology. 2019;150:70–79. doi: 10.1016/j.neuropharm.2019.03.018. [DOI] [PubMed] [Google Scholar]
- 101.Tyganov S. A., Mochalova E., Belova S., Sharlo K., Rozhkov S., et al. Plantar mechanical stimulation attenuates protein synthesis decline in disused skeletal muscle via modulation of nitric oxide level. Sci. Rep. 2021;11:9806. doi: 10.1038/s41598-021-89362-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Tyganov S. A., Mochalova E. P., Melnikov I. Y., Vikhlyantsev I. M., Ulanova A. D., et al. NOS-dependent effects of plantar mechanical stimulation on mechanical characteristics and cytoskeletal proteins in rat soleus muscle during hindlimb suspension. FASEB. J. 2021;35:e21905. doi: 10.1096/fj.202100783R. [DOI] [PubMed] [Google Scholar]
- 103.Petrov A. M., Kravtsova V. V., Matchkov V. V., Vasiliev A. N., Zefirov A. L., et al. Membrane lipid rafts are disturbed in the response of rat skeletal muscle to short-term disuse. Am. J. Physiol. Cell Physiol. 2017;312:C627–C637. doi: 10.1152/ajpcell.00365.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Clements L., Harvey J. Activation of oestrogen receptor alpha induces a novel form of LTP at hippocampal temporoammonic-CA1 synapses. Br. J. Pharmacol. 2020;177:642–655. doi: 10.1111/bph.14880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Brooks S. W., Dykes A. C., Schreurs B. G. A high-cholesterol diet increases 27-hydroxycholesterol and modifies estrogen receptor expression and neurodegeneration in rabbit hippocampus. J. Alzheimer’s Dis. 2017;56:185–196. doi: 10.3233/JAD-160725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Jetten A. M., Takeda Y., Slominski A., Kang H. S. Retinoic acid-related Orphan Receptor gamma (RORgamma): Connecting sterol metabolism to regulation of the immune system and autoimmune disease. Curr. Opin. Toxicol. 2018;8:66–80. doi: 10.1016/j.cotox.2018.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Solt L. A., Griffin P. R., Burris T. P. Ligand regulation of retinoic acid receptor-related orphan receptors: Implications for development of novel therapeutics. Curr. Opin. Lipidol. 2010;21:204–211. doi: 10.1097/MOL.0b013e328338ca18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Journiac N., Jolly S., Jarvis C., Gautheron V., Rogard M., et al. The nuclear receptor ROR(alpha) exerts a bi-directional regulation of IL-6 in resting and reactive astrocytes. Proc. Natl. Acad. Sci. USA. 2009;106:21365–21370. doi: 10.1073/pnas.0911782106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Schaeren-Wiemers N., Andre E., Kapfhammer J. P., Becker-Andre M. The expression pattern of the orphan nuclear receptor RORbeta in the developing and adult rat nervous system suggests a role in the processing of sensory information and in circadian rhythm. Eur. J. Neurosci. 1997;9:2687–2701. doi: 10.1111/j.1460-9568.1997.tb01698.x. [DOI] [PubMed] [Google Scholar]
- 110.Kasimov M. R., Zakyrjanova G. F., Giniatullin A. R., Zefirov A. L., Petrov A. M. Similar oxysterols may lead to opposite effects on synaptic transmission: Olesoxime versus 5alpha-cholestan-3-one at the frog neuromuscular junction. Biochim. Biophys. Acta. 2016;1861:606–616. doi: 10.1016/j.bbalip.2016.04.010. [DOI] [PubMed] [Google Scholar]
- 111.Kasimov M. R., Giniatullin A. R., Zefirov A. L., Petrov A. M. Effects of 5alpha-cholestan-3-one on the synaptic vesicle cycle at the mouse neuromuscular junction. Biochim. Biophys. Acta. 2015;1851:674–685. doi: 10.1016/j.bbalip.2015.02.012. [DOI] [PubMed] [Google Scholar]
- 112.Petrov A. M., Mast N., Li Y., Denker J., Pikuleva I. A. Brain sterol flux mediated by cytochrome P450 46A1 affects membrane properties and membrane-dependent processes. Brain Commun. 2020;2:fcaa043. doi: 10.1093/braincomms/fcaa043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Kobierski J., Wnetrzak A., Chachaj-Brekiesz A., Filiczkowska A., Petelska A. D., et al. How the replacement of cholesterol by 25-hydroxycholesterol affects the interactions with sphingolipids: The Langmuir Monolayer Study complemented with theoretical calculations. J. R. Soc. Interface. 2021;18:20210050. doi: 10.1098/rsif.2021.0050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Olsen B. N., Schlesinger P. H., Ory D. S., Baker N. A. 25-Hydroxycholesterol increases the availability of cholesterol in phospholipid membranes. Biophys. J. 2011;100:948–956. doi: 10.1016/j.bpj.2010.12.3728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Bielska A. A., Olsen B. N., Gale S. E., Mydock-McGrane L., Krishnan K., et al. Side-chain oxysterols modulate cholesterol accessibility through membrane remodeling. Biochemistry. 2014;53:3042–3051. doi: 10.1021/bi5000096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Xu X., London E. The effect of sterol structure on membrane lipid domains reveals how cholesterol can induce lipid domain formation. Biochemistry. 2000;39:843–849. doi: 10.1021/bi992543v. [DOI] [PubMed] [Google Scholar]
- 117.Olsen B. N., Schlesinger P. H., Baker N. A. Perturbations of membrane structure by cholesterol and cholesterol derivatives are determined by sterol orientation. J. Am. Chem. Soc. 2009;131:4854–4865. doi: 10.1021/ja8095224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Domingues M. M., Gomes B., Hollmann A., Santos N. C. 25-hydroxycholesterol effect on membrane structure and mechanical properties. Int. J. Mol. Sci. 2021;22:2574. doi: 10.3390/ijms22052574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Gomes B., Goncalves S., Disalvo A., Hollmann A., Santos N. C. Effect of 25-hydroxycholesterol in viral membrane fusion: Insights on HIV inhibition. Biochim. Biophys. Acta Biomembr. 2018;1860:1171–1178. doi: 10.1016/j.bbamem.2018.02.001. [DOI] [PubMed] [Google Scholar]
- 120.Sibarov D. A., Poguzhelskaya E. E., Antonov S. M. Downregulation of calcium-dependent NMDA receptor desensitization by sodium-calcium exchangers: A role of membrane cholesterol. BMC Neurosci. 2018;19:73. doi: 10.1186/s12868-018-0475-3. [DOI] [PMC free article] [PubMed] [Google Scholar]