In 1510, Leonardo Da Vinci was the first to describe the heart muscle as arranged in longitudinal and transverse fibers and their contraction resulted in shortening and widening of the ventricular cavities.1 Since then, there has been a divergence in the study of cardiac and skeletal muscle cells because of the complexity in structure and function of cardiac myocytes. In this issue of Circulation, du Fay de Lavallaz et al.2, reporting on the findings of the four-hospital based Heart & Muscle BASEL XII Study, have in part circled back by producing conclusive evidence of the shared pathophysiology between these two muscle types with the expression of the cardiac isoform of troponin T in chronic skeletal myopathies and myositis.2
The investigative team examined circulating levels of cardiac troponin T and I (cTnT and I) using 1 cTnT and 3 cTnI widely utilized commercial high-sensitivity (hs) assays among 211 patients with several skeletal muscle diseases (SMD) including myopathy, myositis, neuropathy, myasthenic syndrome, auto-immune disease, and non-specific muscle symptoms enrolled from inpatient (11%) or elective ambulatory clinics (89%) from general medical or specialized neuromuscular and rheumatology services. The investigators found that approximately half of the SMD participants had some form of cardiovascular disease (CVD) and most commonly this was due to coronary heart disease (CHD). There was a dose-response relationship between the severity of known CVD and cardiac myocyte injury with higher levels of both hs-cTnT or I. Hs-cTnT values were above the upper limit of normal (ULN) in 55% of study population with skeletal muscle disease (SMD) with a median level of 16 ng/L versus 5 ng/L for controls (emergency department patients with adjudicated non-cardiac chest pain [n=3,508]), whereas hs-cTnI levels, measured by the three other commercial assays, were at most elevated in 23% among those with SMD and median levels were nearly identical to controls for all 3 assays. This observation was more remarkable when evaluating the subgroup without known CVD; SMD resulted in 37% of the hs-cTnT levels above the ULN versus 2-8% with the hs-cTnI assays. Elevations in hs-cTnT among patients without underlying CVD were mostly found among patients with non-inflammatory myopathy or inflammatory myositis, which correlated with creatine kinase, a biomarker of skeletal muscle damage. In contrast, there was no correlation with cTnI levels measured by any of the three hs-cTnI assays and creatinine kinase. Aiming to establish causal associations, the investigators also measured cTn specific mRNA from muscle biopsies in 88 SMD patients and then 16 controls having elective hip surgery. They found there was an 8-fold upregulation of TNNT2 mRNA expression (coding cTnT) versus controls, which correlated with circulating hs-cTnT concentrations, but there was no upregulation of mRNA coding for cTnI.
The work by du Fay de Lavallaz et al. supports the earlier associations between elevated circulating hs-cTnT levels with various myopathies ascertained by biopsies.3, 4 The elevation in cTnT was previously observed in skeletal myopathies regardless of whether the underlying muscle injury was endocrine, metabolic, autoimmune, genetic, neurally-mediated or caused by drug toxicity.3 While the results are robust and corroborated with prior evidence, as with most well conducted studies, a path is opened to ask more questions about the clinical implications of the dichotomization of diseased skeletal muscle to express cTnT and not cTnI beyond a highly select group of patients with symptomatic skeletal muscle myopathies or myositis. First, as we recently described, there are many asymptomatic patients with malnutrition, sarcopenia, frailty, or even cachexia that phenotypically exhibit interrelated pathophysiological processes leading to different magnitudes of “wasting” and may be susceptible to similar muscle pathophysiologic changes as patients with active chronic muscular complaints.5, 6 Recently an “omics” approach using machine learning across four older adult cohorts showed cTnT was one of the most strongly associated circulating proteins with pre-frailty and frailty with and without disability.7 Second, the impact of the findings by du Fay de Lavallaz on patients with suspected or known SMD during acute presentations needs to be carefully considered. In many cases the levels of both cTnT and cTnI will often be at least several fold higher than the ULN but given that 55% of the study population had levels above the hs-cTnT ULN, levels closer to the 99th percentile established ULN will need appropriate clinical caution and the short-term longitudinal change in circulating levels of patients with SMD may need further study. Third, while biologic plausibility is ascertained in this study, the strength of the association between elevated hs-cTnT and long-term incident CVD outcomes among patients with active muscle complaints remain unknown and warrants additional investigation.
Cardiovascular clinicians are regularly tasked to care for complex patients living with multiple chronic conditions and these conditions often confound clinical decision making. SMD often requires a high level of clinical suspicion, and the interpretation of diagnostic testing among these patients pose challenges. With assimilation of the findings from the present study perhaps most interesting is the (re) interpretation of hs-cTnI and hs-cTnT for prognosis in ambulatory adults. For more than a decade, it has been recognized that cTnT and cTnI measured with hs assays identify ambulatory patients either free of prevalent CVD, with known CHD, or chronic kidney disease who are at increased risk of CVD hospitalization and death.8, 9 High-sensitivity assays for cTnI and cTnT can be used interchangeably for the diagnosis of acute myocardial infarction (AMI) with both assays showing equivalent accuracy and a high correlation (Pearson’s r=0.89), but the same may not be true in ambulatory cohorts.10 In the Atherosclerosis Risk in Communities (ARIC) study evaluating participants without prevalent CVD and the Prevention of Events With Angiotensin-Converting Enzyme Inhibitor Therapy (PEACE) trial evaluating participants with stable CHD without heart failure (HF), the same hs-cTnT and hs-cTnI assay results were only moderately correlated (r=0.47 and r=0.44, respectively).8, 11 Unlike AMI evaluations, this discordance between hs-cTnT and hs-cTnI results among ambulatory participants has translated into differences for predicting some, but not all CVD events. For example, in the large community cohort Generation Scotland Scottish Family Health Study progressively higher baseline hs-cTnI, but not hs-cTnT levels, predicted incident AMI.12 A similar finding for incident AMI was seen in PEACE.8 In contrast, in the same studies both hs-cTnT and hs-cTnI have similar accuracy for the prediction of incident HF.8, 12
In older adults does skeletal muscle production of cTnT detectable by a hs assay account for the discordance between circulating cTnT and cTnI levels? It is provocative that incident HF, which can be driven by systemic co-morbidities (clinical or subclinical), particularly for HF with preserved ejection fraction, may be equally well predicted by a circulating biomarker potentially reflecting cardiac and skeletal muscle disease (cTnT) as a biomarker exclusive to cardiac injury (cTnI). However, a circulating biomarker exclusive to cardiac injury (cTnI) may be a more specific harbinger of CHD events. This doesn’t mean that cTnT may not have an important role in prediction of incident CVD (Figure 1). If future work bears out skeletal muscle production of cTnT in pre-frail, frail, or sarcopenic adults, the biomarker might ultimately find its way as a measure of the efficacy of physical activity on both skeletal and cardiac myocytes; pilot work in sedentary older adults has shown a physical activity program can reduce the longitudinal trajectory of hs-cTnT levels.13 As with most “definitive” well conducted studies, the work du Fay de Lavallaz provides a big step forward with respect to interpreting hs-cTn in those presenting with known MSD, but importantly now challenges and potentially enhances our interpretation of hs-cTn results in a much broader “at-risk” ambulatory population.
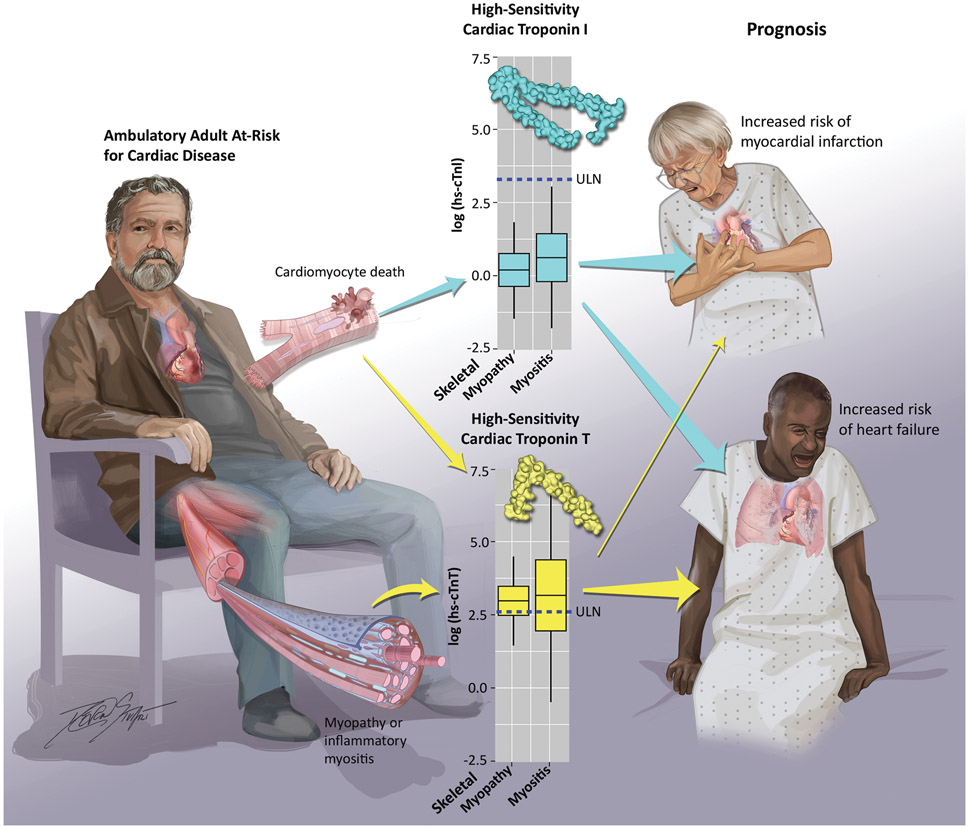
Figure 1.
Adults with pre-frailty, frailty or others with co-morbidities may have skeletal muscle loss or disorders that could result in cTnT production (left panel). Circulating cTnT, but not cTnI is measurable by high sensitivity assays in patients without known cardiovascular disease, but skeletal myopathy or myositis (from du Fay de Lavallaz et al.2) (middle panel). Prior longitudinal ambulatory studies show that both cTnI and cTnT levels are long-term prognosticators for incident heart failure, but hs-cTnI is more prognostic of incident MI (right panel). cTnI, cardiac troponin I; cTnT, cardiac troponin T; hs, high sensitivity; ULN, upper limit of normal (defined as the 99th percentile value in a healthy adult population).
FUNDING:
Dr. Damluji receives research funding from the Pepper Scholars Program of the Johns Hopkins University Claude D. Pepper Older Americans Independence Center funded by the National Institute on Aging P30-AG021334 and mentored patient-oriented research career development award from the National Heart, Lung, and Blood Institute K23-HL153771-01.
Dr deFilippi receives research funding from the National Institute of Health R01HL154768, R01HL151293, R21AG072095 and 1UL1TR003015.
Footnotes
DISCLOSURES: Dr. deFilippi serves as a consultant for Abbott Diagnostics, FujiRebio, Ortho Diagnostics, Quidel, Roche Diagnostics, and Siemens Healthineers which all manufacture cardiac biomarker assays. Dr. Damluji has no relevant interests.
REFERENCES
- 1.Keele KD. Leonardo da Vinci as physiologist. Postgrad Med J. 1952;28:521–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.du Fay de Lavallaz J, Prepoudis A, Wendebourg MJ, Kesenheimer E, Kyburz D, Daikeler T, Haaf P, Wanschitz J, Löscher WN, Schreiner B, et al. Skeletal Muscle Disorders: A Non-cardiac Source of Cardiac Troponin T. Circulation. 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rittoo D, Jones A, Lecky B and Neithercut D. Elevation of Cardiac Troponin T, But Not Cardiac Troponin I, in Patients With Neuromuscular Diseases: Implications for the Diagnosis of Myocardial Infarction. Journal of the American College of Cardiology. 2014;63:2411–2420. [DOI] [PubMed] [Google Scholar]
- 4.Jaffe AS, Vasile VC, Milone M, Saenger AK, Olson KN and Apple FS. Diseased skeletal muscle: a noncardiac source of increased circulating concentrations of cardiac troponin T. J Am Coll Cardiol. 2011;58:1819–24. [DOI] [PubMed] [Google Scholar]
- 5.Ijaz N, Buta B, Xue QL, Mohess DT, Bushan A, Tran H, Batchelor W, deFilippi CR, Walston JD, Bandeen-Roche K, et al. Interventions for Frailty Among Older Adults With Cardiovascular Disease: JACC State-of-the-Art Review. J Am Coll Cardiol. 2022;79:482–503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Damluji AA, Forman DE, van Diepen S, Alexander KP, Page RL 2nd, Hummel SL, Menon V, Katz JN, Albert NM, Afilalo J, et al. Older Adults in the Cardiac Intensive Care Unit: Factoring Geriatric Syndromes in the Management, Prognosis, and Process of Care: A Scientific Statement From the American Heart Association. Circulation. 2019:Cir0000000000000741. [DOI] [PubMed] [Google Scholar]
- 7.Gomez-Cabrero D, Walter S, Abugessaisa I, Miñambres-Herraiz R, Palomares LB, Butcher L, Erusalimsky JD, Garcia-Garcia FJ, Carnicero J, Hardman TC, et al. A robust machine learning framework to identify signatures for frailty: a nested case-control study in four aging European cohorts. Geroscience. 2021;43:1317–1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Omland T, Pfeffer MA, Solomon SD, de Lemos JA, Røsjø H, Šaltytė Benth J, Maggioni A, Domanski MJ, Rouleau JL, Sabatine MS, et al. Prognostic value of cardiac troponin I measured with a highly sensitive assay in patients with stable coronary artery disease. J Am Coll Cardiol. 2013;61:1240–9. [DOI] [PubMed] [Google Scholar]
- 9.Saunders JT, Nambi V, de Lemos JA, Chambless LE, Virani SS, Boerwinkle E, Hoogeveen RC, Liu X, Astor BC, Mosley TH, et al. Cardiac troponin T measured by a highly sensitive assay predicts coronary heart disease, heart failure, and mortality in the Atherosclerosis Risk in Communities Study. Circulation. 2011;123:1367–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.van der Linden N, Wildi K, Twerenbold R, Pickering JW, Than M, Cullen L, Greenslade J, Parsonage W, Nestelberger T, Boeddinghaus J, et al. Combining High-Sensitivity Cardiac Troponin I and Cardiac Troponin T in the Early Diagnosis of Acute Myocardial Infarction. Circulation. 2018;138:989–999. [DOI] [PubMed] [Google Scholar]
- 11.Jia X, Sun W, Hoogeveen RC, Nambi V, Matsushita K, Folsom AR, Heiss G, Couper DJ, Solomon SD, Boerwinkle E, et al. High-Sensitivity Troponin I and Incident Coronary Events, Stroke, Heart Failure Hospitalization, and Mortality in the ARIC Study. Circulation. 2019;139:2642–2653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Welsh P, Preiss D, Hayward C, Shah ASV, McAllister D, Briggs A, Boachie C, McConnachie A, Padmanabhan S, Welsh C, et al. Cardiac Troponin T and Troponin I in the General Population. Circulation. 2019;139:2754–2764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.deFilippi CR, de Lemos JA, Newman AB, Guralnik JM, Christenson RH, Pahor M, Church T, Espeland M, Krithevsky SB, Stafford R, Seliger SL. Impact of moderate physical activity on the longitudinal trajectory of a cardiac specific biomarker of injury: Results from a randomized pilot study of exercise intervention. Am Heart J. 2016;179:151–156 [DOI] [PubMed] [Google Scholar]