Abstract
Background
Globally, infectious diseases are the major cause of death in children under the age of 5 years. Sub-Saharan Africa and South Asia account for 95% of global child mortalities every year, where acute respiratory infections (ARI) remain the leading cause of child morbidity and mortality. The aim of this study is to analyze the risk factors of ARI disease symptoms among children under the age of 5 years in Uganda.
Methods
A cross-sectional design was used to analyze 2016 Uganda Demographic and Health Survey (UDHS) data collected on 13,493 children under the age of 5 years in Uganda. Various methods, such as logistic regression, elastic net logistic regression, decision tree, and random forest, were compared and used to predict 75% of the symptom outcomes of ARI disease. Well-performing methods were used to determine potential risk factors for ARI disease symptoms among children under the age of 5 years.
Results
In Uganda, about 40.3% of children were reported to have ARI disease symptoms in the 2 weeks preceding the survey. Children under the age of 24 months were found to have a high prevalence of ARI disease symptoms. By considering 75% of the sample, the random forest was found to be a well-performing method (accuracy = 88.7%; AUC = 0.951) compared to the logistic regression method (accuracy = 62.0%; AUC = 0.638) and other methods in predicting childhood ARI symptoms. In addition, one-year old children (OR: 1.27; 95% CI: 1.12–1.44), children whose mothers were teenagers (OR: 1.28; 95% CI: 1.06–1.53), and farm workers (1.25; 95% CI: 1.11–1.42) were most likely to have ARI disease symptoms than other categories. Furthermore, children aged 48–59 months (OR: 0.69; 95% CI: 0.60–0.80), breastfed children (OR: 0.83; 95% CI: 0.76–0.92), usage of charcoal in cooking (OR: 0.77; 95% CI: 0.69–0.87), and the rainy season effect (OR: 0.66; 95% CI: 0.61–0.72) showed a low risk of developing ARI disease symptoms among children under the age of 5 years in Uganda.
Conclusion
Policy-makers and health stakeholders should initiate target-oriented approaches to address the problem regarding poor children’s healthcare, improper environmental conditions, and childcare facilities. For the sake of early child care, the government should promote child breastfeeding and maternal education.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12889-022-13532-y.
Keywords: Acute respiratory infections, Risk-factors, Under-five mortality, Uganda
Background
Globally, infant and child mortality rates are critical issues and fundamental indicators of a country’s population’s health, quality of life, and socioeconomic situation [1]. A remarkable decline of 60% in under-five mortality has been observed over the last three decades. However, 7.4 million annual global mortalities are estimated due to preventable and treatable diseases in young children. Besides, 70% of these deaths occur in children under the age of 5 years, and 95% are from South Asia and sub-Saharan Africa, i.e., on average, about 1 in 13 children in sub-Saharan Africa die before the age of five [2]. Various factors may contribute to high mortality rates, such as poor living conditions and other socio-economic factors of countries’ populations where childhood acute respiratory infections (ARI) remain among the top leading morbidities in low-income countries, particularly in sub-Saharan Africa [3].
The ARI disease and its related symptoms are typically caused by contagious viruses and bacterial infections that spread rapidly through droplets from either person-to-person or contaminated food or drinking water due to poor hygiene [4]. According to WHO 2019, ARI diseases are the fourth most common childhood disease among those with a higher rate of morbidity. When combined with malaria, ARI diseases become the top communicable diseases causing more deaths than other comorbidities [5–7]. In addition, the symptoms of ARI disease coincide with those of diarrhea and malaria diseases could lead to childhood death [8].
In Uganda, ARI has remained the leading cause of morbidity and mortality in children under the age of 5 years, accounting for about 9% of the ARI prevalence, with 81.3% in urban areas. The under-five mortality rate accounted for 1 in 16 child deaths, and 42% of these deaths occurred in the neonatal period [9]. The heavy loss of young lives from childhood ARI mortalities poses a heavy burden to families and healthcare providers in Uganda. Therefore, conducting research regarding the assessment of risk factors related to such diseases can greatly help policy decision-making and reduce these morbidity and mortality rates, especially in under-five children.
Traditional analysis methods such as logistic regression and chi-square test approaches are commonly applied in social science and medical literature. However, in diagnosing cardiopulmonary diseases using medical data, machine learning tools have become popular and frequently used in recent research [10]. The appropriate usage of machine learning algorithms has revealed significant performance in the prediction and classification of disease outcomes [11]. This study aims to determine potential risk factors contributing to ARI disease symptoms in children under the age of 5 years in Uganda using well-performed methods to predict the ARI symptom outcomes between traditional and machine learning analysis methods. The study findings could help in making research-based decisions to address the associated risk factors of ARI disease symptoms relevant to the disease’s control and spread among children.
Methods
Data source
This study used secondary data from the recent five-year cross-sectional survey, the Uganda Demographic and Health Survey (UDHS), that was conducted between June and December 2016. The UDHS is conducted by the Ugandan Bureau of Statistics and collaborated with the DHS program to collect up-to-date data for fundamental demographic and health indicators relevant to policymakers and program managers in order to evaluate the national population’s health and nutritional programs [9]. The DHS data collected in different developing countries can be found and downloaded via the website of the DHS program after approval.
Design and sampling
We used a cross-sectional study design in collecting characteristics and information regarding the prevalence of the ARI disease symptoms among children under the age of 5 years in Uganda, using UDHS data collected in 2016. We used a two-stage stratified sampling design to select the sample. The first stage involved selecting 697 geographic areas named enumerated areas (EAs) (535 rural and 162 urban EAs) that covered 130 households on average, and the second stage involved the selection of households to be included in each EA. All the EAs with more than 300 households were segmented into one EA, and the households in the EAs were selected with a probability proportional to the size of the segment [9].
Population and sample
The target population of this study was comprised of male and female children under the age of 5 years from different regions of Uganda. The data and recorded information for 13,493 children were used as the sample for this study. The total sample of children was divided into two groups: 75% for analysis and 25% for testing the performance of the various methods of analysis used in the study.
Variables of interest
In this study, we used various characteristics that were measured in the 2016 UDHS [9] and factors from other related literature, which were included in the survey dataset (Fig. 1). Behavioral, environmental, and social demographic characteristics for children, mothers, and households were used to analyze and determine potential risk factors associated with the symptoms of ARI disease in children under the age of five in Uganda. During the survey, mothers aged 15–49 years who had children under the age of 5 years in the selected households were asked whether their children experienced ARI disease symptoms such as coughing accompanied by short, rapid or difficulty breathing in the 2 weeks before the survey. The responses regarding the ARI disease symptoms were considered subjective since they were mothers’ perceptions without validation from medical personnel. The explanations for the variables used in this study are presented in the supplementary file in Tables A1 and A2.
Fig. 1.
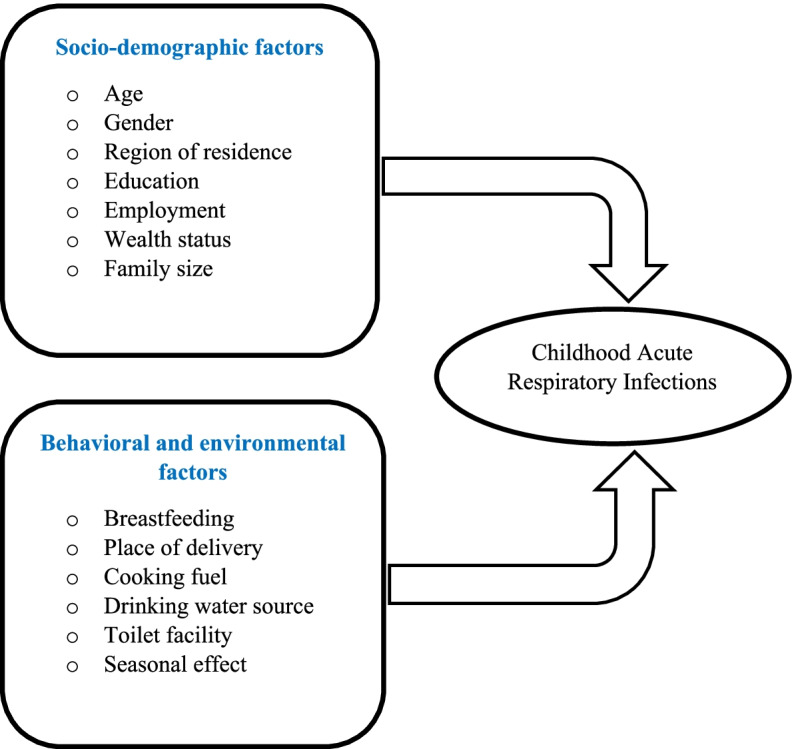
A framework of factors of childhood ARI disease symptoms
Analysis methods
The scope of this study focuses primarily on determining the potential risk factors of ARI disease symptoms based on the well-performing methods between traditional and machine learning methods of analysis mostly applied in the social sciences and medical research [12].
Logistic regression
We used a binary logistic regression (LR) model shown in Eq. 1 to analyze the log-linear association of k variables (i.e., factors), X1,X2, …,Xk with their corresponding b1,b2, …,bk effects on Y outcome of the ARI disease symptoms, 1 if “child had ARI symptoms” and 0 “otherwise” where π indicates the probability that a child had the ARI symptoms [13]. Stepwise variable selection procedures were also used to select influential factors associated with the outcome of interest, i.e., the symptoms of ARI disease.
| 1 |
Elastic net regression
The elastic net logistic regression (EN) model shown in Eq. 2 was used in addition to the previous LR model in Eq. 1 to control the correlation between features in order to solve the problem of overfitting that could exist in the analysis of risk factors associated with the outcomes of the ARI disease symptoms [14]. The EN method penalizes and shrinks b1,b2, …,bk effects of the non-informative x1,x2, …,xk variables using non-negative tuning parameters αϵ[0, 1] and λ with ten-fold cross-validation [2].
| 2 |
Machine learning methods
In addition, machine learning algorithms such as decision tree (DT) and random forest (RF) methods were also used in comparisons with the regression methods to predict the outcomes of ARI disease symptoms in children under the age of 5 years in Uganda [15, 16]. The DT algorithm was particularly used due to its advantages like its tree-like structure, which is simple and easy to learn and interpret, while the RF algorithm approach was used as an extension of the DT method because of its effectiveness in minimizing the variance using its random DT tree-like structures generated from a random sample in the prediction [17].
Measures of evaluation
In the evaluation of the performance of the methods used in this study, we considered various measures or metrics that are applied in the contingency matrix in diagnosing ill patients in most medical research [18]. A total accuracy in Eq. 3 measures the proportion of all children reported as with and without ARI disease symptoms who are correctly predicted by the method in this study; a precision measure in Eq. 4 shows the proportion of children who actually had ARI symptoms and were correctly predicted as having ARI disease symptoms. While the selectivity measure shown in Eq. 5 measured the proportion of children who were actually reported as not having ARI symptoms and correctly predicted by the method as not having ARI symptoms; A recall measure in Eq. 6, also called a sensitivity measure, indicates the proportion of the children who are predicted as symptomatic among all children with ARI symptoms in the study. We also used the area under the curves (AUCs) measure for the receiver operating characteristic (ROC) curves based on the true and predicted outcomes of ARI symptoms [10]. This study used statistical software such as STATA version 17.0 for data management and R software using functions in the Caret package for analyzing data.
| 3 |
| 4 |
| 5 |
| 6 |
Where TP, TN, FP, and FN represent the number of true positives, true negatives, false positives, and false negatives respectively.
Results
In this study, a sample of 13,493 children under the age of 5 years in Uganda was analyzed. Overall, the prevalence of ARI disease symptoms in children with symptoms was found to be 5437 (40.3%) and 8056 (59.7%) for children without ARI disease symptoms (Fig. 2). Tables 1 and 2 show that the symptoms’ prevalence of ARI disease in children was high in males (50.7%) compared to females (49.3%), and about 44.5% of children with ARI symptoms were under 24 months of age, and 33.8% had mothers under 25 years of age and living in a lower-income class (47.4%). About 74.2% of children had mothers who only attended below the secondary level of education, and only 56.6% were breastfed. The majority of children reported were found in households exposed to wood smoke from firewood as cooking energy (77.9%) and 53.4% reported in the dry season.
Fig. 2.
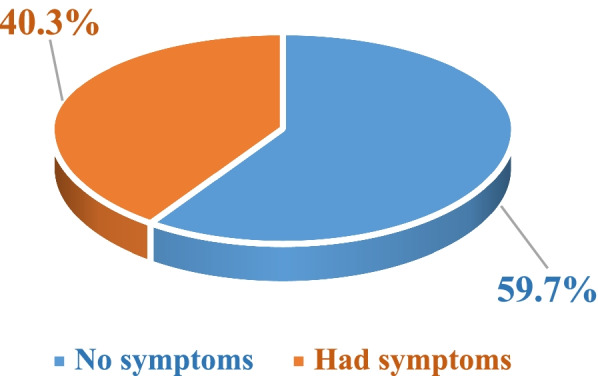
Prevalence of ARI disease symptoms in children under the age of 5 years in Uganda
Table 1.
Distribution of the ARI symptoms’ prevalence based on socio-economic and demographic characteristics
| Characteristics | ARI disease symptoms: n = 13,493 | Sig. | ||
|---|---|---|---|---|
| No (%) | Yes (%) | Total (%) | ||
| Child age (months) | ||||
| 0–11 | 1724 (21.4) | 1149 (21.1) | 2873 (21.3) | < 0.001 |
| 12–23 | 1440 (17.9) | 1273 (23.4) | 2713 (20.1) | |
| 24–35 | 1534 (19.0) | 1124 (20.7) | 2658 (19.7) | |
| 36–47 | 1608 (20.0) | 1008 (18.5) | 2616 (19.4) | |
| 48–59 | 1750 (21.7) | 883 (16.3) | 2633 (19.5) | |
| Child gender | ||||
| Male | 3991 (49.5) | 2757 (50.7) | 6748 (50.0) | 0.183 |
| Female | 4065 (50.5) | 2680 (49.3) | 6745 (50.0) | |
| Region of residence | ||||
| Central | 1358 (16.9) | 1360 (25.0) | 2718 (20.1) | < 0.001 |
| Eastern | 2346 (29.1) | 1517 (27.9) | 3863 (28.6) | |
| Northern | 2023 (25.1) | 1357 (25.0) | 3.380 (25.1) | |
| Western | 2329 (28.9) | 1203 (22.7) | 3532 (26.2) | |
| Mother age (years) | ||||
| 15–19 | 441 (5.5) | 364 (6.7) | 805 (6.0) | < 0.001 |
| 20–24 | 2165 (26.9) | 1474 (27.1) | 3639 (27.0) | |
| 25–29 | 1991 (24.7) | 1466 (27.0) | 3457 (25.6) | |
| 30–34 | 1716 (21.3) | 1072 (19.7) | 2788 (20.7) | |
| 35–39 | 1094 (13.6) | 670 (12.3) | 1764 (13.1) | |
| 40–49 | 649 (8.1) | 391 (7.2) | 1040 (7.7) | |
| Mother education | ||||
| No education | 1097 (13.6) | 705 (13.0) | 1802 (13.4) | 0.002 |
| Primary | 5110 (63.4) | 3328 (61.2) | 8438 (62.5) | |
| Secondary | 1449 (18.0) | 1088 (20.0) | 2537 (18.8) | |
| Tertiary | 400 (5.0) | 316 (5.8) | 716 (5.3) | |
| Mother employment | ||||
| Unemployed | 1481 (18.4) | 698 (12.8) | 2179 (16.1) | < 0.001 |
| Farmer | 3947 (49.0) | 2380 (43.8) | 6327 (46.9) | |
| Other | 2628 (32.6) | 2359 (43.4) | 4987 (37.0) | |
| Mother wealth status | ||||
| Lower | 4012 (49.8) | 2579 (47.4) | 6591 (48.8) | 0.004 |
| Middle | 1557 (19.3) | 1033 (19.0) | 2590 (19.2) | |
| Higher | 2487 (30.9) | 1825 (33.6) | 4312 (32.0) | |
Sig. 5% Chi-square test significance
Table 2.
Distribution of the ARI symptoms’ prevalence based on behavioral and environmental characteristics
| Characteristics | ARI disease symptoms: n = 13,493 | Sig. | ||
|---|---|---|---|---|
| No (%) | Yes (%) | Total (%) | ||
| Family size | ||||
| Not crowded (≤ 5) | 3746 (46.5) | 2581 (47.5) | 6327 (46.9) | 0.267 |
| Crowded (> 5) | 4310 (53.5) | 2856 (52.5) | 7166 (53.1) | |
| Breastfeeding | ||||
| Not breastfed | 3323 (41.3) | 2362 (43.4) | 5685 (42.1) | 0.011 |
| Breastfed | 4733 (58.7) | 3075 (56.6) | 7808 (57.9) | |
| Child received IP drug | ||||
| No | 3615 (44.9) | 2363 (43.5) | 5978 (44.3) | 0.105 |
| Yes | 4441 (55.1) | 3074 (56.5) | 7515 (55.7) | |
| Place of delivery | ||||
| Home | 2257 (28.0) | 1383 (25.4) | 3640 (27.0) | 0.005 |
| Public Hospital | 1464 (18.2) | 1030 (18.9) | 2494 (18.5) | |
| Health Center | 3225 (40.0) | 2205 (40.6) | 5430 (40.2) | |
| Private Hospital | 1110 (13.8) | 819 (15.1) | 1929 (14.3) | |
| Toilet facility | ||||
| With slab | 2320 (28.8) | 1645 (30.3) | 3965(29.4) | 0.122 |
| Without slab | 4893 (60.7) | 3208 (59.0) | 8101(60.0) | |
| No facility | 843 (10.5) | 584 (10.7) | 1427 (10.6) | |
| Cooking fuel | ||||
| Wood | 6370 (79.1) | 4237 (77.9) | 10,607 (78.6) | 0.112 |
| Charcoal | 1686 (20.9) | 1200 (22.1) | 2886 (21.4) | |
| Drinking water source | ||||
| Protected | 6097 (75.7) | 4179 (76.9) | 10,276 (76.2) | 0.115 |
| Unprotected | 1959 (24.3) | 1258 (23.1) | 3217 (23.8) | |
| Season effect | ||||
| Dry | 3436 (42.6) | 2905 (53.4) | 6341 (47.0) | < 0.001 |
| Rainy | 4620 (57.4) | 2532 (46.6) | 7152 (53.0) | |
IP Intestinal Parasites
Comparison of method performances
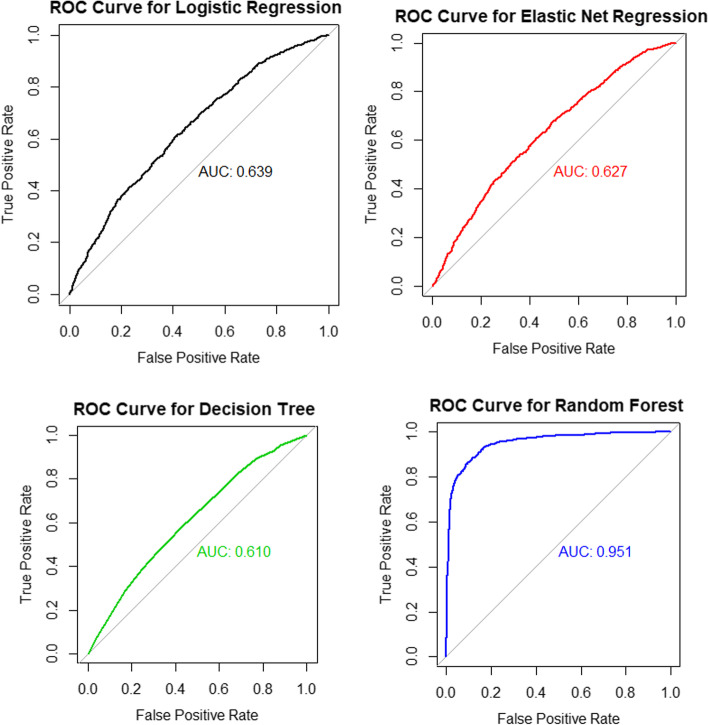
The scope of this study focused primarily on determining the potential risk factors of ARI disease symptoms based on well-performing methods. In the analysis, we used 75% of the total sample as a training sample and the remaining 25% for testing the method’s performance using ten-fold cross-validation. Table 3 shows the results of the performance comparisons between logistic regression (LR), elastic net logistic regression (EN), decision tree (DT), and random forest (RF) methods. The RF method showed the highest accuracy of 88.7 and 93.10% for precision in predicting the childhood ARI symptoms compared to other methods, i.e., about 88.7% of children who actually reported having or not having symptoms of ARI were correctly predicted by the RF method, while 93.1% of children with actual symptoms were also correctly predicted to have ARI symptoms. The LR method is followed with 62.0% accuracy and 88.03% precision. The other methods, such as the EN methods (61.7% accurate and 86.25% precise) and the DT method (61.2% accurate and 83.0% precise), showed the least performance in the prediction of ARI disease symptoms in this study. The AUC results for the receiver operating curves comparing these methods are presented in Fig. 3. Therefore, we used the random forest and logistic regression methods to determine potential risk factors for ARI disease symptoms among children under the age of 5 years in Uganda.
Table 3.
Comparison of predictive performances for the methods
| Method | Recall | Selectivity | Precision | Accuracy | AUC |
|---|---|---|---|---|---|
| Logistic regression (LR) | 63.05% | 57.04% | 88.03% | 62.05% | 0.638 |
| Elastic net regression (EN) | 63.14% | 55.47% | 86.25% | 61.73% | 0.627 |
| Decision tree (DT) | 63.34% | 53.33% | 82.97% | 61.16% | 0.610 |
| Random forest (RF) | 88.57% | 88.93% | 93.10% | 88.70% | 0.951 |
AUC area under the curve
Fig. 3.
ROC curves and AUC values for method performances
Potential risk factors contributing to the childhood ARI disease symptoms
Tables 4 and 5 summarize the logistic regression and random forest methods’ results. A subsample of 75% of all sampled children used in performance comparisons between these methods was used to determine the potential risk factors for ARI disease symptoms among children in Uganda. Adjusting for other factors, the LR model results (Table 4) reveal that children aged 12–23 months had a higher risk of 1.27 times (95% CI: 1.12–1.44) of developing ARI disease symptoms than infants, while children aged 48–59 months showed a low risk of 0.69 times (95% CI: 0.60–0.80) of ARI symptoms compared to children at earlier ages. Children living in other regions different from the central region had a low risk of developing ARI symptoms. Children of teen mothers had a significantly higher risk of having ARI symptoms than children of mothers in their middle ages (21–24 years old), and employed mothers in farming 1.25 times (95% CI: 1.11–1.42) or other employment 1.93 times (95% CI: 1.71–2.19) showed that their children were associated with a high risk of ARI disease symptoms compared to children of unemployed mothers who had enough time to care for them.
Table 4.
Logistic regression estimates of risk factors associated with childhood ARI symptoms in Uganda
| Factors | Frequency (%) | AOR | [95% CI] | Sig. |
|---|---|---|---|---|
| Socio-demographic factors | ||||
| Child age (months) | ||||
| 0–11 | 2131 (21.1) | Ref | – | – |
| 12–23 | 2070 (20.4) | 1.27 | [1.12–1.44] | < 0.001 |
| 24–35 | 2003 (19.8) | 0.98 | [0.85–1.12] | 0.727 |
| 36–47 | 1939 (19.2) | 0.88 | [0.76–1.01] | 0.065 |
| 48–59 | 1977 (19.5) | 0.69 | [0.60–0.80] | < 0.001 |
| Region of residence | ||||
| Central | 2064 (20.4) | Ref | – | – |
| Eastern | 2877 (28.4) | 0.63 | [0.56–0.72] | < 0.001 |
| Northern | 2535 (25.1) | 0.65 | [0.57–0.74] | < 0.001 |
| Western | 2644 (26.1) | 0.51 | [0.44–0.57] | < 0.001 |
| Mother age (years) | ||||
| 15–19 | 595 (5.9) | 1.28 | [1.06–1.53] | 0.009 |
| 20–24 | 2736 (27.0) | Ref | – | – |
| 25–29 | 2586 (25.6) | 1.07 | [0.95–1.19] | 0.268 |
| 30–34 | 2067 (20.4) | 0.94 | [0.83–1.06] | 0.313 |
| 35–39 | 1334 (13.2) | 0.96 | [0.84–1.11] | 0.600 |
| 40–49 | 802 (7.9) | 0.95 | [0.80–1.12] | 0.546 |
| Mother employment | ||||
| Unemployed | 1624 (16.1) | Ref | – | – |
| Farmer | 4780 (47.2) | 1.25 | [1.11–1.42] | < 0.001 |
| Other | 3716 (36.7) | 1.93 | [1.71–2.19] | < 0.001 |
| Behavioral and environmental factors | ||||
| Breastfeeding | ||||
| Not Breastfed | 4247 (42.0) | Ref | – | – |
| Breastfed | 5873 (58.0) | 0.83 | [0.76–0.92] | < 0.001 |
| Cooking energy | ||||
| Wood | 7955 (78.6) | Ref | – | – |
| Charcoal | 2165 (21.4) | 0.77 | [0.69–0.87] | < 0.001 |
| Season effect | ||||
| Dry | 4769 (47.1) | Ref | – | – |
| Rainy | 5351 (52.9) | 0.66 | [0.61–0.72] | < 0.001 |
Sig.: p-value at 5% level of significance
Table 5.
Potential risk factors contributing to the childhood ARI disease in both random forest and logistic regression methods
| Random Forest (RF) | Logistic Regression (LR) | |
|---|---|---|
| Mother employmenta | Region of residence | |
| Season effecta | Mother employment | |
| Region of residencea | Season effect | |
| Cooking energya | Child age | |
| Mother wealth status | Cooking energy | |
| Place of delivery | Breastfeeding | |
| Mother education | Mother age | |
| Accuracy | 88.70% | 62.05% |
aindicates factors in both methods
In behavioral and environmental factors, mothers who breastfed their children had a lower risk of 0.83 times (95% CI: 0.76–0.92) of ARI symptoms compared to those who did not breastfeed, and other factors such as the child’s exposure to charcoal cooking smoke showed a lower risk of 0.77 times (95% CI: 0.69–0.87) of developing ARI disease symptoms than children exposed to firewood smoke, while in the rainy season, children were less likely to 0.66 times (95% CI: 0.61–0.72) of developing symptoms of ARI disease than in the dry season in Uganda.
For the random forest method, the important factors contributing to the prediction of ARI disease symptoms were shown in Table 5, and risk factors such as mother’s employment, season effect, region of residence, cooking energy, mother’s wealth status, place of delivery, and mother’s education were potential risk factors contributing to the ARI disease symptoms among children under the age of 5 years in the random forest methods.
Discussion
This study builds upon the analysis of risk factors for ARI disease symptoms among children under the age of 5 years and compares various methods’ performances in predicting childhood ARI symptom outcomes. Using well-performing methods, we analyzed socio-demographic, behavioral, and environmental factors contributing to childhood ARI disease symptoms in Uganda using the 2016 UDHS dataset. The results revealed that the random forest method performed better in accuracy than other methods considered in the analysis, followed by the logistic regression method (Table 5). As shown in two methods, the employment of mothers in farming activities, the season effect, the region of residence, and the fuel used for cooking, such as firewood and charcoal, were found to be potential risk factors contributing to the childhood ARI disease symptoms in Uganda. In addition, the young ages of mothers and children, breastfeeding, and wealth status were also found to be factors associated with ARI disease symptoms among children in this study.
Other studies conducted in Uganda also showed that these higher prevalence results for childhood ARI diseases were consistent with the current findings [19, 20], and the high risk of childhood ARI disease symptoms due to factors such as season and geographical regions was also in concurrence with other findings from studies conducted in neighboring countries such as Rwanda and Kenya [21–23]. A vulnerable region in Uganda, like the northern region where people were forced to settle in camps because of the civil war in 1986, suffered from overcrowding and poor sanitation that speeded up the disease occurrence [19]. More efforts in sanitation and appropriate health services from the government should be established in highly risk areas to eliminate regional differences against ARI diseases. However, a study conducted in the Gulu district, northern Uganda, reported that children living in urban areas were more likely to develop ARI symptoms than those living in rural areas [24].
The study findings also revealed that the ARI symptoms increased among the children exposed to firewood smoke compared to those exposed to charcoal smoke. These results of the association between wood fuel and ARI symptoms were similar to others conducted in sub-Saharan African countries [23, 25–28]. According to WHO reports, “Children exposed to cooking fuels and parental smoking are more likely to be at a high risk of having pneumonia and other respiratory infection diseases” [8]. The need for parents’ and community education about the dangers of smoking to children must be addressed, especially in places where smoking and firewood are used frequently [29].
The ARI factors, such as the education and employment of mothers, are consistent with other results found in Kenya, Ethiopia, and Rwanda [22, 30, 31]. However, the factors contradict findings from another study conducted in northern Uganda because of the discrepancies in living standards and characteristics of the population studied. In the northern part, people suffered from overcrowding and poor sanitation, and most people were living in camps that encouraged disease occurrence and easy spread [19]. In the current study, children younger than 1 year old showed a higher risk of having ARI disease symptoms than children aged 48–59 months. These findings are supported by similar findings [29, 32–34]. The factors were related to the low rates of immunization in young children, low maternal literacy, and the young mothers in farming activities that do not allow the care of young children, particularly in sub-Saharan African countries, where health facilities and maternal healthcare education have to be improved.
Aside from the foregoing, this study provides evidence on parental behavior factors such as breastfeeding, which contradicts other findings [19, 20]. The current study showed that non-breastfed children whose mothers were teenaged were found to be more likely to develop ARI disease symptoms than breastfed ones, and generally, breastfeeding is more important to the child’s nutrition and the good functionality of the child’s immunity system.
Despite the strengths, limitations also have to be discussed. Parental smoking and childbirth weight factors were found to be significantly associated with ARI disease among children under the age of five in other studies [26, 29, 35, 36]. Due to the much missing information presented in these two variables in the current study, these two risk factors were limited in the 2016 UDHS dataset. In general, smoking harms the natural human defense of the respiratory system [37], especially in low birth-weight children. The government and community campaigns should educate people about the dangers of smoking on people’s health, particularly in young children’s households.
In terms of the analysis methods, we used both new and traditional supervised analysis methods, such as machine learning algorithms and multivariate regression methods, to predict the childhood outcomes of ARI disease symptoms. Furthermore, these findings complement other comparative machine learning findings [38–41] in providing evidence of the better performance of the random forest algorithm (88.7%) than traditional methods of analysis. However, other studies [42, 43] contradicted these findings. Further research is needed to overcome these challenges and compare various analysis methods using nationwide cross-sectional survey datasets like the DHS data. Moreover, longitudinal data analysis can better examine the potential risk factors of ARI disease in children under the age of 5 years.
In summary, this paper revealed that the mother’s employment and age, child age, breastfeeding, wealth status, season effect, region of residence, and cooking fuel such as firewood and charcoal were found to be potential risk factors for ARI disease symptoms in children under the age of 5 years. In this study, non-breastfed children whose mothers were teenagers had a significant effect on the development of ARI disease symptoms. Based on the results, policy-makers and health stakeholders should initiate target-oriented approaches to address the problems regarding poor children’s healthcare, improper environmental conditions, and childcare facilities. The government and child family interventions have to encourage maternal education and especially child breastfeeding. For the sake of early child care, the government should promote child breastfeeding and maternal education.
Supplementary Information
Additional file 1: Table 1. Descriptions of socio-economic and demographic characteristics. Table 2. Descriptions of behavioral and environmental characteristics.
Acknowledgments
Firstly, we express our sincere gratitude to the ICF Institutional Board and DHS protocol for its data provision and use permission. We also thank the East China Normal University advisory committee members for their continuous support to completion.
Abbreviations
- ARI
Acute Respiratory Infections
- UDHS
Uganda Demographic and Health Survey
- EA
Enumeration Area
- LR
Logistic Regression
- EN
Elastic Net Regression
- DT
Decision Tree
- RF
Random Forest
- AOR
Adjusted Odds Ratio
- CI
Confidence Interval
- TP
True Positive
- TN
True Negative
- FP
False Positive
- FN
False Negative
Authors’ contributions
YN: Analysed and extracted the data, interpreted results, and drafted the manuscript. YZ: Designed and proposed the study methodology, and supervised the whole study process. All authors have read and approved the final version of the manuscript.
Funding
This work was sponsored by National Natural Science Foundation of China (project number: 11771146, 11831008).
Availability of data and materials
The data used is openly available to the DHS Program after having access approval at https://www.dhsprogram.com/data/
Declarations
Ethics approval and consent to participate
The ICF Institutional Review Board in collaboration with the Uganda Bureau of Statistics was granted permission and assistance through The 2016 DHS protocol. Prior to enrollment, all participant mothers or guardians of a child who were eligible for the survey provided written informed consent for participation, and all data were gathered anonymously.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.UNDP . Measuring human development: a primer. New York: United Nations Development Programme (UNDP); 2007. [Google Scholar]
- 2.UNIGME . Levels trends in child mortality: report 2020, estimates developed by the United Nations inter-agency Group for Child Mortality Estimation. New York: United Nations inter-agency Group for Child Mortality Estimation (UNIGME); 2020. [Google Scholar]
- 3.WHO . The world health report 2003: shaping the future. Geneva: World Health Organization (WHO); 2003. [Google Scholar]
- 4.WHO . Infection prevention and control of epidemic- and pandemic-prone acute respiratory infections in health care. Geneva: World Health Organization (WHO); 2014. [PubMed] [Google Scholar]
- 5.WHO . The top 10 causes of death. World Health Organisation (WHO); 2020. [Google Scholar]
- 6.WHO . Diarrhoeal disease. World Health Organisation (WHO); 2017. [Google Scholar]
- 7.WHO et al. Ending preventable child deaths from pneumonia and diarrhoea by 2025: the integrated global action plan for pneumonia and diarrhoea (gappd) 2013. [DOI] [PubMed] [Google Scholar]
- 8.Wardlaw TM, Johansson EW, Hodge MJ. Pneumonia: the forgotten killer of children. Geneva: Unicef; 2006. [Google Scholar]
- 9.UBOS, ICF . Uganda demographic and health survey 2016. Kampala: Demographic and Health Survey (DHS) & Uganda Bureau of Statistics (UBOS); 2018. [Google Scholar]
- 10.Sridevi Radhakrishnan DD. A critical study on data mining techniques in healthcare dataset. 2015. [Google Scholar]
- 11.Kirubha V, Priya SM. Survey on data mining algorithms in disease prediction. Int J Comput Trends Technol. 2016;38(3):124–128. doi: 10.14445/22312803/IJCTT-V38P122. [DOI] [Google Scholar]
- 12.Uddin S, Khan A, Hossain ME, Moni MA. Comparing different supervised machine learning algorithms for disease prediction. BMC Med Inform Decis Making. 2019;19(1):1–16. doi: 10.1186/s12911-019-1004-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hosmer DW, Jr, Lemeshow S, Sturdivant RX. Applied logistic regression. New Jersey: Wiley; 2013. [Google Scholar]
- 14.Zou H, Hastie T. Regularization and variable selection via the elastic net. J R Stat Soc Series B Stat Methodol. 2005;67(2):301–320. doi: 10.1111/j.1467-9868.2005.00503.x. [DOI] [Google Scholar]
- 15.Quinlan JR. Induction of decision trees. Mach Learn. 1986;1(1):81–106. [Google Scholar]
- 16.Breiman L. Random forests. Mach Learn. 2001;45(1):5–32. doi: 10.1023/A:1010933404324. [DOI] [Google Scholar]
- 17.Kourou K, Exarchos TP, Exarchos KP, Karamouzis MV, Fotiadis DI. Machine learning applications in cancer prognosis and prediction. Comput Struct Biotechnol J. 2015;13:8–17. doi: 10.1016/j.csbj.2014.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Spencer R, Thabtah F, Abdelhamid N, Thompson M. Exploring feature selection and classification methods for predicting heart disease. Digit Health. 2020;6:2055207620914777. doi: 10.1177/2055207620914777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bbaale E. Determinants of diarrhoea and acute respiratory infection among under-fves in Uganda. Australas Med J. 2011;4(7):400. doi: 10.4066/AMJ.2011.723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cummings MJ, Bakamutumaho B, Kayiwa J, Byaruhanga T, Owor N, Namagambo B, Wolf A, Wamala JF, Morse SS, Lutwama JJ, et al. Epidemiologic and spatiotemporal characterization of influenza and severe acute respiratory infection in Uganda, 2010-2015. Ann Am Thorac Soc. 2016;13(12):2159–2168. doi: 10.1513/AnnalsATS.201607-561OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Murray E, Klein M, Brondi L, McGowan J, Van Mels C, Brooks WA, Kleinbaum D, Goswami D, Ryan P, Bridges C. Rainfall, household crowding, and acute respiratory infections in the tropics. Epidemiol Infect. 2012;140(1):78–86. doi: 10.1017/S0950268811000252. [DOI] [PubMed] [Google Scholar]
- 22.Harerimana J-M, Nyirazinyoye L, Thomson DR, Ntaganira J. Social, economic and environmental risk factors for acute lower respiratory infections among children under five years of age in Rwanda. Arch Public Health. 2016;74(1):1–7. doi: 10.1186/s13690-016-0132-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ramani VK, Pattankar J, Puttahonnappa SK. Acute respiratory infections among under-five age group children at urban slums of Gulbarga city: a longitudinal study. J Clin Diagn Res. 2016;10(5):08. doi: 10.7860/JCDR/2016/15509.7779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lanyero H, Eriksen J, Obua C, Stålsby Lundborg C, Nanzigu S, Katureebe A, Kalyango JN, Ocan M. Use of antibacterials in the management of symptoms of acute respiratory tract infections among children under _ve years in Gulu, northern Uganda: prevalence and determinants. PLoS One. 2020;15(6):0235164. doi: 10.1371/journal.pone.0235164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mathew JL, Patwari AK, Gupta P, Shah D, Gera T, Gogia S, Mohan P, Panda R, Menon S. Acute respiratory infection and pneumonia in India: a systematic review of literature for advocacy and action: Unicef-ph_ series on newborn and child health, India. Indian Pediatr. 2011;48(3):191. doi: 10.1007/s13312-011-0051-8. [DOI] [PubMed] [Google Scholar]
- 26.WHO . Acute respiratory infections in children: case management in small hospitals in developing countries, a manual for doctors and other senior health workers. Technical report. Geneva: World Health Organization; 1990. [Google Scholar]
- 27.Jackson S, Mathews KH, Pulanić D, Falconer R, Rudan I, Campbell H, Nair H. Risk factors for severe acute lower respiratory infections in children_a systematic review and meta-analysis. Croat Med J. 2013;54(2):110–121. doi: 10.3325/cmj.2013.54.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Buchner H, Rehfuess EA. Cooking and season as risk factors for acute lower respiratory infections in African children: a cross-sectional multi-country analysis. PLoS One. 2015;10(6):0128933. doi: 10.1371/journal.pone.0128933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tazinya AA, Halle-Ekane GE, Mbuagbaw LT, Abanda M, Atashili J, Obama MT. Risk factors for acute respiratory infections in children under-five years attending the Bamenda regional hospital in Cameroon. BMC Pulm Med. 2018;18(1):1–8. doi: 10.1186/s12890-018-0579-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fekadu GA, Terefe MW, Alemie GA. Prevalence of pneumonia among under-five children in Este town and the surrounding rural kebeles, Northwest Ethiopia: a community-based cross-sectional study. Sci J Public Health. 2014;2(3):150–155. doi: 10.11648/j.sjph.20140203.12. [DOI] [Google Scholar]
- 31.Onyango D, Kikuvi G, Amukoye E, Omolo J. Risk factors of severe pneumonia among children aged 2-59 months in western Kenya: a case control study. Pan Afr Med J. 2012;13(1):45. [PMC free article] [PubMed] [Google Scholar]
- 32.Banda B, Mazaba M, Mulenga D, Siziya S. Risk factors associated with acute respiratory infections among under-five children admitted to Arthur’s children’s hospital, Ndola, Zambia. J Health Sci. 2016;3:153–159. [Google Scholar]
- 33.Geberetsadik A, Worku A, Berhane Y. Factors associated with acute respiratory infection in children under the age of 5 years: evidence from the 2011 Ethiopia demographic and health survey. Pediatr Health Med Ther. 2015;6:9. doi: 10.2147/PHMT.S77915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Alemayehu S, Kidanu K, Kahsay T, Kassa M. Risk factors of acute respiratory infections among under five children attending public hospitals in southern Tigray, Ethiopia, 2016/2017. BMC Pediatr. 2019;19(1):1–8. doi: 10.1186/s12887-019-1767-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ujunwa F, Ezeonu C. Risk factors for acute respiratory tract infections in under-five children in Enugu Southeast Nigeria. Ann Med Health Sci Res. 2014;4(1):95–99. doi: 10.4103/2141-9248.126610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Arcavi L, Benowitz NL. Cigarette smoking and infection. Arch Intern Med. 2004;164(20):2206–2216. doi: 10.1001/archinte.164.20.2206. [DOI] [PubMed] [Google Scholar]
- 37.Valencia-Gattas M, Conner GE, Fregien NL. Ge_tinib, an egfr tyrosine kinase inhibitor, prevents smoke-mediated ciliated airway epithelial cell loss and promotes their recovery. PLoS One. 2016;11(8):0160216. doi: 10.1371/journal.pone.0160216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bihter D. A comparative study on the performance of classi_cation algorithms for effective diagnosis of most liver diseases. Sakarya Univ J Comput Inform Sci. 2020;3(3):366–375. [Google Scholar]
- 39.Pathan A, Mhaske D, Jadhav S, Bhondave R, Rajeswari K. Comparative study of di_erent classi_cation algorithms on ilpd dataset to predict liver disorder. Int J Res Appl Sci Eng Technol. 2018;6(2):388–394. doi: 10.22214/ijraset.2018.2056. [DOI] [Google Scholar]
- 40.Mani S, Chen Y, Elasy T, Clayton W, Denny J. AMIA Annual Symposium Proceedings. American Medical Informatics Association; 2012. Type 2 diabetes risk forecasting from emr data using machine learning; p. 606. [PMC free article] [PubMed] [Google Scholar]
- 41.Marikani T, Shyamala K. Prediction of heart disease using supervised learning algorithms. Int J Comput Appl. 2017;165(5):41–44. [Google Scholar]
- 42.Islam MM, Wu C-C, Poly TN, Yang H-C, Li Y-CJ. Applications of machine learning in fatty live disease prediction. MIE; 2018. pp. 166–170. [PubMed] [Google Scholar]
- 43.Tapak L, Mahjub H, Hamidi O, Poorolajal J. Real-data comparison of data mining methods in prediction of diabetes in Iran. Healthc Inform Res. 2013;19(3):177. doi: 10.4258/hir.2013.19.3.177. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table 1. Descriptions of socio-economic and demographic characteristics. Table 2. Descriptions of behavioral and environmental characteristics.
Data Availability Statement
The data used is openly available to the DHS Program after having access approval at https://www.dhsprogram.com/data/