Abstract
Aims
Adjuvant chemotherapy (ACT) plays an important role in improving the survival of stage II-III colorectal cancer (CRC) patients after curative surgery. However, the prognostic role of irregular delay of ACT (IDacT) for these patients has been less studied.
Materials and methods
A total of 117 stage II-III CRC patients who underwent radical resection and received at least 3 months ACT were enrolled retrospectively. The significance of IDacT, including total delay (TD) and delay per cycle (DpC), in predicting disease-free survival (DFS) was determined using receiver operating characteristic curve (ROC) analysis. The survival differences between the TD, DpC-short and DpC-long subgroups were tested using Kaplan–Meier analysis, and risk factors for prognosis were determined using a Cox proportional hazards model.
Results
Using 35.50 and 3.27 days as the optimal cut-off points for TD and DpC, respectively, ROC analysis revealed that TD and DpC had sensitivities of 43.60% and 59.00% and specificities of 83.30% and 62.80%, respectively, in predicting DFS (both P < 0.05). No differences in the clinicopathological parameters were found between the TD, DpC-short or -long subgroups except histological differentiation in different TD subgroups and combined T stages in different DpC subgroups (both P = 0.04). Patients in the TD or DpC-long group exhibited significantly worse survival than in the -short group (TD: Log rank = 9.11, P < 0.01; DpC: Log rank = 6.09, P = 0.01). DpC was an independent risk factor for prognosis (HR = 2.54, 95% CI: 1.32–4.88, P = 0.01).
Conclusions
IDacT had a profound effect on the outcome for stage II-III CRC. Although TD and DpC were significant for the prognosis, DpC was more robust, and patients who presented DpC for a long time had a significantly worse DFS.
Keywords: Colorectal cancer, Adjuvant chemotherapy, Delay, Disease-free survival
Introduction
Adjuvant chemotherapy (ACT) improves survival in stage II-III colorectal cancer (CRC) patients, and the efficacy of chemotherapy regimens based on oxaliplatin, such as XELOX (oxalipaltin + capecitabine) and FOLFOX (oxalipaltin + leucovorin + 5-fluorouracil), were extensively validated in a series of clinical trials [1–3]. Six months (m) of ACT was the standard of treatment for these patients, but a shortened duration of ACT of 3 m was acceptable for both disease-free survival (DFS) and overall survival (OS) for certain cases due to the significant adverse effects (AEs) of chemotherapy, as validated in recent studies [2–7]. However, many factors could impair the benefits of ACT in practice.
Delayed ACT (DacT) conventionally refers to the interval between the points of surgery and the initiation of ACT, which is common in the clinic. DacT has been documented in many malignancies, including gastric cancer [8, 9], breast cancer [10, 11], lung cancer [12] and CRC [13–15]. Notably, DacT consistently correlated with poor outcome in these studies [8–15]. Kim et al. investigated the impact of DacT (defined as ≥ 8 weeks (w)) on survival in stage II-III CRC and found that DacT was associated with inferior OS [13]. Becerra et al. used a similar cut-off point to examine the impact of DacT in stage III cases on DFS, and the results indicated that DacT was correlated with poor DFS [14]. The underlying causes for DacT are complicated and include patient factors (e.g., sex, age, comorbidity, postoperative anxiety, and depression), insurance status, socioeconomic status, and treatment-related factors (e.g., emergency surgery, reoperation, and prolonged hospital stay) [11, 13, 16–18]. However, DacT also occurred during the prescribed ACT with high probability due to additional cause-AEs of chemotherapeutic drugs [16]. Although the specific rate of DacT during treatment is largely unknown, those of grade 3 or 4 AEs were 37.6% and 24.2% in the 3-m arms of the FOLFOX and XELOX regimens, respectively, and increased to 56.9% and 36.9% in the 6-m arms according to IDEA research [19]. These AEs resulted in a high probability of dose adjustment and DacT in practice. Notably, the DacT during these treatments may be highly irregular due to the different recovery capabilities of the patients. However, whether this irregular DacT (IDacT) affected the survival of the patients was not addressed.
The present study examined the effect of IDacT, including total delay (TD) and delay per cycle (DpC), on the outcome in stage II-III CRC.
Materials and methods
Patient enrollment
CRC patients treated in Hainan Hospital of Chinese PLA General Hospital were retrospectively enrolled from December 2012 to December 2021. Patients were included if they met the following criteria: 1. curative resection of the primary lesion; 2. pathologically confirmed as stage II-III (according to the 8th edition of American Joint Committee on Cancer) with no suspicious distant metastasis; and 3. a clear ACT record (at least 4 cycles ACT based on XELOX or single capecitabine (X), or at least 6 cycles ACT based on FOLFOX). Patients who met any of the following criteria were excluded: 1. receiving any preoperative neoadjuvant therapies; and 2. loss or refusal to follow-up or a follow-up period less than 36 m. The risk factors, including lymphovascular invasion, histopathological poorly differentiated, T4 or obstruction [20], and other clinicopathological information, were collected as previously described [21, 22]. The study was conducted in accordance with the principles stated in the Declaration of Helsinki and was approved by the ethics committee of Hainan Hospital of Chinese PLA General Hospital (ID: 301HLFYLS15), and informed consent was obtained from the patients or their authorized relatives.
Definition of TD, DpC and DFS
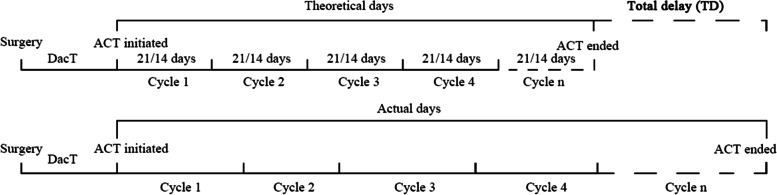
The TD was calculated as the sum of the actual days minus the theoretical days from the initiation of ACT to the first day of the last cycle. The theoretical number of days for which patients underwent regimens such as XELOX or single X was counted every 21 days (d), and it was counted every 14 d for patients receiving FOLFOX (Fig. 1). The days from the day of surgery to the initiation of ACT were not included in the TD calculation. After calculating the TD, the DpC was determined as the TD divided by the actual ACT cycles. DFS was defined from the time of surgery to the date of any recurrence or metastasis or the date of death from any cause. The follow-up was performed as described in a previous report [22], and the latest follow-up point was December 2021.
Fig. 1.
Methods to estimate the total delay
Statistical analysis
Statistical analyses were performed using SPSS 20.0 (SPSS Inc., Chicago, IL, USA). The optimal cut-off points of TD and DpC were calculated using receiver operating characteristic (ROC) curve analysis for DFS. Patients were divided into TD, DpC-short or -long subgroups according to the Youden index. The differences in the collected clinicopathological parameters between these subgroups were estimated using the χ2-test. Survival differences between these subgroups were assessed using Kaplan–Meier (K-M) analysis followed by the log rank tests. Risk factors for the outcome were calculated using a Cox proportional hazards model. A double-sided P < 0.05 was considered statistically significant.
Results
Demographic features of the cohort
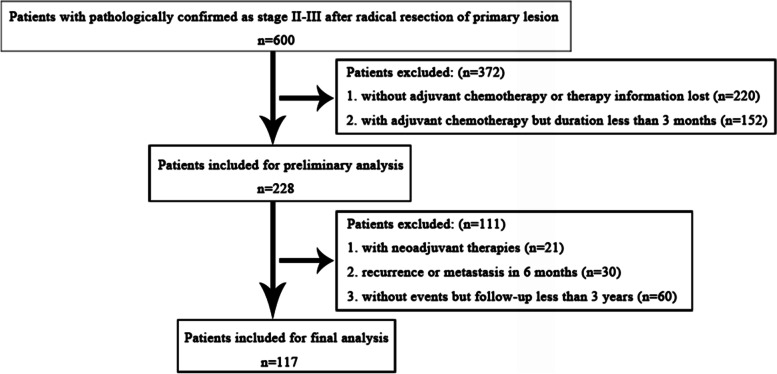
Following the inclusion and exclusion criteria, 117 patients were enrolled for the final analysis (Fig. 2). The basic demographic characteristics are shown in Table 1. Briefly, there were 30 stage II (with 16 cases accompanied at least 1 risk factor) and 87 stage III (with 43 cases accompanied at least 1 risk factor) patients. The mean TD and DpC were 24.18 d (range: 0–117 d) and 3.74 days (range: 0–16.60 d), respectively. Five patients presented a conventional DacT (defined as ≥ 8 w) of 61 d (2 cases), 65 d, 66 d and 70 d (1 case each), and the rest of the cases did not exhibit a delay (mean time from surgery to initiation of ACT: 32.60 d, range: 7–79 d). The mean age of the patients was 53.44 years (y) (range: 24–80 y), and the mean follow-up time was 41.80 m (range: 3–60 m). At the end of the follow-up, 39 events occurred, and the 3-year DFS rate was 66.67% (90.00% for stage II and 58.60% for stage III).
Fig. 2.
Flowchart of case inclusion and exclusion
Table 1.
Basic demographic characteristic of the study
| Parameters | Cases (n/%) |
|---|---|
| Age (y) | |
| < 60 | 81 (69.23) |
| ≥ 60 | 36 (30.67) |
| Sex | |
| Male | 77 (65.81) |
| Female | 40 (34.19) |
| Tumor location | |
| Right | 33 (28.21) |
| Left | 84 (71.79) |
| Tumor morphology | |
| Ulcerated type | 57 (48.72) |
| Protruded type | 21 (17.95) |
| Mixed type | 6 (5.13) |
| Unknown | 33 (28.21) |
| Histological differentiation | |
| Well + moderate | 92 (78.63) |
| Poor | 25 (21.37) |
| Mucinous element | |
| With | 18 (15.39) |
| Without | 99 (84.61) |
| Combined T stages | |
| T1 + T2 + T3 | 94 (80.34) |
| T4 | 23 (19.66) |
| Combined N stages | |
| N0 | 30 (25.64) |
| N1+N2 | 87 (74.32) |
| TNM stages | |
| II | 30 (25.64) |
| III | 87 (74.32) |
| Tumor deposits | |
| With | 20 (17.09) |
| Without | 97 (82.91) |
| RAS mutation | |
| Mutated | 6 (5.13) |
| Wild-type | 11 (9.40) |
| Unknown | 100 (85.47) |
| MSS status | |
| MSI-H | 3 (2.56) |
| MSI-L + MSS | 66 (56.41) |
| Unknown | 48 (41.03) |
| Ki-67 percentage | |
| ≥ 75% | 41 (35.04) |
| 50–75% | 14 (11.97) |
| 0–50% | 7 (5.98) |
| Unknown | 55 (47.01) |
| Risk factors | |
| None | 58 (49.57) |
| 1 | 34 (29.06) |
| 2 | 15 (12.82) |
| 3 | 9 (7.69) |
| 4 | 1 (0.86) |
| Adjuvant therapy regimens | |
| XELOX | 71 (60.83) |
| FOLFOX | 11 (9.40) |
| XELOX + X | 17 (14.53) |
| FOLFOX + X | 1 (0.85) |
| XELOX + FOLFOX | 8 (6.84) |
| X | 1 (0.85) |
| Others | 8 (6.70) |
Predicting the efficacy of TD and DpC for survival and differences in clinicopathological parameters between subgroups
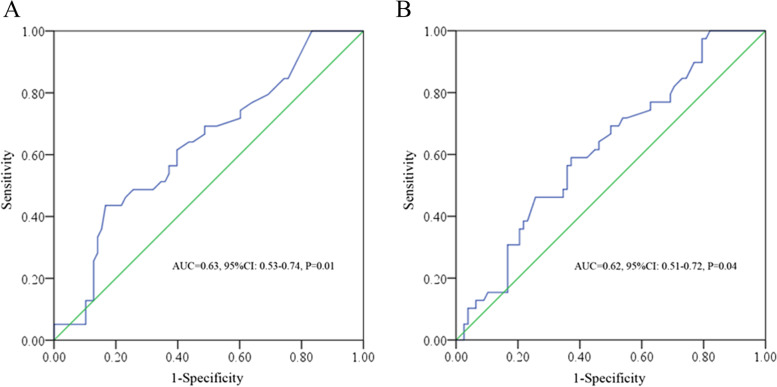
The Youden index from the ROC tests described 35.50 and 3.27 d as the optimal cut-off points for TD and DpC, respectively. The TD and DpC had a sensitivity of 43.60% and 59.00% and a specificity of 83.30% and 62.80% in predicting DFS (the areas under the curve (AUC) = 0.63, P = 0.01; AUC = 0.62, P = 0.04, respectively) (Fig. 3). Patients were divided into TD-short (< 35.50 d) or TD-long (≥ 35.50 d), DpC-short (< 3.27 d) or DpC-long (≥ 3.27 d) subgroups. Notably, no significant differences were found for the collected clinicopathological parameters between these subgroups except histological differentiation in different TD subgroups and combined T stages in different DpC subgroups (Table 2).
Fig. 3.
ROC analysis of TD (A) and DpC (B) in predicting DFS
Table 2.
Differences of clinicopathological parameters among TD, DpC-short and -long subgroups
| TD | DpC | |||||
|---|---|---|---|---|---|---|
| Short | Long | P | Short | Long | P | |
| Age (y) | 0.37 | 0.84 | ||||
| < 60 | 58 | 23 | 45 | 36 | ||
| ≥ 60 | 29 | 7 | 19 | 17 | ||
| Sex | 0.27 | 0.17 | ||||
| Male | 60 | 17 | 46 | 31 | ||
| Female | 27 | 13 | 18 | 22 | ||
| Tumor location | 1.00 | 1.00 | ||||
| Right | 25 | 8 | 18 | 15 | ||
| Left | 62 | 22 | 46 | 38 | ||
| Tumor morphology | 0.26 | 0.23 | ||||
| Ulcerated type | 44 | 13 | 36 | 21 | ||
| Protruded type | 12 | 9 | 9 | 12 | ||
| Mixed type | 5 | 1 | 4 | 2 | ||
| Unknown | 26 | 7 | 15 | 18 | ||
| Histological differentiation | 0.04* | 0.37 | ||||
| Well + moderate | 64 | 28 | 48 | 44 | ||
| Poor | 23 | 2 | 16 | 9 | ||
| Mucinous element | 0.08 | 0.80 | ||||
| With | 10 | 8 | 9 | 9 | ||
| Without | 77 | 22 | 55 | 44 | ||
| Combined T stages | 0.11 | 0.04* | ||||
| T1 + T2 + T3 | 73 | 21 | 56 | 38 | ||
| T4 | 14 | 9 | 8 | 15 | ||
| Combined N stages | 0.81 | 0.84 | ||||
| N0 | 23 | 7 | 17 | 13 | ||
| N1+N2 | 64 | 23 | 47 | 40 | ||
| TNM stages | 0.81 | 0.84 | ||||
| II | 23 | 7 | 17 | 13 | ||
| III | 64 | 23 | 47 | 40 | ||
| Tumor deposits | 0.78 | 0.63 | ||||
| With | 14 | 6 | 12 | 8 | ||
| Without | 73 | 24 | 52 | 45 | ||
| Risk factors | 0.14 | 1.00 | ||||
| None | 40 | 19 | 32 | 27 | ||
| ≥ 1 | 47 | 11 | 32 | 26 | ||
*with significant difference
Survival differences between TD and DpC subgroups
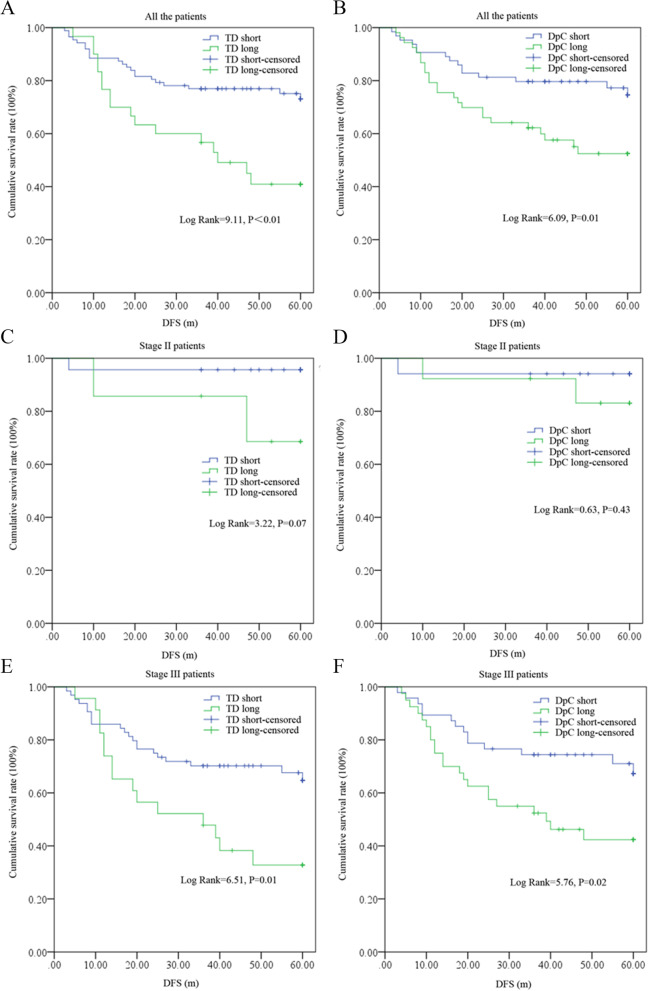
K-M analysis showed significant differences in survival between the TD, DpC-short and -long subgroups, and patients in the TD (Log rank = 9.11, P < 0.01) or DpC (Log rank = 6.09, P = 0.01)-long groups exhibited significantly inferior survival compared with that of the -short groups. Further, when stage II and III cases were analyzed individually, stage III patients presented a consistent statistical differences in survival among TD, DpC-short and -long subgroups; however, no such differences were found in stage II cases but with a trend of difference only in TD-short and -long subgroups (Fig. 4).
Fig. 4.
Survival differences between TD, DpC-short or -long subgroups. A TD in all the patients; B DpC in all the patients; C TD in stage II patients; D DpC in stage II patients; ETD in stage III patients; F DpC in stage III patients
Univariate and multivariate tests of risk factors for prognosis
Univariate tests found that the combined N stage, TNM stage, tumor deposits, TD and DpC were risk factors for prognosis, and when these parameters were included in multivariate tests, DpC was an independent risk factor for prognosis (HR = 2.54, 95% CI: 1.332–4.88, P = 0.01) (Table 3).
Table 3.
Univariate and multivariate analyses of different parameters for DFS
| Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|
| P | HR | 95%CI | P | HR | 95%CI | |
| Age (years) | ||||||
| < 60 | 1 | |||||
| ≥ 60 | 0.19 | 1.54 | 0.81–2.95 | |||
| Sex | ||||||
| Male | 1 | |||||
| Female | 0.08 | 0.57 | 0.30–1.07 | |||
| Tumor location | ||||||
| Right | 1 | |||||
| Left | 0.50 | 0.78 | 0.37–1.63 | |||
| Tumor morphology | ||||||
| Ulcerated type | 1 | |||||
| Protruded type | 0.34 | 1.47 | 0.67–3.28 | |||
| Mixed type | 0.38 | 1.73 | 0.51–5.89 | |||
| Unknown | 0.76 | 0.88 | 0.40–1.96 | |||
| Histological differentiation | ||||||
| Well + moderate | 1 | |||||
| Poor | 0.07 | 2.58 | 0.92–7.26 | |||
| Mucinous element | ||||||
| With | 1 | |||||
| Without | 0.24 | 0.63 | 0.29–1.37 | |||
| T stages | ||||||
| T1 + T2 + T3 | 1 | |||||
| T4 | 0.72 | 1.16 | 0.51–2.63 | |||
| N stages | ||||||
| N0 | 1 | 1 | ||||
| N1+ N2 | 0.01* | 5.07 | 1.56–16.51 | 0.01* | 4.84 | 1.48–15.81 |
| TNM stages | ||||||
| II | 1 | |||||
| III | 0.01* | 5.08 | 1.56–16.51 | |||
| Tumor deposits | ||||||
| With | 1 | 1 | ||||
| Without | 0.01* | 0.27 | 0.14–0.52 | < 0.01* | 0.26 | 0.13–0.51 |
| Risk factors | ||||||
| None | 1 | |||||
| ≥ 1 | 0.54 | 1.22 | 0.65–2.30 | |||
| TD | ||||||
| TD-short | 1 | |||||
| TD-long | < 0.01* | 2.56 | 1.36–4.83 | |||
| DpC | ||||||
| DpC-short | 1 | 1 | ||||
| DpC-long | 0.02* | 2.20 | 1.15–4.20 | 0.01* | 2.54 | 1.32–4.88 |
*with significant difference
TD total delay, DpC delay per cycle
Discussion
The present study demonstrated an obvious IDacT during the treatment of stage II-III CRC patients, and TD and DpC were useful in predicting DFS with relatively high specificities. Patients who presented a long TD or DpC had worse survival than those who presented a short TD or DpC, in particular, in stage III cases. Additionally, the DpC was likely to be a more robust indicator when compared to TD and it was an independent risk factor. To the best of our knowledge, this study is the first report to address the prognostic role of IDacT in CRC.
DacT impairs the outcome in cancer patients, but most studies focused on the period from surgery to the initiation of ACT. Lu et al. performed a systematic review and meta-analysis of 6107 gastric cancer patients after curative resection and found that the initiation of ACT per 4-w delay correlated with significant decreases in DFS and OS [8]. Chen et al. investigated 1520 stage II-III gastric cancer patients and found that patients with DacT (defined as commencement of ACT more than 60 d after surgery) also had significantly worse DFS and OS [9]; additionally, patients in the DacT group were less likely to benefit from ACT [9]. Although the definition of DacT was not consistent in CRC, its role in prognosis was not surprising. For example, Cheung et al. enrolled 5617 stage II-III CRC patients and indicated that DacT (defined as ≥ 3 m) predicted poor OS [23]. Bayraktar et al. performed a study of 186 stage II-III CRC patients and suggested that DacT (defined as ≥ 60 d) was associated with worse OS [24]. Most subsequent studies used 8 w as the cut-off point for DacT and found that it correlated with an inferior DFS or OS in stage II-III cases [13, 14, 25]. The underlying causes of DacT have been extensively studied and include a series of aforementioned factors [11, 13, 16–18]. However, taking into consideration the high probability of grade 3 or 4 AEs during chemotherapy [19], a substantial proportion of patients would experience IDacT in addition to these causes, as demonstrated in DacT. Although no previous studies indicated that IDacT would also impair the outcome of the patients, the consolidated results of DacT in prognosis support such a role of IDacT to some extent because they shared similar causes in practice. When using 8 w as the cut-off point, patients with DacT who completed the prescribed ACT were subsequently regarded as TD long to some extent in our study (although no delay occurred during ACT). When these patients (n = 5) were included in the analysis following this assumption, the AUC of TD in predicting DFS was even larger (AUC = 0.64, P = 0.02, data not shown) in our study.
The underlying mechanisms by which DacT could impair the outcome are largely unknown and commonly attributed to its decreased killing efficacy for micrometastasis. However, the mechanisms of DacT and IDacT may not be identical because chemotherapeutic drugs have an additional profound effect on cancer cells and patients. From this point of review, we speculated that IDacT would impair the outcome for the following reasons. First, it was found that as many as 19.40% of breast cancer patients who underwent ACT could develop hyperglycemia and exhibited a poor DFS [26]; accordingly, the serum insulin level was observed to experience an elevation [27]. In CRC, it was found that 5-fluorouracil (5-Fu) chemotherapy resulted in an elevation of fasting glucose levels [28], which could correspondingly lead to an increase in insulin secretion. Notably, insulin plays multiple roles in the development of CRC. For example, studies have revealed that it can lead to oxaliplatin resistance via activation of the PI3K/Akt pathway in HT-29 cells [29, 30] and contribute to cancer cell progression via the upregulation of ACAT1 [31]; additionally, it can increase proliferation and migration of HCT-116 cells [32]. Based on these results, patients who presented with IDacT were likely to have a high level of serum insulin that could promote disease development, and the residual cancer cells may become refractory to chemotherapeutic drugs due to the delayed administration of the next cycle of treatment. Second, patients with IDacT had a high probability of dose adjustment due to AEs [19]. Although one study indicated that FOLFOX regimens with different doses of 5-Fu as ACT in CRC showed similar efficiency [33], a dose intensity reduction in these regimens could potentially correlate with an attenuated killing effect for the cancer cells and was associated with impaired survival [34]. Third, patients with IDacT were found to exhibit a slow recovery due to various chemotherapy-related hematological toxicities (CRHT). Although no relationship between chemotherapy-associated neutrophil counts and CRC recurrence was found [35], chemotherapy-induced lymphopenia was a validated risk factor for febrile neutropenia and could lead to shorter DFS (< 0.66 × 109/L) and OS (< 0.91 × 109/L) [35] or early death in CRC [36]. In addition, chemotherapy-induced anemia (CIA) was also not uncommon in practice. It was found that anemia (which contributes to tumor hypoxia) can not only lead to chemotherapy resistance but also increase the invasiveness and metastatic potential and decrease the apoptosis of cancer cells [37]. A study indicated that CIA (< 90 g/L) was an independent prognostic factor for both DFS and OS in CRC patients receiving ACT [38]. Based on these reports, it was plausible that patients with IDacT caused by the above reasons would have a poor outcome.
Noticeably, although XELOX and FOLFOX exhibited similar efficacy in ACT [19, 39], the rate of severe AEs and the patients’ cost were obviously higher in FOLFOX than XELOX [19, 39–41]. Moreover, as the XELOX was redelivered every 21 d, and the FOLFOX regimen was redelivered every 14 d [19], we speculated that the XELOX regimen would be the preferred regimen for ACT based on its relatively mild AEs, cost and the lower frequency for chemotherapy booking. In addition, as an increased IDacT resulted in poor survival, some approaches could be used to minimize its impact on survival or decrease its incidence based on our aforementioned reasons. For example, metformin (a classical agent for the treatment of diabetes mellitus) could be used to control the fluctuation of glucose and it was also found to be effective in reversing insulin-induced oxaliplatin resistance in human colon cancer HCT-116 cells [42]; and clinically, metformin was demonstrated to be helpful in improving OS in stage II-III CRC in adjuvant settings [43, 44]. In addition, for CRHT, a recent study indicated that administration of pegfilgrastim on the same day as capecitabine-based chemotherapy in gastrointestinal cancers was safe, as it did not require dose reductions or cause a chemotherapy delay [45]. However, it is also noteworthy that certain causes, such as CIA, are still difficult to manage, irrespective that red blood cell transfusion, erythropoietin-stimulating agents (ESAs) and intravenous iron have been extensively studied in the clinic [46]. Although these approaches were found to play an important role in improving the quality of life of patients who underwent chemotherapy, approaches including blood transfusion or intravenous iron were not helpful in improving the outcome [47, 48]. For ESAs, it was found that approximately 1/3 of the patients did not respond to the treatment alone [49]; moreover, responses were not observed until 4–6 w after therapy initiation even in the responding ones [50]. Based on these results, a tailored therapeutic strategy is of pivotal importance for patients presenting with IDacT caused by CIA. Interestingly, in previous studies, a 12-m duration of single capecitabine ACT was found to yield better DFS and OS results than a 6-m duration in selected patients [51]; and a reduced dosage of oxaliplatin in the FOLFOX regimen for ACT had no obvious impact on DFS and OS [52]. Thus, we speculate that a duration extension with a dosage reduction may be acceptable for these patients. Nonetheless, these speculations need to be validated in prospective studies in the future.
There are some limitations to our study. First, it was a retrospective study that was performed in a single medical center, and the small sample size could lead to biased findings. Second, stage II cases (n = 14) without any of the risk factors were also included in our study; although previous studies have shown that these patients would also benefit from ACT [53–55], a standardized ACT and duration were not well established for such cases [56]. Third, 20 patients with at least one tumor deposit were included (II: n = 2, III: n = 18). Some studies indicated that stage I-III cases with these features would behave similarly to stage IV patients and would have no DFS benefits from ACT, particularly in stage III cases [57, 58]. Based on these studies, patients with these features would exhibit no differences in DFS regardless of the IDacT in our cohort. However, larger sample studies with restricted inclusion criteria would overcome these limitations in the future.
Conclusion
Overall, our study indicated that IDacT impaired survival in stage II-III CRC patients. Although TD and DpC were significant in predicting survival, DpC was an independent prognostic factor, and patients who experienced DpC for a long period had significantly worse DFS.
Acknowledgements
None.
Abbreviations
- ACT
Adjuvant chemotherapy
- CRC
Colorectal cancer
- IDacT
Irregular delay of adjuvant chemotherapy
- TD
Total delay
- DpC
Delay per cycle
- DFS
Disease free survival
- ROC
Receiver operating characteristic curve
- XELOX
Oxalipaltin + capecitabine
- FOLFOX
Oxalipaltin + leucovorin + 5-fluorouracil
- DFS
Disease free survival
- OS
Overall survival
- AEs
Adverse effects
- K-M
Kaplan–Meier
- 5-Fu
5-Fluorouracil
- CRHT
Chemotherapy-related hematological toxicities
- CIA
Chemotherapy-induced anemia
- ESAs
Erythropoietin-stimulating agents
Authors’ contributions
BY was responsible for the conception of the work. YC, MX, QY, JX, TX, and TY obtained the data. TY, LL and BY analyzed the data. YC, MX, QY and BY wrote the manuscript. TY, LL and BY critically revised the manuscript. All authors are accountable for the contents of this work. The authors read and approved the final manuscript.
Funding
The study was conducted with grants from the Chinese Natural Science Foundation (grant no. 81873215 and 82003006) and the Natural Science Foundation of Hainan Province (821QN384).
Availability of data and materials
The datasets generated during the current study are not publicly available due to limitations of ethical approval involving the patient data and anonymity but are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
The study was conducted in accordance with the principles stated in the Declaration of Helsinki and was approved by the ethics committee of Hainan Hospital of Chinese PLA General Hospital (ID: 301HLFYLS15), and informed consent was obtained from the patients or their authorized relatives.
Consent for publication
Not applicable.
Competing interests
There are no conflicts of interest to declare.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yuanyuan Chen, Mingyue Xu and Qianwen Ye contributed equally.
Contributor Information
Long Liu, Email: liul513111@163.com.
Bing Yan, Email: y_bing41@163.com.
References
- 1.Kuebler JP, Wieand HS, O'Connell MJ, Smith RE, Colangelo LH, Yothers G, et al. Oxaliplatin combined with weekly bolus fluorouracil and leucovorin as surgical adjuvant chemotherapy for stage II and III colon cancer: results from NSABP C-07. J Clin Oncol. 2007;25:2198–2204. doi: 10.1200/JCO.2006.08.2974. [DOI] [PubMed] [Google Scholar]
- 2.Souglakos J, Boukovinas I, Kakolyris S, Xynogalos S, Ziras N, Athanasiadis A, et al. Three- versus six-month adjuvant FOLFOX or CAPOX for high-risk stage II and stage III colon cancer patients: the efficacy results of Hellenic Oncology Research Group (HORG) participation to the International Duration Evaluation of Adjuvant Chemotherapy (IDEA) project. Ann Oncol. 2019;30:1304–1310. doi: 10.1093/annonc/mdz193. [DOI] [PubMed] [Google Scholar]
- 3.Yamazaki K, Yamanaka T, Shiozawa M, Manaka D, Kotaka M, Gamoh M, et al. Oxaliplatin-based adjuvant chemotherapy duration (3 versus 6 months) for high-risk stage II colon cancer: the randomized phase III ACHIEVE-2 trial. Ann Oncol. 2021;32:77–84. doi: 10.1016/j.annonc.2020.10.480. [DOI] [PubMed] [Google Scholar]
- 4.Ji WB, Hong KD, Kim JS, Joung SY, Um JW, Min BW. Effect of a shortened duration of FOLFOX chemotherapy on the survival rate of patients with stage II and III colon cancer. Chemotherapy. 2018;63:8–12. doi: 10.1159/000481566. [DOI] [PubMed] [Google Scholar]
- 5.Petrelli F, Rulli E, Labianca R, Lonardi S, Rosati G, Dotti K, et al. Overall survival with 3 or 6 months of adjuvant chemotherapy in Italian TOSCA phase 3 randomised trial. Ann Oncol. 2021;32:66–76. doi: 10.1016/j.annonc.2020.10.477. [DOI] [PubMed] [Google Scholar]
- 6.Iveson TJ, Sobrero AF, Yoshino T, Souglakos I, Ou FS, Meyers JP, et al. Duration of adjuvant doublet chemotherapy (3 or 6 months) in patients with high-risk stage II colorectal cancer. J Clin Oncol. 2021;39:631–641. doi: 10.1200/JCO.20.01330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Iveson T, Sobrero AF, Yoshino T, Sougklakos I, Ou FS, Meyers JP, et al. Prospective pooled analysis of four randomized trials investigating duration of adjuvant (adj) oxaliplatin-based therapy (3 vs 6 months {m}) for patients (pts) with high-risk stage II colorectal cancer (CC) J Clin Oncol. 2019;15(_suppl):3501. doi: 10.1200/JCO.2019.37.15_suppl.3501. [DOI] [Google Scholar]
- 8.Lu H, Zhao B, Zhang J, Huang R, Wang Z, Xu H, et al. Does delayed initiation of adjuvant chemotherapy following the curative resection affect the survival outcome of gastric cancer patients: A systematic review and meta-analysis. Eur J Surg Oncol. 2020;46:1103–1110. doi: 10.1016/j.ejso.2020.01.013. [DOI] [PubMed] [Google Scholar]
- 9.Chen QY, Liu ZY, Zhong Q, Xie JW, Wang JB, Lin JX, et al. Clinical impact of delayed initiation of adjuvant chemotherapy among patients with stage II/III gastric cancer: can we do better? Front Oncol. 2020;10:1149. doi: 10.3389/fonc.2020.01149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Morante Z, Ruiz R, Araujo JM, Pinto JA, Cruz-Ku G, Urrunaga-Pastor D, et al. Impact of the delayed initiation of adjuvant chemotherapy in the outcome of triple negative breast cancer. Clin Breast Cancer. 2021;21:239–46.e4. doi: 10.1016/j.clbc.2020.09.008. [DOI] [PubMed] [Google Scholar]
- 11.Chavez-MacGregor M, Clarke CA, Lichtensztajn DY, Giordano SH. Delayed initiation of adjuvant chemotherapy among patients with breast cancer. JAMA Oncol. 2016;2:322–329. doi: 10.1001/jamaoncol.2015.3856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Salazar MC, Rosen JE, Wang Z, Arnold BN, Thomas DC, Herbst RS, et al. Association of delayed adjuvant chemotherapy with survival after lung cancer surgery. JAMA Oncol. 2017;3:610–619. doi: 10.1001/jamaoncol.2016.5829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kim YW, Choi EH, Kim BR, Ko WA, Do YM, Kim IY. The impact of delayed commencement of adjuvant chemotherapy (eight or more weeks) on survival in stage II and III colon cancer: a national population-based cohort study. Oncotarget. 2017;8:80061–80072. doi: 10.18632/oncotarget.17767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Becerra AZ, Aquina CT, Mohile SG, Tejani MA, Schymura MJ, Boscoe FP, et al. Variation in delayed time to adjuvant chemotherapy and disease-specific survival in stage III colon cancer patients. Ann Surg Oncol. 2017;24:1610–1617. doi: 10.1245/s10434-016-5622-4. [DOI] [PubMed] [Google Scholar]
- 15.Kim IY, Kim BR, Kim YW. Factors affecting use and delay (≥8 weeks) of adjuvant chemotherapy after colorectal cancer surgery and the impact of chemotherapy-use and delay on oncologic outcomes. PLoS ONE. 2015;10:e0138720. doi: 10.1371/journal.pone.0138720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.van der Geest LG, Portielje JE, Wouters MW, Weijl NI, Tanis BC, Tollenaar RA, et al. Complicated postoperative recovery increases omission, delay and discontinuation of adjuvant chemotherapy in patients with stage III colon cancer. Colorectal Dis. 2013;15:e582–e591. doi: 10.1111/codi.12288. [DOI] [PubMed] [Google Scholar]
- 17.Chan A, Woods R, Kennecke H, Gill S. Factors associated with delayed time to adjuvant chemotherapy in stage III colon cancer. Curr Oncol. 2014;21:181–186. doi: 10.3747/co.21.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhu L, Tong YX, Xu XS, Xiao AT, Zhang YJ, Zhang S. High level of unmet needs and anxiety are associated with delayed initiation of adjuvant chemotherapy for colorectal cancer patients. Support Care Cancer. 2020;28:5299–5306. doi: 10.1007/s00520-020-05333-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Grothey A, Sobrero AF, Shields AF, Yoshino T, Paul J, Taieb J, et al. Duration of adjuvant chemotherapy for stage III colon cancer. N Engl J Med. 2018;378:1177–1188. doi: 10.1056/NEJMoa1713709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dienstmann R, Salazar R, Tabernero J. Personalizing colon cancer adjuvant therapy: selecting optimal treatments for individual patients. J Clin Oncol. 2015;33:1787–1796. doi: 10.1200/JCO.2014.60.0213. [DOI] [PubMed] [Google Scholar]
- 21.Xu R, You JH, Li F, Yan B. Postoperative fasting blood glucose predicts prognosis in stage I-III colorectal cancer patients undergoing resection. Gastroenterol Res Pract. 2020;2020:2482409. doi: 10.1155/2020/2482409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang Y, Liu Y, Qiu X, Yan B. Concurrent comparison of the prognostic values of tumor budding, tumor stroma ratio, tumor infiltrating pattern and lymphocyte-to-monocyte ratio in colorectal cancer patients. Technol Cancer Res Treat. 2021;20:15330338211045826. doi: 10.1177/15330338211045826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cheung WY, Neville BA, Earle CC. Etiology of delays in the initiation of adjuvant chemotherapy and their impact on outcomes for stage II and III rectal cancer. Dis Colon Rectum. 2009;52:1054–63. doi: 10.1007/DCR.0b013e3181a51173. [DOI] [PubMed] [Google Scholar]
- 24.Bayraktar UD, Chen E, Bayraktar S, Sands LR, Marchetti F, Montero AJ, et al. Does delay of adjuvant chemotherapy impact survival in patients with resected stage II and III colon adenocarcinoma? Cancer. 2011;117:2364–2370. doi: 10.1002/cncr.25720. [DOI] [PubMed] [Google Scholar]
- 25.Des Guetz G, Nicolas P, Perret GY, Morere JF, Uzzan B. Does delaying adjuvant chemotherapy after curative surgery for colorectal cancer impair survival? A meta-analysis. Eur J Cancer. 2010;46:1049–1055. doi: 10.1016/j.ejca.2010.01.020. [DOI] [PubMed] [Google Scholar]
- 26.Ahn HR, Kang SY, Youn HJ, Jung SH. Hyperglycemia during adjuvant chemotherapy as a prognostic factor in breast cancer patients without diabetes. J Breast Cancer. 2020;23:398–409. doi: 10.4048/jbc.2020.23.e44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Alacacioglu A, Kebapcilar L, Gokgoz Z, Oztekin O, Bozkaya G, Tarhan O, et al. Leptin, insulin and body composition changes during adjuvant taxane based chemotherapy in patients with breast cancer, preliminary study. Indian J Cancer. 2016;53:39–42. doi: 10.4103/0019-509X.180836. [DOI] [PubMed] [Google Scholar]
- 28.Tayek JA, Chlebowski RT. Metabolic response to chemotherapy in colon cancer patients. JPEN J Parenter Enteral Nutr. 1992;16:65–71. doi: 10.1177/014860719201600606. [DOI] [PubMed] [Google Scholar]
- 29.Chen J, Huang XF, Qiao L, Katsifis A. Insulin caused drug resistance to oxaliplatin in colon cancer cell line HT29. J Gastrointest Oncol. 2011;2:27–33. doi: 10.3978/j.issn.2078-6891.2010.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Baricevic I, Roberts DL, Renehan AG. Chronic insulin exposure does not cause insulin resistance but is associated with chemo-resistance in colon cancer cells. Horm Metab Res. 2014;46:85–93. doi: 10.1055/s-0033-1354414. [DOI] [PubMed] [Google Scholar]
- 31.Chen X, Liang H, Song Q, Xu X, Cao D. Insulin promotes progression of colon cancer by upregulation of ACAT1. Lipids Health Dis. 2018;17:122. doi: 10.1186/s12944-018-0773-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lu CC, Chu PY, Hsia SM, Wu CH, Tung YT, Yen GC. Insulin induction instigates cell proliferation and metastasis in human colorectal cancer cells. Int J Oncol. 2017;50:736–744. doi: 10.3892/ijo.2017.3844. [DOI] [PubMed] [Google Scholar]
- 33.Akdeniz N, Kaplan MA, Uncu D, İnanç M, Kaya S, Dane F, et al. The comparison of FOLFOX regimens with different doses of 5-FU for the adjuvant treatment of colorectal cancer: a multicenter study. Int J Colorectal Dis. 2021;36:1311–1319. doi: 10.1007/s00384-021-03888-9. [DOI] [PubMed] [Google Scholar]
- 34.Aspinall SL, Good CB, Zhao X, Cunningham FE, Heron BB, Geraci M, et al. Adjuvant chemotherapy for stage III colon cancer: relative dose intensity and survival among veterans. BMC Cancer. 2015;15:62. doi: 10.1186/s12885-015-1038-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chu-Yuan H, Jing P, Yi-Sheng W, He-Ping P, Hui Y, Chu-Xiong Z, et al. The impact of chemotherapy-associated neutrophil/lymphocyte counts on prognosis of adjuvant chemotherapy in colorectal cancer. BMC Cancer. 2013;13:177. doi: 10.1186/1471-2407-13-177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Borg C, Ray-Coquard I, Philip I, Clapisson G, Bendriss-Vermare N, Menetrier-Caux C, et al. CD4 lymphopenia as a risk factor for febrile neutropenia and early death after cytotoxic chemotherapy in adult patients with cancer. Cancer. 2004;101:2675–2680. doi: 10.1002/cncr.20688. [DOI] [PubMed] [Google Scholar]
- 37.Harrison L, Blackwell K. Hypoxia and anemia: factors in decreased sensitivity to radiation therapy and chemotherapy? Oncologist. 2004;9:31–40. doi: 10.1634/theoncologist.9-90005-31. [DOI] [PubMed] [Google Scholar]
- 38.Wei YS, Zhou YG, Wang GY, Liang ZH, Luo MR, Yang TA, et al. The impact of chemotherapy-associated hemoglobin on prognosis of colorectal cancer patients receiving adjuvant chemotherapy. Cancer Biomark. 2017;20:627–635. doi: 10.3233/CBM-170601. [DOI] [PubMed] [Google Scholar]
- 39.Pectasides D, Karavasilis V, Papaxoinis G, Gourgioti G, Makatsoris T, Raptou G, et al. Randomized phase III clinical trial comparing the combination of capecitabine and oxaliplatin (CAPOX) with the combination of 5-fluorouracil, leucovorin and oxaliplatin (modified FOLFOX6) as adjuvant therapy in patients with operated high-risk stage II or stage III colorectal cancer. BMC Cancer. 2015;15:384. doi: 10.1186/s12885-015-1406-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yoshino T, Yamanaka T, Oki E, Kotaka M, Manaka D, Eto T, et al. Efficacy and long-term peripheral sensory neuropathy of 3 vs 6 months of oxaliplatin-based adjuvant chemotherapy for colon cancer: the achieve phase 3 randomized clinical trial. JAMA Oncol. 2019;5:1574–1581. doi: 10.1001/jamaoncol.2019.2572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ho MY, Chang AY, Ruan JY, Cheung WY. Population-based cost-minimization analysis of CAPOX versus modified FOLFOX6 in the adjuvant treatment of stage III colon cancer. Clin Colorectal Cancer. 2016;15:158–163. doi: 10.1016/j.clcc.2015.09.004. [DOI] [PubMed] [Google Scholar]
- 42.Liu C, Liu Q, Yan A, Chang H, Ding Y, Tao J, et al. Metformin revert insulin-induced oxaliplatin resistance by activating mitochondrial apoptosis pathway in human colon cancer HCT116 cells. Cancer Med. 2020;9:3875–3884. doi: 10.1002/cam4.3029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Christou N, Bergen ES, Canton C, Le Malicot K, Di Bartolomeo M, Galli F, et al. Impact of diabetes and metformin use on recurrence and outcome in stage II-III colon cancer patients-A pooled analysis of three adjuvant trials. Eur J Cancer. 2022;166:100–111. doi: 10.1016/j.ejca.2022.02.005. [DOI] [PubMed] [Google Scholar]
- 44.Coyle C, Cafferty FH, Vale C, Langley RE. Metformin as an adjuvant treatment for cancer: a systematic review and meta-analysis. Ann Oncol. 2016;27:2184–2195. doi: 10.1093/annonc/mdw410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Saif MW, Hakim N, Chi J, Rehman H, Goyal SP, Olazagasti C, et al. First analysis of same-day pegfilgrastim use with concurrent capecitabine-based regimens in patients with gastrointestinal malignancies. Cancer Med J. 2021;4:1–6. [PMC free article] [PubMed] [Google Scholar]
- 46.Abdel-Razeq H, Hashem H. Recent update in the pathogenesis and treatment of chemotherapy and cancer induced anemia. Crit Rev Oncol Hematol. 2020;145:102837. doi: 10.1016/j.critrevonc.2019.102837. [DOI] [PubMed] [Google Scholar]
- 47.Ye X, Liu J, Chen Y, Wang N, Lu R. The impact of hemoglobin level and transfusion on the outcomes of chemotherapy in gastric cancer patients. Int J Clin Exp Med. 2015;8:4228–4235. [PMC free article] [PubMed] [Google Scholar]
- 48.Wilson MJ, Dekker JWT, Buettner S, Harlaar JJ, Jeekel J, Schipperus M, et al. The effect of intravenous iron therapy on long-term survival in anaemic colorectal cancer patients: Results from a matched cohort study. Surg Oncol. 2018;27:192–199. doi: 10.1016/j.suronc.2018.03.005. [DOI] [PubMed] [Google Scholar]
- 49.Ludwig H, Aapro M, Bokemeyer C, Macdonald K, Soubeyran P, Turner M, et al. Treatment patterns and outcomes in the management of anaemia in cancer patients in Europe: findings from the Anaemia Cancer Treatment (ACT) study. Eur J Cancer. 2009;45:1603–1615. doi: 10.1016/j.ejca.2009.02.003. [DOI] [PubMed] [Google Scholar]
- 50.Littlewood TJ, Bajetta E, Nortier JW, Vercammen E, Rapoport B, Epoetin Alfa Study Group Effects of epoetin alfa on hematologic parameters and quality of life in cancer patients receiving nonplatinum chemotherapy: results of a randomized, double-blind, placebo-controlled trial. J Clin Oncol. 2001;19:2865–74. doi: 10.1200/JCO.2001.19.11.2865. [DOI] [PubMed] [Google Scholar]
- 51.Tomita N, Kunieda K, Maeda A, Hamada C, Yamanaka T, Sato T, et al. Phase III randomised trial comparing 6 vs. 12-month of capecitabine as adjuvant chemotherapy for patients with stage III colon cancer: final results of the JFMC37–0801 study. Br J Cancer. 2019;120:689–96. doi: 10.1038/s41416-019-0410-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Park D, Baek SJ, Kwak JM, Kim J, Kim SH. Analysis of reduced-dose administration of oxaliplatin as adjuvant FOLFOX chemotherapy for colorectal cancer. Ann Surg Treat Res. 2018;94:196–202. doi: 10.4174/astr.2018.94.4.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yun HR, Kim HC, Yun SH, Lee WY. Adjuvant chemotherapy increase survival and decrease recurrence in stage IIA colon cancer. Hepatogastroenterology. 2012;59:2466–2471. doi: 10.5754/hge10260. [DOI] [PubMed] [Google Scholar]
- 54.Araujo CS, Venchiarutti Moniz CM, Bonadio RC, Watarai GY, Rojas J, Nogueira PVS, et al. Real-world data for high-risk stage II colorectal cancer-the role of tumor side in the adjuvant setting. Clin Colorectal Cancer. 2021;20:e100–e108. doi: 10.1016/j.clcc.2020.09.009. [DOI] [PubMed] [Google Scholar]
- 55.He Y, Liu P, Zhang Y, Deng X, Meng W, Wei M, et al. Low-dose capecitabine adjuvant chemotherapy in elderly stage II/III colorectal cancer patients (LC-ACEC): study protocol for a randomized controlled trial. Trials. 2015;16:238. doi: 10.1186/s13063-015-0753-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Baxter NN, Kennedy EB, Bergsland E, Berlin J, George TJ, Gill S, et al. Adjuvant therapy for stage II colon cancer: ASCO guideline update. J Clin Oncol. 2022;40:892–910. doi: 10.1200/JCO.21.02538. [DOI] [PubMed] [Google Scholar]
- 57.Lino-Silva LS, Anchondo-Núñez P, Chit-Huerta A, Aguilar-Romero E, Morales-Soto J, Salazar-García JA, et al. Stage I-III colon cancer patients with tumor deposits behave similarly to stage IV patients. cross-section analysis of 392 patients. J Surg Oncol. 2019;120:300–7. doi: 10.1002/jso.25482. [DOI] [PubMed] [Google Scholar]
- 58.Li X, An B, Zhao Q, Qi J, Wang W, Zhang D, et al. Impact of tumor deposits on the prognosis and chemotherapy efficacy in stage III colorectal cancer patients with different lymph node status: A retrospective cohort study in China. Int J Surg. 2018;56:188–194. doi: 10.1016/j.ijsu.2018.06.029. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during the current study are not publicly available due to limitations of ethical approval involving the patient data and anonymity but are available from the corresponding author on reasonable request.