Abstract
The hydrothermal vent tubeworm Riftia pachyptila lacks a mouth and gut and lives in association with intracellular, sulfide-oxidizing chemoautotrophic bacteria. Growth of this tubeworm requires an exogenous source of nitrogen for biosynthesis, and, as determined in previous studies, environmental ammonia and free amino acids appear to be unlikely sources of nitrogen. Nitrate, however, is present in situ (K. Johnson, J. Childress, R. Hessler, C. Sakamoto-Arnold, and C. Beehler, Deep-Sea Res. 35:1723–1744, 1988), is taken up by the host, and can be chemically reduced by the symbionts (U. Hentschel and H. Felbeck, Nature 366:338–340, 1993). Here we report that at an in situ concentration of 40 μM, nitrate is acquired by R. pachyptila at a rate of 3.54 μmol g−1 h−1, while elimination of nitrite and elimination of ammonia occur at much lower rates (0.017 and 0.21 μmol g−1 h−1, respectively). We also observed reduction of nitrite (and accordingly nitrate) to ammonia in the trophosome tissue. When R. pachyptila tubeworms are exposed to constant in situ conditions for 60 h, there is a difference between the amount of nitrogen acquired via nitrate uptake and the amount of nitrogen lost via nitrite and ammonia elimination, which indicates that there is a nitrogen “sink.” Our results demonstrate that storage of nitrate does not account for the observed stoichiometric differences in the amounts of nitrogen. Nitrate uptake was not correlated with sulfide or inorganic carbon flux, suggesting that nitrate is probably not an important oxidant in metabolism of the symbionts. Accordingly, we describe a nitrogen flux model for this association, in which the product of symbiont nitrate reduction, ammonia, is the primary source of nitrogen for the host and the symbionts and fulfills the association's nitrogen needs via incorporation of ammonia into amino acids.
Riftia pachyptila, a hydrothermal vent tubeworm, is a conspicuous member of the hydrothermal vent communities found along the East Pacific Rise, the Guaymas Basin, and the Southern East Pacific Rise (33). This mouthless, gutless vestimentiferan worm received much attention when researchers found that it has chemoautotrophic bacterial symbionts in its trunk (in an organ referred to as the trophosome) (2, 8a). The symbionts were determined to be carbon-fixing, sulfide-oxidizing chemoautotrophs (10) that are present at densities up to 3.7 × 109 cells g of trophosome−1 (2, 29). The symbionts are far from the external milieu, and the metabolites required to sustain sulfide-driven carbon fixation must be provided via the host. R. pachyptila thrives at the interface of vent and bottom-water mixing; thus, its plume (gill) has variable contact with both the cold bottom water and the warmer vent effluent, and the organism has simultaneous access to both reduced and oxidized metabolites (4).
In previous studies workers have focused on the suite of biochemical and physiological adaptations that result in acquisition, storage, and elimination of the reduced and oxidized substrates and end products of carbon fixation and sulfide oxidation (1, 13, 14). However, for growth to occur there must be an exogenous source of nitrogen for biosynthesis, and the absence of a digestive tract in the worm precludes the possibility of particulate ingestion (18). Accordingly, nitrogen must be obtained through absorption from the environment. The in situ concentrations of free amino acids at hydrothermal vent sites along the East Pacific Rise are extremely low (<200 pM) (17), and the results of δ15N stable isotope studies of R. pachyptila tissues have indicated that the source of nitrogen for the tubeworm-bacterium association is not organic (30). In addition, the ammonia concentrations at sites along the East Pacific Rise are low (3 μM) (17), although the ammonia concentrations in the Guaymas Basin can be as high as 15 mM in the sediments around the tubeworms (34). Shipboard studies of intact associations have suggested that R. pachyptila collected from sites along the East Pacific Rise does not take up ammonia in detectable quantities (21). Nitrate, however, occurs in the cold bottom water at in situ concentrations of ca. 40 μM (17), and preliminary studies have suggested that nitrate may be taken up by intact associations (21). R. pachyptila, like all heterotrophic metazoans, is not capable of metabolizing nitrate. Reduction of nitrate by bacteria, however, is common among the chemoautotrophs, such as the filamentous sulfur bacteria (27). In one previous study of isolated R. pachyptila symbionts, the workers demonstrated that nitrate was reduced to nitrite (16). The authors posited that nitrate reduction by the symbionts is a means of sustaining the oxidative requirements of the symbionts during periods of environmental hypoxia. They found no evidence that ammonia was formed during reduction of nitrate, and the possibility that this process could provide a substantial source of reduced nitrogen for the association was not considered. However, in another study the researchers found activities of ammonia assimilatory enzymes in the symbionts of R. pachyptila (22), suggesting that assimilation of ammonia may be the predominant source of nitrogen for symbiont biosynthesis.
No one has quantified the rates of nitrogenous metabolite flux or the rates of flux for other major metabolites by an intact association previously. This is due in large part to the difficulty of maintaining live hydrothermal vent fauna in the laboratory. Our high-pressure respirometry system, which is the result of years of development (20), allows us to maintain animals and to simultaneously measure metabolite flux in pressurized flow-through aquaria. This system also allows us to determine that tubeworms are in “autotrophic” balance (i.e., that they take up inorganic metabolites and eliminate proton equivalents). This system differs from the systems used in previous studies, in which the worms were maintained at atmospheric pressure or at in situ pressures in aquaria filled with surface seawater containing dissolved gases and compounds at concentrations that were not typical of the concentrations found in situ and thus did not support autotrophy.
In this study, we investigated nitrate uptake by intact R. pachyptila during exposure to external nitrate concentrations ranging from 0 to 550 μM. During our experiments, we paid particular attention to the rate and duration of nitrate uptake at in situ nitrate concentrations, as well as the concomitant rates of nitrogenous metabolite loss to the environment. We also examined the relationship among nitrate uptake, carbon uptake, and oxygen uptake when different external nitrate regimens were used. In addition, we investigated whether formation of ammonia was the result of nitrite (and accordingly nitrate) reduction in trophosome tissue.
In a previous study of nitrate reduction by the symbionts (16), the authors demonstrated that the symbionts were able to utilize nitrate, but they did not elucidate the role of nitrate reduction in respiration (dissimilatory nitrate reduction) and biosynthesis (assimilatory nitrate reduction). In this study, we examined the stoichiometry of nitrate reduction and, by comparing the concomitant rates of oxygen uptake and sulfide uptake, also examined the potential role of nitrate reduction in respiration. We posit that reduction of nitrate, which may be assimilatory or dissimilatory, leads to the formation of ammonia, which is assimilated into amino acids by the symbionts and, potentially, by the host.
MATERIALS AND METHODS
Animal collection and maintenance.
Tubeworms were collected from hydrothermal vent sites along the East Pacific Rise (12°48′N, 103°56′W and 9°50′N, 104°18′W) at a depth of about 2,600 m during expeditions in April 1996, November 1997, and November 1998. Worms were collected daily, brought from depth in a thermally insulated container (25), and immediately placed into flow-through, high-pressure respirometer aquaria (20). Unless otherwise stated, all experiments were conducted in high-pressure aquaria at 12°C and 27.5 kPa. Recent modifications to the high-pressure respirometry system include (i) the development of 316 stainless steel-reinforced acrylic sleeves, which permit prolonged operation at pressures up to 34 kPa, (ii) a gas extractor (fabricated of polysulfone) for “stripping” dissolved gases from the aquarium effluents into a helium stream for analysis by membrane inlet mass spectroscopy, and (iii) a LabView-based computer program for data acquisition and system control.
In the experiment in which we examined the relationship between nitrate uptake and environmental oxygen tension (see below), we used worms which had been collected 2 days previously and had been kept in “maintenance” aquaria (14). Worms in these aquaria were maintained under in situ vent conditions (total concentration of all ionic species of inorganic carbon [ΣCO2], 5 to 6 mM; total concentration of all ionic species of sulfide [ΣH2S], 250 to 600 μM; O2 concentration, 100 to 400 μM; NO3− concentration, 40 μM; temperature, 12°C; pressure, 27.5 kPa). The ΣCO2, ΣH2S, and O2 uptake values for these worms and for freshly caught worms did not differ (P = 0.578, P = 0.351, and P = 0.376, respectively, as determined by Mann-Whitney tests; number of worms in each group, 4).
Determination of flux rates by intact associations.
In all of the experiments in which we examined flux rates in intact associations, one to three tubeworms weighing between 5 and 15 g each were placed into two of the high-pressure respirometry system aquaria. A third vessel, which served as a control, did not contain tubeworms. To simulate the conditions found in situ, filtered seawater (pore size, 0.2 μm) was pumped with a metering pump (Cole-Parmer, Inc.) into an acrylic gas equilibration column and bubbled with CO2, H2S, O2, and N2 or He to obtain in situ concentrations. Mass flow controllers (Sierra Instruments, Inc.) were used to regulate the gas flow into the equilibration column. The seawater pH was maintained between 6.2 and 6.5 with a proportional pH controller (Prominent Industries, Inc.) that controlled two metering pumps that pumped 1 M NaOH and 1 M HCl. A 5 mM sodium nitrate solution (sodium nitrate dissolved in filtered seawater [pore size, 0.2 μm]) was also pumped into the equilibration column with a metering pump (Prominent Industries, Inc.) at various rates, which resulted in specific seawater nitrate concentrations between 0 and 550 μM (the seawater used was obtained from the surface and did not contain nitrate at concentrations within the limits of our method of detection [ca. 500 nM]). The resulting seawater was pumped from the equilibration column into each aquarium by using three high-pressure pumps (Hastelloy C diaphragms and check valves; 316 stainless steel pump heads; Lewa America, Inc.). The temperatures in the aquaria were maintained at 15°C by immersing the aquaria in a circulating water bath. The pressure in the aquaria was maintained at 27.5 kPa (4,000 lb/in2) by using pneumatically charged or spring-loaded backpressure valves (Circle Seal, Inc.). Changes in the dissolved gas concentrations in the vessel effluents were analyzed with a residual gas analyzer-mass spectrometer (Hiden Analytical Inc.). Changes in the seawater nitrate and nitrite concentrations were analyzed by performing a spectrophotometric analysis of discrete water samples (17, 23). Seawater urate (uric acid) concentrations were determined by a quantitative enzymatic assay (11). Seawater ammonium concentrations were determined by performing a flow injection analysis with discrete water samples (35). Before experiments were performed, all tubeworms were maintained in respirometer aquaria under in situ conditions until autotrophy was established. This typically required between 12 and 24 h. An autotrophic worm was defined as a worm which exhibited net inorganic carbon, oxygen, and sulfide uptake from the environment, as well as net elimination of proton equivalents into the environment.
To determine the nitrogenous metabolite flux rates of R. pachyptila, three worms collected during the April 1996 expedition (designated the HOT 96 expedition) were placed into two of the pressurized aquaria and maintained until they exhibited autotrophy. Sodium nitrate was added to the incurrent aquarium seawater via the equilibration column at various rates over several hours in order to obtain a series of final seawater nitrate concentrations between 0 and 550 μM. Worms were kept at each incremental step until their inorganic carbon, oxygen, sulfide, and proton flux rates stabilized, typically 10 to 12 h. Other than the variation in the external nitrate concentrations, the worms were kept under constant in situ conditions for the duration of the experiment (ΣCO2, 5 mM; ΣH2S, 250 μM; O2 concentration, 150 μM; pH 5.6; temperature, 12°C; pressure, 27.5 kPa). Water samples were collected from all three aquaria at least hourly and used for nitrate, nitrite and ammonia analyses.
To examine the relationship between nitrate uptake and oxygen uptake, four worms collected during the November 1997 expedition (designated the HOT 97 expedition) were placed in two of the pressurized aquaria and maintained until they exhibited autotrophy. The worms were exposed to 50 μM nitrate and 95 μM dissolved oxygen for 30 h. Subsequently, the worms were exposed to 150 μM nitrate and 95 μM dissolved oxygen for 36 h. The flow of oxygen to the equilibration column was then turned off, and the concentrations of dissolved oxygen in the aquaria decreased to less than 3 μM (below the limit of detection by gas chromatography [3]). Samples of the aquarium effluents were taken from all three aquaria and used for nitrate, nitrite, and ammonia analyses.
To examine the relationship among nitrate uptake, carbon dioxide uptake, sulfide uptake, and oxygen uptake, three worms collected during the November 1998 expedition (designated the LARVE 98 expedition) were placed in two of the pressurized aquaria and maintained in nitrate-free seawater for 2 days. The surface seawater did not contain nitrate at concentrations within the limit of detection (ca. 500 nM). The concentrations of all other compounds were maintained at in situ values. Nitrate was then added to the incurrent seawater in order to obtain a nitrate concentration of 100 μM in the aquaria. The worms were maintained under these conditions for 3 days, during which the dissolved gas and nitrate concentrations were varied incrementally one at a time. The seawater dissolved gas concentrations were measured, and samples of aquarium effluents were taken from all three aquaria and used for nitrate, nitrite, and ammonia analyses.
To examine the relationship between nitrate uptake and inorganic carbon uptake, four worms collected during the November 1997 expedition (the HOT 97 expedition) were placed in two of the pressurized aquaria and maintained in the presence of three different nitrate concentrations (56, 154, and 640 μM) for 4 days. The concentrations of all other compounds were maintained at in situ values. The dissolved gas concentrations were measured, and samples of aquarium effluents were taken and used for nitrate and ammonia analyses.
At the end of each experiment, the worms were removed from the aquaria, quickly separated from their tubes, and weighed with a motion-compensated balance (5). All rates and other parameters were expressed in terms of wet weight. The tubeworms were then dissected, and tissue samples were promptly frozen in liquid nitrogen for later analysis. In most cases, the empty worm tubes were returned to the pressure vessel and subjected to the same experimental conditions to determine what fraction, if any, of the observed flux rates could be attributed to bacterial growth or other phenomena associated with the tubes.
Determination of trophosome nitrogenous metabolite concentrations in vessel-maintained worms.
To examine the changes in tissue nitrogenous metabolite concentrations, 11 R. pachyptila tubeworms collected during the April 1996 expedition (the HOT 96 expedition) were kept in seawater that contained no nitrate for 2 days (all other conditions were in situ conditions; i.e., ΣCO2, 5 mM; ΣH2S, 250 μM; O2 concentration, 150 μM; pH 5.6; temperature, 12°C; pressure, 27.5 kPa). Six worms were removed for analysis. The nitrate was added to a final concentration of 53 μM, and the remaining worms were maintained under the conditions described above for an additional 2 days. Trophosome samples obtained from vessel-maintained worms were quickly dissected and analyzed on the ship or were frozen in liquid nitrogen for later analysis. The trophosome samples used for nitrate analyses were prepared by homogenizing the tissue by using a Dounce ground-glass homogenizer and M9 bacterial medium (31). The nitrate concentrations in the trophosome samples were determined by performing a bacterial bioassay in which reduction of nitrate (in the trophosome homogenate) to nitrite by Escherichia coli was coupled to spectrophotometric determination of nitrite concentrations (28). The trophosome samples used for ammonia analysis were prepared by homogenizing the tissue in a ground-glass homogenizer with an equal volume of methanol, centrifuging the homogenate at 11,000 × g to remove the particulates, and diluting the preparation 1:1 with deionized water. The homogenate was then analyzed by performing a flow injection analysis (35). The trophosome samples used for urate analysis were prepared by homogenizing the tissue in a ground-glass homogenizer by using 2 parts of 1 M perchloric acid for each 1 part of tissue, neutralizing the homogenate with an equal volume of 2.5 mM potassium bicarbonate, and centrifuging the preparation at 11,000 × g to remove the precipitate. The supernatant was analyzed by performing a quantitative enzymatic analysis of the uric acid content (11).
Determination of nitrogenous metabolite production in excised trophosomes.
To determine the product of nitrite reduction, we examined the simultaneous disappearance of nitrite and production of ammonia by performing assays in which we used excised trophosome tissue extracts obtained from R. pachyptila collected during the November 1997 expedition (the HOT 97 expedition). Extracts were prepared as follows. A trophosome from R. pachyptila was homogenized with a Dounce ground-glass homogenizer by using 5 volumes of ice-cold 100 mM Tris buffer–2.5 mM MgCl2–1 mM mercaptoethanol (pH 7.5), and the extract was sonicated on ice and centrifuged for 10 min at 11,000 × g. Supernatants from such extracts were used in subsequent assays. A 50-μl portion of supernatant was added to a 1-ml (final volume) reaction mixture containing 100 mM phosphate, 0.2 or 5 mM sodium nitrite, and 0.2 mM benzyl viologen (an artificial electron donor) at pH 7.4 and 25°C. The reaction was initiated by reducing benzyl viologen by adding sodium dithionite to a final concentration of 2 mM. Nitrite concentrations were determined spectrophotometrically after sulfanilamide and naphthylethylenediamine (12) were added. Ammonia concentrations were measured by performing a flow injection analysis (35).
RESULTS
Rates of metabolite flux.
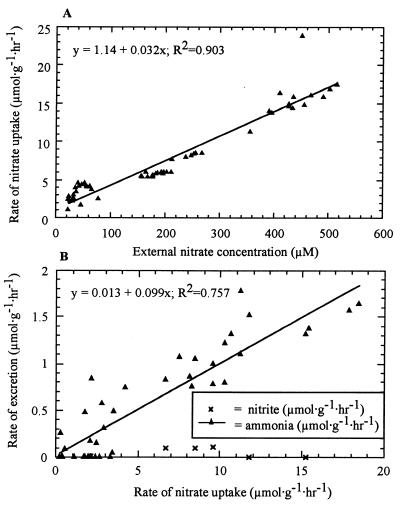
Data obtained from our HOT 96 expedition experiments showed that nitrate uptake by R. pachyptila was strongly correlated with the seawater nitrate content during exposure to nitrate concentrations between 50 and 500 μM (Fig. 1A) (R2 = 0.903; P < 0.0001). The correlation between the seawater nitrate concentration and nitrate uptake remained linear for seawater nitrate concentrations up to 550 μM. During exposure to nitrate concentrations resembling in situ concentrations (e.g., 50 μM nitrate), the average rate of nitrate uptake by R. pachyptila was 3.54 ± 0.403 μmol g−1 h−1 (number of seawater samples, 19).
FIG. 1.
(A) Nitrate uptake by R. pachyptila as a function of the external nitrate concentration. (B) Nitrite and ammonia excretion as a function of nitrate uptake by R. pachyptila. All rates are expressed in terms of wet weight.
Nitrate uptake resulted in the concomitant appearance of nitrite and ammonia in the experimental vessel effluents compared to the control vessel effluents (Fig. 1B). The appearance of nitrite and ammonia in the experimental vessels was attributed to nitrate reduction by the symbionts of R. pachyptila; this appearance of nitrite and ammonia in the vessel seawater is referred to below as nitrite loss and ammonia loss by the tubeworm. At environmentally relevant seawater nitrate concentrations, nitrite loss and ammonia loss occurred at rates of 0.017 and 0.21 μmol g−1 h−1, respectively (Fig. 1B). The rate of nitrite loss by R. pachyptila was always low and was not correlated with nitrate uptake (R2 = 0.016; P = 0.813) (Fig. 1B), while the rate of ammonia loss was correlated with nitrate uptake (R2 = 0.758; P < 0.0001) (Fig. 1B). The rates of nitrite loss and ammonia loss by R. pachyptila were typically 2 and 1 orders of magnitude, respectively, less than the rates of nitrate uptake by R. pachyptila (Fig. 1B). In two experiments performed during our HOT 97 and HOT 98 expeditions, R. pachyptila tubeworms were maintained in the presence of 50 μM nitrate seawater concentrations for more than 60 h. During that time, nitrate uptake by R. pachyptila remained high while nitrite and ammonia loss by R. pachyptila remained low. In addition, the levels of dinitrogen in the experimental vessels were not significantly different than the level in the control (P = 0.802, as determined by the Student t test).
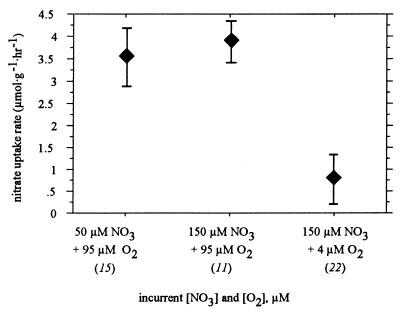
Data obtained during the HOT 97 expedition showed that, during exposure for 30 h, increasing the seawater nitrate concentration from 50 to 150 μM did not result in a significant increase in the average rate of nitrate uptake by R. pachyptila (it increased from 3.56 ± 0.66 to 3.95 ± 0.54 μmol g−1 h−1) (Fig. 2). Reducing the seawater dissolved oxygen concentration to an undetectable level resulted in a significant reduction in the rate of nitrate uptake by R. pachyptila, to an average value of 0.77 ± 0.29 μmol g−1 h−1 (Fig. 2).
FIG. 2.
Nitrate uptake by R. pachyptila when three different oxygen-nitrate regimens were used. The incurrent [NO3] and [O2] values are the concentrations of nitrate and oxygen, respectively, in the seawater. The numbers in parentheses are the numbers of discrete samples used to calculate the rates. The error bars indicate standard errors. All rates are expressed in terms of wet weight.
R. pachyptila tubeworms collected during the HOT 97 expedition were exposed for 12 h at a time to seawater nitrate concentrations of 56, 154, and 640 μM. The rates of nitrate uptake by the worms increased as the nitrate concentration increased, while there was no change in the ΣCO2 uptake rate (Table 1). The difference between the amount of nitrogen acquired as nitrate and the amount of nitrogen lost as other nitrogenous compounds (ΔN) was the amount of nitrogen available to the association. At approximately in situ nitrate concentrations, ΔN was 3.61 μmol of N g−1 h−1. The ratio of ΣCO2 uptake to ΔN was 3.81. This ΣCO2 uptake/ΔN ratio was similar to the association's C/N ratio (3.91) (9).
TABLE 1.
Rates of inorganic carbon and nitrogenous metabolite uptake and elimination by R. pachyptila exposed to three environmental nitrate concentrations
| NO3− concn in aquarium seawater (μM) | Rate of NO3− uptake (μmol · g−1 · h−1) | Sum of NO2− excretion rate and NH3 excretion rate (μmol · g−1 · h−1) | ΔN (μmol · g−1 · h−1) | ΣCO2 uptake rate (μmol · g−1 · h−1) | Ratio of ΣCO2 uptake to NO3− uptake |
|---|---|---|---|---|---|
| 56 ± 8.72 (20)a | 3.78 ± 0.302 (20) | 0.17 ± 0.091 (18) | 3.61 | 14.4 ± 2.73 (33) | 3.81 |
| 154 ± 9.94 (9) | 8.12 ± 0.879 (9) | 0.51 ± 0.276 (8) | 7.61 | 16.9 ± 1.20 (29) | 2.22 |
| 640 ± 16.1 (13) | 12.63 ± 1.04 (13) | 0.77 ± 0.198 (9) | 11.86 | 15.5 ± 0.388 (42) | 1.31 |
The numbers in parentheses are the numbers of mass spectrometer observations or discrete seawater samples collected at a given nitrate concentration. All rates are expressed in terms of wet weight.
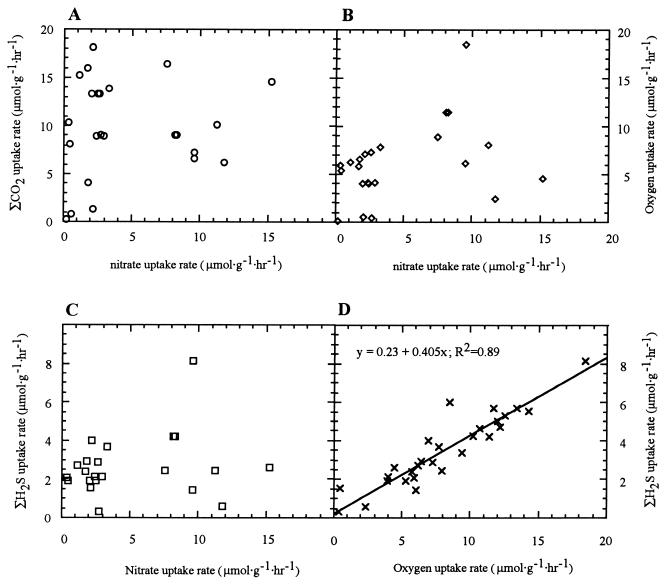
Data obtained during our LARVE 98 expedition showed that nitrate uptake was not correlated with changes in seawater sulfide, oxygen, or inorganic carbon concentrations (Fig. 3). During this same experiment, oxygen uptake by R. pachyptila was strongly correlated with sulfide uptake (R2 = 0.89).
FIG. 3.
(A) Inorganic carbon uptake rate versus nitrate uptake rate. (B) Oxygen uptake rate versus nitrate uptake rate. (C) Sulfide uptake rate versus nitrate uptake rate. (D) Sulfide uptake rate versus oxygen uptake rate in R. pachyptila. Nitrate concentrations were maintained at 100 μM in the aquarium incurrent flows. All rates are expressed in terms of wet weight.
Empty tubeworm tubes that were returned to the vessels after worm removal in order to determine what fractions of the flux rates were attributable to bacterial growth exhibited no flux compared to the control.
Tissue and seawater nitrogenous metabolite concentrations.
During the HOT 96 expedition, the trophosome nitrate, nitrite, and ammonia concentrations in R. pachyptila maintained in aquarium seawater without nitrate were much lower than the concentrations in worms maintained in the presence of nitrate (Table 2). The urate and dinitrogen concentrations, however, did not vary significantly when we compared worms maintained for 2 days with and without nitrate (Table 2).
TABLE 2.
Concentrations of nitrogenous metabolites in excised trophosome tissue of R. pachyptila before and during exposure to 53 μM nitrate
| Metabolite | Concn in trophosome in the absence of nitrate (μM) | Concn in trophosome in the presence of 53 μM nitrate (μM) | Significant difference | P value |
|---|---|---|---|---|
| Nitrate | 27.8 ± 12.2 (6)a | 394.8 ± 84.2 (5) | Yes | 0.0055 |
| Nitrite | 7.4 ± 2.6 (6) | 127.3 ± 25.8 (5) | Yes | 0.0055 |
| Ammonia | 1,363.3 ± 146.0 (4) | 6,242.1 ± 2,041.7 (5) | Yes | 0.0105 |
| Dinitrogen | 427.4 ± 15.6 (4) | 396.2 ± 16.1 (4) | No | 0.2623 |
| Urate | 1,596.2 ± 201.2 (6) | 1,368.8 ± 221.2 (4) | No | 0.2482 |
Mean ± standard error. The numbers in parentheses are the numbers of individual R. pachyptila tubeworms from which discrete samples were taken for analysis.
The concentrations of ammonia in samples of seawater effluent from pressurized aquaria containing four R. pachyptila tubeworms increased significantly during exposure to nitrate (53 μM in the surrounding seawater) (Table 3). The concentrations of nitrite in the seawater effluents, however, did not vary significantly between treatments. Urate was not detected in samples of seawater effluents obtained from pressurized aquaria containing four R. pachyptila tubeworms before and during exposure to nitrate (Table 3).
TABLE 3.
Concentrations of nitrogenous metabolites in the seawater effluents from pressurized aquaria containing four R. pachyptila tubeworms before and during exposure to 53 μM nitrate in the surrounding seawater
| Metabolite | Aquarium effluent concn in the absence of nitrate (μM) | Aquarium effluent concn in the presence of 53 μM nitrate (μM) | Statistical significance (Mann-Whitney U test) | P value |
|---|---|---|---|---|
| Nitrite | 0.089 ± 0.061 (8)a | 0.190 ± 0.107 (6) | No | 0.5635 |
| Ammonia | 2.00 ± 0.65 (3) | 15.48 ± 1.16 (7) | Yes | 0.0015 |
| Dinitrogen | 447.61 ± 29.74 (5) | 429.39 ± 19.55 (7) | No | 0.3756 |
| Urate | NDb | ND |
Mean ± standard error. The numbers in parentheses are the numbers of observations or discrete samples collected from the aquarium effluent for analysis.
ND, not within the detectable range of our technique.
Reduction of nitrite to ammonia in excised trophosomes.
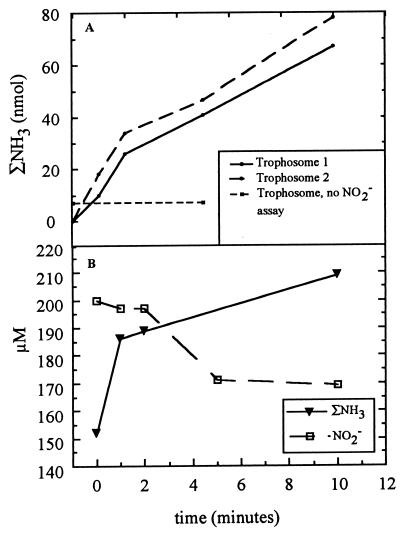
Experiments conducted during the HOT 97 expedition with trophosome tissue extracts obtained from R. pachyptila revealed that the ammonia concentration increased after 5 mM nitrite was added. No increases in the ammonia concentrations were observed in the reaction mixtures to which nitrite was not added (Fig. 4A). These results indicate that the level of nitrite reductase activity was approximately 0.78 to 0.85 nmol mg (fresh weight)−1 min−1 and are consistent with the hypothesis that a nitrite reductase that reduced nitrite to ammonia was present. At a nitrite concentration of 200 μM, a decrease in the nitrite concentration accompanied the production of ammonia (Fig. 4B). Lower nitrite concentrations did not result in detectable ammonia production (data not shown).
FIG. 4.
(A) Assays involving excised trophosomes of R. pachyptila exposed to 5 mM sodium nitrite in Riftia saline. Increases in the ammonia concentration were observed in the presence of 5 mM nitrite. No increases in the ammonia concentration were observed in the reaction mixture containing trophosomes without added nitrite. (B) Assays involving trophosomes of R. pachyptila exposed to 200 μM nitrite (to determine if ammonia production was accompanied by a concomitant disappearance of nitrite). A decrease in the nitrite concentration accompanied the production of ammonia. ΣNH3, both the NH3 and NH4+ ionic species of ammonia.
DISCUSSION
The most readily available form of inorganic nitrogen in the hydrothermal vent environment is nitrate (17). In general, there are two possible uses of nitrate by symbiotic bacteria. Nitrate may be used in lieu of oxygen as a terminal electron acceptor during respiration (generally referred to as dissimilatory nitrate reduction) (6). Nitrate may also be reduced and incorporated into amino acids for biosynthesis (generally referred to as assimilatory nitrate reduction) (6). Lee and Childress (21) demonstrated that in R. pachyptila 15N from 15NO3− was incorporated into organic matter and inferred that the nitrate was reduced to ammonia before incorporation. The technique which these authors used, however, does not allow workers to measure the rates or durations of nitrate uptake into the worm, the concentrations of ammonia and nitrate in the worm and its symbionts, or the loss of nitrite and ammonia by the worm. Consequently, Lee and Childress concluded that reduction of nitrate was the source of the labeled nitrogen, but no definitive inference could be drawn concerning the mechanism of nitrogen acquisition or utilization.
The previous studies were conducted by using closed systems in which the conditions (particularly the O2 concentration, CO2 concentration, ΣH2S, pH, and temperature) were often unstable and were far from the optimum conditions for autotrophic balance in the R. pachyptila symbiosis. Due to the long lag time for initiation of autotrophy (approximately 24 h), it is certain that these previous studies in which much shorter incubation times were used did not involve animals in net autotrophic balance. As indicated above, the experiments described here were performed by using flowing, pressurized seawater aquaria and worms which exhibited signs of autotrophy while they were maintained under in situ conditions.
In our experiments performed with intact associations, we found uptake of nitrate by R. pachyptila occurs at in situ concentrations of nitrate and that a typical rate of 3.54 ± 0.403 μmol g−1 h−1 can be sustained for at least 60 h (Fig. 1A). Our experiments were the first experiments in which the rate of nitrate uptake by an intact vent symbiosis was measured. In a previous study of isolated R. pachyptila symbionts, Hentschel and Felbeck showed that nitrate was taken up from the media at a rate of 10 μM g of protein−1 h−1 (as determined by the rate of nitrite production) (16). We have found that fractions of the internal nitrite and ammonia, which occur only in the presence of nitrate, are eliminated into the external milieu when R. pachyptila tubeworms are exposed to any concentration of nitrate (Fig. 1B). The correlation between nitrate uptake and ammonium elimination suggests that ammonium results from nitrate reduction and may be the end product of nitrate reduction (Fig. 1B). The lack of a correlation between nitrate uptake and nitrite elimination and the low levels of nitrite elimination suggest that nitrite is not a substantial end product of nitrate reduction by the symbionts (under conditions in which the intact symbiosis exhibits autotrophy). Our experiments performed with excised trophosome tissue also showed that ammonia was formed when trophosome tissue was exposed to nitrite (the product of nitrate reduction and the precursor of ammonia synthesis) (Fig. 4). Although uric acid was found in the trophosome and body wall of R. pachyptila, this compound was not eliminated into the seawater. Dinitrogen assimilation or production was not detected in either the blood or tissues of R. pachyptila or in the external milieu (Table 2).
Typically, the sum of the rates of elimination for all of the other nitrogenous metabolites measured did not exceed 10% of the nitrate uptake rate, even after 60 h of exposure to 40 μM nitrate. During 60 h of exposure to 40 μM nitrate, the ΔN was equivalent to a stoichiometric gain of 2.2 μmol of nitrogen g−1 h−1. Although this difference might be attributed to disequilibrium of nitrate pools, our measurements of the tissue nitrate pools suggest that this is unlikely. The nitrate concentrations in the trophosomes of R. pachyptila maintained in nitrate-depleted water were 27.8 ± 12.2 μmol g (wet weight) of tissue−1, which increased to 394.8 ± 84.2 μmol g (wet weight) of tissue−1 when 100 μM nitrate was added to the aquarium seawater (Table 2). At the observed rate of nitrate uptake by R. pachyptila (when it was exposed to 100 μM nitrates), a typical 50-g worm required roughly 2.5 h to accumulate 400 μM nitrate in its trophosome. Our observation that the rates of uptake and elimination mentioned above can be sustained for at least 60 h suggests that the observed differences are not attributable to storage of nitrate. Thus, there is a “sink” for nitrogenous compounds that we believe represents the fraction of ammonia that is assimilated into amino acids. The similarity between the CO2 uptake/NO3 uptake ratio of R. pachyptila and the C/N ratio of the association (9) is evidence that nitrogen is acquired at rates sufficient to meet the biosynthetic needs of this association (Table 1). In addition, activities of the enzymes responsible for incorporation of ammonia (22) have been found in the trophosome, indicating that there is a capacity to assimilate ammonia.
If dissimilatory nitrate reduction is the major mode of symbiont respiration, the rate of uptake of nitrate must be sufficient to meet the demands of the symbionts for an oxidant. Accordingly, one would expect the nitrate uptake rate to be relatively high, comparable to the oxygen uptake rate. One would also expect the availability of nitrate, which is governed both by the rate of nitrate uptake and by the storage of nitrate in the tissues, to affect symbiont oxidation of sulfide and fixation of carbon. If, however, the primary role of nitrate reduction is to be a source of nitrogen for biosynthesis, three major criteria must be met. First, nitrate uptake by R. pachyptila must occur at in situ seawater nitrate concentrations. Second, nitrate reduction by the symbionts must lead to formation of ammonia or another compound that can be assimilated by the symbionts and potentially by the host. Third, there must be a flux of nitrogen into the association (determined by the net flux of all nitrogenous metabolites) at a rate sufficient to support the biosynthetic needs of the association.
In a previous study Hentschel and Felbeck suggested that nitrate reduction by the symbionts sustains the association through periods of environmental hypoxia (16). It is known that dissimilatory nitrate reduction typically occurs in anoxic environments, and given the substantial oxygen uptake by the association and the high concentration of hemoglobin in the blood, it is unlikely that the internal milieu is anoxic. Furthermore, if reduction of nitrate were solely for respiration, one would expect an approximately 1:1 stoichiometric influx and efflux of nitrogenous compounds from the association. The rates of nitrogenous compound efflux measured in our experiments never approached the intake rates, even during and prior to several hours of experimentally induced hypoxia. Even if all of the nitrate is used by the symbionts for respiration and if it is assumed that the redox potential is equal to that of oxygen, the reduction of nitrate for respiration could at best meet 20 to 25% of the respiratory needs of the symbionts (as determined by comparison to the fraction of oxygen which is utilized by the symbionts during oxidation of sulfide) (Fig. 3). Additional compelling evidence is the strong correlation between sulfide uptake and oxygen uptake, which demonstrates the tight coupling between sulfide oxidation (via oxygen) by the symbionts. (Fig. 3D) and the lack of coupling between nitrate uptake and sulfide oxidation (Fig. 3C). In previous experiments we were able to sustain net inorganic carbon acquisition (and presumably net fixation), as well as sulfide uptake in the total absence of nitrate, which indicated that oxygen is a sufficient oxidant for R. pachyptila symbionts (13). These findings do not support the hypothesis that nitrate plays a role as an important oxidant for symbiont respiration.
It is possible that the reduction of nitrate may be coupled to respiration, as a minor oxidant, with concomitant production of ammonia (referred to as dissimilatory nitrate reduction to ammonia). This occurs in members of the genera Beggiatoa and Thioploca, which reduce nitrate during respiration and produce ammonia as an end product (24, 27). Dissimilatory nitrate reduction has also been observed in the Lucinoma aequizonata symbiosis (15). This scenario, although possible, seems unlikely in R. pachyptila given the evidence that dissimilatory nitrate reduction does not occur, as mentioned above.
The role of urate (uric acid) in the association described here has not been definitively resolved. It has been suggested that in R. pachyptila uric acid may play a role in nitrogen storage (7). In our nitrate repletion-depletion studies, the pools of uric acid in both trophosome samples and body wall samples did not differ significantly when we compared pre- and postnitrate treatments, suggesting that, unlike the sizes of the nitrate and ammonia pools, the sizes of the uric acid pools do not vary over several days (Table 2). In addition, uric acid was never found in the aquarium effluent, indicating that uric acid is probably not eliminated into the environment. The stability of the uric acid pools and the presence of uric acid in the host body wall suggest that uric acid may not be a product of bacterial metabolism but rather may be a product of host nitrogen metabolism. In some marine organisms, such as limpets, tissue uric acid concentrations vary seasonally and as a function of diet (32); the uric acid in R. pachyptila may be an intermediate or end product of protein catabolism and may not be utilized by the symbionts. In other symbiotic associations, such as Convoluta roscoffensis, the uric acid serves as a store of nitrogen for the symbionts (8). Experiments to determine the changes in the uric acid pools over a longer time course, as well as the utilization of uric acid by isolated symbiont preparations, are needed to determine the potential for utilization of uric acid by the bacteria. It is also possible that uric acid may also serve as an antioxidant and protect oxidant-sensitive pathways from both oxygen and nitrate (23, 26).
Accordingly, in our model of nitrogen flux for this association, nitrate is taken up by the worm (via an unknown mode of active transport), transported to the trophosome, and reduced by the symbionts to nitrite and then ammonia. We posit that ammonia is the end product of nitrate reduction and that fractions of both the nitrite and the ammonia produced by the symbionts diffuse from the trophosome into the vascular blood of the host, which results in the loss of a small amount of nitrite and ammonia to the environment via passive diffusion. The diffusion of ammonia into the vascular fluid allows for the possibility that the host may incorporate ammonia into amino acids via ammonia assimilatory pathways. This does not preclude the possibility that the host acquires nitrogen through some other means (e.g., digestion of symbionts). A fraction of the nitrogen incorporated by the host leads to formation and storage of urate in both the body wall (which is devoid of symbionts) and the trophosome. We also posit that the symbionts incorporate the majority of the ammonia into amino acids and that this incorporation is most responsible for the discrepancy in the nitrogen flux. At the rates observed under in situ conditions, the reduction of nitrate is sufficient to meet the biosynthetic demands of the association but cannot solely support the oxidative demands of the symbionts. Given the extraordinary biomass of R. pachyptila at many vents (33) and the rates of nitrate reduction inferred from our study, the reduction of nitrate and the subsequent incorporation into the tissues of the worms suggest that R. pachyptila may be the major primary producer (with respect to nitrogen) in the hydrothermal vent community.
ACKNOWLEDGMENTS
We thank the captains and crews of the RV New Horizon, RV Atlantis II and Atlantis, DSRV Alvin, RV Wecoma, RV Nadir, and DSRV Nautile. We also thank S. Goffredi, S. Powell, J. Freytag, M. Delacruz, and Kristie Klose for their tireless efforts, as well as R. Trench and R. Suarez for reviews and revision of the manuscript. Special thanks go to F. Gaill, A. Chave, and D. Manahan, chief scientists of the 1996, 1997, and 1998 expeditions.
Funding for this project was provided by NSF grants OCE-9301374 (to J.J.C.) and OCE-9632861 (to J.J.C.).
REFERENCES
- 1.Arp A J, Childress J J. Sulfide binding by the blood of the hydrothermal vent tube worm Riftia pachyptila. Science. 1983;219:295–297. doi: 10.1126/science.219.4582.295. [DOI] [PubMed] [Google Scholar]
- 2.Cavanaugh C M, Gardiner S L, Jones M L, Jannasch H W, Waterbury J B. Prokaryotic cells in the hydrothermal vent tube worm Riftia pachyptila: possible chemoautotrophic symbionts. Science. 1981;213:340–342. doi: 10.1126/science.213.4505.340. [DOI] [PubMed] [Google Scholar]
- 3.Childress J J, Arp A J, Fisher C R., Jr Metabolic and blood characteristics of the hydrothermal vent tube worm Riftia pachyptila. Mar Biol. 1984;83:109–124. [Google Scholar]
- 4.Childress J J, Fisher C R. The biology of hydrothermal vent animals: physiology, biochemistry, and autotrophic symbioses. Oceanogr Mar Biol Annu Rev. 1992;30:337–441. [Google Scholar]
- 5.Childress J J, Mickel T J. A motion compensated shipboard precision balance system. Deep-Sea Res. 1980;27:965–970. [Google Scholar]
- 6.Cole J A, Ferguson S J. The nitrogen and sulphur cycles. Cambridge, United Kingdom: Cambridge University Press; 1988. [Google Scholar]
- 7.De Cian M, Regnault M, Lallier F. Nitrogenous metabolites in tissues and circulating fluids of Riftia pachyptila. Cah Biol Mar. 1997;38:122. [Google Scholar]
- 8.Douglas A E. Uric-acid utilization in Platymonas-Convolutae and symbiotic Convoluta Roscoffensis. J Mar Biol Assoc U K. 1983;63:435–447. [Google Scholar]
- 8a.Felbeck H, Somero G N, Childress J J. Calvin-Benson cycle and sulfide oxidation enzymes in animals from sulfide-rich habitats. Nature. 1981;293:291–293. [Google Scholar]
- 9.Fisher C R, Childress J J, Arp A J, Brooks J M, Distel D, Favuzzi J A, Macko S A, Newton A, Powell M A, Somero G N, Soto T. Physiology, morphology, and biochemical composition of Riftia pachyptila at Rose Garden in 1985. Deep-Sea Res. 1988;35:1745–1758. [Google Scholar]
- 10.Fisher C R, Childress J J, Minnich E. Autotrophic carbon fixation by the chemoautotrophic symbionts of Riftia pachyptila. Biol Bull (Woods Hole) 1989;177:372–385. [Google Scholar]
- 11.Fossati P, Prencipe L, Berti G. Use of 3,5-dichloro-2-hydroxybenzenesulfonic acid/4-aminophenazone chromogenic system in direct enzymatic assay of uric acid in serum and urine. Clin Chem. 1980;26:227–231. [PubMed] [Google Scholar]
- 12.Gieskes J, Peretsman G. Water chemistry procedures aboard Joides Resolution—some comments. Ocean Drilling Program Tech Note. 1986;5:30–36. [Google Scholar]
- 13.Girguis P R, Childress J J. H+ equivalent elimination by the tubeworm Riftia pachyptila. Cah Biol Mar. 1998;39:295. [Google Scholar]
- 14.Goffredi S K, Childress J J, Desaulniers N T, Lee R W, Lallier F H, Hammond D. Inorganic carbon acquisition by the hydrothermal vent tubeworm Riftia pachyptila depends upon high external PCO2 and upon proton elimination by the worm. J Exp Biol. 1997;200:883–896. doi: 10.1242/jeb.200.5.883. [DOI] [PubMed] [Google Scholar]
- 15.Hentschel U, Hand S C, Felbeck H. The contribution of nitrate respiration to the energy budget of the symbiont containing clam Lucinoma aequizonata: a calorimetric study. J Exp Biol. 1996;199:427–433. doi: 10.1242/jeb.199.2.427. [DOI] [PubMed] [Google Scholar]
- 16.Hentschel U, Felbeck H. Nitrate respiration in the hydrothermal vent tubeworm Riftia pachyptila. Nature. 1993;366:338–340. [Google Scholar]
- 17.Johnson K S, Childress J J, Hessler R R, Sakamoto-Arnold C M, Beehler C L. Chemical and biological interactions in the Rose Garden hydrothermal vent field. Deep-Sea Res. 1988;35:1723–1744. [Google Scholar]
- 18.Jones M L. Riftia pachyptila, new genus, new species, the vestimentiferan tubeworm from the Galapagos Rift geothermal vents. Proc Biol Soc Wash. 1981;93:1295–1313. [Google Scholar]
- 19.Karlsson M, Karlberg B, Olsson R J O. Determination of nitrate in municipal waste water by UV spectroscopy. Anal Chim Acta. 1995;312:107–113. [Google Scholar]
- 20.Kochevar R E, Childress J J, Fisher C R, Minnich L. The methane mussel: roles of symbiont and host in the metabolic utilization of methane. Mar Biol. 1992;112:389–401. [Google Scholar]
- 21.Lee R W, Childress J J. Assimilation of inorganic nitrogen by marine invertebrates and their chemoautotrophic and methanotrophic symbionts. Appl Environ Microbiol. 1994;60:1852–1858. doi: 10.1128/aem.60.6.1852-1858.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee R W, Robinson J J, Cavanaugh C M. Pathways of inorganic nitrogen assimilation in chemoautotrophic bacteria-marine invertebrate symbioses: expression of host and symbiont glutamine synthetase. J Exp Biol. 1999;202:289–300. doi: 10.1242/jeb.202.3.289. [DOI] [PubMed] [Google Scholar]
- 23.Matsuo T, Ishikawa Y. Protective role of uric acid against photooxidative stress in the silkworm, Bombyx mori (Lepidoptera: Bombycidae) Appl Entomol Zool. 1999;34:481–484. [Google Scholar]
- 24.McHatton S C, Barry J P, Jannasch H W, Nelson D C. High nitrate concentrations in vacuolate, autotrophic marine Beggiatoa spp. Appl Environ Microbiol. 1996;62:954–958. doi: 10.1128/aem.62.3.954-958.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mickel T J, Childress J J. Effects of pressure and temperature on the EKG and heart rate of the hydrothermal vent crab Bythograea thermydron (Brachyura) Biol Bull (Woods Hole) 1982;162:70–82. [Google Scholar]
- 26.Mikami T, Yoshino Y, Ito A. Does a relationship exist between the urate pool in the body and lipid peroxidation during exercise? Free Radic Res. 2000;32:31–39. doi: 10.1080/10715760000300041. [DOI] [PubMed] [Google Scholar]
- 27.Nelson D. Physiology and biochemistry of filamentous sulfur bacteria. In: Schlegel H G, Bowien B, editors. Autotrophic bacteria. 1989. pp. 219–238. Science Tech, Madison, Wis. [Google Scholar]
- 28.Pospesel M A, Hentschel U, Felbeck H. Determination of nitrate in the blood of the hydrothermal vent tubeworm Riftia pachyptila using a bacterial nitrate reduction assay. Deep-Sea Res Part A Oceanogr Res Pap. 1998;45:2189–2200. [Google Scholar]
- 29.Powell M A, Somero G N. Adaptations to sulfide by hydrothermal vent animals: sites and mechanisms of detoxification and metabolism. Biol Bull (Woods Hole) 1986;171:274–290. [Google Scholar]
- 30.Rau G. Low 15N/14N in hydrothermal vent animals: ecological implications. Nature. 1981;289:484–485. [Google Scholar]
- 31.Sambrook J, Maniatis T, Fritsch E F. Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory; 1989. [Google Scholar]
- 32.Smith D C, Smith T P. Seasonal variation in soluble uric acid concentration in Littorina saxatilis. Hydrobiologia. 1998;378:187–191. [Google Scholar]
- 33.Tunnicliffe V. The biology of hydrothermal vents: ecology and evolution. Oceanogr Mar Biol Annu Rev. 1991;29:319–407. [Google Scholar]
- 34.Von Damm K L. Seafloor hydrothermal activity: black smoker chemistry and chimneys. Annu Rev Earth Planet Sci. 1990;18:173–204. [Google Scholar]
- 35.Willason S W, Johnson K S. A rapid, highly sensitive technique for the determination of ammonia in seawater. Mar Biol. 1986;91:285–290. [Google Scholar]