Abstract
Background
Irreversible electroporation (IRE) is a new local ablation technique for pancreatic cancer. The aim of this study is to analyse the safety and effectiveness of simultaneous gemcitabine and percutaneous CT-guided IRE for locally advanced pancreatic cancer (LAPC).
Materials and Methods
From October 2016 to January 2018, 61 patients with LAPC who received simultaneous gemcitabine and IRE therapy (GEM-IRE group, n = 31) or IRE alone therapy (IRE group, n = 30). Routine intravenous gemcitabine chemotherapy was performed 2 weeks after IRE in both groups.
Results
Technical success rates were 90.0% (27/30) and 93.3% (28/30) in the GEM-IRE and IRE groups. Compared with the IRE group, the GEM-IRE group exhibited longer overall survival (OS), local tumor progression free survival (LTPFS), and distant disease free survival (DDFS) from IRE (OS, 17.1 vs. 14.2 months, p=0.031; LTPFS, 14.6 vs. 10.2 months, p=0.045; DDFS, 15.4 vs. 11.7 months, p=0.071). Multivariate Cox regression analysis results suggested that tumor volume ≤37 cm3 and simultaneous gemcitabine with IRE were significant independent prognostic factors of OS, LTPFS, and DDFS. Four major adverse reactions occurred; all of them were resolved after symptomatic treatment.
Conclusions
Simultaneous gemcitabine and percutaneous CT-guided IRE therapy model was effective and well-tolerated therapeutic strategy in LAPC patients.
1. Introduction
It is projected that pancreatic cancer will become the second leading cause of death in the United States and Germany within the next decade [1, 2]. Approximately 80% of patients are diagnosed with locally advanced pancreatic cancer (LAPC), and in fewer than 20% of these patients, surgical resection is possible [3, 4], and a 5-year survival rate is less than 8% [5]. First-line chemotherapy regimens for LAPC include FOLFIRINOX, a combination of gemcitabine and albumin-bound paclitaxel, or just gemcitabine. However, the LAPC prognosis remains dismal, even with chemotherapy.
At present, minimally invasive treatments such as radiofrequency, microwave, and cryoablation have been demonstrated to be effective for LAPC patients [6–8]. These types of technology, however, have the potential to damage bile ducts, pancreatic ducts, peripancreatic vessels, and the duodenum, which may result in limited benefit [9, 10]. Irreversible electroporation (IRE) is a novel, minimally invasive ablation technique for soft tissue cancer, which uses high-voltage electrical pulses to develop irreversible small holes on the surface of the cell membrane, causing a disruption of homeostasis and cell death [11–14]. Currently, IRE has demonstrated distinct superiority and potency in the management of LAPC [15–18].
In recent years, there has been an explosion in the use of electrochemical therapy (ECT) in the treatment of solid tumors [19–21]. Usually, ECT combines poorly or not penetrating but strongly available cytotoxic agents with electroporation. Because it strengthens the transmission of molecules to the structure, ECT has a high potential for treating advanced solid tumors [22, 23]. In the reversible electroporation (RE) zone, cell membrane permeability due to electroporation can promote drug diffusion into the cells [24–27], which may improve antitumor efficacy. On the other hand, a preclinical study has shown that IRE enhanced gemcitabine delivery into the RE zone and thereby reduced local recurrence [28].
Based on the above studies, a retrospective study about simultaneous gemcitabine and percutaneous CT-guided IRE for LAPC was conducted. In this study, we evaluated overall survival (OS), local tumor progression free survival (LTPFS), distant disease free survival (DDFS), and objective response rate (ORR) and adverse events after combination therapy, towards a more effective treatment strategy for LAPC.
2. Materials and Methods
2.1. Patients
The patients received either simultaneous gemcitabine and percutaneous CT-guided IRE (GEM-IRE group) or IRE alone (IRE group). We executed this retrospective study with the approval of the Ethics Committee of Fuda Cancer Hospital and strict administration of the Declaration of Helsinki and the Declaration of Good Clinical Practice. We obtained written informed consent from each patient.
2.2. Inclusion and Exclusion Criteria
Inclusion Criteria: (1) patients older than 18 years; (2) expected survival >3 months; (3) tumor diameter ≤5 cm; (4) sufficient hepatic, renal, and bone marrow function were maintained; (5) performance status score of ≤2; (6) patients could not undergo surgical resection, or patients who could undergo surgery but prefer IRE treatment; (7) informed consent. Exclusion Criteria: (1) severe cardiac, pulmonary, and renal insufficiency or inability to tolerate general anesthesia with endotracheal intubation; (2) allergy to contrast media; (3) chemotherapy or radiotherapy within 1 month prior to the procedure; (4) history of epilepsy; and (5) history of heart arrhythmia, implantation of metallic stent or cardiac pacemaker.
2.3. Procedure
In all patients, preoperative bowel preparation was performed and anesthesia was inducted with etomidate (0.3 mg/kg), remifentanil (3–5 μg/kg, 1.2–1.6 mg/h), and cisatracurium besilate (0.1 mg/kg). Anesthesia was administered by injection of intravenous cisatracurium besilate, remifentanil, and propofol with sevoflurane inhalation. To avoid cardiac arrhythmias, an electrocardiogram (ECG)-gating device was synchronized with pulse delivery during the cardiac refractory period. To prevent generalized muscle contractions, complete muscle relaxants were administered immediately before IRE delivery.
In the GEM-IRE group, before IRE treatment, gemcitabine (1000 mg/m2) was given intravenously for 30 minutes. Once-weekly infusions of gemcitabine were administered for the first two weeks, followed by a one-week rest period. Subsequent cycles consisted of weekly injections for three continuous weeks, with a cycle of four weeks each. The therapy was maintained until disease progression, by mRECIST or intolerable toxicity [29]. Upon disease progression, palliative care was given.
During IRE, CT scanning and ultrasound were used to guide percutaneous insertion of the electrode. Pretreatment planning determines electrode-insertion mode, electrode number, and intraoperative parameters. The IRE parameters were set up as follows: 90-microsecond high-voltage (1500–3000 V), delivered between 9 pairs of paired monopolar electrodes with 2 cm exposed tips, totaling 90 pulses. Further treatment includes anti-infection treatments, stomach-preserving and liver protection therapies, and nutritional treatment.
2.4. Carbohydrate Antigen 19-9 (CA19-9)
Carbohydrate antigen 19-9 (CA19-9) was analyzed 30 days postintervention to evaluate the initial effect.
2.5. Technical Success
Technical success was considered to be the ability to successfully deliver pulses (at least 90) to ensure a change in current of at least 5 A relative to the initial 10 pulses.
2.6. Follow-Up and Response Assessment
Imaging follow-up was done using contrast-enhanced CT or magnetic resonance imaging (MRI) at 1, 3, and 6 months postintervention and then every 3 months. Imaging follow-up at 1 month evaluated the success of the technique. Imaging efficacy evaluation was based on mRECIST [29]. Complete response (CR) was considered as the absence of arterial enhancing lesions. A partial response (PR) was indicated by a 30% decrease in the size of the target lesion. Progressive disease (PD) was considered by a decrease of 20% in the size of the lesion. Stable disease (SD) was considered by lack of target lesions reduction to PR or increase to PD. We estimated the objective response rate (ORR) as ORR = (CR + PR)/total number × 100%.
2.7. OS
The OS was the length of time from diagnosis and IRE treatment to death from any cause. The LTPFS was considered as the time from IRE treatment to local tumor progression. DDFS was considered as the time from IRE treatment to distant metastasis.
2.8. Safety
Adverse events were assessed after 24 h using enhanced CT and ultrasound. Any adverse events that occurred within 30 days of treatment were graded depending on the CTCAE V4.0. CTCAE grades III-IV were classified as “severe.” The change of AST, ALT, and ALP and routine blood tests were taken before the procedure and were repeated 1, 3, 7, and 14 days after the procedure to evaluate intervention safety.
2.9. Statistical Analysis
Statistical analyses were performed with GraphPad Prism and SPSS 25.0 (IBM). We used the Fisher's exact test, the Mann–Whitney test, and the log-rank test to compare continuous data, categorical data, and survival curves. Wilcoxon matching was used to compare consecutive data tests in the same group. All statistical tests were two-sided. A P value of 0.05 indicates statistical significance.
3. Results
3.1. Patient Parameters
From October 2016 to January 2018, 84 patients were included in our study (Figure 1). Among them, 23 patients were excluded because of chemoradiation within 1 month prior to the procedure (n = 12), iodine-125 seed treatment (n = 6), with metal duct stent or heart pacemaker (n = 3), and severe arrhythmia (n = 2). Overall, the GEM-IRE group (31 patients) received gemcitabine and IRE simultaneously, and the IRE group (30 patients) received IRE alone. During the follow-up, 1 patient lost to follow in the GEM-IRE group. Finally, 60 patients were analyzed. The clinical features of the 60 included patients are given in Table 1.
Figure 1.
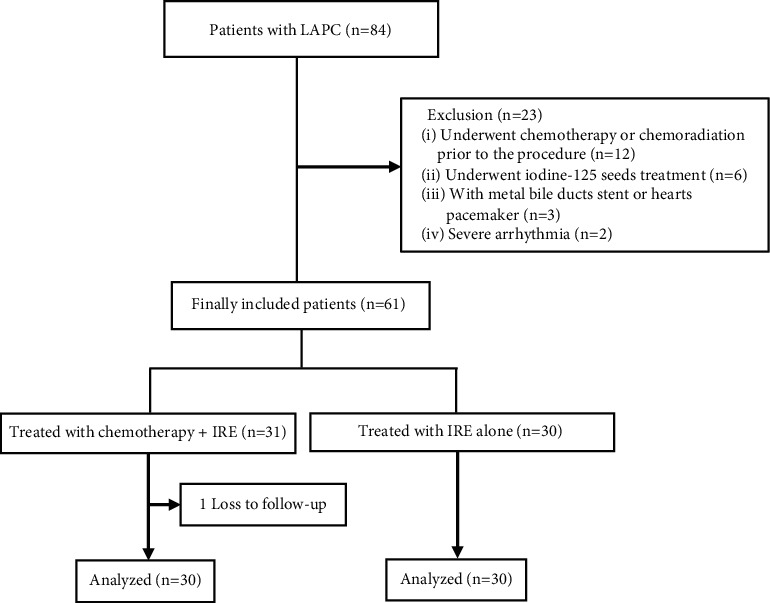
Flow diagram of patients included in the study.
Table 1.
Patients characteristics (n (%)).
| Characteristic | GEM-IRE group (n = 30) | IRE group (n = 30) | P value |
|---|---|---|---|
| Sex | |||
| F | 16 (53.3) | 18 (60.0) | 0.795 |
| M | 14 (46.7) | 12 (40) | |
|
| |||
| Mean age (years) | 61 (43–78) | 67 (47–75) | |
| Lesion size (cm) | 3.8 (2.1–4.7) | 4.1 (2.3–4.6) | |
| Tumor location | 0.763 | ||
| Head | 7 (23.3) | 8 (26.7) | |
| Body | 12 (40.0) | 15 (50.0) | |
| Tail | 7(23.3) | 4 (13.3) | |
| Uncinate process | 4 (13.4) | 3 (10.0) | |
|
| |||
| Preoperative surgical therapy | 0.624 | ||
| None | 23 (76.6) | 25 (83.4) | |
| Gastrojejunostomy | 2 (6.7) | 1 (3.3) | |
| Hepaticojejunostomy | 1 (3.3) | 1 (3.3) | |
| Double bypass | 2 (6.7) | 0 (0.0) | |
| Others | 2 (6.7) | 3 (10.0) | |
|
| |||
| PS | 0.627 | ||
| 0 | 8 (26.7) | 5 (16.7) | |
| 1 | 18 (60.0) | 22 (73.3) | |
| 2 | 4 (13.3) | 3 (10.0) | |
PS: performance status.
3.2. Tumor Markers by CA19-9
The positive serum CA19-9 level rates in the GEM-IRE and IRE groups were 73.3% (22/30) and 80.0% (24/30), respectively. The CA19-9 levels in the GEM-IRE and IRE groups before treatment had significantly decreased by 1 month posttreatment (1981.5 vs. 752.5, p=0.005, and 2376 vs. 1554, p=0.012, respectively); no statistically significant difference in both groups before treatment or 1 month after treatment (p=0.567 and p=0.053, respectively; Figure 2).
Figure 2.
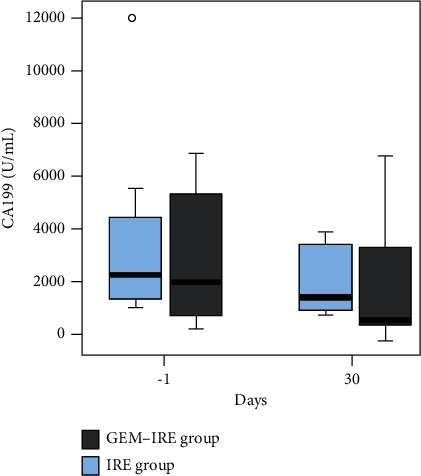
CA19-9 levels before and after treatment.
3.3. Treatment Success Rate and Tumor Response
There was no operative mortality within 90 days. Technical success rates were 90.0% (27/30) in the GEM-IRE group and 93.3% in the IRE group (28/30), (p > 0.05). The 1-, 3-, and 6-month ORRs for the GEM-IRE group vs. the IRE group were 93.3%, 83.3%, and 65.5% vs. 86.6%, 70.0%, and 53.8%, respectively. A representative CT result from a GEM-IRE patient is displayed in Figure 3.
Figure 3.
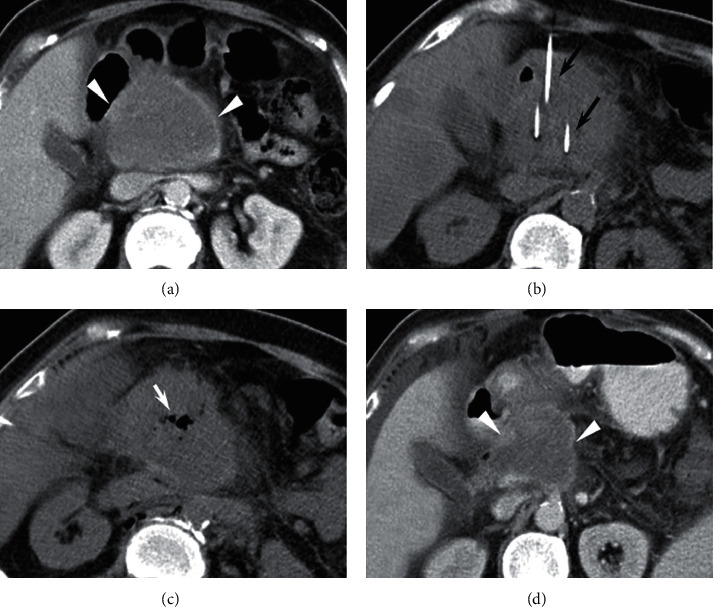
A 66-year-old female with unresectable pancreatic head tumors treated with gemcitabine and irreversible electroporation (GEM-IRE) simultaneously. (a) Preoperative contrast-enhanced CT showed tumor size (7.4 × 6.2 cm). (b) Arrows indicate electrode probes. (c) Bubbles were seen immediately after IRE. (d) 3 months after operation, preoperative contrast-enhanced CT revealed a tumor size of 4.6 × 5.1 cm.
3.4. OS
The median follow-up time was 20.4 months (3.0–28.4 months). The median OS from diagnosis in the GEM-IRE group was significantly longer than that of those in the IRE group (21.5 vs. 16.7 months, respectively; hazard ratio (HR), 0.520; p=0.019; Figure 4(a)). The median OS from IRE was also significantly longer in the GEM-IRE group than that of those in the IRE group (17.1 vs. 14.2 months, respectively; HR, 0.54; p=0.031; Figure 4(b)). According to univariate analysis, tumor size (HR = 1.925, 95% CI, 1.056–3.559, p=0.046), tumor volume (HR = 2.486, 95% CI, 1.245–4.325, p=0.009), local recurrence (HR = 3.522, 95% CI, 1.362–7.023, p=0.006), and IRE treatment (HR = 0.389, 95% CI, 0.178–0.952, p=0.045) were related to OS. In addition, the independent prognostic factors were tumor volume (HR = 2.675, 95% CI, 1.230–7.215, p=0.035) and IRE treatment (HR = 0.422, 95% CI, 0.157–0.958, p=0.007) (Table 2).
Figure 4.
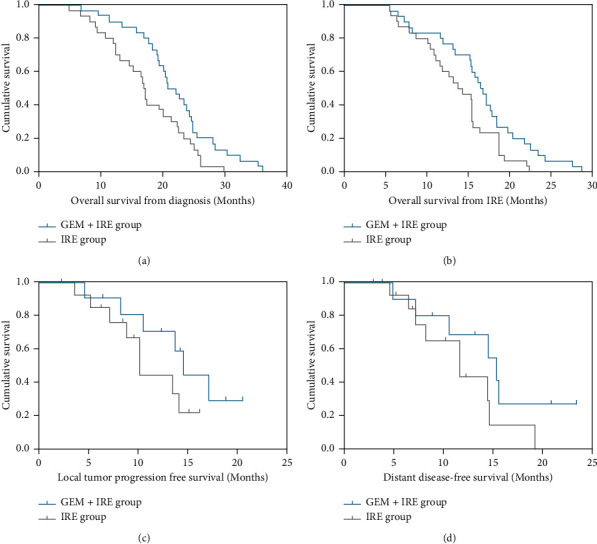
Kaplan–Meier survival curves. (a) The median OS from diagnosis. (b) The median OS from IRE. (c) Local tumor progression free survival from IRE. (d) Distant disease-free survival from IRE.
Table 2.
Univariate and multivariate analyses of overall survival.
| Characteristic | Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | ||
| Age | ≤60/>60 | 1.245 (0.625–2.856) | 0.156 | ||
| Sex | Female/male | 0.653 (0.302–1.127) | 0.129 | ||
| Tumor site | Head/body/tail | 0.936 (0.648–1.582) | 0.832 | ||
| Tumor size (cm) | ≤5/>5 | 1.925 (1.056, 3.559) | 0.046 | 1.369 (0.625, 2.848) | 0.521 |
| Tumor volume (cm3) | ≤37/>37 | 2.486 (1.245–4.325) | 0.009 | 2.675 (1.230–7.215) | 0.035 |
| Local recurrence | ≤6 mo/>6 mo | 3.522 (1.362–7.023) | 0.006 | 0.542 (0.308–2.034) | 0.384 |
| WBC (×109) | ≤10/>10 | 1.352 (0.475–2.322) | 0.789 | ||
| HGB (g/L) | ≤120/>120 | 1.086 (0.365–1.458) | 0.426 | ||
| PLT (× 109) | ≤300/>300 | 1.335 (0.468–3.589) | 0.459 | ||
| ALT (U/L) | ≤40/>40 | 0.895 (0.257–1.752) | 0.658 | ||
| AST (U/L) | ≤40/>40 | 0.235 (0.128–1.329) | 0.457 | ||
| ALP (U/L) | ≤100/>100 | 0.965 (0.042–1.436) | 0.689 | ||
| CA19-9 level | ≤35/>35 | 1.350 (0.618–3.572) | 0.245 | ||
| Complications | Yes/no | 0.621 (0.321–1.126) | 0.096 | ||
| IRE | With/without GEM | 0.389 (0.178–0.952) | 0.045 | 0.422 (0.157–0.958) | 0.007 |
IRE: irreversible electroporation; GEM: gemcitabine; HR: hazard ratio; WBC: white blood cell count; HGB: hemoglobin; PLT: platelet count; ALT: alanine transaminase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; CA19-9: carbohydrate antigen 19-9.
3.5. LTPFS
A total of 25 patients experienced tumor progression, including 11 (36.7%) patients in the GEM-IRE group and 14 (46.6%) patients in the IRE group (p=0.436). The median LTPFS for patients in GEM-IRE and IRE groups were 14.6 months and 10.2 months, respectively (p=0.045, Figure 4(c)). Univariate analysis for LTPFS revealed that tumor volume (HR = 2.356, 95% CI, 1.367–3.161, p=0.001), local recurrence (HR = 0.445, 95% CI, 0.245–0.975, p=0.024), CA19-9 level (HR = 2.156, 95% CI, 1.034–4.265, p=0.045), and IRE treatment (HR = 0.545, 95% CI, 0.326–1.051, p=0.047) were associated with LTPFS. In addition, the independent prognostic factors were tumor volume (HR = 1.683, 95% CI, 1.035–3.191, p=0.032) and IRE treatment (HR = 0.556, 95% CI, 0.315–1.260, p=0.043) (Table 3).
Table 3.
Univariate and multivariate analyses of local tumor progression free survival.
| Characteristic | Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | ||
| Age | ≤60/>60 | 0.654 (0.565–1.818) | 0.185 | ||
| Sex | Female/male | 1.588 (0.927–2.579) | 0.056 | ||
| Tumor site | Head/body/tail | 1.167 (0.6987–1.640) | 0.960 | ||
| Tumor size (cm) | ≤5/>5 | 1.154 (0.776–1.705) | 0.512 | ||
| Tumor volume (cm3) | ≤37/>37 | 2.356 (1.367–3.161) | 0.001 | 1.683 (1.035–3.191) | 0.032 |
| Local recurrence | ≤6 mo/>6 mo | 0.445 (0.245–0.975) | 0.024 | 0.526 (0.225–1.325) | 0.151 |
| WBC (×109) | ≤10/>10 | 1.154 (0.479–2.658) | 0.811 | ||
| HGB (g/L) | ≤120/>120 | 0.673 (0.318–1.535) | 0.369 | ||
| PLT (×109) | ≤300/>300 | 0.623 (0.256–1.338) | 0.206 | ||
| ALT (U/L) | ≤40/>40 | 0.741 (0.325–1.437) | 0.356 | ||
| AST (U/L) | ≤40/>40 | 0.678 (0.328–1.478) | 0.246 | ||
| ALP (U/L) | ≤100/>100 | 0.823 (0.446–1.489) | 0.744 | ||
| CA19-9 level | ≤35/>35 | 2.156 (1.034–4.265) | 0.045 | 1.916 (0.931–3.568) | 0.078 |
| Complications | Yes/no | 0.645 (0.334–1.451) | 0.095 | ||
| IRE | With/without GEM | 0.545 (0.326–1.051) | 0.047 | 0.556 (0.315–1.260) | 0.043 |
IRE: irreversible electroporation; GEM: gemcitabine; HR: hazard ratio; WBC: white blood cell count; HGB: hemoglobin; PLT: platelet count; ALT: alanine transaminase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; CA19-9: carbohydrate antigen 19-9.
3.6. DDFS
The median DDFS were 15.4 months and 11.7 months in the GEM-IRE and IRE groups, respectively (p=0.071, Figure 4(d)). According to the univariate and multivariate analyses, tumor volume (HR = 2.364, 95% CI, 1.059–4.685, p=0.007), local recurrence (HR = 3.432, 95% CI, 1.406–8.125, p=0.006), and IER treatment (HR = 0.326, 95% CI, 0.192–0.752, p=0.014) were associated with DDFS. Moreover, tumor volume (HR = 2.856, 95% CI, 1.196–7.398, p=0.024) and IRE treatment (HR = 0.385, 95% CI, 0.202–0.654, p=0.021) were found to be an independent favourable factor of DDFS (Table 4).
Table 4.
Univariate and multivariate analyses of distance disease-free survival.
| Characteristic | Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|---|
| HR (95% CI) | P value | HR (95% CI) | P value | ||
| Age | ≤60/>60 | 1.214 (0.985–1.125) | 0.421 | ||
| Sex | Female/male | 0.621 (0.34–1.263) | 0.127 | ||
| Tumor site | Head/body/tail | 0.772 (0.438–1.248) | 0.337 | ||
| Tumor size (cm) | 1.187 (0.674–2.062 | 0.543 | |||
| Tumor volume (cm3) | ≤37/>37 | 2.364 (1.059–4.685) | 0.007 | 2.856 (1.196–7.398) | 0.024 |
| Local recurrence | ≤6 mo/>6 mo | 3.432 (1.406–8.125) | 0.006 | 0.523 (0.135–1.420) | 0.231 |
| WBC (×109) | ≤10/>10 | 1.128 (0.364–2.825) | 0.858 | ||
| HGB (g/L) | ≤120/>120 | 0.644 (0.345–1.670) | 0.403 | ||
| PLT (×109) | ≤300/>300 | 1.218 (0.578–2.567) | 0.628 | ||
| ALT (U/L) | ≤40/>40 | 0.767 (0.358–1.716) | 0.504 | ||
| AST (U/L) | ≤40/>40 | 0.484 (0.205–1.561) | 0.176 | ||
| ALP (U/L) | ≤100/>100 | 0.921 (0.546–1.950) | 0.703 | ||
| CA19-9 level | ≤35/>35 | 1.638 (0.720–3.882) | 0.225 | ||
| Complications | Yes/no | 1.382 (0.678–2.821) | 0.363 | ||
| IRE | With/without GEM | 0.326 (0.192–0.752) | 0.014 | 0.385 (0.202–0.654) | 0.021 |
IRE: irreversible electroporation; GEM: gemcitabine; HR: hazard ratio; WBC: white blood cell count; HGB: hemoglobin; PLT: platelet count; ALT: alanine transaminase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; CA19-9: carbohydrate antigen 19-9.
3.7. Safety
Within 30 days post-IRE, there were 16 minor adverse reactions and 4 major adverse reactions (Tables 5 and 6). The major adverse reactions included two cases of pancreatitis (grade III), one case of severe neutropenia (grade IV), and one case of gastroduodenal artery hemorrhage (grade IV) in the GEM-IRE group. The case of severe neutropenia was diagnosed on day 5 and that of gastroduodenal artery hemorrhage on day 11 after IRE. After a subcutaneous injection of 300 μg of recombinant human granulocyte colony-stimulating factor (G-CSF), the neutrophil level increased to 5.6 × 109/L within 24 hours. The gastroduodenal artery hemorrhage cases were controlled by vascular interventional embolization with 560–710 μm of gelatin sponge particles and 2 mm and 3 mm coils. The differences in the frequencies of adverse events were not statistically significant in both groups (p > 0.05 for all; Table 6). The 90-day mortality rate was 0% overall.
Table 5.
The type and treatment of adverse reactions (n = 60).
| Adverse reactions | Grade I/II | Grade III | Grade IV | Treatment |
|---|---|---|---|---|
| Hemoglobin reduction | 37 | 0 | 0 | NA |
| Leukocyte reduction | 6 | 0 | 0 | NA |
| Neutropenia | 4 | 0 | 1 | Human granulocyte colony-stimulating factor |
| Thrombocytopenia | 27 | 0 | 0 | NA |
| Transient elevation of myocardial enzyme | 22 | 0 | 0 | NA |
| Proteinuria | 1 | 0 | 0 | NA |
| Hypokalemia | 3 | 0 | 0 | NA |
| Pancreatitis | 0 | 2 | 0 | Drainage and antibiotics |
| Bleeding from duodenal ulcer | 0 | 0 | 1 | Interventional embolization |
| Fever | 10 | 0 | 0 | Antibiotics |
| Diarrhea | 1 | 0 | 0 | Pancreatic enzyme suppletion |
| Nausea and vomiting | 9 | 0 | 0 | Antiemetics |
| Infection | 3 | 0 | 0 | Antibiotics and drainage |
| Abdominal pain | 39 | 0 | 0 | Oral analgesics |
| Loss of appetite and | 9 | 0 | 0 | Nasojejunal tube feeding |
| Mild ascites | 27 | 0 | 0 | NA |
| Mild pleural effusion | 8 | 0 | 0 | NA |
| Abdominal distention | 12 | 0 | 0 | NA |
NA: no receiving further treatment.
Table 6.
Adverse reaction rates after treatment (n (%)).
| Characteristic | GEM-IRE group (n = 30) | IRE group (n = 30) | P value |
|---|---|---|---|
| Hemoglobin reduction | 22 (73.3) | 15 (50.0) | 0.110 |
| Leukocyte reduction | 4 (13.3) | 2 (6.7) | 0.671 |
| Neutropenia | 2 (6.7) | 3 (10.0) | 1.000 |
| Thrombocytopenia | 15 (50.0) | 12 (40.0) | 0.604 |
| Transient elevation of myocardial enzyme | 14 (46.7) | 8 (26.7) | 0.180 |
| Proteinuria | 1 (3.3) | 0 (0.0) | 1.000 |
| Hypokalemia | 1 (3.3) | 2 (6.7) | 1.000 |
| Pancreatitis | 1 (3.3) | 1 (3.3) | 1.000 |
| Bleeding from duodenal ulcer | 1 (3.3) | 0 (0.0) | 1.000 |
| Fever | 6 (20.0) | 4 (13.3) | 0.731 |
| Diarrhea | 1 (3.3) | 0 (0.0) | 1.000 |
| Nausea and vomiting | 6 (20.0) | 3 (10.0) | 0.472 |
| Infection | 2 (6.7) | 1 (3.3) | 1.000 |
| Abdominal pain | 5 (16.7) | 7 (23.3) | 0.748 |
| Loss of appetite and/or reduced intake | 7 (23.3) | 2 (6.7) | 0.145 |
| Mild ascites | 15 (50.0) | 12 (40.0) | 0.604 |
| Mild pleural effusion | 5 (16.7) | 3 (10.0) | 0.706 |
| Abdominal distention | 8 (26.7) | 4 (13.3) | 0.333 |
For routine blood tests, the postoperative hemoglobin level continued to significantly decrease in both groups on days 3 and 7 (p < 0.000, Figure 5(a)). The neutrophil counts showed a significant increase on day 1 (p < 0.000, Figure 5(b)); however, the levels recovered to preoperative levels by day 3, 7. There were significant differences in the platelet counts (p=0.0035, Figure 5(c)) and white blood cell counts (p=0.0179, Figure 5(d)) after treatment between the two groups at days 3 and 7 (p=0.0035) (Figure 5).
Figure 5.
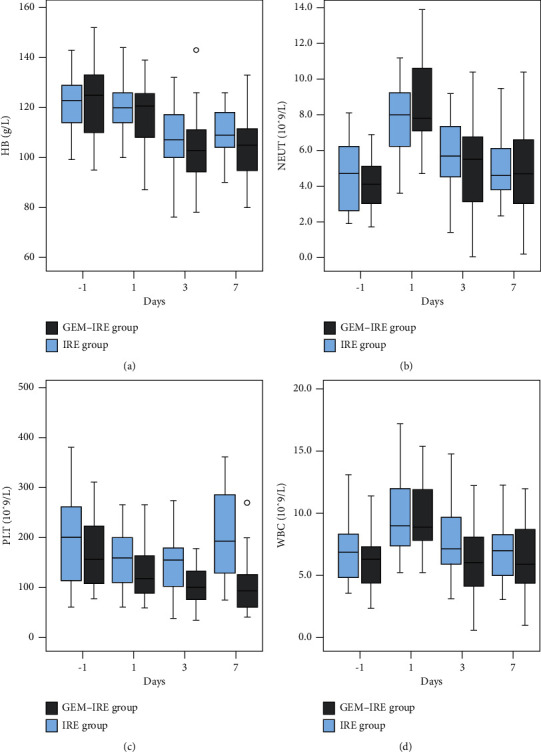
Changes in routine blood tests before and after treatment in both groups. (a) Changes in hemoglobin levels. (b) Changes of neutrophil counts. (c) Changes in platelet count. (d) Changes in white blood cell count.
For liver function, on postoperation day 1, ALT (Figure 6(a)) and AST (Figure 6(b)) values for both groups indicated a significant rising (p < 0.000 for both), but the levels decreased rapidly on day 3 and recovered to preoperative levels by day 7. Interestingly, the two groups did not differ statistically significantly (p > 0.05 for all). No obvious changes in alkaline phosphatase (ALP) (Figure 6(c)) were observed after treatment.
Figure 6.
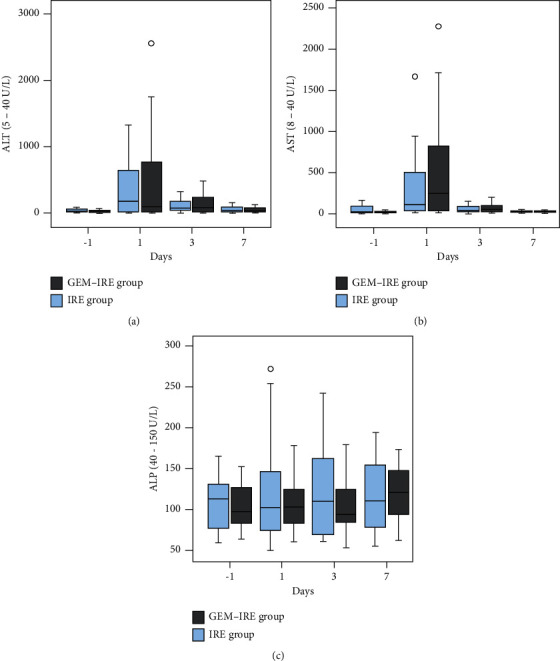
Changes in liver function before and after treatment. (a) ALT levels. (b) AST levels. (c) Changes in ALP levels.
4. Discussion
Recent chemotherapy regimens have improved OS for LAPC patients only by single-digit months [30]. Due to the insensitivity to conventional chemotherapy, gemcitabine administration is suppressed by the peritumor stroma and pancreatic cancer patients with low pancreatic hENT1 levels exhibit a significantly lower response to gemcitabine. To overcome this obstacle, ECT was developed. ECT utilizes high-intensity electrical pulses to enhance cell membrane permeability to injected chemotherapies. Although ECT has been applied to solid tumors for the past 2 decades and has been proven effective [31, 32], this was the first study to investigate simultaneous gemcitabine combination with IRE. Recent studies suggest that IRE combination with chemotherapy improves survival, but they were all induction chemotherapy regimens before IRE or postoperative adjuvant chemotherapy [33–35].
Our study was the first to demonstrate the antitumor effects of simultaneously combining gemcitabine with IRE. Our data showed that the GEM-IRE group had a longer OS (21.5 months). According to statistics, the median OS ranged from 17.9–27.0 months in some previous retrospective studies [34, 36–38]. In the aforementioned retrospective studies, patients who underwent induction chemotherapy achieved stable disease. Thus, simultaneous gemcitabine and IRE treatment had some survival benefit regardless of patient condition. Using percutaneous IRE as first-line treatment for LAPC without prior systemic treatment, Mansson et al. [39] found that the median OS after diagnosis was 13.3 months compared to 21.5 months in our study, indicating that simultaneous gemcitabine and IRE achieved better survival than percutaneous IRE as first-line LAPC treatment.
Interestingly, although the treatment success rate was similar within both groups, the short-term tumor response in the GEM-IRE group was higher than in the IRE group. Therefore, this study showed that the simultaneous treatment of gemcitabine and IRE is a better option for patients with LAPC. The main reason for this phenomenon might be that the peak plasma values of gemcitabine and its deaminated metabolite occurr within 30 minutes of injection. Continued treatment with IRE at this point prolonged its peak value and enhanced drug toxicity. This is consistent with findings by Shamseddine et al. that intra-arterial plasma gemcitabine peaks at 30 minutes [40]. Membrane permeation by electroporation enhances drug entry into the cell, enhancing cytotoxicity [26, 27, 41, 42]. These factors highlight the potential value of ECT in LAPC treatment.
IRE enhances drug transmission to cells via breaking up the dense mesenchymal tissues of tumors [28, 43]. Considering LAPC's high heterogeneity, local control of the tumor by RE ablation followed by neoadjuvant chemotherapy may improve survival rates. Thus, neoadjuvant chemotherapy followed by simultaneous gemcitabine and IRE may benefit LAPC patients more.
In terms of safety, most of the postoperative adverse reactions seen in this study were classified as grade I or II based on CTCAE V4.0 standards, while 4 cases were considered severe (grade III or grade IV). Between the 2 groups, there were no significant differences of adverse reactions (p > 0.05). Although IRE is known to preserve the structure of blood vessels, we observed a case of hemorrhage in the gastroduodenal artery during follow-up. Similar complications have been reported in other studies of about 4–7% [33–35, 44]. This may be because the duodenum was infiltrated by a pancreatic tumor and IRE ablation caused a duodenal ulcer, allowing gastric acid exudation and, ultimately, blood vessel rupture and bleeding. Thus, the combined use of laparotomy and gastrointestinal anastomosis may be an effective preventive method. Additionally, after the first postintervention day, ALT and AST levels increased significantly, suggesting that neither treatment has long-term effects on liver function and that the combination therapy was safe for LAPC treatment.
The findings of our study have a few limitations. First is small sample size. In the future, multicenter, prospective, randomized controlled, large-sample clinical studies are needed to support our results. Second, our study did not directly compare our treatments with conventional chemotherapy, including FOLFIRINOX, gemcitabine-based chemotherapy. Thus, larger, multicenter studies are being developed to support our results.
Acknowledgments
The authors appreciate the support and cooperation of all our patients. The authors are grateful for the assistance of our hospital staff. This study was funded with the support of the International Science Foundation of Fuda Cancer Hospital (No. Y2022-MS-02).
Contributor Information
Mao Lin, Email: linmaogz@163.com.
Lizhi Niu, Email: niuboshi@fudahospital.com.
Data Availability
All data are available upon reasonable request.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Rahib L., Smith B. D., Aizenberg R., Rosenzweig A. B., Fleshman J. M., Matrisian L. M. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Research . 2014;74(11):2913–2921. doi: 10.1158/0008-5472.CAN-14-0155. [DOI] [PubMed] [Google Scholar]
- 2.Quante A. S., Ming C., Rottmann M., et al. Projections of cancer incidence and cancer-related deaths in Germany by 2020 and 2030. Cancer Medicine . 2016;5(9):2649–2656. doi: 10.1002/cam4.767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Al Efishat M., Wolfgang C. L., Weiss M. J. Stage III pancreatic cancer and the role of irreversible electroporation. BMJ . 2015;350:p. h521. doi: 10.1136/bmj.h521. [DOI] [PubMed] [Google Scholar]
- 4.Sugumar K., Hurtado A., Naik I., et al. Multimodal therapy with or without irreversible electroporation for unresectable locally advanced pancreatic adenocarcinoma: a systematic review and meta-analysis. International Hepato-Pancreato-Biliary Association . 2022;24(5):586–595. doi: 10.1016/j.hpb.2021.12.014. [DOI] [PubMed] [Google Scholar]
- 5.Siegel R. L., Miller K. D., Jemal A. Cancer statistics. CA: A Cancer Journal for Clinicians . 2017;67(1):7–30. doi: 10.3322/caac.21387. [DOI] [PubMed] [Google Scholar]
- 6.Iancu I., Bartoș A., Cioltean C. L., Breazu C., Iancu C., Bartoș D. Role of radio-ablative technique for optimizing the survival of patients with locally advanced pancreatic adenocarcinoma (Review) Experimental and Therapeutic Medicine . 2021;22(2):p. 853. doi: 10.3892/etm.2021.10285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Narayanan G., Daye D., Wilson N. M., Noman R., Mahendra A. M., Doshi M. H. Ablation in pancreatic cancer: past, present and future. Cancers . 2021;11:p. 2511. doi: 10.3390/cancers13112511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wu Y., Gu Y., Zhang B., Zhou X., Li Y., Qian Z. Laparoscopic ultrasonography-guided cryoablation of locally advanced pancreatic cancer: a preliminary report. Japanese Journal of Radiology . 2022;40(1):86–93. doi: 10.1007/s11604-021-01175-9. [DOI] [PubMed] [Google Scholar]
- 9.Casadei R., Ricci C., Pezzilli R., et al. A prospective study on radiofrequency ablation locally advanced pancreatic cancer. Hepatobiliary and Pancreatic Diseases International . 2010;9(3):306–311. [PubMed] [Google Scholar]
- 10.Girelli R., Frigerio I., Salvia R., Barbi E., Tinazzi Martini P., Bassi C. Feasibility and safety of radiofrequency ablation for locally advanced pancreatic cancer. British Journal of Surgery . 2010;97(2):220–225. doi: 10.1002/bjs.6800. [DOI] [PubMed] [Google Scholar]
- 11.Lafranceschina S., Brunetti O., Delvecchio A., et al. Systematic review of irreversible electroporation role in management of locally advanced pancreatic cancer. Cancers . 2019;11 doi: 10.3390/cancers11111718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Narayanan G., Bilimoria M. M., Hosein P. J., Su Z., Mortimer K. M., Martin R. C. G. Multicenter randomized controlled trial and registry study to assess the safety and efficacy of the NanoKnife® system for the ablation of stage 3 pancreatic adenocarcinoma: overview of study protocols. BMC Cancer . 2021;21(1):p. 78. doi: 10.1186/s12885-021-08474-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rai Z. L., Feakins R., Pallett L. J., Manas D., Davidson B. R. Irreversible electroporation (IRE) in locally advanced pancreatic cancer: a review of current clinical outcomes, mechanism of action and opportunities for synergistic therapy. Journal of Clinical Medicine . 2021;10(8):p. 1609. doi: 10.3390/jcm10081609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ma Y., Chen Z., Liang B., et al. Irreversible electroporation for hepatocellular carcinoma abutting the diaphragm: a prospective single-center study. Journal of Clinical and Translational Hepatology . 2022;10(2):190–196. doi: 10.14218/jcth.2021.00019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lin M., Zhang X., Liang S., et al. Irreversible electroporation plus allogenic Vγ9Vδ2 T cells enhances antitumor effect for locally advanced pancreatic cancer patients. Signal Transduction and Targeted Therapy . 2020;5(1):p. 215. doi: 10.1038/s41392-020-00260-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ma Y. Y., Leng Y., Xing Y. L., Li H. M., Chen J. B., Niu L. Z. Gemcitabine plus concurrent irreversible electroporation vs gemcitabine alone for locally advanced pancreatic cancer. World Journal of Clinical Cases . 2020;8(22):5564–5575. doi: 10.12998/wjcc.v8.i22.5564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ma Y., Chen Z., Zhu W., et al. Chemotherapy plus concurrent irreversible electroporation improved local tumor control in unresectable hilar cholangiocarcinoma compared with chemotherapy alone. International Journal of Hyperthermia . 2021;38(1):1512–1518. doi: 10.1080/02656736.2021.1991008. [DOI] [PubMed] [Google Scholar]
- 18.Garnier J., Turrini O., Chretien A. S., Olive D. Local ablative therapy associated with immunotherapy in locally advanced pancreatic cancer: a solution to overcome the double trouble?-a comprehensive review. Journal of Clinical Medicine . 2022;11(7):p. 1948. doi: 10.3390/jcm11071948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tafuto S., Von Arx C., De Divitiis C., et al. Electrochemotherapy as a new approach on pancreatic cancer and on liver metastases. International Journal of Surgery . 2015;21:S78–S82. doi: 10.1016/j.ijsu.2015.04.095. [DOI] [PubMed] [Google Scholar]
- 20.Coletti L., Battaglia V., De Simone P., Turturici L., Bartolozzi C., Filipponi F. Safety and feasibility of electrochemotherapy in patients with unresectable colorectal liver metastases: a pilot study. International Journal of Surgery . 2017;44:26–32. doi: 10.1016/j.ijsu.2017.06.033. [DOI] [PubMed] [Google Scholar]
- 21.Matthiessen L. W., Keshtgar M., Curatolo P., et al. Electrochemotherapy for breast cancer-results from the INSPECT database. Clinical Breast Cancer . 2018;18(5):e909–e917. doi: 10.1016/j.clbc.2018.03.007. [DOI] [PubMed] [Google Scholar]
- 22.Cannon R., Ellis S., Hayes D., Narayanan G., Martin R. C. Safety and early efficacy of irreversible electroporation for hepatic tumors in proximity to vital structures. Journal of Surgical Oncology . 2013;107(5):544–549. doi: 10.1002/jso.23280. [DOI] [PubMed] [Google Scholar]
- 23.Martin R. C. G., McFarland K., Ellis S., Velanovich V. Irreversible electroporation in locally advanced pancreatic cancer: potential improved overall survival. Annals of Surgical Oncology . 2013;20:S443–S449. doi: 10.1245/s10434-012-2736-1. [DOI] [PubMed] [Google Scholar]
- 24.Granot Y., Rubinsky B. Mass transfer model for drug delivery in tissue cells with reversible electroporation. International Journal of Heat and Mass Transfer . 2008;51(23-24):5610–5616. doi: 10.1016/j.ijheatmasstransfer.2008.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Escobar-Chavez J. J., Bonilla-Martinez D., Villegas-Gonzalez M. A., Revilla-Vazquez A. L. Electroporation as an efficient physical enhancer for skin drug delivery. The Journal of Clinical Pharmacology . 2009;49(11):1262–1283. doi: 10.1177/0091270009344984. [DOI] [PubMed] [Google Scholar]
- 26.Tamzali Y., Borde L., Rols M. P., Golzio M., Lyazrhi F., Teissie J. Successful treatment of equine sarcoids with cisplatin electrochemotherapy: a retrospective study of 48 cases. Equine Veterinary Journal . 2012;44(2):214–220. doi: 10.1111/j.2042-3306.2011.00425.x. [DOI] [PubMed] [Google Scholar]
- 27.Gibot L., Wasungu L., Teissie J., Rols M. P. Antitumor drug delivery in multicellular spheroids by electropermeabilization. Journal of Controlled Release . 2013;167(2):138–147. doi: 10.1016/j.jconrel.2013.01.021. [DOI] [PubMed] [Google Scholar]
- 28.Bhutiani N., Agle S., Li Y., Li S., Martin R. C. Irreversible electroporation enhances delivery of gemcitabine to pancreatic adenocarcinoma. Journal of Surgical Oncology . 2016;114(2):181–186. doi: 10.1002/jso.24288. [DOI] [PubMed] [Google Scholar]
- 29.Lencioni R., Llovet J. M. Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Seminars in Liver Disease . 2010;30(1):052–060. doi: 10.1055/s-0030-1247132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Long J., Zhang Y., Yu X., et al. Overcoming drug resistance in pancreatic cancer. Expert Opinion on Therapeutic Targets . 2011;15(7):817–828. doi: 10.1517/14728222.2011.566216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Edhemovic I., Gadzijev E. M., Brecelj E., et al. Electrochemotherapy: a new technological approach in treatment of metastases in the liver. Technology in Cancer Research and Treatment . 2011;10(5):475–485. doi: 10.7785/tcrt.2012.500224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Miklavcic D., Sersa G., Brecelj E., et al. Electrochemotherapy: technological advancements for efficient electroporation-based treatment of internal tumors. Medical, and Biological Engineering & Computing . 2012;50(12):1213–1225. doi: 10.1007/s11517-012-0991-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Martin R. C. G., Kwon D., Chalikonda S., et al. Treatment of 200 locally advanced (stage III) pancreatic adenocarcinoma patients with irreversible electroporation: safety and efficacy. Annals of Surgery . 2015;262(3):486–494. doi: 10.1097/SLA.0000000000001441. [DOI] [PubMed] [Google Scholar]
- 34.Mansson C., Brahmstaedt R., Nilsson A., Nygren P., Karlson B. M. Percutaneous irreversible electroporation for treatment of locally advanced pancreatic cancer following chemotherapy or radiochemotherapy. European Journal of Surgical Oncology . 2016;42(9):1401–1406. doi: 10.1016/j.ejso.2016.01.024. [DOI] [PubMed] [Google Scholar]
- 35.Vogel J. A., Rombouts S. J., De Rooij T., et al. Induction chemotherapy followed by resection or irreversible electroporation in locally advanced pancreatic cancer (impala): a prospective cohort study. Annals of Surgical Oncology . 2017;24(9):2734–2743. doi: 10.1245/s10434-017-5900-9. [DOI] [PubMed] [Google Scholar]
- 36.Narayanan G., Hosein P. J., Beulaygue I. C., et al. Percutaneous image-guided irreversible electroporation for the treatment of unresectable, locally advanced pancreatic adenocarcinoma. Journal of Vascular and Interventional Radiology . 2017;28(3):342–348. doi: 10.1016/j.jvir.2016.10.023. [DOI] [PubMed] [Google Scholar]
- 37.Leen E., Picard J., Stebbing J., Abel M., Dhillon T., Wasan H. Percutaneous irreversible electroporation with systemic treatment for locally advanced pancreatic adenocarcinoma. Journal of Gastrointestinal Oncology . 2018;9(2):275–281. doi: 10.21037/jgo.2018.01.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sugimoto K., Moriyasu F., Tsuchiya T., et al. Irreversible electroporation for nonthermal tumor ablation in patients with locally advanced pancreatic cancer: initial clinical experience in Japan. Internal Medicine . 2018;57(22):3225–3231. doi: 10.2169/internalmedicine.0861-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mansson C., Brahmstaedt R., Nygren P., Nilsson A., Urdzik J., Karlson B. M. Percutaneous irreversible electroporation as first-line treatment of locally advanced pancreatic cancer. Anticancer Research . 2019;39(5):2509–2512. doi: 10.21873/anticanres.13371. [DOI] [PubMed] [Google Scholar]
- 40.Shamseddine A. I., Khalifeh M. J., Mourad F. H., et al. Comparative pharmacokinetics and metabolic pathway of gemcitabine during intravenous and intra-arterial delivery in unresectable pancreatic cancer patients. Clinical Pharmacokinetics . 2005;44(9):957–967. doi: 10.2165/00003088-200544090-00005. [DOI] [PubMed] [Google Scholar]
- 41.Quaglino P., Mortera C., Osella-Abate S., et al. Electrochemotherapy with intravenous bleomycin in the local treatment of skin melanoma metastases. Annals of Surgical Oncology . 2008;15(8):2215–2222. doi: 10.1245/s10434-008-9976-0. [DOI] [PubMed] [Google Scholar]
- 42.Tozon N., Pavlin D., Sersa G., Dolinsek T., Cemazar M. Electrochemotherapy with intravenous bleomycin injection: an observational study in superficial squamous cell carcinoma in cats. Journal of Feline Medicine & Surgery . 2014;16(4):291–299. doi: 10.1177/1098612X13507071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Moir J., White S. A., French J. J., Littler P., Manas D. M. Systematic review of irreversible electroporation in the treatment of advanced pancreatic cancer. European Journal of Surgical Oncology . 2014;40(12):1598–1604. doi: 10.1016/j.ejso.2014.08.480. [DOI] [PubMed] [Google Scholar]
- 44.Yan L., Chen Y. L., Su M., et al. A single-institution experience with open irreversible electroporation for locally advanced pancreatic carcinoma. Chinese Medical Journal . 2016;129(24):2920–2925. doi: 10.4103/0366-6999.195476. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data are available upon reasonable request.


