Summary
Background
Few studies have investigated molecular biomarkers of specific executive function (EF) skills in children. We aimed to characterise the prospective associations between metabolome and multiple domains of EF using a bidirectional design.
Methods
This study was conducted within a longitudinal birth cohort, the Growing Up in Singapore Towards healthy Outcomes (GUSTO). Circulating levels of 165 metabolites were quantified using a nuclear magnetic resonance based metabolomics platform (n = 457 (∼6yrs) and n = 524 (∼8yrs)). Parent-reported EF was available for 495 children (∼7yrs). Multivariate linear regression was used to assess the metabolite-EF relationships. We examined the role of body composition, dietary factors, and genetics in the metabolite-EF associations.
Findings
Higher leucine level (∼6yrs) was associated with poorer EF (∼7yrs, Initiate (P = 0.003) and Working Memory (P = 0.004)). EF (∼7yrs) was not associated with leucine (∼8yrs). Importantly, we found weak evidence for associations of dietary factors (∼5yrs) with leucine (∼6yrs) and EF (∼7yrs). Each copy of C allele in rs1260326 (a leucine-related polymorphism) was associated with higher leucine level and poorer Initiate and Working Memory (P < 0.05). Amongst those with less strongly genetically influenced leucine, inverse association between leucine and cognitive regulation were weaker among those with higher BMI.
Interpretation
The observed association between higher leucine level and poorer EF may be determined by genetics and may not be easily amenable to dietary interventions. Further research is needed for validation and to understand mechanisms.
Funding
Singapore National Research Foundation and Agency for Science, Technology and Research.
Keywords: Metabolite, Executive function, Dietary factor, Genetic
Research in context.
Evidence before this study
Early-life plasma metabolites have been associated with childhood cognitive functions such as IQ, but few studies have investigated the molecular biomarkers for specific executive function (EF) skills, which is a set of higher-level cognitive skills responsible for goal-directed behaviours. In addition, it remains an open question as to how early nutritional environments and metabolism are linked to EF.
Added value of this study
We performed a metabolomics analysis of multiple domains of EF using bidirectional design in a longitudinal birth cohort. We examined the relationships between plasma metabolites and three domains of EF skills, i.e., behavioural regulation, cognitive regulation, and emotional regulation. We found evidence for associations of higher circulating leucine level (∼6 years) with poorer EF, especially cognitive regulation (∼7 years), but EF (∼7 years) was not associated with leucine (∼8 years). Additionally, we found that dietary factors were not strongly related to circulating leucine. We also identified genetic association that may have confounded the observational association between higher circulating leucine and poorer cognitive regulation skills.
Implications of all the available evidence
This study suggests that genetically-determined metabolism and, in particular, leucine may be responsible for effects on child cognitive regulation skills, thus dietary interventions may be less efficacious for cognitive development than suggested by previous studies. The role of leucine on neurodevelopment is supported by biological evidence, suggesting our findings may be relevant to risk prediction and drive further research into mechanisms.
Alt-text: Unlabelled box
Introduction
Early life cognitive development has been associated with academic achievement and health outcomes in adulthood, including risks of age-related cognitive decline, depression, and type 2 diabetes.1, 2, 3, 4, 5, 6 Molecular biomarkers for cognitive function have been widely investigated, but mainly in the context of age-related cognitive impairment in later life7 and neurodevelopmental disorders, such as autism8 and attention deficit hyperactivity disorder (ADHD).9,10 Particularly, plasma margaroylcarnitine and indolelactate were lower among children diagnosed with autism,8 while higher cord plasma branched-chain amino acids (BCAAs) and genetic variation in sphingolipid metabolism genes were associated with greater risk of childhood ADHD.9,10 Early-life plasma metabolites have also been associated with childhood IQ, particularly phosphatidylcholine, hydroxy-sphingomyelin, and threonine.11 Nevertheless, few investigated early development of specific executive function (EF) skills, which is a set of higher-level cognitive skills responsible for goal-directed behaviours.12,13 EF are distinct brain functions which map onto the prefrontal cortex14,15 and include neural processes that allow an individual to control impulses (i.e., inhibitory control), hold information in mind (i.e., working memory), shift attention (i.e., cognitive flexibility), etc.13 Together, these processes coordinate learning and development of other cognitive abilities of the brain.16 Thus, children with better self-control were more capable of managing health, financial, and social demands in adulthood, and aged more slowly in both the body and the brain.6
EF development is a complex process influenced by heritable genetic variation and environmental factors. Several studies have reported associations of healthy diets (assessed using various dietary assessment methods such as the Food Frequency Questionnaire (FFQ)) with better executive functioning.17 However, the underlying mechanisms and directionality remain unclear. Dietary factors influence the metabolome18 and thereby may play a role in EF development.17 Metabolomics quantify the levels of a wide range of metabolites to construct a profile associated with a trait of interest. Comprehensive characterisation of metabolomic biomarkers may highlight molecular pathways relevant to EF development. Peripheral blood circulating metabolites may be a reasonable proxy for difficult-to-sample brain metabolites relevant to EF development such as the maturation of the prefrontal cortex, which is intimately involved in EF development.19 On the other hand, EF may influence self-regulation of eating behaviours and thereby dietary intake,20,21 which subsequently affects metabolome.18 Together, blood metabolome may represent a readily accessible source of biomarkers to better understand EF development. Since EF development spans from infancy to early adulthood22 better understanding the role of early metabolism may inform early interventions designed to enhance EF. However, intervention targeting metabolomic biomarkers may not be effective if 1) EF influences dietary factors and thereby metabolomic biomarkers; or 2) genetic components influence metabolism and thereby EF development. Therefore, in this study, we aimed to investigate the metabolomic biomarkers for EF in a deeply phenotyped and multi-ethnic birth cohort in Singapore to characterise the prospective associations between metabolome and multiple domains of EF using bidirectional analysis. We further investigated how the identified signals were related to genotype and early dietary factors.
Materials and methods
Data source
This study was conducted within the Growing Up in Singapore Towards healthy Outcomes (GUSTO) prospective cohort study. Details of the cohort design have been described elsewhere.23 In brief, the GUSTO study recruited pregnant women aged at least 18 years attending their first trimester antenatal ultrasound scan at one of Singapore's two major public maternity units, namely National University Hospital and KK Women's and Children's Hospital, between June 2009 and September 2010. Initially, the main GUSTO cohort recruited 1,450 mothers. By delivery, we excluded 246 mothers lost to follow up, 96 mothers who underwent in vitro ferilisation and 10 pairs of twins. In this study, we included 1,095 mother-child dyads present in the cohort at delivery. Obstetric information was abstracted from medical records using standardized forms. Sociodemographic characteristics were collected using standardized self-report questionnaires. Figure 1a shows the flowchart and sample sizes for data collected at various time points. Figure 1b shows the overlapping sample sizes between different domains of data. Study sample was selected based on data availability for each analysis. As this was a secondary data analyses, data from all available subjects were used for analyses.
Figure 1.
Study design and data availability a) Flowchart for study sample from the Growing Up in Singapore Towards healthy Outcomes (GUSTO) prospective study and sample sizes for data collected at various time points; b) Venn diagram indicating overlapping sample sizes between different domains of data. Dietary factors in b-3 represents individuals with assessment from either food frequency questionnaire (FFQ) or energy consumption tasks.
Metabolome
Peripheral blood was drawn after an overnight fast in 460 and 528 children at age 6 and 8 years, respectively. Blood was immediately fractionated, aliquoted, and stored at -80°C. Circulating metabolites levels were quantified using an automated nuclear magnetic resonance (NMR)-based high throughput metabolomics platform (Nightingale Health Ltd., Helsinki, Finland). This mature technique in metabolomic analysis has the advantage of circumventing batch effects. Quality control procedures examined sample contamination during the collection process and from lab handling. After quality control, metabolomics data were available for 457 and 524 children aged ∼6 and ∼8 years, respectively. Circulating level of 165 metabolites, including cholesterol, triglycerides, lipoprotein subclasses, fatty acids, amino acids, etc, (Supplementary Table 1) were quantified to provide a comprehensive profile of metabolically important biomarkers.24 About 12% of the metabolites had missing values, among which the median missing rate per metabolite was lower than 1% (Supplementary Table 1). We performed analyses using complete data. Raw data were natural log-transformed after adding one. We used per standard deviation (SD) unit of the transformed data for analysis.
Executive function
To assess whether molecular biomarkers are relevant to specific executive function skills, we assessed parent-reported EF based on Behavior Rating Inventory of Executive Function, Second Edition, Parent Form (BRIEF2-PF). BRIEF2-PF evaluates three main composite indices of EF, each comprises of different scales, i.e., behavioural regulation (Inhibit and Self-Monitor), cognitive regulation (Initiate, Working Memory, Plan/Organize, Task-Monitor, and Organization of Materials), and emotional regulation (Shift and Emotional Control).25 At the age of ∼7 years, BRIEF2-PF was administrated to 509 children. After removing potentially invalid response using the internal validation scales provided in the BRIEF2-PF rating, EF assessment was available for 495 children. Supplementary Figure 1 shows that different EF scales were correlated. A global executive composite was generated for 170 children aged ∼4.5 years as an indication of general EF based on the parent-reported BRIEF-Preschool version, which assesses five EF scales, i.e., Inhibit, Working Memory, Plan/Organize, Shift, and Emotional control.25 Raw scores were transformed into T-Scores (mean=50, SD=10) for each EF scale for analysis.25 A higher T-Score indicates more EF impairments. We also performed analyses using domain-specific EF scores for the key findings.
Predictors and confounders of metabolome
We further explored predictors and confounders that may be relevant to leucine, a consistent signal in the metabolomics analysis of EF. Given the role of leucine in protein synthesis,26 we investigated body composition measures (∼6 years), including skinfold (biceps, triceps, subscapular, and suprailiac; mm), height (m), and weight (kg) measured in triplicate by trained study staff, body-mass index (BMI) computed as weight/height2 (kg/m2), and fat and lean mass (kg) measured by quantitative magnetic resonance (QMR). Raw measures were transformed into SD unit for analysis.
Dietary factors investigated as predictors were energy consumption and food intakes. Energy consumption was estimated during an in-lab ad libitum buffet lunch and separate eating in the absence of hunger (EAH) task, where children were exposed to palatable snacks after lunch. Detailed description of the protocol has been described in a previous GUSTO study.27 Here, we focused on 1) total energy consumption (kcal) at lunch; 2) energy consumption during the eating in the absence of hunger (EAH) task after lunch (higher than or equal to median vs lower than median; median=21 kcal). Children who had reported they were hungry after lunch were excluded from this analysis, following standard protocol; and 3) total energy consumption (kcal) during the assessments (at lunch and in the EAH task after lunch). A total of 443 children had available data for at least one of the energy intakes measurements reported at ∼4.5 years.
Food intakes were estimated from FFQ administrated by trained interviewers to the caregivers during clinic visits. Detailed description of the protocol and validation has been described in a previous GUSTO study.28 In this study, we focused on the intakes (gram/day) of 9 types of animal-based proteins (Chicken, Deep-fried chicken, Beef, Processed meats, Fish, Fried fish, Seafood, Boiled egg, and Fried egg) and 6 types of dairy products (Milk, Low fat milk, Malt drinks, Yogurt, Cheese, and Cultured drink) among 716 children at aged ∼5 years (Supplementary Table 2).28
Genetic variants of leucine metabolism were obtained from a genome-wide association study (GWAS) meta-analysis among 24,925 adults of European ancestry.29 Circulating leucine level was quantified using the same NMR-based metabolomics platform as in our cohort. We selected 76 single nucleotide polymorphisms (SNPs) with P-value smaller than 5×10−8, among which 3 independent lead SNPs (rs1260326 (GCKR), rs17789027 (PPM1K), rs12325419 (DDX19B)) remained after removing correlated SNPs (r2>0.001). In GUSTO, 68 out of 76 SNPs were available in the imputed genetic data. Detail of DNA extraction, genotyping, and imputation has been described previously.30 Genotype imputation were performed for each ethnicity separately using 1000G Phase 3 reference panel. Only SNPs with a minor allele frequency higher than 5% and imputation quality higher than 0.5 were included.
Ethics
Ethics approval was obtained from Centralised Institutional Review Board of SingHealth (2018/2767) and the Domain Specific Review Board of Singapore National Healthcare Group (D/2009/021, B/2014/00414). Informed written consent was obtained from each participant.
Statistical analysis
Analyses of this study were designed by stage, in which we started with a bidirectional analysis between metabolites and EF and followed by post-hoc analyses to improve the understanding of the underlying mechanisms. See Supplementary Method for an illustration of the study design.
Stage 1: Metabolomics analysis of executive function
We first compared the study sample with both metabolomics and EF data to the full cohort at delivery regarding maternal and child characteristics using chi-squared test and t-test for categorical and continuous characteristics, respectively.
To investigate the bidirectional association between metabolomics and EF, we performed one set of association analyses between each metabolite and each EF scale (Metabolite (∼6 years) → EF (∼7 years)), and second set of association analyses for the other direction (EF (∼7 years) → Metabolite (∼8 years)) using multivariable linear regression. In Model 1, we adjusted for age at blood draw for metabolite measurement, age at EF assessment, and sex. In Model 2, we additionally adjusted for socio-economic status as surrogated by maternal ethnicity, maternal educational level, and household income at recruitment. In Model 3, we additionally adjusted for child BMI at the time when the corresponding exposure in the bi-directional analysis was measured/assessed. Built on Model 3, we further accounted for pregnancy-related factors (i.e., maternal age at delivery, parity, pre-pregnancy BMI, gestational weight gain rate, gestational age, number of days after birth to discharge) and maternal behaviours (i.e., maternal smoking and maternal pre-pregnancy alcohol consumption) in a sensitivity analysis. We only included pregnancy-related factors and maternal behaviours as a sensitivity analysis because a regression model with a large number of variables but a small sample size can lead to overfitting. Given that EF may influence eating behaviours, thereby dietary intake, which subsequently affects metabolome,18,20,21 we also adjusted for general EF score at ∼4.5 years in a sensitivity analysis and assess the change of effect size (Cohen's f2) in the corresponding associations. See Supplementary Method for model construction.
We presented associations with a P-value < 0.01 threshold to identify patterns in our findings. Due to the correlation between metabolites, Bonferroni correction for the total number of metabolites (n = 165) is not appropriate. Principal component analysis found that the first 13 principal components explained over 95% of variation in our metabolomics data. Therefore, we accounted for multiple comparisons by applying a Bonferroni-corrected significance threshold of P-value < 0.004, i.e., 0.05/13.31
Relation between child weight status and EF has been reported,32 and a recent Mendelian randomization study suggested higher genetically predicted BMI increases the risk of ADHD, but not the other way around.33 Child weight status is also a strong determinant of circulating metabolites such as BCAAs (i.e., leucine, isoleucine, and valine),34 thus it is worth particular attention to modelling its contributions to observed leucine-EF associations. Therefore, we assessed the associations of BCAAs with body composition measures in our cohort.
Stage 2: Dietary factors, circulating leucine level, and executive function
Built on a consistent signal for leucine found in Stage 1, we further investigated the association of dietary factors (∼5 years) as predictors of circulating leucine level and relevant EF indices. Specifically, we investigated the associations of three energy intakes variables and fifteen food intakes variables with circulating leucine level and with behavioural and cognitive regulation using multivariable linear regression and logistic regression model. Main model construction for this stage was similar to Model 3 at Stage 1. We also performed the two sensitivity analyses considering pregnancy-related factors and maternal behaviours, and earlier general EF. See Supplementary Method for model construction. We presented associations with a P-value < 0.01 threshold.
Stage 3: Role of leucine metabolism in executive function
To further investigate whether genetic influences on leucine explained or modified the leucine-EF relationship, we assessed the associations of 68 leucine-related SNPs with circulating leucine level (∼6 years) and the most pertinent EF index, i.e., cognitive regulation (∼7 years), using additive model adjusting for age, sex, and maternal ethnicity. We also performed ethnicity-specific analysis to assess whether the effect is consistent across different ethnic groups. In addition, we performed an analysis of the associations between BCAAs and cognitive regulation stratified by rs1260326, which is a missense variant responsible for proline-to-leucine substitution (C>T, Pro446Leu) in glucokinase regulatory protein (GKRP).
Lastly, we performed causal effect decomposition analyses using regmedint package in R to evaluate a) whether leucine mediated or modified genetic-EF associations; and b) whether BMI mediated or modified leucine-EF associations. We hypothesized that leucine is a mediator for the genetic-EF associations and BMI is a mediator for the leucine-EF associations. A regression-based counterfactual approach accounting for exposure-mediator interactions and adjusting for exposure-mediator, mediator-outcome, and exposure-outcome confounders was implemented to compute pure and total (including interaction) direct effects and pure and total (including mediated-interaction) indirect effects.35 Specifically, we performed three sets of analyses: 1) rs1260326 (CC vs. CT) → Leu (∼6 years) → Cognitive regulation (∼7 years); 2) rs1260326 (CC vs. CT) → BMI (∼6 years) → Cognitive regulation (∼7 years); and 3) Leu (∼6 years) → BMI (∼6 years) → Cognitive regulation (∼7 years). For the third analysis, we also performed stratified analysis by rs1260326 genotype (CC vs. CT). See Supplementary Method for model construction.
Role of funders
None of the funders were directly involved in the design, data collection, analysis, or interpretation, or writing of this report.
Results
Descriptive analysis and metabolomics analysis of executive function (Stage 1)
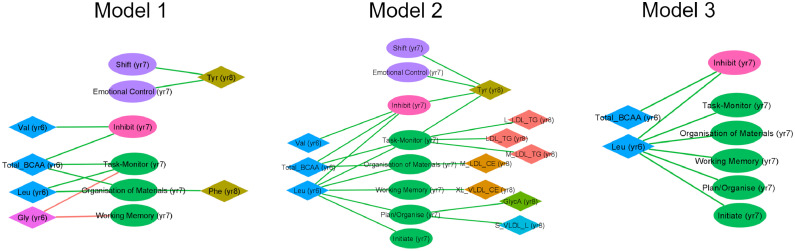
We found no substantial differences between our study sample and the full cohort at delivery, except for under-representation of children of Chinese ethnicity (Table 1). Sample sizes of Models 1 to 3 for all metabolite-EF pairs ranged from 262 to 348. Figure 2 is a network illustration of the bidirectional relationships for all multivariable-adjusted metabolite-EF associations with P-value < 0.01 (see Supplementary Table 3 for detailed results). Emotional regulation (∼7 years) was associated with tyrosine (∼8 years) after adjusting for age, sex, and socio-economic status. However, these associations did not sustain after additionally adjusting for BMI in Model 3. On the other hand, BCAAs (∼6 years) were consistently associated with behavioural and cognitive regulation (∼7 years). The associations of leucine (∼6 years) with cognitive regulation (∼7 years) were the most robust. Table 2 shows the results from Model 3 as illustrated in Figure 2. Only the associations for Leu (∼6 years)-Initiate (∼7 years) and Leu (∼6 years)-Working Memory (∼7 years) passed the Bonferroni correction threshold (P-value < 0.004). Our findings showed that per SD higher log-leucine (equivalent to 1.5% higher in absolute leucine concentration; IQRLeu 0.079 to 0.010 mmol/L) was associated with 1.61 units (95% confidence interval (CI): 0.56-2.66; or 4% (95%CI: 1.2–5.9%) higher in Initiate T-Score and 1.65 units (95%CI: 0.55-2.75; or 3% (95%CI: 1.1–5.7%)) higher in Working Memory T-Scores. Additional adjustment for pregnancy-related factors, maternal smoking, and maternal pre-pregnancy alcohol consumption did not affect the effect size of the association between leucine and Initiate T-Score. However, effect sizes dropped substantially after additionally adjusting for general EF score at ∼4.5 years. Since Initiate and Working Memory are both cognitive regulation skills and were highly correlated in our cohort (r = 0.79, see Supplementary Figure 1), we performed an additional analysis for cognitive regulation specific scores and found that per SD higher log-leucine was associated with 1.68 units (95% CI: 0.64-2.72; or 4% (95%CI: 1.5–6.5%)) higher in cognitive regulation specific scores.
Table 1.
Comparisons of characteristics between the full sample and the subsample with metabolomics and executive function data.
| Characteristics | Category | Full sample at delivery (n = 1095) |
Subsample with metabolomics (yr6) and EF (yr7) (n = 278) |
Subsample with EF (yr7) and metabolomics (yr8) (n = 300) |
|||||
|---|---|---|---|---|---|---|---|---|---|
| N (%) | Mean (SD) | N (%) | Mean (SD) | P-valuea | N (%) | Mean (SD) | P-valuea | ||
| Sex | Girl | 523 (48%) | - | 134 (48%) | - | 0.949 | 152 (51%) | - | 0.408 |
| Boy | 572 (52%) | - | 144 (52%) | - | 148 (49%) | - | |||
| Maternal ethnicity | Chinese | 598 (55%) | - | 132 (47%) | - | 0.006 | 131 (44%) | - | 0.001 |
| Malay | 291 (27%) | - | 101 (36%) | - | 109 (36%) | - | |||
| Indian | 205 (19%) | - | 45 (16%) | - | 59 (20%) | - | |||
| Maternal educational level at recruitment | Primary or below | 60 (5%) | - | 18 (6%) | - | 0.430 | 19 (6%) | - | 0.435 |
| Secondary | 282 (26%) | - | 61 (22%) | - | 73 (24%) | - | |||
| Post-secondary | 384 (35%) | - | 109 (39%) | - | 118 (39%) | - | |||
| University | 355 (32%) | - | 88 (32%) | - | 86 (29%) | - | |||
| Household monthly income at recruitment (SG$) | Below 2000 | 163 (15%) | - | 42 (15%) | - | 0.942 | 49 (16%) | - | 0.781 |
| 2000 to 3999 | 324 (30%) | - | 82 (29%) | - | 95 (32%) | - | |||
| 4000 to 5999 | 255 (23%) | - | 62 (22%) | - | 68 (23%) | - | |||
| More than 6000 | 282 (26%) | - | 77 (28%) | - | 71 (24%) | - | |||
| Child BMI at age 6 years | - | 771 | 15.6 (2.3) | 274 | 15.7 (2.2) | 0.558 | 272 | 15.8 (2.6) | 0.203 |
| Maternal age at delivery | - | 1095 | 30.9 (5.1) | 278 | 30.9 (5.1) | 0.942 | 300 | 31.1 (5.4) | 0.616 |
| Parity | Nulliparous | 468 (43%) | - | 110 (40%) | - | 0.253 | 117 (39%) | - | 0.051 |
| Primiparous | 390 (36%) | - | 95 (34%) | - | 98 (33%) | - | |||
| Multiparous | 237 (22%) | - | 73 (26%) | - | 85 (28%) | - | |||
| Maternal pre-pregnancy BMI | - | 993 | 22.7 (4.4) | 255 | 23.0 (4.5) | 0.380 | 271 | 23.3 (4.6) | 0.049 |
| Gestational weight gain rate | - | 1085 | 0.5 (0.1) | 278 | 0.5 (0.1) | 0.438 | 299 | 0.5 (0.1) | 0.689 |
| Gestational age | - | 1095 | 38.7 (1.6) | 278 | 38.8 (1.5) | 0.658 | 300 | 38.8 (1.2) | 0.464 |
| Number of days after birth to discharge | 1 or below | 342 (31%) | - | 91 (33%) | - | 0.563 | 98 (33%) | - | 0.620 |
| 2 to 3 | 558 (51%) | - | 144 (52%) | - | 156 (52%) | - | |||
| 4 or above | 174 (16%) | - | 37 (13%) | - | 41 (14%) | - | |||
| Maternal pre-pregnancy alcohol consumption | No | 699 (64%) | - | 184 (66%) | - | 0.470 | 205 (68%) | - | 0.177 |
| Yes | 379 (35%) | - | 89 (32%) | - | 91 (30%) | - | |||
| Maternal smoking | Non-smoker | 922 (84%) | - | 236 (85%) | - | 0.898 | 254 (85%) | - | 0.952 |
| Ex-smoker | 125 (11%) | - | 29 (10%) | - | 34 (11%) | - | |||
| Current smoker | 29 (3%) | - | 7 (3%) | - | 7 (2%) | - | |||
Abbreviation: body-mass index (BMI), executive function (EF).
P-values for difference comparing the subsample with the full sample at delivery were estimated from chi-squared test for categorical characteristics and from t-test for continuous characteristics.
Figure 2.
Network illustration for association analyses between metabolites and executive function (Metab.yr6→EF.yr7 and EF.yr7→Metab.yr8). Edges indicate associations with nominal P-value < 0.01. Edge colours indicate direction of associations (Green: positive associations. Pink: inverse associations). Node colours indicate different groups of metabolites or EF scales (Green: cognitive regulation. Pink: behavioural regulation. Purple: emotional regulation). Model 1 adjusted for age at blood draw for metabolite measurement, age at executive function assessment, and sex (n = 277∼348). Model 2 is Model 1 additionally adjusted for maternal ethnicity, maternal educational level, and household income at recruitment (n = 262∼327). Model 3 is Model 2 additionally adjusted for child body-mass index (BMI) at the time of exposure measurement (n = 262∼321). (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
Table 2.
Associations (P-value < 0.01) between circulating metabolite levels, executive function, and dietary factors.
| Analysis (Exposure-Outcome) | Model 3a |
Model 3 additionally adjusting for pregnancy-related factors and maternal behavioursb |
Model 3 additionally adjusting for GEC (yr4.5) |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample size | Beta | SE | Cohen's f2 | P-value | Sample size | Cohen's f2 | P-value | Sample size | Cohen's f2 | P-value | |
| Stage 1 | |||||||||||
| Leu (yr6)-Initiate (yr7) | 264 | 1.61 | 0.54 | 0.04 | 0.0030* | 241 | 0.04 | 0.0049 | 69 | 0.01 | 0.403 |
| Leu (yr6)-Working Memory (yr7) | 264 | 1.65 | 0.56 | 0.03 | 0.0036* | 241 | 0.02 | 0.025 | 69 | 0.01 | 0.490 |
| Leu (yr6)-Organisation of Materials (yr7) | 264 | 1.27 | 0.46 | 0.03 | 0.0058 | 241 | 0.03 | 0.0088 | 69 | 4.8E-4 | 0.874 |
| Leu (yr6)-Task-Monitor (yr7) | 264 | 1.50 | 0.54 | 0.03 | 0.0061 | 241 | 0.02 | 0.038 | 69 | 0.01 | 0.507 |
| Leu (yr6)-Plan/Organise (yr7) | 264 | 1.39 | 0.53 | 0.03 | 0.0085 | 241 | 0.02 | 0.022 | 69 | 2.7E-3 | 0.708 |
| Leu (yr6)-Inhibit (yr7) | 264 | 1.40 | 0.53 | 0.03 | 0.0086 | 241 | 0.02 | 0.028 | 69 | 1.8E-3 | 0.756 |
| Total_BCAA (yr6)-Inhibit (yr7) | 264 | 1.37 | 0.52 | 0.03 | 0.0092 | 241 | 0.02 | 0.026 | 69 | 6.4E-4 | 0.854 |
| Stage 2c | |||||||||||
| Higher energy consumption at EAH (yr4.5)-Inhibit (yr7) | 253 | 3.78 | 1.17 | 0.04 | 0.0014 | 231 | 0.06 | 4.2E-04 | 100 | 0.04 | 0.066 |
| Higher energy consumption at EAH (yr4.5)-Organisation of Materials (yr7) | 253 | 2.75 | 1.03 | 0.03 | 0.0082 | 231 | 0.05 | 0.0010 | 100 | 0.01 | 0.306 |
| Processed meats (yr5)-Organisation of Materials (yr7) | 396 | 0.04 | 0.02 | 0.02 | 0.0089 | 359 | 0.02 | 0.015 | 96 | 0.02 | 0.214 |
Abbreviation: branched-chain amino acid (BCAA), body-mass index (BMI), eating in the absence of hunger (EAH), global executive composite (GEC), leucine (Leu), standard error (SE).
Stage 1 P-value < 0.004, which is the P-value < 0.05 significance level divided by the number of principal components (n = 13) that explained over 95% of variation in the metabolomics data.
Model 3 adjusted for age at exposure measurement, age at outcome measurement, sex, maternal ethnicity, maternal educational level, household income at recruitment, and child BMI at the time of exposure measurement.
Pregnancy-related factors included maternal age at delivery, parity, pre-pregnancy BMI, gestational weight gain rate, gestational age, number of days after birth to discharge. Maternal behaviours included maternal smoking and maternal pre-pregnancy alcohol consumption.
Higher energy consumption was defined as energy consumption during the eating in the absence of hunger (EAH) task after lunch higher than or equal to the median energy consumption of the subjects included in the task (median=21 kcal).
Higher circulating levels of BCAAs were consistently associated with higher subscapular skinfold, BMI, and QMR-measured lean mass at age 6 years (Supplementary Figure 2). To summarise, circulating BCAAs levels at 6 years were associated with body composition at 6 years, but only circulating leucine level was associated with cognitive regulation at 7 years.
Dietary factors, circulating leucine level, and executive function (Stage 2)
Table 2 also shows the findings (P-value < 0.01) of the associations of dietary factors with EF. Higher energy consumption during the eating in the absence of hunger task was associated with 3.78 units (P-value = 0.001, 95%CI: 1.48-6.08; or 4% (95%CI: 1.7–7.0%)) higher in Inhibit T-Score. Per one gram/day higher of processed meat intakes was associated with 0.04 unit (P-value = 0.009, 95%CI: 0.01-0.07; or 2% (95%CI: 0.5–3.2%)) higher in Organisation of Materials T-Scores. Accounting for general EF score at ∼4.5 years did not affect the effect size. Higher energy consumption at the eating in the absence of hunger task (∼4.5 years) and processed meats intakes (∼5 years) were associated with Inhibit and Organisation of Materials at ∼7 years (P-value < 0.01; Table 2); however, they were not associated with circulating leucine level (P-value>0.01; Supplementary Table 4).
Role of leucine metabolism in executive function (Stage 3)
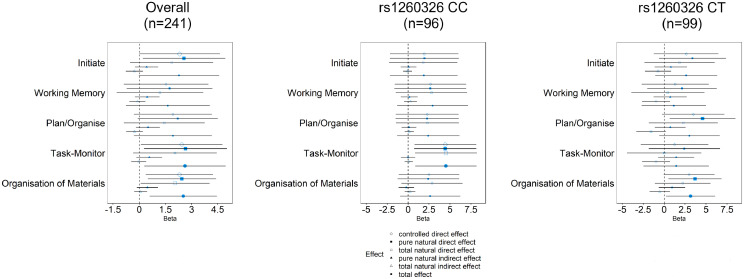
Leucine-related SNPs identified in the European GWAS meta-analysis were associated with circulating leucine level (∼6 years) in GUSTO (Supplementary Table 5). The top hit in our analysis (rs6753534, P-value = 0.023) was correlated with rs1260326 (r2=0.74, which was one of the top hits in the GWAS meta-analysis. In our analysis, per additional copy of C allele in rs1260326 was associated with 0.14 SD higher of natural log-transformed leucine level (P-value = 0.064). The effect size of rs1260326-leucine association was larger in the Indian subgroup, but the directions of effect were consistent in all ethnic groups (242 Chinese, 115 Malay, and 68 Indian; see Supplementary Tables 6). Per additional copy of C allele in rs1260326 was also associated with 1.64, 1.47, and 1.35 units higher in Initiate, Working Memory, and Plan/Organise T-Scores at ∼7 years, respectively (P-values smaller than 0.05; Supplementary Table 7). These associations were also evident among the Chinese subgroup (228 Chinese, 161 Malay, and 75 Indian; see Supplementary Tables 8). The associations of leucine with Initiate, Working Memory, and Task-Monitor had a larger effect size and smaller P-value in rs1260326 CC genotype comparing to CT genotype (Figure 3 and Supplementary Table 9). Taken together, per additional copy of C allele in rs1260326 was associated with higher circulating leucine level (∼6 years) and poorer cognitive regulation (∼7 years), which was consistent with the metabolomic analysis at Stage 1.
Figure 3.
Longitudinal associations of branched-chain amino acids (BCAAs) at age 6 years with executive function (cognitive regulation index) at age 7 years stratified by genotype of rs1260326. Beta indicates the change in executive function T-Score per standard deviation higher of natural log-transformed BCAAs. Model adjusted for age, sex, maternal ethnicity, maternal educational level, household income, body-mass index (BMI) at age 6 years. Sample sizes are 103, 109, and 35 for CC, CT, and TT genotype, respectively. Larger shapes indicate associations that the 95% confidence interval did not cover the null value. Red shapes indicate associations with a P-value smaller than 0.017 (i.e., 0.05/(3 genotype groups)). (Leucine (Leu), isoleucine (Ile), valine (Val)).
Compared to the CT genotype, the CC genotype in rs1260326 was associated with poorer cognitive regulation. But these associations were not mediated via circulating leucine level or BMI (Supplementary Figures 3 and 4). Supplementary Table 10 and Figure 4 show that pure natural direct effect of higher leucine level on Initiate was only found in the overall genotype subgroup (beta=2.56, P-value = 0.034) while pure natural direct effect of higher leucine level on Task-Monitor was found in both the overall genotype subgroup (beta=2.65, P = 0.029) and CT subgroup (beta=4.42, P = 0.020). However, these effects in the overall genotype subgroup disappeared after accounting for leucine-BMI interaction (see total natural direct effect). In CT subgroup, pure natural direct effect was observed for leucine with Plan/Organise and Organisation of Materials. Similarly, these effects disappeared after accounting for leucine-BMI interaction (see total natural direct effect). In addition, indirect effect via BMI was not statistically significant in these analyses.
Figure 4.
Regression-based causal mediation analysis for Leu (yr6; Above median vs Below median) → BMI (yr6) → Cognitive regulation T-Scores (yr7). Pure natural indirect effect is the effect of exposure on outcome due to the mediation only. The difference between the total natural indirect effect and the pure natural indirect effect indicates the effect due to interaction. (Leucine (Leu), body-mass index (BMI), Cognitive Regulation Index (CRI); total effect=pure natural direct effect + total natural indirect effect = pure natural indirect effect + total natural direct effect; larger shapes indicate associations that the 95% confidence interval did not cover the null value).
Discussion
In this study, we performed metabolomics analysis of three domains of EF involved in behavioural, cognitive, and emotional regulation. We found an association of higher circulating leucine level with poorer cognitive regulation. In addition, the relations between leucine, BMI, and EF differed by rs1260326 genotype. Specifically, at Stage 1, we identified circulating leucine as a candidate biomarker for cognitive regulation. Higher energy consumption during eating in the absence of hunger task was associated with poorer Inhibit skills and higher processed meat with poorer Organisation of Materials skills. However, we did not find an association between these dietary factors with circulating leucine levels. As expected, body composition measures (∼6 years) were strongly associated with BCAAs (∼6 years) and might at least partially explain the leucine-EF associations. CC genotype in rs1260326 was associated with higher circulating leucine in this population. Leucine-BMI interaction was observed in the overall genotype and the CT genotype subgroups, suggesting that such interaction may only exist among those with the CT genotype, which was less strongly associated with leucine levels. The inverse association between leucine and cognitive regulation among those with the CT genotype may be weaker among those with higher BMI.
BCAAs are known to stimulate muscle protein synthesis, although the effectiveness of dietary supplement of BCAAs alone is controversial.36,37 The biochemical and functional effects of BCAAs on the brain have also been studied.38 However, to the best of our knowledge, there are no studies reporting effects of BCAAs on cognitive development in children. Some human and animal studies suggest a potential effect of BCAAs on cognitive improvement after traumatic brain injury, albeit evidence has not been strong.39,40 However, a recent study consisting of eight prospective human cohorts reported an association between higher levels of BCAAs and decreased Alzheimer's disease risk,41 which was also supported by a murine experiment of BCAA-supplemented diet.42 It is important to note that patients with traumatic brain injury or Alzheimer's disease, whose brain metabolism is likely altered,43,44 are very different from children at the age of cognitive development. Nevertheless, these findings imply an effect of BCAAs on cognitive function.
In our analysis, circulating BCAAs were associated with body composition, but the association between leucine and poorer EF was more prominent compared to valine and isoleucine.45 The distinct role of leucine may be due to its regulatory role in the mechanistic target of rapamycin (mTOR) pathway.46 The final product of leucine catabolism, acetyl-coenzyme A enhances mTOR complex 1 (mTORC1) activity.46 The mTORC1 activation subsequently inhibits catabolic processes, such as autophagy,47 which regulates brain growth and neuronal connectivity, and potentially affects learning ability, memory function, and neurological disorders.47,48 Catabolism of isoleucine also yields acetyl-coenzyme A; however, leucine has a more pivotal regulatory role in mTOR pathway.46
The decrease in effect sizes additionally adjusting for general EF score at an earlier age suggest that the observed association between leucine and cognitive regulation may be determined by factors in earlier life such as dietary factors and genetics. Nevertheless, we found weak evidence for the associations between dietary factors and EF, and no association between dietary factors and circulating leucine level. Weak correlations of dietary intakes and circulating level of BCAAs have been previously reported.49 This finding suggests the relation between increased leucine and poorer EF may not be easily amenable to dietary interventions. Genetic analysis and mediation analysis showed consistent directionality for the association between circulating leucine level and cognitive regulation. Mediation analysis further suggested leucine-BMI interactions in the CT subgroup where leucine may be more malleable to environment (diet). This may imply two points. First, harmful effects may only present at higher circulating leucine levels observed in the CC subgroup. Second, at relatively lower circulating leucine level in the CT subgroup, higher leucine appears to have a beneficial effect on cognitive regulation at a higher BMI. We did not observed an association in TT subgroup, of which the analysis was limited by a small sample size and the result should be interpreted with caution. Nevertheless, these findings were only exploratory and might be false positive since family wise error rate was not controlled for in the analyses. Further investigation is warranted.
In this study, we performed metabolomics analysis of three domains of EF, followed by a series of post-hoc analyses to improve the understanding of the relationship between leucine metabolism and EF. Nevertheless, this study has some limitations. First, our analyses were based on a subset of the original GUSTO cohort. Although the demographic characteristics of the study sample were not substantially different from the full cohort, small sample sizes limit the power of our analysis, especially for the genetic analysis. Second, although leucine was associated with several EF skills at P-value < 0.01, only the associations of leucine with Initiate and Working Memory passed the Bonferroni-corrected P-value threshold (P-value < 0.004). This is likely due to the small sample sizes. Nevertheless, our findings suggested a considerable effect size for the leucine-EF associations and were supported by numerous accessory findings. Third, general EF score at ∼4.5 years is available in a smaller group of children, which limited the sample size of our additional analysis accounting for earlier EF. Thus, this analysis should be interpreted with caution. Fourth, candidate SNPs associated with leucine were obtained from a European GWAS since no such GWAS is available among Asian population. However, our genetic analysis focused on a missense variant (rs1260326) that is more functionally relevant to the biosynthesis of leucine. Our ethnicity-specific analysis showed that effect sizes of the rs1260326-leucine and rs1260326-EF associations may differ by ethnic groups, but the directions of effect were consistent. Fifth, rs1260326 has also been associated with triglyceride levels, fasting glucose, and risk of type 2 diabetes, suggesting a role in multiple metabolic traits and pleiotropy.50 This suggested rs1260326 is not an ideal instrumental variable for Mendelian randomization but it does not invalidate our causal mediation analysis using the regression-based counterfactual approach. Besides, we did not perform Mendelian randomization given the small sample in our cohort and mediation analysis using Mendelian randomization does not account for exposure-mediator interaction. Sixth, food frequency questionnaire was administrated one year before blood draw for metabolite measurement and two years before EF assessment. Dietary pattern may change during such time lag, especially in children that normally start schooling between age 6-7 years. In addition, food frequency questionnaire may not be accurate for children who spent most of their time in the school, pre-school, and after-school care. Thus, we cannot rule out that measurement error in caregiver-reported dietary patterns may mask true relationships with diet. Lastly, in the case that blood metabolomics may not reflect brain metabolome, we may miss relevant associations thereof. However, metabolites, such as sphingolipids, in blood and brain have been consistently linked to the progression and severity of Alzheimer's disease.51 Leucine passes across blood-brain barrier and biological evidence supports its role in cognitive function. However, in our cohort, sphingomyelin, a type of sphingolipid, was not associated with any executive function scales (P-value>0.05). This may suggest distinct metabolomic profiles are relevant to cognitive development and cognitive degeneration.
Taken together, our metabolomics analysis found an association between higher circulating leucine level with poorer EF, especially cognitive regulation. Effect sizes attenuated after accounting for earlier general EF score suggested that the observed association may be determined by factors in earlier life. Our further investigation showed that this relationship may be determined by genetic factors and may not be easily amenable to dietary interventions. Causal mediation analysis suggested a relatively lower circulating leucine level, leucine-BMI interaction may counteract the harmful effect of leucine on cognitive regulation. While the role of leucine on neurodevelopment is supported by biological evidence, suggesting our findings may be relevant to risk prediction and drive further research into mechanisms, future investigation with a larger sample size should be considered for validation.
Contributors
Jian Huang contributes to conceptualisation, data curation, formal analysis, investigation, methodology, verification of the underlying data, data interpretation, visualisation, drafting, reviewing and editing the manuscript.
Evelyn Law contributes to data curation, data interpretation, funding acquisition, and reviewing and editing the manuscript.
Ibrahim Karaman contributes to methodology, and reviewing and editing the manuscript.
Keri McCrickerd contributes to methodology, data interpretation, and reviewing and editing the manuscript.
Anna Fogel contributes to methodology, data interpretation, and reviewing and editing the manuscript.
Mary FF Chong contributes to methodology, data interpretation, and reviewing and editing the manuscript.
Lourdes Mary Daniel contributes to data interpretation, and reviewing and editing the manuscript.
Patricia Pelufo Silveira contributes to data interpretation, and reviewing and editing the manuscript.
Yap Seng Chong contributes to funding acquisition, and reviewing and editing the manuscript.
Johan G. Eriksson contributes to funding acquisition, and reviewing and editing the manuscript.
Michael J Meaney contributes to funding acquisition, and reviewing and editing the manuscript.
Jonathan Huang contributes to methodology, verification of the underlying data, data interpretation, supervision, and reviewing and editing the manuscript.
All authors read and approved the final version of the manuscript.
Data sharing statement
Deidentified participant data will be made available on request.
Declaration of interests
The authors disclose no conflict of interest.
Acknowledgements
The GUSTO study group includes: Airu Chia, Allan Sheppard, Amutha Chinnadurai, Anna Magdalena Fogel, Anne Eng Neo Goh, Anne Hin Yee Chu, Anne Rifkin-Graboi, Anqi Qiu, Arijit Biswas, Bee Wah Lee, Birit Froukje Philipp Broekman, Bobby Kyungbeom Cheon, Boon Long Quah, Candida Vaz, Chai Kiat Chng, Cheryl Shufen Ngo, Choon Looi Bong, Christiani Jeyakumar Henry, Ciaran Gerard Forde, Claudia Chi, Daniel Yam Thiam Goh, Dawn Xin Ping Koh, Desiree Y. Phua, Doris Ngiuk Lan Loh, E Shyong Tai, Elaine Kwang Hsia Tham, Elaine Phaik Ling Quah, Elizabeth Huiwen Tham, Evelyn Chung Ning Law, Evelyn Xiu Ling Loo, Fabian Kok Peng Yap, Faidon Magkos, Falk Müller-Riemenschneider, George Seow Heong Yeo, Hannah Ee Juen Yong, Helen Yu Chen, Heng Hao Tan, Hong Pan, Hugo P S van Bever, Hui Min Tan, Iliana Magiati, Inez Bik Yun Wong, Ives Yubin Lim, Ivy Yee-Man Lau, Izzuddin Bin Mohd Aris, Jeannie Tay, Jeevesh Kapur, Jenny L. Richmond, Jerry Kok Yen Chan, Jia Xu, Joanna Dawn Holbrook, Joanne Su-Yin Yoong, Joao Nuno Andrade Requicha Ferreira, Johan Gunnar Eriksson, Jonathan Tze Liang Choo, Jonathan Y. Bernard, Jonathan Yinhao Huang, Joshua J. Gooley, Jun Shi Lai, Karen Mei Ling Tan, Keith M. Godfrey, Kenneth Yung Chiang Kwek, Keri McCrickerd, Kok Hian Tan, Kothandaraman Narasimhan, Krishnamoorthy Naiduvaje, Kuan Jin Lee, Leher Singh, Li Chen, Lieng Hsi Ling, Lin Lin Su, Ling-Wei Chen, Lourdes Mary Daniel, Lynette Pei-Chi Shek, Marielle V. Fortier, Mark Hanson, Mary Foong-Fong Chong, Mary Rauff, Mei Chien Chua, Melvin Khee-Shing Leow, Michael J. Meaney, Michelle Zhi Ling Kee, Min Gong, Mya Thway Tint, Navin Michael, Neerja Karnani, Ngee Lek, Oon Hoe Teoh, P. C. Wong, Paulin Tay Straughan, Peter David Gluckman, Pratibha Keshav Agarwal, Priti Mishra, Queenie Ling Jun Li, Rob Martinus van Dam, Salome A. Rebello, Sambasivam Sendhil Velan, Seang Mei Saw, See Ling Loy, Seng Bin Ang, Shang Chee Chong, Sharon Ng, Shiao-Yng Chan, Shirong Cai, Shu-E Soh, Sok Bee Lim, Stella Tsotsi, Stephen Chin-Ying Hsu, Sue-Anne Ee Shiow Toh, Suresh Anand Sadananthan, Swee Chye Quek, Varsha Gupta, Victor Samuel Rajadurai, Walter Stunkel, Wayne Cutfield, Wee Meng Han, Wei Wei Pang, Wen Lun Yuan, Yanan Zhu, Yap Seng Chong, Yin Bun Cheung, Yiong Huak Chan, Yung Seng Lee.
The study is supported by the National Research Foundation (NRF) under the Open Fund-Large Collaborative Grant (OF-LCG; MOH-000504) administered by the Singapore Ministry of Health's National Medical Research Council (NMRC) and the Agency for Science, Technology and Research (A*STAR). In RIE2025, GUSTO is supported by funding from the NRF's Human Health and Potential (HHP) Domain, under the Human Potential Programme.
Footnotes
Supplementary material associated with this article can be found in the online version at doi:10.1016/j.ebiom.2022.104111.
Contributor Information
Jian Huang, Email: huang_jian@sics.a-star.edu.sg.
Jonathan Huang, Email: jonathan_huang@sics.a-star.edu.sg.
Appendix. Supplementary materials
References
- 1.Park DC. Cognitive ability in old age is predetermined by age 20 y. Proc Natl Acad Sci. 2019;116(6):1832–1833. doi: 10.1073/pnas.1821142116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Feinstein L, Bynner J. The importance of cognitive development in middle childhood for adulthood socioeconomic status, mental health, and problem behavior. Child Dev. 2004;75(5):1329–1339. doi: 10.1111/j.1467-8624.2004.00743.x. [DOI] [PubMed] [Google Scholar]
- 3.Olsson GM, Hulting AL, Montgomery SM. Cognitive function in children and subsequent type 2 diabetes. Diabetes Care. 2008;31(3):514–516. doi: 10.2337/dc08-0676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Moffitt TE, Arseneault L, Belsky D, et al. A gradient of childhood self-control predicts health, wealth, and public safety. Proc Natl Acad Sci. 2011;108(7):2693–2698. doi: 10.1073/pnas.1010076108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Robson DA, Allen MS, Howard SJ. Self-regulation in childhood as a predictor of future outcomes: a meta-analytic review. Psychol Bull. 2020;146(4):324–354. doi: 10.1037/bul0000227. [DOI] [PubMed] [Google Scholar]
- 6.Richmond-Rakerd LS, Caspi A, Ambler A, et al. Childhood self-control forecasts the pace of midlife aging and preparedness for old age. Proc Natl Acad Sci. 2021;118(3) doi: 10.1073/pnas.2010211118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Steenland K, Zhao L, Goldstein F, Cellar J, Lah J. Biomarkers for predicting cognitive decline in those with normal cognition. J Alzheimers Dis. 2014;40(3):587–594. doi: 10.3233/JAD-2014-131343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Needham BD, Adame MD, Serena G, et al. Plasma and fecal metabolite profiles in autism spectrum disorder. Biol Psychiatry. 2021;89(5):451–462. doi: 10.1016/j.biopsych.2020.09.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Anand NS, Ji Y, Wang G, et al. Maternal and cord plasma branched-chain amino acids and child risk of attention-deficit hyperactivity disorder: a prospective birth cohort study. J Child Psychol Psychiat. 2021;62(7):868–875. doi: 10.1111/jcpp.13332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Henriquez-Henriquez M, Acosta MT, Martinez AF, et al. Mutations in sphingolipid metabolism genes are associated with ADHD. Transl Psychiat. 2020;10(1):231. doi: 10.1038/s41398-020-00881-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Moreau GB, Ramakrishnan G, Cook HL, et al. Childhood growth and neurocognition are associated with distinct sets of metabolites. EBioMedicine. 2019;44:597–606. doi: 10.1016/j.ebiom.2019.05.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Miller AL, Lee HJ, Lumeng JC. Obesity-associated biomarkers and executive function in children. Pediatr Res. 2015;77(1):143–147. doi: 10.1038/pr.2014.158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Diamond A. Executive functions. Annu Rev Psychol. 2013;64:135–168. doi: 10.1146/annurev-psych-113011-143750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Miyake A, Friedman NP. The nature and organization of individual differences in executive functions: four general conclusions. Curr Dir Psychol Sci. 2012;21(1):8–14. doi: 10.1177/0963721411429458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Diamond A. Executive functions. Annu Rev Psychol. 2013;64(1):135–168. doi: 10.1146/annurev-psych-113011-143750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Toll SW, Van der Ven SH, Kroesbergen EH, Van Luit JE. Executive functions as predictors of math learning disabilities. J Learn Disabil. 2011;44(6):521–532. doi: 10.1177/0022219410387302. [DOI] [PubMed] [Google Scholar]
- 17.Cohen JF, Gorski MT, Gruber SA, Kurdziel LB, Rimm EB. The effect of healthy dietary consumption on executive cognitive functioning in children and adolescents: a systematic review. Br J Nutr. 2016;116(6):989–1000. doi: 10.1017/S0007114516002877. [DOI] [PubMed] [Google Scholar]
- 18.Guasch-Ferre M, Bhupathiraju SN, Hu FB. Use of metabolomics in improving assessment of Dietary Intake. Clin Chem. 2018;64(1):82–98. doi: 10.1373/clinchem.2017.272344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fiske A, Holmboe K. Neural substrates of early executive function development. Dev Rev. 2019;52:42–62. doi: 10.1016/j.dr.2019.100866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Groppe K, Elsner B. Executive function and food approach behavior in middle childhood. Front Psychol. 2014;5:447. doi: 10.3389/fpsyg.2014.00447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dohle S, Diel K, Hofmann W. Executive functions and the self-regulation of eating behavior: a review. Appetite. 2018;124:4–9. doi: 10.1016/j.appet.2017.05.041. [DOI] [PubMed] [Google Scholar]
- 22.Best JR, Miller PH. A developmental perspective on executive function. Child Dev. 2010;81(6):1641–1660. doi: 10.1111/j.1467-8624.2010.01499.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Soh SE, Tint MT, Gluckman PD, et al. Cohort profile: growing up in Singapore towards healthy outcomes (GUSTO) birth cohort study. Int J Epidemiol. 2014;43(5):1401–1409. doi: 10.1093/ije/dyt125. [DOI] [PubMed] [Google Scholar]
- 24.Soininen P, Kangas AJ, Wurtz P, Suna T, Ala-Korpela M. Quantitative serum nuclear magnetic resonance metabolomics in cardiovascular epidemiology and genetics. Circ Cardiovasc Genet. 2015;8(1):192–206. doi: 10.1161/CIRCGENETICS.114.000216. [DOI] [PubMed] [Google Scholar]
- 25.Hendrickson NK, McCrimmon AW. Test review: behavior rating inventory of executive Function®, Second Edition (BRIEF®2) by Gioia GA, Isquith PK, Guy SC, & Kenworthy L. Canad J Sch Psychol. 2018;34(1):73–78. [Google Scholar]
- 26.Blomstrand E, Eliasson J, Karlsson HKR, Köhnke R. Branched-chain amino acids activate key enzymes in protein synthesis after physical exercise. J Nutr. 2006;136(1):269S–273S. doi: 10.1093/jn/136.1.269S. [DOI] [PubMed] [Google Scholar]
- 27.Fogel A, McCrickerd K, Fries LR, et al. Eating in the absence of hunger: stability over time and associations with eating behaviours and body composition in children. Physiol Behav. 2018;192:82–89. doi: 10.1016/j.physbeh.2018.03.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sugianto R, Chan MJ, Wong SF, et al. Evaluation of a quantitative food frequency questionnaire for 5-year-old children in an Asian population. J Acad Nutr Diet. 2020;120(3):437–444. doi: 10.1016/j.jand.2019.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kettunen J, Demirkan A, Wurtz P, et al. Genome-wide study for circulating metabolites identifies 62 loci and reveals novel systemic effects of LPA. Nat Commun. 2016;7:11122. doi: 10.1038/ncomms11122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sampathkumar A, Tan KM, Chen L, et al. Genetic link determining the maternal-fetal circulation of vitamin D. Front Genet. 2021;12 doi: 10.3389/fgene.2021.721488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Proitsi P, Kuh D, Wong A, et al. Lifetime cognition and late midlife blood metabolites: findings from a British birth cohort. Transl Psychiatry. 2018;8(1):203. doi: 10.1038/s41398-018-0253-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Favieri F, Forte G, Casagrande M. The executive functions in overweight and obesity: a systematic review of neuropsychological cross-sectional and longitudinal studies. Front Psychol. 2019;10:2126. doi: 10.3389/fpsyg.2019.02126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Martins-Silva T, Vaz JDS, Hutz MH, et al. Assessing causality in the association between attention-deficit/hyperactivity disorder and obesity: a Mendelian randomization study. Int J Obes. 2019;43(12):2500–2508. doi: 10.1038/s41366-019-0346-8. [DOI] [PubMed] [Google Scholar]
- 34.Newgard CB, An J, Bain JR, et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 2009;9(4):311–326. doi: 10.1016/j.cmet.2009.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.VanderWeele TJ. A unification of mediation and interaction: a 4-way decomposition. Epidemiology. 2014;25(5):749–761. doi: 10.1097/EDE.0000000000000121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Blomstrand E, Eliasson J, Karlsson HK, Kohnke R. Branched-chain amino acids activate key enzymes in protein synthesis after physical exercise. J Nutr. 2006;136(1 Suppl):269S–273S. doi: 10.1093/jn/136.1.269S. [DOI] [PubMed] [Google Scholar]
- 37.Wolfe RR. Branched-chain amino acids and muscle protein synthesis in humans: myth or reality? J Int Soc Sports Nutr. 2017;14:30. doi: 10.1186/s12970-017-0184-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fernstrom JD. Branched-chain amino acids and brain function. J Nutr. 2005;135(6 Suppl):1539S–1546S. doi: 10.1093/jn/135.6.1539S. [DOI] [PubMed] [Google Scholar]
- 39.Institute of Medicine (US) Committee on Nutrition T, and the Brain . National Academies Press (US); Washington (DC): 2011. Nutrition and Traumatic Brain Injury: Improving Acute and Subacute Health Outcomes in Military Personnel: Branched-Chain Amino Acids.https://www.ncbi.nlm.nih.gov/books/NBK209312/ Available from: [PubMed] [Google Scholar]
- 40.Cole JT, Mitala CM, Kundu S, et al. Dietary branched chain amino acids ameliorate injury-induced cognitive impairment. Proc Natl Acad Sci USA. 2010;107(1):366–371. doi: 10.1073/pnas.0910280107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tynkkynen J, Chouraki V, van der Lee SJ, et al. Association of branched-chain amino acids and other circulating metabolites with risk of incident dementia and Alzheimer's disease: a prospective study in eight cohorts. Alzheimers Dement. 2018;14(6):723–733. doi: 10.1016/j.jalz.2018.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tournissac M, Vandal M, Tremblay C, et al. Dietary intake of branched-chain amino acids in a mouse model of Alzheimer's disease: Effects on survival, behavior, and neuropathology. Alzheimers Dement. 2018;4:677–687. doi: 10.1016/j.trci.2018.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Brooks GA, Martin NA. Cerebral metabolism following traumatic brain injury: new discoveries with implications for treatment. Front Neurosci. 2014;8:408. doi: 10.3389/fnins.2014.00408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.de la Monte SM, Tong M. Brain metabolic dysfunction at the core of Alzheimer's disease. Biochem Pharmacol. 2014;88(4):548–559. doi: 10.1016/j.bcp.2013.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yudkoff M. Brain metabolism of branched-chain amino acids. Glia. 1997;21(1):92–98. doi: 10.1002/(sici)1098-1136(199709)21:1<92::aid-glia10>3.0.co;2-w. [DOI] [PubMed] [Google Scholar]
- 46.Son SM, Park SJ, Lee H, et al. Leucine signals to mTORC1 via its metabolite acetyl-coenzyme A. Cell Metab. 2019;29(1) doi: 10.1016/j.cmet.2018.08.013. 192-201 e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Liu GY, Sabatini DM. mTOR at the nexus of nutrition, growth, ageing and disease. Nat Rev Mol Cell Biol. 2020;21(4):183–203. doi: 10.1038/s41580-019-0199-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Marsh D, Dragich JM. Autophagy in mammalian neurodevelopment and implications for childhood neurological disorders. Neurosci Lett. 2019;697:29–33. doi: 10.1016/j.neulet.2018.04.017. [DOI] [PubMed] [Google Scholar]
- 49.de la OV, Zazpe I, Ruiz-Canela M. Effect of branched-chain amino acid supplementation, dietary intake and circulating levels in cardiometabolic diseases: an updated review. Curr Opin Clin Nutr Metab Care. 2020;23(1):35–50. doi: 10.1097/MCO.0000000000000614. [DOI] [PubMed] [Google Scholar]
- 50.Raimondo A, Rees MG, Gloyn AL. Glucokinase regulatory protein: complexity at the crossroads of triglyceride and glucose metabolism. Curr Opin Lipidol. 2015;26(2):88–95. doi: 10.1097/MOL.0000000000000155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Varma VR, Oommen AM, Varma S, et al. Brain and blood metabolite signatures of pathology and progression in Alzheimer disease: a targeted metabolomics study. PLoS Med. 2018;15(1) doi: 10.1371/journal.pmed.1002482. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.