Abstract
Purpose
This study was performed to investigate the effect of drain placement on complicated laparoscopic cholecystectomy (cLC) for acute cholecystitis (AC).
Methods
This single-center retrospective study reviewed patients with AC who underwent cLC between January 2010 and December 2020. cLC was defined as open conversion, subtotal cholecystectomy, adjacent organ injury during surgery, operation time of ≥90 minutes, or estimated blood loss of ≥100 mL. One-to-one propensity score matching was performed to compare the surgical outcomes between patients with and without drain on cLC.
Results
A total of 216 patients (mean age, 65.8 years; 75 female patients [34.7%]) underwent cLC, and 126 (58.3%) underwent intraoperative abdominal drainage. In the propensity score-matched cohort (61 patients in each group), early drain removal (≤postoperative day 3) was performed in 42 patients (68.9%). The overall rate of surgical site infection (SSI) was 10.7%. Late drain removal demonstrated significantly worse surgical outcomes than no drain placement and early drain removal for overall complications (13.1% vs. 21.4% vs. 47.4%, p = 0.006), postoperative hospital stay (3.8 days vs. 4.4 days vs. 12.7 days, p < 0.001), and SSI (4.9% vs. 11.9% vs. 31.6%, p = 0.006). In the multivariate analysis, late drain removal was the most significant risk factor for organ space SSI.
Conclusion
This study demonstrated that drain placement is not routinely recommended, even after cLC for AC. When placing a drain, early drain removal is recommended because late drain removal is associated with a higher risk of organ space SSI.
Keywords: Laparoscopy, Cholecystectomy, Acute cholecystitis, Drainage
INTRODUCTION
Acute cholecystitis (AC) is the most common biliary disease, and laparoscopic cholecystectomy (LC) is recognized as the gold standard for AC [1]. It is widely accepted that prophylactic drainage is useful for removing intraabdominal collections and detecting early postoperative complications after surgery, such as bleeding and leakage. In the setting of contaminated abdominal surgery, such as in LC for AC, abdominal drainage can reduce intraabdominal surgical site infection (SSI) by preventing or reducing intraperitoneal fluid collection. A recent meta-analysis concluded that prophylactic drain placement is ineffective in reducing complications in LC, and postoperative recovery is improved if a drain is not present [2]. Compared with uncomplicated LC, complicated LC with increased blood loss and longer operation time is a significant risk factor for postoperative complications, such as SSI [3]. In clinical practice, the placement of an abdominal drain in complicated LC is a good choice for reducing or preventing postoperative SSI. However, there is controversy regarding the actual effect of abdominal drain placement in complicated LC for AC.
This study aimed to compare the surgical outcomes, particularly the occurrence of postoperative SSI, in patients with and without drain placement following complicated LC for AC.
MATERIALS AND METHODS
Study population
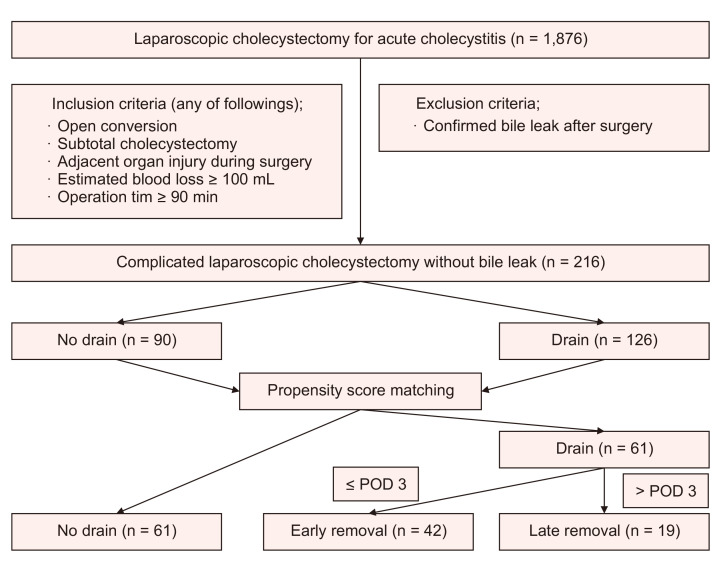
This single-center retrospective study reviewed 1,876 patients with AC who underwent LC in a single institution between January 2010 and December 2020. Patients who underwent complicated LC were included, while those with confirmed bile leak after surgery were excluded. A total of 216 patients were included in this study. Complicated LC was defined as open conversion, subtotal cholecystectomy, adjacent organ injury during surgery, operation time of ≥90 minutes, or estimated blood loss of ≥100 mL. The definition and severity grading of AC were based on the 2018 Tokyo Guidelines (TG 18) [4]. We divided the study population into two groups based on whether a drain was inserted. Propensity scores were used to match the patients with or without a drain. Postoperative day (POD) 3 was selected to distinguish between early and late drain removal (Fig. 1).
Fig. 1.
Study flow diagram. POD, postoperative day.
Perioperative variables
The general condition and physical fitness of each patient before surgery were evaluated using the Charlson age comorbidity index [5] and the American Society of Anesthesiologists physical status (ASA-PS) classification [6]. The results of blood and bile culture tests were classified as positive or negative depending on whether growing bacteria were identified. The operation time was calculated as the time from skin incision to skin closure. Blood loss estimates were obtained from surgical records. Adjacent organ injury was defined as unintended damage (requiring repair) to organs other than the gallbladder such as the bile duct, hepatic artery, duodenum, small bowel, and colon. Subtotal cholecystectomy was defined as making an incision in the gallbladder, aspirating the contents, and removing majority of the gallbladder wall as possible, with the aim of treating the stump instead of removing the entire gallbladder [6]. In principle, only one use of prophylactic antibiotics is recommended before surgery, even in cases of complicated LC in our institution. Exceptionally, in patients with gangrenous or emphysematous cholecystitis, antibiotics were maintained up to POD 2.
Surgical outcomes
Postoperative complications were graded according to the Clavien-Dindo classification [7]. A complication greater than grade III according to the Clavien-Dindo classification was defined as serious complication. The definition and classification of SSI was based on the Centers for Disease Control and Prevention guidelines [8]. Abdominal percutaneous drainage (PCD) was performed when complicated fluid collection, including biloma, was detected. Postoperative pulmonary complications were defined as any complication affecting the respiratory system after anesthesia and surgery, including pneumonia, atelectasis, pleural effusion, pneumothorax, and respiratory failure [9].
Propensity score matching
Propensity score-matched pairs were created by matching patients with and without drains on the logit of the propensity score using 1:1 nearest-neighbor matching without replacement within specified caliper widths. The propensity score was estimated using a logistic regression model with covariates that were expected to affect surgical outcomes: age, sex, body mass index (BMI), ASA-PS classification, Charlson age comorbidity index, previous abdominal surgery, severity of AC according to the TG 18, preoperative percutaneous transhepatic gallbladder drainage (PTGBD), and preoperative endoscopic sphincterotomy (EST). Propensity score matching was performed using the R statistical software, version 3.6.3 (R Foundation for Statistical Computing, Vienna, Austria).
Statistical analysis
Continuous variables were summarized as mean and standard deviation and were compared using Student t-test or analysis of variance. Categorical variables are presented as counts and percentages and were compared using the chi-square test. Multivariate analysis was performed using a logistic regression model to identify the risk factors for organ space SSI. All tests were two-sided, and statistical significance was set at p < 0.05. Analyses were performed using IBM SPSS version 27.0 (IBM Corp., Armonk, NY, USA).
RESULTS
Comparison of patient’s characteristics before and after propensity score matching
The demographic and perioperative variables of the study population before propensity score matching are shown in Table 1. Of the 216 patients included, 126 underwent complicated LC with drainage and 90 underwent complicated LC without drainage. Patients with a drain were older than those without a drain (68.4 years vs. 62.2 years, p = 0.005). Patients with drains had a higher ASA-PS classification (≥III; 37.3% vs. 23.3%, p = 0.029), a higher frequency of preoperative PTGBD (74.6% vs. 58.9%, p = 0.015), and marginally lower BMI (24.6 kg/m2 vs. 25.7 kg/m2, p = 0.057) than those without drains. There were no significant differences in sex (female; 33.3% vs. 36.7%, p = 0.612), history of previous abdominal surgery (26.2% vs. 20.0%, p = 0.291), severity grading of AC (grade I/II/III; 47.6%/43.7%/8.7% vs. 58.9%/36.7%/4.4%, p = 0.193), frequency of preoperative EST (18.3% vs. 17.8%, p = 0.929), positive bile culture (66.0% vs. 72.4%, p = 0.404), and positive blood culture (15.9% vs. 7.6%, p = 0.086) between the groups. Additionally, there were no significant differences in laboratory findings, except hemoglobin (12.9 g/dL vs. 13.5 g/dL, p = 0.016), creatinine (1.11 mg/dL vs. 0.87 mg/dL, p = 0.012), and albumin (3.38 mg/dL vs. 3.63 mg/dL, p = 0.002). Regarding intraoperative variables, patients with a drain had a longer operation time (118.4 minutes vs. 100.9 minutes, p < 0.001), a higher frequency of adjacent organ injury (12.7% vs. 2.2%, p = 0.006), and marginally higher frequency of open conversion (10.3% vs. 3.3%, p = 0.053). There were no significant differences in estimated blood loss (112.8 mL vs. 90.1 mL, p = 0.276) and frequency of subtotal cholecystectomy (4.0% vs. 3.3%, p = 0.808) between the two groups. There were no significant differences between two groups in the pathologic outcomes.
Table 1.
Demographics and perioperative variables of patients with or without drain before propensity score matching
| Variable | Total | Drain | No drain | p value |
|---|---|---|---|---|
| No. of patients | 216 | 126 | 90 | |
| Age (yr) | 65.8 ± 15.9 | 68.4 ± 15.1 | 62.2 ± 16.3 | 0.005 |
| Female sex | 75 (34.7) | 42 (33.3) | 33 (36.7) | 0.612 |
| Body mass index (kg/m2) | 25.0 ± 4.1 | 24.6 ± 3.9 | 25.7 ± 4.2 | 0.057 |
| ASA-PS classification, ≥III | 68 (31.5) | 47 (37.3) | 21 (23.3) | 0.029 |
| CACI, ≥6 | 16 (7.4) | 11 (8.7) | 5 (5.6) | 0.380 |
| Prior abdominal surgery (+) | 51 (23.6) | 33 (26.2) | 18 (20.0) | 0.291 |
| Severity according to the TG 18 | 0.193 | |||
| Mild (grade I) | 113 (52.3) | 60 (47.6) | 53 (58.9) | |
| Moderate (grade II) | 88 (40.7) | 55 (43.7) | 33 (36.7) | |
| Severe (grade III) | 15 (7.0) | 11 (8.7) | 4 (4.4) | |
| Preoperative laboratory findings | ||||
| WBC (×103/mm3) | 13.1 ± 5.6 | 13.3 ± 6.0 | 12.7 ± 5.0 | 0.387 |
| Hemoglobin (g/dL) | 13.2 ± 1.9 | 12.9 ± 1.8 | 13.5 ± 2.0 | 0.016 |
| Platelet (×103/mm3) | 219.7 ± 84.3 | 216.7 ± 85.9 | 223.9 ± 82.3 | 0.531 |
| PT, INR | 1.14 ± 0.20 | 1.16 ± 0.24 | 1.13 ± 0.14 | 0.262 |
| Creatinine (mg/dL) | 1.01 ± 0.68 | 1.11 ± 0.84 | 0.87 ± 0.31 | 0.012 |
| Albumin (g/dL) | 3.48 ± 0.61 | 3.38 ± 0.60 | 3.63 ± 0.58 | 0.002 |
| AST (IU/L) | 103.7 ± 221.6 | 113.7 ± 251.7 | 89.8 ± 171.2 | 0.406 |
| ALT (IU/L) | 88.1 ± 158.2 | 85.7 ± 162.6 | 91.6 ± 152.6 | 0.786 |
| Total bilirubin (mg/dL) | 2.10 ± 2.21 | 2.02 ± 1.77 | 2.20 ± 2.72 | 0.552 |
| C-reactive protein (mg/dL) | 12.3 ± 0.6 | 13.0 ± 11.3 | 11.2 ± 11.5 | 0.262 |
| Preoperative EST | 39 (18.1) | 23 (18.3) | 16 (17.8) | 0.929 |
| Preoperative PTGBD | 147 (68.1) | 94 (74.6) | 53 (58.9) | 0.015 |
| Positive for bile culture | 106/155 (68.4) | 64/97 (66.0) | 42/58 (72.4) | 0.404 |
| Positive for blood culture | 24/192 (12.5) | 18/113 (15.9) | 6/79 (7.6) | 0.086 |
| Operation time (min) | 111.1 ± 35.4 | 118.4 ± 38.5 | 100.9 ± 27.8 | <0.001 |
| Estimated blood loss (mL) | 103.4 ± 160.4 | 112.8 ± 182.7 | 90.1 ± 122.1 | 0.276 |
| Adjacent organ injury detected during surgery | 18 (8.3) | 16 (12.7) | 2 (2.2) | 0.006 |
| Bile duct | 7 (3.2) | 6 (4.8) | 1 (1.1) | |
| Duodenum | 2 (0.9) | 2 (1.6) | 0 (0) | |
| Colon | 1 (0.5) | 1 (0.8) | 0 (0) | |
| Hepatic artery | 5 (2.3) | 5 (4.0) | 0 (0) | |
| Small intestine | 3 (1.4) | 2 (1.6) | 1 (1.1) | |
| Open conversion | 16 (7.4) | 13 (10.3) | 3 (3.3) | 0.053 |
| Subtotal cholecystectomy | 8 (3.7) | 5 (4.0) | 3 (3.3) | 0.808 |
| Pathology | 0.249 | |||
| Chronic cholecystitis | 88 (40.7) | 50 (39.7) | 38 (42.2) | |
| Acute cholecystitis | 82 (38.0) | 46 (36.5) | 36 (40.0) | |
| Gangrenous or emphysematous cholecystitis | 40 (18.5) | 26 (20.6) | 14 (15.6) | |
| Others | 6 (2.8) | 4 (3.2) | 2 (2.2) |
Values are presented as number only, mean ± standard deviation, or number (%).
ASA-PS, American Society of Anesthesiologists physical status; CACI, Charlson age comorbidity index; TG 18, 2018 Tokyo Guidelines; WBC, white blood cell; PT, prothrombin time; INR, international normalized ratio; AST, aspartate transaminase; ALT, alanine transaminase; EST, endoscopic sphincterotomy; PTGBD, percutaneous transhepatic gallbladder drainage.
The characteristics of the study population after propensity score matching are listed in Table 2. After matching (61 patients in each group), there were no significant differences in demographics and perioperative variables of patients with or without drain, except for operation time (119.5 minutes vs. 96.8 minutes, p < 0.001). When comparing patients with early drain removal and patients with late drain removal, there were also no significant differences in demographics and perioperative variables except preoperative albumin level (3.58 g/dL vs. 3.19 g/dL, p = 0.015) and open conversion rate (0.0% vs. 21.1%, p = 0.002).
Table 2.
Demographics and perioperative variables of patients with or without drain and with early removal or with late removal, after propensity score matching
| Variable | Drain placement | p value | Timing of drain removal | p value | ||
|---|---|---|---|---|---|---|
|
|
|
|||||
| Yes (n = 61) | No (n = 61) | Early (n = 42) | Late (n = 19) | |||
| Age (yr) | 67.7 ± 15.7 | 67.6 ± 12.7 | 0.970 | 65.6 ± 16.8 | 72.5 ± 12.0 | 0.110 |
| Female sex | 20 (32.8) | 20 (32.8) | >0.999 | 14 (33.3) | 6 (31.6) | 0.892 |
| Body mass index (kg/m2) | 25.0 ± 3.8 | 24.9 ± 3.5 | 0.894 | 25.3 ± 4.1 | 24.3 ± 3.2 | 0.328 |
| ASA-PS classification, ≥III | 17 (27.9) | 16 (26.2) | 0.839 | 9 (21.4) | 8 (42.1) | 0.095 |
| CACI, ≥6 | 5 (8.2) | 3 (4.9) | 0.464 | 4 (9.5) | 1 (5.3) | 0.574 |
| Prior abdominal surgery (+) | 15 (24.6) | 15 (24.6) | >0.999 | 9 (21.4) | 6 (31.6) | 0.394 |
| Severity according to the TG 18 | 0.254 | 0.265 | ||||
| Mild (grade I) | 30 (49.2) | 39 (63.9) | 23 (54.8) | 7 (36.8) | ||
| Moderate (grade II) | 26 (42.6) | 18 (29.5) | 15 (35.7) | 11 (57.9) | ||
| Severe (grade III) | 5 (8.2) | 4 (6.6) | 4 (9.5) | 1 (5.3) | ||
| Preoperative laboratory findings | ||||||
| WBC (×103/mm3) | 13.6 ± 6.6 | 12.5 ± 5.1 | 0.250 | 13.4 ± 5.6 | 14.1 ± 5.8 | 0.660 |
| Hemoglobin (g/dL) | 13.3 ± 1.7 | 13.6 ± 1.8 | 0.360 | 13.6 ± 1.5 | 12.7 ± 1.9 | 0.075 |
| Platelet (×103/mm3) | 222.8 ± 75.4 | 223.9 ± 86.7 | 0.942 | 226.2 ± 74.8 | 215.5 ± 78.2 | 0.619 |
| PT, INR | 1.11 ± 0.13 | 1.13 ± 0.14 | 0.398 | 1.09 ± 0.12 | 1.16 ± 0.13 | 0.075 |
| Creatinine (mg/dL) | 1.05 ± 0.69 | 0.91 ± 0.33 | 0.176 | 0.92 ± 0.51 | 1.35 ± 0.95 | 0.085 |
| Albumin (g/dL) | 3.46 ± 0.59 | 3.56 ± 0.56 | 0.313 | 3.58 ± 0.62 | 3.19 ± 0.41 | 0.015 |
| AST (IU/L) | 146.7 ± 343.6 | 101.3 ± 200.1 | 0.374 | 185.4 ± 405.2 | 61.2 ± 91.8 | 0.194 |
| ALT (IU/L) | 101.5 ± 198.7 | 104.4 ± 175.3 | 0.931 | 123.6 ± 229.3 | 52.6 ± 90.3 | 0.089 |
| Total bilirubin (mg/dL) | 1.98 ± 1.82 | 2.29 ± 2.98 | 0.483 | 1.91 ± 1.72 | 2.12 ± 2.06 | 0.696 |
| C-reactive protein (mg/dL) | 11.9 ± 11.6 | 11.8 ± 11.3 | 0.985 | 11.7 ± 12.1 | 12.2 ± 10.8 | 0.887 |
| Preoperative EST | 10 (16.4) | 10 (16.4) | >0.999 | 7 (16.7) | 3 (15.8) | 0.932 |
| Preoperative PTGBD | 41 (67.2) | 41 (67.2) | >0.999 | 28 (66.7) | 13 (68.4) | 0.892 |
| Positive for bile culture | 24/44 (54.5) | 33/44 (75.0) | 0.074 | 15/31 (48.4) | 9/13 (69.2) | 0.205 |
| Positive for blood culture | 5/53 (9.4) | 5/53 (9.4) | 1.000 | 4/38 (10.5) | 1/15 (6.7) | 0.665 |
| Operation time (min) | 119.5 ± 43.6 | 96.8 ± 26.5 | <0.001 | 111.8 ± 12.1 | 136.6 ± 67.3 | 0.134 |
| Estimated blood loss (mL) | 102.0 ± 195.1 | 98.5 ± 131.6 | 0.908 | 109.3 ± 230.0 | 85.8 ± 77.6 | 0.556 |
| Adjacent organ injury detected during surgery | 6 (9.8) | 2 (3.3) | 0.143 | 3 (7.1) | 3 (15.8) | 0.294 |
| Bile duct | 2 (3.3) | 1 (1.6) | 1 (2.4) | 1 (5.3) | ||
| Duodenum | 1 (1.6) | 0 (0) | 1 (2.4) | 0 (0) | ||
| Colon | 1 (1.6) | 0 (0) | 0 (0) | 1 (5.3) | ||
| Hepatic artery | 1 (1.6) | 0 (0) | 1 (2.4) | 0 (0) | ||
| Small intestine | 1 (1.6) | 1 (1.6) | 0 (0) | 1 (5.3) | ||
| Open conversion | 4 (6.6) | 3 (4.9) | 0.697 | 0 (0) | 4 (21.1) | 0.002 |
| Subtotal cholecystectomy | 2 (3.3) | 1 (1.6) | 0.559 | 1 (2.4) | 1 (5.3) | 0.558 |
| Pathology | 0.496 | 0.298 | ||||
| Chronic cholecystitis | 23 (37.7) | 28 (45.9) | 19 (45.2) | 4 (21.1) | ||
| Acute cholecystitis | 25 (41.0) | 25 (41.0) | 15 (35.7) | 10 (52.6) | ||
| Gangrenous or emphysematous cholecystitis | 11 (18.0) | 7 (11.5) | 6 (14.3) | 5 (26.3) | ||
| Others | 2 (3.2) | 1 (1.6) | 2 (4.8) | 0 (0) | ||
Values are presented as mean ± standard deviation or number (%).
ASA-PS, American Society of Anesthesiologists physical status; CACI, Charlson age comorbidity index; TG 18, 2018 Tokyo Guidelines; WBC, white blood cell; PT, prothrombin time; INR, international normalized ratio; AST, aspartate transaminase; ALT, alanine transaminase; EST, endoscopic sphincterotomy; PTGBD, percutaneous transhepatic gallbladder drainage.
Comparison of surgical outcomes after propensity score matching
The surgical outcomes for the no drain, early removal, and late removal cohorts after propensity score matching are listed in Table 3. In the propensity score-matched cohort (61 patients in each group), early drain removal (≤POD 3) was performed in 42 patients (68.9%). Late removal of the drain demonstrated significantly worse surgical outcomes when compared to no drain placement and early removal of drain for overall complication (13.1% vs. 21.4% vs. 47.4%, p = 0.006), postoperative hospital stay (3.8 days vs. 4.4 days vs. 12.7 days, p < 0.001), SSI (4.9% vs. 11.9% vs. 31.6%, p = 0.006), and reoperation (0.0% vs. 0.0% vs. 10.5%, p = 0.004). There were no statistically significant differences in serious complications (4.9% vs. 4.8% vs. 15.8%, p = 0.333), in-hospital mortality (0.0% vs. 2.4% vs. 0.0%, p = 0.383), abdominal PCD insertion (3.3% vs. 4.8% vs. 5.3%, p = 0.897), pulmonary complications (1.6% vs. 2.4% vs. 0.0%, p = 0.795), and readmission (3.3% vs. 2.4% vs. 5.3%, p = 0.843) among the three groups.
Table 3.
Surgical outcomes for no drain, early removal, and late removal cohorts
| Variable | Total (n = 122) |
No drain (n = 61) |
Early removal (n = 42) |
Late removal (n = 19) |
p value |
|---|---|---|---|---|---|
| Overall complication | 26 (21.3) | 8 (13.1) | 9 (21.4) | 9 (47.4) | 0.006 |
| Serious complicationa) | 8 (6.6) | 3 (4.9) | 2 (4.8) | 3 (15.8) | 0.333 |
| In-hospital mortality | 1 (0.8) | 0 (0) | 1 (2.4) | 0 (0) | 0.383 |
| Postoperative hospital stay (day) | 5.4 ± 9.2 | 3.8 ± 4.2 | 4.4 ± 3.6 | 12.7 ± 20.4 | <0.001 |
| Surgical site infection | 13 (10.7) | 3 (4.9) | 5 (11.9) | 6 (31.6) | 0.006 |
| Superficial | 1 (0.8) | 0 (0) | 1 (2.4) | 0 (0) | |
| Deep | 1 (0.8) | 0 (0) | 0 (0) | 1 (5.3) | |
| Organ/space | 11 (9.0) | 3 (4.9) | 4 (9.5) | 5 (26.3) | |
| Abdominal PCD insertion | 5 (4.1) | 2 (3.3) | 2 (4.8) | 1 (5.3) | 0.897 |
| Pulmonary complication | 2 (1.6) | 1 (1.6) | 1 (2.4) | 0 (0) | 0.795 |
| Reoperation | 2 (1.6) | 0 (0) | 0 (0) | 2 (10.5) | 0.004 |
| Readmission | 4 (3.3) | 2 (3.3) | 1 (2.4) | 1 (5.3) | 0.843 |
Values are presented as number (%) or mean ± standard deviation.
PCD, percutaneous drainage.
a)Clavien-Dindo classification ≥ III.
Risk factors for organ space surgical site infection
Among the participants, 16 (7.4%) had organ space SSI after surgery. The results of the univariate and multivariate analyses of risk factors for organ space SSI are presented in Table 4. Univariate analyses revealed that severe (grade III) AC, elevated creatinine (≥2.0 mg/dL), positive blood culture, and late drain removal were statistically significant risk factors. In multivariate analyses, the following were identified as significant risk factors for organ space SSI: severe (grade III) AC (odds ratio [OR], 7.047; 95% confidence interval [CI], 1.372–36.201; p = 0.019), positive blood culture (OR, 5.772; 95% CI, 1.499–22.229; p = 0.011), and late drain removal (OR, 7.196; 95% CI, 1.336–38.755; p = 0.022).
Table 4.
Univariate and multivariate analyses of risk factor for organ space surgical site infection in study population
| Factor | Univariate analysis | Multivariate analysis | |||
|---|---|---|---|---|---|
|
|
|
||||
| OR (95% CI) | p value | OR (95% CI) | p value | ||
| Sex | |||||
| Female | 1 (Reference) | ||||
| Male | 1.651 (0.513–5.310) | 0.400 | |||
| Age (yr) | |||||
| <80 | 1 (Reference) | ||||
| ≥80 | 2.071 (0.678–6.327) | 0.202 | |||
| Body mass index (kg/m2) | |||||
| ≥20 | 1 (Reference) | ||||
| <20 | 2.198 (0.575–8.408) | 0.250 | |||
| ASA-PS classification | |||||
| <III | 1 (Reference) | ||||
| ≥III | 0.988 (0.330–2.965) | 0.983 | |||
| CACI | |||||
| <6 | 1 (Reference) | ||||
| ≥6 | 1.898 (0.392–9.196) | 0.426 | |||
| Previous abdominal surgery | |||||
| No | 1 (Reference) | ||||
| Yes | 0.731 (0.200–2.672) | 0.635 | |||
| Severity of acute cholecystitis | |||||
| Mild (grade I) | 1 (Reference) | 1 (Reference) | |||
| Moderate (grade II) | 0.721 (0.204–2.546) | 0.611 | – | ||
| Severe (grade III) | 7.571 (2.026–28.290) | 0.003 | 7.047 (1.372–36.201) | 0.019 | |
| Albumin (mg/dL) | |||||
| ≥3.0 | 1 (Reference) | ||||
| <3.0 | 2.643 (0.904–7.731) | 0.076 | |||
| Total bilirubin (mg/dL) | |||||
| <2.0 | 1 (Reference) | ||||
| ≥2.0 | 1.904 (0.677–5.355) | 0.222 | |||
| Creatinine (mg/dL) | |||||
| <2.0 | 1 (Reference) | 1 (Reference) | |||
| ≥2.0 | 5.306 (1.268–22.191) | 0.022 | 1.563 (0.238–10.279) | 0.642 | |
| CRP (mg/dL) | |||||
| <20.0 | 1 (Reference) | ||||
| ≥20.0 | 0.966 (0.298–3.135) | 0.954 | |||
| Preoperative EST | |||||
| No | 1 (Reference) | ||||
| Yes | 1.571 (0.479–5.160) | 0.456 | |||
| Preoperative PTGBD | |||||
| No | 1 (Reference) | ||||
| Yes | 1.035 (0.345–3.104) | 0.951 | |||
| Bile culture | |||||
| Negative | 1 (Reference) | ||||
| Positive | 2.180 (0.453–10.494) | 0.331 | |||
| Blood culture | |||||
| Negative | 1 (Reference) | 1 (Reference) | |||
| Positive | 5.889 (1.879–18.454) | 0.002 | 5.772 (1.499–22.229) | 0.011 | |
| Operation time (min) | |||||
| <120 | 1 (Reference) | ||||
| ≥120 | 1.191 (0.415–3.417) | 0.745 | |||
| Estimated blood loss (mL) | |||||
| <200 | 1 (Reference) | ||||
| ≥200 | 1.088 (0.294–4.021) | 0.899 | |||
| Subtotal cholecystectomy | |||||
| No | 1 (Reference) | ||||
| Yes | 4.619 (0.853–25.026) | 0.076 | |||
| Open conversion | |||||
| No | 1 (Reference) | ||||
| Yes | 0.822 (0.102–6.658) | 0.854 | |||
| Adjacent organ injury | |||||
| No | 1 (Reference) | ||||
| Yes | 2.484 (0.644–9.584) | 0.187 | |||
| Drain | |||||
| No | 1 (Reference) | 1 (Reference) | |||
| Early removal | 1.768 (0.410–7.636) | 0.445 | – | ||
| Late removal | 7.484 (1.866–30.010) | 0.005 | 7.196 (1.336–38.755) | 0.022 | |
OR, odds ratio; CI, confidence interval; ASA-PS, American Society of Anesthesiologists physical status; CACI, Charlson age comorbidity index; CRP, C-reactive protein; EST, endoscopic sphincterotomy; PTGBD, percutaneous transhepatic gallbladder drainage.
DISCUSSION
According to several recently published studies, routine abdominal drainage after elective uncomplicated LC is not recommended [10–13]. In actual clinical practice, most of these principles are followed. Drain placement after LC is typically performed for patients with AC. In patients with AC, the placement of a drain is determined depending on the intraoperative situation, such as the occurrence of an unintended event during surgery, increased blood loss, and longer operation time. However, no study has investigated the effect of drain placement on complicated LC in AC. Our study is meaningful because it is the first to report the comparison of surgical outcomes in patients with and without drain placement following complicated LC for AC.
Bile leaks are an immediate technical problem in LC. Most bile leaks following LC originate from the cystic duct stump and aberrant branches of the hepatic duct due to intraoperative injury to the bile duct. They mostly present as percutaneous bile drainage through an intraoperatively placed drain [14]. Most of these leaks can be successfully managed with a maintained drain alone. It is difficult to determine the effectiveness of drain management in patients with bile leakage because early drain removal is not possible. Therefore, patients with bile leakage were excluded from the present study.
SSIs are the most common and costly hospital-acquired infections. SSIs after surgery for infectious diseases are associated with an increased length of hospital stay and risk of mortality [15]. Drain placement in the abdominal cavity after various surgical procedures has been beneficial for many decades. Nevertheless, in recent years, doubts about the effectiveness of drain placement have increased, and studies have been conducted on the effects of not placing drains in various surgical procedures [16–18]. In LC for AC, three recently published randomized clinical trials reported that there is no significant benefit in placing a drain for preventing or reducing postoperative morbidities, including SSI [19–21]. In our study on complicated LC, the surgical outcomes were worse in patients with drains than in those without drains in terms of overall postoperative complications, postoperative hospital stay, SSI, and reoperation. Therefore, routine drain placement is not recommended, even in complicated LC for AC.
The timing of drain removal is also very important if a drain is placed. Early drain removal is a frequent practice for LC in the present study. However, 31.1% of patients had late drain removal after complicated LC. According to recently published studies, even after major hepatectomy and pancreatectomy, early drain removal has shown better surgical outcomes [22–24]. In the present study, late drain removal demonstrated significantly worse surgical outcomes than no drain placement and early drain removal for overall complications, postoperative hospital stay, SSI, and reoperation. However, it may not be clear whether the drain was removed late due to an intraabdominal problem or whether the SSI occurred due to the late removal of drain. To analyze this problem, we checked the 19 patients in late removal group after propensity matching. In all patients, no change in the color of drain or signs of infection appeared within POD 3. Median duration of drain in late removal group was 4 days (range, 4–8 days). The reason for the late removal of drain was patient’s poor general condition in many cases, and other reasons were unclear. In one patient who received additional abdominal PCD (POD 7), organ space SSI was confirmed after the drain was removed (POD 4). Therefore, it was confirmed that removal of drain was not delayed due to intraabdominal problems in the late removal of drain group in present study. Furthermore, in the multivariate analysis, late drain removal was identified as the most significant risk factor for organ space SSI. Therefore, when placing a drain in a complicated LC, early drain removal is recommended after checking for bleeding or bile leaks.
This study had several limitations. First, this was a retrospective study with a small sample size. However, we tried to reduce bias through propensity score matching. Nevertheless, there were differences in demographics and perioperative variables (preoperative albumin level and open conversion rate) due to the insufficient number of patients in the subgroup according to the timing of drain removal. Second, the definition of complicated LC in the present study is not generalized. Third, there was no specific algorithm for placing a drain during LC at our institution. Thus, it was left to the individual surgeon whether to place a drain or not. Finally, postoperative treatments, including the use of antibiotics, were not included in the data, which could have been a potential bias in this study.
In conclusion, this study demonstrated that drain placement is not routinely recommended, even after complicated LC for AC. When placing a drain, early drain removal is recommended because late drain removal is associated with a higher risk of organ space SSI.
NOTES
Ethical statements
This study was approved by the Institutional Review Board of Konyang University Hospital, and the requirement for obtaining informed consent was waived due to the retrospective study design (No. 2022-01-021).
Authors’ contributions
Conceptualization, Formal analysis, Methodology, Visualization: SJL, ISC, JIM
Data curation, Investigation: All authors
Writing-original draft: SJL
Writing-review & editing: SJL, ISC, JIM
All authors read and approved the final manuscript.
Conflict of interest
All authors have no conflicts of interest to declare.
Funding/support
None.
REFERENCES
- 1.Strasberg SM. Clinical practice. Acute calculous cholecystitis. N Engl J Med. 2008;358:2804–2811. doi: 10.1056/NEJMcp0800929. [DOI] [PubMed] [Google Scholar]
- 2.Picchio M, De Cesare A, Di Filippo A, Spaziani M, Spaziani E. Prophylactic drainage after laparoscopic cholecystectomy for acute cholecystitis: a systematic review and meta-analysis. Updates Surg. 2019;71:247–254. doi: 10.1007/s13304-019-00648-x. [DOI] [PubMed] [Google Scholar]
- 3.Utsumi M, Sakurai Y, Narusaka T, et al. C-reactive protein to albumin ratio predicts difficult laparoscopic cholecystectomy in patients with acute cholecystitis diagnosed according to the Tokyo Guidelines 2018. Asian J Endosc Surg. 2022 Feb 8; doi: 10.1111/ases.13035. [Epub]. DOI: 10.1111/ases.13035. [DOI] [PubMed] [Google Scholar]
- 4.Yokoe M, Hata J, Takada T, et al. Tokyo Guidelines 2018: diagnostic criteria and severity grading of acute cholecystitis (with videos) J Hepatobiliary Pancreat Sci. 2018;25:41–54.:diagnostic. doi: 10.1002/jhbp.515. [DOI] [PubMed] [Google Scholar]
- 5.Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47:1245–1251. doi: 10.1016/0895-4356(94)90129-5. [DOI] [PubMed] [Google Scholar]
- 6.Wakabayashi G, Iwashita Y, Hibi T, et al. Tokyo Guidelines 2018: surgical management of acute cholecystitis: safe steps in laparoscopic cholecystectomy for acute cholecystitis (with videos) J Hepatobiliary Pancreat Sci. 2018;25:73–86. doi: 10.1002/jhbp.517. [DOI] [PubMed] [Google Scholar]
- 7.Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250:187–196. doi: 10.1097/SLA.0b013e3181b13ca2. [DOI] [PubMed] [Google Scholar]
- 8.Berríos-Torres SI, Umscheid CA, Bratzler DW, et al. Centers for Disease Control and Prevention guideline for the prevention of surgical site infection, 2017. JAMA Surg. 2017;152:784–791. doi: 10.1001/jamasurg.2017.0904. [DOI] [PubMed] [Google Scholar]
- 9.Jammer I, Wickboldt N, Sander M, et al. Standards for definitions and use of outcome measures for clinical effectiveness research in perioperative medicine: European Perioperative Clinical Outcome (EPCO) definitions: a statement from the ESA-ESICM joint taskforce on perioperative outcome measures. Eur J Anaesthesiol. 2015;32:88–105. doi: 10.1097/EJA.0000000000000118. [DOI] [PubMed] [Google Scholar]
- 10.Picchio M, Lucarelli P, Di Filippo A, De Angelis F, Stipa F, Spaziani E. Meta-analysis of drainage versus no drainage after laparoscopic cholecystectomy. JSLS. 2014;18:e2014.00242. doi: 10.4293/JSLS.2014.00242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wong CS, Cousins G, Duddy JC, Walsh SR. Intra-abdominal drainage for laparoscopic cholecystectomy: a systematic review and meta-analysis. Int J Surg. 2015;23(Pt A):87–96. doi: 10.1016/j.ijsu.2015.09.033. [DOI] [PubMed] [Google Scholar]
- 12.Yong L, Guang B. Abdominal drainage versus no abdominal drainage for laparoscopic cholecystectomy: a systematic review with meta-analysis and trial sequential analysis. Int J Surg. 2016;36(Pt A):358–368. doi: 10.1016/j.ijsu.2016.11.083. [DOI] [PubMed] [Google Scholar]
- 13.Bugiantella W, Vedovati MC, Becattini C, Canger RC, Avenia N, Rondelli F. To drain or not to drain elective uncomplicated laparoscopic cholecystectomy? A systematic review and meta-analysis. J Hepatobiliary Pancreat Sci. 2014;21:787–794. doi: 10.1002/jhbp.127. [DOI] [PubMed] [Google Scholar]
- 14.Kim KH, Kim TN. Endoscopic management of bile leakage after cholecystectomy: a single-center experience for 12 years. Clin Endosc. 2014;47:248–253. doi: 10.5946/ce.2014.47.3.248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ban KA, Minei JP, Laronga C, et al. American College of Surgeons and Surgical Infection Society: surgical site infection guidelines, 2016 update. J Am Coll Surg. 2016;2017;224:59–74. doi: 10.1016/j.jamcollsurg.2016.10.029. [DOI] [PubMed] [Google Scholar]
- 16.Lyu Y, Cheng Y, Wang B, Zhao S, Chen L. Peritoneal drainage or no drainage after pancreaticoduodenectomy and/or distal pancreatectomy: a meta-analysis and systematic review. Surg Endosc. 2020;34:4991–5005. doi: 10.1007/s00464-019-07293-w. [DOI] [PubMed] [Google Scholar]
- 17.Anweier N, Apaer S, Zeng Q, et al. Is routine abdominal drainage necessary for patients undergoing elective hepatectomy? A protocol for systematic review and meta-analysis. Medicine (Baltimore) 2021;100:e24689. doi: 10.1097/MD.0000000000024689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li Z, Zhao L, Cheng Y, Cheng N, Deng Y. Abdominal drainage to prevent intra-peritoneal abscess after open appendectomy for complicated appendicitis. Cochrane Database Syst Rev. 2018;5:CD010168. doi: 10.1002/14651858.CD010168.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim EY, Lee SH, Lee JS, et al. Is routine drain insertion after laparoscopic cholecystectomy for acute cholecystitis beneficial? A multicenter, prospective randomized controlled trial. J Hepatobiliary Pancreat Sci. 2015;22:551–557. doi: 10.1002/jhbp.244. [DOI] [PubMed] [Google Scholar]
- 20.Lucarelli P, Picchio M, Martellucci J, et al. Drain after laparoscopic cholecystectomy for acute calculous cholecystitis. A pilot randomized study. Indian J Surg. 2015;77(Suppl 2):288–292. doi: 10.1007/s12262-012-0797-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Park JS, Kim JH, Kim JK, Yoon DS. The role of abdominal drainage to prevent of intra-abdominal complications after laparoscopic cholecystectomy for acute cholecystitis: prospective randomized trial. Surg Endosc. 2015;29:453–457. doi: 10.1007/s00464-014-3685-5. [DOI] [PubMed] [Google Scholar]
- 22.Seykora TF, Liu JB, Maggino L, Pitt HA, Vollmer CM., Jr Drain management following distal pancreatectomy: characterization of contemporary practice and impact of early removal. Ann Surg. 2020;272:1110–1117. doi: 10.1097/SLA.0000000000003205. [DOI] [PubMed] [Google Scholar]
- 23.Sahara K, Tsilimigras DI, Moro A, et al. Variation in drain management among patients undergoing major hepatectomy. J Gastrointest Surg. 2021;25:962–970. doi: 10.1007/s11605-020-04610-w. [DOI] [PubMed] [Google Scholar]
- 24.Dai M, Liu Q, Xing C, et al. Early drain removal is safe in patients with low or intermediate risk of pancreatic fistula after pancreaticoduodenectomy: a multicenter, randomized controlled trial. Ann Surg. 2022;275:e307–e314. doi: 10.1097/SLA.0000000000004992. [DOI] [PubMed] [Google Scholar]