Abstract
Simple Summary
Genome-wide runs of homozygosity (ROH) are excellent in understanding population history, estimating genomic inbreeding, and deciphering genetic architecture of complex traits and diseases, as well as identifying genes of agro-economic traits. ROH are defined as continuous region on the chromosome where an individual is homozygous across the genome. This study identified distribution of ROH in the six selected beef cattle breeds, Chinese Simentaler (CSI), Belgian Blue (BEL), and South African Angus (ANG), Nguni (NGU), Bonsmara (BON), and Simentaler (SIM), using Bovine BeadChip markers. Furthermore, nine candidate genes, CDF9, MARCH1, WDR19, SLOICI, ST7, DOP1B, CFAF9, INHBA, and ADAMTS1, were suggested to be associated with semen QTL traits and reported moderate inbreeding in some breeds with high to low correlation inbreeding between breeds. The study findings will allow proper guidelines for breeder’s societies.
Abstract
In this study, runs of homozygosity (ROH) and quantitative trait locus/association (QTL) for semen parameters in selected Chinese and South African beef cattle breed were estimated. The computed results showed 7516 ROH were observed between classes 0–5 Mb with no ROH observed in classes >40 Mb. Distribution of ROH showed high level of genomic coverage for ANG, NGU, CSI, and BEL breeds. Approximately 13 genomic regions with QTL were controlling sperm motility, sperm concentration, semen volume, sperm count, sperm head abnormalities, sperm tail abnormalities, sperm integrity, and percentage of abnormal sperm traits. Nine candidate genes, CDF9, MARCH1, WDR19, SLOICI, ST7, DOP1B, CFAF9, INHBA, and ADAMTS1, were suggested to be associated with above mentioned QTL traits. The results for inbreeding coefficient showed moderate correlation between FROH vs FHOM at 0.603 and high correlation between FROH 0–5 Mb 0.929, and lowest correlation for 0–>40 Mb 0.400. This study suggested recent inbreeding in CSI, BEL, ANG, BON, SIM, and NGU breeds. Furthermore, it highlighted varied inbreeding levels and identified QTL for semen traits and genes of association. These results can assist in implementation of genetic improvement strategies for bulls and provide awareness and proper guidelines in developing breeding programs.
Keywords: ROH, inbreeding coefficient, quantitative trait locus/association and breeds
1. Introduction
Breeding programs for cattle have been known to be widely implemented in a lot of countries in the world. Several countries have regarded breeding as an important aspect in beef cattle, while some countries have already implemented those programs. These programs include breeds like South African Bonsmara, an indigenous cross between European Shorthorn, Hereford, and Afrikaner [1], and the indigenous Southern African (South Africa, Eswatini, Namibia, Zimbabwe, Botswana, and Angola) Nguni breed a cross between Bos taurus and Bos indicus. The Simmentaler, a cross between German cattle and Swiss cattle is still one of the most utilized breeds in breeding programs. The Scottish Angus [2] has been in South Africa over a hundred years, even part of the South African Studbook. The Belgian blue originally from Belgium and is the first known Belgian breeds [3]. These programs lead to fast genetic progress, but they also lead to the accumulation of inbreeding via heavy impact of a few selected individuals [4]. Therefore, using genome-wide runs of homozygosity (ROH) is excellent in understanding population history and estimating genomic inbreeding, solving genetic architecture of complex traits and diseases, as well as identifying genes of agro-economic traits [5]. ROH can be defined as continuous regions on the chromosome where an individual is homozygous across the genome [5]. They occur when both haplotypes transmitted from parents are identical and inherited from a common ancestry [5]. An increase in homozygous loci and regions of homozygosity may be an indication of loss in genetic diversity [6], or population going through bottleneck.
A few studies have reported the negative impact of high homozygosity on fertility traits, including bull semen quality [7], calving rate [8], stillbirths, and dystocia [9]. A previous study [10] showed distinct differences in the length, quantity, and frequency of ROH between 11 Polish breeds as well as highlighting level of genomic inbreeding within breeds. The authors of [7] reported that longer ROH (>100 Mb) indicate recent inbreeding that has occurred within a population; however, shorter ROH may indicate ancient inbreeding that has occurred in population. Long ROH may persist in out bred individuals, due to unusual mutation, linkage disequilibrium (LD), and recombination rates at certain genomic locations [8]. The investigation of ROH in farm animals also suggested their importance contributions to complex traits [6]. Runs of homozygosity in American Holstein cattle revealed candidate genes associated with reproduction traits affecting fertility [11]. There are several factors which affects the quality of ROH calling rate; these include the marker density, their distribution across the genome, the quality of the genotype calling rates, and minor allele frequency [12]. Single nucleotide polymorphism provide information about both past and more recent demographic variations of a population [12], allowing a comparison of the degree of homozygosity among populations with varying degrees of isolation and inbreeding [5]. The authors [13] stated different approaches to estimating inbreeding at an individual and population level using genetic information. The objective of the study is to use distribution of ROH to identify inbred individuals in both South African and Chinese beef bulls and identify QTL for semen traits and their associated genes. This will assist in understanding population history and reveal genes linked to with semen traits. It will further show levels of inbreeding in commercial breeds, especially beef breeds [10].
2. Materials and Methods
2.1. Sampling, Genotyping and Quality Control
Approximately 144 semen samples were collected from South African and Chinese bulls, and genomic DNA was extracted from South African Bonsmara (n = 21), Angus (n = 22), Nguni (n = 28), and Simmental (n = 25), and from Chinese Belgian blue (n = 24) and Chinese Simmental (n = 24) breeds. Genotyping was conducted using Illumina Bovine 150 K BeadChip (Illumina, Inc, San Diego, CA, USA) for South African breeds and GGP Neogen Bovine 150 K (Neogen, Lansing, MI, USA) was used for Chinese breeds. All samples were processed with Genome Studio 2.0 software (Illumina, Inc, San Diego, CA, USA). Plink v1.07 [14] software was used for quality control to filter data according to the following criteria: (1) call frequency ≥ 90, (2) remove individuals with (MIND) ≤ 0.05, (3) SNPs with missingness (GENO) ≤ 0.05, (4) minor allele frequency (MAF) ≥ 0.05 and (5) Hardy–Weinberg equilibrium (HWE > 0.00001), and (6) samples that did not satisfy these criteria were excluded.
2.2. Estimation of Runs of Homozygosity (ROH)
Runs of homozygosity were estimated using PLINK v1.07 [14] following the criteria: (1) the minimum length was 1000 kb; (2) the proportion of homozygous overlap window was 0.05; (3) the minimum number of SNPs included in a ROH was 100; (4) the minimum SNP density was set to 50 kb/SNP; (5) the maximum gap between continuous homozygous SNPs was 1000 kb; and (6) a maximum of one SNPs with missing genotypes and up to one heterozygous genotype were allowed in a ROH. RStudio software was used, package “detectRUNS” version 0.9.6 was used to summarize ROH into five classes: (0–5 Mb), (5–10 Mb), (10–20 Mb), (20–40 Mb), and >40 for all six breeds. Runs of homozygosity were calculated per breed and ROH coverage in each chromosome was estimated as the sum of the total length of the chromosome covered by ROH of all individuals in a population. Furthermore, the percentage of SNPs present in ROH were calculated based on the frequency of a SNP in a ROH across individuals.
2.3. QTL and Genomic Regions in ROH
PLINK v1.07 [14] (Cambridge, MA, USA) was used to estimate the consensus regions across individuals, which represent ROH pools of overlapping and potentially matching segments. The software Bovine Quantitative Trait Locus (QTL) Analysis (QTL) [15] https://www.animalgenome.org.cgi-bin/qtldb/btsearch (accessed on 28 January 2022) [16] was used for information, gene information, and animal information. Then, National Center for Biotechnology Information (NCBI) https://www.ncbi.nlm.nih.gov (accessed on 27 March 2022) was used to confirm ID of the genes found from published QTL/association and functions. We then used KEGG [17] https://www.genome.jp/kegg-bin/showorganism?org=bta (accessed on 28 January March 2022) to identify the position and length of the genes.
2.4. Classification of Runs and Inbreeding Co-Efficient
Measuring homozygosity per individual was calculated following the method of [18].
LROH is the total length of ROH and LAUTO is the length of the autosomal genome [19]. The ROH length categories were as follows: >0 Mb, >5 Mb, >10 Mb, >20 Mb, and >40 Mb. Furthermore, inbreeding based on homozygous SNPs was determined using PLINK v1.07 software [14]. The inbreeding coefficient for an individual (FHOM) was computed. Correlations of the inbreeding coefficient for two methods were estimated using the Pearson correlation from Minitab ™ version 17.1.0 [20] (State College, PA, USA).
3. Results
3.1. Distribution of Runs of Homozygosity (ROH)
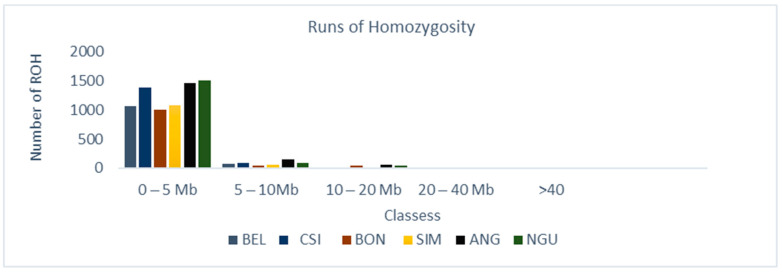
In subsequent quality control steps, 117,042 SNPs and 144 individuals were retained in a genome 2.6 Gb. Approximately 7516 ROH were observed between classes 0–5 Mb, with the most ROH observed in NGU (1507), ANG (1456), CSI (1388), SIM (1083), BEL (1067), and BON (1006), shown on (Figure 1), respectively. In total, 514 ROH were observed between classes 5–10 Mb with ANG (149) and NGU (95) demonstrating the most ROH amongst all breeds. Fewer ROH were observed between classes 10–20 Mb, 20–40 Mb, and >40 Mb for all breeds. Distribution of ROH (Figure 2) coverage was observed on chromosomes 5, 6, 11, and 14 for CSI and BEL on chromosomes 1, 2, 3,5, 6, 7, and 14 for Chinese breeds. On behalf of South African breeds, ROH coverage (Figure 3) was observed on chromosomes 1, 2, 3, 4, 5, 6, 11, and 14 for NGU; chromosomes 1, 4, 7, 11, and 14 for ANG; chromosomes 5, 6, 11, and 14 for SIM; and chromosomes 5, 7, and 14 for BON.
Figure 1.
Classification of ROH were estimated per breed; each ROH length category and average per breed, represented from left to the right: Belgian Blue (BEL), Chinese Simmentaler (CSI), Bonsmara (BON), Nguni (NGU), South African Simmentaler (SIM), and Angus (ANG). All Breeds had the highest ROH between 0–5 Mb with the NGU and AGN showing the highest ROH 0–5 Mb on chromosomes 5, 6, 11, and 14 for SIM, and chromosomes 5, 7, and 14 for BON, respectively, in South African breeds.
Figure 2.
Number of ROH per chromosomes for Chinese CSI and BEL with significant ROH observed on chromosomes 5, 6, 11, and 14 for both breeds.
Figure 3.
Number runs of homozygosity per chromosomes for South Africa BON, NGU, SIM, and ANG with significant ROH observed on chromosomes 1, 2, 3, 4, 5, 6, 7, 11, and 14, with NGU and ANG showing the most significant chromosome coverage.
3.2. The proportion of SNPs in ROH
A Manhattan plot was used to compute the number of significant SNP in a ROH (Figure 4). The results revealed SNP in ROH across all individual breeds (Figure 4A) with BEL showing significant SNP in ROH on BTA 3, 4, and 14. (Figure 4B), CSI on BTA 6 and 14, (Figure 4C); for BON on BTA 6 and 14, (Figure 4F) ANG on BTA 1, 3, 5, 7, 13, 14, and 20, and (Figure 4E) for SIM, significant SNP were detected on BTA 6 and 14, (Figure 4D), with no significant SNP for NGU breeds. The results further highlighted most significant SNP were higher in all breeds on BTA 14, excluding NGU and SIM. The South African ANG had the most significant SNP in ROH based on BTA coverage. Further results supporting Manhattan plot are shown on (Table 1).
Figure 4.
Manhattan plot for SNPs in ROH from the top to bottom (A) BEL, (B) CSI, and (C) BON, with BTA 14 having the most significant SNP in ROH for the three breeds. Manhattan plot for SNPs in ROH from the top to bottom (D) NGU, (E) ANG, and (F) SIM, with ANG having the most significant SNP in ROH on BTA 1,3, 5,7,13,14, and 20 compared to SIM and breeds.3.3. QTL and Identification of Candidate Genes within Runs of Homozygosity.
Table 1.
SNP position, length, chromosome, and number of SNPs ROH.
| Breeds | Start SNP | End SNP | Start Position | End Position | BTA | nSNP |
|---|---|---|---|---|---|---|
| SIM | BovineHD0600010649 | BovineHD0600010935 | 38474338 | 39921321 | 6 | 258 |
| SIM | BovineHD0600010973 | ARS-BFGL-NGS-99026 | 40064733 | 72930338 | 6 | 1491 |
| CSI | BovineHD1400006792 | BovineHD1400006937 | 23394002 | 23917569 | 14 | 60 |
| CSI | BTB-01143619 | BovineHD1400007578 | 26196375 | 26302589 | 14 | 21 |
| CSI | BovineHD4100004545 | BovineHD0600010837 | 38290032 | 39418286 | 6 | 183 |
| BON | BovineHD1400006736 | BovineHD1400006801 | 23240328 | 23407192 | 14 | 55 |
| BON | BovineHD1400007096 | BovineHD1400007272 | 24448641 | 25069487 | 14 | 107 |
| BON | ARS-BFGL-NGS 100816 | BovineHD0500032567 | 111909943 | 112748475 | 5 | 39 |
| BEL | BovineHD1400001029 | BovineHD0300021345 | 7162753 | 73321455 | 3 | 43 |
| BEL | BTA-107777 | BovineHD0300022023 | 73921609 | 75752848 | 3 | 77 |
| BEL | BovineHD0500024941 | BovineHD0500026993 | 87870382 | 95025828 | 5 | 295 |
| BEL | BovineHD1400006790 | BovineHD1400007272 | 233392546 | 25069487 | 14 | 265 |
| BEL | BovineHD1400007377 | BovineHD1400007531 | 25607730 | 26108646 | 14 | 117 |
| BEL | BovineHD0400019201 | BovineHD0400019430 | 6990155 | 70569432 | 4 | 17 |
| ANG | BovineHD0100044669 | Hapmap23088-BTA-151194 | 153400885 | 154349918 | 1 | 47 |
| ANG | BovineHD1300018328 | BovineHD1300018406 | 64228423 | 64621429 | 13 | 10 |
| ANG | BovineHD1400006790 | BovineHD1400006916 | 23392546 | 23831754 | 14 | 31 |
| ANG | BovineHD1400007051 | BovineHD1400007272 | 24315353 | 25069487 | 14 | 55 |
| ANG | BovineHD1400007366 | BovineHD1400007408 | 25480962 | 25583674 | 14 | 31 |
| ANG | BovineHD1400007518 | BovineHD1400007583 | 26051609 | 26938603 | 14 | 49 |
| ANG | BovineHD1400007694 | BovineHD1400024442 | 26700286 | 26938603 | 14 | 52 |
| ANG | BovineHD2000001713 | BovineHD2000001840 | 5497761 | 5839847 | 20 | 81 |
| ANG | BovineHD0300015826 | BovineHD0300015865 | 52418548 | 52539507 | 3 | 32 |
| ANG | BovineHD0500030711 | BovineHD0500030764 | 106905471 | 106988256 | 5 | 47 |
| ANG | BovineHD0700027123 | BovineHD0700027253 | 92797461 | 93307177 | 7 | 53 |
Runs of homozygosity length segments were used to detect QTL for semen traits using cattle QTL online database. QTL were identified for sperm motility, sperm concentration, semen volume, sperm count, sperm head abnormalities, sperm tail abnormalities, sperm integrity, and percentage of abnormal sperm, with the majority QTL suggested for sperm motility accounting for 80% of the results (Table 2). On BTA 6, the study revealed suggestive QTL for sperm motility, sperm head abnormalities, and sperm tail abnormalities located on the WDR 19 gene. MARCH1 gene also shared an association on BTA 6 to suggestive QTL for sperm count, semen volume, and sperm motility, with the majority of QTL on BTA 6 reported for Chinese CSI and South African SIM breeds covering 68 Mb of the genome position. Secondly on BTA 1 suggestive QTL for sperm concentration revealed an association to CFAP9 gene and QTL for sperm motility and sperm count revealed an association to DOP1B gene, with suggestive QTL for sperm motility revealing an associated with ADAMTS1 and CRYZL1 gene. Other suggestive QTL for ANG breed were observed on BTA 7 for sperm acrosome integrity rate, sperm motility and sperm count which revealed an association to GDF9 gene. However, some of the suggestive QTL for percentage of normal sperm on BTA 13 and sperm motility on BTA 3 showed no association to any gene for ANG breeds.
Table 2.
Breeds, QTL, BTA, and gene of association.
| Breed | QTL TRAIT | BTA | Gene |
|---|---|---|---|
| SIM | Sperm motility, Sperm head abnormalities (SPHAB) and Sperm tail abnormalities | 6 | WD repeat domain 19 (WDR 19) |
| Semen volume, sperm motility and sperm count | 6 | MARCHF1 membrane associated ring-CH-type finger 1 (MARCH1) | |
| CSI | Sperm motility, Sperm head abnormalities (SPHAB) and Sperm tail abnormalities (SPTAB) | 6 | WD repeat domain 19 (WDR 19) |
| BON | Sperm motility (SPMOT) | 5 | WD repeat domain 19 (WDR 19) |
| Sperm motility (SPMOT) | 5 | Solute carrier organic anion transporter family member 1C1 (SLCOICI) | |
| BEL | Sperm motility (SPMOT) | 5 | Solute carrier organic anion transporter family member 1C1 (SLCO1C1) WD repeat domain 19 (WDR 19) |
| Sperm count and sperm motility | 4 | suppression of tumorigenicity 7 (ST7) inhibin subunit beta A (INHBA) |
|
| ANG | Sperm motility and sperm count | 1 | DOP1 leucine zipper like protein B (DOP1B) |
| Sperm concentration | 1 | 1 cilia and flagella associated protein 91 CFAP9 | |
| Sperm motility | 1 | crystallin zeta like 1 (CRYZL1) ADAM metallopeptidase with thrombospondin type 1 motif 1 (ADAMTS1) |
|
| Percentage normal sperm | 13 | - | |
| Sperm motility | 3 | - | |
| Sperm acrosome integrity rate, sperm motility and sperm count. | 7 | growth differentiation factor 9 GDF9 |
The South African BON on BTA 5 revealed QTLs for sperm motility and it’s associated with SLCO1C1 gene and BEL on BTA 5 suggested QTL for sperm motility and revealed an association with SLCO1C1 and WDR 19 genes, also INHBA and ST7 genes on BTA 4 found an associated to sperm count. The results reported 13 genomic regions had identified QTL and their association; additionally, BTA 14 had the greatest coverage of SNP in ROH in CSI, BEL, BON, and ANG, but there were no detection of QTL or association on those regions.
3.3. Inbreeding Coefficient FROH vs. FHOM and ROH Regions
Lengths of ROH were further used to estimate inbreeding coefficient (FROH), shown on (Figure 5); inbreeding coefficient (FROH) was highest in NGU (0.16), followed by ANG (0.14), BEL (0.13), CSI (0.12), BON (0.11), and, finally, SIM (0.08). The Pearson correlation method was used to estimate the linear correlation coefficient between five classes and at a genomic level shown on (Table 3). Correlation estimated for FROH vs FHOM was 0.603, showing a significant moderate correlation. However, the highest correlation was observed between Froh _5 vs. Froh _0 at (0.92), and the lowest correlation was observed between Froh _40 vs. Froh _5 (0.40). This shows that the most correlation was observed the first classes (0–5 Mb).
Figure 5.
Inbreeding coefficient at a genomic level for all six breeds.
Table 3.
Correlations runs of homozygosity (ROH) between class category (0, 5, 10, 20, and >40 Mb), inbreeding coefficients in six different bulls.
| Classes | Froh_Class_0 | Froh_Class_5 | Froh_Class_10 | Froh_Class_20 | Froh |
|---|---|---|---|---|---|
| Froh_Class_5 | 0.929 | ||||
| Froh_Class_10 | 0.711 | 0.864 | |||
| Froh_Class_20 | 0.567 | 0.743 | 0.881 | ||
| Froh_Class_40 | 0.400 | 0.527 | 0.599 | 0.665 | |
| Fhom | 0.603 |
4. Discussion
Inbreeding is known to negatively affects the reproductive performance of male animals [21], and when expressed at high levels can cause poor semen quality [22,23,24,25]. However, not all inbreeding reported is harmful; the authors of [26] reported that recent inbreeding is more harmful than ancient inbreeding due to selection decreasing the frequency of deleterious alleles over time. Many studies have explored the genome wide distribution of ROH and inbreeding depression in cattle populations [9,27,28] using high density Illumina BovineHD BeadChip microarrays. This includes studies such as [29], whose findings showed that ROH are frequent across all breeds (Angus, Belgian Blue, Charolais, Friesian, Hereford, Holstein, Holstein-Friesian, Limousin, and Simmental); however, difference in patterns of ROH and variation is based on breed origin and recent management. The authors of [10] reported the distinct differences in length, quantity, and frequency of ROH between breeds and levels of genomic inbreeding. The study highlighted higher levels of inbreeding observed in commercial breeds, especially beef breeds and identified a number of genes confirmed to influence production traits [10]. Homozygous regions of the genome [8] have been reported in different species, not only in cattle [19,30], but these regions have been used to quantify individual inbreeding in humans [18], goats [31], buffalo [32], sheep [33], and pigs [12]; based on results of ROH in these studies, it appears to be more accurate than traditional pedigree-based estimates [13]. In this study, more than 1000 Mb ROH were estimated in six selected Chinese and South African beef cattle breeds at a genomic length of 2.6 Gb, with the highest ROH observed in the NGU, ANG, BEL, CSI, SIM, and BON breeds. Furthermore, the highest ROH were observed between classes 0–5 Mb compared to the rest of the classes (5–10 Mb, 10–20 Mb and 20–40 Mb) with no ROH observed in >40 class. Similar results were reported by [29], who compared European cattle breeds to the European bison, with average ROH observed for Angus and Hereford, and the most ROH were observed for the European bison breeds between classes 1–5 Mb, suggesting limited recent inbreeding for Angus and Hereford, but reporting high levels of inbreeding for European bison. These results confirm earlier studies that reported population history involving a severe bottleneck [29]. Runs of homozygosity can be affected by demographic events [34], e.g., age, geography, breed history and origin, area of distribution, climate, introduction of breeds, environment, production system, etc.; the fact that breeds from this study are distributed in two geographically different environments might explain the inbreeding. The authors of [35] reported that inbred individuals are particularly sensitive to environment changes this can explaining most of the breeds in the study are inbred due to moving from one captive environment to another captive environment. Analysis at a genomic level revealed significant distribution of ROH within the selected six Chinese and South African beef cattle breeds. Chinese CSI and BEL had significant ROH observed on chromosomes 5, 6, 11, and 14 for both breeds. South Africa BON showed the highest ROH on chromosomes 5, 7, and 14, and SIM displayed the most ROH on chromosomes 5, 6, and 14. The highest genome coverage in ROH observed for ANG on chromosomes 1–6, 11, and 14, and NGU on chromosomes 1–3, 5–7, 11, and 14. The study further used length and number of ROH to identify SNPs in those ROH. The authors of [30] reported that greatest number of ROH per chromosomes was observed for chromosome 1 across all 867 animals, and ROH per chromosome tended to decrease with chromosome length. By identifying the number of SNPs in ROH, the study identified overlapping ROH that were above the threshold of 0.05. Chromosome 14 had the most SNPs above the threshold; still, it did not report any QTL or association to any candidate gene in all breeds. Additionally, no SNP in ROH were identified for NGU; possibly markers were standardized for commercial breeds, e.g., (Simentaler) however Nguni is an indigenous Southern African breed. In total, we reported 13 genomic regions that were associated to nine candidate genes (CDF9, MARCH1, WDR19, SLOICI, ST7, DOP1B, CFAF9, INHBA, and ADAMTS1). The authors of [36] reported that genomic regions located on BTA 7, BTA 14, BTA 16, and BTA 18 were characterized by a high frequency of ROH occurrence and included important genes related to immune traits, muscularity, and ease of calving. The authors of [10] reported a similar region, BTA 6, was identified for Polish Red, Limousin, and Simmental breeds. MARCH1 [19] revealed an association with QTL for sperm motility and semen volume on BTA 6 in SIM. The MARCH 1 gene was also reported by [37] to be significantly associated with semen production traits (semen volume per ejaculate, number of sperm per ejaculate, and number of motile sperm per ejaculate). Another gene associated with QTL for sperm motility revealed on BTA 5 is SLCO1C1 [17] for BON and BEL. BTA 7 revealed the growth differentiation GDF9 gene was associated with QTL for sperm motility, sperm count, and sperm integrity. This was also reported by [38] who revealed significant association of GDF9 with sperm quality traits in Holstein bulls. The results also stated that GDF9 is involved in the initiation or maintenance of spermatogenesis; however, further verification is needed. WD repeat domain 19 (WDR 19) gene was detected on BTA 5 and BTA 6 for CSI showed an association to QTL sperm motility, sperm head abnormalities, and sperm tail abnormalities; this gene was reported to be associated to ejaculate volume, sperm concentration, sperm motility, and sperm head and tail anomalies [39]. On BTA 4, ST7 gene was significant to QTLs for sperm count and sperm motility, this gene was also reported by [40] as the nearest genes for poor sperm motility. QTL associated with semen traits were identified and validated by previously published literature. This study highlighted genes within QTL regions of semen for beef bulls. Identification of the QTL regions associated with these traits provides the knowledge necessary to enrich these regions [35]. The authors of [41] reported on three QTL found to be related with abnormal sperm frequencies at a significant p < 0.01. The authors of [40] reported several candidate genes associated with sperm concentration, sperm motility and sperm volume in Holstein-Friesian populations.
The study further showed high to moderate correlation between classes Froh_0 −5 Mb and correlation at Froh_ > 40. Moderate correlation was observed between Froh vs Fhom selected breeds. Several authors reported on strong correction between inbreeding coefficient [9]. It should be underlined that the occurrence of ROH in an individual may be the result of inbreeding events, but they may also be present in outbreed populations as result of other phenomena.
5. Conclusions
The study identified distribution of ROH in the six selected Chinese and south African beef cattle breeds (CSI, BEL, ANG, NGU, BON, and SIM) using Bovine HD Bead Chip makers. The study also showed that ANG, NGU, and CSI showed the highest frequency and length of long ROH (0–5 Mb), indicating higher recent inbreeding in all breeds, including SIM. Several QTL and genes were related to semen traits (CDF9, MARCH1, WDR19, SLOICI, ST7, DOP1B, CFAF9, INHBA, and ADAMTS1). These genes can be used as target genes for future marker-assisted selection.
Acknowledgments
I would like to acknowledge all authors for their contribution and the Department of Agriculture, Land Reform and Rural Development through the Directorate: Sector Education and Training (International Training Programme).
Author Contributions
Writing of original draft, editing, and conceptualization, M.C.M.; conceptualization, K.H.; conceptualization and supervision, K.A.N.; supervision, J.W.; supervision and editing, N.H.; supervision W.L.; editing and conceptualization, B.M. All the authors have contributed and worked together on the manuscript. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was approved by the Animal Research Ethics Committee of the Agricultural Research Council: Animal Production (Ref no: APIEC17/17).
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
Funding is supported by the National Key Research and Development Program of China (Grant Number: 2018YFE0122000) and the National Research Foundation of South Africa (Grant No: 120370).
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Scholtz M.M., Bester J., Mamabolo J.M., Ramsay K.A. Results of the national cattle survey undertaken in South Africa, with emphasis on beef. Appl. Anim. Husb. Rural. Dev. 2008;1:1–9. [Google Scholar]
- 2.Mokolobate M.C. Ph.D. Thesis. University of the Free State; Bloemfontein, South Africa: Apr 28, 2015. Novelty Traits to Improve Cow-Calf Efficiency in Climate Smart Beef Production Systems. [Google Scholar]
- 3.Kolkman I. Ph.D. Thesis. Ghent University. Faculty of Veterinary Medicine; Ghent, Belgium: Jun 6, 2022. Calving problems and calving ability in the phenotypically double muscled Belgian Blue breed. [Google Scholar]
- 4.Peñagaricano F., Weigel K.A., Khatib H. Genome-wide association study identifies candidate markers for bull fertility in Holstein dairy cattle. Anim. Genet. 2012;43:65–71. doi: 10.1111/j.1365-2052.2012.02350.x. [DOI] [PubMed] [Google Scholar]
- 5.Zhao G., Liu Y., Niu Q., Zheng X., Zhang T., Wang Z., Xu L., Zhu B., Gao X., Zhang L., et al. Runs of homozygosity analysis reveals consensus homozygous regions affecting production traits in Chinese Simmental beef cattle. BMC Genom. 2021;22:678. doi: 10.1186/s12864-021-07992-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Peripolli E., Stafuzza N.B., Munari D.P., Lima A.L.F., Irgang R., Machado M.A., Panetto J.C.D.C., Ventura R.V., Baldi F., da Silva M.V.G.B. Assessment of runs of homozygosity islands and estimates of genomic inbreeding in Gyr (Bos indicus) dairy cattle. BMC Genom. 2018;19:34. doi: 10.1186/s12864-017-4365-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kirin M., McQuillan R., Franklin C.S., Campbell H., McKeigue P.M., Wilson J.F. Genomic runs of homozygosity record population history and consanguinity. PLoS ONE. 2010;5:e13996. doi: 10.1371/journal.pone.0013996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gibson J., Morton N.E., Collins A. Extended tracts of homozygosity in outbred human populations. Hum. Mol. Genet. 2006;15:789–795. doi: 10.1093/hmg/ddi493. [DOI] [PubMed] [Google Scholar]
- 9.Broman K.W., Weber J.L. Long homozygous chromosomal segments in reference families from the centre d’Etude du polymorphisme humain. Am. J. Hum. Genet. 1999;65:1493–1500. doi: 10.1086/302661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Szmatoła T., Gurgul A., Jasielczuk I., Ząbek T., Ropka-Molik K., Litwińczuk Z., Bugno-Poniewierska M. A comprehensive analysis of runs of homozygosity of eleven cattle breeds representing different production types. Animals. 2019;9:1024. doi: 10.3390/ani9121024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nani J.P., Peñagaricano F. Whole-genome homozygosity mapping reveals candidate regions affecting bull fertility in US Holstein cattle. BMC Genom. 2020;21:338. doi: 10.1186/s12864-020-6758-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bosse M., Megens H.J., Madsen O., Paudel Y., Frantz L.A., Schook L.B., Groenen M.A. Regions of homozygosity in the porcine genome: Consequence of demography and the recombination landscape. PLoS Genet. 2012;8:e1003100. doi: 10.1371/journal.pgen.1003100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Curik I., Ferenčaković M., Sölkner J. Inbreeding and runs of homozygosity: A possible solution to an old problem. Livest. Sci. 2014;166:26–34. doi: 10.1016/j.livsci.2014.05.034. [DOI] [Google Scholar]
- 14.Purcell S. PLINK (1.07). Documentation. [(accessed on 6 June 2022)]. pp. 1–293. Available online: http://zzz.bwh.harvard.edu/plink/dist/plink-doc-1.07.pdf.
- 15.Childers C.P., Reese J.T., Sundaram J.P., Vile D.C., Dickens C.M., Childs K.L., Salih H., Bennett A.K., Hagen D.E., Adelson D.L., et al. Bovine Genome Database: Integrated tools for genome annotation and discovery. Nucleic Acids Res. 2011;39((Suppl. S1)):D830–D834. doi: 10.1093/nar/gkq1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hu Z.L., Park C.A., Wu X.L., Reecy J.M. Animal QTLdb: An improved database tool for livestock animal QTL/association data dissemination in the post-genome era. Nucleic Acids Res. 2013;41:D871–D879. doi: 10.1093/nar/gks1150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kanehisa M., Goto S. Comprehensive gene and pathway analysis of cervical cancer progression. Nucleic Acids Res. 2000;28:27–30. doi: 10.1093/nar/28.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McQuillan R., Leutenegger A.L., Abdel-Rahman R., Franklin C.S., Pericic M., Barac-Lauc L., Smolej-Narancic N., Janicijevic B., Polasek O., Tenesa A., et al. Runs of homozygosity in European populations. Am. J. Hum. Genet. 2008;83:359–372. doi: 10.1016/j.ajhg.2008.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ferencakovic M., Hamzic E., Gredler B., Curik I., Sölkner J. Runs of homozygosity reveal genome-wide autozygosity in the Austrian Fleckvieh cattle. Agric. Conspec. Sci. 2011;76:325–329. [Google Scholar]
- 20.Alin A. Minitab. Wiley Interdiscip. Rev. Comput. Stat. 2010;2:723–727. doi: 10.1002/wics.113. [DOI] [Google Scholar]
- 21.Ferenčaković M., Sölkner J., Kapš M., Curik I. Genome-wide mapping and estimation of inbreeding depression of semen quality traits in a cattle population. J. Dairy Sci. 2017;100:4721–4730. doi: 10.3168/jds.2016-12164. [DOI] [PubMed] [Google Scholar]
- 22.Margulis S.W., Walsh A. The effects of inbreeding on testicular sperm concentration in Peromyscus polionotus. Reprod. Fertil. Dev. 2002;14:63–67. doi: 10.1071/RD01120. [DOI] [PubMed] [Google Scholar]
- 23.Wildt D.E., Baas E.J., Chakraborty P.K., Wolfle T.L., Stewart A.P. Influence of inbreeding on reproductive performance, ejaculate quality and testicular volume in the dog. Theriogenology. 1982;17:445–452. doi: 10.1016/0093-691X(82)90026-7. [DOI] [PubMed] [Google Scholar]
- 24.Aurich C., Achmann R., Aurich J.E. Semen parameters and level of microsatellite heterozygosity in Noriker draught horse stallions. Theriogenology. 2003;60:371–378. doi: 10.1016/S0093-691X(03)00005-0. [DOI] [PubMed] [Google Scholar]
- 25.Vickers N.J. Animal communication: When i’m calling you, will you answer too? Curr. Biol. 2017;27:R713–R715. doi: 10.1016/j.cub.2017.05.064. [DOI] [PubMed] [Google Scholar]
- 26.Doekes H.P., Veerkamp R.F., Bijma P., de Jong G., Hiemstra S.J., Windig J.J. Inbreeding depression due to recent and ancient inbreeding in Dutch Holstein–Friesian dairy cattle. Genet. Sel. Evol. 2019;51:54. doi: 10.1186/s12711-019-0497-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhao G., Zhang T., Liu Y., Wang Z., Xu L., Zhu B., Gao X., Zhang L., Gao H., Liu G.E., et al. Genome-wide assessment of runs of homozygosity in Chinese wagyu beef cattle. Animals. 2020;10:1425. doi: 10.3390/ani10081425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ferenčaković M. Ph.D. Thesis. University of Zagreb; Faculty of Agriculture, Zagreb, Croatia: Apr 28, 2015. Molecular Dissection of Inbreeding Depression for Semen Quality Traits in Cattle. [Google Scholar]
- 29.Iacolina L., Stronen A.V., Pertoldi C., Tokarska M., Nørgaard L.S., Muñoz J., Kjærsgaard A., Ruiz-Gonzalez A., Kamiński S., Purfield D.C. Novel graphical analyses of runs of homozygosity among species and livestock breeds. Int. J. Genom. 2016;2016:2152847. doi: 10.1155/2016/2152847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Purfield D.C., Berry D.P., McParland S., Bradley D.G. Runs of homozygosity and population history in cattle. BMC Genet. 2012;13:70. doi: 10.1186/1471-2156-13-70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mastrangelo S., Di Gerlando R., Sardina M.T., Sutera A.M., Moscarelli A., Tolone M., Cortellari M., Marletta D., Crepaldi P., Portolano B. Genome-wide patterns of homozygosity reveal the conservation status in five italian goat populations. Animals. 2021;11:1510. doi: 10.3390/ani11061510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Macciotta N.P., Colli L., Cesarani A., Ajmone-Marsan P., Low W.Y., Tearle R., Williams J.L. The distribution of runs of homozygosity in the genome of river and swamp buffaloes reveals a history of adaptation, migration and crossbred events. Genet. Sel. Evol. 2021;53:20. doi: 10.1186/s12711-021-00616-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mastrangelo S., Tolone M., Sardina M.T., Sottile G., Sutera A.M., Di Gerlando R., Portolano B. Genome-wide scan for runs of homozygosity identifies potential candidate genes associated with local adaptation in Valle del Belice sheep. Genet. Sel. Evol. 2017;49:84. doi: 10.1186/s12711-017-0360-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tokarska M., Pertoldi C., Kowalczyk R., Perzanowski K. Genetic status of the European bison Bison bonasus after extinction in the wild and subsequent recovery. Mammal. Rev. 2011;41:151–162. doi: 10.1111/j.1365-2907.2010.00178.x. [DOI] [Google Scholar]
- 35.Leroy G. Inbreeding depression in livestock species: Review and meta-analysis. Anim. Genet. 2014;45:618–628. doi: 10.1111/age.12178. [DOI] [PubMed] [Google Scholar]
- 36.Purfield D.C., McParland S., Wall E., Berry D.P. The distribution of runs of homozygosity and selection signatures in six commercial meat sheep breeds. PLoS ONE. 2017;12:e0176780. doi: 10.1371/journal.pone.0176780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Liu S., Yin H., Li C., Qin C., Cai W., Cao M., Zhang S. Genetic effects of PDGFRB and MARCH1 identified in GWAS revealing strong associations with semen production traits in Chinese Holstein bulls. BMC Genet. 2017;18:63. doi: 10.1186/s12863-017-0527-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tang K.Q., Yang W.C., Zhang X.X., Yang L.G. Effects of polymorphisms in the bovine growth differentiation factor 9 gene on sperm quality in Holstein bulls. Genet. Mol. Res. 2013;12:2189–2195. doi: 10.4238/2013.July.4.1. [DOI] [PubMed] [Google Scholar]
- 39.Hiltpold M., Niu G., Kadri N.K., Crysnanto D., Fang Z.H., Spengeler M., Schmitz-Hsu F., Fuerst C., Schwarzenbacher H., Seefried F.R., et al. Activation of cryptic splicing in bovine WDR19 is associated with reduced semen quality and male fertility. PLoS Genet. 2020;16:e1008804. doi: 10.1371/journal.pgen.1008804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hering D.M., Oleński K., Ruść A., Kaminski S. Genome-wide association study for semen volume and total number of sperm in Holstein-Friesian bulls. Anim. Reprod. Sci. 2014;151:126–130. doi: 10.1016/j.anireprosci.2014.10.022. [DOI] [PubMed] [Google Scholar]
- 41.Druet T., Fritz S., Sellem E., Basso B., Gerard O., Salas-Cortes L., Humblot P., Druart X., Eggen A. Estimation of genetic parameters and genome scan for 15 semen characteristics traits of Holstein bulls. J. Anim. Breed. Genet. 2009;126:269–277. doi: 10.1111/j.1439-0388.2008.00788.x. [DOI] [PubMed] [Google Scholar]