Abstract
Polycystic ovary syndrome (PCOS) is an endocrine systemic disorder mainly characterized by a hormonal and metabolic disbalance that leads to oligo/anovulation, hyperandrogenism and the formation of ovarian cysts. Despite the progress that has been reached in its diagnosis and management, little is known about the molecular mechanisms and signaling pathways underlying the pathogenic mechanisms. In this sense, recent research has suggested that the influence of multiple factors, including age, environment, lifestyle and the disease state environment can change the clinical presentation of PCOS via epigenetic modifications. Variants in the genes encoding for proteins involved in steroidogenesis and glucose homeostasis play a crucial role in the development of the disease. Other genes involved in inflammation and cell proliferation seem to undergo an epigenetic control. Moreover, lifestyle factors influence the PCOS course and prognosis, including diet and physical activity, which are fundamental in reducing oxidative stress, inflammation and in improving metabolic and hormonal parameters. In the present review, literature evidence on molecular and epigenetic mechanisms related to PCOS etiology will be discussed, with a particular attention on the positive influence of diet and physical activity as nonpharmacological ways of intervention in the management of the disease.
Keywords: polycystic ovarian syndrome, diet, physical exercise, epigenetic modifications
1. Polycystic Ovary Syndrome: Definition and Pathogenic Mechanisms
Polycystic ovary syndrome (PCOS) is an endocrine, systemic disorder that affects 5–20% of women in reproductive age and is mainly characterized by a sex hormone imbalance causing the formation of water-retained cysts in the ovarian antral follicles [1]. The hormonal set is characterized by high luteinizing hormone (LH) levels and an elevated production of androgens, with the consequent virilization of female tracts and anovulation/oligovulation [2]. This hyperandrogenism and chronic anovulation are typical of PCOS, and related PCOS phenotypes mirror a self-powering cycle that involves metabolic and endocrine disorders [3]. The excess of androgens causes hyperinsulinemia, insulin resistance and obesity, which in turn negatively affect ovarian function and the hormonal profile [4]. Abnormalities in levels of gonadotropin-releasing hormone and follicle-stimulating hormone (FSH) are also present [5].
It has to be considered that about 75% of PCOS women suffers from anovulatory infertility [6], and those who undergo in vitro fertilization (IVF) have a higher risk of developing ovarian hyperstimulation syndrome. Moreover, the same women present a significantly higher risk of miscarriage and gestational diabetes during pregnancy [7].
Currently, the most used diagnostic criteria are the Rotterdam criteria (2003). According to them, PCOS is present if two of the following three criteria are manifested: oligo/anovulation, hyperandrogenism and the presence of ovarian cysts. Following the same criteria, four phenotypes could be determined [8]:
-
-
oligo/anovulation + hyperandrogenism + polycystic ovaries;
-
-
oligo/anovulation + hyperandrogenism;
-
-
hyperandrogenism + polycystic ovaries;
-
-
oligo/anovulation + polycystic ovaries [8].
Despite these well-characterized phenotypes, the pathogenesis of PCOS remains unclear; it is a multifactorial disorder that involves genetic, epigenetic and environmental variables [9], as well as immunity disorders [10].
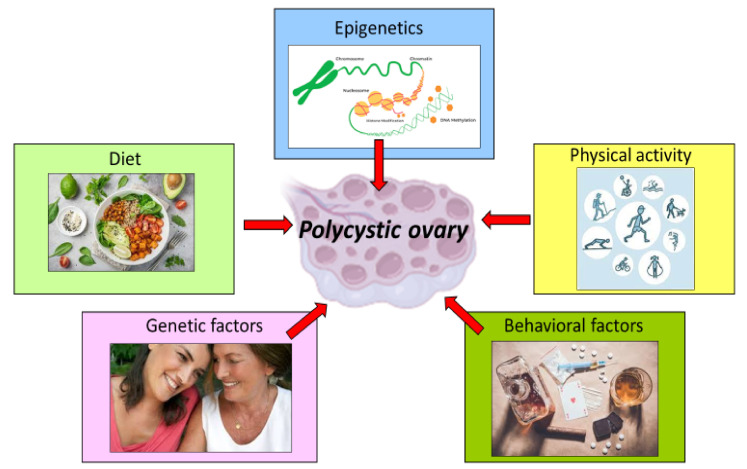
Moreover, lifestyle factors influence the PCOS course and prognosis. Despite the progress that has been reached in its diagnosis and management, little is known about the molecular mechanisms and signaling pathways underlying the pathogenic mechanisms. In this sense, recent research has suggested that the influence of multiple factors, including age, environment, lifestyle and the disease state environment, can change the clinical presentation of PCOS via epigenetic modifications. In the present review, the molecular and epigenetic mechanisms of diet and physical activity are discussed, with a particular attention on their positive influence in the management of the disease and their efficacy as nonpharmacological ways of intervention (Figure 1).
Figure 1.
Genetic, environmental and behavioral factors involved in the pathogenesis of PCOS.
2. Mechanisms Involved in PCOS Pathogenesis
2.1. Molecular Mechanisms
Dysfunctional granulosa cells (GCs), characterized by a poor apoptosis and increased proliferation, have proved to be typical of PCOS and are usually associated with impaired ovulation and folliculogenesis [11]. Elevated levels of circulating insulin could be responsible for GC abnormalities: specifically, it is able to favor the overexpression of mir-93 and the downregulation of mir-145, which in turn enhance cell growth and inhibit apoptosis [12,13].
Dysfunctional GCs also present increased mitochondrial autophagy that leads to mitochondrial injuries with consequent decreased membrane potential and mtDNA content [14]. More generally, elevated levels of androgens in PCOS women causes an overactivation of mitochondria in several tissues within the body favoring an ATP surplus [15]. An excessive amount of ATP molecules acts both on pancreatic β-cells and α-cells causing hyperinsulinemia and insulin resistance through the inhibition of the AMPK signaling pathway in insulin-sensitive tissues [16]. Insulin and insulin growth factor 1 (IGF-1) trigger some pathways in inner theca cells, directly causing its hypertrophy and the overproduction of androgens [17]. Particularly, they activate CYP17A1 and the steroidogenic factor (SF-1), favoring the adrenal steroidogenesis as well as the ovarian one [9]. In addition, hyperinsulinemia and hyperandrogenism affect pituitary LH pulsatility and worsen the release of male hormones [18].
The excess of androgens is directly related to the arrest in the antral follicle development and anovulation: in murine models, androgens have proved to enhance the activity of the C-type natriuretic peptide (CNP) and natriuretic peptide receptor 2 (CNP/NPR2) pathway that in turn stops oocyte meiosis impairing ovulation [19].
Androgens also correlate with GCs dysfunction, favoring in them the accumulation of advanced glycation end products (AGEs): male hormones, in fact, cause the overexpression of the AGE receptor in GCs through the activation of the endoplasmic reticulum stress [20]. In their turn, AGEs impair insulin signaling in GCs, affecting follicle growth [21].
Moreover, GCs undergo the epithelial mesenchymal transition (EMT) during the ovulation period, and this process is regulated by the integrin-interacting protein kindlin 2; it has been demonstrated that an excess of testosterone in PCOS women significantly increased the expression of kindlin 2, thus affecting EMT and ovulation [22]. In addition, EMT has proved to be linked to pathological processes within the endometrium of women suffering from PCOS, causing endometrial dysfunction and reproductive disorders [23].
In this kind of patient, a strict correlation between hyperandrogenism and abdominal adiposity has been highlighted as well. An in vitro study showed that in normal-weight patients, subcutaneous abdominal stem cells developed too early to adipocyte: these new adipose cells were characterized by a high content of lipids and an overexpression of PPARγ and CEBPα, which are two genes involved in adipogenesis processes [24]. In addition, these modifications were positively correlated with higher circulating levels of androgens [24].
2.2. Gene Polymorphisms
The genetic predisposition in PCOS is unquestionable and several genes are demonstrated to be involved in the etiology of the disease [25]. Indeed, due to the multifactorial nature of this disease, and to its polygenic contributions, it is not easy to offer a complete report about the genetic basis of PCOS [26]. Moreover, the different PCOS phenotypes surely complicate the genomic dissection of PCOS etiology. Most of genes involved in ovary functioning are potentially implicated in PCOS etiology, also considering the presence of an endocrine dysregulation and of inflammatory processes [26]. In this sense, the different gene types are often classified in subgroups based on their involvement in secretion and hormone functioning, as summarized in Table 1. With these premises, herein we provide the main genetic studies, also offering a connection with the pathological traits of PCOS (Table 1).
Table 1.
Pivotal genes that have been involved in PCOS etiology.
| Gene | Encoded Protein | Implication | Reference |
|---|---|---|---|
| INSR | Insuline receptor | Insulin secretion and action | [27] |
| CAPN10 | Calpain 10 protein | [28] | |
| IRS1, IRS2 | Insulin receptor substrates IRS1 and IRS2 | [29] | |
| FSHR | Follicle-stimulating hormone receptor | Gonadotropin release regulation | [30] |
| LH | Beta subunit of luteinizing hormone | [31] | |
| AMH | Anti-Müllerian hormone | [32] | |
| CYP21 | 21-hydroxylase enzyme | Synthesis pathways of steroid hormones | [33] |
| CYP11a | A cytochrome P450 enzyme |
[34] | |
| CYP19 | Cytochrome P450 aromatase | Androgen synthesis pathways | [35] |
| CYP17 | Enzyme cytochrome P450-C17 | [36,37] | |
| TNF-α | The cytokine Tumor Necrosis Factor | inflammation | [38] |
| SHBG | Sex Hormone–Binding Globulin | Regulation the androgen level in the body | [39] |
Considering that most PCOS women are at an increased risk of developing glucose intolerance and type 2 diabetes mellitus (T2DM), the gene Calpain 10 (CAPN10), located on the 2q chromosome and usually associated with insulin resistance, has been investigated [40]. In particular, UCSNP-44 polymorphism of the CAPN10 gene seems to have a remarkable role in the onset of the disease [40], while the UCSNP-43 genotype has been related to a worse metabolic profile in PCOS women [41]. Moreover, insulin resistance can be secondary to abnormalities involving insulin receptors, which are members of the tyrosine kinase family and are encoded by the INSR gene (chromosome 19p13.2) [42]. Notably, INSR rs1799817 polymorphism has been found to favor a worse glycemic pattern, as well as to predispose PCOS women to obesity [43]. In this sense, the fat mass and obesity-associated (FTO) gene plays a crucial role in developing PCOS: a recent study showed that the rs1421085, rs17817449 and rs8050136 variants of the FTO gene positively correlate with higher androgens levels and with an obese phenotype [44].
Critical genes that encode for steroidogenic enzymes, including cytochrome P450 enzymes (CYP), have also been investigated. In this regard, polymorphisms in CYP11A, CYP17, CYP19 and CYP21 genes are related to the alteration in the steroidogenesis with an overproduction of androgen hormones [45]. Recent studies have demonstrated that CYP 17 5’-UTR MspA1 (rs743572) and CYP 19 (rs2414096) polymorphisms increase the susceptibility to develop PCOS in young women, altering different hormone genesis pathways [46,47,48].
Since androgen effects are mediated by their receptors, it has been assumed that the androgen receptor (AR) gene is involved in hormonal disbalance. The AR, located on the X-chromosome at Xq11–12, presents a genetic polymorphism in exon 1, defined by a poly-glutamine (CAG) repeat region (8–35 repeats in most cases) [49]: a shorter length of this region has been typically associated with an increased androgen sensitivity; for this reason, CAG repeat polymorphism in the AR gene has been linked to PCOS amplifying women’s response to male hormones and leading to virilization and menstrual disorders [50].
Moreover, polymorphisms that occur in the sex hormone–binding globulin (SHBG) gene (chromosome 17p12—p13) seem to be crucial: it has been shown that a shorter (TAAAA) pentanucleotide repeat is responsible for an altered gene transcription [51]. As a consequence, poor circulating levels of SHBG cause obesity, impaired lipid metabolism, hyperinsulinemia, hyperandrogenism and chronic inflammation in women affected by PCOS [52].
In addition, reproductive anomalies in PCOS suggest the presence of mutations in folliculogenesis-related genes: for example, a recent study demonstrated that luteinizing hormone (LH) and luteinizing hormone receptor (LHR) genes show variants strongly related to PCOS phenotypes [53]. The FSHR (follicle-stimulating hormone receptor) gene also showed polymorphisms in PCOS women: interestingly, the two detected missense mutations, p.Ala307Thr and p.Asn680Ser, negatively affect ovarian FSH response, causing an impaired oocyte maturation, anovulation and consequent infertility [54]. A summary of the reported genetic polymorphisms associated to PCOS is reported in Table 2.
Table 2.
Genetic polymorphisms associated to PCOS.
| Gene | Polymorphisms | Effects on PCOS Phenotype |
|---|---|---|
| Calpain 10 (CAPN10) | UCSNP-44 | PCOS pathogenesis [40] |
| UCSNP-43 | Worse metabolic profile [41] | |
| Insulin receptor gene (INSR) | rs1799817 | Worse glycemic pattern and obesity [42] |
| Fat mass and obesity-associated gene (FTO) | -rs1421085 -rs17817449 -rs8050136 |
High androgen levels [44] |
| Cytochrome P450 enzymes (CYP): | ||
| -CYP 17 | -rs743572 | Steroidogenesis and alteration of hormonal pathways [46,47,48] |
| -CYP 19 | -rs2414096 | |
| Androgen receptor gene (AR) | poly-glutamine (CAG) repeat region | Amplified response to male hormones [50] |
| Sex hormone–binding globulin gene (SHBG) | short (TAAAA)n pentanucleotide repeat | Obesity, impaired lipid metabolism, hyperinsulinemia, hyperandrogenism and chronic inflammation [55] |
|
Follicle-stimulating hormone receptor
(FSHR) |
-p.Ala307Thr -p.Asn680Ser |
Impaired oocyte maturation, anovulation, infertility [54] |
Despite the difficulties to offer a complete report about the genetic basis of PCOS, common genetic risk factors can be identified and linked to metabolic and endocrine dysfunctions associated to PCOS. Next generation genetic analysis will allow incorporating huge, genotyped datasets, thereby detecting a genetic architecture across PCOS diagnostic categories.
2.3. Epigenetic Mechanisms
Epigenetic mechanisms have been recently supposed to participate in PCOS pathogenesis: in particular, a huge amount of data is demonstrating that DNA methylation and microRNAs (miRNAs) have an altered pattern in the blood, serum, adipose tissue, granulose cells and theca of women with PCOS. Thus, PCOS women can have a different epigenetic regulation that seems to trigger and support the progression of the disease [56], possibly regulated by environmental elements, including diet and/or obesity and an adverse intrauterine environment [56]. Herein, we provide a summary of the main evidence related to DNA methylation, miRNAs and chromatin remodeling, connecting these alterations to protein functioning and PCOS pathological traits.
2.3.1. DNA Methylation
DNA methylation consists in an enzymatic reaction that adds a methyl group generally at the carbon in the 5′ position of the pyrimidine ring of a cytosine followed by a guanine called CpG dinucleotides [57]. A huge amount of data reveals that women with PCOS have an altered epigenetic program in part due to this epigenetic modification. Modifications in DNA methylation have been evidenced in tissues involved in PCOS pathogenesis, including ovary, adipose tissue and skeletal muscle [58], but also in umbilical cord blood, suggesting a correlation between the PCOS phenotype and epigenetic changes in cells from systemic and fetal circulation [58].
Furthermore, recent studies have highlighted that the mechanisms of DNA methylation possess a high plasticity across age and DNA methylation, and gene expression and the corresponding phenotype can be modified through therapeutic intervention in animal models, thus opening the possibility of therapeutic interventions in PCOS women as well [58,59,60].
Table 3 summarizes the main studies investigating DNA methylation variations related in PCOS women/animals, with changes in the gene expression and correlations between DNA methylation and PCOS clinical findings.
Table 3.
Differentially methylated genes reported in tissues from patients with PCOS.
| Genes | DNA Methylation Mechanism | Tissue | Clinical Effects Related to DNA Methylation Changes |
References |
|---|---|---|---|---|
|
LY6G6F,
KCTD21, ADCY9, RABL2B, ZNF611, VASH1, FST, LMNA, PPARGC1A |
Hypermethylation | Peripheral blood |
Increased prolactin and estradiol levels in serum, increased free androgen index, insulin resistance, increased triglyceride levels in plasma and risk for metabolic syndrome |
[61,62] |
|
L-1,
TMSB15B, RPF1, DNA2, EPHA8, LHCGR EPHX1 |
Hypomethylation | |||
|
MATN4,
DLGAP2, CDH13, GAREM2, GSC, ANKRD34C, ATP8B2 PPARG |
Hypermethylation | Granulosa cells | Hyperandrogenism | [62,63,64] |
|
L-1, LHCGR,
SMG6, CCR5, LHB, NTN1, ARFGAP1, MDGA1, NCOR1, YAP1, CD9, NR4A1, EDN2, BNIP3, LIF |
Hypomethylation | |||
|
ZZEF1, TPT1,
STUB1, DMAP1, RAB5B, PPARG, SVEP1, SAV1, RORA, RAB6A CNST |
Hypermethylation | Subcutaneous adipose tissue |
Oligomenorrhea; Increased testosterone levels in circulation |
[60] |
|
PUM1,
DIP2C, SNX8, SRGAP3, ZFHX3, OR52W1and BBX |
Hypomethylation | |||
|
TET1
ROBO 1 CDKN1A HDC IGFBPL1 IRS4 |
Hypomethylation | Ovarian tissue | Pathogenesis of PCOS | [65] |
| TNIK | Hypermethylation | Granulosa cells | Altered metabolic profile | [61] |
| TNF | Hypermethylation | Granulosa cells | Inflammation and hyperandrogenism | [66] |
|
AKR1C3
CASR GHRHR RETN MAMLD1 |
Hypomethylation | Granulosa cells | Hyperandrogenism and hormonal disbalance | [66] |
A general DNA methylome profiling of GCs recently has revealed an altered methylation in genes regulating pivotal ovarian functions in PCOS [66]: in particular, few differentially methylated genes, including aldo-keto reductase family 1 member C3, calcium-sensing receptor, growth hormone-releasing hormone receptor and tumor necrosis factor, which predominantly contribute to hyperandrogenism, premature luteolysis and oocyte development defects, can be explored as novel epigenetic candidates in mediating ovarian dysfunction. Overall, this study evidenced that the epigenetic dysregulation of genes involved in important processes associated with follicular development may contribute to ovarian defects observed in women with PCOS.
Several studies investigated the genome-wide DNA methylation levels in the peripheral blood of women with PCOS (for a detailed review, see [58]). Among these findings, Shen and collaborators have shown up to 40 genes with a different pattern of methylation in PCOS samples, mainly correlating with cancer, immune response, transcription regulation and metabolism [67]. Another study has identified additional genes with altered DNA methylation that correlate with inflammation and altered metabolism [68]. Particularly, in this study DNA methylation of specific CpG sites has been shown to be related to pathological clinical parameters in PCOS women (i.e., circulating estradiol and prolactin). In parallel to investigations related to genome-wide DNA methylation, other studies have focused on the DNA methylation pattern of specific genes [62,69,70,71], as reported in Table 3. Overall, the reported changes in DNA methylation are related with the molecular pathways and physiological processes that are dysregulated in PCOS, including follicular development [72], infertility [73], steroidogenesis [74], glucose metabolism and insulin signaling [75]. Of note, the investigation of DNA methylation in blood does not automatically reproduce the status of specific organs or tissues implicated by the disease [58]. Moreover, in these studies, DNA is isolated from different cell types: as a consequence, the methylation profile can be the average of the methylation levels of each cell type [76].
The molecular and morphological changes in the ovarian tissue that are typical of PCOS can be related to epigenetic alterations, as demonstrated in animal models of PCOS.
For example, the fetal programming induced by prenatal testosterone has been proven to be related with defects in the expression of ovarian steroidogenic genes correlated with the insulin pathway in the ovaries of fetal ewes [77]. Similarly, DHEA administration in prepubertal female mice resulted in a decrease of DNA methylation in oocytes [78]. DNA methylation changes in single oocytes has been related with transcriptional regulation and cell division [79]. Using dihydrotestosterone (DHT)-induced prenatally androgenized (PNA) mice as a model of PCOS, 857 differentially methylated genes and 3317 differentially expressed ones have been found compared to control mice [63]. In murine models, DNA hypomethylation regulates the expression of several key genes for developing PCOS; the same genes, involved in DNA demethylation (TET1), axon guidance (ROBO-1), inhibition of cell proliferation (CDKN1A), inflammation (HDC) and insulin signaling (IGFBPL1, IRS4), are found to be hypomethylated in PCOS women as well, and it has been suggested that this epigenetic process could be inheritable, since three of them (ROBO-1, HDC, IGFBPL1) resulted in being hypomethylated in daughters suffering from PCOS [65].
A few studies have stated significant differences in the methylated genes of the ovarian tissue between PCOS women and controls, which correlate with hormone activity (such as AMH), transcriptional regulation, inflammation, glucose metabolism and insulin signaling [71]. Furthermore, alterations in DNA methylation in PCOS women have been proven for different genes associated with the ovary function, and these modifications are related to a dysregulated response to gonadotropins, insulin signaling and steroidogenesis [64,71,80,81]. Similarly, significantly lower levels of DNA methylation in the LHCGR gene promoter with an increase in the gene expression have been reported in granulosa cells in PCOS women [71].
The epigenetic regulation affects also the Traf2- and Nck-interacting kinase (TNIK) usually involved in cell proliferation: the hypermethylation of the corresponding gene upregulates TNIK in PCOS ovarian tissue; moreover, the hyperexpression of TNIK seems to be related to Wnt signaling pathways, finally altering women metabolic profile [61].
In addition, epigenetic mechanisms correlate with the androgen excess. The hypermethylation of tumor necrosis factor (TNF) and the hypomethylation of aldo-keto reductase 1 C3 (AKR1C3), calcium-sensing receptor (CASR), growth hormone-releasing hormone receptor (GHRHR), resistin (RETN) and mastermind-like domain 1 (MAMLD1) lead to hyperandrogenism, causing the hormone disbalance that is crucial for the pathogenesis of PCOS [66]. Interestingly, the hyperandrogenic profile differs between PCOS obese and nonobese women, and the discrepancy occurs under the influence of an epigenetic control: in particular, the gene that encode for luteinizing hormone receptor (LHR) is overexpressed in PCOS nonobese women; in contrast, the insulin receptor gene (INSR) in under expressed in PCOS obese subjects [82]. Hyperandrogenism in PCOS women can be attributed to a decrease in the DNA methylation of the NCOR1 promoter (a nuclear corepressor of PPARG pivotal in reproductive functions and hormonal signaling) and an increase in the PPARGpromoter in granulosa cells (encoding the PPARG that regulates ovarian function) [64].
Moreover, considering that ovarian autophagy is critical for follicular development [83], the genes involved in these mechanisms were investigated. In PNA mice, a global hypomethylation is detected, resulting in an overexpression of genes involved in the mitogen-activated protein kinase (MAPK)/p53 pathway (Mapk14, Mapkapk3 and Trp53) and autophagy (Becn1) [63]. Interestingly, it has been found that the prenatal exposure to DHT (used in this model) is responsible for the hypomethylation, suggesting that hormonal changes influence the epigenetic control [63].
2.3.2. MicroRNA
Together with the DNA methylation, microRNAs (miRNAs), which are small noncoding RNAs, are involved in epigenetic processes regulating gene expression at the post-transcriptional level [84]. The interaction between DNA methylation and miRNAs seems to have an important role in the pathogenesis of PCOS, since the hypermethylation of some miRNA promoter regions (miR-429, miR-141-3p and miR-126-3p′) upregulate the expression of genes controlling cell proliferation, inflammation and apoptosis (XIAP, BRD3, MAPK14 and SLC7A5) [85]. Actually, up to 30 miRNAs are differentially expressed in the PCOS women compared to controls, and are implicated in controlling ovulation, follicles and hormonal levels [86].
Altered miRNA expressions are closely associated with the occurrence of diseases, including endocrine and metabolic disorders. Recently, the roles of miRNAs in PCOS pathology have attracted considerable attention [87]. For example, the expression level of three miRNAs (miR-222, miR-146a and miR-30c) has been demonstrated to be significantly increased in PCOS patients with respect to the controls [88]. In this paper, Liu and colleagues have proved that miR-222 is positively associated with serum insulin, while miR-146a is negatively associated with serum testosterone. Furthermore, bioinformatics analysis indicated that the selected miRNAs are mainly involved in the regulation of cell cycle, apoptosis and endocrine functioning and can represent noninvasive biomarkers for PCOS diagnosis [88].
In addition, different miRNAs have been linked to PCO pathogenesis and different studies have enlightened the importance of miRNAs in cumulus GCs for ovarian follicle development. For example, Cai and collaborators [12] have proved a significant downregulation of miR-145 in the isolated human GCs of PCOS women, which has been related to an increase of cell proliferation and DNA transcription by the activation of the insulin receptor substrate 1 (IRS1) and of the downstream MAPK/ERK signaling pathway [12]. The pharmacological modulation of miR-145 can thus represent a therapeutic strategy to resolve GCs dysfunction in PCOS [12]. Furthermore, human GCs have been found to present significant lower levels of miR-126-5p and miR-29a-5p, along with the overexpression of its target, the klotho protein, which has been suggested to regulate GC survival/apoptosis by modulating the insulin/IGF-1, Wnt1 and Akt signaling pathways [11,89].
Another miRNA involved in GC proliferation is miR-93, whose expression has been proven to be upregulated in PCOS ovarian samples. This miRNA seems to regulate folliculogenesis and GC proliferation via targeting CDKN1A and promoting G1/S transition [13]. An elevated insulin concentration has been supposed to trigger GC proliferation by inducing miR-93 expression and downregulating CDK1A [13].
Similarly, miR-221/miR-222 has been evidenced as downregulated in GCs of PCOS samples and to be related to GC decreased proliferation, probably as a consequence of an elevated androgen level and overexpression of p27/kip1 [90].
A few members of the miR-17-92 cluster (i.e., miR-92a and miR-92b) have been found to be downexpressed in the ovarian tissues of PCOS women [91]. These miRNAs are believed to target the insulin receptor substrate proteins 2 (IRS-2), contributing to the hyperinsulinemia status of PCOS patients [91]. Moreover, miR-92a can bind the GATA binding factor 6 (GATA6), which is involved in androgen release, thus linking the insulin and androgenic signaling pathways in PCOS pathogenesis [91].
Additionally, other miRNAs have been related to the serum concentration of key hormones or of their receptors: for example, Song and collaborators have evidenced a downregulation of serum miR-592, whose targets are represented by luteinizing hormone/chorionic gonadotropin receptor (LHCGR), which regulates follicle development, and by IGF-1 receptor (IGF1R), which has been linked to insulin resistance [92].
2.3.3. Chromatin Modifications
A few studies have studied the putative differences in the chromatin accessibility between PCOS women and controls, examining ovary samples but also samples derived from adipose tissue. For example, Hosseini and colleagues have investigated histone acetylation and methylation levels of cumulus cells (CCs) in infertile PCOS patients, and their correlation with the expression of the ovarian aromatase gene [93]. This study has evidenced a significantly higher incorporation of the histone H3K9ac mark in three analyzed promoters of CYP19A1 in PCOS than in the control group, highlighting an epigenetic contribution to aromatase gene expression in PCOS patients during controlled ovarian stimulation. In contrast, DNA methylation promoters and the histone H3K9me2 levels in promoter PII have been demonstrated to be significantly lower in the CCs of PCOS than those of the control group, suggesting a further epigenetic control of aromatase transcription during ovarian stimulation in PCOS patients [93].
A recent interesting study has evidenced that adipose stem cells (ASCs) of normal-weight PCOS women present an augmented lipid accumulation accompanied by an overexpression of the key master regulator genes of adipogenesis required for adipocyte maintenance/differentiation and specific changes in chromatin accessibility and transcriptional regulation compared to control stem cells [94]. Interestingly, there was an extensive correspondence between increased chromatin accessibility and increased RNA expression of the key genes involved in adipocyte differentiation and function, and in the triacylglycerol synthesis functional group during adipogenesis. Thus, the dynamic chromatin remodeling occurring during adipogenesis in abdominal ASCs of normal-weight PCOS women may improve adipogenic gene expression, finally promoting a greater fat storage [94].
The data presented here below allow concluding that methylation in DNA and miRNAs (and in part modifications in the chromatin status) is altered in PCOS women in the blood, serum, adipose tissue, granulose and theca cells. Thus, women with PCOS present a changed epigenetic regulation, which might be triggered by an adverse intrauterine environment or by postnatal environmental elements, including diet and obesity. These epigenetic alterations can be considered as biomarkers for the diagnosis and treatment of PCOS patients.
2.4. Immune Mechanisms
An autoimmune predisposition could be involved in PCOS pathogenesis [10]. The frequent association with thyroid autoimmune diseases suggests that the autoimmunity could be at the basis of the disease [95]. In this regard, in women suffering from both PCOS and Hashimoto’s thyroiditis, levels of anti-Mullerian hormone (AMH) are negatively correlated with antithyroid peroxidase antibody (anti-TPO) levels, suggesting a decreased ovarian reserve in patients presenting these autoantibodies [96,97].
It has been demonstrated that, in women affected by PCOS, CD4(+), CD25(+) and CD127(−/low) T regulatory cells present a decreased activity with a consequent impaired immunosuppression [98]. Moreover, nonorgan-specific autoantibodies are often found in these women: particularly, antinuclear antibodies (ANA) have proved to be higher in PCOS women and are associated to clinical signs of hyperandrogenism [99].
Antiendometrial antibodies were also found in PCOS women and they have been demonstrated to associate with the presence of oxidized proteins, suggesting a link between the pathogenesis of the disease and the oxidative stress in the uterine microenvironment [100]. Interestingly, in these patients, levels of IgM antibodies against phosphorylcholine (IgM anti-PC), which are natural antibodies protective against atherosclerosis, are found to be lower, predisposing PCOS women to cardiovascular diseases [101].
Recent studies showed the presence of activating autoantibodies (AAb) to the gonadotropin-releasing hormone receptor (GnRHR) [102,103] that seem to be associated with increased levels of testosterone and inflammatory cytokines [102]. The same antibodies, in murine models, are also responsible for increased levels of LH, TNF-α, IL-1α and IL-18, enhancing inflammation and hormonal disbalance [104].
Low levels of LH and FSRH autoantibodies were also found, but their role in enhancing hyperandrogenism in PCOS women is still doubtful [105].
3. Impact of Lifestyle and Behavior in the Management of PCOS
Improving nonpharmacological interventions is fundamental in the management of PCOS. Lifestyle changes aim to achieve better anthropometric parameters that usually reflect better metabolic and hormonal profiles affecting positively ovarian functions [106]. Obesity is considered one of the major risk factors of infertility in women suffering from PCOS [107]. In particular, the adipose tissue, considered as an endocrine organ, produces specific and nonspecific cytokines, including adiponectin (APN), resistin, visfatin, omentin, retinol binding protein-4 (RBP4), lipocalin-2 (LCN2), chemerin, interleukin 6 (IL6), interleukin 1β (IL1β) and tumor necrosis factor α (TNFα), that are able to affect negatively insulin resistance and hormonal release [108].
First of all, identifying and correcting abnormal eating disorders is a successful strategy in providing weight loss [106]. In fact, PCOS women are more susceptible to depressive disorders and altered eating behaviors [109]. Moreover, depression has proved to be a typical tract of PCOS that affects about 40% of these patients [110] and is responsible for a worse metabolic profile: depression, in fact, is considered an inflammatory disorder and the overlap of symptoms between PCOS and depression suggests that mood modifications would be desirable to achieve better PCOS phenotypes [111].
In order to improve the metabolic profile, it has to be considered that sleep duration is crucial in developing worse PCOS phenotypes: a recent study demonstrated that poor sleep is associated with sleep apnea disorders, which in turn are responsible for increased levels of triglycerides, a higher percentage of liver fat and increased waist circumferences in adolescents affected by PCOS [112]. The resulting intermittent hypoxia causes oxidative stress and mitochondrial dysfunction: free radicals are able to activate several kinases, including NK, p38 MAPK and IKK, that trigger pathways involved in the insulin sensitivity [113]. Moreover, in rodent models, the intermittent hypoxia leads to pancreatic apoptosis (via Caspase 3 pathways), causing insulin resistance [114]. In addition, hormones that regulate appetite and insulin resistance follow a circadian rhythm. The disruption of this rhythmicity alters the release of hormones like growth hormone (GH) and leptin; GH-elevated levels in wake periods impair glucose metabolism, while the lowest levels of leptin in sleep deficiency increase appetite [115]. Therefore, interventions aimed to improve sleep quality and quantity could ameliorate women’s metabolic status and contribute to weight loss in PCOS women.
In this perspective, it is clear that diet and physical activity are keystones to achieve lifestyle changes which are effective in obtaining the improvement both in the metabolic and in hormonal profile [116].
4. Dietary Modification
Quantitative and qualitative changes in dietary patterns are needed to have an impact on the management of PCOS [117]. Generally, it has been demonstrated that both ordinary hypocaloric diet and a high-protein/low-glycemic-load hypocaloric diet positively affect the insulin sensitivity and the hormonal disbalance, with a significant reduction of testosterone levels and increased estrogens ones [118]. A modest reduction in carbohydrate (CHO) intake affects β-cell responsiveness, reducing basal β-cell response, fasting insulin, fasting glucose, total testosterone and all cholesterol measures, with a clear positive effect on insulin resistance [119]. As well as improving the metabolic profile, dietary patterns characterized by a lower glycemic index positively influence ovulation, fertility and cardiovascular features in PCOS women [120]. In addition, a high fiber consumption is recommended: in fact, a poor fiber intake means a higher glycemic load that worsens the insulin resistance [121].
The effect of a ketogenic diet on PCOS outcomes has also been investigated. A ketogenic diet is usually characterized by high fat, moderate protein and very low (40–50 g/die) CHO intake [122]. Evidence suggests that this specific pattern is able to improve anthropometric and biochemical parameters (including LH, FSH, SHBG) and reduce abnormal estrogen production deriving from the aromatization of androgens that occur in the adipose tissue, with a consequent improvement of the LH/FSH ratio [122,123]. Considering that in PCOS anovulation and infertility are aggravated by a chronic inflammatory state that affects ovaries and the uterine cavity [124], the importance of a ketogenic diet lies also in its anti-inflammatory properties mediated by the ketone β-hydroxybutyrate (BHB). Indeed, in murine models, this metabolite seems able to block the activation of NLRP3 inflammasome that usually is responsible for IL-1β and IL-18 production in human cells [125].
In this regard, in PCOS women, oral supplementation with natural molecules has been surprisingly useful in reducing inflammation. It has been shown that a magnesium–zinc–calcium–vitamin D cosupplementation reduces oxidative stress and inflammation in women affected by PCOS, increasing antioxidant capacity [126]. This is reflected in a reduced plasma malondialdehyde (MDA), that is a marker of lipid peroxidation [127], and consequently a better metabolic profile [126]. Interestingly, magnesium and zinc supplementation for 12 weeks is even able to interfere with the expression of genes that encode for inflammatory cytokines (IL-1, TNF-α), besides reducing blood high-sensitivity C-reactive protein (hs-CRP), protein carbonyl and increasing antioxidant mechanisms [128]. Moreover, magnesium alone has a remarkable role in regulating glucose metabolism, since magnesium serum levels have been demonstrated to be related to an improved insulin resistance [129].
An enhanced glucose and lipid metabolism has been observed in PCOS women also during vitamin D and calcium supplementation that positively affects triglyceride, cholesterol, testosterone levels, dehydroepiandrosterone sulfate (DHEAS), SHBG and hs-CRP [130]. In addition, vitamin D has anti-inflammatory and antioxidant capacities which reflect in reduced hs-CRP and MDA levels [131]. Because of their well-known anti-inflammatory properties [132], the relation between omega-3 polyunsaturated fatty acids (n3 PUFAs) supplementation and PCOS has also been investigated. Evidence suggests that n3 PUFAs positively affects inflammation, oxidative stress and hormonal parameters, reducing hs-CRP, MDA, total testosterone and increasing antioxidant capacity [133], even though these effects seem to be limited to women over 40 years old and with cardiovascular and metabolic abnormalities [134]. Specifically, n3 PUFAs, including eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), act on inflammation, reducing TNF-α, IL-6 and IL-1β, and interfering with the nuclear factor kappa B (NF-κB) activity, reducing the activation of several proinflammatory genes [135]. Moreover, n3 PUFAs favor the β-oxidation of mitochondrial fatty acids, reducing lipid deposits and free radical genesis [135].
The inositol supplementation seems to be particularly effective in PCOS women. Inositol is a hexahydroxy cyclohexane (six-carbon ring compound) whose two isoforms, myo-inositol (MI) and D-chiro-inositol (DCI), respectively mediate insulin uptake in the ovaries and insulin-dependent androgen production [136]. In particular, starting from animal models, it has been demonstrated that the supplementation that led to 40:1 MI/DCI plasma ratio enhanced insulin sensitivity, reduced testosterone levels and was able to restore ovulation in most cases [136,137]. However, despite the effectiveness on the metabolic profile, the role of the MI: DCI (40:1) treatment is controversial; in fact, it has been suggested that this specific formulation could be a first-line treatment in normal-weight PCOS patients without insulin resistance, since a recent study demonstrated that ovulation and spontaneous pregnancies occur mostly in nonobese PCOS women [138]. Despite this, it is clear that adequate doses of inositol inside cells are fundamental for a correct function of the tissues, and detecting the exact formulation of inositol for each patients (according to their phenotypes) appears crucial for restoring ovulation and improving fertility [139].
The ovarian microenvironment is also influenced by the oral supplementation with L-carnitine; in a murine model, acetyl and propionyl L-carnitine have proved to enhance ovarian functions though the amelioration of the ovarian oxidative stress status, preventing the upregulation of SIRT1, SIRT3 and SOD2. Moreover, the same study demonstrated that L-carnitine acts against glycative molecular pathways [140]. In PCOS women, L-carnitine is also able to improve the metabolic profile, with consequent better levels of insuline, adiponectin, testosterone, LH and FSH [141].
In addition, a nonpharmacological intervention with flavonoids should be considered in women suffering from PCOS because of their large anti-inflammatory and antioxidant activities [142]. In murine models, flavonoids extracted from Nervilia Fordii are able to positively balance FSH, LH, testosterone and insulin levels, preventing JAK2/STAT3 pathway activation and upregulating IL-6 expression [143].
Finally, it must be considered that a balanced diet modulates intestinal biodiversity that, in turn, has an impact on the PCOS phenotype. The gut microbiota refers to a group of nonpathogenic microorganisms with anti-inflammatory, antioxidant, immune and metabolic properties for which a relationship with several diseases, including PCOS, has been established [144]. In murine PCOS models, gut microbiota negatively affects levels of triglycerides, cholesterol, testosterone, TNFα and lipopolysaccharides, increasing the inflammatory status [145]. In general, in women suffering from PCOS, the increased abundance of Escherichia and Shigella leads to an altered production of short-chain fatty acids, while a lower abundance of Lactobacillus and Bifidobacterium species impairs the release of anti-inflammatory metabolites and reduces the immune response [146]. In this regard, dietary patterns that include large fiber consumption seem able to improve PCOS clinical manifestations just acting on gut microbiota; into specifics, a recent study has reported that a 12-week high-fiber diet, or in combination with Acarbose, favors Lactobacillus and Bifidobacterium abundance with a positive effect on LH/FSH ratio, testosterone, homeostasis model assessment-insulin resistance (HOMA-IR), α-1-acid glycoprotein (α-AGP) and leptin [147]. In murine PCPS models, dietary α-linolenic acid, which is an n3 PUFA, has been shown to modulate gut microbiota, increasing Lactobacillus and Bifidobacterium as well as Allobaculum, Butyrivibrio, Desulfovibrio, Faecalibacterium and Parabacteroides, with a consequent reduction of plasma and ovarian inflammatory cytokines, including IL-6,IL-1β, IL-10, IL-17A, TNFα and monocyte chemoattractant protein-1 [148].
Therefore, gut microbiota plays a crucial role in PCOS pathogenesis and the use of probiotics as modulator of intestinal microorganisms should be considered as an alternative therapeutic strategy.
5. Exercise Interventions in PCOS Women
In order to obtain significant lifestyle changes, and in addition to a balanced diet, physical activity is fundamental for improving both metabolic and hormonal parameters as well as reducing risk factors for cardiovascular diseases [149]. As shown in a recent study, distinct types of training (high-intensity interval training, HIIT and continuous aerobic exercise training, CAET) were equally capable to act both on anthropometric and fertility profiles. In particular, after 6 months of exercise, women underwent a significant reduction of BMI and insulin resistance and improved their cardiorespiratory fitness (VO2 max); most importantly, some of them had documented ovulation and achieved pregnancy after the protocol [150]. Another study showed that a 3 month structured training reduced the peak oxygen consumption, body mass index and C-reactive protein, improving the cardiopulmonary function and decreasing the cardiovascular risk [151].
Besides improving BMI and cardiorespiratory functions, aerobic exercise seems able to affect catecholamine-induced lipolysis in PCOS women. A 6-week aerobic training has proved effective in increasing in vitro atrial natriuretic peptide (ANP)/isoproterenol-lipolysis, enhancing the sensitivity to ANP and isoproterenol and the postreceptor response via cAMP-dependent protein kinases [152]. This could favor weight loss, which is crucial in the management of PCOS women. A low volume HIIT is also able to modulate the expression of factors involved in fatty acid metabolism and inflammation; specifically, PCOS women seem to have higher levels of miRNA-27b (c-miR-27b), which is associated with an altered lipid metabolism and inflammatory status: a 16-week low volume HIIT has proven to be effective in reducing levels of c-miR-27b, thus improving the patient metabolic profile [153].
Moreover, an aerobic protocol seems capable to reduce inflammation markers in women affected by PCOS: especially in obese women, the inflammatory status is predominant [154] and often characterized by higher circulating leukocytes. Sixteen weeks of an aerobic training are useful to improve the insulin resistance and decrease the white blood cell (WBC) count. These changes correlated with a reduced serum leptin and the ratio of leptin to high-molecular-weight adiponectin [155]. Regarding exercise effects on hormonal outcomes, in murine models, a 15-day aerobic training has proved to be useful in decreasing inflammation and restoring the hormonal balance acting on levels of leptin, testosterone, estradiol, FSH, AMH, TNF-α, IL-6 and free fatty acids (FFA) [156]. Exercise positively affects the hormonal profile also in women: both continuous and intermittent aerobic training have been demonstrated to be useful in reducing circulating levels of testosterone as well as obesity parameters [157]. Controversial is the role of strength training in PCOS management, since it seems to increase androgen levels [158]. However, after a 4-month strength training, PCOS women had reduced levels of testosterone and a lower testosterone/androstenedione (T/A) ratio [159].
Physical activity also affects mitochondria that have been shown to be involved in PCOS pathogenesis [160]. In PCOS women, the mitochondrial DNA copy number is lower compared to women not affected by PCOS, and several mtDNA mutations occur in response to the insulin resistance and hyperandrogenism which enhance the oxidative stress, causing mtDNA abnormalities [160]. Different kinds of training protocols have demonstrated that exercise improves mitochondrial biogenesis and respiration upregulating PGC-1α expression [161] that is crucial for the transcriptional control of mitochondrial functions [162], even though further studies are needed to evaluate the effective role of exercise on mitochondria in PCOS.
Certainly, physical activity is effective in improving antioxidant defenses. In PCOS women, a 12-week aerobic training has proved to increase superoxide dismutase (SOD) and the total antioxidant capacity, and reduce MDA and AMH [163].
Finally, the role of exercise-induced irisin should be considered. Irisin is a hormone-like myokine whose production in the skeletal muscle is mediated by physical activity and seems to be crucial in improving biochemical aspects of several metabolic diseases, including PCOS [164]. Exercise is responsible for an overexpression of PGC-1α that in turn activates FNDC5 with the consequent release of irisin, which is also produced in response to ATP depletion after exercise [165]. The importance of this hormone-like myokine lies in its capacity to impact glucose homeostasis and reduce systemic inflammation [166]. Indeed, irisin is able to brown the white adipose tissue-activating UCP1 gene and increase the expression of GLUT4 in the brown adipose tissue, leading to an enhanced glucose uptake; it is also involved in lipolysis mechanisms triggering the AMP (cAMP)–protein kinase A (PKA)–perilipin–hormone-sensitive lipase (HSL) pathway [165].
Because of its effect on inflammation, metabolism, endocrine and fertility parameters, physical activity should be always considered primary in the management of PCOS (Figure 2).
Figure 2.
The influence of diet and physical activity on inflammation and ovarian health.
6. Conclusions
PCOS is a multifactorial disorder whose real pathogenesis is still unclear. Continuous interaction between genetic and environmental factors seems to influence different phenotypes of the disease through epigenetic and molecular mechanisms.
Differently from previous literature reviews in the field, the present work does not focus on a single topic, but it offers an overview on PCOS pathogenesis and nonpharmacological treatments, focusing on epigenetic mechanisms related to PCOS pathogenesis and on the reversal effects of diet and physical exercise. These approaches have a predominant role in triggering several pathways that affect PCOS clinical manifestations in a positive way. Next generation genetic analysis will allow incorporating huge, genotyped datasets, possibly generating genetic risk scores enhanced with clinical information, environment and lifestyle data for a precision medicine approach to PCOS diagnosis and treatment. Considering the above, more attention should be focused on these aspects to better improve the nonpharmacological strategies that appear fundamental in the management of PCOS.
Acknowledgments
We thank Elisa Zappelli for technical support with figures and Azzurra Vannucchi for English editing.
Author Contributions
Conceptualization, S.D. and F.F.; writing—original draft preparation, G.S. and J.F.; writing—review and editing, S.D.; supervision, V.C., M.G., C.M. and F.F.; project administration, V.C. and P.G.A.; funding acquisition, P.G.A. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research received no external funding.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Laven J.S.E. Follicle Stimulating Hormone Receptor (FSHR) Polymorphisms and Polycystic Ovary Syndrome (PCOS) Front. Endocrinol. 2019;10:23. doi: 10.3389/fendo.2019.00023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Patel S. Polycystic Ovary Syndrome (PCOS), an Inflammatory, Systemic, Lifestyle Endocrinopathy. J. Steroid Biochem. Mol. Biol. 2018;182:27–36. doi: 10.1016/j.jsbmb.2018.04.008. [DOI] [PubMed] [Google Scholar]
- 3.Witchel S.F., Oberfield S.E., Peña A.S. Polycystic Ovary Syndrome: Pathophysiology, Presentation, and Treatment With Emphasis on Adolescent Girls. J. Endocr. Soc. 2019;3:1545–1573. doi: 10.1210/js.2019-00078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wojciechowska A., Osowski A., Jóźwik M., Górecki R., Rynkiewicz A., Wojtkiewicz J. Inositols’ Importance in the Improvement of the Endocrine–Metabolic Profile in PCOS. IJMS. 2019;20:5787. doi: 10.3390/ijms20225787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ajmal N., Khan S.Z., Shaikh R. Polycystic Ovary Syndrome (PCOS) and Genetic Predisposition: A Review Article. Eur. J. Obstet. Gynecol. Reprod. Biol. X. 2019;3:100060. doi: 10.1016/j.eurox.2019.100060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Costello M.F., Misso M.L., Balen A., Boyle J., Devoto L., Garad R.M., Hart R., Johnson L., Jordan C., Legro R.S., et al. A Brief Update on the Evidence Supporting the Treatment of Infertility in Polycystic Ovary Syndrome. Aust. N. Z. J. Obstet. Gynaecol. 2019;59:867–873. doi: 10.1111/ajo.13051. [DOI] [PubMed] [Google Scholar]
- 7.Hart R. PCOS and Infertility. Panminerva Med. 2008;50:305–314. [PubMed] [Google Scholar]
- 8.Revised 2003 Consensus on Diagnostic Criteria and Long-Term Health Risks Related to Polycystic Ovary Syndrome. Fertil. Steril. 2004;81:19–25. doi: 10.1016/j.fertnstert.2003.10.004. [DOI] [PubMed] [Google Scholar]
- 9.Bednarska S., Siejka A. The Pathogenesis and Treatment of Polycystic Ovary Syndrome: What’s New? Adv. Clin. Exp. Med. 2017;26:359–367. doi: 10.17219/acem/59380. [DOI] [PubMed] [Google Scholar]
- 10.Ganie M., Vasudevan V., Wani I., Baba M., Arif T., Rashid A. Epidemiology, Pathogenesis, Genetics & Management of Polycystic Ovary Syndrome in India. Indian J. Med. Res. 2019;150:333. doi: 10.4103/ijmr.IJMR_1937_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ruiz-Manriquez L.M., Ledesma Pacheco S.J., Medina-Gomez D., Uriostegui-Pena A.G., Estrada-Meza C., Bandyopadhyay A., Pathak S., Banerjee A., Chakraborty S., Srivastava A., et al. A Brief Review on the Regulatory Roles of MicroRNAs in Cystic Diseases and Their Use as Potential Biomarkers. Genes. 2022;13:191. doi: 10.3390/genes13020191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cai G., Ma X., Chen B., Huang Y., Liu S., Yang H., Zou W. MicroRNA-145 Negatively Regulates Cell Proliferation Through Targeting IRS1 in Isolated Ovarian Granulosa Cells From Patients With Polycystic Ovary Syndrome. Reprod. Sci. 2017;24:902–910. doi: 10.1177/1933719116673197. [DOI] [PubMed] [Google Scholar]
- 13.Jiang L., Huang J., Li L., Chen Y., Chen X., Zhao X., Yang D. MicroRNA-93 Promotes Ovarian Granulosa Cells Proliferation through Targeting CDKN1A in Polycystic Ovarian Syndrome. J. Clin. Endocrinol. Metab. 2015;100:E729–E738. doi: 10.1210/jc.2014-3827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yi S., Zheng B., Zhu Y., Cai Y., Sun H., Zhou J. Melatonin Ameliorates Excessive PINK1/Parkin-Mediated Mitophagy by Enhancing SIRT1 Expression in Granulosa Cells of PCOS. Am. J. Physiol. Endocrinol. Metab. 2020;319:E91–E101. doi: 10.1152/ajpendo.00006.2020. [DOI] [PubMed] [Google Scholar]
- 15.Yin L., Luo M., Wang R., Ye J., Wang X. Mitochondria in Sex Hormone-Induced Disorder of Energy Metabolism in Males and Females. Front. Endocrinol. 2021;12:749451. doi: 10.3389/fendo.2021.749451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ye J. Mechanism of Insulin Resistance in Obesity: A Role of ATP. Front. Med. 2021;15:372–382. doi: 10.1007/s11684-021-0862-5. [DOI] [PubMed] [Google Scholar]
- 17.Wu S., Divall S., Nwaopara A., Radovick S., Wondisford F., Ko C., Wolfe A. Obesity-Induced Infertility and Hyperandrogenism Are Corrected by Deletion of the Insulin Receptor in the Ovarian Theca Cell. Diabetes. 2014;63:1270–1282. doi: 10.2337/db13-1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Van Leckwyck M., Kong W., Burton K.J., Amati F., Vionnet N., Pralong F.P. Decreasing Insulin Sensitivity in Women Induces Alterations in LH Pulsatility. J. Clin. Endocrinol. Metab. 2016;101:3240–3249. doi: 10.1210/jc.2016-1727. [DOI] [PubMed] [Google Scholar]
- 19.Wang X., Wang H., Liu W., Zhang Z., Zhang Y., Zhang W., Chen Z., Xia G., Wang C. High Level of C-Type Natriuretic Peptide Induced by Hyperandrogen-Mediated Anovulation in Polycystic Ovary Syndrome Mice. Clin. Sci. 2018;132:759–776. doi: 10.1042/CS20171394. [DOI] [PubMed] [Google Scholar]
- 20.Azhary J.M.K., Harada M., Kunitomi C., Kusamoto A., Takahashi N., Nose E., Oi N., Wada-Hiraike O., Urata Y., Hirata T., et al. Androgens Increase Accumulation of Advanced Glycation End Products in Granulosa Cells by Activating ER Stress in PCOS. Endocrinology. 2020;161:bqaa015. doi: 10.1210/endocr/bqaa015. [DOI] [PubMed] [Google Scholar]
- 21.Diamanti-Kandarakis E., Chatzigeorgiou A., Papageorgiou E., Koundouras D., Koutsilieris M. Advanced Glycation End-Products and Insulin Signaling in Granulosa Cells. Exp. Biol. Med. 2016;241:1438–1445. doi: 10.1177/1535370215584937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yang M., Du J., Lu D., Ren C., Shen H., Qiao J., Chen X., Zhang H. Increased Expression of Kindlin 2 in Luteinized Granulosa Cells Correlates with Androgen Receptor Level in Patients with Polycystic Ovary Syndrome Having Hyperandrogenemia. Reprod. Sci. 2014;21:696–703. doi: 10.1177/1933719113512536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hu M., Zhang Y., Li X., Cui P., Li J., Brännström M., Shao L.R., Billig H. Alterations of Endometrial Epithelial-Mesenchymal Transition and MAPK Signalling Components in Women with PCOS Are Partially Modulated by Metformin in Vitro. Mol. Hum. Reprod. 2020;26:312–326. doi: 10.1093/molehr/gaaa023. [DOI] [PubMed] [Google Scholar]
- 24.Fisch S.C., Nikou A.F., Wright E.A., Phan J.D., Leung K.L., Grogan T.R., Abbott D.H., Chazenbalk G.D., Dumesic D.A. Precocious Subcutaneous Abdominal Stem Cell Development to Adipocytes in Normal-Weight Women with Polycystic Ovary Syndrome. Fertil. Steril. 2018;110:1367–1376. doi: 10.1016/j.fertnstert.2018.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Shaaban Z., Khoradmehr A., Amiri-Yekta A., Nowzari F., Jafarzadeh Shirazi M.R., Tamadon A. Pathophysiologic Mechanisms of Insulin Secretion and Signaling-Related Genes in Etiology of Polycystic Ovary Syndrome. Genet. Res. 2021;2021:1–13. doi: 10.1155/2021/7781823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wawrzkiewicz-Jałowiecka A., Kowalczyk K., Trybek P., Jarosz T., Radosz P., Setlak M., Madej P. In Search of New Therapeutics-Molecular Aspects of the PCOS Pathophysiology: Genetics, Hormones, Metabolism and Beyond. Int. J. Mol. Sci. 2020;21:E7054. doi: 10.3390/ijms21197054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Urbanek M., Woodroffe A., Ewens K.G., Diamanti-Kandarakis E., Legro R.S., Strauss J.F., Dunaif A., Spielman R.S. Candidate Gene Region for Polycystic Ovary Syndrome on Chromosome 19p13.2. J. Clin. Endocrinol. Metab. 2005;90:6623–6629. doi: 10.1210/jc.2005-0622. [DOI] [PubMed] [Google Scholar]
- 28.Anastasia K., Koika V., Roupas N.D., Armeni A., Marioli D., Panidis D., George A., Georgopoulos N.A. Association of Calpain (CAPN) 10 (UCSNP-43, Rs3792267) Gene Polymorphism with Elevated Serum Androgens in Young Women with the Most Severe Phenotype of Polycystic Ovary Syndrome (PCOS) Gynecol. Endocrinol. 2015;31:630–634. doi: 10.3109/09513590.2015.1032932. [DOI] [PubMed] [Google Scholar]
- 29.Dilek S., Ertunc D., Tok E.C., Erdal E.M., Aktas A. Association of Gly972Arg Variant of Insulin Receptor Substrate-1 with Metabolic Features in Women with Polycystic Ovary Syndrome. Fertil. Steril. 2005;84:407–412. doi: 10.1016/j.fertnstert.2005.01.133. [DOI] [PubMed] [Google Scholar]
- 30.Hatfield J., Kasliwal A. Response to “The FSRH Guideline on Conscientious Objection Disrespects Patient Rights and Endangers Their Health”. BMJ Sex. Reprod Health. 2018;44:146. doi: 10.1136/bmjsrh-2018-200103. [DOI] [PubMed] [Google Scholar]
- 31.Chambers A.E., Griffin C., Naif S.A., Mills I., Mills W.E., Syngelaki A., Nicolaides K.H., Banerjee S. Quantitative ELISAs for Serum Soluble LHCGR and HCG-LHCGR Complex: Potential Diagnostics in First Trimester Pregnancy Screening for Stillbirth, Down’s Syndrome, Preterm Delivery and Preeclampsia. Reprod. Biol. Endocrinol. 2012;10:113. doi: 10.1186/1477-7827-10-113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zheng M.-X., Li Y., Hu R., Wang F.-M., Zhang X.-M., Guan B. Anti-Müllerian Hormone Gene Polymorphism Is Associated with Androgen Levels in Chinese Polycystic Ovary Syndrome Patients with Insulin Resistance. J. Assist. Reprod. Genet. 2016;33:199–205. doi: 10.1007/s10815-015-0641-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Witchel S.F., Kahsar-Miller M., Aston C.E., White C., Azziz R. Prevalence of CYP21 Mutations and IRS1 Variant among Women with Polycystic Ovary Syndrome and Adrenal Androgen Excess. Fertil. Steril. 2005;83:371–375. doi: 10.1016/j.fertnstert.2004.10.027. [DOI] [PubMed] [Google Scholar]
- 34.Diamanti-Kandarakis E., Bartzis M.I., Bergiele A.T., Tsianateli T.C., Kouli C.R. Microsatellite Polymorphism (Tttta)(n) at -528 Base Pairs of Gene CYP11alpha Influences Hyperandrogenemia in Patients with Polycystic Ovary Syndrome. Fertil. Steril. 2000;73:735–741. doi: 10.1016/S0015-0282(99)00628-7. [DOI] [PubMed] [Google Scholar]
- 35.Mehdizadeh A., Kalantar S.M., Sheikhha M.H., Aali B.S., Ghanei A. Association of SNP Rs.2414096 CYP19 Gene with Polycystic Ovarian Syndrome in Iranian Women. Int. J. Reprod. Biomed. 2017;15:491–496. doi: 10.29252/ijrm.15.8.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chua A.K., Azziz R., Goodarzi M.O. Association Study of CYP17 and HSD11B1 in Polycystic Ovary Syndrome Utilizing Comprehensive Gene Coverage. Mol. Hum. Reprod. 2012;18:320–324. doi: 10.1093/molehr/gas002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wickenheisser J.K., Quinn P.G., Nelson V.L., Legro R.S., Strauss J.F., McAllister J.M. Differential Activity of the Cytochrome P450 17alpha-Hydroxylase and Steroidogenic Acute Regulatory Protein Gene Promoters in Normal and Polycystic Ovary Syndrome Theca Cells. J. Clin. Endocrinol. Metab. 2000;85:2304–2311. doi: 10.1210/jcem.85.6.6631. [DOI] [PubMed] [Google Scholar]
- 38.Deepika M.L.N., Reddy K.R., Yashwanth A., Rani V.U., Latha K.P., Jahan P. TNF-α Haplotype Association with Polycystic Ovary Syndrome–a South Indian Study. J. Assist. Reprod. Genet. 2013;30:1493–1503. doi: 10.1007/s10815-013-0080-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Deswal R., Yadav A., Dang A.S. Sex Hormone Binding Globulin–an Important Biomarker for Predicting PCOS Risk: A Systematic Review and Meta-Analysis. Syst. Biol. Reprod. Med. 2018;64:12–24. doi: 10.1080/19396368.2017.1410591. [DOI] [PubMed] [Google Scholar]
- 40.Dasgupta S., Sirisha P.V.S., Neelaveni K., Anuradha K., Reddy B.M. Association of CAPN10 SNPs and Haplotypes with Polycystic Ovary Syndrome among South Indian Women. PLoS ONE. 2012;7:e32192. doi: 10.1371/journal.pone.0032192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wiltgen D., Furtado L., Kohek M.B.F., Spritzer P.M. CAPN10 UCSNP-43, UCSNP-19 and UCSNP-63 Polymorphisms and Metabolic Syndrome in Polycystic Ovary Syndrome. Gynecol. Endocrinol. 2007;23:173–178. doi: 10.1080/09513590701233661. [DOI] [PubMed] [Google Scholar]
- 42.Mutib M.T., Hamdan F.B., Al-Salihi A.R. INSR Gene Variation Is Associated with Decreased Insulin Sensitivity in Iraqi Women with PCOs. Iran. J. Reprod. Med. 2014;12:499–506. [PMC free article] [PubMed] [Google Scholar]
- 43.Daghestani M.H. Rs1799817 in INSR Associates with Susceptibility to Polycystic Ovary Syndrome. J. Med. Biochem. 2020;39:149. doi: 10.2478/jomb-2019-0023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Song D.K., Lee H., Oh J.-Y., Hong Y.S., Sung Y.-A. FTO Gene Variants Are Associated with PCOS Susceptibility and Hyperandrogenemia in Young Korean Women. Diabetes Metab. J. 2014;38:302. doi: 10.4093/dmj.2014.38.4.302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chaudhary H., Patel J., Jain N.K., Joshi R. The Role of Polymorphism in Various Potential Genes on Polycystic Ovary Syndrome Susceptibility and Pathogenesis. J. Ovarian Res. 2021;14:125. doi: 10.1186/s13048-021-00879-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ashraf S., Rasool S.U.A., Nabi M., Ganie M.A., Masoodi S.R., Amin S. Impact of Rs2414096 Polymorphism of CYP19 Gene on Susceptibility of Polycystic Ovary Syndrome and Hyperandrogenism in Kashmiri Women. Sci. Rep. 2021;11:12942. doi: 10.1038/s41598-021-92265-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Jin J.-L., Sun J., Ge H.-J., Cao Y.-X., Wu X.-K., Liang F.-J., Sun H.-X., Ke L., Yi L., Wu Z.-W., et al. Association between CYP19 Gene SNP Rs2414096 Polymorphism and Polycystic Ovary Syndrome in Chinese Women. BMC Med. Genet. 2009;10:139. doi: 10.1186/1471-2350-10-139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Munawar Lone N., Babar S., Sultan S., Malik S., Nazeer K., Riaz S. Association of the CYP17 and CYP19 Gene Polymorphisms in Women with Polycystic Ovary Syndrome from Punjab, Pakistan. Gynecol. Endocrinol. 2021;37:456–461. doi: 10.1080/09513590.2020.1822803. [DOI] [PubMed] [Google Scholar]
- 49.Astapova O., Minor B.M.N., Hammes S.R. Physiological and Pathological Androgen Actions in the Ovary. Endocrinology. 2019;160:1166–1174. doi: 10.1210/en.2019-00101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Polat S., Karaburgu S., Unluhizarci K., Dündar M., Özkul Y., Arslan Y.K., Karaca Z., Kelestimur F. The Role of Androgen Receptor CAG Repeat Polymorphism in Androgen Excess Disorder and Idiopathic Hirsutism. J. Endocrinol. Invest. 2020;43:1271–1281. doi: 10.1007/s40618-020-01215-7. [DOI] [PubMed] [Google Scholar]
- 51.Li Y., Fang L., Yan Y., Wang Z., Wu Z., Jia Q., Cheng J.-C., Sun Y.-P. Association between Human SHBG Gene Polymorphisms and Risk of PCOS: A Meta-Analysis. Reprod. BioMed. Online. 2021;42:227–236. doi: 10.1016/j.rbmo.2020.10.003. [DOI] [PubMed] [Google Scholar]
- 52.Zhu J., Chen Z., Feng W., Long S., Mo Z.-C. Sex Hormone-Binding Globulin and Polycystic Ovary Syndrome. Clin. Chim. Acta. 2019;499:142–148. doi: 10.1016/j.cca.2019.09.010. [DOI] [PubMed] [Google Scholar]
- 53.Deswal R., Nanda S., Dang A.S. Association of Luteinizing Hormone and LH Receptor Gene Polymorphism with Susceptibility of Polycystic Ovary Syndrome. Syst. Biol. Reprod. Med. 2019;65:400–408. doi: 10.1080/19396368.2019.1595217. [DOI] [PubMed] [Google Scholar]
- 54.Abutorabi E.S., Rashidi B.H., Irani S., Haghollahi F., Bagheri M. Investigation of the FSHR, CYP11, and INSR Mutations and Polymorphisms in Iranian Infertile Women with Polycystic Ovary Syndrome (PCOS) Rep. Biochem. Mol. Biol. 2021;9:470–477. doi: 10.52547/rbmb.9.4.470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ferk P., Teran N., Gersak K. The (TAAAA)n Microsatellite Polymorphism in the SHBG Gene Influences Serum SHBG Levels in Women with Polycystic Ovary Syndrome. Hum. Reprod. 2007;22:1031–1036. doi: 10.1093/humrep/del457. [DOI] [PubMed] [Google Scholar]
- 56.Concha F., Recabarren S.E., Pérez B.F. Epigenética Del Síndrome de Ovario Poliquístico. Rev. Méd. Chile. 2017;145:907–915. doi: 10.4067/s0034-98872017000700907. [DOI] [PubMed] [Google Scholar]
- 57.Illingworth R., Kerr A., Desousa D., Jørgensen H., Ellis P., Stalker J., Jackson D., Clee C., Plumb R., Rogers J., et al. A Novel CpG Island Set Identifies Tissue-Specific Methylation at Developmental Gene Loci. PLoS Biol. 2008;6:e22. doi: 10.1371/journal.pbio.0060022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Vázquez-Martínez E.R., Gómez-Viais Y.I., García-Gómez E., Reyes-Mayoral C., Reyes-Muñoz E., Camacho-Arroyo I., Cerbón M. DNA Methylation in the Pathogenesis of Polycystic Ovary Syndrome. Reproduction. 2019;158:R27–R40. doi: 10.1530/REP-18-0449. [DOI] [PubMed] [Google Scholar]
- 59.Cui P., Ma T., Tamadon A., Han S., Li B., Chen Z., An X., Shao L.R., Wang Y., Feng Y. Hypothalamic DNA Methylation in Rats with Dihydrotestosterone-Induced Polycystic Ovary Syndrome: Effects of Low-Frequency Electro-Acupuncture. Exp. Physiol. 2018;103:1618–1632. doi: 10.1113/EP087163. [DOI] [PubMed] [Google Scholar]
- 60.Kokosar M., Benrick A., Perfilyev A., Fornes R., Nilsson E., Maliqueo M., Behre C.J., Sazonova A., Ohlsson C., Ling C., et al. Epigenetic and Transcriptional Alterations in Human Adipose Tissue of Polycystic Ovary Syndrome. Sci. Rep. 2016;6:22883. doi: 10.1038/srep22883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Shen H.-R., Qiu L.-H., Zhang Z.-Q., Qin Y.-Y., Cao C., Di W. Genome-Wide Methylated DNA Immunoprecipitation Analysis of Patients with Polycystic Ovary Syndrome. PLoS ONE. 2013;8:e64801. doi: 10.1371/journal.pone.0064801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang X.-X., Wei J.-Z., Jiao J., Jiang S.-Y., Yu D.-H., Li D. Genome-Wide DNA Methylation and Gene Expression Patterns Provide Insight into Polycystic Ovary Syndrome Development. Oncotarget. 2014;5:6603–6610. doi: 10.18632/oncotarget.2224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Qu F., Wang F.-F., Yin R., Ding G.-L., El-prince M., Gao Q., Shi B.-W., Pan H.-H., Huang Y.-T., Jin M., et al. A Molecular Mechanism Underlying Ovarian Dysfunction of Polycystic Ovary Syndrome: Hyperandrogenism Induces Epigenetic Alterations in the Granulosa Cells. Mol. Med. 2012;90:911–923. doi: 10.1007/s00109-012-0881-4. [DOI] [PubMed] [Google Scholar]
- 64.Jiang L.-L., Xie J.-K., Cui J.-Q., Wei D., Yin B.-L., Zhang Y.-N., Chen Y.-H., Han X., Wang Q., Zhang C.-L. Promoter Methylation of Yes-Associated Protein (YAP1) Gene in Polycystic Ovary Syndrome. Medicine. 2017;96:e5768. doi: 10.1097/MD.0000000000005768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Sagvekar P., Kumar P., Mangoli V., Desai S., Mukherjee S. DNA Methylome Profiling of Granulosa Cells Reveals Altered Methylation in Genes Regulating Vital Ovarian Functions in Polycystic Ovary Syndrome. Clin. Epigenet. 2019;11:61. doi: 10.1186/s13148-019-0657-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Li S., Zhu D., Duan H., Ren A., Glintborg D., Andersen M., Skov V., Thomassen M., Kruse T., Tan Q. Differential DNA Methylation Patterns of Polycystic Ovarian Syndrome in Whole Blood of Chinese Women. Oncotarget. 2017;8:20656–20666. doi: 10.18632/oncotarget.9327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Sang Q., Zhang S., Zou S., Wang H., Feng R., Li Q., Jin L., He L., Xing Q., Wang L. Quantitative Analysis of Follistatin (FST) Promoter Methylation in Peripheral Blood of Patients with Polycystic Ovary Syndrome. Reprod. BioMed. Online. 2013;26:157–163. doi: 10.1016/j.rbmo.2012.10.011. [DOI] [PubMed] [Google Scholar]
- 68.Sang Q., Li X., Wang H., Wang H., Zhang S., Feng R., Xu Y., Li Q., Zhao X., Xing Q., et al. Quantitative Methylation Level of the EPHX1 Promoter in Peripheral Blood DNA Is Associated with Polycystic Ovary Syndrome. PLoS ONE. 2014;9:e88013. doi: 10.1371/journal.pone.0088013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ting W., Yanyan Q., Jian H., Keqin H., Duan M. The Relationship Between Insulin Resistance and CpG Island Methylation of LMNA Gene in Polycystic Ovary Syndrome. Cell Biochem. Biophys. 2013;67:1041–1047. doi: 10.1007/s12013-013-9602-z. [DOI] [PubMed] [Google Scholar]
- 70.Teede H., Ng S., Hedger M., Moran L. Follistatin and Activins in Polycystic Ovary Syndrome: Relationship to Metabolic and Hormonal Markers. Metabolism. 2013;62:1394–1400. doi: 10.1016/j.metabol.2013.05.003. [DOI] [PubMed] [Google Scholar]
- 71.Hai L., McGee S.R., Rabideau A.C., Paquet M., Narayan P. Infertility in Female Mice with a Gain-of-Function Mutation in the Luteinizing Hormone Receptor Is Due to Irregular Estrous Cyclicity, Anovulation, Hormonal Alterations, and Polycystic Ovaries1. Biol. Reprod. 2015;93:1–11. doi: 10.1095/biolreprod.115.129072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Korhonen S. Two Exonic Single Nucleotide Polymorphisms in the Microsomal Epoxide Hydrolase Gene Are Associated with Polycystic Ovary Syndrome. Fertil. Steril. 2003;79:1353–1357. doi: 10.1016/S0015-0282(03)00385-6. [DOI] [PubMed] [Google Scholar]
- 73.Vandenbeek R., Khan N.P., Estall J.L. Linking Metabolic Disease With the PGC-1α Gly482Ser Polymorphism. Endocrinology. 2018;159:853–865. doi: 10.1210/en.2017-00872. [DOI] [PubMed] [Google Scholar]
- 74.Titus A.J., Gallimore R.M., Salas L.A., Christensen B.C. Cell-Type Deconvolution from DNA Methylation: A Review of Recent Applications. Hum. Mol. Genet. 2017;26:R216–R224. doi: 10.1093/hmg/ddx275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Luense L.J., Veiga-Lopez A., Padmanabhan V., Christenson L.K. Developmental Programming: Gestational Testosterone Treatment Alters Fetal Ovarian Gene Expression. Endocrinology. 2011;152:4974–4983. doi: 10.1210/en.2011-1182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Eini F., Novin M.G., Joharchi K., Hosseini A., Nazarian H., Piryaei A., Bidadkosh A. Intracytoplasmic Oxidative Stress Reverses Epigenetic Modifications in Polycystic Ovary Syndrome. Reprod. Fertil. Dev. 2017;29:2313. doi: 10.1071/RD16428. [DOI] [PubMed] [Google Scholar]
- 77.Li Q.-N., Guo L., Hou Y., Ou X.-H., Liu Z., Sun Q.-Y. The DNA Methylation Profile of Oocytes in Mice with Hyperinsulinaemia and Hyperandrogenism as Detected by Single-Cell Level Whole Genome Bisulphite Sequencing (SC-WGBS) Technology. Reprod. Fertil. Dev. 2018;30:1713. doi: 10.1071/RD18002. [DOI] [PubMed] [Google Scholar]
- 78.Qin Y., Li T., Zhao H., Mao Z., Ding C., Kang Y. Integrated Transcriptomic and Epigenetic Study of PCOS: Impact of Map3k1 and Map1lc3a Promoter Methylation on Autophagy. Front. Genet. 2021;12:620241. doi: 10.3389/fgene.2021.620241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Mimouni N.E.H., Paiva I., Barbotin A.-L., Timzoura F.E., Plassard D., Le Gras S., Ternier G., Pigny P., Catteau-Jonard S., Simon V., et al. Polycystic Ovary Syndrome Is Transmitted via a Transgenerational Epigenetic Process. Cell Metab. 2021;33:513–530.e8. doi: 10.1016/j.cmet.2021.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pan J.-X., Tan Y.-J., Wang F.-F., Hou N.-N., Xiang Y.-Q., Zhang J.-Y., Liu Y., Qu F., Meng Q., Xu J., et al. Aberrant Expression and DNA Methylation of Lipid Metabolism Genes in PCOS: A New Insight into Its Pathogenesis. Clin. Epigenet. 2018;10:6. doi: 10.1186/s13148-018-0442-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Li D., Jiao J., Zhou Y.-M., Wang X.-X. Epigenetic Regulation of Traf2- and Nck-Interacting Kinase (TNIK) in Polycystic Ovary Syndrome. Am. J. Transl. Res. 2015;7:1152–1160. [PMC free article] [PubMed] [Google Scholar]
- 82.Jones M.R., Brower M.A., Xu N., Cui J., Mengesha E., Chen Y.-D.I., Taylor K.D., Azziz R., Goodarzi M.O. Systems Genetics Reveals the Functional Context of PCOS Loci and Identifies Genetic and Molecular Mechanisms of Disease Heterogeneity. PLoS Genet. 2015;11:e1005455. doi: 10.1371/journal.pgen.1005455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kumariya S., Ubba V., Jha R.K., Gayen J.R. Autophagy in Ovary and Polycystic Ovary Syndrome: Role, Dispute and Future Perspective. Autophagy. 2021;17:2706–2733. doi: 10.1080/15548627.2021.1938914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Chen L., Heikkinen L., Wang C., Yang Y., Sun H., Wong G. Trends in the Development of MiRNA Bioinformatics Tools. Brief. Bioinform. 2019;20:1836–1852. doi: 10.1093/bib/bby054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Mao Z., Li T., Zhao H., Qin Y., Wang X., Kang Y. Identification of Epigenetic Interactions between MicroRNA and DNA Methylation Associated with Polycystic Ovarian Syndrome. J. Hum. Genet. 2021;66:123–137. doi: 10.1038/s10038-020-0819-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Jiang X., Li J., Zhang B., Hu J., Ma J., Cui L., Chen Z.-J. Differential Expression Profile of Plasma Exosomal MicroRNAs in Women with Polycystic Ovary Syndrome. Fertil. Steril. 2021;115:782–792. doi: 10.1016/j.fertnstert.2020.08.019. [DOI] [PubMed] [Google Scholar]
- 87.Chen Z., Ou H., Wu H., Wu P., Mo Z. Role of MicroRNA in the Pathogenesis of Polycystic Ovary Syndrome. DNA Cell Biol. 2019;38:754–762. doi: 10.1089/dna.2019.4622. [DOI] [PubMed] [Google Scholar]
- 88.Long W., Zhao C., Ji C., Ding H., Cui Y., Guo X., Shen R., Liu J. Characterization of Serum MicroRNAs Profile of PCOS and Identification of Novel Non-Invasive Biomarkers. Cell Physiol. Biochem. 2014;33:1304–1315. doi: 10.1159/000358698. [DOI] [PubMed] [Google Scholar]
- 89.Mao Z., Fan L., Yu Q., Luo S., Wu X., Tang J., Kang G., Tang L. Abnormality of Klotho Signaling Is Involved in Polycystic Ovary Syndrome. Reprod. Sci. 2018;25:372–383. doi: 10.1177/1933719117715129. [DOI] [PubMed] [Google Scholar]
- 90.Xu B., Zhang Y.-W., Tong X.-H., Liu Y.-S. Characterization of MicroRNA Profile in Human Cumulus Granulosa Cells: Identification of MicroRNAs That Regulate Notch Signaling and Are Associated with PCOS. Mol. Cell Endocrinol. 2015;404:26–36. doi: 10.1016/j.mce.2015.01.030. [DOI] [PubMed] [Google Scholar]
- 91.Lin L., Du T., Huang J., Huang L.-L., Yang D.-Z. Identification of Differentially Expressed MicroRNAs in the Ovary of Polycystic Ovary Syndrome with Hyperandrogenism and Insulin Resistance. Chin. Med. J. 2015;128:169–174. doi: 10.4103/0366-6999.149189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Song J., Luo S., Li S.-W. MiRNA-592 Is Downregulated and May Target LHCGR in Polycystic Ovary Syndrome Patients. Reprod. Biol. 2015;15:229–237. doi: 10.1016/j.repbio.2015.10.005. [DOI] [PubMed] [Google Scholar]
- 93.Hosseini E., Shahhoseini M., Afsharian P., Karimian L., Ashrafi M., Mehraein F., Afatoonian R. Role of Epigenetic Modifications in the Aberrant CYP19A1 Gene Expression in Polycystic Ovary Syndrome. Arch. Med. Sci. AMS. 2019;15:887–895. doi: 10.5114/aoms.2019.86060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Leung K.L., Sanchita S., Pham C.T., Davis B.A., Okhovat M., Ding X., Dumesic P., Grogan T.R., Williams K.J., Morselli M., et al. Dynamic Changes in Chromatin Accessibility, Altered Adipogenic Gene Expression, and Total versus de Novo Fatty Acid Synthesis in Subcutaneous Adipose Stem Cells of Normal-Weight Polycystic Ovary Syndrome (PCOS) Women during Adipogenesis: Evidence of Cellular Programming. Clin. Epigenet. 2020;12:181. doi: 10.1186/s13148-020-00970-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Romitti M., Fabris V.C., Ziegelmann P.K., Maia A.L., Spritzer P.M. Association between PCOS and Autoimmune Thyroid Disease: A Systematic Review and Meta-Analysis. Endocr. Connect. 2018;7:1158–1167. doi: 10.1530/EC-18-0309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Serin A.N., Birge Ö., Uysal A., Görar S., Tekeli F. Hashimoto’s Thyroiditis Worsens Ovaries in Polycystic Ovary Syndrome Patients Compared to Anti-Müllerian Hormone Levels. BMC Endocr. Disord. 2021;21:44. doi: 10.1186/s12902-021-00706-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Adamska A., Łebkowska A., Krentowska A., Hryniewicka J., Adamski M., Leśniewska M., Polak A.M., Kowalska I. Ovarian Reserve and Serum Concentration of Thyroid Peroxidase Antibodies in Euthyroid Women With Different Polycystic Ovary Syndrome Phenotypes. Front. Endocrinol. 2020;11:440. doi: 10.3389/fendo.2020.00440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Krishna M.B., Joseph A., Subramaniam A.G., Gupta A., Pillai S.M., Laloraya M. Reduced Tregs in Peripheral Blood of PCOS Patients–a Consequence of Aberrant Il2 Signaling. J. Clin. Endocrinol. Metab. 2015;100:282–292. doi: 10.1210/jc.2014-2401. [DOI] [PubMed] [Google Scholar]
- 99.Rashid A., Bhat J.A., Ganie M.A., Wani I.A., Bhat M.H., Shah Z.A., Masoodi S.R., Marwaha R.K. Evaluation of Serum Anti-Nuclear Antibody among Women with PCOS: A Hospital Based Single Center Cross Sectional Study. Gynecol. Endocrinol. 2018;34:965–969. doi: 10.1080/09513590.2018.1469613. [DOI] [PubMed] [Google Scholar]
- 100.Palacio J.R., Iborra A., Ulcova-Gallova Z., Badia R., Martínez P. The Presence of Antibodies to Oxidative Modified Proteins in Serum from Polycystic Ovary Syndrome Patients. Clin. Exp. Immunol. 2006;144:217–222. doi: 10.1111/j.1365-2249.2006.03061.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Gingnell M., Dahlbom I., Lindholm Å., Hudecova M., Arnadottir R., Hansson T., Sundstrom-Poromaa I. Patients with Polycystic Ovary Syndrome Have Lower Levels of IgM Anti-Phosphorylcholine Antibodies than Healthy Women. Gynecol. Endocrinol. 2011;27:486–490. doi: 10.3109/09513590.2010.501880. [DOI] [PubMed] [Google Scholar]
- 102.Li H., Guo Y., Deng J., Fischer H., Weedin E.A., Burks H.R., Craig L.B., Yu X. Increased Testosterone and Proinflammatory Cytokines in Patients with Polycystic Ovary Syndrome Correlate with Elevated GnRH Receptor Autoantibody Activity Assessed by a Fluorescence Resonance Energy Transfer-Based Bioassay. Endocrine. 2021;74:163–171. doi: 10.1007/s12020-021-02761-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Weedin E.A., Burks H.R., Yu X., Li H.L., Aston C.E., Kem D.C., Craig L.B. Elevated Activity Levels of Activating Autoantibodies to the GnRH Receptor in Patients with Polycystic Ovary Syndrome. F&S Rep. 2020;1:299–304. doi: 10.1016/j.xfre.2020.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Li H., Zhang G., Guo Y., Deng J., Fischer H., Craig L.B., Kem D.C., Yu X. Autoimmune Activation of the GnRH Receptor Induces Insulin Resistance Independent of Obesity in a Female Rat Model. Physiol. Rep. 2021;8:e14672. doi: 10.14814/phy2.14672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Schniewind H.A., Sattler L.-M., Haudum C.W., Münzker J., Minich W.B., Obermayer-Pietsch B., Schomburg L. Autoimmunity to the Follicle-Stimulating Hormone Receptor (FSHR) and Luteinizing Hormone Receptor (LHR) in Polycystic Ovarian Syndrome. IJMS. 2021;22:13667. doi: 10.3390/ijms222413667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Panidis D., Tziomalos K., Papadakis E., Vosnakis C., Chatzis P., Katsikis I. Lifestyle Intervention and Anti-Obesity Therapies in the Polycystic Ovary Syndrome: Impact on Metabolism and Fertility. Endocrine. 2013;44:583–590. doi: 10.1007/s12020-013-9971-5. [DOI] [PubMed] [Google Scholar]
- 107.Cena H., Chiovato L., Nappi R.E. Obesity, Polycystic Ovary Syndrome, and Infertility: A New Avenue for GLP-1 Receptor Agonists. J. Clin. Endocrinol. Metab. 2020;105:e2695–e2709. doi: 10.1210/clinem/dgaa285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Silvestris E., de Pergola G., Rosania R., Loverro G. Obesity as Disruptor of the Female Fertility. Reprod. Biol. Endocrinol. 2018;16:22. doi: 10.1186/s12958-018-0336-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Asdaq S.M.B., Jomah S., Hasan R., Al-Baroudi D., Alharbi M., Alsubaie S., Buhamad M.H., Alyahya B., Al-Yamani M.J. Impact of Polycystic Ovary Syndrome on Eating Behavior, Depression and Health Related Quality of Life: A Cross-Sectional Study in Riyadh. Saudi J. Biol. Sci. 2020;27:3342–3347. doi: 10.1016/j.sjbs.2020.08.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sadeeqa S., Mustafa T., Latif S. Polycystic Ovarian Syndrome–Related Depression in Adolescent Girls: A Review. J. Pharm. Bioall. Sci. 2018;10:55. doi: 10.4103/JPBS.JPBS_1_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kolhe J.V., Chhipa A.S., Butani S., Chavda V., Patel S.S. PCOS and Depression: Common Links and Potential Targets. Reprod. Sci. 2021:1–18. doi: 10.1007/s43032-021-00765-2. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 112.Simon S., Rahat H., Carreau A.-M., Garcia-Reyes Y., Halbower A., Pyle L., Nadeau K.J., Cree-Green M. Poor Sleep Is Related to Metabolic Syndrome Severity in Adolescents With PCOS and Obesity. J. Clin. Endocrinol. Metab. 2020;105:e1827–e1834. doi: 10.1210/clinem/dgz285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Monteiro R., Azevedo I. Chronic Inflammation in Obesity and the Metabolic Syndrome. Mediat. Inflamm. 2010;2010:1–10. doi: 10.1155/2010/289645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Mesarwi O.A., Loomba R., Malhotra A. Obstructive Sleep Apnea, Hypoxia, and Nonalcoholic Fatty Liver Disease. Am. J. Respir. Crit. Care Med. 2019;199:830–841. doi: 10.1164/rccm.201806-1109TR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Leproult R., Van Cauter E. Role of Sleep and Sleep Loss in Hormonal Release and Metabolism. In: Loche S., Cappa M., Ghizzoni L., Maghnie M., Savage M.O., editors. Endocrine Development. Volume 17. KARGER; Basel, Switzerland: 2009. pp. 11–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Kite C., Lahart I.M., Afzal I., Broom D.R., Randeva H., Kyrou I., Brown J.E. Exercise, or Exercise and Diet for the Management of Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis. Syst. Rev. 2019;8:51. doi: 10.1186/s13643-019-0962-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Szczuko M., Kikut J., Szczuko U., Szydłowska I., Nawrocka-Rutkowska J., Ziętek M., Verbanac D., Saso L. Nutrition Strategy and Life Style in Polycystic Ovary Syndrome—Narrative Review. Nutrients. 2021;13:2452. doi: 10.3390/nu13072452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Mehrabani H.H., Salehpour S., Amiri Z., Farahani S.J., Meyer B.J., Tahbaz F. Beneficial Effects of a High-Protein, Low-Glycemic-Load Hypocaloric Diet in Overweight and Obese Women with Polycystic Ovary Syndrome: A Randomized Controlled Intervention Study. J. Am. Coll. Nutr. 2012;31:117–125. doi: 10.1080/07315724.2012.10720017. [DOI] [PubMed] [Google Scholar]
- 119.Gower B.A., Chandler-Laney P.C., Ovalle F., Goree L.L., Azziz R., Desmond R.A., Granger W.M., Goss A.M., Bates G.W. Favourable Metabolic Effects of a Eucaloric Lower-Carbohydrate Diet in Women with PCOS. Clin. Endocrinol. 2013;79:550–557. doi: 10.1111/cen.12175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Kazemi M., Hadi A., Pierson R.A., Lujan M.E., Zello G.A., Chilibeck P.D. Effects of Dietary Glycemic Index and Glycemic Load on Cardiometabolic and Reproductive Profiles in Women with Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2021;12:161–178. doi: 10.1093/advances/nmaa092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Cutler D.A., Pride S.M., Cheung A.P. Low Intakes of Dietary Fiber and Magnesium Are Associated with Insulin Resistance and Hyperandrogenism in Polycystic Ovary Syndrome: A Cohort Study. Food Sci. Nutr. 2019;7:1426–1437. doi: 10.1002/fsn3.977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Cincione R.I., Losavio F., Ciolli F., Valenzano A., Cibelli G., Messina G., Polito R. Effects of Mixed of a Ketogenic Diet in Overweight and Obese Women with Polycystic Ovary Syndrome. IJERPH. 2021;18:12490. doi: 10.3390/ijerph182312490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Mavropoulos J.C., Yancy W.S., Hepburn J., Westman E.C. The Effects of a Low-Carbohydrate, Ketogenic Diet on the Polycystic Ovary Syndrome: A Pilot Study. Nutr. Metab. 2005;2:35. doi: 10.1186/1743-7075-2-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Rudnicka E., Suchta K., Grymowicz M., Calik-Ksepka A., Smolarczyk K., Duszewska A.M., Smolarczyk R., Meczekalski B. Chronic Low Grade Inflammation in Pathogenesis of PCOS. IJMS. 2021;22:3789. doi: 10.3390/ijms22073789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Youm Y.-H., Nguyen K.Y., Grant R.W., Goldberg E.L., Bodogai M., Kim D., D’Agostino D., Planavsky N., Lupfer C., Kanneganti T.D., et al. The Ketone Metabolite β-Hydroxybutyrate Blocks NLRP3 Inflammasome–Mediated Inflammatory Disease. Nat. Med. 2015;21:263–269. doi: 10.1038/nm.3804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Maktabi M., Jamilian M., Asemi Z. Magnesium-Zinc-Calcium-Vitamin D Co-Supplementation Improves Hormonal Profiles, Biomarkers of Inflammation and Oxidative Stress in Women with Polycystic Ovary Syndrome: A Randomized, Double-Blind, Placebo-Controlled Trial. Biol. Trace Elem. Res. 2018;182:21–28. doi: 10.1007/s12011-017-1085-0. [DOI] [PubMed] [Google Scholar]
- 127.Mas-Bargues C., Escrivá C., Dromant M., Borrás C., Viña J. Lipid Peroxidation as Measured by Chromatographic Determination of Malondialdehyde. Human Plasma Reference Values in Health and Disease. Arch. Biochem. Biophys. 2021;709:108941. doi: 10.1016/j.abb.2021.108941. [DOI] [PubMed] [Google Scholar]
- 128.Afshar Ebrahimi F., Foroozanfard F., Aghadavod E., Bahmani F., Asemi Z. The Effects of Magnesium and Zinc Co-Supplementation on Biomarkers of Inflammation and Oxidative Stress, and Gene Expression Related to Inflammation in Polycystic Ovary Syndrome: A Randomized Controlled Clinical Trial. Biol. Trace Elem. Res. 2018;184:300–307. doi: 10.1007/s12011-017-1198-5. [DOI] [PubMed] [Google Scholar]
- 129.Hamilton K.P., Zelig R., Parker A.R., Haggag A. Insulin Resistance and Serum Magnesium Concentrations among Women with Polycystic Ovary Syndrome. Curr. Dev. Nutr. 2019;3:nzz108. doi: 10.1093/cdn/nzz108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Guo S., Tal R., Jiang H., Yuan T., Liu Y. Vitamin D Supplementation Ameliorates Metabolic Dysfunction in Patients with PCOS: A SystematicReview of RCTs and Insight into the Underlying Mechanism. Int. J. Endocrinol. 2020;2020:1–18. doi: 10.1155/2020/7850816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Zhao J.-F., Li B.-X., Zhang Q. Vitamin D Improves Levels of Hormonal, Oxidative Stress and Inflammatory Parameters in Polycystic Ovary Syndrome: A Meta-Analysis Study. Ann. Palliat. Med. 2021;10:169–183. doi: 10.21037/apm-20-2201. [DOI] [PubMed] [Google Scholar]
- 132.Costantini L., Molinari R., Farinon B., Merendino N. Impact of Omega-3 Fatty Acids on the Gut Microbiota. IJMS. 2017;18:2645. doi: 10.3390/ijms18122645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Yuan J., Wen X., Jia M. Efficacy of Omega-3 Polyunsaturated Fatty Acids on Hormones, Oxidative Stress, and Inflammatory Parameters among Polycystic Ovary Syndrome: A Systematic Review and Meta-Analysis. Ann. Palliat. Med. 2021;10:8991–9001. doi: 10.21037/apm-21-2018. [DOI] [PubMed] [Google Scholar]
- 134.Unfer V. A Deeper Assessment of Ω3-Poly-Unsaturated Fatty Acids in Polycystic Ovary Syndrome Management. Comment on Regidor et al. Chronic Inflammation in PCOS: The Potential Benefits of Specialized Pro-Resolving Lipid Mediators (SPMs) in the Improvement of the Resolutive Response. Int. J. Mol. Sci. 2021;22:10114. doi: 10.3390/ijms221810114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Salek M., Clark C.C.T., Taghizadeh M., Jafarnejad S. N-3 Fatty Acids as Preventive and Therapeutic Agents in Attenuating PCOS Complications. EXCLI J. 2019;18:558. doi: 10.17179/EXCLI2019-1534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Nordio M., Basciani S., Camajani E. The 40:1 Myo-Inositol/D-Chiro-Inositol Plasma Ratio Is Able to Restore Ovulation in PCOS Patients: Comparison with Other Ratios. Eur. Rev. Med. Pharmacol. Sci. 2019;23:5512–5521. doi: 10.26355/eurrev_201906_18223. [DOI] [PubMed] [Google Scholar]
- 137.Kamenov Z., Gateva A. Inositols in PCOS. Molecules. 2020;25:5566. doi: 10.3390/molecules25235566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Colak E., Ozcimen E.E., Tohma Y.A., Ceran M.U. May Myo-inositol and d -chiro-inositol (40:1) Treatment Be a Good Option on Normal-weighted Polycystic Ovary Syndrome Patients without Insulin Resistance? J. Obstet. Gynaecol. Res. 2020;46:2605–2611. doi: 10.1111/jog.14505. [DOI] [PubMed] [Google Scholar]
- 139.Artini P.G., Malacarne E., Tomatis V., Genazzani A.D. The Relevance of Inositols Treatment for PCOS before and during ART. Eur. Rev. Med. Pharmacol. Sci. 2021;25:4799–4809. doi: 10.26355/eurrev_202107_26393. [DOI] [PubMed] [Google Scholar]
- 140.Di Emidio G., Rea F., Placidi M., Rossi G., Cocciolone D., Virmani A., Macchiarelli G., Palmerini M.G., D’Alessandro A.M., Artini P.G., et al. Regulatory Functions of L-Carnitine, Acetyl, and Propionyl L-Carnitine in a PCOS Mouse Model: Focus on Antioxidant/Antiglycative Molecular Pathways in the Ovarian Microenvironment. Antioxidants. 2020;9:867. doi: 10.3390/antiox9090867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Tauqir S., Israr M., Rauf B., Malik M.O., Habib S.H., Shah F.A., Usman M., Raza M.A., Shah I., Badshah H., et al. Acetyl-l-Carnitine Ameliorates Metabolic and Endocrine Alterations in Women with PCOS: A Double-Blind Randomized Clinical Trial. Adv. Ther. 2021;38:3842–3856. doi: 10.1007/s12325-021-01789-5. [DOI] [PubMed] [Google Scholar]
- 142.Akbaribazm M., Goodarzi N., Rahimi M. Female Infertility and Herbal Medicine: An Overview of the New Findings. Food Sci. Nutr. 2021;9:5869–5882. doi: 10.1002/fsn3.2523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Zhou Y., Lv L., Liu Q., Song J. Total Flavonoids Extracted from Nervilia Fordii Function in Polycystic Ovary Syndrome through IL-6 Mediated JAK2/STAT3 Signaling Pathway. Biosci. Rep. 2019;39:BSR20181380. doi: 10.1042/BSR20181380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Zhao X., Jiang Y., Xi H., Chen L., Feng X. Exploration of the Relationship Between Gut Microbiota and Polycystic Ovary Syndrome (PCOS): A Review. Geburtshilfe Frauenheilkd. 2020;80:161–171. doi: 10.1055/a-1081-2036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Zheng Y., Yu J., Liang C., Li S., Wen X., Li Y. Characterization on Gut Microbiome of PCOS Rats and Its Further Design by Shifts in High-Fat Diet and Dihydrotestosterone Induction in PCOS Rats. Bioprocess. Biosyst. Eng. 2021;44:953–964. doi: 10.1007/s00449-020-02320-w. [DOI] [PubMed] [Google Scholar]
- 146.Giampaolino P., Foreste V., Di Filippo C., Gallo A., Mercorio A., Serafino P., Improda F.P., Verrazzo P., Zara G., Buonfantino C., et al. Microbiome and PCOS: State-of-Art and Future Aspects. IJMS. 2021;22:2048. doi: 10.3390/ijms22042048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Wang X., Xu T., Liu R., Wu G., Gu L., Zhang Y., Zhang F., Fu H., Ling Y., Wei X., et al. High-Fiber Diet or Combined With Acarbose Alleviates Heterogeneous Phenotypes of Polycystic Ovary Syndrome by Regulating Gut Microbiota. Front. Endocrinol. 2022;12:806331. doi: 10.3389/fendo.2021.806331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Wang T., Sha L., Li Y., Zhu L., Wang Z., Li K., Lu H., Bao T., Guo L., Zhang X., et al. Dietary α-Linolenic Acid-Rich Flaxseed Oil Exerts Beneficial Effects on Polycystic Ovary Syndrome Through Sex Steroid Hormones—Microbiota—Inflammation Axis in Rats. Front. Endocrinol. 2020;11:284. doi: 10.3389/fendo.2020.00284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Woodward A., Klonizakis M., Broom D. Exercise and Polycystic Ovary Syndrome. In: Xiao J., editor. Physical Exercise for Human Health. Volume 1228. Springer Singapore; Singapore: 2020. pp. 123–136. Advances in Experimental Medicine and Biology. [DOI] [PubMed] [Google Scholar]
- 150.Benham J.L., Booth J.E., Corenblum B., Doucette S., Friedenreich C.M., Rabi D.M., Sigal R.J. Exercise Training and Reproductive Outcomes in Women with Polycystic Ovary Syndrome: A Pilot Randomized Controlled Trial. Clin. Endocrinol. 2021;95:332–343. doi: 10.1111/cen.14452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Vigorito C., Giallauria F., Palomba S., Cascella T., Manguso F., Lucci R., De Lorenzo A., Tafuri D., Lombardi G., Colao A., et al. Beneficial Effects of a Three-Month Structured Exercise Training Program on Cardiopulmonary Functional Capacity in Young Women with Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2007;92:1379–1384. doi: 10.1210/jc.2006-2794. [DOI] [PubMed] [Google Scholar]
- 152.Moro C., Pasarica M., Elkind-Hirsch K., Redman L.M. Aerobic Exercise Training Improves Atrial Natriuretic Peptide and Catecholamine-Mediated Lipolysis in Obese Women with Polycystic Ovary Syndrome. J. Clin. Endocrinol. Metab. 2009;94:2579–2586. doi: 10.1210/jc.2009-0051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Lionett S., Kiel I.A., Camera D.M., Vanky E., Parr E.B., Lydersen S., Hawley J.A., Moholdt T. Circulating and Adipose Tissue MiRNAs in Women With Polycystic Ovary Syndrome and Responses to High-Intensity Interval Training. Front. Physiol. 2020;11:904. doi: 10.3389/fphys.2020.00904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Visser M., Bouter L.M., McQuillan G.M., Wener M.H., Harris T.B. Elevated C-Reactive Protein Levels in Overweight and Obese Adults. JAMA. 1999;282:2131–2135. doi: 10.1001/jama.282.22.2131. [DOI] [PubMed] [Google Scholar]
- 155.Covington J.D., Tam C.S., Pasarica M., Redman L.M. Higher Circulating Leukocytes in Women with PCOS Is Reversed by Aerobic Exercise. Biochimie. 2016;124:27–33. doi: 10.1016/j.biochi.2014.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Li N., Yang C., Xie H., Liu Y., Liao Y. Effects of Aerobic Exercise on Rats with Hyperandrogenic Polycystic Ovarian Syndrome. Int. J. Endocrinol. 2021;2021:1–12. doi: 10.1155/2021/5561980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Ribeiro V.B., Pedroso D.C.C., Kogure G.S., Lopes I.P., Santana B.A., Dutra de Souza H.C., Ferriani R.A., Calado R.T., Furtado C.L.M., Reis R.M.D. Short-Term Aerobic Exercise Did Not Change Telomere Length While It Reduced Testosterone Levels and Obesity Indexes in PCOS: A Randomized Controlled Clinical Trial Study. Int. J. Environ. Res. Public Health. 2021;18:11274. doi: 10.3390/ijerph182111274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Shele G., Genkil J., Speelman D. A Systematic Review of the Effects of Exercise on Hormones in Women with Polycystic Ovary Syndrome. JFMK. 2020;5:35. doi: 10.3390/jfmk5020035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Ribeiro V.B., Kogure G.S., Reis R.M., Gastaldi A.C., DE Araújo J.E., Mazon J.H., Borghi A., Souza H.C.D. Polycystic Ovary Syndrome Presents Higher Sympathetic Cardiac Autonomic Modulation That Is Not Altered by Strength Training. Int. J. Exerc. Sci. 2016;9:554–566. doi: 10.70252/DGCJ5295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Zhang J., Bao Y., Zhou X., Zheng L. Polycystic Ovary Syndrome and Mitochondrial Dysfunction. Reprod. Biol. Endocrinol. 2019;17:67. doi: 10.1186/s12958-019-0509-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Malamouli M., Levinger I., McAinch A.J., Trewin A.J., Rodgers R.J., Moreno-Asso A. The Mitochondrial Profile in Women with Polycystic Ovary Syndrome: Impact of Exercise. J. Mol. Endocrinol. 2022;68:R11–R23. doi: 10.1530/JME-21-0177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Scarpulla R.C. Metabolic Control of Mitochondrial Biogenesis through the PGC-1 Family Regulatory Network. Biochim. Biophys. Acta BBA Mol. Cell Res. 2011;1813:1269–1278. doi: 10.1016/j.bbamcr.2010.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Wu X., Wu H., Sun W., Wang C. Improvement of Anti-Müllerian Hormone and Oxidative Stress through Regular Exercise in Chinese Women with Polycystic Ovary Syndrome. Hormones. 2021;20:339–345. doi: 10.1007/s42000-020-00233-7. [DOI] [PubMed] [Google Scholar]
- 164.Polyzos S.A., Anastasilakis A.D., Efstathiadou Z.A., Makras P., Perakakis N., Kountouras J., Mantzoros C.S. Irisin in Metabolic Diseases. Endocrine. 2018;59:260–274. doi: 10.1007/s12020-017-1476-1. [DOI] [PubMed] [Google Scholar]
- 165.Perakakis N., Triantafyllou G.A., Fernández-Real J.M., Huh J.Y., Park K.H., Seufert J., Mantzoros C.S. Physiology and Role of Irisin in Glucose Homeostasis. Nat. Rev. Endocrinol. 2017;13:324–337. doi: 10.1038/nrendo.2016.221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Pesce M., Ballerini P., Paolucci T., Puca I., Farzaei M.H., Patruno A. Irisin and Autophagy: First Update. IJMS. 2020;21:7587. doi: 10.3390/ijms21207587. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.