Abstract
Glioblastoma is a common primary brain tumor that has a high mortality rate. Reports of intrathoracic metastases are uncommon, with the most commonly reported site for metastases are the lung and pleura. However, involvement of the mediastinum is not well documented, and few reports of confirmed mediastinal metastases diagnosed by endobronchial ultrasound-transbronchial needle aspiration (EBUS-TBNA) exist. Herein, we report a rare case of metastatic glioblastoma to the thorax. A lady in her 40s has been previously diagnosed with intracranial glioblastoma with multiple incidences of disease recurrence despite treatment with chemoradiotherapy, adjuvant chemotherapy, and repeated surgical resections. She presented with dyspnea and pleural effusion, for which radiological imaging revealed lung, pleural, and mediastinal lesions. Further diagnostic workup with EBUS and pleural fluid sampling confirmed metastatic disease to both sites. The pleural fluid showed highly atypical cells positive for GFAP, and EBUS-TBNA immunostains were GFAP, S100, and synaptophysin positive, giving an overall picture consistent with metastatic glioblastoma. The patient was referred for palliative care, and unfortunately, she passed away after several months.
1. Introduction
Glioblastoma is a known highly aggressive primary brain tumor. It is the most common primary malignant brain tumor reported in the United States in 2013-2017, constituting 14.5% of all brain tumors, and it carries a very poor prognosis with a 5-year mortality rate of 5.8% [1]. Similarly, in Saudi Arabia Eastern Province, one study reported glioblastoma as the most common pathological type between 2010 and 2015 in all age groups, representing 32% of all neuroepithelial brain tumors [2]. In a 10-year single-center study at King Fahad Medical City (KFMC) in the central region of Saudi Arabia, glioblastoma was similarly reported as the most common primary malignant tumor, constituting 25.6% of all primary brain tumors in the adult population (>18 years) [3]. The median overall survival (OS) for patients treated with a combined modality was 19.7 months [3]. Another retrospective analysis from the same center in Saudi Arabia found that the 6-month progression-free survival (PFS) rate was 43% for all patients and 55% for the combined modality group. As for median OS, it was 13.7 months [4].
Extraneural metastatic disease of glioblastomas is uncommon, with a reported incidence of 0.4-0.5% of the cases [5]. The frequency of incidence reported for extracranial metastatic (ECM) disease has increased over the last decades [6]. The site of metastasis of glioblastoma impacted survival prognosis, and lung metastases were reported to have the worst prognosis [5]. The most commonly reported initial metastases sites are the lung and pleura, followed by the lymph nodes (LNs) [7]. In a more recent meta-analysis, bone metastasis was most common (24.3%), followed by lung and LN metastases, 21.7% and 12.2%, respectively [8]. LN metastasis is more frequently reported to involve the cervical area (62%), followed by mediastinal or hilar LN (32%) [7].
Endobronchial ultrasound-transbronchial needle aspiration (EBUS-TBNA) is a minimally invasive diagnostic modality, most utilized in mediastinal staging and lung cancer diagnosis. However, it can be used as a first-line diagnostic modality in diagnosing mediastinal lymphadenopathy of unknown etiology. Here, we report one case of metastatic, recurrent glioblastoma, diagnosed by LN sampling of the mediastinum using EBUS-TBNA.
2. Case Report
Our patient is a 43-year-old lady diagnosed with a right parasagittal brain glioblastoma five years ago. The glioblastoma was WHO (World Health Organization) grade 4 with an unmethylated MGMT (methylguanine-DNA methyltransferase) mutation. She underwent gross resection in July 2016 and received concurrent chemoradiotherapy, which was completed in October 2016. In addition, she received adjuvant TMZ (temozolomide) and high-dose atorvastatin as per KFMC ART study protocol, completed in March 2017 [9]. Unfortunately, her glioblastoma recurred in February of 2018, for which she underwent reresection and received bevacizumab for five months. Despite these interventions, she developed a third recurrence and required resection in November of 2018, and received bevacizumab and irinotecan for six months. In September 2019, she had a fourth recurrence, which was resected, but she had rapid disease progression and was treated with surgical resection and 35 Gy in 10 fractions of radiation therapy. In December 2019, CT scans showed a fifth recurrence of her disease, which was resected again and treated with reirradiation 35 Gy in 10 fractions until February 2020, when her disease progressed again, and she received bevacizumab and lomustine until August 2020. However, her disease progressed again. She had another surgical resection in September 2020 with reirradiation with 35 Gy in 10 fractions in November 2020. She was maintained on bevacizumab and dose dense temozolomide until May 2021, when she complained of shortness of breath.
The patient was referred to our emergency room for admission. She gave a two-week history of shortness of breath associated with productive cough and right-sided pleuritic chest pain. Before coming to the clinic with her new symptoms, she sought medical advice in a nearby clinic and was given a course of antibiotics with no significant improvement in her condition. Her symptoms progressed to the extent that she had severe dyspnea at rest, could not lie flat, and her cough was worse with blood-tinged sputum. She lost her appetite and had a 6-kilogram unintentional weight loss over two months. She did not have any history of fever or exposure history of note. Subsequently, she presented to our emergency department and was found tachypneic, and her other hemodynamics were normal. The patient looked fatigued, and her physical examination was significant for decreased air entry over the right side on chest auscultation with a dull percussion note in the right lower zone.
Her laboratory investigations showed a WBC (white cell count) of 5.7 × 103/μL and two negative COVID-PCR swabs. Her chest computed tomography showed multiple enlarged necrotic mediastinal and bilateral hilar LN, the largest measuring 4.1 × 2.4 cm. She had a moderate right-sided pleural effusion with multiple bilateral pulmonary nodules, with the largest one in the left lower lobe, measuring 2.4 cm. Multiple lytic lesions in the dorsal vertebrae and a sclerotic focus in the manubrium sternum were identified (Figures 1 and 2).
Figure 1.
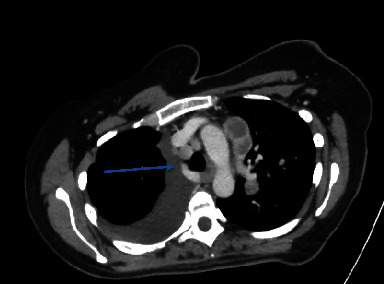
CT chest showing involvement of the right lower paratracheal lymphnodes (Station 4R).
Figure 2.
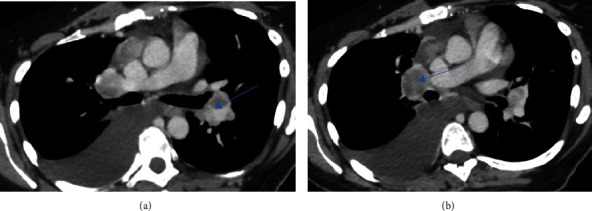
CT chest. (a) Confluent prevascular nodal mass measuring 4.1 × 2.4 cm. (b) Right hilar lymph node measuring 2.5 × 3.1 cm.
Bedside thoracentesis was done, draining around 1.2 L of clear yellow fluid initially. The pleural fluid cell count showed 723 × 103/UL WBC with a lymphocytic predominance (78% lymphocytes). Gram stain showed no acid-fast bacilli, and the polymerase chain reaction for mycobacterium DNA was not detected. Pleural fluid cytology showed highly atypical cells, and after sometime, GFAP (glial fibrillary acidic protein) immune staining was positive in some atypical cells. After pleural fluid drainage, the patient's symptoms improved, and she was able to lie flat.
The patient underwent EBUS-TBNA the following day as this case was time-sensitive and to confirm wether she has LN metastasis (which is very rare as mentioned) vs. another primary malignancy or other cause. Five and six needle passes were performed from the right lower paratracheal and right interlobar LNs, respectively. The rapid onsite examination from both stations was consistent with adequate lymph node tissue aquisition and highly suspicious for malignancy, and the samples were sent for further pathological examination and cultures. Cytological smears and cellblock demonstrate clusters of hyperchromatic neoplastic cells with small nucleoli and oval to elongated nuclei in a necrotic background. Cytoplasmic extensions were appreciated in some areas (Figure 3). The tumor cells were negative for broad-spectrum epithelial markers (a panel of keratins was employed). All site-specific epithelial markers were negative. Tumor cells, however, were immunoreactive for GFAP, synaptophysin, and S100 protein (Figure 4). Due to the patient's prior history and immunohistochemical results, the findings were considered consistent with metastatis from the patient's known glioblastoma.
Figure 3.
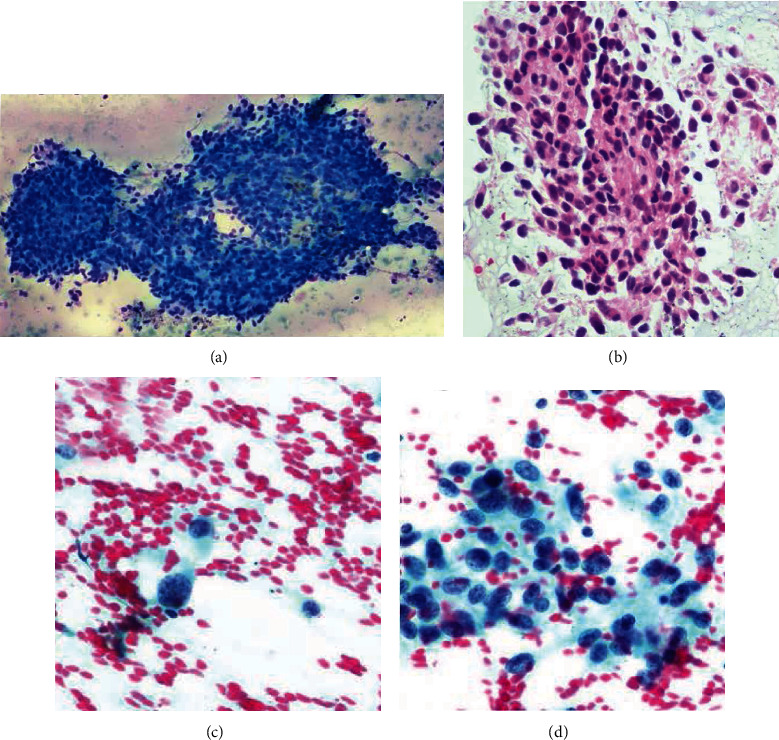
Diff-Quik smear shows clusters of cohesive neoplastic, epithelioid cells with a small amount of cytoplasm (a). The alcoholol-fixed smears demonstrate individual and clustered neoplastic cells with coarse chromatin, ill-defined borders, and nuclear grooves (c, d). The corresponding cellblock material is shown in (b).
Figure 4.
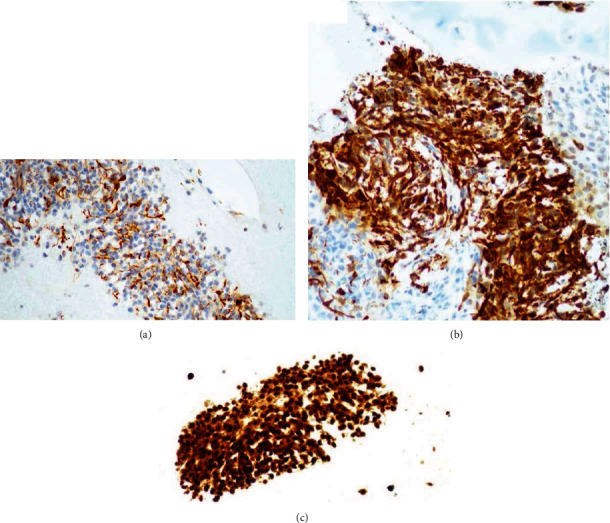
GFAP is immunoreactive in the tumor cells (a). (b) Strong positivity for synaptophysin. S100 protein immunostain demonstrates strong and diffuse positivity (c). Keratin is negative (not shown).
A trial of carboplatin/etoposide was entertained, and the patient received 2 cycles before her extraneural disease progressed. Next-generation sequencing (NGS) was performed on the tissue samples, but no actionable mutations were found. Her performance status declined, and she was transferred under the care of the palliative team in July 2021. Unfortunately, she passed away after several months. Treatment timeline is shown in Figure 5.
Figure 5.
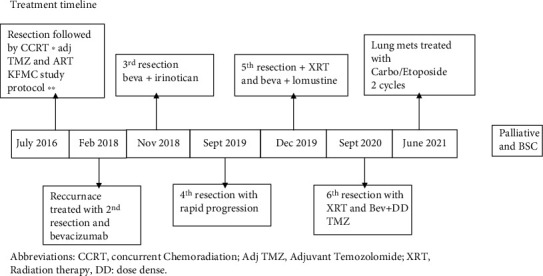
Treatment timeline. Abbreviations: CCRT: concurrent chemoradiation; Adj TMZ: adjuvant temozolomide; XRT: radiation therapy; DD: dose dense.
3. Discussion
Glioblastoma metastatic disease is known to occur mainly through blood and cerebrospinal fluid (CSF) [10]. Another important metastatic route is the lymphatic system; it was initially thought that the cerebrum was devoid of a lymphatic system and spread to LNs occurs through the cervical lymphatic plexus and the cranial perineural spaces [7]. However, new evidence reports the existence of a functioning lymphatic drainage system [11]. Surgical interventions such as shunt placement have been reported to cause metastases [7, 12]. Metastatic glioblastoma generally occurs after craniotomy, but it has been reported in cases without surgical intervention [7, 13]. The higher incidence following surgery is thought to be coincidental rather than metastatogenic [14].
The diagnostic modalities of metastatic intrathoracic glioblastoma depend on the pattern of disease involvement. Intrathoracic involvement can present as lung nodules, lung masses, pleural effusion, mediastinal masses, and mediastinal lymphadenopathy [15–21]. Similar to our case, the utilization of the cell block technique of pleural fluid in the diagnosis of ECM Glioblastoma was previously reported [22]. Bronchoscopic biopsy has also been previously utilized in the diagnosis of metastatic glioblastoma [20, 23, 24]. Endobronchial fine-needle aspiration (FNA) biopsy has been reported in the diagnosis of glioblastoma from a metastatic lung mass [25]. For suspected mediastinal involvement, endoscopic ultrasonography-guided fine-needle aspiration (EUS-FNA) was reported diagnostic of gliosarcoma, a variant of glioblastoma, from a posterior mediastinal mass [26]. In one study of a metastatic glioblastoma to the lung with mediastinal lymphadenopathy, EBUS-TBNA was employed in the sampling of a subcarinal LN, which was nondiagnostic; however, although the patient underwent mediastinoscopy, it was unclear if the sampled LN was positive for glioblastoma (i.e., false-negative TBNA) [27]. The diagnosis by FNA sampling of cervical LN was previously reported [28–33]. To our knowledge, no reports of EBUS-TBNA diagnostic for glioblastoma from mediastinal LN exist.
In conclusion, ECM of glioblastoma incidence is increasing. Minimally invasive, cost-effective diagnostic modalities of FNA for suspected ECM of glioblastoma have been utilized. This case showed that EBUS-TBNA could rapidly and safely diagnose rare ECM glioblastoma mediastinal LN metastases.
Abbreviations
- EBUS-TBNA:
Endobronchial ultrasound-transbronchial needle aspiration
- KFMC:
King Fahad Medical City
- LN:
Lymph nodes
- OS:
Overall survival
- ECM:
Extracranial metastases
- WBC:
White blood cell
- TMZ:
Temozolomide
- GFAP:
Glial fibrillary acidic protein
- FNA:
Fine-needle aspiration
- CCRT:
Concurrent chemoradiation
- Adj TMZ:
Adjuvant temozolomide
- XRT:
Radiation therapy
- DD:
Dose dense
Data Availability
No data were used to support this study.
Conflicts of Interest
The authors declare that they have no conflict of interest.
References
- 1.Ostrom Q. T., Patil N., Cioffi G., Waite K., Kruchko C., Barnholtz-Sloan J. S. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2013-2017. Neuro-Oncology . 2020;22(Supplement_1):iv1–iv96. doi: 10.1093/neuonc/noaa200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Taha M. S., Almsned F. M., Hassen M. A., et al. Demographic and histopathological patterns of neuro-epithelial brain tumors in Eastern Province of Saudi Arabia. Neurosciences . 2018;23(1):18–22. doi: 10.17712/nsj.2018.1.20160543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Almutrafi A., Bashawry Y., Alshakweer W., Al-Harbi M., Altwairgi A., Al-Dandan S. The epidemiology of primary central nervous system tumors at the National Neurologic Institute in Saudi Arabia: a ten-year single-institution study. Journal of Cancer Epidemiology . 2020;2020:9. doi: 10.1155/2020/1429615.1429615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Altwairgi A. K., Algareeb W., Yahya G., et al. Outcome of patients with glioblastoma in Saudi Arabia: single center experience. Molecular and Clinical Oncology . 2016;4(5):756–762. doi: 10.3892/mco.2016.818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lun M., Lok E., Gautam S., Wu E., Wong E. T. The natural history of extracranial metastasis from glioblastoma multiforme. Journal of neuro-oncology . 2011;105(2):261–273. doi: 10.1007/s11060-011-0575-8. [DOI] [PubMed] [Google Scholar]
- 6.Pietschmann S., von Bueren A. O., Kerber M. J., Baumert B. G., Kortmann R. D., Müller K. An individual patient data meta-analysis on characteristics, treatments and outcomes of glioblastoma/gliosarcoma patients with metastases outside of the central nervous system. PLoS One . 2015;10(4):p. e0121592. doi: 10.1371/journal.pone.0121592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Pasquier B., Pasquier D., N'golet A., Panh M. H., Couderc P. Extraneural metastases of astrocytomas and glioblastomas clinicopathological study of two cases and review of literature. Cancer . 1980;45(1):112–125. doi: 10.1002/1097-0142(19800101)45:1<112::AID-CNCR2820450121>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- 8.da Cunha M. L. V., Maldaun M. V. C. Metastasis from glioblastoma multiforme: a meta-analysis. Revista da Associacao Medica Brasileira . 2019;65(3):424–433. doi: 10.1590/1806-9282.65.3.424. [DOI] [PubMed] [Google Scholar]
- 9.Altwairgi A. K., Alghareeb W. A., AlNajjar F. H., et al. Atorvastatin in combination with radiotherapy and temozolomide for glioblastoma: a prospective phase II study. Investigational New Drugs . 2021;39(1):226–231. doi: 10.1007/s10637-020-00992-5. [DOI] [PubMed] [Google Scholar]
- 10.Urbanska K., Sokolowska J., Szmidt M., Sysa P. Glioblastoma multiforme - an overview. Wspolczesna Onkologia . 2014;18(5):307–312. doi: 10.5114/wo.2014.40559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sun B. L., Wang L. H., Yang T., et al. Lymphatic drainage system of the brain: a novel target for intervention of neurological diseases. Progress in Neurobiology . 2018;163-164:118–143. doi: 10.1016/j.pneurobio.2017.08.007. [DOI] [PubMed] [Google Scholar]
- 12.Berger M. S., Baumeister B., Geyer J. R., Milstein J., Kanev P. M., LeRoux P. D. The risks of metastases from shunting in children with primary central nervous system tumors. Journal of Neurosurgery . 1991;74(6):872–877. doi: 10.3171/jns.1991.74.6.0872. [DOI] [PubMed] [Google Scholar]
- 13.Robert M. C., Wastie M. E. Glioblastoma multiforme: a rare manifestation of extensive liver and bone metastases. Biomedical imaging and intervention journal . 2008;4(1) doi: 10.2349/biij.4.1.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rosen J., Blau T., Grau S. J., Barbe M. T., Fink G. R., Galldiks N. Extracranial metastases of a cerebral glioblastoma: a case report and review of the literature. Case reports in oncology . 2018;11(2):591–600. doi: 10.1159/000492111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chretien F., Gray F., Funalot B., et al. Extracerebral metastases of a glioblastoma, in the absence of surgery. Archives D'anatomie et de Cytologie Pathologiques . 1995;43(5-6):342–349. [PubMed] [Google Scholar]
- 16.Swinnen J., Gelin G., Fransis S., Vandevenne J., van Cauter S. Glioblastoma with extracranial parotid, lymph node, and pulmonary metastases: a case report. Radiology Case Reports . 2019;14(11):1334–1347. doi: 10.1016/j.radcr.2019.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Blume C., von Lehe M., van Landeghem F., Greschus S., Boström J. Extracranial glioblastoma with synchronous metastases in the lung, pulmonary lymph nodes, vertebrae, cervical muscles and epidural space in a young patient - case report and review of literature. BMC research notes . 2013;6(1) doi: 10.1186/1756-0500-6-290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tuominen H., Lohi J., Maiche A., Törmänen J., Baumann P. Mediastinal metastasis of glioblastoma multiforme evolving from anaplastic astrocytoma. Journal of neuro-oncology . 2005;75(2):225–226. doi: 10.1007/s11060-005-3395-x. [DOI] [PubMed] [Google Scholar]
- 19.Armanios M. Y., Grossman S. A., Yang S. C., et al. Transmission of glioblastoma multiforme following bilateral lung transplantation from an affected donor: case study and review of the literature. Neuro-Oncology . 2004;6(3):259–263. doi: 10.1215/S1152851703000474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wu Y., Zhang G., Zhang J., Zhang L., Wu Z. Enhanced proliferation and migration capability of epidermal stem cells by PRP and PDGF stimulation. International Journal of Clinical and Experimental Pathology . 2017;10(8):8804–8812. [PMC free article] [PubMed] [Google Scholar]
- 21.Hamilton J. D., Rapp M., Schneiderhan T. M., et al. Glioblastoma multiforme metastasis outside the CNS: three case reports and possible mechanisms of escape. Journal of Clinical Oncology . 2014;32(22):e80–e84. doi: 10.1200/JCO.2013.48.7546. [DOI] [PubMed] [Google Scholar]
- 22.Hori Y. S., Fukuhara T., Aoi M., Oda K., Shinno Y. Extracranial glioblastoma diagnosed by examination of pleural effusion using the cell block technique: case report. Neurosurgical Focus . 2018;44(6):p. E8. doi: 10.3171/2017.8.FOCUS17403. [DOI] [PubMed] [Google Scholar]
- 23.Briones-Claudett K. H., Briones-Claudett M. H., Garcia F. V., et al. Early pulmonary metastasis after a surgical resection of glioblastoma multiforme. A case report. American Journal of Case Reports . 2020;21, article e922976 doi: 10.12659/AJCR.922976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chung Y. H., Wong S. L., Huang H. Y. Endobronchial metastasis of glioblastoma multiforme diagnosed by fiberoptic bronchoscopic biopsy. Journal of the Formosan Medical Association . 1999;98(2):133–135. [PubMed] [Google Scholar]
- 25.Chivukula M., Dincer H. E., Biller J. A., Krouwer H. G., Simon G., Shidham V. FNAB cytology of extracranial metastasis of glioblastoma multiforme may resemble a lung primary: a diagnostic pitfall. Cytojournal . 2005;2 doi: 10.1186/1742-6413-2-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lee J., Rodriguez F., Ali S. Z. Metastatic gliosarcoma: cytopathologic characteristics with histopathologic correlations. Acta Cytologica . 2016;60(5):490–494. doi: 10.1159/000448509. [DOI] [PubMed] [Google Scholar]
- 27.Chen H., Shah A. S., Girgis R. E., Grossman S. A. Transmission of glioblastoma multiforme after bilateral lung transplantation. Journal of Clinical Oncology . 2008;26(19) doi: 10.1007/978-1-59745-183-3. [DOI] [PubMed] [Google Scholar]
- 28.Gestrich C., Cowden D., Harbhajanka A. Cytomorphology of glioblastoma metastic to a cervical lymph node diagnosed by fine needle aspiration (FNA): a case report and review of literature. Diagnostic Cytopathology . 2020;48(6):567–570. doi: 10.1002/dc.24412. [DOI] [PubMed] [Google Scholar]
- 29.Romero-Rojas A. E., Diaz-Perez J. A., Amaro D., Lozano-Castillo A., Chinchilla-Olaya S. I. Glioblastoma metastasis to parotid gland and neck lymph nodes: fine-needle aspiration cytology with histopathologic correlation. Head and Neck Pathology . 2013;7(4):409–415. doi: 10.1007/s12105-013-0448-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhen L., Yufeng C., Zhenyu S., Lei X. Multiple extracranial metastases from secondary glioblastoma multiforme: a case report and review of the literature. Journal of Neuro-Oncology . 2010;97(3):451–457. doi: 10.1007/s11060-009-0044-9. [DOI] [PubMed] [Google Scholar]
- 31.Moon K. S., Jung S., Lee M. C., et al. Metastatic glioblastoma in cervical lymph node after repeated craniotomies: report of a case with diagnosis by fine needle aspiration. Journal of Korean Medical Science . 2004;19(6):911–914. doi: 10.3346/jkms.2004.19.6.911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cámpora R. G., Salaverri C. O., Ramirez F. V., Villadiego M. S., Davidson H. G. Metastatic glioblastoma multiforme in cervical lymph nodes: report of a case with diagnosis by fine needle aspiration. Acta Cytologica . 1993;37(6) [PubMed] [Google Scholar]
- 33.Al-Rikabi A. C., Al-Sohaibani M. O., Jamjoom A., Al-Rayess M. M. Metastatic deposits of a high-grade malignant glioma in cervical lymph nodes diagnosed by fine needle aspiration (FNA) cytology—case report and literature review. Cytopathology . 1997;8(6):421–427. doi: 10.1111/j.1365-2303.1997.tb00573.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data were used to support this study.


