Abstract
Respiratory tract infections (RTIs) are one of the most common reasons for seeking healthcare, but are amongst the most challenging diseases in terms of clinical decision-making. Proper and timely diagnosis is critical in order to optimise management and prevent further emergence of antimicrobial resistance by misuse or overuse of antibiotics. Diagnostic tools for RTIs include those involving syndromic and aetiological diagnosis: from clinical and radiological features to laboratory methods targeting both pathogen detection and host biomarkers, as well as their combinations in terms of clinical algorithms. They also include tools for predicting severity and monitoring treatment response. Unprecedented milestones have been achieved in the context of the COVID-19 pandemic, involving the most recent applications of diagnostic technologies both at genotypic and phenotypic level, which have changed paradigms in infectious respiratory diseases in terms of why, how and where diagnostics are performed. The aim of this review is to discuss advances in diagnostic tools that impact clinical decision-making, surveillance and follow-up of RTIs and tuberculosis. If properly harnessed, recent advances in diagnostic technologies, including omics and digital transformation, emerge as an unprecedented opportunity to tackle ongoing and future epidemics while handling antimicrobial resistance from a One Health perspective.
Short abstract
Unprecedented advances have been achieved by improving and applying the latest technologies to key aspects such as epidemiology, contact tracing and diagnostics of respiratory tract infections that need to be properly addressed for global improvements https://bit.ly/3xnnqqb
General introduction
Respiratory tract infections (RTI) are amongst the most common reasons for seeking healthcare. Despite important advances in the last two decades, clinical management is challenging at several levels. Many RTI are caused by viruses for which antibiotics are not effective and in many cases are self-limiting. Misuse and mismanagement of antibiotics is particularly relevant in RTIs and contribute to the emergence of antimicrobial resistance (AMR) while hindering treatment. The aim of the current review is to discuss advances in diagnostic tools that have an impact on clinical decision-making and follow-up.
Pneumonia is the leading cause of death among children under 5 years old and the fifth cause of death among adults over 69 [1]. By contrast, for decades, the leading cause of death from a single infectious pathogen was tuberculosis (TB) [2]. In 2020, COVID-19 overtook TB as the infectious disease causing the highest mortality; and although global TB control efforts were not on track, even before the advent of the pandemic, TB control strategies have been further impacted by it [3]. According to the World Health Organization (WHO) report in 2021 [4], reduced access to TB diagnosis and treatment in 2020 resulted in the first year-on-year increase (of 5.6%) since 2005: estimated numbers account 1.3 million TB deaths among HIV-negative people and an additional 214 000 among HIV-positive people, with the combined total back to the level of 2017.
Existing surveillance programmes regularly monitor respiratory syndromes with focus on influenza viruses [5] and very efficient initiatives provide a coordinated and agile research response to infectious disease outbreaks [6]. The usefulness of monitoring became evident with the reported clusters of patients with pneumonia of unknown cause in late December 2019 that led to the description of the novel human pathogen SARS-CoV-2 [7]. The initial predictions were that 60% of the global population could be infected [8]. During 2020 and 2021, we witnessed a pandemic with devastating effects on health and economies but simultaneously a historical game-changer. The SARS-CoV-2 is unquestionably a One Health disease and has highlighted the concept of syndemics: a synergistic effect of the pandemic overlapping with endemic diseases and contextual determinants of health such as cultural, socio-economic factors, climate and environment [9]. The influence of noncommunicable diseases is well known and the impact of smoking and air pollution is finally being highlighted [10]. New vulnerable populations are resulting from population ageing [11] and the increased use of immunosuppressive therapies for a wide range of medical conditions [12].
Why do we need diagnostics? The concept of diagnostic tools
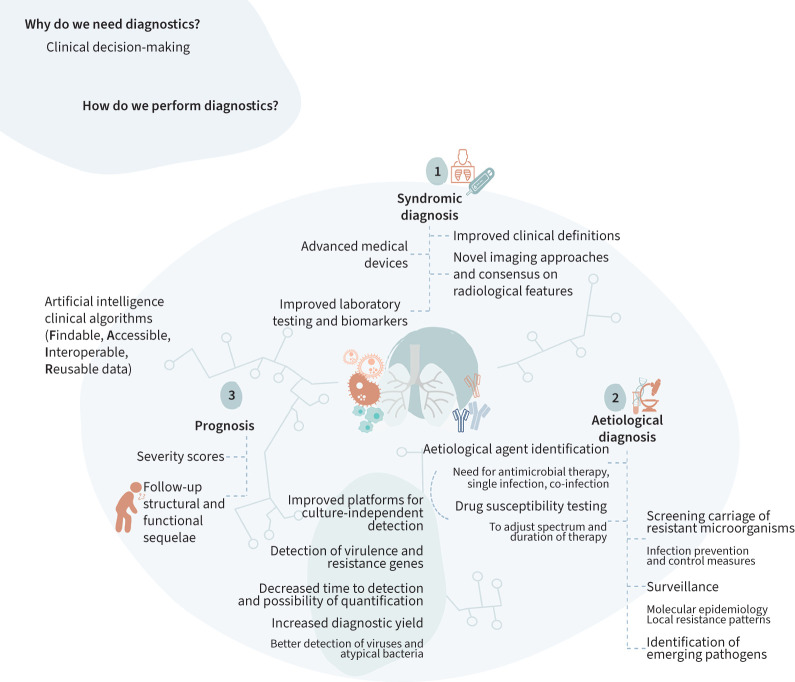
The concept of diagnostics refers to: 1) interpreting the clinical profile to predict the presence of the illness (syndromic diagnosis), 2) finding an explanation for the illness (aetiology) and 3) predicting the course of disease and need for follow-up (prognosis) (figure 1). These three above-mentioned aspects have been reflected in this review.
FIGURE 1.
Three pillars in clinical decision-making: syndromic diagnosis, aetiological diagnosis and prognosis assessment. The most relevant advances regarding methodologies available are presented.
Diagnostic tools must be accurate and need to be properly assessed and monitored, as emphasised by the introduction of the In Vitro Diagnostic Medical Devices Regulation (EU) 2017/746 (IVDR) [13]. Risks and benefits need to be quantified, based on scientific evidence and fit for use in the population for which they are intended (age, comorbidities, immune status). Finally, they need to be used at the right time and interpreted appropriately to affect care optimally.
Never before have we witnessed such social awareness of infectious disease; the importance of nonpharmacological measures, of a test result and of understanding the immunity behind it. COVID-19 provided an unprecedented response from industry during the massive testing and vaccination campaigns that ensued, with more than 1000 new tests, new diagnostic approaches and modification of existing techniques or the development of new ones [14]. While we must continue to monitor the virus closely, it is time to change the dynamics regarding the value of diagnostics of RTI globally. If this is properly addressed, an unprecedented opportunity emerges to face current and future epidemics.
Advances in syndromic diagnosis
One of the first challenges in RTIs is correcting frequently inconsistent case definitions. Clinical signs and symptoms are similar for many lower respiratory tract infections (LRTIs) and several other diseases (e.g. pulmonary embolism, acute heart failure, asthma). Global burden estimations require valid, reliable and timely data [1], so appropriate definitions are essential and important progress has recently been made [15]. Those aspects that have shown advances are highlighted below.
Pneumonia
Pneumonia is defined as an acute illness affecting the lungs, usually presenting with cough, sputum production, and rapid and difficult breathing with a new or worsening pulmonary infiltrate on a chest radiograph. The diagnostic processes range from simple to relatively complex procedures combining clinical, radiological and laboratory features. Specific investigations should be considered in some endemic settings (e.g. TB, mycoses) [16] and also in cases of pneumonia associated with air pollution [17]. Depending on the clinical setting and accessibility, the plain chest radiograph is not always required for diagnosis, nevertheless it still remains a relevant tool. Lung ultrasound has shown better accuracy than chest X-ray for bacterial pneumonia in emergency departments [18] and an advance during the pandemic has been its increased use for both diagnosis and follow-up [19]. The additional value of computed tomography has been widely recognised for COVID-19 pneumonia [20].
Tuberculosis
One of the main advances in definitions is the change from addressing two disease states (latent TB infection or active TB disease) to evaluating a continuous spectrum of metabolic bacterial activity and antagonistic immunological responses by adding two additional clinical states: incipient (asymptomatic phase of early disease, between latent infection and subclinical TB) and subclinical TB (described as having viable and detectable bacteria, but without TB-related symptoms) [21, 22]. Recent WHO guidelines endorse automated nucleic acid amplification tests for detection of TB and resistance to rifampicin and isoniazid, providing more options for early diagnosis of TB [23].
Bronchiolitis
Bronchiolitis is a disorder commonly caused by viral infections in infants, typically beginning with rhinitis and cough, which may progress to tachypnoea, wheezing, rales, use of accessory muscles and/or nasal flaring [24]. Many respiratory viruses cause a similar constellation of signs and symptoms. The most common aetiology is respiratory syncytial virus (RSV), which infects 90% of children in the first 2 years of life, with up to 40% experiencing LRTIs during the initial infection [25]. Recent advances include the definition of different endotypes and the identification of relevant risk factors for recurrent wheezing and asthma development [26].
Bronchiectasis
Bronchiectasis is characterised by a permanent and progressive dilation of the airways because of a vicious cycle of inflammation, infection and repair of the bronchial mucosa, which leads to malfunctioning of the mucociliary system and destruction of the bronchial wall [27]. Awareness of bronchiectasis has increased considerably in recent years [28, 29] and advances include a clearer definition of the disease [30] as well as its exacerbation. Now acute deterioration in three or more of the following symptoms for at least 48 h: cough, sputum volume and/or consistency, sputum purulence, dyspnoea and/or exercise tolerance, fatigue and/or malaise, or haemoptysis, lead clinicians to make changes in bronchiectasis treatment [31].
COPD exacerbation
The definition of COPD exacerbation has also improved by highlighting a spectrum of different exacerbation types that can require different interventions. Identifying exacerbation triggers is of the utmost importance for proper treatment. Best known are viral and bacterial RTIs with a significant proportion of co-infections. However, in one-third of cases, they remain unidentified [32].
Treatable traits
Recently, a novel treatment approach based on “treatable traits” recognises that airway infection is only one of many treatable traits in a given patient that benefit from specific management, such as airway clearance techniques, prompt treatment of exacerbations or oral or inhaled antibiotics. This approach makes it possible to differentiate those individuals in whom symptoms or exacerbations are driven by a different treatable trait that will not benefit from antibiotic treatment [33–35].
Chronic bronchial infection
When potentially pathogenic microorganisms are present over time, a chronic bronchial infection occurs. Chronic bacterial infection can be clinically defined as evidence of positive respiratory tract cultures of the same microorganism by standard microbiology, on two or more occasions, at least 3 months apart over 1 year, while in a stable state [30]. Several novel techniques, such as automated molecular diagnostic systems, might be incorporated into the diagnostic scheme for exacerbations following careful interpretation. Both acute and chronic respiratory infection cause a progression of the disease and a worsening quality of life [29]. This is a very common situation in diseases such as COPD and bronchiectasis, and less frequently it also occurs in asthma. Chronic airway infection has been associated with altered pulmonary immune response and worse clinical outcomes [36, 37].
Spectrum of disease
A major challenge in each of these clinical conditions is correlating syndromic and aetiological diagnosis leading to the concept of spectrum of disease. Diagnosis is straightforward if a primary pathogen such as Mycobacterium tuberculosis, Legionella pneumophila or Pneumocystis jirovecii is identified. In other situations, more careful interpretation is needed such as when identifying viruses (infection or asymptomatic shedding), environmental fungus or environmental mycobacteria (infection, colonisation or contamination). The most frequently isolated bacteria such as Streptococcus pneumoniae, Haemophilus influenzae or Moraxella catarrhalis are potentially commensal. This is also the case for Staphylococcus aureus and Pseudomonas aeruginosa and other nonfermenting bacilli (e.g. Achromobacter xylosoxidans, Stenotrophomonas maltophilia) when underlying disease is present. Nonprimary pathogens might gain access and trigger symptoms or disease (e.g. by activating virulence factors) or remain quiescent (biofilm, intracellular survival), so better understanding of host–pathogen interaction is crucial [38]. Improving microbiological methods and the increased number of vulnerable patients has added Nocardia spp., fungi and environmental mycobacteria to the spectrum of potential pathogens. Recent advances include guidelines to assess definitions of disease stages for environmental mycobacteria [39–41] and also for pulmonary aspergillosis, depending on the interaction between Aspergillus and the host [27].
Advances in diagnostic tools identifying the aetiological agent
Microbiological diagnostics are critical at three levels: 1) identifying the aetiological agent and guiding appropriate therapy, so adjusting spectrum and duration and decreasing misuse and overuse of antibiotics to prevent the emergence of AMR; 2) surveillance of local resistance patterns and screening to identify patients colonised with resistant pathogens and adoption of infection prevention and control measures to prevent spread; and 3) detecting emerging pathogens. An important concept is that detection might refer to screening (e.g. detecting carriers of the resistant pathogen) or to aetiological diagnosis. Independent of the sophistication of the methods, it is crucial to generate timely, understandable results that can inform clinical decisions.
A diagnostic test can be used to demonstrate the presence or absence of infection or to detect evidence of a previous one. Generally speaking, it can be categorised into direct diagnosis, including microscopic examination, culture, antigen detection and molecular detection; and indirect diagnosis, covering immunological tests. The pandemic has brought advances in every single technique [42].
Appropriate samples and methodology
Accurate microbiological diagnosis of RTIs requires good-quality specimens. Samples must be collected taking medical conditions into account, either from the upper (nasal/throat specimens) or lower respiratory tract (bronchoalveolar lavage fluid, tracheal aspirate). Additionally, improved detection methods are making the use of new types of specimens possible and even saliva and oral mucosa may have a role to play in some situations [43–46]. But this is not true for all microorganisms; it depends on the pathogenesis and whether they are primary pathogens or potentially commensal. Particularly for COVID-19 diagnosis, pre-analytical factors have been particularly relevant, such as the swabbing methods and the use of different matrices and viral transport media [47, 48]. Lastly, reliable transportation in optimal conditions (temperature, time) to the laboratory are key to ensuring valid results.
Pathogen identification
For culture-based methods, the arrival of matrix-assisted laser desorption/ionisation-time of flight (MALDI-TOF) mass spectrometry in clinical labs has been a major development in rapid identification, with the additional potential for detection of AMR and even typing, among other possible/putative/future uses. In culture-independent detection, recently antigen detection has increased its identifiable targets. Additionally, and of particular note, molecular methods have increased the diagnostic yield for virus and atypical bacteria, showing unprecedented development during the COVID-19 pandemic. Molecular techniques are also becoming available at point-of-care (POC) or near-POC as different portable molecular diagnostic instruments are being developed [49]. These syndromic multiplex panels can be used to detect the pathogen DNA or RNA most commonly associated with RTIs, including viruses and bacterial atypical pathogens [50–54]. Advantages include decreased time to detection, the possibility of quantification and detection of resistance and virulence genes. Serological diagnosis of atypical pneumonia has been replaced by molecular tests and is now only used in cases such as Q fever. Antibody detection has seen a comeback, mostly for immunoprevalence and vaccine response studies of COVID-19 (enzyme-linked immunosorbent/chemiluminescence/fluorescence microparticle and lateral flow immunoassays). The pandemic has also enabled the development of advanced sensing technologies based on microfluidics, nanotechnology and material science [55] as well as on targeting structures such as serum extracellular vesicles [56]. The main benefits of these assays are that they are portable, miniaturised, low cost and highly integrated POC devices [57]. In the move to a new paradigm of personalised medicine, POC diagnostic testing has been proposed to improve the quality of antibiotic prescription [58] but accuracy is variable and settings need careful adjustment [18].
Susceptibility testing
Advances involving the use of selective chromogenic media shorten the detection time for resistant bacteria. The gold standard is phenotypic testing because it shows the overall phenotype, although multiple resistance mechanisms can lead to difficult phenotype interpretations. The long time to result has been improved by selective culture media and analytical methods starting from a bacterial pellet, which is particularly useful in systemic infections [59] and also applicable to mass spectrometry or nanosensors [60]. Genotypic tests detect resistant genes in a fast and sensitive way, but genes may be present but not expressed, and the tests only detect known resistance genes. As a pragmatic summary, phenotypic testing can tell what antibiotics to prescribe, while genotypic testing can tell what not to prescribe.
Molecular epidemiology
Available typing methods for respiratory pathogens are laborious and time-consuming, but in recent years whole genome sequencing (WGS) has emerged as the gold standard method for detecting outbreaks and preventing clonal dissemination in medical settings. However, this is not so easily implemented in real-time practice. In this sense, the current COVID-19 pandemic has seen unprecedented generation and global sharing of large numbers of SARS-CoV-2 sequences in a record time, with global sharing of variants of concern in common databases, powering influenza surveillance programmes into COVID-19 efforts [61]. The next step involves real-time molecular epidemiology [62]. Genomics have expanded rapidly, bringing the opportunity to move these approaches from academic to public health decisions and surveillance, as well as strengthening global cooperation with other disease control programmes [63]. Phenotypical typing methods are also improving, such as Fourier transform infrared spectroscopy [64].
Extended culture techniques, automated molecular diagnostic systems and DNA sequencing technologies have revealed unknown airway microbiota, including an abundance of species that are refractory to common diagnostic tools. The interactions that occur between these microbial species can profoundly affect the expression of pathogenicity and virulence [27]. Large numbers of microorganisms, including bacteria, fungi and viruses, collectively known as the microbiome, coexist in the lungs of healthy subjects and patients with respiratory diseases [65].
The microbiome
The microbiome is defined as the set of the genes and gene products (RNA, proteins, metabolites) produced by resident microbial communities. Research into the chronic lung disease microbiome began in the early 2000s, when ecological techniques of microbial DNA analysis were applied to sputum samples [66, 67]. Since then, the number of studies of the respiratory microbiome has grown exponentially. Whilst traditionally bacterial infection in the airways has been characterised using culture-based methods, 16S ribosomal RNA and other metagenomic approaches provide a powerful method of determining microbial identities and relative abundances [68].
The impact of omics technologies on precision approaches
General concepts of omics
There is growing clinical interest in understanding biological mechanisms beyond the molecular level to include biological functionality. In this sense, systems biology aims to explore how the interactions between biological components (genes, proteins, metabolites, etc.) contained in a biological tissue, cell, fluid or a whole organism affect its functionality (biological processes) as a whole, thus making it possible to characterise a biological system in a complete and integrated way [69]. Omics cover the set of high-throughput technologies that provide a global vision of a dynamic biological process through the analysis of genes (genomic), ribonucleic acid (RNA) (transcriptomic), proteins (proteomic) or metabolites (metabolomic).
Metagenomics
The first sequencing technique used in respiratory microbiome research was 16S rRNA gene sequencing. It is based on PCR amplification using primers that target the 16S ribosomal gene in variable regions of bacterial genomes, which can be used for taxonomic classification [70]. It is fast, reasonably low cost, and by converging on a specific region of the bacterial genome requires only a limited sequencing depth and allows the study of microbial communities [71]. However, it targets relatively short gene sequences, which are often shared by different closely related species, making distinguishing them a challenge [72]. Another shortcoming is that 16S sequencing analyses are restricted to the detection of bacteria and Archaea because viruses or fungi do not carry 16S rRNA genes. In this sense, shotgun metagenomic sequencing is an approach that allows microbiome characterisation with a much greater resolution than 16S sequencing. The term “shotgun” refers to the untargeted sequencing of all DNA present, in contrast to the targeted amplicon-based approaches. By not limiting sequencing to a single region of DNA, metagenomic sequencing can also provide information on the functional characteristics of the taxa present, including their metabolic traits, and their carriage of antibiotic resistance and pathogenicity features [73]. Furthermore, the inclusive nature of metagenomic sequencing provides information not only on the bacteria present, but also the fungi and DNA viruses, achieving identification to a subspecies or strain level. The principal limitation of shotgun metagenomic approaches is related to costs, but also to host DNA contamination. Finally, current methods cannot differentiate between live and dead microbes, which may be relevant for several reasons, such as assessing the impact of antibiotic therapy in clinical samples [74]. Novel methodologies to integrate bacterial, viral and fungal communities to allow assessment of the “interactome” have been recently developed. Rather than focusing on individual taxa, this approach proposes a role for microbial networks in altering clinical outcomes or treatment responses [75].
Transcriptomics
Transcriptomics involves the analysis of RNA produced by a given genome at a given time and condition, and thus during LRTIs it can provide information regarding pathogen and host dynamics. It can refer to the exploratory analysis of an entire transcriptome, primarily using RNA sequencing, or to a targeted analysis of known RNAs using techniques such as gene expression panels.
Metabolomics
Metabolomics is one of the most powerful bioanalytical strategies to obtain a picture of the metabolites in the course of a biological process and is a phenotyping tool [76]. Metabolomics allows the comparison of a chemical fingerprint present in a cellular system or a biofluid under normal conditions with that of altered states produced by disease, pharmacological treatment, dietary intervention or environmental modulation [77].
Clinical impact of omics
There is enormous interest in the potential of the microbiome to improve the understanding and stratification of respiratory diseases and to serve as a biomarker for clinical management. Host–microbiome interactions probably contribute substantially to differences in clinical phenotypes and disease outcomes [68, 78, 79]. Recent studies conclude that the microbiome could identify subgroups of patients at higher risk of poor outcome, who could benefit from precision treatment strategies [79]. Important questions still need to be examined, including the role of fungi, viruses and mycobacteria, the interactions with the host, and the usefulness of microbiome profiles for selecting antibiotics and to evaluate therapeutic responses [80]. The gut–lung axis affects disease and treatment. The intestinal microbiome influences the pulmonary microbiome and also lung immune responses by directly seeding the airways with bacteria and distributing bacterial metabolites that act as immune modulators [81].
Regarding tuberculosis, advances in systems biology and omics strategies have identified sets of biomarkers with the potential to optimise TB prevention, diagnosis and treatment. Only a few have been evaluated in clinical trials, so applicability in TB management is still limited [82, 83]. Metabolomics has provided insight into the pathogenesis of TB related to detection of infection and disease progression [76, 84–87]. Urinary metabolomic response has also been characterised during community-acquired pneumonia, and different profiles have been identified according to the causative microorganism: atypical bacteria versus pneumococcal or viral [88]; ARDS versus influenza; pneumococcal versus other aetiologies [89]; and pneumococcal versus staphylococcal [90].
Advances in diagnostic tools characterising the host response
The identification of novel biomarkers based on host–pathogen interactions related to the shift from carriage to infection may improve RTI management. There is a need to understand how the pathogens interact with their host to achieve a successful invasion (figure 2). Inflammatory biomarkers and severity scores can contribute to some levels of stratification [38, 66].
FIGURE 2.

Omics for a better characterisation of host, pathogen and host–pathogen interaction factors during RTIs.
Systemic biomarkers, such as the C-reactive protein (CRP) and procalcitonin (PCT), have been widely analysed in patients with LRTIs, although other biomarkers are also being investigated [91, 92]. They are also being implemented at POC. The capacity of biomarkers to distinguish acute bacterial from viral RTIs has enabled their use as tools for guiding antimicrobial therapy [93], but in the outpatient setting they might have a limited capacity [94]. Regardless of whether they are used for diagnostic or monitoring purposes, biomarker analysis should be included as an additional criterion to be integrated into decision-making algorithms.
During the COVID-19 pandemic, besides the identification of the causative agent, the detection and monitoring of the host response has provided insights into the pathogenesis of the disease [95, 96]. In fact, immune tools measuring T-cell responses and detecting IFN-γ in vitro (IGRAs) have been used for some years to diagnose TB and nowadays they are under investigation for SARS-CoV-2. Furthermore, second-generation IGRAs that detect other cytokines, such as IP-10, have proved comparable to IFN-γ. Furthermore, IP-10 cytokine has been shown to be very stable when dried on filter paper, which facilitates sample shipment to reference laboratories [97–100].
Because disease outcome depends on dynamic host–pathogen interactions, specific host genetic signatures, together with pathogen genomics, can be combined in order to identify those individuals with a higher risk of severe disease [101, 102]. Specific host transcriptomic signatures have also been identified for this purpose [103–106]. An interesting additional area of host genomics research is its use during pre-symptomatic stages, which can be important in influenza [107, 108] or TB [109] for predicting disease progression or detecting early forms of the illness. Host determinants for outcome are also related to acquired risk factors. Biological therapies targeting cytokines and/or cell subsets have become essential for the treatment of several immune-mediated diseases and have an impact on RTIs, as reviewed elsewhere [12].
Advances in understanding host immunity have been unprecedented during the pandemic, providing better knowledge of the immune response for other endemic respiratory viruses. Multiple mechanisms might lead to humoral and cellular responses involved in combatting RTIs. While an enormous effort has been put into vaccine development and immunogen design, there are still some knowledge gaps in understanding long-term memory and protective immunity. Advances in methods to assess immunogenicity have been very relevant. Vaccines have been traditionally dependent on humoral response activation by means of B-cells producing neutralising antibodies. However, over the years it has been accepted that cellular immunity mediated by CD4+ and CD8+ T-cells is also critical, evidencing that even a strong antibody response is not sufficient for protection. Recently it has been recognised that local immunity to bacterial or viral RTIs can be mediated by specific cells with a memory phenotype, called tissue-resident memory T-cells (TRMs). These cells are noncirculating, present at the site of infection, and retained in tissues for mediating a protective response in case of reinfection. Studies in mouse models and humans have evidenced that TRMs have a potential role against several respiratory pathogens such as influenza, RSV, M. tuberculosis or SARS-CoV-2 virus [110–115]. It is therefore important to develop new approaches and perform immunological studies to characterise TRMs as a diagnostic tool to monitor new vaccines that enhance these cell populations. Finally, the pandemic accelerated novel vaccine formulations including new viral vectored or nucleic acid-based vaccines, as well as the re-emergence of the concept of trained immunity. Live vaccines such as BCG can confer nonspecific protection against upper and lower RTIs not associated with M. tuberculosis through epigenetic reprogramming during haematopoiesis of innate cells [116–121].
Finally, better understanding of host–pathogen interactions has allowed the identification of potential host targets and therefore the development of host-directed therapies. These methodologies consist of inhibiting host factors indispensable for microorganism replication, intensifying a pathway of the host immune response or decreasing the inflammatory status [122–125]. Metabolic, autophagy and immune response pathways are currently being investigated. Implementation is still a challenge, with a need for accurate patient stratification for tailored interventions that prevent major side effects. Several randomised clinical trials are ongoing, focusing on TB and COVID-19 (https://ClinicalTrials.gov).
Advances in diagnostic tools to predict severity
Clinical assessment of severity, related to involvement of lung parenchyma in the acute phase and prediction of functional sequelae, is relevant for clinical decisions. In radiology, current practices include tedious conventional processes, which rely on specialist technical expertise and are prone to human error. Great progress has been made in deep learning that supports medical radiologists [126–128]. Novel approaches such as radiomics, a high-throughput method extracting a tremendous amount of quantitative imaging data using data-characterisation algorithms, have shown great potential in characterising imaging biomarkers [129]. Also, radiomics-based machine learning signatures have shown the potential to accurately differentiate ground-glass opacities due to COVID-19 pneumonia from those due to other acute lung diseases [130] or distinction between lung adenocarcinoma and tuberculosis granuloma [131]. Another example would be the computer-aided design (CAD) procedure for automatic diagnosis of COVID-19 from chest X-ray images [132].
Several bronchoscopic technologies have emerged over recent years, including thin/ultrathin bronchoscopes, radial probe endobronchial ultrasound (RP-EBUS), virtual navigation bronchoscopy (VBN), electromagnetic navigation bronchoscopy (ENB) and robotic bronchoscopy [133]. Bronchoscopic transparenchymal nodule access (BTPNA) and transbronchial access tool (TBAT) are novel techniques that, combined with navigational bronchoscopic technologies, improve access to lung lesions. The introduction of cryobiopsy has improved tissue sampling. These innovative techniques allow higher diagnostic yield, also in the context of RTIs and TB and other mycobacterial infections during the study of lung nodules and staging of lung cancer [134].
Oxygen saturation monitoring has traditionally been carried out using transmittance pulse oximeters due to their dependability, but they are limited to peripheral regions. Recently, new options being studied include reflectance pulse oximeters that can be used at different body sites (finger, wrist, chest and forehead) and can be scaled down to affordable patches [135].
Regarding prognostic tools, diaphragm ultrasound can be used to diagnose diaphragm dysfunction, assess severity, and monitor disease progression, and could be beneficial both in pneumonia and COVID-19 [16, 136]. Advances have also been made regarding better diagnosis of long-term structural–functional complications and a better follow-up of sequelae by standardising prognostic measures such as quality of life and social impact [137–139].
Final remarks: lessons learned
The pandemic has brought the need for an integrated approach for handling infectious diseases back to the forefront, along with the need for a coordinated effort across multiple disciplines including human, animal and environmental [140]. Unprecedented milestones have been achieved by improving and applying the latest technologies in key areas such as epidemiology, contact tracing, diagnostics and vaccine development.
However, global health inequities in low-income countries related to vaccines and treatments, and also diagnostics, still need to be addressed. The pandemic has additional costs related to the previously identified global crisis in AMR and TB [141] and several aspects have been highlighted as opportunities at different levels [142, 143].
POC technologies have allowed community-based testing to be scaled up and used as a public health tool, but they need to be linked to careful evaluation [144] and to demonstrate their diagnostic accuracy in clinical practice [18]. Timely results also involve careful interpretation and this is facilitated by effective interdisciplinary communication, starting with understandable reporting of results.
While providing emergency healthcare, clinicians and scientists have carried out clinical research and lessons learned underline the importance of pre-established structures and procedures and the need for improved regulatory consensus and globally connected networks [145]. Cost-effectiveness analyses can be used to assess the value of diagnostics in clinical practice, but they need proper design and reporting [146] in the context of pragmatic clinical trials. Digital transformation of health is ongoing through the incorporation of artificial intelligence to support clinical decision processes [147].
After years of reporting clinical diagnosis and microbiologically confirmed diagnosis in different databases, priorities have been set up in data connectivity by maximising efforts in FAIR (Findable, Accessible, Interoperable, Reusable) approaches [148, 149]. Real-world data and real-world evidence are rapidly gaining importance and being formalised in policy frameworks [150].
Summary
Advances in diagnostic tools have been largely accelerated by the pandemic and affect both the design of personalised therapies and public health measures. Transdisciplinary communication is crucial for proper development and implementation of current techniques. Impact of the use of diagnostic tools depends on several contextual factors involving clinical setting, geographical location, connectivity and clinical research resources. It is mandatory to maintain monitoring of pathogenesis of individual RTIs for a timely diagnosis and proper interpretation.
Acknowledgements
The authors wish to thank Raquel Villar-Hernandez, who designed the figures, and Harvey Evans, who reviewed the English grammar.
Contributors: Antoni Rosell, Pneumology Dept, Hospital Universitari Germans Trias i Pujol, Institut d'Investigació Germans Trias i Pujol (IGTP), Universitat Autònoma de Barcelona, Ciber Enfermedades Respiratorias, Badalona, Spain; Carlos Rodrigo Gonzalo de Liria, Pediatrics Dept, Hospital Universitari Germans Trias i Pujol, IGTP, Universitat Autònoma de Barcelona, Badalona, Spain; Maria Méndez, Pediatrics Dept, Hospital Universitari Germans Trias i Pujol, IGTP, Universitat Autònoma de Barcelona, Badalona, Spain; and Raquel Villar-Hernandez, Microbiology, IGTP, Badalona, Spain.
Footnotes
Provenance: Commissioned article, peer reviewed.
Conflict of interest: C. Prat-Aymerich is an associate editor of this journal. The others authors have no conflict of interest, including specific financial interests or relationships or affiliations to the subject matter or materials discussed in the manuscript. All authors have seen and approved the final version of the manuscript; they have contributed significantly to the work and they have taken care to ensure the integrity of the work.
Support statement: No specific funding has been required but the research scope of this review was supported by 1) grants from the Instituto de Salud Carlos III (ISCIII) (PI18/00411, PI19/01408 and CP20/00070), integrated in the Plan Nacional de I+D+I and co-funded by the ISCIII Subdirección General de Evaluación and the Fondo Europeo de Desarrollo Regional; 2) a grant from the Sociedad Española de Neumología y Cirugía Torácica (project 25/2016); 3) from the European Union's Horizon 2020 Research and Innovation Programme under the Marie Skłodowska-Curie grant agreement number 823854 (INNOVA4TB); and 4) Innovative Medicines Initiative 2 Joint Undertaking under grant agreement No 820755 (Value-Dx). This Joint Undertaking receives support from the European Union's Horizon 2020 Research and Innovation Programme and EFPIA and bioMérieux SA, Janssen Pharmaceutica NV, Accelerate Diagnostics S.L., Abbott, Bio-Rad Laboratories, BD Switzerland Sàrl and The Wellcome Trust Limited.
References
- 1.GBD 2019 Diseases and Injuries Collaborators . Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020; 396: 1204–1222. doi: 10.1016/S0140-6736(20)30925-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.World Health Organization. Global Tuberculosis Report 2020 . WHO, Geneva, 2020. [Google Scholar]
- 3.Chakaya J, Khan M, Ntoumi F, et al. . Global Tuberculosis Report 2020 – Reflections on the Global TB burden, treatment and prevention efforts. Int J Infect Dis 2021; 113: Suppl. 1, S7–S12. doi: 10.1016/j.ijid.2021.02.107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.World Health Organization. Global Tuberculosis Report 2021 . WHO, Geneva, 2021. [Google Scholar]
- 5.Global Influenza Surveillance and Response System (GISRS) . World Health Organization. https://www.who.int/initiatives/global-influenza-surveillance-and-response-system/ Date last accessed: 5 July 2022.
- 6.International Severe Acute Respiratory and emerging Infection Consortium. https://isaric.org/ Date last accessed: 5 July 2022.
- 7.Zhu N, Zhang D, Wang W, et al. . A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med 2020; 382: 727–733. doi: 10.1056/NEJMoa2001017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). Geneva: World Health Organization, 2020. https://www.who.int/publications/i/item/report-of-the-who-china-joint-mission-on-coronavirus-disease-2019-(covid-19)/ Date last accessed: 5 July 2022.
- 9.Fronteira I, Sidat M, Magalhaes JP, et al. . The SARS-CoV-2 pandemic: a syndemic perspective. One Health 2021; 12: 100228. doi: 10.1016/j.onehlt.2021.100228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.World Health Organization. 2019. Noncommunicable diseases and air pollution. https://apps.who.int/iris/bitstream/handle/10665/346416/WHO-EURO-2019-3641-43400-60937-eng.pdf?sequence=1&isAllowed=y Date last accessed: 5 July 2022.
- 11.Byng-Maddick R, Noursadeghi M. Does tuberculosis threaten our ageing populations? BMC Infect Dis 2016; 16: 119. doi: 10.1186/s12879-016-1451-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lacoma A, Mateo L, Blanco I, et al. . Impact of host genetics and biological response modifiers on respiratory tract infections. Front Immunol 2019; 10: 1013. doi: 10.3389/fimmu.2019.01013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Regulation (EU) 2017/746 of the European Parliament and of the Council of 5 April 2017 on in vitro diagnostic medical devices and repealing Directive 98/79/EC and Commission Decision 2010/227/EU. https://op.europa.eu/s/waFx/ Date last accessed: 5 July 2022.
- 14.Chau CH, Strope JD, Figg WD. COVID-19 clinical diagnostics and testing technology. Pharmacotherapy 2020; 40: 857–868. doi: 10.1002/phar.2439 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.The World Health Organization Essential Medicines List Antibiotic Book. 2022. https://www.who.int/news-room/questions-and-answers/item/the-who-essential-medicines-list-antibiotic-book/ Date last accessed: 5 July 2022.
- 16.Torres A, Cilloniz C, Niederman MS, et al. . Pneumonia. Nat Rev Dis Primers 2021; 7: 25. doi: 10.1038/s41572-021-00259-0 [DOI] [PubMed] [Google Scholar]
- 17.Nhung NTT, Amini H, Schindler C, et al. . Short-term association between ambient air pollution and pneumonia in children: a systematic review and meta-analysis of time-series and case-crossover studies. Environ Pollut 2017; 230: 1000–1008. doi: 10.1016/j.envpol.2017.07.063 [DOI] [PubMed] [Google Scholar]
- 18.Gentilotti E, De Nardo P, Cremonini E, et al. . Diagnostic accuracy of point-of-care tests in acute community-acquired lower respiratory tract infections. A systematic review and meta-analysis. Clin Microbiol Infect 2022; 28: 13–22. doi: 10.1016/j.cmi.2021.09.025 [DOI] [PubMed] [Google Scholar]
- 19.D'Amato M, Rea G, Carnevale V, et al. . Assessment of thoracic ultrasound in complementary diagnosis and in follow up of community-acquired pneumonia (CAP). BMC Medical Imaging 2017; 17: 52. doi: 10.1186/s12880-017-0225-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shi H, Han X, Jiang N, et al. . Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: a descriptive study. Lancet Infect Dis 2020; 20: 425–434. doi: 10.1016/S1473-3099(20)30086-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Drain PK, Bajema KL, Dowdy D, et al. . Incipient and subclinical tuberculosis: a clinical review of early stages and progression of infection. Clin Microbiol Rev 2018; 31: e00021–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Migliori GB, Ong CWM, Petrone L, et al. . The definition of tuberculosis infection based on the spectrum of tuberculosis disease. Breathe (Sheff) 2021; 17: 210079. doi: 10.1183/20734735.0079-2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.WHO consolidated guidelines on tuberculosis. Module 3: Diagnosis – Rapid diagnostics for tuberculosis detection 2021 update. https://www.who.int/publications/i/item/9789240029415/ Date last accessed: 5 July 2022.
- 24.American Academy of Pediatrics Subcommittee on Diagnosis and Management of Bronchiolitis . Diagnosis and management of bronchiolitis. Pediatrics 2006: 118: 1774–1793. doi: 10.1542/peds.2006-2223 [DOI] [PubMed] [Google Scholar]
- 25.Ralston S, Lieberthal AS, Meissner HC, et al. . Clinical practice guideline: the diagnosis, management, and prevention of bronchiolitis. Pediatrics 2014; 134: e1474–e1502. doi: 10.1542/peds.2014-2742 [DOI] [PubMed] [Google Scholar]
- 26.Teijeiro A, Gómez RM. Wheezing-related relevant factors and the role of viral bronchiolitis. Front Allergy 2021; 2: 726972. doi: 10.3389/falgy.2021.726972 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bronchiectasis. European Respiratory Society, 2018. https://books.ersjournals.com/content/bronchiectasis
- 28.King PT, Holdsworth SR, Freezer NJ, et al. . Microbiologic follow-up study in adult bronchiectasis. Respir Med 2007; 101: 1633–1638. doi: 10.1016/j.rmed.2007.03.009 [DOI] [PubMed] [Google Scholar]
- 29.Polverino E, Goeminne PC, McDonnell MJ, et al. . European Respiratory Society guidelines for the management of adult bronchiectasis. Eur Respir J 2017; 50: 1700629. doi: 10.1183/13993003.00629-2017 [DOI] [PubMed] [Google Scholar]
- 30.Aliberti S, Goeminne PC, O'Donnell AE, et al. . Criteria and definitions for the radiological and clinical diagnosis of bronchiectasis in adults for use in clinical trials: international consensus recommendations. Lancet Respir Med 2022; 10: 298–306. doi: 10.1016/S2213-2600(21)00277-0 [DOI] [PubMed] [Google Scholar]
- 31.Hill AT, Haworth CS, Aliberti S, et al. . Pulmonary exacerbation in adults with bronchiectasis: a consensus definition for clinical research. Eur Respir J 2017; 49: 1700051. doi: 10.1183/13993003.00051-2017 [DOI] [PubMed] [Google Scholar]
- 32.Global Initiative for Chronic Obstructive Lung Disease . Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease. 2022 Report. Available from: https://goldcopd.org/2022-gold-reports-2/
- 33.Boaventura R, Sibila O, Agusti A, et al. . Treatable traits in bronchiectasis. Eur Respir J 2018; 52: 1801269. doi: 10.1183/13993003.01269-2018 [DOI] [PubMed] [Google Scholar]
- 34.Soler-Cataluña JJ, Piñera P, Trigueros JA, et al. . Spanish COPD guidelines (GesEPOC) 2021 update diagnosis and treatment of COPD exacerbation syndrome. Arch Bronconeumol 2022; 58: 159–170. doi: 10.1016/j.arbres.2021.05.011 [DOI] [PubMed] [Google Scholar]
- 35.McDonald VM, Clark VL, Cordova-Rivera L, et al. . Targeting treatable traits in severe asthma: a randomised controlled trial. Eur Respir J 2020; 55: 1901509. doi: 10.1183/13993003.01509-2019 [DOI] [PubMed] [Google Scholar]
- 36.Martínez-García MA, Soler-Cataluña J-J, Perpiñá-Tordera M, et al. . Factors associated with lung function decline in adult patients with stable non-cystic fibrosis bronchiectasis. Chest 2007; 132: 1565–1572. doi: 10.1378/chest.07-0490 [DOI] [PubMed] [Google Scholar]
- 37.Sethi S, Maloney J, Grove L, et al. . Airway inflammation and bronchial bacterial colonization in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2006; 173: 991–998. doi: 10.1164/rccm.200509-1525OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Prat C, Lacoma A. Bacteria in the respiratory tract – how to treat? Or do not treat? Int J Infect Dis 2016; 51: 113–122. doi: 10.1016/j.ijid.2016.09.005 [DOI] [PubMed] [Google Scholar]
- 39.Daley CL, Iaccarino JM, Lange C, et al. . Treatment of nontuberculous mycobacterial pulmonary disease: an official ATS/ERS/ESCMID/IDSA clinical practice guideline. Eur Respir J 2020; 56: 2000535. doi: 10.1183/13993003.00535-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Prevots DR, Loddenkemper R, Sotgiu G, et al. . Nontuberculous mycobacterial pulmonary disease: an increasing burden with substantial costs. Eur Respir J 2017; 49: 1700374. doi: 10.1183/13993003.00374-2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pan S-W, Shu C-C, Lee C-C, et al. . Role of soluble T-cell immunoglobulin mucin domain-3 in differentiating nontuberculous mycobacterial lung disease from pulmonary colonization. Arch Bronconeumol 2022; 58: 547–553. [DOI] [PubMed] [Google Scholar]
- 42.Safiabadi Tali SH, LeBlanc JJ, Sadiq Z, et al. . Tools and techniques for severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)/COVID-19 detection. Clin Microbiol Rev 2021; 34: e00228-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ren A, Sohaei D, Ulndreaj A, et al. . Ultrasensitive assay for saliva-based SARS-CoV-2 antigen detection. Clin Chem Lab Med 2022; 60: 771–777. doi: 10.1515/cclm-2021-1142 [DOI] [PubMed] [Google Scholar]
- 44.Wölfl-Duchek M, Bergmann F, Jorda A, et al. . Sensitivity and specificity of SARS-CoV-2 rapid antigen detection tests using oral, anterior nasal, and nasopharyngeal swabs: a diagnostic accuracy study. Microbiol Spectr 2022; 10: e0202921. doi: 10.1128/spectrum.02029-21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.LaCourse SM, Seko E, Wood R, et al. . Diagnostic performance of oral swabs for non-sputum based TB diagnosis in a TB/HIV endemic setting. PLoS One 2022; 17: e0262123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Molina-Moya B, Ciobanu N, Hernandez M, et al. . Molecular detection of Mycobacterium tuberculosis in oral mucosa from patients with presumptive tuberculosis. J Clin Med 2020; 9: 4124. doi: 10.3390/jcm9124124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Summer S, Schmidt R, Herdina AN, et al. . Detection of SARS-CoV-2 by real-time PCR under challenging pre-analytical conditions reveals independence of swab media and cooling chain. Sci Rep 2021; 11: 13592. doi: 10.1038/s41598-021-93028-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lippi G, Simundic AM, Plebani M. Potential preanalytical and analytical vulnerabilities in the laboratory diagnosis of coronavirus disease 2019 (COVID-19). Clin Chem Lab Med 2020; 58: 1070–1076. doi: 10.1515/cclm-2020-0285 [DOI] [PubMed] [Google Scholar]
- 49.Zidovec Lepej S, Poljak M. Portable molecular diagnostic instruments in microbiology: current status. Clin Microbiol Infect 2020; 26: 411–420. doi: 10.1016/j.cmi.2019.09.017 [DOI] [PubMed] [Google Scholar]
- 50.Rytter H, Jamet A, Coureuil M, et al. . Which current and novel diagnostic avenues for bacterial respiratory diseases? Front Microbiol 2020; 11: 616971. doi: 10.3389/fmicb.2020.616971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Murphy CN, Fowler R, Balada-Llasat JM, et al. . Multicenter evaluation of the BioFire FilmArray Pneumonia/Pneumonia Plus Panel for detection and quantification of agents of lower respiratory tract infection. J Clin Microbiol 2020; 58: e00128-20. doi: 10.1128/JCM.00128-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Leber AL, Lisby JG, Hansen G, et al. . Multicenter evaluation of the QIAstat-Dx respiratory panel for detection of viruses and bacteria in nasopharyngeal swab specimens. Clin Microbiol 2020; 58: e00155-20. doi: 10.1128/JCM.00155-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.van Asten SAV, Boers SA, de Groot JDF, et al. . Evaluation of the Genmark ePlex® and QIAstat-Dx® respiratory pathogen panels in detecting bacterial targets in lower respiratory tract specimens. BMC Microbiol 2021; 21: 236. doi: 10.1186/s12866-021-02289-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Parčina M, Schneider UV, Visseaux B, et al. . Multicenter evaluation of the QIAstat Respiratory Panel – a new rapid highly multiplexed PCR based assay for diagnosis of acute respiratory tract infections. PLoS One 2020; 15: e0230183. doi: 10.1371/journal.pone.0230183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Etienne EE, Nunna BB, Talukder N, et al. . COVID-19 biomarkers and advanced sensing technologies for point-of-care (POC) diagnosis. Bioengineering (Basel) 2021; 8: 98. doi: 10.3390/bioengineering8070098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Mehaffy C, Kruh-Garcia NA, Graham B, et al. . Identification of Mycobacterium tuberculosis peptides in serum extracellular vesicles from persons with latent tuberculosis infection. J Clin Microbiol; 58: e00393-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wang C, Liu M, Wang Z, et al. . Point-of-care diagnostics for infectious diseases: from methods to devices. Nano Today 2021; 37: 101092. doi: 10.1016/j.nantod.2021.101092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Antoñanzas F, Juárez-Castelló CA, Rodríguez-Ibeas R. Using point-of-care diagnostic testing for improved antibiotic prescription: an economic model. Health Econ Rev 2021; 11: 29. doi: 10.1186/s13561-021-00326-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Prod'hom G, Durussel C, Greub G. A simple blood-culture bacterial pellet preparation for faster accurate direct bacterial identification and antibiotic susceptibility testing with the VITEK 2 system. J Med Microbiol 2013; 62: 773–777. doi: 10.1099/jmm.0.049361-0 [DOI] [PubMed] [Google Scholar]
- 60.Stupar P, Opota O, Longo G, et al. . Nanomechanical sensor applied to blood culture pellets: a fast approach to determine the antibiotic susceptibility against agents of bloodstream infections. Clin Microbiol Infect 2017; 23: 400–405. doi: 10.1016/j.cmi.2016.12.028 [DOI] [PubMed] [Google Scholar]
- 61.GISAID . Enabling rapid and open access to epidemic and pandemic virus data. https://www.gisaid.org/about-us/mission/ Date last accessed: 5 July 2022.
- 62.Oude Munnink BB, Worp N, Nieuwenhuijse DF, et al. . The next phase of SARS-CoV-2 surveillance: real-time molecular epidemiology. Nat Med 2021; 27: 1518–1524. doi: 10.1038/s41591-021-01472-w [DOI] [PubMed] [Google Scholar]
- 63.WHO Consultation on the Global Genomic Surveillance Strategy for Pathogens with Pandemic and Epidemic Potential. https://www.who.int/news-room/events/detail/2021/12/08/default-calendar/who-consultation-on-the-global-genomic-surveillance-strategy-for-pathogens-with-pandemic-and-epidemic-potential/ Date last accessed: 5 July 2022. [DOI] [PMC free article] [PubMed]
- 64.Rakovitsky N, Frenk S, Kon H, et al. . Fourier transform infrared spectroscopy is a new option for outbreak investigation: a retrospective analysis of an extended-spectrum-beta-lactamase-producing Klebsiella pneumoniae outbreak in a neonatal intensive care unit. J Clin Microbiol 2020; 58: e00098-20. doi: 10.1128/JCM.00098-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rogers GB, Wesselingh S. Precision respiratory medicine and the microbiome. Lancet Respir Med 2016; 4: 73–82. doi: 10.1016/S2213-2600(15)00476-2 [DOI] [PubMed] [Google Scholar]
- 66.Dickson RP, Erb-Downward JR, Martinez FJ, et al. . The microbiome and the respiratory tract. Annu Rev Physiol 2016; 78: 481–504. doi: 10.1146/annurev-physiol-021115-105238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tiew PY, Mac Aogáin M, Chotirmall SH. The current understanding and future directions for sputum microbiome profiling in chronic obstructive pulmonary disease. Curr Opin Pulm Med 2022; 28: 121-133. doi: 10.1097/MCP.0000000000000850 [DOI] [PubMed] [Google Scholar]
- 68.Faner R, Sibila O, Agustí A, et al. . The microbiome in respiratory medicine: current challenges and future perspectives. Eur Respir J 2017; 49: 1602086. doi: 10.1183/13993003.02086-2016 [DOI] [PubMed] [Google Scholar]
- 69.Pinu FR, Beale DJ, Paten AM, et al. . Systems biology and multi-omics integration: viewpoints from the metabolomics research community. Metabolites 2019; 9: 76. doi: 10.3390/metabo9040076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Huang YJ, Erb-Downward JR, Dickson RP, et al. . Understanding the role of the microbiome in chronic obstructive pulmonary disease: principles, challenges, and future directions. Transl Res 2017; 179: 71–83. doi: 10.1016/j.trsl.2016.06.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Janda JM, Abbott SL. 16S rRNA gene sequencing for bacterial identification in the diagnostic laboratory: pluses, perils, and pitfalls. J Clin Microbiol 2007; 45: 2761–2764. doi: 10.1128/JCM.01228-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Knight R, Vrbanac A, Taylor BC, et al. . Best practices for analysing microbiomes. Nat Rev Microbiol 2018; 16: 410–422. doi: 10.1038/s41579-018-0029-9 [DOI] [PubMed] [Google Scholar]
- 73.Quince C, Walker AW, Simpson JT, et al. . Shotgun metagenomics, from sampling to analysis. Nat Biotechnol 2017; 35: 833–844. doi: 10.1038/nbt.3935 [DOI] [PubMed] [Google Scholar]
- 74.Rogers GB, Marsh P, Stressmann AF, et al. . The exclusion of dead bacterial cells is essential for accurate molecular analysis of clinical samples. Clin Microbiol Infect 2010; 16: 1656–1658. doi: 10.1111/j.1469-0691.2010.03189.x [DOI] [PubMed] [Google Scholar]
- 75.MacAogáin M, Narayana JK, Tiew PY, et al. . Integrative microbiomics in bronchiectasis exacerbations. Nat Med 2021; 27: 688–699. doi: 10.1038/s41591-021-01289-7 [DOI] [PubMed] [Google Scholar]
- 76.Kosmides AK, Kamisoglu K, Calvano SE, et al. . Metabolomic fingerprinting: challenges and opportunities. Crit Rev Biomed Eng 2013; 41: 205–221. doi: 10.1615/CritRevBiomedEng.2013007736 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Nicholson JK, Lindon JC. Metabonomics. Nature 2008; 455: 1054–1056. doi: 10.1038/4551054a [DOI] [PubMed] [Google Scholar]
- 78.Dicker AJ, Huang JTJ, Lonergan M, et al. . The sputum microbiome, airway inflammation, and mortality in chronic obstructive pulmonary disease. J Allergy Clin Immunol 2021; 147: 158–167. doi: 10.1016/j.jaci.2020.02.040 [DOI] [PubMed] [Google Scholar]
- 79.Dicker AJ, Lonergan M, Keir HR, et al. . The sputum microbiome and clinical outcomes in patients with bronchiectasis: a prospective observational study. Lancet Respir Med 2021; 9: 885–896. doi: 10.1016/S2213-2600(20)30557-9 [DOI] [PubMed] [Google Scholar]
- 80.Rogers GB, van der Gast CJ, Cuthbertson L, et al. . Clinical measures of disease in adult non-CF bronchiectasis correlate with airway microbiota composition. Thorax 2013; 68: 731–737. doi: 10.1136/thoraxjnl-2012-203105 [DOI] [PubMed] [Google Scholar]
- 81.Mindt BC, DiGiandomenico A. Microbiome modulation as a novel strategy to treat and prevent respiratory infections. Antibiotics (Basel) 2022; 11: 474. doi: 10.3390/antibiotics11040474 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Matthews H, Hanison J, Nirmalan N. “Omics”-informed drug and biomarker discovery: opportunities, challenges and future perspectives. Proteomes 2016; 4: 28. doi: 10.3390/proteomes4030028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kontsevaya I, Lange C, Comella-del-Barrio P, et al. . Perspectives for systems biology in the management of tuberculosis. Eur Respir Rev 2021; 30: 200377. doi: 10.1183/16000617.0377-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Olivier I, Loots DT. 2011. An overview of tuberculosis treatments and diagnostics. What role could metabolomics play. J Cell Tissue Res 2011; 11: 2655-2671.
- 85.Haas CT, Roe JK, Pollara G, et al. . Diagnostic ‘omics’ for active tuberculosis. BMC Med 2016; 14: 37. doi: 10.1186/s12916-016-0583-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Izquierdo-Garcia JL, Comella-del-Barrio P, Campos-Olivas R, et al. . Discovery and validation of an NMR-based metabolomic profile in urine as TB biomarker. Sci Rep 2020; 10: 22317. doi: 10.1038/s41598-020-78999-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Comella-del-Barrio P, Izquierdo-Garcia JL, Gautier J, et al. . Urine NMR-based TB metabolic fingerprinting for the diagnosis of TB in children. Sci Rep 2021; 11: 12006. doi: 10.1038/s41598-021-91545-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.den Hartog I, Zwep LB, Vestjens SMT, et al. . Metabolomic profiling of microbial disease etiology in community-acquired pneumonia. PLoS One 2021; 16: e0252378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Izquierdo-García JL, Nin N, Cardinal-Fernandez P, et al. . Metabolomic profile of acute respiratory distress syndrome of different etiologies. Intensive Care Med 2019; 45: 1318–1320. doi: 10.1007/s00134-019-05634-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Slupsky CM, Cheypesh A, Chao DV, et al. . Streptococcus pneumoniae and Staphylococcus aureus pneumonia induce distinct metabolic responses. J Proteome Res 2009; 8: 3029–3036. doi: 10.1021/pr900103y [DOI] [PubMed] [Google Scholar]
- 91.Karakioulaki M, Stolz D. Biomarkers in pneumonia – beyond procalcitonin. Int J Mol Sci 2019; 20: 2004. doi: 10.3390/ijms20082004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Carlton HC, Savović J, Dawson S, et al. . Novel point-of-care biomarker combination tests to differentiate acute bacterial from viral respiratory tract infections to guide antibiotic prescribing: a systematic review. Clin Microbiol Infect 2021; 27: 1096–1108. doi: 10.1016/j.cmi.2021.05.018 [DOI] [PubMed] [Google Scholar]
- 93.Haag E, Molitor A, Gregoriano C, et al. . The value of biomarker-guided antibiotic therapy. Expert Rev Mol Diagn 2020; 20: 829–840. doi: 10.1080/14737159.2020.1782193 [DOI] [PubMed] [Google Scholar]
- 94.Pérez-López A, Irwin A, Rodrigo C, et al. . Role of C reactive protein and procalcitonin in the diagnosis of lower respiratory tract infection in children in the outpatient setting. BMJ 2021; 373: n1409. doi: 10.1136/bmj.n1409 [DOI] [PubMed] [Google Scholar]
- 95.Tabernero E, Ruiz LA, España PP, et al. . COVID-19 in young and middle-aged adults: predictors of poor outcome and clinical differences. Infection 2022; 50: 179–189. doi: 10.1007/s15010-021-01684-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Riou C, du Bruyn E, Stek C, et al. . Relationship of SARS-CoV-2-specific CD4 response to COVID-19 severity and impact of HIV-1 and tuberculosis coinfection. J Clin Invest 2021; 131: e149125. doi: 10.1172/JCI149125 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Aabye MG, Latorre I, Diaz J, et al. . Dried plasma spots in the diagnosis of tuberculosis: IP-10 release assay on filter paper. Eur Respir J 2013; 42: 495–503. doi: 10.1183/09031936.00129412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Latorre I, Díaz J, Mialdea I, et al. . IP-10 is an accurate biomarker for the diagnosis of tuberculosis in children. J Infect 2014; 69: 590–599. doi: 10.1016/j.jinf.2014.06.013 [DOI] [PubMed] [Google Scholar]
- 99.Ruhwald M, Dominguez J, Latorre I, et al. . A multicentre evaluation of the accuracy and performance of IP-10 for the diagnosis of infection with M. tuberculosis. Tuberculosis (Edinb) 2011; 91: 260–267. doi: 10.1016/j.tube.2011.01.001 [DOI] [PubMed] [Google Scholar]
- 100.Villar-Hernández R, Latorre I, Mínguez S, et al. . Use of IFN-γ and IP-10 detection in the diagnosis of latent tuberculosis infection in patients with inflammatory rheumatic diseases. J Infect 2017; 75: 315–325. doi: 10.1016/j.jinf.2017.07.004 [DOI] [PubMed] [Google Scholar]
- 101.Lydon EC, Ko ER, Tsalik EL. The host response as a tool for infectious disease diagnosis and management. J Infect 2017; 75: 315–325. doi: 10.1080/14737159.2018.1493 [DOI] [PubMed] [Google Scholar]
- 102.Ross MH, Zick BL, Tsalik EL. Host-based diagnostics for acute respiratory infections. Clin Ther 2019; 41: 1923–1938. doi: 10.1016/j.clinthera.2019.06.007 [DOI] [PubMed] [Google Scholar]
- 103.Herberg JA, Kaforou M, Wright VJ, et al. . Diagnostic test accuracy of a 2-transcript host RNA signature for discriminating bacterial vs viral infection in febrile children. JAMA 2016; 316: 835–845. doi: 10.1001/jama.2016.11236 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Channon-Wells S, O'Connor D. Host gene signature shows promise to distinguish bacterial and viral infections. Lancet Digit Health 2021; 3: e465–e466. doi: 10.1016/S2589-7500(21)00136-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Bhattacharya S, Rosenberg AF, Peterson DR, et al. . Transcriptomic biomarkers to discriminate bacterial from nonbacterial infection in adults hospitalized with respiratory illness. Sci Rep 2017; 7: 6548. doi: 10.1038/s41598-017-06738-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Suarez NM, Bunsow E, Falsey AR, et al. . Superiority of transcriptional profiling over procalcitonin for distinguishing bacterial from viral lower respiratory tract infections in hospitalized adults. J Infect Dis 2015; 212: 213–222. doi: 10.1093/infdis/jiv047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Woods CW, McClain MT, Chen M, et al. . A host transcriptional signature for presymptomatic detection of infection in humans exposed to influenza H1N1 or H3N2. PLoS One 2013; 8: e52198. doi: 10.1371/journal.pone.0052198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Davenport EE, Antrobus RD, Lillie PJ, et al. . Transcriptomic profiling facilitates classification of response to influenza challenge. J Mol Med (Berl) 2015; 93: 105–114. doi: 10.1007/s00109-014-1212-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Zak DE, Penn-Nicholson A, Scriba TJ, et al. . A blood RNA signature for tuberculosis disease risk: a prospective cohort study. Lancet 2016; 387: 2312–2322. doi: 10.1016/S0140-6736(15)01316-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Zheng MZM, Wakim LM. Tissue resident memory T cells in the respiratory tract. Mucosal Immunol 2022; 15: 379–388. doi: 10.1038/s41385-021-00461-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ogongo P, Porterfield JZ, Leslie A. Lung tissue resident memory T-cells in the immune response to Mycobacterium tuberculosis. Front Immunol 2019; 10: 992. doi: 10.3389/fimmu.2019.00992 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Grau-Expósito J, Sánchez-Gaona N, Massana N, et al. . Peripheral and lung resident memory T cell responses against SARS-CoV-2. Nat Commun 2021; 12: 3010. doi: 10.1038/s41467-021-23333-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Luangrath MA, Schmidt ME, Hartwig SM, et al. . Tissue-resident memory T cells in the lungs protect against acute respiratory syncytial virus infection. Immunohorizons 2021; 5: 59–69. doi: 10.4049/immunohorizons.2000067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Ogongo P, Tezera LB, Ardain A, et al. . Tissue-resident-like CD4+ T cells secreting IL-17 control Mycobacterium tuberculosis in the human lung. J Clin Invest 2021; 131: e142014. doi: 10.1172/JCI142014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Yang Q, Zhang M, Chen Q, et al. . Cutting edge: characterization of human tissue-resident memory T cells at different infection sites in patients with tuberculosis. J Immunol 2020; 204: 2331–2336. doi: 10.4049/jimmunol.1901326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Arts RJW, Moorlag S, Novakovic B, et al. . BCG vaccination protects against experimental viral infection in humans through the induction of cytokines associated with trained immunity. Cell Host Microbe 2018; 23: 89–100.e5. doi: 10.1016/j.chom.2017.12.010 [DOI] [PubMed] [Google Scholar]
- 117.Covián C, Ríos M, Berríos-Rojas RV, et al. . Induction of trained immunity by recombinant vaccines. Front Immunol 2021; 11: 611946. doi: 10.3389/fimmu.2020.611946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Netea MG, Giamarellos-Bourboulis EJ, Domínguez-Andrés J, et al. . Trained immunity: a tool for reducing susceptibility to and the severity of SARS-CoV-2 infection. Cell 2020; 181: 969–977. doi: 10.1016/j.cell.2020.04.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.O'Neill LAJ, Netea MG. BCG-induced trained immunity: can it offer protection against COVID-19? Nat Rev Immunol 2020; 20: 335–337. doi: 10.1038/s41577-020-0337-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Pittet LF, Messina NL, Gardiner K, et al. . BCG vaccination to reduce the impact of COVID-19 in healthcare workers: protocol for a randomised controlled trial (BRACE trial). BMJ Open 2021; 11: e052101. doi: 10.1136/bmjopen-2021-052101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Giamarellos-Bourboulis EJ, Tsilika M, Moorlag S, et al. . Activate: randomized clinical trial of BCG vaccination against infection in the elderly. Cell 2020; 183: 315–323.e9. doi: 10.1016/j.cell.2020.08.051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Hancock RE, Nijnik A, Philpott DJ. Modulating immunity as a therapy for bacterial infections. Nat Rev Microbiol 2012; 10: 243–254. doi: 10.1038/nrmicro2745 [DOI] [PubMed] [Google Scholar]
- 123.Kaufmann SHE, Dorhoi A, Hotchkiss RS, et al. . Host-directed therapies for bacterial and viral infections. Nat Rev Drug Discov 2018; 17: 35–56. doi: 10.1038/nrd.2017.162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.van de Veerdonk FL, Giamarellos-Bourboulis E, Pickkers P, et al. . A guide to immunotherapy for COVID-19. Nat Med 2022; 28: 39–50. doi: 10.1038/s41591-021-01643-9 [DOI] [PubMed] [Google Scholar]
- 125.Zumla A, Rao M, Wallis RS, et al. . Host-directed therapies for infectious diseases: current status, recent progress, and future prospects. Lancet Infect Dis 2016; 16: e47–e63. doi: 10.1016/S1473-3099(16)00078-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Nardelli P, Jimenez-Carretero D, Bermejo-Pelaez D, et al. . Pulmonary artery-vein classification in CT images using deep learning. IEEE Trans Med Imaging 2018; 37: 2428–2440. doi: 10.1109/TMI.2018.2833385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Yan C, Wang L, Lin J, et al. . A fully automatic artificial intelligence-based CT image analysis system for accurate detection, diagnosis, and quantitative severity evaluation of pulmonary tuberculosis. Eur Radiol 2022; 32: 2188–2199. doi: 10.1007/s00330-021-08365-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Serena Low WC, Chuah JH, Tee CATH, et al. . An overview of deep learning techniques on chest X-ray and CT scan identification of COVID-19. Comput Math Methods Med 2021; 2021: 5528144. doi: 10.1155/2021/5528144.eCollection2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Frix A-N, Cousin F, Refaee T, et al. . Radiomics in lung diseases imaging: state-of-the-art for clinicians. J Pers Med 2021; 11: 602. doi: 10.3390/jpm11070602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Pizzi AD, Chiarelli AM, Chiacchiaretta P, et al. . Radiomics-based machine learning differentiates “ground-glass” opacities due to COVID-19 from acute non-COVID-19 lung disease. Sci Rep 2021; 11: 17237. doi: 10.1038/s41598-021-96755-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Feng B, Chen X, Chen Y, et al. . Radiomics nomogram for preoperative differentiation of lung tuberculoma from adenocarcinoma in solitary pulmonary solid nodule. Eur J Radiol 2020; 128: 109022. doi: 10.1016/j.ejrad.2020.109022 [DOI] [PubMed] [Google Scholar]
- 132.Chen L, Rezaei T. A new optimal diagnosis system for coronavirus (COVID-19) diagnosis based on Archimedes optimization algorithm on chest X-ray images. Comput Intell Neurosci 2021; 2021: 7788491. doi: 10.1155/2021/7788491.eCollection2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Chen AC, Pastis NJ, Mahajan AK, Jr, et al. . Robotic bronchoscopy for peripheral pulmonary lesions: a multicenter pilot and feasibility study (BENEFIT). Chest 2021; 159: 845–852. doi: 10.1016/j.chest.2020.08.2047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Ishiwata T, Gregor A, Inage T, et al. . Bronchoscopic navigation and tissue diagnosis. Gen Thorac Cardiovasc Surg 2020; 68: 672–678. doi: 10.1007/s11748-019-01241-0 [DOI] [PubMed] [Google Scholar]
- 135.Vijayarangan S, Suresh P, Sp P, et al. . Robust modelling of reflectance pulse oximetry for SpO2 estimation. Annu Int Conf IEEE Eng Med Biol Soc 2020; 2020: 374–377. doi: 10.1109/EMBC44109.2020.9176410 [DOI] [PubMed] [Google Scholar]
- 136.Patel Z, Franz CK, Bharat A, et al. . Diaphragm and phrenic nerve ultrasound in COVID-19 patients and beyond. J Ultrasound Med 2022; 41: 285–299. doi: 10.1002/jum.15706 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Ong CWM, Migliori GB, Raviglione M, et al. . Epidemic and pandemic viral infections: impact on tuberculosis and the lung. Eur Respir J 2020; 56: 2001727. doi: 10.1183/13993003.01727-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Taboada M, Moreno E, Cariñena A, et al. . Quality of life, functional status, and persistent symptoms after intensive care of COVID-19 patients. Br J Anaesth 2021; 126: e110–e113. doi: 10.1016/j.bja.2020.12.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Chams N, Chams S, Badran R, et al. . COVID-19: a multidisciplinary review. Front Public Health 2020; 8: 383. doi: 10.3389/fpubh.2020.00383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.One health. World Health Organization. https://www.euro.who.int/en/health-topics/health-policy/one-health/ Date last accessed: 5 July 2022.
- 141.Naylor NR, Atun R, Zhu N, et al. . Estimating the burden of antimicrobial resistance: a systematic literature review. Antimicrob Resist Infect Control 2018; 7: 58. doi: 10.1186/s13756-018-0336-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Ruhwald M, Carmona S, Pai M. Learning from COVID-19 to reimagine tuberculosis diagnosis. Lancet Microbe 2021; 2: e169–e170. doi: 10.1016/S2666-5247(21)00057-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Stojanovic Z, Prat-Aymerich C. Impact of COVID-19 on tuberculosis: turning challenges into opportunities. Enf Emerg 2021; 20: 151–154. [Google Scholar]
- 144.FIND. Landscape of benchtop immunoassay platforms for near patient testing: The MAPDx Program. https://www.finddx.org/wp-content/uploads/2019/04/Landscape-benchtop-immunoassay-platforms-201890405_FINAL_v2.pdf Date last accessed: 5 July 2022.
- 145.Goossens H, Derde L, Horby P, et al. . The European clinical research response to optimise treatment of patients with COVID-19: lessons learned, future perspective, and recommendations. Lancet Infect Dis 2022; 22: e153–e158. doi: 10.1016/S1473-3099(21)00705-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.van der Pol S, Rojas Garcia P, Antoñanzas Villar F, et al. . Health-economic analyses of diagnostics: guidance on design and reporting. PharmacoEconomics 2021; 39: 1355–1363. doi: 10.1007/s40273-021-01104-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.García-Vidal C, Sanjuán G, Puerta-Alcalde P, et al. . Artificial intelligence to support clinical decision-making processes. EBioMedicine 2019; 46: 27–29. doi: 10.1016/j.ebiom.2019.07.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Wilkinson MD, Dumontier M, Aalbersberg IJ, et al. . The FAIR Guiding Principles for scientific data management and stewardship. Sci Data 2016; 3: 160018. doi: 10.1038/sdata.2016.18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.European Commission, Directorate-General for Research and Innovation, Maxwell L . Maximising investments in health research: FAIR data for a coordinated COVID-19 response: workshop report; 2022. https://op.europa.eu/en/publication-detail/-/publication/f023acef-ba07-11ec-b6f4-01aa75ed71a1/language-en/format-PDF/ Date last accessed: 5 July 2022.
- 150.US Food and Drug Administration. 21st Century Cures Act. https://www.fda.gov/regulatory-information/selected-amendments-fdc-act/21st-century-cures-act/ Date last accessed: 5 July 2022.