Biological models of Alzheimer’s disease (AD): Non-human models have contributed tremendously to the understanding of AD and its underlying pathological processes. These models have aided the investigation of the genetic and environmental risk factors. They also have enabled the progression of candidate therapies into human clinical trials. Because of similarities with human brain anatomy and genetics, rodent models have been used extensively to recapitulate some aspects of AD pathology, measure AD-associated behavioral parameters and related nervous system dysfunctions (Eriksen and Janus, 2007). For instance, transgenic mice overexpressing human amyloid precursor protein have furthered the development of the amyloid cascade hypothesis as a central pillar of familial AD. Although considered as advantageous, mammalian models have practical and ethical problems when it comes to high-throughput screening (HTS) of drugs. Therefore, alternative models, including the use of invertebrates with very short lifecycles, represent rapid, simple, and highly cost-effective platforms for HTS or drug target identification (Artal-Sanz et al., 2006). Indeed, the use of such organisms, as opposed to individual cell models, provides advantages for understanding the wider complexity of human pathology. Thereby, prominent invertebrate model organisms, such as Drosophila melanogaster (fruit fly), and the nematode Caenorhabditis elegans (roundworm), were insightful into the aging process, AD and Parkinson’s disease (Newman et al., 2011). The genome of D. melanogaster is ~70% homologous to that of humans and shows relatively complex brain and neuronal structures. Its eye is an accessible organ for phenotypic characterization, measuring of behavioral deficits, and determining responses to drug compounds. This fruit fly has unique learning and memory behaviors and can induce the gene expression of proteins considered to be the major hallmarks of AD, such as amyloid-beta (Aβ) or tau, both leading to AD pathological features and phenotypes. Furthermore, compounds such as GSK3-β inhibitors can rescue axonal transport and locomotion defects in these models. C. elegans is another invertebrate model organism employed in AD research. Several attempts have been made to use this free-living nematode (natively occurring in temperate soil environments) to generate transgenic AD models. One transgenic C. elegans model expressing Aβ peptides in muscle has a paralysis transgene-induced phenotype (Dostal and Link, 2010). Other models that neuronally express human Aβ exhibit phenotypes such as impaired chemotaxis and deficits in associated learning. Reported defects include impaired odor associative learning behavior, impaired serotonin-stimulated egg-laying, and decreased lifespan evidently resulting from Aβ toxicity.
Introducing Globodera pallida (G. pallida)as a novel model for AD (and potentially other proteinopathies): In this paper, we put forward a non-transgenic invertebrate for newly modeling AD pathology, G. pallida, which is one of the best characterized of the potato cyst nematodes (Althobaiti et al., 2021). G. pallida is an important model for understanding the biology of potato cyst nematodes, which are responsible for substantial global crop losses (Warnock et al., 2017). The lifecycle of G. pallida is described as obligate, involving the cross-fertilization of different hosts, and it has six life stages: egg (or embryo) stage, four juvenile stages (J1 to J4), and a sexually adult stage. Our proposed AD model uses J2s which have a motile, vermiform, round head and fine tail, with bodies ~500 µm in length, styles of 19–25 µm, and tails of 37–55 µm. Nematodes have a structurally simple nervous system but contain a complex array of signaling molecules (e.g., neuropeptides) that perform vital functions such as the maintenance of parasite physiology, feeding, locomotion, and reproduction (Warnock et al., 2017). The nematode ‘brain’ (Figure 1) consists of a circumpharyngeal nerve ring (CNR) which encircles the oesophagus just behind the pharyngeal bulb. The CNR contains multiple neurons which connect several sensing organs and a nerve trunk that runs longitudinally from the CNR to the head and the rest of the body. There are two pairs of lengthwise nerve cords on the dorsal and ventral sides, as well as multiple distinct chemosensory neural cell bodies in the ventral nerve cord. Chemoreceptive organs include amphids, near the head, and phasmids at their posterior. These chemosensory organs are vital to nematode behavior with a large number of neurons because they are sensitive to environmental changes, such as temperature, presence of food, or detection of reproductive mates. It has long been established that the nematode behavior is affected by chemicals exuded from plant roots, and that the sugar mannitol, is a key attractant for G. pallida. It is also known that their chemosensory neurons can assimilate exogenous peptides from the external environment (Warnock et al., 2017) – a feature which we have exploited for the development of G. pallida as a new model for studying AD pathology. Indeed, our peer-reviewed published research has demonstrated, for the first time, the capacity of G. pallida (as a non-genetically modified organism) to naturally assimilate Aβ peptides (Althobaiti et al., 2021). Crucially, these peptides almost exclusively localized to neurons (including the amphid neurons of the CNR), and this causes measurable impairments of their behavior. We have shown that their normal chemosensing of mannitol, as a food source, can be completely disrupted. The unique feature of such a model system is that it is possible to reliably control the dose, duration, and protocol the organisms’ exposure to Aβ; additionally, the impact of each specific Aβ isoform (or combination of isoforms) can be assessed. Chemotaxis is disrupted to the greatest extent by treatment with human Aβ(1–42) (which is also the most neurotoxic form), and we were able to compare this against the relative harm caused by other Aβ forms including (1–40), (17–42), (17–40), (1–28) and (1–16). For Aβ(1–42), we have established a time course for their responsiveness. Pre-incubations for at least 6 hours significantly affect chemosensing, with maximal effect reached by 18–24 hours. Clearly, the absorption and toxicity of Aβ peptides on the neurons and CNR takes a period to manifest as behavioral changes in this nematode. The major contributors to this are the transit times of peptides along neurons (corroborated by immunocytochemical staining), and the time taken for neurotoxicity to occur. In addition to measuring changes in chemosensing, we describe several other quantitative parameters that can be applied in G. pallida as an alternative model nematode of AD. Some of these are discussed below and include: the fluorometric measurement of the production of reactive oxygen species (ROS), fluorometric measurement of oxidized/reduced glutathione (GSSG/GSH), quantification of mitochondrial reductase activity, and the visual assessment of their movement and survival (Figure 1). These methods could be combined into a proposed process for establishing proteinopathy models in G. pallida and for testing these. Oxidative stress is a factor implicated in the occurrence and progression of AD, and Aβ is implicated in the production of ROS. In G. pallida, ROS significantly rose upon exposure to Aβ peptide fragments, particularly Aβ(1–42) and Aβ(17–42), which are known to be most toxic (Joshi et al., 2005). The methionine at position 35 of Aβ is thought to be an important amino acid for neurotoxicity and oxidative stress (Navarro-Yepes et al., 2014). The highest levels of ROS production have been elicited with Aβ(1–16), Aβ(1–28), Aβ(1–40), and Aβ(17–40) peptides. A degree of sensitivity appears to be afforded by measuring ROS in G. pallida, since Aβ(1–16) and Aβ(1–28) isoforms do not aggregate and are rarely considered to be neurotoxic. Other studies suggest that Aβ(1–28) may not induce oxidative stress (Butterfield and Boyd-Kimball, 2005), but Aβ(1–16) has previously been demonstrated to induce ROS formation (Du et al., 2011). In the G. pallida system, Aβ(1–40) and (17–40) induced the lowest amounts of ROS formation, which is in line with the literature. It is postulated that the termination of the peptide sequence at position 40 makes them more soluble, less toxic and less prone to aggregation compared with peptides terminating at position 42. Indeed, it is clear that Aβ(1–42) and (17–42) cause mitochondrial dysfunction induction, lipid and protein oxidation in neuronal cells culture (Butterfield and Boyd-Kimball, 2005), and Aβ(1–42) is thought to impact AD pathology to a much great extent than other smaller fragments of Aβ (Butterfield and Sultana, 2011). GSH plays an important cellular role in the anti-oxidative activity, but when cells are exposed to increased levels of oxidative stress, it is worth noting that the oxidized glutathione GSSG will accumulate, thereby altering the ratio of GSH to GSSG. Measurement of this ratio in G. pallida reinforces the findings of ROS assays. GSSG/GSH ratios were markedly augmented by Aβ(1–42) in a cumulative manner over 24 hours. This has a disease relevance since brain tissues from AD and mild cognitive impairment subjects have lower levels of antioxidant enzymes, perhaps making it more vulnerable to Aβ(1–42) induced-toxicity. Relatively high levels of metal ions, oxygen and unsaturated lipids in the brain also make this organ more sensitive and vulnerable to damage from oxidative stress (Pocernich and Butterfield, 2012).
Figure 1.
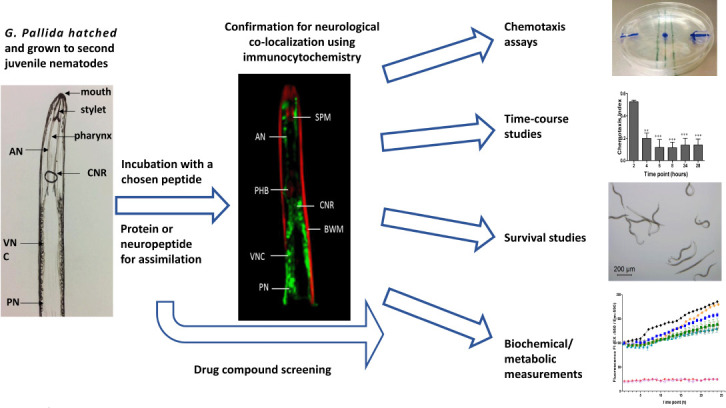
A proposed process for establishing proteinopathy models in G. pallida and testing.
After hatching and growth to J2 juveniles, the potato cyst nematodes were incubated with a selected peptide or protein (e.g., amyloid beta (1–42)). Assimilation and co-localization to neurons were assessed by immunocytochemistry using an appropriate antibody, and the physiological and behavioral effects were evaluated by a battery of tests. Once the model is characterized, the process can be repeated with libraries of candidate drugs or medicinal plant extracts. AN: Amphidial neuron; BWM: body wall muscle; CNR: circumpharyngeal nerve ring; PHB: pharyngeal bulb; PN: phasmid neurons; SPM: stylet protractor muscles; VNC: ventral nerve cord.
Future potential of G. pallida for examining proteinopathies: One might envisage running automated ultra-HTS of thousands of candidate compounds or medicinal plant extracts, in order to evaluate their potential for countering the neurodegenerative effects of amyloidoses, tauopathies, and synucleopathies. AD is a key example of both amyloidosis and tauopathy, and thus, studies with tau and phospho-tau are warranted. Other major synucleopathies include Parkinson’s disease (where alpha-synuclein aggregates in the brain stem), Lewy body disease (where Lewy bodies occur in the cortico-limbic and brainstem), and multiple system atrophy (where alpha-synuclein accumulates in basal ganglia, brainstem, and cerebellum). Taken together, studies that examined the assimilation of alpha-synuclein isoforms should be undertaken. Furthermore, G. pallida model can be potentially used to examine prion diseases, for instance by examining the role of prion protein in the manifestation of Creutzfeldt-Jakob disease. Another distinctive feature of G. pallida is its extreme sensitivity to neuronal RNA interference, which would be a powerful functional genomics tool for studying the underlying mechanisms of proteinopathy toxicity (Dalzell et al., 2010).
Final remarks and perspectives: Although there have been significant advances in understanding neurodegenerative diseases, such as AD, it is worth mentioning that there is still no effective cure. Prevalence of AD has dramatically increased with life expectancy, which has been steadily increasing worldwide. Establishing a novel model system such G. pallida, that is cost-effective, practical, non-transgenic (to ensure data reliability), and that provides an HTS platform for screening potential treatments, will hopefully aid in the discovery of effective therapy with minimal adverse effects. Such a disease model provides a means of modulating the dose, duration, sequence, and type of proteinopathy being investigated, and therefore is worthy of further investigation.
Footnotes
C-Editors: Zhao M, Liu WJ, Wang Lu; T-Editor: Jia Y
References
- 1.Althobaiti NA, Menaa F, Albalawi AE, Dalzell JJ, Warnock ND, Mccammick EM, Alsolais A, Alkhaibari AM, Green BD. Assessment and validation of Globodera pallida as a novel in vivo model for studying Alzheimer's disease. Cells. 2021;10:2481. doi: 10.3390/cells10092481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Artal-Sanz M, de Jong L, Tavernarakis N. Caenorhabditis elegans:a versatile platform for drug discovery. Biotechnol J. 2006;1:1405–1418. doi: 10.1002/biot.200600176. [DOI] [PubMed] [Google Scholar]
- 3.Butterfield DA, Boyd-Kimball D. The critical role of methionine 35 in Alzheimer's amyloid β-peptide (1–42)-induced oxidative stress and neurotoxicity. Biochim Biophys Acta. 2005;1703:149–156. doi: 10.1016/j.bbapap.2004.10.014. [DOI] [PubMed] [Google Scholar]
- 4.Butterfield DA, Sultana R. Methionine-35 of Aβ(1–42):importance for oxidative stress in Alzheimer disease. J Amino Acids. 2011;2011:198430. doi: 10.4061/2011/198430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dalzell JJ, McMaster S, Fleming CC, Maule AG. Short interfering RNA-mediated gene silencing in Globodera pallida and Meloidogyne incognita infective stage juveniles. Int J Parasitol. 2010;40:91–100. doi: 10.1016/j.ijpara.2009.07.003. [DOI] [PubMed] [Google Scholar]
- 6.Dostal V, Link CD. Assaying β-amyloid toxicity using a transgenic C elegans model. J Vis Exp. 2010;2010:e2252. doi: 10.3791/2252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Du XT, Wang L, Wang YJ, Andreasen M, Zhan DW, Feng Y, Li M, Zhao M, Otzen D, Xue D. Aβ1-16 can aggregate and induce the production of reactive oxygen species, nitric oxide, and inflammatory cytokines. J Alzheimers Dis. 2011;27:401–413. doi: 10.3233/JAD-2011-110476. [DOI] [PubMed] [Google Scholar]
- 8.Eriksen JL, Janus CG. Plaques, tangles, and memory loss in mouse models of neurodegeneration. Behav Genet. 2007;37:79–100. doi: 10.1007/s10519-006-9118-z. [DOI] [PubMed] [Google Scholar]
- 9.Joshi G, Sultana R, Perluigi M, Butterfield DA. In vivo protection of synaptosomes from oxidative stress mediated by Fe2+/H2O2 or 2, 2-azobis-(2-amidinopropane) dihydrochloride by the glutathione mimetic tricyclodecan-9-yl-xanthogenate. Free Radic Biol Med. 2005;38:1023–1031. doi: 10.1016/j.freeradbiomed.2004.12.027. [DOI] [PubMed] [Google Scholar]
- 10.Navarro-Yepes J, Zavala-Flores L, Anandhan A, Wang F, Skotak M, Chandra N, Li M, Pappa A, Martinez-Fong D, Del Razo LM. Antioxidant gene therapy against neuronal cell death. Pharmacol Ther. 2014;142:206–230. doi: 10.1016/j.pharmthera.2013.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Newman T, Sinadinos C, Johnston A, Sealey M, Mudher M. Using Drosophila models of neurodegenerative diseases for drug discovery. Expert Opin Drug Discov. 2011;6:129–410. doi: 10.1517/17460441.2011.549124. [DOI] [PubMed] [Google Scholar]
- 12.Pocernich CB, Butterfield DA. Elevation of glutathione as a therapeutic strategy in Alzheimer disease. Biochim Biophys Acta. 2012;1822:625–630. doi: 10.1016/j.bbadis.2011.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Warnock ND, Wilson L, Patten C, Fleming CC, Maule AG, Dalzell JJ. Nematode neuropeptides as transgenic nematicides. PLoS Pathog. 2017;13:e1006237. doi: 10.1371/journal.ppat.1006237. [DOI] [PMC free article] [PubMed] [Google Scholar]


