Abstract
Regulated cell death predominantly involves apoptosis, autophagy, and regulated necrosis. It is vital that we understand how key regulatory signals can control the process of cell death. Pin1 is a cis-trans isomerase that catalyzes the isomerization of phosphorylated serine or threonine-proline motifs of a protein, thereby acting as a crucial molecular switch and regulating the protein functionality and the signaling pathways involved. However, we know very little about how Pin1-associated pathways might play a role in regulated cell death. In this paper, we review the role of Pin1 in regulated cell death and related research progress and summarize Pin1-related pathways in regulated cell death. Aside from the involvement of Pin1 in the apoptosis that accompanies neurodegenerative diseases, accumulating evidence suggests that Pin1 also plays a role in regulated necrosis and autophagy, thereby exhibiting distinct effects, including both neurotoxic and neuroprotective effects. Gaining an enhanced understanding of Pin1 in neuronal death may provide us with new options for the development of therapeutic target for neurodegenerative disorders.
Keywords: apoptosis, autophagy, calpain, central nervous system, necroptosis, necrosis, neurodegenerative diseases, neuron, Pin1, regulated neuronal death
Introduction
Cell death is a fundamental process that is essential for the normal development and homeostasis of organisms (Bai et al., 2020; Moujalled et al., 2021). In general, there are three types of cell death: apoptosis, autophagy, and necrosis (Carloni et al., 2017; Bai et al., 2020). Apoptosis refers to regulated cell death (RCD) that occurs in developing tissues and plays an important role in homeostasis. RCD is characterized by chromatin condensation, nuclear fragmentation, and the formation of apoptotic bodies. Previous studies have reported that a large number of apoptotic proteins play a role in apoptotic signaling pathways, including caspases and B-cell lymphoma 2 (Bcl-2) family members (Bai et al., 2020; Özel et al., 2021). Autophagy is another form of RCD during which cellular contents, such as organelles, are encapsulated by a double-membrane to form auto-phagosomes that are subsequently degraded by lysosomes (Napoletano et al., 2019; Bourdenx et al., 2021). Autophagy plays an important role in maintaining homeostasis and protecting cells against stressful stimuli. Traditionally, necrosis is considered to represent a form of accidental cell death that is characterized by membrane destruction, the release of internal materials, and inflammation (Chen et al., 2021; Hu et al., 2021b). However, recent research has provided evidence that some types of necrosis also can be regulated at the molecular level (Theobald et al., 2021; Tonnus et al., 2021). Other researchers have reported that this form of regulated necrosis (RN) includes necroptosis (Evans and Coyne, 2019; Yuan et al., 2019), pyroptosis (Lammert et al., 2020), and neutrophil cell death (Sung and Hsieh, 2021; Yan et al., 2021). RCD, featuring apoptosis, autophagy and RN, is recognized to be beneficial to the host by eliminating certain intracellular pathogens or stimuli (Guo et al., 2020; Wang et al., 2020b; Yan et al., 2021). It is vital that we know how RCD responds to pathogens or injuries.
Prolyl cis-trans isomerase NIMA-interacting 1 (Pin1) belongs to the parvulin subfamily and is an 18-kDa protein with a C-terminal PPIase domain and a N-terminal WW domain (Kumari et al., 2021; Tonnus et al., 2021; Zheng et al., 2021). Pin1 is able to bind and then isomerize phosphorylated serine-proline or phosphorylated threonine-proline (pSer/Thr-Pro) motifs, thus regulating a series of biological processes (Koikawa et al., 2021; Kumari et al., 2021). The WW domain responds to and binds to pSer/Thr-Pro sequences, while the catalytic PPIase domain isomerizes the prolyl bond in the pSer/Thr-Pro motif; collectively, these mechanisms play an important role in many biological processes, such as the cell cycle, immune responses, and tumorigenesis (Koikawa et al., 2021; Kumari et al., 2021). In addition to binding to the prolyl bond in the pSer/Thr-Pro motif, Pin1 can also specifically regulate the activity of mitotic and nuclear proteins in a phosphorylation-dependent manner (Kumari et al., 2021).
Pin1 is predominantly expressed in cell nuclei and functions as a mitotic regulator, thus playing a vital role in DNA replication and mitosis (Zhang and Zhang, 2019; Kumari et al., 2021). The targets of Pin1 include not only proteins that exert function in transcription and the cell cycle, but also those involved in many physiological and pathological processes, including apoptosis, proliferation, and maintenance of the cytoskeleton (Thorpe et al., 2004; Lepore et al., 2021). Pin1 acts as a context-specific signal transducer based on specific environmental signals (Oster et al., 2021). Researchers have found that Pin1 plays a dual role in cellular apoptosis (Baik et al., 2015; Dubiella et al., 2021; Fagiani et al., 2021). For example, Pin1 was shown to enhance the anti-cell death ability of Bcl-2 and inhibit apoptosis by directly inactivating Bcl-2 associated X (Bax) (Bianchi et al., 2019; Makinwa et al., 2020). However, Pin1 has also been shown to act as a promoter of cell apoptosis by enhancing the expression of pro-apoptotic genes, such as p53 (Balaganapathy et al., 2018; Hleihel et al., 2021). Pin1 has been reported to be distributed in the central nervous system (CNS) and participate in a variety of neurodegenerative diseases, including Alzheimer’s disease (AD), Parkinson’s disease (PD), amyotrophic lateral sclerosis (ALS) and Huntington’s disease (HD) (Ghosh et al., 2013; Carnemolla et al., 2017; Iridoy et al., 2018; Napoletano et al., 2021; Samimi et al., 2021). An increasing body of evidence now supports the fact that Pin1 could exert a neuroprotective role or act as a promoter for neuronal death by upregulating survival-promoting factors or by downregulating death-suppression factors. However, the comprehensive regulatory pathways underlying the specific role of Pin1 in RCD has yet to be elucidated.
In this review, we set out to summarize our current knowledge of Pin1 in neuronal RCD and discuss the critical regulatory mechanisms associated with Pin1 in the CNS (Additional Table 1). In addition, Pin1 is an intriguing target for cancer therapy at present and could represent a desirable pharmaceutical target for neurological therapy. As such, we also summarize research relating to Pin1-specific inhibitors.
Additional Table 1.
Summary of Pin1 changes in neurodegenerative diseases
| Cell death type | Disease | Pin1 changes in disease | Main molecule in disease | Role of Pin in death | Reference |
|---|---|---|---|---|---|
| Apoptosis | AD | Decreased in AD patients | Pinl, GSK-3β, APP, HIF-lα, Aβ, p-Tau | Inhibits apoptosis | Lu et al., l999; Thorpe et al., 2004; Hashemzadeh-Bonehi et al., 2006; Pastorino et al., 2006; Kesavapany et al., 2007; Nowotny et al., 2007; Dakson et al., 20ll; O’Brien and Wong, 20l l; Lonati et al., 20l4; Azimi et al., 20l7; Xu et al., 20l7; Rong et al., 2017; Samimi et al., 202l; Nakhjiri et al., 2020; Li et al., 202la; Wang et al., 2020a; Zheng et al., 2021 |
| TBI | Decreased in TBI models | DAPK1, Pinl, p-Tau | Kim et al., 202l; Qiu et al., 202l; Tonnus et al., 202l | ||
| PD | Increased in PD patients | Pin1, α-synuclein, caspase-3 | Promotes apoptosis | Ghosh et al., 20l3; Matena et al., 20l3; Ibanez et al., 20l4; Ryo et al.,2006 | |
| HD | Increased in HD patients; Decreased in HD models |
Htt, Pinl, iASPP, p53 | Both inhibits and promotes apoptosis | Grison et al., 20ll; Agostoni et al., 20l6; Carnemolla et al., 20l7 | |
| Stroke | Increased in Stroke models | Pinl, FBX7, NICD1, p53 | Promotes apoptosis | Zacchi et al., 2002; Baik et al., 20l5; Arumugam et al., 20l8; Balaganapathy et al., 20l8; Li et al., 20l9 |
|
| SCI | Increased in SCI models | Pinl, Daxx, ASK1, BIMEL, JNK, Mcl-2, BAX, Bcl-2 |
Becker and Bonni, 2006, 2007; Ryo et al., 2007; Barone et al., 2008; Shen et al., 2009; Li et al., 20l7; Oh and Mouradian, 20l8; Zhang et al., 2021 |
||
| Epilepsy | Decreased in epileptic models | CaMKII, AMPA and NMDA receptors | Inhibits apoptosis | Becker and Bonni, 2007; Tang et al., 20l7; Hou et al., 202l | |
| Other neurodegenerative diseases | Decreased in age-related neurodegeneration patients | Lipofuscin | Hashemzadeh-Bonehi et al., 2006 | ||
| RN | Brain ischemia and traumatic injury | Increased in brain ischemia and traumatic injury models | Calcium, DAPK, Pinl | Promotes RN | Bialik and Kimchi, 20ll; Del Rosario et al., 20l5; Chen et al., 20l7; Ibarra et al., 2017; Wangetal., 20l7 |
| Retinal disease | Increased in retinal disease | Calcium, CaM, CaMKII, Pinl, CAST, Calpain2, AIF | Wang etal., 20l7, 20l9a, b; Cheng etal., 20l8; Hou etal., 202l | ||
| Autophagy | AD | Decreased in AD patients | Pinl, Akt, Ubiquitin, GSK-3β, Aβ, LC3-II | Promotes autophagy | Min et al., 200S;_Ma et al., 2012; Del Rosario et al., 20l5; So and Oh, 20l5; Kim et al., 2009; Chao et al., 202l |
AD: Alzheimer’s disease; AIF: apoptosis-induced factor; AMPA: α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; APP: amyloid precursor protein; ASK1: apoptosis signal-regulating kinase 1; Aβ: β-amyloid peptide; BAX: Bcl2-associated X; Bcl-2: B-cell lymphoma 2; BIMEL: Bcl-2-interacting mediator of cell death, extra long; CaM: calmodulin; CaMKII: calmodulin-dependent protein kinase II; CAST: calpastatin; DAPK1: death-associated protein kinase 1; Daxx: death domain associated protein; FBW7: F-box and WD repeat domain containing domain protein 7; GSK-3β: glycogen synthase kinase-3β; HD: Huntington's disease; HIF-1: Hypoxia-inducible transcription factor-1; Htt: Huntingtin; iASPP: inhibitor of apoptosis-stimulating protein of p53; JNK: C-Jun N-terminal kinase; LC3: long chain 3; Mcl-1: myeloid cell leukemia sequence-1; NICD1: Notch intracellular domain 1; NMDA: N-methyl-D-aspartic acid receptor; PD: Parkinson's disease; Pin1: prolyl cistrans isomerase NIMA-interacting 1; RN: regulated necrosis; SCI: spinal cord injury; TBI: traumatic brain injury.
Search Strategy
We searched all literatures published from June 1998 to May 2021 that relates to the role of Pin1 in neuronal death (Figure 1) in this narrative review. We performed a PubMed search using the term “Pin1” AND “neuron” to identify potential neuronal death in neurodegenerative diseases that are mediated by Pin1 dysfunction. We next screened the results by the title, keywords, abstract and excluded non-neuronal death-related articles, and retrieved further papers by citation tracking.
Figure 1.
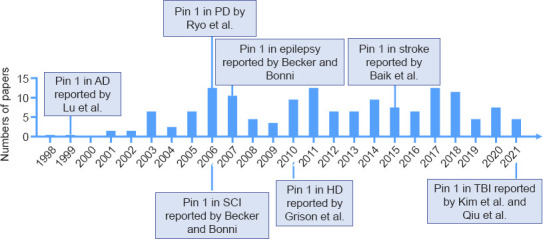
Timeline of the milestones of Pin1-mediated neuronal death in neurodegenerative diseases.
AD: Alzheimer’s disease; HD: Huntington’s disease; PD: Parkinson’s disease; Pin1: Prolyl cistrans isomerase NIMA-interacting 1; SCI: spinal cord injury; TBI: traumatic brain injury.
Apoptosis
Apoptosis is controlled by several apoptotic genes and characterized by RCD (Bai et al., 2020; Hu et al., 2021a; Nascimento et al., 2022). Traditionally, apoptosis can be divided into two pathways, the extrinsic and intrinsic pathways (Carneiro and El-Deiry, 2020; Nascimento et al., 2022). In the extrinsic pathway, apoptosis is induced by extracellular signals which then trigger the activation of caspases and caspase-mediated pathways (Carneiro and El-Deiry, 2020; Nascimento et al., 2022). The intrinsic pathway is activated by intraocular stimulation, such as signals from the mitochondria; this leads to the activation of caspases and eventually causes the degradation of vital proteins, thus inducing apoptosis (Carneiro and El-Deiry, 2020; Nascimento et al., 2022). The role of Pin1 in neuronal apoptosis has been investigated in many studies (Figure 2) (Becker and Bonni, 2007; Moujalled et al., 2021). For example, Pin1 has been reported to mediate neuronal apoptosis by promoting the expression of pro-apoptotic genes, such as p53 (Grison et al., 2011; Balaganapathy et al., 2018). Literature indicates that Pin1 may participate in neuronal survival or apoptosis, depending on the specific development stages of neurons as well as the different tissues and diseases involved. In this regard, herein, we discuss the different roles of Pin1 in apoptosis in different neurodegenerative diseases.
Figure 2.
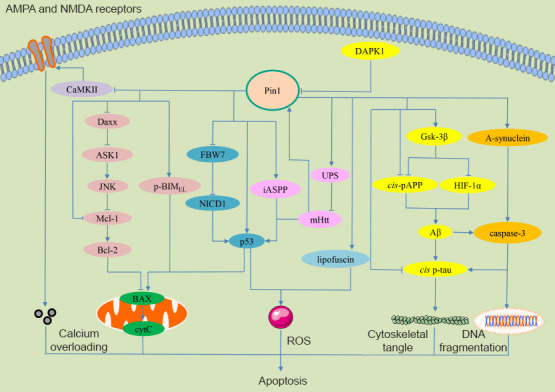
The molecular mechanisms underlying Pin1-regulated apoptosis signaling pathways.
In AD, reduced levels of Pin1 inhibits GSK-3β enzymatic activity; this could inhibit HIF-1 protein degradation and contribute to the expression of the cis isoform of pT668-APP. Next, the increased production of Aβ accelerates the formation of the cis isoform of pT231-Tau, finally promoting the progression of AD. Furthermore, caspase-3 can be activated by the accumulation of Aβ and promotes the formation of the cis isoform of pT231-Tau. In TBI, increased levels of DAPK1 inhibits Pin1 and then promotes the transformation of cis pT231-Tau as well as TBI. In PD, the overexpression of Pin1 facilitates the formation and stability of α-synuclein aggregation and promotes caspase-3 activation, finally leading to PD. In HD, Htt directly increases the activation of p53 and Pin1. Pin1 also interacts with p53 and triggers dissociation from its inhibitor iASPP. In addition, the overexpression of Pin1 leads to a reduction of Htt accumulation via UPS-mediated degradation. In stroke, increased levels of Pin1 promotes the stability of NICD by inhibiting FBW7-mediated poly-ubiquitination, up-regulates p53 transactivation, and enhances the production of ROS. In SCI, Pin1 promotes the degradation of Daxx and then activates the ASK1/JNK signaling pathway; this could promote Mcl-1 degradation and Bcl-2 inhibition. Pin1 also binds with and mediates a conformational change in pSer65-BIMEL to protect pS65-BIMEL from proteasomal degradation, finally leading to neuronal apoptosis. In epilepsy, the deletion of Pin1 enhanced AMPA and NMDA receptors by phosphorylating CaMKII, finally inducing excitotoxic neuronal death. In age-related neurodegeneration, Pin1 is progressively associated with lipofuscin, thus resulting in the generation of ROS; this process can be deleterious to neurons. AD: Alzheimer’s disease; AMPA: α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; APP: amyloid precursor protein; ASK1: apoptosis signal-regulating kinase 1; Aβ: β-amyloid peptide; BAX: Bcl-2-associated X; Bcl-2: B-cell lymphoma 2; BIMEL: Bcl-2-interacting mediator of cell death, extra long; CaMKII: calmodulin-dependent protein kinase II; cyt C: cytochrome C; DAPK1: death-associated protein kinase 1; Daxx: death domain associated protein; FBW7: F-box and WD repeat domain containing domain protein 7; GSK-3β: glycogen synthase kinase-3β; HD: Huntington’s disease; HIF-1: hypoxia-inducible transcription factor-1; Htt: Huntingtin; iASPP: inhibitor of apoptosis-stimulating protein of p53; JNK: C-Jun N-terminal kinase; Mcl-1: myeloid cell leukemia sequence-1; NICD1: Notch intracellular domain 1; NMDA: N-methyl-D-aspartic acid receptor; PD: Parkinson’s disease; Pin1: prolyl cistrans isomerase NIMA-interacting 1; SCI: spinal cord injury; TBI: traumatic brain injury; UPS: ubiquitin-proteasome system.
Pin1 mediates neuronal apoptosis in AD
AD is a leading dementia disease and affects the global population. AD is characterized by the accumulation of β-amyloid peptide (Aβ) with hyper-phosphorylated tau (p-Tau), a microtubule associated protein (Azimi et al., 2017; Gu and Liu, 2020; Li et al., 2021b; Blanchard et al., 2022). Lu et al. (1999) were the first to report that Pin1 was involved in the process of AD. In the normal human brain, Pin1 is mainly distributed in the neuronal nuclei. However, in AD patients, Pin1 is localized in both the neuronal cytoplasm and perikaryal neurofibrillary tangles (NFTs); furthermore, the levels of Pin1 are significantly reduced (by 39%) in the brains of AD patients when compared to normal brains (Pastorino et al., 2006; Samimi et al., 2021). Analysis of Pin1 knockout mice showed that the accumulation of p-Tau, neuronal loss, and behavioral deficits appeared together (Thorpe et al., 2004; Azimi et al., 2017). Other research has shown that Pin1 is able to bind to the phosphorylated Thr231 motif of Tau (pT231-Tau) and can accumulate in NFTs within the neuronal cytoplasm (Lu et al., 1999). In addition, Pin1 can ameliorate p-Tau pathology by isomerizing p-Tau, thus recovering its ability to bind to and restore cytoskeletal integrity (Hashemzadeh-Bonehi et al., 2006; Kesavapany et al., 2007). It has been reported that p-Tau exists in two isoforms, physiological trans and pathological cis (Wang et al., 2020a; Qiu et al., 2021). The trans form of p-Tau exhibits normal functionality and may undergo degradation. In contrast, the cis form of p-Tau is abnormal and cannot be degraded; this form has a high tendency for aggregation (Zheng et al., 2021). Pin1 can accelerate the transformation of the cis isoform to the trans isoform of pT231-Tau and restore its normal function. In fact, cis pT231-Tau is also significantly increased in human AD brains and is strongly correlated with NFTs and reduced levels of Pin1 (Nakhjiri et al., 2020; Wang et al., 2020a; Qiu et al., 2021).
A large body of evidence now suggests that interaction between Aβ and Tau could enhance the process of AD (Selkoe and Hardy, 2016; Shentu et al., 2018). For example, Pastorino et al. (2006) reported that Pin1 could modulate the production of Aβ production. Enhanced Aβ production can also promote the phosphorylation of Tau and the generation of NFTs (Dakson et al., 2011). In particular, Pin1 binds specifically to the phosphorylated Thr668-Pro motif of amyloid precursor protein (APP) (pT668-APP) and catalyzes pT668-APP from cis to trans transformation (Nowotny et al., 2007; Dakson et al., 2011). The cis pT668-APP isoform promotes the processing of Aβ. By catalyzing from a cis to trans isoform, Pin1 could inhibit the processing of Aβ (Nowotny et al., 2007; Dakson et al., 2011). The deletion of Pin1 in mice has been linked to the accumulation of Aβ and neurotoxicity (Wang et al., 2020a; Li et al., 2021a). Some other reports have also indicated that molecules leading to Tau phosphorylation, including caspase-3, can also be activated by Aβ accumulation, finally resulting in neuronal death and the progression of AD process (O’Brien and Wong, 2011; Rong et al., 2017). Glycogen synthase kinase-3β (GSK-3β), a serine/threonine protein kinase, plays a role in several vital cellular functions, such as growth and glycogen metabolism and can also promote hypoxia-inducible transcription factor-1 degradation (Flügel et al., 2012). Recently, Lonati et al. (2014) reported that in cortical neurons, hypoxia-inducible transcription factor-1 was maintained at higher levels in the brains of AD patients; this was coincident with reduced levels of GSK-3β protein. Interestingly, these authors found that reduced levels of Pin1 inhibited GSK-3β enzymatic activity, ultimately inhibiting hypoxia-inducible transcription factor-1 protein degradation and contributing to Aβ production and progression in AD patients (Lonati et al., 2014). As the levels of Pin1 are decreased in AD patients and because Pin1 plays a vital role in AD processing in animal models of AD, it follows that the down-regulation of Pin1 might be an early potential biomarker for the progression of AD (Xu et al., 2017).
Pin1 mediates neuronal apoptosis in TBI
TBI includes closed-head injuries (in which the skull and dura are intact), penetrating injuries (in which skull and dura are broken), and injury to the brain parenchyma (Wood, 2018). TBI is characterized by acute neuronal dysfunction and is thought to be a non-aging risk factor for AD (Wood, 2018). Tau pathology is also a pathophysiological factor in TBI; patients with TBI have higher levels of tau (Shin et al., 2021). Furthermore, recent studies have shown that in the brains of TBI patients, Pin1 is widely distributed in the neurons and correlates with neurodegenerative pathologies (Kim et al., 2021; Qiu et al., 2021). Other research has shown that cis pT231-Tau, but not the trans form, contributes to neuronal pathology and functional impairment in a model of TBI (Albayram et al., 2017, 2018; Qiu et al., 2021; Tonnus et al., 2021). Recent studies showed that death-associated protein kinase 1 (DAPK1) plays a critical role in regulating cis pT231-Tau after TBI (Kim et al., 2021; Qiu et al., 2021). DAPK1 is a calcium/calmodulin-dependent serine/threonine (Ser/Thr) kinase that plays a critical role in neuronal death. DAPK1 has been shown to increase significantly in the brains of a mouse model of TBI and then promotes the induction of cis pT231-Tau (Kim et al., 2021; Qiu et al., 2021). Other research has shown that DAPK1 is a key regulator of the activity of Pin1. DAPK1-mediated cis pT231-Tau transformation has been proven to be regulated by Pin1 in the brain after injury (Kim et al., 2021; Qiu et al., 2021). Following brain damage, the activity of Pin1 is known to decrease; this is accompanied by an increase in the expression of cis pT231-Tau, thus promoting neuronal death (Kim et al., 2021; Qiu et al., 2021). Furthermore, the pharmacological inhibition of DAPK1 activity significantly increases the levels of Pin1 and significantly decreases the expression of cis pT231-Tau and the extent of neuronal injury (Kim et al., 2021; Qiu et al., 2021). Thus, DAPK1-Pin1-cis pT231-Tau is a novel regulatory pathway in TBI, and exerts an important effect on the progression of TBI. Thus, the targeting of this pathway might be a potential therapeutic method for TBI (Tonnus et al., 2021). It should be noted, however, that the levels of Tau change in both AD and TBI (Blennow et al., 2016). In AD, Aβ aggregation induces tau phosphorylation and accumulation. However, in TBI, Aβ has no effect on TBI-related tau accumulation. Thus, the mechanisms of tau accumulation in TBI might be distinct from AD (Blennow et al., 2016).
Pin1 mediates neuronal apoptosis in PD
PD is a disease that is characterized by the loss of dopaminergic neurons and the formation of α-synuclein aggregates in Lewy bodies. Pin1 also has been reported to be implicated in the pathogenesis of PD (Matena et al., 2013; Ibáñez et al., 2014). Ryo et al. (2006) were the first to report that Pin1 is located in the Lewy bodies of PD patients. In particular, the overexpression of Pin1 may facilitate the formation and stability of α-synuclein aggregation in experimental models of PD (Ryo et al., 2006). Other research has shown that Pin1 is highly up-regulated in dopaminergic cells and nigrostriatal cells after Parkinsonian neurotoxicant 1-methyl-4-phenylpyridinium treatment (Ghosh et al., 2013). Furthermore, Pin1 knockdown or functional inhibition almost completely prevented 1-methyl-4-phenylpyridinium-induced caspase-3 activation, DNA fragmentation and neuronal death, thus indicating that Pin1 has a pro-apoptotic role and causes α-synuclein aggregation (Ghosh et al., 2013). However, until now, the precise regulatory mechanism underlying the upregulation of Pin1 in PD has yet to be fully elucidated. Ghosh et al. (2013) reported that the upregulation of Pin1 may be directly due to neurotoxic stress and may then contribute to the death of dopaminergic neurons. Therefore, the overexpression of Pin may mediate apoptosis and may represent a critical neurotoxic event in the pathogenesis of PD.
Pin1 mediates neuronal apoptosis in HD
HD is an autosomal dominant neurodegenerative disease caused by an expanded CAG repeat in the gene encoding huntingtin (Htt) protein; this condition is characterized by the death of medium spiny neurons in the striatum (Tabrizi et al., 2020). It has been reported that mutated Htt binds to p53, increases the levels of p53 protein, enhances the phosphorylation of p53 at Ser46 and evokes DNA damage in the striatum and cerebral cortex of patients with HD (Grison et al., 2011). It is important to note that the functionality of Pin1 plays a key role in stimulating the pro-apoptotic role of p53 (Agostoni et al., 2016). First, mutated Htt promotes the activation of Pin1. Subsequently, Pin1 interacts with p53 and triggers dissociation from its inhibitor (inhibitor of apoptosis-stimulating protein of p53; iASPP), finally promoting the expression of a series of apoptotic genes (Grison et al., 2011). However, in contrast, another study found that Pin1 might act as a negative regulator for Htt accumulation in a genetic mouse model of HD (Carnemolla et al., 2017). These authors found that the overexpression of Pin1 led to a reduction of Htt accumulation and that this might occur via degradation by the ubiquitin-proteasome system (Carnemolla et al., 2017). Therefore, the role of Pin1 in neuronal apoptosis might have a different effect in that it may stimulate Htt clearance-mediated neuronal protection and promote p53 mediated-apoptosis (Grison et al., 2011). Furthermore, by using a genetic model of Pin1 deletion, researchers found that the role of Pin1 in neuronal death might be time-dependent (Agostoni et al., 2016). In the early stages, Pin1 activation reduces the DNA damage response. However, in the middle stages of life, the activation of Pin1 might be triggered by Htt, thus initiating the role of Pin1 in p53-mediated neuronal apoptosis. In aging individuals, Pin1 significantly increases the amount of Htt accumulation (Agostoni et al., 2016). In summary, the role and mechanisms of Pin1 in neuronal apoptosis provides some support for the pathogenesis of HD. However, the role of Pin1 in HD still requires further research.
Pin1 mediates neuronal apoptosis in stroke
Stroke is a disease involving sudden impairment of consciousness and motio; this condition is caused by the obstruction or rupture of blood vessels in the brain (Magnusson et al., 2014). Baik et al. (2015) reported that Pin1 contributes to the process of stroke through Notch signaling. The Notch signaling pathway plays a vital role in the development of the CNS, including neuronal differentiation, determination, and certain neural pathological diseases (Magnusson et al., 2014). Notch signaling has also been implicated in neuronal death (Arumugam et al., 2006; Magnusson et al., 2014). It has been reported that the levels of Pin1 and Notch intracellular domain 1 (NICD1), an active domain of Notch, were both increased following stroke (Baik et al., 2015). Further research showed that the overexpression of Pin1 could promote NICD1 stability and its pro-apoptotic function after stroke (Baik et al., 2015). It was also found that Pin1 binds to and stabilizes NICD1 domain through the WW domain (Baik et al., 2015). Following stroke, Pin1 interacts with NICD1 and enhances its stability by inhibiting F-box and WD repeat domain containing domain protein 7-mediated poly-ubiquitination, ultimately leading to NICD1-induced neuronal apoptosis (Wang et al., 2014; Baik et al., 2015). Other research indicated that Pin1 regulates p53 transactivation and the upregulation of pro-apoptotic genes, such as Bax, via the activation of NICD1, finally promoting neuronal apoptosis and stroke (Balaganapathy et al., 2018). The tumor suppressor p53 is an important decision maker in the cell cycle and apoptosis and responds to a series of stimulations. It has been reported that the phosphorylated form of p53 induced by stress combines with the WW-domain of Pin1 to form a complex (Arumugam et al., 2018; Balaganapathy et al., 2018). Next, phosphorylated p53 undergoes a conformational change that is catalyzed by Pin1, thus enhancing the transactivation activity of p53 (Zacchi et al., 2002; Balaganapathy et al., 2018). The interaction between NICD1 and p53 is an important signaling step that induces neuronal apoptosis in stroke (Arumugam et al., 2018). The disturbance of NICD1/p53 interaction could ameliorate the process of stroke (Balaganapathy et al., 2018). In addition, some other reports indicated that the production of reactive oxygen species (ROS) was a critical effector in the Pin1-Notch-p53 pathway and plays a role in the activation of apoptosis in stroke (Li et al., 2019).
Pin1 mediates neuronal apoptosis in SCI
SCI is a severe trauma that causes severe injury to the function of the motor and sensory neurons of the spinal cord and generally is associated with a poor prognosis (Yuan et al., 2021). Myeloid cell leukemia sequence-1 (Mcl-1), an anti-apoptotic factor of the Bcl-2 family, remains phosphorylated in the basal state (Carlet et al., 2021). In normal neurons, Pin1 binds with pT163-Mcl-1 and inhibits Mcl-1 ubiquitination and degradation (Li et al., 2017). However, after SCI injury, Mcl-1 undergoes degradation following the removal of Pin1, thus resulting in Bcl-2 inhibition and the release of cytochrome C into the cytosol (Li et al., 2007). Furthermore, the activation of c-Jun N-terminal kinase (JNK) induced by SCI can perturb the interaction between Pin1 and phosphorylated Mcl-1, thereby promoting the release of cytochrome C (Li et al., 2007). The JNK pathway plays a complicated role in many physiological and pathological processes, such as cell apoptosis and differentiation, and may be activated by various stress signals and inflammation (Zhang et al., 2021). JNK also can promote the mitochondrial apoptotic pathway by regulating the release of BAX under the control of Pin1 (Shen et al., 2009). Other reports have also indicated that Pin1 binds to the Ser178-Pro motif in phosphorylated death domain associated protein and promotes the degradation of death domain associated protein via ubiquitination (Ryo et al., 2007). Degraded death domain associated protein can interact with apoptosis signal-regulating kinase 1 and then activate the apoptosis signal-regulating kinase 1/JNK signaling pathway (Ryo et al., 2007; Oh and Mouradian, 2018). Most importantly, Pin1 also binds with phosphorylated Bcl-2-interacting mediator of cell death, extra-long (BIMEL) at Ser65 and JNK‑interacting protein 3 to form a complex (Becker and Bonni, 2006, 2007). Pin1 also mediates the isomerization of BIMEL and leads to a conformational change in pSer65-BIMEL (Becker and Bonni, 2006, 2007). It has also been reported that the phosphorylation of BIMEL at Ser65 in non-neural cells can promote the degradation of BIMEL by the proteasome (Ley et al., 2004; Becker and Bonni, 2006). Although the presence of Pin1 in a neuron-specific JNK signaling complex also allows Pin1 to bind with BIMEL, Pin1 could promots a conformational change in pS65-BIMEL and protect pS65-BIMEL from proteasomal degradation in neurons, finally leading to neuronal apoptosis (Becker and Bonni, 2006; Barone et al., 2008). It should be noted that phosphorylated BIMEL elicits distinct effects depending on cell type, thus leading to neuronal apoptosis and the prevention of non-neuronal apoptosis (Becker and Bonni, 2006, 2007). In neurons, Pin1 activates the mitochondrial apoptotic machinery; this might be caused by Pin1 coming into close proximity with its substrate BIMEL, which is localized to the mitochondrial membrane. In non-neuronal cells, Pin1 is mainly localized in the nucleus. Pin1 and BIMEL are localized in distinct cell compartments; this might allow for the degradation of BIMEL (Becker and Bonni, 2006, 2007; Barone et al., 2008).
Pin1 mediates neuronal apoptosis in epilepsy
Epilepsy is a neurodegenerative disorder that is characterized by repeated seizures caused by the uncontrolled and abnormal firing of neurons (Liu et al., 2019b). Early studies indicated that the expression of Pin1 was remarkably reduced in epileptic patients (Becker and Bonni, 2007; Tang et al., 2017). Studies of an epileptic mouse model suggested the interaction of Pin1 with N-methyl-D-aspartic acid receptors and that this might be associated with the excitotoxicity of pyramidal neurons (Tang et al., 2017). Excitotoxicity is a toxic event induced by an excess of excitatory amino acids receptors which leads to excitotoxic neuronal death (Liu et al., 2019b). This report was further confirmed by other researchers who found that Pin1 deletion led to a significant increase in seizure susceptibility in chemical-induced epileptic mouse models (Becker and Bonni, 2007). Pin1 deletion was also shown to enhance the phosphorylation of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors by phosphorylating calcium/calmodulin-dependent protein kinase II (CaMKII) (Becker and Bonni, 2007). In a clinical study, the levels of Pin1 were found to be reduced, while the levels of p-CaMKII and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid were significantly increased in the neocortex of epileptic patients (Tang et al., 2017). Moreover, in a mouse model of epilepsy, the re-expression of Pin1 in Pin1-deletion mice led to a significant down-regulation of p-CaMKII and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; this was accompanied by the effective suppression of seizure susceptibility (Tang et al., 2017). Thus, Pin1 is an important controlling effector in epilepsy; this indicates that Pin1 is an attractive therapeutic strategy for epilepsy (Hou et al., 2021).
Pin1 mediates neuronal apoptosis in other neurodegenerative diseases
It has been reported that Pin plays an important role in age-related neurodegeneration. Lipofuscin is an age-associated pigment and a cytological characteristic of post-mitotic cell aging (Van Houten, 2019). Hashemzadeh-Bonehi et al. (2006) found that the levels of Pin1 were progressively associated with lipofuscin in the intracellular granules of aging neurons. These researchers also found that Pin1 can be modified and become dysfunctional post-translationally. As a result, the binding of Pin1 could be cleared via the endosomal pathway as the micro-environment was not suitable for Pin1 (Hashemzadeh-Bonehi et al., 2006). Eventually, this binding could reduce the amount of soluble Pin1 in neurons, thus resulting in oxidative stress; this could have a deleterious effect on neurons during aging (Hashemzadeh-Bonehi et al., 2006).
Regulated Necrosis
Under chronic neurodegenerative diseases and accident conditions, such as ischemic injury, cells may undergo morphological changes involving necrosis (Charidimou et al., 2020). Traditional views consider that necrosis is a type of passive and uncontrolled cell death that occurs around the damaged tissues. However, in recent years, some reports have indicated that, in addition to the classic form of necrosis, there is a type of procedural necrosis that can be regulated but undergoes classicical necrotic morphological changes (Degterev et al., 2005; Yuan et al., 2019). In 2005, Yuan’s team (Degterev et al., 2005; Yuan et al., 2019) first proposed a new term to define this form of RN, and referred to this as necroptosis, also known as programmed necrosis. RN is known to play vital roles in various physiological and pathophysiology processes, such as neurodegenerative disorders. Thus far, a series of different types of RN have been identified, including receptor interacting protein 3-mediated necroptosis that can be induced by tumor necrosis factor, pyroptosis that relies on the activation of caspase-1 and is triggered by microbial infection, aerastin-mediated ferroptosis that requires excessive iron, and mitochondrial permeability transition dependent RN (Yuan et al., 2019; Alu et al., 2020; Chen et al., 2020; Liao et al., 2021; Tonnus et al., 2021). Although these types of RN are named distinctly, their regulatory signaling pathways exhibit some degree of crosstalk (Alu et al., 2020). For example, in some stimuli, such as excitatory amino injury and ischemia, calcium overloading could result in different types of RN (Ros et al., 2017). However, the precise control mechanisms underlying RN are still unclear and should be further investigated. Over recent years, the relationship between Pin1 and RN has been described in certain neurodegenerative disorders; for example, Pin1 knockout mice showed significantly increased levels of retinal neuronal necrosis (Figure 3) (Kuboki et al., 2009; Wang et al., 2017, 2019a). In this section, we discuss the role of Pin1 in RN and certain neurodegenerative disorders.
Figure 3.
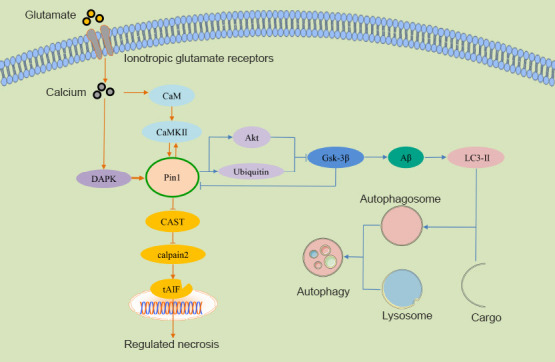
The molecular mechanisms responsible for how Pin1 regulates RN and autophagy signaling pathways.
In RN in brain ischemia and traumatic injury, Pin1 acts as an important down-regulator of DAPK1-induced excitotoxic necrosis. The overexpression of DAPK1 resulted in a strong and significant enhancement of necrotic neurodegeneration in postsynaptic neurons; this was mediated by CaMK activation and the excessive influx of ions into the neurons. The DAPK1-dependent necrosis of postsynaptic neurons can be triggered by an excessive influx of ions. In RN in retinal diseases, Pin1 activation, induced by activated ionotropic glutamate receptor-mediated CaMKII and CaMKII activation, was shown to inhibit and enhance calpain-2 activity; this activated AIF, ultimately resulting in neuronal RN. In autophagy in AD, the inhibition of Pin1 promotes GSK-3β expression by inhibiting proteasome and Akt activation, and stimulating cell death via autophagy, as shown by increased levels of LC3-II. AD: Alzheimer’s disease; AMPA: α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; Aβ: β-amyloid peptide; CaM: calmodulin; CaMKII: calmodulin-dependent protein kinase II; CAST: calpastatin; GSK-3β: glycogen synthase kinase-3β; NMDA: N-methyl-D-aspartic acid receptor; NMDAR: NMDA receptor; Pin1: prolyl cistrans isomerase NIMA-interacting 1; RGCs: retinal ganglion cells; RN: regulated necrosis; tAIF: truncated apoptosis-induced factor.
Pin1 mediates neuronal RN in brain ischemia and traumatic injury
As discussed above, Pin1 has been shown to be an important regulator of neurodegenerative diseases, such as AD. It has been suggested, that in the process of AD, DAPK1 acts up-stream of Pin1 (Bialik and Kimchi, 2011). Studies have found that the over-expression of DAPK1 resulted in a strong and significant enhancement of necrotic neurodegeneration in postsynaptic neurons and that this was mediated by CaMK activation and the excessive influx of ions into the neurons (Del Rosario et al., 2015; Wang et al., 2017). Several studies have reported that DAPK1 is involved in excitotoxicity, although the regulatory mechanisms involved remain unclear. Some reports have indicated that DAPK1 acts as a modulatory adaptor to control the choice between necrosis and autophagy (Prokesch et al., 2017; Singh and Talwar, 2017). However, in acute brain injury conditions, there was no significant change in the levels of light chain 3 in neurons that overexpressed DAPK1, thus demonstrating that DAPK1 may not play a role in autophagy in such acute neurodegenerative conditions (Stevens and Hupp, 2008; Huang et al., 2014; Wang et al., 2017). Consistent with this result, the blockade of autophagy had no significant effects on neurodegeneration in DAPK1 knockout neurons. In this condition, DAPK1-dependent necrosis of the postsynaptic neurons could be triggered by the excessive influx of ions (Wang et al., 2017). These authors also found that Pin1 acts as an important down-regulator in DAPK1-induced excitotoxic necrosis (Wang et al., 2017). Collectively, these results suggested that DAPK1 contributes to cell necrosis but not autophagy, which is Pin1 dependent. Notably, several studies have shown that Pin1 is an up-regulator of DAPK1 in neuronal development (Chen et al., 2017). This may be because the signaling cascades might be dependend on the exact scenario under different conditions (Del Rosario et al., 2015). Future studies now need to determine why the relationship between upstream and downstream of DAPK1 and Pin1 differ in a manner that is dependent on the cellular context (Ibarra et al., 2017).
Pin1 mediates neuronal RN in retinal diseases
The dysfunction of Pin1 is thought to cause retinal neuronal necrosis in glaucoma. Globally, glaucoma is the second most common eye disease that causes blindness and is characterized by the loss of retinal ganglion cells (Erickson, 2021; Huang et al., 2021). Various contributors, such as ischemia, axonal transport failure, and excitatory amino acids toxicity, are thought to be major risks for the death of retinal ganglion cells. In previous studies, Wang et al. (2017, 2019b) found that calpain-2 (m-calpain) played a regulatory role in the necrosis of retinal ganglion cells following glutamate excitotoxicity. Calpains are a ubiquitous form of calcium-dependent protease and have been widely investigated in different types of cellular processes, such as cell survival, proliferation, and death. Calpain-1 (u-calpain) and calpain-2, which require micromolar and millimolar concentrations of calcium for activation respectively, are the two main forms of calpains in the CNS (Wang et al., 2013, 2017). Calpastatin (CAST) is an endogenous specific inhibitor of calpains (Wang et al., 2013; Cheng et al., 2018). Following glutamate excitotoxicity injury, calpain-2, but not calpain-1, is activated and cleaves apoptosis-induced factor which can then translocate to the nucleus and induce DNA degradation, ultimately resulting in neuronal RN (Wang et al., 2017, 2019a). These opposing changes in calpain-1 and calpain-2 may be due to the differential roles of calpain-1 and calpain-2 after glutamate excitotoxicity (Wang et al., 2017, 2019a, b). As Wang et al. (2013, 2017, 2019a, b) reported, calpain-1 is coupled with the synaptic N-methyl-D-aspartic acid receptor and plays a protective role after activation, while calpain-2 is coupled with extrasynaptic N-methyl-D-aspartic acid receptor and is deleterious after activation. Therefore, it is possible that the protective role of calpain-1 may be impaired after glutamate injury. In a further study, Cheng et al. (2018) showed that Pin1 could interact with and inhibit CAST in retinal neurons. Following glutamate injury, the interaction of Pin1 with CAST was enhanced, thus leading to a reductive inhibitory effect of CAST on calpain-2 (Wang et al., 2017). However, the inhibition of Pin1 expression and activation by small interference RNA and juglone (a specific inhibitor of Pin1 was shown to significantly enhance the activation of CAST; in turn, this recovers the inhibitory role on calpain-2 (Wang et al., 2017). Furthermore, Wang et al. (2019a) found that Pin1 is an important down-effector of CaMKII. Following glutamate injury, Pin1 activated ionotropic glutamate receptors and led to calcium overloading (Wang et al., 2019a). Subsequently, the overload of calcium activated CaMKII and CaMKII-meditated Pin1 activation (Wang et al., 2019a). Other research reported that Pin1 can upregulate CaMKII and Pin1 expression and effectively reduce the activation of CaMKII to finally suppress seizure susceptibility (Hou et al., 2021). The upstream and downstream relationship between Pin1 and CaMKII may depend on the cellular and disease context (Hou et al., 2021). Further research should aim to specifically investigate the function of Pin1 in retinal-related diseases.
Autophagy
Autophagy is an important physiological and pathological process that degrades intracellular dysfunctional and degenerated proteins and damaged organelles to become autophagosomes and then fuse with lysosomes to form autolysosomes (Chao et al., 2021). Under normal conditions, autophagy plays a critical role in cellular homeostasis, such as cell survival, renewal and recycling, as autophagy is the primary pathway for recycling biomolecules and organelles by degradation (Chao et al., 2021). Autophagy is also considered as a vital factor in a range of neurodegenerative conditions, such as AD (Levine and Kroemer, 2008; Rami, 2009; Guo et al., 2021). However, in contrast to the role of Pin1 in apoptosis, the molecular mechanisms of Pin1 regulation in autophagy signaling pathways and its role in neurodegeneration still remain unclear and need to be investigated (Figure 3).
Recent studies have revealed the underlying connections between Pin1-mediated autophagy and neuronal toxicity in AD (Del Rosario et al., 2015; Chao et al., 2021). It has been shown that increased levels of APP expression could cause AD. Reduced levels of Pin1 and increased levels GSK-3β, a 47-kDa isoform of the GSK3 family, were previously detected in the brains of AD patients and shown to be involved in the processing of APP processing (Del Rosario et al., 2015). It has also been reported that APP can be phosphorylated by GSK-3β, and that the activation of GSK-3β increases the production of APP and toxicity (Zhou et al., 2021). Further research showed that Pin1 could bind to the phosphorylated Thr330 motif of GSK-3β and inhibits its kinase activity in human neuronal and glioma cells (So and Oh, 2015). Pin1 deficiency or deletion of the binding site in GSK-3β led to an increase in GSK-3β activity, the increased stability of APP, and the production of toxic APP, thus suggesting the important role of Pin1 in neuronal death (Del Rosario et al., 2015). In addition, the over-expression of Pin1 can promote the turnover of APP protein by binding and inhibiting GSK-3β kinase activity, thus providing a novel mechanism for Pin1 in protection against AD (Min et al., 2005; Ma et al., 2012). Akt has also been identified as an intermediary between Pin1 and GSK-3β (Kim et al., 2009). GSK-3β is a target of Akt and can be inhibited by Akt activation (Kim et al., 2009). Eventually, Pin1 could affect the levels of GSK-3β protein by regulating Akt (So et al., 2015). In addition, Pin1 has been considered as a regulator of the proteasome (So and Oh, 2015). The inhibition of Pin1 restored GSK-3β expression by inhibiting proteasome degradation, and stimulated cell death via autophagy, as demonstrated by increased levels of light chain 3-II (So and Oh, 2015). It should be noted that the inhibition of GSK-3β induces the activation of Pin1. Therefore, these data suggest that GSK-3β may also be a mediator for the regulation of Pin1 activity in autophagy (Chao et al., 2021). Collectively, this data suggests that Pin1-GSK-3β pathway-mediated neuronal toxicity acts via autophagy in AD.
Inhibitors of Pin1 and Potential Clinical Applications
Pin1 is a potential therapeutic target for neurological diseases (Zabłocka et al., 2021). By screening a series of compound libraries, a number of Pin1 inhibitors have been identified that could provide protective effects by inhibiting the activity of Pin1 PPIase. According to their specific effects on Pin1 PPIase domain activity, Pin1 inhibitors can be classified as either covalent or non-covalent types (Pinch et al., 2020). After binding to the Pin1 PPIase domain, covalent Pin1 inhibitors, such as Juglone and KPT-6566, can induce a covalent modification of the thiol group of cysteine residues in the Pin1 PPIase domain (Campaner et al., 2017; Bedouhene et al., 2021). Via these covalent modifications, covalent Pin1 inhibitors can irreversibly block the activity of the Pin1 PPIase domain (Pinch et al., 2020). Most of the Pin1 inhibitors are non-covalent types, such as 2-(3-chloro-4-fluoro-phenyl)-isothiazol-3-one (TME-001), diethyl-1,3,6,8-tetrahydro-1,3,6,8-tetraoxobenzol-phenanthroline-2,7-diacetate (PiB), all-trans retinoic acid (ATRA), and arsenic trioxide (ATO) (Mori et al., 2011; Kozono et al., 2018; Bedouhene et al., 2021). Non-covalent Pin1 inhibitors can also bind to the Pin1 PPIase domain, but inhibit Pin1 activity in a competitive manner (Kozono et al., 2018). However, the Pin1 inhibitors identified thus far are associated with limitations that restrict their use in clinical treatments (Additional Table 2). Highly specific and potent Pin1 inhibitors are urgently needed.
Additional Table 2.
Summary of Pin1 inhibitors
| Cell death type | Disease | Pin1 changes in disease | Main molecule in disease | Role of Pin in death | Reference |
|---|---|---|---|---|---|
| Apoptosis | AD | Decreased in AD patients | Pinl, GSK-3β, APP, HIF-lα, Aβ, p-Tau | Inhibits apoptosis | Lu et al., l999; Thorpe et al., 2004; Hashemzadeh-Bonehi et al., 2006; Pastorino et al., 2006; Kesavapany et al., 2007; Nowotny et al., 2007; Dakson et al., 20ll; O’Brien and Wong, 20l l; Lonati et al., 20l4; Azimi et al., 20l7; Xu et al., 20l7; Rong et al., 2017; Samimi et al., 202l; Nakhjiri et al., 2020; Li et al., 202la; Wang et al., 2020a; Zheng et al., 2021 |
| TBI | Decreased in TBI models | DAPK1, Pinl, p-Tau | Kim et al., 202l; Qiu et al., 202l; Tonnus et al., 202l | ||
| PD | Increased in PD patients | Pin1, α-synuclein, caspase-3 | Promotes apoptosis | Ghosh et al., 20l3; Matena et al., 20l3; Ibanez et al., 20l4; Ryo et al.,2006 | |
| HD | Increased in HD patients; Decreased in HD models |
Htt, Pinl, iASPP, p53 | Both inhibits and promotes apoptosis | Grison et al., 20ll; Agostoni et al., 20l6; Carnemolla et al., 20l7 | |
| Stroke | Increased in Stroke models | Pinl, FBX7, NICD1, p53 | Promotes apoptosis | Zacchi et al., 2002; Baik et al., 20l5; Arumugam et al., 20l8; Balaganapathy et al., 20l8; Li et al., 20l9 |
|
| SCI | Increased in SCI models | Pinl, Daxx, ASK1, BIMEL, JNK, Mcl-2, BAX, Bcl-2 |
Becker and Bonni, 2006, 2007; Ryo et al., 2007; Barone et al., 2008; Shen et al., 2009; Li et al., 20l7; Oh and Mouradian, 20l8; Zhang et al., 2021 |
||
| Epilepsy | Decreased in epileptic models | CaMKII, AMPA and NMDA receptors | Inhibits apoptosis | Becker and Bonni, 2007; Tang et al., 20l7; Hou et al., 202l | |
| Other neurodegenerative diseases | Decreased in age-related neurodegeneration patients | Lipofuscin | Hashemzadeh-Bonehi et al., 2006 | ||
| RN | Brain ischemia and traumatic injury | Increased in brain ischemia and traumatic injury models | Calcium, DAPK, Pinl | Promotes RN | Bialik and Kimchi, 20ll; Del Rosario et al., 20l5; Chen et al., 20l7; Ibarra et al., 2017; Wangetal., 20l7 |
| Retinal disease | Increased in retinal disease | Calcium, CaM, CaMKII, Pinl, CAST, Calpain2, AIF | Wang etal., 20l7, 20l9a, b; Cheng etal., 20l8; Hou etal., 202l | ||
| Autophagy | AD | Decreased in AD patients | Pinl, Akt, Ubiquitin, GSK-3β, Aβ, LC3-II | Promotes autophagy | Min et al., 200S;_Ma et al., 2012; Del Rosario et al., 20l5; So and Oh, 20l5; Kim et al., 2009; Chao et al., 202l |
AD: Alzheimer’s disease; AIF: apoptosis-induced factor; AMPA: α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; APP: amyloid precursor protein; ASK1: apoptosis signal-regulating kinase 1; Aβ: β-amyloid peptide; BAX: Bcl2-associated X; Bcl-2: B-cell lymphoma 2; BIMEL: Bcl-2-interacting mediator of cell death, extra long; CaM: calmodulin; CaMKII: calmodulin-dependent protein kinase II; CAST: calpastatin; DAPK1: death-associated protein kinase 1; Daxx: death domain associated protein; FBW7: F-box and WD repeat domain containing domain protein 7; GSK-3β: glycogen synthase kinase-3β; HD: Huntington's disease; HIF-1: Hypoxia-inducible transcription factor-1; Htt: Huntingtin; iASPP: inhibitor of apoptosis-stimulating protein of p53; JNK: C-Jun N-terminal kinase; LC3: long chain 3; Mcl-1: myeloid cell leukemia sequence-1; NICD1: Notch intracellular domain 1; NMDA: N-methyl-D-aspartic acid receptor; PD: Parkinson's disease; Pin1: prolyl cistrans isomerase NIMA-interacting 1; RN: regulated necrosis; SCI: spinal cord injury; TBI: traumatic brain injury.
Juglone was the first identified inhibitor that can inhibit the activity of Pin1. Juglone has been shown to inhibit neuronal damage in some experimental models of disease, such as PD and AD. Juglone irreversibly inhibits Pin1 catalytic domain PPIase activity at high concentrations Although the inhibition of Pin1 by Juglone is effective against neuronal damage both in vitro and in vivo, the lack of Pin1 specificity and toxicity at high concentrations create significant limitations with regards to potential application in clinical treatments. Therefore, further research now needs to evaluate or design potent and specific Pin1 inhibitors for therapeutic use. KPT-6566 is known to degrade Pin1 with higher levels of specificity and can reduce the expression of cyclin D1. Furthermore, KPT-6566 can induce cell apoptosis by generating ROS and inhibits cell proliferation in both breast and prostate cancer. More importantly, KPT-6566 inhibits the growth of cells over-expressing Pin1 overexpressing but not Pin1 knockdown cells in vitro or in vivo, thus suggesting that KPT-6566 can specifically suppress the activity of Pin1. However, further experimental and clinical investigations are now needed to explore the efficacy and safety of KPT-6566 for the treatment of neurological diseases.
TME-001, PiB, epigallocatechin-3-gallate, dipentamethylene thiuram monosulfide, 6,7,4′-trihydroxyisoflavone and 4,6-bis (benzyloxy)-3-phenylbenzofuran have been found to inhibit the growth of cancer cells by inhibiting the Pin1 PPIase catalytic domain in a competitive manner (Tatara et al., 2009; Mori et al., 2011; Lim et al., 2017; Fan et al., 2019; Bedouhene et al., 2021; Della Via et al., 2021). It should be noted that positive PiB uptake in the cerebral cortex has been detected, thus indicating that PiB may be useful for the treatment of neurological diseases (Antonelli et al., 2016; Liu et al., 2019a). Epigallocatechin-3-gallate (EGCG) induces the death of cancer cells through different pathways, such as the release of nitric oxide, and is not limited to Pin1-mediated pathways (Della Via et al., 2021). In addition, only PiB has been reported to be involved in synaptic transmission by inhibiting Pin1 activity in the CNS (Antonelli et al., 2016). A previous study found that 5′-nitro-indirubinoxime can reduce the expression of Pin1 and induce G1 phase cell cycle arrest, finally inhibiting the growth of lung cells (Yoon et al., 2012). 5′-Nitro-indirubinoxime can also block Polo-like kinase 1 signaling (Yoon et al., 2012). API-1 is a newly detected form of Pin1 inhibitor that can strongly inhibit the PPIase domain. Previous research found that hepatoma carcinoma cells were not affected by API-1, possibly may due to low efficacy (Sun et al., 2019). Both ATRA and ATO have been approved by the U. S. Food and Drug Administration for the treatment of patients with acute promyelocytic leukemia (Kozono et al., 2018; Bedouhene et al., 2021). Furthermore, ATO and ATRA exert a synergistic effect to inhibit the growth of breast cancer cells. ATRA and ATO can bind to the Pin1 catalytic PPIase domain in a non-covalent form, degrade the levels of Pin1, and reduce the expression of cyclin D1 (Kozono et al., 2018). However, the short half-life of ATRA may reduce its efficacy against tumors (Bedouhene et al., 2021). Moreover, the lack specificity of Pin1 also impede its application, as ATO can directly degrade cyclin D1 (Kozono et al., 2018). Although these Pin1 inhibitors are effective against proliferation in various cancer cells in vitro, their efficacy for the treatment of neurological diseases has yet to be investigated. Moreover, the poor solubility and specificity of these inhibitors also limit their potential therapeutic ability in the clinic.
Conclusion
RCD, including apoptosis, autophagy, and RN, is a type of controlled cellular death that contributes to the physiological balance in cell clearance and organ homeostasis. Over the past few years, several studies have indicated that the aberration of Pin1 expression is a pathological characteristic and plays a crucial regulatory role in neuronal RCD, especially in apoptosis. Pin1 dysfunction also occurs in patients with various neurodegenerative diseases and is correlated with poor therapeutic efficiency. The inhibition of Pin1 plays a vital role in neuronal survival. Therefore, Pin1 may represent a potential key therapeutic target for related neurodegenerative diseases. Intriguingly, Pin1 acts in both protective and harmful roles to regulate the neuronal death; the precise effects are dependent on different cellular contexts. This is because Pin1 mediates multiple mechanisms by binding with different targets. Then, Pin1 regulates different cellular physiological processes, finally inducing different consequences. The regulatory pathways that mediate Pin1 targets are quite distinct, thus indicating the necessary of using different disease models to investigate disease development and the pharmacological treatment of Pin1-related CNS diseases. Further research is also needed to investigate the potential effect of Pin1 as a biomarker for neurodegenerative diseases and in different stages of disease. Furthermore, although many Pin1 inhibitors have been identified that can inhibit the growth of tumor cells in in vitro, there are obvious limitations with regards to their clinical application, including poor solubility, side effects, specificity, and the lack of investigations in the CNS. Highly specific and potent Pin1 inhibitors now needed to be tested in the CNS. Furthermore, effective methods for Pin1 inhibitor screening are also needed to select potentially useful compounds. For example, Tatara et al. screened chemical compounds containing fused aromatic rings that inhibited Pin1 PPIase activity and identified PiB as an effective inhibitor for Pin1 (Tatara et al., 2009). Molecular modeling by computer simulation technology is also important as this could easily reveal the underlying interactions of the active domain of Pin1 with specific inhibitors. Furthermore, efforts should be made to promote the efficacy of combined targeted therapies and to prevent drug resistance. It should be noted that we must identify specific inhibitors with fewer adverse effects. Finally, we must also develop a highly efficient delivery system to improve the bioaccessibility and bioavailability of Pin1 inhibitors in both pre-clinical and clinical studies.
It should also be noted that the signaling pathways of Pin1 in different neurodegenerative diseases is complicated and that these pathways may interact with each other. Future studies are needed to investigate such interactive networks and gain a better understanding of Pin1-mediated diseases. Remarkably, although many research studies have reported that Pin1 is an important target in apoptosis for the treatment of neurodegenerative diseases, such as AD, there are still many challenges. For example, we still need to investigate the specific role of Pin1 in RN and autophagy.
Additional files:
Additional Table 1: Summary of Pin1 changes in neurodegenerative diseases.
Additional Table 2: Summary of Pin1 inhibitors.
Footnotes
Funding: The work was supported by the National Natural Science Foundation of China, Nos. 81971891 (to KX), 82101126 (to SCW), 81772134 (to KX), 82172196 (to KX), and the Natural Science Foundation of Hunan Province of China, No. 2021JJ40873 (to SCW).
Conflicts of interest: The authors declare that there is no potential conflict of interest.
Editor note: KX is an Editorial Board member of Neural Regeneration Research. He was blinded from reviewing or making decisions on the manuscript. The article was subject to the journal’s standard procedures, with peer review handled independently of this Editorial Board member and their research groups.
Availability of data and materials: All data generated or analyzed during this study are included in this published article and its supplementary information files.
C-Editor: Zhao M; S-Editors: Yu J, Li CH; L-Editors: Yu J, Song LP; T-Editor: Jia Y
References
- 1.Agostoni E, Michelazzi S, Maurutto M, Carnemolla A, Ciani Y, Vatta P, Roncaglia P, Zucchelli S, Leanza G, Mantovani F, Gustincich S, Santoro C, Piazza S, Del Sal G, Persichetti F. Effects of Pin1 loss in Hdh(Q111) knock-in mice. Front Cell Neurosci. 2016;10:110. doi: 10.3389/fncel.2016.00110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Albayram O, Angeli P, Bernstein E, Baxley S, Gao Z, Lu KP, Zhou XZ. Targeting prion-like cis phosphorylated tau pathology in neurodegenerative diseases. J Alzheimers Dis Parkinsonism. 2018;8:443. doi: 10.4172/2161-0460.1000443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Albayram O, Kondo A, Mannix R, Smith C, Tsai CY, Li C, Herbert MK, Qiu J, Monuteaux M, Driver J, Yan S, Gormley W, Puccio AM, Okonkwo DO, Lucke-Wold B, Bailes J, Meehan W, Zeidel M, Lu KP, Zhou XZ. Cis P-tau is induced in clinical and preclinical brain injury and contributes to post-injury sequelae. Nat Commun. 2017;8:1000. doi: 10.1038/s41467-017-01068-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alu A, Han X, Ma X, Wu M, Wei Y, Wei X. The role of lysosome in regulated necrosis. Acta Pharm Sin B. 2020;10:1880–1903. doi: 10.1016/j.apsb.2020.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Antonelli R, De Filippo R, Middei S, Stancheva S, Pastore B, Ammassari-Teule M, Barberis A, Cherubini E, Zacchi P. Pin1 modulates the synaptic content of NMDA receptors via prolyl-isomerization of PSD-95. J Neurosci. 2016;36:5437–5447. doi: 10.1523/JNEUROSCI.3124-15.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Arumugam TV, Baik SH, Balaganapathy P, Sobey CG, Mattson MP, Jo DG. Notch signaling and neuronal death in stroke. Prog Neurobiol. 2018;165:103–116. doi: 10.1016/j.pneurobio.2018.03.002. 167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Arumugam TV, Chan SL, Jo DG, Yilmaz G, Tang SC, Cheng A, Gleichmann M, Okun E, Dixit VD, Chigurupati S, Mughal MR, Ouyang X, Miele L, Magnus T, Poosala S, Granger DN, Mattson MP. Gamma secretase-mediated Notch signaling worsens brain damage and functional outcome in ischemic stroke. Nat Med. 2006;12:621–623. doi: 10.1038/nm1403. [DOI] [PubMed] [Google Scholar]
- 8.Azimi M, Nikanfar M, Khakikhatibi F, Rahbarghazi R, Nourazarian SM, Biray Avci C, Nourazarian A. Investigation of gene expression and serum levels of PIN1 and eNOS with high blood pressure in patients with Alzheimer disease. J Clin Neurosci. 2017;43:77–81. doi: 10.1016/j.jocn.2017.04.025. [DOI] [PubMed] [Google Scholar]
- 9.Bai Y, Lam HC, Lei X. Dissecting programmed cell death with small molecules. Acc Chem Res. 2020;53:1034–1045. doi: 10.1021/acs.accounts.9b00600. [DOI] [PubMed] [Google Scholar]
- 10.Baik SH, Fane M, Park JH, Cheng YL, Yang-Wei Fann D, Yun UJ, Choi Y, Park JS, Chai BH, Park JS, Back SH, Jeong JI, Jang YJ, Bahn G, Lee JY, Li YI, Sobey CG, Uchida T, Park JH, Kim HT, et al. Pin1 promotes neuronal death in stroke by stabilizing Notch intracellular domain. Ann Neurol. 2015;77:504–516. doi: 10.1002/ana.24347. [DOI] [PubMed] [Google Scholar]
- 11.Balaganapathy P, Baik SH, Mallilankaraman K, Sobey CG, Jo DG, Arumugam TV. Interplay between Notch and p53 promotes neuronal cell death in ischemic stroke. J Cereb Blood Flow Metab. 2018;38:1781–1795. doi: 10.1177/0271678X17715956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Barone MC, Desouza LA, Freeman RS. Pin1 promotes cell death in NGF-dependent neurons through a mechanism requiring c-Jun activity. J Neurochem. 2008;106:734–745. doi: 10.1111/j.1471-4159.2008.05427.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Becker EB, Bonni A. Pin1 mediates neural-specific activation of the mitochondrial apoptotic machinery. Neuron. 2006;49:655–662. doi: 10.1016/j.neuron.2006.01.034. [DOI] [PubMed] [Google Scholar]
- 14.Becker EB, Bonni A. Pin1 in neuronal apoptosis. Cell Cycle. 2007;6:1332–1335. doi: 10.4161/cc.6.11.4316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bedouhene S, Liu M, Senani N, Boussetta T, Pintard C, Dang PM, El-Benna J. Prolyl-isomerase Pin1 controls key fMLP-induced neutrophil functions. Biomedicines. 2021;9:1130. doi: 10.3390/biomedicines9091130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bialik S, Kimchi A. Pin-pointing a new DAP kinase function:the peptidyl-proly isomerase Pin1 Is Negatively Regulated by DAP kinase-mediated phosphorylation. Mol Cell. 2011;42:139–141. doi: 10.1016/j.molcel.2011.04.002. [DOI] [PubMed] [Google Scholar]
- 17.Bianchi M, D'Oria V, Braghini MR, Petrini S, Manco M. Liraglutide treatment ameliorates neurotoxicity induced by stable silencing of Pin1. Int J Mol Sci. 2019;20:5064. doi: 10.3390/ijms20205064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Blanchard JW, Victor MB, Tsai LH. Dissecting the complexities of Alzheimer disease with in vitro models of the human brain. Nat Rev Neurol. 2022;18:25–39. doi: 10.1038/s41582-021-00578-6. [DOI] [PubMed] [Google Scholar]
- 19.Blennow K, Brody DL, Kochanek PM, Levin H, McKee A, Ribbers GM, Yaffe K, Zetterberg H. Traumatic brain injuries. Nat Rev Dis Primers. 2016;2:16084. doi: 10.1038/nrdp.2016.84. [DOI] [PubMed] [Google Scholar]
- 20.Bourdenx M, Martín-Segura A, Scrivo A, Rodriguez-Navarro JA, Kaushik S, Tasset I, Diaz A, Storm NJ, Xin Q, Juste YR, Stevenson E, Luengo E, Clement CC, Choi SJ, Krogan NJ, Mosharov EV, Santambrogio L, Grueninger F, Collin L, Swaney DL, et al. Chaperone-mediated autophagy prevents collapse of the neuronal metastable proteome. Cell. 2021;184:2696–2714.e25. doi: 10.1016/j.cell.2021.03.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Campaner E, Rustighi A, Zannini A, Cristiani A, Piazza S, Ciani Y, Kalid O, Golan G, Baloglu E, Shacham S, Valsasina B, Cucchi U, Pippione AC, Lolli ML, Giabbai B, Storici P, Carloni P, Rossetti G, Benvenuti F, Bello E, et al. A covalent PIN1 inhibitor selectively targets cancer cells by a dual mechanism of action. Nat Commun. 2017;8:15772. doi: 10.1038/ncomms15772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Carlet M, Völse K, Vergalli J, Becker M, Herold T, Arner A, Senft D, Jurinovic V, Liu WH, Gao Y, Dill V, Fehse B, Baldus CD, Bastian L, Lenk L, Schewe DM, Bagnoli JW, Vick B, Schmid JP, Wilhelm A, et al. In vivo inducible reverse genetics in patients'tumors to identify individual therapeutic targets. Nat Commun. 2021;12:5655. doi: 10.1038/s41467-021-25963-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Carloni S, Riparini G, Buonocore G, Balduini W. Rapid modulation of the silent information regulator 1 by melatonin after hypoxia-ischemia in the neonatal rat brain. J Pineal Res. 2017;63:e12434. doi: 10.1111/jpi.12434. [DOI] [PubMed] [Google Scholar]
- 24.Carneiro BA, El-Deiry WS. Targeting apoptosis in cancer therapy. Nat Rev Clin Oncol. 2020;17:395–417. doi: 10.1038/s41571-020-0341-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Carnemolla A, Michelazzi S, Agostoni E. PIN1 modulates huntingtin levels and aggregate accumulation:an in vitro model. Front Cell Neurosci. 2017;11:121. doi: 10.3389/fncel.2017.00121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chao X, Wang S, Fulte S, Ma X, Ahamed F, Cui W, Liu Z, Rülicke T, Zatloukal K, Zong WX, Liu W, Ni HM, Ding WX. Hepatocytic p62 suppresses ductular reaction and tumorigenesis in mouse livers with mTORC1 activation and defective autophagy. J Hepatol. 2021 doi: 10.1016/j.jhep.2021.10.014. doi:10.1016/j.jhep.2021.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Charidimou A, Perosa V, Frosch MP, Scherlek AA, Greenberg SM, van Veluw SJ. Neuropathological correlates of cortical superficial siderosis in cerebral amyloid angiopathy. Brain. 2020;143:3343–3351. doi: 10.1093/brain/awaa266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chen F, Chisholm AD, Jin Y. Tissue-specific regulation of alternative polyadenylation represses expression of a neuronal ankyrin isoform in C. elegans epidermal development. Development. 2017;144:698–707. doi: 10.1242/dev.146001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen W, Jiang L, Hu Y, Fang G, Yang B, Li J, Liang N, Wu L, Hussain Z. Nanomedicines, an emerging therapeutic regimen for treatment of ischemic cerebral stroke:a review. J Control Release. 2021;340:342–360. doi: 10.1016/j.jconrel.2021.10.020. [DOI] [PubMed] [Google Scholar]
- 30.Chen Y, Li Y, Guo L, Hong J, Zhao W, Hu X, Chang C, Liu W, Xiong K. Bibliometric analysis of the inflammasome and pyroptosis in brain. Front Pharmacol. 2020;11:626502. doi: 10.3389/fphar.2020.626502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cheng SY, Wang SC, Lei M, Wang Z, Xiong K. Regulatory role of calpain in neuronal death. Neural Regen Res. 2018;13:556–562. doi: 10.4103/1673-5374.228762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dakson A, Yokota O, Esiri M, Bigio EH, Horan M, Pendleton N, Richardson A, Neary D, Snowden JS, Robinson A, Davidson YS, Mann DM. Granular expression of prolyl-peptidyl isomerase PIN1 is a constant and specific feature of Alzheimer's disease pathology and is independent of tau, Aβand TDP-43 pathology. Acta Neuropathol. 2011;121:635–649. doi: 10.1007/s00401-011-0798-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Degterev A, Huang Z, Boyce M, Li Y, Jagtap P, Mizushima N, Cuny GD, Mitchison TJ, Moskowitz MA, Yuan J. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat Chem Biol. 2005;1:112–119. doi: 10.1038/nchembio711. [DOI] [PubMed] [Google Scholar]
- 34.Del Rosario JS, Feldmann KG, Ahmed T, Amjad U, Ko B, An J, Mahmud T, Salama M, Mei S, Asemota D, Mano I. Death associated protein kinase (DAPK) -mediated neurodegenerative mechanisms in nematode excitotoxicity. BMC Neurosci. 2015;16:25. doi: 10.1186/s12868-015-0158-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Della Via FI, Shiraishi RN, Santos I, Ferro KP, Salazar-Terreros MJ, Franchi Junior GC, Rego EM, Saad STO, Torello CO. (-)-Epigallocatechin-3-gallate induces apoptosis and differentiation in leukaemia by targeting reactive oxygen species and PIN1. Sci Rep. 2021;11:9103. doi: 10.1038/s41598-021-88478-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dubiella C, Pinch BJ, Koikawa K, Zaidman D, Poon E, Manz TD, Nabet B, He S, Resnick E, Rogel A, Langer EM, Daniel CJ, Seo HS, Chen Y, Adelmant G, Sharifzadeh S, Ficarro SB, Jamin Y, Martins da Costa B, Zimmerman MW, et al. Sulfopin is a covalent inhibitor of Pin1 that blocks Myc-driven tumors in vivo. Nat Chem Biol. 2021;17:954–963. doi: 10.1038/s41589-021-00786-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Erickson TK. Poem by a woman with glaucoma. JAMA. 2021;326:1875–1875. [Google Scholar]
- 38.Evans AS, Coyne CB. RIPK3:beyond necroptosis. Immunity. 2019;50:1–3. doi: 10.1016/j.immuni.2018.12.031. [DOI] [PubMed] [Google Scholar]
- 39.Fagiani F, Govoni S, Racchi M, Lanni C. The peptidyl-prolyl isomerase Pin1 in neuronal signaling:from neurodevelopment to neurodegeneration. Mol Neurobiol. 2021;58:1062–1073. doi: 10.1007/s12035-020-02179-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fan X, He H, Li J, Luo G, Zheng Y, Zhou JK, He J, Pu W, Zhao Y. Discovery of 4,6-bis(benzyloxy)-3-phenylbenzofuran as a novel Pin1 inhibitor to suppress hepatocellular carcinoma via upregulating microRNA biogenesis. Bioorg Med Chem. 2019;27:2235–2244. doi: 10.1016/j.bmc.2019.04.028. [DOI] [PubMed] [Google Scholar]
- 41.Flügel D, Görlach A, Kietzmann T. GSK-3βregulates cell growth, migration, and angiogenesis via Fbw7 and USP28-dependent degradation of HIF-1α. Blood. 2012;119:1292–1301. doi: 10.1182/blood-2011-08-375014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ghosh A, Saminathan H, Kanthasamy A, Anantharam V, Jin H, Sondarva G, Harischandra DS, Qian Z, Rana A, Kanthasamy AG. The peptidyl-prolyl isomerase Pin1 up-regulation and proapoptotic function in dopaminergic neurons:relevance to the pathogenesis of Parkinson disease. J Biol Chem. 2013;288:21955–21971. doi: 10.1074/jbc.M112.444224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Grison A, Mantovani F, Comel A, Agostoni E, Gustincich S, Persichetti F, Del Sal G. Ser46 phosphorylation and prolyl-isomerase Pin1-mediated isomerization of p53 are key events in p53-dependent apoptosis induced by mutant huntingtin. Proc Natl Acad Sci U S A. 2011;108:17979–17984. doi: 10.1073/pnas.1106198108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gu JL, Liu F. Tau in Alzheimer's disease:pathological alterations and an attractive therapeutic target. Curr Med Sci. 2020;40:1009–1021. doi: 10.1007/s11596-020-2282-1. [DOI] [PubMed] [Google Scholar]
- 45.Guo LM, Wang Z, Li SP, Wang M, Yan WT, Liu FX, Wang CD, Zhang XD, Chen D, Yan J, Xiong K. RIP3/MLKL-mediated neuronal necroptosis induced by methamphetamine at 39°C. Neural Regen Res. 2020;15:865–874. doi: 10.4103/1673-5374.268902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Guo T, Deng YH, Dong LL, Ren L, He HY. Autophagy elicits neuroprotection at the subacute phase of transient cerebral ischaemia but has few effects on neurological outcomes after permanent ischaemic stroke in rats. Curr Med Sci. 2021;41:803–814. doi: 10.1007/s11596-021-2400-8. [DOI] [PubMed] [Google Scholar]
- 47.Hashemzadeh-Bonehi L, Phillips RG, Cairns NJ, Mosaheb S, Thorpe JR. Pin1 protein associates with neuronal lipofuscin:potential consequences in age-related neurodegeneration. Exp Neurol. 2006;199:328–338. doi: 10.1016/j.expneurol.2005.12.030. [DOI] [PubMed] [Google Scholar]
- 48.Hleihel R, El Hajj H, Wu HC, Berthier C, Zhu HH, Massoud R, Chakhachiro Z, El Sabban M, De The H, Bazarbachi A. A Pin1/PML/P53 axis activated by retinoic acid in NPM-1c acute myeloid leukemia. Haematologica. 2021;106:3090–3099. doi: 10.3324/haematol.2020.274878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hou X, Yang F, Li A, Zhao D, Ma N, Chen L, Lin S, Lin Y, Wang L, Yan X, Zheng M, Lee TH, Zhou XZ, Lu KP, Liu H. The Pin1-CaMKII-AMPA receptor axis regulates epileptic susceptibility. Cereb Cortex. 2021;31:3082–3095. doi: 10.1093/cercor/bhab004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hu XM, Zhang Q, Zhou RX, Wu YL, Li ZX, Zhang DY, Yang YC, Yang RH, Hu YJ, Xiong K. Programmed cell death in stem cell-based therapy:Mechanisms and clinical applications. World J Stem Cells. 2021a;13:386–415. doi: 10.4252/wjsc.v13.i5.386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hu XM, Li ZX, Lin RH, Shan JQ, Yu QW, Wang RX, Liao LS, Yan WT, Wang Z, Shang L, Huang Y, Zhang Q, Xiong K. Guidelines for regulated cell death assays:a systematic summary, a categorical comparison, a prospective. Front Cell Dev Biol. 2021b;9:634690. doi: 10.3389/fcell.2021.634690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Huang Y, Chen L, Guo L, Hupp TR, Lin Y. Evaluating DAPK as a therapeutic target. Apoptosis. 2014;19:371–386. doi: 10.1007/s10495-013-0919-2. [DOI] [PubMed] [Google Scholar]
- 53.Huang Y, Wang S, Huang F, Zhang Q, Qin B, Liao L, Wang M, Wan H, Yan W, Chen D, Liu F, Jiang B, Ji D, Xia X, Huang J, Xiong K. c-FLIP regulates pyroptosis in retinal neurons following oxygen-glucose deprivation/recovery via a GSDMD-mediated pathway. Ann Anat. 2021;235:151672. doi: 10.1016/j.aanat.2020.151672. [DOI] [PubMed] [Google Scholar]
- 54.Ibáñez K, Boullosa C, Tabarés-Seisdedos R, Baudot A, Valencia A. Molecular evidence for the inverse comorbidity between central nervous system disorders and cancers detected by transcriptomic meta-analyses. PLoS Genet. 2014;10:e1004173. doi: 10.1371/journal.pgen.1004173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ibarra MS, Borini Etichetti C, Di Benedetto C, Rosano GL, Margarit E, Del Sal G, Mione M, Girardini J. Dynamic regulation of Pin1 expression and function during zebrafish development. PLoS One. 2017;12:e0175939. doi: 10.1371/journal.pone.0175939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Iridoy MO, Zubiri I, Zelaya MV, Martinez L, Ausín K, Lachen-Montes M, Santamaría E, Fernandez-Irigoyen J, Jericó I. Neuroanatomical quantitative proteomics reveals common pathogenic biological routes between amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD) Int J Mol Sci. 2018;20:4. doi: 10.3390/ijms20010004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kesavapany S, Patel V, Zheng YL, Pareek TK, Bjelogrlic M, Albers W, Amin N, Jaffe H, Gutkind JS, Strong MJ, Grant P, Pant HC. Inhibition of Pin1 reduces glutamate-induced perikaryal accumulation of phosphorylated neurofilament-H in neurons. Mol Biol Cell. 2007;18:3645–3655. doi: 10.1091/mbc.E07-03-0237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kim MR, Choi HK, Cho KB, Kim HS, Kang KW. Involvement of Pin1 induction in epithelial-mesenchymal transition of tamoxifen-resistant breast cancer cells. Cancer Sci. 2009;100:1834–1841. doi: 10.1111/j.1349-7006.2009.01260.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kim N, Wang B, Koikawa K, Nezu Y, Qiu C, Lee TH, Zhou XZ. Inhibition of death-associated protein kinase 1 attenuates cis P-tau and neurodegeneration in traumatic brain injury. Prog Neurobiol. 2021;203:102072. doi: 10.1016/j.pneurobio.2021.102072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Koikawa K, Kibe S, Suizu F, Sekino N, Kim N, Manz TD, Pinch BJ, Akshinthala D, Verma A, Gaglia G, Nezu Y, Ke S, Qiu C, Ohuchida K, Oda Y, Lee TH, Wegiel B, Clohessy JG, London N, Santagata S, et al. Targeting Pin1 renders pancreatic cancer eradicable by synergizing with immunochemotherapy. Cell. 2021;184:4753–4771.e27. doi: 10.1016/j.cell.2021.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kozono S, Lin YM, Seo HS, Pinch B, Lian X, Qiu C, Herbert MK, Chen CH, Tan L, Gao ZJ, Massefski W, Doctor ZM, Jackson BP, Chen Y, Dhe-Paganon S, Lu KP, Zhou XZ. Arsenic targets Pin1 and cooperates with retinoic acid to inhibit cancer-driving pathways and tumor-initiating cells. Nat Commun. 2018;9:3069. doi: 10.1038/s41467-018-05402-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kuboki S, Sakai N, Clarke C, Schuster R, Blanchard J, Edwards MJ, Lentsch AB. The peptidyl-prolyl isomerase, Pin1, facilitates NF-kappaB binding in hepatocytes and protects against hepatic ischemia/reperfusion injury. J Hepatol. 2009;51:296–306. doi: 10.1016/j.jhep.2009.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kumari A, Kumar C, Pergu R, Kumar M, Mahale SP, Wasnik N, Mylavarapu SVS. Phosphorylation and Pin1 binding to the LIC1 subunit selectively regulate mitotic dynein functions. J Cell Biol. 2021;220:e202005184. doi: 10.1083/jcb.202005184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lammert CR, Frost EL, Bellinger CE, Bolte AC, McKee CA, Hurt ME, Paysour MJ, Ennerfelt HE, Lukens JR. AIM2 inflammasome surveillance of DNA damage shapes neurodevelopment. Nature. 2020;580:647–652. doi: 10.1038/s41586-020-2174-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lepore A, Choy PM, Lee NCW, Carella MA, Favicchio R, Briones-Orta MA, Glaser SS, Alpini G, D'Santos C, Tooze RM, Lorger M, Syn WK, Papakyriakou A, Giamas G, Bubici C, Papa S. Phosphorylation and stabilization of PIN1 by JNK promote intrahepatic cholangiocarcinoma growth. Hepatology. 2021;74:2561–2579. doi: 10.1002/hep.31983. [DOI] [PubMed] [Google Scholar]
- 66.Levine B, Kroemer G. Autophagy in the pathogenesis of disease. Cell. 2008;132:27–42. doi: 10.1016/j.cell.2007.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ley R, Ewings KE, Hadfield K, Howes E, Balmanno K, Cook SJ. Extracellular signal-regulated kinases 1/2 are serum-stimulated “Bim(EL) kinases”that bind to the BH3-only protein Bim(EL) causing its phosphorylation and turnover. J Biol Chem. 2004;279:8837–8847. doi: 10.1074/jbc.M311578200. [DOI] [PubMed] [Google Scholar]
- 68.Li J, Mo C, Guo Y, Zhang B, Feng X, Si Q, Wu X, Zhao Z, Gong L, He D, Shao J. Roles of peptidyl-prolyl isomerase Pin1 in disease pathogenesis. Theranostics. 2021a;11:3348–3358. doi: 10.7150/thno.45889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Li L, Su Z, Zou Z, Tan H, Cai D, Su L, Gu Z. Ser46 phosphorylation of p53 is an essential event in prolyl-isomerase Pin1-mediated p53-independent apoptosis in response to heat stress. Cell Death Dis. 2019;10:96. doi: 10.1038/s41419-019-1316-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Li QM, Tep C, Yune TY, Zhou XZ, Uchida T, Lu KP, Yoon SO. Opposite regulation of oligodendrocyte apoptosis by JNK3 and Pin1 after spinal cord injury. J Neurosci. 2007;27:8395–8404. doi: 10.1523/JNEUROSCI.2478-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Li R, Yin F, Guo YY, Zhao KC, Ruan Q, Qi YM. Knockdown of ANRIL aggravates H(2)O(2)-induced injury in PC-12 cells by targeting microRNA-125a. Biomed Pharmacother. 2017;92:952–961. doi: 10.1016/j.biopha.2017.05.122. [DOI] [PubMed] [Google Scholar]
- 72.Li YZ, Liu YJ, Zhang W, Luo SF, Zhou X, He GQ. Combined treatment with valproic acid and estrogen has neuroprotective effects in ovariectomized mice with Alzheimer's disease. Neural Regen Res. 2021b;16:2078–2085. doi: 10.4103/1673-5374.308103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Liao LS, Lu S, Yan WT, Wang SC, Guo LM, Yang YD, Huang K, Hu XM, Zhang Q, Yan J, Xiong K. The role of HSP90αin methamphetamine/hyperthermia-induced necroptosis in rat striatal neurons. Front Pharmacol. 2021;12:716394. doi: 10.3389/fphar.2021.716394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lim TG, Lee SY, Duan Z, Lee MH, Chen H, Liu F, Liu K, Jung SK, Kim DJ, Bode AM, Lee KW, Dong Z. The prolyl isomerase Pin1 is a novel target of 6, 7,4'-trihydroxyisoflavone for suppressing esophageal cancer growth. Cancer Prev Res (Phila) 2017;10:308–318. doi: 10.1158/1940-6207.CAPR-16-0318. [DOI] [PubMed] [Google Scholar]
- 75.Liu M, Bedouhene S, Hurtado-Nedelec M, Pintard C, Dang PM, Yu S, El-Benna J. The prolyl isomerase Pin1 controls lipopolysaccharide-induced priming of NADPH oxidase in human neutrophils. Front Immunol. 2019a;10:2567. doi: 10.3389/fimmu.2019.02567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Liu Y, Cheng A, Li YJ, Yang Y, Kishimoto Y, Zhang S, Wang Y, Wan R, Raefsky SM, Lu D, Saito T, Saido T, Zhu J, Wu LJ, Mattson MP. SIRT3 mediates hippocampal synaptic adaptations to intermittent fasting and ameliorates deficits in APP mutant mice. Nat Commun. 2019b;10:1886. doi: 10.1038/s41467-019-09897-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Lonati E, Brambilla A, Milani C, Masserini M, Palestini P, Bulbarelli A. Pin1, a new player in the fate of HIF-1αdegradation:an hypothetical mechanism inside vascular damage as Alzheimer's disease risk factor. Front Cell Neurosci. 2014;8:1. doi: 10.3389/fncel.2014.00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lu PJ, Wulf G, Zhou XZ, Davies P, Lu KP. The prolyl isomerase Pin1 restores the function of Alzheimer-associated phosphorylated tau protein. Nature. 1999;399:784–788. doi: 10.1038/21650. [DOI] [PubMed] [Google Scholar]
- 79.Ma SL, Pastorino L, Zhou XZ, Lu KP. Prolyl isomerase Pin1 promotes amyloid precursor protein (APP) turnover by inhibiting glycogen synthase kinase-3β(GSK3β) activity:novel mechanism for Pin1 to protect against Alzheimer disease. J Biol Chem. 2012;287:6969–6973. doi: 10.1074/jbc.C111.298596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Magnusson JP, Goritz C, Tatarishvili J, Dias DO, Smith EM, Lindvall O, Kokaia Z, Frisen J. A latent neurogenic program in astrocytes regulated by Notch signaling in the mouse. Science. 2014;346:237–241. doi: 10.1126/science.346.6206.237. [DOI] [PubMed] [Google Scholar]
- 81.Makinwa Y, Musich PR, Zou Y. Phosphorylation-dependent Pin1 isomerization of ATR:its role in regulating ATR's anti-apoptotic function at mitochondria, and the implications in cancer. Front Cell Dev Biol. 2020;8:281. doi: 10.3389/fcell.2020.00281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Matena A, Sinnen C, van den Boom J, Wilms C, Dybowski JN, Maltaner R, Mueller JW, Link NM, Hoffmann D, Bayer P. Transient domain interactions enhance the affinity of the mitotic regulator Pin1 toward phosphorylated peptide ligands. Structure. 2013;21:1769–1777. doi: 10.1016/j.str.2013.07.016. [DOI] [PubMed] [Google Scholar]
- 83.Min SH, Cho JS, Oh JH, Shim SB, Hwang DY, Lee SH, Jee SW, Lim HJ, Kim MY, Sheen YY, Lee SH, Kim YK. Tau and GSK3beta dephosphorylations are required for regulating Pin1 phosphorylation. Neurochem Res. 2005;30:955–961. doi: 10.1007/s11064-005-6177-0. [DOI] [PubMed] [Google Scholar]
- 84.Mori T, Hidaka M, Lin YC, Yoshizawa I, Okabe T, Egashira S, Kojima H, Nagano T, Koketsu M, Takamiya M, Uchida T. A dual inhibitor against prolyl isomerase Pin1 and cyclophilin discovered by a novel real-time fluorescence detection method. Biochem Biophys Res Commun. 2011;406:439–443. doi: 10.1016/j.bbrc.2011.02.066. [DOI] [PubMed] [Google Scholar]
- 85.Moujalled D, Strasser A, Liddell JR. Molecular mechanisms of cell death in neurological diseases. Cell Death Differ. 2021;28:2029–2044. doi: 10.1038/s41418-021-00814-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Nakhjiri E, Vafaee MS, Hojjati SMM, Shahabi P, Shahpasand K. Tau pathology triggered by spinal cord injury can play a critical role in the neurotrauma development. Mol Neurobiol. 2020;57:4845–4855. doi: 10.1007/s12035-020-02061-7. [DOI] [PubMed] [Google Scholar]
- 87.Napoletano F, Baron O, Vandenabeele P, Mollereau B, Fanto M. Intersections between regulated cell death and autophagy. Trends Cell Biol. 2019;29:323–338. doi: 10.1016/j.tcb.2018.12.007. [DOI] [PubMed] [Google Scholar]
- 88.Napoletano F, Ferrari Bravo G, Voto IAP, Santin A, Celora L, Campaner E, Dezi C, Bertossi A, Valentino E, Santorsola M, Rustighi A, Fajner V, Maspero E, Ansaloni F, Cancila V, Valenti CF, Santo M, Artimagnella OB, Finaurini S, Gioia U, et al. The prolyl-isomerase PIN1 is essential for nuclear Lamin-B structure and function and protects heterochromatin under mechanical stress. Cell Rep. 2021;36:109694. doi: 10.1016/j.celrep.2021.109694. [DOI] [PubMed] [Google Scholar]
- 89.Nascimento FR, Viktor de Paula Barros Baeta J, Prado de França AA, Braga Rocha EOMA, Pizziolo VR, Aparecida Dos Santos A, Antônio de Oliveira Mendes T, Diaz-Muñoz G, Nogueira Diaz MA. Dibenzoylmethane derivative inhibits melanoma cancer in vitro and in vivo through induction of intrinsic and extrinsic apoptotic pathways. Chem Biol Interact. 2022;351:109734. doi: 10.1016/j.cbi.2021.109734. [DOI] [PubMed] [Google Scholar]
- 90.Nowotny P, Bertelsen S, Hinrichs AL, Kauwe JS, Mayo K, Jacquart S, Morris JC, Goate A. Association studies between common variants in prolyl isomerase Pin1 and the risk for late-onset Alzheimer's disease. Neurosci Lett. 2007;419:15–17. doi: 10.1016/j.neulet.2007.03.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.O'Brien RJ, Wong PC. Amyloid precursor protein processing and Alzheimer's disease. Annu Rev Neurosci. 2011;34:185–204. doi: 10.1146/annurev-neuro-061010-113613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Oh SE, Mouradian MM. Cytoprotective mechanisms of DJ-1 against oxidative stress through modulating ERK1/2 and ASK1 signal transduction. Redox Biol. 2018;14:211–217. doi: 10.1016/j.redox.2017.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Oster M, Reyer H, Gerlinger C, Trakooljul N, Siengdee P, Keiler J, Ponsuksili S, Wolf P, Wimmers K. mRNA profiles of porcine parathyroid glands following variable phosphorus supplies throughout fetal and postnatal life. Biomedicines. 2021;9:454. doi: 10.3390/biomedicines9050454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Özel MN, Simon F, Jafari S, Holguera I, Chen YC, Benhra N, El-Danaf RN, Kapuralin K, Malin JA, Konstantinides N, Desplan C. Neuronal diversity and convergence in a visual system developmental atlas. Nature. 2021;589:88–95. doi: 10.1038/s41586-020-2879-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Pastorino L, Sun A, Lu PJ, Zhou XZ, Balastik M, Finn G, Wulf G, Lim J, Li SH, Li X, Xia W, Nicholson LK, Lu KP. The prolyl isomerase Pin1 regulates amyloid precursor protein processing and amyloid-beta production. Nature. 2006;440:528–534. doi: 10.1038/nature04543. [DOI] [PubMed] [Google Scholar]
- 96.Pinch BJ, Doctor ZM, Nabet B, Browne CM, Seo HS, Mohardt ML, Kozono S, Lian X, Manz TD, Chun Y, Kibe S, Zaidman D, Daitchman D, Yeoh ZC, Vangos NE, Geffken EA, Tan L, Ficarro SB, London N, Marto JA, et al. Identification of a potent and selective covalent Pin1 inhibitor. Nat Chem Biol. 2020;16:979–987. doi: 10.1038/s41589-020-0550-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Prokesch A, Blaschitz A, Bauer T, Moser G, Hiden U, Zadora J, Dechend R, Herse F, Gauster M. Placental DAPK1 and autophagy marker LC3B-II are dysregulated by TNF-αin a gestational age-dependent manner. Histochem Cell Biol. 2017;147:695–705. doi: 10.1007/s00418-016-1537-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Qiu C, Albayram O, Kondo A, Wang B, Kim N, Arai K, Tsai CY, Bassal MA, Herbert MK, Washida K, Angeli P, Kozono S, Stucky JE, Baxley S, Lin YM, Sun Y, Rotenberg A, Caldarone BJ, Bigio EH, Chen X, et al. Cis P-tau underlies vascular contribution to cognitive impairment and dementia and can be effectively targeted by immunotherapy in mice. Sci Transl Med. 2021;13:eaaz7615. doi: 10.1126/scitranslmed.aaz7615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Rami A. Review:autophagy in neurodegeneration:firefighter and/or incendiarist? Neuropathol Appl Neurobiol. 2009;35:449–461. doi: 10.1111/j.1365-2990.2009.01034.x. [DOI] [PubMed] [Google Scholar]
- 100.Rong XF, Sun YN, Liu DM, Yin HJ, Peng Y, Xu SF, Wang L, Wang XL. The pathological roles of NDRG2 in Alzheimer's disease, a study using animal models and APPwt-overexpressed cells. CNS Neurosci Ther. 2017;23:667–679. doi: 10.1111/cns.12716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Ros U, Peña-Blanco A, Hänggi K, Kunzendorf U, Krautwald S, Wong WW, García-Sáez AJ. Necroptosis execution is mediated by plasma membrane nanopores independent of calcium. Cell Rep. 2017;19:175–187. doi: 10.1016/j.celrep.2017.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Ryo A, Hirai A, Nishi M, Liou YC, Perrem K, Lin SC, Hirano H, Lee SW, Aoki I. A suppressive role of the prolyl isomerase Pin1 in cellular apoptosis mediated by the death-associated protein Daxx. J Biol Chem. 2007;282:36671–36681. doi: 10.1074/jbc.M704145200. [DOI] [PubMed] [Google Scholar]
- 103.Ryo A, Togo T, Nakai T, Hirai A, Nishi M, Yamaguchi A, Suzuki K, Hirayasu Y, Kobayashi H, Perrem K, Liou YC, Aoki I. Prolyl-isomerase Pin1 accumulates in lewy bodies of parkinson disease and facilitates formation of alpha-synuclein inclusions. J Biol Chem. 2006;281:4117–4125. doi: 10.1074/jbc.M507026200. [DOI] [PubMed] [Google Scholar]
- 104.Samimi N, Sharma G, Kimura T, Matsubara T, Huo A, Chiba K, Saito Y, Murayama S, Akatsu H, Hashizume Y, Hasegawa M, Farjam M, Shahpasand K, Ando K, Hisanaga SI. Distinct phosphorylation profiles of tau in brains of patients with different tauopathies. Neurobiol Aging. 2021;108:72–79. doi: 10.1016/j.neurobiolaging.2021.08.011. [DOI] [PubMed] [Google Scholar]
- 105.Selkoe DJ, Hardy J. The amyloid hypothesis of Alzheimer's disease at 25 years. EMBO Mol Med. 2016;8:595–608. doi: 10.15252/emmm.201606210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Shen ZJ, Esnault S, Schinzel A, Borner C, Malter JS. The peptidyl-prolyl isomerase Pin1 facilitates cytokine-induced survival of eosinophils by suppressing Bax activation. Nat Immunol. 2009;10:257–265. doi: 10.1038/ni.1697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Shentu YP, Huo Y, Feng XL, Gilbert J, Zhang Q, Liuyang ZY, Wang XL, Wang G, Zhou H, Wang XC, Wang JZ, Lu YM, Westermarck J, Man HY, Liu R. CIP2A causes Tau/APP phosphorylation, synaptopathy, and memory deficits in Alzheimer's disease. Cell Rep. 2018;24:713–723. doi: 10.1016/j.celrep.2018.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Shin MK, Vázquez-Rosa E, Koh Y, Dhar M, Chaubey K, Cintrón-Pérez CJ, Barker S, Miller E, Franke K, Noterman MF, Seth D, Allen RS, Motz CT, Rao SR, Skelton LA, Pardue MT, Fliesler SJ, Wang C, Tracy TE, Gan L, et al. Reducing acetylated tau is neuroprotective in brain injury. Cell. 2021;184:2715–2732.e23. doi: 10.1016/j.cell.2021.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Singh P, Talwar P. Exploring putative inhibitors of death associated protein kinase 1 (DAPK1) via targeting Gly-Glu-Leu (GEL) and Pro-Glu-Asn (PEN) substrate recognition motifs. J Mol Graph Model. 2017;77:153–167. doi: 10.1016/j.jmgm.2017.08.001. [DOI] [PubMed] [Google Scholar]
- 110.So KY, Oh SH. Prolyl isomerase Pin1 regulates cadmium-induced autophagy via ubiquitin-mediated post-translational stabilization of phospho-Ser GSK3αβin human hepatocellular carcinoma cells. Biochem Pharmacol. 2015;98:511–521. doi: 10.1016/j.bcp.2015.09.007. [DOI] [PubMed] [Google Scholar]
- 111.So KY, Ahn SG, Oh SH. Autophagy regulated by prolyl isomerase Pin1 and phospho-Ser-GSK3αβinvolved in protection of oral squamous cell carcinoma against cadmium toxicity. Biochem Biophys Res Commun. 2015;466:541–546. doi: 10.1016/j.bbrc.2015.09.066. [DOI] [PubMed] [Google Scholar]
- 112.Stevens C, Hupp TR. Novel insights into DAPK autophagic signalling using peptide aptamer combinatorial protein-interaction screens. Autophagy. 2008;4:531–533. doi: 10.4161/auto.5940. [DOI] [PubMed] [Google Scholar]
- 113.Sun D, Tan S, Xiong Y, Pu W, Li J, Wei W, Huang C, Wei YQ, Peng Y. MicroRNA biogenesis is enhanced by liposome-encapsulated Pin1 inhibitor in hepatocellular carcinoma. Theranostics. 2019;9:4704–4716. doi: 10.7150/thno.34588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Sung PS, Hsieh SL. C-type lectins and extracellular vesicles in virus-induced NETosis. J Biomed Sci. 2021;28:46. doi: 10.1186/s12929-021-00741-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Tabrizi SJ, Flower MD, Ross CA, Wild EJ. Huntington disease:new insights into molecular pathogenesis and therapeutic opportunities. Nat Rev Neurol. 2020;16:529–546. doi: 10.1038/s41582-020-0389-4. [DOI] [PubMed] [Google Scholar]
- 116.Tang L, Zhang Y, Chen G, Xiong Y, Wang X, Zhu B. Down-regulation of Pin1 in Temporal Lobe Epilepsy Patients and Mouse Model. Neurochem Res. 2017;42:1211–1218. doi: 10.1007/s11064-016-2158-8. [DOI] [PubMed] [Google Scholar]
- 117.Tatara Y, Lin YC, Bamba Y, Mori T, Uchida T. Dipentamethylene thiuram monosulfide is a novel inhibitor of Pin1. Biochem Biophys Res Commun. 2009;384:394–398. doi: 10.1016/j.bbrc.2009.04.144. [DOI] [PubMed] [Google Scholar]
- 118.Theobald SJ, Gräb J, Fritsch M, Suárez I, Eisfeld HS, Winter S, Koch M, Hölscher C, Pasparakis M, Kashkar H, Rybniker J. Gasdermin D mediates host cell death but not interleukin-1βsecretion in Mycobacterium tuberculosis-infected macrophages. Cell Death Discov. 2021;7:327. doi: 10.1038/s41420-021-00716-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Thorpe JR, Mosaheb S, Hashemzadeh-Bonehi L, Cairns NJ, Kay JE, Morley SJ, Rulten SL. Shortfalls in the peptidyl-prolyl cis-trans isomerase protein Pin1 in neurons are associated with frontotemporal dementias. Neurobiol Dis. 2004;17:237–249. doi: 10.1016/j.nbd.2004.07.008. [DOI] [PubMed] [Google Scholar]
- 120.Tonnus W, Belavgeni A, Beuschlein F, Eisenhofer G, Fassnacht M, Kroiss M, Krone NP, Reincke M, Bornstein SR, Linkermann A. The role of regulated necrosis in endocrine diseases. Nat Rev Endocrinol. 2021;17:497–510. doi: 10.1038/s41574-021-00499-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Van Houten B. Huntington's disease:astrocytes shift to fatty acid metabolism. Trends Endocrinol Metab. 2019;30:575–577. doi: 10.1016/j.tem.2019.07.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Wang L, Zhou Y, Chen D, Lee TH. Peptidyl-prolyl cis/trans isomerase Pin1 and Alzheimer's disease. Front Cell Dev Biol. 2020a;8:355. doi: 10.3389/fcell.2020.00355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Wang L, Ye X, Liu Y, Wei W, Wang Z. Aberrant regulation of FBW7 in cancer. Oncotarget. 2014;5:2000–2015. doi: 10.18632/oncotarget.1859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Wang M, Wan H, Wang S, Liao L, Huang Y, Guo L, Liu F, Shang L, Huang J, Ji D, Xia X, Jiang B, Chen D, Xiong K. RSK3 mediates necroptosis by regulating phosphorylation of RIP3 in rat retinal ganglion cells. J Anat. 2020b;237:29–47. doi: 10.1111/joa.13185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Wang S, Liao L, Huang Y, Wang M, Zhou H, Chen D, Liu F, Ji D, Xia X, Jiang B, Huang J, Xiong K. Pin1 is regulated by CaMKII activation in glutamate-induced retinal neuronal regulated necrosis. Front Cell Neurosci. 2019a;13:276. doi: 10.3389/fncel.2019.00276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Wang S, Liao L, Wang M, Zhou H, Huang Y, Wang Z, Chen D, Ji D, Xia X, Wang Y, Liu F, Huang J, Xiong K. Pin1 promotes regulated necrosis induced by glutamate in rat retinal neurons via CAST/Calpain2 pathway. Front Cell Neurosci. 2017;11:425. doi: 10.3389/fncel.2017.00425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Wang S, Huang Y, Yan Y, Zhou H, Wang M, Liao L, Wang Z, Chen D, Ji D, Xia X, Liu F, Huang J, Xiong K. Calpain2 but not calpain1 mediated by calpastatin following glutamate-induced regulated necrosis in rat retinal neurons. Ann Anat. 2019b;221:57–67. doi: 10.1016/j.aanat.2018.08.005. [DOI] [PubMed] [Google Scholar]
- 128.Wang Y, Briz V, Chishti A, Bi X, Baudry M. Distinct roles for μ-calpain and m-calpain in synaptic NMDAR-mediated neuroprotection and extrasynaptic NMDAR-mediated neurodegeneration. J Neurosci. 2013;33:18880–18892. doi: 10.1523/JNEUROSCI.3293-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Wood H. Traumatic brain injury induces transmissible tau pathology. Nat Rev Neurol. 2018;14:570–571. doi: 10.1038/s41582-018-0062-3. [DOI] [PubMed] [Google Scholar]
- 130.Xu L, Ren Z, Chow FE, Tsai R, Liu T, Rizzolio F, Boffo S, Xu Y, Huang S, Lippa CF, Gong Y. Pathological role of peptidyl-prolyl isomerase Pin1 in the disruption of synaptic plasticity in Alzheimer's disease. Neural Plast. 2017;2017:3270725. doi: 10.1155/2017/3270725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Yan WT, Lu S, Yang YD, Ning WY, Cai Y, Hu XM, Zhang Q, Xiong K. Research trends, hot spots and prospects for necroptosis in the field of neuroscience. Neural Regen Res. 2021;16:1628–1637. doi: 10.4103/1673-5374.303032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Yan WT, Yang YD, Hu XM, Ning WY, Liao LS, Lu S, Zhao WJ, Zhang Q, Xiong K. Do pyroptosis, apoptosis, and necroptosis (PANoptosis) exist in cerebral ischemia?Evidence from cell and rodent studies. Neural Regen Res. 2022;17:1761–1768. doi: 10.4103/1673-5374.331539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Yoon HE, Kim SA, Choi HS, Ahn MY, Yoon JH, Ahn SG. Inhibition of Plk1 and Pin1 by 5'-nitro-indirubinoxime suppresses human lung cancer cells. Cancer Lett. 2012;316:97–104. doi: 10.1016/j.canlet.2011.10.029. [DOI] [PubMed] [Google Scholar]
- 134.Yuan J, Amin P, Ofengeim D. Necroptosis and RIPK1-mediated neuroinflammation in CNS diseases. Nat Rev Neurosci. 2019;20:19–33. doi: 10.1038/s41583-018-0093-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Yuan T, Shao Y, Zhou X, Liu Q, Zhu Z, Zhou B, Dong Y, Stephanopoulos N, Gui S, Yan H, Liu D. Highly permeable DNA supramolecular hydrogel promotes neurogenesis and functional recovery after completely transected spinal cord injury. Adv Mater. 2021;33:e2102428. doi: 10.1002/adma.202102428. [DOI] [PubMed] [Google Scholar]
- 136.Zabłocka A, Kazana W, Sochocka M, Stańczykiewicz B, Janusz M, Leszek J, Orzechowska B. Inverse correlation between Alzheimer's disease and cancer:short overview. Mol Neurobiol. 2021;58:6335–6349. doi: 10.1007/s12035-021-02544-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Zacchi P, Gostissa M, Uchida T, Salvagno C, Avolio F, Volinia S, Ronai Z, Blandino G, Schneider C, Del Sal G. The prolyl isomerase Pin1 reveals a mechanism to control p53 functions after genotoxic insults. Nature. 2002;419:853–857. doi: 10.1038/nature01120. [DOI] [PubMed] [Google Scholar]
- 138.Zhang H, Jiao W, Cui H, Sun Q, Fan H. Combined exposure of alumina nanoparticles and chronic stress exacerbates hippocampal neuronal ferroptosis via activating IFN-γ/ASK1/JNK signaling pathway in rats. J Hazard Mater. 2021;411:125179. doi: 10.1016/j.jhazmat.2021.125179. [DOI] [PubMed] [Google Scholar]
- 139.Zhang S, Zhang X. Shrimp miRNA suppresses the stemness of human cancer stem cells via the PIN1 pathway. FASEB J. 2019;33:10767–10779. doi: 10.1096/fj.201900395RR. [DOI] [PubMed] [Google Scholar]
- 140.Zheng F, Li Y, Zhang F, Sun Y, Zheng C, Luo Z, Wang YL, Aschner M, Zheng H, Lin L, Cai P, Shao W, Guo Z, Zheng M, Zhou XZ, Lu KP, Wu S, Li H. Cobalt induces neurodegenerative damages through Pin1 inactivation in mice and human neuroglioma cells. J Hazard Mater. 2021;419:126378. doi: 10.1016/j.jhazmat.2021.126378. [DOI] [PubMed] [Google Scholar]
- 141.Zhou HH, Luo L, Zhai XD, Chen L, Wang G, Qin LQ, Yu Z, Xin LL, Wan Z. Sex-specific neurotoxicity of dietary advanced glycation end products in APP/PS1 mice and protective roles of trehalose by inhibiting tau phosphorylation via GSK-3β-TFEB. Mol Nutr Food Res. 2021;65:e2100464. doi: 10.1002/mnfr.202100464. [DOI] [PubMed] [Google Scholar]


