Abstract
MiR-4295, located on chromosome 10q25.2, is a unique miRNA with a wide range of biological functions. miR-4295 is widely expressed in vivo, participating in the biological processes of multiple cancers. Although miR-4295 is dysregulated in various cancers, it has also been found to have the function of inhibiting cancer. At the same time, the expression of miR-4295 is related to prognosis and can be affected by numerous factors connecting to the therapeutic effects of various drugs. This article is to better summarize the role of miR-4295 in cancer and review the potential diagnostic, prognostic, and therapeutic value of miR-4295, which may provide insight into subsequent research.
Keywords: Cancer, Cancer cells, miR-4295, Molecular function, Prognosis, Tumor suppressor
Introduction
MicroRNAs (miRNAs) are small non-coding RNAs, usually 20–22 nucleotides in length.1 miRNA generates pri-miRNA through transcription, which is then recognized by the Drosha/DGCR8 complex and cleaved into pre-miRNA, which is transported to the cytoplasm through Exportin-5.2 Pre-miRNA is further cleaved by Dicer enzyme and cofactors to form mature miRNA. The RNA-induced silencing complex (RISC) and mature miRNA bind to the Argonaute (AGO) protein to guide the complex to its target mRNA.3 Most miRNAs inhibit gene translation by recognizing and binding the 3′-UTR of the gene.4 miRNAs can regulate various biological processes such as cell differentiation, apoptosis, cell migration, and cell cycle.5,6 miRNAs can be artificially divided into two types of miRNAs, miRNA with tumor suppressor function and miRNA with cancer-promoting function.7 However, sometimes two mature miRNAs of the same pre-miRNA may have opposite effects in cancer.8 A mature miRNA may also have a dual impact on cancer development.9 In addition, more and more pieces of evidence show that miRNAs are not only reliable diagnostic markers of diseases7,10 but also potential therapeutic targets.11
miR-4295 is located in the intron region of the VTI1A gene. The precursor miR-4295 (85 bp) can be cleaved by the Dicer enzyme to form an active miRNA (miR-4295-5P, 18 bp).12 The apoptosis protein X-linked inhibitor of apoptosis protein (XIAP) targets the VTI1A promoter region through the transcription factor SP1 to promote the expression of miR-4295.13 miR-4295 can target to inhibit the expression of downstream genes14 and has abnormal expression in a variety of cancers including prostate cancer, gastric cancer, bladder cancer.15, 16, 17 Here, this review summarizes the biological functions of miR-4295 and its role in cancer pathogenesis and treatment.
The biological function of miR-4295
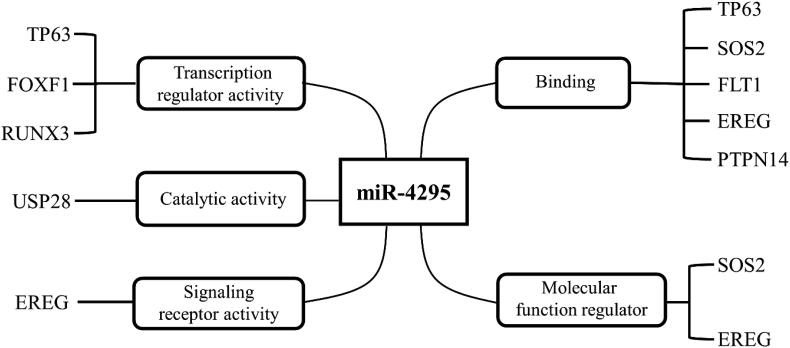
Currently, target genes of miR-4295 include TP63, USP28, RUNX3, GPC5, CDKN1A, BTG1, NPTX1, LRIG1, and PTPN14. In addition, downstream genes of miR-4295 also include SOS2, EREG, VEGF, VEGF-A, FLT1, FLT2, and FOXF1. The relationship among these genes and miR-4295 remains to be studied, and it will be an interesting question that whether there are potential intermediate pathways between these genes and miR-4295. We summarize the molecular functions of these downstream genes, indicating that miR-4295 has a wide range of biological functions (Fig. 1).
Figure 1.
Molecular functions of the downstream genes of miR-4295.
Binding function, molecular modulator and signal receptor activity of miR-4295 related genes
In the urothelial cell UROTsa, XIAP increases miR-4295 expression through the transcription factor SP1. It is well known that miR-4295 targets the expression of TP63 (p63α), which ultimately down-regulates the expression of epidermal growth factor (EGF). Low expression of EGF eventually induces malignant transformation of cells.13 TP63 is a gene on chromosome 3q27-29, which has strong homology with the tumor suppressor gene p53 and the related gene p73.18 Studies have shown that TP63 can bind to the anti-apoptotic factor BclxL through its DNA binding domain (p63-DBD).19
Analysis of KEGG and GEO databases revealed that low expression of miR-4295 reduced the expression of SOS2 and EREG, which can activate the ERBB2 pathway to promote cancer.20 Both SOS2 and EREG are related proteins of the ERBB2 pathway, and EREG is a ligand for EGFR.20 SOS2 forms a molecular complex with GRB2, thereby coupling EGFR to RAS activation.21
The expression of FLT1 is regulated by miR-4295 as the overexpression of miR-4295 in human umbilical vein endothelial cells (HUVEC) significantly upregulates the protein level of FLT1. Propranolol can down-regulate the expression of fms-associated tyrosine kinase 1 (FLT1) by down-regulating the expression of miR-4295, which exerts a tumor suppressor effect.22 FLT1 is a receptor for VEGF/PIGF and has a protein binding function. It is expressed on vascular endothelial cells and plays a key role in angiogenesis and subsequent cancer progression.23
The miR-4295/PTPN14/YAP axis plays an important role in the development of osteosarcoma. In osteosarcoma tissue, dual luciferase experiments showed that overexpressed miR-4295 can target the expression of Non-receptor tyrosine phosphatase 14 (PTPN14).24 The protein encoded by PTPN14 is a member of the protein tyrosine phosphatase (PTP) family, which is widely present in the nuclear and cytoplasmic localization.25 It mainly functions through a linker that includes a FERM domain, a linker domain, and a C-terminal catalytic PTP domain. The two PPxY motifs contained in the linker can bind to the WW domain of YAP and regulate its phosphorylation. Inactivation of YAP phosphorylation is an important mechanism by which PTPN14 inhibits tumor proliferation and metastasis and exerts a tumor suppressive effect.26
Catalytic activity of miR-4295 target gene
miR-4295 directly targets USP28 expression, and reduces the proliferation of non-small cell lung cancer (NSCLC) cells and induce its cell apoptosis.14 In addition, the stability of the oncoprotein MYC is regulated by SCFFbw7 ubiquitin ligase-mediated protein regulation of degradation. Since USP28 and SCFFbw7 bind to the same phosphorylation motif,27 USP28 can antagonize the protein degradation activity of SCFFbw7 and promote the growth of cancer cells.28,29 USP28 antagonizes SCFFbw7 and can also stabilize HIF-1α, which plays an important role in cell growth, migration, and angiogenesis.30 USP28 can stabilize the squamous cell carcinoma cell protein by removing the ubiquitin chain interaction between K48 and ΔNp63 protein, and the catalytic domain of USP28 is required for this activity.31 USP28 is also related to cell apoptosis caused by DNA damage.32 USP28 antagonizes cell apoptosis induced by ubiquitination by binding to the complex formed by Chk2 and ubiquitin E3 ligase PIRH2.33 As mentioned earlier, the miR-4295 expression is down-regulated in NSCLC tissues.14 Due to the low expression of miR-4295, the inhibitory effect of this miRNA on the expression of USP28 is relieved. As a target gene of miR-4295, the high expression of USP28 will promote the occurrence of non-small cell lung cancer,14 promote the proliferation, metastasis, and angiogenesis of cancer cells, and inhibit the apoptosis of cancer cells.34 To sum up, the low expression of miR-4295 will weaken the targeted inhibitory effect on USP28, thus showing a cancer-promoting effect.
Transcription regulator activity of miR-4295 related genes
miR-4295 affects the transcriptional regulatory activity of RNUX3, FOXF1, and TP63. RUNX3 expression is regulated by a variety of microRNAs including miR-4295.35, 36, 37 The RUNX3-encoded protein can form a protein complex that binds to the core DNA sequence 5′-PYGPYGGT-3′ of enhancers or promoters and then activates or inhibits gene transcription. In addition, RUNX3 interacts with other transcription factors and acts as a tumor suppressor.38 Li X et al discovered a potential signaling pathway of N-myc/miR-4295/RUNX 3. Among them, the up-regulation of miR-4295 can directly inhibit RUNX3 expression and its tumor suppressor function.37 Studies have shown that FOXF1 acts as a transcription factor and can activate the transcription of genes such as VEGF-A and SNAL1D.39,40 Similarly, propranolol down-regulates the expression of FOXF1 by down-regulating miR-4295 and inhibiting its transcriptional activation, exerting the anticancer function of miR-4295.22 In addition, TP63, which has transcription factor activity that binds to DNA, can be targeted and inhibited by miR-4295.13
The dual function of miR-4295 in cancer
As shown in Table 1, the expression of miR-4295 in various cancers is not completely consistent. miR-4295 is down-regulated in NSCLC tissues of 10 smoking patients14,20 while it is up-regulated in bladder cancer tissue, bladder cancer cell lines (Hcv29, UM-UC-3, BIu87, T24, and Hbc) and UROtsa cells. At the same time, it is up-regulated in the drug-treated bladder cancer cells (T24T cells).13,17,41 miR-4295 is also upregulated in the prostate cancer cell lines (PC3 and DU145)16 and in various pancreatic ductal adenocarcinoma (PDAC) cell lines (AsPC-1, Panc-1, BxPC-3, and SW1990).42 Besides, miR-4295 is up-regulated in various gastric cancer cell lines (MKN-28, NCI-N87, SGC-7901, MKN-45, and BGC-823)15 and 24 infantile hemangioma tissues and HUVEC cell lines.22 miR-4295 is also up-regulated in 20 gliomas tissues and the gliomas cell lines (U87 and U251).37 Beyond that, miR-4295 is up-regulated in 92 head and neck squamous cell carcinoma tissues and various head and neck squamous cell carcinoma (HNSCC) cell lines (FaDu, Hep2, SCC22B, and SCC154).43 miR-4295 is also up-regulated in 25 osteosarcoma tissues.24 Moreover, low expression of miR-4295 is currently only found in NSCLC tissues and cell lines, which requires more validation studies, but at least we have discovered the possibility of miR-4295 as a double-edged miRNA. In addition, the different effects of miR-4295 on prognosis also provide evidence for this.
Table 1.
Dysregulated miR-4295 in various cancers.
| System | Type | Level | Expression | Reference |
|---|---|---|---|---|
| Respiratory system | NSCLC | 10 tumor tissues of smokers, 10 tumor tissues of former smokers and 9 tumor tissues of non-smokers from GEO (GSE53519) | Downregulation | 14,20 |
| Urinary system | BC | Paired tumor and normal tissues; Hcv29, UM-UC-3, BIu87, T24, Hbc, UROtsa, and ISO-treated T24T cell lines | Upregulation | 13,17,41 |
| PC | PC3 and DU145 cell lines | Upregulation | 16 | |
| Digestive system | PDAC | AsPC-1, Panc-1, BxPC-3, and SW1990 cell lines | Upregulation | 42 |
| GC | MKN-28, NCI-N87, SGC-7901, MKN-45, and BGC-823 cell lines | Upregulation | 15 | |
| Circulatory system | IH | 24 tumor tissues and matched adjacent tissues; HUVEC cell line | Upregulation | 22 |
| Nervous system | Gliomas | 20 tumor tissues and matched normal tissues; U87 and U251 cell lines | Upregulation | 37 |
| Others | HNSCC | 92 tumor tissues and adjacent normal tissues; FaDu, Hep2, SCC22B, and SCC154 cell lines | Upregulation | 43 |
| Osteosarcoma | 25 paired tumor and adjacent normal tissues | Upregulation | 24 |
NSCLC: non-small cell lung cancer; BC: bladder cancer; ISO: Isorhapontigenin; PC: prostate cancer; PDAC: pancreatic ductal adenocarcinoma; GC: gastric cancer; IH: infantile hemangioma; HNSCC: head and neck squamous cell carcinoma; HUVEC: human umbilical vein endothelial cells.
Tumor-promoting function of miR-4295 related genes
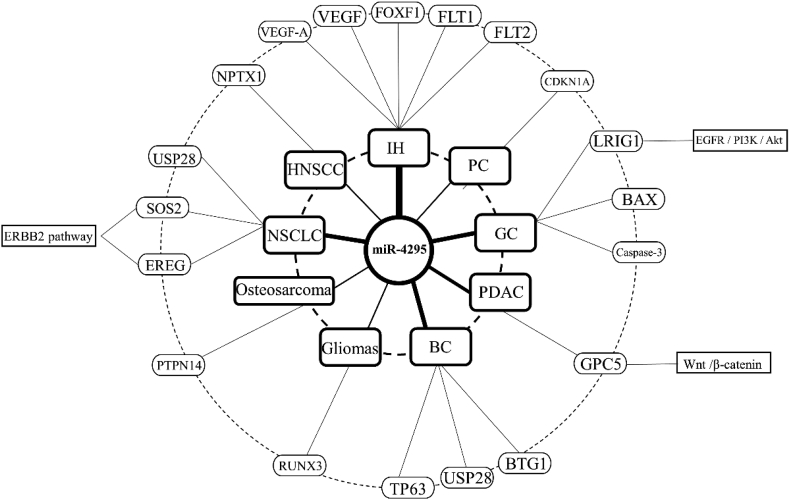
miR-4295 has been found to be involved in multiple cancer pathways (Fig. 2), and at the same time, it promotes cancer by affecting seven cellular activities including cell proliferation, cell migration, cell cycle, cell invasion, epithelial–mesenchymal transition (EMT) and apoptosis. In detail, miR-4295 suppresses or promotes the expression of specific genes primarily, and then through certain pathways taking effect at the cellular level. Finally, miR-4295 can promote the development of cancer. The genes and pathways related to miR-4295 are shown in Fig. 3. The association of these genes with miR-4295 has been experimentally proven, and we will describe them below.
Figure 2.
miR-4295 plays a role in multiple cancers through different pathways. miR-4295 inhibits the expression of TP63 and BTG1 in bladder cancer, and inhibites the expression of USP28 to exert a tumor suppressing effect. miR-4295 also promotes the expression of CDKN1A in prostate cancer. In gastric cancer, miR-4295 down-regulates the expression of BAX and Caspase-3 and inhibits EREG/PI3K/Akt signaling pathway by targeted down-regulation of LRIG1. miR-4295 inhibits GPC5 expression and promotes Wnt/β-catenin signaling pathway in pancreatic ductal adenocarcinoma. In IH, miR-4295 up-regulates the expression of VEGF-A, VEGF, FLT1, FOXF1 and FLT2. In gliomas, miR-4295 reduces the expression of RUNX3. In HNSCC, miR-4295 reduced the expression of NPTX1. In osteosarcoma, up-regulation of miR-4295 reduce the expression of PTPN14 to promote oncogenesis. In non-small cell lung cancer, low expression of miR-4295 reduces the expression of SOS2 and EREG, whereas miR-4295 inhibits the expression of USP28 in non-small cell lung cancer, and acting as a tumor suppressor.
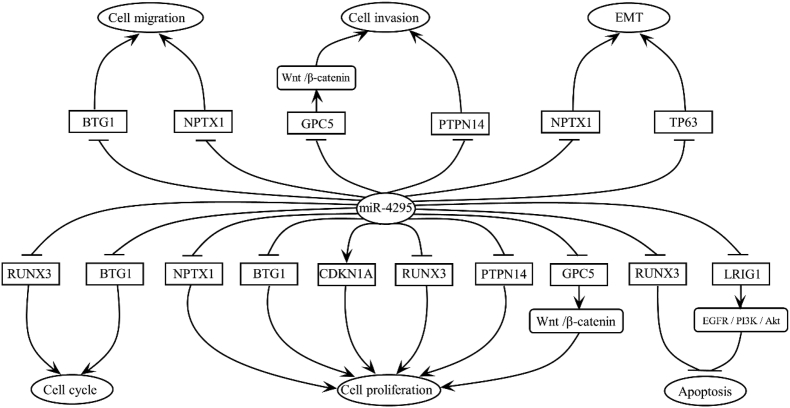
Figure 3.
Regulation of miR-4295 on biological behavior of cancer cells through a variety of pathway. miR-4295 exerts multiple effects on cancer cells through different target genes. miR-4295 promotes cell migration by inhibiting the expression of BTG1 and NPTX1. miR-4295 promotes cell invasion and EMT by inhibiting the expression of GPC5, NPTX1, TP63, and PTPN14, respectively. Furthermore, miR-4295 promotes cell cycle by inhibiting the expression of RUNX3 and BTG1, and promotes cell proliferation by inhibiting the expression of 5 genes (NPTX1, BTG1, RUNX3, GPC5, and PTPN14) and promoting the expression of CNKN1A. Moreover, miR-4295 can also inhibit apoptosis by inhibiting the expression of RUNX3 and LRIG1.
Cell proliferation regulation of miR-4295 related genes
In the PDAC cell lines (AsPC-1 and BxPC-3), overexpression of miR-4295 down-regulated the expression of GPC5 and promoted cell proliferation by promoting Wnt/β-catenin signaling.42 In the human glioma cell lines (U87 and U251), N-myc binds to the promoter of miR-4295 to up-regulate its expression, and over-expression of miR-4295 will inhibit the expression of RUNX3 and promotes colony formation and cell growth viability subsequently.37 In prostate cancer cell lines (PC3 and DU145), ginsenoside Rh2 down-regulates the expression of miR-4295, and it is proved that miR-4295 can directly inhibit CDKN1A and reduces cell proliferation of oncocytes.16 In bladder cancer cell lines (T24 and Hbc), overexpression of miR-4295 down-regulates BTG1 expression and significantly promotes cell proliferation in bladder cancer.44 In the HNSCC cell line, overexpression of miR-4295 down-regulates the expression of NPTX1 and promotes cell proliferation.43 In osteosarcoma Saos-2 and MG-63 cell lines, inhibition of miR-4195 expression increased PTPN14 expression and significantly inhibited the proliferation of these two cells. This shows that miR-4295 promotes cancer cell proliferation through targeted inhibition of PTPN14.24 The specific mechanism of PTPN14 inhibiting cancer cell proliferation is related to YAP. The Hippo signaling pathway is involved in regulating cell proliferation, and YAP is one of its family members. The combination of PTPN14 and YAP promotes YAP phosphorylation, prevents YAP nuclear translocation, thereby inhibiting YAP-mediated transcriptional activity and inhibiting cell proliferation.45
Cell cycle regulation of miR-4295 related genes
In the human glioma cell lines (U87 and U251), knockout of miR-4295 will significantly inhibit the cell cycle.37 Similarly, in the human bladder cancer cell lines (T24 and Hbc), inhibition of miR-4295 expression reduces the percentage of cells in the G1/G0 phase observably.17 These results indicate that miR-4295 can regulate the G0/G1 transition and accelerate the cell cycle.
Cell migration regulation of miR-4295 related genes
In human bladder cancer cells (T24 and Hbc), miR-4295 not only accelerates the cell cycle by down-regulating BTG1 but also promotes the migration of cancer cells.17 At the same time, targeted inhibition of NPTX1 by miR-4295 can also promote the migration of HNSCC cells (FaDu and Hep2).43 In addition, propranolol can down-regulates the expression of miR-4295 and this kind of inhibition finally reduces the cell migration of HUVEC.22 These indicate that miR-4295 plays an important role in promoting cancer cell migration.
Malignant EMT regulation of miR-4295 related genes
As previously described, in urothelial tumor UROtsa cells, miR-4295 binds the 3′ UTR of TP63 mRNA to inhibit its expression,13 and the down-regulation of TP63 is critical for malignant EMT.46 In the HNSCC cell lines (FaDu and Hep2), after treated with the miR-4295 inhibitor, the expression of mesenchymal markers (N-cadherin and vimentin) was decreased, while the expression of epithelial marker (E-cadherin) was up-regulated.43 These results indicate that miR-4295 can promote the EMT process.
Cell invasion regulation of miR-4295 related genes
In PDAC cells, miR-4295 down-regulates the expression of GPC5 and then promotes the Wnt/β-catenin signal transduction pathway and increases cancer cell invasion, at the same time, down-regulation of miR-4295 expression also inhibits the cell invasion of PDAC oncocytes.42 In osteosarcoma Saos-2 and MG-63 cell lines, overexpressed miR-4295 and targeted inhibition of PTPN14 significantly promoted cancer cell migration. We already know that PTPN14 is involved in cytoplasmic localization, which inhibits metastasis by changing protein transport. Inhibition of PTPN14 increases the secretion of growth factors and cytokines, and increases the expression of EGFR and FLT4 on the cell surface.47 Interestingly, miR-4295 also promotes the production of EGFR through other pathways such as increasing the expression of EREG, indicating that miR-4295 is a complex network for tumor regulation.15 Besides, PTPN14 can promote cancer cell invasion by inhibiting YAP activity in the Hippo signaling pathway.26
Apoptosis regulation of miR-4295 related genes
In the human glioma cell lines (U87 and U251), up-regulated miR-4295 expression can not only promotes proliferation and cell cycle, but also inhibits apoptosis causing glioma ultimately.37 In pediatric hemangiomas, miR-4295 can also inhibits apoptosis in HUVEC cells.22 At the same time, in the treatment of gastric cancer, cisplatin can reduce the expression of miR-4295 and promote the expression of LRIG1. Inhibiting of miR-4295 expression impedes the EGFR/PI3K/Akt signaling pathway, which finally induce apoptosis in gastric cancer cells (MKN-28 and MKN-45).15 In other words, miR-4295 play a tumor-promoting role in apoptosis regulation.
Cancer inhibition function of miR-4295 related genes
At present, it has been reported that miR-4295 also has a tumor-suppressing effect, which is related to USP28 (Fig. 4). As a carcinogenic factor, USP28 is involved in the development of cancers such as gastric cancer34 and bladder cancer.48 In non-small cell lung cancer, Zhang L et al first discovered that miR-4295 targets USP28 and promotes cell proliferation and induces apoptosis,14 while Luo Y et al found that isosaponin (ISO) can increases the expression of Dicer protein, causing the over-expression of miR-4295. Subsequently, over-expressed miR-4295 targets USP28 to inhibit invasion of bladder cancer cells, thereby demonstrating a tumor suppressor effect.41 Interestingly, miR-4295 exhibits dual function in bladder cancer (BC) cells. Specifically, it inhibits stem cell-like properties of cancer cells at an early stage, thereby exerting a tumor suppressor effect41; and then promotes cancer progression in advanced bladder cancer.17 At present, whether miR-4295 will produce anti-cancer effects through other genes is an important direction for future research.
Figure 4.
miR-4295 inhibits cancer by targeting USP28. miR-4295 can directly inhibit the expression of USP28, preventing non-small cell lung cancer and bladder cancer by promoting apoptosis and cell proliferation, and inhibiting cell invasion.
Antioxidative damage of miR-4295 related genes
There is a research shows that, in human trabecular meshwork (HTM) cells, lycium barbarum polysaccharides (LBP) treatment can up-regulate the expression of miR-4295, thereby activating PI3K/AKT and ERK signaling pathways and preventing oxidative damage caused by high concentrations of H2O2.49 miR-4295 plays an important role in the treatment of glaucoma with LBP. Oxidative damage caused by H2O2 is not only related to glaucoma but also has a complex effect in the development of cancer. On the one hand, low concentrations of H2O2 promote cancer cell proliferation50 and invasion51; on the other hand, excessive H2O2-mediated oxidative damage causes nuclear and mitochondrial DNA damage, and up-regulates Caspase-3 and pro-apoptotic protein BAX, leading to apoptosis in cancer cells.52 Interestingly, the expression of Caspase-3 and BAX was also up-regulated in gastric cancer cell lines (MKN-28 and MKN-45) with suppressed expression of miR-4295.15 However, there are no study on miR-4295 and oxidative damage in cancer currently. Oxidative damage is the driving factor for cancer cell apoptosis.53 By activating the PI3K/AKT and ERK signaling pathways, miR-4295 can antagonize the oxidative damage49 of HTM cells in glaucoma caused by high concentrations of H2O2. In gastric cancer cell lines MKN-28 and MKN-45, miR-4295 can activate EGFR/PI3K/AKT signaling pathway to inhibit cell apoptosis.15 It can be seen that the PI3K/AKT pathway is a common pathway for miR-4295 to antagonize H2O2 oxidative damage and inhibit cell apoptosis. The downstream effector of the PI3K/AKT pathway is mTOR, which can inhibit ROS and protect cancer cells from oxidative stress.54 In summary, miR-4295 inhibits cell apoptosis through the PI3K/AKT pathway may be related to the inhibition of oxidative damage of cancer cells, which also implies that miR-4295 has oncogenic properties that inhibit cancer cell apoptosis. However, whether miR-4295 induces apoptosis through oxidative stress-related pathways remains to be verified.
miR-4295 has important functions in diagnosis, prognosis, and treatment
The expression of miR-4295 in bladder cancer,17 infantile hemangioma,22 glioma37 and head and neck squamous cell carcinoma43 was significantly higher than that in adjacent tissues. These results suggest that miR-2495 may be a diagnostic indicator for a variety of cancers. However, it is worth noting that miR-4295 is down-regulated in lung cancer,20 and the effects of expression in different stages of bladder cancer are also inconsistent.17,41 A new clinical study55 shows that miR-4295 can be an effective prognostic indicator for propranolol and pingyangmycin combined with pulsed dye laser in the treatment of childhood hemangiomas. Interestingly, the expression of miR-4295 varies in different types of hemangiomas, and the expression of miR-4295 in cavernous hemangioma is significantly higher than that of capillary hemangioma.55 These results suggest that miR-4295 may be a promising indicator for future diagnosis and treatment of cancer.
miR-4295 plays a dual role in cancer and it also exhibits different prognostic effects (Table 2). First, promoting the expression of miR-4295 can shorten the survival of patients with NSCLC and therefore has a poor prognosis.20 In addition, miR-4295 expression was significantly associated with tumor size, TNM stage, and lymph node metastasis of HNSCC, and high expression of miR-4295 was a prognostic risk factor for HNSCC patients.43 In glioma, miR-4295 overexpression was positively correlated with clinical stage, and miR-4295 expression in stage IV tumors was higher than stage I tumors and normal tissues.37 In HNSCC, miR-4295 is significantly elevated in clinical stages III and IV, while high miR-4295 expression means lower overall survival (OS).43 On the other hand, Luo Y et al compared the OS of bladder cancer patients with different miR-4295 expression and found that the high expression of miR-4295 was positively correlated with the survival time of patients with BC.41 And up-regulation of miR-4295 may improve OS in patients with NSCLC.14 All these results indicate that miR-4295 is involved in cancer development and cancer prognosis.
Table 2.
Prognostic role of miR-4295 in different cancers.
| Cancer type | Sample | Factors | Relationship | Reference |
|---|---|---|---|---|
| NSCLC | Survival of NSCLC patients from Kaplan–Meier Plotter database | OS | Negative correlation | 20 |
| Gliomas | 20 matched patients | Clinical stage | Positive correlation | 37 |
| HNSCC | 92 matched patients | Clinical stage, Tumor size, TNM stage, lymphatic metastasis OS |
Positive correlation Negative correlation |
43 43 |
| BC | 564 high miR-4295 expression patients and 186 lower expression of miR-4295 patients from Kaplan–Meier Plotter database | OS | Positive correlation | 41 |
| NSCLC | 53 high USP28 expression patients and 17 lower expression of USP28 | OS | Positive correlation | 14 |
NSCLC: non-small cell lung cancer; BC: bladder cancer; HNSCC: head and neck squamous cell carcinoma; OS: overall survival.
miRNA transcription is regulated by RNA Pol II-related transcription factors and epigenetic regulatory factors.56 Several studies have reported that the expression of miR-4295 is regulated and can be used for disease treatment. For example, GRh2 alters the expression of miR-4295 at the transcriptional level and inhibits the growth of prostate cancer cells.16 Propranolol was found to reduce the expression of miR-4295 in HUVEC, thereby inhibiting the development of IH.22 Decreasing the expression of miR-4295 can promote the sensitivity of GC patients to the chemotherapy drug cisplatin. In addition, the proto-oncogene N-myc promotes glioma development by binding to the miR-4295 promoter.37 ISO also promotes the development of bladder cancer by promoting the expression of Dicer protein to promote the maturation of miR-4295.41 LBP treatment can up-regulate the expression of miR-4295 to prevent oxidative damage caused by high concentrations of H2O2.49 These results suggest that miR-4295 may be a novel therapeutic target for disease.
Interaction of miR-4295 related genes and various drugs
We also screened for the drugs related to the downstream genes of miR-4295. Hypermethylation of CpG island in RUNX3 may cause colorectal cancer patients to be resistant to two chemotherapeutic drugs, irinotecan and leucovorin.57 While the effect of miR-4295 on RUNX3 is consistent with the methylation of RUNX3 CpG island, all of which down-regulate the expression of RUNX3,37 suggesting that miR-4295 may be one of the causes of chemotherapy drug resistance in patients with colorectal cancer. It is worth noting that the down-regulation of miR-4295 can enhance the sensitivity of gastric cancer patients to the chemotherapeutic drug cisplatin.15
The high transcription level of EREG is a predictor of good prognosis in the treatment of metastatic colorectal cancer with cetuximab.58 The protein expression level of EREG is a predictive marker of the therapeutic effect of panitumumab in advanced colorectal cancer patients with RAS wild-type.59 The expression of EREG is also elevated during the down-regulation of miR-4295 by the chemotherapy drug cisplatin to promote apoptosis in gastric cancer cells.15 This suggests that miR-4295 may affect the efficacy of the drug by affecting the expression of EREG.
Pentosan polysulfate sodium is an anti-VEGF drug60 that treats prostate cancer by reducing the expression of VEGF in prostate cancer. Ranibizumab, bevacizumab, aflibercept, and other drugs are VEGF inhibitors, and their mechanism for treating IH is similar to that of propranolol, which reduces the expression of VEGF.22 miR-4295 up-regulates VEGF expression in IH, so miR-4295 may affect the effects of these anti-VEGF drugs. While phenytoin, cilastazol, and gentamicin can increase VEGF expression, miR-4295 may have a synergistic effect on them. FLT-1 and FLT-2 are the VEGF receptor and fibroblast growth factor receptor, respectively. They have been found to be useful in a variety of cancer therapies61, 62, 63 such as lung cancer,64 thyroid cancer,65 prostate cancer, etc.66 The up-regulation of miR-4295 on these two genes may affect the effects of these antagonists. Therefore, miR-4295 may play a role in the treatment of diseases as a new target.
Conclusions and future perspectives
There is increasing evidence that miRNAs are involved in a variety of biological processes.5 Over-expressed miR-4295 promotes tumor development by directly targeting TP63, RUNX3, GPC5, CNKN1A, BTG1, NPTX1, LRIG1 and PTPN14, and functions as an oncogene. However, miR-4295 also targets USP28 to inhibit the progression of non-small cell lung cancer. miR-4295 is over-expressed in most of the cancer tissues, and the prognosis of patients is significantly correlated with the level of miR-4295. Propranolol, lycium barbarum polysaccharides, ginsenoside, isorhapontigenin, cisplatin, and other drugs can affect the expression of miR-4295, and the biological effects of downstream genes suggest that miR-4295 may be a potential therapeutic target.
We have identified three signaling pathways related to miR-4295, including N-myc/miR-4295/RUNX3, miR-4295/GPC5/Wnt/β-catenin, and miR-4295/EGFR/PI3K/Akt, but whether the existence of other signaling pathways related to miR-4295 remains to be explored. In the current study, miR-4295 is down-regulated in tumors such as NSCLC and up-regulated in other cancers, such as BC and PC, which indicate the paradoxical roles of miR-4295 in human cancer. In addition, the role of non-coding RNA (ncRNA) on the abnormal expression of miR-4295 in cancer cannot be ignored. Although there is no research report on ncRNA targeting miR-4295, the study of the ncRNA/miRNA/mRNA signaling pathway is of great significance for understanding the role of miRNA-4295 in cancer. However, more research is needed on a large number of different cancer samples to determine this observation. In addition, miR-4295 may interact with various drugs through related genes, which will make miR-4295 a new target for disease treatment. Due to concerns about the safety of miRNAs, attempts to use miRNAs as a cancer gene therapy tool are still few.67 In this review, we present the potential diagnostic, prognostic, and therapeutic value of miR-4295 in cancer, which may provide insight into future research.
Funding
The research was supported by K. C. Wong Magna Fund in Ningbo University (Ningbo, Zhejiang, China).
Conflict of interests
The authors declare that they have no conflict of interest.
Acknowledgements
SD and LY contributed to the conception, design and final approval of the submitted version. LY, YZ1, YZ2, and SD contributed to manuscript writing. All the authors had read and approved the final manuscript.
Footnotes
Peer review under responsibility of Chongqing Medical University.
References
- 1.Acunzo M., Romano G., Wernicke D., Croce C.M. MicroRNA and cancer-a brief overview. Adv Biol Regul. 2015;57:1–9. doi: 10.1016/j.jbior.2014.09.013. [DOI] [PubMed] [Google Scholar]
- 2.Lin S., Gregory R.I. MicroRNA biogenesis pathways in cancer. Nat Rev Cancer. 2015;15(6):321–333. doi: 10.1038/nrc3932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Correia de Sousa M., Gjorgjieva M., Dolicka D., Sobolewski C., Foti M. Deciphering miRNAs' action through miRNA editing. Int J Mol Sci. 2019;20(24):6249. doi: 10.3390/ijms20246249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mohr A.M., Mott J.L. Overview of microRNA biology. Semin Liver Dis. 2015;35(1):3–11. doi: 10.1055/s-0034-1397344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee Y.S., Dutta A. MicroRNAs in cancer. Annu Rev Pathol. 2009;4:199–227. doi: 10.1146/annurev.pathol.4.110807.092222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ran X., Xiao C.H., Xiang G.M., Ran X.Z. Regulation of embryonic stem cell self-renewal and differentiation by microRNAs. Cell Reprogram. 2017;19(3):150–158. doi: 10.1089/cell.2016.0048. [DOI] [PubMed] [Google Scholar]
- 7.Bertoli G., Cava C., Castiglioni I. MicroRNAs: new biomarkers for diagnosis, prognosis, therapy prediction and therapeutic tools for breast cancer. Theranostics. 2015;5(10):1122–1143. doi: 10.7150/thno.11543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lv Y., Yang H., Ma X., Wu G. Strand-specific miR-28-3p and miR-28-5p have differential effects on nasopharyngeal cancer cells proliferation, apoptosis, migration and invasion. Cancer Cell Int. 2019;19:187. doi: 10.1186/s12935-019-0915-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wen R., Umeano A.C., Essegian D.J., Sabitaliyevich U.Y., Wang K., Farooqi A.A. Role of microRNA-410 in molecular oncology: a double edged sword. J Cell Biochem. 2018;119(11):8737–8742. doi: 10.1002/jcb.27251. [DOI] [PubMed] [Google Scholar]
- 10.Kiselev F.L. MicroRNA and cancer. Mol Biol (Mosk) 2014;48(2):232–242. [PubMed] [Google Scholar]
- 11.Hayes J., Peruzzi P.P., Lawler S. MicroRNAs in cancer: biomarkers, functions and therapy. Trends Mol Med. 2014;20(8):460–469. doi: 10.1016/j.molmed.2014.06.005. [DOI] [PubMed] [Google Scholar]
- 12.Griffiths-Jones S., Grocock R.J., van Dongen S., Bateman A., Enright A.J. miRBase: microRNA sequences, targets and gene nomenclature. Nucleic Acids Res. 2006;34(Database issue):D140–D144. doi: 10.1093/nar/gkj112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jin H., Xu J., Guo X., et al. XIAP RING domain mediates miR-4295 expression and subsequently inhibiting p63alpha protein translation and promoting transformation of bladder epithelial cells. Oncotarget. 2016;7(35):56540–56557. doi: 10.18632/oncotarget.10645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhang L., Xu B., Qiang Y., et al. Overexpression of deubiquitinating enzyme USP28 promoted non-small cell lung cancer growth. J Cell Mol Med. 2015;19(4):799–805. doi: 10.1111/jcmm.12426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yan R., Li K., Yuan D.W., et al. Downregulation of microRNA-4295 enhances cisplatin-induced gastric cancer cell apoptosis through the EGFR/PI3K/Akt signaling pathway by targeting LRIG1. Int J Oncol. 2018;53(6):2566–2578. doi: 10.3892/ijo.2018.4595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gao Q., Zheng J. Ginsenoside Rh2 inhibits prostate cancer cell growth through suppression of microRNA-4295 that activates CDKN1A. Cell Prolif. 2018;51(3) doi: 10.1111/cpr.12438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nan Y.H., Wang J., Wang Y., et al. MiR-4295 promotes cell growth in bladder cancer by targeting BTG1. Am J Transl Res. 2016;8(11):4892–4901. [PMC free article] [PubMed] [Google Scholar]
- 18.Yang A., Kaghad M., Wang Y., et al. p63, a p53 homolog at 3q27-29, encodes multiple products with transactivating, death-inducing, and dominant-negative activities. Mol Cell. 1998;2(3):305–316. doi: 10.1016/s1097-2765(00)80275-0. [DOI] [PubMed] [Google Scholar]
- 19.Enthart A., Klein C., Dehner A., et al. Solution structure and binding specificity of the p63 DNA binding domain. Sci Rep. 2016;6:26707. doi: 10.1038/srep26707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang L., Wang H., Wang C. Persistence of smoking induced non-small cell lung carcinogenesis by decreasing ERBB pathway-related microRNA expression. Thorac Cancer. 2019;10(4):890–897. doi: 10.1111/1759-7714.13020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Buday L., Downward J. Epidermal growth factor regulates p21ras through the formation of a complex of receptor, Grb2 adapter protein, and Sos nucleotide exchange factor. Cell. 1993;73(3):611–620. doi: 10.1016/0092-8674(93)90146-h. [DOI] [PubMed] [Google Scholar]
- 22.Zhao F., Yang X., Xu G., Bi J., Lv R., Huo R. Propranolol suppresses HUVEC viability, migration, VEGF expression, and promotes apoptosis by downregulation of miR-4295. J Cell Biochem. 2019;120(4):6614–6623. doi: 10.1002/jcb.27957. [DOI] [PubMed] [Google Scholar]
- 23.Ferrara N., Gerber H.P., LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9(6):669–676. doi: 10.1038/nm0603-669. [DOI] [PubMed] [Google Scholar]
- 24.Liang G., Duan C., He J., Ma W., Dai X. PTPN14, a target gene of miR-4295, restricts the growth and invasion of osteosarcoma cells through inactivation of YAP1 signalling. Clin Exp Pharmacol Physiol. 2020;47(7):1301–1310. doi: 10.1111/1440-1681.13296. [DOI] [PubMed] [Google Scholar]
- 25.Bottini A., Wu D.J., Ai R., et al. PTPN14 phosphatase and YAP promote TGFbeta signalling in rheumatoid synoviocytes. Ann Rheum Dis. 2019;78(5):600–609. doi: 10.1136/annrheumdis-2018-213799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liu X., Yang N., Figel S.A., et al. PTPN14 interacts with and negatively regulates the oncogenic function of YAP. Oncogene. 2013;32(10):1266–1273. doi: 10.1038/onc.2012.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Diefenbacher M.E., Chakraborty A., Blake S.M., et al. Usp28 counteracts Fbw7 in intestinal homeostasis and cancer. Cancer Res. 2015;75(7):1181–1186. doi: 10.1158/0008-5472.CAN-14-1726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sacco J.J., Coulson J.M., Clague M.J., Urbe S. Emerging roles of deubiquitinases in cancer-associated pathways. IUBMB Life. 2010;62(2):140–157. doi: 10.1002/iub.300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Diefenbacher M.E., Popov N., Blake S.M., et al. The deubiquitinase USP28 controls intestinal homeostasis and promotes colorectal cancer. J Clin Invest. 2014;124(8):3407–3418. doi: 10.1172/JCI73733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Flugel D., Gorlach A., Kietzmann T. GSK-3beta regulates cell growth, migration, and angiogenesis via Fbw7 and USP28-dependent degradation of HIF-1alpha. Blood. 2012;119(5):1292–1301. doi: 10.1182/blood-2011-08-375014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Prieto-Garcia C., Hartmann O., Reissland M., et al. Maintaining protein stability of Np63 via USP28 is required by squamous cancer cells. EMBO Mol Med. 2020;12(4) doi: 10.15252/emmm.201911101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhang D., Zaugg K., Mak T.W., Elledge S.J. A role for the deubiquitinating enzyme USP28 in control of the DNA-damage response. Cell. 2006;126(3):529–542. doi: 10.1016/j.cell.2006.06.039. [DOI] [PubMed] [Google Scholar]
- 33.Wang X., Liu Z., Zhang L., et al. Targeting deubiquitinase USP28 for cancer therapy. Cell Death Dis. 2018;9(2):186. doi: 10.1038/s41419-017-0208-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhao L.J., Zhang T., Feng X.J., et al. USP28 contributes to the proliferation and metastasis of gastric cancer. J Cell Biochem. 2018;120(5):7657–7666. doi: 10.1002/jcb.28040. [DOI] [PubMed] [Google Scholar]
- 35.Yu J., Tian X., Chang J., Liu P., Zhang R. RUNX3 inhibits the proliferation and metastasis of gastric cancer through regulating miR-182/HOXA9. Biomed Pharmacother. 2017;96:782–791. doi: 10.1016/j.biopha.2017.08.144. [DOI] [PubMed] [Google Scholar]
- 36.Qiu Y.Y., Zhang Y.W., Qian X.F., Bian T. miR-371, miR-138, miR-544, miR-145, and miR-214 could modulate Th1/Th2 balance in asthma through the combinatorial regulation of Runx3. Am J Transl Res. 2017;9(7):3184–3199. [PMC free article] [PubMed] [Google Scholar]
- 37.Li X., Zheng J., Diao H., Liu Y. RUNX3 is down-regulated in glioma by Myc-regulated miR-4295. J Cell Mol Med. 2016;20(3):518–525. doi: 10.1111/jcmm.12736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lee J.W., van Wijnen A., Bae S.C. RUNX3 and p53: how two tumor suppressors cooperate against oncogenic ras? Adv Exp Med Biol. 2017;962:321–332. doi: 10.1007/978-981-10-3233-2_20. [DOI] [PubMed] [Google Scholar]
- 39.Wang S., Yan S., Zhu S., et al. FOXF1 induces epithelial-mesenchymal transition in colorectal cancer metastasis by transcriptionally activating SNAI1. Neoplasia. 2018;20(10):996–1007. doi: 10.1016/j.neo.2018.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang S., Xiao Z., Hong Z., et al. FOXF1 promotes angiogenesis and accelerates bevacizumab resistance in colorectal cancer by transcriptionally activating VEGFA. Cancer Lett. 2018;439:78–90. doi: 10.1016/j.canlet.2018.09.026. [DOI] [PubMed] [Google Scholar]
- 41.Luo Y., Tian Z., Hua X., et al. Isorhapontigenin (ISO) inhibits stem cell-like properties and invasion of bladder cancer cell by attenuating CD44 expression. Cell Mol Life Sci. 2020;77(2):351–363. doi: 10.1007/s00018-019-03185-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yuan Q., Zhang Y., Li J., Cao G., Yang W. High expression of microRNA-4295 contributes to cell proliferation and invasion of pancreatic ductal adenocarcinoma by the down-regulation of Glypican-5. Biochem Biophys Res Commun. 2018;497(1):73–79. doi: 10.1016/j.bbrc.2018.02.023. [DOI] [PubMed] [Google Scholar]
- 43.Lu S., Zhou C., Zou B., Zhang H., Feng M. MiR-4295 facilitates cell proliferation and metastasis in head and neck squamous cell carcinoma by targeting NPTX1. Gene Immun. 2020;21(1):4–12. doi: 10.1038/s41435-019-0081-0. [DOI] [PubMed] [Google Scholar]
- 44.Nan Y.H., Wang J., Wang Y., et al. Erratum: MiR-4295 promotes cell growth in bladder cancer by targeting BTG1. Am J Transl Res. 2017;9(4):1960. [PMC free article] [PubMed] [Google Scholar]
- 45.Meng Z., Moroishi T., Guan K.L. Mechanisms of Hippo pathway regulation. Genes Dev. 2016;30(1):1–17. doi: 10.1101/gad.274027.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Stacy A.J., Craig M.P., Sakaram S., Kadakia M. DeltaNp63alpha and microRNAs: leveraging the epithelial-mesenchymal transition. Oncotarget. 2017;8(2):2114–2129. doi: 10.18632/oncotarget.13797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Belle L., Ali N., Lonic A., et al. The tyrosine phosphatase PTPN14 (Pez) inhibits metastasis by altering protein trafficking. Sci Signal. 2015;8(364):ra18. doi: 10.1126/scisignal.2005547. [DOI] [PubMed] [Google Scholar]
- 48.Guo G., Xu Y., Gong M., Cao Y., An R. USP28 is a potential prognostic marker for bladder cancer. Tumour Biol. 2014;35(5):4017–4022. doi: 10.1007/s13277-013-1525-1. [DOI] [PubMed] [Google Scholar]
- 49.Liu Y., Zhang Y. Lycium barbarum polysaccharides alleviate hydrogen peroxide-induced injury by up-regulation of miR-4295 in human trabecular meshwork cells. Exp Mol Pathol. 2019;106:109–115. doi: 10.1016/j.yexmp.2018.12.007. [DOI] [PubMed] [Google Scholar]
- 50.Liu S.L., Lin X., Shi D.Y., Cheng J., Wu C.Q., Zhang Y.D. Reactive oxygen species stimulated human hepatoma cell proliferation via cross-talk between PI3-K/PKB and JNK signaling pathways. Arch Biochem Biophys. 2002;406(2):173–182. doi: 10.1016/s0003-9861(02)00430-7. [DOI] [PubMed] [Google Scholar]
- 51.Nishikawa M., Hashida M., Takakura Y. Catalase delivery for inhibiting ROS-mediated tissue injury and tumor metastasis. Adv Drug Deliv Rev. 2009;61(4):319–326. doi: 10.1016/j.addr.2009.01.001. [DOI] [PubMed] [Google Scholar]
- 52.Cao L., Liu J., Zhang L., Xiao X., Li W. Curcumin inhibits H2O2-induced invasion and migration of human pancreatic cancer via suppression of the ERK/NF-kappaB pathway. Oncol Rep. 2016;36(4):2245–2251. doi: 10.3892/or.2016.5044. [DOI] [PubMed] [Google Scholar]
- 53.Lang J.Y., Ma K., Guo J.X., Sun H. Oxidative stress induces B lymphocyte DNA damage and apoptosis by upregulating p66shc. Eur Rev Med Pharmacol Sci. 2018;22(4):1051–1060. doi: 10.26355/eurrev_201802_14388. [DOI] [PubMed] [Google Scholar]
- 54.Sun Y., Ai J.Z., Jin X., et al. IL-8 protects prostate cancer cells from GSK-3beta-induced oxidative stress by activating the mTOR signaling pathway. Prostate. 2019;79(10):1180–1190. doi: 10.1002/pros.23836. [DOI] [PubMed] [Google Scholar]
- 55.Huang Z., Zhang N., Cai H., Luo K. Efficacy of propranolol and pingyangmycin, respectively, combined with pulsed dye laser on children with hemangioma. Exp Ther Med. 2020;19(2):1197–1202. doi: 10.3892/etm.2019.8314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Ha M., Kim V.N. Regulation of microRNA biogenesis. Nat Rev Mol Cell Biol. 2014;15(8):509–524. doi: 10.1038/nrm3838. [DOI] [PubMed] [Google Scholar]
- 57.Ogino S., Meyerhardt J.A., Kawasaki T., et al. CpG island methylation, response to combination chemotherapy, and patient survival in advanced microsatellite stable colorectal carcinoma. Virchows Arch. 2007;450(5):529–537. doi: 10.1007/s00428-007-0398-3. [DOI] [PubMed] [Google Scholar]
- 58.Pentheroudakis G., Kotoula V., De Roock W., et al. Biomarkers of benefit from cetuximab-based therapy in metastatic colorectal cancer: interaction of EGFR ligand expression with RAS/RAF, PIK3CA genotypes. BMC Cancer. 2013;13:49. doi: 10.1186/1471-2407-13-49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Seligmann J.F., Elliott F., Richman S.D., et al. Combined epiregulin and amphiregulin expression levels as a predictive biomarker for Panitumumab therapy benefit or lack of benefit in patients with RAS wild-type Advanced colorectal cancer. JAMA Oncol. 2016;2(5):633–642. doi: 10.1001/jamaoncol.2015.6065. [DOI] [PubMed] [Google Scholar]
- 60.Zaslau S., Sparks S., Riggs D., Jackson B., Kandzari S.J. Pentosan polysulfate (Elmiron): in vitro effects on prostate cancer cells regarding cell growth and vascular endothelial growth factor production. Am J Surg. 2006;192(5):640–643. doi: 10.1016/j.amjsurg.2006.08.008. [DOI] [PubMed] [Google Scholar]
- 61.Harding T.C., Long L., Palencia S., et al. Blockade of nonhormonal fibroblast growth factors by FP-1039 inhibits growth of multiple types of cancer. Sci Transl Med. 2013;5(178) doi: 10.1126/scitranslmed.3005414. 178ra139. [DOI] [PubMed] [Google Scholar]
- 62.Gozgit J.M., Wong M.J., Moran L., et al. Ponatinib (AP24534), a multitargeted pan-FGFR inhibitor with activity in multiple FGFR-amplified or mutated cancer models. Mol Cancer Ther. 2012;11(3):690–699. doi: 10.1158/1535-7163.MCT-11-0450. [DOI] [PubMed] [Google Scholar]
- 63.Kulthanan K., Chiawsirikajorn Y., Jiamton S. Acute urticaria: etiologies, clinical course and quality of life. Asian Pac J Allergy Immunol. 2008;26(1):1–9. [PubMed] [Google Scholar]
- 64.Singleton K.R., Hinz T.K., Kleczko E.K., et al. Kinome RNAi screens reveal synergistic targeting of MTOR and FGFR1 pathways for treatment of lung cancer and HNSCC. Cancer Res. 2015;75(20):4398–4406. doi: 10.1158/0008-5472.CAN-15-0509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Cohen E.E., Rosen L.S., Vokes E.E., et al. Axitinib is an active treatment for all histologic subtypes of advanced thyroid cancer: results from a phase II study. J Clin Oncol. 2008;26(29):4708–4713. doi: 10.1200/JCO.2007.15.9566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Saez C., Gonzalez-Baena A.C., Japon M.A., et al. Expression of basic fibroblast growth factor and its receptors FGFR1 and FGFR2 in human benign prostatic hyperplasia treated with finasteride. Prostate. 1999;40(2):83–88. doi: 10.1002/(sici)1097-0045(19990701)40:2<83::aid-pros3>3.0.co;2-n. [DOI] [PubMed] [Google Scholar]
- 67.Wang Z., Sha H.H., Li H.J. Functions and mechanisms of miR-186 in human cancer. Biomed Pharmacother. 2019;119:109428. doi: 10.1016/j.biopha.2019.109428. [DOI] [PubMed] [Google Scholar]