Abstract
Recent guidelines recommend that colorectal cancer (CRC) screening after age 75 be considered on an individualized basis, and discourage screening for people over 85 due to competing causes of mortality. Given the heterogeneity in the health of older individuals, and lack of data within current guidelines for personalized CRC screening approaches, there remains a need for a clearer framework to inform clinical decision-making. A revision of the current approach to CRC screening in older adults is even more compelling given the improvements in CRC treatment, post-treatment survival, and increasing life expectancy in the population. In this review, we aim to examine the personalization of CRC screening cessation based on specific factors influencing life and health expectancy such as comorbidity, frailty, and cognitive status. We will also review screening modalities and endoscopic technique for minimizing risk, the risks of screening unique to older adults, and CRC treatment outcomes in older patients, in order to provide important information to aid CRC screening decisions for this age group. This review article offers a unique approach to this topic from both the gastroenterologist and geriatrician perspective by reviewing the use of specific clinical assessment tools, and addressing technical aspects of screening colonoscopy and periprocedural management to mitigate screening-related complications.
Keywords: Colorectal cancer, Colonoscopy, Cancer screening, Early detection of cancer, Aged, Elderly
Core Tip: Clinical guidelines do not recommend colorectal cancer (CRC) screening after age 75. Given the improvements in CRC treatment and post-treatment survival, and increasing life expectancy in the population, the current approach to CRC screening in older adults needs to be revised. This review examines the personalization of CRC screening in older individuals based on specific factors influencing life and health expectancy. Screening modalities, techniques, and risks are also discussed.
INTRODUCTION
Colorectal cancer (CRC) is the third most common cancer in both men and women in the United States, with almost 150000 total cases in 2020[1]. CRC is predominantly a disease of older adults, as the risk increases markedly with age[2]. CRC incidence is 59.5 per 100000 for individuals aged 50 to 54 years, but increases to 197.5 per 100000 for adults aged 75 to 79 years[1]. The relative survival rate for CRC is 64% at 5 years following diagnosis and 58% at 10 years. The most important predictor of CRC survival is stage at diagnosis. The 5-year survival rate is 90% for patients diagnosed with localized-stage disease, but declines to 71% and 14% for those diagnosed with regional and distant stages, respectively[1]. The percentage of CRC deaths is highest among people aged 65 to 74 years of age at 24.2%. 23.6% of CRC deaths occur among people aged 75 to 84 years, and 20.3% among people 84 years and older[3].
CRC screening is an opportunity to both decrease incidence and increase survival by detection and removal of growths at early stages. Recent guidelines recommend that CRC screening after age 75 be considered on an individualized basis, and that providers engage in shared decision making for screening individuals in this category[4-6]. CRC screening is discouraged for people over 85 due to competing causes of mortality[5,6]. Although these guidelines provide a general framework for screening older individuals, the evidence provided is not robust, as most of the studies in older patients referenced by the guidelines are either microsimulation, meta-analysis, or studies with few patients above 80 years of age[4,7,8]. Guidelines acknowledge that few empirical data exist on when best to stop offering screening, yet continue to use 75 years and 85 years as screening upper age limits. Given the heterogeneity in the health of older individuals and lack of data for personalized CRC screening approaches, there remains a need for a clearer framework to inform clinical decision-making. While CRC diagnoses and mortality are still highest in older adults, treatment of CRC has evolved dramatically, with studies showing improved survival in older patients. The current US population has over 13 million individuals between ages 75 and 85, and is expected to increase to over 28 million by 2050[9,10]. The expansion of this age group coupled with increasing life expectancy requires reevaluation of the current approach to CRC screening decisions in the older population.
Currently, for CRC, there is lack of consistency in best practices to screen older individuals. Some studies indicate that screening is underutilized in older individuals and those with comorbidities[11,12]; other studies indicate that this population may be more likely to undergo screening, potentially due to frequent contact with the healthcare system[11-13]. Nevertheless, this inconsistency in the application of consensus guidelines to clinical practice indicates that knowledge gaps persist when deciding which older patients should be offered CRC screening.
A basic tenet of cancer screening is that the benefits of screening should outweigh the harms. This necessitates a reliable means for assessing the risks and estimating if a person has enough of a life and health expectancy to realize screening benefit. CRC screening is associated with short-term risks, including complications from colonoscopy, overdiagnosis, and treatment of tumors that may not have led to symptoms. Appropriate utilization of screening in older adults requires a balance between short-term screening risks and long-term benefits of early detection. Another important consideration in recommending screening is patient preference, as a patient’s values may dictate willingness to be screened.
In this review, we will examine the personalization of CRC screening in older individuals based on specific factors influencing life and health expectancy. This includes age, frailty, cognitive status, and disease burden, as these factors more accurately reflect life expectancy rather than age alone. We will also review the risks of screening unique to older adults, screening modalities and endoscopic technique for minimizing risk, and CRC treatment outcomes in older patients, in order to provide important information to aid CRC screening decisions for this age group (Figure 1). This review article offers a unique approach to this topic from both the gastroenterologist and geriatrician perspective by reviewing the use of specific clinical assessment tools, and addressing technical aspects of screening colonoscopy and periprocedural management to mitigate screening-related complications.
Figure 1.
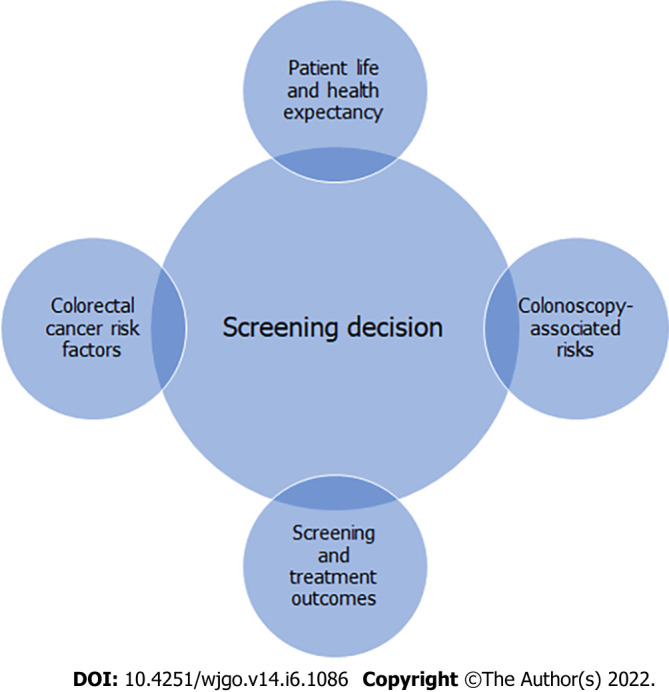
Practical considerations for colorectal cancer screening in older adults.
FACTORS FOR DECIDING WHO TO SCREEN
Guidelines currently recommend that patients with reduced life expectancy should not be screened for CRC[4-6]; however, they do not provide a method to estimate life expectancy in the clinical setting[10,14] (Table 1). A consistent method to estimate life expectancy is critical to determine the patient population who would benefit from screening. Randomized trials have suggested that the mortality difference between screened and unscreened patients becomes noticeable only after 5 or more years after screening[15,16]. Similarly, the benefits of polypectomy are delayed by 7–10 years after screening has occurred[17] and, thus, screening is of limited benefit for those with lower life expectancy. Conversely, patients with a longer life expectancy could gain more years of life from screening.
Table 1.
Comments on colorectal cancer screening across professional organizations
|
Professional organization
|
Recommended ages for screening
|
Other considerations
|
| American College of Gastroenterology (2021) | 50-75 | Screening after age 75 should be considered on an individualized basis; providers must engage in shared decision making |
| United States Preventive Services Task Force (2021) | 45-75 | Screening adults aged 76-85 should be conducted on an individualized basis; do not screen adults age 86 years and above |
| United States Multi-Society Task Force on Colorectal Cancer (2021) | 45-75 | Consider discontinuation when persons up to date with screening, who have prior negative screening reach age 75 or have < 10 yr of life expectancy. Persons without prior screening should be considered for screening up to age 85, depending on age and comorbidities. |
| Canadian Task Force on Preventive Health Care (2016) | 50-74 | Recommend not screening adults aged 75 yr and older. (Weak recommendation; low-quality evidence) |
| American College of Physicians (2019) | 50-75 | Discontinue screening in average-risk adults older than 75 yr or in adults with a life expectancy of 10 yr or less |
| American Cancer Society (2018) | 45-75 | Screening adults aged 76-85 should be conducted on an individualized basis; screening discouraged above age 85 |
Adapted from Ref. [14].
The research regarding CRC screening in older individuals is limited, many studies either do not take factors beyond age into account, or were conducted in patients younger than 75 years. The lack of high quality data that are stratified by more than age is significant. For example, Lin et al[8] found that patients aged 80 years or older had a mean extension of life expectancy with screening colonoscopy of only 0.13 years, compared to an average gain of 0.85 years for patients 50 to 54 years-old, a 6.5 fold difference. However, this study only took age into account, and did not consider comorbidity, frailty, or any other modifying factors that affect life expectancy. The conclusion that older age is associated with less years gained from screening may or may not be applicable to a given older individual, depending on their overall health and functional status.
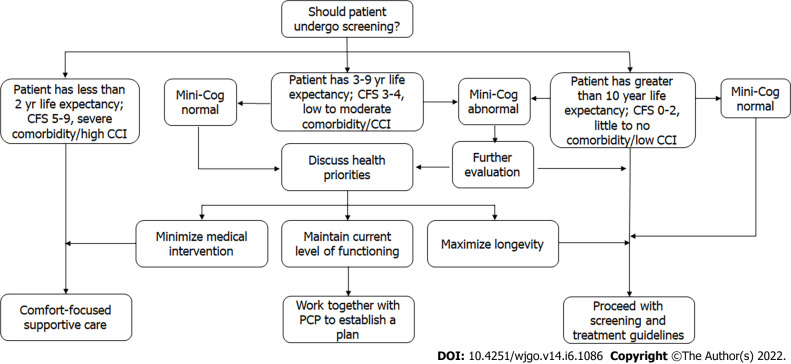
When considering screening an older patient, chronological age must be supplemented. Factors such as comorbid status, frailty, cognitive function, and patient priorities[18,19] are important tools for doing so (Figure 2).
Figure 2.
Personalized approach to colorectal cancer screening in an older patient. Adapted from Ref. [18,19]. CCI: Charlson comorbidity index; CFS: Clinical frailty scale; PCP: Primary care provider.
Comorbidity
Comorbidity status has a strong influence on life expectancy in older adults[20]. A study by DuGoff et al[21] evaluating chronic conditions and life expectancy found that the average marginal decline in life expectancy is 1.8 years with each additional chronic condition. The study found that a 67-year-old individual with no chronic conditions will live on average 22.6 additional years, while a 67-year-old individual with 5 chronic conditions will live 7.7 fewer years, and for greater than 10 chronic conditions, 17.6 fewer years[21].
Another study by Cho et al[20] evaluating Medicare claims data and the Charlson Comorbidity Index (CCI) found that persons with higher levels of comorbidity had shorter life expectancies, whereas those with no comorbid conditions, including oldest adults, had favorable life expectancies relative to an average person of the same chronological age. The study found substantial variation between estimated life expectancies for healthy persons without comorbidity and those with high levels of comorbidity, or with specific conditions, such as diabetes, CHF, and COPD. Details about the CCI are summarized in Table 2.
Table 2.
Charlson comorbidity index conditions and scoring
|
Condition
|
Value
|
Score
|
| Age | < 50, 50-59, 60-69, 70-79, 80+ | 0, +1, +2, +3, +4 |
| Myocardial infarction | Yes/no | +1 |
| CHF | Yes/no | +1 |
| PVD | Yes/no | +1 |
| CVA or TIA | Yes/no | +1 |
| Dementia | Yes/no | +1 |
| COPD | Yes/no | +1 |
| Connective tissue disease | Yes/no | +1 |
| Peptic ulcer disease | Yes/no | +1 |
| Liver disease | None/mild/severe | +1 (mild), +3 (moderate-severe) |
| Diabetes mellitus | None or diet controlled /uncomplicated/end-organ damage | +1 (uncomplicated), +2 (end-organ damage) |
| Hemiplegia | Yes/no | +2 |
| CKD | Yes/no | +2 |
| Solid tumor | None/localized/metastatic | +2 (local), +6 (metastatic) |
| Leukemia | Yes/no | +2 |
| Lymphoma | Yes/no | +2 |
| AIDS | Yes/no | +6 |
CHF: Congestive heart failure; PVD: Peripheral vascular disease; CVA: Cerebrovascular accident; TIA: Transient ischemic attack; COPD: Chronic obstructive pulmonary disorder; CKD: Chronic kidney disease; AIDS: Acquired immunodeficiency syndrome.
The total number of chronic illnesses as well as specific comorbid conditions should also be considered prior to initiating a screening colonoscopy. A systematic review by Søgaard et al[22] found that there is a 1.2- to 4.8-fold higher 5-year mortality for CRC patients with comorbidity versus without comorbidity. The total number of chronic illnesses as well as specific comorbid conditions should also be considered prior to initiating a screening colonoscopy. Even if a patient’s comorbid conditions do not specifically affect the procedure, a larger number of comorbidities may indicate decreased ability to compensate for and recover from stresses. These comorbidities, combined with normal physiologic changes of aging, create a state of homeostenosis, or decreased physiologic reserve[23]. This renders patients at higher risk for complications from even low-risk procedures, such as screening colonoscopy.
Coexisting chronic illness is associated with a substantial reduction in life expectancy after diagnosis of early-stage CRC[22]. Screening colonoscopies may also put these patients at higher risk of complications[23]. This, along with the decrease in life expectancy associated with comorbid conditions in general, should be considered when deciding whether to screen older persons.
Frailty
Frailty is a reflection of homeostenosis. It is a state of vulnerability to stressors and increased risk of adverse health outcomes due to multisystem decline in physiologic reserve and function[24]. Clinically, it presents as slow gait speed, exhaustion, weight loss, and decreased grip strength and physical activity. The presence of frailty has been shown to be predictive of falls, worsening mobility, ADL disability, hospitalization, and death. It provides important information about functional status and longevity[24,25]. Cancer patients defined as being functionally dependent according to the validated instrumental activities of daily living were found to have a 2- to 3-fold increased risk of postoperative morbidity compared with those defined as independent[26]. In the perioperative setting, the presence of frailty has been shown to independently predict postoperative complications, length of stay, likelihood of discharge to subacute nursing facilities, and mortality at 30 d and 6 mo[27].
Given that frailty has been associated with poor survival, evaluating an individual’s degree of frailty is critical when making decisions to continue CRC screening[19]. There are various published frailty scoring tools, but none have been established as the standard or best method in this setting. The most well-known is the Fried score, which measures specific criteria for unintentional weight loss, self-reported exhaustion, weakness, slow walking speed, and low physical activity[24]. However, the details needed for the Fried and many other frailty scores are often time-consuming and may require special training or tools beyond the scope of a typical outpatient office[28].
Other frailty and risk assessment tools, such as gait speed, have been evaluated and validated. Of these, the authors feel the most accessible tool in the primary care or preoperative setting is the clinical frailty scale (CFS). The CFS has been developed for rapid frailty screening without the need for specific geriatric expertise or functional testing and has been validated against the very detailed Canadian Study of Health and Aging 70-item frailty index[29]. A recent multicenter, prospective study evaluated CFS score and postoperative complications in CRC patients over the age of 80. The study found that postoperative complications were significantly more common in patients with a CFS score of 3 and above. Interestingly, age was not found to affect postoperative outcomes[30]. Details about how to apply and interpret the CFS are provided in Figure 3.
Figure 3.
Applying and interpreting the clinical frailty score. Citation: Rockwood K, Song X, MacKnight C, Bergman H, Hogan DB, McDowell I, Mitnitski A. A global clinical measure of fitness and frailty in elderly people. CMAJ 2005; 173: 489-495. Copyright © CMA Impact Inc.
ePrognosis
ePrognosis is a web-based tool that facilitates shared decision-making among clinicians and patients about colorectal and breast cancer screening. It uses a point system that assigns points based on health behaviors, disease burden, and function that are most highly associated with 10 years chance of death. Its calculator is based on data collected from The National Health Interview Survey and the Health and Retirement Survey. After data are collected, a pictograph representation of the risks versus benefits of screening is provided to give a more concrete understanding of the choice. While ePrognosis provides a framework for discussion about screening pros and cons for most patients, it does not yield a result in favor of screening anyone over the age of 75. This poses difficulty for the clinician attempting to aid an older, non-frail patient deciding whether to continue screening if they fall into this age category.
Cognitive status
Although not a normal part of aging, cognitive decline and dementia are common geriatric syndromes that may result in loss of functional status. Low ADL score and suspected dementia are associated with increased mortality in older individuals[31]. One study by Katz et al[32] found that increases in senile cognitive decline (SCD) were associated with higher risk of mortality, as were dementia and amnestic mild cognitive impairment. The study posited that the association of SCD with mortality may be due to the association of SCD with clinical cognitive status. Another study by Lv et al[33] found that faster cognitive decline was associated with higher mortality, independent of initial cognitive function, especially among those aged 65-79 years and those with normal cognitive function at baseline. This association indicates the practical significance of monitoring cognitive change in older adults, and for cancer screening purposes, provides another basis for informed screening decisions based on life expectancy.
Cognitive evaluation provides insight about the patient’s baseline functioning, ability to understand discussion of the medical issues and ability to follow instructions. Screening for cognitive impairment can be accomplished with the Mini-Cog. The Mini-Cog is a simple cognitive screening tool that is relatively uninfluenced by level of education or language. It has three steps: 3-word registration, clock drawing, and 3-word recall, and is scored on a scale of 0–5 based on number of words recalled and accuracy of the clock[34]. It may reveal previously undetected cognitive concerns, as well as inform risk assessment. This is significant in the context of minor procedures such as screening colonoscopy, as even mild cognitive impairment confers a higher risk of delirium with anesthesia[35]. Furthermore, if cognitive impairment is detected it will be important to ensure an adequate support system to help review pre- and post-procedure instructions[19].
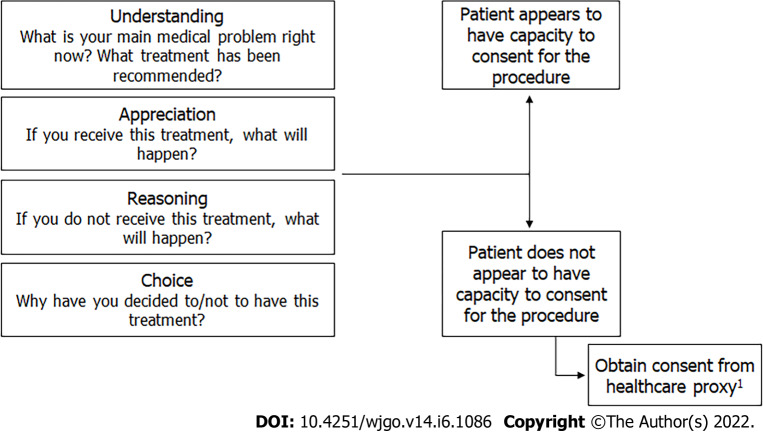
Another necessary component of cognitive evaluation is confirming medical decision-making capacity[36]. Even individuals with mild dementia can have capacity to make medical decisions, which may include decisions about screening and treatment. As these decisions necessitate weighing risks and benefits of screening, it is essential to ensure patients understand the pros and cons, as well as implications of a positive screening test. Achieving this involves conducting a capacity evaluation. The MacArthur Competence Assessment Tool-Treatment is a tool which assesses the ability to understand, appreciate, reason, and express a choice[37]. Details about how to assess medical decision-making capacity are outlined in Figure 4.
Figure 4.
Steps in the evaluation of decisional capacity. 1Include patient in process.
Given that cognitive decline and functional dependence are associated with increased mortality in older patients, overall functional status and medical capacity must be taken into consideration when making CRC screening decisions.
Personal choices
Screening decisions must be patient-centered, and should involve a discussion of patient priorities. This begins with exploring which health outcomes are most important. For some older adults, this may be longevity or surviving to a specific family event. For others, the priority may be maintaining independence, or avoiding worry and having peace of mind. Others may prioritize minimizing interventions and time spent at doctors’ offices or avoiding pain. Within the context of a patient’s health-priority outcomes, the clinician can better assist in making individual screening decisions[19].
For a patient that prefers to minimize medical intervention, but has a life expectancy exceeding ten years, a discussion may involve stool-based screening as an initial consideration, with plans to follow up a positive test with colonoscopy. For a patient that prefers to minimize pain, and has a life expectancy of only a few years, a discussion of screening cessation would be appropriate. Discussing patient health priorities allows the clinician to aid the patient in realistic goal-setting and guide decision-making.
RISK FACTORS FOR DEVELOPING CRC
There are both modifiable and non-modifiable risk factors for developing CRC, including lifestyle, a history of certain medical conditions, family history, and prior screening[1].
In the United States, more than half of all CRCs are attributable to lifestyle factors, such as unhealthy diet, sedentary lifestyle, heavy alcohol consumption, and smoking[38]. Numerous studies have shown that people with healthy lifestyle behaviors have a 27% to 52% lower risk of CRC compared to those without these behaviors[39].
Certain conditions that increase risk include a history of type 2 diabetes, personal history of CRC, or history of other cancer types due to the carcinogenic effects of some cancer treatments[1].
People with a first-degree relative who has been diagnosed with CRC have 2 to 4 times the risk of developing the disease compared to people without this family history[40]. A history of CRC among more distant relatives also increases risk, as does a family history of adenomas[41]. However, the rate of CRC in patients 80 years and above with a family history is significantly lower than that in younger age groups with a family history of CRC[42]. This study by Miller et al[42] found that family history should not be used as an inclusionary criterion for CRC screening in the 80 years and above age group for this reason.
Another risk factor to consider is screening history. Individuals with prior negative screening history may have reduced risk, whereas unscreened individuals are at a higher risk for CRC than adequately screened patients. Screening should be initiated for those who have never been screened, even after the age of 75 years for individuals without significant comorbidities[43].
Previous colonoscopy findings are also important when considering risk, as a personal history of advanced adenomatous polyps, especially multiple or large polyps, increases the risk of CRC[44,45]. Additionally, polyp shape and histology are significant[44,45]. Hyperplastic polyps have a relatively low risk for malignancy, and have little to no risk when found in the left colon[46]. Inflammatory polyps, or pseudopolyps, are benign and generally do not carry the risk of developing into CRC. Sessile serrated lesions frequently exhibit dysplasia, and disproportionally contribute to interval CRCs[47]. Villous adenomas also carry a high risk of malignancy[47].
Studies have found that individuals with advanced adenomas (≥ 10 mm, high-grade dysplasia, or tubulovillous or villous histology) and large (≥ 10 mm) serrated polyps have a significantly higher risk of CRC as compared to those without adenomas[44,45,48,49] . This is important to consider when factoring in an individual’s previous colonoscopy findings.
OUTCOMES
Colonoscopy outcomes in older adults
It has been shown that colonoscopies decrease both morbidity and mortality related to CRC. In particular, a meta-analysis identified nearly 1.5 million patients from observational studies that evaluated CRC incidence and mortality[50]. Pooled analysis of this data showed that colonoscopies are associated with a 61% relative risk reduction in CRC incidence (RR = 0.39; 95%CI: 0.26-0.60) and a 61% reduction in CRC mortality (RR = 0.39; 95%CI: 0.35-0.43) in patients who underwent a colonoscopy with non-malignant findings.
There have been recent studies assessing performing colonoscopies in older patients. A nationwide retrospective data analysis of the Department of Veterans Affairs electronic medical record identified United States male veterans at least 80 years of age that underwent a colonoscopy. They found that of nearly 81,946 patients who received a colonoscopy, 9,365 were diagnosed with CRC[51]. Of the total patients that received a colonoscopy, they found that 13% that were diagnosed with CRC were 80-84 years old, 10.2% were 85-90 years old, and 12.6% were over 90 years old. Overall, their findings suggest that there is a large number of colonoscopies being performed in patients who are at least 80 years of age and that CRC is diagnosed in both octogenarians and nonagenarians in spite of guidelines suggesting cessation of screening at 75 years of age. Other studies have shown that with older patients in particular, colonoscopies prove to be beneficial. In two large, prospective cohort studies, continuation of screening lower endoscopy after 75 years of age, regardless of screening history, was associated with reduced risk of CRC incidence and mortality[52]. These findings raise the importance of considering other factors besides age when deciding to screen a patient for CRC.
Adverse events associated with colonoscopy in older adults
Adverse effects and complications from a colonoscopy, while rare, are still something to consider when recommending colonoscopy for older patients. Studies have suggested that the benefits of endoscopy in older adults may be compromised due to a higher risk of inadequate bowel preparation, lower procedural completion rates, and higher complication rates such as perforation, bleeding, and cardiovascular adverse events[52,53]. However, these risks are associated with comorbidity[52]. A population-based matched cohort study assessed adverse events in outpatient colonoscopies among a random 5% sample of Medicare beneficiaries age 66-95 years of age from 2001-2005[54]. This study found that rates of adverse events after colonoscopy increased with age, and that patients having a polypectomy had a high risk for all adverse events compared to the matched group as well as the screening and diagnostic colonoscopy groups. This study also found that comorbid conditions increased the risk of adverse events such as history of stroke, COPD, atrial fibrillation, or CHF. Overall, this study did find the risk for adverse events to be low, with a risk of 0.6 per 1000 procedures for perforation and a risk of 2.1 per 1000 procedures for gastrointestinal bleeding for the screening group and 8.7 per 1000 procedures for the polypectomy group[54]. Other studies demonstrated that colonoscopies are safe in older patients with overall low complication rates, however these rates are still higher than they are in younger patients[55]. A systematic review found that patients 80 years of age and older experienced higher rates of cumulative gastrointestinal (GI) adverse events (incidence rate ratio 1.7; 95%CI: 1.5–1.9) and had a greater risk of perforation (incidence rate ratio 1.6, 95%CI: 1.2–2.1) compared with younger patients undergoing colonoscopy[53]. A large, population-based retrospective cohort study found the overall risk of post-colonoscopy complications to be twice as high in patients aged 75 years or older compared with that of patients aged 50 to 74 years at 30 d post procedure. While advanced age was associated with increased risk of post-colonoscopy complications, baseline comorbidities were independently associated with complications, regardless of age. Specific independent risk factors for post-colonoscopy complications were the presence of anemia, cardiac arrhythmia, heart failure, hypertension, chronic kidney disease, smoking history, liver disease, and obesity[52]. A retrospective study evaluated the complication rates of colonoscopies in patients > 90 years old compared to those 75-79 years old age[56]. Interestingly, this study found that while the complication rates in patients who were at least 90 years of age were higher, the yield for advanced neoplasia (28.4% vs 6.4% of controls, P < 0.001) as well as any neoplasia (P < 0.001 vs controls) were higher in this age group as well. The majority of the post-colonoscopy complications were cardiopulmonary events. This study sheds light on the importance of carefully judging the benefits and risks of performing a colonoscopy in oldest older patients.
Outcomes of foregoing colonoscopy
Despite some of the risks that the procedure poses in older patients, the continuation of screening endoscopy among many adults older than 75 years prevents CRC incidence and death, especially among those who do not have significant comorbidities. It is important to assess an individual’s risks for CRC in order to decide whether pursuing a screening colonoscopy is the best choice as outcomes of not performing a colonoscopy include very advanced metastatic disease. A retrospective cohort study assessed data in Saudi Arabia, where there is no population-based CRC screening, to see if there is any association between sex and metastatic CRC adjusting for patient covariates. They found over just one thousand CRC patients, and found that female patients were 20% more likely than males to present with a metastatic tumor (RR = 1.20, 95%CI: 1.04-1.38)[57]. Their reasoning for this discrepancy was that women had less access to healthcare compared to men. Another study performed at Massachusetts General Hospital assessed outcome differences in women of screening age presenting for surgical treatment for CRC[58]. This study found that unscreened women had a significantly higher risk for having high-grade tumors (RR = 1.61), lymph node metastasis (RR = 1.36), and distant metastasis on pathology (RR = 2.26). Overall, the findings in these studies suggest that patients who do not receive proper screening are at increased risk of advanced, metastatic disease. Given that there is an increase in incidence of CRC in patients beyond 75 years of age, this is an important factor to consider.
CRC surgery outcomes
While older studies have found that older patients have worse CRC surgical outcomes and survival than their younger counterparts[59], recent studies have shown otherwise[60-67]. Improvements in perioperative care, surgical techniques and the introduction of multimodal treatments have made surgery feasible for the majority of patients, including many older cancer patients[26].
These perioperative improvements include risk assessments, pre-habilitation, fast-track programs, medication list review, and patient education, and optimize the procedure[68].
More recent studies have been published that evaluate the feasibility, safety and advantages of the laparoscopic approach for colonic cancer in older patients. These have supported that minimally invasive surgery in older CRC patients reduces overall mortality and morbidity when compared to a laparotomy, and correlates with shorter hospital stays and faster functional recovery. Furthermore, it has been clearly demonstrated that postoperative outcomes in advanced age do not significantly differ from those of younger CRC patients[60-67]. One study found that intraoperative and postoperative complications were equally distributed among CRC patients over 75 years of age and younger patients[60]. Virk et al[69] found that a substantial number of octogenarians undergo surgery for treatment of stages 0–2 of CRC and have comparable 5–10-year survival (25.45%) with their younger counterparts (34.40% and 30.86% in ages 60–69 and 70–79). This study concluded that despite a statistically significant difference between age groups, the survival rates were surprisingly similar from a clinical perspective, and show that CRC surgery should still be part of the conversation in octogenarians[68].
Additionally, Flynn et al performed a single institution, retrospective cohort study of patients who underwent resection of CRC from 2010-2018 and concluded that resection of CRC in patients over the age of 85 is safe and effective and that age alone is not a sufficient reason to withhold surgical treatment in this patient cohort[67]. The study also found that both the short-term post-operative outcomes as well as 30-d mortality was similar among patients who received open surgery versus laparoscopic intervention.
Another important point is that comorbidity may play a more significant part in CRC treatment outcomes than age alone. Patients with greater comorbid medical conditions have lower survival rates after an initial diagnosis of CRC, poorer survival after chemotherapy and prolonged hospitalizations as a consequence of their CRC[53].
Various studies have demonstrated the association between Charlson Comorbidity Index (CCI) and poor CRC surgical outcomes. Zingmond et al[70] found that, of 56621 CRC patients undergoing tumor resection, those with a higher CCI were significantly associated with postoperative complications. Tan et al[71] showed that the CCI was an independent predictor of morbidity in a population of 121 octogenarians undergoing CRC surgery. Ouellette et al[72] demonstrated that CCI was associated with a longer length of stay, perioperative mortality, and overall mortality in 239 CRC patients. Please see Table 2 for more details about the CCI.
CRC surgery in the aged was previously considered high risk. Recent studies have shown that this is not necessarily the case, while advances in minimally invasive surgical technique and perioperative management have lowered the risk of postoperative complications and improved recovery.
PRACTICAL CONSIDERATIONS FOR SCREENING
Modality
Screening can be done via direct visualization methods such as colonoscopy, CT colonography, colon capsule, or flexible sigmoidoscopy, or via stool-based tests such as guaiac-based fecal occult blood test, FIT, or multitargeted stool DNA test, or via serology test such as SEPT9 DNA test[4,73].
A stool-based test such as FIT may be offered as an initial, non-invasive screening method. However, stool-based tests should not be used in older patients that are unable to undergo a follow up colonoscopy in the event that the test is positive[4]. CT colonography is another non-invasive screening technique that offers visualization that stool based tests do not. Nevertheless, it still requires bowel preparation, exposes patients to ionizing radiation, and requires follow-up colonoscopy for positive findings[4]. There is also risk of incidental findings, as CT colonography provides images of the entire abdomen and pelvis. Studies report extracolonic incidental findings in 15%-69% of imaging, and the risk likely increases with age[74]. These findings may result in unnecessary testing, burdening the patient with more cost, radiation exposure, and mental distress[74].
Colonoscopy is the gold standard, as it has the highest sensitivity among available screening modalities[75], and offers both detection and treatment of precancerous lesions at the time of the procedure. The regional distribution of CRC changes with age, with proximal colon cancer being more common in older patients, and distal colon cancer predominating in younger patients[76,77]. This is significant when considering screening modalities in older patients, as sigmoidoscopy cannot reach the proximal colon. Therefore, colonoscopy is the superior direct visualization method in older patients.
All screening tests other than colonoscopy are two-step tests. A limitation of non–colonoscopy-based CRC screening tests is that a positive test requires a follow-up colonoscopy. This must be kept in mind when deciding whether to screen an older patient, regardless of the initial screening modality. On the other hand, a major advantage of stool-based screening as a first step is that invasive screening colonoscopy is limited to those individuals with a higher pretest probability of cancer. This is important in the balance of risks and benefits, as the potential benefit of screening colonoscopy is higher in individuals with a positive FIT or other stool-based screening method.
Prevention and management of colonoscopy-associated risks in older patients
Screening colonoscopy carries the risk of major adverse events such as perforation, bleeding and cardiopulmonary complications. In addition to risks of the procedure, there are risks associated with the bowel preparation and sedation required for the colonoscopy. These risks are affected by age and influenced by additional factors such as comorbid conditions[19,52,53]. When considering colonoscopy for older individuals, caution should be taken during every step including the pre-procedure preparation, the analgesia and sedation, and the procedure itself[78] (Table 3).
Table 3.
Colonoscopy-associated risks in older adults
|
Risk
|
Associated problems
|
Mitigation
|
| Perforation | Bleed, infection, necrotic bowel | Endoscopic technique (carbon dioxide insufflation, use of pediatric endoscopic equipment, careful navigation of diverticular disease), adequate bowel preparation |
| Bleeding | Post-polypectomy bleed | Hemoclip placement for bleeding prevention when appropriate, diluted epinephrine injection, use of detachable snare, thermal coagulation |
| Cardiovascular event | Arrythmia | Medication review, screen for high-risk medications, confirm dosing appropriate for renal function; adjustment of anesthesia |
| Anticoagulation therapy interruption | Risk of thrombosis, MI, CVA | Liaise with prescribing physician; avoid colonoscopy during high-risk period; avoid interruption if possible |
| Delirium | Cognitive impairment | Risk assessment; optimize medication list, avoid holding medications with withdrawal potential on morning of procedure |
| Medication interaction | Polypharmacy increases sensitivity to anesthesia | Medication review; adjustment of anesthesia |
| Dehydration | Electrolyte disturbances | Appropriate counseling prior to colonoscopy prep; caretaker supervision to ensure patient safety during prep |
MI: Myocardial infarction; CVA: Cerebrovascular accident.
During the pre-procedure preparation of a colonoscopy, bowel preparation is essential and obtained either through polyethylene glycol-based solutions or sodium phosphate solutions[78]. Sodium phosphate has an osmotic mechanism of action, which results in fluid shifts and electrolyte abnormalities such hyperphosphatemia, hypernatremia, and hypokalemia[78]. The geriatric populations, especially those with renal or cardiac disease, are more sensitive to these changes and should therefore avoid sodium phosphate solutions[78]. Older individuals are at greater risk of dehydration as well, which can commonly occur in any bowel preparation. This can be managed with adequate hydration throughout the pre-procedure preparation[78]. Another concern throughout this process is the large volume of oral preparations required for adequate colonic preparation. Older patients may not be able to consume such a large volume and are therefore at risk for poor colonic preparation[78]. This can have major consequences including failure to complete the procedure, missed lesions, and prolonged procedure time with increased risk for complications[79]. Safe and adequate bowel preparation is very important in planning colonoscopies for older patients.
Age-related effects on the body also put the geriatric population at higher risk for adverse effects from sedation. GI colonoscopies are generally preformed under conscious sedation with numerous guidelines published for monitoring during the procedure[78]. Physiologic changes such as decreased arterial oxygenation, delayed or blunted cardiorespiratory stimulation in response to the body’s requirements, increased risk of aspiration, and reduced hepatic and renal clearance mechanisms are factors that make the choice of sedation more difficult when managing an older adult[78,80]. Due to increased sensitivity to effects of sedatives, it is recommended to administer fewer agents at a slower rate and lower dose for safe sedation during colonoscopies[78]. Endoscopies without sedation are not commonly performed in the United States, but are an option for higher risk patients[19]. In older patients with multiple comorbid medical conditions, there is greater risk of experiencing an adverse event if the colonoscopy is performed under general anesthesia[53,81]. Minimizing sedation during procedures reduces risk for anesthesia-related adverse events. During the procedure, standard monitoring procedures should be followed during procedures in the geriatric population. Maintaining a low threshold for beginning supplemental oxygen therapy is important as older patients are at higher risk of oxygen desaturation[82]. Another serious adverse event is delirium. Older adults are at an increased risk for experiencing delirium related to sedation, and this risk is further increased in older patients with underlying cognitive impairment and polypharmacy[68]. Delirium is a poor prognostic factor for survival among hospitalized older[83], and is associated with nursing home placement within the year after surgery[84].
In addition to the risks of bowel preparation and sedation, the screening colonoscopy procedure is also carries risk of perforation, bleeding, and cardiopulmonary complications. Of all adverse events associated with colonoscopy, the greatest risk associated with age is perforation. Studies show that older patients have a 30% higher risk of experiencing a perforation than younger patients undergoing colonoscopy and a 14-fold higher risk of having a perforation than patients of the same age who do not undergo the procedure[53]. Bowel perforation can be a surgical emergency and puts the patient at significant risk of life-threatening infection. Older patients are at an increased risk of perforation due to the higher prevalence of diverticulosis, tortuosity of the intestines, inadequate bowel preparation, and post-surgical adhesions or strictures, which threaten the integrity of the tissue and make the procedure more technically challenging[78,80,85,86]. Furthermore, there is a higher risk of mortality associated with perforation in older individuals[19,87]. For this reason, it is critical to weigh the risk of complications and comorbid conditions when considering colonoscopy in older patients.
Endoscopic technique
Endoscopists use a variety of techniques during colonoscopies to effectively screen for CRC. The most widely used techniques include air insufflation, carbon dioxide insufflation, water exchange, and water immersion[88]. Air insufflation, the oldest colonoscopy technique, allows for distension of colonic lumen and visualization during colonoscopy[88,89]. However, air insufflation results in unabsorbed air in the colon and prolonged bowel distension and subsequently can cause symptoms of pain and nausea[89]. This can also increase risk of postprocedural bowel ischemia and spasms, and patients may require unnecessary hospitalization or testing to rule out colonic perforation[89]. The risks of air insufflation are important to consider in older individuals as they are at higher risk of complications.
Air insufflation was followed by introduction of carbon dioxide insufflation[88]. Numerous studies have demonstrated reduction in abdominal pain during and following the completion of colonoscopy with carbon dioxide insufflation compared to air insufflation[89]. The reduction in pain is likely related to faster absorption of carbon dioxide by intestinal mucosa compared to air. The rapid absorption was confirmed early on by a study that looked at abdominal radiographs at 1 h[89]. The benefits of the reabsorption of carbon dioxide include less bowel distension, improved abdominal pain, and decreased interference in colonic mucosal blood flow[89]. However, concerns have been raised related to increased blood carbon dioxide concentrations. One study showed end-tidal carbon dioxide was increased in both carbon dioxide and air insufflation groups, suggesting this was likely related to hypoventilation from sedation rather than carbon dioxide insufflation[90]. Further studies are still required to determine risk of hypercapnia from carbon dioxide insufflation.
More recently, newer water-assisted methods including water immersion and water exchange have been used to reduce colonic spasms, facilitate cecal intubation, and lower patient discomfort and need for sedation[91]. Water immersion involves removing infused water during withdrawal while water exchange involves removing infused water during insertion[92]. The water immersion technique was initially used for patients with severe diverticulosis, as the water opened and straightened the sigmoid colon to distinguish the real lumen[91]. As older individuals are commonly affected by diverticulosis, utilizing this technique can allow for efficient completion of the procedure. Another study that investigated the results of water-assisted colonoscopies without sedation showed that this technique resulted in less reported abdominal pain and faster cecal intubation compared to air insufflation[91]. Older individuals with more comorbidities are at high risk for adverse events from sedation; this population may therefore benefit from a water-assisted colonoscopy performed with no sedation. In addition, minimizing sedation allows patients to indicate pain during the procedure, which can be indicative of risk of perforation[91,93]. Both the water immersion and water exchange techniques allow for decreased pain with insertion and completion of even difficult colonoscopies, with the advantages more pronounced in the water exchange technique[92]. Water exchange is also associated with increase in adenoma detection rate[92]. The benefits of these newer colonoscopy techniques may allow for feasible procedures in older individuals with decreased pain and risk of adverse events.
Management of anticoagulation
Over 6 million patients in the United States receive long-term anticoagulation therapy for the prevention of thromboembolism due to atrial fibrillation, mechanical heart-valve prosthesis, or venous thromboembolism[94]. Atrial fibrillation specifically affects over 33 million people worldwide and the prevalence is increasing with the aging population[95]. Older individuals with atrial fibrillation are at higher risk of stroke, as evidenced by the CHA2DS2-VASc score[95,96]. The CHA2DS2-VASc score calculates stroke risk in patients with atrial fibrillation and takes age into account, with 2 points added for individuals older than 75 and 1 point for individuals between 65-74[96]. Those at high risk are commonly started on anticoagulation, which may be a concern if a colonoscopy is required.
When considering colonoscopy in patients on anticoagulation, multiple factors need to be taken into consideration. This includes the patient’s risk of thromboembolism, the bleeding risk of the procedure, the features of the anticoagulant the patient is taking, and the urgency of the procedure[97,98]. Regarding bleeding risk of the procedure, diagnostic colonoscopy, mucosal biopsy sampling, and polypectomy less than 1 cm have low risk of bleeding[99]. However, procedures that involve an endoscopic treatment are at higher risk of bleeding; this includes polypectomy greater than or equal to 1 cm, tumor ablation, and endoscopic submucosal dissection[99]. The severity of the bleeding risk after polypectomy is between 0.3% to 10% depending on factors such as polyp size, location, morphology, resection technique, and cautery used[99]. Based on this, providers should consider the likelihood of a patient having polyps when decided how to manage the anticoagulation. Providers may consider a colonoscopy and biopsy on anticoagulation if there is low likelihood of polyps but should hold the medication if large polyps are suspected.
While the risks and benefits of stopping anticoagulation prior to colonoscopy need to be weighed for every patient, it is of utmost importance in the older population. With older individuals, they may have more comorbidities with higher risk of thromboembolism or stroke with stopping anticoagulation. If a patient is still in their high-risk window of thromboembolism, colonoscopy may be delayed until this period has passed. It is important for physicians to exercise a considerable amount of clinical judgement and consult with prescribing providers as necessary.
CONCLUSION
Screening extends life by detecting disease at an earlier stage, when there may be better chance of survival after treatment than in the absence of screening. The current approach to CRC screening in older individuals, in which the decision to offer screening is often based primarily on age, is inefficient, resulting in underuse of screening for some and overuse of screening for others.
Life and health expectancy should be used to inform cancer screening guidelines to ensure that patients live long enough to benefit from early detection. There is considerable heterogeneity of life expectancy beyond that described by chronological age, and subjective estimation of life expectancy has been shown to be inaccurate. Therefore, maximizing the potential benefits of CRC screening while minimizing potential harms requires attention to comorbidity, frailty, cognitive status, and patient preferences. The Charlson Comorbidity Index, Clinical Frailty Score, and Mini-Cog are helpful tools for evaluating these elements. Screening history and previous colonoscopy findings are also important to take into consideration when evaluating CRC risk.
Despite some of the risks that colonoscopy poses in older patients, the continuation of screening endoscopy among many adults older than 75 years prevents CRC incidence and death, especially among those who do not have significant comorbidities. Recent studies also show that fit older patients fair similarly to younger patients undergoing CRC treatment, while advances in laparoscopic surgical technique and adjuvant therapies have improved outcomes for CRC patients as a whole.
Colonoscopy-associated risks can be mitigated in older patients by conducting thorough medication review, coordinating with the prescribing provider, educating the patient about bowel preparation, adjusting sedation, and using endoscopic techniques that minimize perforation and bleed risk.
Altogether, this review emphasizes the importance of patient assessment, with chronological age being just one aspect of that evaluation, and highlights important considerations for CRC screening in older patients that age-based screening guidance may overlook.
Footnotes
Conflict-of-interest statement: The authors disclose no conflicts of interest or external funding for this publication.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Peer-review started: December 13, 2021
First decision: March 13, 2022
Article in press: April 30, 2022
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Farouk S, Egypt; Kanat O, Turkey S-Editor: Ma YJ L-Editor: A P-Editor: Ma YJ
Contributor Information
Dana Gornick, Albany Medical College, Albany Medical College, Albany, NY 12208, United States.
Anusri Kadakuntla, Albany Medical College, Albany Medical College, Albany, NY 12208, United States.
Alexa Trovato, Albany Medical College, Albany Medical College, Albany, NY 12208, United States.
Rebecca Stetzer, Division of Geriatrics, Albany Medical Center, Albany, NY 12208, United States.
Micheal Tadros, Division of Gastroenterology, Albany Medical Center, Albany, NY 12208, United States. tadrosm1@amc.edu.
References
- 1.American Cancer Society. Colorectal Cancer Facts and Figures 2020-2022. Atlanta: American Cancer Society 2020. [Google Scholar]
- 2.National Cancer Institute. SEER Cancer Statistics Review, 1975-2018. [cited September 21, 2021] Available from: https://seer.cancer.gov/csr/1975_2018/
- 3.National Cancer Institute. Percent of Deaths by Age Group: Colorectal Cancer. [cited November 15, 2021] Available from: https://seer.cancer.gov/statfacts/html/colorect.html .
- 4.Shaukat A, Kahi CJ, Burke CA, Rabeneck L, Sauer BG, Rex DK. ACG Clinical Guidelines: Colorectal Cancer Screening 2021. Am J Gastroenterol. 2021;116:458–479. doi: 10.14309/ajg.0000000000001122. [DOI] [PubMed] [Google Scholar]
- 5.USPSTF U.S. Preventative Services Task Force. In: Final Recommendation Statement. 2021. [cited December 13, 2021] Available from: https://www.uspreventiveservicestaskforce.org/uspstf/recommendation/colorectal-cancer-screening .
- 6.Wolf AMD, Fontham ETH, Church TR, Flowers CR, Guerra CE, LaMonte SJ, Etzioni R, McKenna MT, Oeffinger KC, Shih YT, Walter LC, Andrews KS, Brawley OW, Brooks D, Fedewa SA, Manassaram-Baptiste D, Siegel RL, Wender RC, Smith RA. Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society. CA Cancer J Clin. 2018;68:250–281. doi: 10.3322/caac.21457. [DOI] [PubMed] [Google Scholar]
- 7.Ko CW, Sonnenberg A. Comparing risks and benefits of colorectal cancer screening in elderly patients. Gastroenterology. 2005;129:1163–1170. doi: 10.1053/j.gastro.2005.07.027. [DOI] [PubMed] [Google Scholar]
- 8.Lin OS, Kozarek RA, Schembre DB, Ayub K, Gluck M, Drennan F, Soon MS, Rabeneck L. Screening colonoscopy in very elderly patients: prevalence of neoplasia and estimated impact on life expectancy. JAMA. 2006;295:2357–2365. doi: 10.1001/jama.295.20.2357. [DOI] [PubMed] [Google Scholar]
- 9.U.S. Census Bureau. Projections of the Population and Components of Change for the United States: 2015 to 2060. 2021. [Google Scholar]
- 10.Lansdorp-Vogelaar I, Gulati R, Mariotto AB, Schechter CB, de Carvalho TM, Knudsen AB, van Ravesteyn NT, Heijnsdijk EA, Pabiniak C, van Ballegooijen M, Rutter CM, Kuntz KM, Feuer EJ, Etzioni R, de Koning HJ, Zauber AG, Mandelblatt JS. Personalizing age of cancer screening cessation based on comorbid conditions: model estimates of harms and benefits. Ann Intern Med. 2014;161:104–112. doi: 10.7326/M13-2867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Klabunde CN, Schenck AP, Davis WW. Barriers to colorectal cancer screening among Medicare consumers. Am J Prev Med. 2006;30:313–319. doi: 10.1016/j.amepre.2005.11.006. [DOI] [PubMed] [Google Scholar]
- 12.Shapiro JA, Seeff LC, Thompson TD, Nadel MR, Klabunde CN, Vernon SW. Colorectal cancer test use from the 2005 National Health Interview Survey. Cancer Epidemiol Biomarkers Prev. 2008;17:1623–1630. doi: 10.1158/1055-9965.EPI-07-2838. [DOI] [PubMed] [Google Scholar]
- 13.Deshpande AD, McQueen A, Coups EJ. Different effects of multiple health status indicators on breast and colorectal cancer screening in a nationally representative US sample. Cancer Epidemiol. 2012;36:270–275. doi: 10.1016/j.canep.2011.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Done JZ, Fang SH. Young-onset colorectal cancer: A review. World J Gastrointest Oncol. 2021;13:856–866. doi: 10.4251/wjgo.v13.i8.856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kronborg O, Fenger C, Olsen J, Jørgensen OD, Søndergaard O. Randomised study of screening for colorectal cancer with faecal-occult-blood test. Lancet. 1996;348:1467–1471. doi: 10.1016/S0140-6736(96)03430-7. [DOI] [PubMed] [Google Scholar]
- 16.Hardcastle JD, Chamberlain JO, Robinson MH, Moss SM, Amar SS, Balfour TW, James PD, Mangham CM. Randomised controlled trial of faecal-occult-blood screening for colorectal cancer. Lancet. 1996;348:1472–1477. doi: 10.1016/S0140-6736(96)03386-7. [DOI] [PubMed] [Google Scholar]
- 17.Lee SJ, Boscardin WJ, Stijacic-Cenzer I, Conell-Price J, O'Brien S, Walter LC. Time lag to benefit after screening for breast and colorectal cancer: meta-analysis of survival data from the United States, Sweden, United Kingdom, and Denmark. BMJ. 2013;346:e8441. doi: 10.1136/bmj.e8441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Blaum CS, Rosen J, Naik AD, Smith CD, Dindo L, Vo L, Hernandez-Bigos K, Esterson J, Geda M, Ferris R, Costello D, Acampora D, Meehan T, Tinetti ME. Feasibility of Implementing Patient Priorities Care for Older Adults with Multiple Chronic Conditions. J Am Geriatr Soc. 2018;66:2009–2016. doi: 10.1111/jgs.15465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stetzer RJ, Al-Tarbsheh AH. Occult GI Bleeding: Considerations in Older Adults. Management of Occult GI Bleed. Clinical Gastroenterology Series: Humana Press; 2021. [Google Scholar]
- 20.Cho H, Klabunde CN, Yabroff KR, Wang Z, Meekins A, Lansdorp-Vogelaar I, Mariotto AB. Comorbidity-adjusted life expectancy: a new tool to inform recommendations for optimal screening strategies. Ann Intern Med. 2013;159:667–676. doi: 10.7326/0003-4819-159-10-201311190-00005. [DOI] [PubMed] [Google Scholar]
- 21.DuGoff EH, Canudas-Romo V, Buttorff C, Leff B, Anderson GF. Multiple chronic conditions and life expectancy: a life table analysis. Med Care. 2014;52:688–694. doi: 10.1097/MLR.0000000000000166. [DOI] [PubMed] [Google Scholar]
- 22.Søgaard M, Thomsen RW, Bossen KS, Sørensen HT, Nørgaard M. The impact of comorbidity on cancer survival: a review. Clin Epidemiol. 2013;5:3–29. doi: 10.2147/CLEP.S47150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Halter JB, Studenski S, High KP, Asthana S, Woolard N, Ritchie CS, Supiano MA. Aging and Homeostatic Regulation. Hazzard's Geriatric Medicine and Gerontology. 7th ed., 2016: p2096. [Google Scholar]
- 24.Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, Seeman T, Tracy R, Kop WJ, Burke G, McBurnie MA Cardiovascular Health Study Collaborative Research Group. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56:M146–M156. doi: 10.1093/gerona/56.3.m146. [DOI] [PubMed] [Google Scholar]
- 25.Shamliyan T, Talley KM, Ramakrishnan R, Kane RL. Association of frailty with survival: a systematic literature review. Ageing Res Rev. 2013;12:719–736. doi: 10.1016/j.arr.2012.03.001. [DOI] [PubMed] [Google Scholar]
- 26.Ugolini G, Ghignone F, Zattoni D, Veronese G, Montroni I. Personalized surgical management of colorectal cancer in elderly population. World J Gastroenterol. 2014;20:3762–3777. doi: 10.3748/wjg.v20.i14.3762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Partridge JS, Harari D, Dhesi JK. Frailty in the older surgical patient: a review. Age Ageing. 2012;41:142–147. doi: 10.1093/ageing/afr182. [DOI] [PubMed] [Google Scholar]
- 28.Huisman MG, Kok M, de Bock GH, van Leeuwen BL. Delivering tailored surgery to older cancer patients: Preoperative geriatric assessment domains and screening tools - A systematic review of systematic reviews. Eur J Surg Oncol. 2017;43:1–14. doi: 10.1016/j.ejso.2016.06.003. [DOI] [PubMed] [Google Scholar]
- 29.Rockwood K, Song X, MacKnight C, Bergman H, Hogan DB, McDowell I, Mitnitski A. A global clinical measure of fitness and frailty in elderly people. CMAJ. 2005;173:489–495. doi: 10.1503/cmaj.050051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Niemeläinen S, Huhtala H, Andersen J, Ehrlich A, Haukijärvi E, Koikkalainen S, Koskensalo S, Kössi J, Mattila A, Pinta T, Uotila-Nieminen M, Vihervaara H, Hyöty M, Jämsen E. The Clinical Frailty Scale is a useful tool for predicting postoperative complications following elective colon cancer surgery at the age of 80 years and above: A prospective, multicentre observational study. Colorectal Dis. 2021;23:1824–1836. doi: 10.1111/codi.15689. [DOI] [PubMed] [Google Scholar]
- 31.Bahat G, Tufan F, Bahat Z, Tufan A, Aydin Y, Akpinar TS, Erten N, Karan MA. Observational cohort study on correlates of mortality in older community-dwelling outpatients: The value of functional assessment. Geriatr Gerontol Int. 2015;15:1219–1226. doi: 10.1111/ggi.12422. [DOI] [PubMed] [Google Scholar]
- 32.Katz MJ, Wang C, Derby CA, Lipton RB, Zimmerman ME, Sliwinski MJ, Rabin LA. Subjective Cognitive Decline Prediction of Mortality: Results from the Einstein Aging Study. J Alzheimers Dis. 2018;66:239–248. doi: 10.3233/JAD-180335. [DOI] [PubMed] [Google Scholar]
- 33.Lv X, Li W, Ma Y, Chen H, Zeng Y, Yu X, Hofman A, Wang H. Cognitive decline and mortality among community-dwelling Chinese older people. BMC Med. 2019;17:63. doi: 10.1186/s12916-019-1295-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Borson S, Scanlan JM, Watanabe J, Tu SP, Lessig M. Improving identification of cognitive impairment in primary care. Int J Geriatr Psychiatry. 2006;21:349–355. doi: 10.1002/gps.1470. [DOI] [PubMed] [Google Scholar]
- 35.Pompei P, Foreman M, Rudberg MA, Inouye SK, Braund V, Cassel CK. Delirium in hospitalized older persons: outcomes and predictors. J Am Geriatr Soc. 1994;42:809–815. doi: 10.1111/j.1532-5415.1994.tb06551.x. [DOI] [PubMed] [Google Scholar]
- 36.Etchells E, Darzins P, Silberfeld M, Singer PA, McKenny J, Naglie G, Katz M, Guyatt GH, Molloy DW, Strang D. Assessment of patient capacity to consent to treatment. J Gen Intern Med. 1999;14:27–34. doi: 10.1046/j.1525-1497.1999.00277.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Appelbaum PS. Clinical practice. Assessment of patients' competence to consent to treatment. N Engl J Med. 2007;357:1834–1840. doi: 10.1056/NEJMcp074045. [DOI] [PubMed] [Google Scholar]
- 38.Islami F, Goding Sauer A, Miller KD, Siegel RL, Fedewa SA, Jacobs EJ, McCullough ML, Patel AV, Ma J, Soerjomataram I, Flanders WD, Brawley OW, Gapstur SM, Jemal A. Proportion and number of cancer cases and deaths attributable to potentially modifiable risk factors in the United States. CA Cancer J Clin. 2018;68:31–54. doi: 10.3322/caac.21440. [DOI] [PubMed] [Google Scholar]
- 39.Kohler LN, Garcia DO, Harris RB, Oren E, Roe DJ, Jacobs ET. Adherence to Diet and Physical Activity Cancer Prevention Guidelines and Cancer Outcomes: A Systematic Review. Cancer Epidemiol Biomarkers Prev. 2016;25:1018–1028. doi: 10.1158/1055-9965.EPI-16-0121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lowery JT, Ahnen DJ, Schroy PC 3rd, Hampel H, Baxter N, Boland CR, Burt RW, Butterly L, Doerr M, Doroshenk M, Feero WG, Henrikson N, Ladabaum U, Lieberman D, McFarland EG, Peterson SK, Raymond M, Samadder NJ, Syngal S, Weber TK, Zauber AG, Smith R. Understanding the contribution of family history to colorectal cancer risk and its clinical implications: A state-of-the-science review. Cancer. 2016;122:2633–2645. doi: 10.1002/cncr.30080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tuohy TM, Rowe KG, Mineau GP, Pimentel R, Burt RW, Samadder NJ. Risk of colorectal cancer and adenomas in the families of patients with adenomas: a population-based study in Utah. Cancer. 2014;120:35–42. doi: 10.1002/cncr.28227. [DOI] [PubMed] [Google Scholar]
- 42.Miller DR, Averbukh L, Virk G, Jafri M, Tadros M. The Value of Family History in Colorectal Screening Decisions for Oldest Old Geriatric Populations. Cureus. 2021;13:e12815. doi: 10.7759/cureus.12815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ma W, Wang K, Nguyen LH, Joshi A, Cao Y, Nishihara R, Wu K, Ogino S, Giovannucci EL, Song M, Chan AT. Association of Screening Lower Endoscopy With Colorectal Cancer Incidence and Mortality in Adults Older Than 75 Years. JAMA Oncol. 2021;7:985–992. doi: 10.1001/jamaoncol.2021.1364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lieberman DA, Weiss DG, Harford WV, Ahnen DJ, Provenzale D, Sontag SJ, Schnell TG, Chejfec G, Campbell DR, Kidao J, Bond JH, Nelson DB, Triadafilopoulos G, Ramirez FC, Collins JF, Johnston TK, McQuaid KR, Garewal H, Sampliner RE, Esquivel R, Robertson D. Five-year colon surveillance after screening colonoscopy. Gastroenterology. 2007;133:1077–1085. doi: 10.1053/j.gastro.2007.07.006. [DOI] [PubMed] [Google Scholar]
- 45.Løberg M, Kalager M, Holme Ø, Hoff G, Adami HO, Bretthauer M. Long-term colorectal-cancer mortality after adenoma removal. N Engl J Med. 2014;371:799–807. doi: 10.1056/NEJMoa1315870. [DOI] [PubMed] [Google Scholar]
- 46.Lin OS, Schembre DB, McCormick SE, Gluck M, Patterson DJ, Jiranek GC, Soon MS, Kozarek RA. Risk of proximal colorectal neoplasia among asymptomatic patients with distal hyperplastic polyps. Am J Med. 2005;118:1113–1119. doi: 10.1016/j.amjmed.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 47.Heitman SJ, Ronksley PE, Hilsden RJ, Manns BJ, Rostom A, Hemmelgarn BR. Prevalence of adenomas and colorectal cancer in average risk individuals: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2009;7:1272–1278. doi: 10.1016/j.cgh.2009.05.032. [DOI] [PubMed] [Google Scholar]
- 48.He X, Hang D, Wu K, Nayor J, Drew DA, Giovannucci EL, Ogino S, Chan AT, Song M. Long-term Risk of Colorectal Cancer After Removal of Conventional Adenomas and Serrated Polyps. Gastroenterology. 2020;158:852–861.e4. doi: 10.1053/j.gastro.2019.06.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Click B, Pinsky PF, Hickey T, Doroudi M, Schoen RE. Association of Colonoscopy Adenoma Findings With Long-term Colorectal Cancer Incidence. JAMA. 2018;319:2021–2031. doi: 10.1001/jama.2018.5809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pan J, Xin L, Ma YF, Hu LH, Li ZS. Colonoscopy Reduces Colorectal Cancer Incidence and Mortality in Patients With Non-Malignant Findings: A Meta-Analysis. Am J Gastroenterol. 2016;111:355–365. doi: 10.1038/ajg.2015.418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Virk GS, Jafri M, Ashley C. Colonoscopy and colorectal cancer rates among octogenarians and nonagenarians: nationwide study of US veterans. Clin Interv Aging. 2019;14:609–614. doi: 10.2147/CIA.S192497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Causada-Calo N, Bishay K, Albashir S, Al Mazroui A, Armstrong D. Association Between Age and Complications After Outpatient Colonoscopy. JAMA Netw Open. 2020;3:e208958. doi: 10.1001/jamanetworkopen.2020.8958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Day LW, Kwon A, Inadomi JM, Walter LC, Somsouk M. Adverse events in older patients undergoing colonoscopy: a systematic review and meta-analysis. Gastrointest Endosc. 2011;74:885–896. doi: 10.1016/j.gie.2011.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Warren JL, Klabunde CN, Mariotto AB, Meekins A, Topor M, Brown ML, Ransohoff DF. Adverse events after outpatient colonoscopy in the Medicare population. Ann Intern Med. 2009;150:849–857, W152. doi: 10.7326/0003-4819-150-12-200906160-00008. [DOI] [PubMed] [Google Scholar]
- 55.Lippert E, Herfarth HH, Grunert N, Endlicher E, Klebl F. Gastrointestinal endoscopy in patients aged 75 years and older: risks, complications, and findings--a retrospective study. Int J Colorectal Dis. 2015;30:363–366. doi: 10.1007/s00384-014-2088-3. [DOI] [PubMed] [Google Scholar]
- 56.Cha JM, Kozarek RA, La Selva D, Gluck M, Ross A, Chiorean M, Koch J, Lin OS. Risks and Benefits of Colonoscopy in Patients 90 Years or Older, Compared With Younger Patients. Clin Gastroenterol Hepatol. 2016;14:80–86.e1. doi: 10.1016/j.cgh.2015.06.036. [DOI] [PubMed] [Google Scholar]
- 57.Alyabsi M, Sabatin F, Jazieh AR. The Outcome of Unscreened Population in Colorectal Cancer: The Impact of Sex and Other Determinants on Cancer Stage. Cancer Manag Res. 2020;12:12319–12327. doi: 10.2147/CMAR.S268823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Amri R, Bordeianou LG, Sylla P, Berger DL. The fate of unscreened women in colon cancer: impact on staging and prognosis. Am J Surg. 2015;209:927–934. doi: 10.1016/j.amjsurg.2014.09.033. [DOI] [PubMed] [Google Scholar]
- 59.Rabeneck L, Davila JA, Thompson M, El-Serag HB. Outcomes in elderly patients following surgery for colorectal cancer in the veterans affairs health care system. Aliment Pharmacol Ther. 2004;20:1115–1124. doi: 10.1111/j.1365-2036.2004.02215.x. [DOI] [PubMed] [Google Scholar]
- 60.Scheidbach H, Schneider C, Hügel O, Yildirim C, Lippert H, Köckerling F. Laparoscopic surgery in the old patient: do indications and outcomes differ? Langenbecks Arch Surg. 2005;390:328–332. doi: 10.1007/s00423-005-0560-9. [DOI] [PubMed] [Google Scholar]
- 61.Iroatulam AJ, Chen HH, Potenti FM, Parameswaran S, Wexner SD. Laparoscopic colectomy yields similar morbidity and disability regardless of patient age. Int J Colorectal Dis. 1999;14:155–157. doi: 10.1007/s003840050202. [DOI] [PubMed] [Google Scholar]
- 62.She WH, Poon JT, Fan JK, Lo OS, Law WL. Outcome of laparoscopic colectomy for cancer in elderly patients. Surg Endosc. 2013;27:308–312. doi: 10.1007/s00464-012-2466-2. [DOI] [PubMed] [Google Scholar]
- 63.Poon JT, Law WL, Chow LC, Fan JK, Lo SH. Outcome of laparoscopic resection for colorectal cancer in patients with high operative risk. Ann Surg Oncol. 2011;18:1884–1890. doi: 10.1245/s10434-010-1530-1. [DOI] [PubMed] [Google Scholar]
- 64.Cheung HY, Chung CC, Fung JT, Wong JC, Yau KK, Li MK. Laparoscopic resection for colorectal cancer in octogenarians: results in a decade. Dis Colon Rectum. 2007;50:1905–1910. doi: 10.1007/s10350-007-9070-x. [DOI] [PubMed] [Google Scholar]
- 65.Isbister WH. Colorectal surgery in the elderly: an audit of surgery in octogenarians. Aust N Z J Surg. 1997;67:557–561. doi: 10.1111/j.1445-2197.1997.tb02038.x. [DOI] [PubMed] [Google Scholar]
- 66.Law WL, Chu KW, Tung PH. Laparoscopic colorectal resection: a safe option for elderly patients. J Am Coll Surg. 2002;195:768–773. doi: 10.1016/s1072-7515(02)01483-7. [DOI] [PubMed] [Google Scholar]
- 67.Flynn DE, Mao D, Yerkovich S, Franz R, Iswariah H, Hughes A, Shaw I, Tam D, Chandrasegaram M. Should we resect colorectal cancer in patients over the age of 85? World J Gastrointest Oncol. 2021;13:185–196. doi: 10.4251/wjgo.v13.i3.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Cheema FN, Abraham NS, Berger DH, Albo D, Taffet GE, Naik AD. Novel approaches to perioperative assessment and intervention may improve long-term outcomes after colorectal cancer resection in older adults. Ann Surg. 2011;253:867–874. doi: 10.1097/SLA.0b013e318208faf0. [DOI] [PubMed] [Google Scholar]
- 69.Virk GS, Jafri M, Mehdi S, Ashley C. Staging and survival of colorectal cancer (CRC) in octogenarians: Nationwide Study of US Veterans. J Gastrointest Oncol. 2019;10:12–18. doi: 10.21037/jgo.2018.09.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Zingmond D, Maggard M, O'Connell J, Liu J, Etzioni D, Ko C. What predicts serious complications in colorectal cancer resection? Am Surg. 2003;69:969–974. [PubMed] [Google Scholar]
- 71.Tan KY, Kawamura YJ, Tokomitsu A, Tang T. Assessment for frailty is useful for predicting morbidity in elderly patients undergoing colorectal cancer resection whose comorbidities are already optimized. Am J Surg. 2012;204:139–143. doi: 10.1016/j.amjsurg.2011.08.012. [DOI] [PubMed] [Google Scholar]
- 72.Ouellette JR, Small DG, Termuhlen PM. Evaluation of Charlson-Age Comorbidity Index as predictor of morbidity and mortality in patients with colorectal carcinoma. J Gastrointest Surg. 2004;8:1061–1067. doi: 10.1016/j.gassur.2004.09.045. [DOI] [PubMed] [Google Scholar]
- 73.U . Screening for colorectal cancer: recommendation and rationale. Ann Intern Med. 2002;137:129–131. doi: 10.7326/0003-4819-137-2-200207160-00014. [DOI] [PubMed] [Google Scholar]
- 74.Moore JS, Aulet TH. Colorectal Cancer Screening. Surg Clin North Am. 2017;97:487–502. doi: 10.1016/j.suc.2017.01.001. [DOI] [PubMed] [Google Scholar]
- 75.Lin JS, Piper MA, Perdue LA, Rutter CM, Webber EM, O'Connor E, Smith N, Whitlock EP. Screening for Colorectal Cancer: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. 2016;315:2576–2594. doi: 10.1001/jama.2016.3332. [DOI] [PubMed] [Google Scholar]
- 76.Tadros M, Mago S, Miller D, Ungemack JA, Anderson JC, Swede H. The rise of proximal colorectal cancer: a trend analysis of subsite specific primary colorectal cancer in the SEER database. Ann Gastroenterol. 2021;34:559–567. doi: 10.20524/aog.2021.0608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Reif de Paula T, Simon HL, Profeta da Luz MM, Keller DS. Right sided colorectal cancer increases with age and screening should be tailored to reflect this: a national cancer database study. Tech Coloproctol. 2021;25:81–89. doi: 10.1007/s10151-020-02329-z. [DOI] [PubMed] [Google Scholar]
- 78.ASGE Standards of Practice Committee. Chandrasekhara V, Early DS, Acosta RD, Chathadi KV, Decker GA, Evans JA, Fanelli RD, Fisher DA, Foley KQ, Fonkalsrud L, Hwang JH, Jue T, Khashab MA, Lightdale JR, Muthusamy VR, Pasha SF, Saltzman JR, Sharaf R, Shergill AK, Cash BD. Modifications in endoscopic practice for the elderly. Gastrointest Endosc. 2013;78:1–7. doi: 10.1016/j.gie.2013.04.161. [DOI] [PubMed] [Google Scholar]
- 79.Rex DK, Imperiale TF, Latinovich DR, Bratcher LL. Impact of bowel preparation on efficiency and cost of colonoscopy. Am J Gastroenterol. 2002;97:1696–1700. doi: 10.1111/j.1572-0241.2002.05827.x. [DOI] [PubMed] [Google Scholar]
- 80.Travis AC, Pievsky D, Saltzman JR. Endoscopy in the elderly. Am J Gastroenterol. 2012;107:1495–1501; quiz 1494, 1502. doi: 10.1038/ajg.2012.246. [DOI] [PubMed] [Google Scholar]
- 81.Cooper GS, Kou TD, Rex DK. Complications following colonoscopy with anesthesia assistance: a population-based analysis. JAMA Intern Med. 2013;173:551–556. doi: 10.1001/jamainternmed.2013.2908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Reed MW, O'Leary DP, Duncan JL, Majeed AW, Wright B, Reilly CS. Effects of sedation and supplemental oxygen during upper alimentary tract endoscopy. Scand J Gastroenterol. 1993;28:319–322. doi: 10.3109/00365529309090249. [DOI] [PubMed] [Google Scholar]
- 83.Kiely DK, Marcantonio ER, Inouye SK, Shaffer ML, Bergmann MA, Yang FM, Fearing MA, Jones RN. Persistent delirium predicts greater mortality. J Am Geriatr Soc. 2009;57:55–61. doi: 10.1111/j.1532-5415.2008.02092.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.McAvay GJ, Van Ness PH, Bogardus ST Jr, Zhang Y, Leslie DL, Leo-Summers LS, Inouye SK. Older adults discharged from the hospital with delirium: 1-year outcomes. J Am Geriatr Soc. 2006;54:1245–1250. doi: 10.1111/j.1532-5415.2006.00815.x. [DOI] [PubMed] [Google Scholar]
- 85.Jafri SM, Monkemuller K, Lukens FJ. Endoscopy in the elderly: a review of the efficacy and safety of colonoscopy, esophagogastroduodenoscopy, and endoscopic retrograde cholangiopancreatography. J Clin Gastroenterol. 2010;44:161–166. doi: 10.1097/MCG.0b013e3181c64d64. [DOI] [PubMed] [Google Scholar]
- 86.Lin OS. Performing colonoscopy in elderly and very elderly patients: Risks, costs and benefits. World J Gastrointest Endosc. 2014;6:220–226. doi: 10.4253/wjge.v6.i6.220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Gatto NM, Frucht H, Sundararajan V, Jacobson JS, Grann VR, Neugut AI. Risk of perforation after colonoscopy and sigmoidoscopy: a population-based study. J Natl Cancer Inst. 2003;95:230–236. doi: 10.1093/jnci/95.3.230. [DOI] [PubMed] [Google Scholar]
- 88.Zhang Z, Wu Y, Sun G, Zhang J, Li J, Qiu C, Zheng X, Wang B, Yang L, Wang X. Bayesian network meta-analysis: Efficacy of air insufflation, CO2 insufflation, water exchange, and water immersion in colonoscopy. Dig Endosc. 2018;30:321–331. doi: 10.1111/den.13012. [DOI] [PubMed] [Google Scholar]
- 89.Memon MA, Memon B, Yunus RM, Khan S. Carbon Dioxide Versus Air Insufflation for Elective Colonoscopy: A Meta-Analysis and Systematic Review of Randomized Controlled Trials. Surg Laparosc Endosc Percutan Tech. 2016;26:102–116. doi: 10.1097/SLE.0000000000000243. [DOI] [PubMed] [Google Scholar]
- 90.Bretthauer M, Lynge AB, Thiis-Evensen E, Hoff G, Fausa O, Aabakken L. Carbon dioxide insufflation in colonoscopy: safe and effective in sedated patients. Endoscopy. 2005;37:706–709. doi: 10.1055/s-2005-870154. [DOI] [PubMed] [Google Scholar]
- 91.Sugimoto S, Mizukami T. Diagnostic and therapeutic applications of water-immersion colonoscopy. World J Gastroenterol. 2015;21:6451–6459. doi: 10.3748/wjg.v21.i21.6451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Cadoni S, Leung FW. Water-Assisted Colonoscopy. Curr Treat Options Gastroenterol. 2017;15:135–154. doi: 10.1007/s11938-017-0119-1. [DOI] [PubMed] [Google Scholar]
- 93.Leung CW, Kaltenbach T, Soetikno R, Wu KK, Leung FW, Friedland S. Water immersion versus standard colonoscopy insertion technique: randomized trial shows promise for minimal sedation. Endoscopy. 2010;42:557–563. doi: 10.1055/s-0029-1244231. [DOI] [PubMed] [Google Scholar]
- 94.Roger VL, Go AS, Lloyd-Jones DM, Benjamin EJ, Berry JD, Borden WB, Bravata DM, Dai S, Ford ES, Fox CS, Fullerton HJ, Gillespie C, Hailpern SM, Heit JA, Howard VJ, Kissela BM, Kittner SJ, Lackland DT, Lichtman JH, Lisabeth LD, Makuc DM, Marcus GM, Marelli A, Matchar DB, Moy CS, Mozaffarian D, Mussolino ME, Nichol G, Paynter NP, Soliman EZ, Sorlie PD, Sotoodehnia N, Turan TN, Virani SS, Wong ND, Woo D, Turner MB American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics--2012 update: a report from the American Heart Association. Circulation. 2012;125:e2–e220. doi: 10.1161/CIR.0b013e31823ac046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Kato ET, Goto S, Giugliano RP. Overview of oral antithrombotic treatment in elderly patients with atrial fibrillation. Ageing Res Rev. 2019;49:115–124. doi: 10.1016/j.arr.2018.10.006. [DOI] [PubMed] [Google Scholar]
- 96.Lip GY, Nieuwlaat R, Pisters R, Lane DA, Crijns HJ. Refining clinical risk stratification for predicting stroke and thromboembolism in atrial fibrillation using a novel risk factor-based approach: the euro heart survey on atrial fibrillation. Chest. 2010;137:263–272. doi: 10.1378/chest.09-1584. [DOI] [PubMed] [Google Scholar]
- 97.Baron TH, Kamath PS, McBane RD. New anticoagulant and antiplatelet agents: a primer for the gastroenterologist. Clin Gastroenterol Hepatol. 2014;12:187–195. doi: 10.1016/j.cgh.2013.05.020. [DOI] [PubMed] [Google Scholar]
- 98.Baron TH, Kamath PS, McBane RD. Management of antithrombotic therapy in patients undergoing invasive procedures. N Engl J Med. 2013;368:2113–2124. doi: 10.1056/NEJMra1206531. [DOI] [PubMed] [Google Scholar]
- 99.ASGE Standards of Practice Committee. Acosta RD, Abraham NS, Chandrasekhara V, Chathadi KV, Early DS, Eloubeidi MA, Evans JA, Faulx AL, Fisher DA, Fonkalsrud L, Hwang JH, Khashab MA, Lightdale JR, Muthusamy VR, Pasha SF, Saltzman JR, Shaukat A, Shergill AK, Wang A, Cash BD, DeWitt JM. The management of antithrombotic agents for patients undergoing GI endoscopy. Gastrointest Endosc. 2016;83:3–16. doi: 10.1016/j.gie.2015.09.035. [DOI] [PubMed] [Google Scholar]