Abstract
Gastric cancer is strongly associated with atrophic gastritis associated with Helicobacter pylori infection. The eradication of H. pylori has been reported to improve inflammation of the gastric mucosa, atrophy, and intestinal metaplasia and has also been shown to inhibit the development and growth of gastric cancer. Advanced gastric cancer from H. pylori-negative mucosa without inflammation, atrophy, or intestinal epithelialization is rarely found. We report on two cases of advanced gastric cancer without endoscopic evidence of gastric mucosal atrophy. One case was in the gastric angle 9 years after H. pylori eradication and the other case was in the pylorus of an uninfected stomach. Although gastric cancer is strongly associated with atrophic gastritis caused by H. pylori infection, postoperative histopathological examination of the stomach in both cases showed no inflammation, atrophy, or intestinal metaplasia. Poorly differentiated adenocarcinomas were confirmed at the microscopic level, and both cases were determined to be type 4 using the Borrmann classification. There is a body of evidence showing that H. pylori infection can cause gastric cancer and is the most prevalent infection-induced cancer in the world. The 2 cases here displayed different carcinogenesis than gastric mucosal atrophy caused by H. pylori infection. With the spread of H. pylori eradication treatments and an increasing number of uninfected patients, these case reports can contribute to future strategies for the diagnosis of gastric cancer.
Keywords: Gastric cancer, Helicobacter pylori infection, Atrophic gastritis, Endoscopy, Carcinogenesis
Introduction
Gastric cancer (GC) is a major leading cause of cancer deaths worldwide and is associated with high morbidity. Numerous studies have shown that GC is strongly associated with atrophic gastritis from Helicobacter pylori infection [1]. H. pylori is confirmed as a class I carcinogen of GC development by the World Health Organization and causes chronic gastric inflammation resulting in precancerous changes of atrophic gastritis and intestinal metaplasia. The severity and extent of these changes are associated with a higher risk of GC [2].
The eradication of H. pylori has been reported to improve the inflammation of the gastric mucosa as well as atrophy and intestinal metaplasia [3] and has also been shown to inhibit the development [4] and growth of GC [5]. On the other hand, GCs in uninfected stomachs are extremely rare [6]. When they do occur, they are highly differentiated, and most are diagnosed as early-stage cancers [7], with few reports of advanced GC [8]. This is a report on 2 cases of advanced GC in which histopathological examination of the resected stomach showed no inflammation, atrophy, or intestinal epithelialization of the gastric mucosa. We believe that these cases are valuable for future GC diagnosis strategies.
Case Presentation
Case 1
The patient was a female in her seventies who underwent an ABC classification [9] GC screening, which combined serum pepsinogen (PG) and H. pylori antibodies. The results (PGI: 111.9 ng/mL, PGII: 44.1 ng/mL, I/II ratio: 2.5, H. pylori antibodies: 29.4 U/mL) indicated a group B diagnosis. She did not receive a detailed examination at that time but did undergo an upper gastrointestinal endoscopy the following year and received eradication therapy based on a diagnosis of gastritis. Seven years after the eradication therapy, blood tests showed a successful eradication (PGI: 30 ng/mL, PGII: 4.5 ng/mL, I/II ratio: 6.7, H. pylori antibodies: <3 U/mL). After that, the patient underwent outpatient treatment at a local doctor with a diagnosis of dyslipidemia and hypertension, but no upper gastrointestinal endoscopy was performed.
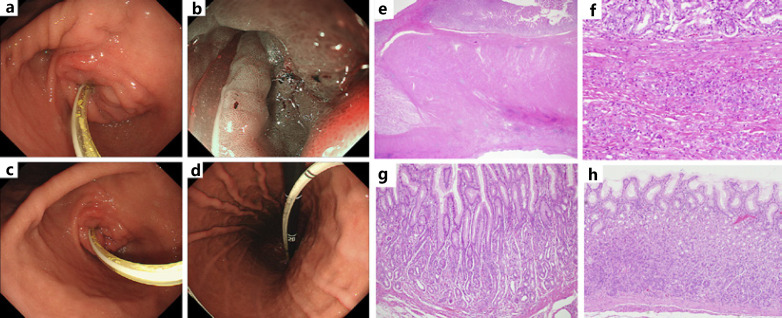
Nine years after the eradication therapy, she developed mild anemia (hemoglobin: 9.0 g/mL), an endoscopy discovered GC, and she was then referred to us. Figure 1a-d shows the findings of an upper gastrointestinal endoscopy at our hospital. There was a retraction in the posterior wall of the gastric angle (Fig. 1a), some ulceration was observed in the proximal image, and the mucosa on the side of the large fold was extensively faded and raised in a trapezoidal shape (Fig. 1b). The mucosa of the antrum and gastric body showed no atrophic changes, such as visible blood vessels (Fig. 1c, d). A gastric biopsy revealed group 5, poorly differentiated adenocarcinoma. The preoperative diagnosis was scirrhous-type GC, stage IV, and a total gastrectomy was performed. Figure 1e-h shows the histopathological images of the tumor area and background mucosa. It was poorly differentiated adenocarcinoma (por2) with marked fibrosis (Fig. 1f), and the tumor cells had invaded the serosa (Fig. 1e). The tumor was 14.0 × 8.0 cm, and the final diagnosis was type 4 GC, pT4a (SE), por > sig, pN1, M0, INFc, ly0, V2, and stage IIIB according to Japanese GC guidelines. In the background mucosal examination, there was no inflammatory cell infiltration, no atrophy, or intestinal metaplasia within either the pyloric or gastric fundic gland mucosa (Fig. 1g, h).
Fig. 1.
Endoscopic findings and histopathological findings of the resected stomach of case 1. An ulcerative lesion with sclerosis was found on the posterior wall of the gastric angle (a, b). There were no atrophic changes in the pyloric (c) or corpus mucosa (d), suggesting an apparently uninfected stomach. The tumor cells invaded the serosa with marked fibrosis (e). There was a diffuse intramucosal growth of undifferentiated tumor cells (f). Histopathological findings of the mucosa in the pyloric (g) and fundic gland regions (h) showed no inflammation, no mucosal atrophy, and no intestinal metaplasia (e–h: H&E staining, (e): 2 × 10, (f–h): 20 × 10).
Case 2
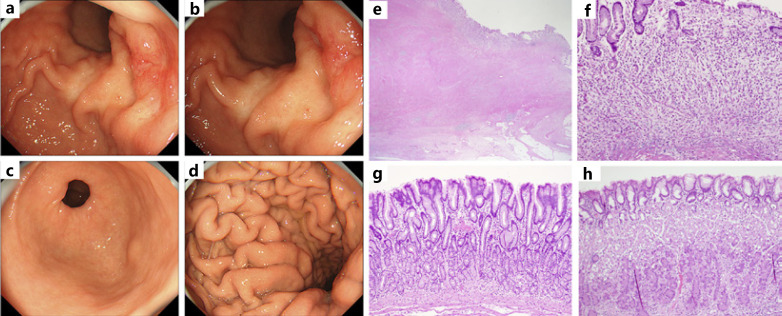
The female patient visited her local doctor for nausea and vomiting. Thirteen years prior, she had a surgical operation for colorectal cancer (stage IIIa, moderately differentiated adenocarcinoma). An upper gastrointestinal endoscopy showed stenosis in the pylorus, and after the insertion of a drainage tube, the patient was referred to our hospital. A gastric biopsy at the local doctor's office showed no malignancy or H. pylori infection. Figure 2a-d shows the findings of the upper gastrointestinal endoscopy at our hospital. The pyloric region was stenotic with folds (Fig. 2a), and some ulcerative lesions were found in the proximal view (Fig. 2b). A gastric biopsy revealed group 5, poorly differentiated adenocarcinoma. The pyloric gland mucosa in front of the stenosis and the mucosa of the gastric body did not show any atrophic changes, such as vascular permeability (Fig. 2c, d). The patient was diagnosed with pyloric stenosis due to GC (stage IV) and underwent a subtotal gastrectomy. The tumor was poorly differentiated adenocarcinoma (por2) with collagen fiber proliferation (Fig. 2e, f), and the tumor cells had invaded the serosa. The tumor was 6.5 × 4.3 cm, and the final diagnosis was type 4 GC (por2 adenocarcinoma), and the tumor stage was pT3, pN0, INFc, Ly0, V1b, and pstage IIB. The background mucosal examination revealed that both the pyloric and fundic glands were intact, and there was no inflammation of the gastric mucosa or intestinal metaplasia (Fig. 2g, h).
Fig. 2.
Endoscopic findings and histopathological findings of the resected stomach of case 2. A drainage tube was inserted due to the pyloric stenosis (a). There was a circumferential stenosis in the pylorus, with ring-shaped mucosal folds in the proximal view and ulceration in some areas (b). The mucosa of the angle and gastric body in front of the stenosis did not show any atrophic changes, such as vascular permeability (c, d). Undifferentiated tumor cells proliferated mainly in the submucosa (e), with marked fibrosis, and invaded the serosa (f). Histopathological findings of the mucosa in the pyloric (g) and gastric fundic glands (h) showed no inflammation, no atrophy, and no intestinal metaplasia (e–h: H&E staining (e): 2 × 10, (f): 40 × 10, (g, h): 20 × 10).
Discussion
Above, we reported on the details of two cases of H. pylori-negative advanced GC arising from the gastric mucosa without inflammation, atrophy, or intestinal metaplasia. Both cases were poorly differentiated adenocarcinoma and morphologically were type 4 GC. It has been shown that the eradication of H. pylori can reduce the incidence of GC [4] and may also inhibit its growth by improving gastritis [5]. However, it is also true that GC does occur even after the eradication of H. pylori, and there are still questions about how long patients should be followed up with after eradication and what kinds of patients have the highest risk of developing GC after eradication. These questions are the newest challenge in the prevention and diagnosis of GC, now that H. pylori eradication is widely available. Most post-eradication GCs are detected in an early stage [7], but advanced cancers have been found after eradication [8]. The high-risk groups for post-eradication GC are those with a high degree of atrophy of the gastric mucosa and intestinal metaplasia [10], and those with map-like redness after eradication [11].
Case 1 was first suspected to be GC in an uninfected stomach because the endoscopy did not show any H. pylori-infected gastritis, gastric mucosal atrophy, or further intestinal metaplasia. However, it was discovered that the patient had previously had her H. pylori eradicated. Such eradication has been reported to not only improve inflammation of the gastric mucosa but also atrophy and intestinal metaplasia in the long term. The complete improvement of atrophy and intestinal metaplasia may take a long time, but when the atrophy and metaplasia are mild before eradication, the stomach may later be mistaken as never having had any H. pylori infection, as it was in case 1. It is important for clinicians to learn from cases like this that after the H. pylori is gone, GC can still develop even after the inflammation and atrophy have completely disappeared [12].
Case 2 is thought to have occurred in a non-H. pylori-infected stomach. First, metastasis of the prior colorectal cancer had to be ruled out. The previous colorectal cancer surgical records showed a stage II adenocarcinoma with no lymph node metastasis and a differentiated adenocarcinoma, which was different from the histopathology seen in the stomach. Along with esophagogastric junction cancer and EV virus-infected GC, H. pylori is a leading cause of GC [1]. The occurrence of cancer in the gastric mucosa without H. pylori infection, inflammation, atrophy, or intestinal metaplasia is thought to be extremely rare [5], but reports of these exact conditions have been increasing [13]. Currently, most GCs are diagnosed in their early stages, and the frequency of advanced GC is thought to be very low [14, 15].
Among the few reports, Okano et al. [14] reported a case of H. pylori-uninfected advanced GC with no inflammation, atrophy, or intestinal metaplasia in the pyloric and fundic gland regions. The lesion was located in the pylorus and was an undifferentiated adenocarcinoma. Although Takagi et al. [15] reported on a seemingly H. pylori-uninfected undifferentiated advanced GC with massive eosinophilia, past H. pylori infection could not be excluded because the background mucosal inflammation, atrophy, and intestinal metaplasia were not examined endoscopically or histopathologically. In conclusion, we reported on two clinically relevant cases of type 4 GC from mucosa uninfected with H. pylori and without inflammation, atrophy, or intestinal metaplasia. Case 1 illustrates the need for regular endoscopic examination, even in patients whose gastric mucosal inflammation and atrophy have improved after H. pylori eradication. Case 2 suggests the need for GC screening even in those who never had any H. pylori infection.
Statement of Ethics
We reported this case in compliance with the principles of the Declaration of Helsinki. The cases were reviewed and approved by the Institutional Ethics Committee of Kawasaki Medical School. The approval number was 5537-00. Written informed consents were obtained from the patients for publication of this case report and any accompanying images.
Conflict of Interest Statement
The authors declare that Hirofimi Kawamoto received advisory fees from Gadelius Medical. Other authors have no competing interests.
Funding Sources
None of the authors have any funding to disclose.
Author Contributions
Jiro Hayashi contributed to manuscript writing, methodology, and literature review. Tomoki Yamatsuji contributed to supervision and visualization. Mitsuhiko Suehiro contributed to data analysis and literature review. Ken Haruma, corresponding author, contributed to conceptualization, supervision, and manuscript review. Ryo Katsumata contributed to manuscript writing, visualization, and project administration. Hirofumi Kawamoto contributed to data collecting and supervision. Tomoari Kamada contributed to data collecting and literature review. Kazuhiro Yoshida contributed to manuscript review and editing. Masamichi Yoneda contributed to data collecting and analysis. Yasumasa Monobe contributed to data collecting and literature review. Yoshio Naomoto contributed to conceptualization and supervision. Jiro Hayashi, Tomoki Yamatsuji, Mitsuhiko Suehiro, Ken Haruma, Ryo Katsumata, Hirofumi Kawamoto, Tomoari Kamada, Kazuhiro Yoshida, Masamichi Yoneda, Yasumasa Monobe, and Yoshio Naomoto read and approved the final manuscript.
Data Availability Statement
All data generated or analyzed during this study are included in this article. Further inquiries can be directed to the corresponding author.
References
- 1.Komoto K, Haruma K, Kamada T, Tanaka S, Yoshihara M, Sumii K, et al. Helicobacter pylori infection and gastric neoplasia: correlations with histological gastritis and tumor histology. Am J Gastroenterol. 1998 Aug;93((8)):1271–6. doi: 10.1111/j.1572-0241.1998.00408.x. [DOI] [PubMed] [Google Scholar]
- 2.Shin HR, Shin A, Woo H, Fox K, Walsh N, Lo YR, et al. Prevention of infection-related cancers in the WHO Western Pacific Region. Jpn J Clin Oncol. 2016 Jan;46((1)):13–22. doi: 10.1093/jjco/hyv092. [DOI] [PubMed] [Google Scholar]
- 3.Kodama M, Okimoto T, Ogawa R, Mizukami K, Murakami K. Endoscopic atrophic classification before and after H. pylori eradication is closely associated with histological atrophy and intestinal metaplasia. Endosc Int Open. 2015 Aug;3((4)):E311–7. doi: 10.1055/s-0034-1392090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, et al. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001 Sep;345((11)):784–9. doi: 10.1056/NEJMoa001999. [DOI] [PubMed] [Google Scholar]
- 5.Sasaki A, Kitadai Y, Ito M, Sumii M, Tanaka S, Yoshihara M, et al. Helicobacter pylori infection influences tumor growth of human gastric carcinomas. Scand J Gastroenterol. 2003 Feb;38((2)):153–8. doi: 10.1080/00365520310000636. [DOI] [PubMed] [Google Scholar]
- 6.Matsuo T, Ito M, Takata S, Tanaka S, Yoshihara M, Chayama K. Low prevalence of Helicobacter pylori-negative gastric cancer among Japanese. Helicobacter. 2011 Dec;16((6)):415–9. doi: 10.1111/j.1523-5378.2011.00889.x. [DOI] [PubMed] [Google Scholar]
- 7.Mori G, Nakajima T, Asada K, Shimazu T, Yamamichi N, Maekita T, et al. Incidence of and risk factors for metachronous gastric cancer after endoscopic resection and successful Helicobacter pylori eradication: results of a large-scale, multicenter cohort study in Japan. Gastric Cancer. 2016 Jul;19((3)):911–8. doi: 10.1007/s10120-015-0544-6. [DOI] [PubMed] [Google Scholar]
- 8.Kamada T, Hata J, Sugiu K, Kusunoki H, Ito M, Tanaka S, et al. Clinical features of gastric cancer discovered after successful eradication of Helicobacter pylori: results from a 9-year prospective follow-up study in Japan. Aliment Pharmacol Ther. 2005 May;21((9)):1121–6. doi: 10.1111/j.1365-2036.2005.02459.x. [DOI] [PubMed] [Google Scholar]
- 9.Inui M, Ohwada S, Inui Y, Kondo Y, Moro A, Sasaki K. Evaluating the accuracy of the endoscopic ABC classification system in diagnosing Helicobacter pylori-infected gastritis. Digestion. 2020 May;101((3)):298–307. doi: 10.1159/000498966. [DOI] [PubMed] [Google Scholar]
- 10.Obayashi Y, Kawano S, Sakae H, Abe M, Kono Y, Kanzaki H, et al. Risk factors for gastric cancer after the eradication of Helicobacter pylori evaluated based on the background gastric mucosa: a propensity score-matched case-control study. Intern Med. 2021 Apr;60((7)):969–76. doi: 10.2169/internalmedicine.5486-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ohno A, Miyoshi J, Kato A, Miyamoto N, Yatagai T, Hada Y, et al. Endoscopic severe mucosal atrophy indicates the presence of gastric cancer after Helicobacter pylori eradication -analysis based on the Kyoto classification. BMC Gastroenterol. 2020 Jul;20((1)):232. doi: 10.1186/s12876-020-01375-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Choi IJ, Kook MC, Kim YI, Cho SJ, Lee JY, Kim CG, et al. Helicobacter pylori therapy for the prevention of metachronous gastric cancer. N Engl J Med. 2018 Mar;378((12)):1085–95. doi: 10.1056/NEJMoa1708423. [DOI] [PubMed] [Google Scholar]
- 13.Yamamoto Y, Fujisaki J, Omae M, Hirasawa T, Igarashi M. Helicobacter pylori-negative gastric cancer: characteristics and endoscopic findings. Dig Endosc. 2015 Jul;27((5)):551–61. doi: 10.1111/den.12471. [DOI] [PubMed] [Google Scholar]
- 14.Okano A, Kato S, Ohana M. Helicobacter pylori-negative gastric cancer: advanced-stage undifferentiated adenocarcinoma located in the pyloric gland area. Clin J Gastroenterol. 2017 Feb;10((1)):13–7. doi: 10.1007/s12328-016-0696-x. [DOI] [PubMed] [Google Scholar]
- 15.Takagi A, Ozawa H, Oki M, Yanagi H, Nabeshima K, Nakamura N. Helicobacter pylori-negative advanced gastric cancer with massive eosinophilia. Intern Med. 2018 Jun;57((12)):1715–8. doi: 10.2169/internalmedicine.0013-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this article. Further inquiries can be directed to the corresponding author.