Abstract
Background
Medical plants confer various benefits to human health and their bioconversion through microbial fermentation can increase efficacy, reduce toxicity, conserve resources and produce new chemical components. In this study, the cholesterol-lowering monacolin K genes and content produced by Monascus species were identified. The high-yield monacolin K strain further fermented with various medicinal plants. The antioxidant and anti-inflammatory activities, red pigment and monacolin K content, total phenolic content, and metabolites in the fermented products were analyzed.
Results
Monacolin K was detected in Monascus pilosus (BCRC 38072), and Monascus ruber (BCRC 31533, 31523, 31534, 31535, and 33323). It responded to the highly homologous mokA and mokE genes encoding polyketide synthase and dehydrogenase. The high-yield monacolin K strain, M. ruber BCRC 31535, was used for fermentation with various medicinal plants. A positive relationship between the antioxidant capacity and total phenol content of the fermented products was observed after 60 days of fermentation, and both declined after 120 days of fermentation. By contrast, red pigment and monacolin K accumulated over time during fermentation, and the highest monacolin K content was observed in the fermentation of Glycyrrhiza uralensis, as confirmed by RT-qPCR. Moreover, Monascus-fermented medicinal plants including Paeonia lactiflora, Alpinia oxyphylla, G. uralensis, and rice were not cytotoxic. Only the product of Monascus-fermented G. uralensis significantly exhibited the anti-inflammatory capacity in a dose-dependent manner in lipopolysaccharide-induced Raw264.7 cells. The metabolites of G. uralensis with and without fermentation (60 days) were compared by LC/MS. 2,3-Dihydroxybenzoic acid, 3,4-dihydroxyphenylglycol, and 3-amino-4-hydroxybenzoate were considered to enhance the antioxidant and anti-inflammatory ability.
Conclusions
Given that highly homologous monacolin K and citrinin genes can be observed in Monascus spp., monacolin K produced by Monascus species without citrinin genes can be detected through the complementary methods of PCR and HPLC. In addition, the optimal fermentation time was important to the acquisition of antioxidants, red pigment and monacolin K. These bioactive substances were significantly affected by medicinal plants over fermentation time. Consequently, Monascus-fermented G. uralensis had a broad spectrum of biological activities.
Supplementary Information
The online version contains supplementary material available at 10.1186/s40529-022-00351-y.
Keywords: Monascus, Monacolin K, Antioxidant capacity, Anti-inflammatory capacity, Glycyrrhiza uralensis
Background
Monascus species are traditionally used in fermenting red yeast rice or red koji and widely used in the food industry in Asian countries (Farkouh and Baumgartel 2019). According to the National Center for Biotechnology Information, 19 typical Monascus species have been identified. Monascus purpureus, Monasucs pilosus, and Monascus ruber are frequently used in research. Many pigments, such as rubropunctatin, monascorubrin, rubropunctamine, monascorubramine, monascin, and ankaflavin, are detected in Monascus species (Liu et al. 2018). They are important food colorant additives. Moreover, the pharmacological efficacy of Monascus species has attracted considerable interest for the improvement of health in lipid metabolism. Monacolin K, also known as lovastatin, has a cholesterol-lowering effect and can be found in Monascus species (Yanli and Xiang 2020). In addition to lowering lipid level, monacolin K prevents many diseases, such as colon, gastric, breast, lung, and thyroid cancer; acute myeloid leukemia; Parkinson’s disease; schizophrenia; depression; and type I neurofibromatosis (Chen et al. 2015; Hong et al. 2008; Lin et al. 2015; Xiong et al. 2019; Zhang et al. 2019b). Therefore, monacolin K production through the addition of linoleic acid, non-ionic surfactant, and glutamic acid has been extensively explored (Huang et al. 2018; Yang et al. 2021; Zhang et al. 2019a). A monacolin K biosynthetic gene cluster containing nine genes (mokA–mokI) is homologous to the lovastatin gene cluster from Aspergillus terreus (Chen et al. 2008b). Monacolin K production by Monascus pilosus can be enhanced by mokH overexpression and up-regulation of monacolin K biosynthetic genes (Chen et al. 2010b).
Probiotic-fermented food offers health benefits through the bioconversion of dairy products (Lee et al. 2021). Rice or different grains are generally used as substrates for Monascus fermentation to improve secondary metabolites. Extracts from Monascus-fermented soybean have antioxidant capacities and inhibitory activities against enzymes related to skin aging (Jin and Pyo 2017). The antioxidant activities of scavenging free radicals have been observed in Monascus-fermented coix seed (Zeng et al. 2021). Fish bone as an antioxidant-active peptide source can be fermented by M. purpureus, which increases 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2, 2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid)(ABTS) radical scavenging ability (Chen et al. 2021). Additionally, Monascus-submerged fermentation using agro-industrial residues, such as rice flour and molasses, is a promising method for red pigment production (Da Silva et al. 2021). Different cereal substrates through the solid-state fermentation of M. purpureus have been developed for pigment production (Srianta et al. 2016). Besides, millet is a good candidate for monacolin K production using Monascus species (Maric et al. 2019; Zhang et al. 2018a).
Medicinal plants offer various benefits to human health. Medicinal plant bioconversion through fermentation can enhance medicinal effects by increasing efficacy, reducing toxicity, conserving resources, and producing new chemical components (Li et al. 2020a). Microbes used in the fermentation of medicinal plants include Bacillus, lactic acid bacteria, yeast, and filamentous fungi. By contrast, natural fermentation without additional microbes is a common method but less effective and specific than other known methods. In this study, a high-yield monacolin K strain from 18 Monascus spp. was screened and identified. Subsequently, medicinal plants for treating diarrhea, dementia, pain, cold, inflammation, and immune disorders were used as substrates for Monascus fermentation. The fermentation products were used in analyzing antioxidant and anti-inflammatory activities, red pigment, monacolin K, and total phenolic content. Metabolites from the fermentation product with the best efficacy were further analyzed through LC/MS.
Materials and methods
Monascus spp., medicinal plants, and growth conditions
Monascus spp. listed in Table 1 were used in this study. To detect a high-yield monacolin K strain, the medium with 7% glycerol, 3% glucose, 3% monosodium glutamate, 1.2% polypetone, 0.2% NaNO3, and 0.1% MgSO4 ·7H2O was performed for Monascus spp. incubation. Mycelia were harvested after 14 days of cultivation at 25 ℃ for DNA manipulation. Medicinal plants including Angelica pubescens, Pogostemon cablin, Paeonia lactiflora, Alpinia oxyphylla, Melaleuca leucadendron, Lavandula angustifolia, Osmanthus fragrans, Glycyrrhiza uralensis, Phellodendron chinense, and rice as the control were utilized for the fermentation materials (Beijing Tongrentang Traditional Chinese Medicine Co., Ltd, Chengdu, China). Two grams of medicinal plants as the sole substrate ground into a powder with 50 mL reverse osmosis water were sterilized at 121 ℃ for 30 min. Spores of Monascus spp. harvested from potato dextrose agar plate were added into the medicinal plant liquids for submerged batch-fermentation. Monascus spp. with medicinal plants were stood for 60 and 120 days at 25 ℃. After 60 and 120 days of submerged fermentation, the culture was centrifugated and filtered, and the suspension was freeze-dried. The freeze-dried powders were carried out for the bioactive assays.
Table 1.
Strains used in this study and their monacolin K characteristic
| Strain | Speciesa | Polyketide synthase (mokA)b |
Dehydrogenase (mokE) |
Monacolin K production (mg/g mycelia) |
|---|---|---|---|---|
| 1. BCRC 31,502 (ATCC 16363) | Monascus pilosus, Type | + | + | — |
| 2. BCRC 38072 | Monascus pilosus | + | + | 0.151 ± 0.111 |
| 3. BCRC 31533 (ATCC 16246) | Monascus ruber, Type | + | + | 0.156 ± 0.031 |
| 4. BCRC 31523 (ATCC 16378) | Monascus ruber | + | + | 0.019 ± 0.032 |
| 5. BCRC 31534 (ATCC 16366) | Monascus ruber | + | + | 0.180 ± 0.137 |
| 6. BCRC 31535 (ATCC 18199) | Monascus ruber | + | + | 0.924 ± 0.262 |
| 7. BCRC 33314 (ATCC 16371) | Monascus ruber | + | + | — |
| 8. BCRC 33323 (ATCC 18199) | Monascus ruber | + | + | 0.282 ± 0.068 |
| 9. BCRC 31542 (ATCC 16365) | Monascus purpureus, Type | — | — | — |
| 10. BCRC 31541 (ATCC 16379) | Monascus purpureus | — | — | — |
| 11. BCRC 33325 (IFO 30873) | Monascus purpureus | — | — | — |
| 12. BCRC 31615 (DSM 1379) | Monascus purpureus | — | — | — |
| 13. BCRC 31506 (CBS 302.78) | Monascus kaoliang, Type | — | — | — |
| 14. BCRC 33446 (ATCC 200613) | Monascus sanguineus, Type | — | — | — |
| 15. BCRC 33309 (ATCC 16966) | Monascus barkeri | + | + | — |
| 16. BCRC 33310 (IMI 282587) | Monascus floridanus, Type | — | — | — |
| 17. BCRC 33640 (ATCC 204397) | Monascus lunisporas, Type | — | — | — |
| 18. BCRC 33641 (ATCC 200612) | Monascus pallens, Type | — | — | — |
| 19. BCRC 32670 (ATCC 20542) | Aspergillus terreus | + | + | 0.228 ± 0.199 |
a“Type” indicates type strain
b+, positive; −, negative
DNA manipulation
Approximately 0.5 g Monascus mycelia were ground using a mortar and pestle by liquid nitrogen. DNA was extracted by phenol and chloroform, and precipitated by isopropanol. DNA was finally dissolved in TE buffer. PCR was implemented according to the condition by Chen et al. (2008a). The primer sets of mokA and mokE genes involved in the monacolin K biosynthesis were mokA-F, ATCATTCTTTCCNCGCTCCA, mokA-R, CGGGCTATTGTCGGCCATAG; mokE-F, GTGGTGGACTCGACGTTGGT, and mokE-R, TTCTCGCAGTACACGGTCAC. PCR was performed by an ABI 2700 PCR (Applied Biosystems, Thermo Fisher Scientific, Waltham, MA) and the reaction condition was as follows, 96 ℃ for 5 min by 1 cycle, 96 ℃ for 1 min, 50 ℃ for 1 min, and 72 ℃ for 1 or 2 min by 30 cycles, and 72 ℃ for 10 min with a final extension. The PCR products were recovered from agarose for DNA sequencing.
Monacolin K detection by HPLC
The culture of Monascus spp. fermentation was filtered by a 0.2 mm filter. Monacolin K was determined by a high-performance liquid chromatography (HPLC) (Shimadzu, Kyoto, Japan) fitted with a reverse-phase C18 column (InsertSustain, 5 μm, 4.6 × 150 mm)(GL science Inc., Tokyo, Japan). The HPLC reaction was followed by 0.1% phosphorus acid in water with 35% and methanol with 65% at a flow rate of 1 mL/min. Monacolin K was scanned by a UV spectroscopy from 210 to 400 nm.
Antioxidant analysis of fermentation product
The radical scavenging assay of DPPH and ABTS was used to estimate the antioxidant capacity of the fermentation product. The fermentation product of Monascus spp., 3,4-dihydroxyphenylglycol, and 3-amino-4-hydroxybenzoate were respectively mixed with 0.2 mM DPPH solution for 30 min and the mixture was measured by an enzyme-linked immunosorbent assay (ELISA) reader (Molecular Devices, Sunnyvale, CA) at OD517. The ABTS radical scavenging capacity was evaluated by the T-AOC Assay Kit (Beyotime Biotechnology, Shanghai, China). The reaction was detected by the absorbance of OD405. The radical scavenging activity of DPPH and ABTS (%) was calculated as follows, the radical scavenging activity (%) = ([OD517 or 405 of control-OD517 or 405 of fermentation product]/OD517 or 405 of control ) × 100. The sterile H2O was utilized as the control.
Cell viability of fermentation product
The Raw264.7 macrophages were carried out to implement the 3-(4,5-cimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay. The cell in a 96-well plate was incubated at 37 °C for 24 h with 5% CO2 incubation. The fermentation products, 3,4-dihydroxyphenylglycol, and 3-amino-4-hydroxybenzoate were respectively added into the cells while the sterile H2O was utilized as the control. After 24 h incubation, the supernatant medium was removed and the fresh medium containing 10 µL MTT (5 mg/mL) was added into the 96-well plate of the cells for 4 h incubation. Then, the supernatant medium was discarded, and dimethyl sulfoxide was added to the 96-well plate. The mixture was detected by the absorbance of OD570.
Anti-inflammatory analysis of fermentation product
The Raw264.7 macrophages were incubated at 37 °C for 24 h and 5% CO2 incubation in a 96-well plate. The fermentation products, 3,4-dihydroxyphenylglycol, and 3-amino-4-hydroxybenzoate were respectively added into the 96-well plate of the cells for 1 h. To stimulate the inflammation response, 1 µg/mL lipopolysaccharide was added into the cells. After 24 h incubation, the nitrite of the medium was determined by the nitrite detection kit (Beyotime Biotechnology) using the Griess reagent. The mixture was detected by the absorbance of OD540. The NO decreasing rate was evaluated as follows, NO decreasing rate (%) = ([OD540 of control-OD540 of fermentation products, 3,4-dihydroxyphenylglycol, and 3-amino-4-hydroxybenzoate]/OD540 absorbance of control) × 100. The sterile H2O was utilized as the control.
Red pigment analysis of fermentation product
The freeze-dried products of Monascus spp. fermentation was dissolved by 1 mL 70% ethanol and centrifugated at 15,000×g (Hettich, Mikro 220R, Germany) for 10 min. The supernatant was obtained and detected by the absorbance of OD505. The red pigment was calculated as follows, red pigment (U/g) = OD505 of fermentation product × (1/gram) × (1/volume) × dilution factor.
Monacolin K biosynthetic gene expression by RT-qPCR
Approximately 0.5 g Monascus mycelia were ground and performed by TRIzol reagent (Thermo Fisher Scientific). Total RNA was extracted by chloroform and treated with RNase-free DNase. Finally, total RNA was preserved in DEPC H2O. HiFi-MMLV cDNA kit (Beijing ComWin Biotech Co., Ltd., Beijing, China) with oligo (dT) and random hexamers was used to carry out the first-strand cDNA. The qPCR in a final volume of 25 µL was implemented using UltraSYBR Mixture kit (Beijing ComWin Biotech) by the Roche LightCycler® 480 System (Roche Group, Switzerland). The primer sets of monacolin K biosynthetic genes were utilized as follows, qmokA-F, CTGGTGCAGACACAGTACGACAT, qmokA-R, GGAACCATCGCCGACAAAT; qmokB-F, GGGACCCTGAGTTTCGAACA, qmokB-R, GCACTTTTTCACCCCGTTGA; qmokC-F, GAGGCCAGCGCGACAAT, qmokC-R, GTGACAGTGCGTGTCACCAAA; qmokD-F, CCGCTTTACGGGAAGACTTT, qmokD-R, GAACCCTCGAACCAGGTGTA; qmokE-F, GCGACGATTGTGATGCAGAT, qmokE-R, ATCTTCTGCGCCGTGCTTT; qmokF-F, AACGGAGAAGCAGATGAACCA. qmokF-R, TCCCACCAAGCCCAAAACT; qmokG-F, CGTCCGGAAGGTCCTGAAG, qmokG-R, TGAACCCCCCCATACTACCA; qmokH-F, GGAGTGGCCAAAACAGGAAA, qmokH-R, TGCGGGTGTTGGATTGTTG; qmokI-F, TGCTGGGAGGTGCTTTTACC, and qmokI-R, AATGTGGATGGCGAGAAGGA. 18 S rRNA was used as the internal control and the primer set was mpF18S rRNA, TCTCGTAATCGGAATGAGAACGA, and mpR18S rRNA, TACGCTATTGGAGCTGGAATTACC. The three replicates of gene expression were done and calculated by the 2−ΔΔCT method.
Analysis of total phenol and metabolites from the fermentation product
Total phenol of fermentation product was measured by the Folin-Ciocalteu method. The fermentation product was added into Folin-Ciocalteu reagent for 5 min reaction, and then 10% sodium carbonate solution was used for color development in darkness. The gallic acid of different concentrations as a positive control was utilized for the calculation of the standard curve. The mixture was centrifugated at 15,000×g for 10 min and the supernatant was measured by the absorbance of OD760. Thermo Vanquish system equipped with an ACQUITY UPLC® HSS T3 (150 × 2.1 mm, 1.8 μm, Waters Corporation, Milford, MA) column was accomplished for the analysis of fermentation product. The UPLC reaction was followed by 0.1% formic acid in acetonitrile (A) and 5 mM ammonium formate in acetonitrile (B) at a flow rate of 0.25 mL/min. The gradient of solvent A/B (v/v) was set as follows, 2% A/B from 0 to 1 min; 2-50% A/B from 1 to 9 min; 50–98% A/B from 9 to 12 min; 98% A/B from 12 to 13.5 min; 98–2% A/B from 13.5 to 14 min; 2% A from 14 to 20 min for positive model (2% B from 14 to 17 min for negative model). The ESI-MS was implemented by the Q Exactive Plus mass spectrometer (Thermo Fisher Scientific) with the spray voltage of 3.5 kV and − 2.5 kV in positive and negative modes, respectively. Data-dependent acquisition MS/MS was carried out with the HCD scan and the normalized collision energy was 30 eV. Unnecessary information in MS/MS spectra was removed by dynamic exclusion. The metabolites were screened by the accurate molecular weight (molecular weight error < 30 ppm) and identified by Metlin (http://metlin.scripps.edu), Xue et al. (2020) MoNA (https://mona.fiehnlab.ucdavis.edu//) and the database (built by Azenta Life Sciences, Suzhou, China) according to MS/MS fragment mode.
Statistical analysis
Duncan’s multiple range test, Pearson correlation, and repeated-measures analysis of variances (ANOVAs) were performed by IBM SPSS Statistics v20 software package (SPSS Inc. Chicago, USA) at a confidence level of 95%. The mean and standard deviation of three replicates were shown.
Results
Identification of monacolin K biosynthetic genes and content from Monascus spp.
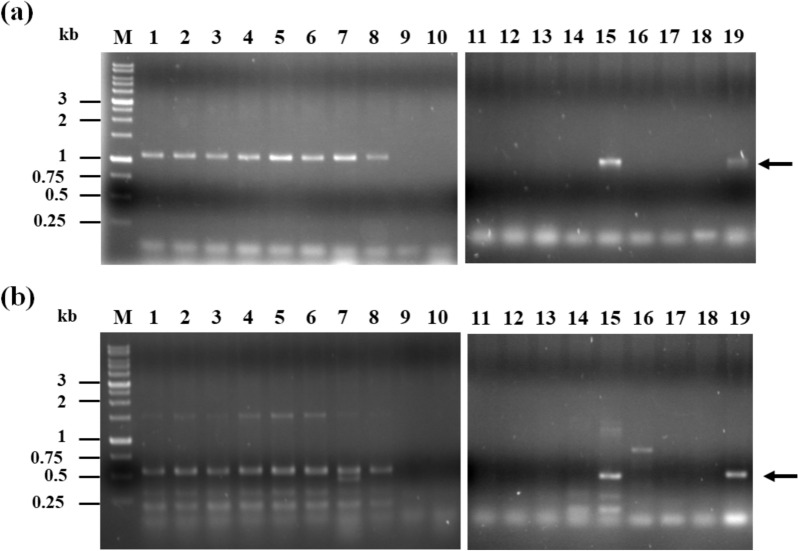
mokA and mokE genes encoding polyketide synthase and dehydrogenase involved in monacolin K biosynthesis from 18 Monascus spp. and one Aspergillus terreus were amplified through PCR. The expected bands of approximately 1 and 0.5 kb were detected (Fig. 1). The partial mokA and mokE genes can be obtained from M. pilosus (BCRC 31502 and 38072), M. ruber (BCRC 31533, 31523, 31534, 31535, 33314, and 33323), and Monascus barkeri (BCRC 33309). A similar result was obtained in A. terreus BCRC 32670, which can produce lovastatin and has lov genes. The amplified PCR products were further sequenced. Highly homologous DNA sequences were obtained in the partial mokA and mokE genes (Additional file 1: Fig. S1). However, lovB and lovC genes from A. terreus showed some differences from those of Monascus spp. mlcA and mlcG derived from the compactin biosynthetic gene cluster of Penicillium citrinum, which had a gene cluster and cholesterol-inhibiting efficacy similar to monacolin K, were different from mokA and mokE (Abe et al. 2002).
Fig. 1.
PCR analyses of monacolin K-related genes. The partial mokA (a) and mokE (b) genes were detected. Lanes 1–19 revealed M. pilosus BCRC 31502, and 38072; M. ruber BCRC 31533, 31523, 31534, 31535, 33314, and 33323; M. purpureus BCRC 31542, 31541, 33325, and 31615; M. kaoliang BCRC 31506; M. sanguineus BCRC 33446; M. barkeri BCRC 33309; M. floridanus BCRC 33310; M. lunisporas BCRC 33640; M. pallens BCRC 33641; A. terreus BCRC 32670. The arrows indicated the expected DNA bands by PCR
Monascus spp. and A. terreus BCRC 32670 were incubated for 14 days, and monacolin K content was determined (Table 1). Monacolin K was detected in M. pilosus (BCRC 38072), M. ruber (BCRC 31533, 31523, 31534, 31535, and 33323), and A. terreus BCRC 32670 in response to the PCR results. M. ruber BCRC 31535 had the highest monacolin K content. Hence, it was used in submerged fermentation with different medicinal plants.
Antioxidant analysis of M. ruber BCRC 31535-fermented product
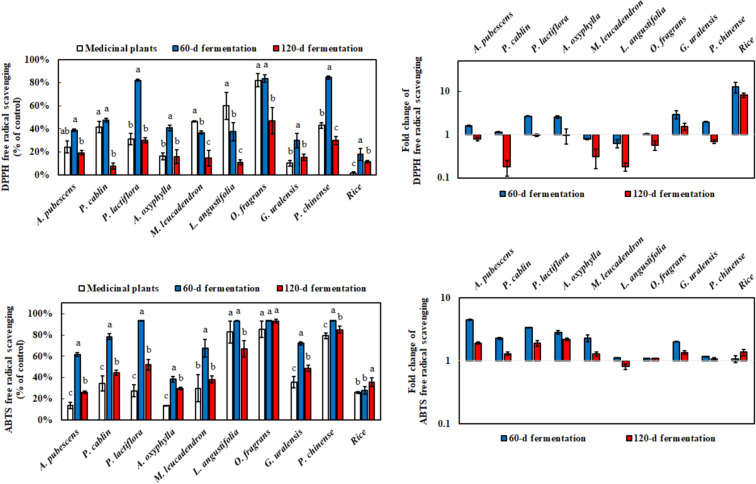
Nine medicinal plants and one rice were used in the submerged fermentation of M. ruber BCRC 31535 for antioxidation, anti-inflammation, and metabolite analysis. The extracts of nine medicinal plants without M. ruber BCRC 31535 fermentation exhibited antioxidant capacity, especially in L. angustifolia and O. fragrans, which scavenged over 50% of DPPH free radicals at 0.5 mg/mL (Fig. 2). However, rare antioxidation was detected in rice. On the other hand, the effect of these products at 0.5 mg/mL on the antioxidant activity of ABTS free radical were not compared because all of them had ABTS scavenging rates of over 90%. Nevertheless, the ABTS scavenging capacity of the medicinal plants had a similar antioxidant profile to DPPH result at 0.0625 mg/mL. After the 60-day fermentation of M. ruber BCRC 31535, the DPPH scavenging ability of the fermentation product was elevated, except in M. leucadendron, and L. angustifolia. In addition, the order of the DPPH scavenging capacity by fold change was rice > G. uralensis > P. lactiflora, whereas that of ABTS was A. pubescens > P. lactiflora > A. oxyphylla in the 60-day fermentation. However, the ABTS and DPPH scavenging capacities significantly declined after 120 days submerged fermentation.
Fig. 2.
Antioxidant capacity determined using DPPH and ABTS scavenging methods with or without the fermentation of medicinal plants at 0.5 and 0.0625 mg/mL by M. ruber BCRC 31,535. The ratio of 60 and 120 days of fermentation to medical plants was depicted on the right side of the figure. Results are mean ± S.D. (n = 3)
Anti-inflammatory analysis of M. ruber BCRC 31535-fermented product
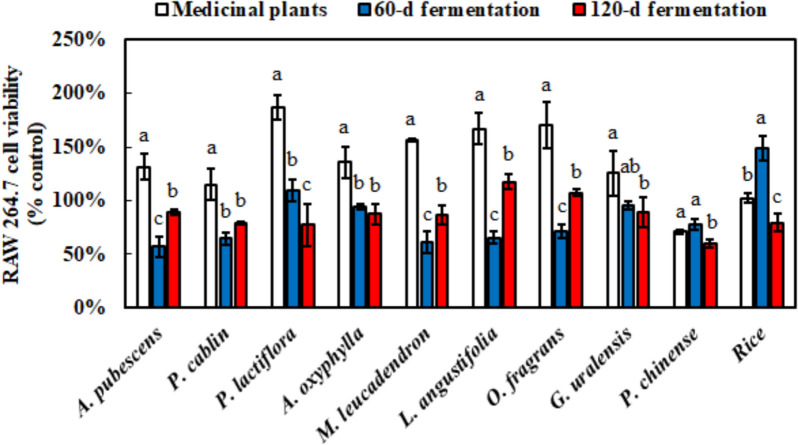
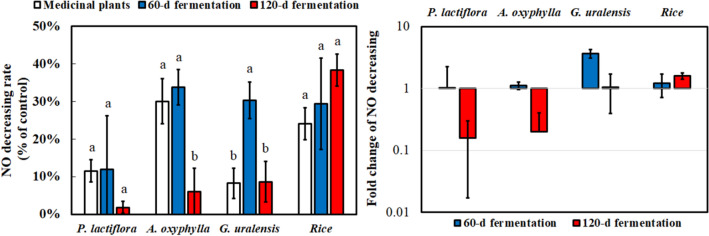
To analyze the anti-inflammatory capacity of M. ruber BCRC 31535-fermented product, the cell viability of the fermentation product was explored (Fig. 3). Most medicinal plants without fermentation were harmless to Raw264.7 cells, and cell viability below 80% was observed only in P. chinense. However, the cell viability of the fermentation products significantly decreased, and M. ruber BCRC 31535-fermented P. lactiflora, A. oxyphylla, G. uralensis, and rice had survival rates of over 90% after 60 days of fermentation. Thus, their anti-inflammatory effects were further investigated using their fermentation products (Fig. 4). A. oxyphylla without fermentation showed the best anti-inflammatory effect. After the fermentation of M. ruber BCRC 31535, the fermented G. uralensis significantly improved anti-inflammatory capacity. The trend of anti-inflammatory effect of the fermentation products was similar to that of their antioxidant ability. After 120 days of submerged fermentation, anti-inflammation obviously decreased.
Fig. 3.
Raw264.7 cell viability using MTT method with or without fermentation of medicinal plants at 0.5 mg/mL by M. ruber BCRC 31535. Results are mean ± S.D. (n = 3)
Fig. 4.
Anti-inflammatory capacity using LPS-stimulated Raw264.7 cell with or without fermentation of medicinal plants at 0.5 mg/mL by M. ruber BCRC 31535. The ratio of 60 and 120 days of fermentation to medical plants was depicted on the right side of the figure. Results are mean ± S.D. (n = 3)
Red pigment and monacolin K analysis of M. ruber BCRC 31535-fermented product
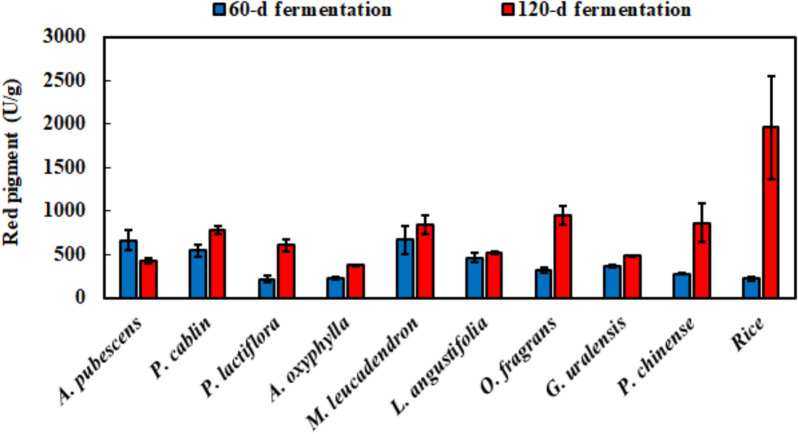
Red pigment and monacolin K derived through polyketide synthesis can be produced by M. ruber BCRC 31,535 and these of fermentation product were further determined. The red pigment of the fermentation product increased with fermentation time, except in A. pubescens (Fig. 5). The red pigment of the 120-day fermentation product from rice was 8.9-fold that of the 60-day fermentation product.
Fig. 5.
Red pigment with the fermentation of medicinal plants by M. ruber BCRC 31,535 and detected by the absorbance of OD505. Results are mean ± S.D. (n = 3)
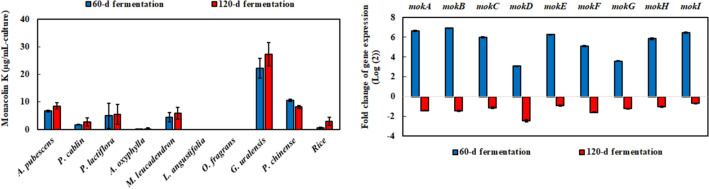
Monacolin K was obviously detected in the fermentation of A. pubescens, P. cablin, P. lactiflora, M. leucadendron, G. uralensis, P. chinense, and rice (Fig. 6). It was not detected during the fermentation of L. angustifolia and O. fragrans. Monacolin K content in these fermentation products increased, similar to that of the red pigment during fermentation, except in P. chinense. The content of monacolin K derived through the 60 and 120 days fermentation of G. uralensis was the highest, which was 41.9-fold and 9.1-fold that in rice, respectively. Thus, the gene expression of monacolin K containing mokA-mokI genes was further analyzed by comparing M. ruber BCRC 31535-fermented G. uralensis and rice. The results indicated that the gene expression levels of monacolin K obtained through the 60-day fermentation of G. uralensis were higher than those in rice. However, monacolin K gene expression level after 120 days G. uralensis fermentation was obviously lower than that in rice. This result was consistent with monacolin K content. The content after 120 days G. uralensis fermentation was only 1.2 times higher than that after 60 days of fermentation. Monacolin K content in rice after 120 days of fermentation was 5.7-fold that after 60 days of fermentation.
Fig. 6.
Monacolin K with the fermentation of medicinal plants by M. ruber BCRC 31535 and its gene expression of M. ruber BCRC 31535-fermented G. uralensis and rice by RT-qPCR. The log(2) fold change of M. ruber BCRC 31,535-fermented G. uralensis to M. ruber BCRC 31,535-fermented rice by monacolin K gene expression was depicted on the right side of the figure. The 18 S rRNA was used as the positive control to normalize the data. Results are mean ± S.D. (n = 3)
Total phenolic content and metabolites analysis of M. ruber BCRC 31535-fermented product
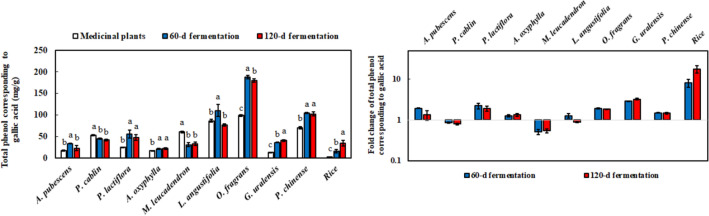
The antioxidant effect of the 60-day fermentation product significantly increased relative to the antioxidant effects of medicinal plants without fermentation. Accordingly, the contribution of total phenolic content to antioxidant capacity was evaluated. The result showed that the total phenolic content was elevated after M. ruber BCRC 31535 fermentation, except in P. cablin, and M. leucadendron (Fig. 7). The highest increase in total phenolic content was found in rice after fermentation, followed by that in G. uralensis. This result was consistent with the DPPH scavenging capacity.
Fig. 7.
Total phenols using Folin-Ciocalteu method with or without fermentation of medicinal plants by M. ruber BCRC 31535. The ratio of 60 and 120 days of fermentation to medical plants was depicted on the right side of the figure. Results are mean ± S.D. (n = 3)
The 60-day fermentation of G. uralensis enhanced DPPH scavenging, anti-inflammatory ability, monacolin K content, and total phenolic content. The metabolites of G. uralensis with or without fermentation were determined through LC/MS (Additional file 2: Fig. S2). The result indicated that the five compounds with the highest increases after 60 days of fermentation were 11beta-hydroxyandrost-4-ene-3,17-dione (steroid), 3,4-dihydroxyphenylglycol (phenol), leucine (amino acid), 2,3-dihydroxybenzoic acid (iron chelator), and 3-amino-4-hydroxybenzoate (monohydroxybenzoic acid), as shown in Table 2 and Additional file 3: Fig. S3. The order of relative intensity was 2,3-dihydroxybenzoic acid > leucine > 3-amino-4-hydroxybenzoate > 3,4-dihydroxyphenylglycol. The antioxidant and anti-inflammatory capacities of 2,3-dihydroxybenzoic acid against DPPH free radicals and lipopolysaccharide-induced inflammation have been demonstrated (Adjimani and Asare 2015; Alvarez Cilleros et al. 2020). Thus, 3-amino-4-hydroxybenzoate and 3,4-dihydroxyphenylglycol were further used to verify their antioxidant and anti-inflammatory abilities.
Table 2.
The metabolites using LC/MS analysis with most increase after the 60-day G. uralensis fermentation by M. ruber BCRC 31,535
| Compounds | 60-day fermentation (Relative intensity) |
G. uralensis
(Relative intensity) |
Fold change | Log2(fold change) |
|---|---|---|---|---|
| 11beta-Hydroxyandrost-4-ene-3,17-dione | 1.7995 ± 0.1111 | 0.0023 ± 0.0006 | 771.82 | 9.5921 |
| 3,4-Dihydroxyphenylglycol | 13.5409 ± 0.9084 | 0.0465 ± 0.02575 | 291.41 | 8.1869 |
| Leucine | 30.0989 ± 1.8297 | 0.1727 ± 0.0992 | 174.3 | 7.4455 |
| 2,3-Dihydroxybenzoic acid | 55.09045 ± 4.9859 | 0.382895 ± 0.2036 | 143.88 | 7.1687 |
| 3-Amino-4-hydroxybenzoate | 14.7540 ± 0.8378 | 0.019943 ± 0.0197 | 89.138 | 6.478 |
| Scopoletin | 4.3951 ± 0.2914 | 0.1874 ± 0.0148 | 23.448 | 4.554 |
| N-Butyryl-l-homoserine lactone | 5.2381 ± 1.1549 | 0.2710 ± 0.0255 | 19.326 | 4.2725 |
| Allantoic acid | 2.9601 ± 0.1018 | 0.1624 ± 0.1627 | 18.229 | 4.1882 |
| 16-Hydroxy hexadecanoic acid | 3.5948 ± 0.4486 | 0.2038 ± 0.0670 | 17.641 | 4.1408 |
| Histamine | 1.4013 ± 0.1212 | 0.0799 ± 0.0045 | 17.542 | 4.1327 |
Antioxidant and anti-inflammatory analysis of 3,4-dihydroxyphenylglycol, and 3-amino-4-hydroxybenzoate
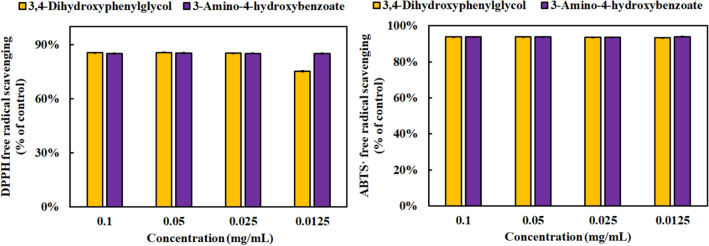
The antioxidant capacities of 3,4-dihydroxyphenylglycol and 3-amino-4-hydroxybenzoate were analyzed with DPPH and ABTS methods (Fig. 8). The result showed that over 75% DPPH scavenging activity was observed in 3,4-dihydroxyphenylglycol and 3-amino-4-hydroxybenzoate at 0.0125 mg/mL. However, 3-amino-4-hydroxybenzoate showed better antioxidation than 3,4-dihydroxyphenylglycol. The removal rate of ABTS free radical was over 90% at 0.0125 mg/mL in both compounds.
Fig. 8.
Antioxidant capacity of 3,4-dihydroxyphenylglycol and 3-amino-4-hydroxybenzoate using DPPH and ABTS scavenging methods. Results are mean ± S.D. (n = 3)
The anti-inflammatory capacities of 3,4-dihydroxyphenylglycol and 3-amino-4-hydroxybenzoate were explored on the basis of cell viability (Fig. 9). The survival rates of Raw264.7 cells treated with 3,4-dihydroxyphenylglycol and 3-amino-4-hydroxybenzoate below 0.1 mg/mL were over 90%, and no cytotoxicity was observed. Therefore, the anti-inflammatory capacities of the two compounds were further measured. The anti-inflammatory capacity of 3,4-dihydroxyphenylglycol was observed in a dose-dependent manner. Over 56% decrease in NO was detected at 0.05 mg/mL. However, the anti-inflammatory effects of 3-amino-4-hydroxybenzoate were not significant in a dose-dependent manner.
Fig. 9.
Raw264.7 cell viability and anti-inflammatory capacity of 3,4-dihydroxyphenylglycol and 3-amino-4-hydroxybenzoate using MTT method and LPS-stimulated Raw264.7 cells. Results are mean ± S.D. (n = 3)
Discussion
Monascus species offer various health benefits to human beings. However, hepato-nephrotoxic citrinin of polyketide metabolites is present in Monascus species (de Oliveira Filho et al. 2017). Hence, many studies have focused on improving the ratio of monacolin K to citrinin through optimal cultivation or fermentation. Given that citrinin and monacolin K biosynthetic gene clusters are present in Monascus species (Chen et al. 2008b; Shimizu et al. 2005), considerable effort has been devoted to identifying these genes in different Monascus strains. According to our previous study, pksCT, ctnA, and orf3 genes, which encode citrinin polyketide synthase, a major activator, and oxygenase, were distributed in M. purpureus and Monascus kaoliang, but not in M. pilousus, M. ruber, M. barkeri, Monascus floridanus, Monascus lunisporas, and Monascus pallens (Chen et al. 2008a). Interestingly, in this study, mokA and mokE genes were detected in M. pilousus, M. ruber, and M. barkeri only, and no monacolin K content was detected in M. barkeri. Further investigation into the citrinin and monacoin K genes of Monascus has shown that the distribution of citrinin and monacolin K is not restricted in the specific Monascus species. Some Monascus species simultaneously have two gene clusters, but some have only one (Chen et al. 2008a; Li et al. 2020b; Yang et al. 2015). Information about the evolutionary process of the polyketide gene cluster in fungi is currently limited, and thus gene duplication or horizontal gene transfer is worthy of further study (Wisecaver et al. 2014). On the other hand, gene sequencing similarity of monacolin K among Monascus species was highly homologous. This finding is in agreement with the study of Dai et al., who compared the monacolin K gene cluster of seven M. pilosus and M. ruber strains (Dai et al. 2021). The highly homologous citrinin genes in Monascus spp. can be also observed (Chen et al. 2008a; Li et al. 2020b). Therefore, Monascus species-produced monacolin K without citrinin biosynthetic genes can be screened with the complementary methods of PCR and HPLC. Consequently, M. pilosus BCRC 38072 and M. ruber BCRC 31533, 31523, 31534, 31535, and 33323 producing monacolin K without citrinin can be safely applied to the food industry.
Raw materials, such as rice and grains, provide carbohydrates, protein, and inorganic elements for traditional fermentation. Thus, considerable attention has been devoted to the fermentation of Monascus species with rice and grains (Jin and Pyo 2017; Maric et al. 2019; Srianta et al. 2016; Zhang et al. 2018a). Given that medicinal plants themselves have certain efficacy, fermentation through the biotransformation of microorganisms can improve the original properties of the plants and result in the production of novel chemical components (Li et al. 2020a). Scant attention has been devoted to the relationship between the fermentation of medicinal plants and Monascus species. Hence, several medicinal plants and rice were used in the fermentation of M. ruber BCRC 31535 for 60 and 120 days. Most medicinal plants and their extracts have antioxidant capacities (Chen et al. 2017; Kim et al. 2017, 2019; Soheili and Salami 2019; Surh and Yun 2012; Wu et al. 2021, 2022; Yuan et al. 2020; Zhang et al. 2018b). In this study, after the 60-day fermentation of M. ruber BCRC 31535 with medicinal plants, the DPPH scavenging rate significantly improved, except in M. leucadendron and L. angustifolia as raw materials. M. leucadendron and L. angustifolia contributed to antifungal activity and probably affected M. ruber BCRC 31535 fermentation (Abd Rashed et al. 2021; Valdes et al. 2008). Additionally, the long-term fermentation of medicinal plants (120 days) decreased antioxidant activity in the DPPH and ABTS assays. This effect may have promoted the formation of acids from antioxidant compounds, such as phenols, through long-term fermentation (Lee et al. 2019). Phenols are responsible for antioxidant activity, and the relationship between total phenolic content and antioxidant capacity was demonstrated (Additional file 4: Table S1, Additional file 5: Table S2, Additional file 6: Table S3). According to the Pearson correlation between total phenolic content and antioxidant activity, the significant effects of total phenolic content on DPPH and ABTS scavenging abilities were observed. Thus, the optimal fermentation time was important to the acquisition of antioxidants and prevention of further degradation by microbes.
Before the anti-inflammatory capacities of fermentations product generated by lipopolysaccharide-induced Raw264.7 cells were determined, cell viability was analyzed with the MTT method. Most medicinal plants were not cytotoxic. The effects of medicinal plants and fermentation products on cell viability across temporal change was evaluated by repeated-measures ANOVAs (Additional file 7: Table S4). The result indicated that cell viability was significantly affected by medicinal plants over time. This result was contrary to our expectation that toxicity would be reduced after fermentation (Li et al. 2020a). However, a survival rate of over 90% was still observed in the fermentation of P. lactiflora, A. oxyphylla, G. uralensis, and rice, which originally had potential anti-inflammatory activities (Okonogi et al. 2018; Xin et al. 2019; Yin et al. 2018; Zhang et al. 2018b). Thus, these medicinal plants and fermentation products were further used in the analysis of anti-inflammatory capacity. Only 60-day G. uralensis fermentation revealed a significant effect on NO decreasing rate. This benefit was consistent with the fermentation of ginger and tea by M. pilosus and M. purpureus, respectively (Chen et al. 2010a; Deng et al. 2021).
Red pigment and monacolin K derived from polyketides were the major characteristics in Monascus species. The amounts of red pigment and monacolin K increased with fermentation time in most fermentation products, and their maximum amounts were obtained after the fermentation of rice and G. uralensis, respectively. The repeated-measures ANOVAs demonstrated that the red pigment and monacolin K were significantly affected by medicinal plants over fermentation time (Additional file 8: Table S5). The trend was different from the trends of antioxidant and anti-inflammatory capacities. The stable production of red pigment and monacolin K was similar to the different studies using agro-industrial residues, cereal, and millet as fermented substrates (Da Silva et al. 2021; Maric et al. 2019; Srianta et al. 2016; Zhang et al. 2018a). In addition, the transcription level of monacolin K biosynthetic genes between the fermentation of G. uralensis and rice was further verified with RT-qPCR. As expected, the expression levels of monacolin K biosynthetic genes in the 60-day G. uralensis fermentation were higher than those in rice. Furthermore, no up-regulation was observed in the 120-day G. uralensis fermentation, and rate of increase in monacolin K content was lower than that in the 120-day rice fermentation. Moreover, MokG encoding HMG-CoA reductase may confer resistance on monacolin K (Abe et al. 2002; Chen et al. 2008b); therefore, its relative gene expression was lower than the expression levels of other genes.
LC/MS was used in analyzing difference between metabolites with or without fermentation of M. ruber BCRC 31533 in G. uralensis. The relative abundance of glutamic acid in G. uralensis without fermentation was 3.1-fold higher than that in 60 days fermentation. This result suggested that the consumption of glutamic acid promoted monacolin K production (Zhang et al. 2019a). In addition, G. uralensis, commonly known as licorice, is usually used in the food industry as a sweetener and contains various bioactive constituents, such as liquiritin, isoliquiritin, liquiritigenin, and isoliquiritigenin (Kao et al. 2014). Liquiritigenin and isoliquiritigenin are the aglycone forms of liquiritin and isoliquiritin, respectively. The relative abundance rates of liquiritin and liquiritigenin were high in G. uralensis and were 632.9- and 3.8-fold those in 60-day fermentation, respectively. This result suggested that the bioactive substances were metabolized and consumed by M. ruber BCRC 31,535. This result was different from that of Kim et al., who reported that liquiritigenin and isoliquiritigenin content improved after the fermentation of Monascus albidulus (Kim et al. 2020). Furthermore, 2,3-dihydroxybenzoic acid had the highest content, which increased 143.9-fold after 60-day G. uralensis fermentation. According to the previous study, 2,3-dihydroxybenzoic acid displayed good antioxidant and anti-inflammatory capacities (Adjimani and Asare 2015; Alvarez Cilleros et al. 2020). Moreover, 3,4-dihydroxyphenylglycol and 3-amino-4-hydroxybenzoate increased 291.4- and 89.1-fold through the biotransformation of M. ruber BCRC 31,535, respectively. The two compounds had high antioxidant capacities according to the DPPH and ABTS assays. The NO rate of 3,4-dihydroxyphenylglycol decreased in a dose-dependent manner. These results implied that 2,3-dihydroxybenzoic acid, 3,4-dihydroxyphenylglycol, and 3-amino-4-hydroxybenzoate improved antioxidative and anti-inflammatory effects in the 60-day fermentation of G. uralensis.
Conclusions
Traditionally, the effective substance, new compound, resource conservation, and toxic reduction of medicinal plants can be enhanced by means of microbial fermentation. In this study, the antioxidant capacity and total phenolic content of medicinal plants increased after 60 days of fermentation by M. ruber BCRC31535. However, cytotoxicity was also elevated but not influenced in P. lactiflora, A. oxyphylla, G. uralensis, and rice. The positive correlation between total phenolic content and DPPH and ABTS scavenging activities was verified by repeated-measures ANOVAs. Long-term fermentation (120 days) decreased antioxidant efficacy and cytotoxicity. Red pigment and monacolin K derived from M. ruber BCRC31535 biosynthesis accumulated over time during fermentation. Among these medicinal plants, M. ruber BCRC31535-fermented G. uralensis had a broad spectrum of efficacy with antioxidative and anti-inflammatory capacities, and monacolin K contributed by the increase in 2,3-dihydroxybenzoic acid, 3,4-dihydroxyphenylglycol and 3-amino-4-hydroxybenzoate levels and glutamic acid consumption rates. To the best of our knowledge, this study is the first to report the antioxidative capacities of 3-amino-4-hydroxybenzoate for DPPH and ATBS scavenging activities.
Supplementary Information
Additional file 1: Figure S1. Phylogeny of the partial mokA (a) and mokE (b) from Monascus species, and the related genes from A. terreus, and P. citrinum.
Additional file 2: Figure S2. LC/MS analysis with or without M. ruber BCRC 31535-fermented G. uralensis in positive and negative modes.
Additional file 3: Figure S3. The metabolites of the ten compounds with the most increase after 60-day G. uralensis fermentation in MS/MS spectrum.
Additional file 4: Table S1. Pearson correlation between total phenols and antioxidant activity by medicinal plants without fermentation.
Additional file 5: Table S3. Pearson correlation between total phenols and antioxidant activity by 120 days fermentation of M. ruber BCRC 31535.
Additional file 6: Table S2. Pearson correlation between total phenols and antioxidant activity by 60 days fermentation of M. ruber BCRC 31535.
Additional file 7: Table S4. Significance of p-value for repeated measures ANOVA on cell viability from the M. ruber BCRC 31535-fermentation of various medicinal plants.
Additional file 8 Table S5. Significance of p-value for repeated measures ANOVA on red pigment and monacolin K from the M. ruber BCRC 31535-fermentation of various medicinal plants and rice.
Author contributions
Y-PC designed the experiment, analyzed the data, and prepared the writing-original draft. All authors collected the samples and the data. Y-PC and H-TW analyzed the data and revised the manuscript. Y-PC and Y-CK designed the experiment and reviewed the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by the Ministry of Science and Technology, Taiwan (110WFA0911781), and the Fujian provincial health technology project from Fujian Provincial Health Commission (2019-ZQNB-20), Fujian, China.
Data availability
The data used and analyzed in this study can be provided from the corresponding author for scientific, non-profit purpose.
Declarations
Ethics approval and consent to participate
Not applicable, the study involves no human participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Abd Rashed A, Rathi DG, Ahmad Nasir NAH, Abd Rahman AZ. Antifungal properties of essential oils and their compounds for application in skin fungal infections: conventional and nonconventional approaches. Molecules. 2021;26:1093. doi: 10.3390/molecules26041093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abe Y, Suzuki T, Mizuno T, Ono C, Iwamoto K, Hosobuchi M, Yoshikawa H. Effect of increased dosage of the ML-236B (compactin) biosynthetic gene cluster on ML-236B production in Penicillium citrinum. Mol Genet Genomics. 2002;268:130–137. doi: 10.1007/s00438-002-0736-8. [DOI] [PubMed] [Google Scholar]
- Adjimani JP, Asare P. Antioxidant and free radical scavenging activity of iron chelators. Toxicol Rep. 2015;2:721–728. doi: 10.1016/j.toxrep.2015.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alvarez Cilleros D, Lopez-Oliva ME, Martin MA, Ramos S. (-)-Epicatechin and the colonic metabolite 2,3-dihydroxybenzoic acid protect against high glucose and lipopolysaccharide-induced inflammation in renal proximal tubular cells through NOX-4/p38 signalling. Food Funct. 2020;11:8811–8824. doi: 10.1039/D0FO01805H. [DOI] [PubMed] [Google Scholar]
- Chen YP, Tseng CP, Chien IL, Wang WY, Liaw LL, Yuan GF. Exploring the distribution of citrinin biosynthesis related genes among Monascus species. J Agric Food Chem. 2008;56:11767–11772. doi: 10.1021/jf802371b. [DOI] [PubMed] [Google Scholar]
- Chen YP, Tseng CP, Liaw LL, Wang CL, Chen IC, Wu WJ, Wu MD, Yuan GF. Cloning and characterization of monacolin K biosynthetic gene cluster from Monascus pilosus. J Agric Food Chem. 2008;56:5639–5646. doi: 10.1021/jf800595k. [DOI] [PubMed] [Google Scholar]
- Chen CC, Chyau CC, Liao CC, Hu TJ, Kuo CF. Enhanced anti-inflammatory activities of Monascus pilosus fermented products by addition of ginger to the medium. J Agric Food Chem. 2010;58:12006–12013. doi: 10.1021/jf103070m. [DOI] [PubMed] [Google Scholar]
- Chen YP, Yuan GF, Hsieh SY, Lin YS, Wang WY, Liaw LL, Tseng CP. Identification of the mokH gene encoding transcription factor for the upregulation of monacolin K biosynthesis in Monascus pilosus. J Agric Food Chem. 2010;58:287–293. doi: 10.1021/jf903139x. [DOI] [PubMed] [Google Scholar]
- Chen CC, Wu ML, Ho CT, Huang TC. Blockade of the Ras/Raf/ERK and Ras/PI3K/Akt pathways by monacolin K reduces the expression of GLO1 and induces apoptosis in U937 cells. J Agric Food Chem. 2015;63:1186–1195. doi: 10.1021/jf505275s. [DOI] [PubMed] [Google Scholar]
- Chen J, Li WC, Gu XL. Optimized extraction, preliminary characterization, and in vitro antioxidant activity of polysaccharides from Glycyrrhiza Uralensis Fisch. Med Sci Monit. 2017;23:1783–1791. doi: 10.12659/MSM.900471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen YT, Hsieh SL, Gao WS, Yin LJ, Dong CD, Chen CW, Singhania RR, Hsieh S, Chen SJ. Evaluation of chemical compositions, antioxidant capacity and intracellular antioxidant action in fish bone fermented with Monascus purpureus. Molecules. 2021;26:5288. doi: 10.3390/molecules26175288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Da Silva VL, Ienczak JL, Moritz D. Agro-industrial residues for the production of red biopigment by Monascus ruber: rice flour and sugarcane molasses. Braz J Microbiol. 2021;52:587–596. doi: 10.1007/s42770-021-00456-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai W, Shao Y, Chen F. Production of monacolin K in Monascus pilosus: Comparison between industrial strains and analysis of its gene clusters. Microorganisms. 2021;9:747. doi: 10.3390/microorganisms9040747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Oliveira Filho JWG, Islam MT, Ali ES, Uddin SJ, Santos JVO, de Alencar M, Junior ALG, Paz M, de Brito M, JMC ES, Shaw S, de Medeiros M, Dantas S, Rolim HML, Ferreira PMP, Kamal MA, Pieczynska MD, Das N, Gupta VK, Mocan A, Dos Santos Andrade TJA, Singh BN, Mishra SK, Atanasov AG, Melo-Cavalcante AAC. A comprehensive review on biological properties of citrinin. Food Chem Toxicol. 2017;110:130–141. doi: 10.1016/j.fct.2017.10.002. [DOI] [PubMed] [Google Scholar]
- Deng X, Hou Y, Zhou H, Li Y, Xue Z, Xue X, Huang G, Huang K, He X, Xu W. Hypolipidemic, anti-inflammatory, and anti-atherosclerotic effects of tea before and after microbial fermentation. Food Sci Nutr. 2021;9:1160–1170. doi: 10.1002/fsn3.2096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farkouh A, Baumgartel C. Mini-review: medication safety of red yeast rice products. Int J Gen Med. 2019;12:167–171. doi: 10.2147/IJGM.S202446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong MY, Seeram NP, Zhang Y, Heber D. Anticancer effects of Chinese red yeast rice versus monacolin K alone on colon cancer cells. J Nutr Biochem. 2008;19:448–458. doi: 10.1016/j.jnutbio.2007.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang J, Liao N, Li H. Linoleic acid enhance the production of moncolin K and red pigments in Monascus ruber by activating mokH and mokA, and by accelerating cAMP-PkA pathway. Int J Biol Macromol. 2018;109:950–954. doi: 10.1016/j.ijbiomac.2017.11.074. [DOI] [PubMed] [Google Scholar]
- Jin YJ, Pyo YH. Effect of Monascus-fermented soybean extracts on antioxidant and skin aging-related enzymes inhibitory activities. Prev Nutr Food Sci. 2017;22:376–380. doi: 10.3746/pnf.2017.22.4.376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kao TC, Wu CH, Yen GC. Bioactivity and potential health benefits of licorice. J Agric Food Chem. 2014;62:542–553. doi: 10.1021/jf404939f. [DOI] [PubMed] [Google Scholar]
- Kim YJ, Lim HS, Kim Y, Lee J, Kim BY, Jeong SJ. Phytochemical quantification and the in vitro acetylcholinesterase inhibitory activity of Phellodendron chinense and its components. Molecules. 2017;22:925. doi: 10.3390/molecules22060925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim EK, Kim JH, Jeong S, Choi YW, Choi HJ, Kim CY, Kim YM. Pachypodol, a methoxyflavonoid isolated from Pogostemon cablin Bentham exerts antioxidant and cytoprotective effects in HepG2 cells: possible role of ERK-dependent Nrf2 activation. Int J Mol Sci. 2019;20:4082. doi: 10.3390/ijms20174082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim KU, Lee SJ, Lee I. Development of an Improved menopausal symptom-alleviating licorice (Glycyrrhiza uralensis) by biotransformation using Monascus albidulus. J Microbiol Biotechnol. 2020;30:178–186. doi: 10.4014/jmb.1909.09037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee SM, Kim SB, Kim YS. Determination of key volatile compounds related to long-term fermentation of soy sauce. J Food Sci. 2019;84:2758–2776. doi: 10.1111/1750-3841.14771. [DOI] [PubMed] [Google Scholar]
- Lee Y, Yoon Y, Choi KH. Probiotics-mediated bioconversion and periodontitis. Food Sci Anim Resour. 2021;41:905–922. doi: 10.5851/kosfa.2021.e57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li L, Wang L, Fan W, Jiang Y, Zhang C, Li J, Peng W, Wu C. The application of fermentation technology in traditional chinese medicine: a review. Am J Chin Med. 2020;48:899–921. doi: 10.1142/S0192415X20500433. [DOI] [PubMed] [Google Scholar]
- Li Z, Liu Y, Li Y, Lin F, Wu L. Screening and identification of Monascus strains with high-yield monacolin K and undetectable citrinin by integration of HPLC analysis and pksCT and ctnA genes amplification. J Appl Microbiol. 2020;129:1410–1418. doi: 10.1111/jam.14689. [DOI] [PubMed] [Google Scholar]
- Lin CM, Lin YT, Lin RD, Huang WJ, Lee MH. Neurocytoprotective effects of aliphatic hydroxamates from lovastatin, a secondary metabolite from Monascus-fermented red mold rice, in 6-hydroxydopamine (6-OHDA)-treated nerve growth factor (NGF)-differentiated PC12 cells. ACS Chem Neurosci. 2015;6:716–724. doi: 10.1021/cn500275k. [DOI] [PubMed] [Google Scholar]
- Liu L, Zhao J, Huang Y, Xin Q, Wang Z. Diversifying of chemical structure of native Monascus pigments. Front Microbiol. 2018;9:3143. doi: 10.3389/fmicb.2018.03143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maric A, Skocaj M, Likar M, Sepcic K, Cigic IK, Grundner M, Gregori A. Comparison of lovastatin, citrinin and pigment production of different Monascus purpureus strains grown on rice and millet. J Food Sci Technol. 2019;56:3364–3373. doi: 10.1007/s13197-019-03820-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okonogi S, Kaewpinta A, Junmahasathien T, Yotsawimonwat S. Effect of rice variety and modification on antioxidant and anti-inflammatory activities. Drug Discov Ther. 2018;12:206–213. doi: 10.5582/ddt.2018.01041. [DOI] [PubMed] [Google Scholar]
- Shimizu T, Kinoshita H, Ishihara S, Sakai K, Nagai S, Nihira T. Polyketide synthase gene responsible for citrinin biosynthesis in Monascus purpureus. Appl Environ Microbiol. 2005;71:3453–3457. doi: 10.1128/AEM.71.7.3453-3457.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soheili M, Salami M. Lavandula angustifolia biological characteristics: an in vitro study. J Cell Physiol. 2019;234(9):16424–30. doi: 10.1002/jcp.28311. [DOI] [PubMed] [Google Scholar]
- Srianta I, Zubaidah E, Estiasih T, Yamada M, Harijono Comparison of Monascus purpureus growth, pigment production and composition on different cereal substrates with solid state fermentation. Biocatal Agric Biotechnol. 2016;7:181–186. doi: 10.1016/j.bcab.2016.05.011. [DOI] [Google Scholar]
- Surh J, Yun JM. Antioxidant and anti-inflammatory activities of butanol extract of Melaleuca leucadendron L. Prev Nutr Food Sci. 2012;17:22–28. doi: 10.3746/pnf.2012.17.1.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valdes AF, Martinez JM, Lizama RS, Vermeersch M, Cos P, Maes L. In vitro anti-microbial activity of the Cuban medicinal plants Simarouba glauca DC, Melaleuca leucadendron L and Artemisia absinthium L. Mem Inst Oswaldo Cruz. 2008;103:615–618. doi: 10.1590/S0074-02762008000600019. [DOI] [PubMed] [Google Scholar]
- Wisecaver JH, Slot JC, Rokas A. The evolution of fungal metabolic pathways. PLoS Genet. 2014;10:e1004816. doi: 10.1371/journal.pgen.1004816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Y, Jiang Y, Zhang L, Zhou J, Yu Y, Zhou Y, Kang T. Chemical profiling and antioxidant evaluation of Paeonia lactiflora Pall. “Zhongjiang” by HPLC-ESI-MS combined with DPPH assay. J Chromatogr Sci. 2021;59:795–805. doi: 10.1093/chromsci/bmab005. [DOI] [PubMed] [Google Scholar]
- Wu L, Liu J, Huang W, Wang Y, Chen Q, Lu B. Exploration of Osmanthus fragrans Lour.‘s composition, nutraceutical functions and applications. Food Chem. 2022;377:131853. doi: 10.1016/j.foodchem.2021.131853. [DOI] [PubMed] [Google Scholar]
- Xin Q, Yuan R, Shi W, Zhu Z, Wang Y, Cong W. A review for the anti-inflammatory effects of paeoniflorin in inflammatory disorders. Life Sci. 2019;237:116925. doi: 10.1016/j.lfs.2019.116925. [DOI] [PubMed] [Google Scholar]
- Xiong Z, Cao X, Wen Q, Chen Z, Cheng Z, Huang X, Zhang Y, Long C, Zhang Y, Huang Z. An overview of the bioactivity of monacolin K / lovastatin. Food Chem Toxicol. 2019;131:110585. doi: 10.1016/j.fct.2019.110585. [DOI] [PubMed] [Google Scholar]
- Xue J, Guijas C, Benton HP, Warth B, Siuzdak G. METLIN MS(2) molecular standards database: a broad chemical and biological resource. Nat Methods. 2020;17:953–954. doi: 10.1038/s41592-020-0942-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X, Xiang L, Zhang C, Cao Y, Wang C. Promotion of monacolin K production in Monascus extractive fermentation: the variation in fungal morphology and in the expression levels of biosynthetic gene clusters. J Sci Food Agric. 2021;101:5652–5659. doi: 10.1002/jsfa.11218. [DOI] [PubMed] [Google Scholar]
- Yang Y, Liu B, Du X, Li P, Liang B, Cheng X, Du L, Huang D, Wang L, Wang S. Complete genome sequence and transcriptomics analyses reveal pigment biosynthesis and regulatory mechanisms in an industrial strain, Monascus purpureus YY-1. Sci Rep. 2015;5:8331. doi: 10.1038/srep08331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanli F, Xiang Y. Perspectives on functional red mold rice: functional ingredients, production, and application. Front Microbiol. 2020;11:606959. doi: 10.3389/fmicb.2020.606959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin L, Guan E, Zhang Y, Shu Z, Wang B, Wu X, Chen J, Liu J, Fu X, Sun W, Liu M. Chemical profile and anti-inflammatory activity of total flavonoids from Glycyrrhiza uralensis Fisch. Iran J Pharm Res. 2018;17:726–734. [PMC free article] [PubMed] [Google Scholar]
- Yuan Q, Zhang J, Xiao C, Harqin C, Ma M, Long T, Li Z, Yang Y, Liu J, Zhao L. Structural characterization of a low-molecular-weight polysaccharide from Angelica pubescens Maxim. f. biserrata Shan et Yuan root and evaluation of its antioxidant activity. Carbohydr Polym. 2020;236:116047. doi: 10.1016/j.carbpol.2020.116047. [DOI] [PubMed] [Google Scholar]
- Zeng H, Qin L, Liu X, Miao S. Increases of lipophilic antioxidants and anticancer activity of coix seed fermented by Monascus purpureus. Foods. 2021;10(3):566. doi: 10.3390/foods10030566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang BB, Xing HB, Jiang BJ, Chen L, Xu GR, Jiang Y, Zhang DY. Using millet as substrate for efficient production of monacolin K by solid-state fermentation of Monascus ruber. J Biosci Bioeng. 2018;125:333–338. doi: 10.1016/j.jbiosc.2017.10.011. [DOI] [PubMed] [Google Scholar]
- Zhang Q, Zheng Y, Hu X, Hu X, Lv W, Lv D, Chen J, Wu M, Song Q, Shentu J. Ethnopharmacological uses, phytochemistry, biological activities, and therapeutic applications of Alpinia oxyphylla Miquel: a review. J Ethnopharmacol. 2018;224:149–168. doi: 10.1016/j.jep.2018.05.002. [DOI] [PubMed] [Google Scholar]
- Zhang C, Chai S, Hao S, Zhang A, Zhu Q, Zhang H, Wang C. Effects of glutamic acid on the production of monacolin K in four high-yield monacolin K strains in Monascus. Appl Microbiol Biotechnol. 2019;103:5301–5310. doi: 10.1007/s00253-019-09752-9. [DOI] [PubMed] [Google Scholar]
- Zhang L, Kang W, Lu X, Ma S, Dong L, Zou B. Weighted gene co-expression network analysis and connectivity map identifies lovastatin as a treatment option of gastric cancer by inhibiting HDAC2. Gene. 2019;681:15–25. doi: 10.1016/j.gene.2018.09.040. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Figure S1. Phylogeny of the partial mokA (a) and mokE (b) from Monascus species, and the related genes from A. terreus, and P. citrinum.
Additional file 2: Figure S2. LC/MS analysis with or without M. ruber BCRC 31535-fermented G. uralensis in positive and negative modes.
Additional file 3: Figure S3. The metabolites of the ten compounds with the most increase after 60-day G. uralensis fermentation in MS/MS spectrum.
Additional file 4: Table S1. Pearson correlation between total phenols and antioxidant activity by medicinal plants without fermentation.
Additional file 5: Table S3. Pearson correlation between total phenols and antioxidant activity by 120 days fermentation of M. ruber BCRC 31535.
Additional file 6: Table S2. Pearson correlation between total phenols and antioxidant activity by 60 days fermentation of M. ruber BCRC 31535.
Additional file 7: Table S4. Significance of p-value for repeated measures ANOVA on cell viability from the M. ruber BCRC 31535-fermentation of various medicinal plants.
Additional file 8 Table S5. Significance of p-value for repeated measures ANOVA on red pigment and monacolin K from the M. ruber BCRC 31535-fermentation of various medicinal plants and rice.
Data Availability Statement
The data used and analyzed in this study can be provided from the corresponding author for scientific, non-profit purpose.