Abstract
Robotic esophageal surgery is becoming more widely adopted. Several publications on the feasibility, short-term outcomes and technical aspects are available. Most of these articles used either the da Vinci® SI system or in newer series the Xi System. The da Vinci® X system is generally considered less suited for multiquadrant access like in esophageal surgery, hence only limited data is available. Here we describe our initial experience with 16 Ivor-Lewis robotic assisted minimally invasive esophagectomies (RAMIE) in patients with esophageal adenocarcinoma. The da Vinci® X system was installed in our department in 2019; the robotic program comprises colorectal, pancreatic and esophageal surgery. The first two patients were operated in the presence of a proctor. An operative standard was established including fluorescence angiography (Firefly®). Technical aspects with focus on the characteristics of the da Vinci® X system, operating room setup, and short-term outcomes are discussed.
Keywords: Esophageal adenocarcinoma, Robotic Assisted Minimally Invasive Esophagectomy (RAMIE)
INTRODUCTION
The poor prognosis and rising incidence of esophageal cancer demonstrate the necessity for improved treatment options.1 The current 10-year overall survival rate in Germany is approximately 16%.2 Adenocarcinoma is the predominant esophageal cancer in developed nations, and important risk factors include chronic gastroesophageal reflux disease, obesity, and smoking.3 Localized tumors can be treated with endoscopic mucosal resection, whereas more advanced tumors need multidisciplinary treatment including surgery.4 However, the esophagus is difficult to reach in open surgery, requiring a simultaneous abdominal and thoracic approach causing significant morbidity and mortality.5 Minimally invasive esophagectomy has been shown to improve perioperative outcomes in resectable esophageal cancer.6–10 Since minimally invasive surgery of the esophagus is technically challenging using standard laparoscopic instruments, robotic surgery is a promising approach to overcome the limitations of laparoscopic/thoracoscopic surgery. The majority of recent publications on esophageal surgery used the da Vinci® Xi system which is considered the most suitable choice for multiquadrant access.11 Here we describe our experience using the more affordable da Vinci® X system for robotic assisted minimally invasive esophagectomy (RAMIE) in 16 patients with adenocarcinoma of the esophagus.
The da Vinci X® System was installed in our clinic in March 2019 and is the first robotic system used in our hospital. The urology and visceral surgery departments alternate using the system. The primary robotic surgeon (HA) has acquired experience in robotic surgery since 2013 primarily in colorectal, pancreatic, and liver surgery. However, his experience in robotic upper gastrointestinal (GI) surgery was limited.
Since the implementation of robotic surgery in the department of visceral surgery at Klinikum Robert-Koch, the authors have performed 163 robotic operations between March 1, 2019 to October 31, 2021. After training the entire team and four months experience in our department, we felt competent at handling the system and confident to start with robotic esophagectomy. The first and second robotic esophagectomies were performed in the presence of a proctor (Prof. Dr. Jan-Hendrik Egberts, Israelitisches Krankenhaus, Hamburg).
In this case report, we describe our experience with the establishment of an advanced robotic-assisted procedure in a district hospital. Moreover, we present our standard procedure using the da Vinci X® system, which was adapted by our team over time.
METHODS
Treatment of patients with esophageal cancer is undertaken in accordance with national and international guidelines:2 patients with esophageal cancer in our national board-certified cancer center undergo routine staging including, but not limited to, endoscopy, endoscopic ultrasound, computed tomography scans (thorax and abdomen), as well as pulmonary function testing with spirometry. Additionally, bronchoscopy is performed in some patients to rule out tracheobronchial infiltration.12
Characteristics of the da Vinci X® System and Comparison of the Robotic Systems
The advantages of robotic over laparoscopic surgery are well described.13 In brief, robotic surgery offers improved dexterity, elimination of the fulcrum effect and physiological tremor, a surgeon- controlled camera, improved range of motion with seven degrees of freedom, and the ability to scale motions of the instruments.17 Three-dimensional vision and near-infrared fluorescence imaging (Firefly®) are also available for laparoscopic surgery and therefore not specific to the da Vinci® system.
The da Vinci Xi® system has been available since 2014. Compared to the SI® system, it has a wider range of motion of the arms since they are mounted onto an overhead boom. This is beneficial in multiquadrant surgery, e.g., colorectal and esophageal resections. The arms are thinner, which makes arm collisions less frequent, and they have one extra joint to facilitate intraoperative handling of arm collisions. The instruments are longer, enabling a more flexible port placement and better access to distant regions like the deep pelvis or the thoracic inlet in esophageal surgery. The camera is longer as well; it is 80.5 mm in diameter and can be placed in any of the trocars (port hopping) which allows for a better view in difficult situations or an alternative angle on delicate structures.14,15 The Xi® System is therefore considered the high-end device; however, the investment remains high.
In 2017, Intuitive introduced the more affordable da Vinci X® system. It has many features of the Xi® systems (thinner arms, 8 mm camera, longer instruments, Firefly®, same console). Compared with the Si® system, the thinner arms and the longer instruments make port placement more flexible and easier. Arm collisions occur considerably less frequently and are often manageable without undocking. In contrast to the Xi® system, the arms of the X® system are directly mounted to the patient cart and not to the overhead boom. Theoretically, this setup sacrifices some of the versatility that the overhead design offers for multiquadrant access. However, we did not encounter any serious problems in multiquadrant approaches, e.g., rectal surgery, which requires the widest range of motion in general surgery, and therefore felt encouraged to use the system for esophageal surgery as well.
Operating Room Setup and Anesthesia Considerations
The current operating room (OR) setup is shown in Figures 1 and 2. During the introduction of RAMIE, there was a certain evolution of the setup because space is limited in our OR (40 m2). In the first 10 cases in contrast to Figures 1 and 2, the video cart was on the other side of the patient cart, and the patient table was rotated 270° when shifting from the abdominal to the thoracic part since the patient table was fixed to the ground and could be rotated in one direction only. This procedure was time-consuming and could not be optimized until the mobile table was available in our department. With the new setup, the patient cart still has to be moved, but far less than in the primary approach.
Figure 1.
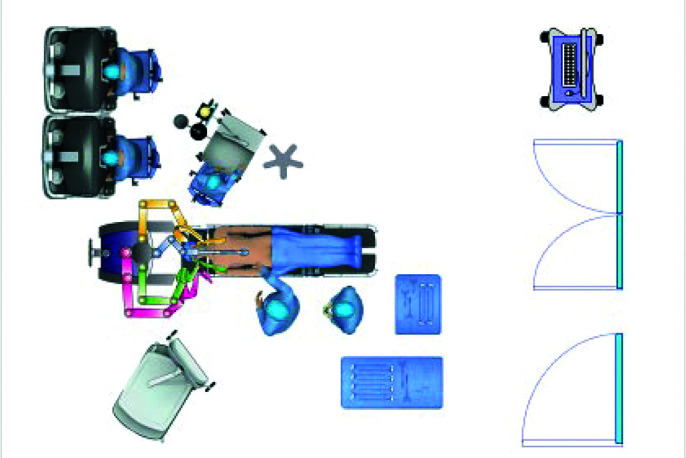
Operating room setup for the abdominal part of robotic assisted minimally invasive esophagectomy.
Figure 2.
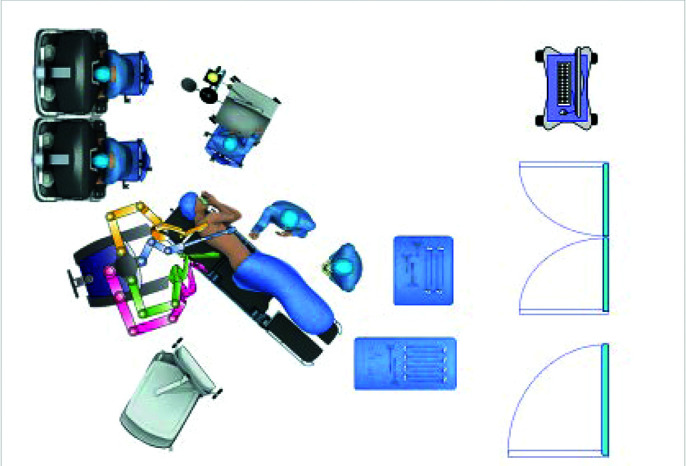
Operating room setup for the thoracic part of robotic assisted minimally invasive esophagectomy.
Our port placement (Figure 3 and 4) does not differ from approaches using the Xi® System and a study on this approach was published by Egberts et al.16 The main difference is the direction of docking. The da Vinci Xi® system can be docked from the patient’s side since the boom can be rotated in any direction. The robotic arms of the da Vinci X® are fixed to the patient cart, the optimal docking direction for the abdominal part is cranio-caudally over the patient’s head. Therefore, access to the patient’s head is markedly impaired.
Figure 3.
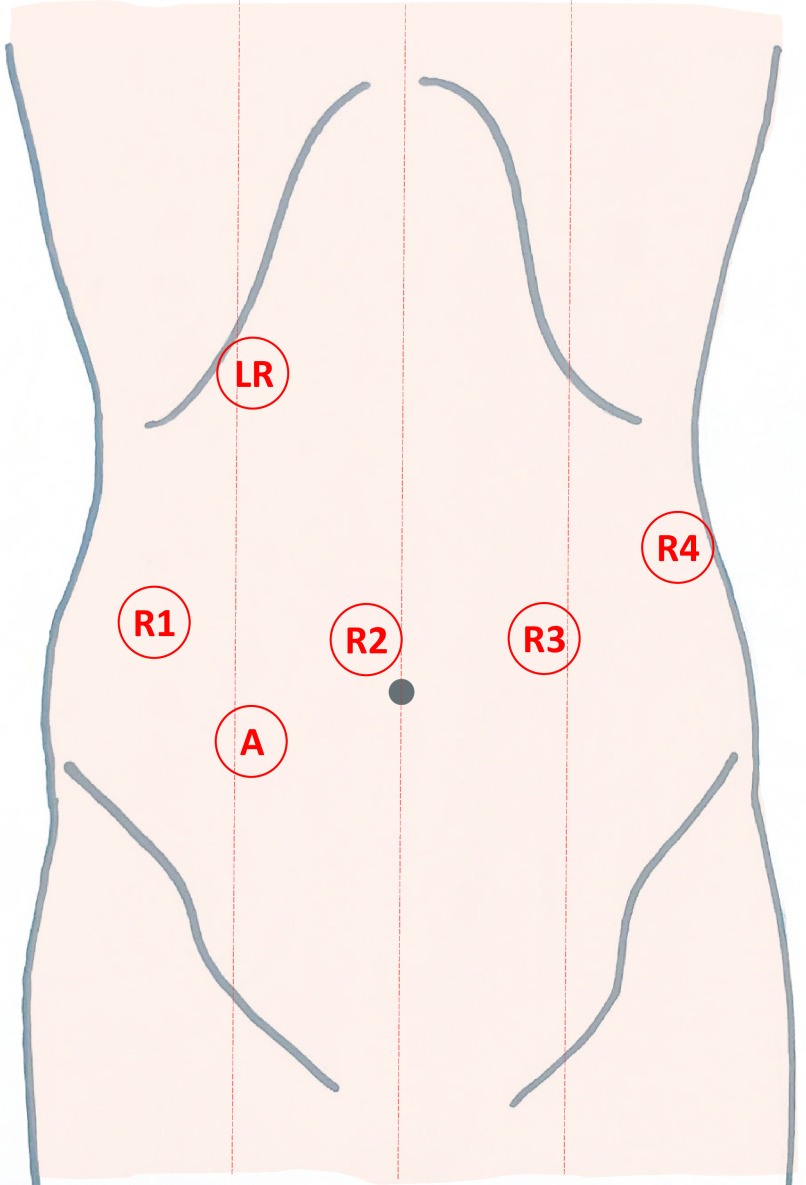
Port placement for the abdominal phase (R1 – R4: Robotic Arms 1 – 4, A: Assistant port, LR: Liver retractor).
Figure 4.
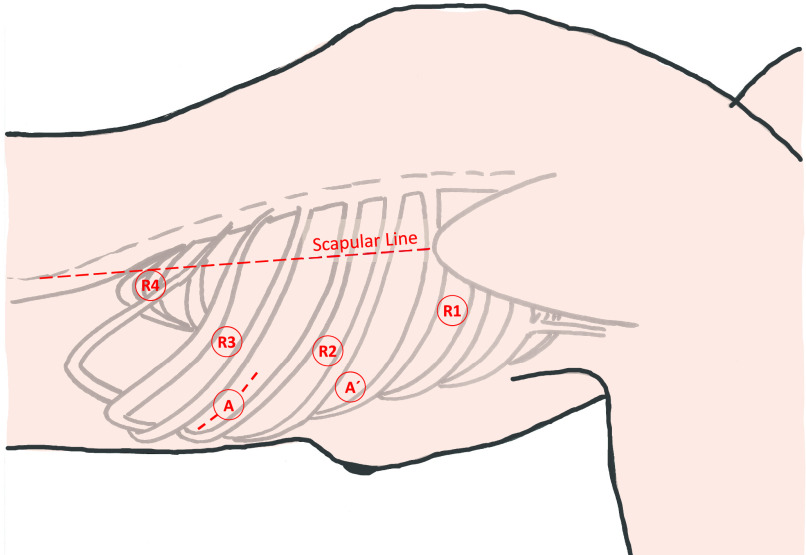
Port placement for the thoracic phase (R1 – R4: Robotic arms 1 – 4, A: Assistant port specimen removal/stapler entry, A´: Assistant port and specimen removal if anastomosis is close to the thoracic inlet).
Surgery is performed under general anesthesia. Prior to the induction of anesthesia, a thoracic epidural catheter for perioperative pain management is inserted and initially loaded with ropivacaine and sufentanil followed by continuous application of ropivacaine. After induction of anesthesia, the patient is placed in supine position. A double lumen endotracheal tube with a built-in video camera is inserted to continuously monitor theposition of the tip (VivaSight 2 DLT, Ambu, Bad Nauheim, Germany). Monitoring is performed using a central venous and arterial catheter and a urinary catheter with temperature probe. The patient’s hypnotic state and relaxation are continuously monitored (Narcotrend, Narcotrend-Gruppe, Hanover, Germany; ToFscan®, Dräger, Lübeck, Germany) to avoid any unintended movement of the patient since this can lead to severe damage to the abdominal and thoracic wall.
Due to overhead docking of the patient cart, access to the patient’s head and arms is limited. Therefore, all lines and the endotracheal tube are sufficiently extended and meticulously secured. Maintaining the patient’s body temperature during surgery lasting up to 8 hours is challenging. We used upper body warming during the abdominal part and lower body warming during the thoracic phase (3M Bair Hugger, 3M Deutschland, Neuss), and an infusion warming system (level 1 HOTLINE Blood and Fluid Warmer, Smiths Medical, Grasbrunn, Germany).
Regarding marginal blood loss, it is usually sufficient for restrictive fluid management to use balanced electrolyte solutions.
During the thoracic phase, the patient is placed in a left lateral decubitus position with the right arm in a swimmer’s position nearly covering the patient’s head. Access to the patients head again is impaired; however, access to the arms is easier. The patient and especially the head must be carefully secured for the table is tilted at least 15° – 20° to the left. Intraoperative manipulation of the nasogastric tube is challenging but necessary when the esophagus is transected and during/after completion of the esophagogastrostomy to ensure the tip of the nasogastric tube being placed at least 5 – 10 cm in the gastric conduit. After surgery, the patients wake up in the OR and are transferred to the intensive care unit.
Abdominal Part
The patient is placed on a vacuum mattress in a supine position with a 20° – 25° reverse Trendelenburg and a 10° right tilt. The 8 mm da Vinci ports are placed on a straight line slightly above the umbilicus (see Figure 3). Port 2 (R2) is usually placed in the midline, port 4 slightly cranial to the other ports and as lateral as possible. Port 3 is placed on the left midclavicular line. Port 1 (R1) is placed lateral to the right midclavicular line. A 12 mm robotic port is inserted if a da Vinci stapler is used for gastric conduit construction. If a laparoscopic stapler is used, an 8 mm port is inserted in R1. The 12 mm assistant port is placed caudally and between 1 and 2 (see above). A liver retractor is inserted subcostally on the midclavicular line.
Dissection starts with the division of the lesser omentum close to the liver until the hiatus is reached. If an accessory left hepatic artery is encountered, it can usually be preserved, and the left gastric branches are divided directly at the origin from this vessel. D2-lymph node dissection is carried out starting at the left border of the hepatoduodenal ligament retrograde along the proper and common hepatic artery at the superior border of the pancreas to the celiac axis. The right gastric artery is preserved. The right crus of the diaphragm is identified. Lymph node dissection proceeds along the splenic artery until the splenic hilum. The left gastric vein is divided at the crossing over the common hepatic artery. The left gastric artery is encircled and divided at the origin from the celiac trunc. The lymph node package is dissected en bloc and left connected to the lesser curve of the stomach. The esophago-gastric junction is encircled and the left crus is separated. The gastric fundus is identified and lifted up to the anterior abdominal wall. By doing so, the upper short gastric vessels can be divided using the vessel sealer. This facilitates the dissection of the remaining short gastric vessels later on. The right crus is subtotally incised and the distal esophagus is encircled.
To dissect the greater curve, the lesser sac is entered by dividing the gastrocolic ligament close to the transverse colon left of the midline. Identification of the gastroepiploic arcade can be facilitated using near-infrared fluorescence (Firefly®) (Figure 5). This is also helpful to identify anastomotic arcades between the right and left gastroepiploic artery in the greater omentum which then can be preserved. The gastrocolic ligament is divided to the left and the remaining short gastric vessels are divided.
Figure 5.

Visualization of the gastroepiploic arcade before dividing the greater omentum using near infrared fluorescence angiography (Firefly®).
Further to the right side, the gastroepiploic fat body is separated from the omentum and the mesocolon and the duodenum is identified. A partial Kocher maneuver is carried out. About 5 cm proximal of the pylorus, the lesser curve of the stomach is sceletonized and the construction of the gastric conduit is started using either the EndoWrist® da Vinci Linear Stapler or a laparoscopic stapler (we used Echelon 45 mm, Ethicon, Norderstedt, Germany). At the gastric fundus, a small bridge is left between the conduit and the lesser curve, which is stapled after the conduit is pulled up into the thorax during the thoracic phase. Initially, we used a 29 mm circular stapler for the anastomosis; therefore, the conduit was created with a diameter of 4 – 5 cm. In our latest cases, we switched to a handsewn anastomosis, and the diameter of the conduit was rather 3 – 4 cm.
A Blake drainage is inserted in R1 and placed under the left lobe of the liver through the hiatus as far as possible into the mediastinum. The patient cart is then undocked and the instruments are kept sterile.
Thoracic Part
After wound closure and sterile dressings, the whole table is rotated as shown in Figure 1. The patient is then placed in a left lateral decubitus position with the right arm in a swimmer’s position. The vacuum mattress is deflated and care is taken to form an adequate cavity for the left arm to prevent radial nerve palsy. Extra sustainers are placed on the sternum and the right upper limb. The table is tilted anteriorly until the scapular line is the apex of the patient. A reverse Trendelenburg tilt is applied until the lateral wall of the chest is nearly straight and parallel to the ground.
Ports are placed as shown in Figures 3 and 4. The port placement does not differ significantly from recommendations previously described for the Xi® system.16 The da Vinci ports are placed semicircularly between the scapular and anterior axillary line: R4 in the fourth intercostal space (ICS) 1 cm medial to the scapula, R3 slightly medial to R4 in the sixth ICS, R2 on the level of R4 in the eight ICS and R1 in the 10th ICS on the scapular line. If a laparoscopic stapler is used, all ports are 8 mm. In case a da Vinci EndoWrist® Stapler is used, a 12 mm cannula is introduced in R1. A 12 mm assistant port (A) is usually inserted in the seventh intercoastal space. If the anastomosis is expected to be rather high, the assistant port can be inserted in the fifth ICS (A). The incision can be extended to a mini thoracotomy for specimen removal and introduction of the circular stapler.
After dividing the right pulmonary ligament and mobilizing the lower lobe of the lung until the pulmonary vein, dissection starts with the incision of the pleura covering the azygos vein. The azygos vein is divided either between clips or a vascular stapler. The pleura is incised along the medial margin of the azygos vein and the lymphatic tissue en bloc with the esophagus is removed from the aorta, the left pleura, the pericardium, and the left and right main bronchus. The subcarinal lymph nodes are usually included, and the small supplying artery is clipped. In one patient, we encountered an infiltration of the right pleura and the lower lobe of the right lung, which was dissected using the vessel sealer and oversewn with a monofilament absorbable suture (4/0 PDS, Ethicon, Norderstedt, Germany). The vagus nerves are divided distally to the bronchial branches, which are preserved, if possible. The esophagus is encircled slightly cranially to the azygos vein and 2 – 3 cm below using scissors without coagulation in order to allow for a frozen section. The esophagus is divided with a safety margin of at least 5 cm proximal to tumor.
The stomach is then pulled up into the thorax carefully to avoid breaking of the staple line or damaging the serosa. This step is supported by the table assistant after extension of the assistant port to a mini thoracotomy and insertion of an Alexis® Wound Retractor (Applied Medical, Rancho Santa Margarita, CA, USA). Twisting of the gastric tube is thoroughly avoided; therefore, the gastroepiploic arcade is kept dorsally. In the first cases, we utilized a 29 mm circular stapler for reconstruction. To ensure a full thickness anastomosis, the esophageal mucosa is secured by four to six interrupted sutures using PDS 5/0 (see Figure 6). The anvil of the stapler is introduced into the esophageal stump and secured with a purse string suture using Stratafix Spiral® 3/0. Perfusion of the gastric conduit was validated by injecting indocyanine green (ICG) in the Firefly mode. The circular stapler was introduced via a small incision of the lesser curve of the gastric part of the specimen and forwarded into the gastric conduit. After construction of the end-to-side esophago-gastrostomy, the gastric conduit is completed using either the laparoscopic or EndoWrist® stapler. The anastomosis was reinforced with a circular suture (Stratafix® 4/0). The linear suture line was only oversewn when considered necessary, but not routinely.
Figure 6.

Interrupted sutures for fixation of the esophageal mucosa to ensure a full thickness anastomosis (stapled or hand-sewn anastomosis).
Since it is often challenging to introduce the circular stapler through the intercostal space and find the correct position in the gastric conduit, we switched to a robotic-assisted handsewn end-to-side anastomosis in our later series. This anastomosis is carried out after validation of the perfusion by injection of ICG. If a demarcation line can be detected, the anastomosis is performed in the well perfused area distal to the demarcation line. The conduit is then completed using the linear stapler and the specimen is removed. To secure the esophageal mucosa, four to six interrupted stay sutures are applied using PDS 5/0 (Ethicon, Norderstedt, Germany). The posterior wall of the gastric conduit is incised vertically to the greater curve at least 2 cm distant to any staple line. 4/0 Stratafix® (Ethicon, Norderstedt, Germany) is used for the running anastomosis (Figure 7). If necessary, the anastomosis is reinforced using interrupted sutures with PDS 4/0 (Ethicon, Norderstedt, Germany). A nasogastric tube is forwarded approximately 5 cm distal to the anastomosis.
Figure 7.

End-to-Side esophagogastrostomy using a 3/0 barbed suture.
If possible, the anastomosis is wrapped with a flap of the omentum, which is fixed with a suture. The Blake drainage is placed in the esophageal bed posterior to the gastric conduit with the tip close to the anastomosis. Two chest drains are placed, one next to the anastomosis, one caudally to the diaphragm.
RESULTS
In the period from May 2019 to August 2021 (27 months), we carried out 16 robotic-assisted esophagectomies in patients with esophageal adenocarcinoma. The median hospital stay in our series was 16 days, one patient was released on day 9 and most patients on day 13, however due to the small number of cases the patients with complications (see Figure 8) increased the average hospitalization time. No deaths occurred; therefore, in-house mortality was 0%. There were 2 female patients and 14 male patients. The average number of harvested lymph nodes was 23. The average body mass index (BMI) was 26, with a range of 20 to 32. The body height of the patients was 143 cm to 190 cm, average height was 177 cm. Neoadjuvant therapy was performed in 13 patients. Two patients had previous abdominal surgery (one laparoscopic sigmoid resection and one open feeding jejunostomy). We did not encounter any difficulties in these patients although minor adhesiolysis was necessary. Therefore, we did not observe a prolonged operative time or a change in operative strategy. The average operating time of all cases is 516 minutes. The mean time for port placement was 10 minutes. The average docking time was seven minutes for the abdominal phase and eight minutes for the thoracic phase.
Figure 8.
Table of complications.
The average time required to change from the abdominal part to the thoracic part by rotating the table and repositioning the patient from supine position to a left lateral decubitus position with the right arm in swimmer’s position was 29 min. In the first 10 cases when we used the rigid patient table, the average time was 34 minutes. We were able to improve this time with the mobile patient table to an average time of 21 minutes. If we divide the cases in two groups consisting of 10 cases without and six cases with the mobile table, the average operating time in the first group is 526 minutes and in the other group 495 minutes. Anastomotic leakage of the esophago-gastrostomy occurred in two patients, accounting for a leakage rate of 12.5% (2/16). We did not observe any leakage in robotic handsewn anastomoses 0/4. Both patients with anastomotic leakage were treated successfully using endoscopic vacuum therapy. Both had no major complications during the treatment or in the close follow-up. One of these patients was discharged on day 22 of hospital stay and the other on day 33.
Although intraoperative fluorescence angiography revealed rapid enhancement in the conduit, one patient developed mucosal congestion and partial necrosis of the gastric mucosa in the proximal 2 cm of the conduit. However, anastomotic leakage was not observed in repeated endoscopic controls.
One patient suffered from pleural empyema/abscess with no apparent leakage. He was treated by percutaneous drainage. However, on day 16, a leakage of the longitudinal staple line was detected during repeated endoscopy after clinical deterioration. The leakage could be successfully treated by endoscopic vacuum therapy.
In two cases, a conversion to open surgery was necessary. In the first case due to acute hypotension combined with upper inflow congestion. We therefore performed emergency undocking of the robot and a limited median laparotomy to exclude suspected pericardial tamponade. In conclusion, acute hypotension was caused by an anaphylactic reaction to medication. The patient recovered after administration of glucocorticoids and suprarenine. Surgery was terminated for this day and successfully performed in as an open procedure six days later. The second case of conversion was in a patient with obesity and a BMI of 33. After diagnostic laparoscopy, we decided to perform the abdominal part in an open manner to respect the fact of “safety first” in a situs without a secure overview. The thoracic part was then successfully performed with the robot.
DISCUSSION
To start with Robotic esophagectomy in our clinic was facilitated by the fact that the primary surgeon (HA) had gained experience in robotic surgery in advance.17–20 The team had prior experience in laparoscopic and open esophageal surgery; therefore, we felt confident to start with RAMIE. The first and second case were performed in the presence of a proctor and following a standardized approach previously described.16 Proctorship is considered to shorten the learning phase and is able to safely implement complex surgery at acceptable complication rates as Kwon et al. could show for robotic prostatectomy.21 In the presence of a dual console, the proctoring environment has significantly improved and allows for proctoring without prolonging operative time and compromising operative safety.22 Although fellowship training remains the strongest way for acquisition of robotic surgical skills, at present, proctorship remains a viable option to safely introduce new complex surgical techniques.23
The operative time in the first patients was longer due to limited space in our OR, and the team although experienced with Robotic surgery, was not familiar with robotic esophagectomy. Therefore, rotating the table and bringing the patient from the supine into the left lateral decubitus position was time consuming. However, we were able to reduce the time needed for this step and could decrease the average operating time by approximately 30 minutes comparing the first 10 patients with the last six patients. Switching to a mobile patient table reduced the time needed to change from the abdominal part to the thoracic part by 13 minutes on average. Moreover, the procedure turned out to be safer because we minimized the risk of dislocation of the endotracheal tube or the vascular access since the mobile table allowed for an optimized OR setup as described above. Due to local conditions, the fixed table had to be rotated for 270° and the anesthesia equipment had to follow the table. Whereas using the mobile table rotation could be reduced to 30° – 45° and, more important, the anesthesia equipment could nearly be left untouched. The da Vinci patient cart and video cart still had to be moved, but far less than with the initial approach.
In the initial cases, we performed a circular stapler anastomosis as previously described.16 This technique appeared challenging regarding the introduction of the circular stapler through a mini-thoracotomy, especially in male and obese patients. The correct placement of the stapler into the gastric conduit is difficult since only the upper margin of the anastomosis can be seen even if the camera is placed in the most apical port. Furthermore, we routinely over sew the anastomosis with a barbed suture. In patient 9 we encountered a stapler dysfunction which forced us to perform a hand sewn end-to-side anastomosis. This type of anastomosis felt quite convenient and the overview was better compared to the stapled technique. This led us to the reevaluate our concept, so that after discussion and planning, we performed robotic hand-sewn anastomosis starting with patient 14 and for the remaining operations. Although robotic handsewn anastomosis is considered more time-consuming compared to the stapler time, the average operating time in the last 4 cases was slowly decreasing. We believe the decrease in operative time was due to the increased experience in this procedure the entire team has gained over time. The time needed for completion of the robotic hand sewn anastomosis, although these times were not documented appeared comparable to the stapled approach. The two anastomotic leakages were observed in the circular stapler group (2/12) whereas we did not see any leakage in the four handsewn anastomoses (0/4). In the literature, there is still controversy regarding the ideal technique for reconstruction after Ivor-Lewis esophagectomy. The stapled anastomosis has a lower leakage rate but a higher incidence of strictures and pulmonary complications.24–26
Minimally invasive esophagectomy (MIE) has been shown to be advantageous over open surgery. The TIME Trial demonstrated less pulmonary complications, less pain, less blood loss, a shorter hospital stay, and a better quality of life after MIE compared to open surgery. Consistent with these findings, no blood transfusions were required in our patients, the median hospital stay was 16 days. As expected, operative time was longer in the MIE group, whereas oncological parameters e.g., R0 resection rate, lymph node harvest, and three-year survival were comparable in both groups.7–9 Several authors confirm these findings; however, a higher reintervention and reoperation rate was found in database studies from the United States and Japan.10,12 Further research is needed to clarify whether a learning curve effect or the technique itself accounts for these higher complication rates. Many of the well-known limitations of standard minimally invasive surgery can be overcome by the robotic system and this approach may reduce the complication rate after RAMIE. Although single center experiences with RAMIE are encouraging, there is only one propensity score matched study with 66 patients comparing RAMIE and MIE.27
We used ICG fluorescence angiography (FA) (Firefly®) in all patients to determine the anastomotic area in the conduit and verify the blood flow in the anastomosis. After provisional placement of the conduit and determination of the assumed localization of the anastomosis, ICG was injected and the inflow observed. If a demarcation line could be seen, the anastomosis was performed at least 2 cm distal to this line. We did not encounter any length problems in the gastric conduit in any patient; therefore, all anastomoses could be performed tension free. We did not use any quantification of the fluorescent dye e.g., time to inflow or intensity of fluorescence in the present series.
ICG-FA has been shown to significantly lower the anastomotic leakage rate as well as the reoperation rate in low anterior resections for rectal cancer.28 In esophageal resections, early publications used ICG-FA in a qualitative way to identify poorly perfused areas of the proximal conduit and/or a demarcation line as we did in our patients.29 In a systematic review the authors found an overall anastomotic leakage rate of 8.4% in the ICG-FA group and of 18.5% in the control (non-ICG-FA) group.30 In conclusion, ICG-FA is a useful tool to assess perfusion of the gastric conduit before performing any type of anastomosis. The routine use may be helpful to minimize the anastomotic leakage rate. The best way to perform ICG-FA (qualitative or quantitative) remains to be established.
Compared to the da Vinci® Xi system which is considered the high-end model, the more affordable da Vinci® X system has some limitations.31 The robotic arms are directly mounted to the patient cart whereas the arms of the Xi systems are mounted to an overhead boom offering more flexibility. Based upon our experience, the potential disadvantages of the da Vinci X are not clinically relevant. Collisions of the robotic arms can easily be managed intraoperatively by shifting arm 2 and 3 from one side to the other as needed. In the vast majority of cases, undocking of the patient cart can be avoided. Therefore, even low anterior resections of the rectum (LAR) can be performed in a single docking technique although this procedure has the widest operative field in general surgery.
We did not encounter any technical difficulties, neither during the abdominal nor the thoracic part of RAMIE. The port placement did not differ from approaches previously described using the da Vinci® Xi system.16 The operative field during the thoracic phase can be nearly as wide as during LAR ranging from the diaphragm to the upper thoracic inlet. Although in two of our patients the anastomosis was located cranially of the azygos vein close to the thoracic inlet, we did not observe any collisions of the robotic arms or other technical limitations requiring redocking or even conversion to open surgery. Therefore, we believe that the da Vinci® X system is a good and cost-effective alternative in RAMIE.
CONCLUSION
RAMIE can be safely performed by using the da Vinci® X system. In our series, we did not encounter any system-specific limitations that required conversion to open surgery. Arm collisions can easily be managed intraoperatively without the need for redocking even during the thoracic part with the widest operative field.
Robotic esophagectomy can be safely introduced after initial proctoring if the team as well as the hospital is experienced in both robotic surgery and esophageal surgery.
Footnotes
Disclosure: HA is proctor for Intuitive Surgical Inc, Sunnyvale, CA.
Funding sources: None.
Conflict of interests: None.
Informed consent: Dr. Juri Bergmann declares that written informed consent was obtained from the patient/s for publication of this study/report and any accompanying images.
Contributor Information
Juri Bergmann, General-, Visceral and Vascular Surgery, KRH Klinikum Robert Koch, Gehrden, Germany..
Barbara Lehmann-Dorl, Anesthesiology, KRH Klinikum Robert Koch, Gehrden, Germany..
Lars Witt, Anesthesiology, KRH Klinikum Robert Koch, Gehrden, Germany..
Heiko Aselmann, General-, Visceral and Vascular Surgery, KRH Klinikum Robert Koch, Gehrden, Germany.; General-, Visceral and Minimally Invasive Surgery, KRH Klinikum Siloah, Hannover, Germany.
References:
- 1.Huang FL, Yu SJ. Esophageal cancer: risk factors, genetic association, and treatment. Asian J Surg. 2018;41(3):210–215. [DOI] [PubMed] [Google Scholar]
- 2.Holscher AH, Gockel I, Porschen R. [Updated German S3 guidelines on esophageal cancer and supplements from a surgical perspective]. Chirurg. 2019;90(5):398–402. [DOI] [PubMed] [Google Scholar]
- 3.Short MW, Burgers KG, Fry VT. Esophageal cancer. Am Fam Physician. 2017;95(1):22–28. [PubMed] [Google Scholar]
- 4.Shah MA, Kennedy EB, Catenacci DV, et al. Treatment of locally advanced esophageal carcinoma: ASCO Guideline. J Clin Oncol. 2020;38(23):2677–2694. [DOI] [PubMed] [Google Scholar]
- 5.Schieman C, Wigle DA, Deschamps C, et al. Patterns of operative mortality following esophagectomy. Dis Esophagus. 2012;25(7):645–651. [DOI] [PubMed] [Google Scholar]
- 6.Yibulayin W, Abulizi S, Lv H, Sun W. Minimally invasive oesophagectomy versus open esophagectomy for resectable esophageal cancer: a meta-analysis. World J Surg Oncol. 2016;14(1):304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Biere SS, van Berge Henegouwen MI, Maas KW, et al. Minimally invasive versus open oesophagectomy for patients with oesophageal cancer: a multicentre, open-label, randomised controlled trial. Lancet. 2012;379(9829):1887–1892. [DOI] [PubMed] [Google Scholar]
- 8.Sihag S, Kosinski AS, Gaissert HA, Wright CD, Schipper PH. Minimally invasive versus open esophagectomy for esophageal cancer: a comparison of early surgical outcomes from the Society of Thoracic Surgeons National Database. Ann Thorac Surg. 2016;101(4):1281–1288; discussion 1288-1289. [DOI] [PubMed] [Google Scholar]
- 9.Straatman J, van der Wielen N, Cuesta MA, et al. Minimally invasive versus open esophageal resection: three-year follow-up of the previously reported randomized controlled trial: the TIME Trial. Ann Surg. 2017;266(2):232–236. [DOI] [PubMed] [Google Scholar]
- 10.Takeuchi H, Miyata H, Ozawa S, et al. Comparison of short-term outcomes between open and minimally invasive esophagectomy for esophageal cancer using a nationwide database in Japan. Ann Surg Oncol. 2017;24(7):1821–1827. [DOI] [PubMed] [Google Scholar]
- 11.de Groot EM, Goense L, Ruurda JP, van Hillegersberg R. State of the art in esophagectomy: robotic assistance in the abdominal phase. Updates Surg. 2021;73(3):823–830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fuchs HF, Muller DT, Leers JM, Schroder W, Bruns CJ. Modular step-up approach to robot-assisted transthoracic esophagectomy-experience of a German high volume center. Transl Gastroenterol Hepatol. 2019;4:62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Potscher A, Bittermann C, Langle F. Robot-assisted esophageal surgery using the da Vinci® Xi system: operative technique and initial experiences. J Robot Surg. 2019;13(3):469–474. [DOI] [PubMed] [Google Scholar]
- 14.Morelli L, Di Franco G, Lorenzoni V, et al. Structured cost analysis of robotic TME resection for rectal cancer: a comparison between the da Vinci Si and Xi in a single surgeon's experience. Surg Endosc. 2019;33(6):1858–1869. [DOI] [PubMed] [Google Scholar]
- 15.Protyniak B, Jorden J, Farmer R. Multiquadrant robotic colorectal surgery: the da Vinci Xi vs Si comparison. J Robot Surg. 2018;12(1):67–74. [DOI] [PubMed] [Google Scholar]
- 16.Egberts JH, Biebl M, Perez DR, et al. Robot-assisted oesophagectomy: recommendations towards a standardised Ivor Lewis procedure. J Gastrointest Surg. 2019;23(7):1485–1492. [DOI] [PubMed] [Google Scholar]
- 17.Aselmann H, Egberts JH, Beckmann JH, et al. Robotic pylorus-preserving pancreaticoduodenectomy: Video article]. Chirurg. 2017;88(5):411–421. [DOI] [PubMed] [Google Scholar]
- 18.Aselmann H, Kersebaum JN, Bernsmeier A, et al. Robotic-assisted total mesorectal excision (TME) for rectal cancer results in a significantly higher quality of TME specimen compared to the laparoscopic approach-report of a single-center experience. Int J Colorectal Dis. 2018;33(11):1575–1581. [DOI] [PubMed] [Google Scholar]
- 19.Aselmann H, Moller T, Kersebaum JN, et al. Robot-assisted liver resection. Chirurg. 2017;88(6):476–483. [DOI] [PubMed] [Google Scholar]
- 20.Egberts JH, Aselmann H, Hauser C, et al. Robotic-assisted oesophageal surgery]. Zentralbl Chir. 2016;141(2):145–153. [DOI] [PubMed] [Google Scholar]
- 21.Kwon EO, Bautista TC, Blumberg JM, et al. Rapid implementation of a robot-assisted prostatectomy program in a large health maintenance organization setting. J Endourol. 2010;24(3):461–465. [DOI] [PubMed] [Google Scholar]
- 22.Smith AL, Scott EM, Krivak TC, Olawaiye AB, Chu T, Richard SD. Dual-console robotic surgery: a new teaching paradigm. J Robot Surg. 2013;7(2):113–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Santok GD, Raheem AA, Kim LH, et al. Proctorship and mentoring: its backbone and application in robotic surgery. Investig Clin Urol. 2016;57(Suppl 2):S114–S120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Castro PM, Ribeiro FP, Rocha Ade F, Mazzurana M, Alvarez GA. Hand-sewn versus stapler esophagogastric anastomosis after esophageal ressection: systematic review and meta-analysis. Abcd, Arq Bras Cir Dig. 2014;27(3):216–221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Harustiak T, Pazdro A, Snajdauf M, Stolz A, Lischke R. Anastomotic leak and stricture after hand-sewn versus linear-stapled intrathoracic oesophagogastric anastomosis: single-centre analysis of 415 oesophagectomies. Eur J Cardiothorac Surg. 2016;49(6):1650–1659. [DOI] [PubMed] [Google Scholar]
- 26.Law S, Fok M, Chu KM, Wong J. Comparison of hand-sewn and stapled esophagogastric anastomosis after esophageal resection for cancer: a prospective randomized controlled trial. Ann Surg. 1997;226(2):169–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang Y, Han Y, Gan Q, et al. Early outcomes of robot-assisted versus thoracoscopic-assisted Ivor Lewis esophagectomy for esophageal cancer: a propensity score-matched study. Ann Surg Oncol. 2019;26(5):1284–1291. [DOI] [PubMed] [Google Scholar]
- 28.Watanabe J, Ishibe A, Suwa Y, et al. Indocyanine green fluorescence imaging to reduce the risk of anastomotic leakage in laparoscopic low anterior resection for rectal cancer: a propensity score-matched cohort study. Surg Endosc. 2020;34(1):202–208. [DOI] [PubMed] [Google Scholar]
- 29.Turner SR, Molena DR. The role of intraoperative fluorescence imaging during esophagectomy. Thorac Surg Clin. 2018;28(4):567–571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Koyanagi K, Ozawa S, Ninomiya Y, et al. Indocyanine green fluorescence imaging for evaluating blood flow in the reconstructed conduit after esophageal cancer surgery. Surg Today. 2022;52(3):369–376. [DOI] [PubMed] [Google Scholar]
- 31.Shin HR, Lee K, Yu HW, et al. Comparison of perioperative outcomes using the da Vinci S, Si, X, and Xi robotic platforms for BABA robotic thyroidectomy. Medicina (Kaunas). 2021;57(10):1130. [DOI] [PMC free article] [PubMed] [Google Scholar]