Abstract
Background/Aims
Gut-barrier dysfunction is well recognized in pathogenesis of both non-alcoholic fatty liver disease (NAFLD) and alcoholic liver disease (ALD). However, comparison of components of this dysfunction between the two etiologies remains unexplored especially in early stages of NAFLD.
Methods
Components of gut-barrier dysfunction like alterations in intestinal permeability (IP) by lactulose mannitol ratio (LMR) in urine, systemic endotoxemia (IgG and IgM anti-endotoxin antibodies), systemic inflammation (serum tumor necrosis factor alpha [TNF-α] and interleukin-1 [IL-1] levels), tight junction (TJ) proteins expression in duodenal biopsy and stool microbiota composition using Oxford Nanopore MinION device were prospectively evaluated in patients with NAFLD (n = 34) with no cirrhosis, ALD (n = 28) and were compared with disease free controls (n = 20).
Results
Patients with ALD had more advanced disease than those with NAFLD (median liver stiffness -NAFLD:7.1 kPa [5.9–8.9] vs. ALD:14.3 kPa [9.6–24], P < 0.001]. Median LMR was significantly higher in NAFLD and ALD group when compared to controls (NAFLD 0.054 [0.037–0.17] vs. controls 0.027 [0.021–0.045] (P = 0.001)) and ALD 0.043 [0.03–0.068] vs. controls 0.027 [0.021–0.045] (P = 0.019)]. Anti-endotoxin antibody titer (IgM) (MMU/mL) was lowest in NAFLD 72.9 [3.2–1089.5] compared to ALD 120.6 [20.1–728]) (P = 0.042) and controls 155.3 [23.8–442.9]) (P = 0.021). Median TNF-α (pg/mL) levels were elevated in patients with NAFLD (53.3 [24.5–115]) compared to controls (16.1 [10.8–33.3]) (P < 0.001) and ALD (12.3 [10.1–42.7]) (P < 0.001). Expression of zonulin-1 and claudin-3 in duodenal mucosa was lowest in NAFLD. On principal co-ordinate analysis (PCoA), the global bacterial composition was significantly different across the three groups (PERMANOVA test, P < 0.001).
Conclusion
While remaining activated in both etiologies, gut-barrier dysfunction abnormalities were more pronounced in NAFLD at early stages compared to ALD despite more advanced disease in the latter.
Keywords: intestinal permeability, endotoxemia, tight junction protein, non-alcoholic fatty liver disease, alcoholic liver disease
Abbreviations: ALD, alcoholic liver disease; ALT, alanine transaminase; AST, aspartate transaminase; IP, intestinal permeability; IL-1, interleukin-1; KFT, kidney function test; LFT, liver function test; LMR, lactulose mannitol ratio; NAFLD, non-alcoholic fatty liver disease; TNF, tumor necrosis factor
A bidirectional communication between liver and the gastrointestinal tract is now recognized as the key to maintenance of physiological functions in health, and its disruption can contribute to pathogenesis of several diseases affecting the liver, including non-alcoholic fatty liver disease (NAFLD) and alcoholic liver disease (ALD).1 The basic components of this bidirectional communication are the portal circulation which carries intestinal microbiome derived products to the liver and bile along with systemic circulation, which transmit signals to the intestinal tract via bile acids, inflammatory mediators, metabolites, and antibodies respectively. Transfer via portal circulation in turn depends on the intestinal microbiota composition, and integrity of the intestinal epithelial barrier and the gut-vascular endothelial barrier, the two barriers which control entry of bacterial products from the intestinal lumen to the portal circulation and constitute what is called the gut barrier.2,3 Gut-barrier is one of the major determinants of the gut–liver axis. The intestinal epithelial barrier regulates translocation of microbial products at both transcellular and paracellular levels.4 Paracellular pathway is controlled by several multiprotein junctional complexes of which tight junction proteins (TJPs) are the most relevant.5 Perturbations of the gut that disrupt critical interactions between the host and gut microbes, namely gut dysbiosis may translate into alterations in TJPs, as evidenced by disruption of tight junctions on electron microscopy6 and decreased expression of TJP on immunohistochemistry, essentially leading to increased intestinal permeability (IP).7 This interaction leads to increased bacterial products translocation and possibly liver injury via activation of inflammatory pathways. Gut-liver axis dysfunction especially gut-barrier dysfunction is central to the pathogenesis of NAFLD and ALD.1,8 While there is an evidence of existence of this gut-barrier dysfunction in cirrhosis9,10 especially in patients with decompensations,11,12 the components of this dysfunction and their relative activation in totality have seldom been studied in precirrhotic (early) stages of NAFLD.
Several studies have demonstrated a change in fecal microbiota composition in both, patients with non-alcoholic fatty liver disease (NAFLD) and alcoholic liver disease (ALD), specifically increase in Proteobacteria and a decrease in Firmicutes and Bacteroides,13,14 although significant discordance has been documented in patients with non-alcoholic steatohepatitis (NASH)14, 15, 16, 17 with findings varying from increase in Bacteroidetes18 and no change in the representation of Bacteroides and Firmicutes to decrease in Bacteroides levels.17 However, it is still unclear, how these changes in intestinal microbiome leads to increased bacterial translocation and translates to characteristic pathological changes in the liver especially in early stages of chronic liver disease (CLD) without any clinical decompensations.
To better understand the similarities as well as differences in gut-barrier dysfunction in both NAFLD and ALD, we in this study demonstrate changes in intestinal microbiota in conjunction with gut-barrier dysfunction, its consequence (bacterial products translocation) and the systemic response to these products in early stages of CLD (precirrhotic liver disease) of NAFLD and compared it with those of ALD. We choose to study precirrhotic stage in NAFLD because this is a state which is driven more by etiology of the disease, than by the impact of cirrhosis and different clinical decompensations associated with it.
Materials and methods
Patients
This was a prospective, single center cross-sectional observational study conducted at a tertiary care institute in North India catering to patients with liver diseases, including those with NAFLD and ALD. Patients of CLD without any present or past clinical decompensation like ascites, variceal bleeding and overt hepatic encephalopathy presenting over the period from December 2016 till January 2018 were evaluated for inclusion in the study. For the present study, there was no patient and public involvement. The diagnoses of NAFLD and ALD were made in included patients as described below. In addition, liver dysfunction due to other etiologies such as viral hepatitis, autoimmune hepatitis, Wilson's disease and hemochromatosis was ruled out in all patients using careful history taking and with appropriate laboratory investigations and imaging. Additionally, those with age more than 65 or less than 18 years, bilirubin more than 8 mg/dL, and those with hepatocellular carcinoma (HCC) were excluded from this study. Patients who had used drugs such as antibiotics, proton pump inhibitors, prokinetics, probiotics, prebiotics and laxatives such as lactulose within the last 4 weeks and those with pregnancy, renal dysfunction, history of bariatric surgery, active infection (evaluated by relevant clinical history followed by directed laboratory and/or radiological investigations), diarrhea or conditions primarily associated with increased intestinal permeability such as celiac disease and inflammatory bowel disease were also excluded from this study.
All the subjects underwent complete blood counts, liver and kidney function tests (LFT-KFT) and testing for prothrombin time-international normalized ratio (PT-INR). Furthermore, all patients underwent ultrasonography of the abdomen and transient elastography. Esophagogastroduodenoscopy was done to screen for varices in all patients. Liver biopsy was performed in majority of patients with NAFLD. Additional blood samples were collected for further analysis at the time of enrolment in the study. Serum was separated from blood within 1 h by centrifugation and stored at −20°C till analysis. This was analyzed for IgM and IgG anti-endotoxin antibody, IL-1 and TNF-α by ELISA. Patients were also given lactulose and mannitol solution and urine was collected according to prespecified protocol to assess IP. For patients with NAFLD, assessment of investigations were done prior to institution of lifestyle modifications and dietary changes prescribed in their management.
The control group consisted of patients with functional gastrointestinal disease presenting to gastroenterology-outpatient department (GE-OPD) who were planned for esophagogastroduodenoscopy (EGD) for additional indications. Those who had no pathological findings on EGD and had no evidence of liver disease were included and their duodenal biopsy specimens were collected as for included patients after written informed consent.
Diagnosis of NAFLD/ALD
For inclusion in the NAFLD group, patients presenting to GE-OPD with deranged liver function test (LFT) and evidence of fatty liver on histology or on imaging were evaluated. Patients with a history of significant alcohol intake (21 drinks/week in men and 14 drinks/week in women) were excluded. Diagnosis of NAFLD was based on histological evidence of steatosis and inflammation on liver biopsy or radiological evidence of fatty liver by ultrasound or using transient elastography (TE) (using a controlled attenuation parameter [CAP] value of greater than 275 dB/m as cut-off).19 Exclusion criteria specific to NAFLD cohort included consumption of drugs known to cause fatty liver such as tamoxifen, methotrexate, steroids, amiodarone, valproate and antiretroviral therapy.
For alcoholic liver disease (ALD) group, patients with active alcohol intake within past 6 months at the time of presentation were considered. The diagnosis of alcohol use disorder was made based on Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM- V) criteria, and the diagnosis of advanced ALD was established on clinical, radiological, transient elastographic (TE), and endoscopic evidence.20 Patients with severe alcoholic hepatitis (Maddrey's discriminant factor ≥32) were excluded from the study. Patients were advised to abstain from alcohol 7 days prior to assessment.21
The study protocol was approved by the institutional ethical committee. Written informed consent was taken from all patients and all procedures performed in the study were as per the ethical standards of the institutional ethics committee and the 1975 Helsinki declaration and its subsequent amendments or comparable ethical standards. Assessment of systemic inflammation, systemic endotoxemia, tight-junction protein expression in duodenal biopsies and stool microbiota analysis were done prior to intestinal permeability assessment.
Measurement of Intestinal Permeability
Intestinal permeability (IP) was based on estimation of lactulose mannitol ratio (LMR) using nuclear magnetic resonance (NMR) spectroscopy.18 The patients had an overnight fasting for 8 h. After evacuation of bladder, they were administered the test solution containing 5 g of lactulose and 2 g of mannitol orally. No food or drink other than water was allowed till the completion of the test. All the urine passed in the subsequent 5 h was collected in a plastic can of 2.5 L capacity containing 20% chlorhexidine as preservative. The total volume of urine was noted and aliquots of the collected urine were stored at –20°C until analysis. The collected urine was then analyzed using NMR spectroscopy to measure the concentration of lactulose and mannitol.
Serum Anti-endotoxin Antibodies
Endotoxemia was assessed as a surrogate marker indirectly by measuring by IgG anti-endotoxin antibodies and IgM anti-endotoxin antibodies. The antibodies were measured using the EndoCAb® ELISA kit (Hycult Biotechnology, Netherlands). This serum-based assay is a solid-phase sandwich enzyme-linked immunosorbent assay (ELISA) and detects and quantifies antibodies against endotoxin as an indirect marker of systemic endotoxemia. Anti-endotoxin core antibodies are captured by solid phase antigen which is then bound to peroxidase conjugated antihuman IgG antibody which then reacts with tetramethylbenzidine (TMB) for spectrophotometric assessment. A decreased titer suggests increased binding of systemic endotoxin to the antibody.
Systemic Inflammatory Markers
Tumor necrosis factor-alpha (TNF-α) and interleukin-1 (IL-1) were measured by ELISA (Elabscience, China) as per the manufacturer's instructions. The tests used a sandwich-ELISA with biotinylated detection antibodies specific for TNF-α and IL-1, respectively. In the presence of the cytokine being measured by a specific kit, biotinylated detection antibody and avidin-horseradish peroxidase (HRP) conjugate, the well turned blue. The reaction was terminated by addition of the stop solution and the well-turned yellow. The optical density (OD) was proportional to the concentration of cytokine being measured and was assessed spectrophotometrically at a wavelength of 450 ± 2 nm.
16 S rRNA Gene Library Preparation and Gene Sequencing
Microbiota analysis was done in 10 patients of NAFLD, ALD and disease free controls. Fresh stool was collected from subjects and were immediately stored at −80°C at the time of enrolment in the study. At the time of analysis, stool was thawed and DNA extraction was done using Power Soil DNA Isolation Kit (MoBio, Carlsbad, CA, USA).
DNA quality-check: Extracted DNA from the samples were subjected to Nano Drop and GEL Check before being taken for polymerase chain reaction (PCR) amplification. DNA's quality was assessed with the Nano Drop readings of 260/280 at an ∼ value of 1.8–2.
PCR Amplicon quality check. Forty nanogram of extracted DNA was used for amplification in PCR reactions with 16S primers 16F:-5′ AGAGTTTGATCMTGGCTCAG 3′ and 16R:- 5′ TACGGYTACCTTGTTACGACTT. The amplified 16s PCR Product is purified and subjected to GEL Check and Nanodrop QC. The Nano Drop readings of 260/280 at an ∼ value of 1.8–2 is used to determine the DNA's quality.
Nanopore sequencing was performed with 1 μg of DNA template. For each sample, repair and dA-tailing was done using NEBNext End-Repair and NEBNext dA-Tailing modules (New England Biolabs) as per the manufacturer's instructions. Purification was done using AMPure XP bead binding. Sequencing was performed using a MinION device (ONT) on a SpotON FLO-MIN106 flowcell (R9.4) (Oxford Nanopore Technologies).
Bioinformatics Protocol
The cloud-based or local data analysis platform EPI2ME 16S analysis of workflow which allows users to perform genus level identification from single reads with access to base-called files for detailed investigations at the species and subspecies level was used for bioinformatics. The phylogeny analysis of query sequence with the closely related sequence of blast results was performed followed by multiple sequence alignment.
The workflow was designed to BLAST base-called sequence against the National Center for Biotechnology Information (NCBI) 16S bacterial database, which contains 16S sequences from different organisms. Each read was classified based on percentage coverage and identity.
Expression of Tight Junction Proteins (TJP) on Immunohistochemistry
Biopsies were kept in formalin vials and processed. Briefly, sections cut from tissue blocks were boiled in 0.01 M citrate buffer ethylene-diamine-tetra-acetic acid (EDTA), non-specific antibody binding was blocked, and primary monoclonal antibodies against claudin-3 (1:50) and zonulin-1 (1:400) (Zymed, San Francisco, CA) was used in standard overnight incubation. Universal polymer-based peroxidase-labeled secondary antibody (Envision real system; Dako, Denmark) was used and reaction color was developed using DAB (3,3′-Diaminobenzidine).
Immunohistochemistry (IHC) expression was then analyzed in both the mucosal villous and crypt epithelium and the stain distribution and intensity were subgraded. The stain distribution was subgraded as follows: 0: <10% mucosal epithelial surface positivity, 1+: 11%–20% surface area positivity); 2+: 21%–40% surface area; 3+: 41%–60% surface area; 4+: 61%–80% surface area and 5+: >80% surface area positivity. The staining intensity was categorized as grade 1 (very light stain), grade 3 (stain intensity equal to in-house standardized positive control biopsies), and grade 2 (intermediate intensity). The H-score was calculated separately for mucosal villous and crypt epithelium by multiplying the stain distribution and intensity scores and the overall H-scores ranged from 0 to 15. Laboratory personnel and pathologists performing IHC analyses were blinded to the group assignment and the clinical details of the patients, and all laboratory procedures were performed by the trained personnel in a single laboratory using a standard predefined protocol. All biopsies were read by two independent pathologists who were blinded to the clinical details and were graded in a single sitting, with disagreements in interpretation resolved through mutual consensus.
Outcomes
The outcomes of this study were: (1) the comparison of intestinal permeability (IP), systemic inflammation, and systemic endotoxemia among NAFLD, ALD, and controls; (2) exploratory analysis of expression in duodenal biopsies from NAFLD, ALD, and controls; and (3) exploratory analysis of differences in stool microbiota among NAFLD, ALD, and controls.
Statistical Analysis
The baseline data was recorded as number (%) or mean ± SD/median (Q1-Q3) as appropriate, based on normality of distribution. Baseline parameters were compared using chi-square test/Fischer's Exact test for categorical variables and Student's t-test for continuous variables with normal distribution. Continuous variables with non-normal distribution were compared using independent samples Kruskal–Wallis test. Variables distributed across more than two groups were tested using pairwise comparisons with adjustment for multiple comparisons to identify differences in individual pairs. For all statistical tests, a P value < 0.05 was considered statistically significant.
Box-plots were used to represent differences in different continuous parameters representing IP, bacterial translocation, inflammatory response and gut microbiota among patients with NAFLD, ALD and controls. Variations in gut microbiota at genus level were further compared using principal components analysis, with 3 major principal components represented graphically. The alpha diversity metrices (Shannon diversity indices), beta diversity measure (Principal co-ordinate analysis [PCoA]), and relative abundance of bacteria at phyla, genus and species level were compared among the three groups.
All data was entered using Microsoft Excel 2011 and was analyzed using RStudio. In addition to the base packages in R, ggpubr, readxl and tidyverse packages were used.
Results
A total of 34 patients of NAFLD and 28 patients of ALD were included in this study after evaluating 119 and 117 patients, respectively over the study period (Supplementary Figure-1), in addition to 20 disease free controls. The baseline characteristics of the overall cohort and subgroups are shown in Table 1. The mean age was comparable in all three groups. Patients with ALD were more likely to be male (males: females 27:1, 13:5 and 13:7 in ALD, NAFLD and controls respectively, P < 0.001) while those in NAFLD group had more frequent metabolic syndrome as indicated by higher mean body mass index (BMI) [26.7 ± 4 kg/m2 in NAFLD vs. 23.6 ± 3.7 kg/m2 in ALD; P < 0.001], higher serum triglycerides (median 167 mg/dL (IQR: 117–230) vs. 126 mg/dL (IQR:110–213) in ALD, P = 0.02) and a higher frequency of diabetes mellitus (30.6% vs. 3.6% in ALD; P < 0.001). The ALD group had more advanced liver disease compared to those with NAFLD as indicated by significantly higher serum bilirubin (median 1.2 mg/dL (IQR 0.7–5.4) vs. 0.8 mg/dL (IQR: 0.6–1.3) in NAFLD; P = 0.02) and lower serum albumin (4 g/dL ± 0.8 g/dL vs. 4.8 ± 0.4 g/dL in NAFLD; P < 0.001). The median duration of alcohol consumption and amount of alcohol consumption was 10 (IQR: 5–13) years and 120 (IQR:75–150) grams per day in patients with ALD. Liver biopsy was performed in 31 NAFLD patients. Histological cirrhosis (F4 fibrosis) was seen in none out of 31 patients. Patients with ALD did not undergo biopsy. However, they had a higher liver stiffness on transient elastography than those with NAFLD [median LSM -NAFLD: 7.1 kPa (IQR:5.9–8.9) vs. ALD: 14.3 kPa (IQR:9.6–24), P < 0.001]. Twenty eight out of 31 NAFLD patients (90%) had a non-alcoholic fatty liver disease activity score (NAS)- non-alcoholic Clinical Research Network (CRN) score of ≤4.
Table 1.
Baseline Characteristics of Studied Patients.
| Variable | Group 1 Controls (n = 20) | Group 2 NAFLD (n = 34) | Group 3 ALD (n = 28) |
P value |
|---|---|---|---|---|
| Age (years) | 33.6 ± 10.4 | 38.6 ± 10.7 | 40 ± 7.7 | 0.1 |
| BMI (kg/m2) | 22.4 ± 2 | 26.7 ± 4 | 23.6 ± 3.7 | <0.001 |
| Gender (M: F) | 13:7 | 13:5 | 27:1 | <0.001 |
| Diabetes (n%) | 0 | 11 (30.6) | 1 (3.57%) | <0.001 |
| Hypertension (n%) | 0 | 6 (16.67) | 2 (7.54%) | 0.15 |
| Antidiabetic drugs | 0 | 6 | 0 | na |
| Hemoglobin (g/dL) |
13.6 ± 1.4 | 13.8 ± 1.9 | 12.9 ± 2.2 | 0.18 |
| TLC (1000/ml) | 6.5 ± 1.3 | 7.2 ± 2 | 7.6 ± 2 | 0.12 |
| Platelet (lakh/mL) |
1.6 (1.3–2.3) | 1.6 (1.2–2.2) |
1.5 (1.2–2.3) |
0.80 |
| Urea (mg/dL) | 22.3 ± 6.3 | 23 ± 6.1 | 22.6 ± 8.4 | 0.82 |
| Creatinine (mg/dL) | 0.8 ± 0.2 | 0.8 ± 0.2 | 0.75 ± 0.2 | 0.53 |
| ALT (IU/mL) | 25 (21–34) | 79 (54–105) |
62 (47–94) |
<0.001 |
| AST (IU/mL) | 25 | 46.5 | 80 | <0.001 |
| (17–38) | (39–70) | (58–126) | ||
| ALP (IU/mL) | 197 | 236.5 | 256 | 0.56 |
| (147–256) | (141–297) | (154–312) | ||
| Bilirubin (mg/dL) | 0.7 | 0.8 | 1.2 | 0.02 |
| (0.5–0.8) | (0.6–1.3) | (0.7–5.4) | ||
| Protein (g/dL) | 7.2 ± 0.4 | 7.5 ± 0.4 | 7.5 ± 0.6 | 0.11 |
| Albumin (g/dL) | 4.6 ± 0.3 | 4.8 ± 0.4 | 4 ± 0.8 | <0.001 |
| INR | – | 1 (0.9–1.2) | 1.1 (0.9–1.2) | 0.09 |
| HDL (mg/dL) | 42.9 ± 9.1 | 44.4 ± 21.2 | 37.4 ± 12.1 | 0.23 |
| LDL (mg/dL) | 108 ± 22.8 | 120.6 ± 31.3 | 108.1 ± 26.9 | 0.14 |
| VLDL (mg/dL) | 17.5 | 21 (15–40) | 24 | 0.23 |
| (14–23) | (14–32) | |||
| Cholesterol (mg/dL) | 171.2 ± 32.1 | 185.9 ± 52 | 171.3 ± 41.9 | 0.36 |
| TG (mg/dL) | 105 (87–134) | 167 (117–230) | 126 (110–213) | 0.02 |
| LSM (kPa) | 4.8 (4.3–5.3) | 7.1 (5.9–8.9) | 14.3 (9.6–24) | <0.001 |
| CAP (db/m) | 206.3 ± 33.6 | 304.8 ± 49.8 | 268.1 ± 40.5 | <0.001 |
Data is expressed in Frequency (Percentage), Mean ± SD and Median (inter-quartile range) format. P value comparing the three groups with value < 0.05 has been considered as significant. Abbreviations: ALP, Alkaline Phosphatase; ALT, Alanine transaminases; AST, Aspartate transaminases; BMI, Body mass index; CAP, Controlled attenuation parameter; HDL, High density lipoprotein; LDL, Low density lipoprotein; LSM, Liver stiffness measurement: TLC, Total Leucocyte count; VLDL, Very low density lipoprotein; TG, triglycerides.
Intestinal Permeability
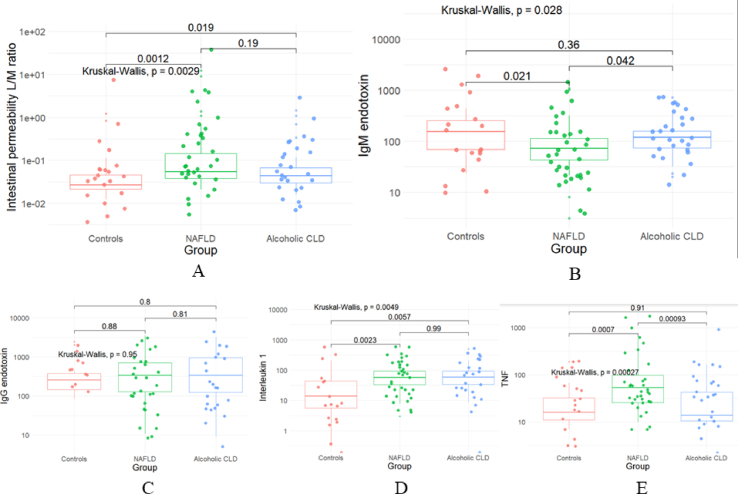
Median lactulose mannitol ratio (LMR) was highest in NAFLD patients [0.054 (IQR: (0.037–0.17)] and lowest in controls [0.027 (0.021–0.045)]; with intermediate value in ALD group [0.043 (0.03–0.068)]. The difference was significant on comparing NAFLD group with controls (P = 0.001) and ALD group with controls (P = 0.019), though the difference between NAFLD and ALD groups was not significant (Table 2, Figure- 1A).
Table 2.
Comparison of Intestinal Permeability, Systemic Endotoxemia, and Systemic Inflammatory Markers Among Studied Patients.
| Parameters | Group 1 Control (n = 20) |
Group 2 NAFLD (n = 34) |
Group 3 ALD (n = 28) | P value | Pairwise comparison |
||
|---|---|---|---|---|---|---|---|
| Group 1 vs. 2 |
Group 1 vs. 3 |
Group 2 vs. 3 |
|||||
| LMR | 0.027 (0.021–0.045) | 0.054 (0.037–0.17) | 0.043 (0.03–0.068) | 0.003 | 0.001 | 0.019 | 0.19 |
| IgM anti-endotoxin antibody (MMU/mL) |
155.3 | 72.9 | 120.6 | 0.028 | 0.021 | 0.36 | 0.042 |
| (23.8–442.9) | (3.2–1089.5) | (20.1–728) | |||||
| IgG anti-endotoxin antibody (GMU/mL) |
138 | 264.4 | 215 | 0.95 | 0.28 | 0.3 | 1 |
| (0–2460.4) | (0–3555.6) | (0–3176.4) | |||||
| TNF-α (pg/mL) | 16.1 (10.8–33.3) | 53.3 (24.5–115) | 12.3 (10.1–42.7) | <0.001 | <0.001 | 0.91 | <0.001 |
| IL-1 (pg/mL) | 13.61 (3.79–43.6) | 57.3 (30.54–95.5) | 58.19 (28.55–92.28) | 0.005 | 0.002 | 0.0057 | 0.99 |
Data is expressed in Mean ± SD and Median (inter-quartile range) format. P < 0.05 is significant. LMR, Lactulose Mannitol ratio; TNF- α, Tumor Necrosis Factor – α; IL-1, Interleukin-1.
Figure 1.
Profile plots representing comparison of A) Intestinal permeability using lactulose mannitol (LMR) and B) systemic endotoxemia using serum IgM anti-endotoxin antibody and C) IgG anti-endotoxin antibody levels and D) systemic inflammatory response by measuring serum IL-1 and E) TNF-α levels among controls, NAFLD and ALD patients.
Systemic Endotoxemia
Median IgM anti-endotoxin antibody levels were lowest in the NAFLD group [72.9 MMU/mL (IQR: 3.2–1089.5)] and highest in the control group [155.3 MMU/mL (IQR: 23.8–442.9)]. The ALD group was found to have intermediate levels [120.6 MMU/mL (IQR: 20.1–728) (Table 2, Figure-1B). The difference was found to be significant on comparing NAFLD group with controls (P = 0.021), as well as on comparing NAFLD group with ALD group (P = 0.042). Though median IgM anti-endotoxin antibody levels were lower in ALD as compared to controls, this was not significant (P = 0.36) [Table 2, Figure-1C]. Median IgG anti-endotoxin levels were non-significantly elevated in patients with NAFLD (Table- 2).
Systemic Inflammatory Markers
Median TNF-α levels were significantly higher in patients with NAFLD [53.3 pg/mL (IQR: 24.5–115)] when compared to both controls [16.1 pg/mL (IQR: 10.8–33.3)] and ALD group [12.3 pg/mL (IQR: 10.1–42.7) (Table 2, Figure-1E). IL-1 levels were also significantly higher in patients with NAFLD [57.3 pg/mL (IQR: 30.5–95.5)] and ALD [58.2 pg/mL (28.6–92.3)] as compared to controls [13.6 pg/mL (3.8–43.6)] (P value = 0.0023 and 0.0057 respectively). However, the levels of IL-1 were comparable between NAFLD and ALD (Table 2, Figure-1D).
Microbiota Analysis
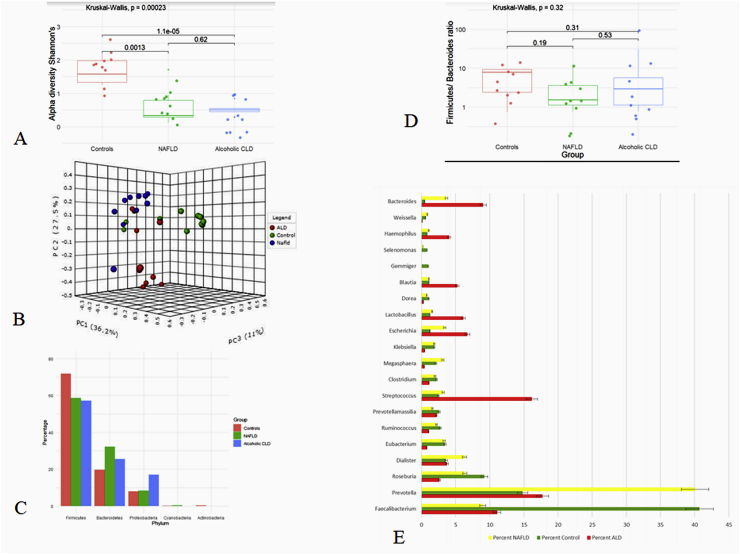
The Shannon alpha diversity index evaluated at a genus level was significantly different among the three groups (P < 0.001) with significant decrease in diversity in patients with NAFLD [0.33 (0.28–0.85)] and ALD [0.50 (0.45–0.55)] as compared to controls [1.57 (1.31–2.08)]. Although there was a trend towards decreased diversity in NAFLD group as compared to ALD group this was not significant (Figure-2A).
Figure 2.
Comparison of the relative abundance at different taxonomic levels among 10 NASH patients, 10 ALD and 10 controls (A) Comparison of Alpha diversity among the three cohorts (Shannon diversity). (B) Principal component analysis showing the overall structure of gut microbiota in the three groups, showing three different clusters with slight overlaps. (C) Abundance variation of the 6 major phyla across the three cohorts (D) Comparison of Bacteroides/Firmicutes ratio across 3 cohorts (E) Comparison of genus composition across the three cohorts.
On Principal Coordinate analysis, the global bacterial composition was significantly different across the three groups, as indicated by distinct clustering with slight overlap on the PCoA graph (PERMANOVA test P < 0.001) (Figure- 2B).
Fecal microbiota was first compared at phyla and genus level among patients with NAFLD, ALD and controls (Figure- 2E, Figure- 3). At phyla level, both patients with NAFLD and ALD had lower proportion of Firmicutes, lowest being in the ALD group. This was accompanied by a rise in Bacteroidetes levels in the NAFLD and ALD group with the levels being the highest in the NAFLD group. Proportion of Proteobacteria was significantly higher in patients with ALD when compared to those with NAFLD and controls.
Figure 3.
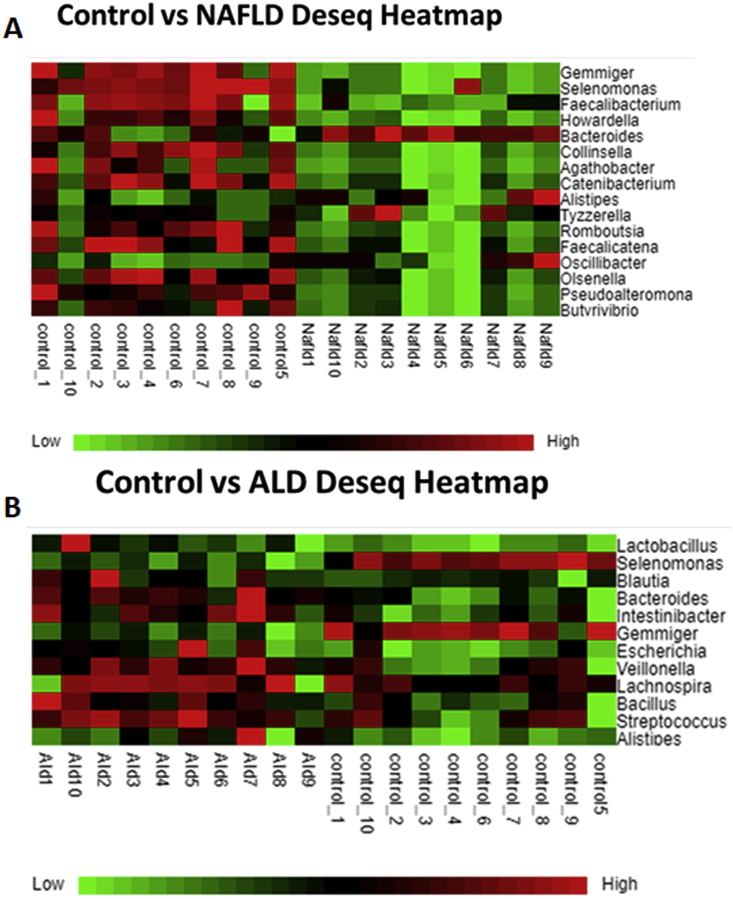
Heatmaps showing the comparison of relative abundance of various genera between A) NAFLD and controls and B) ALD and controls.
Specific Enrichment of Microbiota in Different Groups
On comparing patients of NAFLD with controls, a significant enrichment of genus Prevotella, Alistepes and Bacteroides (members of Bacteroidetes phylum) along with a few members of Firmicutes phylum such as Tyzzerella and Oscillibacter. Members of the Firmicutes phyla consisting of genus Faecalibacterium and Gemmiger (family Ruminococcaceae), Agathobacter, Butyrivibrio and Faecalicatena (Lachnospiraceae), Selenomonas, Howardella, Catenibacterium, Ramboutsia were depleted in NASH group, along with Collinsella and Olsenella, both members of phylum Actinobacteria.
On comparing microbiota in ALD group with controls (Figure-3B), ALD patients had enrichment of genus Escherichia and Haemophilus, both members of phyla Proteobacteria, Alistepes and Bacteroides of phyla Bacteroidetes, and certain selective genera of Firmicute phylum such as Blautia and Lachnospira (family Lachnospiraceae), Veillonella (Veillonellaceae), Intetstinibacter (Peptostreptococci) and Streptococci, Lactobacillus and Bacillus. Members of the Firmicutes phyla consisting of genus Faecalibacterium, Ruminococcus and Gemmiger (Ruminococcaceae), Roseburia (Lachnospiraceae) and Selenomonas were depleted in ALD group as compared to controls.
On comparing NAFLD group with ALD group directly, NAFLD group was found to be enriched in several genera of the Firmicutes phyla namely Oscillibacter and Ruminiclostridium (family Ruminococcaceae), Megasphaera (Viollenallecae), Tyzerella (Lachnospiraceae) and Eubacter (Eubacteriaceae) and Suturella member of family Proteobacteria. ALD group demonstrated an enrichment of the genus Burkholderia a member of Proteobacteria phylum and Prevotella, Streptococcus, Blautia and Intestinibacter, all being members of the Firmicute phyla. Though Escherichia and Haemophilus were represented more in ALD group as compared to NAFLD, this did not reach statistical significance.
TJP Expression in Duodenal Biopsy
The expression of Claudin-3 in duodenal crypts (H score) was found to be significantly lower in NAFLD group [2 (IQR: 0–4)] and demonstrated a strong trend towards decreased expression in ALD group [3 (IQR: 3–5)] when compared to controls [6 (IQR: 4–8)] (P value = 0.033 and 0.054 respectively) (Table 3). Claudin-3 expression was also evaluated in duodenal villi and though it was found to be lower in both NAFLD [2 (IQR 0–3)] and ALD [3 (IQR 2–4)] as compared to controls [6 (IQR 4–8)], none of these differences were statistically significant (Table 3).
Table 3.
Comparison of Tight Junction Protein Expression Using H Score in Duodenal Mucosa in Study Subjects.
| Tight junction protein | Group 1 Control (n = 20) | Group 2 NAFLD (n = 34) | Group 3 ALD (n = 28) | P value | Pairwise comparison |
||
|---|---|---|---|---|---|---|---|
| Group 1 vs. 2 | Group 1 vs. 3 | Group 2 vs. 3 | |||||
| Claudin-3 villus | 6 (4–8) | 2 (0–4) | 3 (3–5) | 0.15 | 0.098 | 0.16 | 0.32 |
| Claudin-3 crypt | 6 (4–8) | 2 (0–3) | 3 (2–4) | 0.043 | 0.033 | 0.054 | 0.28 |
| Zonulin-1 villus | 11 (6–12) | 5 (0–8) | 8 (8–14) | 0.043 | 0.051 | 0.94 | 0.026 |
| Zonulin-1 crypt | 3 (1–10) | 2 (1–4) | 5 (3–12) | 0.4 | 0.48 | 0.51 | 0.2 |
There was a decreased expression of Zo-1 in duodenal villus, with median H score levels being lowest in NAFLD group [5 (IQR 0–8)], intermediate in ALD [8 (IQR 8–14)] and highest in controls [11 (IQR 6–12)], these differences being statistically significant (Table 3). Zo-1 expression in duodenal crypt was highest in the ALD group [5 (IQR 3–12)] and lowest in the NAFLD group [2 (IQR 1–4)] with intermediate values in controls [3 (IQR 1–10)], none of the differences being statistically significant (Table 3).
Discussion
Our study focused on demonstration of abnormalities of gut-barrier, an important component of gut–liver axis. The result of our study demonstrates that dysfunction of gut-barrier exists even in early stages of NAFLD. Among components of this dysfunction, patients with precirrhotic (early) NAFLD had a higher degree of endotoxemia (Median IgM anti-endotoxin antibody levels were lowest in the NAFLD group), reduced expression of duodenal mucosal TJPs like Zo-1 and claudin-3 and a higher systemic inflammatory flux with a similar increase in intestinal permeability when compared to those with ALD. These abnormalities were seen despite a presence of more advanced disease in ALD. Both NAFLD and ALD groups also had a significantly altered intestinal microbiota at both phylum and genus level even at this stage of the disease with an increase in the enrichment of Proteobacteria in ALD and increase in SCFA producing bacteria in NAFLD.
In health, the liver is constantly exposed to relatively small amounts of bacterial components such as endotoxin, also a lipopolysaccharide (LPS) and bacterial mRNA, which are rapidly neutralized.22 An increase in IP is potentially associated with an increase in bacterial translocation, endotoxemia, which act on the toll-like receptor 4 (TLR4) receptors to activate resident liver mononuclear cells and subsequent trigger an inflammatory cascade ultimately translating into hepatocyte necrosis, apoptosis, and induction of liver fibrosis.23, 24, 25 Our study demonstrated significantly increased intestinal permeability in both NAFLD and ALD which was in line with previous studies of IP in these patient groups. We used nuclear magnetic resonance (NMR) spectroscopy for measuring concentration of lactulose and mannitol in urine samples, which is currently the technique of choice for simultaneous detection of multiple metabolites in complex biological mixtures in view of its rapid, quantitative and high-throughput technique with highly reproducible results.18 Our study also demonstrated increased bacterial product translocation in patients with NAFLD as evidenced by lower IgM anti-endotoxin antibodies. The EndoCab IgG and IgM assays measure endotoxin core antibodies and were chosen as they reflect the immune response against persistent endotoxin exposure and are more stable then LPS in serum which has a short half-life (as low as 2 h) and may not be a true reflection of portal LPS.26 It is interesting that IgM and IgG endotoxin levels show trends in opposite directions. This might be explained by relative distribution of IgG and IgM antibodies in the intravascular and extravascular compartment. Depletion of circulating anti-endotoxin antibody levels as the LPS–antibody complex is cleared may be more marked in the case of IgM antibodies than IgG antibodies, as only 20% of IgG is in circulation and will be supplemented from the 80% extravascular pool of IgG.27 Though previous studies have demonstrated increased endotoxemia in patients with ALD when compared to controls,28 our study only showed a trend in that direction, the values of IgG and IgM anti-endotoxin being statistically similar to controls.
The increased systemic inflammation demonstrated in both groups may well have reflected inflammatory activity going on in liver than a component of gut-barrier dysfunction in both entities. However, exclusion of alcoholic hepatitis in ALD and absence of histological non-alcoholic steatohepatitis (NASH) in more than 90% of patients suggest that this is unlikely and systemic inflammation demonstrated in this study appears a likely manifestation of gut-barrier dysfunction.
Exploratory analysis of tight junction proteins (TJPs), claudin-3 and zonulin-1 in duodenal mucosa also demonstrated a trend towards decreased expression in both NAFLD and ALD patients. Claudin-3 and zonulins and occludin protein 1 are sealing TJPs which help in maintaining the integrity of the gut-epithelial barrier. Similar to the changes in intestinal permeability, the decrease in expression of TJPs was more pronounced in patients with NAFLD when compared to ALD. This indicates that decreased expression of TJP could be one mechanism which contributes to increased intestinal permeability (IP) and further liver injury.
Gut dysbiosis may be an important contributor to alterations in gut mucosal barrier, and our study indeed showed significant differences in stool microbiota composition among patients with NASH, ALD, and controls at different taxonomic levels. First, there was decreased representation of Firmicutes and increased representation of Bacteroides in both NAFLD and ALD cohorts as compared to controls, with the Firmicutes: Bacteroides ratio being lowest in patients with NAFLD. Our results were in line with other studies in patients with NAFLD17,29,30 although our findings were in contradiction with previous studies in ALD which indicated a decrease in both Firmicutes and Bacteroides.13,14,31 This was perhaps due to the different study population in these studies which consisted of patients with alcoholic hepatitis unlike our population where all patients with ALD had compensated advanced disease with none having alcoholic hepatitis.
There is gut dysbiosis in both NAFLD and advanced ALD. However, short-chain fatty acid (SCFA)-producing bacteria of Ruminococcacceae and Lachnospiraceae family were further decreased in ALD group while Proteobacteria were found to be increased, hinting towards increased representation of lipopolysaccharide (LPS)-producing bacteria and a decrease in bacteria producing enterocyte protective SCFA in the same.
Overall, this comprehensive assessment of gut-barrier dysfunction with inclusion of luminal abnormalities (microbiome changes), structural components (decreased expression of tight junction proteins on gut mucosa), functional component (increased IP), increased bacterial translocation (higher endotoxemia) and consequent systemic inflammation suggest that gut–liver axis derangement occurs even before the onset of cirrhosis and its complications in NAFLD. While biopsy proven precirrhotic stage of NAFLD was included, the same cannot be said about ALD as reflected by their significantly higher LSM values. On comparison between two etiologies, there was similar increase in IP with demonstration of increased endotoxemia, inflammatory flux and reduced expression of TJPs in early stages of NAFLD. Our findings suggest that gut-barrier dysfunction in prominent in NAFLD even before the onset of cirrhosis compared with ALD despite presence of more advanced liver disease in ALD. One potential implication of the present findings may be the role that gut-barrier dysfunction plays in the pathogenesis of two diseases. Alcohol is metabolized in liver and its products may promote a dual hit with both direct hepatic injury and a second injury to the gut integrity which may be responsible for pathogenesis of disease. The increased dysfunction of gut-barrier with consequent increased endotoxemia and systemic inflammation may suggest this to be a triggering event in the pathogenesis of NAFLD with consequent hepatic inflammation. However, this remains to be explored in further studies. Another aspect that remains unexplored is the role of endogenous alcohol on gut-barrier dysfunction in patients with NAFLD. Endogenous alcohol production in NAFLD has been related to ethanol producing bacteria which can subsequently trigger dysfunction in gut-barrier, endotoxemia, hepatic inflammation and fibrosis.29 While the study lacks real-time structural assessment of gut-epithelial and vascular barrier (best seen with electron microscopy), the results of this study sheds light on individual components seldom studied previously in totality in these diseases. However, the lack correlation of the increasing dysfunction of the gut barrier with different stages of NAFLD deter us from ascertaining if these abnormalities truly contribute to progression/pathogenesis of the liver disease.
Our study has several limitations. First, the sample size of the patient population was small and many parameters may not have reached statistical significance. This was essentially a proof-of-concept study demonstrating changes in gut-barrier at early stages of NAFLD. Therefore, no predetermined sample size was estimated for this study. Also, our analysis of stool microbiota was restricted to a small number of patients and controls, and a larger number of subjects would be needed to further assess relationships between changes in IP and endotoxemia with gut microbiota, especially at genus level. These differences may have reflected the inter-individual difference than etiology specific differences. In terms of anthropometry, patient populations were significantly different, at baseline. In terms of anthropometry, population was significantly different. However, patients with NAFLD are more likely to be obese and hence matching is not possible in this regard. Second, we restricted our analysis of intestinal barrier dysfunction to TJPs, which are only one of the many contributors to bacterial translocation, and the role of other players, especially gut immune cells, paracellular pathways and endothelial-vascular barrier were not studied. Our assessment of bacterial translocation was also indirect; via systemic anti-endotoxin antibody levels, which might again be an inadequate representation of actual spillover of microbial products in portal circulation. This may be the reason for no clear correlation between IP and anti-endotoxin antibody levels. Third, we did not study the gut-immunological barrier, hence our assessment of gut-barrier may still be incomplete and requires further studies in this regard. Fourth, patients with ALD had a more advanced liver disease than NAFLD. In the absence of biopsy, patients with ALD were categorized with the use of LSM, which has a modest correlation in ALD with advanced fibrosis. Although, this is a comparison of two heterogeneous population, our results show that despite being at an earlier stage of disease, gut-barrier dysfunction is more pronounced in NAFLD than ALD. Fifth, although we studied the functional surrogate of gut-barrier dysfunction in the form of lactulose mannitol ratio (LMR), a concomitant study of circulating biomarkers like intestinal fatty acid binding protein and zonulin-1 would have allowed to assess this dysfunction in a more comprehensive manner. Sixth, while patients with ALD were abstinent from alcohol at the time of assessment, the same could not be said about the intake of fructose in NAFLD, which itself could lead to these derangements. Seventh, our assessment of systemic inflammation may have been incomplete as we did not study other robust markers like interleukin-6 (IL-6), C-reactive protein (CRP), complement factors, and leptin-binding protein (LBP) levels.
To summarize, our study shows components of gut-barrier and gut dysbiosis to be more pronounced in patients with NAFLD when compared to those with advanced ALD. Whether this translates to progression of disease remains to be investigated with other different aspects of gut liver axis to better understand its contribution to the pathogenesis.
Credit authorship contribution statement
Kanav Kaushal: Data collection, writing of draft and critical revision of draft. Samagra Agarwal: Statistical analysis, writing of draft and critical revision of draft. Sanchit Sharma: Writing of draft and critical revision of draft. Pooja Goswami: Data collection and conductance of procedures. Namrata Singh: Critical revision of draft. Vikas Sachdev: Analysis of systemic inflammatory markers and anti-endotoxin antibodies. Shekhar Poudel: Data collection. Prasenjit Das: Analysis of duodenal biopsy and critical revision of draft. Rajni Yadav: Analysis of duodenal biopsy and critical revision of draft. Dinesh Kumar: Conductance of NMR spectroscopy for LMR. Gaurav Pandey: Conductance of NMR spectroscopy for LMR. Deepak Gunjan: Critical revision of draft. Anoop Saraya: Conceptualization, supervision, critical revision of draft, administrative support and vouching for integrity of manuscript.
Conflicts of interest
The authors have none to declare.
Acknowledgment
We would like to acknowledge Dr. Saurabh Kedia Assistant Professor, Department of Gastroenterology, AIIMS, New Delhi for review of the bioinformatics part of the manuscript.
Funding
None.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.jceh.2022.01.006.
Appendix A. Supplementary data
The following is the supplementary data to this article:
References
- 1.Albillos A., de Gottardi A., Rescigno M. The gut-liver axis in liver disease: pathophysiological basis for therapy. J Hepatol. 2020;72:558–577. doi: 10.1016/j.jhep.2019.10.003. [DOI] [PubMed] [Google Scholar]
- 2.Mu Q., Kirby J., Reilly C.M., Luo X.M. Leaky gut as a danger signal for autoimmune diseases. Front Immunol. 2017;8:598. doi: 10.3389/fimmu.2017.00598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Spadoni I., Zagato E., Bertocchi A., et al. A gut-vascular barrier controls the systemic dissemination of bacteria. Science. 2015;350:830–834. doi: 10.1126/science.aad0135. [DOI] [PubMed] [Google Scholar]
- 4.MacFie J. Current status of bacterial translocation as a cause of surgical sepsis. Br Med Bull. 2005;71:1–11. doi: 10.1093/bmb/ldh029. [DOI] [PubMed] [Google Scholar]
- 5.Zihni C., Mills C., Matter K., Balda M.S. Tight junctions: from simple barriers to multifunctional molecular gates. Nat Rev Mol Cell Biol. 2016;17:564–580. doi: 10.1038/nrm.2016.80. [DOI] [PubMed] [Google Scholar]
- 6.Jiang W., Wu N., Wang X., et al. Dysbiosis gut microbiota associated with inflammation and impaired mucosal immune function in intestine of humans with non-alcoholic fatty liver disease. Sci Rep. 2015;5:8096. doi: 10.1038/srep08096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Miele L., Valenza V., La Torre G., et al. Increased intestinal permeability and tight junction alterations in nonalcoholic fatty liver disease. Hepatology. 2009;49:1877–1887. doi: 10.1002/hep.22848. [DOI] [PubMed] [Google Scholar]
- 8.Tranah T.H., Edwards L.A., Schnabl B., Shawcross D.L. Targeting the gut-liver-immune axis to treat cirrhosis. Gut. 2021;70:982–994. doi: 10.1136/gutjnl-2020-320786. [DOI] [PubMed] [Google Scholar]
- 9.Such J., Guardiola J.V., de Juan J., et al. Ultrastructural characteristics of distal duodenum mucosa in patients with cirrhosis. Eur J Gastroenterol Hepatol. 2002;14:371–376. doi: 10.1097/00042737-200204000-00006. [DOI] [PubMed] [Google Scholar]
- 10.Assimakopoulos S.F., Tsamandas A.C., Tsiaoussis G.I., et al. Altered intestinal tight junctions' expression in patients with liver cirrhosis: a pathogenetic mechanism of intestinal hyperpermeability. Eur J Clin Invest. 2012;42:439–446. doi: 10.1111/j.1365-2362.2011.02609.x. [DOI] [PubMed] [Google Scholar]
- 11.Haderer M., Neubert P., Rinner E., et al. Novel pathomechanism for spontaneous bacterial peritonitis: disruption of cell junctions by cellular and bacterial proteases. Gut. 2021;11 doi: 10.1136/gutjnl-2020-321663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Garcia-Tsao G., Lee F.Y., Barden G.E., Cartun R., West A.B. Bacterial translocation to mesenteric lymph nodes is increased in cirrhotic rats with ascites. Gastroenterology. 1995;108:1835–1841. doi: 10.1016/0016-5085(95)90147-7. [DOI] [PubMed] [Google Scholar]
- 13.Jacobs J.P., Dong T.S., Agopian V., et al. Microbiome and bile acid profiles in duodenal aspirates from patients with liver cirrhosis: the Microbiome, Microbial Markers and Liver Disease Study. Hepatol Res. 2018;48:1108–1117. doi: 10.1111/hepr.13207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen Y., Yang F., Lu H., et al. Characterization of fecal microbial communities in patients with liver cirrhosis. Hepatology. 2011;54:562–572. doi: 10.1002/hep.24423. [DOI] [PubMed] [Google Scholar]
- 15.Turnbaugh P.J., Ley R.E., Mahowald M.A., Magrini V., Mardis E.R., Gordon J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444:1027–1031. doi: 10.1038/nature05414. [DOI] [PubMed] [Google Scholar]
- 16.Loomba R., Seguritan V., Li W., et al. Gut microbiome based metagenomic signature for non-invasive detection of advanced fibrosis in human nonalcoholic fatty liver disease. Cell Metabol. 2017;25:1054–1062. doi: 10.1016/j.cmet.2017.04.001. e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mouzaki M., Comelli E.M., Arendt B.M., et al. Intestinal microbiota in patients with nonalcoholic fatty liver disease. Hepatology. 2013;58:120–127. doi: 10.1002/hep.26319. [DOI] [PubMed] [Google Scholar]
- 18.Kumar D., Pandey G., Bansal D., et al. NMR-based urinary profiling of lactulose/mannitol ratio used to assess the altered intestinal permeability in acute on chronic liver failure (ACLF) patients. Magn Reson Chem. 2017;55:289–296. doi: 10.1002/mrc.4525. [DOI] [PubMed] [Google Scholar]
- 19.null Shalimar, Kumar R., Rout G., et al. Body mass index-based controlled attenuation parameter cut-offs for assessment of hepatic steatosis in non-alcoholic fatty liver disease. Indian J Gastroenterol. 2020;39:32–41. doi: 10.1007/s12664-019-00991-2. [DOI] [PubMed] [Google Scholar]
- 20.Crabb D.W., Im G.Y., Szabo G., Mellinger J.L., Lucey M.R. Diagnosis and treatment of alcohol-associated liver diseases: 2019 practice guidance from the American association for the study of liver diseases. Hepatology. 2020;71:306–333. doi: 10.1002/hep.30866. [DOI] [PubMed] [Google Scholar]
- 21.Keshavarzian A., Fields J.Z., Vaeth J., Holmes E.W. The differing effects of acute and chronic alcohol on gastric and intestinal permeability. Am J Gastroenterol. 1994;89:2205–2211. [PubMed] [Google Scholar]
- 22.Brandl K., Kumar V., Eckmann L. Gut-liver axis at the frontier of host-microbial interactions. Am J Physiol Gastrointest Liver Physiol. 2017;312:G413–G419. doi: 10.1152/ajpgi.00361.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Csak T., Ganz M., Pespisa J., Kodys K., Dolganiuc A., Szabo G. Fatty acid and endotoxin activate inflammasomes in mouse hepatocytes that release danger signals to stimulate immune cells. Hepatology. 2011;54:133–144. doi: 10.1002/hep.24341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nicoletti A., Ponziani F.R., Biolato M., et al. Intestinal permeability in the pathogenesis of liver damage: from non-alcoholic fatty liver disease to liver transplantation. World J Gastroenterol. 2019;25:4814–4834. doi: 10.3748/wjg.v25.i33.4814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Uesugi T., Froh M., Arteel G.E., Bradford B.U., Thurman R.G. Toll-like receptor 4 is involved in the mechanism of early alcohol-induced liver injury in mice. Hepatology. 2001;34:101–108. doi: 10.1053/jhep.2001.25350. [DOI] [PubMed] [Google Scholar]
- 26.Kitabatake H., Tanaka N., Fujimori N., et al. Association between endotoxemia and histological features of nonalcoholic fatty liver disease. World J Gastroenterol. 2017;23:712–722. doi: 10.3748/wjg.v23.i4.712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pirofsky B., Campbell S.M., Montanaro A. Individual patient variations in the kinetics of intravenous immune globulin administration. J Clin Immunol. 1982;2:7S–14S. doi: 10.1007/BF00918361. [DOI] [PubMed] [Google Scholar]
- 28.Parlesak A., Schäfer C., Schütz T., Bode J.C., Bode C. Increased intestinal permeability to macromolecules and endotoxemia in patients with chronic alcohol abuse in different stages of alcohol-induced liver disease. J Hepatol. 2000;32:742–747. doi: 10.1016/S0168-8278(00)80242-1. [DOI] [PubMed] [Google Scholar]
- 29.Zhu L., Baker S.S., Gill C., et al. Characterization of gut microbiomes in nonalcoholic steatohepatitis (NASH) patients: a connection between endogenous alcohol and NASH. Hepatology. 2013;57:601–609. doi: 10.1002/hep.26093. [DOI] [PubMed] [Google Scholar]
- 30.Boursier J., Mueller O., Barret M., et al. The severity of nonalcoholic fatty liver disease is associated with gut dysbiosis and shift in the metabolic function of the gut microbiota. Hepatology. 2016;63:764–775. doi: 10.1002/hep.28356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Smirnova E., Puri P., Muthiah M.D., et al. Fecal microbiome distinguishes alcohol consumption from alcoholic hepatitis but does not discriminate disease severity. Hepatology. 2020;72:271–286. doi: 10.1002/hep.31178. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.