Abstract
OBJECTIVE:
β-lactams are the cornerstone of empiric and targeted antibiotic therapy for critically ill patients. Recently, there have been calls to use β-lactam therapeutic drug monitoring (TDM) within 24–48 hours after the initiation of therapy in critically ill patients. In this article, we review the dynamic physiology of critically ill patients, β-lactam dose response in critically ill patients, the impact of pathogen minimum inhibitory concentration (MIC) on β-lactam TDM, and pharmacokinetics in critically ill patients. Additionally, we highlight available clinical data to better inform β-lactam TDM for critically ill patients.
DATA SOURCES:
We retrospectively analyzed patients admitted for sepsis or septic shock at a single academic medical center who were treated with β-lactam antibiotics.
STUDY SELECTION:
Indexed studies in PubMed in English language were selected for review on topics relative to critical care physiology, β-lactams, pharmacokinetics/pharmacodynamics, TDM, and antibiotic susceptibility.
DATA EXTRACTION:
We reviewed potentially related studies on β-lactams and TDM and summarized their design, patients, and results. This is a synthetic, nonsystematic, review.
DATA SYNTHESIS:
In the retrospective analysis of patients treated with β-lactam antibiotics, approximately one-third of patients received less than 48 hours of β-lactam therapy. Of those who continued beyond 48 hours, only 13.7% had patient-specific factors (augmented renal clearance, fluid overload, morbid obesity, and/or surgical drain), suggesting a potential benefit of β-lactam TDM.
CONCLUSIONS:
These data indicate that a strategy of comprehensive β-lactam TDM for critically ill patients is unwarranted as it has not been shown yet to improve patient-oriented outcomes. This review demonstrates that β-lactam TDM in the ICU, while laudable, layers ambiguous β-lactam exposure thresholds upon uncertain/unknown MIC data within a dynamic, unpredictable patient population for whom TDM results will not be available fast enough to significantly affect care. Judicious, targeted TDM for those with risk factors for β-lactam over- or underexposure is a better approach but requires further study. Clinically, choosing the correct antibiotic and dosing β-lactams aggressively, which have a wide therapeutic index, to overcome critical illness factors appears to give critically ill patients the best likelihood of survival.
Keywords: beta-lactam antibiotics, critically ill, intensive care unit, pharmacodynamics, pharmacokinetics, therapeutic drug monitoring
Of all the antimicrobial classes, β-lactams are the most widely used and successful agents. They remain the backbone of any hospital antimicrobial armamentarium. Resistance emergence is often countered through β-lactam protection with β-lactamase inhibitors and/or novel dosing with high dose, prolonged, or continuous infusion (CI). The clinical implementation of these approaches is well-founded through rigorous basic, translational, and clinical research, demonstrating patient benefit and workflow integration ease. A recent concept to further improve patient outcomes with β-lactams is the use of therapeutic drug monitoring (TDM) in specific populations, primarily critically ill patients. In this review, we examine the evidence for β-lactam TDM, limitations for its clinical implementation, and critically ill populations in whom potential benefit requires further study.
THE ICU PATIENT: A CASE STUDY IN VARIABILITY
Daily, 70% of ICU patients are administered at least one antibiotic, most frequently β-lactams (1). The physiology of the sepsis patient is complex and dynamic and along with it comes changes in β-lactam pharmacokinetics (PK). Patients in the ICU with sepsis are likely to receive noninvasive or invasive mechanical ventilatory support, intermittent or continuous renal replacement therapy (CRRT), circulatory support via pharmacologic or mechanical means, and/or extracorporeal membrane oxygenation (ECMO). It is common for patients to receive several of these organ support therapies simultaneously. Alternatively, the ICU patient could be on the precipice of requiring these therapeutic techniques or at the other end of the spectrum: toward organ system recovery such that the supportive maneuvers are no longer necessary.
A position paper from several critical care and infectious diseases societies recommends routine TDM should be performed for β-lactam antibiotics (in addition to other antimicrobials) (2). Justification for this recommendation stems from both the risk of underexposure (efficacy) and overexposure (safety) in critically ill patients due to altered PK. The therapeutic target the panel recommends for β-lactams is a trough concentration (Cmin) greater than the minimum inhibitory concentration (MIC; 100% TFREE > MIC) for intermittent or prolonged infusions and a steady-state concentration greater than the MIC for CI’s (Css > MIC). This recommendation begs the question: will β-lactam TDM produce data that are robust and actionable given the rapid fluctuations in the septic patient’s condition and correspondingly improve outcomes beyond current practice?
DOSE RESPONSE OF β-LACTAMS: THE OPTIMAL TIME ABOVE MIC TO IMPROVE CRITICALLY ILL PATIENT OUTCOMES IS UNKNOWN
β-lactams are time-dependent killing antibiotics, but their PK/pharmacodynamics (PD) target varies by infusion strategy and β-lactam class.(3–5) In vitro and animal models indicate that the amount of time a β-lactam concentration exceeds the MIC is important (4, 6–9). Preclinical data suggest β-lactam kill rate is maximized when steady-state drug concentrations are maintained at four to eight times the MIC for CI’s and maximized when serum concentrations exceed the MIC (TFREE > MIC) for 40–70% of the dosing interval of intermittent infusions (5). During intermittent infusion, β-lactam concentrations greater than or equal to 4 times the MIC do not appear to increase the rate or extent of bacterial killing (10). In fact, excessive β-lactam concentrations may induce a paradoxical reduction in bacterial killing known as the “Eagle Effect” (10, 11). Although the molecular mechanisms of this paradoxical effect vary by antibiotics, excessive β-lactam concentration may induce β-lactamase and/or alter target-site binding efficiency (12). The β-lactam Eagle Effect has been well described in animal models, but the clinical evidence of suboptimal outcomes with aggressive dosing remains circumstantial (13).
Curiously—provided the reproducible in vitro data—human trials maximizing concentration above MIC have not necessarily been associated with improved clinical outcomes, and data supporting improved clinical outcomes when achieving a β-lactam concentration greater than or equal to 4 times the MIC are sparse (14–18). Notably, many patients in observational β-lactam TDM studies have extensive and variable TFREE > MIC. Thus, the optimal clinical TFREE > MIC parameter is unknown. No research has explored optimization of β-lactam dosing for the treatment of highly susceptible, low MIC pathogens, which account for the majority of pathogens in patients, including those in the ICU (19). One possible explanation for the lack of supportive data is the wide therapeutic index of β-lactam antibiotics, specifically that these antibiotics work well at a variety of doses and serum concentrations when the MIC is low.
The observation that PK/PD efficacy indices established using in vitro models, animal models, and/or through various predictive modeling approaches may not translate clinically applies to other antibiotics as well. Levofloxacin has failed to meet established Cmax:MIC ratios against Streptococcus pneumoniae, but patients with pneumonia improve (20). Clinical cure is also possible for bacterial meningitis with CNS antibiotic concentrations at or below the MIC (21, 22). This may be due to a reliance on bloodstream infections when developing PK/PD models and antibiotic efficacy indices; these data may not extrapolate appropriately to other sites of infection. Conversely, higher exposures are needed for those with impaired host immunity. β-lactam efficacy in vivo is due in part to postantibiotic leukocyte enhancement, which may be diminished or absent in immunocompromised patients (23). A recent multicenter, prospective observational study found vancomycin PK/PD target attainment in patients with methicillin-resistant Staphylococcus aureus bacteremia was not associated with a lower risk of treatment failure, although other observational studies suggest otherwise (24–27).
PATHOGEN MINIMUM INHIBITORY CONCENTRATION AND ITS IMPACT ON TDM
The central argument behind β-lactam TDM is optimization of the PK/PD parameter time above organism MIC (4, 6–9). The underlying denominator, MIC, is a significant factor in this response; however, there are several limitations to the reported MIC’s clinical utility when attempting to personalize a patient’s β-lactam regimen.
By nature, microorganisms are not precise; they vary widely—and rapidly—in their metabolism, response to stressors (including antibiotics), and their environment. Some have recently questioned the “true MIC” in vivo where host and bacterial factors may differ from the in vitro environment (28, 29). In addition, several studies indicate that the inoculum has important implications for antibiotic activity, with high inocula, biofilm-producing cultures reducing susceptibility beyond what can be replicated in the clinical microbiology laboratory (30–32). For resistant infections and/or for patients who are critically ill and deteriorating, clinicians could select alternative antibiotic therapy (e.g., a different β-lactam and/or a non- β-lactam) instead of trying to optimize the current regimen with TDM. When no alternative antibiotic exists, which is currently unlikely given several new agents available for multi-drug resistant pathogens, then β-lactam TDM is a reasonable approach (33).
There are many different MIC testing platforms available, and there is noted variability among them. Although determining susceptible, intermediate, or resistance (i.e., within defined breakpoints) is consistent among the many automated platforms, the reported MIC among these can vary by up to two dilution factors (34, 35). Other contributing factors to MIC variance include interstrain and interlaboratory differences (36). Recent studies suggest that antibiotic activity in vitro may be media- and/or condition-dependent for β-lactams (37, 38). One notable example of this is with metallo-β-lactamase (MBL) producing Enterobacterales, which require one or two zinc atoms at their active site to enable hydrolysis of the β-lactam ring and avert its activity (39). Standard antimicrobial susceptibility testing medium contains high levels of zinc, allowing MBL producers to maximally exert carbapenem hydrolysis. However, in zinc-depleted media, more analogous to human zinc concentrations, these organisms are more sensitive to carbapenems and better predict in vivo outcomes (40). Conversely, Morrison et al (41) demonstrated among respiratory and blood Pseudomonas aeruginosa isolates recovered from pediatric patients that some had lower aminoglycoside and minocycline MICs in traditional medium than in serum-based media. The impact of these newly identified conditional differences in MIC on the patient PK/PD response has not been identified, but this is an area of further interest to study.
Finally, there remains a lack of MIC data in real time or even at all in some patients. In the ICU, patients may either not have an identifiable organism, or an MIC is reported greater than 48 hours after initiation of therapy. Several studies have shown that early, appropriate antimicrobial therapy (within the first 24 hr) is crucial for success in infected, critically ill patients (42–46). Tailoring therapy or dose optimization after the first few days appears to have limited added value (47). In this critical time period, using the correct β-lactam antibiotic and dosing aggressively provides the best opportunity to achieve PK/PD targets, even for pathogens with higher MICs. During this crucial intervention period, clinicians also have at their disposal validated PK/PD modeling software designed to dose-optimize antibiotics with consideration given to conditions associated with altered PK, infection source, and the suspected/documented pathogen (48). However, when done without any patient-specific β-lactam concentration input, this approach relies completely on the population PK model, and many models have not yet been validated in the critically ill population of interest. By including patient concentrations, Bayesian estimation allows for such simulations to rely less on the models and more on individual patients. One could argue that obtaining patient β-lactam serum concentrations would further benefit PK/PD simulation. Yet, these concentrations are not available in real time at most centers, at least in the United States. As an alternative to individual serum levels, such software can also be used in conjunction with local MIC data to provide a more personalized β-lactam dosing strategy (49). Although these computer modeling programs may rely on organism epidemiologic cutoff values, local antibiogram data, or the patient’s past cultures from previous infections, this approach offers a method to address dose-optimization needs more quickly, efficiently, and less invasively than TDM in the critical care population.
β-LACTAM PHARMACOKINETIC VARIATION IN THE ICU
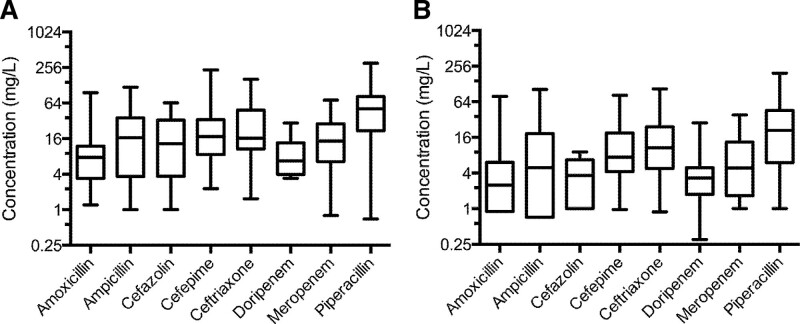
There is widespread pharmacokinetic variation among critically ill patients due to the complex pathophysiology and clinical manifestations of sepsis. The TDM process proposes all patients in the heterogeneous critically ill population need to achieve the same PK/PD target to ensure a positive clinical outcome. A single, multicenter point-prevalence study of serum antibiotic exposure in critically ill patients (Defining Antibiotic Levels in Intensive Care Unit Patients [DALI]) is often cited to justify TDM in this population (50). Of DALI subjects with a pathogen recovered (73%), only 25% had an MIC reported (17% of the entire cohort). When unavailable, the MIC was defined using the European Committee on Antimicrobial Susceptibility Testing MIC90 data. For those without a pathogen (27%), the highest MIC for susceptible bacteria was used. This “worst case scenario” approach for missing MIC data biases the results toward a lower likelihood of PK/PD target attainment, especially as only 33% of patients received prolonged antibiotic infusions—likely lower than to be expected in current critical care practice based on European data; however, contemporary U.S. data area lacking (51, 52). More importantly, only 25% of DALI subjects had β-lactam serum concentrations obtained within 24 hours of antibiotic initiation, which is the most important time to optimize antibiotic and dose (50, 53, 54). Despite these limitations, pharmacokinetic variation was nominal. The median Cmin concentrations were approximately 16, 4, and 6 mg/L for piperacillin, meropenem, and cefepime, respectively (Fig. 1), at, above, and near the Clinical Laboratory Standards Institute breakpoints for each of these agents for presumably the entire dosing interval against Gram-negative pathogens, respectively (55). As noted previously, the position paper on TDM in critically ill adults recommends targeting a β-lactam Cmin greater than the MIC (2). These data from the DALI study do not suggest there is meaningful β-lactam PK/PD variation in critically ill patients necessitating TDM. Still, these data compared serum concentrations with a target PK/PD goal. It remains prudent to determine if β-lactam TDM improves patient outcomes.
Figure 1.
β-lactam serum concentration data from the DALI study. The boxplot of antibiotic concentrations observed at 50% (A) and 100% (B) of the dosing interval. Median, interquartile range, and range are presented. The y-axes are presented on a log2 scale. Used with permission from Clinical Infectious Diseases (50). DALI = defining antibiotic levels in intensive care unit patients.
CLINICAL IMPACT AND LOGISTICAL CHALLENGES OF β-LACTAM TDM IMPLEMENTATION
TDM appears to consistently increase the likelihood of β-lactam PK/PD target attainment in observational studies (56–59). Likewise, two small sample randomized controlled trials (RCTs) have demonstrated significantly improved β-lactam target attainment in patients allocated to TDM-based dosing compared with standardized, traditional dosing without TDM (60, 61). Neither of the RCTs demonstrated improved clinical outcomes, though this was not either study’s primary intent. The benefit of TDM on clinical outcomes thus far is limited to retrospective, observational analyses: in which sufficient TFREE > MIC was associated with clinical cure, microbial eradication, and suppression of resistance as well as with hospital survival among patients with certain modified Acute Physiology and Chronic Health Evaluation scores (62, 63). It is imperative to acknowledge these types of data but not rely on them exclusively. To confirm the clinical value of β-lactam TDM, we must rely upon RCT data. Recently, the first randomized trial in 249 patients to evaluate outcomes of β-lactam TDM (piperacillin/tazobactam [TZP]) was published. This is the largest study to date, examining the impact of TDM on clinical outcomes, and the majority of patients were categorized as having septic shock. Patients were randomized to receive 13.5 g of TZP per day with or without daily TDM, and the groups were well matched; TZP doses were reduced for those with renal impairment. Putting the lower than recommended total daily TZP dose for sepsis and shock (13.5 g/d instead of 18 g/d) aside, the primary outcome selected—mean daily Sequential Organ Failure Assessment (SOFA) score over 10 days’ time—is not an outcome typically associated with adequate antibiotic exposure in a direct manner. There was no difference in the mean SOFA score with and without TDM (7.9 points; 95% CI, 7.1–8.7; and 8.2 points, 95% CI, 7.5–9.0, respectively; p = 0.39). However, looking at secondary outcomes studied by the authors, which are directly related to antibiotic exposure, namely, clinical and microbiologic cures, there was also no difference between the groups (odds ratio [OR], 1.9; 95% CI, 0.5–6.2; p = 0.30 and OR, 2.4; 95% CI, 0.7–7.4; p = 0.12 for clinical and microbiologic cures, respectively). Curiously, the authors found no difference in the mean daily SOFA score—a mortality prediction score, yet heavily discuss the observed, nonstatistically significant, numeric difference in 28-day mortality between the two groups, which favored TDM (21.6% vs 25.8% without TDM; p = 0.44), as a reason for TDM optimism and further study (64). If TDM did not impact the mean change in the SOFA score, a severity of illness score that predicts mortality, it seems implausible that TDM led to the observed, numeric reduction in mortality.
Beyond these clinical and methodological observations from the Therapeutic drug monitoring-based dose optimisation of piperacillin/tazobactam to improve outcome in patients with sepsis (TARGET) trial, the logistical observations with TDM from this study are striking and reinforce the arguments advanced in the current article. First, patient enrollment was challenging: more patients were excluded (n = 305) than enrolled and randomized due to difficulties in obtaining TZP within 24 hours of sepsis or the initial dose. Second, the rate of TZP discontinuation with each subsequent day of study enrollment reinforces how few patients with sepsis or septic shock will remain on antibiotic therapy or their initially prescribed antibiotic(s) after a few days’ time, rendering any initial TDM measurements inconsequential (we buttress this observation with local, point-prevalence data later in the article). The median duration of TZP therapy was less than 5 days in both groups. Additionally, only 65.6% of patients had a causative pathogen identified, and for those receiving TDM, the median time to MIC data was 2 days. Thus, during the initial days of antibiotic exposure in the ICU, there appear to be limited opportunities for PK/PD optimization based on MIC data. Finally, 53.9% of those in the TDM group had a TZP dose adjustment, yet there were no appreciable between-group differences in the mean total daily TZP dose in grams (10.3 ± 5.6 and 9.8 ± 2.5 with and without TDM, respectively; p = 0.12) nor the median TZP serum concentration (mg/L) up to day 10 (74.9 [60.6–91] and 79 [43.8–143.6] with and without TDM, respectively; p = 0.38). This could be a result of ineffective or heterogeneous clinician response to TDM results. Further, TZP dose and serum level data suggest that β-lactam TDM may not impact antibiotic exposure and/or that clinicians in the ICU are currently unprepared to respond to TDM levels appropriately. Although the rate of serum level target attainment was higher with TDM than without (37.3% and 14.6%, respectively; p < 0.001), it was still low based on the effort expended on TDM during the study. This suggests an inappropriate serum level target or the inability of TDM to appreciably impact target attainment. Either way, the results of TARGET, in which the majority of patients had septic shock not sepsis, reinforce our current arguments against universal β-lactam TDM in the ICU. A second RCT in critically ill patients is ongoing. As we although we await further results, it is important to discuss the logistical challenges clinicians face when attempting to implement β-lactam TDM into everyday practice (65).
Currently, TDM assays are not available at most U.S. hospitals, though may be more commonly available in other parts of the world (e.g., Europe and Australia), and initiation and maintenance costs are prohibitively expensive. “Send out” TDM levels are available with an approximately 8–24-hour turnaround time (TAT) from sample receipt (66). However, there may be variation in “send out” TDM level availability across the globe. In the United States, these tests can range from $65 to $200 (U.S. dollars; Atlantic Diagnostic Laboratories, 66). Naturally, lengthy TAT limits TDM impact (67, 68). This is especially pertinent in critically ill patients where acute changes in organ function that affect drug clearance and frequent coadministration of therapies affecting drug distribution (e.g., vasopressors, inotropes, and IV fluids) render single historically collected (in the critically ill sense) β-lactam samples meaningless. Additionally, antibiotic therapy changes frequently in the ICU; thus, serum levels obtained may be rendered inconsequential following antibiotic therapy changes. “Send out” tests also require considerable supervision and coordination by local staff, coupled with different PK/PD targets for the multiple β-lactam infusion strategies previously mentioned.
Given the procedural differences across studies, it becomes a challenge to implement a β-lactam TDM program at the local level. Future TDM studies should prioritize broadly generalizable TDM methods and examine patient-oriented outcomes as the primary end point, not just “target attainment.” Additionally, an institution with on-site TDM specialization retrospectively found early β-lactam TDM has better outcomes than delayed TDM (62). The lack of readily available, on-site TDM at nearly every healthcare institution should encourage a collective investment in research that will allow for real-time β-lactam TDM assays at an affordable cost.
PATIENT POPULATIONS IN WHOM TDM COULD BENEFIT
A prospective cohort study revealed that approximately 75% of ICU patients receiving β-lactams required an adjustment of the initially selected dose to achieve a pharmacodynamic target of 100% TFREE > 4–5 × MIC (trough concentrations at 4–5× the concentration of the MIC of the known or suspected pathogen) (69). Focusing specifically on two commonly administered antibiotics in the ICU, TZP, and meropenem, roughly 50% of patients required a dose increase, 25% required a decrease in dose, and 25% had doses maintained. A deeper dive into the study reveals most dose increases were performed in patients with augmented renal clearance (ARC) and patients with surgical drains who did not require renal replacement therapy (RRT). ARC is defined as a creatinine clearance above 130 mL/min/1.73 m2 and can be associated with younger age and polytrauma, including those with those with burns or significant wounds (70). ARC has been associated with subtherapeutic β-lactam concentrations in observational studies (71, 72). However, there have been conflicting results in studies that have attempted to associate ARC with worsened clinical outcomes (73–76).
Other populations for whom β-lactam TDM could be of benefit include those on RRT, ECMO, and those at the extremes of body weight (e.g., obesity). However, similar to ARC, this presumption is based on the known β-lactam PK variation in these populations not any documented impact of β-lactam TDM on clinical outcomes for these patients. Obesity is associated with antibiotic pharmacokinetic variation in the ICU (77). Although obese patients may benefit from β-lactam TDM, PK, and TDM, interventional data for these patients remain sparse. Obese patients were underrepresented in the TARGET study: the mean body mass index (BMI) in which was 27 ± 7 kg/m2 (78). The number of obese patients continues to increase worldwide, making this a key population for which to better understand the potential role of TDM. Absent compelling data focused on patient-oriented outcomes it is challenging to recommend TDM for all obese patients in the ICU given the large number of patients this would entail. CRRT is also associated with antibiotic PK variability and subtherapeutic exposures (79). As this is a smaller group of ICU patients at high risk for mortality, it is reasonable to consider β-lactam TDM for those on CRRT. ECMO can also impact β-lactam PK, but the impact appears to be drug-specific rather than an antibiotic class effect (e.g., all β-lactams) (80). It is also worth noting that ECMO is not as common in the ICU as CRRT or obesity. The ever-changing nature of ECMO circuitry also makes application of available data to one’s practice challenging. Additionally, we do not believe target β-lactam serum levels for those on ECMO are known, nor are β-lactam serum levels for those not on ECMO appropriate for those on ECMO.
The recent multisociety TDM position paper recommends one sample, trough concentration (Cmin) monitoring 24–48 hours after the initiation of therapy, with incorporation of the laboratory result into dosing software to perform Bayesian estimation and regimen adjustment (if needed) followed by repeat TDM within 1–2 days to assess for target attainment (2). Additional monitoring is warranted when there is evidence of PK changes significant enough to potentially alter drug concentrations. Although we disagree with this recommendation for all ICU patients, we believe there may be smaller groups of ICU patients at risk for β-lactam PK variation for whom TDM could be of benefit. The RCT of TZP TDM by Hagel et al (78) described above perhaps could provide some insight. However, without additional subgroup analyses, it is hard to identify unique patients from TARGET for whom TDM may have been more beneficial (e.g., ECMO and RRT). Judicious, targeted TDM for those with risk factors for β-lactam over- or underexposure is a better approach, including those with ARC, those with burns or polytrauma, those with surgical drains and fluid overload, those on CRRT, and those on ECMO and/or morbid obesity. Prospective studies focusing on patient-oriented outcome should be conducted to determine the value of beta-lactam TDM for these vulnerable patients. Until then, TDM remains a clinical strategy with a potential for benefit in the populations.
REAL-WORLD IDENTIFICATION OF PATIENTS FOR WHOM TDM COULD BENEFIT
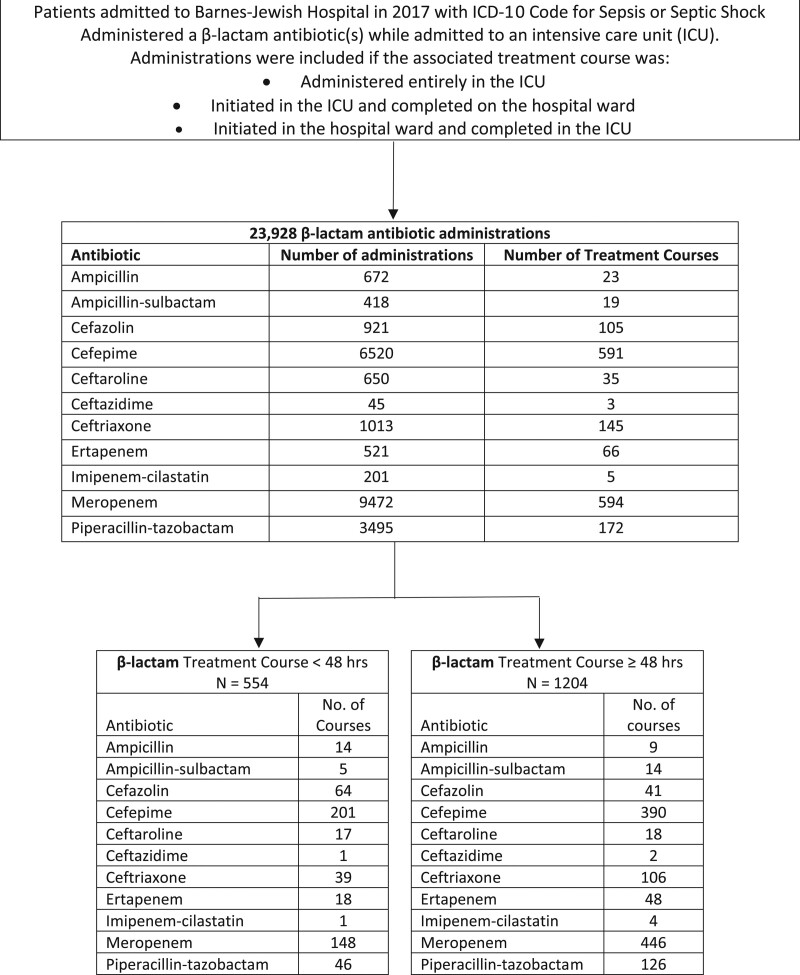
Contemporary information about the frequency of pharmacokinetic alterations observed in patients with septic shock can aid the development of strategies to implement a β-lactam TDM program that optimizes resource allocation. We conducted a cross-sectional, point-prevalence evaluation of all patients admitted to Barnes-Jewish Hospital (an academic medical center in St. Louis, MO) in 2017 for sepsis or septic shock based on International Classification of Diseases (ICD), 9th Edition and 10th Edition codings. (Washington University institutional review board [IRB] number 201153035 and University of Health Sciences and Pharmacy IRB number 201631). Prevalence of treatment courses in which patients were observed to have altered pharmacokinetic variables alone, or in combinations of two PK variables, was the main outcome measure. Pharmacokinetic variables associated with increased risk of underdosing was defined estimated glomerular filtration rate (eGFR) greater than or equal to 120 mL/min based on Cockcroft-Gault calculation, fluid overload, morbid obesity (BMI >40), and presence of a surgical drain (81). Variables associated with increased risk of overdosing included eGFR less than 20 mL/min or receipt of CRRT (69). A total of 1,758 treatment courses associated with 23,928 administrations of various β-lactam antibiotics in the ICU were observed (Fig. 2). The proportion of treatment courses less than 48 hours in duration was 30.7% (n = 554). The frequency of treatment courses greater than 48 hours duration in which there was one altered PK variable associated with risk of underdosing was 38.9% (n = 468) and overdosing 24.1% (n = 290) (Table 1). Further, estimates of the risk for underdosing when considering two patient-specific factors, in combination (surgical drain placement, fluid overload, morbid obesity, ARC, or normal eGFR 60–119 mL/min in combination), were observed in 13.7% (n = 165) (Table 1). Altogether, this snapshot refutes the need for β-lactam TDM in all patients as roughly one-third of treatment courses are less than 48 hours duration and under 15% of courses occur in patients with multiple PK variable alterations. Considering the healthcare personnel time required to identify these patients, followed by the acquisition and analysis of drug levels and the TDM workflow integration into a complicated and rapidly changing patient care environment requiring other urgent interventional care, it is difficult to justify universal β-lactam TDM in the ICU without definitive data for outcome improvement.
Figure 2.
Flow diagram. ICD-10 = International Classification of Diseases, 10th Edition.
TABLE 1.
Frequency Matrix of Factors That Could Influence Antibiotic Pharmacokinetics in β-Lactam Treatment Courses Greater Than or Equal to 48 hr (n = 1,204).
| Vasopressor | Inotrope | Mechanical Circulation | eGFR <20 | eGFR 21-59 | CRRT | eGFR 60-119 | eGFR ≥120 | Fluid Overload | Morbid Obesity | Surgical Drain | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Vasopressor | 836 | 241 | 33 | 42 | 296 | 205 | 274 | 12 | 139 | 105 | 103 |
| Inotrope | 259 | 31 | 9 | 91 | 91 | 66 | 1 | 48 | 27 | 27 | |
| Mechanical circulation | 36 | 0 | 13 | 6 | 17 | 0 | 4 | 2 | 2 | ||
| eGFR < 20 mL/min | 61a (5.1) | 0 | 0 | 0 | 0 | 9 | 11 | 13 | |||
| eGFR 21–59 mL/min | 443 | 0 | 0 | 0 | 60 | 60 | 50 | ||||
| CRRT | 229a (19.0) | 0 | 0 | 38 | 31 | 19 | |||||
| eGFR 60–119 mL/min | 421 | 0 | 46b (3.8) | 37b (3.1) | 45b (3.7) | ||||||
| eGFR ≥ 120 mL/min | 38c (3.2) | 2b (0.2) | 4b (0.3) | 4b (0.3) | |||||||
| Fluid overload | 154c (12.8) | 5b (0.4) | 22b (1.8) | ||||||||
| Morbid obesity | 143c (11.9) | 16b (1.3) | |||||||||
| Surgical drain | 133c (11.0) | ||||||||||
CRRT = continuous renal replacement therapy, eGFR = estimated glomerular filtration rate.
aOne pharmacokinetics (PK) variable with increased risk of overdosing.
bCombination of two PK variables with increased risk of underdosing.
cOne PK variable with increased risk of underdosing.
All data presented as number or number (%).
Number with percentage indicates the patients at risk for underdosing who could potentially benefit from β-lactam therapeutic drug monitoring. For example, the number of patients with eGFR greater than or equal to 120 mL/min in combination with fluid overload was two.
CAN TDM REDUCE TOXICITY FOR WIDE THERAPEUTIC INDEX DRUGS?
An argument can be made for TDM wholistically for its potential to reduce drug toxicity. This has been used for antimicrobials including vancomycin, aminoglycosides, triazole antifungals, and other classes anecdotally with varying results (82–84). However, β-lactam antibiotics are much different from these antibiotics with defined therapeutic concentration ranges. First, β-lactams, for the great benefit to patients, have a wide therapeutic index. For most patients in the ICU, moderate-to-high dose, prolonged infusion β-lactams optimize the PD of these drugs. Second, toxicities that are derived from elevated β-lactams concentrations, for example, neurologic toxicity, occur primarily in patients with preexisting neurologic conditions and those with impaired renal function (85). In the latter, β-lactam doses are reduced according to well-established dosing guidelines. β-lactam antibiotics can be associated with acute kidney injury, and this is often in context with vancomycin (86). It is difficult to parse out the causal differences in this scenario and link to β-lactam exposure thresholds. Finally, β-lactam TDM is founded from serum concentrations, which may be a poor surrogate in an individual patient for either infected tissue concentrations of interest (e.g., lung) or, in the case of toxicity, the CNS. In a pertinent example, observational studies examining the relationship between cefepime exposure and neurotoxicity cite a wide range of serum concentrations associated with neurotoxicity (87–89). The lack of agreement between these datasets suggests a current inability to draw universal conclusions between β-lactam exposure and toxicity in the serum, an observation recently highlighted by Barreto et al (90). Additional studies of serum and site concentrations correlating with toxicity would be highly valuable toward therapeutic toxicodynamic implications with β-lactams.
CONCLUSIONS
β-lactams are a cornerstone of inpatient antibiotic therapy, and no effort should be spared to ensure their prolonged efficacy and safety. This review demonstrates that β-lactam TDM in the ICU, while laudable, layers ambiguous β-lactam exposure thresholds upon uncertain MIC data within a dynamic, unpredictable patient population for whom TDM results will not be available fast enough to significantly affect care. PK/PD is the foundation upon which the best way to dose β-lactams rests. PK/PD led us to prolonged β-lactam infusions based on positive outcome studies. Although not based on the strongest quality of evidence, prolonged β-lactam infusions are now the “standard” in many hospitals and recognized by pharmaceutical regulatory bodies (e.g., the U.S. Food and Drug Administration) as a viable and often preferred method of β-lactam administration due to their improved ability to achieve PK/PD targets compared with intermittent infusions. Prolonged infusions are easier to implement than TDM: once a workflow is established, the process operates without substantial human effort and coordination. PK/PD modeling software has further advanced this application through design of empiric drug dosing for complex patient situations where the risks of subtherapeutic pharmacokinetic exposure exist. Individual β-lactam TDM will require considerable clinician effort to achieve the desired concentration, but, based on the evidence to date, no appreciable improvements in patient-oriented outcomes exist.
In summation, TDM should be goal-driven: to improve efficacy, reduce toxicity risk, and/or prevent resistance. There are few published studies comparing outcomes in those with and without beta-lactam TDM, and findings have not demonstrated a difference. There may be a role for targeted TDM use in select patients most likely to benefit from such data as we have outlined here; however, randomized clinical trial data with patient-oriented outcomes are needed to confirm such a benefit, even in a selected patient population(s) before the logistical and operational effort is put toward such activity. There remain opportunities to improve PK/PD models and increase the likelihood that in vitro and animal methods will translate to patient experience as well as make dose adjustments to drastically reduce antibiotic exposure in highly susceptible infections. For now, those opportunities remain a point of research focus and are not ready for widespread clinical application. PK/PD application to a population shows benefit, but, as we outlined, there are difficulties for such application at the individual patient level. Clinically, choosing the correct antibiotic and dosing appropriately to overcome ARC associated with critical illness appears to give patients the best likelihood of survival. A dynamic, hypermetabolic environment seems to belie the idea of TDM precision. Clinical success at subtherapeutic levels is possible; there is no guarantee that achieving therapeutic or supratherapeutic concentrations improves patient care. Therefore, what is the goal of β-lactam TDM in the ICU? It may be a solution in search of a problem.
Footnotes
Dr. Schulz reports grant funding from Merck. Dr. Micek reports being a coinvestigator on a grant from Merck. Dr. Kollef reports salary support from the Barnes-Jewish Hospital Foundation. Dr. Rose reports research funding from Merck and Paratek and personal consulting fees from Paratek and Visante. Dr. Dilworth has disclosed that he does not have any potential conflicts of interest.
REFERENCES
- 1.Vincent JL, Sakr Y, Singer M, et al. ; EPIC III Investigators: Prevalence and outcomes of infection among patients in intensive care units in 2017. JAMA 2020; 323:1478–1487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Abdul-Aziz MH, Alffenaar JC, Bassetti M, et al. ; Infection Section of European Society of Intensive Care Medicine (ESICM); Pharmacokinetic/pharmacodynamic and Critically Ill Patient Study Groups of European Society of Clinical Microbiology and Infectious Diseases (ESCMID); Infectious Diseases Group of International Association of Therapeutic Drug Monitoring and Clinical Toxicology (IATDMCT); Infections in the ICU and Sepsis Working Group of International Society of Antimicrobial Chemotherapy (ISAC): Antimicrobial therapeutic drug monitoring in critically ill adult patients: A position paper. Intensive Care Med 2020; 46:1127–1153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Craig WA: Pharmacokinetic/pharmacodynamic parameters: Rationale for antibacterial dosing of mice and men. Clin Infect Dis 1998; 26:1–10 [DOI] [PubMed] [Google Scholar]
- 4.Felton TW, Goodwin J, O’Connor L, et al. : Impact of Bolus dosing versus continuous infusion of Piperacillin and Tazobactam on the development of antimicrobial resistance in Pseudomonas aeruginosa. Antimicrob Agents Chemother 2013; 57:5811–5819 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dhaese S, Heffernan A, Liu D, et al. : Prolonged versus intermittent infusion of β-lactam antibiotics: A systematic review and meta-regression of bacterial killing in preclinical infection models. Clin Pharmacokinet 2020; 59:1237–1250 [DOI] [PubMed] [Google Scholar]
- 6.Roberts JA, Abdul-Aziz MH, Davis JS, et al. : Continuous versus intermittent β-lactam infusion in severe sepsis. A meta-analysis of individual patient data from randomized trials. Am J Respir Crit Care Med 2016; 194:681–691 [DOI] [PubMed] [Google Scholar]
- 7.Dulhunty JM, Roberts JA, Davis JS, et al. : Continuous infusion of beta-lactam antibiotics in severe sepsis: A multicenter double-blind, randomized controlled trial. Clin Infect Dis 2013; 56:236–244 [DOI] [PubMed] [Google Scholar]
- 8.Bulik CC, Christensen H, Li P, et al. : Comparison of the activity of a human simulated, high-dose, prolonged infusion of meropenem against Klebsiella pneumoniae producing the KPC carbapenemase versus that against Pseudomonas aeruginosa in an in vitro pharmacodynamic model. Antimicrob Agents Chemother 2010; 54:804–810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Vourli S, Tsala M, Kotsakis S, et al. : Comparison of short versus prolonged infusion of standard dose of meropenem against carbapenemase-producing Klebsiella pneumoniae isolates in different patient groups: A pharmacokinetic-pharmacodynamic approach. J Pharm Sci 2016; 105:1513–1518 [DOI] [PubMed] [Google Scholar]
- 10.Craig WA, Ebert SC: Killing and regrowth of bacteria in vitro: A review. Scand J Infect Dis Suppl 1990; 74:63–70 [PubMed] [Google Scholar]
- 11.Eagle H, Musselman AD: The rate of bactericidal action of penicillin in vitro as a function of its concentration, and its paradoxically reduced activity at high concentrations against certain organisms. J Exp Med 1948; 88:99–131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ikeda Y, Fukuoka Y, Motomura K, et al. : Paradoxical activity of beta-lactam antibiotics against Proteus vulgaris in experimental infection in mice. Antimicrob Agents Chemother 1990; 34:94–97 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Prasetyoputri A, Jarrad AM, Cooper MA, et al. : The Eagle effect and antibiotic-induced persistence: Two sides of the same coin? Trends Microbiol 2019; 27:339–354 [DOI] [PubMed] [Google Scholar]
- 14.Muller AE, Punt N, Mouton JW: Optimal exposures of ceftazidime predict the probability of microbiological and clinical outcome in the treatment of nosocomial pneumonia. J Antimicrob Chemother 2013; 68:900–906 [DOI] [PubMed] [Google Scholar]
- 15.Ariano RE, Nyhlén A, Donnelly JP, et al. : Pharmacokinetics and pharmacodynamics of meropenem in febrile neutropenic patients with bacteremia. Ann Pharmacother 2005; 39:32–38 [DOI] [PubMed] [Google Scholar]
- 16.Muller AE, Punt N, Mouton JW: Exposure to ceftobiprole is associated with microbiological eradication and clinical cure in patients with nosocomial pneumonia. Antimicrob Agents Chemother 2014; 58:2512–2519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li C, Du X, Kuti JL, et al. : Clinical pharmacodynamics of meropenem in patients with lower respiratory tract infections. Antimicrob Agents Chemother 2007; 51:1725–1730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tam VH, McKinnon PS, Akins RL, et al. : Pharmacodynamics of cefepime in patients with Gram-negative infections. J Antimicrob Chemother 2002; 50:425–428 [DOI] [PubMed] [Google Scholar]
- 19.Sader HS, Castanheira M, Mendes RE, et al. : Frequency and antimicrobial susceptibility of Gram-negative bacteria isolated from patients with pneumonia hospitalized in ICUs of US medical centres (2015-17). J Antimicrob Chemother 2018; 73:3053–3059 [DOI] [PubMed] [Google Scholar]
- 20.Amsden GW, Graci DM, Cabelus LJ, et al. : A randomized, crossover design study of the pharmacology of extended-spectrum fluoroquinolones for pneumococcal infections. Chest 1999; 116:115–119 [DOI] [PubMed] [Google Scholar]
- 21.Sakoulas G, Nonejuie P, Kullar R, et al. : Examining the use of ceftaroline in the treatment of Streptococcus pneumoniae meningitis with reference to human cathelicidin LL-37. Antimicrob Agents Chemother 2015; 59:2428–2431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nau R, Sörgel F, Eiffert H: Penetration of drugs through the blood-cerebrospinal fluid/blood-brain barrier for treatment of central nervous system infections. Clin Microbiol Rev 2010; 23:858–883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Turnidge JD: The pharmacodynamics of beta-lactams. Clin Infect Dis 1998; 27:10–22 [DOI] [PubMed] [Google Scholar]
- 24.Lodise TP, Rosenkranz SL, Finnemeyer M, et al. : The emperor’s new clothes: Prospective observational evaluation of the association between initial vancomycin exposure and failure rates among adult hospitalized Patients with Methicillin-resistant Staphylococcus aureus Bloodstream Infections (PROVIDE). Clin Infect Dis 2020; 70:1536–1545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Moise-Broder PA, Forrest A, Birmingham MC, et al. : Pharmacodynamics of vancomycin and other antimicrobials in patients with Staphylococcus aureus lower respiratory tract infections. Clin Pharmacokinet 2004; 43:925–942 [DOI] [PubMed] [Google Scholar]
- 26.Ghosh N, Chavada R, Maley M, et al. : Impact of source of infection and vancomycin AUC0-24/MICBMD targets on treatment failure in patients with methicillin-resistant Staphylococcus aureus bacteraemia. Clin Microbiol Infect 2014; 20:O1098–O1105 [DOI] [PubMed] [Google Scholar]
- 27.Men P, Li HB, Zhai SD, et al. : Association between the AUC0-24/MIC ratio of vancomycin and its clinical effectiveness: A systematic review and meta-analysis. PLoS One 2016; 11:e0146224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nicoloff H, Hjort K, Levin BR, et al. : The high prevalence of antibiotic heteroresistance in pathogenic bacteria is mainly caused by gene amplification. Nat Microbiol 2019; 4:504–514 [DOI] [PubMed] [Google Scholar]
- 29.Dewachter L, Fauvart M, Michiels J: Bacterial heterogeneity and antibiotic survival: understanding and combatting persistence and heteroresistance. Mol Cell 2019; 76:255–267 [DOI] [PubMed] [Google Scholar]
- 30.Zhao C, Wistrand-Yuen P, Lagerbäck P, et al. : Combination of polymyxin B and minocycline against multidrug-resistant Klebsiella pneumoniae: Interaction quantified by pharmacokinetic/pharmacodynamic modelling from in vitro data. Int J Antimicrob Agents 2020; 55:105941. [DOI] [PubMed] [Google Scholar]
- 31.Canovas J, Petitjean G, Chau F, et al. : Expression of CTX-M-15 limits the efficacy of ceftolozane/tazobactam against Escherichia coli in a high-inoculum murine peritonitis model. Clin Microbiol Infect 2020; 26:1416.e5–1416.e9 [DOI] [PubMed] [Google Scholar]
- 32.McNeil JC, Sommer LM, Boyle M, et al. : Cefazolin inoculum effect and methicillin-susceptible Staphylococcus aureus osteoarticular infections in children. Antimicrob Agents Chemother 2020; 64:e00703–e00720 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Talbot GH, Jezek A, Murray BE, et al. ; Infectious Diseases Society of America: The Infectious Diseases Society of America’s 10 × ‘20 initiative (10 new systemic antibacterial agents US Food And Drug Administration approved by 2020): Is 20 × ‘20 a possibility? Clin Infect Dis 2019; 69:1–11 [DOI] [PubMed] [Google Scholar]
- 34.Sader HS, Fritsche TR, Jones RN: Accuracy of three automated systems (MicroScan WalkAway, VITEK, and VITEK 2) for susceptibility testing of Pseudomonas aeruginosa against five broad-spectrum beta-lactam agents. J Clin Microbiol 2006; 44:1101–1104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhou M, Wang Y, Liu C, et al. : Comparison of five commonly used automated susceptibility testing methods for accuracy in the China Antimicrobial Resistance Surveillance System (CARSS) hospitals. Infect Drug Resist 2018; 11:1347–1358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Mouton JW, Meletiadis J, Voss A, et al. : Variation of MIC measurements: The contribution of strain and laboratory variability to measurement precision. J Antimicrob Chemother 2018; 73:2374–2379 [DOI] [PubMed] [Google Scholar]
- 37.Kubicek-Sutherland JZ, Heithoff DM, Ersoy SC, et al. : Host-dependent induction of transient antibiotic resistance: A prelude to treatment failure. EBioMedicine 2015; 2:1169–1178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ersoy SC, Heithoff DM, Barnes L, 5th, et al. : Correcting a fundamental flaw in the paradigm for antimicrobial susceptibility testing. EBioMedicine 2017; 20:173–181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Palzkill T: Metallo-β-lactamase structure and function. Ann N Y Acad Sci 2013; 1277:91–104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Abdelraouf K, Reyes S, Nicolau DP: The paradoxical in vivo activity of β-lactams against metallo-β-lactamase-producing Enterobacterales is not restricted to carbapenems. J Antimicrob Chemother 2021; 76:684–691 [DOI] [PubMed] [Google Scholar]
- 41.Morrison JM, Chojnacki M, Fadrowski JJ, et al. : Serum-associated antibiotic tolerance in pediatric clinical isolates of pseudomonas aeruginosa. J Pediatric Infect Dis Soc 2020; 9:671–679 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ibrahim EH, Sherman G, Ward S, et al. : The influence of inadequate antimicrobial treatment of bloodstream infections on patient outcomes in the ICU setting. Chest 2000; 118:146–155 [DOI] [PubMed] [Google Scholar]
- 43.Kollef MH, Sherman G, Ward S, et al. : Inadequate antimicrobial treatment of infections: A risk factor for hospital mortality among critically ill patients. Chest 1999; 115:462–474 [DOI] [PubMed] [Google Scholar]
- 44.Zilberberg MD, Shorr AF, Micek ST, et al. : Hospitalizations with healthcare-associated complicated skin and skin structure infections: Impact of inappropriate empiric therapy on outcomes. J Hosp Med 2010; 5:535–540 [DOI] [PubMed] [Google Scholar]
- 45.Vardakas KZ, Rafailidis PI, Konstantelias AA, et al. : Predictors of mortality in patients with infections due to multi-drug resistant Gram negative bacteria: The study, the patient, the bug or the drug? J Infect 2013; 66:401–414 [DOI] [PubMed] [Google Scholar]
- 46.Vazquez-Guillamet C, Scolari M, Zilberberg MD, et al. : Using the number needed to treat to assess appropriate antimicrobial therapy as a determinant of outcome in severe sepsis and septic shock. Crit Care Med 2014; 42:2342–2349 [DOI] [PubMed] [Google Scholar]
- 47.Zilberberg MD, Shorr AF, Micek ST, et al. : Antimicrobial therapy escalation and hospital mortality among patients with health-care-associated pneumonia: A single-center experience. Chest 2008; 134:963–968 [DOI] [PubMed] [Google Scholar]
- 48.Bulik CC, Bader JC, Zhang L, et al. : PK-PD Compass: Bringing infectious diseases pharmacometrics to the patient’s bedside. J Pharmacokinet Pharmacodyn 2017; 44:161–177 [DOI] [PubMed] [Google Scholar]
- 49.Landmesser KB, Clark JA, Burgess DS: Time above all else: Pharmacodynamic analysis of β-lactams in critically ill patients. J Clin Pharmacol 2022; 62:479–485 [DOI] [PubMed] [Google Scholar]
- 50.Roberts JA, Paul SK, Akova M, et al. ; DALI Study: DALI: defining antibiotic levels in intensive care unit patients: Are current β-lactam antibiotic doses sufficient for critically ill patients? Clin Infect Dis 2014; 58:1072–1083 [DOI] [PubMed] [Google Scholar]
- 51.Buyle FM, Decruyenaere J, De Waele J, et al. : A survey of beta-lactam antibiotics and vancomycin dosing strategies in intensive care units and general wards in Belgian hospitals. Eur J Clin Microbiol Infect Dis 2013; 32:763–768 [DOI] [PubMed] [Google Scholar]
- 52.Liebchen U, Paal M, Scharf C, et al. : The ONTAI study - a survey on antimicrobial dosing and the practice of therapeutic drug monitoring in German intensive care units. J Crit Care 2020; 60:260–266 [DOI] [PubMed] [Google Scholar]
- 53.Crass RL, Rodvold KA, Mueller BA, et al. : Renal dosing of antibiotics: Are we jumping the gun? Clin Infect Dis 2019; 68:1596–1602 [DOI] [PubMed] [Google Scholar]
- 54.Kumar A, Roberts D, Wood KE, et al. : Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med 2006; 34:1589–1596 [DOI] [PubMed] [Google Scholar]
- 55.CLSI. Performance Standards for Antimicrobial Susceptibility Testing, 31st Edition. CLSI Supplement M100. Wayne, PA, Clinical and Laboratory Standards Institute, 2021. Available at: https://clsi.org/standards/products/microbiology/documents/m100/. Accessed November 22, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wong G, Briscoe S, McWhinney B, et al. : Therapeutic drug monitoring of β-lactam antibiotics in the critically ill: Direct measurement of unbound drug concentrations to achieve appropriate drug exposures. J Antimicrob Chemother 2018; 73:3087–3094 [DOI] [PubMed] [Google Scholar]
- 57.Machado AS, Oliveira MS, Sanches C, et al. : Clinical outcome and antimicrobial therapeutic drug monitoring for the treatment of infections in acute burn patients. Clin Ther 2017; 39:1649–1657.e3 [DOI] [PubMed] [Google Scholar]
- 58.Fournier A, Eggimann P, Pagani JL, et al. : Impact of the introduction of real-time therapeutic drug monitoring on empirical doses of carbapenems in critically ill burn patients. Burns 2015; 41:956–968 [DOI] [PubMed] [Google Scholar]
- 59.Fratoni AJ, Nicolau DP, Kuti JL: A guide to therapeutic drug monitoring of β-lactam antibiotics. Pharmacotherapy 2021; 41:220–233 [DOI] [PubMed] [Google Scholar]
- 60.Fournier A, Eggimann P, Pantet O, et al. : Impact of real-time therapeutic drug monitoring on the prescription of antibiotics in burn patients requiring admission to the intensive care unit. Antimicrob Agents Chemother 2018; 62:e01818–e01817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.De Waele JJ, Carrette S, Carlier M, et al. : Therapeutic drug monitoring-based dose optimisation of piperacillin and meropenem: A randomised controlled trial. Intensive Care Med 2014; 40:380–387 [DOI] [PubMed] [Google Scholar]
- 62.Al-Shaer MH, Rubido E, Cherabuddi K, et al. : Early therapeutic monitoring of β-lactams and associated therapy outcomes in critically ill patients. J Antimicrob Chemother 2020; 75:3644–3651 [DOI] [PubMed] [Google Scholar]
- 63.Miglis C, Rhodes NJ, Kuti JL, et al. : Defining the impact of severity of illness on time above the MIC threshold for cefepime in Gram-negative bacteraemia: A ‘Goldilocks’ window. Int J Antimicrob Agents 2017; 50:487–490 [DOI] [PubMed] [Google Scholar]
- 64.Vincent JL, de Mendonça A, Cantraine F, et al. : Use of the SOFA score to assess the incidence of organ dysfunction/failure in intensive care units: Results of a multicenter, prospective study. Working group on “sepsis-related problems” of the European Society of Intensive Care Medicine. Crit Care Med 1998; 26:1793–1800 [DOI] [PubMed] [Google Scholar]
- 65.Abdulla A, Ewoldt TMJ, Hunfeld NGM, et al. : The effect of therapeutic drug monitoring of beta-lactam and fluoroquinolones on clinical outcome in critically ill patients: The DOLPHIN trial protocol of a multi-centre randomised controlled trial. BMC Infect Dis 2020; 20:57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Atlantic Diagnostic Laboratories. Anti-Infective Therapeutic Drug Monitoring. 2022. Available at: https://atlanticdiagnosticlaboratories.com/wp-content/uploads/2020/12/Ai-TDM-406-Sell-Sheet-12172020p.pdf. Accessed June 20, 2022. [Google Scholar]
- 67.Schoenenberger-Arnaiz JA, Ahmad-Diaz F, Miralbes-Torner M, et al. : Usefulness of therapeutic drug monitoring of piperacillin and meropenem in routine clinical practice: A prospective cohort study in critically ill patients. Eur J Hosp Pharm 2020; 27:e30–e35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sandaradura I, Wojciechowski J, Marriott DJE, et al. : Model-optimized fluconazole dose selection for critically ill patients improves early pharmacodynamic target attainment without the need for therapeutic drug monitoring. Antimicrob Agents Chemother 2021; 65:e02019–e02020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Roberts JA, Ulldemolins M, Roberts MS, et al. : Therapeutic drug monitoring of beta-lactams in critically ill patients: Proof of concept. Int J Antimicrob Agents 2010; 36:332–339 [DOI] [PubMed] [Google Scholar]
- 70.Bilbao-Meseguer I, Rodríguez-Gascón A, Barrasa H, et al. : Augmented renal clearance in critically ill patients: A systematic review. Clin Pharmacokinet 2018; 57:1107–1121 [DOI] [PubMed] [Google Scholar]
- 71.De Waele JJ, Lipman J, Akova M, et al. : Risk factors for target non-attainment during empirical treatment with β-lactam antibiotics in critically ill patients. Intensive Care Med 2014; 40:1340–1351 [DOI] [PubMed] [Google Scholar]
- 72.Carlier M, Carrette S, Roberts JA, et al. : Meropenem and piperacillin/tazobactam prescribing in critically ill patients: Does augmented renal clearance affect pharmacokinetic/pharmacodynamic target attainment when extended infusions are used? Crit Care 2013; 17:R84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Burnham JP, Micek ST, Kollef MH: Augmented renal clearance is not a risk factor for mortality in Enterobacteriaceae bloodstream infections treated with appropriate empiric antimicrobials. PLoS One 2017; 12:e0180247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Udy AA, Dulhunty JM, Roberts JA, et al. ; BLING-II Investigators; ANZICS Clinical Trials Group: Association between augmented renal clearance and clinical outcomes in patients receiving β-lactam antibiotic therapy by continuous or intermittent infusion: A nested cohort study of the BLING-II randomised, placebo-controlled, clinical trial. Int J Antimicrob Agents 2017; 49:624–630 [DOI] [PubMed] [Google Scholar]
- 75.Huttner A, Von Dach E, Renzoni A, et al. : Augmented renal clearance, low β-lactam concentrations and clinical outcomes in the critically ill: An observational prospective cohort study. Int J Antimicrob Agents 2015; 45:385–392 [DOI] [PubMed] [Google Scholar]
- 76.Claus BO, Hoste EA, Colpaert K, et al. : Augmented renal clearance is a common finding with worse clinical outcome in critically ill patients receiving antimicrobial therapy. J Crit Care 2013; 28:695–700 [DOI] [PubMed] [Google Scholar]
- 77.Hites M, Taccone FS: Optimization of antibiotic therapy in the obese, critically ill patient. Réanimation 2015; 24:278–294 [Google Scholar]
- 78.Hagel S, Bach F, Brenner T, et al. : Effect of therapeutic drug monitoring-based dose optimization of piperacillin/tazobactam on sepsis-related organ dysfunction in patients with sepsis: A randomized controlled trial. Intensive Care Med 2022; 48:311–321 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Roberts DM, Roberts JA, Roberts MS, et al. ; RENAL Replacement Therapy Study Investigators: Variability of antibiotic concentrations in critically ill patients receiving continuous renal replacement therapy: A multicentre pharmacokinetic study. Crit Care Med 2012; 40:1523–1528 [DOI] [PubMed] [Google Scholar]
- 80.Hahn J, Choi JH, Chang MJ: Pharmacokinetic changes of antibiotic, antiviral, antituberculosis and antifungal agents during extracorporeal membrane oxygenation in critically ill adult patients. J Clin Pharm Ther 2017; 42:661–671 [DOI] [PubMed] [Google Scholar]
- 81.Pařízková RČ, Martínková J, Havel E, et al. : Impact of cumulative fluid balance on the pharmacokinetics of extended infusion meropenem in critically ill patients with sepsis. Crit Care 2021; 25:251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Rybak MJ, Le J, Lodise TP, et al. : Therapeutic monitoring of vancomycin for serious methicillin-resistant Staphylococcus aureus infections: A revised consensus guideline and review by the American Society of Health-system Pharmacists, the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the Society of Infectious Diseases Pharmacists. Clin Infect Dis 2020; 71:1361–1364 [DOI] [PubMed] [Google Scholar]
- 83.Maglio D, Nightingale CH, Nicolau DP: Extended interval aminoglycoside dosing: From concept to clinic. Int J Antimicrob Agents 2002; 19:341–348 [DOI] [PubMed] [Google Scholar]
- 84.Ashbee HR, Barnes RA, Johnson EM, et al. : Therapeutic drug monitoring (TDM) of antifungal agents: Guidelines from the British Society for Medical Mycology. J Antimicrob Chemother 2014; 69:1162–1176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Chow KM, Hui AC, Szeto CC: Neurotoxicity induced by beta-lactam antibiotics: From bench to bedside. Eur J Clin Microbiol Infect Dis 2005; 24:649–653 [DOI] [PubMed] [Google Scholar]
- 86.Avedissian SN, Pais GM, Liu J, et al. : Piperacillin-tazobactam added to vancomycin increases risk for acute kidney injury: Fact or fiction? Clin Infect Dis 2020; 71:426–432 [DOI] [PubMed] [Google Scholar]
- 87.Boschung-Pasquier L, Atkinson A, Kastner LK, et al. : Cefepime neurotoxicity: Thresholds and risk factors. A retrospective cohort study. Clin Microbiol Infect 2020; 26:333–339 [DOI] [PubMed] [Google Scholar]
- 88.Huwyler T, Lenggenhager L, Abbas M, et al. : Cefepime plasma concentrations and clinical toxicity: A retrospective cohort study. Clin Microbiol Infect 2017; 23:454–459 [DOI] [PubMed] [Google Scholar]
- 89.Vercheval C, Sadzot B, Maes N, et al. : Continuous infusion of cefepime and neurotoxicity: A retrospective cohort study. Clin Microbiol Infect 2021; 27–731–735. [DOI] [PubMed] [Google Scholar]
- 90.Barreto EF, Webb AJ, Pais GM, et al. : Setting the beta-lactam therapeutic range for critically ill patients: Is there a floor or even a ceiling? Crit Care Explor 2021; 3:e0446. [DOI] [PMC free article] [PubMed] [Google Scholar]