Abstract
PCR was used to identify genes encoding aminoglycoside-modifying enzymes in 422 veterinary isolates of Salmonella enterica serotype Typhimurium. The identities of extra-integron genes encoding resistance to streptomycin, gentamicin, kanamycin, and apramycin were evaluated. Gentamicin resistance was conferred by the aadB gene. Kanamycin resistance was encoded by either the aphA1-Iab gene or the Kn gene. Apramycin resistance was determined by the aacC4 gene. Analysis of gene distribution did not reveal significant differences with regard to phage type, host species, or region except for the Kn gene, which was found mostly in nonclinical isolates. The data from this study indicate that pentaresistant DT104 does not acquire extra-integron genes in species- or geography-related foci, which supports the hypothesis that clonal expansion is the method of spread of this organism.
Infections with Salmonella are an important health problem worldwide (12). Of particular concern is a distinct strain of Salmonella enterica serotype Typhimurium, characterized as definitive type 104 (DT104), that is commonly resistant to five antibiotics (ampicillin, chloramphenicol, streptomycin, sulfonamides, and tetracycline; ACSSuT antibiogram)(10).
DT104 can develop resistance to antibiotics by acquiring the genes that confer antibiotic resistance. This can be facilitated by genetic elements such as plasmids, transposons, and mobile cassettes of DNA called integrons (7, 9). Recent studies have identified a genetic arrangement in the chromosome of DT104 for the ACSSuT antibiotic resistance pattern (2, 10, 11). This genetic arrangement is composed largely of two integrons containing the following genes: PSE-1, a cmlA homologue, aadA2, sull, and tetA, which encode resistance to ampicillin, chloramphenicol, streptomycin-spectinomycin, sulfonamides, and tetracycline, respectively. Our study was conducted to determine the “extra-integron” genes conferring resistance to several aminoglycoside antibiotics (and a related aminocyclitol antibiotic) in various animal isolates from the United States. Specific differences between host species or geographic areas may reveal differences in modes of spread or gene acquisition among phage types. Aminoglycoside-aminocyclitol resistance genes were the focus of this study since these antibiotics are widely utilized in clinical settings and since aminoglycoside resistance, apart from streptomycin resistance, conferred by genes outside the integrons. Amikacin resistance, genes were not examined due to the paucity of resistance in our pool of isolates (no strain was resistant).
The sample pool consisted of 422 pentaresistant S. enterica serotype Typhimurium isolates submitted in 1998 to the USDA-APHIS National Veterinary Services Laboratory, Ames, Iowa. The isolates used in the survey were obtained from ruminant species, swine, avian species, companion animals (including horses), and nonclinical settings (Food Safety and Inspection Service [FSIS]). The phage types included DT104 (n = 312), U302 (n = 54), DT193 (n = 33), DT208 (n = 13), and DT120 (n = 10). The isolates were submitted from 32 different states, and the isolate pool is summarized in Table 1. The resistance breakpoints for the antibiotics used were those established by the National Antimicrobial Susceptibility Monitoring Program (6) and were determined as previously described (3, 4). DNA isolation was performed as previously described (5). PCR was performed with an automated thermocycler (Hybaid, Teddington, United Kingdom) as previously described (3) by using the primers shown in Table 2. All of the gentamicin-resistant isolates harbored the aadB gene that codes for 2-aminoglycoside acetyltransferase (accession number AF078527). Similarly, a single gene, aacC4, which encodes for the 6-N-aminoglycoside acetyltransferase (accession number AJ009820), conferred all of the apramycin resistance. Kanamycin resistance was conferred by either of two genes, aphA1-Iab (111 isolates) (accession number AF093572) or Kn (11 isolates) (accession number U66885); both of these genes code for an aminoglycoside 3′ phosphotransferase product, although there is only 54% identity between the two genes.
TABLE 1.
Sample pool of pentaresistant S. typhimurium isolates submitted in 1998 to the National Veterinary Services Laboratory in Ames, Iowa
| Regiona | Phage typeb | No. of isolates from:
|
No. of companion isolates | Total no. of isolates | |||
|---|---|---|---|---|---|---|---|
| Ruminants | Swine | Nonclinical sources | Birds | ||||
| Eastern | DT104 | 24 | 48 | 9 | 22 | 4 | 107 |
| U302 | 2 | 12 | 2 | 3 | 0 | 19 | |
| DT193 | 7 | 0 | 0 | 1 | 0 | 8 | |
| DT208 | 1 | 0 | 0 | 0 | 1 | 2 | |
| DT120 | 0 | 3 | 0 | 1 | 0 | 4 | |
| Subtotal | 34 | 63 | 11 | 27 | 5 | 140 | |
| Middle | DT104 | 42 | 47 | 33 | 4 | 8 | 134 |
| U302 | 16 | 4 | 9 | 1 | 1 | 31 | |
| DT193 | 4 | 0 | 1 | 5 | 0 | 10 | |
| DT208 | 9 | 1 | 0 | 0 | 0 | 10 | |
| DT120 | 2 | 1 | 1 | 0 | 0 | 4 | |
| Subtotal | 73 | 53 | 44 | 10 | 9 | 189 | |
| Western | DT104 | 21 | 2 | 25 | 0 | 8 | 56 |
| U302 | 0 | 0 | 3 | 0 | 0 | 3 | |
| DT193 | 2 | 0 | 9 | 0 | 1 | 12 | |
| DT208 | 1 | 0 | 0 | 0 | 0 | 1 | |
| DT120 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Subtotal | 24 | 2 | 37 | 0 | 9 | 72 | |
| Unclassifiedc | DT104 | 1 | 1 | 13 | 0 | 0 | 15 |
| U302 | 0 | 0 | 1 | 0 | 0 | 1 | |
| DT193 | 0 | 0 | 3 | 0 | 0 | 3 | |
| DT208 | 0 | 0 | 0 | 0 | 0 | 0 | |
| DT120 | 1 | 0 | 1 | 0 | 0 | 2 | |
| Subtotal | 2 | 1 | 18 | 0 | 0 | 21 | |
| Total | 133 | 119 | 110 | 37 | 23 | 422 | |
The eastern, middle, and western regions are described in the text.
A total of 315, 54, 33, 13, and 10 isolates belonged to phage types DT104, U302, DT193, DT208, and DT120, respectively.
The states from which the isolates were obtained were not identified; 18 strains were from the FSIS, two strains were from foreign sources, and the origin of one strain was not recorded.
TABLE 2.
Nucleotide sequences of oligonucleotide primers used in PCR assays for gentamicin, kanamycin, apramycin, and streptomycin resistance genes
| Gene | Resistance | Accesion no. | Primersa |
|---|---|---|---|
| aac(3)-Ia | Gentamicin | U90945 | F, TTGATCTTTTCGGTCGTGAGT |
| R, TAAGCCGCGAGAGCGCCAACA | |||
| aacC1 | Gentamicin | U12338 | F, ATGTTACGCAGCAGCAACGAT |
| R, TAAGCCGCGAGAGCGCCAACA | |||
| aac(6′)-Ib | Gentamicin | U90945 | F, GTTACTGGCGAATGCATCACA |
| R, TGTTTGAACCATGTACACGGC | |||
| aadB | Gentamicin | AF078527 | F, GAGCGAAATCTGCCGCTCTGG |
| R, CTGTTACAACGGACTGGCCGC | |||
| aaC3 | Gentamicin | X55652 | F, AAACTGGTGGCAATAGAAGGAT |
| R, CTATCCGTATGACGCTGAGTC | |||
| grm | Gentamicin | M55521 | F, AAGCGCACGAAGCGCGGGCTG |
| R, AAGGCGGGCCTCAAGGAGGTC | |||
| aacC2 | Gentamicin | X51534 | F, GGCAATAACGGAGGCAATTCGA |
| R, CTCGATGGCGACCGAGCTTCA | |||
| aacA-aphD | Gentamicin | M18086 | F, AGCCAAATTATTGGAGTAAAGGAA |
| R, AACTGGCAATATCTCGTTTTA | |||
| aph(2")-Id | Gentamicin | AF016483 | F, CAATACAATAGAGATTTCAGGAG |
| R, ATGCTTCTGATTTCATTTAATCCC | |||
| aadD | Kanamycin | AF051917 | F, ATATTGGATAAATATGGGGAT |
| R, TCCACCTTCCACTCACCGGTT | |||
| aphA-3 | Kanamycin | U51474 | F, CTGTTCCAAAGGTCCTGCACT |
| R, CAATTCGGCTAAGCGGCTGTC | |||
| aphA/aph(3′)-Id | Kanamycin | Z48231 | F, ATGGGCGCCTATCACAATTGG |
| R, TCGCCTCCAGCTCTTCGTAGA | |||
| aphA-7 | Kanamycin | M29953 | F, GGAAACACATAGATTGCTTTA |
| R, CTCCACATCTTGCCAAATCAT | |||
| aphAI-IAB | Kanamycin | AF093572 | F, AAACGTCTTGCTCGAGGC |
| R, CAAACCGTTATTCATTCGTGA | |||
| Kn | Kanamycin | U66885 | F, ACTGGCTGCTATTGGGCGA |
| R, CGTCAAGAAGGCGATAGAAGG | |||
| Kan | Kanamycin | K02552 | F, GTGTTTATGGCTCTCTTGGTC |
| R, CCGTGTCGTTCTGTCCACTCC | |||
| kamB | Apramycin | M64625 | F, TGCCCAACCTGCTGTACCTGT |
| R, CCCCACGACGGCAGCTGCGGT | |||
| kamC | Apramycin | M64626 | F, TGCGCCGAACCTGCTCTCTGCGT |
| R, AAGTACCGCGCCTCCTCCA | |||
| aacC4 | Apramycin | AJ009820 | F, ACTGAGCATGACCTTGCGATGCTCTA |
| R, TACCTTGCCTCTCAAACCCCGCTT | |||
| kamA | Apramycin | D13170 | F, AGGACCCAGGCGGCCA |
| R, ATCGAGGACACCCGCATCCAC | |||
| aadA2 | Streptomycin | AF071555 | F, CGGTGACCATCGAAATTTCG |
| R, CTATAGCGCGGAGCGTCTCGC |
F, forward; R, reverse.
The results of a chi-square analysis and multivariate logistic regression are shown in Table 3. No significant gene differences were identified with regard to host, geographic region, or phage type, except for the Kn gene. Of the 11 isolates found to contain the Kn gene, 8 were nonclinical in origin. Streptomycin resistance is explained in phage type DT104 by the presence of an integron containing the aadA2 gene (2). All non-DT104 phage types were tested for the presence of the DT104-specific integron. Only phage type DT208 isolates lacked this element, as determined by the cmlA-tetR amplicon (5), but all DT208 isolates contained the aadA2 gene (Fig. 1). Geographic regions of the United States were arbitrarily classified as eastern (including Alabama, Connecticut, Delaware, Florida, Georgia, Indiana, Kentucky, Massachusett Maryland, Maine, Michigan, Mississippi, North Carolina, New Hampshire, New Jersey, New York, Ohio, Pennsylvania, Rhode Island, South Carolina, Tennessee, Virginia, Vermont, and West Virginia), middle (Arkansas, Iowa, Illinois, Kansas, Louisiana, Minnesota, Missouri, North Dakota, Nebraska, Oklahoma, South Dakota, Texas, and Wisconsin), and western (Alaska, Arizona, California, Colorado, Hawaii, Idaho, Montana, New Mexico, Nevada, Oregon, Utah, Washington, and Wyoming). Twenty-one isolates were not classified by region. P values of <0.05 were considered significant. Analysis was performed by using JMP IN (SAS Institute Inc.). Gene comparison was performed by using DNASIS (Hitachi Software).
TABLE 3.
Chi-square analyses for genes found to confer resistance to gentamicin, kanamycin, and apramycin by phage type, species, region, and multivariate regression
| Gene | Chi-square analyses
|
|||
|---|---|---|---|---|
| Phage type | Species | Regiona | Multivariate regressionab | |
| aadB | 0.2911 | 0.6521 | 0.4553 | 0.5902 |
| aphAI-IAB | 0.6835 | 0.4627 | 0.5874 | 0.6066 |
| Kn | 0.6184 | 0.0101 | 0.0626 | 0.0089 |
| aacC4 | 0.9037 | 0.5943 | 0.1585 | 0.7257 |
Unclassified isolates were not included in the analysis.
Phage type, species, and region were included in the analysis.
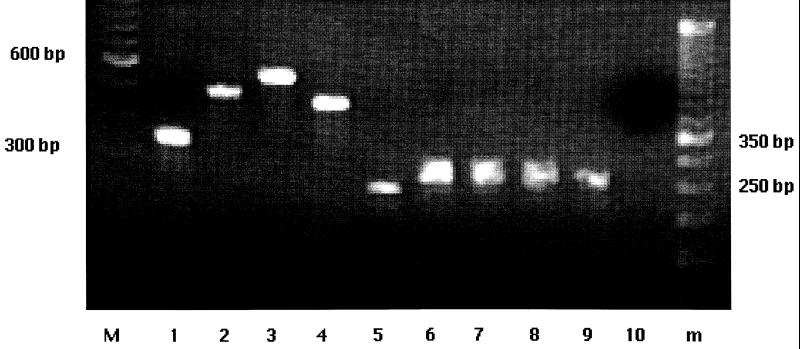
FIG. 1.
Agarose gel electrophoresis of amplicons following PCR for aminoglycoside resistance genes of multiresistant S. enterica serotype Typhimurium. Lanes M and m contained 100- and 50-bp molecular weight standards (GIBCO BRL), respectively. Lane 1, aadB amplicon obtained by using template DNA from a representative gentamicin-resistant strain; lanes 2 and 3, aphA-lab and Kn amplicons, respectively, obtained by using template DNA from representative kanamycin-resistant strains; lane 4, aacC4 amplicon obtained by using template DNA from a representative apramycin-resistant strain; lane 5, aadA2 amplicon obtained by using template DNA from a representative phage type DT208 isolate (all isolates yielded this amplicon [data not shown]); lanes 6 through 10, cmlA-tetR amplicons, or lack of these amplicons, obtained by using template DNA from representatives of phage type DT104 (lane 6), U302 (lane 7), DT193 (lane 8), DT120 (lane 9), and DT208 (lane 10). The positions of specific molecular weight standards and amplicon sizes are indicated on the left and right.
The results of this study indicate that single genes are responsible for conferring gentamicin resistance and apramycin resistance, and no host, geographic (Fig. 2), or phage type differences were noted. These findings suggest two possibilities regarding gentamicin and apramycin resistance genes in DT104. First, although many genes can confer gentamicin or apramycin resistance, only a small subset of gentamicin or apramycin resistance genes or enzymes are compatible with vitality of pathogenic microorganisms. For example, the G+C content or enzyme kinetics may dictate, and thus limit, potential acquisition candidates. Second, DT104 is abundantly exposed to microbes that possess and transfer these two specific genes. This seems less likely since intestinal microfloras are diverse and since the isolates were obtained from a wide array of hosts. We favor the former possibility since DT104 resistance genes can be found in fish pathogens (1, 2, 8, 13), suggesting that certain resistance genes have a predilection for moving from bacterium to bacterium.
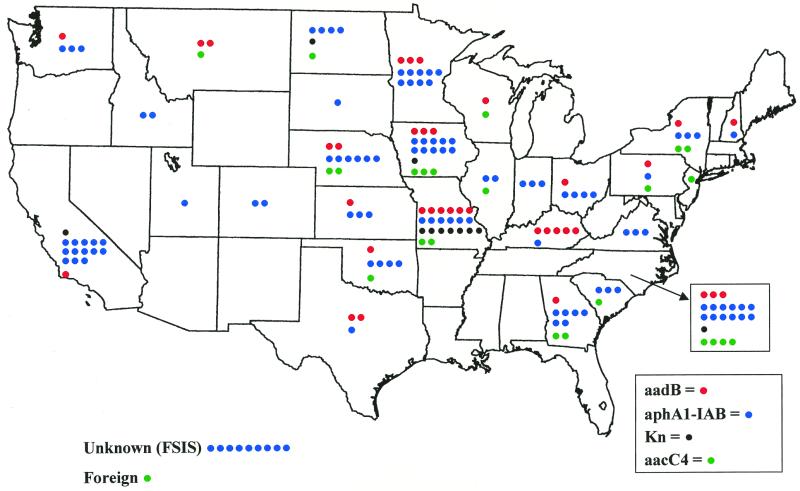
FIG. 2.
Geographic distribution of strains possessing genes encoding aminoglycoside-modifying enzymes. Individual isolates are indicated on the basis of their genes. (red, aadB; green, aacC4; black, Kn; blue, aphA-lab). For isolates with two or more genes, there is a colored dot for each gene. Nine isolates, all obtained from FSIS, were not attributed to a geographic location. One isolate was obtained from outside the United States.
Two separate genes, however, conferred kanamycin resistance, although a single gene conferred kanamycin resistance in the clinical isolates. The Kn gene was much more likely to be found in nonclinical isolates, suggesting that its presence and/or expression is not compatible with overt virulence. This is consistent with our recent finding that certain kanamycin-resistant DT104 isolates are less invasive and less pathogenic than the rest of the group (3). Alternatively, it is possible that the Kn gene is abundant in slaughterhouse environments and that its presence in the nonclinical isolates represents a postharvest transfer or acquisition event.
Recently, we reported that a DT104-related phage type (U302) and a phage type not related to DT104 (DT120) are capable of possessing the integrons found in DT104 (4). In this study we demonstrated that another phage type not related to DT104 (DT193) also has this capability. However, it appears that multiresistant DT208 does not contain the genetic array characterized for the ACSSuT pattern but does have the aadA2 gene typically associated with the DT104-specific integron. This suggests that DT208 may contain an uncharacterized integron harboring this gene and perhaps other antibiotic resistance genes.
Our study, which was aimed at detecting integron-independent genes encoding aminoglycoside-modifying enzymes, did not reveal significant differences in the genes conferring gentamicin, kanamycin, and apramycin resistance in clinical isolates. This suggests that DT104 did not acquire these genes in a geography- or species-specific manner. Our data support the theory of clonal dissemination of DT104, although it is possible that the overwhelming prevalence of single aminoglycoside resistance genes is due to physiochemical compatibilities between DT104 metabolic properties and aminoglycoside-modifying enzymes. Our results should be compared to the results of similar studies performed in the future and similar studies performed with human isolates. The results should provide an understanding of gene acquisition and the mode of spread of multiresistant DT104 in order to provide insight into the emergence of DT104 as an important health concern worldwide.
Acknowledgments
We thank Ruth Willson for technical assistance, Kathy Ferris for contributing strains, Sandy Johnson for secretarial assistance, and Jeff Zimmerman, Irene Wesley, and James Roth for reading the manuscript.
REFERENCES
- 1.Bolton L, Kelley L, Lee M, Fedorka-Cray P, Maurer J. Detection of multidrug-resistant Salmonella enterica serotype typhimurium DT104 based on a gene which confers cross-resistance to florfenicol and chloramphenicol. J Clin Microbiol. 1999;37:1348–1351. doi: 10.1128/jcm.37.5.1348-1351.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Briggs C E, Fratamico P M. Molecular characterization of an antibiotic resistance gene cluster of Salmonella typhimurium DT104. Antimicrob Agents Chemother. 1999;43:846–849. doi: 10.1128/aac.43.4.846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Carlson S A, Browning M, Ferris K E, Jones B D. Identification of diminished tissue culture invasiveness among multiple antibiotic resistant Salmonella typhimurium DT104. Microb Pathogen. 2000;28:37–44. doi: 10.1006/mpat.1999.0322. [DOI] [PubMed] [Google Scholar]
- 4.Carlson S A, Willson R M, Crane A J, Ferris K E. Evaluation of invasion-conferring genotypes and antibiotic-induced hyperinvasive phenotypes in multiple antibiotic resistant Salmonella typhimurium DT104. Microb Pathogen. 2000;28:373–378. doi: 10.1006/mpat.2000.0355. [DOI] [PubMed] [Google Scholar]
- 5.Carlson S A, Bolton L F, Briggs C E, Hurd H S, Sharma V K, Fedorka-Cray P, Jones B D. Detection of Salmonella typhimurium DT104 using multiplex and fluorogenic PCR. Mol Cell Probes. 1999;13:213–222. doi: 10.1006/mcpr.1999.0240. [DOI] [PubMed] [Google Scholar]
- 6.Fedorka-Cray P J, Miller M, Tollefson L, Dargatz D A, Wineland N E. National Antimicrobial Susceptibility Monitoring Program— veterinary isolates. Washington, D.C.: United States Government Printing office; 1998. [Google Scholar]
- 7.Hall R M, Stokes H W. Integrons: novel DNA elements which capture genes by site-specific recombination. Genetica. 1993;90:115–132. doi: 10.1007/BF01435034. [DOI] [PubMed] [Google Scholar]
- 8.Kim E H, Aoki T. Drug resistance and broad geographical distribution of identical R plasmids of Pasteurella piscicida isolated from cultured yellowtail in Japan. Microbiol Immunol. 1993;37:103–109. doi: 10.1111/j.1348-0421.1993.tb03186.x. [DOI] [PubMed] [Google Scholar]
- 9.Recchia G D, Hall R M. Origins of the mobile gene cassettes found in integrons. Trends Microbiol. 1997;5:389–394. doi: 10.1016/S0966-842X(97)01123-2. [DOI] [PubMed] [Google Scholar]
- 10.Ridley A, Threlfall E J. Molecular epidemiology of antibiotic resistance genes in multiresistant epidemic Salmonella typhimurium DT 104. Microb Drug Resist. 1998;4:113–118. doi: 10.1089/mdr.1998.4.113. [DOI] [PubMed] [Google Scholar]
- 11.Sandvang D, Aarestrup F M, Jensen L B. Characterisation of integrons and antibiotic resistance genes in Danish multiresistant Salmonella enterica Typhimurium DT104. FEMS Microbiol Lett. 1998;160:37–41. doi: 10.1111/j.1574-6968.1998.tb12887.x. [DOI] [PubMed] [Google Scholar]
- 12.Slutsker L, Altekruse S F, Swerdlow D L. Foodborne diseases. Emerging pathogens and trends. Infect Dis Clin N Am. 1998;12:199–216. doi: 10.1016/s0891-5520(05)70418-9. [DOI] [PubMed] [Google Scholar]
- 13.Zhao J, Aoki T. Nucleotide sequence analysis of the class G tetracycline resistance determinant from Vibrio anguillarum. Microbiol Immunol. 1992;36:1051–1060. doi: 10.1111/j.1348-0421.1992.tb02109.x. [DOI] [PubMed] [Google Scholar]