Abstract
Metabolic reprogramming is an important characteristic of tumor cells. Tumor cells reprogram their metabolic pathways to meet the material, energy and redox force needs for rapid proliferation. Metabolic reprogramming changes the level or type of specific metabolites inside and outside cells, and promotes tumor growth by affecting gene expression, cell state and the tumor microenvironment. Glucose metabolism, glutamine metabolism and lipid metabolism are significant metabolic pathways in tumors. Targeting metabolic reprogramming can significantly inhibit tumor growth and induce apoptosis. Metabolic reprogramming also plays an important role in maintaining the growth advantage of tumor cells and enhancing the chemotherapy tolerance of lung cancer. This review summarizes abnormal changes in the metabolism of glucose, fat and amino acids in lung cancer, and the underlying molecular mechanism, with the aim of providing novel ideas for the prevention, early diagnosis and treatment of lung cancer.
Keywords: metabolic reprogramming, lung cancer
1. Introduction
Lung cancer is a disease with high incidence and mortality rates worldwide. At present, the pathogenic factors of lung cancer have not been completely clarified. The most common pathogenic factor of lung cancer is long-term and high-frequency smoking (1). In addition, long-term exposure to carcinogens, air pollution, human immune status, genetic factors and metabolic activities have been associated with lung cancer (2). Lung cancer is mainly divided into small cell lung cancer (SCLC) and non-SCLC (NSCLC). The latter includes lung squamous cell carcinoma, lung adenocarcinoma (LUAD) and large cell cancer, accounting for ~85% of all lung cancer cases (3). Although considerable progress has been achieved in the targeted treatment of lung cancer in recent years, drug resistance, recurrence and metastasis have brought great difficulties (4). At present, surgical resection is still performed for tumors that limit themselves to the primary location, but 80–85% of patients are already in the unresectable stage at early diagnosis (5). In addition, the progress of lung cancer treatment is limited by the adverse reactions of chemotherapeutic drugs, the high resistance rate of targeted drugs and the immune tolerance microenvironment of the tumor. Therefore, novel strategies for lung cancer treatment need to be developed.
Metabolism is the energy and material basis of life activities. Under normal circumstances, metabolism occurs in the body in an orderly manner to ensure physiological functions (6). However, tumor growth is a multi-factor and multi-stage dynamic process. Some studies have shown that the metabolic pattern of tumor cells is different from that of normal cells (7–9). Metabolic change, also called metabolic reprogramming, plays an important role in regulating the occurrence and development of tumors. To meet the needs of rapid proliferation and growth in a tumor microenvironment with poor blood vessels and nutrition, tumor cells undergo metabolic reprogramming to provide energy and raw materials, maintain the steady state of cell redox and regulate intracellular signal transduction (10). However, the mechanisms underlying tumorigenesis and development remain to be elucidated. Various metabolism-related genes serve as oncogenes and tumor suppressor genes (11). It has been shown that oncogenes such as MYC, NF-κB and AKT can regulate the enzymes in the glycolysis and glutaminolysis pathways (12–14). MYC increases the transcription rate of GLUT transporter and hexokinase-2, which enhances glucose uptake and retention (15).
Therefore, the present review summarizes the abnormal changes in the metabolism of glucose, fat and amino acids in lung cancer, and the molecular mechanisms behind them to provide novel ideas for the prevention, early diagnosis and treatment of lung cancer (Fig. 1).
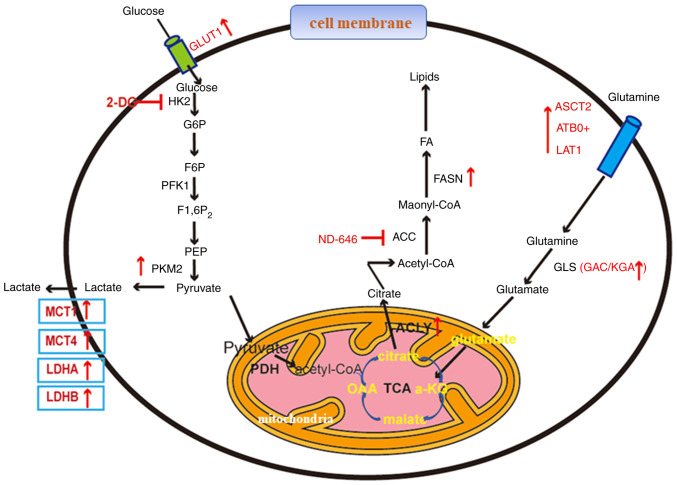
Figure 1.
Tumor metabolism in lung cancer. An overview of key metabolic pathways and their associated crucial molecules in lung cancer cells is presented. Red arrows represent upregulated genes or metabolites involved in tumor metabolism, and the developed inhibitors are indicated. GLUT1, glucose transporter 1; HK2, hexokinase 2; G6P, glucose-6-phosphate; PFK1, phosphofructokinase 1; PEP, phosphoenolpyruvic acid; PKM2, pyruvate kinase M2; MCT, monocarboxylate transporter; LDH, lactate dehydrogenase; FA, fatty acid; FASN, fatty acid synthase; ACC, acetyl-CoA carboxylase; ACLY, ATP-citrate lyase; GAC, glutaminase C; ASCT2, alanine-serine-cysteine transporter 2; ATB0+, amino acid transporter B0,+; LAT1, Human L-type amino acid transporter 1; α-KG, α-ketoglutarate; OAA, oxaloacetate; PDH, pyruvate dehydrogenase; GLS, glutaminase; KGA, kidney-type glutaminase.
2. Glucose metabolism
Metabolic reprogramming is an important marker of cancer (16). A number of metabolic pathways in tumor cells change to meet the material, energy and redox force needs for rapid and continuous cell proliferation (17). In the 1920s, Otto Warburg reported that cancer cells consume a large amount of glucose even in the presence of oxygen, and most of this is metabolized into lactic acid. This phenomenon is called aerobic glycolysis or the Warburg effect (18). This process is one of the important metabolic differences between cancer and most normal tissues, and it is also the first tumor metabolic reprogramming process. Under aerobic conditions, normal cells consume oxygen through glycolysis, the tricarboxylic acid (TCA) cycle and oxidative phosphorylation, thus completely decomposing the glucose and producing a large amount of ATP (34 mol ATP/mol glucose). Under anaerobic conditions, the lack of oxygen prevents the complete degradation of intracellular glucose. Glucose is partially degraded to pyruvate and converted to lactic acid. The energy generated by this process (2 mol ATP/mol glucose) is much lower than that generated by oxidative phosphorylation (19). Although enhanced glycolysis in lung cancer consumes a large amount of glucose and leads to lactic acid accumulation, high levels of glucose have also been found in the serum samples of patients with lung cancer. Although glycolysis accelerates the decomposition of glucose, several reactions in the glycolytic pathway are reversible and participate in glucose resynthesis. Gluconeogenesis, carbohydrate metabolism and lipogenesis can store and provide energy by synthesizing glucose, which may be the reason for the increase in glucose level in lung cancer (20).
Tumor cells produce intermediate metabolites through glycolysis. These intermediate metabolites provide raw materials for biosynthetic pathways, such as nucleotides, lipids, amino acids and reduced nicotinamide adenine dinucleotide phosphate (NADPH), to meet the needs of rapid cell proliferation (21). These metabolic pathways include the pentose phosphate pathway for the production of RNA and NADPH, the hexosamine pathway for protein glycosylation and glycogen production, and the serine biosynthesis pathway (22–24).
The first step in glycolysis is glucose entering the cell through the plasma membrane. Glucose transporters (GLUTs) are important carrier proteins responsible for transporting glucose. Treatment of A549 lung cancer cells with WZB117, an irreversible inhibitor of GLUT1, reduces GLUT1 expression and glucose uptake; it also inhibits the growth of lung cancer cells in cooperation with cisplatin and paclitaxel. The addition of exogenous ATP alleviates this inhibitory effect, which indicates that the inhibition of GLUT1 prevents tumor growth by blocking ATP synthesis (25).
After glucose enters the cell, the first irreversible reaction is the production of glucose-6-phosphate catalyzed by hexokinase 2 (HK2). Tumor cells promote glucose metabolism by increasing glucose uptake and inducing high HK2 expression (26). Targeting HK can reverse the drug resistance of tumor cells (27). 2-Deoxy-D-glucose (2-DG) is a small-molecule inhibitor targeting HK. 2-DG combined with Adriamycin or paclitaxel can significantly delay tumor growth and prolong the survival time of mice with NSCLC (28). Moreover, inhibition of glycolysis by 2-DG can improve the sensitivity of NSCLC with T790M secondary drug resistance mutation to epidermal growth factor receptor (EGFR)-tyrosine kinase inhibitor (TKI) (29).
In the second step, fructose-1,6-diphosphate is catalyzed by phosphofructokinase 1 (PFK1). Fascin promotes the transcription of PFKFB3 by promoting the binding of YAP1 to a TEAD1/4 binding site, which activates the expression of PFK1 and mediates glycolysis in lung cancer (30). Not only is PFK expression upregulated in malignant tumors, platelet-type PFK can also regulate the glycolysis level of lung cancer and promote cell proliferation (31).
In the third step, pyruvate kinase catalyzes the conversion of phosphoenolpyruvic acid and ADP to pyruvate and ATP, respectively. Low-affinity pyruvate kinase M2 (PKM2) is highly expressed in lung cancer, and its upregulation is often associated with the hypomethylation of the PKM2 gene promoter. Silencing PKM2 can improve the sensitivity of lung cancer to the chemotherapy drugs cisplatin and docetaxel by increasing apoptosis and inhibiting proliferation. PKM2 is a potential adjuvant therapeutic target (32,33). However, PKM1 rather than PKM2 shows tumor-promoting function in pulmonary neuroendocrine tumors, including SCLC. This finding challenges the view that PKM2 limiting glucose metabolism is a prerequisite for tumorigenesis and provides a theoretical basis for PKM1 as a therapeutic target in SCLC (34).
The attenuation of the last reaction step promotes the entry of intermediate metabolites into the above biosynthetic pathway (35). However, the expression of lactate dehydrogenase (LDH) in lung cancer cells is upregulated (36), which can convert pyruvate into lactate. In addition, lactate secretion is increased, which is characterized by a high expression of monocarboxylate transporter (MCT), as lactate retained in cells inhibits the expression of PFK1 (37). Lactate secretion into the environment can also promote the development of tumors (38). Lactate, as a signal transduction substance under hypoxia, activates the signal pathway associated with tumor cell survival (39). Targeted lactate production is a possible strategy to overcome tumor drug resistance. Inhibition of LDH activity by small interfering RNA or oxalate can overcome the drug resistance of tumor cells to paclitaxel and trastuzumab (40,41). In mice and patients with lung cancer, lactate in tissues and circulation provides an equivalent carbon source for the aerobic oxidation of normal and tumor tissues, and its contribution to mitochondrial metabolism is no less than that of glucose (38,42). Faubert et al (42) injected 13C-labeled lactate into patients with lung cancer and found that lactate can be used as the carbon source of the TCA cycle in patients with different lung cancer types, and that the expression levels of MCT1, MCT4, LDHA and LDHB in tumors are upregulated. In the transplanted tumor model of human lung cancer cells in mice, the levels of isotopic-labeled lactic acid and isotopic-labeled TCA cycle intermediate metabolites at the tumor site are increased. Knockout of MCT1 in mice could reduce the uptake of lactic acid by tumors (43).
The upregulated expression of these key glycolytic enzymes in lung cancer cells is often closely associated with the abnormally activated oncogenes and cancer-promoting signaling pathways in cells. Hypoxia inducible factor-1α (HIF-1α) is an important transcription factor involved in glycolysis regulation in cells, which can directly transcribe and regulate the expression of multiple key glycolysis enzymes (44). The expression of aldolase A in the glycolysis pathway is increased in NSCLC, which consequently increases lactate activity to inhibit prolyl hydroxylase activity and further induce HIF-1α (45). The positive feedback of lactate further promotes the aerobic glycolysis of tumor cells. MYC is an important oncogene that promotes the glycolysis of lung cancer cells by transcriptionally regulating the expression of multiple key glycolytic enzymes (46).
3. Amino acid metabolism
Amino acid metabolic abnormalities include those in glutamine, serine and glycine, among others, the most important of which are the glutamine metabolic abnormalities (47). In addition to glycolysis, numerous tumor cells also rely on glutamine to meet their bioenergy and metabolic needs. Although glutamine is an important non-essential amino acid required for cell proliferation, it is also an essential amino acid under specific circumstances (48). Glutamine depends on glutamine metabolism, provides metabolic energy for rapidly proliferating tumor cells, provides carbon and nitrogen sources for the synthesis and metabolism of substances, such as nucleotides, amino acids and fatty acids, and maintains the balance and stability of cellular reactive oxygen species (49). Therefore, similar to glucose, glutamine is considered the main nutrient to promote tumor proliferation.
In tumor cells, glutamine can be transported into cells by amino acid transporters as a substrate. Glutamine is then converted into glutamate in the mitochondria and enters the TCA cycle. Some amino acid transporters, such as alanine-serine-cysteine transporter 2, amino acid transporter B0,+ and Human L-type amino acid transporter 1, are overexpressed in lung cancer and upregulate the intake of glutamine by cancer cells (50). Meanwhile, glutamine and glutamate play important roles in the growth of pC9/IR-resistant cells. The growth of erlotinib-resistant NSCLC depends on glutamine (51).
Clinical studies have found high plasma glutamine levels in patients with malignant tumors (52). Glutamine can be converted into glutamate under the action of glutaminase (GLS) for the synthesis of fatty acids and glutathione. Antioxidant glutathione maintains the balance of redox reaction in tumor cells, helps tumor cells resist oxidative stress and prolongs tumor cell survival (53).
GLS is the starting and rate-limiting enzyme for glutamine catabolism (54); it can be divided into renal GLS1 and hepatic GLS2. Selective cleavage of GLS1 precursor mRNA produces two subtypes, glutaminase C (GAC) and renal glutaminase (KGA), which have different regulatory activities (55,56). Some studies have shown that the expression of GAC increases in various types of cancer, such as breast cancer (57), lung cancer (58) and acute myeloid leukemia (59), suggesting that GLS1 is closely associated with glutamine metabolism reprogramming in various tumors.
Van den Heuvel et al(60) analyzed the GAC/KGA ratio in 45 NSCLC tissues and matched normal lung cancer tissues. The study found that the ratio increases significantly in lung cancer tissues, indicating that GAC plays a key role in tumor metabolism. Transfection of the oncogene KRAS induces the dependence of cells on glutamine. However, different KRAS mutation sites may play different roles. For example, lung cancer cells carrying KRAS-G21V mutation are less glutamine-dependent than G12C or G12D mutant cells (60). Recent studies have shown that KRAS and LKB1 co-mutants lead to the invasion and metastasis of NSCLC, accompanied by metabolic reprogramming (61).
4. Lipid metabolism
Lipids contain thousands of different types of molecules, including glycerophosphingolipids, glycerides, fatty acids, sphingolipids, sterol lipids, pregnenolone lipids, glycolipids and polyketones. Lipids are widely distributed in organelles and are a key component of all membranes (62). Lipid metabolism provides energy for tumor cells, cell proliferation and signaling molecule generation (63).
Fatty acid metabolism and lung cancer
In normal cells, the de novo synthesis of fatty acids generally occurs only in specific tissues, such as liver tissue, adipose tissue and breast tissue during lactation (64). Rapidly proliferating cells increase fatty acid synthesis, provide lipids for membrane components, and are conducive to β-oxidation and protein acyl modification. Therefore, increased fat synthesis is extremely important for highly proliferating cancer cells. When the glucose level is low, tumor cells absorb fatty acids in the surrounding environment for fatty acid metabolism, produce metabolic intermediates, and then enter the TCA cycle to produce ATP and nutrients required for tumor growth (65). Fatty acids are usually stored in lipid droplets in the form of triglycerides (TGs), and free fatty acids are released through the action of adipose TG lipase, hormone-sensitive lipase and monoacylglycerol lipase.
The biosynthesis of fatty acids begins with acetyl-CoA carboxylase (ACC) carboxylating acetyl CoA in the cytoplasm to produce malonyl CoA. Silencing ACC accelerates the growth of lung cancer cells by promoting the redox balance of NADPH (53). ND-646, an allosteric inhibitor of ACC, also exerts an antitumor effect on NSCLC cells (66).
Fatty acid synthase (FASN) catalyzes the assembly of malonyl CoA or acetyl CoA into fatty acids; it is also one of the key enzymes in fatty acid anabolism. The increase in fatty acid synthesis is due to the increase in FASN level, which is closely associated with a poor prognosis, as confirmed in various tumors, such as pancreatic and breast cancer (67,68). Currie et al (69) found a high expression level of fatty acid synthase in NSCLC. A high expression level of FASN not only promotes the growth of tumor cells but also improves the metastatic ability of NSCLC cells and cisplatin resistance (70). Ali et al (71) found that the palmitoylation of EGFR is specifically expressed in mutated EGFR NSCLC with acquired TKI resistance. Mutated EGFR activates FASN mediated by sterol regulatory element-binding proteins (SREBPs), which consequently promote the palmitoylation of EGFR. To produce TKI resistance, orlistat, a selective inhibitor of FASN, can inhibit this effect. Therefore, FASN is an attractive therapeutic target. Most tumor cells rely on the FASN-mediated de novo synthesis of fatty acids, whereas most non-tumor cells rely on exogenous fatty acids. The first targeted drug of FASN, TVB-2640, has entered clinical trials. Combined with paclitaxel, it can stabilize the disease progression in the medium and long term (72,73).
Fatty acid synthesis can also be performed through reductive carboxylation. Glutamine-derived α-ketoglutarate is catalyzed by isocitrate dehydrogenase (IDH) to form citric acid (49). IDH mutations are associated with various tumor types, including gliomas, myeloid malignancies and myelodysplastic syndormes (74). Genetic hybridization of IDH2 mutant mice with carcinogenic FLT3 or NRAS alleles can promote leukemia transformation by inhibiting myeloid cell differentiation (75). The expression levels of FASN, ACC and ATP-citrate lyase (ACLY) are upregulated in fatty acid synthesis in various cancer types, including prostate cancer, breast cancer, colorectal cancer, lung cancer, hepatocellular carcinoma, pancreatic cancer, gastric cancer and multiple myeloma. Inhibition of these enzymes prevents tumor growth in vitro and in vivo (76–78).
ACLY is the most critical enzyme in glucose catabolism, and cholesterol and fatty acid anabolism. In the presence of ATP and CoA, it catalyzes citric acid in the cytoplasm to produce acetyl CoA and oxaloacetate. Acetyl CoA is not only an important raw material for the de novo synthesis of cholesterol and fatty acids, but also a substrate for protein acetylation modification. ACLY knockdown or inhibition with SB-204990 changes the metabolic pathway to reduce the occurrence of mouse tumors and the formation of transplanted human tumor cells (79,80). Acetylation of 540, 546 and 554 lysine residues (3K) of ACLY polypeptide chain promotes lipid biosynthesis and tumor growth. The acetylation level of ACLY is significantly increased in lung cancer tissues (81). Further investigations have confirmed that ACLY 3K is also a ubiquitin modification site, and a competitive association exists between them. Under high-glucose conditions, P300/calcium-binding protein-associated factor acetyltransferase is activated to promote ACLY acetylation, block ACLY ubiquitination and degradation, improve its stability, and promote de novo lipid synthesis and tumor growth (81). ACLY is a promising therapeutic target for lung cancer; its product, acetyl CoA, is not only an important metabolite, but also a substrate for the acetylation of proteins and nucleic acids. Therefore, inhibiting its production affects de novo fatty acid biosynthesis.
Fatty acids entering the bioactive pool must be activated by acetyl CoA synthetase (ACS) to produce fatty acid CoA. Bioactive fatty acids contribute to protein palmitoylation, a post-translational modification that is particularly important in certain tumors (82). Triacsin C, a chemical inhibitor of ACS, induces the apoptosis of lung cancer cells, and the expression of ACS is negatively correlated with the overall survival of patients with lung cancer (83).
The polyunsaturated fatty acid metabolism and biotransformation pathways have a great impact on tumor apoptosis and proliferation. A number of tumor cells highly express clyclooxygenase (COX), lipoxygenase (LOX) and cytochrome P450. These enzymes transform ω-6 polyunsaturated fatty acids into highly active arachidonic acid (AA) to regulate the proliferation and apoptosis of tumor cells (84). Various ω-3 metabolites of polyunsaturated fatty acids inhibit tumorigenic pathways; for example, the eicosapentaenoic acid metabolite ω-3,17,18-epoxide cascade activates anti-proliferation and pro-apoptosis pathways (85). Resolvins produced by LOX metabolites are effective inhibitors of tumor-derived inflammatory pathways (86). The cytosolic phospholipase A2-AA-COX-2 pathway is an important signaling pathway of inflammation, and some key factors of the pathway are associated with lung cancer. Overexpression of COX-2 initiates and promotes lung cancer development (87,88).
Cholesterol metabolism and lung cancer
Cholesterol homeostasis is strictly regulated by a complex protein grid, including its intake, synthesis, efflux, metabolism and esterification. High levels of cholesterol are found in prostate, breast, liver, gastric, colorectal and lung cancer (89,90).
3-Hydroxy-3-methyl glutaryl CoA reductase is the rate-limiting enzyme of cholesterol biosynthesis; it is upregulated in lung cancer and the target of statins regulating plasma cholesterol (91).
In a number of tissues and organs, cholesterol can be converted to 27-hydroxycholersterol (27HC) through sterol 27 hydroxylase (CYP27A1). 27HC is the most abundant hydroxyl cholesterol substance in the circulating blood; its role in cancer mainly depends on the properties of endogenous SREBP and the function of liver X receptor (LXR) regulator.
In normal cells, the rate of cholesterol production is low, occurring through the transcriptional regulation of key genes involved in lipid biosynthesis. Tumor cells can regulate and activate SREBP through the PI3K/Akt/mTOR, MAPK/ERK1/2, HIF-1α, p53 and SHH pathways to increase the signal activity of growth factors or steroid hormone receptors (92). In vivo, 27HC inhibits cholesterol synthesis by regulating SREBP (93).
LXRs are members of the nuclear receptor family of ligand-dependent transcription factors. Activation of LXRs affects cancer progression in various ways. Specifically, LXRs inhibit the proliferation, migration and invasion of breast, prostate, ovarian, lung, skin and colorectal cancer cells (94,95). 27HC is a risk factor for a number of cancer types; it triggers a tumor response to endocrine therapy and reduces the activation of estrogen receptor (ER) to promote angiogenesis, proliferation, invasion and migration (96). By contrast, 27HC inhibits cell viability, proliferation, invasion and migration by activating LXRs in EGFR-mutated malignant tumors, such as lung cancer and colon cancer (97,98). Lung cancer tissues are rich in 27HC, and the expression of 27HC synthase CYP27A1 in lung cancer cells is also higher than that in normal lung cells. 27HC may be the main source of lung cancer occurrence and development (99). Existing research results show that 27HC promotes lung cancer cells proliferation in an ERβ-dependent manner. The role of 27HC is not affected by the membrane-bound ER G protein-coupled receptor 30. Conversely, the role of 27HC is not associated with the activation of EGFR or MAPK, but is mediated by the PI3K/Akt signaling pathway. In other respects, 27HC promotes the production of osteoclasts in the microenvironment of LUAD by inhibiting the expression of miR-139 and activating the STAT3/c-Fos/NFATc1 pathway to accelerate bone metastasis (100). A high-cholesterol diet upregulates the level of 27HC in vivo, whereas 27HC downregulates the cholesterol level of ER-negative NSCLC A549 cells by activating the LXR signaling pathway, and inhibits the activity and proliferation of A549 cells.
Cholesterol is also involved in the drug resistance of tumor cells. For example, Chen et al (101) observed that the cholesterol level is significantly higher in the lipid raft of gefitinib-resistant cells than in that of gefitinib-sensitive cells in NSCLC. After depletion of lipid raft cholesterol, the cells recover their sensitivity to gefitinib. Colenemine and betulin improve the sensitivity of patients with NSCLC to gefitinib by inhibiting SREBP/SCAP or SREBP (102).
5. Conclusion
Carcinogenesis is a complex process involving multiple genes and steps. In cell carcinogenesis, the imbalance in cell metabolism is not only an important biochemical basis for maintaining various malignant phenotypes, such as tumor cell growth, proliferation and apoptosis, but also a result of the imbalance of the intracellular regulation grid; it involves the activation of oncogenes, the inactivation of tumor suppressor genes and gene mutations. With the continuous development of metabolomics, genomics, proteomics and other associated disciplines and technologies, great breakthroughs have been achieved in the research into the metabolic reprogramming of lung cancer in recent years, and the molecular mechanisms underlying metabolic reprogramming have been gradually understood. However, tumor metabolic reprogramming includes different metabolic pathways that usually involve several regulatory genes, metabolic enzymes and signaling pathways. The research progress associated with lung cancer metabolism is far less than expected, and each metabolic pathway and its specific regulatory mechanism need to be further studied. The identification of the specific metabolic enzymes or metabolites involved in the occurrence and development of lung cancer, and the elucidation of their roles and mechanisms in tumorigenesis warrant further investigation.
Acknowledgements
Not applicable.
Funding Statement
This research was supported by a grant from the Scientific Research Program of Tianjin Education Commission (no. 2020KJ151).
Availability of data and materials
Not applicable.
Authors' contributions
XL was responsible for the conception of the manuscript, and ML was responsible for consulting relevant literature and manuscript drafting. JC and HL are responsible for designing and overseeing the study, and reviewing the manuscript. All authors read and approved the final manuscript. Data authentication is not applicable.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Tammemagi MC, Berg CD, Riley TL, Cunningham CR, Taylor KL. Impact of lung cancer screening results on smoking cessation. J Natl Cancer Inst. 2014;106:dju084. doi: 10.1093/jnci/dju084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sun S, Schiller JH, Gazdar AF. Lung cancer in never smokers-a different disease. Nat Rev Cancer. 2007;7:778–790. doi: 10.1038/nrc2190. [DOI] [PubMed] [Google Scholar]
- 3.Relli V, Trerotola M, Guerra E, Alberti S. Abandoning the notion of non-small cell lung cancer. Trends Mol Med. 2019;25:585–594. doi: 10.1016/j.molmed.2019.04.012. [DOI] [PubMed] [Google Scholar]
- 4.Zheng H, Zhan Y, Liu S, Lu J, Luo J, Feng J, Fan S. The roles of tumor-derived exosomes in non-small cell lung cancer and their clinical implications. J Exp Clin Cancer Res. 2018;37:226. doi: 10.1186/s13046-018-0901-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fernandez Y, Viesca M, Arvanitakis M. Early diagnosis and management of malignant distal biliary obstruction: A review on current recommendations and guidelines. Clin Exp Gastroenterol. 2019;12:415–432. doi: 10.2147/CEG.S195714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Murphy RM, Watt MJ, Febbraio MA. Metabolic communication during exercise. Nat Metab. 2020;2:805–816. doi: 10.1038/s42255-020-0258-x. [DOI] [PubMed] [Google Scholar]
- 7.Gururaja Rao S. Mitochondrial changes in cancer. Handb Exp Pharmacol. 2017;240:211–227. doi: 10.1007/164_2016_40. [DOI] [PubMed] [Google Scholar]
- 8.Chen Y, Chen Z, Feng JH, Chen YB, Liao NS, Su Y, Zou CY. Metabolic profiling of normal hepatocyte and hepatocellular carcinoma cells via 1H nuclear magnetic resonance spectroscopy. Cell Biol Int. 2018;42:425–434. doi: 10.1002/cbin.10911. [DOI] [PubMed] [Google Scholar]
- 9.Guppy M. The hypoxic core: A possible answer to the cancer paradox. Biochem Biophys Res Commun. 2002;299:676–680. doi: 10.1016/S0006-291X(02)02710-9. [DOI] [PubMed] [Google Scholar]
- 10.Kalyanaraman B. Teaching the basics of cancer metabolism: Developing antitumor strategies by exploiting the differences between normal and cancer cell metabolism. Redox Biol. 2017;12:833–842. doi: 10.1016/j.redox.2017.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Levine AJ, Puzio-Kuter AM. The control of the metabolic switch in cancers by oncogenes and tumor suppressor genes. Science. 2010;330:1340–1344. doi: 10.1126/science.1193494. [DOI] [PubMed] [Google Scholar]
- 12.Stine ZE, Walton ZE, Altman BJ, Hsieh AL, Dang CV. MYC, metabolism, and cancer. Cancer Discov. 2015;5:1024–1039. doi: 10.1158/2159-8290.CD-15-0507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Moretti M, Bennett J, Tornatore L, Thotakura AK, Franzoso G. Cancer: NF-κB regulates energy metabolism. Int J Biochem Cell Biol. 2012;44:2238–2243. doi: 10.1016/j.biocel.2012.08.002. [DOI] [PubMed] [Google Scholar]
- 14.Courtnay R, Ngo DC, Malik N, Ververis K, Tortorella SM, Karagiannis TC. Cancer metabolism and the Warburg effect: The role of HIF-1 and PI3K. Mol Biol Rep. 2015;42:841–851. doi: 10.1007/s11033-015-3858-x. [DOI] [PubMed] [Google Scholar]
- 15.Christofk HR, Vander Heiden MG, Harris MH, Ramanathan A, Gerszten RE, Wei R, Fleming MD, Schreiber SL, Cantley LC. The M2 splice isoform of pyruvate kinase is important for cancer metabolism and tumour growth. Nature. 2008;452:230–233. doi: 10.1038/nature06734. [DOI] [PubMed] [Google Scholar]
- 16.Ward PS, Thompson CB. Metabolic reprogramming: A cancer hallmark even warburg did not anticipate. Cancer Cell. 2012;21:297–308. doi: 10.1016/j.ccr.2012.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pavlova NN, Thompson CB. The emerging hallmarks of cancer metabolism. Cell Metab. 2016;23:27–47. doi: 10.1016/j.cmet.2015.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liberti MV, Locasale JW. The warburg effect: How does it benefit cancer cells? Trends Biochem Sci. 2016;41:211–218. doi: 10.1016/j.tibs.2016.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nagao A, Kobayashi M, Koyasu S, Chow CCT, Harada H. HIF-1-dependent reprogramming of glucose metabolic pathway of cancer cells and its therapeutic significance. Int J Mol Sci. 2019;20:238. doi: 10.3390/ijms20020238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Musharraf SG, Mazhar S, Choudhary MI, Rizi N, Atta-ur-Rahman Plasma metabolite profiling and chemometric analyses of lung cancer along with three controls through gas chromatography-mass spectrometry. Sci Rep. 2015;5:8607. doi: 10.1038/srep08607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: The metabolic requirements of cell proliferation. Science. 2009;324:1029–1033. doi: 10.1126/science.1160809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Patra KC, Hay N. The pentose phosphate pathway and cancer. Trends Biochem Sci. 2014;39:347–354. doi: 10.1016/j.tibs.2014.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lam C, Low JY, Tran PT, Wang H. The hexosamine biosynthetic pathway and cancer: Current knowledge and future therapeutic strategies. Cancer Lett. 2021;503:11–18. doi: 10.1016/j.canlet.2021.01.010. [DOI] [PubMed] [Google Scholar]
- 24.Amelio I, Cutruzzola F, Antonov A, Agostini M, Melino G. Serine and glycine metabolism in cancer. Trends Biochem Sci. 2014;39:191–198. doi: 10.1016/j.tibs.2014.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liu Y, Cao Y, Zhang W, Bergmeier S, Qian Y, Akbar H, Colvin R, Ding J, Tong L, Wu S, et al. A small-molecule inhibitor of glucose transporter 1 downregulates glycolysis, induces cell-cycle arrest, and inhibits cancer cell growth in vitro and in vivo. Mol Cancer Ther. 2012;11:1672–1682. doi: 10.1158/1535-7163.MCT-12-0131. [DOI] [PubMed] [Google Scholar]
- 26.Xu S, Herschman HR. A tumor agnostic therapeutic strategy for hexokinase 1-Null/Hexokinase 2-positive cancers. Cancer Res. 2019;79:5907–5914. doi: 10.1158/0008-5472.CAN-19-1789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhang XY, Zhang M, Cong Q, Zhang MX, Zhang MY, Lu YY, Xu CJ. Hexokinase 2 confers resistance to cisplatin in ovarian cancer cells by enhancing cisplatin-induced autophagy. Int J Biochem Cell Biol. 2018;95:9–16. doi: 10.1016/j.biocel.2017.12.010. [DOI] [PubMed] [Google Scholar]
- 28.Maschek G, Savaraj N, Priebe W, Braunschweiger P, Hamilton K, Tidmarsh GF, De Young LR, Lampidis TJ. 2-deoxy-D-glucose increases the efficacy of adriamycin and paclitaxel in human osteosarcoma and non-small cell lung cancers in vivo. Cancer Res. 2004;64:31–34. doi: 10.1158/0008-5472.CAN-03-3294. [DOI] [PubMed] [Google Scholar]
- 29.Kim SM, Yun MR, Hong YK, Solca F, Kim JH, Kim HJ, Cho BC. Glycolysis inhibition sensitizes non-small cell lung cancer with T790M mutation to irreversible EGFR inhibitors via translational suppression of Mcl-1 by AMPK activation. Mol Cancer Ther. 2013;12:2145–2156. doi: 10.1158/1535-7163.MCT-12-1188. [DOI] [PubMed] [Google Scholar]
- 30.Lin S, Li Y, Wang D, Huang C, Marino D, Bollt O, Wu C, Taylor MD, Li W, DeNicola GM, et al. Fascin promotes lung cancer growth and metastasis by enhancing glycolysis and PFKFB3 expression. Cancer Lett. 2021;518:230–242. doi: 10.1016/j.canlet.2021.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shen J, Jin Z, Lv H, Jin K, Jonas K, Zhu C, Chen B. PFKP is highly expressed in lung cancer and regulates glucose metabolism. Cell Oncol (Dordr) 2020;43:617–629. doi: 10.1007/s13402-020-00508-6. [DOI] [PubMed] [Google Scholar]
- 32.Guo W, Zhang Y, Chen T, Wang Y, Xue J, Zhang Y, Xiao W, Mo X, Lu Y. Efficacy of RNAi targeting of pyruvate kinase M2 combined with cisplatin in a lung cancer model. J Cancer Res Clin Oncol. 2011;137:65–72. doi: 10.1007/s00432-010-0860-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shi HS, Li D, Zhang J, Wang YS, Yang L, Zhang HL, Wang XH, Mu B, Wang W, Ma Y, et al. Silencing of pkm2 increases the efficacy of docetaxel in human lung cancer xenografts in mice. Cancer Sci. 2010;101:1447–1453. doi: 10.1111/j.1349-7006.2010.01562.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Morita M, Sato T, Nomura M, Sakamoto Y, Inoue Y, Tanaka R, Ito S, Kurosawa K, Yamaguchi K, Sugiura Y, et al. PKM1 confers metabolic advantages and promotes cell-autonomous tumor cell growth. Cancer Cell. 2018;33:355–367. e7. doi: 10.1016/j.ccell.2018.02.004. [DOI] [PubMed] [Google Scholar]
- 35.Israelsen WJ, Vander Heiden MG. Pyruvate kinase: Function, regulation and role in cancer. Semin Cell Dev Biol. 2015;43:43–51. doi: 10.1016/j.semcdb.2015.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang X, Guo M, Fan J, Lv Z, Huang Q, Han J, Wu F, Hu G, Xu J, Jin Y. Prognostic significance of serum LDH in small cell lung cancer: A systematic review with meta-analysis. Cancer Biomark. 2016;16:415–423. doi: 10.3233/CBM-160580. [DOI] [PubMed] [Google Scholar]
- 37.Costa Leite T, Da Silva D, Guimaraes Coelho R, Zancan P, Sola-Penna M. Lactate favours the dissociation of skeletal muscle 6-phosphofructo-1-kinase tetramers down-regulating the enzyme and muscle glycolysis. Biochem J. 2007;408:123–130. doi: 10.1042/BJ20070687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hui S, Ghergurovich JM, Morscher RJ, Jang C, Teng X, Lu W, Esparza LA, Reya T, Le Zhan, Yanxiang Guo J, et al. Glucose feeds the TCA cycle via circulating lactate. Nature. 2017;551:115–118. doi: 10.1038/nature24057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Colegio OR, Chu NQ, Szabo AL, Chu T, Rhebergen AM, Jairam V, Cyrus N, Brokowski CE, Eisenbarth SC, Phillips GM, et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature. 2014;513:559–563. doi: 10.1038/nature13490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhao Y, Liu H, Liu Z, Ding Y, Ledoux SP, Wilson GL, Voellmy R, Lin Y, Lin W, Nahta R, et al. Overcoming trastuzumab resistance in breast cancer by targeting dysregulated glucose metabolism. Cancer Res. 2011;71:4585–4597. doi: 10.1158/0008-5472.CAN-11-0127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhou M, Zhao Y, Ding Y, Liu H, Liu Z, Fodstad O, Riker AI, Kamarajugadda S, Lu J, Owen LB, et al. Warburg effect in chemosensitivity: Targeting lactate dehydrogenase-A re-sensitizes taxol-resistant cancer cells to taxol. Mol Cancer. 2010;9:33. doi: 10.1186/1476-4598-9-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Faubert B, Li KY, Cai L, Hensley CT, Kim J, Zacharias LG, Yang C, Do QN, Doucette S, Burguete D, et al. Lactate metabolism in human lung tumors. Cell. 2017;171:358–371. e9. doi: 10.1016/j.cell.2017.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hensley CT, Faubert B, Yuan Q, Lev-Cohain N, Jin E, Kim J, Jiang L, Ko B, Skelton R, Loudat L, et al. Metabolic heterogeneity in human lung tumors. Cell. 2016;164:681–694. doi: 10.1016/j.cell.2015.12.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xie N, Tan Z, Banerjee S, Cui H, Ge J, Liu RM, Bernard K, Thannickal VJ, Liu G. Glycolytic reprogramming in myofibroblast differentiation and lung fibrosis. Am J Respir Crit Care Med. 2015;192:1462–1474. doi: 10.1164/rccm.201504-0780OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chang YC, Chan YC, Chang WM, Lin YF, Yang CJ, Su CY, Huang MS, Wu ATH, Hsiao M. Feedback regulation of ALDOA activates the HIF-1α/MMP9 axis to promote lung cancer progression. Cancer Lett. 2017;403:28–36. doi: 10.1016/j.canlet.2017.06.001. [DOI] [PubMed] [Google Scholar]
- 46.Romero OA, Torres-Diz M, Pros E, Savola S, Gomez A, Moran S, Saez C, Iwakawa R, Villanueva A, Montuenga LM, et al. MAX inactivation in small cell lung cancer disrupts MYC-SWI/SNF programs and is synthetic lethal with BRG1. Cancer Discov. 2014;4:292–303. doi: 10.1158/2159-8290.CD-13-0799. [DOI] [PubMed] [Google Scholar]
- 47.de Koning TJ. Amino acid synthesis deficiencies. J Inherit Metab Dis. 2017;40:609–620. doi: 10.1007/s10545-017-0063-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cluntun AA, Lukey MJ, Cerione RA, Locasale JW. Glutamine metabolism in cancer: Understanding the heterogeneity. Trends Cancer. 2017;3:169–180. doi: 10.1016/j.trecan.2017.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Altman BJ, Stine ZE, Dang CV. From Krebs to clinic: Glutamine metabolism to cancer therapy. Nat Rev Cancer. 2016;16:773. doi: 10.1038/nrc.2016.131. [DOI] [PubMed] [Google Scholar]
- 50.Scalise M, Pochini L, Galluccio M, Console L, Indiveri C. Glutamine transport and mitochondrial metabolism in cancer cell growth. Front Oncol. 2017;7:306. doi: 10.3389/fonc.2017.00306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Serizawa M, Kusuhara M, Zangiacomi V, Urakami K, Watanabe M, Takahashi T, Yamaguchi K, Yamamoto N, Koh Y. Identification of metabolic signatures associated with erlotinib resistance of non-small cell lung cancer cells. Anticancer Res. 2014;34:2779–2787. [PubMed] [Google Scholar]
- 52.Dunphy MPS, Harding JJ, Venneti S, Zhang H, Burnazi EM, Bromberg J, Omuro AM, Hsieh JJ, Mellinghoff IK, Staton K, et al. In vivo PET assay of tumor glutamine flux and metabolism: In-Human Trial of 18F-(2S,4R)-4-Fluoroglutamine. Radiology. 2018;287:667–675. doi: 10.1148/radiol.2017162610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Jeon SM, Chandel NS, Hay N. AMPK regulates NADPH homeostasis to promote tumour cell survival during energy stress. Nature. 2012;485:661–665. doi: 10.1038/nature11066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yu Y, Newman H, Shen L, Sharma D, Hu G, Mirando AJ, Zhang H, Knudsen E, Zhang GF, Hilton MJ, Karner CM. Glutamine metabolism regulates proliferation and lineage allocation in skeletal stem cells. Cell Metab. 2019;29:966–978. e4. doi: 10.1016/j.cmet.2019.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Simon J, Nunez-Garcia M, Fernandez-Tussy P, Barbier-Torres L, Fernández-Ramos D, Gómez-Santos B, Buqué X, Lopitz-Otsoa F, Goikoetxea-Usandizaga N, Serrano-Macia M, et al. Targeting hepatic glutaminase 1 ameliorates non-alcoholic steatohepatitis by restoring very-low-density lipoprotein triglyceride assembly. Cell Metab. 2020;31:605–622. e10. doi: 10.1016/j.cmet.2020.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Mates JM, Campos-Sandoval JA, Marquez J. Glutaminase isoenzymes in the metabolic therapy of cancer. Biochim Biophys Acta Rev Cancer. 2018;1870:158–164. doi: 10.1016/j.bbcan.2018.07.007. [DOI] [PubMed] [Google Scholar]
- 57.Daemen A, Liu B, Song K, Kwong M, Gao M, Hong R, Nannini M, Peterson D, Liederer BM, de la Cruz C, et al. Pan-Cancer metabolic signature predicts co-dependency on glutaminase and de novo glutathione synthesis linked to a high-mesenchymal cell state. Cell Metab. 2018;28:383–399. e389. doi: 10.1016/j.cmet.2018.06.003. [DOI] [PubMed] [Google Scholar]
- 58.Han T, Zhan W, Gan M, Liu F, Yu B, Chin YE, Wang JB. Phosphorylation of glutaminase by PKCε is essential for its enzymatic activity and critically contributes to tumorigenesis. Cell Res. 2018;28:655–669. doi: 10.1038/s41422-018-0021-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Jacque N, Ronchetti AM, Larrue C, Meunier G, Birsen R, Willems L, Saland E, Decroocq J, Maciel TT, Lambert M, et al. Targeting glutaminolysis has antileukemic activity in acute myeloid leukemia and synergizes with BCL-2 inhibition. Blood. 2015;126:1346–1356. doi: 10.1182/blood-2015-01-621870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.van den Heuvel AP, Jing J, Wooster RF, Bachman KE. Analysis of glutamine dependency in non-small cell lung cancer: GLS1 splice variant GAC is essential for cancer cell growth. Cancer Biol Ther. 2012;13:1185–1194. doi: 10.4161/cbt.21348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kim J, Lee HM, Cai F, Ko B, Yang C, Lieu EL, Muhammad N, Rhyne S, Li K, Haloul M, et al. The hexosamine biosynthesis pathway is a targetable liability in KRAS/LKB1 mutant lung cancer. Nat Metab. 2020;2:1401–1412. doi: 10.1038/s42255-020-00316-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sezgin E, Levental I, Mayor S, Eggeling C. The mystery of membrane organization: Composition, regulation and roles of lipid rafts. Nat Rev Mol Cell Biol. 2017;18:361–374. doi: 10.1038/nrm.2017.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Snaebjornsson MT, Janaki-Raman S, Schulze A. Greasing the wheels of the cancer machine: The role of lipid metabolism in cancer. Cell Metab. 2020;31:62–76. doi: 10.1016/j.cmet.2019.11.010. [DOI] [PubMed] [Google Scholar]
- 64.Menendez JA, Lupu R. Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat Rev Cancer. 2007;7:763–777. doi: 10.1038/nrc2222. [DOI] [PubMed] [Google Scholar]
- 65.Corbet C, Feron O. Cancer cell metabolism and mitochondria: Nutrient plasticity for TCA cycle fueling. Biochim Biophys Acta Rev Cancer. 2017;1868:7–15. doi: 10.1016/j.bbcan.2017.01.002. [DOI] [PubMed] [Google Scholar]
- 66.Svensson RU, Parker SJ, Eichner LJ, Kolar MJ, Wallace M, Brun SN, Lombardo PS, Van Nostrand JL, Hutchins A, Vera L, et al. Inhibition of acetyl-CoA carboxylase suppresses fatty acid synthesis and tumor growth of non-small-cell lung cancer in preclinical models. Nat Med. 2016;22:1108–1119. doi: 10.1038/nm.4181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tadros S, Shukla SK, King RJ, Gunda V, Vernucci E, Abrego J, Chaika NV, Yu F, Lazenby AJ, Berim L, et al. De novo lipid synthesis facilitates gemcitabine resistance through endoplasmic reticulum stress in pancreatic cancer. Cancer Res. 2017;77:5503–5517. doi: 10.1158/0008-5472.CAN-16-3062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Buckley D, Duke G, Heuer TS, O'Farrell M, Wagman AS, McCulloch W, Kemble G. Fatty acid synthase-Modern tumor cell biology insights into a classical oncology target. Pharmacol Ther. 2017;177:23–31. doi: 10.1016/j.pharmthera.2017.02.021. [DOI] [PubMed] [Google Scholar]
- 69.Currie E, Schulze A, Zechner R, Walther TC, Farese RV., Jr Cellular fatty acid metabolism and cancer. Cell Metab. 2013;18:153–161. doi: 10.1016/j.cmet.2013.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Shen M, Tsai Y, Zhu R, Keng PC, Chen Y, Chen Y, Lee SO. FASN-TGF-β1-PD-L1 axis contributes to the development of resistance to NK cell cytotoxicity of cisplatin-resistant lung cancer cells. Biochim Biophys Acta Mol Cell Biol Lipids. 2018;1863:313–322. doi: 10.1016/j.bbalip.2017.12.012. [DOI] [PubMed] [Google Scholar]
- 71.Ali A, Levantini E, Teo JT, Goggi J, Clohessy JG, Wu CS, Chen L, Yang H, Krishnan I, Kocher O, et al. Fatty acid synthase mediates EGFR palmitoylation in EGFR mutated non-small cell lung cancer. EMBO Mol Med. 2018;10:e8313. doi: 10.15252/emmm.201708313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Falchook G, Infante J, Arkenau HT, Patel MR, Dean E, Borazanci E, Brenner A, Cook N, Lopez J, Pant S, et al. First-in-human study of the safety, pharmacokinetics, and pharmacodynamics of first-in-class fatty acid synthase inhibitor TVB-2640 alone and with a taxane in advanced tumors. EClinicalMedicine. 2021;34:100797. doi: 10.1016/j.eclinm.2021.100797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Jones SF, Infante JR. Molecular pathways: Fatty acid synthase. Clin Cancer Res. 2015;21:5434–5438. doi: 10.1158/1078-0432.CCR-15-0126. [DOI] [PubMed] [Google Scholar]
- 74.Dang L, Yen K, Attar EC. IDH mutations in cancer and progress toward development of targeted therapeutics. Ann Oncol. 2016;27:599–608. doi: 10.1093/annonc/mdw013. [DOI] [PubMed] [Google Scholar]
- 75.Chen C, Liu Y, Lu C, Cross JR, Morris JP IV, Shroff AS, Ward PS, Bradner JE, Thompson C, Lowe SW. Cancer-associated IDH2 mutants drive an acute myeloid leukemia that is susceptible to Brd4 inhibition. Genes Dev. 2013;27:1974–1985. doi: 10.1101/gad.226613.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Migita T, Narita T, Nomura K, Miyagi E, Inazuka F, Matsuura M, Ushijima M, Mashima T, Seimiya H, Satoh Y, et al. ATP citrate lyase: Activation and therapeutic implications in non-small cell lung cancer. Cancer Res. 2008;68:8547–8554. doi: 10.1158/0008-5472.CAN-08-1235. [DOI] [PubMed] [Google Scholar]
- 77.Chajes V, Cambot M, Moreau K, Lenoir GM, Joulin V. Acetyl-CoA carboxylase alpha is essential to breast cancer cell survival. Cancer Res. 2006;66:5287–5294. doi: 10.1158/0008-5472.CAN-05-1489. [DOI] [PubMed] [Google Scholar]
- 78.Carrer A, Trefely S, Zhao S, Campbell SL, Norgard RJ, Schultz KC, Sidoli S, Parris JLD, Affronti HC, Sivanand S, et al. Acetyl-CoA metabolism supports multistep pancreatic tumorigenesis. Cancer Discov. 2019;9:416–435. doi: 10.1158/2159-8290.CD-18-0567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zaidi N, Swinnen JV, Smans K. ATP-citrate lyase: A key player in cancer metabolism. Cancer Res. 2012;72:3709–3714. doi: 10.1158/0008-5472.CAN-11-4112. [DOI] [PubMed] [Google Scholar]
- 80.Hatzivassiliou G, Zhao F, Bauer DE, Andreadis C, Shaw AN, Dhanak D, Hingorani SR, Tuveson DA, Thompson CB. ATP citrate lyase inhibition can suppress tumor cell growth. Cancer Cell. 2005;8:311–321. doi: 10.1016/j.ccr.2005.09.008. [DOI] [PubMed] [Google Scholar]
- 81.Lin R, Tao R, Gao X, Li T, Zhou X, Guan KL, Xiong Y, Lei QY. Acetylation stabilizes ATP-citrate lyase to promote lipid biosynthesis and tumor growth. Mol Cell. 2013;51:506–518. doi: 10.1016/j.molcel.2013.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Niu J, Sun Y, Chen B, Zheng B, Jarugumilli GK, Walker SR, Hata AN, Mino-Kenudson M, Frank DA, Wu X. Fatty acids and cancer-amplified ZDHHC19 promote STAT3 activation through S-palmitoylation. Nature. 2019;573:139–143. doi: 10.1038/s41586-019-1511-x. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 83.Mashima T, Oh-hara T, Sato S, Mochizuki M, Sugimoto Y, Yamazaki K, Hamada J, Tada M, Moriuchi T, Ishikawa Y, et al. p53-defective tumors with a functional apoptosome-mediated pathway: A new therapeutic target. J Natl Cancer Inst. 2005;97:765–777. doi: 10.1093/jnci/dji133. [DOI] [PubMed] [Google Scholar]
- 84.Yarla NS, Bishayee A, Sethi G, Reddanna P, Kalle AM, Dhananjaya BL, Dowluru KS, Chintala R, Duddukuri GR. Targeting arachidonic acid pathway by natural products for cancer prevention and therapy. Semin Cancer Biol. 2016;40–41:48–81. doi: 10.1016/j.semcancer.2016.02.001. [DOI] [PubMed] [Google Scholar]
- 85.Murray M, Hraiki A, Bebawy M, Pazderka C, Rawling T. Anti-tumor activities of lipids and lipid analogues and their development as potential anticancer drugs. Pharmacol Ther. 2015;150:109–128. doi: 10.1016/j.pharmthera.2015.01.008. [DOI] [PubMed] [Google Scholar]
- 86.Vannitamby A, Saad MI, Aloe C, Wang H, Kumar B, Vlahos R, Selemidis S, Irving L, Steinfort D, Jenkins BJ, Bozinovski S. Aspirin-triggered resolvin D1 reduces proliferation and the neutrophil to lymphocyte ratio in a mutant KRAS-driven lung adenocarcinoma model. Cancers (Basel) 2021;13:3224. doi: 10.3390/cancers13133224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Riedl K, Krysan K, Pold M, Dalwadi H, Heuze-Vourc'h N, Dohadwala M, Liu M, Cui X, Figlin R, Mao JT, et al. Multifaceted roles of cyclooxygenase-2 in lung cancer. Drug Resist Updat. 2004;7:169–184. doi: 10.1016/j.drup.2004.04.003. [DOI] [PubMed] [Google Scholar]
- 88.Xin C, Chu L, Zhang L, Geng D, Wang Y, Sun D, Sui P, Zhao X, Gong Z, Sui M, Zhang W. Expression of cytosolic phospholipase A2 (cPLA2)-arachidonic acid (AA)-Cyclooxygenase-2 (COX-2) pathway factors in lung cancer patients and its implication in lung cancer early detection and prognosis. Med Sci Monit. 2019;25:5543–5551. doi: 10.12659/MSM.915314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Yue S, Li J, Lee SY, Lee HJ, Shao T, Song B, Cheng L, Masterson TA, Liu X, Ratliff TL, Cheng JX. Cholesteryl ester accumulation induced by PTEN loss and PI3K/AKT activation underlies human prostate cancer aggressiveness. Cell Metab. 2014;19:393–406. doi: 10.1016/j.cmet.2014.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kopecka J, Trouillas P, Gasparovic AC, Gazzano E, Assaraf YG, Riganti C. Phospholipids and cholesterol: Inducers of cancer multidrug resistance and therapeutic targets. Drug Resist Updat. 2020;49:100670. doi: 10.1016/j.drup.2019.100670. [DOI] [PubMed] [Google Scholar]
- 91.Yeganeh B, Wiechec E, Ande SR, Sharma P, Moghadam AR, Post M, Freed DH, Hashemi M, Shojaei S, Zeki AA, Ghavami S. Targeting the mevalonate cascade as a new therapeutic approach in heart disease, cancer and pulmonary disease. Pharmacol Ther. 2014;143:87–110. doi: 10.1016/j.pharmthera.2014.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Gorin A, Gabitova L, Astsaturov I. Regulation of cholesterol biosynthesis and cancer signaling. Curr Opin Pharmacol. 2012;12:710–716. doi: 10.1016/j.coph.2012.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Li D, Long W, Huang R, Chen Y, Xia M. 27-Hydroxycholesterol inhibits sterol regulatory element-binding protein 1 activation and hepatic lipid accumulation in mice. Obesity (Silver Spring) 2018;26:713–722. doi: 10.1002/oby.22130. [DOI] [PubMed] [Google Scholar]
- 94.Nelson ER, Wardell SE, Jasper JS, Park S, Suchindran S, Howe MK, Carver NJ, Pillai RV, Sullivan PM, Sondhi V, et al. 27-Hydroxycholesterol links hypercholesterolemia and breast cancer pathophysiology. Science. 2013;342:1094–1098. doi: 10.1126/science.1241908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wang B, Tontonoz P. Liver X receptors in lipid signalling and membrane homeostasis. Nat Rev Endocrinol. 2018;14:452–463. doi: 10.1038/s41574-018-0037-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Gibson DA, Collins F, Cousins FL, Esnal Zufiaurre A, Saunders PTK. The impact of 27-hydroxycholesterol on endometrial cancer proliferation. Endocr Relat Cancer. 2018;25:381–391. doi: 10.1530/ERC-17-0449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Wu Y, Yu DD, Hu Y, Cao HX, Yu SR, Liu SW, Feng JF. LXR ligands sensitize EGFR-TKI-resistant human lung cancer cells in vitro by inhibiting Akt activation. Biochem Biophys Res Commun. 2015;467:900–905. doi: 10.1016/j.bbrc.2015.10.047. [DOI] [PubMed] [Google Scholar]
- 98.Lo Sasso G, Bovenga F, Murzilli S, Salvatore L, Di Tullio G, Martelli N, D'Orazio A, Rainaldi S, Vacca M, Mangia A, et al. Liver X receptors inhibit proliferation of human colorectal cancer cells and growth of intestinal tumors in mice. Gastroenterology. 2013;144:1497–1507. 1507, e1–13. doi: 10.1053/j.gastro.2013.02.005. [DOI] [PubMed] [Google Scholar]
- 99.Hiramitsu S, Ishikawa T, Lee WR, Khan T, Crumbley C, Khwaja N, Zamanian F, Asghari A, Sen M, Zhang Y, et al. Estrogen receptor beta-mediated modulation of lung cancer cell proliferation by 27-hydroxycholesterol. Front Endocrinol (Lausanne) 2018;9:470. doi: 10.3389/fendo.2018.00470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Zhang L, Liu M, Liu J, Li X, Yang M, Su B, Lin Y. 27-Hydroxycholesterol enhanced osteoclastogenesis in lung adenocarcinoma microenvironment. J Cell Physiol. 2019;234:12692–12700. doi: 10.1002/jcp.27883. [DOI] [PubMed] [Google Scholar]
- 101.Chen Q, Pan Z, Zhao M, Wang Q, Qiao C, Miao L, Ding X. High cholesterol in lipid rafts reduces the sensitivity to EGFR-TKI therapy in non-small cell lung cancer. J Cell Physiol. 2018;233:6722–6732. doi: 10.1002/jcp.26351. [DOI] [PubMed] [Google Scholar]
- 102.Li J, Yan H, Zhao L, Jia W, Yang H, Liu L, Zhou X, Miao P, Sun X, Song S, et al. Inhibition of SREBP increases gefitinib sensitivity in non-small cell lung cancer cells. Oncotarget. 2016;7:52392–52403. doi: 10.18632/oncotarget.10721. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.