Abstract
Patients with polymerase epsilon (POLE) exonuclease domain mutation (EDM) exhibits distinct clinical characteristics and extremely high tumor mutation burden (TMB). There is a paucity of data on the therapeutic efficacy of immune checkpoint inhibitors (ICIs) for the treatment of colorectal cancer liver metastases (CRLM) patients with POLE EDM. Clinical characteristics, radiological and pathological response, as well as oncological outcomes of four CRLM patients harboring POLE EDM and treated by ICI plus chemotherapy were retrospectively collected and analyzed. TMB and genomic mutation profiling were also assessed in resected CRLM patients harboring different molecular characteristics. The four CRLM patients received toripalimab or sintilimab plus chemotherapy (FOLFOX or FOLFIRI or XELOX) with or without bevacizumab after POLE EDM were detected. All four patients achieved a radiological partial response. Staged or simultaneous complete surgical resection of the primary tumor and liver metastases was conducted. Pathological complete response was achieved in all four patients. After a median follow-up of 14 (range 9–20) months, all four patients maintained non-evidence of disease status until the last follow-up. POLE EDM patients showed a larger set of mutational genes compared with non-POLE EDM patients. TMB of patients harboring POLE EDM was significantly higher than those with microsatellite instability-high (median, 313.92 vs 42.24 mutations/Mb, p<0.05), POLE non-EDM (313.92 vs 4.80, p<0.001), and MSS subtypes (313.92 vs 4.80, p<0.001). Despite being a rare phenotype, CRLM patients with POLE EDM exhibit ultra-high TMB and, more importantly, significant clinical response to ICI-based combination therapy. Therefore, the complete sequencing of POLE exonuclease domains is recommended in CRLM patients clinically.
Keywords: Biomarkers, Tumor; Gastrointestinal Neoplasms; Immunotherapy
Background
The clinical outcomes of patients with colorectal cancer liver metastases (CRLM) exhibit great heterogeneity. Previous studies revealed that more than 50% of CRLM patients would experience recurrence after initial liver resection.1 2 CRLM patients who respond well to preoperative treatment typically have a favorable prognosis, especially those who achieve pathological complete response (pCR).3 Immune checkpoint inhibitors (ICIs) directed against programmed death-1 (PD1) protein are highly effective and have become the preferred choice for the treatment of patients with mismatch repair-deficient (dMMR) metastatic colorectal cancer.4 However, given the small portion of patients with dMMR, more markers that could predict ICI response are urgently needed.
DNA polymerase epsilon (POLE) encodes the catalytic subunit of DNA polymerase epsilon, an enzyme involved in DNA replication and repair.5 Exonuclease domain mutations of POLE (POLE EDM) are detected in a variety of malignant tumors, most frequently in endometrial (6%–12%) and colorectal cancer (1%–2%).6 7 POLE EDM endometrial cancer is characterized by a robust intratumoral T-cell response, possibly because of an abundance of neoantigens caused by extremely high tumor mutation, ultimately leading to an excellent prognosis.8 In stage II colorectal cancer, somatic POLE EDM display enhanced intratumoral immune responses, evidenced by increased CD8 +tumor infiltration lymphocytes and CD8A expression.9
This study examined four CRLM patients harboring the POLE EDM who responed to ICI-based combination treatment. The clinical treatment, radiological response, postoperative pathological findings, and oncological outcomes were reported and assessed. Furthermore, we analyzed CD3, CD8, and programmed death-ligand 1 (PDL1) expression levels in tumor tissues as well as the dynamic change of circulating cell-free DNA methylation during treatment. The gene mutation profiling and tumor mutation burden (TMB) of POLE EDM patients with resected CRLM patients harboring distinct molecular features, that is, POLE non-EDM, microsatellite instability-high (MSI-H) and microsatellite stable (MSS) were also compared.
Methods
Study design
The design of this study is shown in online supplemental figure S1. A total of four patients diagnosed with CRLM harboring POLE EDM and treated by ICI plus chemotherapy between 2019 and 2020 at Sun Yat-sen University Cancer Center, Guangzhou, China, were retrospectively included. Clinical treatment course, radiological response, pathological findings (tumor regression, lymphocyte infiltration), as well as circulating free DNA (cfDNA) methylation dynamics of the four patients were collected and shown (online supplemental figure S1A). Additionally, gene profiles and TMB of these four patients with POLE EDM were showed and compared with other 292 resected CRLM patients harboring distinct molecular features, that is, POLE non-EDM, MSI-H, and MSS, treated between 2010 and 2020 at our center (online supplemental figure S1B). Patients were categorized into the above four molecular subgroups based on their POLE mutation sites (oncogenic mutation or not) and microsatellite status. POLE EDM was defined as oncogenic mutations in P286R, S459F, V411L, etc, referring to the list of the oncogenic POLE alterations at OncoKB (https://www.oncokb.org/gene/POLE).10POLE non-EDM refers to mutations in the POLE non-oncogenic sites, for example, P1832F, R1390H, A25V, etc. We also analyzed the molecular characteristics and TMB of metastatic colorectal cancer from the Memorial Sloan Kettering Cancer Center (MSKCC) cohort.11
jitc-2022-004487supp001.pdf (85.9KB, pdf)
Treatment
Patients harboring POLE EDM received ICI plus chemotherapy with or without bevacizumab every 2–3 weeks. The ICI used was one of the two humanized IgG4 monoclonal PD-1 antibodies, toripalimab (JS-001, Shanghai Junshi Bioscience)12 or sintilimab (IBI308, Innovent Biologics and Eli Lilly and Company),13 both of which were developed and approved for the treatment of several solid tumors in China. Chemotherapy regimens consisted of fluorouracil and oxaliplatin (FOLFOX or XELOX) or irinotecan (FOLFIRI). Radiological response was assessed using the Response Evaluation Criteria in Solid Tumors V.1.1.14 Adverse events were graded according to the National Cancer Institute Common Terminology Criteria, V.4.0.
Pathology assessment and immunohistochemistry analysis
A thorough pathological examination of the resected tumors was conducted. No residual tumor cells in resected specimens (primary tumor, regional lymph nodes, and liver metastases) by routine H&E staining was defined as pCR. Formalin-fixed, paraffin-embedded sections were obtained from both pretreatment biopsies and resection specimens for further examination of CD3+ (ZSGS-BIO, Beijing, China; Catalog No. ZA0503) and CD8+ (ZSGS-BIO, Beijing, China; Catalog No. ZA0508) tumor-infiltrating T lymphocytes using specific antibodies and immunohistochemistry (IHC) analysis. To quantitatively analyze lymphocytes infiltration, we used ImageJ software (https://imagej.nih.gov/ij/, National Institute of Health, Bethesda, Maryland, USA) to calculate the number of CD3 +and CD8+T cells and compared the infiltration level before and after ICI plus chemotherapy by counting the average number of staining lymphocytes in ten random fields (×20). The expression of PD-L1 protein on pretreatment biopsy tissues was determined using mouse monoclonal antibody clone 22C3 pharmDx (Dako).
NPY/SEPT9/WIF1 methylation levels in circulating free DNA
Blood samples were collected at different time points (pretreatment, post-treatment, and postoperatively), and NPY/SEPT9/WIF1 methylation levels in circulating free DNA (meth-cfDNA) were tested. cfDNA was extracted from 2 mL of serum using the QIAamp DNA Blood Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s protocol. DNA bisulfite conversion was then performed using the EZ DNA Methylation Kit (Zymo Research, USA). Finally, a total volume of 20 µL of modified DNA was obtained, which was used immediately for PCR or stored for later use at −20°C. The serum DNA methylation analyses were performed using the QX200TM Droplet DigitalTM PCR System (Bio-Rad).
Next-generation sequencing
All samples subjected to next-generation sequencing (NGS) analysis were required to have >10% of tumor cells identified by IHC. Patients were subjected to a wide panel genomic sequencing (pan-cancer 1021-gene panel, Geneplus Technology) for simultaneous detection of MSI status and mutations in 1021 cancer-related genes, including KRAS, NRAS, BRAF, HER2, TP53, SMAD4, POLE, and other genes related to tumor development and carcinogenesis. TMB was defined as the number of somatic non‐synonymous mutations per megabase of genome examined. MSI was assessed using MSIsensor.15
Statistical analysis
Statistical analyses were performed using R software V.3.5.1 (http://www.r-project.org). Comparison of TMB between different molecular groups and lymphocyte infiltration before and after ICI treatment were evaluated by the nonparametric Wilcxon test. p<0.05 at two sides was considered statistically significant.
Results
Clinical characteristics of the four patients with POLE EDM
The clinical characteristics of the four CRLM patients harboring POLE EDM are shown in table 1. All four patients were male and three of them were less than 40 years of age. They all harbored the oncogenic POLE P286R mutation in the exonuclease domain. The POLE P286R mutation was detected at initial diagnosis in two patients and progressed on first-line systematic chemotherapy in other two patients. The stage of four tumors according to the American Joint Committee on Cancer (eighth edition) in initial diagnosis was cT3-4aN1-2M1a, IVa. The primary tumor was located in the rectum, ascending colon, sigmoid colon, and transverse colon, respectively. Synchronous liver metastases were the only metastatic site in all four patients. The number of liver metastases was 8, 7, 1, and 5, respectively. The diameter of the largest lesion of liver metastases at initial diagnosis was 8.5 cm, 5.9 cm, 14.6 cm, and 6.7 cm, respectively. Hepatobiliary surgeon-assessed resectability of liver metastases at initial diagnosis were unresectable in one patient, potentially resectable in two patients and resectable in one patient. Fong’s Clinical Risk Score16 amounted to a score of 4 in three patients and 3 in one patient (case 4).
Table 1.
Clinicopathological feature, treatment, and outcome of four CRLM patients harboring POLE EDM
| Characteristics | Case 1 | Case 2 | Case 3 | Case 4 |
| Age | Late 30s | Early 70s | Late 20s | Early 30s |
| Gender | Male | Male | Male | Male |
| Primary tumor site | Rectum | Ascending colon | Sigmoid | Transverse colon |
| Stage* | cT3N1M1a, IVa | cT4aN1M1a, IVa | cT3N1M1a, IVa | cT4aN2M1a, IVa |
| Metastatic site | Liver only | Liver only | Liver only | Liver only |
| Clinical Risk Score | 4 | 4 | 4 | 3 |
| RAS/RAF status | KRAS G13D | w.t. | w.t. | w.t. |
| POLE mutation site | P286R | P286R | P286R | P286R |
| TMB (mutations/Mb) | 453.12 | 255.36 | 320.64 | 307.20 |
| Microsatellite status | MSI-H | MSS | MSS | MSS |
| Liver metastases† | ||||
| No | 8 | 7 | 1 | 5 |
| Diameter (largest lesion, cm) | 8.5 | 5.9 | 14.6 | 6.7 |
| Resectability | Unresectable | Resectable | Potential resectable | Potential resectable |
| Treatment before ICI | ||||
| Systematic (response‡) | None | FOLFOX (PD) | Bev +FOLFOX (PR) | None |
| Surgery | None | None | Colectomy, first liver metastasectomy | None |
| ICI combined treatment | ||||
| Line | First | Second | Second | First |
| Regimen | Toripalimab+FOLFOX | Sintilimab+FOLFIRI | Sintilimab+FOLFIRI | Toripalimab+Bev+XELOX |
| Cycles | 7 | 5 | 5 | 5 |
| Radiological response | PR | PR | PR | PR |
| Surgical treatment (after ICI) | ||||
| Primary site | Dixon | Colectomy | - § | Colectomy |
| Liver metastases | Resection +ablation | Resection | Resection | Resection |
| Pathological response | ||||
| Primary tumor | pCR | pCR | - § | pCR |
| Reginal lymph nodes | pCR | pCR | - § | pCR |
| Liver metastases | pCR | pCR | pCR | pCR |
| Postoperative treatment | Toripalimab | None | Sintilimab | Toripalimab |
| Survival status | Alive (NED) | Alive (NED) | Alive (NED) | Alive (NED) |
| OS (months)¶ | 12 | 18 | 7 | 14 |
*According to the American Joint Committee on Cancer (8th edition).
†At initial diagnosis.
‡Assessed by the Response Evaluation Criteria in Solid Tumors 1.1 criteria.
§Colectomy was conducted before ICI combined treatment.
¶Defined from initial ICI combined treatment to last follow-up.
Bev, bevacizumab; CRLM, colorectal cancer liver metastases; EDM, exonuclease domain mutation; FOLFIRI, fluorouracil+irinotecan+leucovorin calcium; FOLFOX, fluorouracil+oxaliplatin+leucovorin calcium; ICI, immune checkpoint inhibitor; MSI-H, microsatellite instability-high; MSS, microsatellite stable; NED, no evidence of disease; OS, overall survival; pCR, pathological complete response; PD, progressive disease; POLE, polymerase epsilon; PR, partial response; w.t., wild type; XELOX, capecitabine+oxaliplatin.
Clinical treatment and radiological response
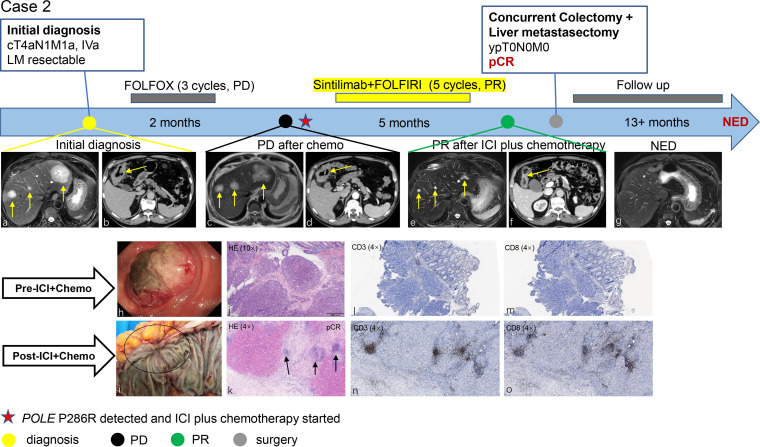
All four patients received ICI plus chemotherapy after POLE P286R mutation was detected. Case 1 received first-line toripalimab plus FOLFOX for 7 cycles and sharp tumor burden reduction was achieved in both liver lesions and primary tumor (online supplemental figure S2A). Case 2 received first-line FOLFOX and the liver lesions progressed after 3 cycles of treatment. He then received second-line sintilimab and FOLFIRI after POLE P286R mutation was detected. A very good partial response (decrease of 80% in diameter) was achieved (figure 1). Case 3 received first-line FOLFOX plus bevacizumab, radiological partial response was achieved and liver metastasectomy was conducted. However, liver recurrence occurred shortly (3 months after liver resection) and second-line sintilimab and FOLFIRI were started after POLE P286R mutation was detected (online supplemental figure S2B). Case 4 received first-line toripalimab plus XELOX and bevacizumab for five cycles (online supplemental figure S2C). Both case 3 and case 4 achieved a small partial response (approximately 35% decrease in diameter) after ICI combined treatment.
Figure 1.
Treatment timeline, radiological and pathological response to second-line sintilimab plus FOLFIRI in a 71-year-old man with stage IVa/cT4aN1M1a, right-sided CRLM harboring POLE EDM (case 2). Radiographic imaging shows resectable multiple metastases (A) in different liver segments originating from the ascending colon (B) at initial diagnoses. The patient received first-line chemotherapy (FOLFOX), which progressed shortly. (C, D) Then he received second-line ICI plus chemotherapy (sintilimab +FOLFIRI) and a notable tumor regression could be seen both in liver metastases (E) and primary tumor (F). simultaneous colectomy and liver metastasectomy were then conducted (G). primary tumor at initial diagnosis was observed using colonoscopy (H) and resection specimens of obvious shrunken colon cancer (I) after ICI plus chemotherapy treatment. H&E staining shows primary tumor at initial diagnosis (J) and pathological complete regression of liver metastases (K) after ICI plus chemotherapy. fibrosis and an infiltration with viable density of many lymphocytes (K, arrowheads) could be seen. IHC showing more CD3 and CD8 tumor-infiltrating lymphocytes (TILs) using a serial section following treatment (N, O) tumor tissue compared with pretreatment (I, M). The comparisons were statistically significant (p<0.01). IHC, immnuohistochemistry; CRLM, colorectal cancer liver metastases; ICI, immune checkpoint inhibitor; pCR, pathological complete response; PD, progressive disease; PR, partial response.
jitc-2022-004487supp002.pdf (23.9MB, pdf)
After systematic ICI plus chemotherapy, all four patients received staged or simultaneous complete surgical resection for primary tumor and liver metastases (case 3 only received liver metastasectomy because he had primary tumor resected before ICI treatment). Three of four patients received postoperative ICI monotherapy continually for a total of 1 year (preoperative plus postoperative) and one patient (case 2) refused any postoperative treatment.
Pathological findings
A thorough pathological examination of the resected tumors was conducted. Massive necrosis tissue and lymphocytes infiltration were detected, while no residual cancer cells were found microscopically both in primary tumor, regional lymphnodes, and metastatic liver lesions in all four patients, indicating pCR was achieved after ICI-based combination treatment (figure 1, online supplemental figure S2).
High CD3 +and CD8+T cell infiltration could be observed in resected specimens after ICI plus chemotherapy. In case 2 and case 3, a significant pretreatment to post-treatment increase in CD3 +and CD8+lymphocytes infiltration was found (p<0.05, Wilcoxon). Weak expression of PDL1 was found in only one patient (case 1, Combine Positve Score=5) (online supplemental figure S2A), while the other 3 patients showed negative PD-L1 expression.
NPY/SEPT9/WIF1 meth-cfDNA dynamics
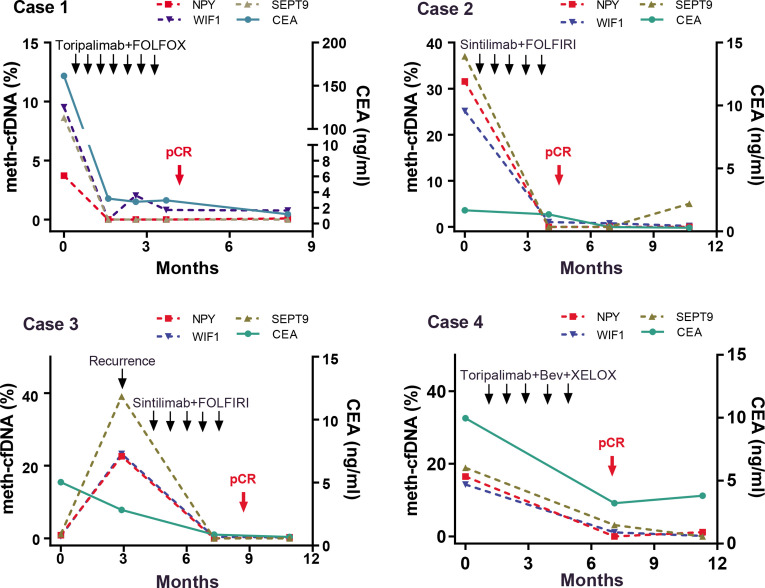
Plasma NPY/SEPT9/WIF1 meth-cfDNA was detected in different timepoints (pre-ICI treatment, post-ICI treatment, and postoperatively). The percentage of NPY, SEPT9, and WIF1 meth-cfDNA in the four patients at baseline ranged from 8.64% to 39.05%, from 9.53% to 25.14%, and from 3.73% to 31.55%, respectively. A sharp reduction in NPY/SEPT9/WIF1 meth-cfDNA was seen after ICI-based treatment in all patients. NPY/SEPT9/WIF1 meth-cfDNA level decreased to an almost non-detectable level (nearly 0%) before surgical intervention and remained at a very low level (0%–1%) postoperatively, and thereafter. The dynamic change of NPY/SEPT9/WIF1 meth-cfDNA in plasma was consistent with tumor burden and treatment response. Plasma carcinoembryonic antigen (CEA) changes during treatment in cases 1 and 4 were consistent with NPY/SEPT9/WIF1 meth-cfDNA, while in cases 2 and 3, plasma CEA was within normal limits during the entire disease course (figure 2).
Figure 2.
Dynamic changes in serum CEA and circulating NPY/SEPT9/WIF1 DNA methylation of 4 CRLM patients. Bev, bevacizumab; CEA, carcinoembryonic antigen; chemo, chemotherapy; CRLM, colorectal cancer liver metastases; ICI, immune checkpoint inhibitor; meth-cfDNA, circulating cell-free DNA methylation; pCR, pathological complete response.
Oncological outcomes and adverse events
After a median follow-up of 14 months (range 9–20), all four patients maintained a non-evidence of disease status until the last follow-up in December 2021. The progression-free survival (defined as from surgery to last follow-up) was 5+, 16+, 4+, and 9+months, respectively. The overall survival (from initial ICI treatment till the last follow-up) was 14+, 20+, 9+, and 16+months, respectively.
The perioperative ICI-based combination treatment was well tolerated. One patient (case 1) developed grade 3 neutropenia, and another patient (case 3) developed grade 3 anemia. No patient developed immune-related adverse events. All patients underwent radical resection within 1 month after the last cycle of preoperative treatment, and there were no reported surgery-related adverse events.
TMB and gene mutation profiling of POLE EDM compared with resected CRLM with other molecular subtypes
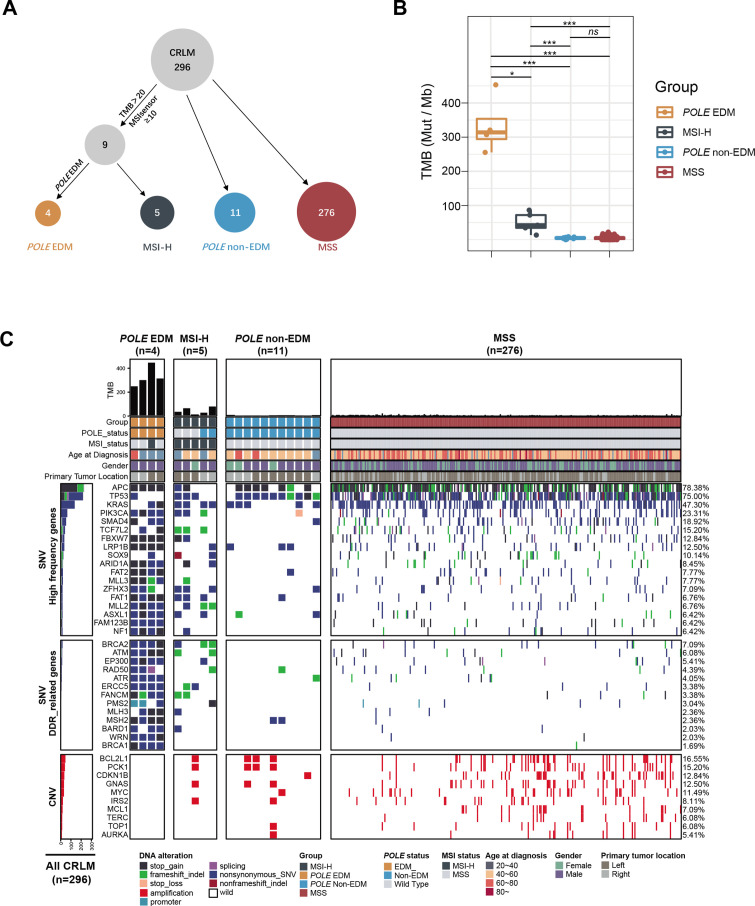
The 296 resected CRLM were classified into four molecular subgroups, POLE EDM (4, 1.4%), MSI-H (5, 1.7%), POLE non-EDM (11, 3.7%), and MSS (276, 93.2%) according to POLE and microsatellite status (figure 3A). Demographic and clinicopathological characteristics of the four molecular subgroups were shown in online supplemental table S1. TMB in the four CRLM tumors harboring POLE EDM were 453.12, 255.36, 320.64, and 307.20 mutations/Mb, respectively, which was significantly higher than those with MSI-H (median, 313.92 vs 42.24 mutations/Mb, p<0.05, Wilcoxon), POLE non-EDM (median, 313.92 vs 4.80 mutations/Mb, p<0.001, Wilcoxon), and MSS tumors (median, 313.92 vs 4.80 mutations/Mb, p<0.001, Wilcoxon). TMB of MSI-H tumors was significantly higher than POLE non-EDM (median, 42.24 vs 4.80 mutations/Mb, p<0.001, Wilcoxon) and MSS tumors (median, 42.24 vs 4.80 mutations/Mb, p<0.001, Wilcoxon). TMB was comparable between POLE non-EDM and MSS tumors (median, 4.80 vs 4.80 mutations/Mb, p>0.05, Wilcoxon) (figure 3B).
Figure 3.
Molecular classification scheme, tumor mutation burden, and altered genes in different molecular subtypes in 296 CRLM patients by next-generation sequencing. (A) Classification scheme for molecular subtypes: POLE-EDM, MSI-H, POLE non-EDM, and MSS. (B) Tumor mutation burden in four molecular subtypes in 296 CRLM patients. (C) Heatmap for altered genes in four molecular subtypes. CNV, copy number variation; CRLM, colorectal cancer liver metastases; DDR, DNA damage repair; EDM, exonuclease domain mutation; MSI-H, microsatellite instability-high; MSS, microsatellite stable; SNV, single-nucleotide variant.
jitc-2022-004487supp004.pdf (68.5KB, pdf)
A heatmap of the genetic mutation profiling of the four molecular subgroups was constructed (figure 3C). A large set of mutational genes could be seen in POLE EDM tumors. PI3KCA (4/4, 100%) and SMAD 4 (3/4, 75%) mutations were higher in POLE EDM tumors compared with other subgroups. DNA damage repair-related genes, including BRCA2, BRCA1, ATM, ATR, etc., were generally mutated in POLE EDM tumors (all nearly 100%) but were less significant in MSI-H, POLE non-EDM or MSS tumors. High APC mutations were found across all subgroups (100% in POLE EDM tumors and 78.38% among all 296 tumors). Besides, KRAS mutations were comparable between POLE EDM tumors (50%) and other subgroups (47%). TP53 mutation was only found in 1 of the 4 (25%) tumors with POLE EDM, which was lower than other subgroups (3/5 in MSI-H and 10/11 in POLE non-EDM). Interestingly, copy number variation of a series of genes, including MYC, BCL2L1, CDKN1B, BRCA2, etc, were common in POLE non-EDM and MSS tumors but rarely in POLE EDM tumors.
Similar TMB difference and genetic characteristics could be seen in 979 metastatic colorectal tumors with different molecular subtypes in the MSKCC cohort (online supplemental figure S3).
jitc-2022-004487supp003.pdf (387.7KB, pdf)
Discussion
The advent of immunotherapy has greatly changed the treatment landscape across a variety of malignancies, including metastatic colorectal cancer. In addition to dMMR and MSI-H, POLE gene variants have been suggested as another potential marker for responsiveness to immunotherapy17 due to their significant association with ultrahigh TMB,18 which is an effective indicator for response prediction to ICI. The clinical characteristics of patients harboring POLE EDM have been reported in different studies.19 20 However, due to the rarity of POLE EDM in colorectal cancer, limited data reporting the clinical efficacy of ICI in POLE EDM patients is available. Here, we present four CRLM patients harboring POLE EDM who benefited from ICI-based combination treatment. The radiological overall response rate was 100%, and all four patients (100%) received subsequent complete surgical resection of primary tumor and liver metastases successfully. pCR was achieved both in primary tumor and liver metastases, and the relapse-free status was maintained until the last follow-up.
CRC with pathogenic EDM in the POLE gene is characterized by distinct clinical features, tumor microenvironment, and genetic profile.9 11 18 Compared with MSS or MSI-H patients, we found that male gender and younger age were more common among patients with POLE EDM, which was in line with previous studies.11 13 In stage II colorectal cancer, increased CD8 +cytotoxic T lymphocytes and CD45RO+memory immune cells in POLE EDM were observed compared with POLE wild type or non-EDMs.9 Our study also observed a high density of CD3 +and/or CD8 +T lymphocytes, especially in postoperative tumor tissues. At the genetic level, upregulation of a large set of immune-related genes, for example, CD8A, EOMES, GZMA, and CXCL9, were observed in POLE EDM CRC from the TCGA database.9
Both POLE EDM and MSI-H lead to ultrahigh TMB in CRC. In the MSKCC cohort, TMB in POLE EDM CRC was much higher than in the MSI-H group,11 in line with our results. POLE EDM and MSI-H seemed mutually exclusive in metastatic CRC. But in stage II CRC, patients with POLE EDMs were more likely to be MSI-H (33.3% vs 11.2%, p=0.043).9 In our study, 1 in 4 CRLM patients with POLE EDM was MSI-H (25%). PDL1 expression often correlates with a good response to ICIs.21 In lung cancer and endometrial cancer, a certain extent of PDL1 expression could be observed in patients with POLE EDM.22 23 However, PDL1 expression was only weakly positive (CPS 5% in one patient) or negative (3 patients) in the CRLM patients of our study.
Hypermutated tumors, for instance, MSI-H, POLE EDM, are sensitive to immunotherapy. KEYNOTE 1774 revealed that pembrolizumab alone showed superior efficacy compared with chemotherapy for the treatment of MSI-H advanced colorectal cancer. However, according ro RESICT criteria, the proportion of progressed disease in the pembrolizumab arm was 29.4%, which is much higher than 12.3% in the chemotherapy arm, indicating immunotherapy alone maybe not sufficient for the treatment of some hypermutated colorectal cancer. Additionally, chemotherapeutic drugs may enhance tumor immunogenicity and facilitate antitumor immunity by inducing immunogenic cell death, terms synergistic effect of tumor chemoimmunotherapy.24 Therefore, ICI plus chemotherapy might be a choice for CRLM with POLE EDM. On one hand, considering two patients in our cohort were potentially resectable for liver metastases initially which means intensive and effective preoperative treatment was needed to achieve the best tumor radiological regression. On the other hand, ICI had been demonstrated well tolerated when combined with chemotherapy for the treatment of gastrointestinal cancer or thoracic cancer.25–27 As a result, the combination of ICI and chemotherapy (with or without bevacizumab) rather than ICI alone was used after POLE EDM was detected for these four CRLM patients. Our data showed that the sharp tumor regression and increased lymphocyte infiltration were the result of the combination of ICI and chemotherapy. Whether a single ICI could achieve a similar radiological or pathological response is yet to know. However, we prefer to believe immunotherapy may play a more important role. In fact, two patients progressed after first-line cytotoxic agents and were later rescued with second line agents including ICI, suggesting that like MSI tumors, chemotherapy may not be as efficient in this setting compared with other low TMB CRC.
In our cohort, we observed not-good accordance between radiological and pathological response in these patients, especially in case 3 and case 4, both of whom achieved complete response pathologically but only mild partial response radiologically after anti-PD1 combination treatment. Therefore, to find out robust biomarkers to dynamically monitor the efficacy of treatment is of great importance. DNA methylation is an epigenetic regulator of gene expression and cfDNA-bearing cancer-specific methylation patterns have been investigated as feasible biomarkers in diagnostic, prognostic, as well as treatment response monitoring of cancer. Bhangu et al found that serial measurements of CRC-associated methylation markers could be a particularly valuable tool for early response assessment in patients receiving neoadjuvant chemotherapy for CRLM.28 In our cohort, NPY/SEPT9/WIF1 methylation dynamics in cfDNA seemed to be correlated well with actual tumor burden changes since NPY/SEPT9/WIF1 meth-cfDNA decreased from a high level at baseline to a nearly undetectable level after treatment in all four patients. NPY/SEPT9/WIF1 meth-cfDNA was also more sensitive than the traditional CRC tumor marker, CEA, in this study. NPY/SEPT9/WIF1 meth-cfDNA clearance could become a potential indicator of pCR in CRLM patients with POLE EDM receiving anti-PD1 treatment, even if a radiological complete response is not reached. Similar to the ‘watch and wait’ strategy in locally advanced rectal cancer who achieved pCR after chemoradiation,29 we think that delayed surgical intervention might be a choice for CRLM patients with POLE EDM who could reach pCR after ICI treatment, especially considering the extensive and complexity of surgical resection for both primary colorectal cancer and liver metastases.
Based on the excellent clinical efficacy of immunotherapy from the present case series and previous case report,30 we suggest that the complete sequencing of POLE exonuclease domains by NGS or PCR is recommended in clinical practice before the treatment of metastatic colorectal cancer. One limitation of this study is the small number of patients due to the rarity of POLE EDM in colorectal cancer. As a result, findings from this study should be interpreted with caution and warrant further verification in such patients in the future.
Acknowledgments
The authors would like to thank Prof. Gong Chen (Department of Colorectal Surgery, Sun Yat sen University Cancer Center, Guangzhou, China.) and Dr. Xiaoyun Liu (Department of Molecular Diagnostics, Sun Yat sen University Cancer Center, Guangzhou, China) for their substantial support to this study.
Footnotes
LW and ZC contributed equally.
Contributors: YL, LW, and DW contributed to conception and design; LW, QS, and XJ contributed to the methodology; LW, CR, QS, XJ, and ZC contributed to acquisition of data; LW and ZC contributed to analysis and interpretation of data; LW contributed to the writing of the manuscript; YL, WPF, BL, YY and DW contributed to review and revision of the manuscript; YL, DW and YY contributed to study supervision. All authors approved the final version of the manuscript, including the authorship list.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: No, there are no competing interests.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
The dataset used during the study are available from the corresponding author on a reasonable request.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
The current study was approved by the institutional ethical review board of Sun Yat-sen University Cancer Center and was conducted in accordance with the Helsinki declaration of the World Medical Association.
References
- 1.Abdalla EK, Vauthey J-N, Ellis LM, et al. Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg 2004;239:818–27. discussion 25-7. 10.1097/01.sla.0000128305.90650.71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.D'Angelica M, Kornprat P, Gonen M, et al. Effect on outcome of recurrence patterns after hepatectomy for colorectal metastases. Ann Surg Oncol 2011;18:1096–103. 10.1245/s10434-010-1409-1 [DOI] [PubMed] [Google Scholar]
- 3.Tanaka K, Takakura H, Takeda K, et al. Importance of complete pathologic response to prehepatectomy chemotherapy in treating colorectal cancer metastases. Ann Surg 2009;250:935–42. 10.1097/SLA.0b013e3181b0c6e4 [DOI] [PubMed] [Google Scholar]
- 4.André T, Shiu K-K, Kim TW, et al. Pembrolizumab in microsatellite-instability-high advanced colorectal cancer. N Engl J Med 2020;383:2207–18. 10.1056/NEJMoa2017699 [DOI] [PubMed] [Google Scholar]
- 5.Pursell ZF, Isoz I, Lundström E-B, et al. Yeast DNA polymerase epsilon participates in leading-strand DNA replication. Science 2007;317:127–30. 10.1126/science.1144067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cancer Genome Atlas Research Network, Kandoth C, Schultz N, et al. Integrated genomic characterization of endometrial carcinoma. Nature 2013;497:67–73. 10.1038/nature12113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cancer Genome Atlas Network . Comprehensive molecular characterization of human colon and rectal cancer. Nature 2012;487:330–7. 10.1038/nature11252 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.van Gool IC, Eggink FA, Freeman-Mills L, et al. Pole proofreading mutations elicit an antitumor immune response in endometrial cancer. Clin Cancer Res 2015;21:3347–55. 10.1158/1078-0432.CCR-15-0057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mo S, Ma X, Li Y, et al. Somatic pole exonuclease domain mutations elicit enhanced intratumoral immune responses in stage II colorectal cancer. J Immunother Cancer 2020;8:e000881. 10.1136/jitc-2020-000881 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. OncoKB. Available: https://www.oncokb.org/gene/POLE
- 11.Yaeger R, Chatila WK, Lipsyc MD, et al. Clinical sequencing defines the genomic landscape of metastatic colorectal cancer. Cancer Cell 2018;33:125–36. 10.1016/j.ccell.2017.12.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang F, Wei XL, Wang FH, et al. Safety, efficacy and tumor mutational burden as a biomarker of overall survival benefit in chemo-refractory gastric cancer treated with toripalimab, a PD-1 antibody in phase Ib/II clinical trial NCT02915432. Ann Oncol 2019;30:1479–86. 10.1093/annonc/mdz197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shi Y, Su H, Song Y, et al. Safety and activity of sintilimab in patients with relapsed or refractory classical Hodgkin lymphoma (ORIENT-1): a multicentre, single-arm, phase 2 trial. Lancet Haematol 2019;6:e12–19. 10.1016/S2352-3026(18)30192-3 [DOI] [PubMed] [Google Scholar]
- 14.Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009;45:228–47. 10.1016/j.ejca.2008.10.026 [DOI] [PubMed] [Google Scholar]
- 15.Niu B, Ye K, Zhang Q, et al. MSIsensor: microsatellite instability detection using paired tumor-normal sequence data. Bioinformatics 2014;30:1015–6. 10.1093/bioinformatics/btt755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fong Y, Fortner J, Sun RL, et al. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg 1999;230:309–18. discussion 18-21. 10.1097/00000658-199909000-00004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang F, Zhao Q, Wang Y-N, et al. Evaluation of pole and POLD1 mutations as biomarkers for immunotherapy outcomes across multiple cancer types. JAMA Oncol 2019;5:1504–6. 10.1001/jamaoncol.2019.2963 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.He J, Ouyang W, Zhao W, et al. Distinctive genomic characteristics in POLE/POLD1-mutant cancers can potentially predict beneficial clinical outcomes in patients who receive immune checkpoint inhibitor. Ann Transl Med 2021;9:129. 10.21037/atm-20-7553 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Domingo E, Freeman-Mills L, Rayner E, et al. Somatic pole proofreading domain mutation, immune response, and prognosis in colorectal cancer: a retrospective, pooled biomarker study. Lancet Gastroenterol Hepatol 2016;1:207–16. 10.1016/S2468-1253(16)30014-0 [DOI] [PubMed] [Google Scholar]
- 20.Hu H, Cai W, Wu D, et al. Ultra-mutated colorectal cancer patients with pole driver mutations exhibit distinct clinical patterns. Cancer Med 2021;10:135–42. 10.1002/cam4.3579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Aguilar EJ, Ricciuti B, Gainor JF, et al. Outcomes to first-line pembrolizumab in patients with non-small-cell lung cancer and very high PD-L1 expression. Ann Oncol 2019;30:1653–9. 10.1093/annonc/mdz288 [DOI] [PubMed] [Google Scholar]
- 22.Song Z, Cheng G, Xu C, et al. Clinicopathological characteristics of pole mutation in patients with non-small-cell lung cancer. Lung Cancer 2018;118:57–61. 10.1016/j.lungcan.2018.02.004 [DOI] [PubMed] [Google Scholar]
- 23.Howitt BE, Shukla SA, Sholl LM, et al. Association of polymerase e-Mutated and Microsatellite-Instable endometrial cancers with neoantigen load, number of tumor-infiltrating lymphocytes, and expression of PD-1 and PD-L1. JAMA Oncol 2015;1:1319–23. 10.1001/jamaoncol.2015.2151 [DOI] [PubMed] [Google Scholar]
- 24.Qi J, Jin F, You Y, et al. Synergistic effect of tumor chemo-immunotherapy induced by leukocyte-hitchhiking thermal-sensitive micelles. Nat Commun 2021;12:4755. 10.1038/s41467-021-24902-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Boku N, Ryu M-H, Kato K, et al. Safety and efficacy of nivolumab in combination with S-1/capecitabine plus oxaliplatin in patients with previously untreated, unresectable, advanced, or recurrent gastric/gastroesophageal junction cancer: interim results of a randomized, phase II trial (ATTRACTION-4). Ann Oncol 2019;30:250–8. 10.1093/annonc/mdy540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gandhi L, Rodríguez-Abreu D, Gadgeel S, et al. Pembrolizumab plus chemotherapy in metastatic non-small-cell lung cancer. N Engl J Med 2018;378:2078–92. 10.1056/NEJMoa1801005 [DOI] [PubMed] [Google Scholar]
- 27.Paz-Ares L, Luft A, Vicente D, et al. Pembrolizumab plus chemotherapy for squamous non-small-cell lung cancer. N Engl J Med 2018;379:2040–51. 10.1056/NEJMoa1810865 [DOI] [PubMed] [Google Scholar]
- 28.Bhangu JS, Beer A, Mittlböck M, et al. Circulating free methylated tumor DNA markers for sensitive assessment of tumor burden and early response monitoring in patients receiving systemic chemotherapy for colorectal cancer liver metastasis. Ann Surg 2018;268:894–902. 10.1097/SLA.0000000000002901 [DOI] [PubMed] [Google Scholar]
- 29.Dossa F, Chesney TR, Acuna SA, et al. A watch-and-wait approach for locally advanced rectal cancer after a clinical complete response following neoadjuvant chemoradiation: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol 2017;2:501–13. 10.1016/S2468-1253(17)30074-2 [DOI] [PubMed] [Google Scholar]
- 30.Silberman R, F Steiner D, Lo AA, et al. Complete and prolonged response to immune checkpoint blockade in POLE-mutated colorectal cancer. JCO Precis Oncol 2019;3:1–5. 10.1200/PO.18.00214 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jitc-2022-004487supp001.pdf (85.9KB, pdf)
jitc-2022-004487supp002.pdf (23.9MB, pdf)
jitc-2022-004487supp004.pdf (68.5KB, pdf)
jitc-2022-004487supp003.pdf (387.7KB, pdf)
Data Availability Statement
The dataset used during the study are available from the corresponding author on a reasonable request.