Abstract
Methylmercury (MeHg) is a widely known environmental pollutant that causes severe neurotoxicity. MeHg-induced neurotoxicity depends on various cellular conditions, including differences in the characteristics of tissues and cells, exposure age (fetal, childhood, or adulthood), and exposure levels. Research has highlighted the importance of oxidative stress in the pathogenesis of MeHg-induced toxicity and the site- and cell-specific nature of MeHg-induced neurotoxicity. The cerebellar granule cells and deeper layer cerebrocortical neurons are vulnerable to MeHg. In contrast, the hippocampal neurons are resistant to MeHg, even at high mercury accumulation levels. This review summarizes the mechanisms underlying MeHg-mediated intracellular events that lead to site-specific neurotoxicity. Specifically, we discuss the mechanisms associated with the redox ability, neural outgrowth and synapse formation, cellular signaling pathways, epigenetics, and the inflammatory conditions of microglia.
Keywords: methylmercury, site-specific neurotoxicity, redox ability, neural hyperactivation, mitogen-activated protein kinase cascade, Rho/ROCK signaling, ER stress, microglia
1. Introduction
Methylmercury (MeHg) is a well-known neurotoxicant that causes serious and irreversible neurological dysfunction, depending on the cellular context and developmental phase. MeHg can pass through the blood–brain barrier, which blocks almost all toxic substances. MeHg causes lesions in both the central and peripheral nervous systems; these lesions are associated with the exposure concentration, the exposed age, and injured neuronal cell types, leading to characteristic neurological dysfunction. Minamata disease is MeHg-intoxicated neurological disorder with two characteristic clinical forms—fetal and adult. Fetal-type Minamata disease is caused by exposure to MeHg in utero and is characterized by extensive brain lesions leading to cerebral palsy-like clinical features with delayed psychomotor development [1]. Contrastingly, adult-type is caused by MeHg intoxication in adulthood and shows Hunter-Russell syndrome-like features, with the primary lesions involving the central nervous system (cerebellum, cerebrum, and dorsal root ganglia) and peripheral sensory nervous system [2,3]. Patients with this condition show neurological signs associated with site-specific pathological lesions.
To date, many studies on the pathogenic processes of MeHg exposure have been conducted using various cells and animal models. The above-mentioned pathological changes in MeHg toxicity do not directly correspond to the accumulation of mercury (Hg) in tissues. In a MeHg-exposed subacute rat model (fed 20 ppm MeHg in drinking water every day for 28 days), the most acute pathology was seen in the cerebellum with lower Hg concentrations than that in the liver and kidneys, which displayed fewer pathological changes despite the high Hg content [4]. Furthermore, it is known that the MeHg-induced pathological changes differ among animal species. In a MeHg-exposed mouse model, the mice showed pathological changes in the cerebra, but not in the cerebellums, which is different from the changes observed in rats. In animal MeHg exposure experiments, attention must be paid to the selection of animals for experimental purposes including their target organs. Site-and cell-specific pathological changes are characteristic of MeHg toxicity. It has been demonstrated that the deep layers of cerebrocortical neurons, cerebellar granule cells, dorsal root ganglia, and dorsal root nerves are all susceptible to MeHg, while the shallow layers of cerebrocortical neurons, cerebellar Purkinje cells, ventral root nerve, and the hippocampal neurons are resistant [5,6,7,8]. MeHg-mediated neurotoxicity depends on various cellular conditions and cellular responses, which play crucial roles in the development of MeHg-induced pathogenic changes. Many researchers have noted the importance of oxidative stress in the pathogenesis of MeHg-induced toxicity. Various MeHg intoxication studies have reported the early incidence of reactive oxygen species (ROS) induction and that the elimination of ROS can prevent MeHg cytotoxicity in vitro and in vivo. Recent studies have clarified that the molecular mechanisms underlying MeHg-mediated cellular damage involve MeHg-mediated oxidative stress [9].
Since different cellular redox abilities and cellular signaling systems play important roles in MeHg neurotoxicity, we focus on the cellular conditions responsible for MeHg-mediated site- and cell-specific pathological changes in this review.
2. MeHg-Mediated Neurotoxicity in Cerebellum
2.1. Involvement of Local Redox Ability in Site-Specific Cerebellar Neurotoxicity
The cerebellum is one of the main lesion sites of MeHg-mediated neurotoxicity, and these lesions are known to be site-specific. The cerebellar cortex consists of three geometrically ordered layers. The innermost layer is densely packed with numerous small neurons called granule cells. The middle layer contains Purkinje cells, and the outermost molecular layer contains stellate and basket cells. Among these cerebellar neurons, cerebellar granule cells, which are known to be excitatory cells [10], are the most vulnerable to MeHg in humans [11].
These site-specific cerebellar lesions can be produced in MeHg-intoxicated animal models [5,12]. Treatment with antioxidants can prevent MeHg-induced neurotoxicity in rats, which suggests that oxidative stress plays a role in MeHg-mediated neural toxicity [4,13]. Antioxidative enzymes, such as copper, zinc superoxide dismutase (Cu, Zn-SOD), manganese superoxide dismutase (Mn-SOD), glutathione peroxidase 1 (GPx1), thioredoxin reductase 1 (TRxR1), and catalase, are intrinsic antioxidants that prevent cellular oxidative stress. A previous study reported whether these intrinsic antioxidants play a role in causing these site-specific cerebellar lesions [5]. In this study, each cell type was isolated from the three cerebellar layers using a microdissection system and performed real-time PCR analysis of antioxidative enzyme-encoding genes. Results demonstrated that the basal abilities of antioxidative enzymes differed among these three cerebellar layers of neurons. Cerebellar granule cells exhibited much lower in situ expression of Mn-SOD, GPx1, and TRxR1 mRNAs than cells in the cerebellar molecular layer or the Purkinje cells. Furthermore, immunohistochemistry showed stronger expression of Mn-SOD, GPx1, and TRxR1 in Purkinje cells and molecular layers of neurons than in granule cell layers, which was consistent with the mRNA expression pattern. Since Mn-SOD, GPx1, and TRxR1 are critical factors in protecting cells against MeHg-induced oxidative stress [9,14], these results suggest that low basal expression of these antioxidative enzymes contributes to MeHg-mediated site-specific neurotoxicity in the cerebellum.
2.2. Involvement of Disruption of Intracellular Ca2+ Regulation in Cerebellar Neurotoxicity
MeHg causes severe disruption of intracellular Ca2+ regulation, which contributes to cell death of primary cultures of rat cerebellar granule neurons [15]. Cerebellar granule neurons appear to be more susceptible to MeHg-induced dysregulation of divalent cation homeostasis and subsequent cell death when compared to purkinje neurons [16]. It has been demonstrated that MeHg-induced Ca2+ dysregulation and subsequent neurotoxicity of cerebellar granule cells are caused by the activation of M3 muscarinic receptor-linked pathway [15]. Cerebellar granule cells express the highest density of M3 muscarinic receptors [17], suggesting that the specific localization of M3 muscarinic receptors also contributes to MeHg-mediated site-specific neurotoxicity. The recent study added the findings that multiple sources of Ca2+ contribute to an increased frequency of MeHg-induced spontaneous inhibitory synaptic responses in rat cerebellar slices [18].
2.3. Impairment of Neurite Outgrowth in the Developing Cerebellum
The brains of human fetuses and neonates are extremely vulnerable to MeHg even at low exposure levels [1]. The previous report demonstrated that exposure to low levels of MeHg (5 ppm in drinking water) during the prenatal and lactation stages reduces the postnatal expression of pre- and post-synaptic proteins through reduced neurite outgrowth in the rat cerebellum; the newborn rats exhibited impaired motor coordination, while any neurotoxicity was not observed in maternal brain [19]. These results suggested that MeHg disturbs synaptic development and impairs motor coordination. Studies on the underlying mechanism demonstrated that 5 ppm MeHg exposure from gestation days 1 to 20 suppressed the tropomyosin receptor kinase (Trk), which disturbed the neuronal differentiation and synapse formation in fetal rats. In addition, downregulation of eukaryotic elongation factor 1A1 (eEF1A1) was observed on postnatal day (PND) 1 [20]. The role of eEF1A1 was confirmed via a knockdown study using differentiating PC12 cells. Knockdown of eEF1A1 in differentiating PC12 cells caused impaired neurite outgrowth and synaptic protein expression, similar to the results of prenatal exposure to MeHg in the developing cerebellum. Taken together, MeHg downregulates the TrkA pathway and eEF1A, thereby impairing synaptic formation in the developing cerebellum.
Recently, it has been suggested that the toxic effects of chemicals on fetal development may be related to epigenetic alterations, such as DNA methylation and histone modification. The epigenetic effects of low-level MeHg exposure on neuronal development were evaluated using in vitro and in vivo experiments [21]. The results showed reduced acetylated histone H3 (AcH3) levels and increased histone deacetylase (HDAC) 3 and HDAC6 levels, both in vitro and in vivo, with the MeHg-mediated suppression of neurite outgrowth. In addition, increased levels of DNA methylation and DNA methyltransferase 1 (DNMT1) were observed. These results suggest that MeHg-induced epigenetic changes may also contribute to the impairment of neurite outgrowth in the developing cerebellum.
2.4. Cerebellar Synaptic and Neuritic Remodeling in Pregnant Rats
Although it is well established that fetuses are extremely susceptible to MeHg toxicity [1], little is known about the effects of a very a low-dose of MeHg exposure on maternal neurons. The recent study demonstrated that pregnant rats exposed to very low levels of MeHg (1 ppm in drinking water) showed temporal cerebellar synaptic and neuritic remodeling during the perinatal period between gestational day 20 and PND 1, while vehicle-exposed pregnant rats exhibited no change [22]. These changes were transient and were reversed by PND 21. To elucidate the mechanisms underlying the perinatal changes observed in MeHg-exposed pregnant rats, the proteins related to synapse formation and neurite outgrowth were assessed. The result identified suppression of the TrkA pathway and reduced expression of the activity-regulated cytoskeleton-associated protein (Arc). Furthermore, an increase in plasma corticosterone levels and a decrease in estradiol levels were observed in MeHg-exposed pregnant rats compared with those in the vehicle-exposed pregnant control rats. The changes in TrkA pathway activity, Arc expression, and plasma hormone levels were normalized in parallel with restoration of synaptic and neuritic changes. These results suggest that very low levels of MeHg affect perinatal synaptic and neuritic remodeling in the maternal cerebellum through modulation of the TrkA pathway and Arc expression, which may be caused by MeHg-induced maternal hormonal changes. Although the importance of this phenomenon has not been so far clear enough, it may be involved in the biological defense response to MeHg toxicity in the mother’s brain [23].
3. MeHg-Mediated Neurotoxicity in the Cerebral Cortex
3.1. Involvement of Local Redox Ability in Site-Specific Neurodegeneration
The cerebral cortex consists of a six-layered neocortex, numbered I to VI, from the outermost layer I to the innermost layer VI. Autopsy studies of adult-type Minamata disease have shown that MeHg-induced cerebral lesions are localized to the deeper layers of the cerebral cortex, especially layer IV [2].
Such site-specific cerebral lesions can be reproduced in a MeHg-intoxicated mouse model [6,24]. The cause of this site-specific MeHg neurotoxicity in model mice was investigated with a focus on in situ antioxidative systems because of their critical role in MeHg intoxication [6,25,26]. In situ analyses of antioxidative enzyme expression were performed in laser-microdissected cerebrocortical neurons of shallower layers (sl-CCNs) and deeper layers (dl-CCNs). RT-qPCR analyses showed lower basal mRNA expression levels of Mn-SOD and GPx1 in the dl-CCNs than in the sl-CNNs. Subsequently, antioxidative enzymes activity analyses for the separated cerebral cortex showed that Mn-SOD and GPx1 activities were lower in dl-CCNs than in sl-CCNs.
These results suggest that low basal expression of these antioxidative enzymes may contribute to MeHg-mediated site-specific neurotoxicity in the cerebral cortex, which is similar to the layer-specific sensitivity seen in the cerebellum as already mentioned in Section 2.1 in this review.
3.2. MeHg-Mediated Neural Hyperactivation in the Deep Layers of Cerebrocortical Neurons
Cerebrocortical neurons of layer IV are mainly composed of excitatory cells, similar to the cerebellar granule cells. This suggests that excitatory cells respond readily to MeHg-induced neural hyperactivation, leading to neuronal damage. Genes that are activated transiently and rapidly in response to various cellular stimuli are referred to as immediate early genes (IEGs). Among IEGs, c-fos and brain-derived neurotrophic factor (BDNF) are potent markers of neural activity. In particular, c-fos has been extensively used to identify site-specific neural activity in the brain [27,28].
The relation between the MeHg-induced changes in site-specific expression of IEGs and neural activity has been investigated in a MeHg-intoxicated mouse model that showed neurodegeneration in dl-CCNs, especially in layer IV. Immunohistochemical and Western blot analyses demonstrated that site-specific upregulation of c-fos and BDNF in dl-CCNs preceded neuropathological changes in the cerebral somatosensory cortex [24]. Western blot results showed that c-fos and BDNF expressed in parallel with the induction of cAMP response element-binding protein (CREB) via phosphorylation, a regulator of c-fos and BDNF. These changes were triggered by activation of the p44/42 MAPK, p38 MAPK, and cAMP-dependent protein kinase (PKA) pathways. This expression of c-fos and BDNF through activation of the MAPK and PKA/CREB pathways was not observed in the hippocampus or cerebellum, where neuronal degeneration was never observed in mice despite MeHg accumulation. MeHg-induced oxidative stress activates the MAPK pathway, leading to various cellular stress responses [24]. Taken together, these findings suggest that the MeHg-induced cerebrocortical neuronal degeneration was caused by site-specific neural hyperactivity, which was triggered by activation of the MAPK and PKA/CREB pathways, followed by c-fos and BDNF upregulation.
Further investigation of MeHg-induced oxidative stress and the subsequent signaling pathways causing cerebrocortical neural hyperactivity and cell death was performed using all-trans retinoic acid-differentiated SH-SY5Y cells. These cells showed neuron-like morphological changes and the expression of neurons/synapse markers for cerebrocortical neurons [29]. Time-course studies clarified that MeHg-induced upregulation of c-fos preceded neuronal cell death, which was similar to the result observed in the cerebra of MeHg-intoxicated mice. The SH-SY5Y model showed that early induction of oxidative stress was followed by the activation of p44/42 MAPK and p38 MAPK and an increase in CREB expression. The antioxidative drugs Trolox and Edarabone significantly suppressed MeHg-induced oxidative stress, the p38 MAPK-CREB pathway, and neuronal cell death. Furthermore, treatment with SB203580, a specific inhibitor of p38 MAPK, significantly blocked the regulation of c-fos and neuronal cell death, whereas treatment with the p44/p42 MAPK inhibitors PD98059 and U0126 did not [29]. These results suggest that the MeHg-induced oxidative stress and subsequent activation of the p38 MAPK-CREB pathway contributes to neuronal hyperactivity and subsequent neuronal cell death.
3.3. Role of the Rho-ROCK Signaling Pathway in MeHg-Mediated Axonal Degeneration
It has been reported that the ROCK signaling pathway is involved in various biological reactions such as neural axonal degeneration, microglial activation, and apoptotic cell death [30]. ROCK is expressed as two homologs, ROCK1 and ROCK2, which share similar structures and functions. In the brain, it has been reported that ROCK1 is distributed in glia, whereas ROCK2 in neurons [31].
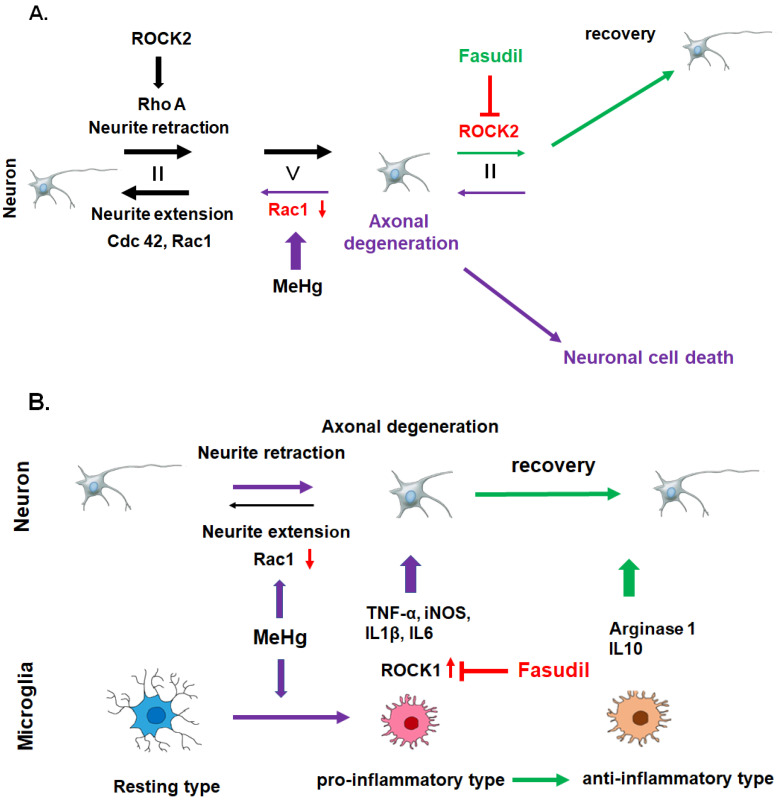
Neurite degeneration prior to neuronal cell death from MeHg exposure has been demonstrated in studies with cultured cerebral cortical neurons [20,32,33]. Previous studies demonstrated that the Rho-ROCK signaling pathway plays a role in this phenomenon both in vitro and in vivo [32,33,34,35]. Rho family proteins, including Rho, Ras-related C3 botulinum toxin substrate (Rac), and cell division control protein 42 homolog (Cdc42), are associated with neurite regulation and apoptotic cell death in neurons. Among the Rho family of proteins, Rac1 and Cdc42 are associated with neurite extension. In contrast, neurite retraction is promoted by RhoA as well as ROCK1 and 2, which are key effectors of RhoA [36]. Using cultured cerebrocortical neurons, we showed that MeHg-induced downregulation of Rac1, a protein related to neurite outgrowth, is the first event that triggers axonal degeneration and subsequent apoptotic neuronal cell death [32]. In contrast, the expression of RhoA, which is associated with neurite contraction, is unaffected by MeHg exposure. These results suggested that disturbances in the Rho/ROCK pathway result in a neurite outgrowth/contraction imbalance, which leads to MeHg-induced axonal degeneration. Several endogenous inhibitors, including myelin-related glycoproteins, NOGO, and oligodendrocyte-myelin glycoproteins, have been reported to interfere with axonal regeneration in the central nervous system (CNS) via the Rho/ROCK pathway [37]. It has been reported that modifiers of the Rho/ROCK pathway, including the Rho inhibitor C3 toxin, and the two ROCK inhibitors, fasudil and Y-27632 (selective ROCK1, 2 inhibitors), significantly protect cultured cortical neurons from MeHg-induced axonal degeneration and apoptotic neuronal cell death [32]. Furthermore, in vivo treatment with fasudil partially prevented pathological changes in the dorsal root ganglia, dorsal root nerve, and posterior column of the spinal cord by inhibiting ROCK2 in the subacute phase of a MeHg-intoxicated rat model [34]. Figure 1A summarizes the MeHg-mediated axonal degeneration caused by a neurite outgrowth/contraction imbalance and its recovery via Rho/ROCK pathway mediators.
Figure 1.
MeHg-mediated axonal degeneration is caused by a neurite outgrowth/contraction imbalance, and the effects are reversed by fasudil treatment. There are two mechanisms behind the therapeutic effect of fasudil on MeHg-induced neurodegeneration in the subacute phase (A) and in the chronic phase (B) in a MeHg-intoxicated rat model. (A) MeHg-induced downregulation of Rac1 causes a neurite outgrowth/contraction imbalance and axonal degeneration. ROCK2 inhibition by fasudil modifies the neurite outgrowth/contraction imbalance and protects cortical neurons from axonal degeneration and apoptotic neuronal cell death. (B) Fasudil suppresses the ROCK1 activity of invaded microglia, followed by downregulation of pro-inflammatory factors, including TNFα, iNOS, IL-1β, and IL-6, and upregulation of the anti-inflammatory factors arginase1 and IL-10. The Rho/ROCK pathway of microglia is essential for the chronic phase of MeHg-induced cerebrocortical neurodegeneration.
Recently, much attention has been paid to blocking the effects of the Rho/ROCK signaling pathway on neurite outgrowth and apoptosis after CNS injury [38]. Regulation of the Rho/ROCK pathway may be a potential target to prevent MeHg-mediated axonal degeneration and apoptotic neuronal cell death. The recent study also demonstrated the therapeutic effect of fasudil in a chronic MeHg-intoxicated rat model, even after the onset of neuropathological changes and clinical signs [35]. Results demonstrated the recovery effect of fasudil on MeHg-induced axonal degeneration and clinical signs during the chronic neurotoxicity stage. The ROCK analysis study demonstrated that the therapeutic effect of fasudil was due to the transition of invaded microglia from a pro-inflammatory to an anti-inflammatory phenotype. Under MeHg exposure, the invaded microglia were pro-inflammatory and showed an increase in ROCK1 expression. Fasudil suppressed the ROCK activity of invaded microglia, which downregulated pro-inflammatory factors, including tumor necrosis factor α (TNFα), inducible nitric oxide synthetase (iNOS), IL-1β, and IL-6, and upregulated the anti-inflammatory factors arginase-1 and IL-10 [35]. These results suggest that changes in the levels of microglia-secreted cytokines have a recovery effect on MeHg-induced axonal degeneration and clinical signs. Figure 1B summarizes the mechanism of the in vivo therapeutic effect of fasudil in MeHg-induced neurodegeneration during the chronic phase of the MeHg-intoxicated rat model. Another study has corroborated the role of microglial ROCK1 in a chronic MeHg-induced mouse cerebrocortical neurodegeneration model [39].
3.4. MeHg-Mediated Activation of the c-Jun N-Terminal Kinase (JNK) Pathway Causes Neurodegeneration in Cerebrocortical Neurons
The microtubule-associated protein tau plays important roles in stabilizing microtubules and axonal transport in neurons. Although tau phosphorylation regulates axonal transport through its conformation under physiological conditions, under pathological conditions, hyperphosphorylation reduces the affinity of tau for microtubules and impairs axonal transport. Furthermore, hyperphosphorylated tau easily aggregates into paired helical filaments and neurofibrillary tangles, subsequently leading to neuronal death [40].
Tau hyperphosphorylation can be observed in the brains of mice exposed to MeHg (30 ppm MeHg in drinking water for eight weeks) [41]. Neuropathological studies have shown reduction in the number of neurons, neuronal necrosis, and apoptosis and increase in the number of astrocytes and microglia/macrophages in the cerebral cortex. Western blot analyses have demonstrated that MeHg exposure increases tau phosphorylation at Thr-205, Ser-396, and Ser-422 in the cerebral cortex, consistent with the phosphorylation patterns noted in Alzheimer’s disease and frontotemporal dementia. Furthermore, immunohistochemical analyses have shown that the distribution of tau-phosphorylated (Thr-205) neurons corresponds with areas showing considerable neuropathological changes. The activation of mitogen-activated protein kinase kinase 4 (MKK4) and JNK is particularly considered to be responsible for tau phosphorylation. Additionally, neuropathological changes and tau hyperphosphorylation are not detected in the hippocampus even if Hg concentration is twice that in the cerebral cortex. These results suggest that MeHg-induced tau phosphorylation is mainly triggered by the activation of JNK pathways and contributes to neuropathological changes in the cerebral cortex [41].
Meanwhile, JNK pathway has been reported to be related to autophagy induction and play an important role in neurotoxicity [42]. Beclin1 and vacuolar sorting protein 34 (Vps34), a class III phosphatidylinositol-3 kinase are known to play an essential role in the cellular process of autophagy [43]. A recent study demonstrated that MeHg-induced JNK activation promotes the interaction between Beclin1 and Vsp34 and autophagy induction in cultured rat cerebrocortical neurons [44]. Although it has been reported that autophagy plays a protective role in cadmium- or manganese-induced neurotoxicity [45,46], the impairment of lysosomal function by MeHg promoted autophagosome accumulation and neuronal cell death [44]. MeHg-induced autophagy via JNK/Vps34 complex pathway also plays a role in neurotoxicity in the cerebral cortex.
3.5. Role of Endoplasmic Reticulum (ER) Stress in MeHg-Mediated Cerebrocortical Neurotoxicity
The ER is a membranous organelle that specializes in the folding and post-translational maturation of proteins. The ER redox stage is linked to ER protein folding homeostasis. Disulfide bond formation in the ER lumen is highly sensitive to an altered redox balance, and the disruption of protein folding causes ER stress.
A previous study showed that the failure to protect cells from MeHg-mediated early oxidative stress triggers ER stress and apoptosis [47]. A subsequent study showed that pretreatment with the antioxidant Trolox significantly blocked the MeHg-induced ER stress, thereby preventing unfolded protein response (UPR) activation, mitochondrial dysfunction, and apoptosis in primary cultured cortical neurons [48]. These results suggest that MeHg induces neuronal apoptosis through ROS-mediated ER stress and mitochondrial apoptosis pathways. Furthermore, a recent study showed site- and cell type-specific ER stress in the MeHg-exposed brains of ER stress-activated indicator (ERAI) transgenic mice [49]. The transgenic ERAI was constructed by fusing the gene encoding a variant of the green fluorescent protein Venus as a reporter downstream of a partial sequence of the human X-box binding protein 1 (XBP1), including an ER stress-specific intron [50]. XBP1 is a transcription factor spliced by inositol-requiring protein 1 (IRE1), a stress sensor of the UPR. UPR is a cytoprotective pathway against ER stress. The transgenic mice with this pathway proteins present with an ERAI signal when the UPR is activated by ER stress. Spatiotemporal time-course analyses of ER stress using these ERAI-Venus mice demonstrated that MeHg exposure (30 ppm MeHg in drinking water) induced the largest ERAI fluorescence signal in the somatosensory cortex three weeks after the start of exposure. Dual immunohistochemical analyses showed that almost all ERAI signals were predominantly localized in the neurons exposed to MeHg for three weeks. Time-course analysis revealed a stronger ERAI signal in the early phase, suggesting this was the period when the UPR pathway was activated. Downstream signaling of the IRE1-XBP1 axis was also observed in the early phase. These results indicate that MeHg induces ER stress that activates the UPR pathway in cerebrocortical neuronal cells in the early phase after exposure.
4. MeHg-Mediated Neurotoxicity in the Primary Afferent Nervous System
Pain can be divided into nociceptive and neuropathic types. While nociceptive pain is caused by inflamed or damaged tissues activating specialized pain sensors called nociceptors, neuropathic pain is caused by damage to or malfunction of the nervous system [51]. Neuropathological changes in the peripheral nerves, spinal cord dorsal roots, spinal cord dorsal ganglia, and cerebral cortex areas, corresponding to damaged sensory systems, have been reported in human and animal MeHg toxicity [3,52,53]. Mercuric compounds have been reported to cause neuropathic pain in humans [54,55], which suggests that the MeHg-induced neuropathological changes in primary afferent nerves may also trigger neuropathic pain.
The recent study demonstrated the pathophysiological mechanism of MeHg-mediated neuropathic pain (hyperalgesia/allodynia) using a rat model of MeHg exposure (20 ppm in drinking water for three weeks) [56]. Histopathological studies using these rats demonstrated axonal degeneration in the primary afferent nervous system (peripheral sural nerve, dorsal root ganglion, dorsal root nerve, and dorsal column of the spinal cord), while no neuropathological changes were observed in the somatosensory cortex and thalamus [53,56]. The rats showed hyperalgesia/allodynia, evidenced by a significant decrease in the threshold of mechanical pain evaluated using an algometer with calibrated forceps [56]. Immunohistochemistry also revealed the accumulation of activated microglia in the dorsal root nerve, dorsal column, and dorsal horn of the spinal cord. An increase in the levels of inflammotoxic and inflammatory cytokines (TNFα, Il-1β, and IL-6), which are related to Iba1-positive inflammatory microglia, and phospho-CRE binding protein (CREB), which is related to neuronal activation, was revealed by Western blot analysis. These results suggest that dorsal horn neuronal activation is mediated by microglia-excreted inflammatory factors and that these changes alter nociceptive transmission within the spinal cord. Furthermore, analyses of the cerebral cortex demonstrated increased expression of phospho-CREB and astrocyte-released thrombospondin-1, which are important factors for excitatory synapse formation, specifically in the somatosensory cortical area. The expression of pre- and post-synaptic markers also increased in this cortical area. These observations suggest that this new cortical circuit is specifically wired in the somatosensory cortex. MeHg-mediated neuropathic pain may be caused by neuronal activation with inflammatory microglia in the dorsal horn and new cortical circuit formation in the somatosensory cortex [56].
5. Conclusions
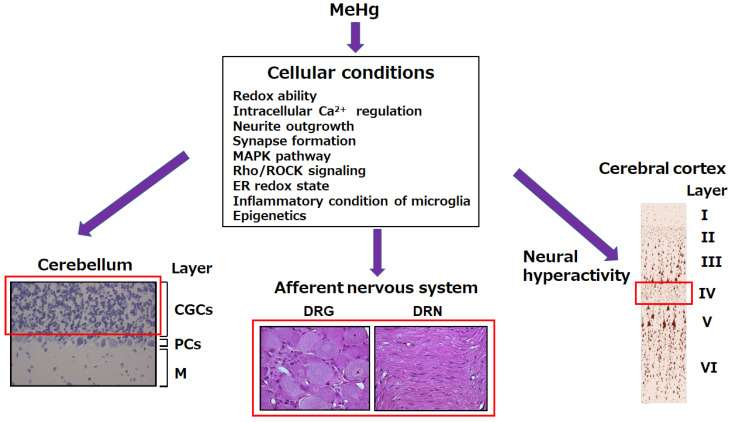
The pathological changes in MeHg toxicity do not directly correspond to the accumulation of Hg in the tissues, and various cellular conditions modify the MeHg-mediated toxicological processes. Oxidative stress in the early phase after exposure to MeHg plays a role in the pathogenesis of MeHg neurotoxicity. MeHg-mediated oxidative stress arises from the disruption of individual cellular redox systems and affects various cellular processes. The MeHg neurotoxicity involves neurite outgrowth and synapse formation, various cellular signaling pathways, the ER redox state, epigenetics, and the inflammation of microglia. An imbalance in cellular protective systems against MeHg toxicity leads to characteristic site-specific MeHg toxicity. To protect cells against MeHg-mediated cellular damage, an understanding of the mechanisms underlying MeHg toxicity is essential. A schematic overview of the cellular conditions that lead to MeHg-mediated site-specific neurotoxicity is presented in Figure 2.
Figure 2.
Cellular conditions responsible for MeHg-mediated site-specific neurotoxicity. MeHg-mediated site-specific neurotoxicity involves cerebellar granule cells, the primary afferent nervous system, and deep layers of cerebrocortical neurons (especially layer IV), which are surrounded by a red frame. The cellular conditions indicated here are responsible for the occurrence of MeHg-mediated site-specific neurotoxicity. CGCs, cerebellar granule cells; PCs, Purkinje cells; M, molecular; DRG, dorsal root ganglia; DRN, dorsal root nerve.
Acknowledgments
We are grateful to Yoshimi Tsuruda, Ayumi Onitsuka, Michiko Fuchigami, and Kaori Nakahara for their technical assistance.
Abbreviations
| MeHg | Methylmercury |
| Hg | Mercury |
| ROS | Reactive oxygen species |
| MAPK | Mitogen-activated protein kinase |
| Rho | Ras homolog gene |
| ROCK | Rho-associated coiled coil-forming protein kinase |
| Cu: Zn-SOD | Copper, zinc-superoxide dismutase |
| Mn-SOD | Manganese-superoxide dismutase |
| GPx1 | Glutathione peroxidase 1 |
| TRxR1 | Thioredoxin reductase 1 |
| PCR | Polymerase chain reaction |
| Trk | Tropomyosin receptor kinase |
| eEF1A1 | Eukaryotic elongation factor 1A1 |
| PND | Postnatal day |
| Arc | Activity-regulated cytoskeleton-associated protein |
| Sl-CCNs | shallower layers of cerebrocortical neurons |
| dl-CCNs | deep layers of cerebrocortical neurons |
| IEGs | Immediate early genes |
| BDNF | Brain-derived neurotrophic factor |
| CREB | cAMP response element binding protein |
| PKA | cAMP-dependent protein kinase |
| Rac | Ras-related C3 botulinum toxin substrate |
| Cdc42 | Cell division control protein 42 homolog |
| CNS | Central nervous system |
| JNK | c-Jun N-terminal kinase |
| MKK4 | Activated protein kinase kinase 4 |
| Vsp34 | Vacuolar sorting protein 34 |
| ER | Endoplasmic reticulum |
| UPR | Unfolded protein response |
| ERAI | ER stress-activated indicator |
| XBP1 | X-box binding protein 1 |
| IRE1 | Inositol-requiring protein 1 |
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare that there are no conflict of interest. The funders had no role in the study design, collection, analyses, interpretation of the data; writing of the manuscript; or decision to publish the results.
Funding Statement
This research received no external funding.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Harada M., Akagi H., Tsuda T., Kizaki T., Ohno H. Methylmercury level in umbilical cords from patients with congenital Minamata disease. Sci. Total Environ. 1999;234:59–62. doi: 10.1016/S0048-9697(99)00255-7. [DOI] [PubMed] [Google Scholar]
- 2.Eto K. Pathology of Minamata disease. Toxicol. Pathol. 1997;25:614–623. doi: 10.1177/019262339702500612. [DOI] [PubMed] [Google Scholar]
- 3.Eto K., Tokunaga H., Nagashima K., Takeuchi T. An autopsy case of minamata disease (methylmercury poisoning)—Pathological viewpoints of peripheral nerves. Toxicol. Pathol. 2002;30:714–722. doi: 10.1080/01926230290166805. [DOI] [PubMed] [Google Scholar]
- 4.Usuki F., Yasutake A., Umehara F., Tokunaga H., Matsumoto M., Eto K., Ishiura S., Higuchi I. In vivo protection of a water-soluble derivative of vitamin E, Trolox, against methylmercury-intoxication in the rat. Neurosci. Lett. 2001;304:199–203. doi: 10.1016/S0304-3940(01)01764-5. [DOI] [PubMed] [Google Scholar]
- 5.Fujimura M., Usuki F. Low in situ expression of antioxidative enzymes in rat cerebellar granular cells susceptible to methylmercury. Arch. Toxicol. 2014;88:109–113. doi: 10.1007/s00204-013-1089-2. [DOI] [PubMed] [Google Scholar]
- 6.Fujimura M., Usuki F. In situ different antioxidative systems contribute to the site-specific methylmercury neurotoxicity in mice. Toxicology. 2017;392:55–63. doi: 10.1016/j.tox.2017.10.004. [DOI] [PubMed] [Google Scholar]
- 7.Shinoda Y., Ehara S., Tatsumi S., Yoshida E., Takahashi T., Eto K., Kaji T., Fujiwara Y. Methylmercury-induced neural degeneration in rat dorsal root ganglion is associated with the accumulation of microglia/macrophages and the proliferation of Schwann cells. J. Toxicol. Sci. 2019;44:191–199. doi: 10.2131/jts.44.191. [DOI] [PubMed] [Google Scholar]
- 8.Karri V., Professor M.S., Kumar V. A systems toxicology approach to compare the heavy metal mixtures (Pb, As, MeHg) impact in neurodegenerative diseases. Food Chem. Toxicol. 2020;139:111257. doi: 10.1016/j.fct.2020.111257. [DOI] [PubMed] [Google Scholar]
- 9.Fujimura M., Usuki F. Methylmercury-mediated oxidative stress and activation of the cellular protective system. Antioxidants. 2020;9:1004. doi: 10.3390/antiox9101004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Palacios E.R., Houghton C., Chadderton P. Accounting for uncertainty: Inhibition for neural inference in the cerebellum. Proc. Biol. Sci. 2021;288:20210276. doi: 10.1098/rspb.2021.0276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.O’Donoghue J.L., Watson G.E., Brewer R., Zareba G., Eto K., Takahashi H., Marumoto M., Love T., Harrington D., Myers G.J. Neuropathology associated with exposure to different concentrations and species of mercury: A review of autopsy cases and the literature. Neurotoxicology. 2020;78:88–98. doi: 10.1016/j.neuro.2020.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ihara H., Kasamatsu S., Kitamura A., Nishimura A., Tsutsuki H., Ida T., Ishizaki K., Toyama T., Yoshida E., Abdul Hamid H., et al. Exposure to electrophiles impairs reactive persulfide-dependent redox signaling in neuronal cells. Chem. Res. Toxicol. 2017;30:1673–1684. doi: 10.1021/acs.chemrestox.7b00120. [DOI] [PubMed] [Google Scholar]
- 13.Yamashita T., Ando Y., Nakamura M., Obayashi K., Terazaki H., Haraoka K., Guo S.X., Ueda M., Uchino M. Inhibitory effect of α-tocopherol on methylmercury-induced oxidative steress. Environ. Health Prev. Med. 2004;9:111–117. doi: 10.1007/BF02898069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tucker E.K., Nowak R.A. Methylmercury alters proliferation, migration, and antioxidant capacity in human HTR8/SV-neo trophoblast cells. Reprod. Toxicol. 2018;78:60–68. doi: 10.1016/j.reprotox.2018.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Limke T.L., Bearss J.J., Atchison W.D. Acute exposure to methylmercury causes Ca2+ dysregulation and neuronal death in rat cerebellar granule cells through an M3 muscarinic receptor-linked pathway. Toxicol. Sci. 2004;80:60–68. doi: 10.1093/toxsci/kfh131. [DOI] [PubMed] [Google Scholar]
- 16.Edwards J.R., Marty M.S., Atchison W.D. Comparative sensitivity of rat cerebellar neurons to dysregulation of divalent cation homeostasis and cytotoxicity caused by methylmercury. Toxicol. Appl. Pharmacol. 2005;208:222–232. doi: 10.1016/j.taap.2005.02.015. [DOI] [PubMed] [Google Scholar]
- 17.Neustadt A., Frostholm A., Rotter A. Topographical distribution of muscarinic cholinergic receptors in the cerebellar cortex of the mouse, rat, guinea pig, and rabbit: A species comparison. J. Comp. Neurol. 1988;272:317–330. doi: 10.1002/cne.902720303. [DOI] [PubMed] [Google Scholar]
- 18.Yuan Y., Atchison W.D. Multiple sources of Ca2+ contribute to methylmercury-induced increased frequency of spontaneous inhibitory synaptic responses in cerebellar slices of rat. Toxicol. Sci. 2016;150:117–130. doi: 10.1093/toxsci/kfv314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fujimura M., Cheng J., Zhao W. Perinatal exposure to low-dose methylmercury induces dysfunction of motor coordination with decreases in synaptophysin expression in the cerebellar granule cells of rats. Brain Res. 2012;1464:1–7. doi: 10.1016/j.brainres.2012.05.012. [DOI] [PubMed] [Google Scholar]
- 20.Fujimura M., Usuki F., Cheng J., Zhao W. Prenatal low-dose methylmercury exposure impairs neurite outgrowth and synaptic protein expression and suppresses TrkA pathway activity and eEF1A1 expression in the rat cerebellum. Toxicol. Appl. Pharmacol. 2016;298:1–8. doi: 10.1016/j.taap.2016.03.002. [DOI] [PubMed] [Google Scholar]
- 21.Go S., Kurita H., Hatano M., Matsumoto K., Nogawa H., Fujimura M., Inden M., Hozumi I. DNA methyltransferase- and histone deacetylase-mediated epigenetic alterations induced by low-level methylmercury exposure disrupt neuronal devel-opment. Arch. Toxicol. 2021;95:1227–1239. doi: 10.1007/s00204-021-02984-7. [DOI] [PubMed] [Google Scholar]
- 22.Fujimura M., Usuki F. Pregnant rats exposed to low level methylmercury exhibit cerebellar synaptic and neuritic remodeling during the perinatal period. Arch. Toxicol. 2020;94:1335–1347. doi: 10.1007/s00204-020-02696-4. [DOI] [PubMed] [Google Scholar]
- 23.Hoekzema E., Barba-Müller E., Pozzobon C., Picado M., Lucco F., García-García D., Soliva J.C., Tobeña A., Desco M., Crone E.A., et al. Pregnancy leads to long-lasting changes in human brain structure. Nat. Neurosci. 2017;20:287–296. doi: 10.1038/nn.4458. [DOI] [PubMed] [Google Scholar]
- 24.Fujimura M., Usuki F. Site-specific neural hyperactivity via the activation of MAPK and PKA/CREB pathways triggers neuronal degeneration in methylmercury-intoxicated mice. Toxicol. Lett. 2017;271:66–73. doi: 10.1016/j.toxlet.2017.03.001. [DOI] [PubMed] [Google Scholar]
- 25.Meinerz D.F., Branco V., Aschner M., Carvalho C., Rocha J.B.T. Diphenyl diselenide protects against methylmercury-induced inhibition of thioredoxin reductase and glutathione peroxidase in human neuroblastoma cells: A comparison with ebselen. J. Appl. Toxicol. 2017;37:1073–1081. doi: 10.1002/jat.3458. [DOI] [PubMed] [Google Scholar]
- 26.Allen J.W., Mutkus L.A., Aschner M. Methylmercury-mediated inhibition of 3H-D-aspartate transport in cultured astrocytes is reversed by the antioxidant catalase. Brain Res. 2001;902:92–100. doi: 10.1016/S0006-8993(01)02375-7. [DOI] [PubMed] [Google Scholar]
- 27.Cheng J., Fujimura M., Zhao W., Wang W. Neurobehavioral effects, c-Fos/Jun expression and tissue distribution in rat offspring prenatally co-exposed to MeHg and PFOA: PFOA impairs Hg retention. Chemosphere. 2013;91:758–764. doi: 10.1016/j.chemosphere.2013.02.016. [DOI] [PubMed] [Google Scholar]
- 28.Flores V.L., Parmet T., Mukherjee N., Nelson S., Katz D.B., Levitan D. The role of the gustatory cortex in incidental experience-evoked enhancement of later taste learning. Learn. Mem. 2018;25:587–600. doi: 10.1101/lm.048181.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fujimura M., Usuki F. Methylmercury induces oxidative stress and subsequent neural hyperactivity leading to cell death through the p38 MAPK-CREB pathway in differentiated SH-SY5Y cells. Neurotoxicology. 2018;67:226–233. doi: 10.1016/j.neuro.2018.06.008. [DOI] [PubMed] [Google Scholar]
- 30.Borrajo A., Rodriguez-Perez A.I., Villar-Cheda B., Guerra M.J., Labandeira-Garcia J.L. Inhibition of the microglial response is essential for the neuroprotective effects of Rho-kinase inhibitors on MPTP-induced dopaminergic cell death. Neuropharmacology. 2014;85:1–8. doi: 10.1016/j.neuropharm.2014.05.021. [DOI] [PubMed] [Google Scholar]
- 31.Iizuka M., Kimura K., Wang S., Kato K., Amano M., Kaibuchi K., Mizoguchi A. Distinct distribution and localization of Rho-kinase in mouse epithelial, muscle and neural tissues. Cell Struct. Funct. 2012;37:155–175. doi: 10.1247/csf.12018. [DOI] [PubMed] [Google Scholar]
- 32.Fujimura M., Usuki F., Sawada M., Rostene W., Godefroy D., Takashima A. Methylmercury exposure downregulates the expression of Racl and leads to neuritic degeneration and ultimately apoptosis in cerebrocortical neurons. Neurotoxicology. 2009;30:16–22. doi: 10.1016/j.neuro.2008.10.002. [DOI] [PubMed] [Google Scholar]
- 33.Fujimura M., Usuki F. Differing effects of toxicants (methylmercury, inorganic mercury, lead, amyloid β and rotenone) on cultured rat cerebrocortical neurons: Differential expression of Rho proteins associated with neurotoxicity. Toxicol. Sci. 2012;126:506–514. doi: 10.1093/toxsci/kfr352. [DOI] [PubMed] [Google Scholar]
- 34.Fujimura M., Usuki F., Kawamura M., Izumo S. Inhibition of the Rho/ROCK pathway prevents neuronal degeneration in vitro and in vivo following methylmercury exposure. Toxicol. Appl. Pharmacol. 2011;250:1–9. doi: 10.1016/j.taap.2010.09.011. [DOI] [PubMed] [Google Scholar]
- 35.Fujimura M., Usuki F., Nakamura A. Fasudil, a Rho-associated coiled coil-forming protein kinase inhibitor, recovers methylmercury-induced axonal degeneration by changing microglial phenotype in rats. Toxicol. Sci. 2019;168:126–136. doi: 10.1093/toxsci/kfy281. [DOI] [PubMed] [Google Scholar]
- 36.Choi E.K., Kim J.G., Kim H.J., Cho J.Y., Jeong H., Park Y., Islam R., Cap C.K., Park J.B. Regulation of RhoA GTPase and novel target proteins for ROCK. Small GTPases. 2020;11:95–102. doi: 10.1080/21541248.2017.1364831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang L., Xu J., Guo D., Zhou X., Jiang W., Wang J., Tang J., Zou Y., Bi M., Li Q. Fasudil alleviates brain damage in rats after carbon monoxide poisoning through regulating neurite outgrowth inhibitor/oligodendrocytemyelin glycoprotein signalling pathway. Basic Clin. Pharmacol. Toxicol. 2019;125:152–165. doi: 10.1111/bcpt.13233. [DOI] [PubMed] [Google Scholar]
- 38.Jacob S., Thangarajan S. Fisetin impedes developmental methylmercury neurotoxicity via downregulating apoptotic signal-ling pathway and upregulating Rho GTPase signalling pathway in hippocampus of F1 generation rats. Int. J. Dev. Neurosci. 2018;69:88–96. doi: 10.1016/j.ijdevneu.2018.07.002. [DOI] [PubMed] [Google Scholar]
- 39.Shinozaki Y., Danjo Y., Koizumi S. Microglial ROCK is essential for chronic methylmercury-induced neurodegeneration. J. Neurochem. 2019;151:64–78. doi: 10.1111/jnc.14817. [DOI] [PubMed] [Google Scholar]
- 40.Pernègre C., Duquette A., Leclerc N. Tau secretion: Good and bad for neurons. Front. Neurosci. 2019;13:649. doi: 10.3389/fnins.2019.00649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fujimura M., Usuki F., Sawada M., Takashima A. Methylmercury induces neuropathological changes with tau hyperphos-phorylation mainly through the activation of the c-jun-N-terminal kinase pathway in the cerebral cortex, but not in the hip-pocampus of the mouse brain. Neurotoxicology. 2009;30:1000–1007. doi: 10.1016/j.neuro.2009.08.001. [DOI] [PubMed] [Google Scholar]
- 42.Lu Z., Miao Y., Muhammad I., Tian E., Hu W., Wang J., Wang B., Li R., Li J. Colistin-induced autophagy and apoptosis involves the JNK-Bcl2-Bax signaling pathway and JNK-p53-ROS positive feedback loop in PC-12 cells. Chem. Biol. Interact. 2017;277:62–73. doi: 10.1016/j.cbi.2017.08.011. [DOI] [PubMed] [Google Scholar]
- 43.Funderburk S.F., Wang Q.J., Yue Z. The Beclin 1-VPS34 complex--at the crossroads of autophagy and beyond. Trends Cell Biol. 2010;20:355–362. doi: 10.1016/j.tcb.2010.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lin T., Ruan S., Huang D., Meng X., Li W., Wang B., Zou F. MeHg-induced autophagy via JNK/Vps34 complex pathway promotes autophagosome accumulation and neuronal cell death. Cell Death Dis. 2019;10:399. doi: 10.1038/s41419-019-1632-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wang Q.W., Wang Y., Wang T., Zhang K.B., Jiang C.Y., Hu F.F., Yuan Y., Bian J.C., Liu X.Z., Gu J.H., et al. Cadmium-induced autophagy promotes survival of rat cerebral cortical neurons by activating class III phosphoinositide 3-kinase/beclin-1/B-cell lymphoma 2 signaling pathways. Mol. Med. Rep. 2015;12:2912–2918. doi: 10.3892/mmr.2015.3755. [DOI] [PubMed] [Google Scholar]
- 46.Zhang J., Cao R., Cai T., Aschner M., Zhao F., Yao T., Chen Y., Cao Z., Luo W., Chen J. The role of autophagy dysregula-tion in manganese-induced dopaminergic neurodegeneration. Neurotox. Res. 2013;24:478–490. doi: 10.1007/s12640-013-9392-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Usuki F., Fujita E., Sasagawa N. Methylmercury activates ASK1/JNK signaling pathways, leading to apoptosis due to both mitochondria- and endoplasmic reticulum (ER)-generated processes in myogenic cell lines. Neurotoxicology. 2008;29:22–30. doi: 10.1016/j.neuro.2007.08.011. [DOI] [PubMed] [Google Scholar]
- 48.Liu W., Yang T., Xu Z., Xu B., Deng Y. Methyl-mercury induces apoptosis through ROS-mediated endoplasmic reticulum stress and mitochondrial apoptosis pathways activation in rat cortical neurons. Free Radic. Res. 2019;53:26–44. doi: 10.1080/10715762.2018.1546852. [DOI] [PubMed] [Google Scholar]
- 49.Hiraoka H., Nomura R., Takasugi N., Akai R., Iwawaki T., Kumagai Y., Fujimura M., Uehara T. Spatiotemporal analysis of the UPR transition induced by methylmercury in the mouse brain. Arch. Toxicol. 2021;95:1241–1250. doi: 10.1007/s00204-021-02982-9. [DOI] [PubMed] [Google Scholar]
- 50.Iwawaki T., Akai R., Kohno K., Miura M. A transgenic mouse model for monitoring endoplasmic reticulum stress. Nat. Med. 2004;10:98–102. doi: 10.1038/nm970. [DOI] [PubMed] [Google Scholar]
- 51.Colloca L., Ludman T., Bouhassira D., Baron R., Dickenson A.H., Yarnitsky D., Freeman R., Truini A., Attal N., Finnerup N.B., et al. Neuropathic pain. Nat. Rev. Dis. Primers. 2017;3:17002. doi: 10.1038/nrdp.2017.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Igata A. Neurological aspects of methylmercury poisoning in Minamata. In: Tsu-baki T., Takahashi H., editors. Recent Advances in Minamata Disease Studies. Kodansha Ltd.; Tokyo, Japan: 1986. pp. 41–57. [Google Scholar]
- 53.Usuki F., Yasutake A., Umehara F., Higuchi I. Beneficial effects of mild lifelong dietary restriction on skeletal muscle: Prevention of age-related mitochondrial damage, morphological changes, and vulnerability to a chemical toxin. Acta Neuropathol. 2004;108:1–9. doi: 10.1007/s00401-004-0844-0. [DOI] [PubMed] [Google Scholar]
- 54.Gnanashanmugam G., Balakrishnan R., Somasundaram S.P., Parimalam N., Rajmohan P., Pranesh M.B. Mercury toxicity following unauthorized siddha medicine intake—A mimicker of acquired neuromyotonia—Report of 32 cases. Ann. Indian. Acad. Neurol. 2018;21:49–56. doi: 10.4103/aian.AIAN_274_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Khodashenas E., Aelami M., Balali-Mood M. Mercury poisoning in two 13-year-old twin sisters. J. Res. Med. Sci. 2015;20:308–311. [PMC free article] [PubMed] [Google Scholar]
- 56.Fujimura M., Usuki F., Nakamura A. Methylmercury induces hyperalgesia and allodynia through spinal cord dorsal horn neuronal activation and subsequent somatosensory cortical circuit formation in rats. Arch. Toxicol. 2021;95:2151–2162. doi: 10.1007/s00204-021-03047-7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.