Abstract
Nanocellulose has lately emerged as one of the most promising “green” materials due to its unique properties. Nanocellulose can be mainly divided into three types, i.e., cellulose nanocrystals (CNCs), cellulose nanofibrils (CNFs), and bacterial cellulose (BC). With the rapid development of technology, nanocellulose has been designed into multidimensional structures, including 1D (nanofibers, microparticles), 2D (films), and 3D (hydrogels, aerogels) materials. Due to its adaptable surface chemistry, high surface area, biocompatibility, and biodegradability, nanocellulose-based composite materials can be further transformed as drug delivery carriers. Herein, nanocellulose-based composite material used for drug delivery was reviewed. The typical drug release behaviors and the drug release mechanisms of nanocellulose-based composite materials were further summarized, and the potential application of nanocellulose-based composite materials was prospected as well.
Keywords: nanocellulose, drug delivery, microparticles, film, hydrogel, aerogel
1. Introduction
Drug delivery systems refer to the advanced technologies used for targeted delivery and/or controlled release of therapeutic drugs [1]. In the past few decades, drug delivery systems have received much attention because they offer potential benefits, such as reducing side effects, improving therapeutic effects, and possibly reducing doses of drugs [2]. There are three key factors required in an effective drug delivery system, including drug carriers, drug-loading ratio, and controlled release rate [3]. A major problem lies in the selection of suitable, natural, nontoxic and inexpensive materials, while the material should maintain good biological activity and fewer side effects. Commercially available poly(lactide-co-glycolide) (PLGA)-based materials are often used for particle drug release formulations [4]. However, due to their large burst release and acidic degradation behaviors, they are often limited to a certain extent. Nowadays, many natural polymers such as cellulose, starch, and glycogen have been extensive studied for drug delivery applications [5,6,7].
Cellulose is one of the main components in natural plants, with good renewability and biodegradability. It is intertwined with lignin and hemicellulose, which helps to maintain good stability and good strength of the plant [8]. With the development of technology, cellulose has attracted great interest in its new form of “nanocellulose” [9]. Nanocellulose can be mainly divided into three categories, including cellulose nanocrystals (CNCs), cellulose nanofibrils (CNFs), and bacterial cellulose (BC) [10]. Among them, CNCs and CNFs are commonly extracted from a variety of plants or algae through a so-called “Top-down” method. Yet, BC is produced by bacteria, which is a so-called “Bottom-up” approach. Nowadays, nanocellulose has attracted extensive attention for the applications in the fields of drug delivery, because of its large specific surface area, good mechanical strength, stiffness, biocompatibility, and renewability [11]. These characteristics enable nanocellulose with good drug loading and binding capacities [12,13].
Recently, nanocellulose-based materials with different types such as single, hybrid, or nanocomposite systems have been fabricated for application in the drug delivery system [14]. As shown in Figure 1, nanocellulose in types of microparticles, films, hydrogels, and aerogels can be utilized as different drug carriers. This review focuses on nanocellulose-based composite materials used in drug delivery systems, which have different dimensions (1D, 2D, 3D). Hydrophilic and hydrophobic drug release behaviors of nanocellulose-based materials are systematically summarized for the first time. The relationships between the structures of nanocellulose-based materials and drug release behaviors are also emphasized. Moreover, the latest research work on nanocellulose-based materials used in drug delivery is introduced in a general overview. The future perspectives with global market value of nanocellulose materials are also systematically summarized.
Figure 1.
Schematic illustration of different nanocellulose-based materials used for drug delivery.
2. Designed Nanocellulose-Based Materials for Drug Delivery
2.1. Structures and Characteristics of Nanocellulose-Based Materials
Nanocellulose is a unique and promising natural material derived from native cellulose or bacteria [15,16]. Table 1 summarizes the fundamental physiochemical properties of different types of nanocellulose. The morphology, size, and mechanical properties of different nanocelluloses are dependent on the fibrous raw materials, the isolation methods, and the processing conditions as well as the possible pre- or post-treatments [17,18]. CNFs can be prepared from the pulped form of wood/plants by the methods of combining chemical, enzymatic treatment, and mechanical pressure stratification [19]. CNFs commonly have both amorphous and crystalline regions with diameters ranging from 3 nm to 60 nm [20]. They also have flexible and gel-like consistency properties due to their amorphous regions and micrometer lengths [21]. Moreover, CNCs with elongated crystalline rod-like fragments are commonly obtained by strong acid hydrolysis, especially sulfuric acid hydrolysis [19]. As shown in Table 1, CNCs have shorter lengths and higher crystallinity compared to CNFs [22]. By contrast, BC is synthesized and secreted by a variety of bacteria, such as Acetobacter, Pseudomonas, and Agrobacterium [23]. Compared with plant-derived CNFs and CNCs, BC only contains cellulose and does not contain other components such as hemicellulose and lignin [24]. The diameter of BC ranges from 20 to 100 nm, with a high aspect ratio, high crystallinity (84–89%), and good biocompatibility [25] (Table 1). Moreover, BC also has other unique physical properties, such as high degree of polymerization (molecular weight up to 8000 Da), strong water retention capacity (water content up to 99%), plasticity, and hydrophilicity [26]. Therefore, the three-dimensional layered nanostructure of nanocellulose and its physicochemical properties on the nanoscale have opened up new prospects for its application in many fields.
Table 1.
Comparison of structure parameters of different types of nanocellulose.
| Types of Nanocellulose |
Structure Properties | Mechanical Properties | |||
|---|---|---|---|---|---|
| Diameter (nm) | Length (nm) | Crystallinity (%) |
Young’s Modulus (GPa) |
Tensile Strength (GPa) | |
| CNCs | 3–50 | 100–500 | ~90 | 50–140 | 8–10 |
| CNFs | 3–60 | ≥103 | 50–90 | 50–160 | 0.8–1 |
| BC | 20–100 | ≥103 | 84–89 | 78 | 0.2–2 |
2.2. Properties Required for the Nanocellulose-Based Materials Utilized in Drug Delivery
Materials used in drug delivery systems need many required properties, such as good drug-loading capacity, biocompatibility, and biodegradability [27]. The drugs must be released with correct concentrations under a proper rate [28,29]. Nanocellulose-based materials are usually used as a drug delivery matrix and drug excipients [30,31]. Herein, the specific requirements of nanocellulose-based materials used for drug delivery are summarized. These requirements can be divided into physical properties, surface chemistry, and biological properties.
2.2.1. Mechanical Properties
Mechanical properties of nanocellulose (such as Young’s modulus, tensile strength, and toughness) play an important role in sustained drug delivery systems [32]. Because of its disordered amorphous regions and ordered crystalline regions, nanocellulose-based composite materials have good mechanical properties [33,34]. The amorphous regions can contribute to the plasticity and flexibility of nanocellulose-based composite materials [35]. In contrast, the crystalline regions determine the elasticity and stiffness of the materials. Moreover, different types of nanocellulose contain different proportions of amorphous and crystalline domains [34,36]. For example, the stiffness of CNCs is caused by the high crystalline region ratio compared to CNFs and BC [23,37]. Moreover, hydrogen bonding plays an important role in the physical properties of nanocellulose-based composite materials [38]. According to the previous theoretical calculations, as hydrogen bonding is taken into consideration, the longitudinal modulus of cellulose I is about 173 GPa, whereas it reduces to 71 GPa without intramolecular hydrogen bonding [39].
2.2.2. Surface Chemistry
The surface properties of nanocellulose can determine the duration and destination of the prepared drug carriers for the drug delivery [40,41]. Pristine nanocellulose cannot be effectively used as a drug carrier due to its limited water solubility and lack of stability in various buffer solutions [42]. The hydroxyl groups in nanocellulose can offer a broad range of surface functionalization and generate the reactive-charged nanocellulose composites [43,44]. In general, the main objective of the surface modification is to introduce new functional groups into the framework of nanocellulose to attach drugs without altering the morphologies, structures, and crystallinities of nanocellulose-based materials [45,46].
2.2.3. Biocompatibility and Toxicity
Biocompatibility is an essential requirement for biomedical materials [47]. It refers to foreign substances embedded in the body being able to exist consistently with tissues without causing injurious changes [48]. It is reported that CNFs prepared by enzymatic hydrolysis have no cytotoxicity at tested concentrations (~10–1000 μg/mL) [49]. In other CNF fabrication processes, it is exposed that some physical, chemical, and even mechanical changes may affect its cytotoxicity to cells. However, many studies have confirmed that there are no signs of toxicity in pure CNFs [50,51], and other studies have reported low toxicity or no obvious toxicity [52]. The biocompatibility of CNFs may be due to their unique 3D nanofibrous network structure, which supports cell penetration and proliferation [53]. De Loid et al. [54] performed the toxicological analyses of depleted CNCs. In vitro experiments showed that there were no significant changes in serum markers, hematology, or histopathology between the control group and the CNC suspension-fed rats. The experiment suggested that ingested CNCs were basically nontoxic and may be harmless when ingested in small amounts. However, the long-term effects of the materials and their effects in vivo have yet to be revealed [55]. Many polymer-based nanoparticles may have side effects on cells, which may take a long time to observe. BC is reported to be nontoxic and does not show any sign of cytotoxicity in mouse subcutaneous tissue. It is useful in the production of tissue-engineered grafts [56]. Since most toxicity studies are conducted through cell tissue culture, it is impossible to give an accurate image of the compatibility of the selected nanocellulose-based materials [57].
Moreover, the biocompatibility of nanocellulose depends on its structural characteristics, application concentrations, research models, cell type, and exposure times. The uptake of nanocellulose uptake into cells is usually low, which will not induce oxidative stress, and will not produce obvious cytotoxic and genotoxic effects. However, macrophages can internalize rod-shaped CNCs due to their phagocytic function, which can lead to moderate to severe inflammatory response. The response is mainly dependent on the functionalization of CNCs [58]. By introducing different chemical groups on the surface of nanocellulose, the proinflammatory response of nanocellulose can be significantly reduced [59]. Therefore, it is necessary to conduct additional immunological studies on nanocellulose-based materials to better understand its impact on innate and adaptive immunity. However, compared with other materials, nanocellulose-based materials are still preferable because their cytotoxicity is relatively low.
2.2.4. Biodegradability
Besides biocompatibility and nontoxicity, biodegradability is another requirement for materials in biomedical applications [60]. A biodegradable material must be degraded in time that matches the regeneration [61]. However, synthetic biopolymers require high energy and temperature to decompose [62]. Cellulose is a well-known natural polymer with biodegradability. It is generally believed that cellulose does undergo chemical decomposition due to an elevated temperature. One of the main volatile decomposition products is levoglucosan (LGA). This process usually leaves a solid carbon residue whose chemical and physical composition are mostly unknown [63]. Moreover, the nanodimensions of cellulose have not lost their biodegradable nature [64]. However, the biodegradability of nanocellulose in animal and human tissues is not clear, since cells are not able to synthesize cellulases. Nonenzymatic, spontaneous biodegradability of cellulose chains may perhaps explain the slow breakdown of unaltered cellulose within the human body [65]. However, this is admittedly conjecture and it has not been adequately studied [65].
2.3. Strategies to Prepare Designed Nanocellulose-Based Materials for Drug Delivery
As discussed above, due to the broad advantages of nanocellulose, including its nanoscale size, high surface area, surface tunable chemistry, good mechanical strength, and biocompatibility, researchers have extensively investigated different nanocellulose-based materials for drug delivery applications [66,67]. Intense efforts have been devoted to improving the properties of nanocellulose-based functional materials to fulfill the demands in drug delivery systems. With the rapid development of technology, nanocellulose has been engineered into multidimensional architectures including 1D, 2D, and 3D, which are further transformed into drug carriers with tailorable structures and properties for different purposes.
2.3.1. Strategies to Prepare 1D Nanocellulose-Based Materials
Commonly, drug carriers should have stable storage of drugs and present controlled-drug-release behavior. Therefore, it should be designed to better control the drug release rate, improve the utilization rate, and reduce the side effects of drugs [68,69]. Nanocellulose-based microspheres have large surface areas and good affinity. They have become a key topic in the research of drug sustained-release systems. Moreover, a variety of methods can be used to modify nanocellulose because of its abundance of surface hydroxyl groups. Surface modification provides a valuable opportunity for controlling the structure–function relationship of nanocellulose [70]. It also can be used to modulate the drug-loading ratio toward nonionized hydrophobic drugs [71]. Ullah et al. [72] fabricated drug-loaded BC microparticles. They found that the drug release rate could be controlled. The microparticles could be tuned for use in many different biomedical applications. Lin et al. [73] used CNCs as chemicals in calcium crosslinked alginate microspheres. The addition of the CNCs stabilized the crosslinked alginate matrix, producing a higher drug encapsulation efficiency of theophylline and obtaining controlled-drug-release behavior.
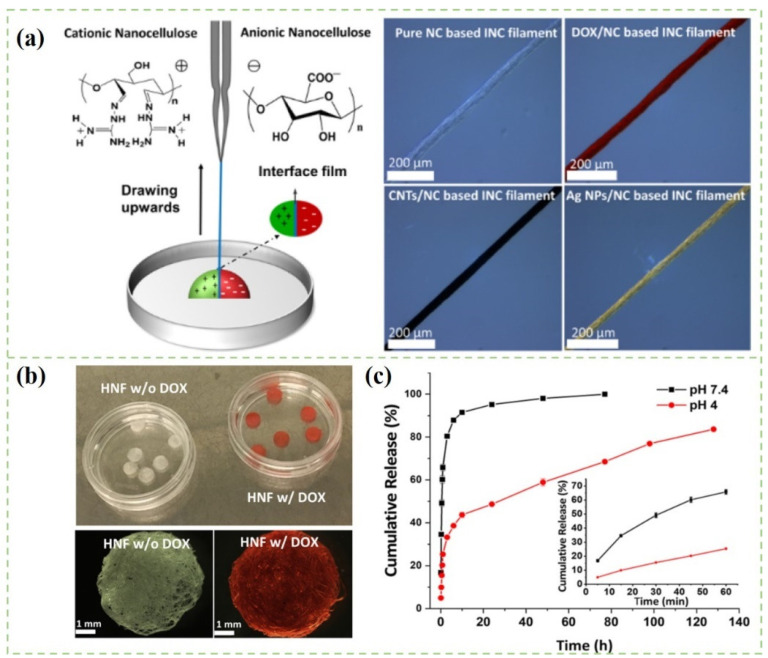
The emulsion method is widely used to prepare drug nanoparticles. The emulsion system is considered as a promising drug delivery system because of its unique characteristics, such as colloidal stability and easy encapsulation of different compounds [74]. It is commonly emulsified and polymerized in aqueous medium by using high-performance surfactant and emulsion stabilizer [75]. Moreover, the encapsulation of drugs in emulsion also helps to preserve and protect drugs from gastrointestinal hydrolysis and enzymatic degradation. As shown in Figure 2, nanocomposite filaments with diverse functions (drug-loading performance, conductivity, or antibacterial properties) were fabricated by the approach of interfacial colloidal nanoparticle complexation (INC), which was fabricated from oppositely charged colloidal nanocelluloses [76]. Figure 2c depicts the cumulative release curves of doxorubicin hydrochloride (DOX) in different pH media. Obviously, the drug release rate was affected by the pH value of the medium solution. The drug release rate of DOX at physiological pH (pH 7.4) was faster than that under acidic conditions (pH 4.0). The differences in the initial burst release behavior in pH 7.4 and 4.0 were presumably attributed to the different swelling ability of the INC filaments in these aqueous buffers. Low et al. [77] reported a Fe3O4@cellulose nanocrystals-stabilized Pickering emulsion containing curcumin for magnetically triggered drug release. The drug release rate was increased after the stimulation of the external magnetic field. An MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) experiment demonstrated that the curcumin-loaded nanomaterials could effectively inhibit human colon cancer cell growth in the presence of a magnetic field. The results suggested that CNCs-based nanomaterials could be used as a promising colloidal drug carrier.
Figure 2.
(a) Schematic illustration of nanocomposite filament; (b) Photography and optical microscopy images of hydroentangled CNCs-based INC filament; (c) Cumulative release profiles of DOX-loaded CNCs-based INC filaments in different pH values at 37 °C. The inset shows the cumulative drug release in the first 1 h [76].
2.3.2. Strategies to Prepare 2D Nanocellulose-Based Materials
Nanocellulose is an excellent film-forming material with a series of properties such as degradability, good biocompatibility, good permeability, and excellent mechanical properties [74,78]. Taking nanocellulose as a film-forming material and supplemented by other functional components, the composite materials with different types can be prepared, such as drug sustained-release films, hemostatic films, and bone repair films [79]. Therefore, nanocellulose usually appears in the form of blend films or composite films as drug carriers [5]. Many antibiotic, antiviral, and anti-inflammatory drugs can be loaded in the nanocellulose-based composite films [80,81].
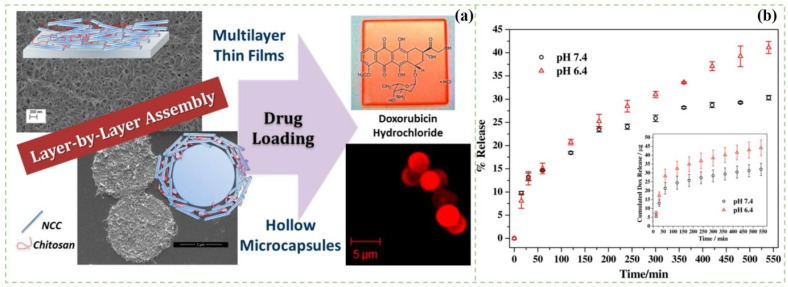
However, the drug-loading ratios and the drug release capabilities are the two important factors which should be considered in designing the nanocellulose-based composite films [82]. As shown in Figure 3, Mohanta et al. prepared a layer-by-layer (LBL)-assembled film for drug delivery by using the complementary electrostatic and hydrogen bond interaction between positively charged chitosan and negatively charged CNCs [83]. The films consisted of porous nanofibers and can be loaded with a large amount of doxorubicin. The loaded doxorubicin hydrochloride was released in a sustained manner in a physiological condition mimicked by PBS buffer of pH 7.4 and pH 6.4. Saidi et al. [84] prepared the BC/poly(N-methacryloyl glycine) (PMGly) composite films for the controlled delivery of diclofenac. The composite films were prepared by in situ polymerization of methacryloyl glycine monomer within the BC network. Diclofenac drugs were loaded into BC/PMGly films by simply immersing wet composite films in the drug solution. The composite films presented a slower drug release rate (9%) under the pH of 2.1 within 24 h, whereas it showed a much higher release (85%) under the pH of 7.4 in the same duration. Therefore, the composite film had controlled and pH-sensitive drug delivery properties, which have potential to be used in both transdermal drug delivery and oral delivery. Poonguzhali et al. [85] prepared alginate/CNCs composite films for in vitro drug release. The results found that CNCs could improve the swelling and mechanical properties of the composite films. Moreover, the composite films exhibited sustained drug release behaviors. In a recent study, authors incorporated honey in the CNC composite films [86]. The drug release ratio was gradually increased and remained consistent for about 48 h in vitro. Moreover, the drug release behaviors of the composite films followed first-order kinetics. Therefore, nanocellulose films have important research value in the areas of sustained and targeted drug delivery.
Figure 3.
(a) Schematic illustration of the preparation of the nanocrystalline/chitosan; (b) Release profile of doxorubicin hydrochloride from the composite film at 37 °C using PBS buffer as release media [83].
2.3.3. Strategies to Prepare 3D Nanocellulose-Based Materials
Nanocellulose-based composite hydrogels are similar to extracellular matrices (ECMs), with highly porous structures, high water retention, good mechanical strength, high specific surface area, and good biocompatibility [87]. The drug molecules can be embedded in the network of the nanocellulose-based composite gels [88]. In the following text, nanocellulose-based composite hydrogels and aerogels used for drug delivery are summarized.
Nanocellulose-Based Composite Hydrogels
Generally, nanocellulose-based composite hydrogels are commonly formed by physical crosslinking [89] or chemical crosslinking methods [90]. Crosslinked biomaterials for drug delivery are commonly needed to have biodegradability, biocompatibility, and adjustable physicochemical properties [91,92,93]. Physical crosslinking methods typically include electrostatic/ionic interactions, hydrophobic interactions, and π–π stacking interactions [94]. Function groups such as hydroxyl groups and carboxyl groups play an important role in the physical crosslinking process [95]. Importantly, the physical crosslinking method can avoid the usage of lethal crosslinking agents which are required in chemical crosslinking methods. Moreover, chemical crosslinking methods typically consist of free radical polymerization, condensation reactions, and aldehyde-mediated reactions [96,97]. Therefore, the abundant active groups such as hydroxyl and carboxyl groups in the nanocellulose backbones make them ideal materials to prepare nanocellulose-based composite hydrogels [98,99].
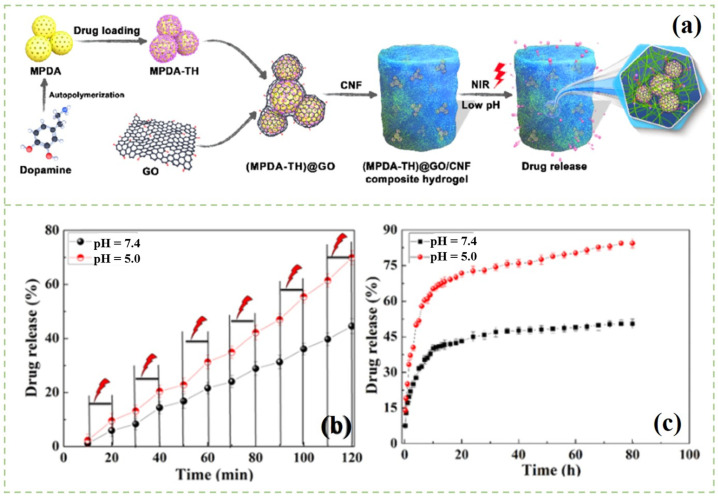
Different types of hydrogels have distinct morphological structures and functional groups, which can affect the drug diffusion paths during the adsorption and release process [100]. In various types of composite hydrogels, smart responsive hydrogels can be used as biomaterials in continuous and targeted drug delivery systems [101]. When the external environment changes, such as temperature, pH, light, and electric field, the smart responsive hydrogel will shrink or expand as required due to the introduction of hydrogen bonds, complexation, ions, noncovalent interactions, and electrostatic interactions [102]. Therefore, the drug molecules loaded in the smart responsive hydrogels can be released from the hydrogels during the above process. As reported previously, Treesuppharat et al. [103] synthesized the composite hydrogel by copolymerization of BC and gelatin. Due to the uniform shape and the size of the BC chains, the prepared hydrogels had thermal stability and the required mechanical properties. Müller et al. [104] fabricated a BC hydrogel as a carrier for the loading of bovine serum albumin. It was found that lyophilized BC hydrogels had lower bovine serum albumin uptake than that of undried BC hydrogels. The drug was released through diffusion and swelling-controlled processes. Moreover, the researchers continued to use luciferase as a model of protein. They found that the activity of the protein may remain unchanged during the binding and releasing from BC hydrogels. Anin, with other collaborators, prepared a BC/acrylic acid (AA) hydrogel [105]. The water absorption results revealed that the maximum swelling of BC/AA composite hydrogels was achieved at a pH of 7 even after 48 h. When the pH was changed to 10, the equilibrium was attained in 24 h. They further found that the composite hydrogels had both pH and thermoresponsive drug release properties [101]. Liu et al. fabricated a porous polydopamine (MPDA)@graphene oxide (GO)/CNFs composite hydrogel using the physical crosslinking method for controllable drug release [106] (Figure 4). They found that near-infrared (NIR) light irradiation and pH change could accelerate the drug release rate. The pH responsiveness may allow the composite hydrogel to release drugs in the bacteria-infected sites under acidic conditions.
Figure 4.
(a) Schematic illustration of the fabrication of the MPDA@GO/CNFs composite hydrogel; (b) NIR-light-triggered drug release behavior from MPDA@GO/CNFs composite hydrogel in different PBS solution at 37 °C; (c) In vitro drug release profiles of the MPDA@GO/CNFs composite hydrogel [106].
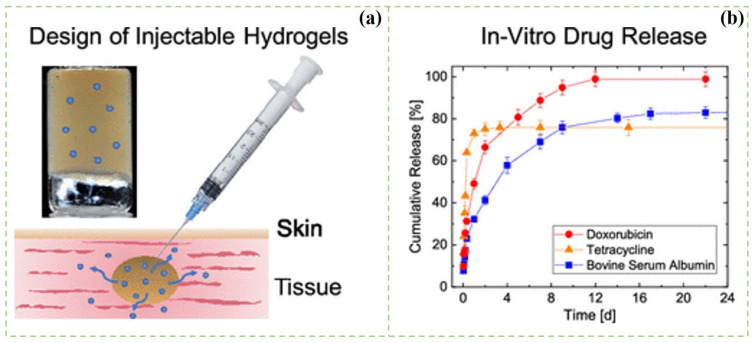
Compared with ordinary hydrogels, injectable hydrogels also have been widely explored as an important part in drug delivery systems. Injectable hydrogels have appeared as promising drug delivery materials because of their properties such as similarity to the ECM, ability to access deep-seated areas, highly porous structure, and capability of enclosing cells within the matrix. A polymer solution with a low viscosity and gel ability after injection can be used to obtain injectable hydrogel. The nucleophilic substitution method can be used to prepare injectable hydrogels for drug delivery and tissue-engineering applications [107,108,109,110]. Injectable hydrogels present a free-flow behavior before injection, but spontaneously change to semisolid hydrogel after gel formation due to the chemical or physical crosslinking reactions. Bertsch et al. prepared the CNC composite injectable hydrogels by salt-induced charge screening (Figure 5). The injectability of the hydrogel was evaluated by the combination of shear rheology and capillary rheology, which showed that the CNC hydrogel was transported through the plug flow in the capillary [111]. The CNC hydrogels were used as drug carriers for the in vitro release of bovine serum albumin (BSA), tetracycline (TC), and doxorubicin (DOX) in normal saline and simulated gastric juice. For TC, a burst release was observed within 2 days, whereas BSA and DOX both showed a sustained release for 2 weeks. The different release behaviors were attributed to drug size, solubility, and specific drug–CNCs interactions. Orasugh et al. [112] added CNCs in the triblock poloxamer copolymer (PM) to obtain composite injectable hydrogels. They found that the PM/CNC hydrogel had good strength and a lower gel temperature, which was attributed to the formation of intermolecular hydrogen bonds between the free hydroxyl group of the CNC molecule and PM molecule. Moreover, injectable hydrogels can undergo reversible phase transitions triggered by pH, temperature, solvent composition, electric field, ionic strength, or light [113].
Figure 5.
(a) Schematic illustration of the preparation of the injectable CNCs hydrogels; (b) In vitro drug release from CNCs hydrogels [111].
Magnetic hydrogel can keep the drug under the electromotive force for a long time [114]. Commonly, the magnetic drug carrier improves the electromotive force of the affected pathological site, and the drug release behavior is controlled by electromotive force [115,116]. Magnetic drug-loaded hydrogels can improve the drug-loading efficacy, reduce the drug dosage, and reduce the damage to normal organs. Supramaniam et al. [117] synthesized magnetic CNCs (m-CNCs) for the loading of ibuprofen. Then, the composite was merged with alginate to fabricate composite hydrogels. The burst release amount (45–60%) was observed from 0 to 30 min. A sustained drug release period was observed from 30 to 330 min.
Nanocellulose-Based Composite Aerogels
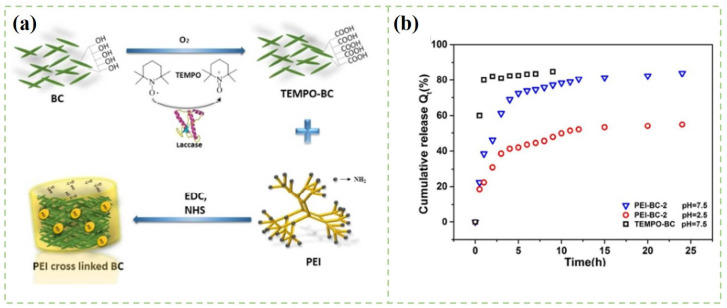
Aerogels are commonly referred to as the porous material with high porosity and low density, which are suitable for the storage of various drugs [118]. Aerogels with adjustable pore size and large pore volume can prevent rapid drug release in undesired areas [119,120]. Nanocellulose is a suitable building block to form aerogels, because of its combined light weight and toughness [121]. Moreover, nanocellulose-based aerogels or their derivatives are unique among solid materials due to their low density, high porosity, and good biocompatibility [122]. In brief, the fabrication process of nanocellulose-based aerogels begins with the formation of hydrogels through the chemical or physical crosslinking method [123]. Additionally, then, the hydrogels are converted into aerogel by different drying techniques such as evaporation, supercritical drying, and the freeze-drying method [124]. Before the drying process, the physical and chemical crosslinking processes are crucial to control the formation of the three-dimensional network and the properties of the porous material [125,126]. Compared with the materials obtained by the physical crosslinking method, chemical crosslinking materials have better mechanical stiffness and structural stability [127]. As shown in Figure 6, Chen et al. fabricated a TEMPO-mediated BC composite aerogel with polyethyleneimine (PEI) as a crosslinker [128]. Aspirin was loaded into the composite aerogel by simple adsorption. The drug release behaviors were investigated in the simulated intestinal fluid (SIF) solutions. The accumulative release ratio of aspirin from composite aerogel was 80.6% in SIF condition under pH of 7.5.
Figure 6.
(a) Schematic illustration of the preparation of the PEI-BC composite aerogels; (b) The cumulative release curves of aspirin [128].
Moreover, the multifunctional nanocellulose-based aerogel can be prepared by grafting functional groups onto cellulose nanofibrils. Controlled-drug-release behaviors can be obtained from different pH/temperature/light conditions [129]. For example, Zhao et al. [130] grafted polyethyleneimine on the surface of CNFs to obtain a composite aerogel used for drug delivery. The composite aerogels had a high drug load capacity (287.39 mg g−1). Due to the amounts of amine group contained in the polyethyleneimine molecule, the obtained composite aerogels presented good temperature and pH-responsive drug release properties.
3. Drug Release Behaviors of Nanocellulose-Based Materials
Drug release behavior is one of the most important properties for drug carriers [131]. The specific surface area of drug carriers plays an important role in controlling the drug-releasing rate [132]. Drug carriers can be used to load hydrophilic or hydrophobic drugs. The hydrophilicity and hydrophobicity of drug carriers not only determine the absorption, distribution, metabolism, and excretion of drugs in vivo, but they also directly affect the value of drugs [133]. For hydrophilic drug carriers, the drug release rate is commonly fast because the surface tension of hydrophilic drug carriers is easy to collapse in aqueous solution. In contrast, the drug release rate of hydrophobic drug carriers is relatively slow [134]. As the structure of hydrophobic materials is more stable in water, the drug release rate of hydrophobic drugs is mainly affected by drug diffusion [135]. In this part, hydrophobic and hydrophilic drug release behaviors of nanocellulose-based materials are summarized.
3.1. Hydrophilic-Drug Release Behaviors
Hydrophilic drugs refer to the drugs that can be dissolved in water. Nanocellulose-based nanocarriers exhibit negative interface charges and high specific surface area, which make them suitable as hydrophilic drug carriers [136]. However, due to their limited water solubility, water sensitivity, and lack of stability in various buffer solutions, the original nanocellulose cannot be effectively used as a drug carrier [35]. Therefore, various methods, such as pretreatment and surface modification, have been developed to overcome these limitations and improve specific properties [137]. Commonly, hydrophilic drugs loaded in the nanocellulose-based carriers via electrostatic attractions or covalent-binding reactions [95]. In addition, hydrophilic drugs usually have problems of low intracellular absorption, enzyme degradation, rapid clearance, poor distribution, drug resistance, poor pharmacokinetics, and low treatment index [138]. Therefore, lower encapsulation efficiency and rapid-release behaviors of hydrophilic drugs in nanocellulose-based carriers are the main problems. To overcome the above problems, many strategies have been developed. Generally, hydrophilic molecules can be first loaded into nanospheres; then, the drug-loaded nanospheres are added in the structure of nanocellulose-based hydrogel materials [106]. Table 2 summarizes the nanocellulose-based materials used in hydrophilic-drug delivery applications. Besides delivering the small drug molecule as listed in Table 2, nanocellulose-based materials can be used to deliver proteins and nucleic acids, because nanocellulose-based materials can meet the strict medical requirements of appropriate carriers for protein and nucleic acid fixation. Basu et al. developed calcium-chloride-crosslinked CNF hydrogels for transporting biomolecules [139]. Bovine serum albumin protein was loaded into the hydrogel by the simple immersion method. The large positively charged proteins promote the sustained drug release behavior of CNFs. The electrostatic interaction between the protein and hydrogel was the main factor to promote the physical adsorption of hydrogel structure stability and activity. Therefore, calcium-crosslinked CNFs hydrogels can transport proteins without affecting their activity.
Table 2.
Nanocellulose-based materials used in hydrophilic-drug delivery applications.
| Dimensions | Drug Delivery Carriers | Hydrophilic-Drug Models |
Drug Release Behaviors | Drug Release Mechanism |
References |
|---|---|---|---|---|---|
| 1D | Poly(lactic acid)/CNCs nanocomposite fibers | Columbia blue | Little burst release (<5%) in the first 4 h. | Fickian diffusion | [140] |
| CNCs-hordein/ zein fibers |
Riboflavin | After 24 h, the cumulative release amount was 26.99%. | - | [141] | |
| CNFs/poly(N-isopropylacrylamide) hybrid microspheres | 5-Fluorouracil | The cumulative drug released amount was 89% within 1 h at 22 °C. | Fickian diffusion | [142] | |
| CNCs/chitosan particles |
Procaine hydrochloride |
In the first 10 min, drug release rate was relatively fast; then, it became slower in the next 1 h. | - | [143] | |
| 2D | Nanocellulose/ pectin films |
Hydroxychloroquine | In the first 2 h, the drug release amount from the pectin films containing CNCs and CNFs was approximately 65% and 95%, respectively. |
Fick’s diffusion | [144] |
| Chitosan/CNCs films | Doxorubicin | Under acidic pH conditions, the drug release amount is higher. |
Fickian diffusion | [83] | |
| CNFs/polyvinyl alcohol films |
Acetaminophen | - | Diffusion controlled and burst release |
[145] | |
| BC composite membranes |
Tetracycline hydrochloride |
The drug release amount was 90% within 10 h in HEPES buffers. | - | [146] | |
| 3D | Polyacrylamide/ CNFs hybrid hydrogels |
Niacinamide | The cumulative drug release amount was 45% with 350 min. | Pseudo-Fickian diffusion |
[147] |
| CNFs/polydopamine composite hydrogels |
Tetracycline hydrochloride |
In acid PBS solution, 70% of the loaded drugs were released after 15 h. |
Anomalous transport | [148] | |
| CNFs/polyethylenimine aerogels | Sodium salicylate | In SIF condition with a pH of 7.4, the cumulative drug release amount was 78.49%. |
Pseudo-second-order release |
[130] | |
| CNFs aerogel | Bendamustine hydrochloride |
The cumulative drug release amount was 78% ± 2.28% in 24 h. | Non-Fickian mechanism | [149] | |
| CNFs/ polyethyleneimine-N/ isopropylacrylamide aerogel |
Doxorubicin | The cumulative drug release amount was 59.45% at pH of 3 and 37 °C. | - | [150] | |
| CNFs/hydroxypropylmethylcellulose nanocomposites |
Ketorolac tromethamine |
The cumulative drug release amount was 95.12% after 8 h under PBS conditions of 7.4. | Non-Fickian diffusion | [151] | |
| Mesoporous polydopamine@ graphene oxide/CNFs composite hydrogel |
Tetracycline hydrochloride |
In the first 1 h, burst release amount was 14% in PBS 7.4 solution. The maximum TH release (84.3%) was achieved in 72 h in PBS 5.0 solution. |
Anomalous transport | [106] |
In vitro drug release study is the basic evaluation experiments to determine the suitability of nanocellulose-based drug delivery materials, which are carried out in either phosphate-buffered saline or mimicking-release medium [151]. Cellulose-based sheet materials with antibacterial and wound-healing properties have been reported [152]. Liu et al. [153] reported a nanocellulose-based hydrogel with a packaging structure for on-demand drug delivery. Zeolite imidazolate skeleton-8 (ZIF-8) was grown on the surface of polydopamine (PDA) to obtain PDA@ZIF-8 nanocomposites. Then, PDA@ZIF-8 nanocomposites were added to the CNF network to prepare PDA@ZIF-8/CNF composite hydrogels. Slight burst release behaviors were observed at the beginning. The composite hydrogel also presented a pH-dependent drug release behavior. Moreover, NIR light irradiation can accelerate the drug delivery rate.
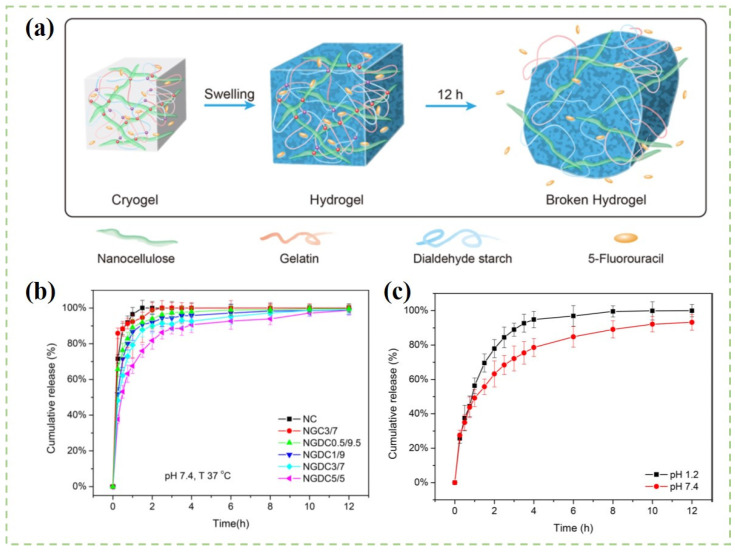
In order to effectively adsorb drugs onto the surface of nanocellulose drug carriers, the physicochemical properties of drugs and nanocellulose materials should be considered. Bhandari et al. [149] found that CNF aerogel was suitable for loading water-soluble bendamustine hydrochloride drugs because of the physical adsorption properties of CNF aerogel. The drug-loading ratio of CNF aerogel was 18.98%. The drug release amount was different in different pH conditions. Approximately 69.2% of the drug was released in 24 h at the pH of 1.2, whereas 78% of the drug was released at the pH of 7.4 within 7.5 h. As shown in Figure 7, Li and coworkers [154] synthesized CNF/gelatin aerogels through the chemical-crosslinking method. The composite aerogels were further used as carriers for the controlled released of 5-fluorouracil (5-FU). The 5-FU powder was added into the CNF solution before hydrogel formation, and the maximum encapsulation efficiency of the drug was about 40%. The cumulative release of composite aerogels was close to 100% after 12 h. Moreover, the controlled and continuous release of drugs was realized, as the drugs should be dissolved from the carrier matrix and then diffused through the structure of the network. Cacicedo et al. [155] mixed lipid nanoparticles loaded with doxorubicin into BC composite hydrogels. They found that the drug encapsulation efficiency of neutral doxorubicin was two times bigger than that of cationic doxorubicin. Moreover, the composite hydrogel showed a sustained drug release behavior toward neutral doxorubicin. In vivo analysis showed that the growth of tumors and metastatic events were decreased by a combination of these two drugs.
Figure 7.
(a) Schematic illustration of the preparation of the 5-fluorouracil-loaded CNF/gelation hydrogels; (b) Drug release profiles of different hydrogels; (c) Drug release profiles of the hydrogel at different pH environments [154].
3.2. Hydrophobic-Drug Release Behaviors
Hydrophobic drugs refer to the substances that are less water-soluble but soluble in organic solvents [156]. It is estimated that about 40% of marketed drugs and 60% of compounds in development status are poorly water-soluble [157]. The lower drug-loading ratio is one of the major challenges which hinders their practical applications [158]. As the presence of reactive functional groups on the nanocellulose backbones, a wide range of surface modifications have been carried out to improve their binding properties toward hydrophobic drugs. Table 3 summarizes the nanocellulose-based materials used in hydrophobic-drug delivery applications.
Table 3.
Nanocellulose-based materials used in hydrophobic-drug delivery applications.
| Dimensions | Drug Delivery Carriers | Hydrophobic-Drug Models | Drug Release Behaviors | Drug Release Mechanism |
References |
|---|---|---|---|---|---|
| 1D | CNCs–cetyltrimethylammonium bromide suspensions |
Paclitaxel, docetaxel, and etoposide |
A total of 75% of the drug was released over 4 days. | - | [159] |
| CNCs/rarasaponin particles |
Tetracycline | More drugs released from neutral condition than in acid condition. |
Pseudo-first-order | [160] | |
| 2D | BC/hyaluronic acid/ diclofenac films |
Diclofenac sodium |
The maximum cumulative release was 90% which was obtained after 4 min in simulated salivary fluid. |
Non-Fickian transport |
[161] |
| CNFs/poly(glycerol sebacate)/polypyrrole patches |
Curcumin | The cumulative drug-released amount was less than 2% with five months in PBS under pH of 7.4. | - | [162] | |
| BC/polyvinyl alcohol films | Vanillin | The diffusion process reached equilibrium after 1 h in water. |
Fickian diffusion |
[163] | |
| 3D | BC/sodium alginate hybrid hydrogels | Ibuprofen | During the first 2 h, the drug release amount was less than 10% in acidic conditions with the pH condition of 1.5. |
Non-Fickian diffusion |
[164] |
| Polyethylenimine/BC aerogels | Aspirin | The cumulative drug release was 80.6% with 25 h in pH condition of 7.5. | - | [128] | |
| Zeolitic imidazolate framework-8@PCNFs composite hydrogel | Curcumin | Under pH condition of 2.5, the maximum curcumin release amount was 90%. |
Anomalous transport |
[165] |
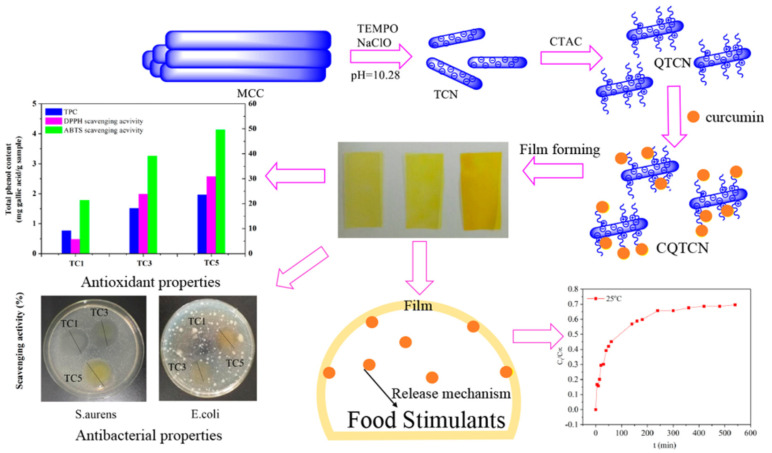
Due to the problems of poor solubility, poor dispersion, lack of uniformity, low bioavailability, and lack of stability, the therapeutic effect of many hydrophobic drugs is limited [166]. However, when drugs are uploaded into the hydrogel system, these defects can be improved to some extent, resulting in solubilization, slow release or controlled release effects, and enhanced stability and biological activity. As shown in Figure 8, Ma et al. added CNCs loaded with curcumin into tara gum/polyvinyl alcohol blend membrane to prepare antioxidant and antibacterial membranes [167]. The release test revealed that curcumin was initially released rapidly into 50% ethanol solution and then released more slowly into the bulk. Moreover, higher temperature could accelerate the release of curcumin. Liu et al. [165] used the one-pot method to grow ZIF-8 and curcumin (Cur) on the surface of PDA-modified CNFs composite hydrogel (PCNFs) to form ZIF-8@PCNFs-Cur composite hydrogel. The loading of ZIF-8 nanoparticles in the PCNFs composite hydrogel structure was expected to increase the loading rate of hydrophobic drugs and prolong the drug release time. The maximum drug encapsulation efficiency and drug-loading ratio of the composite hydrogel were 82 wt% and 4.5 wt%, respectively. It was found that the composite hydrogel had good mechanical properties and sustained drug release properties. In addition, lower pH condition and near-infrared light irradiation could accelerate the drug release behavior. The maximum drug release time of ZIF-8@PCNFs composite hydrogel was 107 h. The mechanism was confirmed as abnormal transport. Plappert et al. [168] found that the surface charge density and carboxylate content in CNF membranes can increase the adsorption ratio of hydrophobic piroxicam. In vitro drug release time was prolonged under simulated human skin conditions. Their findings confirmed that nanofibrous membranes can be potentially used as transdermal drug delivery patches.
Figure 8.
Schematic illustration of curcumin-loaded membrane and the drug release curve of curcumin [167].
Targeted drug delivery and controlled drug release rate are attractive methods to avoid the sudden drug release behaviors [169]. Anirudhan et al. [170] prepared a nanocellulose-based carrier for effective delivery of curcumin. Under the pH of 8.0, the drug-loading ratio was as high as 89.2%. At acidic pH conditions, almost 91.0% of the drug was released within 48 h. Due to the protonation of imine, carboxyl, and hydroxyl groups in the material, the curcumin release amount at pH 5.5 was higher than that at pH 1.2 and 7.4. The electron repulsion may cause expansion in the material and promote the release of curcumin [171]. Luo et al. [172] synthesized BC/graphene oxide composites via a biosynthesis technique as a novel drug delivery carrier for ibuprofen release. The results showed that the presence of graphene oxide promoted the drug-loading ratio and prolonged the sustained release time. The reasons may be related to the increased specific surface area. In vitro drug release studies indicated that the drug release behavior of the composite material followed a non-Fickian diffusion mechanism.
4. Drug Release Mechanisms and Mathematical Models of Nanocellulose-Based Materials
Generally, drug release refers to the process in which drug molecules are transferred from the interior to the outer surface of the drug carriers, and finally released into the surrounding environment [173]. The process is governed by the random motion of drug molecules, driven by chemical potential gradients and convection created by osmotic pressure [174]. For the drug carriers with degradability, the drug release rate is mainly controlled by the diffusion of the carrier’s network. As for nondegradable drug carriers, diffusion is the main driving force for drug release. As for nanocellulose-based carriers, drug release can be predicted by the diffusion rate of the carrier [175]. Moreover, the interactions between drug models and nanocellulose backbones as well as the drug solubility in dissolution medium are the main reasons for the determination of the drug release kinetics. As is shown in Table 2 and Table 3, the sustained drug release mechanisms involved in different drug carriers are different. Kolakovic et al. [176] fabricated CNF films with different drugs including indomethacin, itraconazole, and beclomethasone for the sustained drug release applications. They found that the model drugs could be released from the carriers continuously at an interval of three months. The tight fiber network formed by CNFs around drug particles protected them from the impact of the liquid medium environment. The CNF network is also used to maintain the dissolution of drug molecules and create obstacles for the drug’s diffusion. However, the drug release kinetics of different drug models were different. The release kinetics of the indomethacin drug was diffusion-limiting kinetics. However, the release kinetics of itraconazole and beclomethasone were zero-order release. Considering the slower release rate of drugs, the system can be used in parenteral (implant), local (transdermal patch), or ocular applications.
Mathematical models of drug release are aimed to predict drug release rates and drug diffusion behaviors [177]. It helps to optimize drug release kinetics and determine the physical mechanisms by comparing experimental data with mathematical models [178]. Mathematical models can explain the effects of various parameters on the drug release rate, such as the size, shape, and composition of the drug release system. After fitting by a mathematical model, identified drug release behaviors can be used to predict more effective drug formulations and accurate dosing plans [179]. At present, a series of mathematical models such as the zero-order model (Equation (1)), the first-order model (Equation (2)), the Higuchi model (Equation (3)), and the Korsmeyer–Peppas model (Equation (4)) have been proposed and used to predict and explain drug release behaviors [148,180]. However, these models do not limit the shapes, properties, and structures of different drug carriers. The errors may exist in the simulation of drug release behavior [181]. Therefore, more research is needed to reveal the mechanism of nanocellulose-based drug carriers to achieve better drug release behaviors, which will be one of the future research directions.
| (1) |
| (2) |
| (3) |
| (4) |
In all the above models, M∞ and Mt are the cumulative drug release amount at time t and infinite time. is the cumulative drug release fraction. n is the drug release exponents from the drug release mechanism (Table 4). K0, K1, K2, and K3 are the drug release rate constants of Equations (1)–(4), respectively.
Table 4.
Exponent n and the drug release mechanism from the controlled-drug-delivery carriers of different geometry [148].
| Exponent, n | Drug Release Mechanisms | ||
|---|---|---|---|
| Thin Films | Cylinders | Spheres | |
| 0.5 | 0.45 | 0.43 | Fickian diffusion |
| 0.5 < n < 1 | 0.45 < n < 0.89 | 0.43 < n < 0.85 | Anomalous transport |
| 1 | 0.89 | 0.85 | Case-II transport |
5. Conclusions and Perspectives
Nanocellulose has been proved to be one of the most prominent green materials in various applications, and it has attracted great interest in academic research and industrial applications, as evidenced by more than 4500 relevant patents and commercial products around the world. This review comprehensively introduced the latest research progress of different nanocellulose-based composites in drug delivery. The unique properties of the materials used in drug delivery systems are discussed systematically. As a natural material, nanocellulose has the potential to be involved in future medical applications. The current research is still at the scientific research stage, and we need to focus on in vivo drug release research on animals in the future. More long-term studies are needed to analyze and assess the potential effects of nanocellulose on humans. Nonetheless, it is difficult for the human body to degrade nanocellulose-based materials, and the interaction mechanism between nanocellulose and cells is still unclear. In the future, it is necessary to explore whether the introduction of nanocellulose is potentially harmful to the skin, such as causing skin hyperplasia, scars, or other complications.
From a scientific and economic point of view, nanocellulose is a resource and gift provided by nature. Driven by the recent extraordinary activities in the field of biomedical applications, nanocellulose will make a breakthrough in drug delivery systems. Moreover, with the continuous optimization and commercialization of nanocellulose, nanocellulose-based composite materials with better-designed structures and multifunctions (e.g., pH or NIR responses) will be undoubtedly extensively used in the field of drug delivery. Despite nanocellulose-based composites having already shown great promise in biomedical fields, the large-scale commercial applications of nanocellulose are closely related to the structure and performance of the materials. Thus, we need commit to solving the current difficulties and challenges. Therefore, with the continuous optimization of nanocellulose production, modification, industrialization, and commercialization, nanocellulose will undoubtedly shine in more fields.
Author Contributions
Conceptualization, Y.H. and Y.L.; redraw the figures, Y.H. and M.X.; drafting the manuscript, Y.H., Y.L., M.X. and H.D.; review and editing, Z.L., B.L. and H.L.; proof reading, Y.L., B.L. and H.L. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research was funded by National Natural Science Foundation of China (No. 32001281); Natural Science Foundation of Tianjin (No. 20JCQNJC00110); Guangxi Key Laboratory of Clean Pulp & Papermaking and Pollution Control; College of Light Industry and Food Engineering; Guangxi University (No. 2019KF10); Shandong Provincial Natural Science Foundation for Distinguished Young Scholars (China) (grant number ZR2019JQ10); Qingdao independent innovation major project (grant number 21-1-2-23-hz); Shandong Energy Institute Research Foundation (SEI S202106); as well as Key Laboratory of Pulp and Paper Science & Technology of Ministry of Education of China (No. KF202010).
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Das S., Ghosh B., Sarkar K. Nanocellulose as sustainable biomaterials for drug delivery. Sens. Int. 2022;3:100135. doi: 10.1016/j.sintl.2021.100135. [DOI] [Google Scholar]
- 2.Hasan N., Rahman L., Kim S.-H., Cao J., Arjuna A., Lallo S., Jhun B.H., Yoo J.-W. Recent advances of nanocellulose in drug delivery systems. J. Pharm. Investig. 2020;50:553–572. doi: 10.1007/s40005-020-00499-4. [DOI] [Google Scholar]
- 3.Li X., Liu Y., Yu Y., Chen W., Liu Y., Yu H. Nanoformulations of quercetin and cellulose nanofibers as healthcare supplements with sustained antioxidant activity. Carbohydr. Polym. 2019;207:106–168. doi: 10.1016/j.carbpol.2018.11.084. [DOI] [PubMed] [Google Scholar]
- 4.Miles C.E., Gwin C., Zubris K. Tyrosol derived poly(ester-arylate)s for sustained drug delivery from microparticles. ACS Biomater. Sci. Eng. 2021;7:2580–2591. doi: 10.1021/acsbiomaterials.1c00448. [DOI] [PubMed] [Google Scholar]
- 5.Lin N., Dufresne A. Nanocellulose in biomedicine: Current status and future prospect. Eur. Polym. J. 2014;59:302–325. doi: 10.1016/j.eurpolymj.2014.07.025. [DOI] [Google Scholar]
- 6.Ahmad S.I., Ahmad R., Khan M.S., Kant R., Shahid S., Gautam L., Hasan G.M., Hassan M.I. Chitin and its derivatives: Structural properties and biomedical applications. Int. J. Biol. Macromol. 2020;164:526–539. doi: 10.1016/j.ijbiomac.2020.07.098. [DOI] [PubMed] [Google Scholar]
- 7.Srivastava N., Richa, Choudhury A.R. Recent advances in composite hydrogels prepared solely from polysaccharides. Colloids Surf. B Biointerfaces. 2021;205:111891. doi: 10.1016/j.colsurfb.2021.111891. [DOI] [PubMed] [Google Scholar]
- 8.Zaman A., Huang F., Jiang M., Wei W., Zhou Z. Preparation, properties, and applications of natural cellulosic aerogels: A review. Energy Built Environ. 2020;1:60–76. doi: 10.1016/j.enbenv.2019.09.002. [DOI] [Google Scholar]
- 9.Karimian A., Parsian H., Majidinia M., Rahimi M., Mir S.M., Samadi Kafil H., Shafiei-Irannejad V., Kheyrollah M., Ostadi H., Yousefi B. Nanocrystalline cellulose: Preparation, physicochemical properties, and applications in drug delivery systems. Int. J. Biol. Macromol. 2019;133:850–859. doi: 10.1016/j.ijbiomac.2019.04.117. [DOI] [PubMed] [Google Scholar]
- 10.Thomas B., Raj M.C., Athira K.B., Rubiyah M.H., Joy J., Moores A., Drisko G.L., Sanchez C. Nanocellulose, a versatile green platform: From biosources to materials and their applications. Chem. Rev. 2018;118:11575–11625. doi: 10.1021/acs.chemrev.7b00627. [DOI] [PubMed] [Google Scholar]
- 11.Paukkonen H., Kunnari M., Lauren P., Hakkarainen T., Auvinen V.V., Oksanen T., Koivuniemi R., Yliperttula M., Laaksonen T. Nanofibrillar cellulose hydrogels and reconstructed hydrogels as matrices for controlled drug release. Int. J. Pharm. 2017;532:269–280. doi: 10.1016/j.ijpharm.2017.09.002. [DOI] [PubMed] [Google Scholar]
- 12.Wahid F., Huang L.H., Zhao X.Q., Li W.C., Wang Y.Y., Jia S.R., Zhong C. Bacterial cellulose and its potential for biomedical applications. Biotechnol. Adv. 2021;53:107856. doi: 10.1016/j.biotechadv.2021.107856. [DOI] [PubMed] [Google Scholar]
- 13.Seabra A.B., Bernardes J.S., Favaro W.J., Paula A.J., Duran N. Cellulose nanocrystals as carriers in medicine and their toxicities: A review. Carbohydr. Polym. 2018;181:514–527. doi: 10.1016/j.carbpol.2017.12.014. [DOI] [PubMed] [Google Scholar]
- 14.Khalid M.Y., Rashid A.A., Arif Z.U., Ahmed W., Arshad H. Recent advances in nanocellulose-based different biomaterials: Types, properties, and emerging applications. J. Mater. Res. Technol. 2021;14:2601–2623. doi: 10.1016/j.jmrt.2021.07.128. [DOI] [Google Scholar]
- 15.Ghasemlou M., Daver F., Ivanova E.P., Habibi Y., Adhikari B. Surface modifications of nanocellulose: From synthesis to high-performance nanocomposites. Prog. Polym. Sci. 2021;119:101418. doi: 10.1016/j.progpolymsci.2021.101418. [DOI] [Google Scholar]
- 16.Voisin H., Bergstrom L., Liu P., Mathew A.P. Nanocellulose-Based Materials for Water Purification. Nanomaterials. 2017;7:57. doi: 10.3390/nano7030057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Menon M.P., Selvakumar R., Kumar P.S., Ramakrishna S. Extraction and modification of cellulose nanofibers derived from biomass for environmental application. RSC Adv. 2017;7:42750–42773. doi: 10.1039/C7RA06713E. [DOI] [Google Scholar]
- 18.Lavoine N., Desloges I., Dufresne A., Bras J. Microfibrillated cellulose—Its barrier properties and applications in cellulosic materials: A review. Carbohydr. Polym. 2012;90:735–764. doi: 10.1016/j.carbpol.2012.05.026. [DOI] [PubMed] [Google Scholar]
- 19.Kwon G., Lee K., Kim D., Jeon Y., Kim U.J., You J. Cellulose nanocrystal-coated TEMPO-oxidized cellulose nanofiber films for high performance all-cellulose nanocomposites. J. Hazard. Mater. 2020;398:123100. doi: 10.1016/j.jhazmat.2020.123100. [DOI] [PubMed] [Google Scholar]
- 20.Xue Y., Mou Z., Xiao H. Nanocellulose as sustainable biomass material: Structure, properties, present status and future prospects in biomedical applications. Nanoscale. 2017;9:14758–14781. doi: 10.1039/C7NR04994C. [DOI] [PubMed] [Google Scholar]
- 21.Saito T., Kimura S., Nishiyama Y., Isogai A. Cellulose nanofibers prepared by TEMPO-mediated oxidation of native cellulose. Biomacromolecules. 2007;8:2485. doi: 10.1021/bm0703970. [DOI] [PubMed] [Google Scholar]
- 22.Habibi Y., Lucia L.A., Rojas O.J. Cellulose nanocrystals: Chemistry, self-assembly, and applications. Chem. Rev. 2010;110:3479–3500. doi: 10.1021/cr900339w. [DOI] [PubMed] [Google Scholar]
- 23.Huang Y., Zhu C., Yang J., Nie Y., Chen C., Sun D. Recent advances in bacterial cellulose. Cellulose. 2014;21:1–30. doi: 10.1007/s10570-013-0088-z. [DOI] [Google Scholar]
- 24.Liu W., Du H., Zhang M., Liu K., Liu H., Xie H., Zhang X., Si C. Bacterial cellulose-based composite scaffolds for biomedical applications: A review. ACS Sustain. Chem. Eng. 2020;8:7536–7562. doi: 10.1021/acssuschemeng.0c00125. [DOI] [Google Scholar]
- 25.Klemm D., Kramer F., Moritz S., Lindstrom T., Ankerfors M., Gray D., Dorris A. Nanocelluloses: A new family of nature-based materials. Angew. Chem. 2011;50:5438–5466. doi: 10.1002/anie.201001273. [DOI] [PubMed] [Google Scholar]
- 26.Gorgieva S., Trček J. Bacterial cellulose: Production, modification and perspectives in biomedical applications. Nanomaterials. 2019;9:1352. doi: 10.3390/nano9101352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sunasee R., Hemraz U.D., Ckless K. Cellulose nanocrystals: A versatile nanoplatform for emerging biomedical applications. Expert Opin. Drug Deliv. 2016;13:1243–1256. doi: 10.1080/17425247.2016.1182491. [DOI] [PubMed] [Google Scholar]
- 28.Dash R., Ragauskas A.J. Synthesis of a novel cellulose nanowhisker-based drug delivery system. RCS Adv. 2012;2:3403–3409. doi: 10.1039/c2ra01071b. [DOI] [Google Scholar]
- 29.Grishkewich N., Mohammed N., Tang J., Tam K.C. Recent advances in the application of cellulose nanocrystals. Curr. Opin. Colloid Interface Sci. 2017;29:32–45. doi: 10.1016/j.cocis.2017.01.005. [DOI] [Google Scholar]
- 30.Fakes M.G., Vakkalagadda B.J., Qian F., Desikan S., Gandhi R.B., Lai C., Hsieh A., Franchini M.K., Toale H., Brown J. Enhancement of oral bioavailability of an HIV-attachment inhibitor by nanosizing and amorphous formulation approaches. Int. J. Pharm. 2009;370:167–174. doi: 10.1016/j.ijpharm.2008.11.018. [DOI] [PubMed] [Google Scholar]
- 31.Babu R.P., O’Connor K., Seeram R. Current progress on bio-based polymers and their future trends. Prog. Biomater. 2013;2:8. doi: 10.1186/2194-0517-2-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Clark A., Milbrandt T.A., Hilt J.Z., Puleo D.A. Mechanical properties and dual drug delivery application of poly(lactic-co-glycolic acid) scaffolds fabricated with a poly(β-amino ester) porogen. Acta Biomater. 2014;10:2125–2132. doi: 10.1016/j.actbio.2013.12.061. [DOI] [PubMed] [Google Scholar]
- 33.Patil T.V., Patel D.K., Dutta S.D., Ganguly K., Santra T.S., Lim K.T. Nanocellulose, a versatile platform: From the delivery of active molecules to tissue engineering applications. Bioact. Mater. 2022;9:566–589. doi: 10.1016/j.bioactmat.2021.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Plackett D.V., Lwetchford K., Jackson J.K., Burt H.M. A review of nanocellulose as a novel vehicle for drug delivery. Nord. Pulp Pap. Res. J. 2014;29:105–118. doi: 10.3183/npprj-2014-29-01-p105-118. [DOI] [Google Scholar]
- 35.Trache D., Tarchoun A.F., Derradji M., Hamidon T.S., Hussin M.H. Nanocellulose: From fundamentals to advanced applications. Front. Chem. 2020;8:392. doi: 10.3389/fchem.2020.00392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jonoobi M., Oladi R., Davoudpour Y., Oksman K., Dufresne A., Hamzeh Y., Davoodi R. Different preparation methods and properties of nanostructured cellulose from various natural resources and residues: A review. Cellulose. 2015;22:935–969. doi: 10.1007/s10570-015-0551-0. [DOI] [Google Scholar]
- 37.Aulin C., Ahola S., Josefsson P., Nishino T., Wagberg L. Nanoscale cellulose films with different crystallinities and mesostructures—Their surface properties and interaction with water. Langmuir. 2009;25:7675–7685. doi: 10.1021/la900323n. [DOI] [PubMed] [Google Scholar]
- 38.Wohlert M., Benselfelt T., Wågberg L., Furó I., Berglund L.A., Wohlert J. Cellulose and the role of hydrogen bonds: Not in charge of everything. Cellulose. 2021;29:1–23. doi: 10.1007/s10570-021-04325-4. [DOI] [Google Scholar]
- 39.Tashiro K., Kobayashi M. Calculation of crystallite modulus of native cellulose. Polym. Bull. 1985;14:213–218. doi: 10.1007/BF00254940. [DOI] [Google Scholar]
- 40.Mitchell M.J., Billingsley M.M., Haley R.M., Wechsler M.E., Langer R. Engineering precision nanoparticles for drug delivery. Nat. Rev. Drug Discov. 2020;20:101–124. doi: 10.1038/s41573-020-0090-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lunardi V.B., Soetaredjo F.E., Putro J.N., Santoso S.P., Yuliana M., Sunarso J., Ju Y.H., Ismadji S. Nanocelluloses: Sources, pretreatment, isolations, modification, and its application as the drug carriers. Polymers. 2021;13:2052. doi: 10.3390/polym13132052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Araki J. Electrostatic or steric—Preparations and characterizations of well-dispersed systems containing rod-like nanowhiskers of crystalline polysaccharides. Soft Matter. 2013;9:4125–4141. doi: 10.1039/c3sm27514k. [DOI] [Google Scholar]
- 43.Iwamoto S., Kai W., Isogai A., Iwata T. Elastic modulus of single cellulose microfibrils from tunicate measured by atomic force microscopy. Biomacromolecules. 2009;10:2571–2576. doi: 10.1021/bm900520n. [DOI] [PubMed] [Google Scholar]
- 44.Hare J.I., Lammers T., Ashford M.B., Puri S., Storm G., Barry S.T. Challenges and strategies in anti-cancer nanomedicine development: An industry perspective. Adv. Drug Deliv. Rev. 2016;108:25–38. doi: 10.1016/j.addr.2016.04.025. [DOI] [PubMed] [Google Scholar]
- 45.Habibi Y. Key advances in the chemical modification of nanocelluloses. Chem. Soc. Rev. 2014;45:1519–1542. doi: 10.1039/C3CS60204D. [DOI] [PubMed] [Google Scholar]
- 46.Chandel A.K.S., Shimizu A., Hasegawa K., Ito T. Advancement of biomaterial-based postoperative adhesion barriers. Macromol. Biosci. 2021;21:2000395. doi: 10.1002/mabi.202000395. [DOI] [PubMed] [Google Scholar]
- 47.Dugan J.M., Gough J.E., Eichhorn S.J. Bacterial cellulose scaffolds and cellulose nanowhiskers for tissue engineering. Nanomedicine. 2013;8:287–298. doi: 10.2217/nnm.12.211. [DOI] [PubMed] [Google Scholar]
- 48.Dugan J.M., Collins R.F., Gough J.E., Eichhorn S.J. Oriented surfaces of adsorbed cellulose nanowhiskers promote skeletal muscle myogenesis. Acta Biomater. 2013;9:4707–4715. doi: 10.1016/j.actbio.2012.08.050. [DOI] [PubMed] [Google Scholar]
- 49.Kumari P., Pathak G., Gupta R., Sharma D., Meena A. Cellulose nanofibers from lignocellulosic biomass of lemongrass using enzymatic hydrolysis: Characterization and cytotoxicity assessment. Daru. 2019;27:683–693. doi: 10.1007/s40199-019-00303-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Alexandrescu L., Syverud K., Gatti A., Chinga-Carrasco G. Cytotoxicity tests of cellulose nanofibril-based structures. Cellulose. 2013;20:1765–1775. doi: 10.1007/s10570-013-9948-9. [DOI] [Google Scholar]
- 51.Souza S.F., Mariano M., Reis D., Lombello C.B., Ferreira M., Sain M. Cell interactions and cytotoxic studies of cellulose nanofibers from Curaua natural fibers. Carbohydr. Polym. 2018;201:87–95. doi: 10.1016/j.carbpol.2018.08.056. [DOI] [PubMed] [Google Scholar]
- 52.Wang X., Cheng F., Liu J., Smatt J.H., Gepperth D., Lastusaari M., Xu C., Hupa L. Biocomposites of copper-containing mesoporous bioactive glass and nanofibrillated cellulose: Biocompatibility and angiogenic promotion in chronic wound healing application. Acta Biomater. 2016;46:286–298. doi: 10.1016/j.actbio.2016.09.021. [DOI] [PubMed] [Google Scholar]
- 53.Czaja W., Krystynowicz A., Bielecki S., Brown R.M., Jr. Microbial cellulose—The natural power to heal wounds. Biomaterials. 2006;27:145–151. doi: 10.1016/j.biomaterials.2005.07.035. [DOI] [PubMed] [Google Scholar]
- 54.DeLoid G.M., Cao X., Molina R.M., Silva D.I., Bhattacharya K., Ng K.W., Loo S.C.J., Brain J.D., Demokritou P. Toxicological effects of ingested nanocellulose in in vitro intestinal epithelium and in vivo rat models. Environ. Sci. Nano. 2019;6:2105–2115. doi: 10.1039/C9EN00184K. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hadrup N., Knudsen K.B., Berthing T., Wolff H., Bengtson S., Kofoed C., Espersen R., Højgaard C., Winther J.R., Willemoes M., et al. Pulmonary effects of nanofibrillated celluloses in mice suggest that carboxylation lowers the inflammatory and acute phase responses. Environ. Toxicol. Pharmacol. 2019;66:116–125. doi: 10.1016/j.etap.2019.01.003. [DOI] [PubMed] [Google Scholar]
- 56.Pertile R.A., Moreira S., Gil da Costa R.M., Correia A., Guardao L., Gartner F., Vilanova M., Gama M. Bacterial cellulose: Long-term biocompatibility studies. J. Biomater. Sci. Polym. Ed. 2012;23:1339–1354. doi: 10.1163/092050611X581516. [DOI] [PubMed] [Google Scholar]
- 57.Ventura C., Pinto F., Lourenço A.F., Ferreira P.J.T., Louro H., Silva M.J. On the toxicity of cellulose nanocrystals and nanofibrils in animal and cellular models. Cellulose. 2020;27:5509–5544. doi: 10.1007/s10570-020-03176-9. [DOI] [Google Scholar]
- 58.Abitbol T., Rivkin A., Cao Y., Nevo Y., Abraham E., Ben-Shalom T., Lapidot S., Shoseyov O. Nanocellulose, a tiny fiber with huge applications. Curr. Opin. Biotech. 2016;39:76–88. doi: 10.1016/j.copbio.2016.01.002. [DOI] [PubMed] [Google Scholar]
- 59.Čolić M., Tomić S., Bekić M. Immunological aspects of nanocellulose. Immunol. Lett. 2020;222:80–89. doi: 10.1016/j.imlet.2020.04.004. [DOI] [PubMed] [Google Scholar]
- 60.Petersen N., Gatenholm P. Bacterial cellulose-based materials and medical devices: Current state and perspectives. Appl. Microbiol. Biotechnol. 2011;91:1277–1286. doi: 10.1007/s00253-011-3432-y. [DOI] [PubMed] [Google Scholar]
- 61.Laurencin N. Biodegradable polymers as biomaterials. Prog. Polym. Sci. 2007;6:775–795. doi: 10.1016/j.progpolymsci.2007.05.017. [DOI] [Google Scholar]
- 62.Muneer F., Nadeem H., Arif A., Zaheer W. Bioplastics from Biopolymers: An eco-friendly and sustainable solution of plastic pollution. Polym. Sci. Ser. C. 2021;63:47–63. doi: 10.1134/S1811238221010057. [DOI] [Google Scholar]
- 63.Paajanen A., Vaari J. High-temperature decomposition of the cellulose molecule: A stochastic molecular dynamics study. Cellulose. 2017;24:2713–2725. doi: 10.1007/s10570-017-1325-7. [DOI] [Google Scholar]
- 64.Favi P.M., Ospina S.P., Kachole M., Gao M., Atehortua L., Webster T.J. Preparation and characterization of biodegradable nano hydroxyapatite–bacterial cellulose composites with well-defined honeycomb pore arrays for bone tissue engineering applications. Cellulose. 2016;23:1263–1282. doi: 10.1007/s10570-016-0867-4. [DOI] [Google Scholar]
- 65.Czaja W.K., Young D.J., Kawecki M., Brown R.M. The future prospects of microbial cellulose in biomedical applications. Biomacromolecules. 2007;8:1–12. doi: 10.1021/bm060620d. [DOI] [PubMed] [Google Scholar]
- 66.Duran N., Paula Lemes A., Seabra A.B. Review of cellulose nanocrystals patents: Preparation, composites and general applications. Recent. Pat. Nanotech. 2012;6:16–28. doi: 10.2174/187221012798109255. [DOI] [PubMed] [Google Scholar]
- 67.Sheikhi A., Hayashi J., Eichenbaum J., Gutin M., Kuntjoro N., Khorsandi D., Khademhosseini A. Recent advances in nanoengineering cellulose for cargo delivery—ScienceDirect. J. Control. Release. 2019;294:53–76. doi: 10.1016/j.jconrel.2018.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Al-Lohedan H.A., Arfin T., Mohammad F. Sustained drug release and electrochemical performance of ethyl cellulose-magnesium hydrogen phosphate composite. Mater. Sci. Eng. C Mater. Biol. Appl. 2017;71:735–743. doi: 10.1016/j.msec.2016.10.062. [DOI] [PubMed] [Google Scholar]
- 69.Xie J., Wang C., Ning Q., Gao Q., Gao C., Gou Z., Ye J. A new strategy to sustained release of ocular drugs by one-step drug-loaded microcapsule manufacturing in hydrogel punctal plugs. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017;255:2173–2184. doi: 10.1007/s00417-017-3755-1. [DOI] [PubMed] [Google Scholar]
- 70.Mondal S. Preparation, properties and applications of nanocellulosic materials. Carbohyd. Polym. 2017;163:301–316. doi: 10.1016/j.carbpol.2016.12.050. [DOI] [PubMed] [Google Scholar]
- 71.Tortorella S., Buratti V.V., Maturi M., Sambri L., Locatelli E. Surface-modified nanocellulose for application in biomedical engineering and nanomedicine: A review. Int. J. Nanomed. 2020;15:9909–9937. doi: 10.2147/IJN.S266103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ullah H., Badshah M., Correia A., Wahid F., Santos H.A., Khan T. Functionalized bacterial cellulose microparticles for drug delivery in biomedical applications. Curr. Pharm. Des. 2019;25:3692–3701. doi: 10.2174/1381612825666191011103851. [DOI] [PubMed] [Google Scholar]
- 73.Lin N., Huang J., Chang P.R., Feng L., Yu J. Effect of polysaccharide nanocrystals on structure, properties, and drug release kinetics of alginate-based microspheres. Colloids Surf. B Biointerfaces. 2011;85:270–279. doi: 10.1016/j.colsurfb.2011.02.039. [DOI] [PubMed] [Google Scholar]
- 74.Kim J.K., Kim H.J., Chung J.-Y., Lee J.H., Young S.B., Kim Y.-H. Natural and synthetic biomaterials for controlled drug delivery. Arch. Pharm. Res. 2013;37:60–68. doi: 10.1007/s12272-013-0280-6. [DOI] [PubMed] [Google Scholar]
- 75.Ulbrich K., Holá K., Šubr V., Bakandritsos A., Tuček J., Zbořil R. Targeted drug delivery with polymers and magnetic nanoparticles: Covalent and noncovalent approaches, release control, and clinical studies. Chem. Rev. 2016;116:5338–5431. doi: 10.1021/acs.chemrev.5b00589. [DOI] [PubMed] [Google Scholar]
- 76.Zhang K., Hujaya S.D., Jarvinen T. Interfacial nanoparticle complexation of oppositely charged nanocelluloses into functional filaments with conductive, drug release or antimicrobial property. ACS Appl. Mater. Interfaces. 2020;12:1765–1774. doi: 10.1021/acsami.9b15555. [DOI] [PubMed] [Google Scholar]
- 77.Low L.E., Tan L.T., Goh B.H., Tey B.T., Ong B.H., Tang S.Y. Magnetic cellulose nanocrystal stabilized Pickering emulsions for enhanced bioactive release and human colon cancer therapy. Int. J. Biol. Macromol. 2019;127:76–84. doi: 10.1016/j.ijbiomac.2019.01.037. [DOI] [PubMed] [Google Scholar]
- 78.Tan H.F., Ooi B.S., Leo C.P. Future perspectives of nanocellulose-based membrane for water treatment. J. Water Process Eng. 2020;37:101502. doi: 10.1016/j.jwpe.2020.101502. [DOI] [Google Scholar]
- 79.Anton-Sales I., D’Antin J.C., Fernández-Engroba J., Charoenrook V., Laromaine A., Roig A., Ralph M. Bacterial nanocellulose as a corneal bandage material: A comparison with amniotic membrane. Biomater. Sci. 2020;8:2921–2939. doi: 10.1039/D0BM00083C. [DOI] [PubMed] [Google Scholar]
- 80.Harris D., Robinson J.R. Drug delivery via the mucous membranes of the oral cavity. J. Pharm. Sci. 2010;81:1–10. doi: 10.1002/jps.2600810102. [DOI] [PubMed] [Google Scholar]
- 81.Corbo D.C., Liu J.C., Chien Y.W. Drug Absorption through mucosal membranes: Effect of mucosal route and penetrant hydrophilicity. Pharm. Res. 1989;6:848–852. doi: 10.1023/A:1015952320372. [DOI] [PubMed] [Google Scholar]
- 82.Jeong W.Y., Kwon M., Choi H.E., Kim K.S. Recent advances in transdermal drug delivery systems: A review. Biomater. Res. 2021;25:1–24. doi: 10.1186/s40824-021-00226-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Mohanta V., Madras G., Patil S. Layer-by-layer assembled thin films and microcapsules of nanocrystalline cellulose for hydrophobic drug delivery. ACS Appl. Mater. Interfaces. 2014;6:20093–20101. doi: 10.1021/am505681e. [DOI] [PubMed] [Google Scholar]
- 84.Saidi L., Vilela C., Oliveira H., Silvestre A.J.D., Freire C.S.R. Poly(N-methacryloyl glycine)/nanocellulose composites as pH-sensitive systems for controlled release of diclofenac. Carbohydr. Polym. 2017;169:357–365. doi: 10.1016/j.carbpol.2017.04.030. [DOI] [PubMed] [Google Scholar]
- 85.Poonguzhali R., Khaleel Basha S., Sugantha Kumari V. Synthesis of alginate/nanocellulose bionanocomposite for in vitro delivery of ampicillin. Polym. Bull. 2017;75:4165–4173. doi: 10.1007/s00289-017-2253-2. [DOI] [Google Scholar]
- 86.Md Abu T., Zahan K.A., Rajaie M.A., Leong C.R., Ab Rashid S., Hamin N.S.M.H., Tan W.N., Tong W.Y. Nanocellulose as drug delivery system for honey as antimicrobial wound dressing. Mater. Today Proc. 2020;31:14–17. doi: 10.1016/j.matpr.2020.01.076. [DOI] [Google Scholar]
- 87.Ajdary R., Huan S., Ezazi N.Z., Xiang W., Rojas O.J. Acetylated nanocellulose for single-component bioinks and cell proliferation on 3D-printed scaffolds. Biomacromolecules. 2019;20:2770–2778. doi: 10.1021/acs.biomac.9b00527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lobmann K., Wohlert J., Mullertz A., Wagberg L., Svagan A.J. Cellulose nanopaper and nanofoam for patient-tailored drug delivery. Adv. Mater. Interfaces. 2017;4:1600655. doi: 10.1002/admi.201600655. [DOI] [Google Scholar]
- 89.Zhang X., Elsayed I., Navarathna C., Schueneman G.T., Hassan E.B. Biohybrid hydrogel and aerogel from self-assembled nanocellulose and nanochitin as a high-efficiency adsorbent for water purification. ACS Appl. Mater. Interfaces. 2019;11:46714–46725. doi: 10.1021/acsami.9b15139. [DOI] [PubMed] [Google Scholar]
- 90.Jiang F., Hsieh Y.L. Cellulose nanofibril aerogels: Synergistic improvement of hydrophobicity, strength, and thermal stability via cross-linking with diisocyanate. ACS Appl. Mater. Interfaces. 2017;9:2825. doi: 10.1021/acsami.6b13577. [DOI] [PubMed] [Google Scholar]
- 91.Chandel A.K., Kumar C.U., Jewrajka S.K. Effect of polyethylene glycol on properties and drug encapsulation-release performance of biodegradable/cytocompatible agarose-polyethylene glycol-polycaprolactone amphiphilic co-network gels. ACS Appl. Mater. Interfaces. 2016;10:3182–3192. doi: 10.1021/acsami.5b10675. [DOI] [PubMed] [Google Scholar]
- 92.Bera A., Singh A.K., Uday Kumar C., Jewrajka S.K. Degradable/cytocompatible and pH responsive amphiphilic conetwork gels based on agarose-graft copolymers and polycaprolactone. J. Mater. Chem. B. 2015;21:8548–8557. doi: 10.1039/C5TB01251A. [DOI] [PubMed] [Google Scholar]
- 93.Singh A.K., Anupam B., Bhingaradiya N., Jewrajka S.K. Reactive compatibilizer mediated precise synthesis and application of stimuli responsive polysaccharides-polycaprola-ctone amphiphilic co-network gels. Polymer. 2016;99:470–479. doi: 10.1016/j.polymer.2016.07.033. [DOI] [Google Scholar]
- 94.Hu W., Wang Z., Xiao Y., Zhang S., Wang J. Advances in crosslinking strategies of biomedical hydrogels. Biomater. Sci. 2019;7:843–855. doi: 10.1039/C8BM01246F. [DOI] [PubMed] [Google Scholar]
- 95.Nicu R., Ciolacu F., Ciolacu D.E. Advanced functional materials based on nanocellulose for pharmaceutical/medical applications. Pharmaceutics. 2021;13:1125. doi: 10.3390/pharmaceutics13081125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Hennink W.E., van Nostrum C.F. Novel crosslinking methods to design hydrogels. Adv. Drug Deliv. Rev. 2012;64:223–236. doi: 10.1016/j.addr.2012.09.009. [DOI] [PubMed] [Google Scholar]
- 97.Pellá M.C.G., Lima-Tenório M.K., Tenório-Neto E.T., Guilherme M.R., Muniz E.C., Rubira A.F. Chitosan-based hydrogels: From preparation to biomedical applications. Carbohydr. Polym. 2018;196:233–245. doi: 10.1016/j.carbpol.2018.05.033. [DOI] [PubMed] [Google Scholar]
- 98.Curvello R., Raghuwanshi V.S., Garnier G. Engineering nanocellulose hydrogels for biomedical applications. Adv. Colloid Interface Sci. 2019;267:47–61. doi: 10.1016/j.cis.2019.03.002. [DOI] [PubMed] [Google Scholar]
- 99.Xu T., Du H., Liu H., Zhang X., Si C. Advanced nanocellulose-based composites for flexible functional energy storage devices. Adv. Mater. 2021;33:2101368. doi: 10.1002/adma.202101368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Li Z., Chen Z., Chen H., Chen K., Tao W., Ouyang X.K., Mei L., Zeng X. Polyphenol-based hydrogels: Pyramid evolution from crosslinked structures to biomedical applications and the reverse design. Bioact. Mater. 2022;17:49–70. doi: 10.1016/j.bioactmat.2022.01.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Mantha S., Pillai S., Khayambashi P., Upadhyay A., Tran S.D. Smart hydrogels in tissue engineering and regenerative medicine. Materials. 2019;12:3323. doi: 10.3390/ma12203323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hu S., Zhi Y., Shan S., Ni Y. Research progress of smart response composite hydrogels based on nanocellulose. Carbohydr. Polym. 2022;275:118741. doi: 10.1016/j.carbpol.2021.118741. [DOI] [PubMed] [Google Scholar]
- 103.Treesuppharat W., Rojanapanthu P., Siangsanoh C., Manuspiya H., Ummartyotin S. Synthesis and characterization of bacterial cellulose and gelatin-based hydrogel composites for drug-delivery systems. Biotechnol. Rep. 2017;15:84–91. doi: 10.1016/j.btre.2017.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Muller A., Ni Z., Hessler N., Wesarg F., Muller F.A., Kralisch D., Fischer D. The biopolymer bacterial nanocellulose as drug delivery system: Investigation of drug loading and release using the model protein albumin. J. Pharm. Sci. 2013;102:579–592. doi: 10.1002/jps.23385. [DOI] [PubMed] [Google Scholar]
- 105.Mohd Amin M.C.I., Ahmad N., Halib N., Ahmad I. Synthesis and characterization of thermo- and pH-responsive bacterial cellulose/acrylic acid hydrogels for drug delivery. Carbohydr. Polym. 2012;88:465–473. doi: 10.1016/j.carbpol.2011.12.022. [DOI] [Google Scholar]
- 106.Liu Y., Fan Q., Huo Y., Liu C., Li B., Li Y. Construction of a mesoporous polydopamine@GO/cellulose nanofibril composite hydrogel with an encapsulation structure for controllable drug release and toxicity shielding. ACS Appl. Mater. Interfaces. 2020;12:57410–57420. doi: 10.1021/acsami.0c15465. [DOI] [PubMed] [Google Scholar]
- 107.Chandel A.K.S., Kannan D., Nutan B., Singh S., Jewrajka S.K. Dually crosslinked injectable hydrogels of poly (ethylene glycol) and poly [(2-dimethylamino) ethyl methacrylate]-b-poly (N-isopropyl acrylamide) as a wound healing promoter. J. Mater. Chem. B. 2017;5:4955–4965. doi: 10.1039/C7TB00848A. [DOI] [PubMed] [Google Scholar]
- 108.Chandel A.K.S., Nutan B., Raval I.H., Jewrajka S.K. Self-assembly of partially alkylated dextran-graft-poly [(2-dimethylamino) ethyl methacrylate] copolymer facilitating hydrophobic/hydrophilic drug delivery and improving conetwork hydrogel properties. Biomacromolecules. 2018;19:1142–1153. doi: 10.1021/acs.biomac.8b00015. [DOI] [PubMed] [Google Scholar]
- 109.Nutan B., Chandel A.K.S., Jewrajka S.K. Liquid prepolymer-based in situ formation of degradable poly (ethylene glycol)-linked-poly (caprolactone)-linked-poly (2-dimethylaminoethyl) methacrylate amphiphilic conetwork gels showing polarity driven gelation and bioadhesion. ACS Appl. Bio Mater. 2018;1:1606–1619. doi: 10.1021/acsabm.8b00461. [DOI] [PubMed] [Google Scholar]
- 110.Nutan B., Chandel A.K.S., Bhalani D.V., Jewrajka S.K. Synthesis and tailoring the degradation of multi-responsive amphiphilic conetwork gels and hydrogels of poly (β-amino ester) and poly (amido amine) Polymer. 2017;111:265–274. doi: 10.1016/j.polymer.2017.01.057. [DOI] [Google Scholar]
- 111.Bertsch P., Schneider L., Bovone G. Injectable biocompatible hydrogels from cellulose nanocrystals for locally targeted sustained drug release. ACS Appl. Mater. Interfaces. 2019;2019:38578–38585. doi: 10.1021/acsami.9b15896. [DOI] [PubMed] [Google Scholar]
- 112.Orasugh J.T., Sarkar G., Saha N.R., Das B., Bhattacharyya A., Das S., Mishra R., Roy I., Chattoapadhyay A., Ghosh S.K. Effect of cellulose nanocrystals on the performance of drug loaded in situ gelling thermo-responsive ophthalmic formulations. Int. J. Biol. Macromol. 2018;124:235–245. doi: 10.1016/j.ijbiomac.2018.11.217. [DOI] [PubMed] [Google Scholar]
- 113.Yu L., Ding J. Injectable hydrogels as unique biomedical materials. Chem. Soc. Rev. 2008;37:1473–1481. doi: 10.1039/b713009k. [DOI] [PubMed] [Google Scholar]
- 114.Lin F., Zheng J., Guo W., Zhu Z., Lu B. Smart cellulose-derived magnetic hydrogel with rapid swelling and deswelling properties for remotely controlled drug release. Cellulose. 2019;26:6861–6877. doi: 10.1007/s10570-019-02572-0. [DOI] [Google Scholar]
- 115.Cunha A.G., Mougel J.B., Cathala B., Berglund L.A., Capron I. Preparation of double Pickering emulsions stabilized by chemically tailored nanocelluloses. Langmuir. 2014;30:9327–9335. doi: 10.1021/la5017577. [DOI] [PubMed] [Google Scholar]
- 116.Pooresmaeil M., Namazi H. Fabrication of a smart and biocompatible brush copolymer decorated on magnetic graphene oxide hybrid nanostructure for drug delivery application. Eur. Polym. J. 2020;142:110126. doi: 10.1016/j.eurpolymj.2020.110126. [DOI] [Google Scholar]
- 117.Supramaniam J., Adnan R., Mohd Kaus N.H., Bushra R. Magnetic nanocellulose alginate hydrogel beads as potential drug delivery system. Int. J. Biol. Macromol. 2018;118:640–648. doi: 10.1016/j.ijbiomac.2018.06.043. [DOI] [PubMed] [Google Scholar]
- 118.Khalil H., Adnan A.S., Yahya E.B., Olaiya N.G., Pasquini D. A review on plant cellulose nanofibre-based aerogels for biomedical applications. Polymers. 2020;12:1759. doi: 10.3390/polym12081759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Horcajada P., Chalati T., Serre C., Gillet B., Sebrie C., Baati T., Eubank J.F., Heurtaux D., Clayette P., Kreuz C. Porous metal–organic-framework nanoscale carriers as a potential platform for drug delivery and imaging. Nat. Mater. 2010;9:172–178. doi: 10.1038/nmat2608. [DOI] [PubMed] [Google Scholar]
- 120.Zhao S., Malfait W.J., Guerrero-Alburquerque N., Koebel M.M., Nyström G. Biopolymer aerogels and foams: Chemistry, properties, and applications. Angew. Chem. 2018;57:7580–7608. doi: 10.1002/anie.201709014. [DOI] [PubMed] [Google Scholar]
- 121.Rudaz C., Courson R., Bonnet L., Calas-Etienne S., Budtova T. Aeropectin: Fully biomass-based mechanically strong and thermal superinsulating aerogel. Biomacromolecules. 2014;15:2188–2195. doi: 10.1021/bm500345u. [DOI] [PubMed] [Google Scholar]
- 122.Preibisch I., Niemeyer P., Yusufoglu Y., Gurikov P., Smirnova I. Polysaccharide-based aerogel bead production via jet cutting method. Materials. 2018;11:1287. doi: 10.3390/ma11081287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Nishiguchi A., Taguchi T. A Thixotropic, cell-infiltrative nanocellulose hydrogel that promotes in vivo tissue remodeling. ACS Biomater. Sci. Eng. 2020;6:946–958. doi: 10.1021/acsbiomaterials.9b01549. [DOI] [PubMed] [Google Scholar]
- 124.Barrios E., Fox D., Sip Y., Catarata R., Calderon J.E., Azim N., Afrin S., Zhang Z., Zhai L. Nanomaterials in advanced, high-performance aerogel composites: A review. Polymers. 2019;11:726. doi: 10.3390/polym11040726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Seera S., Kundu D., Banerjee T. Physical and chemical crosslinked microcrystalline cellulose-polyvinyl alcohol hydrogel: Freeze–thaw mediated synthesis, characterization and in vitro delivery of 5-fluorouracil. Cellulose. 2020;27:6521–6535. doi: 10.1007/s10570-020-03249-9. [DOI] [Google Scholar]
- 126.Nascimento A.M., Nunes Y.L., Fifuriredo M.C.B., Dufresne A. Nanocellulose nanocomposite hydrogels: Technological and environmental issues. Green Chem. 2018;20:2428–2448. doi: 10.1039/C8GC00205C. [DOI] [Google Scholar]
- 127.Shen X., Shamshina J.L., Berton P., Gurau G., Rogers R.D. Hydrogels based on cellulose and chitin: Fabrication, properties, and applications. Green Chem. 2015;18:53–75. doi: 10.1039/C5GC02396C. [DOI] [Google Scholar]
- 128.Chen X., Xu X., Li W., Sun B., Yan J., Chen C., Liu J., Qian J., Sun D. Effective drug carrier based on polyethylenimine-functionalized bacterial cellulose with controllable release properties. ACS Appl. Bio Mater. 2018;1:42–50. doi: 10.1021/acsabm.8b00004. [DOI] [Google Scholar]
- 129.Zhu Y., Zeng Q., Zhang Q., Li K., Shi X., Liang F., Han D. Temperature/near-infrared light-responsive conductive hydrogels for controlled drug release and real-time monitoring. Nanoscale. 2020;12:8679–8686. doi: 10.1039/D0NR01736A. [DOI] [PubMed] [Google Scholar]
- 130.Zhao J., Lu C., He X., Zhang X., Zhang W., Zhang X. Polyethylenimine-grafted cellulose nanofibril aerogels as versatile vehicles for drug delivery. ACS Appl. Mater. Interfaces. 2015;7:2607–2615. doi: 10.1021/am507601m. [DOI] [PubMed] [Google Scholar]
- 131.Liu Z., Zhang S., He B., Wang S., Kong F. Synthesis of cellulose aerogels as promising carriers for drug delivery: A review. Cellulose. 2021;28:2697–2714. doi: 10.1007/s10570-021-03734-9. [DOI] [Google Scholar]
- 132.Dickhoff B., Boer A., Lambregts D., Frijlink H.W. The effect of carrier surface and bulk properties on drug particle detachment from crystalline lactose carrier particles during inhalation, as function of carrier payload and mixing time. Eur. J. Pharm. Biopharm. 2003;56:291–302. doi: 10.1016/S0939-6411(03)00109-7. [DOI] [PubMed] [Google Scholar]
- 133.Corsaro C., Mallamace D., Neri G., Fazio E. Hydrophilicity and hydrophobicity: Key aspects for biomedical and technological purposes. Phys. A. 2021;580:126189. doi: 10.1016/j.physa.2021.126189. [DOI] [Google Scholar]
- 134.Kalepu S., Nekkanti V. Insoluble drug delivery strategies: Review of recent advances and business prospects. Acta Pharm. Sin. B. 2015;12:442–453. doi: 10.1016/j.apsb.2015.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Venkatalaxmi A., Padmavathi B.S., Amaranath T. A general solution of unsteady Stokes equations. Fluid Dyn. Res. 2004;35:229–236. doi: 10.1016/j.fluiddyn.2004.06.001. [DOI] [Google Scholar]
- 136.Khojastehfar A., Mahjoub S. Application of nanocellulose derivatives as drug carriers; a novel approach in drug delivery. Anti-Cancer Agents Med. Chem. 2021;11:692–702. doi: 10.2174/1871520620666200811111547. [DOI] [PubMed] [Google Scholar]
- 137.Eyley S., Thielemans W. Surface modification of cellulose nanocrystals. Nanoscale. 2014;6:7764–7779. doi: 10.1039/C4NR01756K. [DOI] [PubMed] [Google Scholar]
- 138.Eloy J.O., Marina C., Petrilli R., Barcellos J., Lee R.J., Marchetti J.M. Liposomes as carriers of hydrophilic small molecule drugs: Strategies to enhance encapsulation and delivery. Colloids Surf. B Biointerfaces. 2014;123:345–363. doi: 10.1016/j.colsurfb.2014.09.029. [DOI] [PubMed] [Google Scholar]
- 139.Alex B., Maria S., Natalia F. Towards tunable protein-carrier wound dressings based on nanocellulose hydrogels crosslinked with calcium ions. Nanomaterials. 2018;8:550. doi: 10.3390/nano8070550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Xiang C., Taylor A.G., Hinestroza J.P., Frey M.W. Controlled release of nonionic compounds from poly(lactic acid)/cellulose nanocrystal nanocomposite fibers. J. Appl. Polym. Sci. 2013;127:79–86. doi: 10.1002/app.36943. [DOI] [Google Scholar]
- 141.Wang Y., Chen L. Cellulose nanowhiskers and fiber alignment greatly improve mechanical properties of electrospun prolamin protein fibers. ACS Appl. Mater. Interfaces. 2014;6:1709–1718. doi: 10.1021/am404624z. [DOI] [PubMed] [Google Scholar]
- 142.Zhang F., Wu W., Zhang X., Meng X., Tong G., Deng Y. Temperature-sensitive poly-NIPAm modified cellulose nanofibril cryogel microspheres for controlled drug release. Cellulose. 2015;23:415–425. doi: 10.1007/s10570-015-0799-4. [DOI] [Google Scholar]
- 143.Akhlaghi S.P., Berry R.C., Tam K.C. Surface modification of cellulose nanocrystal with chitosan oligosaccharide for drug delivery applications. Cellulose. 2013;20:1747–1764. doi: 10.1007/s10570-013-9954-y. [DOI] [Google Scholar]
- 144.Zambuzi G.C., Camargos C.H.M., Ferreira M.P., Rezende C.A., de Freitas O., Francisco K.R. Modulating the controlled release of hydroxychloroquine mobilized on pectin films through film-forming pH and incorporation of nanocellulose. Carbohydr. Polym. Technol. Appl. 2021;2:100140. doi: 10.1016/j.carpta.2021.100140. [DOI] [Google Scholar]
- 145.O’Donnell K.L., Oporto-Velasquez G.S., Comolli N. Evaluation of acetaminophen release from biodegradable poly (vinyl alcohol) (PVA) and nanocellulose films using a multiphase release mechanism. Nanomaterials. 2020;10:301. doi: 10.3390/nano10020301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Orasugh J.T., Saha N.R., Rana D., Sarkar G., Mollick M.M.R., Chattoapadhyay A., Mitra B.C., Mondal D., Ghosh S.K., Chattopadhyay D. Jute cellulose nano-fibrils/hydroxypropylmethylcellulose nanocomposite: A novel material with potential for application in packaging and transdermal drug delivery system. Ind. Crops Prod. 2018;112:633–643. doi: 10.1016/j.indcrop.2017.12.069. [DOI] [Google Scholar]
- 147.Park D., Kim J.W., Shin K., Kim J.W. Bacterial cellulose nanofibrils-reinforced composite hydrogels for mechanical compression-responsive on-demand drug release. Carbohydr. Polym. 2021;272:118459. doi: 10.1016/j.carbpol.2021.118459. [DOI] [PubMed] [Google Scholar]
- 148.Liu Y., Sui Y., Liu C., Liu C., Wu M., Li B., Li Y. A physically crosslinked polydopamine/nanocellulose hydrogel as potential versatile vehicles for drug delivery and wound healing. Carbohydr. Polym. 2018;188:27–36. doi: 10.1016/j.carbpol.2018.01.093. [DOI] [PubMed] [Google Scholar]
- 149.Bhandari J., Mishra H., Mishra P.K., Wimmer R., Ahmad F.J., Talegaonkar S. Cellulose nanofiber aerogel as a promising biomaterial for customized oral drug delivery. Int. J. Nanomed. 2017;12:2021–2031. doi: 10.2147/IJN.S124318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Liang Y., Zhu H., Wang L., He H., Wang S. Biocompatible smart cellulose nanofibres for sustained drug release via pH and temperature dual-responsive mechanism. Carbohydr. Polym. 2020;249:116876. doi: 10.1016/j.carbpol.2020.116876. [DOI] [PubMed] [Google Scholar]
- 151.Shao W., Liu H., Wang S., Wu J., Huang M., Min H., Liu X. Controlled release and antibacterial activity of tetracycline hydrochloride-loaded bacterial cellulose composite membranes. Carbohydr. Polym. 2016;145:114–120. doi: 10.1016/j.carbpol.2016.02.065. [DOI] [PubMed] [Google Scholar]
- 152.Ohta S., Mitsuhashi K., Chandel A.K.S., Qi P., Nakamura N., Nakamichi A., Yoshida H., Yamaguchi G., Hara Y., Sasaki R. Silver-loaded carboxymethyl cellulose nonwoven sheet with controlled counterions for infected wound healing. Carbohydr. Polym. 2022;286:119289. doi: 10.1016/j.carbpol.2022.119289. [DOI] [PubMed] [Google Scholar]
- 153.Liu Y., Huo Y., Fan Q., Li M., Liu H., Li B., Li Y. Cellulose nanofibrils composite hydrogel with polydopamine@zeolitic imidazolate framework-8 encapsulated in used as efficient vehicles for controlled drug release. J. Ind. Eng. Chem. 2021;102:343–350. doi: 10.1016/j.jiec.2021.07.023. [DOI] [Google Scholar]
- 154.Li J., Wang Y., Zhang L., Xu Z., Dai H., Wu W. Nanocellulose/gelatin composite cryogels for controlled drug release. ACS Sustain. Chem. Eng. 2019;7:6381–6389. doi: 10.1021/acssuschemeng.9b00161. [DOI] [Google Scholar]
- 155.Cacicedo M.L., Islan G.A., Leon I.E., Alvarez V.A., Chourpa I., Allard-Vannier E., Garcia-Aranda N., Diaz-Riascos Z.V., Fernandez Y., Schwartz S., Jr., et al. Bacterial cellulose hydrogel loaded with lipid nanoparticles for localized cancer treatment. Colloids Surf. B Biointerfaces. 2018;170:596–608. doi: 10.1016/j.colsurfb.2018.06.056. [DOI] [PubMed] [Google Scholar]
- 156.Wischke C., Schwendeman S.P. Principles of encapsulating hydrophobic drugs in PLA/PLGA microparticles. Int. J. Pharm. 2008;364:298–327. doi: 10.1016/j.ijpharm.2008.04.042. [DOI] [PubMed] [Google Scholar]
- 157.Douroumis D., Fahr A. Drug Delivery Strategies for Poorly Water-Soluble Drugs. John Wiley & Sons; Hoboken, NJ, USA: 2013. pp. 265–286. [DOI] [Google Scholar]
- 158.Larraneta E., Stewart S., Ervine M., Al-Kasasbeh R., Donnelly R.F. Hydrogels for hydrophobic drug delivery. Classification, synthesis and applications. J. Funct. Biomater. 2018;9:13. doi: 10.3390/jfb9010013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Jackson J.K., Letchford K., Wasserman B.Z., Ye L., Hamad W.Y., Burt H.M. The use of nanocrystalline cellulose for the binding and controlled release of drugs. Int. J. Nanomed. 2011;6:321–330. doi: 10.2147/IJN.S16749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Bundjaja V., Sari T.M., Soetaredjo F.E., Yuliana M., Angkawijaya A.E., Ismadji S., Cheng K.C., Santoso S.P. Aqueous sorption of tetracycline using rarasaponin-modified nanocrystalline cellulose. J. Mol. Liq. 2020;301:112433. doi: 10.1016/j.molliq.2019.112433. [DOI] [Google Scholar]
- 161.Carvalho J.P.F., Silva A.C.Q., Bastos V., Oliveira H., Pinto R.J.B., Silvestre A.J.D., Vilela C., Freire C.S.R. Nanocellulose-based patches loaded with hyaluronic acid and diclofenac towards aphthous stomatitis treatment. Nanomaterials. 2020;10:628. doi: 10.3390/nano10040628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Ajdary R., Ezazi N.Z., Correia A., Kemell M., Huan S., Ruskoaho H.J., Hirvonen J., Santos H.A., Rojas O.J. Multifunctional 3D-printed patches for long-term drug release therapies after myocardial infarction. Adv. Funct. Mater. 2020;30:2003440. doi: 10.1002/adfm.202003440. [DOI] [Google Scholar]
- 163.Stroescu M., Stoica-Guzun A., Jipa I.M. Vanillin release from poly(vinyl alcohol)-bacterial cellulose mono and multilayer films. J. Food Eng. 2013;114:153–157. doi: 10.1016/j.jfoodeng.2012.08.023. [DOI] [Google Scholar]
- 164.Shi X., Zheng Y., Wang G., Lin Q., Fan J. pH- and electro-response characteristics of bacterial cellulose nanofiber/sodium alginate hybrid hydrogels for dual controlled drug delivery. RSC Adv. 2014;4:47056–47065. doi: 10.1039/C4RA09640A. [DOI] [Google Scholar]
- 165.Liu Y., Huo Y., Li M., Qin C., Liu H. Synthesis of metal–organic-frameworks on polydopamine modified cellulose nanofibril hydrogels: Constructing versatile vehicles for hydrophobic drug delivery. Cellulose. 2022;29:379–393. doi: 10.1007/s10570-021-04267-x. [DOI] [Google Scholar]
- 166.Wang Y., Khan A., Liu Y., Feng J., Ni Y. Chitosan oligosaccharide-based dual pH responsive nano-micelles for targeted delivery of hydrophobic drugs. Carbohydr. Polym. 2019;223:115061–115071. doi: 10.1016/j.carbpol.2019.115061. [DOI] [PubMed] [Google Scholar]
- 167.Qianyun M., Lele C., Tieqiang L. Active tara gum/PVA blend films with curcumin-loaded CTAC brush-TEMPO-oxidized cellulose nanocrystals. ACS Sustain. Chem. Eng. 2018;6:8926–8934. doi: 10.1021/acssuschemeng.8b01281. [DOI] [Google Scholar]
- 168.Plappert S.F., Liebner F.W., Konnerth J., Nedelec J.M. Anisotropic nanocellulose gel-membranes for drug delivery: Tailoring structure and interface by sequential periodate-chlorite oxidation. Carbohydr. Polym. 2019;226:115306. doi: 10.1016/j.carbpol.2019.115306. [DOI] [PubMed] [Google Scholar]
- 169.Garg T., Rath G., Goyal A.K. Colloidal drug delivery systems: Current status and future directions. Crit. Rev. Ther. Drug Carr. Syst. 2015;32:89. doi: 10.1615/CritRevTherDrugCarrierSyst.2015010159. [DOI] [PubMed] [Google Scholar]
- 170.Anirudhan T.S., Manjusha V., Chithra Sekhar V. A new biodegradable nano cellulose-based drug delivery system for pH-controlled delivery of curcumin. Int. J. Biol. Macromol. 2021;183:2044–2054. doi: 10.1016/j.ijbiomac.2021.06.010. [DOI] [PubMed] [Google Scholar]
- 171.Anirudhan T.S., Sekhar V.C., Athira V.S. Graphene oxide based functionalized chitosan polyelectrolyte nanocomposite for targeted and pH responsive drug delivery. Int. J. Biol. Macromol. 2020;150:468–479. doi: 10.1016/j.ijbiomac.2020.02.053. [DOI] [PubMed] [Google Scholar]
- 172.Luo H., Ao H., Li G., Li W., Xiong G., Zhu Y., Wan Y. Bacterial cellulose/graphene oxide nanocomposite as a novel drug delivery system. Curr. Appl. Phys. 2017;17:249–254. doi: 10.1016/j.cap.2016.12.001. [DOI] [Google Scholar]
- 173.Lauroba J., Diez I., Rius M., Peraire C., Domenech J. Study of the release process of drugs: Suppositories of paracetamol. Int. J. Clin. Pharmacol. Ther. 1990;28:118. doi: 10.1016/0162-3109(90)90049-K. [DOI] [PubMed] [Google Scholar]
- 174.Torkkeli M., Laaksonen T., Peltonen L., Serimaa R., Valo H., Linder M.B. Drug release from nanoparticles embedded in four different nanofibrillar cellulose aerogels. Eur. J. Pharm. Biopharm. 2013;50:69–77. doi: 10.1016/j.ejps.2013.02.023. [DOI] [PubMed] [Google Scholar]
- 175.Khalil H.P.S.A., Bhat A.H., Yusra A.F.I. Green composites from sustainable cellulose nanofibrils: A review. Carbohyd polym. 2012;87:963–979. doi: 10.1016/j.carbpol.2011.08.078. [DOI] [Google Scholar]
- 176.Kolakovic R., Peltonen L., Laukkanen A., Hirvonen J., Laaksonen T. Nanofibrillar cellulose films for controlled drug delivery. Eur. J. Pharm. Biopharm. 2012;82:308–315. doi: 10.1016/j.ejpb.2012.06.011. [DOI] [PubMed] [Google Scholar]
- 177.Manga R.D., Jha P.K. Mathematical models for controlled drug release through pH-responsive polymeric hydrogels. J. Pharm. Sci. 2017;106:629–638. doi: 10.1016/j.xphs.2016.10.019. [DOI] [PubMed] [Google Scholar]
- 178.Pontrelli G., Toniolo G., Mcginty S., Peri D., Chatgilialoglu C. Mathematical modelling of drug delivery from pH-responsive nanocontainers. Comput. Biol. Med. 2021;131:104238. doi: 10.1016/j.compbiomed.2021.104238. [DOI] [PubMed] [Google Scholar]
- 179.Baishya H. Application of mathematical models in drug release kinetics of carbidopa and levodopa ER tablets. J. Dev. Drugs. 2017;6:2. doi: 10.4172/2329-6631.1000171. [DOI] [Google Scholar]
- 180.Mohammadi A., Moghaddas J. Mesoporous tablet-shaped potato starch aerogels for loading and release of the poorly water-soluble drug celecoxib. Chin. J. Chem. Eng. 2020;28:1778–1787. doi: 10.1016/j.cjche.2020.03.040. [DOI] [Google Scholar]
- 181.Ayazi H., Akhavan O., Raoufi M., Varshochian R., Hosseini Motlagh N.S., Atyabi F. Graphene aerogel nanoparticles for in-situ loading/pH sensitive releasing anticancer drugs. Colloids Surf. B Biointerfaces. 2020;186:110712. doi: 10.1016/j.colsurfb.2019.110712. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.