Abstract
Purpose
This large population-based study is the first to analyze the association between coronary artery disease (CAD) and osteoporosis (OP) from the National Health Insurance Research Database (NHIRD) in Taiwan to determine if CAD is associated with OP.
Methods
Data from NHIRD, a national, population-based, retrospective, matched cohort study of 23 million patients, were collected to recruit two matched cohorts: with (n = 192,367) and without (n = 192,367) CAD. The Cox model was used to compare the incidence rate ratio and crude hazard ratio (HR) between the two cohorts for osteoporotic fracture and OP.
Results
The CAD cohort had a significantly increased risk for vertebral compression fracture, with an adjusted HR of 1.74 (95% CI, 1.60–1.89). The cumulative incidence of OP was also statistically higher in the cohort versus without CAD (11.6% vs. 5.6%; p ≤ 0.0001, log-rank) during the 10-year follow-up period. The Cox model showed a 2.04-fold increase in the incidence of OP in the CAD cohort, with an adjusted HR of 2.04 (95% confidence interval [CI], 1.99–2.08).
Conclusions
A positive association exists between CAD and development of subsequent osteoporotic fracture and OP. Patients with CAD have a significantly increased risk of developing vertebral compression fracture and a higher incident rate ratio of OP.
Trial registration
IRB approval number: No. C108094 on February 19, 2020.
Keywords: Coronary artery disease, Osteoporosis, National Health Insurance Research Database, Taiwan
Introduction
Coronary artery disease (CAD) and osteoporosis (OP) are major causes of mortality and morbidity in the older adult population [1–3]. The prevalence of both diseases is expected to rise due to increased life expectancies [4–7]. By 2050, the world’s population age 60 years and older is predicted to increase almost double from 13 to 25%, according to the World Health Organization [8]. Because OP may progress without symptoms and is associated with age-related fractures, it is now recognized as a major threat, and Taiwan policymaking to address the prevalence of OP reached a critical point in 2005 [9]. According to the Taiwan Ministry of Health and Welfare in 2015, the prevalence of OP was 17.4% in 2001 and 25% in 2011 [9], and CAD ranked the second highest cause of death among women in Taiwan [10]. Both diseases can independently progress to more severe conditions, which will result in a large socioeconomic burden [11, 12].
Previously, CAD and OP were viewed as unrelated and independent of each other, until recent years when epidemiological and biological studies formed concepts and evidence of an association [13–15]. For CAD, a process called atherosclerosis, which describes the buildup of fats, cholesterol, and other substances within and outside the artery wall, is influenced by several risk factors, such as obesity, smoking, hypertension, and diabetes, mainly affecting women after menopause [3]. The pathophysiologic mechanism for bone loss in OP is similar in that the process may be accelerated by age. Both diseases involve an endothelium-lined lumen from the artery walls and the osteon from the cortical bone; therefore, it is possible that endothelial dysfunction may be a common precursor to OP and CAD. Previous studies also showed that noninflammatory and inflammatory factors such as elevated C-reactive protein, tumor necrosis factor-alpha (TNF-α), and interleukin (IL)-6 levels have aggravated both atherogenesis and bone resorption [16]. Another possible link seen between CAD and OP is the high concentrations of nitric oxide, which may play a role in bone metabolism through osteoblastic activity, spreading osteoclasts, and bone resorption.
A retrospective study analyzing asymptomatic postmenopausal women in Korea reported that high coronary artery calcium (CAC) scores and obstructive CAD were associated with OP [17]. A community-based study in China concluded that CAD was independently and significantly associated with OP [3]. However, the most recent meta-analysis of cross-sectional studies in 2020 supported that low bone mineral density (BMD) was not associated with the prevalence of CAD. With an increase in the correlation supporting a possible link between the two comparisons that could only be explained by age-accelerated alone, studies have suggested that no relationship exists between OP and CAD. Although advanced science has improved the understanding of pathophysiologic factors and associations shared by both diseases, these two diseases have yet to be sufficiently investigated with regard to the etiologic role of CAD associated with OP. Therefore, the purpose of this study was to examine if CAD is associated with osteoporotic fracture and OP. We hypothesized that CAD correlates with an increased incidence of OP and osteoporotic fracture.
Methods
Data source and ethical considerations
Datasets were retrieved from the National Health Insurance Research Database (NHIRD) in Taiwan, which represents > 99% of the Taiwanese population (approximately 23 million individuals), if not all [18]. By providing abundant information for inpatients and outpatients, including detailed demographic information; initiation date for health care; total medical expenses from ambulatory, prescription, and medication services; and more, the NHIRD has often been used by many researchers for hundreds of published studies [19]. The NHIRD diagnosis codes are the International Classification of Diseases, Ninth Revision; Clinical Modification (ICD-9-CM); and the Tenth Revision (ICD-10-CM) in Taiwan which was fully adopted from January 1, 2016. Following the NHIRD ethical guidelines, personal patient information was anonymized before accessing; therefore, the Research Ethics Committee waived the requirements for informed consent. This study was approved by the Institutional Review Board and Ethics Committee of Fu Jen Catholic University (IRB approval number: No. C108094).
Study population
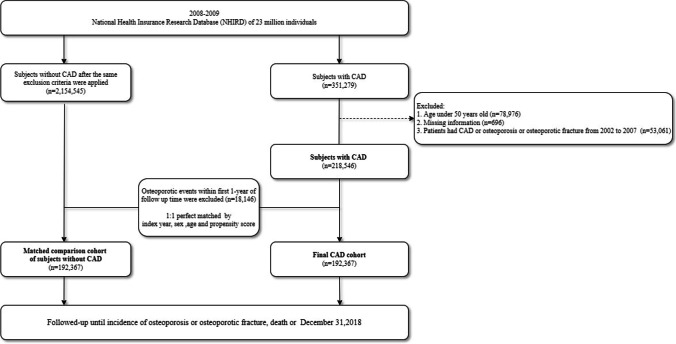
The study design is a retrospective, population-based, database cohort study. Inclusion criteria were age older than 50 years with physician-diagnosed CAD (ICD-9-CM Code 410 to 414) from January 1, 2008, to December 31, 2009. A total of 192,367 CAD cases were identified after excluding patients meeting any of these criteria: age younger than 50 years (n = 78,976); any missing data (n = 696); and CAD or OP or osteoporotic fracture (including hip fracture and vertebral compression fracture) from 2002 to 2007 (n = 53,061) (Fig. 1). For the comparison group, 192,367 patients without CAD were matched after applying the same exclusion criteria used for the CAD cohort. To minimize any allocation bias, the non-CAD cohort was verified to maintain CAD-free status in the dataset throughout the entire follow-up period. Additionally, the first CAD date was defined as the index date. Patients who were diagnosed with OP after the index date within 1 year were excluded from the study to avoid reverse causation bias. To reduce sampling bias, this study used a 1:1 ratio for propensity score matching with index year, age, and sex. After propensity score matching, the final CAD cohort contained 192,367 patients and the comparison cohort contained the same number of patients. All participants in both cohorts were followed up until the occurrence of OP or OP fracture, death, or December 31, 2018, whichever occurred first.
Fig. 1.
Consort diagram showing detailed steps for assembling the two study cohorts
Outcome measure and confounding variables
The primary outcome measure was the incidence of thoracolumbar vertebral compression fracture. The secondary outcome measure was physician-diagnosed OP, defined as at least three different medical claims issued in the outpatient setting or at least one claim in the inpatient setting. To avoid the reverse causation phenomenon (protopathic bias), patients with OP outcomes during the first year of follow-up were excluded from the overall risk calculation. In Taiwan, the gold standard for diagnosis of OP is BMD measurement in the spine and hip with a value of 2.5 or more standard deviation (SD) below the young adult female reference mean (T score − 2.5 SD or less), based on a white female normative database as the T score reference.
Table 1 shows that the two cohorts were balanced for index date, age, and sex after matching. We used categorized insurance premium as a proxy for the participants’ socioeconomic status. Comorbidity data were collected for hypertension, hyperlipidemia, chronic obstructive pulmonary disease (COPD), diabetes mellitus (DM), liver disease, peripheral vascular disease (PVD), congestive heart failure (CHF), stroke, rheumatoid arthritis (RA), and morbid obesity. The Charlson Comorbidity Index (CCI) was added as shown in Table 1.
Table 1.
Demographics and comorbidities at baseline between the CAD cohort and the age-, sex-, and index date-matched comparison cohort without CAD
| Case (N = 192,367) | Compare (N = 192,367) | p | |||
|---|---|---|---|---|---|
| n | % | n | % | ||
| Year | |||||
| 2008 | 97,267 | 50.56 | 97,564 | 50.72 | 0.3382 |
| 2009 | 95,100 | 49.44 | 94,803 | 49.28 | |
| Sex | |||||
| Female | 87,140 | 45.29 | 87,139 | 45.29 | 1 |
| Male | 105,227 | 54.71 | 105,228 | 54.71 | |
| Age | |||||
| 50–59 | 80,787 | 42 | 80,787 | 42 | 1 |
| 60–69 | 57,304 | 29.79 | 57,304 | 29.79 | |
| 70–79 | 39,173 | 20.36 | 39,173 | 20.36 | |
| 80–89 | 15,103 | 7.85 | 15,103 | 7.85 | |
| Baseline comorbidity | |||||
| Hypertension | 14,700 | 7.64 | 4633 | 2.41 | < 0.001 |
| Hyperlipidemia | 48,616 | 25.27 | 16,353 | 8.5 | < 0.001 |
| COPD | 35,698 | 18.56 | 14,669 | 7.63 | < 0.001 |
| Diabetes mellitus | 36,928 | 19.2 | 14,898 | 7.74 | < 0.001 |
| PVD | 10,668 | 5.55 | 3839 | 2.03 | < 0.001 |
| CHF | 23,026 | 11.97 | 5477 | 2.85 | < 0.001 |
| Stroke | 25,245 | 13.12 | 10,529 | 5.47 | < 0.001 |
| RA | 142 | 0.07 | 63 | 0.03 | < 0.001 |
| Morbid obesity | 106 | 0.06 | 28 | 0.01 | < 0.001 |
| Heart failure | 6354 | 3.3 | 1557 | 0.81 | < 0.001 |
| Liver cirrhosis | 2667 | 1.39 | 1938 | 1.01 | < 0.001 |
| CCI | |||||
| 0 | 110,464 | 57.42 | 159,349 | 82.84 | < 0.001 |
| 1 | 25,320 | 13.16 | 9318 | 4.84 | |
| 2 | 21,669 | 11.26 | 8439 | 4.39 | |
| 3 | 14,693 | 7.64 | 5920 | 3.08 | |
| 4 | 9041 | 4.7 | 3863 | 2.01 | |
| 5 | 5051 | 2.63 | 2220 | 1.15 | |
| 6 + | 6129 | 3.19 | 3258 | 1.69 | |
| Mean (SD) | 1.1384 (1.7817) | 0.4963 (0.4963) | < 0.001 | ||
| Mortality | 40,089 | 20.84 | 46,934 | 24.4 | < 0.001 |
SD standard deviation, CAD coronary artery disease, COPD chronic lung disease, PVD peripheral vascular disease, CHF congestive heart failure, RA rheumatic arthritis, CCI Charlson Comorbidity Index
Statistical analysis
The basic demographic characteristics were defined as categorical variables, presented as N (%), and continuous variables, presented as mean ± SD. Sex and age between the case and the control groups were compared using the chi-squared and t tests. Multivariate adjusted Cox proportional hazard regression was calculated for the adjusted hazard ratios (HRs) and 95% confidence intervals (CIs) to estimate the incidences for the case and control groups. Kaplan–Meier curve analyses were conducted to evaluate the cumulative incidences of OP; a log-rank test was used to determine the significance between cases and controls. For all analyses, a two-tailed p value of < 0.05 was considered statistically significant. All statistical analyses were conducted using SAS version 9.4 (SAS Institute, Cary, NC, USA).
Results
Table 1 shows the baseline characteristics of the study participants. A total of 192,367 patients were assessed, with a mean age of 64.26 years. Several significant differences between cohorts were found for the comorbidities of hypertension, hyperlipidemia, COPD, DM, liver disease, PVD, CHF, stroke, and morbid obesity; RA was the only comorbidity without a significant difference (p = 0.4234). The mean CCI showed a significant difference between patients with CAD and controls (p < 0.001).
For the primary outcome of thoracolumbar vertebral compression fracture, the CAD cohort had a higher cumulative incidence rate than the non-CAD cohort (0.9% vs.0.47%; p < 0.001) (Table 2). The secondary outcome of physician-diagnosed OP was present for 22,309 patients with CAD and 10,771controls (cumulative incidence rate 11.6% vs. 5.6%; p < 0.001) during the follow-up period (2009–2018).
Table 2.
Incidence of thoracolumbar vertebral compression fracture, osteoporosis, and the crude and adjusted hazard ratios (HRs) derived from the Cox model for the CAD cohort compared with that from the comparison non-CAD cohort stratified by patient characteristics
| Case (N = 192,367) | Compare (N = 192,367) | |||||
| N | % | Incidence rate | N | % | Incidence rate | |
| Compression fracture | 1740 | 0.9 | 100.8 | 913 | 0.47 | 53.4 |
| Osteoporosis | 22,309 | 11.6 | 1420.4 | 10,771 | 5.6 | 657.4 |
| Case vs compare | ||||||
| Crude hazard ratio (95% CI) | p value | Adjusted HR (95% CI) | p value | |||
| Compression fracture | 1.91 (1.76–2.06) | < 0.001 | 1.74 (1.60–1.89) | < 0.001 | ||
| Osteoporosis | 2.13 (2.09–2.18) | < 0.001 | 2.04 (1.99–2.08) | < 0.001 | ||
Rate incidence per 100,000 PYs
PYs person-years
For the primary outcome measure, the CAD cohort had a significant increase in the risk for thoracolumbar vertebral compression fracture, with a crude HR and adjusted HR of 1.91 (95% CI, 1.76–2.06) and 1.74 (95% CI, 1.60–1.89), respectively (Table 2). At 10-year follow-up, 22,309 patients with CAD and 10,771 controls received consistent diagnoses of OP given by a physician. The incidence rates of OP per 100,000 patients per year were 1420 in the CAD cohort and 657 in the comparison cohort, with an incidence rate ratio of 2.16 (Table 2). The crude HR was also statistically significant, showing a 113% increase in the risk with an HR of 2.13 (95% CI, 2.08–2.18). This significant increase of the risk was sustained even after the multivariate Cox model adjusting for the aforementioned confounding factors with an adjusted HR of 2.04 (95% CI, 1.99–2.08; p < 0.001) (Table 2).
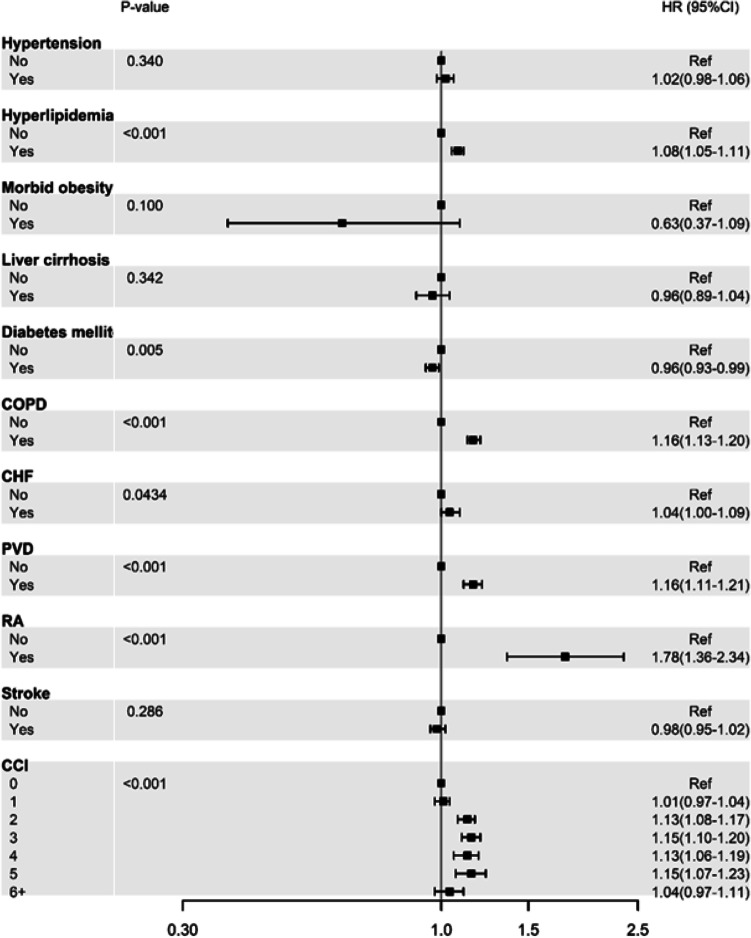
Figure 2 shows the multivariate Cox model analysis of the differential risk among different categorizations, such as hyperlipidemia, for patient characteristics, with the adjusted HR of OP stratified by each patient characteristic. The patients with hyperlipidemia had an increased risk of OP (adjusted HR 1.08 95% CI, 1.05–1.11), as shown in Fig. 2. Moreover, patients with COPD, PVD, and RA had an increased risk of OP compared with those who did not have these comorbidities (Fig. 2). Additionally, patients with a CCI score of 2–5 also had a significantly increased risk of OP compared with patients who had a CCI score of 0.
Fig. 2.
Cox model–derived crude and multivariate adjusted hazard ratios (HRs) of osteoporosis development stratified by patient characteristics
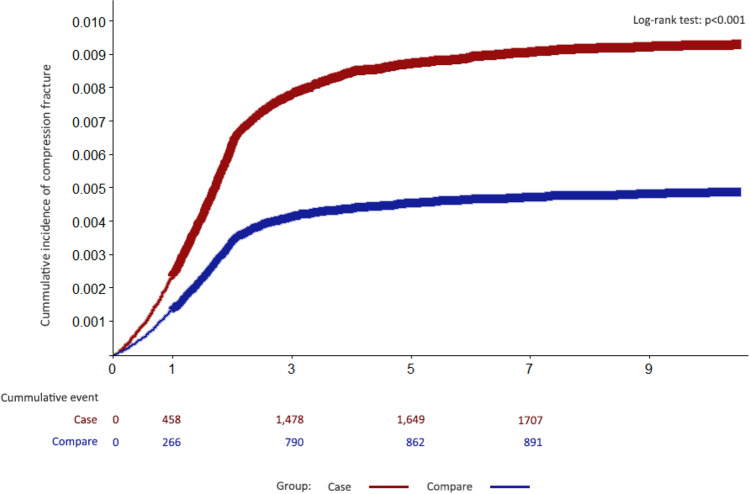
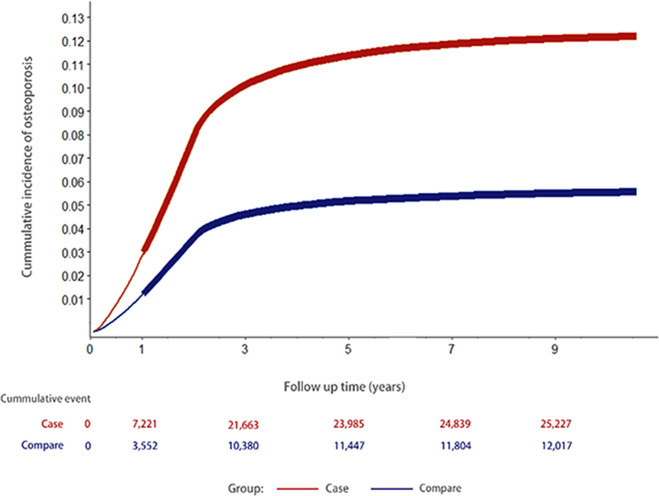
We constructed a multivariate Cox model to derive the cumulative incidence function excluding the first year of follow-up events. The cumulative incidence of osteoporotic fracture is 0.9% and 0.47% (p < 0.001, log-rank) for the CAD and non-CAD cohorts (Fig. 3). For the secondary outcome, Fig. 4 shows the cumulative incidence of OP for the CAD and non-CAD cohorts, which is 11.6% and 5.6%, respectively (p < 0.001, log-rank).
Fig. 3.
Cumulative incidence of thoracolumbar vertebral compression fracture, which is 0.9% and 0.47%, respectively, in the CAD and non-CAD cohorts (p < 0.0001, compared with log-rank)
Fig. 4.
Cumulative incidence of osteoporosis, which is 11.6% and 5.8%, respectively, in the CAD and non-CAD cohorts (p < 0.0001, compared with log-rank)
Discussion
The understanding of the pathophysiologic factors for and associations shared by CAD and OP has increased over the recent past years. Unfortunately, no consistent results have been shown to date, and both diseases have yet to be investigated adequately with regard to the etiologic role of CAD as a possible risk factor for OP. To our knowledge, this study is the first to examine data from the NHIRD as a national population-based study to analyze the association between OP and CAD in Taiwan. Our study discovered a significant increase in the risk of developing a vertebral compression fracture for patients with CAD compared with their non-CAD counterparts based on the multivariate Cox model (adjusted HR 1.74). Additionally, the CAD group also had a higher incident rate of osteoporosis than the control group. Overall, a positive association between CAD and subsequent OP development was shown in this study.
Previous studies have demonstrated that age-related bone loss and cardiovascular disease may be related to chronic inflammation [20, 21]. Elevation of osteoprotegerin, sclerostin, or FGF-23 levels may explain and predict the occurrence of both OP fractures and cardiovascular events [13, 22, 23]. On the basis of these pathophysiologic studies, there is an association between the OP and CAD. Our study also provided evidence of an association between the two diseases. However, to clarify the mechanisms for and the association between CAD and OP, a more well-designed study is needed to establish stronger data for the pathophysiologic association.
Two recent meta-analyses have revealed controversial results for the association between OP and CAD. One meta-analysis assessed 4170 participants from 11 studies. The pooled odds ratio (OR) for the incidence of CAD in patients with low BMD versus patients with normal BMD was 1.58 (95% CI, 0.99–2.52; p = 0.06), and no statistical difference was found between the subgroups of men and women. After analyzing the confounding age, the combined OR was 1.60 (95% CI, 0.69–3.72; p = 0.27) [16]. Therefore, their conclusion was that the low BMD was not associated with the prevalence of CAD. The other meta-analysis assessed 10,299 patients from 25 studies. They found that the incidence of atherosclerotic vascular abnormalities was significantly increased in patients with low BMD, compared with patients with normal BMD (OR 1.81; 95% CI, 1.01–2.19; p < 0.00001). After the study adjusted for age, sex, body mass index (BMI), and other vascular risk factors, decreased BMD remained significantly associated with the incidence of atherosclerotic vascular abnormalities (OR 2.96; 95% CI, 2.25–3.88; p < 0.00001) [24]. These findings showed that decreased BMD was an independent predictor for the development of atherosclerosis in older adults.
Some cohort studies evaluated whether lower bone mass affects cardiovascular events. One cohort study consisted of 5590 consecutive at-risk patients without known CAD, age 57 ± 12 years, and 69% male, who underwent nonenhanced cardiac computed tomography, followed for an average of 8 years. Analysis of the clinical outcomes of CAC scanning and thoracic BMD measurement showed a significant link between low BMD levels and CAC with an increased risk of mortality in both sexes across ethnicities. They claimed that the lower BMD levels are independently associated with the severity of CAC that predicts mortality [15]. Another prospective cohort study in France included 744 men older than 50 years with a 7.5-year follow-up. They found 40 patients had several myocardial infarction, and 43 patients had a stroke [25]. After adjusting for risk factors, the patients whose BMD was in the lowest quartile or whose bone resorption markers were in the highest quartile had a twofold higher risk of cardiovascular events. Furthermore, after adjustment, BMD was significantly lower in the patients with stroke. Last, a prospective cohort study in Sweden evaluated 6872 men and women for 5.7 years, during which time 196 patients experienced myocardial infarction [26]. Myocardial infarction was then concluded to be significantly associated with low hip BMD (women, OR 1.33; 95% CI, 1.08–1.66; men, OR 1.74; 95% CI, 1.34–2.28).
Our current study had several strengths, including the large sample size, randomization, and sufficient follow-up duration. The data originated from a national population dataset, which provided the appropriate statistical power for detecting the association between the two diseases. Additionally, when analyzing a random matching of the non-CAD cohort, it was well balanced for the index date, sex, and age. However, this study has several limitations. First, possible coding errors and other coding problems may occur in the administrative databases. For our study, the definition of CAD was based on the coding in the database, but the accuracy between the diagnosis and coding was not verified. To minimize the interference, we added the restriction that the CAD diagnosis must exist at or more two ambulatory care visits or at one or more inpatient visits and must be made by certain specialists. Second, data were limited or unavailable for serum 25-hydroxyvitamin D; serum parathyroid hormone; proinflammatory cytokines such as IL-1, IL-6, and TNF-α; and serial BMD values, so these factors could not be analyzed further. Additionally, BMI and lifestyle, such as individual dietary habits and physical activity, data related to CAD and OP were not recorded in the dataset. Because this study showed a positive association between CAD and OP, it is worthy of initiating well-designed in vivo or in vitro studies to investigate causation and pathophysiologic link between the two diseases in the future.
Conclusions
In conclusion, patients with CAD have a significant increase in the risk of developing vertebral compression fracture and osteoporosis than those without CAD. This study has demonstrated the positive association between CAD and subsequent development of vertebral compression fracture and OP. To prevent the complications of OP, patients with CAD should receive a personalized risk assessment and screening for OP.
Author contribution
De-Kai Syu: conceptualization, methodology, investigation, writing — original draft, data curation; Shu-Hua Hsu: conceptualization, methodology; Ping-Chun Yeh: conceptualization, methodology; Yen-Chun Huang: software, formal analysis; Yu-Feng Kuo: writing — original draft, writing — reviewing and editing; Ching-Chuan Jiang: supervision, writing — reviewing and editing; Mingchih Chen: project administration, supervision, writing — reviewing and editing. All authors have made substantial contributions to all of the following: (1) the conception and design of the study, or acquisition of data, or analysis and interpretation of data; (2) drafting of the article or critical revision for important intellectual content; (3) final approval of the version to be submitted.
Funding
This study was funded by Fu Jen Catholic University (A0109152) and Ministry of Science and Technology (109–2221-E-030–011-MY3).
Data availability
These data were available to us as staffs of the Department of Orthopedics at Fu Jen Catholic University Hospital and at Fu Jen Catholic University, using the National Hospital Research Database (NHIRD). These data are protected by the Ministry of Health and Welfare and patient privacy laws in Taiwan; no public links are available to these protected health information datasets. These data will be made available to others after appropriate data privacy and human subject approvals needed by the institution. Requests should be sent to 081438@mail.fju.edu.tw.
Declarations
Ethics approval and consent to participate
This study was approved by the Institutional Review Board and Ethics Committee of Fu Jen Catholic University (IRB approval number: C108094). There was no consent in this study. All data collected in this study were all encrypted using the same encryption algorithm to cross-link the data while protecting the privacy of the patients.
Consent for publication
No individual person’s data were presented in any form in this study; therefore, no consent to publish was required.
Conflict of interest
None.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Ching-Chuan Jiang, Email: ccj@ntu.edu.tw.
Mingchih Chen, Email: 081438@mail.fju.edu.tw.
References
- 1.Miller PD. Management of severe osteoporosis. Expert Opin Pharmacother. 2016;17(4):473–488. doi: 10.1517/14656566.2016.1124856. [DOI] [PubMed] [Google Scholar]
- 2.Malakar AK, Choudhury D, Halder B, Paul P, Uddin A, Chakraborty S. A review on coronary artery disease, its risk factors, and therapeutics. J Cell Physiol. 2019;234(10):16812–16823. doi: 10.1002/jcp.28350. [DOI] [PubMed] [Google Scholar]
- 3.Liu N, Chen J, Zhang K, Tang Z. A community-based study of the relationship between coronary artery disease and osteoporosis in Chinese postmenopausal women. Coron Artery Dis. 2016;27(1):59–64. doi: 10.1097/MCA.0000000000000306. [DOI] [PubMed] [Google Scholar]
- 4.Epstein FH. Cardiovascular disease epidemiology: a journey from the past into the future. Circulation. 1996;93:1755–1764. doi: 10.1161/01.CIR.93.9.1755. [DOI] [PubMed] [Google Scholar]
- 5.Cooper C, Atkinson EJ, Jacobsen SJ, O’Fallon WM, Melton LJ., 3rd Population-based study of survival after osteoporotic fractures. Am J Epidemiol. 1993;137:1001–5. doi: 10.1093/oxfordjournals.aje.a116756. [DOI] [PubMed] [Google Scholar]
- 6.Lerner DJ, Kannel WB. Patterns of coronary heart disease morbidity and mortality in the sexes: a 26-year follow-up of the Framingham population. Am Heart J. 1986;111:383–390. doi: 10.1016/0002-8703(86)90155-9. [DOI] [PubMed] [Google Scholar]
- 7.Greenland P, Reicher-Reiss H, Goldbourt U, Behar S. In-hospital and 1-year mortality in 1,524 women after myocardial infarction. Comparison with 4,315 men. Circulation. 1991;83:484–91. doi: 10.1161/01.CIR.83.2.484. [DOI] [PubMed] [Google Scholar]
- 8.Stambler I, Jin K, Lederman S, Barzilai N, Olshansky SJ, Omokaro E, Barratt J, Anisimov VN, Rattan S, Yang S. Aging health and R&D for healthy longevity must be included into the WHO work program. Aging Dis. 2018;9(2):331–333. doi: 10.14336/AD.2017.1120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen FP, Huang TS, Fu TS, Sun CC, Chao AS, Tsai TL. Secular trends in incidence of osteoporosis in Taiwan: a nationwide population-based study. Biomed J. 2018;41(5):314–320. doi: 10.1016/j.bj.2018.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tsai C-C, Hsieh M-H, Yang H-Y, Chan P, Jeng C. Predictors of coronary artery disease in middle-aged Taiwanese women at premenopause, postmenopause and after undergoing hysterectomy. J Clin Nurs. 2016;25(17–18):2438–2449. doi: 10.1111/jocn.13258. [DOI] [PubMed] [Google Scholar]
- 11.NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy Osteoporosis Prevention, Diagnosis, and Therapy. JAMA. 2001;285:785–95. doi: 10.1001/jama.285.6.785. [DOI] [PubMed] [Google Scholar]
- 12.Aronow WS, Silent MI. Prevalence and prognosis in older patients diagnosed by routine electrocardiograms. Geriatrics. 2003;58:24–6. [PubMed] [Google Scholar]
- 13.Laroche M, Pécourneau V, Blain H, Breuil V, Chapurlat R, Cortet B, Sutter B, Degboe Y. Osteoporosis and ischemic cardiovascular disease. Joint Bone Spine. 2017;84(4):427–432. doi: 10.1016/j.jbspin.2016.09.022. [DOI] [PubMed] [Google Scholar]
- 14.Crepaldi G, Maggi S. Epidemiologic link between osteoporosis and cardiovascular disease. J Endocrinol Invest. 2009;32(4 Suppl):2–5. [PubMed] [Google Scholar]
- 15.Ahmadi N, Mao SS, Hajsadeghi F, Arnold B, Kiramijyan S, Gao Y, Flores F, Azen S, Budoff M. The relation of low levels of bone mineral density with coronary artery calcium and mortality. Osteoporos Int. 2018;29(7):1609–1616. doi: 10.1007/s00198-018-4524-7. [DOI] [PubMed] [Google Scholar]
- 16.Zhang Y, He B, Wang H, Shi J, Liang H. Associations between bone mineral density and coronary artery disease: a meta-analysis of cross-sectional studies. Arch Osteoporos. 2020;15(1):24. doi: 10.1007/s11657-020-0691-1. [DOI] [PubMed] [Google Scholar]
- 17.Lee SN, Cho JY, Eun YM, Song SW, Moon KW. Associations between osteoporosis and coronary artery disease in postmenopausal women. Climacteric. 2016;19(5):458–462. doi: 10.1080/13697137.2016.1200550. [DOI] [PubMed] [Google Scholar]
- 18.Lin AC, Chao E, Yang CM, Wen HC, Ma HL, Lu TC. Costs of staged versus simultaneous bilateral total knee arthroplasty: a population-based study of the Taiwanese National Health Insurance Database. J Orthop Surg Res. 2014;9:59. doi: 10.1186/s13018-014-0059-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hsiao F-Y, Huang Y-T, Huang W-F. Using Taiwan’s National Health Insurance Research Databases for pharmacoepidemiology research. J Food Drug Anal. 2007;15(2):99–108. [Google Scholar]
- 20.Lencel P, Magne D. Inflammaging: the driving force in osteoporosis? Med Hypotheses. 2011;76:317–321. doi: 10.1016/j.mehy.2010.09.023. [DOI] [PubMed] [Google Scholar]
- 21.Shao JS, Cheng SL, Sadhu J, et al. Inflammation and the osteogenic regulation of vascular calcification: a review and perspective. Hypertension. 2010;55:579–592. doi: 10.1161/HYPERTENSIONAHA.109.134205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Libby P. Inflammation in atherosclerosis. Nature. 2002;420:868–874. doi: 10.1038/nature01323. [DOI] [PubMed] [Google Scholar]
- 23.Pfeilschifter J, Koditz R, Pfohl M, et al. Changes in pro inflammatory cytokine activity after menopause. Endocr Rev. 2002;23:90–119. doi: 10.1210/edrv.23.1.0456. [DOI] [PubMed] [Google Scholar]
- 24.Ye C, Xu M, Wang S, Jiang S, Chen X, Zhou X, He R (2016) Decreased bone mineral density is an independent predictor for the development of atherosclerosis: a systematic review and meta-analysis. PLoS One 11(5):e0154740. 10.1371/journal.pone.0154740 [DOI] [PMC free article] [PubMed]
- 25.Szulc P, Samelson EJ, Kiel DP, et al. Increased bone resorption is associated with increased risk of cardiovascular events in men: the MINOS study. J Bone Miner Res. 2009;24:2023–2031. doi: 10.1359/jbmr.090531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wiklund P, Nordstrom A, Jansson JH, et al. Low bone mineral density is associated with increased risk for myocardial infarction in men and women. Osteoporosis Int. 2012;23:963. doi: 10.1007/s00198-011-1631-0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
These data were available to us as staffs of the Department of Orthopedics at Fu Jen Catholic University Hospital and at Fu Jen Catholic University, using the National Hospital Research Database (NHIRD). These data are protected by the Ministry of Health and Welfare and patient privacy laws in Taiwan; no public links are available to these protected health information datasets. These data will be made available to others after appropriate data privacy and human subject approvals needed by the institution. Requests should be sent to 081438@mail.fju.edu.tw.