Abstract
Background
Congenital toxoplasmosis is considered a rare but potentially severe infection. Prenatal education about congenital toxoplasmosis could be the most efficient and least harmful intervention, yet its effectiveness is uncertain.
Objectives
To assess the effects of prenatal education for preventing congenital toxoplasmosis.
Search methods
We searched the Cochrane Pregnancy and Childbirth Group's Trials Register (31 May 2015), and reference lists of relevant papers, reviews and websites.
Selection criteria
Randomized and quasi‐randomized controlled trials of all types of prenatal education on toxoplasmosis infection during pregnancy. Cluster‐randomized trials were eligible for inclusion.
Data collection and analysis
Two review authors independently assessed trials for inclusion and risk of bias, extracted data and checked them for accuracy.
Main results
Two cluster‐randomized controlled trials (RCTs) (involving a total of 5455 women) met the inclusion criteria. The two included trials measured the effectiveness of the intervention in different ways, which meant that meta‐analysis of the results was not possible. The overall quality of the two studies, as assessed using the GRADE approach, was low, with high risk of detection and attrition bias in both included trials.
One trial (432 women enrolled) conducted in Canada was judged of low methodological quality. This trial did not report on any of the review's pre‐specified primary outcomes and the secondary outcomes reported results only as P values. Moreover, losses to follow‐up were high (34%, 147 out of 432 women initially enrolled). The authors concluded that prenatal education can effectively change pregnant women's behavior as it increased pet, personal and food hygiene. The second trial conducted in France was also judged of low methodological quality. Losses to follow‐up were also high (44.5%, 2233 out of 5023 women initially enrolled) and differential (40% in the intervention group and 52% in the control group). The authors concluded that prenatal education for congenital toxoplasmoses has a significant effect on improving women's knowledge, whereas it has no effect on changing women's behavior. In this trial 17/3949 pregnant women seroconverted for toxoplasmosis: 13/2591 (0.5%) in the intervention group and 4/1358 (0.3%) in the control group. The rate of seroconversion detected during the study did not differ between groups (risk ratio (RR) 1.70, 95% confidence interval (CI) 0.56 to 5.21; participants = 3949; studies = one, low quality evidence). The number of events was too small to reach conclusions about the effect of prenatal education on seroconversion rate during pregnancy.
No other randomized trials on the effect of prenatal education on congenital toxoplasmosis rate, or toxoplasmosis seroconversion rate during pregnancy were detected.
Authors' conclusions
Even though primary prevention of congenital toxoplasmosis is considered a desirable intervention, given the lack of related risks compared to secondary and tertiary prevention, its effectiveness has not been adequately evaluated. There is very little evidence from RCTs that prenatal education is effective in reducing congenital toxoplasmosis even though evidence from observational studies suggests it is. Given the lack of good evidence supporting prenatal education for congenital toxoplasmosis prevention, further RCTs are needed to confirm any potential benefits and to further quantify the impact of different sets of educational intervention.
Plain language summary
Prenatal education for congenital toxoplasmosis
Toxoplasmosis infection is caused by a parasite, Toxoplasma gondii. Eating or handling raw or insufficiently‐cooked meat, not washing hands thoroughly after gardening, handling contaminated soil or water, or contact with cats' faeces can cause infection. Usually it is asymptomatic and self‐limiting. Primary prevention involves educating the general public, filtering water and improving farm hygiene to reduce animal infection.
If pregnant women have not previously been exposed to the parasite and develop antibodies while pregnant, the infection can be transmitted from the mother to the fetus (congenital toxoplasmosis). This is rare but potentially has serious consequences such as malformation, mental retardation, deafness and blindness of the infected infant, intrauterine death or stillbirth. The probability of infection is greater during the third trimester but the risk of the fetus developing major clinical signs is greater early in pregnancy. Pregnant women are often unaware of risk factors for congenital toxoplasmosis. Offering prenatal education could mean that women adopt simple behavioral measures to avoid toxoplasmosis.
This review included two randomized controlled trials (involving 5455 women). Data could not be combined because each trial measured effectiveness in different ways. One study was from Canada and involved 432 women randomly assigned to a 10‐minute presentation during a prenatal class about toxoplasmosis prevention or to a usual prenatal class. Losses to follow‐up were high and 285 completed the post‐test questionnaire in the third trimester. Only 5% of the intervention women recalled having obtained information on toxoplasmosis prevention during prenatal classes. However, the authors concluded that prenatal education can effectively change pregnant women's behavior as it increased pet, personal and food hygiene. The other trial conducted in France involved 5023 pregnant women with no evidence of toxoplasmosis infection (seronegative) who were randomly assigned to receive a brochure and an audiotape containing information for toxoplasmosis prevention, or to a usual prenatal class. Losses to follow‐up were high and 2790 completed both pre‐test and post‐test questionnaire on behavior (44.5% loss to follow‐up), whereas 3949 women were tested for blood antibodies (22.4% loss to follow‐up). Women's behavior did not change after the intervention. Similarly, the seroconversion rate did not differ between groups (13 out of 2591 women seroconverted in the intervention and four out of 1358 in the control group).
Both trials were judged as having low methodological quality as assessed by the GRADE approach. This limits our confidence in the results. Evidence supporting prenatal education to prevent congenital toxoplasmosis is therefore limited.
Summary of findings
Background
Readers may wish to refer to the following Cochrane systematic review for further information about toxoplasmosis in pregnancy: 'Treatments for toxoplasmosis in pregnancy' (Peyron 1999).
Description of the condition
Congenital toxoplasmosis is a rare (NSC 2001), but potentially severe parasitic infection that can lead to intrauterine death or stillbirth, malformation, mental retardation, deafness and blindness of the infected infant (Montoya 2004). Incidence of congenital toxoplasmosis varies from one to 10 per 10,000 live children in Western countries (Gilbert 1999; Schmidt 2006; Signorell 2006), to 16 per 1000 in Brazil (Reis 1999). The infection is caused by Toxoplasma gondii (T. gondii). T. gondii is one of the most common infectious pathogenic animal parasites of man, belonging to the phylum apicomplexa group (Montoya 2004). Other members of this phylum include known human pathogens such as Plasmodium (malaria) and Cryptosporidium. It is acquired by ingesting oocysts excreted by cats, contaminated soil or water, or by eating the undercooked meat of infected animals, which contain tissue cysts (Gilbert 2002; Montoya 2004). Most cases of toxoplasmosis infection are asymptomatic and self‐limited except for congenital infection and immunocompromised patients (Montoya 2004); hence many cases remain undiagnosed. The incubation period of acquired infection is estimated to be within the range of four to 21 days (seven days on average) (Rorman 2006). Serological surveys demonstrate that worldwide exposure to T. gondii is high (30% in USA and 50% to 80% in Europe) (Rorman 2006). The susceptibility of pregnant women (that is the rate of seronegative pregnant women) to toxoplasmosis varies between countries. It is up to 90% in northern Europe, where T. gondii is not so common (Allain 1998; Gilbert 2002). When infection does occur during pregnancy, T. gondii can be transmitted from the mother to the fetus (vertical transmission) and can lead to congenital toxoplasmosis. Multiple factors are associated with the occurrence of congenital toxoplasmosis infection, including route of transmission, climate, cultural behavior, eating habits and hygenic standards (Rorman 2006). The probability of transmission of the parasite to the fetus varies according to the gestational age and the risk is greater during the third trimester (from 5% at 12 weeks to 80% just before delivery) (Dunn 1999). Conversely, the severity of the condition, that is the risk of the fetus developing major clinical signs, decreases with increasing gestational age (from 60% at 12 weeks to 5% just before delivery) (Dunn 1999). Clinical features include hydrocephalus (excessive accumulation of cerebrospinal fluid within the cranium), microcephaly (abnormal smallness of the head, usually associated with mental retardation), deafness, cerebral calcifications, seizures and psychomotor retardation. Signs of a systemic infection may also be present at birth, including fever, rash, and enlargement of liver and spleen. Fetal infection can cause inflammatory lesions of the retina and choroids that can lead to visual impairment. Moreover, it can cause lesions of the brain leading to mental damage; more rarely, the infection can cause the death of the fetus or the newborn (Gilbert 2002; Montoya 2004; NSC 2001). Severe damage in infancy occurs in 5% of congenital toxoplasmosis cases, while intracranial or ocular lesions are observed in 20% to 30% of cases by three years of age (Gilbert 2001).
Although there is no consensus on the most appropriate screening or treatment for congenital toxoplasmosis, three possible approaches have been proposed: prenatal screening, neonatal screening and primary prevention (Gilbert 2002). This systematic review will focus on primary prevention through prenatal education of pregnant women. Other possibilities are discussed below.
Prenatal screening
Prenatal screening (secondary prevention) is offered in some European countries, for example, France, Switzerland, Germany, Austria and Italy where the incidence of T. gondii maternal infection is more frequent, and is based on the timely detection of the mother's infection by a serum test for toxoplasma Immunoglobulin G (IgG) and Immunoglobulin M (IgM) (NSC 2001). If the first prenatal test shows signs of recent infection or a seroconversion is detected during pregnancy, a confirmatory test is required before starting treatment with spiramycin or pyrimethamine‐sulfadoxine, or both (Foulon 1999a; Gilbert 2002; Montoya 2004). Diagnosis of fetal infection is performed by amniocentesis, which is known to be associated with a 1% risk of miscarriage (Alfirevic 2003), and testing of the amniotic fluid for the detection of the parasite or of toxoplasma DNA by polymerase chain reaction (PCR) technique. Congenital toxoplasmosis can also be diagnosed by cordocentesis, that consists of drawing fetal blood from the umbilical cord, and the detection of the parasite or specific immunoglobulin (IgM and IgA) in the fetal blood, but the risk of complications due to the procedure is higher (Bader 1997; Foulon 1999b; Gilbert 2002). If fetal infection is confirmed, the parents can decide either to terminate the pregnancy or to opt for drug treatment. Prenatal screening, although advocated by some as essential for reducing congenital toxoplasmosis (Boyer 2005), has several limitations: false‐positive toxoplasma IgM results are common, false‐positive toxoplasma IgG are less common but also possible (Liesenfeld 1997; Montoya 2004); moreover, the rate of false‐positive test results can increase, notably in settings where local prevalence of the infection is lower; there can be organizational problems or problems of acceptability due to the need to repeat the serum test every four to six weeks in seronegative women (Bader 1997); there is no evidence that antenatal treatment is effective in reducing transmission to the fetus nor in improving neonatal outcomes or reducing functional impairment in later childhood (Gilbert 2003; Peyron 1999; SYROCOT 2007); there are problems concerning the accuracy of the diagnostic test for fetal infection, particularly the lack of a standardized technique for PCR (Chabbert 2004; Foulon 1999b; Thalib 2005). Finally, this strategy causes additional fetal losses of healthy fetuses due to amniocentesis and to elective terminations of pregnancy. It has been estimated that the number of additional losses necessary to prevent one additional case of toxoplasmosis can be as high as 18.5 in cases of universal screening in a setting with a low incidence of T. gondii maternal infection, such as the USA (Bader 1997).
Neonatal screening
Neonatal screening (tertiary prevention), adopted in Poland, Denmark and some areas of the USA (NSC 2001), consists of the diagnosis of newborn infection through detection of toxoplasma specific IgM on Guthrie card blood spots. In fact, up to 90% of infected infants are asymptomatic at birth and will show clinical symptoms only in later life (Gilbert 2001; Wilson 1980). Current guidelines suggest that infected infants should receive treatment with pyrimethamine and sulfadiazine for up to one year, regardless of symptoms (Gilbert 2002). Even if this strategy is technically feasible and less costly than prenatal screening, it has been proven to have a low sensitivity, even when the test is performed on serum samples, which are more valid than the test on filter paper blood samples currently used: neonatal screening is not able to detect almost half of all infected infants (Gilbert 2007). Moreover, there is no evidence that treating the infected children has any effect (Gilbert 2002; Lebech 1999). Finally, this approach is ineffective on irreversible damage already present at birth. Considering such limitations, neonatal screening should be adopted only in places where other options are not available; the implications of such a policy should be fully discussed with the parents of the tested newborn.
Primary prevention
Primary prevention can involve the whole population by educating the general public and filtering water, and veterinary public health interventions (such as labeling to indicate toxoplasma‐free meat and improved farm hygiene to reduce animal infection). This will reduce the protozoan circulation and could be an option but up to now there is not enough research to determine the feasibility and efficacy of this approach (Gilbert 2002; NSC 2001). Another possibility is primary prevention based on prenatal education of pregnant women or women of reproductive age to avoid toxoplasmosis in pregnancy (Gilbert 2002).
Description of the intervention
This review evaluates the effectiveness of prenatal education for reducing the risk to develop toxoplasmosis infection during pregnancy. Various types of prenatal education exist and can be proposed in different settings, either as a stand alone intervention or as a component of a multifaceted intervention program and they are all assessed in this review. Prenatal educational interventions could include: antenatal classes provided to pregnant women, distribution of leaflets to pregnant women or to women of reproductive age, irrespective of their pregnant status, one‐to‐one or group counseling from different professionals (nurses, midwives, obstetricians and gynecologists, social workers, counselors, teachers, trained lay people, etc), educational intervention in schools, mass‐media campaign and others.
How the intervention might work
Primary prevention based on prenatal education, if proven to be effective, could be a good strategy to reduce congenital toxoplasmosis, since it will not involve any of the problems linked to secondary and tertiary prevention strategies. In fact, sources and risk factors for contracting toxoplasmosis are well known (Cook 2000), and can be avoided by adopting simple behavioral measures such as not eating raw or insufficiently cooked meat, washing hands thoroughly after handling raw meat and after gardening, avoiding contact with cats' faeces (directly or indirectly through the soil, or possibly contaminated raw vegetables or fruits) (Cook 2000; Gilbert 2002). Nevertheless, pregnant women are often unaware of risk factors for congenital toxoplasmosis (Ferguson 2011). If adequately informed, women can be more prone to change their own behavior during pregnancy than during any other period of their life; for this reason several intervention to promote healthy behavior are implemented during pregnancy (see other Cochrane systematic review on pregnancy and education Chamberlain 2013).
Why it is important to do this review
When this systematic review was first published in 2009, we aimed to synthesize available evidence on effective intervention to reduce congenital toxoplasmosis, a rare but potentially severe condition for which effective treatment is still lacking. Primary prevention was theoretically considered to be, and still is, the most valuable and safe intervention to reduce the incidence and prevalence of this congenital infection. Unfortunately, evidence of its effectiveness resulted to be scanty and studies poorly conducted or not adequately reported. A further update of the review, although permitted to add one more study to the list of included studies (Wallon 2006) did not change the conclusion: prenatal education seem to be a promising intervention to reduce congenital toxoplasmosis but strong evidence is lacking. This review calls for further research on this relevant issue that is very likely to have an important impact on our confidence in the estimate of effect and is likely to change our conclusions; moreover, the systematic review provides methodological hints for producing valuable evidence that matter for policy makers, health professionals, and women.
Objectives
The primary objectives of this review were to assess the efficacy of prenatal education to reduce the rate of:
new cases of congenital toxoplasmosis;
toxoplasmosis seroconversion during pregnancy.
Secondary objectives were to assess the efficacy of prenatal education to increase the rate of:
pregnant women's knowledge of risk factors for acquiring toxoplasmosis infection;
pregnant women's awareness of the importance of avoiding toxoplasmosis infection during pregnancy;
pregnant women's behavior with respect to avoidance of risk factors for toxoplasmosis infection during pregnancy.
Methods
Criteria for considering studies for this review
Types of studies
Randomized and quasi‐randomized controlled trials evaluating any kind of prenatal educational intervention dealing with toxoplasmosis infection in pregnancy, and how to avoid it, were assessed for inclusion. Studies where the control group included an alternative intervention or no intervention were also considered for inclusion. Studies where the unit of randomization was a group of women (cluster‐randomization) were assessed for inclusion and analyzed as a separate group. Interventions, exclusively focused on toxoplasmosis or interventions not exclusively focused on toxoplasmosis infection but where toxoplasmosis was included among a series of different topics, were also eligible for inclusion.
Types of participants
Trials of women of reproductive age, irrespective of their pregnant status were included. Since a screening policy for toxoplasmosis infection is not universally adopted, studies including women, irrespective of their toxoplasmosis seropositive status were included.
Types of interventions
Any kind of prenatal education on toxoplasmosis infection during pregnancy. Prenatal educational interventions could include: antenatal classes provided to pregnant women, distribution of leaflets to pregnant women or to women of reproductive age, irrespective of their pregnant status, one‐to‐one or group counseling from different professionals (nurses, midwives, obstetricians and gynecologists, social workers, counselors, teachers, trained lay people, etc), educational intervention in schools, mass‐media campaign and others.
Types of outcome measures
Primary outcomes
Rate of congenital toxoplasmosis, defined by persistence of specific IgG antibodies beyond 11 months of age (Lebech 1996).
-
Rate of toxoplasmosis seroconversion in pregnant women, defined by:
an increase in specific IgG from paired sera in pregnant woman previously seronegative;
a rising IgG titre, low IgG avidity, IgA antibodies, or a combination of these in pregnant women who were IgG and IgM positive at their first prenatal test (Gilbert 2002).
Secondary outcomes
Pregnant women's knowledge of risk factors for acquiring toxoplasmosis infection as objectively measured (quantitative score) through specific questionnaire.
Pregnant women's awareness of the importance of avoiding toxoplasmosis infection during pregnancy as objectively measured (quantitative score) through specific questionnaire.
Pregnant women's behavior with respect to the avoidance of risk factors for toxoplasmosis infection during pregnancy as objectively measured (quantitative score) through specific questionnaire.
Search methods for identification of studies
The following methods section of this review is based on a standard template used by the Cochrane Pregnancy and Childbirth Group.
Electronic searches
We searched the Cochrane Pregnancy and Childbirth Group’s Trials Register by contacting the Trials Search Co‐ordinator (31 May 2015).
The Cochrane Pregnancy and Childbirth Group’s Trials Register is maintained by the Trials Search Co‐ordinator and contains trials identified from:
monthly searches of the Cochrane Central Register of Controlled Trials (CENTRAL);
weekly searches of MEDLINE (Ovid);
weekly searches of Embase (Ovid);
monthly searches of CINAHL (EBSCO);
handsearches of 30 journals and the proceedings of major conferences;
weekly current awareness alerts for a further 44 journals plus monthly BioMed Central email alerts.
Details of the search strategies for CENTRAL, MEDLINE, Embase and CINAHL, the list of handsearched journals and conference proceedings, and the list of journals reviewed via the current awareness service can be found in the ‘Specialized Register’ section within the editorial information about the Cochrane Pregnancy and Childbirth Group.
Trials identified through the searching activities described above are each assigned to a review topic (or topics). The Trials Search Co‐ordinator searches the register for each review using the topic list rather than keywords.
For additional search strategy used in the previous version of this review (Di Mario 2013), see Appendix 1.
Searching other resources
We searched the reference lists of relevant papers, reviews and websites and contacted researchers working in the field for information on any relevant studies and for any additional published or unpublished studies.
We did not apply any language or date restrictions.
Data collection and analysis
For methods used in the previous version of this review, seeDi Mario 2013.
For this update, no new reports were identified for assessment but we assessed the quality of evidence of the existing studies using the GRADE approach as outlined in the GRADE Handbook. In order to assess the quality of the body of evidence relating to the following outcomes for the two comparisons: 'educational prenatal classes compared with usual prenatal classes and brochure and an audiotape compared with standard care'.
Rate of congenital toxoplasmosis.
Rate of toxoplasmosis seroconversion during pregnancy.
Pregnant women's knowledge of risk factors for acquiring toxoplasmosis infection.
Pregnant women's awareness of the importance of avoiding toxoplasmosis infection during pregnancy.
Pregnant women's behavior with respect to avoidance of risk factors for toxoplasmosis infection during pregnancy.
GRADEpro Guideline Development Tool was used to import data from Review Manager 5.3 (RevMan 2014) in order to create ’Summary of findings’ tables. A summary of the intervention effect and a measure of quality for each of the above outcomes has been produced using the GRADE approach. The GRADE approach uses five considerations (study limitations, consistency of effect, imprecision, indirectness and publication bias) to assess the quality of the body of evidence for each outcome. The evidence can be downgraded from 'high quality' by one level for serious (or by two levels for very serious) limitations, depending on assessments for risk of bias, indirectness of evidence, serious inconsistency, imprecision of effect estimates or potential publication bias.
In future updates, if new reports are identified, we will use the methods described in Appendix 2.
Results
Description of studies
Results of the search
We identified 925 reports that met the initial criteria for hard copy scrutiny. Five studies (nine reports) met the predetermined baseline criteria of assessing an educational intervention for toxoplasmosis prevention. Two studies (four reports) (involving 5455 women) have been included and three studies (five reports) have been excluded. SeeFigure 1 for diagram of the studies selection.
1.
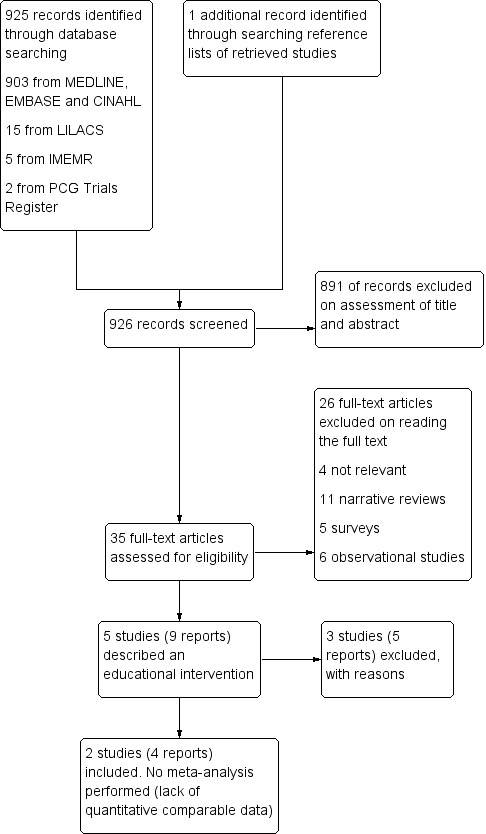
Study flow diagram.
Included studies
The first study compared two randomly allocated groups of women and was therefore included in the review (Carter 1989). The study was conducted in Ontario (Canada) and involved 432 pregnant women attending early prenatal classes in six centers. It was a cluster‐randomized trial, that is, a trial where groups rather than individuals are randomized between or among comparator interventions; in this case, the units of randomization were the prenatal classes: 26 prenatal classes were randomly assigned to the intervention and 26 prenatal classes were assigned to control intervention (usual prenatal classes). A three‐page handout was prepared along with a display poster and resource materials for teachers. Prenatal class instructors received one‐hour basic training on toxoplasmosis prevention. The intervention group women were offered a 10‐minute presentation focused on toxoplasmosis prevention during the first prenatal class. The contents of the intervention focused on cats, food, and personal hygiene.
The second study was a cluster‐randomized trial conducted in France (Wallon 2006). Women attending prenatal clinics in the area of Lyon were enrolled if they tested seronegative for toxoplasma antibodies. The unit of randomization was the city. Physicians in experimental cities provided women with a 20‐page brochure containing four pages of information of toxoplasmosis plus an audiotape containing a conversation between a physician and his/her patient on issues relevant to pregnancy, including questions on toxoplasmosis. Women attending prenatal clinics in control cities received usual care. Knowledge and behavior were assessed through a questionnaire administered at baseline (usually during the third and fourth month of gestation) and at follow‐up (after delivery). Only women who completed both questionnaires (2790 out of 5023) were included in the multivariate analysis to identify any association between the intervention and change in knowledge and behavior.
Excluded studies
The remaining three studies were surveys conducted at the population level, without a control group (before and after studies) and were, therefore, excluded from the review as they were not randomized controlled trials. The details of the three studies are shown in Table 3. One study was from Belgium (Breugelmans 2004), one from Poland (Pawlowski 2001), and one from Cuba (Molé 1992). In the Breugelmans study the intervention changed over time: the first intervention adopted (from 1983 to 1990) consisted of providing pregnant women with a list of recommended hygiene measures to avoid toxoplasma infection during pregnancy; thereafter (from 1991 to 2001) a leaflet containing information on congenital toxoplasmosis and how to avoid it was given to all pregnant women in addition to the list of recommendations and was assessed for effectiveness. Seroconvertion rate during pregnancy in the intervention period was compared with the seroconversion rate in the baseline period, when no intervention on toxoplasmosis prevention was in place (from 1979 to 1982) (Breugelmans 2004). The Pawlowski 2001 study used a multifaceted intervention including providing information to pregnant women, refresher training for health professionals, media campaigns, and the training of biology teachers in secondary schools, over six years and their effectiveness was assessed. It was impossible to obtain the full text of the study conducted in Cuba (Molé 1992): data retrieved from the abstract indicate that an educational intervention about how to avoid toxoplasmosis infection during pregnancy was delivered to seronegative pregnant women. All pregnant women were enrolled at their first prenatal visit. The number of women included in the surveys ranged from 1246 to 16,541. The Breugelmans 2004 study contained data previously published in Foulon (Foulon 1988; Foulon 1994).
1. Description of the excluded studies.
| Reference | Study design/Method | Setting/Participants | Intervention | Outcomes | Results | Notes |
| Breugelmans 2004 | Prospective survey during 3 study periods. Pre‐post study. Serological test (IgG and IgM against toxoplasma) at first prenatal visit. Seronegative women were retested every 6 weeks, in addition, a test on cord blood was done at birth. Definition of seroconversion: women initially IgG negative with subsequent IgG detection. | Belgium. Hospital setting. 23 years of observation (from 1979 to 2001). 27,827 pregnant women: 2986 pregnant women before the intervention; 8300 pregnant women after the first round of intervention; 16,541 pregnant women after the second round of intervention. | First period (from 1979 to 1982): control. Incidence of toxoplasmosis studied in 2986 consecutive pregnant women. Second period (from 1983 to 1990): first round of intervention. 8300 pregnant women received at first prenatal visit, and subsequently during monthly antenatal classes, a list of recommendations to avoid toxoplasma infection. Third period (from 1991 to 2001): second round of intervention. 16,541 pregnant women received leaflets explaining what toxoplasmosis is and what measures should be taken to avoid it in pregnancy. Reiteration of the information during antenatal classes. | Seroconversion rate during pregnancy. | I period: 1.43%. II period: 0.53%. III period: 0.09%. | In this population the incidence of toxoplasmosis in pregnancy is 8/1,000 pregnancies, and the prevalence of congenital toxoplasmosis is 2/1,000 live births. |
| Molé 1992 | Intervention study; the definition of the control group, if any, is not reported in the abstract. Serological test at first prenatal visit. Seronegative women were regularly retested. An educational intervention on how to avoid infection during pregnancy was provided to all seronegative pregnant women. |
Cuba. Hospital and ambulatory setting of 4 municipalities in Manzanillo. 2058 pregnant women. |
Prenatal education on how to avoid toxoplasmosis infection during pregnancy was delivered to seronegative pregnant women (84% of the total sample). | Seroconversion rate during pregnancy. | 1.38%. | Full text of the article not available. Data reported here have been obtained from the published abstract. |
| Pawlowski 2001 | Survey during 2 study periods. Pre‐post study. Questionnaire about knowledge of toxoplasmosis and correct behavior administered to pregnant women in 1991 to 1992 and 1997. | Poland. Hospital setting. 4311 pregnant women in the first period (1991 to 1992); 2710 women in the second period (1997). | Starting from 1991 various educational activities were implemented (training of health professionals, printed material given to pregnant women, mass media campaign, school education programs). | Good knowledge of toxoplasmosis in pregnant women. Correct behavior in pregnant women. | I period: 24.3%. II period: 45.3%. II period: 55.2%. | It is not possible to compare the change in correct behavior, measured only in the second survey in 1997. |
Risk of bias in included studies
We assessed the methodological quality of the included studies as recommended in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). See Figure 2 and Figure 3 for a summary of 'Risk of bias' assessments.
2.
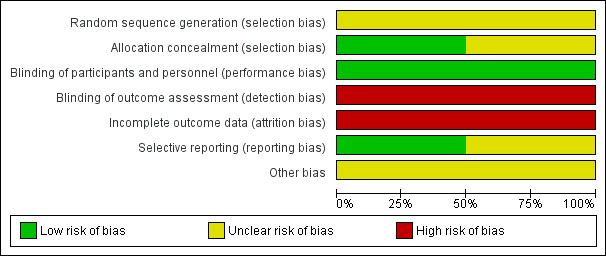
Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
The Canadian trial (Carter 1989), was a cluster‐randomized trial of low quality (Hahn 2005; Puffer 2003). The study reported that the groups were randomized but the randomizations method was not specified. Nevertheless, selection bias of the cluster is unlikely: the six centers provided almost all of the prenatal education available in that jurisdictional area; the study only lasted six months; an equal number of classes in each center received the experimental and the control intervention; authors stated that women in the experimental and control groups did not differ for demographic characteristics (even though a table reporting baseline data of the two groups was not provided), thus there is no strong reason to suspect that women attending prenatal classes in one period of time differ significantly from women attending prenatal classes in another period of time.
The second trial included (Wallon 2006) was also a cluster‐randomized trial of low quality: randomization method was not specified, therefore selection bias can not be excluded. The study, first used for a master thesis, was published in a short form as a poster, and thereafter included in a systematic review. In neither of the publications was the statistical plan of analysis described.
Blinding
In both trials, performance bias was assessed as being at low risk: in both trials (Carter 1989; Wallon 2006), blinding of the women was reported, but blinding of the personnel was not possible, but this is unlikely to influence the outcome. Outcome measures referred to changes in behavior occurring between pre‐test (usually in the first‐second term of pregnancy in Carter 1989 and usually in the third and fourth month of pregnancy in Wallon 2006) and post‐test questionnaire (in the third term of pregnancy in Carter 1989 and at delivery in Wallon 2006) in respect of hygiene measures to avoid toxoplasmosis. Since outcome measures were self‐reported, detection bias can not be totally excluded in both of the included trials, even if women were blinded to the objective of the study.
Incomplete outcome data
In Carter 1989, losses to follow‐up at the level of women participating in prenatal classes were 34% (432 women completed the pre‐test questionnaire and 285 completed the post‐test questionnaire): women in experimental group were more likely to be lost at follow‐up than women in control group, therefore attrition bias can not be excluded (Puffer 2003).
In Wallon 2006 overall losses to follow‐up were 44.5%: women in the control group were more likely to be lost to follow‐up (52% were lost) than women in intervention group (40% were lost), therefore also in the second included trial attrition bias can not be excluded (Puffer 2003).
Selective reporting
It was not clear in the two trials the extent of outcome data that were collected and therefore, unclear whether the outcomes were selectively reported. The primary outcomes were not reported in one study (Carter 1989), whereas it was reported in the more recent study (Wallon 2006).
Other potential sources of bias
Since the prenatal class instructors of the experimental and the control group in Carter 1989 were the same, it is not possible to exclude some contamination, even if such contaminations are usually not an issue in cluster trials (Torgerson 2001), and in any case, it would have acted in the sense of reducing the effect. The total sample, considering the unit of randomization, was low (26 prenatal classes in the intervention group and 26 prenatal classes in the control group). It is not clear from the paper if a statistical analysis was conducted to take into account the effect of intracluster correlation. The authors did not report raw data (number or proportion): only the P value for differences was reported. We also contacted the authors for the original data but we did not receive a reply. In Wallon 2006, the study method and statistical plan of analysis were not described in detail, therefore it is not possible to exclude other bias.
Effects of interventions
Summary of findings for the main comparison. Educational prenatal classes compared with usual prenatal classes for prenatal education for congenital toxoplasmosis.
| Educational prenatal classes compared with usual prenatal classes for prenatal education for congenital toxoplasmosis | |
| Patient or population: women of reproductive age, irrespective of their pregnant status Settings: Ontario, Canada Intervention: educational prenatal classes Comparison: usual prenatal classes | |
| Outcomes | Comments |
| Rate of congenital toxoplasmosis, defined by persistence of specific IgG antibodies beyond 11 months of age | These outcomes were either not reported or were reported in unusable formate for 'Summary of findings' tables. |
| Rate of toxoplasmosis seroconversion in pregnant women | |
| Pregnant women knowledge of risk factors for acquiring toxoplasmosis infection as objectively measured through specific questionnaire | |
| Pregnant women's awareness of the importance of avoiding toxoplasmosis during pregnancy as objectively measured through specific questionnaire | |
| Pregnant women behavior with respect to the avoidance of risk factors for toxoplasmosis infection during pregnancy as objectively measured through specific questionnaire | |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; | |
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | |
Summary of findings 2. Brochure and an audiotape compared with standard care for prenatal education for congenital toxoplasmosis.
| Brochure and an audiotape compared with standard care for prenatal education for congenital toxoplasmosis | ||||||
| Patient or population: women of reproductive age, irrespective of their pregnant status Settings: France Intervention: brochure and an audiotape Comparison: standard care | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No of participants (studies) | Quality of the evidence (GRADE) | Comments | |
| Assumed risk | Corresponding risk | |||||
| Standard care | brochure and an audiotape | |||||
| Rate of toxoplasmosis seroconversion in pregnant women | Study population | RR 1.70 (0.56 to 5.21) | 3949 (1 RCT) | ⊕⊕⊝⊝ LOW 1,2 | ||
| 3 per 1000 | 5 per 1000 (2 to 15) | |||||
| Pregnant women behavior with respect to the avoidance of risk factors for toxoplasmosis infection during pregnancy (no consumption of uncooked meat of any type) | OR 1.21 (0.98 to 1.50) | (1 RCT) | ⊕⊕⊕⊝ MODERATE 1 | Events were not available for these two outcomes. OR for these two outcomes were obtained from a review that included this study. | ||
| Pregnant women behavior with respect to the avoidance of risk factors for toxoplasmosis during pregnancy (hand washing after contact with transmission factor and before meals) | OR 1.01 (0.38 to 1.22) | (1 RCT) | ⊕⊕⊝⊝ LOW 1,2 | |||
| Rate of congenital toxoplasmosis defined by persistence of IgG antibodies beyond 11 months | None of these outcomes were reported in the included study. If in future updates data on these outcomes are available we will include them in this SOF. | |||||
| Pregnant women knowledge of risk factors for acquiring toxoplasmosis infection as objectively measured through specific questionnaire | ||||||
| Pregnant women awareness of the importance of avoiding toxoplasmosis infection during pregnancy as objectively measured through specific questionnaire | ||||||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; OR: odds ratio; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | ||||||
1Evidence from one study with high risk of bias
2Wide CI crossing the line of no effect
Comparison
1) Educational prenatal classes compared with usual prenatal classes
Primary outcomes
The study did not report any of the review pre‐specified outcomes.
Secondary outcomes
The first trial included in the review (Carter 1989), reported the following changes in behavior in the intervention group:
pet hygiene behavior: intervention classes reported to behave significantly better than the control class (P value reported < 0.05);
food hygiene behavior: intervention classes reported to behave significantly better than the control class with respect to cooking roast beef (P value reported < 0.05) and hamburgers (P value reported < 0.01); remaining items were already good at the pre‐test;
personal hygiene behavior intervention classes reported to behave significantly better than the control class only in the subgroup of women who had professional occupations (P value reported < 0.05); remaining professional groups and other items considered were already good at the pre‐test.
Only 5% of the women in the intervention group recall having obtained specific information on toxoplasmosis prevention during prenatal classes.
2) Brochure and an audiotape compared with standard care
Primary outcomes
The second trial included in the review (Wallon 2006) reported the following results:
seroconvertions for toxoplasmosis detected during the study did not differ between groups (risk ratio (RR) 1.70, 95% confidence interval (CI) 0.56 to 5.21; participants = 3949; one study) (Analysis 1.1): there were 13 cases out of 2591 pregnant women (0.5%) in the intervention group and four cases out of 1358 pregnant women (0.3%) in the control group (P = 0.35).
The data above have not been adjusted for cluster design and so it is reasonable to assume the confidence intervals have been underestimated.
1.1. Analysis.
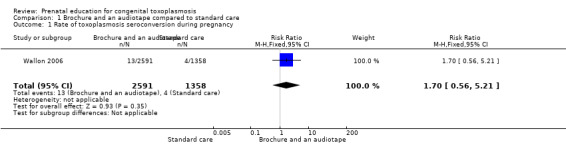
Comparison 1 Brochure and an audiotape compared to standard care, Outcome 1 Rate of toxoplasmosis seroconversion during pregnancy.
Secondary outcomes
These results were taken directly from the published report and odds ratios (OR) appear to have been adjusted for cluster design.
Prenatal education on congenital toxoplasmosis was not significantly associated to the outcome "no consumption of undercooked meat of any type" (multiple logistic regression, odds ratio (OR) 1.21; 95% CI 0.98 to 1.50).
prenatal education on congenital toxoplasmosis was not significantly associated to the outcome "handwashing after contact with transmission factor and before meals" (multiple logistic regression, OR 1.01; 95% CI 0.83 to 1.22).
baseline behaviors concerning toxoplasmosis, smoking and alcohol consumption were significantly associated with both the outcomes measured, after controlling for baseline knowledge.
Discussion
Summary of main results
Two cluster‐randomized trials on primary prevention of congenital toxoplasmosis suggest that providing specific information during antenatal classes or prenatal visits about toxoplasmosis infection and how to avoid it can improve pregnant women's behavior (Carter 1989; Wallon 2006), but the overall quality of the trials was low.
Overall completeness and applicability of evidence
Given the scarcity of evidence supporting the implementation of antenatal classes for congenital toxoplasmosis, and considering the current lack of evidence that alternative interventions such as screening and early treatment of infected pregnant women can reduce the risk of congenital toxoplasmosis, further research to quantify the impact of different educational interventions is needed.
Quality of the evidence
All prespecified outcomes for two comparisons were assessed and assigned a quality rating using the GRADE methods. For the first comparison 'educational prenatal classes compared with usual prenatal classes', outcomes were either not reported or were reported in unusable format for 'Summary of findings' tables. Evidence from one trial for the comparison of brochure and an audiotape compared with standard care for the outcome of rate of toxoplasmosis seroconversion during pregnancy was assessed as of low quality. Evidence for the outcome of pregnant women's behavior with respect to avoidance of risk factors for no consumption of uncooked meat of any type was graded to be of moderate quality, while, for hand washing after contact with transmission factor and before meals was graded to be of low quality. Downgrading decisions were based in part on the high risk of bias of included trials and imprecise results.
Potential biases in the review process
We have followed standard Cochrane methods to identify and evaluate the studies contributing to this review, and are confident that we have not missed any significant trials. We have sought missing or incomplete data, and have contacted authors where possible to clarify our interpretation of their work.
Agreements and disagreements with other studies or reviews
When the protocol for this systematic review was firstly published in 2006 no other systematic reviews on the effectiveness of prenatal education for toxoplasmosis congenital prevention were available. In 2008, a systematic review was conducted (Golloub 2008) on this topic: six studies were described (Breugelmans 2004; Carter 1989; Foulon 2000; Nguyen 2004; Pawlowski 2001; Wallon 2006), two of which were randomized controlled trials (Carter 1989; Wallon 2006), whereas the remaining trials were observational studies (Breugelmans 2004; Foulon 2000; Nguyen 2004; Pawlowski 2001). The authors did not provided a meta‐analysis of the data due to inconsistent reporting of results; their conclusions are that there is some evidence that prenatal education can reduce toxoplasmosis infection during pregnancy but that more research on this topic is needed. In addition to the studies included in the Golloub 2008 review, one more observational study suggests that prenatal education might have a positive impact on reducing congenital toxoplasmosis (Molé 1992).
Authors' conclusions
Implications for practice.
Evidence supporting prenatal education for preventing congenital toxoplasmosis is sparse and of low quality. Prenatal education could have a positive effect in terms of improving women's behavior and reducing seroconversion during pregnancy but strong evidence is still lacking. In settings where prenatal educative interventions are already in place, it could be beneficial to consider offering a specific session on how to avoid toxoplasmosis infection and provide printed materials that are informative.
A good surveillance system should be in place whenever a prenatal education activity is implemented to monitor the prevalence of seropositivity among pregnant women and to detect cases of congenital toxoplasmosis among the offspring of women who seroconverted during pregnancy.
Implications for research.
Given the limited evidence supporting prenatal education for congenital toxoplasmosis prevention, further randomized controlled trials are needed. Given the nature of the intervention, those trials would most probably be cluster‐randomized trials, that is a trial were groups rather than individuals are randomized between or among comparator interventions. The studies should focus on assessing the impact of different sets of intervention. The adequate sample size to detect a reduction of the incidence of congenital toxoplasmosis can be calculated considering at least two possible scenarios depending on the background incidence and hypothesized effect of intervention. In countries with a high incidence rate like Brazil (16/1000) (Reis 1999), and in countries with low incidence rate as in western Europe (10/10,000) (Gilbert 1999), predicting a 50% reduction of incidence in the intervention arm of the trial (Breugelmans 2004), considering a 95% confidence level and a 80% study power, and after doubling the sample to take in account the design effect of the cluster‐randomized trial, the needed sample will be (EpiInfo 6):
countries with high incidence like Brazil: 6300 women per arm;
countries with low incidence as in western Europe: 102,000 women per arm.
As results from this raw calculation, only large studies will be able to detect a difference in terms of incidence of congenital toxoplasmosis; therefore, careful consideration should be given in valuing the balance between benefits and costs of such trials. Trials assessing the effect of a multilevel prenatal educational intervention also including toxoplasmosis prevention among others, and measuring the change in behavior or better pregnancy outcome as a composite outcome, might be more practical and preferable. Finally, we also suggest that in planning such trials, a checklist for good quality reporting of cluster‐randomized trials, for example, a modified Consolidated Standards Of Reporting Trials (CONSORT) (Campbell 2004), should be followed to ensure better quality and to provide valuable information.
What's new
| Date | Event | Description |
|---|---|---|
| 31 May 2015 | New search has been performed | Search updated and no new trial reports identified. 'Summary of findings' table incorporated. |
| 31 May 2015 | New citation required but conclusions have not changed | Review updated. |
History
Protocol first published: Issue 4, 2006 Review first published: Issue 1, 2009
| Date | Event | Description |
|---|---|---|
| 15 January 2012 | New citation required but conclusions have not changed | This review has been updated. The addition of one more included study has not changed the results and conclusions. |
| 15 January 2012 | New search has been performed | Search updated. No new studies identified. One study (Wallon 2006), previously classified as pending has been included. This review now contains two included studies (involving a total of 5455 women). The methods have been updated. |
| 16 January 2008 | Amended | Converted to new review format. |
Acknowledgements
Nasreen Aflaifel for her support in the creation of the 'Summary of findings' tables for the 2015 update. Nasreen Aflaifel's work was financially supported by the UNDP/UNFPA/UNICEF/WHO/World Bank Special Programme of Research, Development and Research Training in Human Reproduction (HRP), Department of Reproductive Health and Research (RHR), World Health Organization. The named authors alone are responsible for the views expressed in this publication.
The World Health Organization and Simona Di Mario, Vittorio Basevi, Carlo Gagliotti, Daniela Spettoli, Gianfranco Gori and Roberto D'Amico retain copyright and all other rights in their respective contributions to the manuscript of this Review as submitted for publication.
We acknowledge Dr Chiara Bassi, CeVEAS, Modena, for designing and running the search strategy, and Dr Sara Balduzzi, Modena and Reggio Emilia University, for providing inputs and comments on the 2013 updated version of the review.
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure funding to Cochrane Pregnancy and Childbirth. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS or the Department of Health.
Appendices
Appendix 1. Search strategy
Search strategies written and run by authors:
PubMed (1966 to 15 January 2012):
#1. toxoplasm* #2. 'toxoplasma'/exp #3. 'toxoplasmosis'/exp #4. #1 OR #2 OR #3 #5. neonat* #6. antenat* #7. infant* #8. ('baby'/exp OR 'baby') #9. babies #10. foet* #11. fetus #12. fetal* #13. p?ediatr* #14. prenat* #15. 'infant'/exp #16. 'fetus'/exp #17. 'pediatrics'/exp #18. OR/ #5‐#17 #19. #4 AND #18 #20.'mass media'/exp OR 'mass media' #21. communication* #22. multimedia* #23. 'multi media' #24. 'multimedia'/exp #25. 'mass communication' #26. ('audiovisual equipment'/exp OR 'audiovisual equipment') #27. ('patient information'/exp OR 'patient information') #28. ('visual information'/exp OR 'visual information') #29. ('radio'/exp OR 'radio') #30. ('television'/exp OR 'television') #31. leaflet* #32. poster* #33. pamphlet* #34. 'print media' #35. 'printed media' #36. 'broadcast' #37. film* #38. ('telecommunication'/exp OR telecommunication*) #39. counsel* #40. educat* #41. empower* #42. knowledge* #43. skill* #44. 'public health'/exp #45. 'preventive medicine'/exp #46. 'preventive health service'/exp #47. 'primary health care'/exp #48. 'health care delivery'/exp #49. 'patient attitude'/exp #50. 'primary prevention'/exp #51. 'health promotion'/exp #52. 'health education'/exp #53. 'patient education'/exp #54. 'education'/exp #55. 'attitude'/exp #56. 'cognition'/exp #57. 'health behavior'/exp #58. 'decision making'/exp #59 OR/#20‐#58 #60 #19 AND #59
EMBASE (Embase.com) (1980 to 15 January 2012)
#1. toxoplasm* #2. 'toxoplasma'/exp #3. 'toxoplasmosis'/exp #4. #1 OR #2 OR #3 #5. neonat* #6. antenat* #7. infant* #8. ('baby'/exp OR 'baby') #9. babies #10. foet* #11. fetus #12. fetal* #13. p?ediatr* #14. prenat* #15. 'infant'/exp #16. 'fetus'/exp #17. 'pediatrics'/exp #18. OR/ #5‐#17 #19. #4 AND #18 #20.'mass medium'/exp OR 'mass media' #21. communication* #22. multimedia* #23. 'multi media' #24. 'mass communication' #25. ('audiovisual equipment'/exp OR 'audiovisual equipment') #26. ('patient information'/exp OR 'patient information') #27. ('visual information'/exp OR 'visual information') #28. radio #29. ('television'/exp OR 'television') #30. leaflet* #31. poster* #32. pamphlet* #33. 'print media' #34. 'printed media' #35. 'broadcast' #36. film* #37. ('telecommunication'/exp OR telecommunication*) #38. counsel* #39. educat* #40. empower* #41. knowledge* #42. skill* #43. 'public health'/exp #44. 'preventive medicine'/exp #45. 'preventive health service'/exp #46. 'primary health care'/exp #47. 'health care delivery'/exp #48. 'patient attitude'/exp #49. 'primary prevention'/exp #50. 'health promotion'/exp #51. 'health education'/exp #52. 'patient education'/exp #53. 'education'/exp #54. 'attitude'/exp #55. 'cognition'/exp #56. 'health behavior'/exp #57. 'decision making'/exp #58 OR/#20‐#57 #59 #19 AND #58
CINAHL (via EBSCO) (1982 to 15 January 2012)
#1. toxoplasm* #2. MH 'toxoplasmosis' #3. #1 OR #2 #4. neonat* #5. antenat* #6. infant* #7. baby #8. babies #9. foet* #10. fetus #11. fetal* #12. p?ediatr* #13. prenat* #14. MH 'infant' #15. MH 'fetus' #16. MH 'pediatrics' #17. OR/ #4‐#16 #18. #3 AND #17 #19. MH 'communication media' OR 'mass media' #20. communication* #21. multimedia* #22. 'multi media' #23. 'mass communication' #24. MH 'audiovisuals' OR 'audiovisual equipment' #25. 'patient information' #26. 'visual information' #27. MH 'radio' OR 'radio' #28. MH 'television' OR 'television' #29. leaflet* #30. poster* #31. pamphlet* #32. 'print media' #33. 'printed media' #34. 'broadcast' #35. film* #36. MH 'telecommunications' OR telecommunication* #37. counsel* #38. educat* #39. empower* #40. knowledge* #41. skill* #42. 'public health'/exp #43. MH 'preventive health care' #44. MH 'primary health care' #45. MH 'health care delivery' #46. MH 'patient attitudes' #47. MH 'primary prevention' #48. MH 'health services needs and demand’ #49. MH 'health education' #50. MH 'patient education' #51. MH 'education’ #52. MH 'attitude' #53. MH 'cognition' #54. MH 'health behavior' #55. MH 'decision making' #56 OR/#19‐#55 #67 #18 AND #56
LILACS (1982 to 15 January 2012) and IMEMR (1984 to 15 January 2012):
#1 congenital toxoplasmosis
Appendix 2. Methods to be used in future updates
The following methods section of this review is based on a standard template used by the Cochrane Pregnancy and Childbirth Group.
Selection of studies
Two review authors will independently assess for inclusion all the potential studies identified as a result of the search strategy. We will resolve any disagreement through discussion or, if required, we will consult the third review author.
Data extraction and management
We will design a form to extract data. For eligible studies, two review authors will extract the data using the agreed form. We will resolve discrepancies through discussion or, if required, we will consult the third review author. Data will be entered into Review Manager software (RevMan 2014) and checked for accuracy.
When information regarding any of the above was unclear, we plan to contact authors of the original reports to provide further details.
Assessment of risk of bias in included studies
Two review authors will independently assess risk of bias for each study using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011). Any disagreement will be resolved by discussion or by involving a third assessor.
(1) Random sequence generation (checking for possible selection bias)
We will describe for each included study the method used to generate the allocation sequence in sufficient detail to allow an assessment of whether it should produce comparable groups.
We will assess the method as:
low risk of bias (any truly random process, e.g. random number table; computer random number generator);
high risk of bias (any non‐random process, e.g. odd or even date of birth; hospital or clinic record number);
unclear risk of bias.
(2) Allocation concealment (checking for possible selection bias)
We will describe for each included study the method used to conceal allocation to interventions prior to assignment and assess whether intervention allocation could have been foreseen in advance of, or during recruitment, or changed after assignment.
We will assess the methods as:
low risk of bias (e.g. telephone or central randomization; consecutively numbered sealed opaque envelopes);
high risk of bias (open random allocation; unsealed or non‐opaque envelopes, alternation; date of birth);
unclear risk of bias.
(3.1) Blinding of participants and personnel (checking for possible performance bias)
We will describe for each included study the methods used, if any, to blind study participants and personnel from knowledge of which intervention a participant received. We will consider that studies are at low risk of bias if they were blinded, or if we judged that the lack of blinding unlikely to affect results. We will assess blinding separately for different outcomes or classes of outcomes.
We will assess the methods as:
low, high or unclear risk of bias for participants;
low, high or unclear risk of bias for personnel.
(3.2) Blinding of outcome assessment (checking for possible detection bias)
We will describe for each included study the methods used, if any, to blind outcome assessors from knowledge of which intervention a participant received. We will assess blinding separately for different outcomes or classes of outcomes.
We will assess methods used to blind outcome assessment as:
low, high or unclear risk of bias.
(4) Incomplete outcome data (checking for possible attrition bias due to the amount, nature and handling of incomplete outcome data)
We will describe for each included study, and for each outcome or class of outcomes, the completeness of data including attrition and exclusions from the analysis. We will state whether attrition and exclusions were reported and the numbers included in the analysis at each stage (compared with the total randomized participants), reasons for attrition or exclusion where reported, and whether missing data are balanced across groups or are related to outcomes. Where sufficient information is reported, or could be supplied by the trial authors, we plan to re‐include missing data in the analyses which we undertake.
We will assess methods as:
low risk of bias (e.g. no missing outcome data; missing outcome data balanced across groups);
high risk of bias (e.g. numbers or reasons for missing data imbalanced across groups; ‘as treated’ analysis done with substantial departure of intervention received from that assigned at randomization);
unclear risk of bias.
(5) Selective reporting (checking for reporting bias)
We will describe for each included study how we investigated the possibility of selective outcome reporting bias and what we found.
We will assess the methods as:
low risk of bias (where it is clear that all of the study’s pre‐specified outcomes and all expected outcomes of interest to the review have been reported);
high risk of bias (where not all the study’s pre‐specified outcomes have been reported; one or more reported primary outcomes were not pre‐specified; outcomes of interest are reported incompletely and so cannot be used; study fails to include results of a key outcome that would have been expected to have been reported);
unclear risk of bias.
(6) Other bias (checking for bias due to problems not covered by (1) to (5) above)
We will describe for each included study any important concerns we had about other possible sources of bias.
(7) Overall risk of bias
We will make explicit judgements about whether studies were at high risk of bias, according to the criteria given in the Handbook (Higgins 2011). With reference to (1) to (6) above, we plan to assess the likely magnitude and direction of the bias and whether we consider it is likely to impact on the findings. We will explore the impact of the level of bias through undertaking sensitivity analyses ‐ see Sensitivity analysis.
Measures of treatment effect
Dichotomous data
For dichotomous data, we will present results as summary risk ratio with 95% confidence intervals.
Continuous data
We will use the mean difference if outcomes were measured in the same way between trials. We will use the standardised mean difference to combine trials that measured the same outcome, but used different methods.
Unit of analysis issues
Cluster‐randomized trials
If we identify any cluster‐randomized trials, we will include them in the analyses along with individually‐randomized trials. We will adjust their sample sizes using the methods described in the Cochrane Handbook for Systematic Reviews of Intervention [Section 16.3.4] using an estimate of the intracluster correlation co‐efficient (ICC) derived from similar trial or from a study of a similar population. We will also conduct sensitivity analyses to investigate the effect of variation in the ICC. If we identify both cluster‐randomized trials and individually‐randomized trials, we plan to synthesize the relevant information. We will consider it reasonable to combine the results from both if there is little heterogeneity between the study designs and the interaction between the effect of intervention and the choice of randomization unit is considered to be unlikely.
We will also acknowledge heterogeneity in the randomization unit and perform a subgroup analysis to investigate the effects of the randomization unit.
Cross‐over trials
Other unit of analysis issues
It is unlikely that cross‐over designs will be a valid study design for Pregnancy and Childbirth reviews and so, if identified, we will exclude them.
Dealing with missing data
For included studies, we will note levels of attrition. If more eligible studies are included, we will explore the impact of including studies with high levels of missing data in the overall assessment of treatment effect by using sensitivity analysis.
For all outcomes, we will carry out analyses, as far as possible, on an intention‐to‐treat basis, i.e. we will attempt to include all participants randomized to each group in the analyses. The denominator for each outcome in each trial will be the number randomized minus any participants whose outcomes are known to be missing.
Assessment of heterogeneity
We will assess statistical heterogeneity in each meta‐analysis using the Tau², I² and Chi² statistics. We will regard heterogeneity as substantial if an I² is greater than 30% and either the Tau² is greater than zero, or there is a low P value (less than 0.10) in the Chi² test for heterogeneity. If we identify substantial heterogeneity (above 30%), we plan to explore it by pre‐specified subgroup analysis.
Assessment of reporting biases
If there are 10 or more studies in the meta‐analysis, we will investigate reporting biases (such as publication bias) using funnel plots. We will assess funnel plot asymmetry visually. If asymmetry is suggested by a visual assessment, we will perform exploratory analyses to investigate it.
Data synthesis
We will carry out statistical analysis using the Review Manager software (RevMan 2014). We will use fixed‐effect meta‐analysis for combining data where it is reasonable to assume that studies are estimating the same underlying treatment effect, i.e. where trials are examining the same intervention, and the trials’ populations and methods are judged sufficiently similar.
If there is clinical heterogeneity sufficient to expect that the underlying treatment effects differ between trials, or if substantial statistical heterogeneity is detected, we will use random‐effects meta‐analysis to produce an overall summary, if an average treatment effect across trials is considered clinically meaningful. The random‐effects summary will be treated as the average range of possible treatment effects and we will discuss the clinical implications of treatment effects differing between trials. If the average treatment effect is not clinically meaningful, we will not combine trials. If we use random‐effects analyses, the results will be presented as the average treatment effect with 95% confidence intervals, and the estimates of Tau² and I².
Subgroup analysis and investigation of heterogeneity
If we identify substantial heterogeneity, we will investigate it using subgroup analyses and sensitivity analyses. We will consider whether an overall summary was meaningful, and if it is, we will use random‐effects analysis to produce it.
We plan to carry out all or part of the following subgroup analyses depending on the characteristics of the studies that we will identify:
background risk of infection (studies conducted in area with low incidence rate of congenital toxoplasmosis and studies conducted in areas with high incidence rate of congenital toxoplasmosis);
health professionals providing the education (counselors, gynecologists, midwives, etc);
information strategy used (printed materials, face‐to‐face information, class groups, videotapes, etc).
Sugroup analyses will be restricted to primary outcomes.
We will assess subgroup differences by interaction tests available within RevMan (RevMan 2014). We will report the results of subgroup analyses quoting the Chi² statistic and P value, and the interaction test I² value.
Sensitivity analysis
We plan to carry out sensitivity analyses to explore the effect of trial quality assessed by concealment of allocation, high attrition rates, or both, with poor quality studies being excluded from the analyses in order to assess whether this makes any difference to the overall result.
Data and analyses
Comparison 1. Brochure and an audiotape compared to standard care.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Rate of toxoplasmosis seroconversion during pregnancy | 1 | 3949 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.70 [0.56, 5.21] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Carter 1989.
| Methods | Cluster‐randomized trial. Unit of randomization was the antenatal class. | |
| Participants | 432 pregnant women attending 52 prenatal classes offered for free by a public health agency in Ontario. Completed both the pre‐test and the post‐test questionnaire 285 women (122 in the experimental arm and 163 in the control arm). Loss to follow‐up was 34%. | |
| Interventions | 10‐minute teaching session during the first prenatal class on how to avoid toxoplasmosis infection. | |
| Outcomes | Pet hygiene behavior of cat owners. Food hygiene behaviors. Personal hygiene behaviors. | |
| Notes | A behavior change scoring system was developed: a woman gained 1 point for every change toward a useful behavior, while she lost 1 point for every change toward a dangerous behavior. The authors did not report raw data (number or proportion): only the significance test data (P value) for differences were reported. We have contacted the authors for the original data but have not yet received a reply. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not described in the publication. |
| Allocation concealment (selection bias) | Low risk | Not described in the publication. However, as all the centers present in that jurisdictional area were involved, an equal number of classes in each center received the experimental and the control intervention, and this is a cluster‐randomized trial, thus the units of randomization are the prenatal groups, the lack of allocation concealment is unlikely to influence the outcome. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Blinding of the women reported, blinding of the personnel was not possible, but unlikely to influence the outcome. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Measure of knowledge and behavior were self‐reported. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Losses to follow‐up were 34% (432 women completed the pretest questionnaire and 285 completed the post‐test questionnaire): women in experimental group were more likely to be lost at follow‐up than women in control group. |
| Selective reporting (reporting bias) | Unclear risk | All of the pre‐specified outcomes have been reported. However, the study did not report seroconversion rate during pregnancy, which is an outcome of interest. |
| Other bias | Unclear risk | Contamination between groups can not be excluded. |
Wallon 2006.
| Methods | Cluster‐randomized trial. Units of randomization were prenatal clinics. | |
| Participants | 5023 seronegative pregnant women attending prenatal clinics in 7 counties in the area of Lyon (France). Completed both the pre‐test and the post‐test questionnaire 2790 women (1953 in the experimental arm and 837 in the control arm). Loss to follow‐up was 44.5%. | |
| Interventions | Physicians in experimental cities were instructed to give their patients: ‐ a 20‐page brochure containing relevant information about different aspects of pregnancy, including 4 pages on toxoplasmosis prevention and transmission; ‐ an audiotape containing frequently asked questions during pregnancy, including questions on toxoplasmosis. |
|
| Outcomes | Knowledge and behavior change including: ‐ consumption of undercooked meat of any type; ‐ handwashing after contact with transmission factors (soil, raw meat, unwashed vegetables) and before meals. Seroconvertion for toxoplasmosis. |
|
| Notes | Knowledge and behavior was measured through a questionnaire at baseline (during the third and fourth months of pregnancy) and at follow‐up (at delivery). There was a large and differential loss‐to‐follow‐up: completed the follow‐up questionnaire and were therefore included in the analysis 60% of the women enrolled in the experimental group and 48% of the women enrolled in the control group. A multivariate analysis was conducted but it was not specified if the statistical plan of analysis considered to correct for cluster design. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not described in the publication. |
| Allocation concealment (selection bias) | Unclear risk | Not described in the publication. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Blinding of the women reported, blinding of the personnel was not possible, but unlikely to influence the outcome as the outcome assessed by the personnel was seroconversion rate. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Measure of knowledge and behavior were self‐reported. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Overall losses to follow‐up were 44.5%: women in the control group were more likely to be lost at follow‐up (52% were lost) than women in intervention group (40% were lost). |
| Selective reporting (reporting bias) | Low risk | All of the study’s pre‐specified outcomes and all expected outcomes of interest to the review (including seroconversion rate) have been reported. |
| Other bias | Unclear risk | Study method and statistical plan of analysis were not described in details, therefore it is not possible to exclude other bias. |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Breugelmans 2004 | Not randomized. For details of study, seeTable 3. |
| Molé 1992 | Not randomized. For details of study, seeTable 3. |
| Pawlowski 2001 | Not randomized. For details of study, seeTable 3. |
Differences between protocol and review
The methods have been updated to reflect the latest Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011) and the current standard methods text of Cochrane Pregnancy and Childbirth Group. Two 'Summary of findings' tables have been incorporated.
Contributions of authors
First version of the review: Simona Di Mario (SDM), Vittorio Basevi (VB) and Daniela Spettoli (DS) were responsible for the conception of the study. SDM, DS, Carlo Gagliotti (CG) and Gianfranco Gori selected the studies and assessed the quality. SDM, DS and CG collected and analyzed the data. VB, Nicola Magrini and Roberto D'Amico (RDA) provided input for writing the protocol and review. All authors provided comments on earlier drafts, revised and approved the initial version of the review. SDM, CG and RDA assessed the new studies for inclusion and performed the data extraction. SDM drafted the changes to the text. All authors provided comments, revised and approved the updated version of the review.
For the 2015 update, Simona Di Mario (SDM) rearranged the text of the review according to new standard suggested from the Cochrane Pregnancy and Childbirth Group, adding new paragraphs and additional other references; Vittorio Basevi (VB), Daniela Spettoli (DS) and Carlo Gagliotti (CG) revised the text, Gianfranco Gori (GG), Nicola Magrini (NM) and Roberto D'Amico (RDA) read and approved the final version of the review.
Sources of support
Internal sources
SaPeRiDoc, Direzione generale sanità e politiche sociali, Regione Emilia‐Romagna, Bologna, Italy.
External sources
UNDP/UNFPA/UNICEF/WHO/World Bank Special Programme of Research, Development and Research Training in Human Reproduction (HRP), Department of Reproductive Health and Research (RHR), World Health Organization, Switzerland.
Declarations of interest
None known.
New search for studies and content updated (no change to conclusions)
References
References to studies included in this review
Carter 1989 {published data only}
- Carter AO, Gelmon SB, Wells GA, Toepell AP. The effectiveness of a prenatal education programme for the prevention of congenital toxoplasmosis. Epidemiology and Infection 1989;103:539‐45. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wallon 2006 {published data only}
- Gollub EL, Leroy V, Gilbert R, Chêne G, Wallon M, the European Toxoprevention Study Group (EUROTOXO). Effectiveness of health education on Toxoplasma‐related knowledge, behaviour, and risk of seroconversion in pregnancy. European Journal of Obstetric & Gynecology and Reproductive Biology 2008;136:137‐45. [DOI] [PubMed] [Google Scholar]
- Nguyen DT. Evolution des connaissances et des comportements au cours d'un programme d'éducation prénatal pour la prévention primaire de la toxoplasmose congénitale chez les femmes enceintes séronégatif pour la toxoplasmose. Règion de Lyon, 1994‐1995 [Impact on knowledge and behaviour of a prenatal education programme to prevent congenital toxoplasmosis among seronegative pregnant women. Lyon region, 1994‐1995] [Masters thesis]. Bordeaux: University of Bordeaux, Juillet 2004. [Google Scholar]
- Wallon M, Nguyen Hoang Hanh DT, Peyron F, Chene G. Impact of health education for the primary prevention of toxoplasma infection in pregnancy: lessons from the ERIS study. 16th European Congress of Clinical Microbiology and Infectious Diseases (ECCMID); 2006 April 1‐4; Nice, France. 2006:Abstract no: P876.
References to studies excluded from this review
Breugelmans 2004 {published data only}
- Breugelmans M, Naessens A, Foulon W. Prevention of toxoplasmosis during pregnancy ‐ an epidemiologic survey over 22 consecutive years. Journal of Perinatal Medicine 2004;32:211‐4. [DOI] [PubMed] [Google Scholar]
- Foulon W, Naessens A, Derde MP. Evaluation of the possibilities for preventing congenital toxoplasmosis. American Journal of Perinatology 1994;11:57‐62. [DOI] [PubMed] [Google Scholar]
- Foulon W, Naessens A, Lauwers S, Meuter F, Amy JJ. Impact of primary prevention on the incidence of toxoplasmosis during pregnancy. Obstetrics & Gynaecology 1988;72:363‐6. [PubMed] [Google Scholar]
Molé 1992 {published data only}
- Molé JR, Suárez D, Alonso F, Piquera G. This is what we did to prevent congenital toxoplasmosis in Manzanillo [Lo que hacemos para prevenir la toxoplasmosis congénita en Manzanillo]. Revista Cubana de Medicina General Integral 1992;8(4):330‐2. [Google Scholar]
Pawlowski 2001 {published data only}
- Pawlowski ZS, Gromadecka‐Sutkiewicz M, Skommer J, Paul M, Rokossowski H, Suchocka E, et al. Impact of health education on knowledge and prevention behavior for congenital tooplasmosis: the experience in Poznan, Poland. Health Education Research 2001;16(4):493‐502. [DOI] [PubMed] [Google Scholar]
Additional references
Alfirevic 2003
- Alfirevic Z, Sundberg K, Brigham S. Amniocentesis and chorionic villus sampling for prenatal diagnosis. Cochrane Database of Systematic Reviews 2003, Issue 3. [DOI: 10.1002/14651858.CD003252] [DOI] [PMC free article] [PubMed] [Google Scholar]
Allain 1998
- Allain JP, Palmer CR, Pearson G. Epidemiological study of latent and recent infection by toxoplasma gondii in pregnant women from a regional population in the UK. Journal of Infection 1998;36:189‐96. [DOI] [PubMed] [Google Scholar]
Bader 1997
- Bader TJ, Macones GA, Asch DA. Prenatal screening for toxoplasmosis. Obstetrics & Gynecology 1997;90:457‐64. [DOI] [PubMed] [Google Scholar]
Boyer 2005
- Boyer KM, Holfels E, Roizen N, Swisher C, Mack D, Remington J, et al. Risk factors for toxoplasma gondii infection in mothers of infants with congenital toxoplasmosis: implications for prenatal management and screening. American Journal of Obstetrics and Gynecology 2005;192:564‐71. [DOI] [PubMed] [Google Scholar]
Campbell 2004
- Campbell MK, Elbourne DR, Altman DG for the CONSORT Group. CONSORT statement: extension to cluster randomised trials. BMJ 2004;328:702‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Chabbert 2004
- Chabbert E, Lachaud L, Crobu L, Bastien P. Comparison of two widely used PCR primer systems for detection of toxoplasma in amniotic fluid, blood, and tissues. Clinical Microbiology 2004;42:1719‐22. [DOI] [PMC free article] [PubMed] [Google Scholar]
Chamberlain 2013
- Chamberlain C, O’Mara‐Eves A, Oliver S, Caird JR, Perlen SM, Eades SJ, et al. Psychosocial interventions for supporting women to stop smoking in pregnancy. Cochrane Database of Systematic Reviews 2013, Issue 10. [DOI: 10.1002/14651858.CD001055.pub4] [DOI] [PMC free article] [PubMed] [Google Scholar]
Cook 2000
- Cook AJ, Gilbert RE, Buffolano W, Zufferey J, Petersen E, Jenum PA, et al. Sources of toxoplasma infection in pregnant women: European multicentre case‐control study. European Research Network on Congenital Toxoplasmosis. BMJ 2000;321:142‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Dunn 1999
- Dunn D. Mother to child transmission of toxoplasmosis: risks estimates or clinical counselling. Lancet 1999;353:1829‐33. [DOI] [PubMed] [Google Scholar]
EpiInfo 6 [Computer program]
- Dean AG, Dean JA, Coulombier D, Brendel KA, Smith DC, Burton AH, et al. EpiInfo, Version 6: a word processing, database, and statistics programs for epidemiology on microcomputers. Atlanta: Centers for Disease Control and Prevention, 1994.
Ferguson 2011
- Ferguson W, Mayne PD, Cafferkey M, Butler K. Lack of awareness of risk factors for primary toxoplasmosis in pregnancy. Irish Journal of Medical Science 2011;180:807‐11. [DOI] [PubMed] [Google Scholar]
Foulon 1988
- Foulon W, Naessens A, Lauwers S, Meuter F, Amy JJ. Impact of primary prevention on the incidence of toxoplasmosis during pregnancy. Obstetrics & Gynecology 1988;72:363‐6. [PubMed] [Google Scholar]
Foulon 1994
- Foulon W, Naessens A, Derde MP. Evaluation of the possibilities for preventing congenital toxoplasmosis. American Journal of Perinatology 1994;11:57‐62. [DOI] [PubMed] [Google Scholar]
Foulon 1999a
- Foulon W, Villena I, Stray‐Pederson B, Decoster A, Lappalainen M, Pinon JM, et al. Treatment of toxoplasmosis during pregnancy: a multicenter study of impact on fetal transmission and children sequelae at age 1 year. American Journal of Obstetrics and Gynecology 1999;180:410‐5. [DOI] [PubMed] [Google Scholar]
Foulon 1999b
- Foulon W, Pinon JM, Stray‐Pederson B, Pollak A, Lappalainen M, Decoster A, et al. Prenatal diagnosis of congenital toxoplasmosis: a multicenter evaluation of different diagnostic parameters. American Journal of Obstetrics and Gynecology 1999;181:843‐7. [DOI] [PubMed] [Google Scholar]
Foulon 2000
- Foulon W, Naessens A, Ho‐Yen D. Prevention of congenital toxoplasmosis. Journal of Perinatal Medicine 2000;28:337‐45. [DOI] [PubMed] [Google Scholar]
Gilbert 1999
- Gilbert RE. Epidemiology of infection in pregnant women. In: Petersen E, Amboise‐Thomas P editor(s). Congenital Toxoplasmosis: Scientific Background, Clinical Management and Control. Paris: Springer‐Verlag France, 1999. [Google Scholar]
Gilbert 2001
- Gilbert R, Dunn D, Wallon M, Hayde M, Prusa A, Lebech M, et al. Ecological comparison of the risks of mother‐to‐child transmission and clinical manifestations of congenital toxoplasmosis according to prenatal treatment protocol. Epidemiology and Infection 2001;127:113‐20. [DOI] [PMC free article] [PubMed] [Google Scholar]
Gilbert 2002
- Gilbert RE, Peckham CS. Congenital toxoplasmosis in the United Kingdom: to screen or not to screen?. Journal of Medical Screening 2002;9:135‐41. [DOI] [PubMed] [Google Scholar]
Gilbert 2003
- Gilbert R, Gras L. European multicentre study on congenital toxoplasmosis. Effect of timing and type of treatment on the risk of mother to child transmission of Toxoplasma gondii. BJOG: an international journal of obstetrics and gynaecology 2003;110:112‐20. [DOI] [PubMed] [Google Scholar]
Gilbert 2007
- Gilbert RE, Thalib L, Tan HK, Paul M, Wallon M, Petersen E. Screening for congenital toxoplasmosis: accuracy of immunoglobulin M and immunoglobulin A tests after birth. Journal of Medical Screening 2007;14:8‐13. [DOI] [PubMed] [Google Scholar]
Golloub 2008
- Gollub EL, Leroy V, Gilbert R, Chêne G, Wallon M, European ToxopreventionStudy Group (EUROTOXO). Effectiveness of health education on Toxoplasma‐relatedknowledge, behaviour, and risk of seroconversion in pregnancy. European Journal of Obstetrics, Gynecology, and Reproductive Biology 2008;136:137‐45. [DOI] [PubMed] [Google Scholar]
Hahn 2005
- Hahn S, Puffer S, Torgerson DJ, Watson J. Methodological bias in cluster randomised trials. BMC Medical Research Methodology 2005;5:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Higgins JPT, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. The Cochrane Collaboration, 2011. Available from www.cochrane‐handbook.org.
Lebech 1996
- Lebech M, Joynson DH, Seitz HM, Thulliez P, Gilbert RE, Dutton GN, et al. Classification system and case definitions of toxoplasma gondii infection in immunocompetent pregnant women and their congenitally infected offspring. European Research Network on Congenital Toxoplasmosis. European Journal of Clinical Microbiology and Infectious Diseases 1996;15:799‐805. [DOI] [PubMed] [Google Scholar]
Lebech 1999
- Lebech M, Andersen O, Christensen NC, Hertel J, Nielsen HE, Petersen B, et al. Feasibility of neonatal screening for toxoplasma infection in the absence of prenatal treatment. Danish Congenital Toxoplasmosis Study Group. Lancet 1999;353:1834‐7. [DOI] [PubMed] [Google Scholar]
Liesenfeld 1997
- Liesenfeld O, Press C, Montoya JG, Gill R, Isaac‐Renton JL, Hedman K, et al. False‐positive results in immunoglobulin M (IgM) toxoplasma antibody tests and importance of confirmatory testing: the Platelia toxo IgM test. Journal of Clinical Microbiology 1997;35:174‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Montoya 2004
- Montoya JG, Liesenfeld O. Toxoplasmosis. Lancet 2004;363:1965‐76. [DOI] [PubMed] [Google Scholar]
Nguyen 2004
- Nguyen DT. Evolution of knowledge and behavior during a prenatal education program for primary prevention of congenital toxoplasmosis in pregnant women seronegative for toxoplasmosis [Evolution des connaissances et des comportements au cours d’un programme d’éducation prénatal pour la prévention primaire de la toxoplasmose congénitale chez les femmes enceintes séronégatif pour la toxoplasmose]. Région de Lyon, 1994–1995. Juillet 2004.
NSC 2001
- National Screening Committee Working Group. Antenatal and Newborn Screening for Toxoplasmosis. Report of the Working Group ‐‐ October 2001. National Screening Committee, 2001. [Google Scholar]
Peyron 1999
- Peyron F, Wallon M, Liou C, Garner P. Treatments for toxoplasmosis in pregnancy. Cochrane Database of Systematic Reviews 1999, Issue 3. [DOI: 10.1002/14651858.CD001684] [DOI] [PMC free article] [PubMed] [Google Scholar]
Puffer 2003
- Puffer S, Torgerson D, Watson J. Evidence for risk of bias in cluster randomised trials: review of recent trials published in three general medical journals. BMJ 2003;327:785‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Reis 1999
- Reis R, Lösch R, Lago EG. Primary prevention of congenital toxoplsmosis [Prevenção primária da toxoplasmose congênita]. Acta Médica 1999;20:704‐20. [Google Scholar]
RevMan 2014 [Computer program]
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Rorman 2006
- Rorman E, Stein Zamir C, Rilkis I, Ben‐David H. Congenital toxoplasmosis ‐ prenatal aspects of toxoplasma gondii infection. Reproductive Toxicology 2006;21(4):458‐72. [DOI] [PubMed] [Google Scholar]
Schmidt 2006
- Schmidt DR, Hogh B, Andersen O, Fuchs J, Fledelius H, Petersen E. The national neonatal screening programme for congenital toxoplasmosis in Denmark: results from the initial four years, 1999‐2002. Archives of Disease in Childhood 2006;91:661‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Signorell 2006
- Signorell LM, Seitz D, Merkel S, Berger R, Rudin C. Cord blood screening for congenital toxoplasmosis in northwestern Switzerland, 1982‐1999. Pediatric Infectious Disease Journal 2006;25:123‐8. [DOI] [PubMed] [Google Scholar]
SYROCOT 2007
- Thiébaut R, Leproust S, Chêne G, Gilbert R. Effectiveness of prenatal treatment for congenital toxoplasmosis: a meta‐analysis of individual patients' data. Lancet 2007;369:115‐22. [DOI] [PubMed] [Google Scholar]
Thalib 2005
- Thalib L, Gras L, Romand S, Prusa A, Bessieres MH, Petersen E, et al. Prediction of congenital toxoplasmosis by polymerase chain reaction analysis of amniotic fluid. BJOG: an international journal of obstetrics and gynaecology 2005;112:567‐74. [DOI] [PubMed] [Google Scholar]
Torgerson 2001
- Torgerson DJ. Contamination in trials: is cluster randomisation the answer?. BMJ 2001;322:355‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wilson 1980
- Wilson CB, Remington JS, Stagno S, Reynolds DW. Development of adverse sequelae in children born with subclinical congenital Toxoplasma infection. Pediatrics 1980;66:767‐74. [PubMed] [Google Scholar]
References to other published versions of this review
Di Mario 2006
- Mario S, Basevi V, Gagliotti C, Spettoli D, Gori G, D'Amico R, et al. Prenatal education for congenital toxoplasmosis. Cochrane Database of Systematic Reviews 2006, Issue 4. [DOI: 10.1002/14651858.CD006171] [DOI] [Google Scholar]
Di Mario 2009
- Mario S, Basevi V, Gagliotti C, Spettoli D, Gori G, D'Amico R, et al. Prenatal education for congenital toxoplasmosis. Cochrane Database of Systematic Reviews 2009, Issue 1. [DOI: 10.1002/14651858.CD006171.pub2] [DOI] [PubMed] [Google Scholar]
Di Mario 2013
- Mario S, Basevi V, Gagliotti C, Spettoli D, Gori G, D'Amico R, Magrini N. Prenatal education for congenital toxoplasmosis. Cochrane Database of Systematic Reviews 2013, Issue 2. [DOI: 10.1002/14651858.CD006171.pub3] [DOI] [PubMed] [Google Scholar]


