Abstract
Cancers of the upper gastrointestinal tract are a leading cause of cancer-related death world-wide and historically have a poor prognosis. The incidence and histology of these cancers have varied temporally and geographically over the last three decades, with an emerging understanding of the differences in the molecular and genetic profiles across different subgroups. Management of oesophagogastric cancers is by a multidisciplinary team with utilisation of surgery, radiotherapy and systemic treatments in combinations where appropriate. Immune checkpoint inhibition (ICI) has drastically changed the treatment landscape of multiple solid malignancies in the last 5 years. In oesophagogastric cancer, clinical trials have only recently shown activity that is often associated with the molecular characteristics of these tumours, in particular PD-L1 scores or microsatellite instability (MSI-H). This review looks to present the pivotal trials in this space, discuss the complexities between trials that may explain the disparate results and assess the benefit ICI offers in the treatment landscape at present.
Subject terms: Cancer immunotherapy, Gastric cancer, Oesophageal cancer
Introduction
Oesophagogastric cancers, originating in the stomach, oesophagus and gastro-oesophageal junction (GOJ), have a poor prognosis and despite similarities in how they are treated, this group of malignancies shows significant heterogeneity, with geographical and temporal variations in both histology and incidence [1]. Globally, gastric and oesophageal cancer represents the third and sixth leading causes of cancer-related death, respectively [2].
In previous decades, treatment of all oesophagogastric malignancies has centred around various combinations of three main chemotherapy agents: platinum compounds, fluoropyrimidines and taxanes. At present, recommended first-line systemic treatment for the metastatic disease by international consensus groups is a combination of fluoropyridine and platinum chemotherapy. For metastatic gastric cancer patients, this yields a median overall survival (mOS) of only 6–12 months [3]. Even the management of localised oesophageal cancer, involving multimodality perioperative treatment and surgery, achieves a five-year survival of less than 50% [4]. Human epidermal growth factor receptor 2 (HER2) positivity has been the only established druggable target over the last decade. Reports vary regarding the prognostic value of HER2 overexpression on survival; however, the use of HER2-directed trastuzumab in combination with chemotherapy has improved survival outcomes in gastric and gastro-oesophageal cancer patients [5]. These outcomes highlight the need for a deepened understanding of the heterogeneity of these diseases and a novel, personalised treatment approach.
Whilst targeted therapies and chemotherapy predominantly act directly on cancer cells, more recent trials have focused on Immune Checkpoint Inhibitors (ICI). ICI’s achieve cancer control by enhancing anti-tumour immune responses. Therapeutic monoclonal antibodies (mAb’s) disrupt co-inhibitory T cell signals (immune checkpoints), such as programmed death-1 (PD-1), programmed death ligand 1 (PD-L1) and cytotoxic T lymphocyte-associated antigen-4 (CTLA-4). Immunotherapy mAb’s targeting these checkpoints have achieved deep and durable tumour responses in melanoma [6], non-small cell lung cancer [7] and other solid tumours. The high tumour mutational burden of esophagogastric cancers, and their development from chronic inflammatory conditions, has led to an interest in how ICI might diversify the treatment algorithm for oesophagogastric cancers. This review looks to expound on the clinical trials done in this space and to analyse the complexities that may explain the divergent responses seen across trials.
Immunotherapy in the first-line metastatic setting
Despite the geographical variation and difference in risk factors and aetiology, prior to recent years, the treatment of all advanced esophagogastric cancer comprised of a few different chemotherapy regimens, with histological subtype [Adenocarcinoma (AC) and Squamous Cell Carcinoma (SCC)] not appearing to influence the observed response rates or survival [8]. In the last decade, however, The Cancer Genome Atlas (TCGA) Research Network’s genomic profiling has demonstrated marked differences between histological subtypes. Oesophageal SCC (ESCC) harbour distinctly different genomic amplifications and a molecular profile mirroring that of SCC from other primaries [9], while oesophageal AC (EAC) are very similar to chromosomally unstable gastric AC, one of the four molecular classified subtypes of gastric cancer [10]. Since the first trials looking at ICI in esophagogastric cancers, an exaggerated response in SCC compared to AC has been observed when treated with anti-programmed cell death-1 (PD-1) mAb’s. This indicates a difference in the histology’s immunogenicity which is likely explained by these distinct molecular and genomic profiles.
Squamous cell carcinoma
Until recently, the only trial in the first-line metastatic setting that enrolled SCC was KEYNOTE 590, a large international phase 3 trial randomising 749 patients with unresectable/metastatic ESCC, EAC or GOJ (Siewert type 1) AC to either pembrolizumab (200 mg 3 weekly) or placebo, plus 5-fluorouracil (5FU) 800 mg/m2 Day 1–5 and cisplatin 80 mg/m2 day 1 every 3 weeks. The interim analysis was presented at ESMO 2020 after 13 months follow-up. The baseline characteristics were predominantly patients from Asian countries (52%), ESCC (73.5%) and an even proportion of PD-L1 positive (defined by CPS ≥ 10, see Fig. 1 for CPS definition) and negative. The primary end-point of OS in ESCC with CPS≥ 10 was superior with pembrolizumab at 13.9 compared to 8.8 months with placebo (HR 0.57, p < 0.001) and in the total ESCC population; 12.6 and 9.8 months, respectively (HR 0.72, p = 0.0006) [11]. The above two positive results allowed for OS in all CPS ≥ 10 to be assessed, demonstrating mOS of 13.5 months with pembrolizumab compared to 9.4 months with placebo (HR 0.62, p < 0.0001). Median PFS (mPFS) also favoured pembrolizumab in all subgroups. Pembrolizumab plus chemotherapy had an ORR of 45% compared to only 29% in the placebo arm (p < 0.0001), the duration of response (DOR) was also longer at 8.3 compared to 6.0 months. With respect to safety, grade 3 or higher treatment-related adverse events (TRAE) were similar between groups. The benefits across OS and PFS were seen irrespective of histology, CPS ≥10 or <10 and country of enrolment (Asian or non-Asian country), although the benefit seemed to be greater for ESCC, CPS ≥10 and patients enrolled in Asian countries. This trial gives the impression that pembrolizumab plus chemotherapy may be beneficial for EAC or Siewert type 1 GOJ AC were CPS ≥ 10; however, the results are limited by small patient numbers and this specific patient subset analysis is yet to be published. CHECKMATE 648 and ESCORT-1st were subsequently presented at the ASCO 2021 annual scientific meeting and are two large phase 3 trials further adding to this body of evidence. CHECKMATE 648 took 970 treatment-naive unresectable/advanced metastatic PD-L1 unselected ESCC and randomised them 1:1:1 to; chemotherapy alone (5FU plus cisplatin), chemotherapy plus nivolumab or combination ICI [nivolumab 3 mg/kg Q2W plus ipilimumab 1 mg/kg Q6W(NIVO3 + IPI1)]. The trial design looked at dual primary end-points of OS and PFS in PD-L1 > 1% (TPS, see Fig. 1 for definition) for nivolumab plus chemotherapy or combination ICI, versus chemotherapy alone. The baseline characteristics across groups were well balanced; PD-L1≥1% (~50%) with predominantly male (>75%), Asian (70%) and de novo metastatic disease (~60%) [12]. At a minimum follow-up of 12.9 months, for the comparison of nivolumab plus chemotherapy versus chemotherapy alone, all end-points [OS, PFS, overall response rate (ORR) and duration of response (DOR)] favoured nivolumab plus chemotherapy. The mOS being 15.4 months for nivolumab plus chemotherapy compared to 9.1 months for chemotherapy alone (HR 0.54). This positive result allowed for hierarchical testing of mOS in all randomised patients which also favoured nivolumab and chemotherapy; 13.2 months versus 10.7 months (HR 0.74). Subgroup analysis revealed a greater benefit in patients with PD-L1≥1% (HR 0.55) than PD-L1 < 1% (HR 0.98). The comparison of combination ICI to chemotherapy also yielded a mOS benefit in patients that were PD-L1 ≥ 1% of 13.7 months compared to 9.1 (HR 0.64). This benefit persisted in the assessment of all randomised patients, mOS of 12.8 months compared to 10.7 (HR 0.78) although the subgroup analysis again showed more benefit in patients with PD-L1≥1% (HR 0.63) as opposed to PD-L1 < 1% (HR 0.96). Unlike nivolumab and chemotherapy, combined ICI did not show PFS benefit and demonstrated increased ORR only in patients with PD-L1≥1% and comparable ORR to chemotherapy alone in all randomised patients. Toxicity was similar across arms, with the highest grade 3–4 treatment-related adverse events (TRAE) in the nivolumab and chemotherapy arm (47%) whilst combined ICI had only 32%. ESCORT-1st took 596 similar phenotype patients from entirely Asian countries and randomised them 1:1 to chemotherapy (cisplatin 75 mg/m2 and paclitaxel 175 mg/m2, Q3W, up to 6 cycles) plus anti-PD-1 agent camrelizumab (200 mg 3 weekly) or placebo. At a median follow-up of only 10.8 months, both primary end-points favoured combination camrelizumab and chemotherapy; mOS 15.3 versus 12 months (HR 0.7, p = 0.001) and PFS 6.9 versus 5.6 months (HR 0.56, p < 0.001) [13]. Subgroup analysis shows all patients benefit, irrespective of PD-L1 (TPS). Both arms had grade 3 or higher TRAE in >60% of patients, the most common of which being reactive cutaneous capillary endothelial proliferation (RCCEP, a unique skin toxicity secondary to camrelizumab) and anaemia. This trial included health-related quality of life (HRQoL) assessments and demonstrated improved QoL scores (QLQ-C30 and QLQ-QES18) in the camrelizumab arm.
Fig. 1.
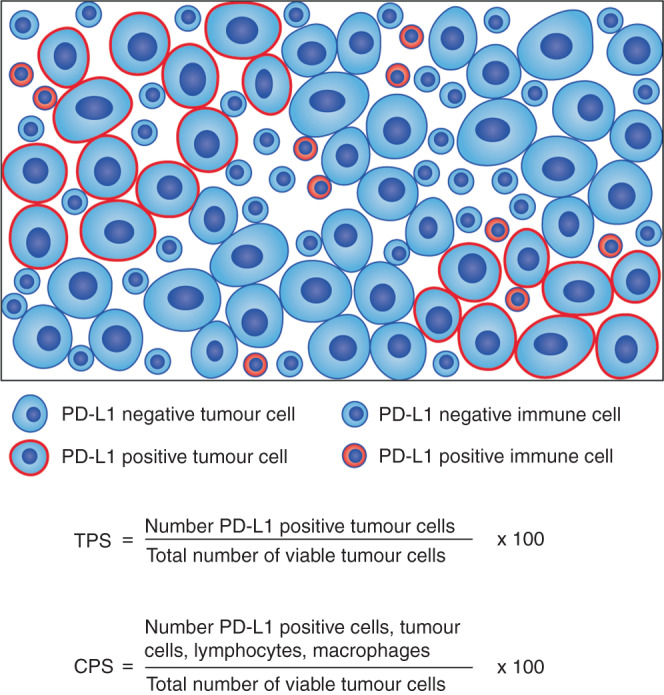
Definition of PD-L1 positivity. PD-L1, Definition of PD-L1 positivity; TPS and CPS.
Based on these results, combination chemotherapy plus anti-PD-1 therapy represents a valuable treatment option for metastatic ESCC, with OS benefit across all three trials and thus a new standard of care. The greatest benefit however seems to be observed in patients with PD-L1 enriched tumours (variably defined) thus further research is needed to aid in appropriate patient selection. The results of CHECKMATE 648 also support the use of combined ICI in the first-line setting; however, there was the familiar lack of PFS benefit and early crossing of survival curves which again indicates the need for refined patient selection for this treatment approach. The evidence for EAC and GOJ AC in KEYNOTE 590 needs to be interpreted in combination with the trials discussed below.
Adenocarcinoma
Outside of the KEYNOTE 590 trial, the only other study which included EAC was the CHECKMATE-649, also presented at ESMO 2020, with an updated efficacy and safety presented at ASCO 2021. CHECKMATE-649 is a three-armed randomised controlled trial (RCT) that enrolled unresectable/metastatic HER2 non-positive (HER2-negative or unknown) AC of gastric/GOJ or oesophagus to either chemotherapy alone (XELOX or FOLFOX), chemotherapy plus nivolumab (NIVO 360 mg 3 weekly + XELOX or NIVO 240 mg 2 weekly + FOLFOX) or nivolumab plus ipilimumab (NIVO1 + IPI3: nivolumab 1 mg/kg + ipilimumab 3 mg/kg 2 weekly). The baseline characteristics of patients were well balanced between groups, with predominantly non-Asian (~76%) and gastric cancer (~70%) cohorts. There was a small proportion of MSI high tumours in each group, 3% in both arms. Regarding chemotherapy received in each arm, there was even prescription of XELOX and FOLFOX. The dual primary end-points were OS and PFS in PD-L1 CPS ≥5 patients within NIVO plus chemo versus chemo alone arms. This PD-L1 cut-off was selected based on early correlative studies done in the phase 1/2 CHECKMATE 032 trial whereby higher CPS [5–10] scores demonstrated a stronger association with ICI efficacy [14], although this observed association was made on limited (<40) patients. At a minimum follow-up of 24 months, the primary end-point favoured nivolumab plus chemotherapy; mOS 14.4 versus 11.1 months (HR 0.70, p < 0.0001) [15, 16]. Similar results were seen between groups when considering CPS ≥ 1 (HR 0.77) and in all patients randomised i.e. PD-L1 unselected (HR 0.8, p = 0.0002). The superiority of OS was consistent across all subgroups (region of enrolment Asian versus non-Asian, site of primary) however patients with PD-L1 enriched tumours looked to derive greater benefit; CPS≥1 (HR 0.76) versus CPS < 1 (HR 0.92) and same for CPS ≥5 or <5. Superiority in PFS was also demonstrated in CPS≥5 at 7.7 months with the combination versus only 6 months with chemotherapy alone (HR 0.68, p < 0.0001). Like KEYNOTE 590, there was an increase in ORR and mDOR with the combination compared to chemotherapy alone. Grade 3 or 4 TRAE was higher in the combination arm at 59% compared to only 44%, with the most common being nausea, diarrhoea and peripheral neuropathy. Despite this, time to symptoms deterioration was NR in the combination arm compared to 21 months in the chemotherapy alone and patients QoL as assessed by FACT-Ga GP5 questionnaire was comparable between groups [16]. Contrary to this, recruitment to the NIVO1 + IPI3 arm was halted early due to high morbidity and mortality [17] and at a minimum follow-up of 35.7 months the results of 409 patients in NIVO1 + IPI3 versus chemotherapy arms showed early crossover of the survival curves and no difference in mOS compared to chemotherapy alone.
This trial demonstrated the clinically and statistically meaningful benefits of adding nivolumab to first-line chemotherapy (XELOX/FOLFOX). Furthermore, unlike some of the earlier ICI studies, the benefit is possible irrespective of PD-L1 status (as determined by either TPS or CPS) given the OS benefit observed in all randomised patients. Although like other studies, the exploratory subgroup analysis comparing PD-L1 enriched groups by various cut-offs (CPS ≥5 or ≥1, TPS ≥1%) gives the impression that the benefit seen in all randomised patients may be driven by the benefit of patients with CPS ≥ 5. This trial provides level 1 evidence for the use of nivolumab plus chemotherapy in achieving increased ORR, PFS, OS and appropriate safety as first-line treatment for esophagogastric adenocarcinoma, the utility of CPS in patient selection needs to be further evaluated.
The phase 3 portion of ATTRACTION-4 was the third large phase 3 trial presented at ESMO 2020 with a similar design to CHECKMATE-649, although this trial did not include oesophageal primaries and the cohort was entirely from Asian countries with primary end-points independent of PD-L1 expression. Patients were randomised 1:1 to nivolumab (360 mg 3 weekly) or placebo, plus standard of care chemotherapy, which was either CAPOX or SOX (S-1, 40 mg/m2 twice daily for 14 days and oxaliplatin 130 mg/m2 every 3 weeks). Baseline characteristics were even between groups, although the overall cohort was predominantly TPS < 1% (84%) and ~50% had diffuse-type gastric cancer [18, 19]. Co-primary end-point of PFS significantly favoured combination with mPFS 10.45 months compared to 8.34 (HR 0.68), similar to the phase 2 KEYNOTE 659 looking at the same combination [20]. Unfortunately, there was no difference in OS, with mOS of 17.45 versus 17.15 months respectively. Combination treatment, however, did yield a higher ORR and a longer DOR, something observed in almost all studies in this space. Another small phase 2 trial KEYNOTE 059, was a multicohort study looking at safety, tolerance and efficacy of pembrolizumab as either third-line monotherapy treatment, first-line monotherapy or first-line in combination with chemotherapy [cisplatin 800 mg/m2 on day 1 and 5FU (800 mg/m2 Day 1–5)/Capecitabine (1000 mg/m2 twice daily day 1–14) every 3 weeks]. Cohort 1; pembrolizumab monotherapy in 3rd line treatment is discussed later. Cohort 2 and 3 were published together in 2019, showing that ORR in the combination cohort favoured PD-L1 CPS≥1 whilst mOS was actually higher in CPS < 1 [21]. The weight of this study’s findings however is limited by the small sample size (Cohort 2: 25, Cohort 3 = 31).
The lack of OS benefit in ATTRACTION-4 could be explained by multiple factors; patient selection, which included enrolment of PD-L1 unselected patients and subsequent assessment of PD-L1 by TPS rather than CPS, showing 84% of patients to be TPS < 1%. Furthermore 50% of patient’s had diffuse histology, a feature of the Asian Cancer Research Group (ACRG) molecular subgroup classification which is associated with advanced-stage disease, poor prognosis and poor response to treatment [22]. Finally, patients from Asian countries tend to have a higher uptake of multiple lines of therapy and in ATTRACTION-4, most patients received subsequent lines of treatment, with 27% of the placebo arm, receiving anti-PD-1 therapy upon progression [18]. Differing response to treatment between Asian and Non-Asian gastric cancers is not a new observation, particularly with targeted treatment [23, 24]. Recent assessments of gene expression profiles have demonstrated distinct differences between these groups. Relevant to the observed negative results of ATTRACTION-4, a large meta-analysis looking at 1600 gastric cancer samples demonstrated that non-Asian gastric cancers have more tumour infiltrating T cells, and enriched T cell gene expression signatures, including CTLA-4 signalling [25], this difference in tumour immunity may explain why ATTRACTION-4 which was an entirely Asian cohort, has seen a lack of OS benefit. Contrary to this however is the greater benefit of ICI in Asian subgroups comparable to non-Asian subgroups observed in other studies [11, 26, 27]. Efforts into understanding the differences between these groups are ongoing.
The only first-line phase 3 trial to have an ICI monotherapy arm is KEYNOTE 062, which randomised 763 HER2-negative, PD-L1-positive (CPS ≥ 1) advanced gastric/GOJ AC patients from predominantly non-Asian countries (~75%) to receive one of three arms of treatment; pembrolizumab monotherapy, pembrolizumab plus chemotherapy (5-fluorouracil (5FU) 800 mg/m2 Day 1–5 or capecitabine 1000 mg/m2 day 1–14 and cisplatin 80 mg/m2 day 1, every 3 weeks) or chemotherapy plus placebo. Primary end-points were OS in PD-L1 CPS ≥1 and ≥10 and PFS in CPS ≥1, comparing both ICI-containing arm to chemotherapy alone. With regards to baseline characteristics, most patient’s had PD-L1 CPS < 10 and a gastric primary [27]. This trial found pembrolizumab monotherapy to be non-inferior to chemotherapy in the intention to treat population; CPS≥1 (HR 0.91) and with a favourable toxicity profile. In the CPS≥10 subgroup analysis, OS favoured pembrolizumab with mOS of 17.4 versus only 10.8 months (HR 0.69) and 2-year survival of 39% compared to only 22% however statistical design meant these results could not be statistically verified. Similarly, combination pembrolizumab and chemotherapy did not show superiority over chemotherapy alone in CPS≥1 or CPS≥10 patients, except for those with microsatellite instability (MSI-H) tumours which favoured the combination, irrespective of PD-L1 expression.
These results are unique as they offer insight into the utility of first-line pembrolizumab monotherapy, which offered favourable toxicity and comparable OS to first-line chemotherapy in patients with CPS ≥ 1, the Kaplan–Meier survival curves however crossed and thus single-agent pembrolizumab needs to be evaluated further before being considered a viable treatment option. The addition of pembrolizumab to chemotherapy, however, failed to show superiority over chemotherapy alone in all subgroups. One theory for this differing result could be the chemotherapy backbone [3]. CHECKMATE-649, apart from including EAC (12–13%), had similar baseline patient characteristics to KEYNOTE 062; however, the chemotherapy backbone was oxaliplatin rather than cisplatin. Clinical and pre-clinical histological studies have demonstrated that oxaliplatin works via immunogenic cell death and has been associated with increased intra-tumoral CD8 + T cells [28] thus offering synergy with ICI. This notion, however, is challenged by the negative results of other trials that used oxaliplatin [12, 18].
Outside of combination first-line ICI with chemotherapy, the JAVELIN GASTRIC 100 also looked at the role anti-PD-L1 antibody avelumab might offer as maintenance treatment following induction chemotherapy. The trial design enrolled 499 advanced or metastatic gastric/GOJ AC patients who had completed 12 weeks of first-line chemotherapy with fluoropyrimidine and oxaliplatin and showed no progressive disease. They were then randomised to continue chemotherapy or start maintenance avelumab 10 mg/kg every 2 weeks. The patients were predominantly western (57%), had a gastric primary (70%) and PD-L1 TPS < 1% (78%) [29]. The primary end-point of mOS in TPS ≥ 1% was numerically higher in avelumab group 19 versus 15 months with chemotherapy (HR 1.13). This is the only study to have looked at ICI in the maintenance role and although the toxicity was favourable, the results do not support its use in clinical practice.
As can be appreciated in Table 1, interpreting these results as a group is difficult given the differing ICI, chemotherapy backbone, patient populations and biomarker cut-off used in patient selection. Despite this, these trials have changed clinical practice, with first-line combination ICI and chemotherapy the new SOC. For ESCC the KEYNOTE 590, ESCORT-1st and CHECKMATE 648 provide evidence for the addition of ICI to first-line chemotherapy irrespective of PD-L1 expression, whilst for AC of the stomach/GOJ, the CHECKMATE-649 supports the use of nivolumab in combination with first-line oxaliplatin-based chemotherapy irrespective of PD-L1 in the primary analysis. The subgroup analysis however casts doubt on the value of nivolumab in the CPS < 5 patients. Furthermore, there are conflicting negative results amongst other studies where the primary end-point was considered in CPS≥1 [20, 21, 27] which makes the value of ICI in combination with chemotherapy for AC unclear were CPS = 1–4 and thus the use of nivolumab in combination with chemotherapy could be reserved for esophagogastric AC patients whom CPS ≥ 5 until more data is available.
Table 1.
Pivotal trials: baseline characteristics, survival outcomes and PD-L1 subgroup definitions.
Immunotherapy in later lines of treatment
All trials supporting the use of IO in later lines of therapy were conducted before the above-mentioned studies and thus this data is relevant to patients who have not had prior ICI treatment. With ongoing trials better defining the benefit of ICI therapies in the first-line combination setting, the use of ICI after progression, either in novel combinations or as a rechallenge, will need to be defined.
Adenocarcinoma
The evidence for ICI in esophagogastric cancers started with ATTRACTION-2, the pivotal phase 3 trial with 493 predominantly Japanese and heavily pre-treated unresectable/metastatic gastric or GOJ AC patients who were randomised 2:1 to receive either nivolumab 3 mg/kg (NIVO3) or placebo every 2 weeks until disease progression or toxicity necessitating discontinuation. OS favoured nivolumab; 5.26 months compared to 4.14 months (HR 0.63, p < 0.0001) [30]. More striking was the 12-month survival rate of 26.2% which was more than double that of placebo (10%) and the median duration of response of 9.53 months. This trial enrolled irrespective of PD-L1 status however <40% of each arm had tissue available for PD-L1 assessment, so whilst they reported an exploratory post hoc analysis concluding that survival benefit of nivolumab was independent of PD-L1 (assessed by TPS only), this has little true value in the biomarker discussion. The treatment of gastric cancer in Japan includes access and widespread use of second-line paclitaxel and ramucirumab so due to previous reports of anti-VEGF treatments changing the tumour microenvironment [31], a subsequent study analysed the Japanese subpopulation from ATTRACTION-2 and found that they had OS benefit compared to the overall population [32] and that ramucirumab exposure did not alter the clinical activity of nivolumab.
Following this, CHECKMATE 032 a phase 1/2 trial looked at response rates and efficacy of NIVO3 or nivolumab plus ipilimumab [either nivolumab 3 mg/kg + ipilimumab 1 mg/kg (NIVO3 + IPI1) or NIVO1 + IPI3] in a range of solid tumour types. The esophagogastric cancer cohort included patients with unresectable/advanced gastric, GOJ or oesophageal AC who had progressed on ≥1 prior chemotherapy, including HER2 positive tumours were the patient had received trastuzumab. This was a small study, but it did show response rates (12%), OS (6.2 months) and PFS (1.4 months) of NIVO3 in a PD-L1 unselected western patient cohort comparable to those seen in ATTRACTION-2 and similarly irrespective of PD-L1 status(TPS). The use of combination NIVO1 + IPI3, resulted in higher ORR then NIVO3 alone or NIVO3 + IPI1 but with increased toxicity and no difference in PFS or OS. The lack of PFS or OS benefit was attributed to an increased number of PD-L1 positive (TPS≥1%) and MSI high patients in the nivolumab group compared to the NIVO1 + IPI3 cohort; however, these results were mirrored in the larger NIVO1 + IPI3 arm of CHECKMATE-649, a predominantly gastric cancer (70%) cohort indicating further research is needed to evaluate the role of combined ICI in these patients.
Cohort 1 of KEYNOTE 059, although a small single-arm phase 2 trial, looked at a further 259 heavily pre-treated patients (≥2 lines previous treatment) with similar characteristics as ATTRACTION-2 and provided safety and clinical activity evidence for pembrolizumab. This trial differed from ATTRACTION-2 in that it had a predominantly non-Asian cohort and ORR was higher in patient’s whom PD-L1 CPS ≥1. A similar trend of PD-L1 dependant response/benefit was seen in KEYNOTE 061, which was the first study looking at second-line pembrolizumab and the first ICI trial in esophagogastric cancers with an active comparator arm. This phase 3 trial enrolled predominately western patients with unresectable/metastatic gastric or GOJ adenocarcinoma who had progressed on first-line platinum and fluoropyrimidine doublet (and trastuzumab if HER amplified) and randomised them to either pembrolizumab or paclitaxel (80 mg/m2 D1,8,15 every 4 weeks). Patients were enrolled irrespective of their PD-L1 status initially however the independent data monitoring committee amended this after 83% enrolment due to poorer outcomes of patients with CPS < 1. The primary end-point; OS in the CPS ≥ 1 was not significant between groups; pembrolizumab mOS of 9.1 months versus Paclitaxel of 8.4 months (HR 0.82, p = 0.045, with a pre-specified significance level needed of p = 0.0135) [33]. In patients with CPS < 1 (stratified subgroup), the median OS for pembrolizumab was 4.8 months compared to 8.4 months with paclitaxel. Interestingly a post hoc analysis comparing CPS ≥10 or <10 demonstrated a difference in OS favouring pembrolizumab of 10.4 versus 8 months, similarly those with MSI high tumours had OS Not Reached (NR) compared to 8.1 months. ORR were similar between arms, whilst pembrolizumab offered a much greater duration of response; 18 versus 5.2 months. Although this was a negative study overall, it did support the observation that with increasing PD-L1 expression, there is greater efficacy with anti-PD-1 treatments. Furthermore, the crossover of the Kaplan–Meier curves at approximately 8 months and prolonged response with pembrolizumab highlights the need for refined patient selection of those who will benefit from ICI. Similarly, the small population of MSI high patients in this study showed significant clinical benefit with pembrolizumab, in keeping with other gastric cancer cohorts in phase 1/2 studies [34, 35].
Like the above avelumab study, JAVELIN Gastric 300 was a negative study, looking at the use of avelumab in the third-line setting, 371 patients with advanced/unresectable gastric or GOJ adenocarcinoma were randomised to either avelumab 10 mg/kg two weekly or physician’s choice chemotherapy (irinotecan or paclitaxel). With well balance groups at baseline, all end-points; OS, PFS, ORR numerically favoured chemotherapy over avelumab, with no differential response seen against subgroups [36].
Squamous cell carcinoma
The two trials that included SCC both involve pembrolizumab, in the second-line setting KEYNOTE 181 or the third or later line setting KEYNOTE 180. Firstly, KEYNOTE 180 was a single-arm phase 2 trial, which enrolled 121 patients with either ESCC, EAC or GOJ (Siewert 1) adenocarcinoma who had progressed through ≥2 lines of prior treatment. They received pembrolizumab until progression, withdrawal or maximum of 2 years of treatment. The study population was predominantly non-Asian (68%), SCC (52%) and PD-L1 negative by CPS < 10 (52%). The primary end-point of ORR was 9.9%, which when assessed by histological subtype was 14.3% in ESCC and only 5.2% in adenocarcinomas [37]. PFS and mOS were limited at 2 and 5.8 months, respectively. As was seen with the above studies, PD-L1 positive patients had a higher response rate and a longer DOR. Median PFS was comparable between histology’s and PD-L1 groups, whilst OS favoured ESCC; 6.8 versus only 3.9 months for AC. The mOS for PD-L1 positive was marginally higher 6.3 versus 5.4 months. The response rate in the ESCC population in this trial is similar to that of another small phase 2 Japanese trial of nivolumab in heavily pre-treated ESCC, which saw ORR of 17% and mOS of 10.8 months [38].
The large phase 3 KEYNOTE 181, enrolled 628 advanced/metastatic ESCC, AEC or GOJ adenocarcinoma (Siewert 1 and HER2-negative) patients who had progressed on first-line treatment and randomised them 1:1 to pembrolizumab or investigators choice chemotherapy (paclitaxel, docetaxel or irinotecan). The primary end-point was OS in PD-L1 CPS ≥10, ESCC and in all patients. The patient’s enrolled were predominantly male (86%), non-Asian (61.5%), with ESCC (63%), had received 1 prior line of treatment (96.5%) and CPS < 10(64%). At final analysis, the mOS in patients with CPS≥10 was 9.3 months with pembrolizumab compared to 6.7 with chemotherapy (HR 0.69, p = 0.0074) [39]. The co-primary end-point of mOS in ESCC showed a statistically insignificant improvement, 8.2 months with pembrolizumab versus 7.1 with chemotherapy; however, if considering only the ESCC patients with CPS ≥ 10 (42%), this difference was more marked at 10.3 versus 6.7 months with chemotherapy. As with previous studies, pembrolizumab had a much high ORR (23%) compared to chemotherapy (6%) in the CPS≥10 patients, regardless of histology and a longer DOR but no difference in PFS in all groups. This trial led to the KEYNOTE 590 study discussed above.
HER2-positive disease
HER2 overexpression, also called HER2-positive and defined as immunohistochemistry 3+ or 2+ and fluorescence in-situ hybridisation ratio positive, is seen in ~18% of oesophagogastric cancers. For patients with metastatic HER2-positive gastric or GOJ adenocarcinoma, adding trastuzumab to first-line chemotherapy offers 2.7 months OS benefit compared to chemotherapy alone [5]. Analysis of tumour tissue in patients who have progressed on trastuzumab have subsequently shown increase PD-L1 expression and immune infiltration [40] which is a part of the mechanistic theory as to why combination HER2 and PD-L1 inhibition may have a synergistic anti-tumour effect.
The mEGA study was a small phase 2 single-arm trial that looked at the safety and efficacy of pembrolizumab in combination with standard of care trastuzumab plus chemotherapy in the first-line setting for 37 patients with unresectable/metastatic HER2-positive (IHC 3+ or FISH positive) oesophageal, GOJ or gastric adenocarcinoma. The baseline characteristics of patients were predominantly white race (86%) with an even representation of tumour locations; oesophageal 38%, GOJ 32%, gastric 30% [41]. The primary end-point of 6 months PFS was 70% and the observed mPFS was 13 months, which translated into a mOS of 27.3 months, furthermore DCR was 100% and 17% achieved a CR. These impressive results came at the cost of 97% of patients experiencing a TRAE, for which all of the required dose reduction and an 11% rate of iRAE that led to discontinuation and 5% rate of serious AE (nephritis).
The majority of the first-line treatment studies excluded patients with her HER2-positive disease however the first results of KEYNOTE 811, a phase 3 RCT comparing first-line pembrolizumab or placebo with SOC; trastuzumab plus chemotherapy (CAPOX or 5FU plus cisplatin) in HER2-positive gastric or GOJ cancer patients was presented at ASCO 2021. The patients were global, with predominantly gastric primaries, 20% diffuse histology and >85% PD-L1 CPS ≥1 in both arms. The ORR was presented confirming an increase in ORR to 74.4% with pembrolizumab compared to only 51.9% in the placebo arm and a complete response (CR) rate of 11% [42]. The all-cause AE’s where however comparable between arms, with an expected increase in immune-mediated AE’s in the pembrolizumab arm. These results are promising, we await the dual primary end-points OS and PFS to determine whether this combination will become a new standard of care for HER-positive patients. Also, on the horizon is INTEGA(NCT03409848), a Phase 2 trial comparing Ipilimumab or FOLFOX in combination with nivolumab and Trastuzumab in first-line HER2-positive GOJ AC.
Neoadjuvant therapy/adjuvant therapy
At present the management of localised and locally advanced oesophagogastric cancer involves an individualised multimodal approach decided upon in a multidisciplinary meeting and based upon the location and histology of the tumour, along with patient’s comorbidities. The current SOC treatment for stage 2–3 operable oesophageal and GOJ cancers regardless of histology is neoadjuvant (NA) chemotherapy, chemoradiotherapy (CRT) or perioperative chemotherapy and resection. Prior to the results of CHECKMATE 577, patients receiving NA CRT would then enter surveillance post operatively, irrespective of pathological response to treatment. Pathological response however is prognostic, with some analysis estimating mOS >70 months when achieving pathological complete response (pCR), compared to only half that if residual disease [43]. Given the poor prognosis in this subgroup of patients, CHECKMATE 577 sought to evaluate 12 months of adjuvant nivolumab in reducing the risk of disease recurrence. The trial randomised 1085 adult patients with stage 2–3 oesophageal or GOJ cancer who had an R0 resection showing residual disease after trimodality treatment (NA CRT followed by resection) in a 2:1 ratio to receive nivolumab or placebo for 12 months. Patients were enrolled irrespective of histological subtype (AC or SCC) or PD-L1 status, though patients were stratified by these features along with pathological nodal status (denoted by ypLN). The patient population was predominantly white (84%) males (81%) from Europe or north America (67%) and majority were stage 3 (66%), ypLN positive (57%) and PD-L1 < 1% (70%), as determined by TPS (see Fig. 1) [44]. At a median follow-up of 24.4 months, the primary end-point; median disease-free survival (DFS: time from the date of randomisation to the first date of disease recurrence or death) was 22.4 months in the nivolumab arm compared to only 11 months in the placebo arm (HR 0.69). A pre-specified subgroup analysis demonstrated no difference in outcomes between patients with PD-L1 (TPS) < 1% and those ≥1%. Similarly in a post hoc analysis, when classifying patients according to CPS (Fig. 1) <5 or ≥5, both groups benefited however CPS≥5 subgroup seemed to obtain a greater effect (HR 0.62 compared to 0.89). There was also greater benefit with nivolumab seen in patients with SCC compared to AC, ypLN+ disease and oesophageal tumours compared to GOJ tumours. Whilst the overall survival data of CHECKMATE 577 is awaited, quality of life (QOL) data was presented at ASCO 2021 and found that FACT-E scores were similar between groups [45]. The use of DFS as a primary end-point in an adjuvant study is accepted based on the meta-analysis supporting this surrogate end-point in gastric cancer studies, noting this was from a purely chemotherapy era [46]. In ICI trials, however, the use of response-based surrogate end-points such as PFS has not always correlated with OS benefit [47]. Some ICI studies that have not demonstrated PFS, go on to show significant OS benefit. So although we await OS data from CHECKMATE 577, with such impressive DFS outcomes, there is an expectation of an OS result.
Subsequent to CHECKMATE 577, the results of a small phase 2 trial (NCT02998268) were presented at ASCO 2021 looking at the safety and efficacy of pembrolizumab plus NA CRT in operable stage 2B-3 EAC and ongoing pembrolizumab for 12 months post-resection. The design of this trial involved initial randomisation of 39 patients to 2 cycles of induction chemotherapy (CROSS regimen) with or without pembrolizumab, followed by all patients receiving the same concurrent CRT plus 3-weekly pembrolizumab. Biopsies pre and post the induction phase of treatment, to assess the correlative relationship between the tumours microenvironment and achieving major pathological response(MPR). The primary end-point was the MPR, which is defined as CR or <10% viable tumour cells remaining post-NA treatment. The use of concurrent pembrolizumab was associated with an increase in MPR of 48.7% compared to historical control of 30% and oesophageal tumours were far more likely to achieve MPR, 73.3% compared to only 33.3% in GOJ tumours [48]. This translated into a 1-year OS of 93.8% for those achieving MPR compared to only 62.5% for those without MPR. The authors observed tumours enriched with monocytes and cytotoxic CD8 + T cells were more likely to achieve MPR. This approach is also being evaluated in the similar Neo-CREATE trial looking at Avelumab in addition to NA CRT and in a large phase 3 trial KEYNOTE 975, other ongoing trials are discussed below.
Discussion
The above-described studies represent a paradigm shift in the management of esophagogastric cancers. The varied responses to ICI highlight the underlying molecular heterogeneity of these cancers. Many patients with oesophagogastric cancer do not respond to ICI treatment, even when combined with chemotherapy. An important aspect of patient selection in both trial design and clinical practice is the identification of appropriate predictive biomarkers. In other tumour types such as non-small-cell lung cancer, the predictive biomarker for ICI efficacy is largely PD-L1 TPS, which as illustrated in Fig. 1 represents all viable tumour cells showing membrane staining for PD-L1 and is expressed as a percentage. For oesophagogastric cancers, however, membranous PD-L1 expression is uncommon and in early studies did not correlate with ICI response [49]. Instead, PD-L1 expression is prominent on tumour infiltrating myeloid cells, for which CPS has been developed as an alternative biomarker for PD-L1 positivity. CPS is calculated by all PD-L1 staining tumour cells, lymphocytes and macrophages, which is then divided by the number of viable tumour cells and multiplied by 100, expressed as a figure not percentage (Fig. 1). Most of the above trials used CPS PD-L1 cut-offs in their primary analysis. These PD-L1 thresholds however are crude for several reasons; firstly, there is inter-pathologist variability in its assessment, with the highest agreement between pathologist (99.2%) observed when determining tumours to be CPS≥10 as opposed to ≥1 (94.7%) [50]. Secondly, there is both temporal and spatial variation in PD-L1 expression within a tumour sample from the same patient, and between primary cancer and metastases, and pre and post-chemotherapy [51]. In the early trials looking at second or later line ICI monotherapy, PD-L1 expression was mostly done retrospectively on archival tissue and the availability of PD-L1 tissue in some studies was <50% which limits the confidence of any conclusions about the association of response and PD-L1 positivity. Some of the pivotal first-line trials used CPS≥1 as their biomarker and failed to demonstrate ICI benefit [20, 21, 27]. There is also a consistent trend across the data that patients with CPS≥10 either by primary analysis, stratification or post hoc analysis, have a greater benefit with ICI. This observation, along with the exploratory subgroup analysis of CHECKMATE-649 and 648 using various CPS cut-offs, tells us the higher the PD-L1 expression by CPS the more likely to benefit from ICI. The ideal threshold however has not been defined.
Regarding other potential biomarkers, in 2014, TCGA characterised gastric cancer into four key molecular subtypes, of which tumours positive for EBV was one. EBV antigens within tumours has been shown to facilitate neoepitope presentation and in small studies, increased anti-tumour immune response. Although studies are looking at the role of EBV as a potential biomarker for ICI response, EBV expression is only seen in 9% of gastric cancers and these tumours also express high PD-L1, thus the utility of EBV positivity as a predictive biomarker needs to be further defined. Another of the defined molecular subgroups by TCGA is MSI-H tumours, which are also associated with increased PD-L1 expression and a higher tumour mutational burden (TMB). Pembrolizumab was approved by the FDA for use in all tumours that are MSI-H based on results of a few phase 1/2 trials showing impressive and durable clinical activity in heavily pre-treated patients. These trials included several gastric and GOJ cancer patients, thus supporting the use of ICI in MSI-H esophagogastric cancers. Furthermore, CHECKMATE 032, CHECKMATE-649, KEYNOTE 061 and KEYNOTE 062 all report on the outcomes of small cohorts of MSI-H patient’s, demonstrating high ORR, durable responses and HR for death of <0.35 [52]. The MSI-H subgroup represents up to 22% of gastric cancer patients and thus its recommended that all patients with advanced esophagogastric cancer have MSI assessment given the predictive value MSI has shown in demonstrating response to pembrolizumab. Correlative biomarker sub-studies are underway, as are a number of trials to better define the role of ICI in these cancers.
For gastric cancer, KEYNOTE 585 is an ongoing large phase 3 trial looking at physicians’ choice perioperative chemotherapy plus concurrent pembrolizumab, additionally there is a small phase 1/2 trial looking at combination nivolumab plus ipilimumab with chemoradiotherapy in operable gastric cancer (NCT03776487). Combined ICI is also being tested in a large phase 2/3 trial concurrently with CRT for operable EAC or GOJ AC(NCT03604991).
As well as the use of ICI in early-stage and HER2 amplified disease, as outlined above, there are several clinical trials on the horizon looking at novel targeted treatment and ICI combinations. Many of these combinations are largely looking at the role of novel targeted treatments in optimising the tumour microenvironment in order to synergistically improve response rates and outcomes with ICI in patients with oesophagogastric cancer. One such trial is the phase 3 INTEGRATE 2b (NCT02773524) which is evaluating third-line combination regorafenib plus nivolumab versus SOC chemotherapy in EAC and GOJ AC in patients can have progressed on ICI therapy. So whilst focus goes in to determining the optimal biomarker for patient selection in trials, these novel combinations may improve the efficacy of ICI more holistically.
Conclusion
The above trial landscape sees a heterogenous group of diseases treated with ICI monotherapy and combination ICI with chemotherapy, with varying results. Despite recent developments in understanding the genetic and molecular differences within this spectrum of diseases, our method for patient selection remains an undefined CPS threshold for PD-L1 positivity. Nevertheless, these pivotal trials have demonstrated clinical activity, durable responses and appropriate safety of both anti-PD-1 monotherapy in later lines and in combination with first-line chemotherapy in selected cases and in combination with anti-CTLA-4 in ESCC. We eagerly await the ongoing trials in this space, in order to refine patient selection and assess the potential role of novel targeted treatment combinations with immune checkpoint inhibition in optimising patients responses.
Author contributions
AS: co-developed concept, analysis of data, manuscript writing, manuscript review. AR: analysis of data, manuscript writing, manuscript review. CK: analysis of data, manuscript writing, manuscript review. VB: analysis of data, manuscript writing, manuscript review. TP: co-developed concept, analysis of data, manuscript writing, manuscript review.
Competing interests
TP reports uncompensated advisory board BMS, compensated advisory board MSD. The remaining authors declare no competing interests.
Ethics approval and consent to Participate
Not Applicable
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Annabel Smith, Email: Annabel.smith@sa.gov.au.
Timothy Price, Email: Timothy.Price@sa.gov.au.
References
- 1.Arnold M, Ferlay J, van Berge Henegouwen MI, Soerjomataram I. Global burden of oesophageal and gastric cancer by histology and subsite in 2018. Gut. 2020;69:1564–71. doi: 10.1136/gutjnl-2020-321600. [DOI] [PubMed] [Google Scholar]
- 2.Napier KJ, Scheerer M, Misra S. Esophageal cancer: A Review of epidemiology, pathogenesis, staging workup and treatment modalities. World J Gastrointest Oncol. 2014;6:112. doi: 10.4251/wjgo.v6.i5.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wagner AD, Syn NLX, Moehler M, Grothe W, Yong WP, Tai B, et al. Chemotherapy for advanced gastric cancer. Cochr Database Syst Rev. 2017. https://www.cochranelibrary.com/cdsr/doi/10.1002/14651858.CD004064.pub4/pdf/full. [DOI] [PMC free article] [PubMed]
- 4.Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7–33. doi: 10.3322/caac.21654. [DOI] [PubMed] [Google Scholar]
- 5.Bang Y-J, van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376:687–97. doi: 10.1016/S0140-6736(10)61121-X. [DOI] [PubMed] [Google Scholar]
- 6.Larkin J, Chiarion-Sileni VGR. Five-year overall survival outcomes of the Check-Mate 067 phase 3 trial of nivolumab plus ipilimumab combination therapy in advanced melanoma. N Engl J Med. 2019;381:1535–46. doi: 10.1056/NEJMoa1910836. [DOI] [PubMed] [Google Scholar]
- 7.Herbst RS, Garon EB, Kim DW, Chul Cho B, Pérez Gracia JL, Han JY, et al. LBA4 Long-term follow-up in the KEYNOTE-010 study of pembrolizumab (pembro) for advanced NSCLC, including in patients (pts) who completed 2 years of pembro and pts who received a second course of pembro. Ann Oncol. 2018;29:mdy511–003. [Google Scholar]
- 8.Chau I, Norman AR, Cunningham D, Oates J, Hawkins R, Iveson T, et al. The impact of primary tumour origins in patients with advanced oesophageal, oesophago–gastric junction and gastric adenocarcinoma—individual patient data from 1775 patients in four randomised controlled trials. Ann Oncol. 2009;20:885–91. doi: 10.1093/annonc/mdn716. [DOI] [PubMed] [Google Scholar]
- 9.Network CGAR. Integrated genomic characterization of oesophageal carcinoma. Nature. 2017;541:169. doi: 10.1038/nature20805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bass AJ, Thorsson V, Shmulevich I, Reynolds SM, Miller M, Bernard B, et al. Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513:202. doi: 10.1038/nature13480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kato K, Sun J, Shah M, Enzinger PC, Adenis A, Doi T, et al. LBA8_PR-Pembrolizumab plus chemotherapy versus chemotherapy as first-line therapy in patients with advanced esophageal cancer: The phase 3 KEYNOTE-590 study. Annals of Oncology. 2020;31:S1192–3.
- 12.Chau I, Doki Y, A.Ajani J, Xu J, Wyrwicz L, Motoyama S, et al. Nivolumab plus ipilimumab or nivolumab plus chemotherapy versus chemotherapy as first-line treatment for advanced esophageal squamous cell carcinoma: first results of CheckMate 648 study. In: Nivolumab plus ipilimumab or nivolumab plus chemotherapy versus chemotherapy as first-line treatment for advanced esophageal squamous cell carcinoma: first results of CheckMate 648 study. 2021 ASCO Annual Meeting. Abstract. Vol. 4001. 2021.
- 13.Xu R, Luo H, Bai Y, Mao T, Wang J, Fan Q, et al. ESCORT-1st: a randomized, doubleblind, placebo-controlled, phase 3 trial of camrelizumab plus chemotherapy vx chemotherapy in patients with untreated advanced or metastatic esophageal squamous cell carcinoma (ESCC) J Clin Oncol. 2021;39:4000. [Google Scholar]
- 14.Lei M, Siemers N, Pandya D, Chang H, Sanchez T, Dorange C, et al. association of PD-L1 combined positive score and immune gene signatures with efficacy of nivolumab (NIVO) ± ipilimumab (IPI) in patients with metastatic gastroesophageal cancer (mGEC). AACR; 2019.
- 15.Moehler M, Shitara K, Garrido M, Salman P, Shen L, Wyrwicz K, et al. LBA6_PR -Nivolumab (nivo) plus chemotherapy (chemo) versus chemo as first-line (1 L) treatment for advanced gastric cancer/gastroesophageal junction cancer (GC/GEJC)/esophageal adenocarcinoma (EAC): First results of the CheckMate 649 study. Annals of Oncology. 2020;31:S1191. [Google Scholar]
- 16.Janjigian YY, Ajani JA, Moehler M, Garrido M, Gallardo C, Shen L, et al. LBA7 Nivolumab (NIVO) plus chemotherapy (Chemo) or ipilimumab (IPI) vs chemo as first-line (1L) treatment for advanced gastric cancer/gastroesophageal junction cancer/esophageal adenocarcinoma (GC/GEJC/EAC): CheckMate 649 study. Ann Oncol. 2021;32:S1329–30. [Google Scholar]
- 17.Harada K, Abdelhakeem AAF, Ajani JA. A balancing act: Dual immune-checkpoint inhibition for oesophagogastric cancer. Nat Rev Clin Oncol. 2019;16:9–10. doi: 10.1038/s41571-018-0108-x. [DOI] [PubMed] [Google Scholar]
- 18.Boku N, Ryu M-H, Oh D, Al EA. randomised, multicenter, phase 2/3 study of nivolumab (Nivo) plus chemotherapy in patient (Pts) with previously untreated advanced or recurrent gastric (G) or gastroesophageal junction (GEJ) cancer. Annals of Oncology. 2020;31:S1142–S1215. doi: 10.6084/m9.figshare.hgv.1920. [DOI] [Google Scholar]
- 19.Boku N, Ryu M-H, Kato K, Chung HC, Minashi K, Lee K-W, et al. Safety and efficacy of nivolumab in combination with S-1/capecitabine plus oxaliplatin in patients with previously untreated, unresectable, advanced, or recurrent gastric/gastroesophageal junction cancer: interim results of a randomized, phase II trial. Ann Oncol. 2019;30:250–8. [DOI] [PMC free article] [PubMed]
- 20.Kawazoe A, Yamaguchi K, Yasui H, Negoro Y, Azuma M, Amagai K, et al. Safety and efficacy of pembrolizumab in combination with S-1 plus oxaliplatin as a first-line treatment in patients with advanced gastric/gastroesophageal junction cancer: Cohort 1 data from the KEYNOTE-659 phase IIb study. Eur J Cancer. 2020;129:97–106. doi: 10.1016/j.ejca.2020.02.002. [DOI] [PubMed] [Google Scholar]
- 21.Bang Y-J, Kang Y-K, Catenacci DV, Muro K, Fuchs CS, Geva R, et al. Pembrolizumab alone or in combination with chemotherapy as first-line therapy for patients with advanced gastric or gastroesophageal junction adenocarcinoma: results from the phase II nonrandomized KEYNOTE-059 study. Gastric Cancer. 2019;22:828–37. doi: 10.1007/s10120-018-00909-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Garcia‐Pelaez J, Barbosa‐Matos R, Gullo I, Carneiro F, Oliveira C. Histological and mutational profile of diffuse gastric cancer: current knowledge and future challenges. Mol Oncol. 2021;15:2841–67. doi: 10.1002/1878-0261.12948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ohtsu A, Shah MA, van Cutsem E, Rha SY, Sawaki A, Park SR, et al. Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III study. J Clin Oncol. 2011;29:3968–76. doi: 10.1200/JCO.2011.36.2236. [DOI] [PubMed] [Google Scholar]
- 24.Hecht JR, Bang Y-J, Qin S, Chung H-C, Xu J-M, Park JO, et al. Lapatinib in combination with capecitabine plus oxaliplatin (CapeOx) in HER2-positive advanced or metastatic gastric, esophageal, or gastroesophageal adenocarcinoma (AC): The TRIO-013/LOGiC Trial. J Clin Oncol. 2013;31. https://ascopubs.org/doi/abs/10.1200/jco.2013.31.18_suppl.lba4001. [DOI] [PubMed]
- 25.Lin SJ, Gagnon-Bartsch JA, Tan IB, Earle S, Ruff L, Pettinger K, et al. Signatures of tumour immunity distinguish Asian and non-Asian gastric adenocarcinomas. Gut. 2015;64:1721–31. doi: 10.1136/gutjnl-2014-308252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Moehler M, Shitara K, Garrido M, Salman P, Shen L, Wyrwicz L, et al. First-line (1L) nivolumab (NIVO) plus chemotherapy (chemo) versus chemo in advanced gastric cancer/gastroesophageal junction cancer/esophageal adenocarcinoma (GC/GEJC/EAC): expanded efficacy and safety data from CheckMate 649. J Clin Oncol. 2021;39:4002. [Google Scholar]
- 27.Shitara K, van Cutsem E, Bang Y-J, Fuchs C, Wyrwicz L, Lee K-W, et al. Efficacy and safety of pembrolizumab or pembrolizumab plus chemotherapy vs chemotherapy alone for patients with first-line, advanced gastric cancer: the KEYNOTE-062 phase 3 randomized clinical trial. JAMA Oncol. 2020;6:1571–80. doi: 10.1001/jamaoncol.2020.3370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li J-Y, Chen Y-P, Li Y-Q, Liu N, Ma J. Chemotherapeutic and targeted agents can modulate the tumor microenvironment and increase the efficacy of immune checkpoint blockades. Mol Cancer. 2021;20:1–21. doi: 10.1186/s12943-021-01317-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Moehler M, Dvorkin M, Boku N, Özgüroğlu M, Ryu M-H, Muntean AS, et al. Phase III trial of avelumab maintenance after first-line induction chemotherapy versus continuation of chemotherapy in patients with gastric cancers: results from JAVELIN gastric 100. J Clin Oncol. 2021;39:966–77. doi: 10.1200/JCO.20.00892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen L-T, Satoh T, Ryu M-H, Chao Y, Kato K, Chung HC, et al. A phase 3 study of nivolumab in previously treated advanced gastric or gastroesophageal junction cancer (ATTRACTION-2): 2-year update data. Gastric Cancer. 2019;23:510–19. doi: 10.1007/s10120-019-01034-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Takahashi A, Kono K, Ichihara F, Sugai H, Fujii H, Matsumoto Y. Vascular endothelial growth factor inhibits maturation of dendritic cells induced by lipopolysaccharide, but not by proinflammatory cytokines. Cancer Immunol Immunother. 2004;53:543–50. doi: 10.1007/s00262-003-0466-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kato K, Satoh T, Muro K, Yoshikawa T, Tamura T, Hamamoto Y, et al. A subanalysis of Japanese patients in a randomized, double-blind, placebo-controlled, phase 3 trial of nivolumab for patients with advanced gastric or gastro-esophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens. Gastric Cancer. 2019;22:344–54. doi: 10.1007/s10120-018-0899-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Shitara K, Özgüroğlu M, Bang Y-J, di Bartolomeo M, Mandalà M, Ryu M-H, et al. Pembrolizumab versus paclitaxel for previously treated, advanced gastric or gastro-oesophageal junction cancer (KEYNOTE-061): a randomised, open-label, controlled, phase 3 trial. Lancet. 2018;392:123–33. doi: 10.1016/S0140-6736(18)31257-1. [DOI] [PubMed] [Google Scholar]
- 34.Le DT, Uram JN, Wang H, Bartlett BR, Kemberling H, Eyring AD, et al. PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med. 2015;372:2509–20. doi: 10.1056/NEJMoa1500596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Marabelle A, Le DT, Ascierto PA, di Giacomo AM, de Jesus-Acosta A, Delord J-P, et al. Efficacy of pembrolizumab in patients with noncolorectal high microsatellite instability/mismatch repair–deficient cancer: results from the phase II KEYNOTE-158 study. J Clin Oncol. 2020;38:1–10. doi: 10.1200/JCO.19.02105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bang Y-J, Ruiz EY, van Cutsem E, Lee K-W, Wyrwicz L, Schenker M, et al. Phase III, randomised trial of avelumab versus physician’s choice of chemotherapy as third-line treatment of patients with advanced gastric or gastro-oesophageal junction cancer: primary analysis of JAVELIN Gastric 300. Ann Oncol. 2018;29:2052–60. doi: 10.1093/annonc/mdy264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shah MA, Kojima T, Hochhauser D, Enzinger P, Raimbourg J, Hollebecque A, et al. Efficacy and safety of pembrolizumab for heavily pretreated patients with advanced, metastatic adenocarcinoma or squamous cell carcinoma of the esophagus: the phase 2 KEYNOTE-180 study. JAMA Oncol. 2019;5:546–50. doi: 10.1001/jamaoncol.2018.5441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kudo T, Hamamoto Y, Kato K, Ura T, Kojima T, Tsushima T, et al. Nivolumab treatment for oesophageal squamous-cell carcinoma: an open-label, multicentre, phase 2 trial. Lancet Oncol. 2017;18:631–9. doi: 10.1016/S1470-2045(17)30181-X. [DOI] [PubMed] [Google Scholar]
- 39.Kojima T, Shah MA, Muro K, Francois E, Adenis A, Hsu C-H, et al. Randomized phase III KEYNOTE-181 study of pembrolizumab versus chemotherapy in advanced esophageal cancer. J Clin Oncol. 2020;38:4138–48. doi: 10.1200/JCO.20.01888. [DOI] [PubMed] [Google Scholar]
- 40.Chaganty BKR, Qiu S, Gest A, Lu Y, Ivan C, Calin GA, et al. Trastuzumab upregulates PD-L1 as a potential mechanism of trastuzumab resistance through engagement of immune effector cells and stimulation of IFNγ secretion. Cancer Lett. 2018;430:47–56. doi: 10.1016/j.canlet.2018.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Janjigian YY, Maron SB, Chatila WK, Millang B, Chavan SS, Alterman C, et al. First-line pembrolizumab and trastuzumab in HER2-positive oesophageal, gastric, or gastro-oesophageal junction cancer: an open-label, single-arm, phase 2 trial. Lancet Oncol. 2020;21:821–31. doi: 10.1016/S1470-2045(20)30169-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Janjigian YY, Kawazoe A, Yanez PE, Luo S, Lonardi S, Kolesnik O, et al. Pembrolizumab plus trastuzumab and chemotherapy for HER2+metastatic gastric or gastroesophageal junction (G/GEJ) cancer: Initial findings of the global phase 3 KEYNOTE-811 study. Wolters Kluwer Health; 2021.
- 43.Blum Murphy M, Xiao L, Patel VR, Maru DM, Correa AM, G. Amlashi F, et al. Pathological complete response in patients with esophageal cancer after the trimodality approach: The association with baseline variables and survival—The University of Texas MD Anderson Cancer Center experience. Cancer. 2017;123:4106–13. doi: 10.1002/cncr.30953. [DOI] [PubMed] [Google Scholar]
- 44.Kelly RJ, Ajani JA, Kuzdzal J, Zander T, van Cutsem E, Piessen G, et al. Adjuvant nivolumab in resected esophageal or gastroesophageal junction cancer. N Engl J Med. 2021;384:1191–203. doi: 10.1056/NEJMoa2032125. [DOI] [PubMed] [Google Scholar]
- 45.Kelly RJ, Ajani JA, Kuzdzal J, Zander T, van Cutsem E, Piessen G, et al. Adjuvant nivolumab in resected esophageal or gastroesophageal junction cancer following neoadjuvant chemoradiotherapy: expanded efficacy and safety analyses from CheckMate 577. ASCO; 2021.
- 46.Oba K, Paoletti X, Alberts S, Bang Y-J, Benedetti J, Bleiberg H, et al. Disease-free survival as a surrogate for overall survival in adjuvant trials of gastric cancer: a meta-analysis. J Natl Cancer Inst. 2013;105:1600–7. doi: 10.1093/jnci/djt270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ye J, Ji X, Dennis PA, Abdullah H, Mukhopadhyay P. Relationship between progression‐free survival, objective response rate, and overall survival in clinical trials of PD‐1/PD‐L1 immune checkpoint blockade: a meta‐analysis. Clin Pharmacol Ther. 2020;108:1274–88. doi: 10.1002/cpt.1956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Shah MA, Almhanna K, Iqbal S, Thakkar P, Schneider BJ, Yantiss R, et al. Multicenter, randomized phase II study of neoadjuvant pembrolizumab plus chemotherapy and chemoradiotherapy in esophageal adenocarcinoma (EAC). Wolters Kluwer Health; 2021.
- 49.Muro K, Chung HC, Shankaran V, Geva R, Catenacci D, Gupta S, et al. Pembrolizumab for patients with PD-L1-positive advanced gastric cancer (KEYNOTE-012): a multicentre, open-label, phase 1b trial. Lancet Oncol. 2016;17:717–26. doi: 10.1016/S1470-2045(16)00175-3. [DOI] [PubMed] [Google Scholar]
- 50.Park Y, Koh J, Na HY, Kwak Y, Lee K-W, Ahn S-H, et al. PD-L1 testing in gastric cancer by the combined positive score of the 22C3 PharmDx and SP263 assay with clinically relevant cut-offs. Cancer Res Treat. 2020;52:661. doi: 10.4143/crt.2019.718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhou KI, Peterson B, Serritella A, Thomas J, Reizine N, Moya S, et al. Spatial and temporal heterogeneity of PD-L1 expression and tumor mutational burden in gastroesophageal adenocarcinoma at baseline diagnosis and after chemotherapy. Clin Cancer Res. 2020;26:6453–63. doi: 10.1158/1078-0432.CCR-20-2085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Chao J, Fuchs CS, Shitara K, Tabernero J, Muro K, Van Cutsem E, et al. Assessment of pembrolizumab therapy for the treatment of microsatellite instability–high gastric or gastroesophageal junction cancer among patients in the KEYNOTE-059, KEYNOTE-061, and KEYNOTE-062 Clinical Trials. JAMA. 2021;7:895–902. doi: 10.1001/jamaoncol.2021.0275. [DOI] [PMC free article] [PubMed] [Google Scholar]



