Abstract
Immunotherapy using programmed cell death 1 (PD1) inhibitors has shown great efficacy in colorectal cancer patients harboring mismatch-repair-deficient (dMMR) and microsatellite instability-high (MSI-H) alterations. We previously showed a negative correlation of zymogen granule protein 16 (ZG16) with programmed death-ligand 1 (PD-L1) expression in patients with colorectal cancer. However, how ZG16 regulates PD-L1 expression is unclear. In this study, we showed that ZG16 can directly bind to glycosylated PD-L1 through its lectin domain, leading to PD-L1 degradation. Mutations on the lectin domain of ZG16 largely inhibit the interaction between ZG16 and PD-L1. Importantly, ZG16 overexpression suppressed tumor growth in two syngeneic mouse models through blockage of PD-L1 expression in cancer cells meanwhile suppression of PD1 expression in T cells. We also showed that ZG16 could improve the effect of chemotherapy and may be delivered as a protein to serve as an immune checkpoint inhibitor to promote T-cell mediated immunity.
Supplementary Information
The online version contains supplementary material available at 10.1186/s40364-022-00396-y.
Keywords: ZG16, PD-L1, PD1, Colorectal cancer, T cells
To the Editor,
Colorectal cancer (CRC) is a heterogeneous disease with a wide variety of genetic alterations, including mismatch-repair-deficient (dMMR) and microsatellite instability-high (MSI-H). Defects in MMR can lead to MSI-H, which can be found in many types of cancer. MSI-H or mismatch repair deficient (dMMR) tumors have an accumulation of errors in genetic sequences that are normally repeated (called microsatellites). Immunotherapy using checkpoint inhibitors that targets the PD-1/PD-L1 pathway, pembrolizumab, and nivolumab, has shown great efficacy in CRC patients harboring dMMR and MSI-H alterations, possibly attributable to a high level of immune checkpoint genes, including PD-1, PD-L1, CTLA-4, LAG-3, and IDO in such MSI-H tumors [1, 2]. Human zymogen granule protein 16 (ZG16) is highly expressed in mucus-secreting cells and characterized by a Jacalin-like lectin domain [3]. We previously showed a negative correlation of ZG16 with PD-L1 expression in patients with CRC and blockage of PD-L1 expression by ZG16 in CRC cells [4]. Surprisingly, we did not observe any change on the PD-L1 RNA after overexpression of ZG16, suggesting that PD-L1 is not regulated at transcriptional level [4]. So far, how ZG16 regulates PD-L1 expression is unclear.
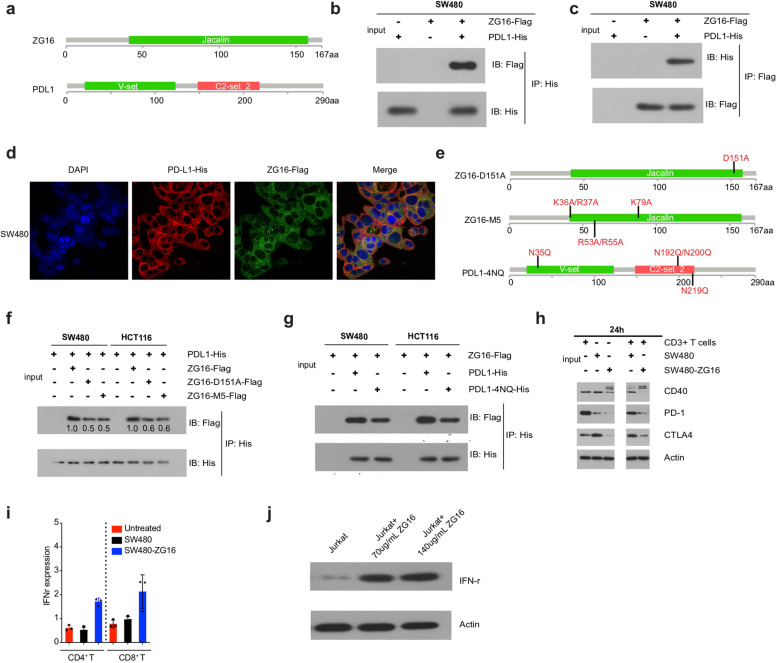
Studies have shown that the activity of PD-L1 is regulated by N-glycosylation, and targeting glycosylated PD-L1 (gPD-L1) by monoclonal antibody blocks PD-L1/PD-1 interaction resulting in PD-L1 degradation [5, 6]. Interestingly, ZG16 contains a lectin domain and lectins are carbohydrate-binding proteins that can specifically select for glycosylated proteins (Fig. 1a and Supplementary Fig. 1a) [7]. Based on these findings, we hypothesized that ZG16 may directly bind to glycosylated PD-L1 through its lectin domain, leading to PD-L1 degradation.
Fig. 1.
ZG16 binds to glycosylated PD-L1 through its lectin domain a Structure of ZG16 and PD-L1. b-c Co-immunoprecipitation of Flag-tagged ZG16 (ZG16-Flag) with His-tagged PD-L1 (PD-L1-His). Plasmid construct of ZG16-Flag was co-transfected with PD-L1-His into SW480 cells. Single vectors expressing each tag (Flag, His) were used as negative controls. d Immunofluorescence of SW480 cells transfected with ZG16-Flag and PD-L1-His, alone or in combination, for 24 h. Cells were subsequently stained with antibodies against His-tag (Red) and Flag-tag (Green) and DAPI (blue; nuclei). e Structure of ZG16-D151A, ZG16-M5, and PD-L1-4NQ f Co-immunoprecipitation of Flag-tagged ZG16 (ZG16-Flag, ZG16-D151A-Flag or ZG16-M5-Flag) with His-tagged PD-L1 (PD-L1-His). Plasmid constructs of ZG16-Flag, ZG16-D151A-Flag, or ZG16-M5-Flag were co-transfected with PD-L1-His into SW480 cells. Single vectors expressing each tag (Flag, His) were used as negative controls. g Co-immunoprecipitation of His-tagged PD-L1 (PDL1-His or PDL1-4NQ-His) with Flag-tagged ZG16 (ZG16-Flag). Plasmid constructs of PDL1-His or PDL1-4NQ-His were co-transfected with ZG16-Flag into SW480 or HCT118 cells. Single vectors expressing each tag (Flag, His) were used as negative controls. h Immunoblots of CD3+T cells cocultured withSW480 or SW480-ZG16 for 24 h. i INF-r expression of CD4+T cells or CD8+T cells cocultured withSW480 or SW480-ZG16. j Jurkat cells were treated with purified ZG16 protein for 48 h and then western blot was performed to detect IFNr expression
To test this hypothesis, we first build a 3D model using iCn3D to detect their interactions. The 3D modeling supported the interaction of ZG16 with PD-L1 (Supplementary Fig. 1b). Encouraged by these observations, we constructed two overexpression plasmids expressing Flag-tagged ZG16 and His-tagged PD-L1. To detect their direct binding in cells, we co-transfected Flag-tagged ZG16 plasmid and His-tagged PD-L1 plasmid into SW480 cells. qRT-PCR and western blot confirmed the overexpression of ZG16 and PD-L1 in SW480 cells after co-transfection (Supplementary Fig. 2a and b). We then performed a co-immunoprecipitation (co-IP) assay to detect the binding. Very surprisingly, we observed a direct binding between ZG16 and PD-L1 in SW480 cells co-transfected with two tagged plasmids (Fig. 1b and c). To exclude the possibility that this interaction between ZG16 and PD-L1 is cell line-specific, we performed co-IP assay in another colon cancer cell line HCT116 after co-transfection. Consistently, the direct interaction between ZG16 and PD-L1 was observed in HCT116 (Supplementary Fig. 3a and b), indicating that ZG16 binding to PD-L1 is not cell line-specific. To further confirm the direct interaction between ZG16 and PD-L1 in colon cells, we performed a colocalization assay in two different cell lines co-transfected with two plasmids expressing Flag-tagged ZG16 and His-tagged PD-L1. In consistency with the co-IP assay, we observed a direct binding between ZG16 and PD-L1 (Fig. 1d and Supplementary Fig. 3c). Together, these data suggest that ZG16 can directly bind to PD-L1 in colon cancer cells.
To determine whether the interaction between ZG16 and PD-L1 is dependent on the lectin domain, we constructed two Flag-tagged ZG16 overexpression plasmids which contain mutations in the lection domain. The first plasmid contains D151A mutation (ZG16-D151A) and the second plasmid contains 5 different mutations (ZG16-M5: K36A, R37A, K79A, R53A, and R55A). We then co-transfected the two plasmids expressing different mutant ZG16 with the plasmid expressing His-tagged PD-L1 into SW480 and HCT116 cells and performed co-IP (Fig. 1e). Very importantly, we observed a significant reduction of the binding between ZG16 and PD-L1 when the mutations were introduced into the lectin domain of ZG16, indicating that the lectin domain is required for the interaction (Fig. 1f and Supplementary Fig. 4a-b). The fact that the interaction was not completely blocked by the mutation suggests that more mutations may be required to completely eliminate the binding. In 2016, Chia-Wei Li and his colleagues demonstrated that PD-L1 glycosylation was completely ablated in the PD-L1 4NQ mutant5. To determine whether the binding is dependent on the glycosylation of PD-L1, we introduced 4 different mutations in PD-L1 (PD-L1-4NQ: N35Q, N192Q, N200Q, and N219Q) which has shown to affect the glycosylation of PD-L1 (Fig. 1e). Again, we observed the reduction of the binding between ZG16 and PD-L1 in the two cell lines with PD-L1 mutations, suggesting that glycosylation of PD-L1 is necessary for the binding (Fig. 1g and Supplementary Fig. 4c). Clearly, these data demonstrate that ZG16 can directly bind to glycosylated PD-L1 through its lectin domain.
As a secret protein primarily in the mucosa layer of the colon, we were also wondering if ZG16 is involved in the defense system, with a focus on the regulation of primary T cells. We investigated whether overexpression of ZG16 in colon cancer cells affects the gene expression of stimulatory and inhibitory checkpoint molecules, including CD40, PD1, and CTLA4. We co-cultured primary CD3+ T cells with ZG16 over-expressed SW480 cells (SW480-ZG16) for different time points. Western blots showed that additional isoforms of CD40 were induced, probably due to the ligand-binding or post-translational modification. Importantly, the expression of PD1 and CTLA4 was significantly decreased (Fig. 1h and Supplementary Fig. 5a). We observed an increased level of IFN-r in both CD4+ cells and CD8+T cells when co-cultured with the SW480-ZG16 cells (Fig. 1i and Supplementary Fig. 5b), suggesting that the T cells were activated. To confirm our finding, we treated Jurkat cells with purified ZG16 protein for 48 h and then performed FACS analysis to detect PD1 and CTLA4. Importantly, both expression of PD1 and CTLA4 were significantly decreased after ZG16 treatment in Jurkat cells (Figure S5c and S5d). in addition, we also performed western blot to detect IFNr expression in Jurkat cells. Consistently, IFNr expression were significantly increased after ZG16 treatment in Jurkat cells, indicating that the Jurkat cells were activated by ZG16 (Fig. 1j). These results support that ZG16 serves as an immune checkpoint inhibitor to activate the T cells by blocking the gene expression of PD1 and CTLA4.
Intrigued by our promising cell lines-based results, we further investigated in vivo efficacy of ZG16 overexpression in CRC xenografts. We subcutaneously implanted murine CT26 cells and CT26 cells with ZG16 overexpression (CT26-ZG16) into the right flank of female BALB/c mice to generate syngeneic mouse models. We followed these xenografts for 35 days. We found that ZG16 overexpression significantly suppressed tumor growth (Fig. 2a and b). Immunohistochemistry (IHC) analysis of residual tumors showed that the ZG16 overexpression resulted in more pronounced CD3 and significantly decreased PD-L1 and PD1 (Fig. 2c), suggesting that ZG16 overexpression blocked PD-L1 expression in cancer cells meanwhile stimulated T cell activation by suppression of PD1 expression, which in turn contributes to the inhibition of tumor growth.
Fig. 2.
ZG16 improves the effect of chemotherapy and promotes T-cell mediated immunity a Growth curve of CT26 and CT26-ZG16 xenografts (n = 6). Data are shown as mean ± s.d.. **P < 0.01 by two-way ANOVA with Tukey’s multiple comparisons test. b the Tumor volume of CT26 and CT26-ZG16 xenografts. c H& E and IHC analysis (ZG16, CD3, PD1, PD-L1) of CT26 and CT26-ZG16 xenografts. Scale bar = 400 μm. d-e Tumor volume and growth curve of MC38 and MC38-ZG16 xenografts treated with PBS or 5-FU(n = 3). Data are shown as mean ± s.d..*P < 0.05, ***P < 0.001 by two-way ANOVA with Tukey's multiple comparisons tests. f-g Tumor volume and growth curve of MC38 xenografts treated with 5-FU, ZG16 protein or their combination (n = 3). Data are shown as mean ± s.d.***P < 0.001 by two-way ANOVA with Tukey’s multiple comparisons test
Finally, we investigated whether ZG16 overexpression could improve the effectiveness of chemotherapy in CRC cancer xenografts. We subcutaneously implanted murine MC38 cells and MC38 cells with ZG16 overexpression (MC38-ZG16) into the right flank of female C57BL/6 mice. The MC38-ZG16 or MC38 xenografts were treated with PBS or 5-FU for indicated time points and the mice were monitored for 30 days. Notably, ZG16 overexpression significantly suppressed tumor growth and showed a similar effect as chemotherapy (Fig. 2d and Supplementary Fig. 6a, 7a). Most importantly, the combination of ZG16 overexpression and 5-FU resulted in significantly greater suppression of tumor growth (Fig. 2e).
As a potential immune checkpoint inhibitor, we investigated whether ZG16 can be functional when delivered as a protein. We purified ZG16 protein and then tested the in vivo efficacy of ZG16 protein delivered by tail vein injection in MC38 xenografts (Fig. 2f and Supplementary Fig. 6b). We found that ZG16 when delivered as protein significantly inhibited tumor growth (Fig. 2g and Supplementary Fig. 6b). We did not observe a synergistic effect between single agent and their combination, probably due to the low dose of ZG16 protein (Fig. 2g). To investigate whether the T cells were involved in the tumor suppression, we measured the percentage of both CD4+ T cells and CD8+ T cells in the tumor, spleen, and blood. We observed an increased number of CD4+ T cells and CD8+ T cells in the combination group (Supplementary Fig. 7b and c). Collectively, these results demonstrate that ZG16 could improve the effect of chemotherapy and may be delivered as a protein to serve as an immune checkpoint inhibitor to activate the T cells.
In conclusion, our study for the first time demonstrated that ZG16 can promote T-cell mediated immunity through direct binding to glycosylated PD-L1 in Colon Cancer. Overexpression of ZG16 significantly suppressed tumor growth and improved the effect of chemotherapy. Most importantly from a clinical standpoint, ZG16 can be delivered as a protein to activate the T cells by blocking the gene expression of PD1 and CTLA4. We envision that our findings may also be applied to other types of cancer. Our results may lead to the discovery of novel immune checkpoint inhibitors, which will provide new routes of immunotherapy for cancer treatment.
Supplementary Information
Acknowledgements
This study was supported by Henan Science and Technology Planning Project (142300410204) to HM.Project of Henan Science and Technology 2018 (project#2018020006) to HM.
Abbreviations
- ZG16
Zymogen granule protein 16
- CRC
Colorectal cancer
- PD-L1
Programmed cell death-ligand 1
- PD-L1
Programmed death-ligand 1
Authors' contributions
H. Meng designed and performed the experiments, analyzed data and wrote the manuscript. L. Wang analyzed the data and provided conceptual inputs. MZ Zhang performed data analysis. W Yao helped with animal experiments. YH Yin provided technical assistance of histological, YZ Li and Y Ding helped with the cell culture experiments. The author(s) read and approved the final manuscript.
Funding
This study was supported by Henan Science and Technology Planning Project (142300410204) to HM and Project of Henan Science and Technology 2018 (project#2018020006) to HM.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
All animal experiments were conducted with the approval of the Institutional Animal Care and Use Committee (First Affiliated Hospital of Zhengzhou university) (study number 2021-KY-0681–003). The design and performance of the study are in accordance with the Declaration of First Affiliated Hospital of Zhengzhou university. Signed informed consent was obtained from all participants before inclusion, allowing analysis of tumor tissue, blood samples and clinical data.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hui Meng and Mingzhi Zhang contributed equally to this work.
Contributor Information
Hui Meng, Email: menghui2000@zzu.edu.cn.
Mingzhi Zhang, Email: mingzhi_zhang@126.com.
References
- 1.Llosa NJ, Cruise M, Tam A, Wicks EC, Hechenbleikner EM, Taube JM, Blosser RL, Fan H, Wang H, Luber BS, et al. The vigorous immune microenvironment of microsatellite instable colon cancer is balanced by multiple counter-inhibitory checkpoints. Cancer Discov. 2015;5(1):43–51. doi: 10.1158/2159-8290.CD-14-0863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ganesh K, Stadler ZK, Cercek A, Mendelsohn RB, Shia J, Segal NH, Diaz LA., Jr Immunotherapy in colorectal cancer: rationale, challenges and potential. Nat Rev Gastroenterol Hepatol. 2019;16(6):361–375. doi: 10.1038/s41575-019-0126-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tateno H, Yabe R, Sato T, Shibazaki A, Shikanai T, Gonoi T, Narimatsu H, Hirabayashi J. Human ZG16p recognizes pathogenic fungi through non-self polyvalent mannose in the digestive system. Glycobiology. 2012;22(2):210–220. doi: 10.1093/glycob/cwr130. [DOI] [PubMed] [Google Scholar]
- 4.Meng H, Ding Y, Liu E, Li W, Wang L. ZG16 regulates PD-L1 expression and promotes local immunity in colon cancer. Transl Oncol. 2021;14(2):101003. doi: 10.1016/j.tranon.2020.101003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li CW, Lim SO, Chung EM, Kim YS, Park AH, Yao J, Cha JH, Xia W, Chan LC, Kim T, et al. Eradication of Triple-Negative Breast Cancer Cells by Targeting Glycosylated PD-L1. Cancer Cell. 2018;33(2):187–201. doi: 10.1016/j.ccell.2018.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li CW, Lim SO, Xia W, Lee HH, Chan LC, Kuo CW, Khoo KH, Chang SS, Cha JH, Kim T, et al. Glycosylation and stabilization of programmed death ligand-1 suppresses T-cell activity. Nat Commun. 2016;7:12632. doi: 10.1038/ncomms12632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Van Damme EJ. Lectins as tools to select for glycosylated proteins. Methods Mol Biol. 2011;753:289–297. doi: 10.1007/978-1-61779-148-2_19. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.