Non-pharmaceutical interventions (NPIs) and societal behavioural changes during the COVID-19 pandemic altered not only the spread of SARS-CoV-2, but also the predictable seasonal circulation patterns of many endemic viral illnesses in children.1 Before 2020, respiratory syncytial virus (RSV) and non-pandemic influenza viruses peaked in the winter in northern and southern hemispheres outside of tropical areas.2 In temperate climates, non-polio enteroviruses circulated in the summer to autumn3 in cyclical patterns.4 The COVID-19 pandemic has led to a departure from these patterns and, in many locations, usual circulation of these viruses was absent for more than a year only to resurge in unexpected ways. Past and present pandemic disruptions make it essential to prepare for further uncertainty in future endemic virus circulation among children.
After the relaxation of the major NPI measures in many settings, upper respiratory tract illnesses due to human rhinoviruses were first to re-emerge in many areas and persist5, 6 (appendix). There was no typical winter surge in hospitalisations related to RSV among children in 2020. As NPI measures were further relaxed, interseasonal RSV outbreaks began the following spring,7 with waves of disease affecting older than the typical median age for childhood RSV-associated respiratory infections.8 After global influenza circulation plummeted in early 2020, non-seasonal outbreaks of influenza A extended into the summer of 2022 in the northern hemisphere,9 while the Yamagata strain of influenza B remained absent.10 Expected seasonal non-polio enterovirus activity, including a predicted outbreak of enterovirus D68-associated acute flaccid myelitis,11 did not occur in 2020,12 with enterovirus D68 re-emerging in the autumn of 2021 in Europe outside established biennial cycles.13 Crucially, the patterns of these returning viral outbreaks have been heterogeneous across locations, populations, and pathogens, making predictions and preparations challenging.
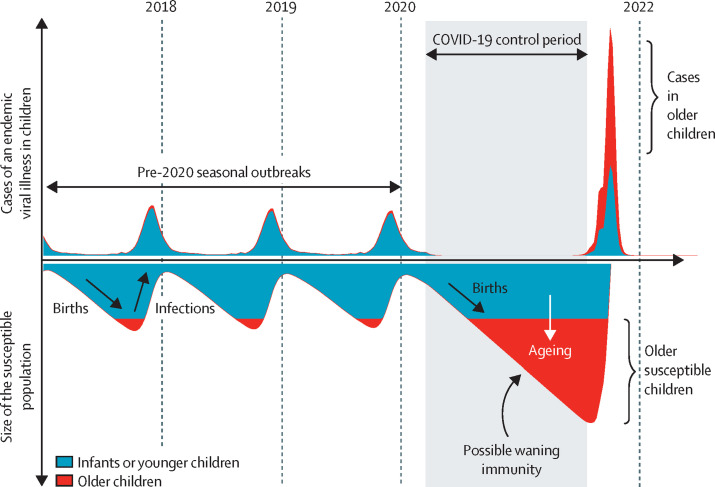
Although many infections and their associated morbidity and mortality were prevented by NPIs, decreased exposure to endemic viruses created an immunity gap—a group of susceptible individuals who avoided infection and therefore lack pathogen-specific immunity to protect against future infection. Decreases in childhood vaccinations with pandemic disruptions to health-care delivery contribute to this immunity gap for vaccine-preventable diseases, such as influenza, measles, and polio.14 The cumulative effect of new susceptible birth cohorts, waning immunity over time with decreased exposures to usual endemic viruses, and lagging vaccination rates in some settings widens this immunity gap and increases the potential for future outbreaks of endemic viruses (figure ).
Figure.
Modelling of endemic virus circulation in children following COVID-19 pandemic disruption
Schemata depicts the possible trajectory of seasonal outbreaks of cases of an endemic viral illness in children pre-2020 and post-2020 following COVID-19 control period with non-pharmaceutical interventions (NPIs) and alterations in societal behaviours due to the COVID-19 pandemic. Model is based on data from Baker and colleagues' study2 and age categorisations are illustrative. COVID-19 NPIs dampen transmission of endemic viruses leading to an immunity gap in a larger, older population of susceptible children that might change the age structure of return outbreaks.
Health systems must prepare for the potential of larger-than-typical non-seasonal outbreaks in the future due to larger susceptible populations of children being simultaneously exposed to endemic viruses. The size and timing of impending outbreaks of specific pathogens are difficult to predict because they depend on many dynamic factors, including the strength and duration of NPIs and pathogen seasonality and transmissibility. Despite moves to reduce or eliminate NPIs, the footprint of COVID-19 on societal behaviours, including ongoing mask wearing in public, increased work-from-home practices, and COVID-19 mitigation strategies in schools, is likely to remain to varying degrees for some time. Changes in NPIs or societal behaviours leading to even moderate alterations in future viral transmission rates could greatly impact the magnitude of future outbreaks of endemic viral illnesses in children.
Despite this dynamic variability, mathematical models allow qualitative predictions of ranges of possible future outbreak patterns. The ongoing natural experiment after unprecedented disruption during the COVID-19 pandemic provides a unique window of insight into understanding pathogen dynamics. Observed differences between pathogens, such as the relative impact of NPIs and the timing of re-emergence, might help elucidate the role of behavioural factors, climate, and immunity in driving the transmission of endemic viral illnesses among children. Narrowing the uncertainty in these epidemiological variables will be crucial to the precision and accuracy of future outbreak predictions.
Another likely effect of delayed circulation and the resultant immunity gap will be a temporary shift in the age distribution of endemic viral infections. Susceptible children might be exposed for the first time at older ages (figure). Decreased maternal exposures and immunity to usual endemic viruses,15 leading to a lack of transferred transplacental antibodies, might also leave young infants more vulnerable to viral infections. Age-related differences in disease presentations are likely to vary by pathogen. For example, the risk of severe RSV disease is higher in younger children,16 therefore delayed exposure might reduce the disease burden in children infected at older ages, whereas young infants unprotected by maternal antibodies might be more likely to develop severe disease. It remains to be seen whether delayed exposure to enterovirus D68 during the COVID-19 pandemic could lead to the increased risk of severe paralytic disease that was observed with increased age at primary infection with poliovirus.17
As the immunity gap closes with reduced use of NPIs leading to resumed viral circulation, many infections are likely to eventually return to pre-pandemic endemic patterns dictated by climate-driven seasonal forcing of transmission rates, thereby preventing out-of-season epidemics. With the re-emergence of these endemic viruses, further study is warranted to determine potential interactions with SARS-CoV-2, whether synergistic or antagonistic, and potential impacts on the severity of clinical presentations or circulation patterns.18, 19 Ongoing phylodynamic studies20 are needed to determine whether the bottlenecks of reduced genetic diversity being observed during the COVID-19 pandemic7, 10 will lead to persistent alterations in viral evolution, including elimination of certain strains, and the impact these evolutionary dynamics could have on future outbreaks.
Epidemiological modelling of the disrupted circulation of endemic viruses in children and the resulting immunity gap suggests that health-care systems must prepare for further uncertainty, including the possibility of larger outbreaks occurring out of season among older children and with atypical presentations. Preparedness depends on improved real-time pathogen and syndromic surveillance21 to provide early warnings to health-care systems of unexpected disease patterns. Seroepidemiological studies22 and host immunological surveillance23 should be used to assist public health authorities in anticipating outbreaks24 and researchers in developing countermeasures.25 Crucially, strengthened implementation of routine childhood immunisation programmes globally is needed to close the immunity gap in the safest way possible and prevent impacts of the COVID-19 pandemic from making children vulnerable to future epidemics of vaccine-preventable diseases.
This online publication has been corrected. The corrected version first appeared at thelancet.com on July 18, 2022
Acknowledgments
JRC serves in an unpaid position on the Board of Directors for the non-profit organisation Immunize Colorado. HN-T and KM receive funding from the US National Institute of Allergy and Infectious Diseases (NIAID) Vaccine Research Center for the Pandemic Response Repository through Microbial and Immune Surveillance and Epidemiology (PREMISE): Enterovirus D68 (EV-D68) Pilot Study. KM also receives grant funding from an NIAID K23 Patient Oriented Research Career Development Award. The other authors declare no competing interests. We thank Rachael Hemmert who provided the data for the supplementary appendix figure from the BIOFIRE Syndromic Trends program and Mark J Abzug and Samuel R Dominguez for their critical review and input to this Comment.
Supplementary Material
References
- 1.Nawrocki J, Olin K, Holdrege MC, et al. The effects of social distancing policies on non-SARS-CoV-2 respiratory pathogens. Open Forum Infect Dis. 2021;8 doi: 10.1093/ofid/ofab133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Baker RE, Park SW, Yang W, Vecchi GA, Metcalf CJE, Grenfell BT. The impact of COVID-19 nonpharmaceutical interventions on the future dynamics of endemic infections. Proc Natl Acad Sci USA. 2020;117:30547–30553. doi: 10.1073/pnas.2013182117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pons-Salort M, Oberste MS, Pallansch MA, et al. The seasonality of nonpolio enteroviruses in the United States: patterns and drivers. Proc Natl Acad Sci USA. 2018;115:3078–3083. doi: 10.1073/pnas.1721159115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pons-Salort M, Grassly NC. Serotype-specific immunity explains the incidence of diseases caused by human enteroviruses. Science. 2018;361:800–803. doi: 10.1126/science.aat6777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fong MW, Leung NHL, Cowling BJ, Wu P. Upper respiratory infections in schools and childcare centers reopening after COVID-19 dismissals, Hong Kong. Emerg Infect Dis. 2021;27:1525–1527. doi: 10.3201/eid2705.210277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rodgers L, Sheppard M, Smith A, et al. Changes in seasonal respiratory illnesses in the United States during the coronavirus disease 2019 (COVID-19) pandemic. Clin Infect Dis. 2021;73(suppl 1):S110–S117. doi: 10.1093/cid/ciab311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Eden JS, Sikazwe C, Xie R, et al. Off-season RSV epidemics in Australia after easing of COVID-19 restrictions. Nat Commun. 2022;13 doi: 10.1038/s41467-022-30485-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Foley DA, Phuong LK, Peplinski J, et al. Examining the interseasonal resurgence of respiratory syncytial virus in Western Australia. Arch Dis Childhood. 2022;107:e7. doi: 10.1136/archdischild-2021-322507. [DOI] [PubMed] [Google Scholar]
- 9.Kuitunen I, Artama M, Haapanen M, Renko M. Respiratory virus circulation in children after relaxation of COVID-19 restrictions in fall 2021—a nationwide register study in Finland. J Med Virol. 2022 doi: 10.1002/jmv.27857. published online May 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dhanasekaran V, Sullivan S, Edwards KM, et al. Human seasonal influenza under COVID-19 and the potential consequences of influenza lineage elimination. Nat Commun. 2022;13 doi: 10.1038/s41467-022-29402-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Park SW, Pons-Salort M, Messacar K, et al. Epidemiological dynamics of enterovirus D68 in the United States and implications for acute flaccid myelitis. Sci Transl Med. 2021;13 doi: 10.1126/scitranslmed.abd2400. [DOI] [PubMed] [Google Scholar]
- 12.Kuo SC, Tsou HH, Wu HY, et al. Nonpolio enterovirus activity during the COVID-19 pandemic, Taiwan, 2020. Emerg Infect Dis. 2021;27:306–308. doi: 10.3201/eid2701.203394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Benschop KS, Albert J, Anton A, et al. Re-emergence of enterovirus D68 in Europe after easing the COVID-19 lockdown, September 2021. Euro Surveill. 2021;26 doi: 10.2807/1560-7917.ES.2021.26.45.2100998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.SeyedAlinaghi S, Karimi A, Mojdeganlou H, et al. Impact of COVID-19 pandemic on routine vaccination coverage of children and adolescents: a systematic review. Health Sci Rep. 2022;5 doi: 10.1002/hsr2.516. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 15.Reicherz F, Xu RY, Abu-Raya B, et al. Waning immunity against respiratory syncytial virus during the COVID-19 pandemic. J Infect Dis. 2022 doi: 10.1093/infdis/jiac192. published online May 7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Johannesen CK, van Wijhe M, Tong S, et al. Age-specific estimates of respiratory syncytial virus-associated hospitalizations in 6 European countries: a time series analysis. J Infect Dis. 2022 doi: 10.1093/infdis/jiac150. published online June 24. [DOI] [PubMed] [Google Scholar]
- 17.US Centers for Disease Control and Prevention . 14th edn. Public Health Foundation; Washington, DC: 2021. Epidemiology and prevention of vaccine-preventable diseases. [Google Scholar]
- 18.Pinky L, Dobrovolny HM. Epidemiological consequences of viral interference: a mathematical modeling study of two interacting viruses. Front Microbiol. 2022;13 doi: 10.3389/fmicb.2022.830423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Piret J, Boivin G. Viral interference between respiratory viruses. Emerg Infect Dis. 2022;28:273–281. doi: 10.3201/eid2802.211727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Grenfell BT, Pybus OG, Gog JR, et al. Unifying the epidemiological and evolutionary dynamics of pathogens. Science. 2004;303:327–332. doi: 10.1126/science.1090727. [DOI] [PubMed] [Google Scholar]
- 21.Meyers L, Ginocchio CC, Faucett AN, et al. Automated real-time collection of pathogen-specific diagnostic data: syndromic infectious disease epidemiology. JMIR Public Health Surveill. 2018;4:e59. doi: 10.2196/publichealth.9876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Metcalf CJE, Farrar J, Cutts FT, et al. Use of serological surveys to generate key insights into the changing global landscape of infectious disease. Lancet. 2016;388:728–730. doi: 10.1016/S0140-6736(16)30164-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mina MJ, Metcalf CJE, McDermott AB, Douek DC, Farrar J, Grenfell BT. A global immunological observatory to meet a time of pandemics. Elife. 2020;9 doi: 10.7554/eLife.58989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Metcalf CJE, Wesolowski A, Winter AK, et al. Using serology to anticipate measles post-honeymoon period outbreaks. Trends Microbiol. 2020;28:597–600. doi: 10.1016/j.tim.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nguyen-Tran H, Park SW, Messacar K, et al. Enterovirus D68: a test case for the use of immunological surveillance to develop tools to mitigate the pandemic potential of emerging pathogens. Lancet Microbe. 2022;3:e83–e85. doi: 10.1016/S2666-5247(21)00312-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.