Abstract
Background
While laparoscopic liver surgery has become a standard procedure, experience with robotic liver surgery is still limited. The aim of this prospective study was to evaluate safety and feasibility of robotic liver surgery and compare outcomes with conventional laparoscopy.
Methods
We here report the results of a single-center, prospective, post-marketing observational study (DRKS00017229) investigating the safety and feasibility of robotic liver surgery. Baseline characteristics, surgical complexity (using the IWATE score), and postoperative outcomes were then compared to laparoscopic liver resections performed at our center between January 2015 and December 2020. A propensity score-based matching (PSM) was applied to control for selection bias.
Results
One hundred twenty nine robotic liver resections were performed using the da Vinci Xi surgical system (Intuitive) in this prospective study and were compared to 471 consecutive laparoscopic liver resections. After PSM, both groups comprised 129 cases with similar baseline characteristics and surgical complexity. There were no significant differences in intraoperative variables, such as need for red blood cell transfusion, duration of surgery, or conversion to open surgery. Postoperative complications were comparable after robotic and laparoscopic surgery (Clavien–Dindo ≥ 3a: 23% vs. 19%, p = 0.625); however, there were more bile leakages grade B–C in the robotic group (17% vs. 7%, p = 0.006). Length of stay and oncological short-term outcomes were comparable.
Conclusions
We propose robotic liver resection as a safe and feasible alternative to established laparoscopic techniques. The object of future studies must be to define interventions where robotic techniques are superior to conventional laparoscopy.
Keywords: Robotic liver surgery, Laparoscopic liver surgery, Surgical robot, Robotic surgery
The advantages of the minimally invasive approach to liver resection have been demonstrated in numerous studies [1–3]. Compared to the conventional open approach, laparoscopic liver surgery is associated with a lower complication rate, less postoperative pain, shorter hospital stay and a higher quality of life [2, 4]. According to a recent randomized trial, these benefits can be achieved without compromising oncologic long-term results [5]. Laparoscopy has therefore become the standard technique at our center and many others worldwide, with conventional open liver surgery being reserved for extended indications, e.g., resections with vascular or biliary reconstruction.
By enabling 3-D visualization and permitting a greater range of motion, robotic liver surgery has been reported to improve surgeon ergonomics, surgical accuracy and to reduce surgeon fatigue [6, 7]. In this respect, the robotic technique might be particularly suitable for highly complex liver resections and could offer potential advantages over laparoscopy. Even though the first case series on robotic liver surgery were published back in the early 2000s, the robotic technique has surprisingly not spread widely [6, 7]. This is certainly associated, at least in part, with the high initial costs of robotic surgical systems and the high running costs [8, 9].
However, interest in implementing robotic techniques in liver surgery has increased dramatically in the last few years. Since recent series have produced promising results, it is now time to safely accompany the development of robotic liver surgery within the framework of prospective studies [10–13]. Suggested benefits need to be verified and validated, especially with regard to an improved case selection and potential technical limitations that may require further refinement and innovation for robotic surgical systems. In this study, we, therefore, present our data from a prospective single-center observational study investigating the safety and feasibility of robotic liver surgery and compare results to established laparoscopic techniques.
Materials and methods
Study design and data collection
We here report the results of a single-center, prospective, post-marketing observational study (DRKS00017229) with the objective of investigating clinical outcomes of robotic liver surgery using the da Vinci Xi surgical system (Intuitive, Sunnyvale, CA, USA). All patients who underwent robotic liver resection at the Department of Surgery, Campus Charité Mitte and Campus Virchow-Klinikum, Charité-Universitätsmedizin Berlin, Germany, between July 31st, 2017 and December 15th, 2020, were included. Baseline characteristics, intraoperative technical details including dissection devices, duration of surgery and console time, as well as postoperative complications were prospectively recorded and analyzed.
All included patients gave informed consent to the collection of their personal and medical data and its use for research purposes. All data were collected, stored, and processed according to the General Data Protection Regulation and local data protection laws. The study was conducted in accord with the ethical standards of the Helsinki Declaration of 1975. The Charité Institutional Review Board (IRB) approved of the study (EA4/084/17).
In a second step, baseline characteristics, surgical complexity and postoperative outcomes of all consecutive laparoscopic liver resections performed at our center between January 2015 and December 2020 were retrospectively analyzed and compared to the robotic approach. Parts of these data have been published previously [14, 15]. The clinical data were handled according to the same ethical and data protection guidelines after prior IRB approval (EA2/006/16). A propensity score-based matching (PSM) was performed to control for selection bias.
Patient selection and perioperative management
Liver resections were preferentially scheduled for minimally invasive surgery in the absence of specific reasons for open resection. These could be patient’s preference or technical issues, e.g., the need for vascular or biliary reconstruction. Before the surgical robot was installed in our program and this prospective observational study was started, all minimally invasive liver resections were performed laparoscopically. After obtaining initial experience with minor resections, the robotic approach was increasingly applied for all extents of resection. The decision to use laparoscopic or robotic approach was not based on defined selection criteria. Preoperative staging included helical computed tomography (CT) scanning of the chest, abdomen and pelvis, which was individually supplemented or replaced by magnetic resonance imaging (MRI) of the liver. All cases of suspected primary or secondary malignant tumors were discussed at one of our weekly multidisciplinary tumor boards. Before extended resection, liver augmentation was induced as individually indicated.
Surgical techniques
Laparoscopic liver resection was performed as reported previously [14–16]. Port strategies included multi-incisional laparoscopic surgery (MILS) and in the early phase single incision laparoscopic surgery (SILS) and hand-assisted laparoscopic surgery (HALS).
For robotic surgery, the da Vinci Xi surgical system (Intuitive, Sunnyvale, CA, USA) was used. Patients were positioned in reverse Trendelenburg position. Four 8 mm robotic trocars were supplemented by two 12 mm (± one 5 mm) assist trocars. A standard operating procedure for the positioning of the patient, port placement, setting of the robotic arms and differences to the laparoscopic approach has been described by our group in detail elsewhere [17].
In both laparoscopic and robotic surgery, ultrasound was routinely performed intraoperatively to confirm the exact tumor location, boarders, proximity to vascular and biliary structures and to rule out further intrahepatic lesions. In preparation for an extracorporeal intermittent Pringle maneuver (IPM), the hepatoduodenal ligament was isolated and threaded, and 250 mg methylprednisolone was administered intravenously prior to parenchymal resection. IPM was applied according to the surgeon's individual decision with 15-min intervals interrupted by 5 min of reperfusion each. In case of iCC, hilar lymph node dissection and intraoperative frozen section analysis were performed routinely, irrespective of the access technique.
In laparoscopy, superficial parenchymal dissection was performed by using energy shears (Harmonic ACE, Ethicon, Inc., Somerville, NJ, USA and THUNDERBEAT, Olympus K.K., Tokyo, Japan). Options for deeper parenchymal dissection included laparoscopic cavitron ultrasonic surgical aspirator (CUSA, Valleylab Boulder, CO, USA), waterjet (ERBEJET, Elektromedizin GmbH, Tübingen, Germany) and in some cases during the learning curve also vascular staplers (Echelon, Ethicon, Somerville, NJ, USA). As these options are not available for use with the da Vinci Xi surgical system, parenchymal transection was performed in a modified clamp crush technique using Harmonic ACE curved shears. Both in laparoscopic and robotic liver surgery, large vessels were either clipped or transected using staplers.
Routinely, a drain was inserted in the abdomen, which was removed on the second postoperative day in case of normal fluids. Patients with liver cirrhosis and after major resections were referred to the intensive care unit (ICU) postoperatively, while individual decisions were made for patients after minor resections.
Clinical parameters and classifications
The preoperative general status of our patients was assessed using the American Society for Anesthesiologists physical status classification (ASA). The underlying pathology of the resected tumor was determined by the staff pathologist in all cases. In case of a malignant tumor, TNM status and resection margin (R status) were specified. Presence of liver fibrosis was also described by histopathology and graded according to Desmet et al. [18]. Furthermore, clinical stage of liver cirrhosis was classified according to the Child–Pugh score [19].
The assessment of surgical complexity was based on the IWATE criteria [20] and the extent of resection (major resection vs. minor resection). Major resection was defined as resection of three segments or more [21]. Furthermore, tumor size (< 3 cm vs. ≥ 3 cm) and tumor location were reported as categorized by the IWATE classification system. Here, liver segments are grouped by the difficulty of access and awarded between one and five points within the scoring system [22].
Postoperative complications within 90 days after surgery were classified according to Clavien–Dindo [23]. Bile leakage was graded according to the International Study Group of Liver Surgery (ISGLS) definition [24]. Further intra- and postoperative outcome parameters included duration of surgery, intraoperative need for transfusion of red blood cells (RBCs), length of stay in the ICU and length of stay in the hospital (LOS). Serum levels of bilirubin, alanine aminotransferase (ALT), aspartate aminotransferase (AST), bilirubin and the international normalized ratio (INR) are reported as preoperatively, on the first postoperative day and the last value before discharge.
Statistical analysis
IBM SPSS Statistics version 25 (IBM Corp., Armonk, NY, USA) and R version 4.03 (R Foundation for Statistical Computing, Vienna, Austria) were used for data analysis. Categorical data were analyzed using Pearson’s χ2 test and reported as frequencies and percentages. Continuous data were tested for normality using the Shapiro–Wilk test and analyzed accordingly by Mann–Whitney U test or Student’s t test. Nonparametric data are presented as median and range (minimum–maximum), parametric data are reported as mean and standard deviation. A p value < 0.05 was considered statistically significant. The propensity score matching was performed with R using the package “Matchit”, with nearest neighbor one to one matching, without replacement and a caliper of 0.2. The following variables were included in the propensity score model: tumor size ≥ 3 cm, tumor location according to IWATE score, extent of resection (major vs. minor), presence of liver cirrhosis, body mass index and severe systemic disease (ASA ≥ 3).
Results
Prospective analysis of robotic liver resections
From the first robotic liver resection in April 2018 until the end of our study in December 2020, 129 liver resections were performed using the da Vinci Xi surgical system and included in our prospective observational study. Most patients (81%) had malignant primary or secondary liver tumors. The most common tumor entity was colorectal liver metastasis (CRLM, 28%), followed by hepatocellular carcinoma (HCC, 23%) and intrahepatic cholangiocarcinoma (iCC, 12%). All baseline characteristics are described in Table 1.
Table 1.
Baseline characteristics before and after propensity score matching
| Pre-PSM | Post-PSM | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Laparoscopic | Robotic | p value | Laparoscopic | Robotic | p value | |||||
| n = 471 | n = 129 | n = 129 | n = 129 | |||||||
| Age (years) | 62 | (19–88) | 64 | (22–85) | 0.836 | 62 | (19–86) | 64 | (22–85) | 0.802 |
| Sex (female) | 211 | (45%) | 63 | (49%) | 0.426 | 59 | (46%) | 63 | (49%) | 0.708 |
| BMI (kg/m2) | 26 | (17–44) | 25 | (17–40) | 0.201 | 25 | (17–44) | 25 | (17–40) | 0.613 |
| ASA | ||||||||||
| < 3 | 246 | (52%) | 65 | (50%) | 0.766 | 58 | (45%) | 65 | (50%) | 0.455 |
| ≥ 3 | 225 | (48%) | 64 | (50%) | 71 | (55%) | 64 | (50%) | ||
| Liver cirrhosis | ||||||||||
| No cirrhosis | 366 | (78%) | 112 | (87%) | 0.052 | 112 | (87%) | 112 | (87%) | 0.597 |
| Child–Pugh A | 99 | (21%) | 17 | (13%) | 16 | (12%) | 17 | (13%) | ||
| Child–Pugh B | 5 | (1%) | 0 | 1 | (1%) | 0 | ||||
| Pathology | ||||||||||
| HCC | 121 | (26%) | 30 | (23%) | 0.057 | 26 | (20%) | 30 | (23%) | 0.359 |
| CRLM | 166 | (35%) | 36 | (28%) | 51 | (40%) | 36 | (28%) | ||
| iCC | 31 | (6%) | 16 | (12%) | 12 | (9%) | 16 | (12%) | ||
| Other-malignant | 56 | (12%) | 23 | (18%) | 17 | (13%) | 23 | (18%) | ||
| Benign | 97 | (21%) | 24 | (19%) | 23 | (18%) | 24 | (19%) | ||
Pre-PSM all patients before propensity score matching, Post-PSM after propensity score matching, BMI body mass index, ASA American Society for Anesthesiologists physical status classification, HCC hepatocellular carcinoma, CRLM colorectal liver metastasis, iCC intrahepatic cholangiocarcinoma
Roughly half (47%) of the robotic liver resections were major resections (Table 2). According to the IWATE classification, the median surgical difficulty score was nine, corresponding to “advanced” difficulty, with 36% of cases classified as > 10, corresponding to “expert” level [20].
Table 2.
Complexity of surgery before and after propensity score matching
| Pre-PSM | Post-PSM | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Laparoscopic | Robotic | p value | Laparoscopic | Robotic | p value | ||||||
| n = 471 | n = 129 | n = 129 | n = 129 | ||||||||
| Extent of resection | |||||||||||
| Minor | 347 | (74%) | 68 | (53%) | < 0.001 | 68 | (53%) | 68 | (53%) | 1.000 | |
| Major | Right | 87 | (18%) | 40 | (31%) | 40 | (31%) | 40 | (31%) | ||
| Left | 37 | (8%) | 21 | (16%) | 21 | (16%) | 21 | (16%) | |||
| IWATE score | 7 | (1–11) | 9 | (1–12) | < 0.001 | 8 | (2–11) | 9 | (1–12) | 0.337 | |
| Tumor ≥ 3 cm | 248 | (53%) | 89 | (69%) | 0.001 | 84 | (65%) | 89 | (69%) | 0.596 | |
| Location score (IWATE) | |||||||||||
| 1 | Segment III | 22 | (5%) | 3 | (2%) | 0.001 | 4 | (3%) | 3 | (2%) | 0.257 |
| 2 | Segment II/VI | 99 | (21%) | 12 | (9%) | 19 | (15%) | 12 | (9%) | ||
| 3 | Segment IVb/V | 67 | (14%) | 24 | (19%) | 16 | (12%) | 24 | (19%) | ||
| 4 | Segment I/IVa | 45 | (10%) | 25 | (19%) | 17 | (13%) | 25 | (19%) | ||
| 5 | Segment VII/VIII | 238 | (50%) | 65 | (50%) | 51 | (57%) | 65 | (50%) | ||
p values <0.05 are indicates in italic
Pre-PSM all patients before propensity score matching. Post-PSM after propensity score matching
The median duration of surgery was 260 min (83–568), with 167 min median console time (47–384). In 56% of the cases, IPM was applied, with a median total duration of 16 min (3–68) (Table 3). For 37%, a vascular stapler was used for transection of large vessels. In seven cases (5%), the surgery was converted to open or laparoscopic technique. Six patients received RBC intraoperatively (5%). Seventy-six percent of cases with malignant tumors were resected with histological declaration of tumor-free margin (R0).
Table 3.
Intraoperative and postoperative outcome criteria before and after propensity score matching
| Pre-PSM | Post-PSM | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Laparoscopic | Robotic | p value | Laparoscopic | Robotic | p value | |||||
| n = 471 | n = 129 | n = 129 | n = 129 | |||||||
| Duration of surgery (min) | 244 | (45–758) | 260 | (83–568) | 0.135 | 270 | (57–580) | 260 | (83–568) | 0.613 |
| Port strategy | ||||||||||
| SILS | 21 | (4%) | 3 | (2%) | ||||||
| HALS | 106 | (23%) | 35 | (27%) | ||||||
| Laparoscopic MILS | 339 | (73%) | 91 | (71%) | ||||||
| Robotic MILS | 129 | (100%) | 129 | (100%) | ||||||
| Use of IPM | 219 | (47%) | 72 | (56%) | 0.084 | 70 | (54%) | 72 | (56%) | 0.900 |
| Total duration of IPM | 25 | (5–113) | 16 | (3–68) | 0.003 | 26 | (5–113) | 16 | (3–68) | 0.050 |
| Conversion | 16 | (3%) | 7 | (5%) | 0.302 | 6 | (5%) | 7 | (5%) | 1.000 |
| Intraoperative RBC transfusion | 35 | (8%) | 6 | (5%) | 0.327 | 11 | (9%) | 6 | (5%) | 0.315 |
| LOS: ICU (days) | 1 | (0–43) | 1 | (0–81) | 0.125 | 1 | (0–24) | 1 | (0–81) | 0.405 |
| LOS: hospital (days) | 8 | (3–59) | 8 | (4–94) | 0.471 | 8 | (3–52) | 8 | (4–94) | 0.816 |
| R0 resection | 302 | (81%) | 81 | (76%) | 0.118 | 87 | (82%) | 81 | (76%) | 0.407 |
| 90-day complicationsa | ||||||||||
| None | 296 | (63%) | 80 | (62%) | 83 | (64%) | 80 | (62%) | ||
| 1–2 | 103 | (22%) | 19 | (15%) | 0.042 | 22 | (17%) | 19 | (15%) | 0.625 |
| 3–5 | 72 | (15%) | 30 | (23%) | 24 | (19%) | 30 | (23%) | ||
| Mortality | 5 | (1%) | 1 | (1%) | 1.000 | 2 | (2%) | 1 | (1%) | 1.000 |
| Bile leakage | 49 | (10%) | 22 | (17%) | 0.045 | 15 | (12%) | 22 | (17%) | 0.286 |
| Bile leakage: Gradeb | ||||||||||
| None | 422 | (90%) | 107 | (83%) | < 0.001 | 114 | (88%) | 107 | (83%) | 0.006 |
| A | 24 | (5%) | 0 | 6 | (5%) | 0 | ||||
| B | 21 | (5%) | 20 | (15%) | 7 | (5%) | 20 | (15%) | ||
| C | 4 | (1%) | 2 | (2%) | 2 | (2%) | 2 | (2%) | ||
p values <0.05 are indicates in italic
Pre-PSM all patients before propensity score matching, Post-PSM after propensity score matching, SILS single incision laparoscopic surgery, HALS hand-assisted laparoscopic surgery, MILS multi-incisional laparoscopic surgery, RBC red blood cells, ICU intensive care unit, LOS length of stay, IPM Intermittent Pringle Maneuver
aAccording to Clavien–Dindo classification [23]
bAccording to the International Study Group of Liver Surgery definition [24]
After robotic liver resections, patients spent a median of one night at the ICU, and were discharged from hospital after a median of 8 days. Ninety-day postoperative overall morbidity was 38%, with severe complications (Clavien–Dindo ≥ 3) occurring in 23% of all cases. The most frequent type of complication was bile leakage, which was detected in 22 cases (17%, Table 3). Two of these cases required surgery, classifying as grade C bile leakage, while the remaining 20 cases were grade B. One patient (1%) died after robotic liver resection. This cirrhotic patient, who also had a pre-existing pulmonary disease, suffered a bile leakage grade C after right hepatectomy and subsequently developed pneumonia and multiorgan failure. All severe complications (Clavien–Dindo ≥ 3) are listed in Table 4.
Table 4.
Severe complications according to Clavien–Dindo classification (≥ 3)
| Post-PSM | ||||
|---|---|---|---|---|
| Laparoscopic | Robotic | |||
| n = 24 | n = 30 | |||
| Bile leakage | 8 | (34%) | 17 | (57%) |
| Abdominal fluid collection | 5 | (21%) | 6 | (20%) |
| Pleural effusion | 3 | (13%) | ||
| Pneumothorax | 1 | (3%) | ||
| Intestinal perforation | 1 | (4%) | 1 | (3%) |
| Fascial dehiscence | 2 | (8%) | 1 | (3%) |
| Hematoma | 2 | (8%) | 1 | (3%) |
| Acute pancreatitis | 1 | (3%) | ||
| Wound infection | 1 | (4%) | 1 | (3%) |
| Multi-organ failure, death | 2 | (8%) | 1 | (3%) |
Robotic vs. laparoscopic liver surgery: baseline characteristics
Between January 2015 and December 2020, 471 laparoscopic liver resections were performed at our center. All clinical data were retrospectively analyzed and compared with patients who underwent robotic liver resection. A PSM was performed to control for selection bias and potential confounding factors, including denominators of surgical difficulty (tumor size, tumor location score and extent of resection) and baseline characteristics (BMI, ASA score and presence of liver cirrhosis). After matching, 129 cases were included in each group.
Baseline characteristics were similar in patients who underwent laparoscopic and those who were scheduled for robotic surgery (Table 1). Eighty percent of all patients were operated for malignant and 20% for benign liver diseases, with no differences between both groups. Before matching, there was a trend toward more CRLM in the laparoscopic and more iCC in the robotic group, with liver cirrhosis less commonly seen in the robotic group; however, those differences were short of statistical significance. After PSM, these parameters were similar in both groups.
Robotic vs. laparoscopic liver surgery: surgical complexity
When comparing all minimally invasive liver resections before matching, those in the robotic group were considerably more extensive and complex when compared to the laparoscopic group (Table 2). Forty-seven percent of robotic liver resections were major resections, compared to 26% in the laparoscopic group (p < 0.001). The median IWATE score as a measurement for surgical complexity was nine for robotic and seven for laparoscopic resections (p < 0.001). The percentage of large tumors (defined as ≥ 3 cm) was significantly higher in the robotic group than in the laparoscopic group (69% vs. 53%, p = 0.001). Regarding the tumor location score of the IWATE criteria, robotic liver surgeries were more likely performed for tumors in the segments classified as more complex (3–4 points: segments I, IV or V), while laparoscopically resected tumors were more often in the lower graded segments (1–2 points: segments II, III or VI).
Robotic vs. laparoscopic liver surgery: Intraoperative outcome
After PSM, duration of surgery was comparable between both surgical approaches (robotic 260 vs. laparoscopic 270 min, n.s.). Intraoperative need for RBC transfusion was also similar in both groups (5% robotic vs. 9% laparoscopic, n.s.). Rates of conversion to open (or laparoscopic, in case of robotic) surgery were 5% during both surgical approaches. The resection status in malignant tumors was similar in both groups with 76% R0 status in the robotic and 82% in the laparoscopic group. Intraoperative and postoperative outcome criteria after robotic vs. laparoscopic liver resection both unmatched and after PSM are detailed in Table 3.
Robotic vs. laparoscopic liver surgery: postoperative outcome
After surgery, median LOS was similar in both groups with one night at the ICU and 8 days in the hospital. After matching for surgical complexity, both overall morbidity and severity of complications were comparable in the robotic and laparoscopic groups.
The overall incidence of bile leakage was similar after robotic and laparoscopic liver resections after PSM (12% vs. 17%, n.s.). However, when considering the severity according to ISGLS criteria, cases of bile leakage after robotic surgery were graded significantly higher than in the laparoscopic group. After PSM, this difference remained significant with all cases in the robotic group graded B or C compared to 9 out of 15 (60%) in the laparoscopic group (p = 0.006), representing the only notable distinction in measured outcome criteria between the robotic and the laparoscopic surgical approaches. When classifying severe bile leakage (B–C) according to localization, 77% of bile leakage after robotic surgery were localized in the parenchymal dissection plane vs. 67% after laparoscopic resection (n.s.).
The causes and frequencies of severe complications (Clavien–Dindo ≥ 3a) of both groups after PSM are detailed in Table 4.
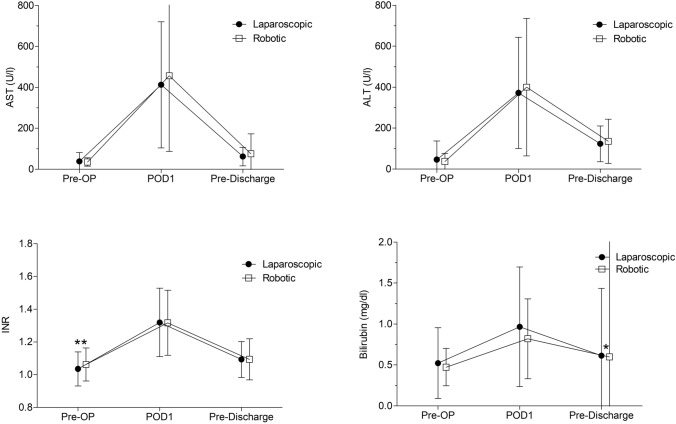
There were similar levels of serum markers of liver injury and liver function (ALT, AST, INR, bilirubin) preoperatively, with a slightly higher INR in the robotic group (1.06 robotic vs. 1.04 laparoscopic, p = 0.006, Fig. 1). On the first postoperative day there was no difference between groups. AST, ALT or INR also did not show any difference in the last test before release from the hospital. However, serum bilirubin was slightly, but statistically significantly lower in the robotic group at this time point, when compared to the laparoscopic group (0.60 mg/dl vs. 0.61 mg/dl, p = 0.034).
Fig. 1.
Serum levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and bilirubin and international normalized ratio (INR) preoperatively (Pre-OP), on the first postoperative day (POD1), and before discharge from the hospital (Pre-Discharge). U/l units per liter, mg/dl milligram per deciliter. *p < 0.05, **p < 0.01
Discussion
We here report our center’s experience in robotic liver surgery with the da Vinci Xi system as investigated in a prospective study. In synopsis with other series, our data indicate that robotic liver surgery is technically feasible and that implementation at a center for minimally invasive hepatopancreatobiliary surgery is safely possible within a short period of time [9, 25, 26]. The high average complexity of the interventions with around fifty percent major liver resections suggests that robotic assistance may be particularly advantageous in technically demanding procedures. Compared to our retrospective data from laparoscopic liver surgery, there are no clinically relevant differences in terms of overall perioperative morbidity or short-term oncologic outcomes.
The implementation of robotic liver surgery built on our experience of 471 laparoscopic liver resections since 2015. Profound technical and strategic experience in laparoscopic liver surgery was helpful in safely implementing the robotic program at our center. Well-known advantages of robotic assistance, such as 3-D visualization or instrument triangulation, were another important factor to safely switch from laparoscopic to robotic liver surgery and quickly led to a preference of the da Vinci Xi system especially for large tumors and complex resections. Accordingly, both the percentage of major liver resections and surgical complexity were higher in the robotic than in the laparoscopic group before matching.
Other groups also reported the perceived benefits of the robotic technique; however, they do not appear to translate into improved short-term outcomes compared to established minimally invasive techniques. Both the intraoperative and postoperative metrics are in the range of what previous studies have described for robotic liver surgery [25, 27]. In accordance, there were no significant perioperative improvements to our data obtained in laparoscopic liver surgery. Intraoperative characteristics such as duration of surgery, need for RBC transfusion or conversion to open surgery were comparable between both groups. Further, no significant differences to laparoscopic liver surgery were found with regard to postoperative complication rates, hospital stay, and short-term oncologic data.
While some other groups report similarly increased risk of bile leakage with robotic surgery [27, 28], a recent meta-analysis comprising 26 non-randomized studies found no significant difference to laparoscopic surgery [29]. In our study, bile leakage was more likely to be grade B or C after robotic liver surgery when compared to the laparoscopic group. This is an important finding, as these complications might require interventional or surgical treatment. It is conceivable that the transection technique in robotic surgery is causative, which is limited to a modified clamp crush technique due to a lack of alternatives. In this respect, there is an urgent need for the development of compatible dissection techniques in future robot generations, e.g., CUSA and waterjet. The higher median surgical complexity with higher frequency of iCC in the robotic group could also add to the higher rate of clinically relevant bile leakage. Another potential contributing factor may be the learning curve, as the era of robotic surgery within our study period was considerably shorter than of laparoscopy.
Our study has obvious limitations. Data in robotic surgery were collected prospectively as part of a study. Nevertheless, the comparison to laparoscopic surgery was only possible on the basis of retrospective data with inherent higher risk of confounding factors. Further, the results of the robotic cases must be interpreted with caution because they were collected within the learning curve, which is also true for the early laparoscopic cases. Although we did not establish clearly defined indications for using the robotic or the laparoscopic approach, a selection bias to either minimally invasive approach must be considered for the era of robotic surgery, which we tried to minimize by propensity score matching. Consequently, a final comparison of the techniques would require a prospective randomized study after completion of the learning curve which will also have to address the issue of bile leakage.
In conclusion, robotic liver resection can be safely established within a manageable period. This seems to be due to a shorter learning curve for robotic when compared to laparoscopic surgery [30, 31]. Whether the robotic technique is generally superior to the laparoscopic technique cannot be concluded based on our data. There are even some disadvantages, such as the currently limited choice of dissection techniques. However, there is the subjective impression that the strengths come into play especially in complex resections. In this respect, we are convinced that the technology will be a valuable addition to established minimally invasive techniques.
Acknowledgements
We acknowledge the support of Intuitive Surgical Deutschland GmbH for providing a research grant for the equipment to perform robotic surgeries and enable this study. We greatly appreciate the contribution of Andrea Köpke, Birgül Ertugrul, and Sabine Boas-Knoop to the study management and data collection. We further acknowledge the support from Deutsche Forschungsgemeinschaft (DFG) to L.F. (FE14.34-2.1) and to M.S. (SCHM2661/3-2) and the support by the German Academic Exchange Service (DAAD) for a Doctoral Scholarship for SAOG. LF and FK were participants in the BIH-Charité Clinician Scientist Program and SM is participant in the Junior Clinician Scientist Program funded by Charité –Universitätsmedizin Berlin and Berlin Institute of Health.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was supported with a Research Grant by Intuitive Surgical Deutschland GmbH.
Disclosures
Moritz Schmelzle reports personal fees or other support outside of the submitted work from Merck, Bayer, ERBE, Amgen, Johnson & Johnson, Takeda, Olympus, Medtronic, and Intuitive. Johann Pratschke reports a research grant from Intuitive Surgical Deutschland GmbH for the conduction of the present study. Outside of the submitted work, he reports personal fees or non-financial support from Johnson & Johnson, Medtronic, AFS Medical, Astellas, CHG Meridian, Chiesi, Falk Foundation, La Fource Group, Merck, Neovii, NOGGO, pharma consult Peterson, and promedicis. Linda Feldbrügge, Santiago Andres Ortiz Galindo, Simon Moosburner, Anika Kästner, Felix Krenzien, Christian Benzing, Matthias Biebl, Robert Öllinger, Thomas Malinka, and Wenzel Schöning have no conflict of interest or financial ties to disclose.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Moritz Schmelzle and Linda Feldbrügge have contributed equally and share first authorship.
References
- 1.Andreou A, Struecker B, Raschzok N, Krenzien F, Haber P, Wabitsch S, Waldbaur C, Touet EM, Eichelberg AC, Atanasov G, Biebl M, Bahra M, Öllinger R, Schmelzle M, Pratschke J. Minimal-invasive versus open hepatectomy for hepatocellular carcinoma: comparison of postoperative outcomes and long-term survivals using propensity score matching analysis. Surg Oncol. 2018;27:751–758. doi: 10.1016/j.suronc.2018.10.005. [DOI] [PubMed] [Google Scholar]
- 2.Fretland ÅA, Dagenborg VJ, Bjørnelv GMW, Kazaryan AM, Kristiansen R, Fagerland MW, Hausken J, Tønnessen TI, Abildgaard A, Barkhatov L, Yaqub S, Røsok BI, Bjørnbeth BA, Andersen MH, Flatmark K, Aas E, Edwin B. Laparoscopic versus open resection for colorectal liver metastases: the OSLO-COMET randomized controlled trial. Ann Surg. 2018;267:199–207. doi: 10.1097/SLA.0000000000002353. [DOI] [PubMed] [Google Scholar]
- 3.Knitter S, Andreou A, Kradolfer D, Beierle AS, Pesthy S, Eichelberg AC, Kästner A, Feldbrügge L, Krenzien F, Schulz M, Banz V, Lachenmayer A, Biebl M, Schöning W, Candinas D, Pratschke J, Beldi G, Schmelzle M. Minimal-invasive versus open hepatectomy for colorectal liver metastases: bicentric analysis of postoperative outcomes and long-term survival using propensity score matching analysis. J Clin Med. 2020;9(12):4027. doi: 10.3390/jcm9124027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Aghayan DL, Fretland ÅA, Kazaryan AM, Sahakyan MA, Dagenborg VJ, Bjørnbeth BA, Flatmark K, Kristiansen R, Edwin B. Laparoscopic versus open liver resection in the posterosuperior segments: a sub-group analysis from the OSLO-COMET randomized controlled trial. HPB (Oxf) 2019;21:1485–1490. doi: 10.1016/j.hpb.2019.03.358. [DOI] [PubMed] [Google Scholar]
- 5.Aghayan DL, Kazaryan AM, Dagenborg VJ, Røsok BI, Fagerland MW, Bjørnelv GMW, Kristiansen R, Flatmark K, Fretland ÅA, Edwin B. Long-term oncologic outcomes after laparoscopic versus open resection for colorectal liver metastases : a randomized trial. Ann Intern Med. 2020;174(2):175–182. doi: 10.7326/M20-4011. [DOI] [PubMed] [Google Scholar]
- 6.Velayutham V, Fuks D, Nomi T, Kawaguchi Y, Gayet B. 3D visualization reduces operating time when compared to high-definition 2D in laparoscopic liver resection: a case-matched study. Surg Endosc. 2016;30:147–153. doi: 10.1007/s00464-015-4174-1. [DOI] [PubMed] [Google Scholar]
- 7.Lafaro KJ, Stewart C, Fong A, Fong Y. Robotic liver resection. Surg Clin N Am. 2020;100:265–281. doi: 10.1016/j.suc.2019.11.003. [DOI] [PubMed] [Google Scholar]
- 8.Ciria R, Berardi G, Alconchel F, Briceño J, Choi GH, Wu YM, Sugioka A, Troisi RI, Salloum C, Soubrane O, Pratschke J, Martinie J, Tsung A, Araujo R, Sucandy I, Tang CN, Wakabayashi G. The impact of robotics in liver surgery: a worldwide systematic review and short-term outcomes meta-analysis on 2,728 cases. J Hepatobiliary Pancreat Sci. 2020 doi: 10.1002/jhbp.869. [DOI] [PubMed] [Google Scholar]
- 9.Zhang L, Yuan Q, Xu Y, Wang W. Comparative clinical outcomes of robot-assisted liver resection versus laparoscopic liver resection: a meta-analysis. PLoS ONE. 2020;15:e0240593. doi: 10.1371/journal.pone.0240593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Goh BKP, Lee LS, Lee SY, Chow PKH, Chan CY, Chiow AKH. Initial experience with robotic hepatectomy in Singapore: analysis of 48 resections in 43 consecutive patients. ANZ J Surg. 2019;89:201–205. doi: 10.1111/ans.14417. [DOI] [PubMed] [Google Scholar]
- 11.Magistri P, Pecchi A, Franceschini E, Pesi B, Guadagni S, Catellani B, Assirati G, Guidetti C, Guerrini GP, Tarantino G, Ballarin R, Codeluppi M, Morelli L, Coratti A, Di Benedetto F. Not just minor resections: robotic approach for cystic echinococcosis of the liver. Infection. 2019;47:973–979. doi: 10.1007/s15010-019-01333-2. [DOI] [PubMed] [Google Scholar]
- 12.Salloum C, Lim C, Lahat E, Gavara CG, Levesque E, Compagnon P, Azoulay D. Robotic-assisted versus laparoscopic left lateral sectionectomy: analysis of surgical outcomes and costs by a propensity score matched cohort study. World J Surg. 2017;41:516–524. doi: 10.1007/s00268-016-3736-2. [DOI] [PubMed] [Google Scholar]
- 13.Araujo RLC, Sanctis MA, Barroti LC, Coelho TRV. Robotic approach as a valid strategy to improve the access to posterosuperior hepatic segments—case series and review of literature. J Surg Oncol. 2020;121:873–880. doi: 10.1002/jso.25831. [DOI] [PubMed] [Google Scholar]
- 14.Schmelzle M, Wabitsch S, Haber PK, Krenzien F, Kästner A, Biebl M, Öllinger R, Pratschke J. Laparoskopische Leberchirurgie – Berliner Zentrumserfahrungen aus 250 konsekutiven Fällen. Zent Chir. 2019;144:145–152. doi: 10.1055/a-0712-5434. [DOI] [PubMed] [Google Scholar]
- 15.Wabitsch S, Schöning W, Kästner A, Haber PK, Benzing C, Krenzien F, Lenz K, Schmelzle M, Pratschke J. A propensity-matched study of full laparoscopic versus hand-assisted minimal-invasive liver surgery. Surg Endosc. 2020;35(5):2021–2028. doi: 10.1007/s00464-020-07597-2. [DOI] [PubMed] [Google Scholar]
- 16.Struecker B, Haber P, Öllinger R, Bahra M, Pascher A, Pratschke J, Schmelzle M. Comparison of single-port versus standard multiport left lateral liver sectionectomy. Surg Innov. 2018;25:136–141. doi: 10.1177/1553350617752010. [DOI] [PubMed] [Google Scholar]
- 17.Schmelzle M, Schöning W, Pratschke J. Leberchirurgie – setup, port placement, strukturierte OP-Schritte. Zent Chir. 2020;145:246–251. doi: 10.1055/a-1135-9162. [DOI] [PubMed] [Google Scholar]
- 18.Desmet VJ, Gerber M, Hoofnagle JH, Manns M, Scheuer PJ. Classification of chronic hepatitis: diagnosis, grading and staging. Hepatology. 1994;19:1513–1520. doi: 10.1002/hep.1840190629. [DOI] [PubMed] [Google Scholar]
- 19.Cassiman D, van Pelt J, de Vos R, van Lommel F, Desmet V, Yap S-H, Roskams T. Synaptophysin: a novel marker for human and rat hepatic stellate cells. Am J Pathol. 1999;155:1831–1839. doi: 10.1016/S0002-9440(10)65501-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Krenzien F, Wabitsch S, Haber P, Kamali C, Brunnbauer P, Benzing C, Atanasov G, Wakabayashi G, Öllinger R, Pratschke J, Schmelzle M. Validity of the IWATE criteria for patients with hepatocellular carcinoma undergoing minimally invasive liver resection. J Hepatobiliary Pancreat Sci. 2018;25:403–411. doi: 10.1002/jhbp.576. [DOI] [PubMed] [Google Scholar]
- 21.Strasberg SM. Nomenclature of hepatic anatomy and resections: a review of the Brisbane 2000 system. J Hepatobiliary Pancreat Surg. 2005;12:351–355. doi: 10.1007/s00534-005-0999-7. [DOI] [PubMed] [Google Scholar]
- 22.Wakabayashi G. What has changed after the Morioka consensus conference 2014 on laparoscopic liver resection? Hepatobiliary Surg Nutr. 2016;5:281–289. doi: 10.21037/hbsn.2016.03.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dindo D, Demartines N, Clavien P-A. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–213. doi: 10.1097/01.sla.0000133083.54934.ae. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Koch M, Garden OJ, Padbury R, Rahbari NN, Adam R, Capussotti L, Fan ST, Yokoyama Y, Crawford M, Makuuchi M, Christophi C, Banting S, Brooke-Smith M, Usatoff V, Nagino M, Maddern G, Hugh TJ, Vauthey JN, Greig P, Rees M, Nimura Y, Figueras J, DeMatteo RP, Büchler MW, Weitz J. Bile leakage after hepatobiliary and pancreatic surgery: a definition and grading of severity by the International Study Group of Liver Surgery. Surgery. 2011;149:680–688. doi: 10.1016/j.surg.2010.12.002. [DOI] [PubMed] [Google Scholar]
- 25.Sucandy I, Luberice K, Lippert T, Castro M, Krill E, Ross S, Rosemurgy A. Robotic major hepatectomy: an institutional experience and clinical outcomes. Ann Surg Oncol. 2020;27:4970–4979. doi: 10.1245/s10434-020-08845-4. [DOI] [PubMed] [Google Scholar]
- 26.Beard RE, Khan S, Troisi RI, Montalti R, Vanlander A, Fong Y, Kingham TP, Boerner T, Berber E, Kahramangil B, Buell JF, Martinie JB, Vrochides D, Shen C, Molinari M, Geller DA, Tsung A. Long-term and oncologic outcomes of robotic versus laparoscopic liver resection for metastatic colorectal cancer: a multicenter, propensity score matching analysis. World J Surg. 2020;44:887–895. doi: 10.1007/s00268-019-05270-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Magistri P, Tarantino G, Guidetti C, Assirati G, Olivieri T, Ballarin R, Coratti A, Di Benedetto F. Laparoscopic versus robotic surgery for hepatocellular carcinoma: the first 46 consecutive cases. J Surg Res. 2017;217:92–99. doi: 10.1016/j.jss.2017.05.005. [DOI] [PubMed] [Google Scholar]
- 28.Lee KF, Chong C, Cheung S, Wong J, Fung A, Lok HT, Lo E, Lai P. Robotic versus open hemihepatectomy: a propensity score-matched study. Surg Endosc. 2020;35(5):2316–2323. doi: 10.1007/s00464-020-07645-x. [DOI] [PubMed] [Google Scholar]
- 29.Kamarajah SK, Bundred J, Manas D, Jiao LR, Hilal MA, White SA. Robotic versus conventional laparoscopic liver resections: a systematic review and meta-analysis. Scand J Surg. 2020 doi: 10.1177/1457496920925637. [DOI] [PubMed] [Google Scholar]
- 30.Magistri P, Guerrini GP, Ballarin R, Assirati G, Tarantino G, Di Benedetto F. Improving outcomes defending patient safety: the learning journey in robotic liver resections. Biomed Res Int. 2019;2019:1835085. doi: 10.1155/2019/1835085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gall TMH, Alrawashdeh W, Soomro N, White S, Jiao LR. Shortening surgical training through robotics: randomized clinical trial of laparoscopic versus robotic surgical learning curves. BJS Open. 2020;4:1100–1108. doi: 10.1002/bjs5.50353. [DOI] [PMC free article] [PubMed] [Google Scholar]