Abstract
Background
Many antidepressants are substrates of P‐glycoprotein, an efflux transporter in the blood‐brain‐barrier encoded by the ABCB1 gene. Genetic variations might influence the transport rate of antidepressants and hence their pharmacological effects. This study investigates the influence of eight polymorphisms in the ABCB1 gene on antidepressant treatment response.
Method
152 patients were included from psychiatric departments of the Mental Health Research Institute in Tomsk. The difference in Hamilton‐Depression‐Rating‐Scale (HAMD‐17)‐scores between baseline and week two, week two and four, and baseline and week four was used to estimate timing of improvement of depression. Associations between the ABCB1 gene‐polymorphisms and reduction in HAMD‐17 score were assessed using independent t‐test and multiple linear regression.
Results
Tricyclic antidepressants were associated with a higher reduction of HAMD‐17 score when compared to SSRIs. The SNP rs2235040 A‐allele had a significant positive influence on the ΔHAMD‐17(0→2W) score but a significant negative influence on the ΔHAMD‐17(2→4W) score. The rs4148739 G‐allele had a significant negative influence on the ΔHAMD‐17(0→2W) score but a significant positive influence on the ΔHAMD‐17(2→4W) score. The SNP rs2235015 T‐allele is significant negatively related to the ΔHAMD‐17(2→4W) score.
Conclusion
ABCB1 Genetic variations appear to affect speed but not magnitude of antidepressant drug response.
Keywords: ABCB1 gene, antidepressants, Hamilton Depression Rating Scale, P‐glycoprotein
1. INTRODUCTION
Major depressive disorder (MDD) is a disabling mental disorder with a high incidence and prevalence rate (Murray & Lopez, 1996). In 2017 worldwide, more than 163 million people were affected by MDD. The number of years lived with disability (YLD's) attributable to a major depressive disorder increased by 12.6% between 2007 and 2017 (GBD 2017 Oral Disorders Collaborators et al., 2020).
In the treatment of MDD, antidepressants are generally indicated (Bauer et al., 2013) but their response rate and clinical efficacy is suboptimal. The main purpose of treatment is remission of depressive symptoms but the response rate of first line treatment is relatively low (Bauer et al., 2013; Rush et al., 2006).
Lack of response to antidepressants may be explained by multiple factors. One important factor is suboptimal treatment caused by inadequate transport into the central nervous system (CNS). The CNS is protected against xenobiotic substances by the blood brain barrier (BBB), which contains multiple transport proteins whose function is to drive xenobiotics back into the bloodstream. One of these trans‐membrane transport proteins is P‐glycoprotein, expressed at the blood facing side of the blood brain barrier (Cordon‐Cardo et al., 1989). Its primary function is the active transport of penetrated chemicals from the brain back into the bloodstream (Ambudkar et al., 1999). Several antidepressants are known substrates of the P‐gp transporter, for example nortriptyline (Ejsing et al., 2006; Uhr, et al., 2000), amitriptyline (Uhr et al., 2000), venlafaxine (Uhr et al., 2003), paroxetine (Uhr et al., 2003), citalopram (Doran et al., 2005; Uhr & Grauer, 2003), sertraline (Wang et al., 2008), and trimipramine (Fabbri et al., 2013; Porcelli et al., 2011; Uhr & Grauer, 2003).
The P‐gp efflux transporter is encoded by the ‘ATP binding cassette subfamily B member 1’ (ABCB1) gene (Ambudkar et al., 1999). Genetic variations in the ABCB1 gene might influence the functionality of the transporter and therefore the transport rate of antidepressants into the brain (Porcelli et al., 2011). Several studies have investigated the influence of single nucleotide polymorphisms (SNPs) in the ABCB1 gene on antidepressant treatment outcome (Breitenstein et al., 2015; Kato et al., 2008; Uhr et al., 2008). The primary SNP's under investigation are rs1045642 (C3435T) and rs2032582 (G2677T), mainly because these are linked to alterations in the P‐gp expression and functionality (Eichelbaum et al., 2004; Hoffmeyer et al., 2000; Schwab et al., 2003).
In studies investigating the relationship between several ABCB1 SNPs and antidepressant response, conflicting results have been found. While several studies have shown a significant relation between SNPs and antidepressant treatment response (Breitenstein et al., 2015; Kato et al., 2008; Nikisch et al., 2008; Sarginson et al., 2010; Singh et al., 2012; Uhr et al., 2008a), others have been unable to determine a significant association (Gex‐Fabry et al., 2008; Perlis et al., 2010; Porcelli et al., 2011; Simoons et al., 2020). Most of these studies focused on newer groups of antidepressants, such as the selective serotonin reuptake inhibitors (SSRIs). However, tricyclic antidepressants (TCAs) like nortriptyline and amitriptyline are also known P‐gp substrates, so one might also expect an effect of SNPs on treatment response for this group (Ejsing et al., 2006; Uhr et al., 2000). The type of antidepressant or affinity of the drug for the P‐gp transporter might influence the contradictory results. Conflicting results are however also found for individual drugs, such as paroxetine (Gex‐Fabry et al., 2008; Kato et al., 2008; Uhr et al., 2008).
The reported results might be influenced by the lack of consensus regarding the clinical efficacy of antidepressants during the first two weeks. Traditionally, a delay in treatment response of several weeks is hypothesized for antidepressants (Quitkin et al., 1984; van Calker et al., 2009). In this theory, early effects of antidepressant treatment are merely attributed to placebo‐like responses. Other studies however have reported more immediate effects of antidepressants compared to placebo (Posternak & Zimmerman, 2005; Stassen et al., 1996, 2007; Taylor, 2007), making the interpretation of “true” drug effect more difficult.
To further investigate the above contradiction, this naturalistic prospective cohort study investigates the influence of eight polymorphisms in the ABCB1 gene on the timing of antidepressant treatment response. The influence of the polymorphisms is investigated over a time period of four weeks in patients with moderate or severe depressive disorders using the calculated difference in HAMD‐17 scores. To determine whether the outcome is influenced by the type of antidepressant or the affinity of the antidepressant for the P‐gp receptor, the patient population will be stratified based on the type of antidepressant treatment and affinity for the P‐gp transporter. To examine the supposed delay in treatment response, improvement in HAMD‐17 scores was compared between the first 2 weeks, the last two weeks and over the total period of 4 weeks.
2. METHOD AND MATERIALS
2.1. Patients
For this study, 186 patients were included from psychiatric departments of the Mental Health Research Institute (MHRI) in Tomsk, Russian Federation. Written informed consent was provided before inclusion. The time period in which patients were included is from April 2012 until January 2015.
The inclusion criteria for patients in the study were an at least moderately severe depressive episode (HAMD‐17 score > 17) caused by a depressive episode (ICD: F32) or recurrent depressive disorder (ICD: F33) (Sheehan et al., 1998). Patients were between 18 and 70 years old and free of treatment with antidepressant drugs for at least 6 months before inclusion.
The study population and methods applied are partially described earlier by Ochi et al. (2019). Patients with schizophrenia, a decompensated personality disorders, of non‐Caucasian ethnicity, with relevant pharmacological withdrawal symptoms or organic brain disorders (i.e., epilepsy, Parkinson's disease) were excluded from participation. In addition, individuals with relevant serious or unstable physical conditions (including abnormal liver and kidney function) were excluded, as were pregnant patients, patients with relevant gynecological or endocrine disorder or patients using antidopaminergic drugs (antipsychotic or antiemetic drugs).
The study procedures were reviewed and approved by the Institutional Medical Review Board of the MHRI (protocol 49 from April 23, 2012). The study was carried out in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki 1975, revised in Fortaleza, Brazil, 2013) for experiments involving humans.
2.2. Depression rating
The 17‐item Hamilton Depression Rating Scale (HAMD‐17) (Hamilton, 1960) was used to score the severity of depression before treatment, after two weeks and after four weeks of treatment with antidepressants. After inclusion in the study, the severity of the depressive episode of the patient was scored with the HAMD‐17 score within 2 days. Treatment was initiated immediately after finishing the HAMD‐17 test.
2.3. Blood sampling and genotyping
Venous blood samples for DNA extraction were collected after 8 h of overnight fasting following the local procedure. Blood samples were collected in EDTA tubes and stored at −20°C until DNA isolation. DNA was isolated by the standard phenol‐chloroform method, checking of the quality and quantity of DNA was carried out in the Laboratory of Molecular Genetics and Biochemistry of the Mental Health Research Institute using NanoDrop™ 8000 Spectrophotometer (Thermo Fisher Scientific). All DNA samples were genotyped simultaneously for eight SNPs within the ABCB1 gene (Supplementary Table 1) in the Laboratory of Genetics of the University of Groningen with the MassARRAY® System (Agena Bioscience™) during March‐May 2015. Genotyping was performed on a Sequenom MassARRAY Analyzer 4 using the SEQUENOM Consumables iPLEX Gold 384. DNA sample preparation for SEQUENOM MassARRAY® Analyzer 4 includes several steps: multiplex PCR, SAP reaction (purification of PCR products), iPLEX Gold reaction (primer extension reaction) and then placing the samples on a special chip (SpectroCHIP Array) using NanoDispenser RS1000 prior to loading them into the analyzer.
2.4. Statistical analysis
From the original dataset, patients with missing data on HAMD‐17 scores and/or genotype information were excluded. After exclusion of missing values, three new variables were created, characterizing the difference in HAMD‐17 score between the second week of treatment and baseline (before treatment) (ΔHAMD‐170‐2W), between the second and fourth week (ΔHAMD‐172‐4W) and between the fourth week of treatment with antidepressants and baseline (ΔHAMD‐170‐4W) (Ochi et al., 2019). A lower HAMD‐17 score indicates a better clinical outcome. Due to the limited population size, the homozygous recessive genotypes were limited in number for six of the eight tested genotypes and were combined with heterozygous genotypes (i.e.,, rs2032583, rs2235040, rs28401781, rs2235015, rs4148739, rs928256). The deviation from Hardy‐Weinberg equilibrium (HWE) was tested for all genotypes.
Patient characteristics were determined with descriptive statistics. Independent t‐test was used to compare the ΔHAMD‐170‐2W, ΔHAMD‐172‐4W and ΔHAMD‐170‐4W values between patients using medication with high affinity for the P‐gp receptor (sertraline/paroxetine and escitalopram/citalopram) and patients using medication with low or unknown P‐gp affinity like fluoxetine or mirtazapine. To determine the variance in the independent t‐test, Levene's test for equality was used. Further, independent T‐tests compared the ΔHAMD‐170‐2W and ΔHAMD‐172‐4W values between different types of antidepressants (i.e., SSRI, TCA, SNRI, NaSSa, Agomelatine). To determine the relation between the ABCB1 SNPs and the ΔHAMD‐170‐2W, ΔHAMD‐172‐4W and ΔHAMD‐170‐4W score, multiple linear regression analysis was performed to determine the association between ABCB1 genotypes and ΔHAMD‐17 scores, including covariates of sex, episode status (single vs. multiple depressive episode) and antidepressant treatment. SPSS software (release 25.0) was used for the statistical analyses. p‐values <0.05 were considered significant.
3. RESULTS
3.1. Patients
After exclusion of patients with missing data on HAMD‐17 scores and/or genotype information, a total of 152 patients (134 female/18 male) were included in this study. The mean age of the male patients was 49.8 ± 9.7 years and for female patients, 49.5 ± 11.2 years. For six of the eight tested genotypes the recessive homozygous genotype was combined with the heterozygous genotypes due to a limited number of patients (<5%) with the genotype in the study population. A total of 92 (60.5%) patients were treated with SSRIs, 23 (15.1%) patients received a TCA, 14 (9.2%) patients were treated with SNRIs, 12 (7.8%) patients used agomelatine and 11 (7.2%) patients received NaSSa. The clinical and demographic characteristics of the study population are presented in Table 1.
TABLE 1.
Patient characteristics
| Parameter | Number |
|---|---|
| Sex (N) | 152 |
| Male | 18 |
| Female | 134 |
| Age (Years) + SD | |
| Male | 49.8 ± 9.7 |
| Female | 49.5 ± 11.2 |
| SSRI (N) | 92 (60.5%) |
| Sertraline | 24 |
| Paroxetine | 22 |
| Escitalopram | 16 |
| Fluoxetine | 12 |
| Fluvoxamine | 12 |
| Citalopram | 4 |
| Trazodone | 1 |
| Trazodone + paroxetine | 1 |
| TCA (N) | 23 (15.1%) |
| Clomipramine | 16 |
| Pipofezine | 6 |
| Amitriptyline | 1 |
| SNRI (N) | 14 (9.2%) |
| Venlafaxine | 10 |
| Duloxetine | 4 |
| NaSSa (N) | 11 (7.2%) |
| Mirtazapine | 7 |
| Mianserin | 4 |
| Agomelatine | 12 (7.8%) |
The genotype distribution of all SNP's was consistent with the Hardy–Weinberg equilibrium (Supplementary Tables 2 and 3). The HAMD‐17 scores improved in each subgroup and genotype tested over time. The improvement was for all subgroups and genotypes greater in the first two weeks compared to the second 2 weeks.
3.2. Differences between antidepressant treatment and ΔHAMD‐17
Independent t‐test analysis was performed to analyze the potential difference in ΔHAMD‐170‐2W and ΔHAMD‐172‐4W and ΔHAMD‐170‐4W scores between different groups of antidepressants. No significant difference in ΔHAMD was determined between the sertraline/paroxetine or escitalopram/citalopram and other antidepressants across all study periods.
3.3. ABCB1 and HAMD‐17 analyses
To study the influence of the different ABCB1 genotypes encoding for P‐gp on the ΔHAMD‐170‐2W, ΔHAMD‐172‐4W and ΔHAMD‐170‐4W scores, multivariate linear regression analyses were performed with stratification for antidepressant treatment (sertraline/paroxetine vs. other antidepressants). A significant association was found in SNPs rs2235040 and rs4148739 and ΔHAMD‐17 scores (rs2235040 ΔHAMD‐170‐2W: B = 10.55 [95% CI: 0.71 – 20.39], p = 0.036; ΔHAMD‐172‐4W: B = −10.03 [95% CI: −19.19 – −0.87], p = 0.036 || rs4148739 ΔHAMD‐170‐2W: B = −11.33 [95% CI: −21.27 – −1.38], p = 0.026; ΔHAMD‐172‐4W: B = 11.19 [95% CI: 1.94 – 20.44], p = 0.018). The associations were no longer significant when analyzed with ΔHAMD‐170‐4W score.
The analysis was repeated with the escitalopram/citalopram study population. Significance was determined in the same SNPs as the previous analysis (rs2235040 ΔHAMD‐170‐2W: B = 10.97 [95% CI: 1.22 – 20.72], p = 0.028; ΔHAMD‐172‐4W: B = −10.28 [95% CI: −19.36 – −1.20], p = 0.027 || rs4148739 ΔHAMD‐170‐2W: B = −11.86 [95% CI: −21.62 – −2.10], p = 0.018; ΔHAMD‐172‐4W: B = 11.59 [95% CI: 2.50 – 20.68], p = 0.013). Proceeding with ΔHAMD‐170‐4W score analyses resulted in similar findings to sertraline/paroxetine as there was no significance found (Figure 1).
FIGURE 1.
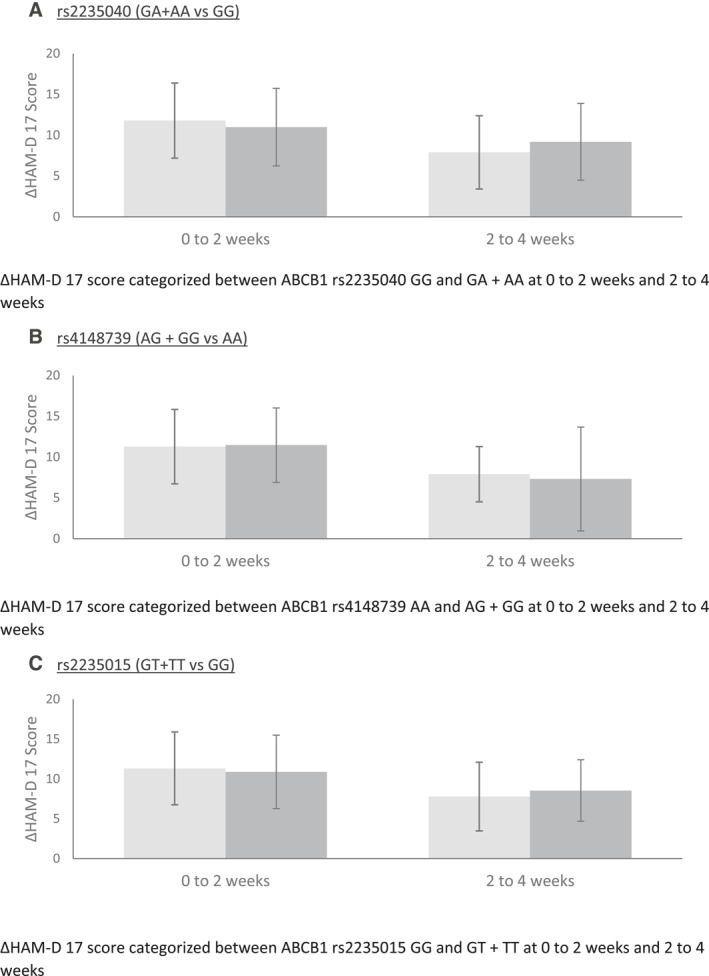
ΔHAM‐D 17 Scores of the SNPs rs2235040, rs4148739 and rs2235015
The study population was stratified based on antidepressant types (i.e., SSRI, TCA, SNRI, NaSSa, agomelatine). Rs2235040 and rs4148739 were found again demonstrating significant differences in ΔHAMD‐17 for time periods time of initiation to two weeks and two weeks and four weeks (rs2235040 ΔHAMD‐170‐2W: B = 10.80 [95% CI: 1.43 – 20.16], p = 0.024; ΔHAMD‐172‐4W: B = −11.14 [95% CI: −19.63 – −2.65], p = 0.011 || rs4148739 ΔHAMD‐170‐2W: B = −11.26 [95% CI: −20.67 – −1.84], p = 0.02; ΔHAMD‐172‐4W: B = 12.46 [95% CI: 3.92 – 21.00], p = 0.005). For ΔHAMD‐172‐4W, rs2235015 was determined to be significant (B = −2.93 [95% CI: −5.46 – −0.39], p = 0.024). No SNPs were found with a significant association in ΔHAMD‐170‐4W (Table 2).
TABLE 2.
Linear regression of covariates age, gender, ABCB1 genotypes, type of antidepressant for baseline/week two, week two/four and baseline and week 4
| Period | 0–2 weeks | 2–4 weeks | 0–4 weeks | |||
|---|---|---|---|---|---|---|
| Baseline predictors | B | p‐value | B | p‐value | B | p‐value |
| Sex | 0.150 | 0.915 | 2.171 | 0.092 | 2.321 | 0.181 |
| Age | −0.018 | 0.647 | 0.015 | 0.690 | −0.004 | 0.937 |
| RS1045642 C | 0.083 | 0.961 | 1.151 | 0.456 | 1.234 | 0.554 |
| RS1045642 TC | −0.286 | 0.793 | −0.314 | 0.750 | −0.600 | 0.652 |
| RS2235040 AGA | 10.799 | 0.024* | −11.143 | 0.011* | −0.345 | 0.953 |
| RS2235015 TGT | 1.075 | 0.447 | −2.926 | 0.024* | −1.851 | 0.286 |
| RS4148739 GGA | −11.258 | 0.020* | 12.462 | 0.005** | 1.204 | 0.836 |
| RS9282564 GGA | −0.346 | 0.750 | −0.119 | 0.904 | −0.465 | 0.727 |
| RS2032582 A | 0.618 | 0.693 | −1.411 | 0.322 | −0.793 | 0.680 |
| RS2032582 CA | 0.282 | 0.825 | −0.898 | 0.438 | −0.617 | 0.693 |
| TCAs | 3.922 | 0.002** | 2.222 | 0.047* | 6.145 | 0.000** |
| SNRIs | −0.378 | 0.787 | 1.625 | 0.201 | 1.248 | 0.467 |
| Agomelatine | 3.085 | 0.065 | −4.998 | 0.001** | −1.912 | 0.347 |
| NaSSAs | −0.033 | 0.986 | −1.252 | 0.463 | −1.285 | 0.577 |
Abbreviations: B, regression coefficients; NaSSAs, noradrenergic and specific serotonergic antidepressants; SNRIs, serotonin–norepinephrine reuptake inhibitors; TCAs, tricyclic antidepressants.
*p < 0.05; **p < 0.01.
Difference ΔHAMD‐17 between men and women was found to be significant in the analysis for sertraline/paroxetine (s/p) and escitalopram/citalopram (e/p) during week two and week four (s/p: B = 2.76 [95% CI: 0.74 – 5.45], p = 0.044 || e/c: B = 2.76 [95% CI: 0.73 – 5.44], p = 0.044). Difference in antidepressant response was found in the analysis between antidepressant types. Agomelatine was associated with a significant influence on the ΔHAMD‐172‐4W score (B = −5.00 [95% CI: −7.97 – −2.02], p = 0.001). Tricyclic antidepressants were found to have a significant positive association compared to SSRIs across the study periods HAMD‐17 (ΔHAM‐D170‐2W B = 3.92 [95% CI: 1.50 – 6.33], p = 0.002; ΔHAM‐D172‐4W B = 2.22 [95% CI: 0.32 – 4.41], p = 0.047; ΔHAM‐D170‐4W B = 6.15 [95% CI: 3.18 – 9.10], p = 0.0003).
When conducting the multiple linear regression analyses for the different ABCB1 genotypes and ΔHAMD‐17, two SNP's (rs28401781 and rs2032583) were excluded from the analysis by the statistical program. These were corroborated by SPSS due to the lack of added value to the equation, as the variance in the SNP was already contained in the equation. A linkage disequilibrium plot was made to check for possible linkage between the SNP's. A high linkage disequilibrium was denoted between rs28401781, rs2032583, rs4148739, rs2235040, rs2235015 and rs9282564, supplementing the rationale of exclusion of the SNP's rs28401781 and rs2032583.
3.4. ΔHAMD‐17 difference between single and multiple depressive episode patients
The independent t‐test analysis was performed to analyze the potential difference in ΔHAMD‐170‐2W, ΔHAMD‐172‐4W and ΔHAMD‐170‐4W scores between patients with single and multiple depressive episodes. No significance concerning response within the two depressive episodes was found, denoting a lack of response difference between the two major depression types in this study.
4. DISCUSSION
In this study, the influence of eight polymorphisms in the ABCB1 gene on antidepressant treatment response was studied in freshly admitted, treatment‐free in‐patients with moderate or severe depressive disorders using the calculated difference in HAMD‐17 scores. The improvement in HAMD‐17 scores was compared between the first two weeks, the last two weeks and in the total period of four weeks. The SNP rs2235040 A allele was found to have a significant positive relation with the ΔHAMD‐170‐2W score but a significant negative influence on the ΔHAMD‐172‐4W score. The rs4148739 G allele in contrast seems to have a significant negative influence on the ΔHAMD‐170‐2W score but a significant positive influence on the ΔHAMD‐172‐4W score. The SNP rs2235015 T allele was also found to be negatively related to the ΔHAMD‐172‐4W score. The analysis between the ABCB1 genotypes and the ΔHAMD‐170‐4W scores failed to demonstrate a significant relationship in all cases. Also no significant difference in ΔHAMD score was found between sertraline/paroxetine or escitalopram/citalopram and other antidepressants across all study periods.
When the study population was stratified based on antidepressants type, TCA's showed a significant positive influence on the calculated difference in HAMD‐17 scores compared to SSRIs across all study periods. Also agomelatine was associated with a significant influence on the ΔHAM‐D172‐4W score.
SSRIs are often first choice antidepressants for major depressive disorder since they have a more favorable side effect profile compared to TCAs (Anderson, 2000; Trimbos instituut, 2013). The effectiveness of the two groups does not seem to differ substantially (Anderson, 2000; Williams et al., 2000). Our study in contrast, showed that TCAs have a higher response rate in all analyzed time periods compared to SSRI's. This might be explained by the fact that all patients in this study were clinically treated and is in line with a large meta‐analysis study conducted by Anderson (2000). This study states that TCA's are more effective in in‐patients compared with SSRI's. TCAs are therefore often first choice in this patient group.
A peculiarity of the current study is also that the response to treatment is very high: the mean HAMD‐17 scores decrease in 4 weeks from 24.3 ± 5.2 to 5.0 ± 3.9 (Ochi et al., 2019). This cannot be entirely attributed to the effect of admission to a clinical setting. An important factor is probably that more than half of the patients had never been treated with antidepressants and none of them had taken an antidepressant during the last 6 months before admission. This criterion has been very unusual in this type of research in recent decades and is likely to have a major impact on response size by elimination of negative carry‐over effects of previous treatments.
The linear regression analysis between the eight SNP's and ΔHAMD‐17 scores across all study periods showed several significant relations. First, the SNP rs2235015 T allele was found to have a significant negative relation with the ΔHAMD‐172‐4W score. Uhr et al. (2008) previously showed that patients carrying the minor C allele of SNP rs2032583 and the minor T allele of rs2235015 had significantly higher remission rates in comparison to non‐carriers. Other studies however found no predictive value of rs2235015 on clinical improvement (Sarginson et al., 2010; Schatzberg et al., 2015). Our study showed a significant negative relation between the minor rs2235015 T allele and improvement of ΔHAMD‐172‐4W score. This effect was previously also shown by Shan et al. (2019) who found a significant relation between the rs2235015 GG genotype and lower HAMD‐17 scores in patients using SNRIs. This indicates that the functionality of the P‐gp receptor may be reduced in carriers of the rs2235015 G allele, resulting in a better effect of the antidepressant treatment.
The SNP rs2235040 A allele was found to have a significant positive relation with the ΔHAMD‐170‐2W score but a significant negative influence on the ΔHAMD‐172‐4W score. No significant relation between the rs2235040 A allele and the improvement in the ΔHAMD‐17 score in the total period of four weeks was found. A comparable result was found for the SNP rs4148739; the rs4148739 G allele seems to have a significant negative influence on the ΔHAMD‐170‐2W score but a significant positive influence on the ΔHAMD‐172‐4W score. Also no significant relation between rs4148739 G allele and the improvement in the ΔHAMD‐170‐4W was found.
Both results indicate that the rs2235040 A allele and rs4148739 G allele mainly influence the speed of the antidepressant response in our study population and not the long‐term response rate. The functionality of the P‐gp receptor seems less in carriers of the rs2235040 A allele resulting in a faster antidepressant response. The functionality of the P‐gp seem significant better in for carriers of the rs4148739 G allele resulting in a slower antidepressant response.
Some original studies performed after the introduction of tricyclic antidepressants and monoamine oxidase inhibitors, have suggested a lag time in the true antidepressant response of two to four weeks (Quitkin, et al., 1984). Several other studies however disagreed with these findings and report no delayed onset of antidepressant action (Stassen et al., 2007; Taylor, 2007). To gain more clarity on this, we examined the first two weeks and the last two weeks separately. We observed that the response was greater during the first two weeks than during the last; therefore no question of a lag time existed. Whether the early antidepressant effects observed in the first two weeks of treatment are specific or non‐specific can be considered a debatable matter. In recent decades, the criteria regarding major depressive disorders have changed substantially. The characteristics of a major depressive disorder according to DSM‐IV criteria differ considerably from endogenous major depressive disorder (MDD) according to original criteria as for example described in the research diagnostic criteria (RDC) and have become far less specific (Loonen, 1997). As a result, the symptom profiles of patients currently diagnosed with MDD are more heterogeneous then those with endogenous depressive disorders in the past.
We hypothesize the antidepressant effects observed are mainly caused by activity reduction within a ventral extrapyramidal circuit regulating the intensity of distress‐avoiding behavior (Loonen & Ivanova, 2016). Our observation that certain ABCB1 gene polymorphisms are associated with the timing of the antidepressant response does suggest that a pharmacological effect contributes to the clinical response: P‐gp determines the rate at which cerebral receptors are reached and the level of concentration.
A limitation of our study is the relatively short observation period of only four weeks. This may be considered a too short period when looking for specific antidepressant effects (Quitkin, et al., 1984). However, the HAMD‐17 score improved within this time‐period to such an extent that enlargement of the observation time would probably have flawed the results by limiting the room for further improvement. We have also limited ourselves to studying the influence of the ABCB1 gene, although it may be minor compared to the well‐known influence of CYP2C19 and CYP2D6 genes, because the influence of this gene is mechanistically very interesting.
Another limitation is the naturalistic design and moderate population size. Small differences in the overall response could easily be missed, what might explain these negative results. Moreover, this also resulted in small numbers of included patients in some genotype groups. As we did not measure plasma levels, non‐compliance could not be discovered and inter‐individual differences in the extent of absorption and elimination of the antidepressants are not detected. There is however no reason to assume that non‐compliance was not randomly distributed between the studied genotypes and all patients were treated as in‐patients. Another relative limitation is the predominance of women in the sample of patients (n = 134), which is associated with a more frequent admission to the hospital in connection with affective disorders of women, rather than men. But in the context of the issue under discussion, this circumstance was verified not to play a significant role in assessing the results obtained. A strong point of our study is that all patients were treatment free for at least six months, which excludes all carry‐over effects of recently used antidepressants on the measured ΔHAMD‐17 improvement. More than half of our patients had never used an antidepressant in their life, so they can be considered to be completely antidepressant treatment naïve. Our study, in which we specifically addressed the timing of antidepressant response, is intended as a hypothesis generating study, and needs verification in a randomized design.
5. CONCLUSION
In conclusion, tricyclic antidepressants showed a higher improvement of treatment outcome over four weeks compared to SSRIs in our patient population. Polymorphisms rs2235040 and rs4148739 were shown to affect the timing of the antidepressant response. The improvement in ΔHAMD‐17 score was significantly larger in the first two weeks of antidepressant treatment for carriers of the rs2235040 A allele and significantly smaller for carriers of the rs4148739 G allele. The improvement in ΔHAMD‐17 score in the second two weeks of treatment was in contrast significant higher for carriers of the rs4148739 G allele and smaller for carriers of the rs2235040 A and rs2235015 T allele. Since no significant difference in the improvement in the ΔHAMD‐17 score in the total period of four weeks was found for any of the SNP's, the SNP's seem to mainly influence the speed of antidepressant response rather than the response rate. Our findings indicate that in our population pharmacological effects do contribute to this early antidepressant response (i.e., are not solely attributable to a placebo effect).
CONFLICT OF INTEREST
The authors declare no conflict of interest.
AUTHOR CONTRIBUTIONS
Conceptualization: A.J.M. Loonen (AJML), S.A. Ivanova (SAI) and D.J. Touw (DJT); Methodology: AJML, SAI, Taichi Ochi (TO), Lisanne M. Geers (LMG); Validation: AJML, SAI, DJT; Formal Analysis: TO; Investigation: German G. Simutkin (GGS), Natalya M. Vyalova (NMV), Innokentiy S.Losenkov (ISL), I.V.Pozhidaev (IVP), D.Z.Paderina (DZP); Resources: SAI, B. Wilffert (BW); Data Curation: ISL, TO, LMG; Writing ‐ Original Draft: LMG, TO, AJML; Writing ‐ Review & Editing: AJML, SAI, DJT; Visualization: TO, LMG; Supervision: AJML, SAI, BW, DJT, N.A. Bokhan (NAB); Project administration: SAI, BW, DJT. All authors read the paper, when necessary commented on it and agree with its content. Their role justifies their (co)‐authorship to this paper.
Supporting information
Supplementary Material 1
ACKNOWLEDGMENTS
This work resulted from a collaboration between the Mental Health Research Institute (Tomsk National Research Medical Center of the Russian Academy of Sciences) in Tomsk and the Groningen Research Institute of Pharmacy (GRIP) of the University of Groningen. The Russian part is carried out within the framework of Tomsk Polytechnic University Competitiveness Enhancement Program without financial support. This research did not receive any specific grant from funding agencies in the public, commercial, or not‐for‐profit sectors.
Geers, L. M. , Ochi, T. , Vyalova, N. M. , Losenkov, I. S. , Paderina, D. Z. , Pozhidaev, I. V. , Simutkin, G. G. , Bokhan, N. A. , Wilffert, B. , Touw, D. J. , Loonen, A. J. M. , & Ivanova, S. A. (2022). Influence of eight ABCB1 polymorphisms on antidepressant response in a prospective cohort of treatment‐free Russian patients with moderate or severe depression: An explorative psychopharmacological study with naturalistic design. Human Psychopharmacology: Clinical and Experimental, 37(3), e2826. 10.1002/hup.2826
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- Ambudkar, S. V. , Dey, S. , Hrycyna, C. A. , Ramachandra, M. , Pastan, I. , & Gottesman, M. M. (1999). Biochemical, cellular, and pharmacological aspects of the multidrug transporter. Annual Review of Pharmacology and Toxicology, 39, 361–398. 10.1146/annurev.pharmtox.39.1.361 [DOI] [PubMed] [Google Scholar]
- Anderson, I. M. (2000). Selective serotonin reuptake inhibitors versus tricyclic antidepressants: A meta‐analysis of efficacy and tolerability. Journal of Affective Disorders, 58, 19–36. 10.1016/s0165-0327(99)00092-0 [DOI] [PubMed] [Google Scholar]
- Bauer, M. , Pfennig, A. , Severus, E. , Whybrow, P. C. , Angst, J. , & Moeller, H. , & World Federation of Societies of Biological Psychiatry ‐ Task Force on Unipolar Depressive Disorders . (2013). World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for biological treatment of unipolar depressive disorders, part 1: Update 2013 on the acute and continuation treatment of unipolar depressive disorders. World Journal of Biological Psychiatry, 14, 334–385. 10.3109/15622975.2013.804195 [DOI] [PubMed] [Google Scholar]
- Breitenstein, B. , Brückl, T. M. , Ising, M. , Müller‐Myhsok, B. , Holsboer, F. , & Czamara, D. (2015). ABCB1 gene variants and antidepressant treatment outcome: A meta‐analysis. American Journal of Medical Genetics Part B Neuropsychiatric Genetics, 168B, 274–283. 10.1002/ajmg.b.32309 [DOI] [PubMed] [Google Scholar]
- Cordon‐Cardo, C. , O'Brien, J. P. , Casals, D. , Rittman‐Grauer, L. , Biedler, J. L. , Melamed, M. R. , & Bertino, J. R. (1989). Multidrug‐resistance gene (P‐glycoprotein) is expressed by endothelial cells at blood‐brain barrier sites. Proceedings of the National Academy of Sciences of the United States of America, 86, 695–698. 10.1073/pnas.86.2.695 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doran, A. , Obach, R. S. , Smith, B. J. , Hosea, N. A. , Becker, S. , Callegari, E. , Chen, C. , Chen, X. , Choo, E. , Cianfrogna, J. , Cox, L. M. , Gibbs, J. P. , Gibbs, M. A. , Hatch, H. , Hop, C. E. , Kasman, I. N. , Laperle, J. , Liu, J. , Liu, X. , … Zhang, C. (2005). The impact of P‐glycoprotein on the disposition of drugs targeted for indications of the central nervous system: Evaluation using the MDR1A/1B knockout mouse model. Drug Metabolism & Disposition, 33, 165–174. 10.1124/dmd.104.001230 [DOI] [PubMed] [Google Scholar]
- Eichelbaum, M. , Fromm, M. F. , & Schwab, M. (2004). Clinical aspects of the MDR1 (ABCB1) gene polymorphism. Therapeutic Drug Monitoring, 26, 180–185. 10.1097/00007691-200404000-00017 [DOI] [PubMed] [Google Scholar]
- Ejsing, T. B. , Hasselstrøm, J. , & Linnet, K. (2006). The influence of P‐glycoprotein on cerebral and hepatic concentrations of nortriptyline and its metabolites. Drug Metabolism and Drug Interactions, 21, 139–162. 10.1515/dmdi.2006.21.3-4 [DOI] [PubMed] [Google Scholar]
- Fabbri, C. , Di Girolamo, G. , & Serretti, A. (2013). Pharmacogenetics of antidepressant drugs: An update after almost 20 years of research. American Journal of Medical Genetics Part B: Neuropsychiatric Genetics, 162, 487–520. 10.1002/ajmg.b.32184 [DOI] [PubMed] [Google Scholar]
- GBD 2017 Oral Disorders Collaborators , Bernabe, E. , Marcenes, W. , Hernandez, C. R. , Bailey, J. , Abreu, L. G. , Alipour, V. , Amini, S. , Arabloo, J. , Arefi, Z. , Arora, A. , Ayanore, M. A. , Barnighausen, T. W. , Bijani, A. , Cho, D. Y. , Chu, D. T. , Crowe, C. S. , Demoz, G. T. , Demsie, D. G. , Dibaji Forooshani, Z. S. , … Kassebaum, N. J. (2020). Global, regional, and national levels and trends in burden of oral conditions from 1990 to 2017: A systematic analysis for the global burden of disease 2017 study. Journal of Dental Research, 99, 362–373. 10.1177/0022034520908533 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gex‐Fabry, M. , Eap, C. B. , Oneda, B. , Gervasoni, N. , Aubry, J. , Bondolfi, G. , & Bertschy, G. (2008). CYP2D6 and ABCB1 genetic variability: Influence on paroxetine plasma level and therapeutic response. Therapeutic Drug Monitoring, 30, 474–482. 10.1097/FTD.0b013e31817d6f5d [DOI] [PubMed] [Google Scholar]
- Hoffmeyer, S. , Burk, O. , von Richter, O. , Arnold, H. P. , Brockmoller, J. , Johne, A. , Cascorbi, I. , Gerloff, T. , Roots, I. , Eichelbaum, M. , & Brinkmann, U. (2000). Functional polymorphisms of the human multidrug‐resistance gene: Multiple sequence variations and correlation of one allele with P‐glycoprotein expression and activity in vivo. Proceedings of the National Academy of Sciences of the United States of America, 97, 3473–3478. 10.1073/pnas.050585397 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato, M. , Fukuda, T. , Serretti, A. , Wakeno, M. , Okugawa, G. , Ikenaga, Y. , Hosoi, Y. , Takekita, Y. , Mandelli, L. , Azuma, J. , & Kinoshita, T. (2008). ABCB1 (MDR1) gene polymorphisms are associated with the clinical response to paroxetine in patients with major depressive disorder. Progress In Neuro‐Psychopharmacology & Biological Psychiatry, 32, 398–404. 10.1016/j.pnpbp.2007.09.003 [DOI] [PubMed] [Google Scholar]
- Loonen, A. J. M. (1997). Are SSRIs true antidepressant drugs? A plea for the re‐evaluation of their therapeutic potential and safety. Pharmacy World and Science, 19, 70–72. 10.1023/a:1008669823508 [DOI] [PubMed] [Google Scholar]
- Loonen, A. J. M. , & Ivanova, S. A. (2016). Circuits regulating pleasure and happiness in major depression. Medical Hypotheses, 87, 14–21. 10.1016/j.mehy.2015.12.013 [DOI] [PubMed] [Google Scholar]
- Murray, C. J. , & Lopez, A. D. (1996). Evidence‐based health policy‐‐lessons from the global burden of disease study. Science, 274, 740–743. 10.1126/science.274.5288.740 [DOI] [PubMed] [Google Scholar]
- Nikisch, G. , Eap, C. B. , & Baumann, P. (2008). Citalopram enantiomers in plasma and cerebrospinal fluid of ABCB1 genotyped depressive patients and clinical response: A pilot study. Pharmacological Research, 58, 344–347. 10.1016/j.phrs.2008.09.010 [DOI] [PubMed] [Google Scholar]
- Ochi, T. , Vyalova, N. M. , Losenkov, I. S. , Levchuk, L. A. , Osmanova, D. Z. , Mikhalitskaya, E. V. , Loonen, A. J. , Bosker, F. J. , Simutkin, G. G. , Bokhan, N. A. , Wilffert, B. , & Ivanova, S. A. (2019). Investigating the potential role of BDNF and PRL genotypes on antidepressant response in depression patients: A prospective inception cohort study in treatment‐free patients. Journal of Affective Disorders, 259, 432–439. 10.1016/j.jad.2019.08.058 [DOI] [PubMed] [Google Scholar]
- Perlis, R. H. , Fijal, B. , Dharia, S. , Heinloth, A. N. , & Houston, J. P. (2010). Failure to replicate genetic associations with antidepressant treatment response in duloxetine‐treated patients. Biological Psychiatry, 67, 1110–1113. 10.1016/j.biopsych.2009.12.010 [DOI] [PubMed] [Google Scholar]
- Porcelli, S. , Fabbri, C. , Drago, A. , Gibiino, S. , De Ronchi, D. , & Serretti, A. (2011). Genetics and antidepressants: Where we are. Clin. Neuropsychiatry, 8, 99–150. [Google Scholar]
- Posternak, M. A. , & Zimmerman, M. (2005). Is there a delay in the antidepressant effect? A meta‐analysis. Journal of Clinical Psychiatry, 66, 148–158. 10.4088/jcp.v66n0201 [DOI] [PubMed] [Google Scholar]
- Quitkin, F. M. , Rabkin, J. G. , Ross, D. , & Stewart, J. W. (1984). Identification of true drug response to antidepressants: Use of pattern analysis. Archives of General Psychiatry, 41, 782–786. 10.1001/archpsyc.1984.01790190056007 [DOI] [PubMed] [Google Scholar]
- Rush, A. J. , Trivedi, M. H. , Wisniewski, S. R. , Nierenberg, A. A. , Stewart, J. W. , Warden, D. , Niederehe, G. , Thase, M. E. , Lavori, P. W. , Lebowitz, B. D. , McGrath, P. J. , Rosenbaum, J. F. , Sackeim, H. A. , Kupfer, D. J. , Luther, J. , & Fava, M. (2006). Acute and longer‐term outcomes in depressed outpatients requiring one or several treatment steps: A STAR* D report. American Journal of Psychiatry, 163, 1905–1917. 10.1176/ajp.2006.163.11.1905 [DOI] [PubMed] [Google Scholar]
- Sarginson, J. E. , Lazzeroni, L. C. , Ryan, H. S. , Ershoff, B. D. , Schatzberg, A. F. , & Murphy, G. M., Jr (2010). ABCB1 (MDR1) polymorphisms and antidepressant response in geriatric depression. Pharmacogenetics and Genomics, 20, 467–475. 10.1097/FPC.0b013e32833b593a [DOI] [PubMed] [Google Scholar]
- Schatzberg, A. F. , DeBattista, C. , Lazzeroni, L. C. , Etkin, A. , Murphy, G. M., Jr , & Williams, L. M. (2015). ABCB1 genetic effects on antidepressant outcomes: A report from the iSPOT‐D trial. American Journal of Psychiatry, 172, 751–759. 10.1176/appi.ajp.2015.14050680 [DOI] [PubMed] [Google Scholar]
- Schwab, M. , Eichelbaum, M. , & Fromm, M. F. (2003). Genetic polymorphisms of the human MDR1 drug transporter. Annual Review of Pharmacology and Toxicology, 43, 285–307. 10.1146/annurev.pharmtox.43.100901.140233 [DOI] [PubMed] [Google Scholar]
- Shan, X. X. , Qiu, Y. , Xie, W. W. , Wu, R. R. , Yu, Y. , Wu, H. S. , & Li, L. H. (2019). ABCB1 gene is associated with clinical response to SNRIs in a local Chinese han population. Frontiers in Pharmacology, 10, 761. 10.3389/fphar.2019.00761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheehan, D. V. , Lecrubier, Y. , Sheehan, K. H. , Amorim, P. , Janavs, J. , Weiller, E. , Hergueta, T. , Baker, R. , & Dunbar, G. C. (1998). The mini‐international neuropsychiatric interview (MINI): The development and validation of a structured diagnostic psychiatric interview for DSM‐IV and ICD‐10. Journal of Clinical Psychiatry, 59(Suppl 20), 22–33. quiz 34‐57. [PubMed] [Google Scholar]
- Simoons, M. , Mulder, H. , Appeldoorn, J. T. , Risselada, A. J. , Schene, A. H. , van Schaik, R. H. , van Roon, E. N. , & Ruhé, E. G. (2020). Modification of the association between paroxetine serum concentration and SERT‐occupancy by ABCB1 (P‐glycoprotein) polymorphisms in major depressive disorder. Psychiatric Genetics, 30, 19–29. 10.1097/YPG.0000000000000244 [DOI] [PubMed] [Google Scholar]
- Singh, A. , Bousman, C. , Ng, C. , Byron, K. , & Berk, M. (2012). ABCB1 polymorphism predicts escitalopram dose needed for remission in major depression. Translational Psychiatry, 2, e198. 10.1038/tp.2012.115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stassen, H. H. , Angst, J. , & Delini‐Stula, A. (1996). Delayed onset of action of antidepressant drugs? Survey of results of Zurich meta‐analyses. Pharmacopsychiatry, 29(3), 87–96. 10.1055/s-2007-979551 [DOI] [PubMed] [Google Scholar]
- Stassen, H. H. , Angst, J. , Hell, D. , Scharfetter, C. , & Szegedi, A. (2007). Is there a common resilience mechanism underlying antidepressant drug response? Evidence from 2848 patients. Journal of Clinical Psychiatry, 68, 1195–1205. 10.4088/jcp.v68n0805 [DOI] [PubMed] [Google Scholar]
- Taylor, M. J. (2007). Rapid onset of true antidepressant action. Current Psychiatry Reports, 9, 475–479. 10.1007/s11920-007-0064-0 [DOI] [PubMed] [Google Scholar]
- Trimbos instituut (2013). GGZ‐Richtlijnen ‐ Multidisciplinaire Richtlijn Depressie 2020 (3e revisie).
- Uhr, M. , & Grauer, M. T. (2003). abcb1ab P‐glycoprotein is involved in the uptake of citalopram and trimipramine into the brain of mice. Journal of Psychiatric Research, 37, 179–185. 10.1016/s0022-3956(03)00022-0 [DOI] [PubMed] [Google Scholar]
- Uhr, M. , Grauer, M. T. , & Holsboer, F. (2003). Differential enhancement of antidepressant penetration into the brain in mice with abcb1ab (mdr1ab) P‐glycoprotein gene disruption. Biological Psychiatry, 54, 840–846. 10.1016/s0006-3223(03)00074-x [DOI] [PubMed] [Google Scholar]
- Uhr, M. , Steckler, T. , Yassouridis, A. , & Holsboer, F. (2000). Penetration of amitriptyline, but not of fluoxetine, into brain is enhanced in mice with blood‐brain barrier deficiency due to mdr1a P‐glycoprotein gene disruption. Neuropsychopharmacology, 22, 380–387. 10.1016/S0893-133X(99)00095-0 [DOI] [PubMed] [Google Scholar]
- Uhr, M. , Tontsch, A. , Namendorf, C. , Ripke, S. , Lucae, S. , Ising, M. , Dose, T. , Ebinger, M. , Rosenhagen, M. , Kohli, M. , Kloiber, S. , Salyakina, D. , Bettecken, T. , Specht, M. , Pütz, B. , Binder, E. B. , Müller‐Myhsok, B. , & Holsboer, F. (2008). Polymorphisms in the drug transporter gene ABCB1 predict antidepressant treatment response in depression. Neuron, 57, 203–209. 10.1016/j.neuron.2007.11.017 [DOI] [PubMed] [Google Scholar]
- van Calker, D. , Zobel, I. , Dykierek, P. , Deimel, C. M. , Kech, S. , Lieb, K. , Berger, M. , & Schramm, E. (2009). Time course of response to antidepressants: Predictive value of early improvement and effect of additional psychotherapy. Journal of Affective Disorders, 114, 243–253. 10.1016/j.jad.2008.07.023 [DOI] [PubMed] [Google Scholar]
- Wang, J. , Zhu, H. , Gibson, B. B. , Markowitz, J. S. , Donovan, J. L. , & DeVane, C. L. (2008). Sertraline and its metabolite desmethylsertraline, but not bupropion or its three major metabolites, have high affinity for P‐glycoprotein. Biological and Pharmaceutical Bulletin, 31, 231–234. 10.1248/bpb.31.231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams, J. W. , Mulrow, C. D. , Chiquette, E. , Noel, P. H. , Aguilar, C. , & Cornell, J. (2000). A systematic review of newer pharmacotherapies for depression in adults: Evidence report summary: Clinical guideline, part 2. Annals of Internal Medicine, 132, 743–756. 10.7326/0003-4819-132-9-200005020-00011 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material 1
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


