With approximately 540 million cases, of which 226 million have occurred in Europe as of the end of June 2022 (https://covid19.who.int/), COVID-19 is a global disease. To date, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection has significantly impacted the activity of nuclear medicine departments because of the large number of patients infected, including staff caregivers [1] and because of the necessary reorganization of medical procedures and hospital circuits [2]. Moreover, new disease entities have emerged with long COVID [3]. The definition of long COVID has evolved over time with increasing knowledge and the updating of national and international recommendations, particularly aspects concerning the type and duration of symptoms, the time from symptom onset, and the necessity of biological COVID confirmation. Finally, in October 2021, the World Health Organization (WHO) defined long COVID as a condition that occurs in individuals with a history of probable or biologically confirmed SARS-CoV-2 infection (to not exclude patients without access to tests), initially symptomatic at the acute phase, with numerous symptoms lasting for at least 2 months, usually 3 months from the onset of COVID-19, that cannot be explained by an alternative diagnosis (https://www.who.int/publications/i/item/WHO-2019-nCoV-Post_COVID-19_condition-Clinical_case_definition-2021.1). By affecting approximately 10–15% of patients [3], long COVID represents a serious global public health issue. These patients are mostly rather young, previously active, and a majority of women; they potentially present chronic forms with a disability leading to significant medico-economic costs [4].
While long COVID is viewed as a multisystemic disease due to the ubiquitous distribution of ACE2 receptors as a virus entry pathway into cells, cerebral impairment has received special attention because of symptoms of possible brain origin: loss of smell and taste, cognitive complaints of brain “fog,” impairment in attentional and memory functions, sleep disturbances, pain, emotion disorders [5], and other symptoms that might be related to dysautonomia, such as breathlessness, tachycardia, and orthostatic intolerance, including orthostatic hypotension [6]. These heterogeneous long COVID symptoms, which also potentially fluctuate over time, are firstly not exclusively specific to genuine brain involvement, and may also correspond in some cases to another disease potentially unmasked by the infection. For example, extracerebral explanations might be assigned to a loss of smell in individuals with a nasal obstruction, to dyspnoea in individuals with lung sequelae, and even in individuals with cognitive complaints presenting severe asthenia. On the other hand, several hypotheses have been proposed to anticipate potential cerebral damage related to the infection [5, 6]. Indeed, acute neurological complications associated with COVID-19 [7, 8], such as stroke or encephalopathy/encephalitis, suggest a possible direct or indirect effect of SARS-CoV-2 on the brain. Moreover, other SARS viruses were similarly associated with neuropsychiatric presentations [9]. Consistent with these findings, a recent meta-analysis showed that the post-illness stage of previous infections with SARS viruses was characterized by memory impairment in 19% of patients, irritability in 13%, insomnia and anxiety in 12%, and a depressed mood in 10% of patients [9]. These associations with neuropsychiatric symptoms were even observed in patients infected with other respiratory viruses, such as the “Spanish” pandemic flu virus in adults and children [10]. Some authors consequently argued for possible direct viral brain invasion [11], while others proposed that cerebral damage might be related to an extensive inflammatory/immune response related to the infection [12]. The two hypotheses may also be linked and could suggest a possible brain pathway. A trans-ethmoidal propagation from the olfactory bulbs through the lamina cribrosa to connected cerebral regions, namely, from the nose to the brain, has been proposed, while alternative or complementary pathways might involve pulmonary innervation or the vascular compartment [13]. Notably, an impairment of olfactory brain areas and their projections to the limbic/paralimbic system, brainstem nuclei, and cerebellum might explain several symptoms of long COVID in sensorial, cognitive, motor, and autonomic dimensions, as well as potentially those in the emotion spectrum. Patients indeed also present psychological and psychiatric symptoms, which are potentially related to the stress associated with the pandemic context, carelessness, chronic evolution, and disability, but which might also be related to brain damages in emotion networks [14]. Overall, neurological and psychiatric symptoms presented by patients with long COVID could at least partially be related to the impairment of the same brain network [15]: the olfactory symptoms with the impairment of the olfactory bulbs; the memory/attention deficits with those of the hippocampus and cingulum; the psychological and psychiatric disorders such as irritability, depression, or anxiety with changes in amygdalae; tachycardia, orthostatic intolerance, or breathlessness as a dysfunction of the autonomic nervous system on brainstem nuclei (dysautonomia); and motor symptoms with the impairment of the cerebellum. Consistent with this hypothesis, the olfactory bulbs have indeed been proposed as a possible pathway in this implicated brain network, as all involved regions are anatomically linked to these olfactory structures [16].
Given the subtle clinical presentations and the heterogeneity of individual profiles, a diagnostic flow chart for a positive diagnosis of long COVID is currently difficult to define. In the future, this diagnosis might be based on a composite definition of clinical, biological, and imaging signatures. At present, biomarkers should first be considered to exclude other underlying aetiologies with respect to the current WHO definition. These biomarkers could also specify the extent of the disease, especially the possible impairment of the brain, which may correspond to both a specific prognosis and management. Nuclear medicine using brain 2-[18F]-FDG (FDG) PET imaging potentially fills these medical needs; however, the observation of cortical metabolic abnormalities and their interpretation at the single-patient level should also be carefully considered based on the patient’s clinical history and potential confounders. The EANM Neuroimaging Committee hereby aims to summarize and discuss potential applications, pitfalls, and prospects of the use of FDG PET to image brain involvement in patients with long COVID.
Brain FDG PET imaging of patients with long COVID in the literature
Brain FDG PET was first employed in patients with post-COVID-19 syndrome at the individual and group levels in monocentric semi-quantitative studies [17–20] including relatively small cohorts of patients [21 for a review]. A hypometabolic long COVID pattern was reported in the study including a larger number of 35 patients with persistent functional complaints at an average of 14 weeks after the initial infection compared to healthy subjects. This hypometabolic profile involves the fronto-orbital and olfactory regions, other limbic/paralimbic regions, the brainstem, and the cerebellum [18]. At this relatively early time from symptom onset, this metabolic pattern was observed in all patients at the individual level. The included patients presented functional complaints associated with the possible brain origin but were free from any neuropsychiatric antecedent and had normal morphological imaging data. Interestingly, a statistical association was observed between anosmia and memory impairment, as well as between the number of symptoms and the severity of hypometabolism. Similarly in this study, patients who had nasal irrigations in the acute phase presented less-pronounced olfactory hypometabolism, whereas those treated with ACE drugs had more-pronounced olfactory hypometabolism. Without prejudging the mechanism (viral, inflammatory, or immune) and its potential reversibility, these results reinforced the hypothesis of a “connected” impairment related to olfactory propagation from ACE2 receptors. The two other PET studies of a series of adult patients included 13 and 14 patients scanned at 19 and 28 weeks on average from symptom onset, respectively [19, 20]. The first study showed a decrease in metabolism in the right parahippocampal gyrus and thalamus compared to control patients without long COVID, associations between decreased metabolism in parahippocampal and orbitofrontal areas and persistent anosmia, and associations between decreased metabolism in the parahippocampal gyrus, brainstem, and thalamus and persistent fatigue [19]. The second study performed at a later time from symptom onset did not observe any metabolic decrease in patients with subjective complaints at the group or at the individual levels. In this last study (and interestingly not in the previously mentioned studies), comparisons were performed by normalizing the brain metabolism on the white matter [20]. The hypometabolic long COVID pattern was finally described in 7 paediatric patients who presented symptoms with a possible brain origin several weeks after COVID compared to control paediatric patients presenting functional complaints before the pandemic, with a similar brain impairment as those found in adults, involving the right olfactory gyrus, bilateral medial temporal lobes, the brainstem, and cerebellum [22].
It is worth noting that the comparability of the results provided by these studies is limited by differences both in their included populations and in the technical parameters used. The studied populations were heterogeneous as they included in- or outpatients, presenting complaints or deficits, with or without structural brain lesions. On the other hand, the choice of the population to compare the brain metabolism of patients may have also interfered with the different results obtained across these studies. Healthy subjects or normal controls (patients with apparent normal metabolism) served as references, and PET images were obtained with or without the same PET camera as the one used for patients with long COVID, along with the same or a distinct protocol of acquisition and reconstruction. Notably, brain analyses were also performed across these studies using different methodological strategies, especially related to the aforementioned activity normalization methods. Finally, the time from symptom onset is certainly the major factor interfering with symptoms and PET results, considering possible brain recovery [23]. All these points are in favour of future larger multicentre studies with harmonized and standardized protocols for inclusion, data acquisition, and image processing.
Nevertheless, a similar pattern of long COVID-19-affected brain areas as those reported with brain FDG PET was observed in a large-scale MRI morphometric longitudinal study of 401 patients who underwent two morphological MRIs before the pandemic and after diagnosis with COVID-19 at an average time from symptom onset of 141 days and compared 384 longitudinal noninfected controls [24]. The authors reported a global reduction in brain size after SARS-CoV-2 infection, with greater tissue damage in areas that are functionally connected to the primary olfactory cortex and a more marked reduction in grey matter thickness in the orbitofrontal cortex and parahippocampal gyrus [24]. This study confirms that the observed abnormalities did not result from a predisposing condition before COVID. Taken together, the results of the metabolic PET studies, the results of the large-scale longitudinal MRI study, and a systematic review integrating neuroimaging and neuropathological findings of 90 previous studies in patients with COVID-19 [25] suggest the involvement of a common brain network and a possible olfactory pathway, without speculating on the exact mechanism and its reversibility.
What is the significance of the long COVID hypometabolic pattern in FDG PET images?
Although the hypometabolic long COVID brain profile described above seems consistent with hypotheses and findings obtained for other viruses before the COVID-19 outbreak, with symptoms reported by patients with long COVID, and with results obtained from other diagnostic approaches assessing long COVID, the significance of these metabolic abnormalities in the individual patients remains to be determined. Specifically, questions have even been raised to clarify whether these abnormalities might be the consequences of the symptoms and not the causes. We believe that this last question may mostly result from a possible misunderstanding or confusion between the pathological process associated with individual metabolic PET findings in the resting state and the functional consequence of symptoms and deficits revealed by multiple scan activation paradigm using MRI. The functional dimension that is mainly investigated in neuroscience with fMRI is undoubtedly captured using various methods, including morphological and molecular imaging [26], but always on a multiple image scale because of low contrast. In contrast, and in clinical settings, brain FDG PET is an individual biomarker of high contrast recommended to explore pathological processes [27]. Therefore, individual PET hypometabolism should correspond more to a genuine brain impairment, again without prejudging its reversibility, than to a simple consequence of the symptoms. To add to this discussion, recently, at an individual level and using a visual interpretation, a French multicentre study was conducted. Here, in 143 patients diagnosed in accordance with the current WHO definition of post-COVID syndrome, a hypometabolic long COVID pattern was observed in 47% of patients presenting persistent symptoms at an average of 10.9 months after COVID-19 [28]. Interestingly, the fact that at the individual level, approximately half of patients with symptoms had no brain metabolic abnormalities at this time [28], an incomplete hypometabolic pattern [19], or no metabolic abnormalities [20] support the other above-cited studies in small cohorts of patients with suspicious long COVID which indicate that this hypometabolic pattern is not simply a functional consequence of symptoms, i.e. a deafferentation from the olfactory bulbs [16], but again more likely the signature of a genuine brain impairment.
We can also argue that no or few abnormalities have been reported at the individual level upon a visual interpretation of images from patients with psychiatric disorders, and especially no individual abnormalities in the brainstem and cerebellum [29]. In addition to psychiatric explanations, some authors have linked long COVID symptoms with a similar functional explanation to possible deconditioning related to the lockdown. Interestingly, the reported hypometabolic long COVID pattern differs from that of the deconditioning state reported in the COVID-19 lockdown in a study including approximately 500 patients, with less than 9% concordance between the two conditions [30].
Brain FDG PET imaging of long COVID: clinical implications
In addition to being an efficient tool in the framework of European recommendations for the differential diagnosis of a neurodegenerative disorder, encephalitis/encephalopathy (including post-infective) [27], brain FDG PET might thus be a promising biomarker to objectively assess brain involvement in patients with a suspicion of long COVID [28]. However, several red flags should be known by nuclear physicians for the correct interpretation of brain FDG PET scans at the individual level in this context. Nuclear physicians should be aware of specific brain areas involved in the hypometabolic long COVID pattern that are sometimes difficult to individualize anatomically and require experienced readers. We should cautiously interpret findings from the fronto-basal regions, especially the olfactory bulbs and their nervous extension from the gyrus rectus, at best on coronal slices, but also the brainstem and the cerebellum by searching for hypometabolism with adapted contrast on colour scales, as well as the medial temporal lobe (see Figs. 1 and 2 for details). Moreover, subtle changes in metabolism and, a fortiori, a partial hypometabolism in one of these brain structures, especially in less specific limbic regions, should not systematically be reported as related to long COVID [28]. Although the hypometabolic long COVID pattern has been described in children [22], an extra-careful, high level-experienced interpretation is needed in paediatric patients since the relative hypometabolism of internal temporal areas, pons and cerebellum, is classically observed (and significantly varies with the age of the child) in such patients with brain FDG PET [31]. Importantly, intensity normalization should preferably be obtained at the global cortical level or centred on the maximal cortical voxel since other references, such as white matter in MRI studies [32] and pons or cerebellum in MRI and PET studies [17, 22, 24, 28], may be involved in long COVID [33]. In addition to the previously mentioned impairment of the brainstem and cerebellum in imaging studies [17, 24, 28], post-mortem autopsy studies documented microglial activation in white matter of patients with COVID [34], limiting the option in employing this location as reference region for intensity normalization. Semi-quantitative analyses should be a helpful complement to visual analysis, especially for less-experienced readers, considering the nature of the control database, which must be age-matched to the patient and obtained with a PET system with similar physical performances [35]. An excessively restrictive brain inclusion mask should not be used for this purpose, as it will potentially limit the interpretation of the gyrus rectus and olfactory nerve projections.
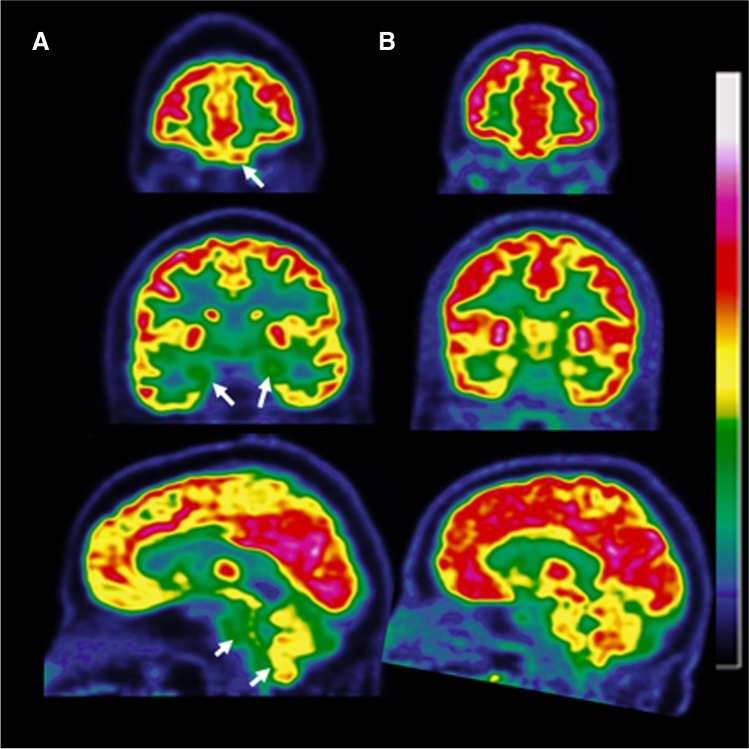
Fig. 1.
Hypometabolic pattern in a patient with long COVID-19 presenting normal MRI (A) and normal brain FDG PET scans of an age-matched control subject (B). The hypometabolic long COVID pattern involves the fronto-orbital and olfactory regions and other limbic/paralimbic regions as visualized in coronal slices, and the brainstem and the cerebellum as visualized in sagittal slices (white arrows in A)
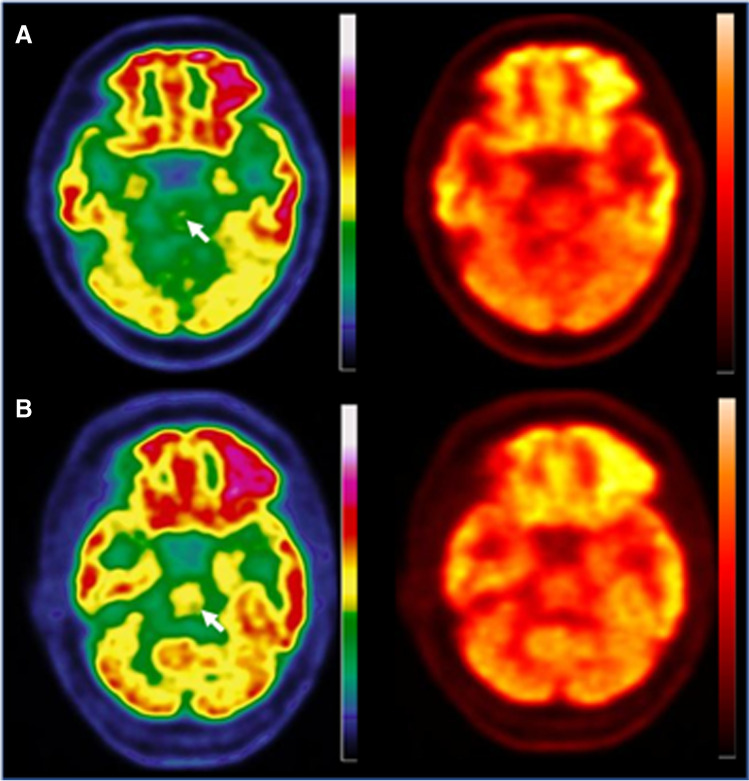
Fig. 2.
Axial slices of a brain FDG PET scan of a patient with a hypometabolic long COVID pattern (A) and in a patient with a normal brain scan (B). A discontinuous colour scale (left column) is better suited than a continuous scale (right column) to indicate hypometabolism in the pons (white arrow)
Notably, in this potentially large population with a suspicion of long COVID, the discovery of a pre-existing neurodegenerative or psychiatric disorder may not be infrequent. Nuclear physicians should be aware of the risk of overdiagnosis with another pathology or pre-existing pathology falsely attributed to COVID. Indeed, in many patients, including those with Alzheimer’s disease, the hypometabolic pattern is expected to differ from the hypometabolic profile described for long COVID [27]. If a more inclusive or partially overlapping pattern is observed, a tight correlation with the clinical presentation (including first symptom onset) is needed. Clinical and, in more complex cases, imaging follow-ups might be considered to establish the final in vivo diagnosis. Although measurable residuals have been reported, some significant recovery of regional neuronal dysfunction and cognition has been reported in patients who have recovered from COVID [7, 8]. In contrast, a risk of underdiagnosis also exists because of the high prevalence of the long COVID pathology (from 10 to 15% of patients having presented COVID, [3]). This risk is noted for patients who are referred to a brain FDG PET scan for a suspicion of another pathology in whom a long COVID pattern is fortuitously discovered.
Overall, brain FDG PET might be used in long COVID patients to exclude a different diagnosis, especially in individuals with atypical presentations, and in positive cases, to identify a brain involvement. This result could potentially contribute to the social and medical recognition of the patient’s disability by the health care system. Multiple rehabilitation procedures would be available for these patients, who are often young and socially active, focusing on cerebral networks observed to be affected in neurological long COVID, i.e. the olfactory pathway, as well as the limbic, cognitive, and motor pathways [36]. More broadly, brain FDG PET might be an additional tool to monitor these rehabilitation procedures [27].
Perspectives
To provide higher-level scientific evidence supporting the above assumptions, especially for a composite positive diagnosis including PET imaging, large-scale prospective studies, ideally with post-mortem histopathological comparisons, should be encouraged. Novel PET radiotracers should also be employed in research to assess neurological long COVID. As neuroinflammation is one of the main hypotheses to explain brain damage in patients with long COVID [12], neuroinflammation PET targeting, for instance, the translocator protein 18 kDa (TSPO)—using full kinetic modelling as no adequate reference region is available—might be a good candidate [37]. In addition to the current burden of long COVID on our patients, society might even experience a larger issue in the near future, as neuroinflammation is a commonly accepted risk factor for neurodegenerative diseases [38]. Furthermore, significantly increased plasma levels of neuronal biomarkers, including amyloid beta, neurofilament light chain, neurogranin, total tau, and p-T181-tau, were detected in patients 1 to 3 months after recovery from COVID-19 infection compared to historic controls [39]. The presence of increased levels of neuronal biomarkers associated with COVID-19 infection motivates to carry out investigation with amyloid and tau PET imaging. In addition, a nigrostriatal dopaminergic deficit was suspected in patients 2 to 8 weeks after COVID-19 with 123I-FPCIT SPECT [40, 41] and 18F-FDOPA PET [42]. The association of neurological long COVID and neurodegenerative disorders clearly deserves further studies using longitudinal follow-ups. Additionally, an exploration of the potential reversibility of brain involvement using follow-up FDG PET is necessary [8, 43] to specify the possible transition from inflammatory hypermetabolism to hypometabolic dysfunctions [42]. Generally, more research is needed to improve our understanding on the incidence and individual risk to develop neurological sequelae after a COVID infection [44]. However, due to the large number of patients with COVID, the absolute numbers of individuals with neurological sequelae might be high.
Regarding the hypothesis of brain entry through the olfactory bulbs, intranasal therapy for long COVID is also under investigation [45], and exploring the effect of this treatment on the olfactory network using PET imaging is a line of future research.
Finally, brain FDG PET imaging might also be a biomarker of neuronal dysfunction in individuals with other infections for which cerebral involvement is suspected through similar mechanisms, as in other SARS viruses [9].
Conclusions and recommendations
Using brain FDG PET, a hypometabolic profile has been observed in a relevant portion of patients with long COVID, suggesting network-based involvement. Brain FDG PET might therefore be a promising approach to objectify brain involvement in individuals with long COVID, which should correspond to distinct prognoses and management strategies. In addition, brain FDG PET potentially provides a differential diagnosis for clinical symptoms related to neurodegenerative diseases, encephalitis/encephalopathy, or psychiatric disorders. In our opinion, and as previously proposed by Meyer et al. [21], this examination should be considered in these two frameworks only after a dedicated clinical evaluation and after a significant delay (3 to 6 months) from the initial infection, or in case of worsening [19]. Clinicians should remember that cerebral symptoms of long COVID are not limited to cognitive complaints, and consequently, a broader evaluation should be considered beyond the neuropsychometric assessment for these PET indications, especially considering dysautonomia. A multicentre international collaboration among clinicians and patients is expected to improve the brain FDG PET imaging evaluation of long COVID.
Declarations
Conflict of interest.
AV has received speaker honoraria from GE Healthcare and Philips.
HB has received reader honoraria from LMI and speaker honoraria from AAA/Novartis.
FF has received speaker honoraria and receives research funding from Siemens Healthineers.
SM has received speaker honoraria from GE Healthcare, Roche, and LMI and is an advisor of LMI.
EG has received consultant and speaker honoraria from GE Healthcare and CIS Bio International; and consultant honoraria from Advanced Accelerator Applications
The other authors declare that they have no conflict of interest
Research involving human participants and/or animals.
Not applicable.
Informed consent.
Not applicable.
Footnotes
This article is part of the Topical Collection on Neurology.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Guedj E, Verger A, Cammilleri S. PET imaging of COVID-19: the target and the number. Eur J Nucl Med Mol Imaging. 2020;47:1636–1637. doi: 10.1007/s00259-020-04820-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Morbelli S, Ekmekcioglu O, Barthel H, Albert NL, Boellaard R, Cecchin D, et al. COVID-19 and the brain: impact on nuclear medicine in neurology. Eur J Nucl Med Mol Imaging. 2020;47:2487–2492. doi: 10.1007/s00259-020-04965-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Greenhalgh T, Knight M, A’Court C, Buxton M, Husain L. Management of post-acute covid-19 in primary care. BMJ. 2020;370:m3026. doi: 10.1136/bmj.m3026. [DOI] [PubMed] [Google Scholar]
- 4.Chen C, Haupert SR, Zimmermann L, Shi X, Fritsche LG, Mukherjee B. Global prevalence of post COVID-19 condition or long COVID: a meta-analysis and systematic review. J Infect Dis. 2022;jiac136. [DOI] [PMC free article] [PubMed]
- 5.Saleki K, Banazadeh M, Saghazadeh A, Rezaei N. The involvement of the central nervous system in patients with COVID-19. Rev Neurosci. 2020;31:453–456. doi: 10.1515/revneuro-2020-0026. [DOI] [PubMed] [Google Scholar]
- 6.the Management Group of the EAN Dementia and Cognitive Disorders Scientific Panel, Toniolo S, Scarioni M, Di Lorenzo F, Hort J, Georges J, et al. Dementia and COVID-19, a bidirectional liaison: risk factors, biomarkers, and optimal health care. Bonanni L, editor. J Alzheimers Dis. 2021;82:883–98. [DOI] [PubMed]
- 7.Kas A, Soret M, Pyatigoskaya N, Habert M-O, Hesters A, Le Guennec L, et al. The cerebral network of COVID-19-related encephalopathy: a longitudinal voxel-based 18F-FDG-PET study. Eur J Nucl Med Mol Imaging. 2021;48:2543–2557. doi: 10.1007/s00259-020-05178-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Blazhenets G, Schroeter N, Bormann T, Thurow J, Wagner D, Frings L, et al. Slow but evident recovery from neocortical dysfunction and cognitive impairment in a series of chronic COVID-19 patients. J Nucl Med. 2021;62:910–915. doi: 10.2967/jnumed.121.262128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rogers JP, Chesney E, Oliver D, Pollak TA, McGuire P, Fusar-Poli P, et al. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry. 2020;7:611–627. doi: 10.1016/S2215-0366(20)30203-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vilensky JA, Foley P, Gilman S. Children and encephalitis lethargica: a historical review. Pediatr Neurol. 2007;37:79–84. doi: 10.1016/j.pediatrneurol.2007.04.012. [DOI] [PubMed] [Google Scholar]
- 11.de Melo GD, Lazarini F, Levallois S, Hautefort C, Michel V, Larrous F, et al. COVID-19–related anosmia is associated with viral persistence and inflammation in human olfactory epithelium and brain infection in hamsters. Sci Transl Med. 2021;13:eabf8396. [DOI] [PMC free article] [PubMed]
- 12.Schurink B, Roos E, Radonic T, Barbe E, Bouman CSC, de Boer HH, et al. Viral presence and immunopathology in patients with lethal COVID-19: a prospective autopsy cohort study. Lancet Microbe. 2020;1:e290–e299. doi: 10.1016/S2666-5247(20)30144-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Iadecola C, Anrather J, Kamel H. Effects of COVID-19 on the nervous system. Cell. 2020;183:16–27.e1. doi: 10.1016/j.cell.2020.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fond G, Masson M, Lançon C, Richieri R, Guedj E. The neuroinflammatory pathways of post-SARS-CoV-2 psychiatric disorders. L’Encéphale. 2021;47:399–400. doi: 10.1016/j.encep.2021.08.001. [DOI] [PubMed] [Google Scholar]
- 15.Guedj E, Lazarini F, Morbelli S, Ceccaldi M, Hautefort C, Kas A, et al. Long COVID and the brain network of Proust’s madeleine: targeting the olfactory pathway. Clin Microbiol Infect Off Publ Eur Soc Clin Microbiol Infect Dis. 2021;27:1196–1198. doi: 10.1016/j.cmi.2021.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Morbelli S, Chiola S, Donegani MI, Arnaldi D, Pardini M, Mancini R, et al. Metabolic correlates of olfactory dysfunction in COVID-19 and Parkinson’s disease (PD) do not overlap. Eur J Nucl Med Mol Imaging [Internet]. 2022 [cited 2022 Jan 19]; Available from: https://link.springer.com/10.1007/s00259-021-05666-9. [DOI] [PMC free article] [PubMed]
- 17.Guedj E, Million M, Dudouet P, Tissot-Dupont H, Bregeon F, Cammilleri S, et al. 18F-FDG brain PET hypometabolism in post-SARS-CoV-2 infection: substrate for persistent/delayed disorders? Eur J Nucl Med Mol Imaging. 2020. [DOI] [PMC free article] [PubMed]
- 18.Guedj E, Campion JY, Dudouet P, Kaphan E, Bregeon F, Tissot-Dupont H, et al. 18F-FDG brain PET hypometabolism in patients with long COVID. Eur J Nucl Med Mol Imaging. 2021;48(9):2823–2833. doi: 10.1007/s00259-021-05215-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sollini M, Morbelli S, Ciccarelli M, Cecconi M, Aghemo A, Morelli P, et al. Long COVID hallmarks on [18F]FDG-PET/CT: a case-control study. Eur J Nucl Med Mol Imaging. 2021;48:3187–3197. doi: 10.1007/s00259-021-05294-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dressing A, Bormann T, Blazhenets G, Schroeter N, Walter LI, Thurow J, et al. Neuropsychological profiles and cerebral glucose metabolism in neurocognitive Long COVID-syndrome. J Nucl Med. 2021;jnumed.121.262677. [DOI] [PMC free article] [PubMed]
- 21.Meyer PT, Hellwig S, Blazhenets G, Hosp JA. Molecular imaging findings on acute and long-term effects of COVID-19 on the brain: a systematic review. J Nucl Med. 2022;63(7):971–980. doi: 10.2967/jnumed.121.263085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Morand A, Campion J-Y, Lepine A, Bosdure E, Luciani L, Cammilleri S, et al. Similar patterns of [18F]-FDG brain PET hypometabolism in paediatric and adult patients with long COVID: a paediatric case series. Eur J Nucl Med Mol Imaging. 2022;49:913–920. doi: 10.1007/s00259-021-05528-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tran V-T, Porcher R, Pane I, Ravaud P. Course of post COVID-19 disease symptoms over time in the ComPaRe long COVID prospective e-cohort. Nat Commun. 2022;13:1812. doi: 10.1038/s41467-022-29513-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Douaud G, Lee S, Alfaro-Almagro F, Arthofer C, Wang C, McCarthy P, et al. SARS-CoV-2 is associated with changes in brain structure in UK Biobank. Nature. 2022;604:697–707. doi: 10.1038/s41586-022-04569-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Manca R, De Marco M, Ince PG, Venneri A. Heterogeneity in regional damage detected by neuroimaging and neuropathological studies in older adults with COVID-19: a cognitive-neuroscience systematic review to inform the long-term impact of the virus on neurocognitive trajectories. Front Aging Neurosci. 2021;13:646908. doi: 10.3389/fnagi.2021.646908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Maguire EA, Woollett K, Spiers HJ. London taxi drivers and bus drivers: a structural MRI and neuropsychological analysis. Hippocampus. 2006;16:1091–1101. doi: 10.1002/hipo.20233. [DOI] [PubMed] [Google Scholar]
- 27.Guedj E, Varrone A, Boellaard R, Albert NL, Barthel H, van Berckel B, et al. EANM procedure guidelines for brain PET imaging using [18F]FDG, version 3. Eur J Nucl Med Mol Imaging [Internet]. 2021 [cited 2021 Dec 10]; Available from: https://link.springer.com/10.1007/s00259-021-05603-w. [DOI] [PMC free article] [PubMed]
- 28.Verger A, Kas A, Dudouet P, Goehringer F, Salmon-Ceron D, Guedj E. Visual interpretation of brain hypometabolism related to neurological long COVID: a French multicentric experience. Eur J Nucl Med Mol Imaging. 2022. [DOI] [PMC free article] [PubMed]
- 29.Guedj E, Tastevin M, Verger A, Richieri R. Brain PET imaging in psychiatric disorders. Ref Module Biomed Sci [Internet]. Elsevier; 2021 [cited 2021 Dec 10]. p. B9780128229606000000. Available from: https://linkinghub.elsevier.com/retrieve/pii/B9780128229606000909.
- 30.Guedj E, Campion J, Horowitz T, Barthelemy F, Cammilleri S, Ceccaldi M. The impact of COVID-19 lockdown on brain metabolism. Hum Brain Mapp. 2022;43:593–597. doi: 10.1002/hbm.25673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Turpin S, Martineau P, Levasseur M-A, Lambert R. Modeling the effects of age and sex on normal pediatric brain metabolism using 18 F-FDG PET/CT. J Nucl Med. 2018;59:1118–1124. doi: 10.2967/jnumed.117.201889. [DOI] [PubMed] [Google Scholar]
- 32.Lu Y, Li X, Geng D, Mei N, Wu P-Y, Huang C-C, et al. Cerebral micro-structural changes in COVID-19 patients – an MRI-based 3-month follow-up study. EClinicalMedicine. 2020;25:100484. doi: 10.1016/j.eclinm.2020.100484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Verger A, Doyen M, Campion JY, Guedj E. The pons as reference region for intensity normalization in semi-quantitative analysis of brain 18FDG PET: application to metabolic changes related to ageing in conventional and digital control databases. EJNMMI Res. 2021;11:31. doi: 10.1186/s13550-021-00771-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Poloni TE, Medici V, Moretti M, Visonà SD, Cirrincione A, Carlos AF, et al. COVID‐19‐related neuropathology and microglial activation in elderly with and without dementia. Brain Pathol [Internet]. 2021 [cited 2022 Jun 28];31. Available from: https://onlinelibrary.wiley.com/doi/10.1111/bpa.12997. [DOI] [PMC free article] [PubMed]
- 35.Mairal E, Doyen M, Rivasseau-Jonveaux T, Malaplate C, Guedj E, Verger A. Clinical impact of digital and conventional PET control databases for semi-quantitative analysis of brain 18F-FDG digital PET scans. EJNMMI Res. 2020;10:144. doi: 10.1186/s13550-020-00733-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rolin S, Chakales A, Verduzco-Gutierrez M. Rehabilitation strategies for cognitive and neuropsychiatric manifestations of COVID-19. Curr Phys Med Rehabil Rep [Internet]. 2022 [cited 2022 Jun 28]; Available from: https://link.springer.com/10.1007/s40141-022-00352-9. [DOI] [PMC free article] [PubMed]
- 37.Jain P, Chaney AM, Carlson ML, Jackson IM, Rao A, James ML. Neuroinflammation PET imaging: current opinion and future directions. J Nucl Med Off Publ Soc Nucl Med. 2020;61:1107–1112. doi: 10.2967/jnumed.119.229443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen W-W, Zhang X, Huang W-J. Role of neuroinflammation in neurodegenerative diseases (Review) Mol Med Rep. 2016;13:3391–3396. doi: 10.3892/mmr.2016.4948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sun B, Tang N, Peluso MJ, Iyer NS, Torres L, Donatelli JL, et al. Characterization and biomarker analyses of post-COVID-19 complications and neurological manifestations. Cells. 2021;10:386. doi: 10.3390/cells10020386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Méndez-Guerrero A, Laespada-García MI, Gómez-Grande A, Ruiz-Ortiz M, Blanco-Palmero VA, Azcarate-Diaz FJ, et al. Acute hypokinetic-rigid syndrome following SARS-CoV-2 infection. Neurology. 2020;95:e2109–e2118. doi: 10.1212/WNL.0000000000010282. [DOI] [PubMed] [Google Scholar]
- 41.Morassi M, Palmerini F, Nici S, Magni E, Savelli G, Guerra UP, et al. SARS-CoV-2-related encephalitis with prominent parkinsonism: clinical and FDG-PET correlates in two patients. J Neurol. 2021;268:3980–3987. doi: 10.1007/s00415-021-10560-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cohen ME, Eichel R, Steiner-Birmanns B, Janah A, Ioshpa M, Bar-Shalom R, et al. A case of probable Parkinson’s disease after SARS-CoV-2 infection. Lancet Neurol. 2020;19:804–805. doi: 10.1016/S1474-4422(20)30305-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hosp JA, Dressing A, Blazhenets G, Bormann T, Rau A, Schwabenland M, et al. Cognitive impairment and altered cerebral glucose metabolism in the subacute stage of COVID-19. Brain. 2021;144:1263–1276. doi: 10.1093/brain/awab009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Guedj E, Morbelli S, Kaphan E, Campion J-Y, Dudouet P, Ceccaldi M, et al. From early limbic inflammation to long COVID sequelae. Brain. 2021;144:e65–e65. doi: 10.1093/brain/awab215. [DOI] [PubMed] [Google Scholar]
- 45.Backer V, Sjöbring U, Sonne J, Weiss A, Hostrup M, Johansen HK, et al. A randomized, double-blind, placebo-controlled phase 1 trial of inhaled and intranasal niclosamide: a broad spectrum antiviral candidate for treatment of COVID-19. Lancet Reg Health - Eur. 2021;4:100084. doi: 10.1016/j.lanepe.2021.100084. [DOI] [PMC free article] [PubMed] [Google Scholar]