Abstract
Malignant mixed mesodermal sarcomas (myxoid leiomyosarcomas – MLMS) are a rare form of uterine cancer developed from the smooth muscles of the uterus. It usually affects women in the postmenopausal period and has an aggressive character with an unfavorable evolution and prognosis. This paper presents a case where MLMS was postoperatively confirmed with the aid of the histopathological (HP) examination coupled with specific immunolabeling techniques. In addition, we reviewed modern literature to compare our results. Clinically, patients may present with a pelvic tumor, vaginal bleeding, or abdominal pressure. Imagistic investigations, such as pelvic ultrasonography (US), computed tomography (CT), magnetic resonance imaging (MRI), and positron emission tomography (PET)–CT may support the diagnosis. Nevertheless, solely the HP examination establishes it. Macroscopically, MLMS is soft and gelatinous, unlike the conventional rigid and spiral leiomyoma appearance. Furthermore, the infiltrative, irregular tumor margin is characteristic of MLMS. From a microscopic point of view, the following are present: tumor cell necrosis, nuclear pleomorphism, and variable mitotic activity. With classical Hematoxylin–Eosin (HE) staining, myometrium presents a leiomyomatous structure and multiple nodular formations with the aspect of malignant tumor proliferation, most likely mesenchymal. We used multiple special immunolabeling techniques. Thus, we observed the intense reactivity of the cells to the anti-vimentin antibody, which immunolabeled type III intermediate filament (IF) protein expressed in mesenchymal cells, thus demonstrating tumor mesenchymal affiliation. Smooth cell positivity for alpha-smooth muscle actin (α-SMA) demonstrates that the tumor is present in its whole myometrial structure. Tumor cells also underwent mutations involving the p53 tumor suppressor gene demonstrated by the number of tumoral cells in division immunolabeled with anti-Ki67 proliferation antibody. Tumor development was demonstrated by protein activation of cyclin-dependent kinase (CDK) and the presence of c-Kit-bound hematopoietic stem cells, immunolabeled with the anti-cluster of differentiation 117 (anti-CD117) antibodies. The anti-desmin antibody demonstrates, along with α-SMA, the involvement of myocytes in the tumoral process. The following microscopic characteristics laid the foundation for the diagnosis of MLMS: irregular myometrial invasion, rare mitosis on high-power fields (HPFs), cell pleomorphism, predominant myxoid component that gave a hypocellular appearance, the matrix rich in proteoglycans and glycosaminoglycans, especially hyaluronic acid.
Keywords: cell pleomorphism, myxoid component, hypocellular appearance, rare mitosis
⧉ Introduction
Uterine sarcomas account for approximately 3% of all uterine tumors and less than 1% of malignant tumors of the female genital tract. The most common types of uterine sarcomas are leiomyosarcomas (LMS), malignant mixed mesodermal sarcomas (myxoid leiomyosarcomas – MLMS), and endometrial stromal sarcomas [1].
Less than 90 cases of uterine MLMS have been reported in the English literature up to the present [2,3,4,5,6,7,8].
MLMS is a tumor variant of the smooth muscle, extremely rare, with an annual incidence of 0.64/100 000 women [9]. It is characterized by a myxoid appearance, reduced mitotic activity, and minimal or no atypia, despite its very own very high aggressive evolution [2, 10].
Aim
This study aims to present the clinical, imaging, morphopathological features and make a positive diagnosis of a rare case of uterine MLMS.
⧉ Clinical, imaging and risk factors involved in MLMS
Clinically, most patients are in the postmenopausal period and may present a pelvic tumor, vaginal bleeding, or abdominal pressure. Prior pelvic radiation therapy [11] and tamoxifen treatment in breast cancer patients [12,13] were incriminated as the main risk factors.
The diagnosis of MLMS may be suggested by certain investigations, such as ultrasonography (US), computed tomography (CT), magnetic resonance imaging (MRI), and positron emission tomography (PET)–CT, each with benefits and downsides. According to the literature, MRI can differentiate between leiomyomas and LMS. Multiple research attempts have been carried out to define the imaging characteristics for LMS to help guide the imaging diagnosis of MLMS. They analyzed the undefined margins, intense signal on T1- and T2-weighted images, intratumoral hemorrhage, central tumor necrosis, specific features of diffusion-weighted imaging (DWI) and examined tumor texture by histogram. Furthermore, attempts have been made to define the imaging characteristics of classical leiomyomas compared to LMS [14,15,16,17,18,19,20,21,22,23,24,25].
⧉ Positive and differential diagnosis of MLMS
The combined evaluation makes the diagnosis of uterine LMS of three factors: tumor cell necrosis (TCN), the number of cell divisions evaluated on 10 high-power fields (HPFs) and the presence of cellular atypia (cell pleomorphism). These criteria must be adapted according to the tumor subtype, myxoid and epithelioid tumors of the myometrium. MLMS is a very rare form [26].
The differential diagnosis of benign (leiomyoma) and malignant (LMS) myometrial tumors is based on the Stanford Criteria proposed by Bell et al. [27], which forms the basis of the current classification of the World Health Organization (WHO) for uterine smooth muscle tumors [27]. The presence of leiomyomas does not represent a cause of malignant transformation into LMS [28].
MLMS causes irregular myometrial invasion with or without lymphovascular invasion. Cell mitosis is rare, and cytology may be unclear due to pleomorphism. The minimum number of mitotic divisions (MDs) to diagnose MLMS varies between 0–4/10 HPFs [26].
⧉ Prognosis and treatment in MLMS
The rarity of this tumor subtype cannot support a definite prognosis, thus remaining inconclusive. Some studies have shown a poor prognosis of MLMS [27], while previous studies have suggested that MLMS is less aggressive than conventional uterine LMS [11,12].
The standard treatment for LMS is total abdominal hysterectomy with bilateral adnexectomy [29], with tissue referral to the Service of Pathological Anatomy for histopathological (HP) examination. Post-interventional overall survival rates vary between 17% and 65%, regardless of the stage of the disease [30,31,32,33]. Aggressive post-interventional adjuvant therapy should be performed with careful selection of patients as it determines significant side effects without a definite benefit in terms of survival rate [29, 34,35].
⧉ Morphopathological and immunohistochemical examinations of MLMS
Over time, several antibodies have been used for HP confirmation regarding the differential diagnosis between malignant and benign smooth muscle tumors [36,37,38,39,40]. However, the diagnostic value of immunohistochemical (IHC) reactivity has not been investigated explicitly for MLMS.
MLMS presents a myxoid matrix and proliferations myofibroblastic, endometrial stromal, and epithelial. The term “myxoid” refers to an extracellular matrix (ECM) rich in proteoglycans (PG) and glycosaminoglycans (GAG), especially hyaluronic acid (HA) [41]. The myxoid transformation appears as a pale blue material in the Hematoxylin–Eosin (HE) classical staining, surrounding the neoplastic transformed cells, individually dispersed or arranged in clusters or bundles. The myxoid ECM is usually prominent, providing a hypocellular appearance and a fleshy, soft feature on macroscopic examination. Certain phenomena can simulate the myxoid appearance, such as edema that causes the separation of cellular elements, with hypocellular appearance, which also occurs in leiomyomas. Mucinous carcinomas can also be associated with extracellular mucin accumulation, and when the epithelial component cannot be easily identified, there is a risk of confusion with a myxoid tumor. Histological staining with Periodic Acid Schiff–Hematoxylin (PAS–H) or Alcian Blue (AB) is applied to prevent these difficulties because it highlights the components of the myxoid ECM [42].
Smooth uterine muscle tumors with myxoid ECM have been classified as myxoid tumor variants, with many details unknown about myxoid leiomyoma or myxoid smooth muscle tumor of uncertain malignant potential (STUMP). Therefore, for clinical and diagnostic purposes, it is essential to differentiate between malignant and benign lesions [43].
MLMS is a malignant tumor of the smooth muscle, found mainly in postmenopausal women. Compared to conventional LMS, MLMS has a more aggressive character with an unfavorable evolution and prognosis [8, 44,45]. Atkins et al. [46] used a ≥30% cut-off in terms of the density of the myxoid ECM, and Burch & Tavassoli [3] suggested a ≥60% cut-off for the myxoid matrix. Recent studies have established a 50% value of the myxoid component to diagnose MLMS [34]. However, the HP diagnosis of MLMS is made when there is a myxoid extracellular component with a cut-off of 30–50% [43].
Macroscopically, MLMS is soft and gelatinous, unlike conventional leiomyoma, with a hard, spiral appearance. Furthermore, the infiltrative, irregular tumor margin is characteristic of MLMS. From a microscopic point of view, the following are present: TCN, nuclear pleomorphism, and variable mitotic activity. From an IHC point of view, MLMS showed positive staining for alpha-smooth muscle actin (α-SMA), desmin, vimentin [47]. The aggressive form is supported by the suppression of tumor suppressor genes, the activation of receptors involved in the cell cycle, and increased tumor proliferation [48,49]. Also, intratumoral vascularization and the inflammatory process support a favorable environment for tumor development [50].
⧉ Case presentation
A 68-year-old patient, with a medical history of two vaginal births and three abortions on request, with type II diabetes under Metformin treatment presented in the Emergency Room for pelvic–abdominal pain and vaginal bleeding. After a complete clinical examination, biochemical and imagistic examinations were carried out. The patient underwent hysterectomy and bilateral adnexectomy surgery for its condition. Tissue fragments were sent for extemporaneous examination, and the excised internal genitalia was considered for definitive HP examination.
The tissue fragments were fixed in 10% neutral buffered formalin solution and sent to the Department of Pathological Anatomy. They were processed according to the pre-established paraffin inclusion technique. All blocks were sectioned using the Microm HM325 rotary microtome, and 5-μm thick sections were applied to single slides and slides treated with poly-L-lysine. HE, Masson’s trichrome (MT), PAS–H, and AB classical histological staining was used, as well as specific IHC procedures.
Before being stained, the slides were deparaffined with xylene in three baths (15 minutes each), rehydrated with successive alcohol baths with decreasing concentrations of 100%, 90%, 70% (3–5 minutes each), and then with distilled water (dH2O) (three baths for 5 minutes each). After hydration, the classical HE, MT, and PAS–H stainings were applied according to the predetermined techniques and the special IHC stainings. These involve antigenic exposure, performed with Ethylenediaminetetraacetic acid (EDTA) pH 9 or with Citrate pH 6, tissue endogenous peroxidase deactivation with the aid of 2% oxygenated water (H2O2) (30 minutes), introduction of slides into 3% powdered milk solution followed by application of the primary antibody (Table 1) for at least 18 hours at 4°C. The next day, after being left at room temperature for 30 minutes, the slides were washed in phosphate-buffered saline (PBS) to remove the primary antibody. The specific secondary antibody [mouse/rabbit immunoglobulin G (IgG) antibody, VC002-025, R&D Systems, VisUCyte Horseradish Peroxidase (HRP) Polymer] was then applied for one hour. The secondary antibody was subsequently removed using PBS, and the slides were developed with the aid of 3,3’-Diaminobenzidine (DAB) (Dako), and the nuclei were labeled with a Hematoxylin solution. Finally, the obtained slides were dehydrated with increasing 70%, 90%, and 100% alcohol concentrations (5 minutes each), clarified in xylene (3×15 minutes/bath). In the end, the slides were mounted with the help of a Canada balm conditioner. The HP preparations were made and scanned in the Research Center for Microscopic Morphology and Immunology, University of Medicine and Pharmacy of Craiova, Romania.
Table 1.
Immunohistochemical panel of antibodies
|
Antibody |
Manufacturer |
Clone |
Antigenic exposure |
Secondary antibody |
Dilution |
Labeling |
|
Anti-CK7 |
Dako |
OV-TL 12/30 |
Citrate, pH 6 |
Monoclonal mouse anti-human CK7 |
1:50 |
Glandular epithelia |
|
Anti-pan-CK AE1/AE3 |
Dako |
AE1/AE3 |
Citrate, pH 6 |
Monoclonal mouse anti-human CK |
1:50 |
Epithelial tissues |
|
Anti-CD34 |
Dako |
QBEnd/10 |
Citrate, pH 6 |
Monoclonal mouse anti-human CD34 Class II |
1:50 |
Neoformed blood vessels |
|
Anti-α-SMA |
Dako |
1A4 |
Citrate, pH 6 |
Monoclonal mouse anti-human SMA |
1:100 |
α-SMA |
|
Anti-VIM |
Dako |
V9 |
Citrate, pH 6 |
Monoclonal mouse anti-VIM |
1:50 |
Mesenchymal cells |
|
Anti-ER |
Dako |
1D5 |
EDTA, pH 9 |
Monoclonal mouse anti-human ERα |
1:50 |
ERα |
|
Anti-PR |
Dako |
PgR 636 |
EDTA, pH 9 |
Monoclonal mouse anti-human PR |
1:50 |
PR |
|
Anti-Ki67 |
Dako |
MIB-1 |
EDTA, pH 9 |
Monoclonal mouse anti-human Ki67 |
1:50 |
Cells in division in the G1, S, G2, and M phase |
|
Anti-p16 |
Invitrogen |
1D7D2 |
Citrate, pH 6 |
Monoclonal mouse anti-p16INK4a |
1:50 |
Human protein CDKN2A – present in HPV infection |
|
Anti-p53 |
Dako |
DO-7 |
EDTA, pH 9 |
Monoclonal mouse anti-human p53 protein |
1:50 |
Nuclear marker |
|
Anti-HMB45 |
Dako |
HMB45 |
Citrate, pH 6 |
Monoclonal mouse anti-human melanosome |
1:50 |
Intracytoplasmic antigen expressed by most melanomas (melanotic and amelanotic) |
|
Anti-CD68 |
Dako |
KP1 |
Citrate, pH 6 |
Monoclonal mouse anti-human CD68 |
1:100 |
Macrophages |
|
Anti-tryptase |
Dako |
AA1 |
Citrate, pH 6 |
Monoclonal mouse anti-human mast cell tryptase |
1:500 |
Mast cells |
|
Anti-CD3 |
Dako |
Polyclonal |
Citrate, pH 6 |
Polyclonal rabbit anti-human CD3 |
1:50 |
T-lymphocytes |
|
Anti-CD20 |
Dako |
L26 |
Citrate, pH 6 |
Monoclonal mouse anti-human CD20cy |
1:50 |
B-lymphocytes |
|
Anti-CD117 |
Dako |
– |
EDTA, pH 9 |
Polyclonal rabbit anti-human CD117 (c-Kit) |
1:400 |
Stem cell factor receptor |
|
Anti-desmin |
Dako |
D33 |
Citrate, pH 6 |
Monoclonal mouse anti-human desmin |
1:50 |
Muscle-specific, type III intermediate filament |
|
Anti-cyclin D1 |
Dako |
DCS-6 |
Citrate, pH 6 |
Monoclonal mouse anti-human cyclin D1 |
1:50 |
Involved in regulating cell cycle progression |
α-SMA: Alpha-smooth muscle actin; CD: Cluster of differentiation; CDKN2A: Cyclin-dependent kinase inhibitor 2A; CK: Cytokeratin; EDTA: Ethylenediaminetetraacetic acid; ER: Estrogen receptor; HMB45: Human melanoma black 45; HPV: Human papillomavirus; PR: Progesterone receptor; VIM: Vimentin
Clinical examination
At the time of admission, the patient had an acceptable general condition, accusing mild pain in the lower abdominal and fever. A stiff, irregular, immobile tumor was noted occupying the entire hypogastrium and lateral flanks, with a maximum diameter of approximately 15 cm. No particular modifications were observed during the speculum examination. In addition, at the mammary examination, a tumor formation of approximately 2–3 cm was noted in the inferior–internal quadrant of the left breast.
At the mammography exam performed in two planes, heterogeneous adipose breasts were described, without foci of microcalcification, without spiky opacifications. Instead, bilateral calcifications with benign characters were noted. In addition, an oval opacity was detected at the left breast evaluation, with 29/17 mm dimensions, irregular contour, heterogenous intensity, and included calcifications. Breast Imaging Reporting and Data System (BI-RADS) score was 4. Due to these suspicious characteristics, an US-guided biopsy puncture was performed, and biopsy fragments were sent to the Laboratory of Pathology for HP examination. From a microscopic point of view, breast parenchyma with dense collagen stroma was observed, which includes ducts with areas of simple epithelial hyperplasia and duct–acinar structures of various shapes and sizes.
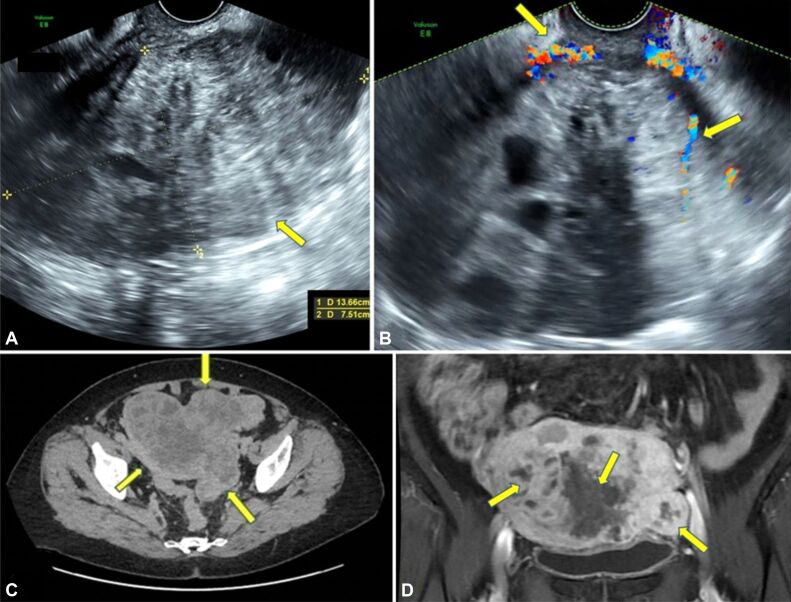
US examination revealed a large multilobular pelvic–abdominal mass, apparently developed from the uterus, inhomogeneous, with a predominant solid structure, high vascularized on the edges, measuring approximately 13 cm (Figure 1, A and B). No fluid was detected in the abdomen or pelvis. The ovaries could not be detected.
Figure 1.
Imagistic aspects of the tumor formation, highlighted by yellow arrows: (A) Ultrasound aspect of the pelvic–abdominal tumor formation – voluminous, multilobular, heterogeneous, with solid structure, approximately 13 cm, probably of uterine affiliation; (B) Echo-Doppler examination shows poor vascularization of the tumor, especially in the periphery; (C) CT aspect of the tumor formation, voluminous, polylobulated, relatively well delimited, with heterogeneous structure and iodophilia, extensive intralesional necrosis areas and multiple iodophilic septa, with dimensions of approximately 138/134/72 mm, belonging to the uterine body, with compressive effect on the adjacent intestinal loops, without infiltrating them; (D) MRI appearance shows polylobed tumor block, heterogeneous, with tissue and cystic components, with an ancillary origin, approximately 108/140/94 mm that imprints the uterus and moves it to the right side, the tumor block includes both adnexa, present peritoneal fluid. The uterus appears compressed by the tumor mass, possibly adherent to it, measuring 77/16 mm, with several focal fibrotic remodeling at the myometrial level, including 16 mm uterine fundus fibromatous nodules. CT: Computed tomography; MRI: Magnetic resonance imaging
The CT examination revealed a 12/20/17 mm tumoral formation, located in the superior–internal quadrant of the left mammary gland, non-iodophilic, well delimited, with internal microcalcifications. Also, a voluminous 138/134/72 mm, irregular-polylobulated pelvic tumor was detected, relatively well delimited, with heterogeneous structure and iodophilia, extensive intralesional necrosis areas, and multiple iodophilic septa, with dimensions of approximately, belonging to the level uterine body. The described formation had a mass effect on adjacent intestinal loops but retained cleavage space with them (Figure 1C).
MRI examination completed the imaging characterization of the tumors. The left breast formation was described as well delimited, showing dystrophic calcifications up to 6 mm, and no associated axillary or internal breast adenopathies. The appearance was suggestive of a fibroadenoma. A pelvic heterogeneous irregular-polylobed tumor block was detected, with solid and cystic components, ancillary origin, measuring 108/140/94 mm. It formed a tumor block that included both adnexa and intestinal loops. The uterus appeared compressed by the tumoral mass, possibly adherent to it, measuring 77/16 mm, with focal fibrotic myometrial changes and small fundus fibromatous nodules of 16 mm. Intrapelvic fluid was in a minimal–moderate amount located between the intestinal loops and in the Douglas sac. No signs of bladder or rectal invasion were detected (Figure 1D).
Immunological tests had normal values (Table 2), cancer antigen 125 (CA125) 9.1 U/mL (normal values: 0–35 U/mL), and serological tests: hemoglobin 12.98 g/dL, glycemia 124 g/dL.
Table 2.
Immunological tests values
|
Marker |
Result value |
Normal reference value |
|
CA15-3 [U/mL] |
23.58 |
0–25 |
|
CA19-9 [U/mL] |
16.21 |
0–39 |
|
CEA [ng/mL] |
1.92 |
0.0–4.7 |
CA: Cancer antigen; CEA: Carcinoembryonic antigen
The patient’s cardiac examination highlighted first-grade hypertension with high cardiovascular risk. Therefore, antihypertensive and hypocholesterolemic medications were recommended.
Oncology consultation recommended laparoscopy or exploratory laparotomy, tumor biopsy, and reassessment after the HP result for the therapeutic decision.
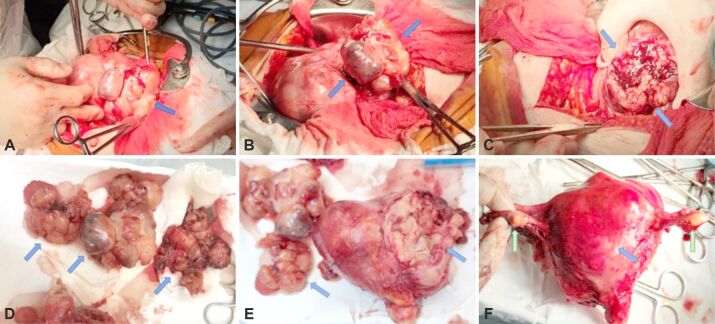
Laparotomy was performed and found a large, fatty, irregular, friable, easily detachable uterine tumor (Figure 2A,2B,2C). Three tumor fragments were sectioned and sent to the extemporaneous HP examination (Figure 2D). Total hysterectomy with bilateral adnexectomy was performed, and internal genitalia was sent for HP examination (Figure 2, E and F). It revealed numerous gray, hemorrhagic tumor fragments, varying in size from 2 cm to 8/7/5 cm. The uterus had dimensions of 11/8/13 cm. The cervix showed Naboth cysts, low polypoidal endocrine proliferation, and fibrous consistency. The uterine body macroscopic evaluation revealed a heterogeneous appearance, with cystic mucoid component and nodular tumor formations varying from 2 cm to 8/7/13 cm, whiteish, translucent, and yellow areas. Nodular formations were present up to the level of the isthmic region. Endocavitary, a polypoid tumor formation was identified, with a length of 3 cm and a thickness of 0.5 cm with a microcystic appearance. Both ovaries were atrophic, sclerotic, with a diameter of 1 cm. Both salpinges had a typical 5 cm and 6 cm length, with a small, uniform caliber.
Figure 2.
Intraoperative and postoperative macroscopic aspects of the uterine tumor, highlighted by the blue arrows: (A and B) Multilobed tumor mass, greasy, friable, easily detachable; (C) The remaining tumor after partial tumor excision, with areas of necrosis and intratumoral hemorrhages (red areas); (D) Tumor fragments detached from the tumor and sent to the extemporaneous histopathological examination, with whitish appearance, elastic consistency, lumpy, with small hemorrhagic foci; (E) Anterior aspect of the specimen, which shows on the left side the detachment area of the tumor biopsy; (F) Posterior aspect of the operatory piece, with bilateral adnexectomy – small, atrophic ovaries are observed, highlighted with green arrows
At the macroscopic extemporaneous examination, a tumor formation of 7/5/2 cm was noted, with a whitish interior aspect after being sectioned, elastic consistency, lumpy, with small hemorrhagic foci, and other two fragments with the same appearance, measuring 5/3.5/1 cm and 3/2.5/1.5 cm. In addition, tumoral proliferation was observed microscopically, with marked cell and nuclear pleomorphism, solid and fasciculate pattern, moderate mitotic activity, diagnosis of poorly differentiated malignant tumor with sarcomatoid appearance.
Postoperatively, the patient’s evolution was favorable under antibiotic, anticoagulant, analgesic, and laxative treatment. When the patient was discharged, she was advised and directed to the regional medical oncology center to undergo further oncological investigation until the final HP result was obtained.
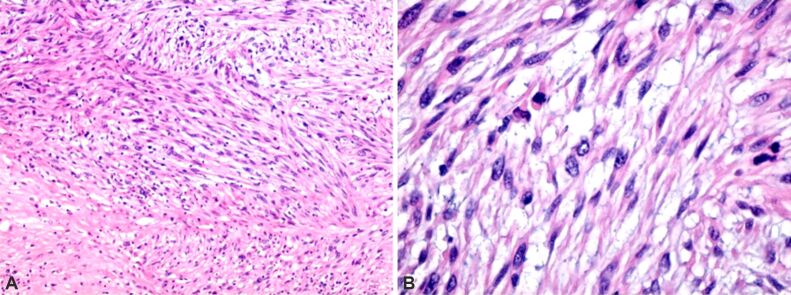
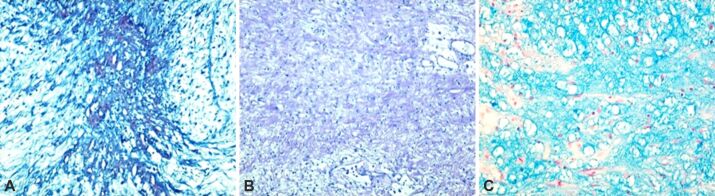
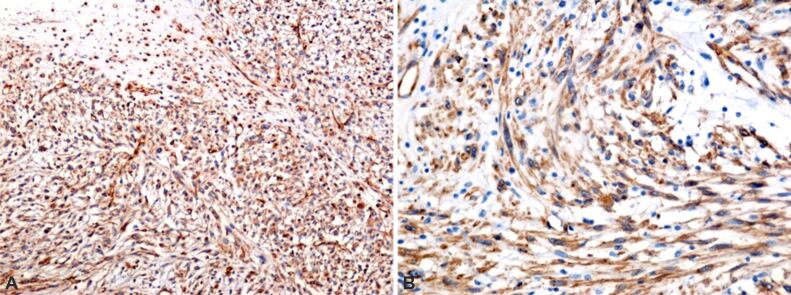
On classical HE staining, myometrium showed a leiomyomatous structure, multiple nodular formations with malignant tumor proliferation, most likely mesenchymal, and areas of hyalinization (Figure 3, A and B). Also, a hyperplastic polyp with regressive changes was identified. These aspects required IHC examination to identify the tumor subtype. Tissue fragments were specifically immunohistochemically labeled and established the tumor subtype (Table 3). Using the classical MT staining, we noticed the presence of the collagenic component in the tumor structure, marked in blue, but also the presence of intratumoral necrosis (stained in red-brown (Figure 4, A and B) and with AB (stained in deep blue) (Figure 4C), highlighted in more than the 50% cut-off of the tumoral mass.
Figure 3.
(A and B) Histopathological section through uterine tumor mass. Tumor proliferation with marked cell and nuclear pleomorphism, with a solid and fasciculate pattern, moderate mitotic activity, suggesting a malignant tumor, most likely mesenchymal and poorly differentiated. Hematoxylin–Eosin (HE) staining: (A) ×100; (B) ×200
Table 3.
IHC reaction intensity of the tumor
|
Antibody |
Reactivity |
|
Anti-ER |
--- |
|
Anti-PR |
--- |
|
Anti-p16 |
+++ |
|
Anti-VIM |
+++ |
|
Anti-CK7 |
--- |
|
Anti-HMB45 |
--- |
|
Anti-p53 |
+ |
|
Anti-pan-CK AE1/AE3 |
--- |
|
Anti-CD117 |
+ |
|
Anti-desmin |
+ |
|
Anti-CD34 |
+ |
|
Anti-cyclin D1 |
++ |
|
Anti-CD68 |
++ |
|
Anti-CD20 |
+ |
|
Anti-CD3 |
+ |
|
Anti-tryptase |
+ |
|
Anti-α-SMA |
+++ |
|
Anti-Ki67 |
+++ |
---: Negative reaction; +: Weakly positive reaction; ++: Moderately positive reaction; +++: Intense positive reaction; α-SMA: Alpha-smooth muscle actin; CD: Cluster of differentiation; CK: Cytokeratin; IHC: Immunohistochemical; ER: Estrogen receptor; HMB45: Human melanoma black 45; PR: Progesterone receptor; VIM: Vimentin
Figure 4.
Microscopic aspects of the tumor: (A) The density of collagen fibers in the tumor structure and asymmetrical disposition is observed, and an area of intratumoral necrosis; (B) The myxoid tumor structure is observed, highlighted by PAS–H; (C) The myxoid tumor structure is observed, highlighted by the AB staining. MT staining: (A) ×100. PAS–H staining: (B) ×100. AB staining: (C) ×200. AB: Alcian Blue; MT: Masson’s trichrome; PAS–H: Periodic Acid Schiff–Hematoxylin
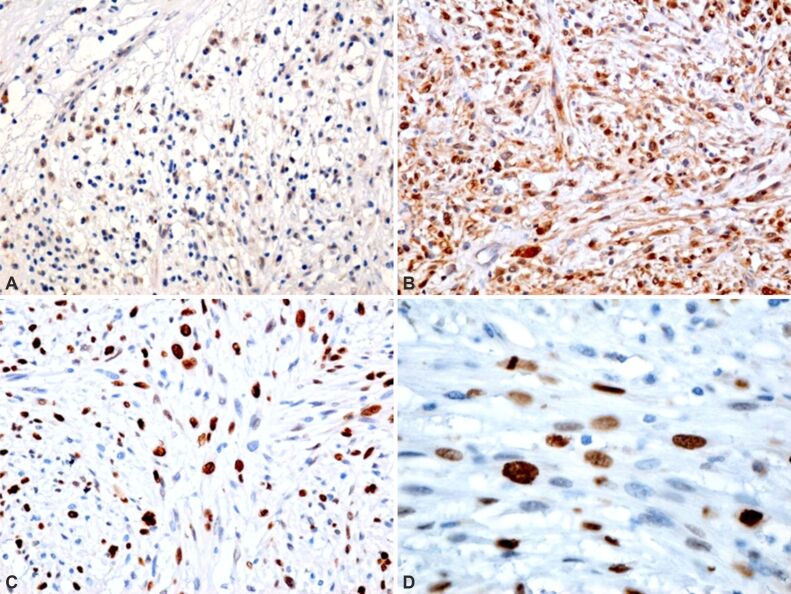
With the help of special IHC staining techniques, we observed the intense reactivity of the cells to the anti-vimentin antibody, which marks type III intermediate filament (IF) protein, expressed in mesenchymal cells, thus demonstrating tumor mesenchymal affiliation (Figure 5A). Smooth cell positivity for α-SMA demonstrates that the tumor is present in its entire myometrial structure (Figure 5B). Tumor cells also underwent mutations involving the p53 tumor suppressor gene (Figure 6A), inactivating the cyclin-dependent kinase inhibitor 2A (CDKN2A) tumor suppressor gene, thus increasing tumor aggression (Figure 6B). This is also demonstrated by the number division of tumor cells in the division, with an average of 2.6 cells/×400 (±0.96 cells/×400), immunolabeled with Ki67 proliferation marker (Figure 6, C and D).
Figure 5.
Microscopic aspects of the tumor: (A) There is an intense reactivity of the anti-VIM antibody; (B) Intense reactivity of myocytes to α-SMA is observed. Immunomarking with anti-VIM antibody: (A) ×100. Immunomarking with anti-α-SMA antibody: (B) ×200. α-SMA: Alpha-smooth muscle actin; VIM: Vimentin
Figure 6.
Microscopic aspects of the tumor: (A) Cells that have undergone mutations in the p53 tumor suppressor gene, immunolabeled in brown; (B) Heavily reactive tumor cells are observed at p16, suggesting a high degree of inactivation of the tumor suppressor gene CDKN2A and an increase in the degree of tumor aggressiveness; (C and D) The density of cells in cell division, stained in brown, is observed. Immunomarking with anti-p53 antibody: (A) ×100. Immunomarking with anti-p16 antibody: (B) ×100. Immunomarking with anti-Ki67 antibody: (C) ×200; (D) ×400. CDKN2A: Cyclin-dependent kinase inhibitor 2A
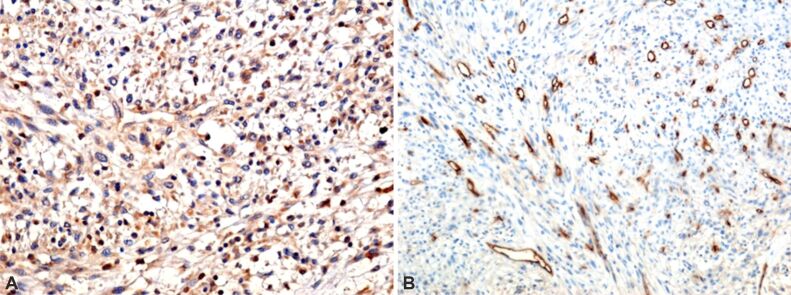
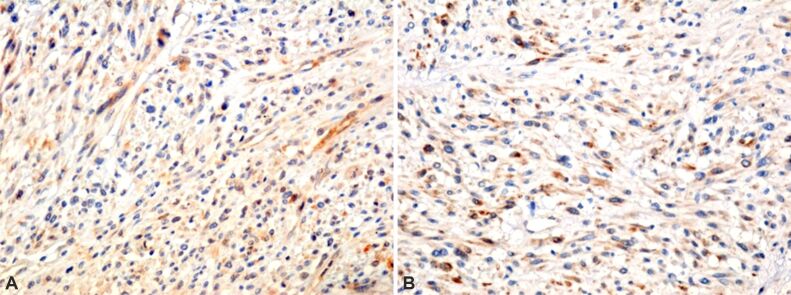
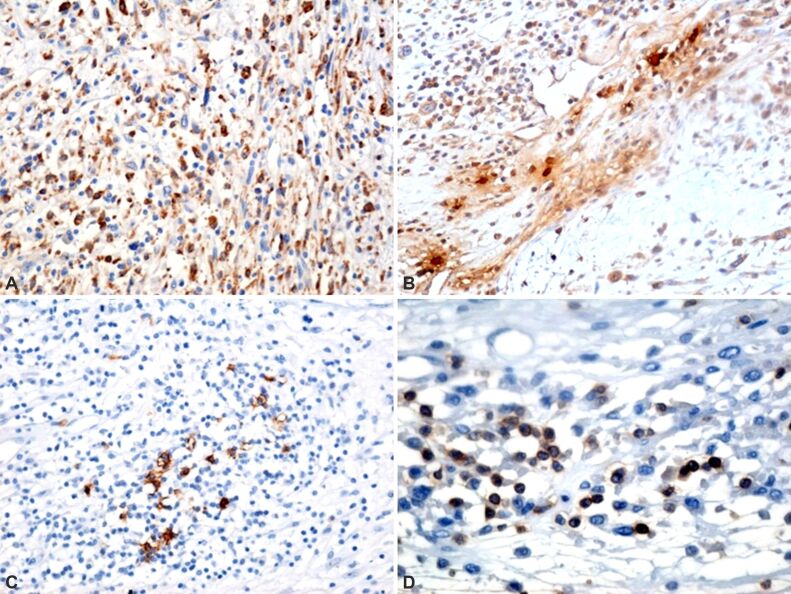
This tumor proliferation is also supported by protein activation of cyclin-dependent kinase (CDK) enzymes required for accelerated cell cycle, demonstrated by cell immunolabeling with the anti-cyclin D1 antibody (Figure 7A). Tumor aggression is inversely correlated with poor vascularization, especially at the edges. The capillary endothelium was immunolabeled with the anti-cluster of differentiation 34 (anti-CD34) antibody (Figure 7B). The tumor substrate is also maintained by the presence of c-Kit-bound hematopoietic stem cells, immunolabeled with the anti-cluster of differentiation 117 (anti-CD117) antibody – tyrosine kinase growth factor receptor, which formed a dimer that regulates the intrinsic activity of tyrosine kinase, involved in tumor development (Figure 8A). Anti-desmin antibody, the fibrous protein that enters the cytoskeleton of muscle cells, demonstrates, along with α-SMA, the involvement of myocytes in the tumor process (Figure 8B). Intratumorally, the cells of the inflammatory system were also highlighted: increased number of macrophages (Figure 9A), mast cells (Figure 9B), B-lymphocytes (Figure 9C), and T-lymphocytes (Figure 9D).
Figure 7.
Microscopic aspects of the tumor: (A) The cellular reactivity that demonstrates the process of protein activation of CDK enzymes required by the cell is observed; (B) The poor intratumoral vascular density is observed by staining (in brown) the capillary endothelium. Immunomarking with anti-cyclin D1 antibody: (A) ×100. Immunomarking with anti-CD34 antibody: (B) ×100. CD34: Cluster of differentiation 34; CDK: Cyclin-dependent kinase
Figure 8.
Microscopic aspects of the tumor: (A) Moderate reactivity of cells with the presence of the CD117 transmembrane protein; (B) Cellular reactivity for desmin is observed, a fibrous protein that enters the cytoskeleton of muscle cells, demonstrating the involvement of myocytes in the tumor process. Immunomarking with anti-CD117 antibody: (A) ×100. Immunomarking with anti-desmin antibody: (B) ×100. CD117: Cluster of differentiation 117
Figure 9.
Microscopic inflammatory aspects involved in tumor development: (A) The increased number of macrophages present in the tumor mass is observed; (B) The mast cells involved in the tumor mass are identified; (C) B-lymphocytes in low numbers, present in the tumor mass, as an adaptive humoral response; (D) T-lymphocytes involved in intratumoral cellular immunity have been identified. Immunomarking with anti-CD68 antibody: (A) ×100. Immunomarking with anti-tryptase antibody: (B) ×100. Immunomarking with anti-CD20 antibody: (C) ×100. Immunomarking with anti-CD3 antibody: (D) ×200. CD: Cluster of differentiation
⧉ Discussions
According to several studies, the incidence of LMS in symptomatic patients undergoing hysterectomy was 0.37%. Corscaden & Singh [51] reported 32 cases of LMS out of a total of 15 000 leiomyomas diagnosed, representing 0.21%, of which 19 died, reaching a mortality rate of 59% [51]. According to other studies, out of 13 000 operated fibroids, 38 were LMS representing 0.29%, and 18 of them died, reaching a mortality rate of 47% [52]. The preoperative diagnosis of uterine LMS is challenging to establish. Vardi & Tovell [31] diagnosed uterine LMS by biopsy uterine curettage in five of the 24 (20.8%) patients and Gallup & Corday [53] in two of eight (25%) patients. Leibsohn et al. [54] established the diagnosis preoperatively by endometrial biopsy in three of eight (37.8%) patients. Two of them showed large, polypoid tumors attached to the uterine wall by a narrow pedicle and an intracavitary extension; the LMS location in the third case was submucosal [54]. In the case we presented, the tumor formation presented imagistic characteristics similar to those described in the literature.
Surgical treatment, when possible, represents the first therapeutic step by performing a total abdominal hysterectomy with or without bilateral adnexectomy. Tumor excision or reduction can also be performed supplemented with radiotherapy/chemotherapy depending on the biological conditions of the patients. Furthermore, chemotherapy can be added only in relapse cases for post-interventional therapeutic purposes [2, 10, 55,56].
King et al. [2] argued that MLMS might not respond to chemotherapy or radiotherapy due to low mitotic index and high amount of intercellular myxoid tissue. Therefore, the effect of adjuvant therapy in preventing recurrence is unclear [57,58].
MLMS is a very aggressive tumor that cannot be controlled locally or systemically despite a proper therapeutic approach. The frequent recurrences represent a major issue. These aspects are shown in various studies, where recurrence occurred from three months to 10 years after treatment [2, 10, 55,56]. MLMS has an unfavorable prognosis, with only 26 cases reported in the literature by King et al. until 1982 [2, 10, 55,56]. In our case, the recurrence did not occur in the first six months post-operatively, during the time this study was published.
Studies have shown that MLMS frequently metastasizes to the lungs, liver, and brain [2].
Patients reported with MLMS had pelvic tumor mass, pelvic pain, and abnormal vaginal bleeding [2, 10, 55,56], similar to the case described by us.
Some authors have analyzed serum levels of CA125 in MLMS, but the significance of this marker has not been sufficiently established [10, 59,60]. Studies on the usefulness of serum CA125 in differentiating MLMS and LMS are not yet available. The serum level of CA125 in our patient was not high at the time of diagnosis (9.1 U/mL), which may have contributed to the lack of recurrence in the first six months postoperatively. This parameter and IHC investigation of the mitotic index lay the groundwork for future diagnostic and therapeutic advances in MLMS.
Most published studies agree that the number of mitoses per 10 HPFs serves as a valuable prognostic and diagnostic factor in LMS. For example, Montague et al. [52] noted that when they were between 2–5 mitoses per 10 HPFs present, 100% of patients survived one year, and 77% survived five years. However, in patients who had 5–10 mitoses per 10 HPFs, the survival rate decreased to 35% at five years, and no patients survived five years when there were more than 10 mitoses per 10 HPFs [52].
About the mitotic index in MLMS, studies have shown that at least 1/10 HPFs or 2/10 HPFs MDs must be present [2,3, 61], but rarely can a high mitotic index be >57/50 HPFs, as reported by Burch & Tavassoli [3]. Moreover, the vascular invasion was present in over 30% of MLMS cases [3], and tumor margins were invasive [8]. All these aspects underline the infiltrative and dissemination potential of the tumor, thus increasing the malignant potential of MLMS. Regarding these findings [2,3, 8, 61], we noticed tumoral infiltration of the entire uterine wall and past the uterine muscle with poor intratumoral vascularization and a high number of blood vessels at the periphery, highlighted by anti-CD34 antibody immunolabeling. The echo-Doppler findings (poor vascularization inside the tumor and high vascularization in the periphery) did not suggest a malignant tumor, but the HP examination confirmed the malignant etiology.
MLMS may be devoid of mitotic figures, causing diagnostic difficulties due to the inability to assess tumoral margin and the occurrence of possible tumor invasion. This aspect is usually found in low-grade tumors. Furthermore, there may be benign leiomyomatous tumors found in the structure of low-grade malignant tumors, as reported in a case of association between leiomyoma and fusiform cell-transformed leiomyoma by an English study [59]. However, this transformation is rare and statistically unlikely, as only 1/800 uterine smooth muscle tumors are malignant [54].
A specific feature of MLMS is the gelatinous consistency and well-circumscribed tumor margins. From a microscopic point of view, it has spindle-shaped cells with low or low atypia, a low mitotic index at 10 HPFs, often <5, and a large amount of intercellular myxoid tissue. Increased numerical density of inflammatory cells is a significant indicator of malignancy [2, 57].
It is challenging to make the differential diagnosis between typical LMS and MLMS because the main criteria of malignancy (nuclear atypia, prominent mitotic figures, and hypercellularity) are rare. Thus, most cases are diagnosed retrospectively according to evolution [10, 59].
The classical HP examination and immunohistochemistry allowed us to achieve and confirm the phenogenic type and the malignancy indicator. Similarly, in the studies performed by Mittal et al. [59], Ki67 contributed to the indication of tumor malignancy (mitotic index).
The detailed immunoprofile of MLMS has rarely been addressed, which has led us to apply a wide range of immunolabeling. In addition to the numerically determined mitotic index and the presence of increased vascular density, we wanted to show the involvement of other mechanisms in the emergence and development of MLMS. Thus, according to other studies, we used the expression of the p53 tumor protein previously examined by five authors, who did not provide additional information regarding the diagnosis of MLMS [48, 59, 62,63], being weakly positive in the tumor formation analyzed in our paper.
Toki et al. [62] have shown that estrogen receptor (ER) and progesterone receptor (PR) may be present in young patients with MLMS, suggesting the possibility of hormone therapy with gonadotropin-releasing hormone (GnRH) and progesterone agonists [62]. However, other authors have shown that ER and PR are no longer present in post-menopause, thus changing the therapeutic methods [55]. For the diagnosis, management, and prognosis of MLMS, it is recommended to perform immunohistochemistry, thus achieving the differential diagnosis between typical LMS and MLMS [59]. In our case, the patient did not show ER and PR present at the tumor level, being at menopause, similar to previous studies, which prevented the administration of hormone therapy.
IHC studies performed with the help of anti-desmin and anti-α-SMA antibodies highlight the belonging of this tumor with a starting point in the myometrial smooth muscles, making the differential diagnosis with other tumors with myxoid modification. Desmin is a type III IF protein specific to the heart, skeletal, and smooth muscles, which integrates the cellular elements and stabilizes its architecture [63,64,65]. α-SMA emphasizes the muscle phenotype of the tumor by immunolabeling muscle-specific actin myofilaments. In the present case, the immune reactivity positive for desmin and intensely positive for α-SMA dictates the muscular phenotypic diagnosis.
Immunostaining for the CD117/c-Kit protein was performed to make a differential diagnosis with a possible gastrointestinal stromal tumor (GIST) [66]. c-Kit is a cytokine receptor expressed on the cell surface of stem cells. It may also be present in other cells, such as mast cells, melanocytes, and interstitial cells of Cajal in the digestive tract but also tumor-transformed cells. In our study, we used this marker to differentiate the uterine tumor from GIST. Because of a weak positive reaction at the cellular level, probably due to the mast cells, the positive immunolabeling of these cells was achieved with the help of anti-tryptase antibody, specific for mast cells. On the other hand, in the case of GIST, the immunolabeling for desmin is negative and positive for MLMS, as in the present case [67].
To date, little is known about the significance of MLMS-associated macrophages. These represent cells that come from the blood cells that have crossed the capillary wall and passed into the interstitial space, participating in various physiological and pathological processes. For example, they play a role in inflammation, immune response, and local tissue remodeling. In addition, several studies show that macrophages play an essential role in the process of tumor development and progression [68,69,70,71,72].
Tumor-associated macrophages interact with tumor-transformed cells by releasing various cell growth factors and cytokines, which can initiate and cause cancer progression, providing an advantage in tumor survival and increasing angiogenesis [69,70, 73]. They also release a series of proteases that contribute to the remodeling of the ECM by the breakdown of different layers of connective tissue, thus allowing increased cell mobility in the tumor structure, penetrating different compartments of tissue. According to previous studies, the presence of a numerically increased macrophage population is associated with an unfavorable prognosis [74,75,76,77,78,79]. We found a moderate number of macrophage and mast cells immunolabeled with anti-CD68/anti-tryptase antibodies and a low number of B- and T-lymphocyte cells immunolabeled with anti-CD20/anti-CD3 antibodies. Lymphocytes contribute both to an optimal pro-inflammatory environment for tumor development through an immune response (B-cells) and a cellular response (T-cells), accelerating cell development through local immune processes [80,81,82].
Cytokeratin (CK) 7 is a type II keratin, positive in several glandular or transitional epithelia in the structure of organs or blood vessels [83]. Our study used it to make a differential diagnosis with other tumors with an epithelial origin, such as endometrial cancer, which would be reacted positively to CK7.
Pan-CK AE1/AE3 is a cocktail of CK that commonly marks the epithelial tissues of the body. It is often used to immunolabel malignantly transformed epithelia from several organs. Therefore, it is helpful for the differential diagnosis with non-malignant tumors [84]. Our study used it to make the differential diagnosis of MLMS, which was negatively labeled in response to pan-CK AE1/AE3, with a malignantly transformed epithelial tumor, where the immunoreaction was to be positive.
To make the differential diagnosis with a melanocyte-starting tumor, we used the anti-human melanoma black (anti-HMB45) antibody, which would have positively marked the melanocytes. This IHC marker reacts positively against fetal melanocytes and junctional nevus cells [85]. However, it is non-reactive with almost all non-melanoma malignancies, except for a few rare tumors, such as clear cell sarcoma or pigmented schwannoma, which show signs of melanogenesis [86]. In MLMS, the immunoreaction to the anti-HMB45 antibody was negative.
Cyclin D1 is a protein expressed in all adult human tissues, lacking in cells from the lymphoid and myeloid lines of bone marrow stem cells [87,88]. This protein functions as a regulator of CDKs, temporarily coordinating each mitotic event. Cyclin D1 has been shown to interact with the retinoblastoma (Rb) tumor suppressor protein, and mutations in this gene lead to cell cycle modification and progression in various tumors, contributing to tumorigenesis, especially in the breast [89,90]. Overexpression of cyclin D1 may also decrease Fas expression, increasing the resistance of the cells against apoptosis and oncological chemotherapeutic treatment [91]. Intense immunolabeling positivity was also reported in the case of MLMS described by us, which suggests that the survival of the patient with this type of cancer may be shorter and has an increased risk of being associated with metastases [92].
Vimentin is a type III IF protein present in mesenchymal cells, where it is a major cytoskeletal component. Therefore, this protein is used as a tumor marker to identify mesenchyma. The described tumor cells were intensely immunolabeled upon immunoreaction with the anti-vimentin antibody, thus demonstrating tumor belonging to mesenchymal origin [93].
p16 or p16INK4a is a CDKN2A. It is a tumor suppressor that slows the progression of the cell cycle from phase G1 to phase S. Mutations in the gene encoding this tumor suppressor leads to cell cycle acceleration and progression of different types of cancer. MLMS overexpression shows that this tumor has an oncological transformation with accelerated cell cycles, which determines an unfavorable prognosis [94,95]. In gynecological pathology, this marker is widely used. Nuclear and cytoplasmic expressions are strongly associated with human papillomavirus (HPV) infection and neoplasms of cervical origin. p16 can be expressed in other malignant tumors and many normal human tissues [96,97].
⧉ Conclusions
The status of the menopausal patient, the pelvic symptomatology, the macroscopic tumor appearance, with fleshy, soft features pleaded for a malignancy pathology. The microscopic features were represented by the irregular myometrial invasion, rare mitosis on HPFs, cell pleomorphism, predominant myxoid component. This hypocellular appearance, with a matrix rich in PG and GAG, especially HA, laid the foundation for the diagnosis of MLMS.
Conflict of interest
The authors declare that they have no conflict of interests.
Authors’ contribution
Anca-Maria Istrate-Ofiţeru and George-Lucian Zorilă equally contributed to this article.
Acknowledgments
Microscopic images have been acquired in the Research Center for Microscopic Morphology and Immunology, University of Medicine and Pharmacy of Craiova, Romania (Manager: Professor Laurenţiu Mogoantă, MD, PhD).
References
- 1.Papadopoulos AJ, Kenney A. Solid malignant uterine tumours. Curr Obstet Gynaecol. 2001;11(5):296–301. https://www.sciencedirect.com/science/article/abs/pii/S0957584701901994?via%3Dihub [Google Scholar]
- 2.King ME, Dickersin GR, Scully RE. Myxoid leiomyosarcoma of the uterus. A report of six cases. Am J Surg Pathol. 1982;6(7):589–598. doi: 10.1097/00000478-198210000-00001. [DOI] [PubMed] [Google Scholar]
- 3.Burch DM, Tavassoli FA. Myxoid leiomyosarcoma of the uterus. Histopathology. 2011;59(6):1144–1155. doi: 10.1111/j.1365-2559.2011.04053.x. [DOI] [PubMed] [Google Scholar]
- 4.Imai H, Yagi H, Okugawa K, Kenjo H, Ohgami T, Kawano Y, Kaneki E, Ichinoe A, Asanoma K, Yahata H, Sonoda K, Kobayashi H, Kaku T, Kato K. Uterine myxoid leiomyosarcoma with tumor embolism extending into the right atrium. Case Rep Obstet Gynecol. 2015;2015:316262–316262. doi: 10.1155/2015/316262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mitrache LE, Dumitru VA, Simion G, Cirstoiu M, Sajin M. A rare case of uterine myxoid leiomyosarcoma: a case report. Maedica (Bucharest) 2014;9(3):269–271. [PMC free article] [PubMed] [Google Scholar]
- 6.Shintaku M, Ashihara T, Koyama T. Myxoid leiomyosarcoma of the uterus with mature adipocytes and numerous bizarre multinucleated giant cells. Pathol Int. 2015;65(4):205–207. doi: 10.1111/pin.12251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Barone A, Ambrosio MR, Rocca BJ, Mastrogiulio MG, Ambrosio A, Santopietro R. Myxoid leiomyosarcoma of the uterus: a case report. Eur J Gynaecol Oncol. 2014;35(3):322–324. [PubMed] [Google Scholar]
- 8.Parra-Herran C, Schoolmeester JK, Yuan L, Dal Cin, Fletcher CDM, Quade BJ, Nucci MR. Myxoid leiomyosarcoma of the uterus: a clinicopathologic analysis of 30 cases and review of the literature with reappraisal of its distinction from other uterine myxoid mesenchymal neoplasms. Am J Surg Pathol. 2016;40(3):285–301. doi: 10.1097/PAS.0000000000000593. [DOI] [PubMed] [Google Scholar]
- 9.Harlow BL, Weiss NS, Lofton S. The epidemiology of sarcomas of the uterus. J Natl Cancer Inst. 1986;76(3):399–402. [PubMed] [Google Scholar]
- 10.Kagami S, Kashimura M, Toki N, Katuhata Y. Myxoid leiomyosarcoma of the uterus with subsequent pregnancy and delivery. Gynecol Oncol. 2002;85(3):538–542. doi: 10.1006/gyno.2002.6634. [DOI] [PubMed] [Google Scholar]
- 11.Lurain JR. In: Novak’s Gynecology. 12. Berek JS, Adashi EY, Hillard PA, editors. Baltimore USA: Williams & Wilkins; 1996. Uterine cancer; pp. 1094–1095.https://www.worldcat.org/title/novaks-gynecology/oclc/1150935563 [Google Scholar]
- 12.Silva EG, Tornos CS, Follen-Mitchell M. Malignant neoplasms of the uterine corpus in patients treated for breast carcinoma: the effects of Tamoxifen. Int J Gynecol Pathol. 1994;13(3):248–258. doi: 10.1097/00004347-199407000-00009. [DOI] [PubMed] [Google Scholar]
- 13.McCluggage WG, Varma M, Weir P, Bharucha H. Uterine leiomyosarcoma in patient receiving Tamoxifen therapy. Acta Obstet Gynecol Scand. 1996;75(6):593–595. doi: 10.3109/00016349609054678. [DOI] [PubMed] [Google Scholar]
- 14.Kaganov H, Ades A, Fraser DS. Preoperative magnetic resonance imaging diagnostic features of uterine leiomyosarcomas: a systematic review. Int J Technol Assess Health Care. 2018;34(2):172–179. doi: 10.1017/S0266462318000168. [DOI] [PubMed] [Google Scholar]
- 15.Lakhman Y, Veeraraghavan H, Chaim J, Feier D, Goldman DA, Moskowitz CS, Nougaret S, Sosa RE, Vargas HA, Soslow RA, Abu-Rustum NR, Hricak H, Sala E. Differentiation of uterine leiomyosarcoma from atypical leiomyoma: diagnostic accuracy of qualitative MR imaging features and feasibility of texture analysis. Eur Radiol. 2017;27(7):2903–2915. doi: 10.1007/s00330-016-4623-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Thomassin-Naggara I, Dechoux S, Bonneau C, Morel A, Rouzier R, Carette MF, Daraï E, Bazot M. How to differentiate benign from malignant myometrial tumours using MR imaging. Eur Radiol. 2013;23(8):2306–2314. doi: 10.1007/s00330-013-2819-9. [DOI] [PubMed] [Google Scholar]
- 17.Gerges L, Popiolek D, Rosenkrantz AB. Explorative investigation of whole-lesion histogram MRI metrics for differentiating uterine leiomyomas and leiomyosarcomas. AJR Am J Roentgenol. 2018;210(5):1172–1177. doi: 10.2214/AJR.17.18605. [DOI] [PubMed] [Google Scholar]
- 18.Li HM, Liu J, Qiang JW, Zhang H, Zhang GF, Ma FH. Diffusion-weighted imaging for differentiating uterine leiomyosarcoma from degenerated leiomyoma. J Comput Assist Tomogr. 2017;41(4):599–606. doi: 10.1097/RCT.0000000000000565. [DOI] [PubMed] [Google Scholar]
- 19.Skorstad M, Kent A, Lieng M. Preoperative evaluation in women with uterine leiomyosarcoma. A nationwide cohort study. Acta Obstet Gynecol Scand. 2016;95(11):1228–1234. doi: 10.1111/aogs.13008. [DOI] [PubMed] [Google Scholar]
- 20.Lin G, Yang LY, Huang YT, Ng KK, Ng SH, Ueng SH, Chao A, Yen TC, Chang TC, Lai CH. Comparison of the diagnostic accuracy of contrast-enhanced MRI and diffusion-weighted MRI in the differentiation between uterine leiomyosarcoma/smooth muscle tumor with uncertain malignant potential and benign leiomyoma. J Magn Reson Imaging. 2016;43(2):333–342. doi: 10.1002/jmri.24998. [DOI] [PubMed] [Google Scholar]
- 21.Jansen JF, Lu Y, Gupta G, Lee NY, Stambuk HE, Mazaheri Y, Deasy JO, Shukla-Dave A. Texture analysis on parametric maps derived from dynamic contrast-enhanced magnetic resonance imaging in head and neck cancer. World J Radiol. 2016;8(1):90–97. doi: 10.4329/wjr.v8.i1.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gaetke-Udager K, McLean K, Sciallis AP, Alves T, Maturen KE, Mervak BM, Moore AG, Wasnik AP, Erba J, Davenport MS. Diagnostic accuracy of ultrasound, contrast-enhanced CT, and conventional MRI for differentiating leiomyoma from leiomyosarcoma. Acad Radiol. 2016;23(10):1290–1297. doi: 10.1016/j.acra.2016.06.004. [DOI] [PubMed] [Google Scholar]
- 23.Sato K, Yuasa N, Fujita M, Fukushima Y. Clinical application of diffusion-weighted imaging for preoperative differentiation between uterine leiomyoma and leiomyosarcoma. Am J Obstet Gynecol. 2014;210(4):368e1–368e8. doi: 10.1016/j.ajog.2013.12.028. [DOI] [PubMed] [Google Scholar]
- 24.Namimoto T, Yamashita Y, Awai K, Nakaura T, Yanaga Y, Hirai T, Saito T, Katabuchi H. Combined use of T2-weighted and diffusion-weighted 3-T MR imaging for differentiating uterine sarcomas from benign leiomyomas. Eur Radiol. 2009;19(11):2756–2764. doi: 10.1007/s00330-009-1471-x. [DOI] [PubMed] [Google Scholar]
- 25.Tamai K, Koyama T, Saga T, Morisawa N, Fujimoto K, Mikami Y, Togashi K. The utility of diffusion-weighted MR imaging for differentiating uterine sarcomas from benign leiomyomas. Eur Radiol. 2008;18(4):723–730. doi: 10.1007/s00330-007-0787-7. [DOI] [PubMed] [Google Scholar]
- 26.Oliva W, Carcangiu ML, Carinelli SG, Herrington CS, Young RH. In: World Health Organization (WHO) Classification of female reproductive organs. 4. Kurman RJ, Carcangiu ML, Herrington S, Young RH, editors. Vol. 6. Lyon France: International Agency for Research on Cancer (IARC) Press; 2014. Mesenchymal tumours. Chapter 5: Tumours of the uterine corpus; pp. 135–147.https://publications.iarc.fr/Book-And-Report-Series/Who-Classification-Of-Tumours/WHO-Classification-Of-Tumours-Of-Female-Reproductive-Organs-2014 [Google Scholar]
- 27.Bell SW, Kempson RL, Hendrickson MR. Problematic uterine smooth muscle neoplasms. A clinicopathologic study of 213 cases. Am J Surg Pathol. 1994;18(6):535–558. [PubMed] [Google Scholar]
- 28.Rosai J. Rosai and Ackerman’s surgical pathology. 10. New York, USA: Mosby–Elsevier Inc; 2011. https://www.elsevier.com/books/rosai-and-ackermans-surgical-pathology-10e/rosai/978-81-312-2984-2 [Google Scholar]
- 29.Berchuck A, Rubin SC, Hoskins WJ, Saigo PE, Pierce VK, Lewis JL. Treatment of uterine leiomyosarcoma. Obstet Gynecol. 1988;71(6 Pt 1):845–850. [PubMed] [Google Scholar]
- 30.Barter JF, Smith EB, Szpak CA, Hinshaw W, Clarke-Pearson DL, Creasman WT. Leiomyosarcoma of the uterus. Clinicopathologic study of 21 cases. Gynecol Oncol. 1985;21(2):220–227. doi: 10.1016/0090-8258(85)90256-2. [DOI] [PubMed] [Google Scholar]
- 31.Vardi JR, Tovell HM. Leiomyosarcoma of the uterus: clinico-pathologic study. Obstet Gynecol. 1980;56(4):428–434. [PubMed] [Google Scholar]
- 32.Mayerhofer K, Obermair A, Windbichler G, Petru E, Kaider A, Hefler L, Czerwenka K, Leodolter S, Kainz C. Leiomyosarcoma of the uterus: a clinicopathologic multicenter study of 71 cases. Gynecol Oncol. 1999;74(2):196–201. doi: 10.1006/gyno.1999.5436. [DOI] [PubMed] [Google Scholar]
- 33.Burns B, Curry RH, Bell ME. Morphologic features of prognostic significance in uterine smooth muscle tumors: a review of eighty-four cases. Am J Obstet Gynecol. 1979;135(1):109–114. [PubMed] [Google Scholar]
- 34.Zaloudek C, Norris HJ. In: Blaustein’s pathology of the female genital tract. 4. Kurman RJ, editor. New York USA: Springer-Verlag; 1994. Mesenchymal tumors of the uterus; pp. 487–528.https://www.worldcat.org/title/blausteins-pathology-of-the-female-genital-tract/oclc/902907632?referer=di&ht=edition [Google Scholar]
- 35.Friedrich M, Villena-Heinsen C, Mink D, Hell K, Schmidt W. Leiomyosarcomas of the female genital tract: a clinical and histopathologic study. Eur J Gynaecol Oncol. 1998;19(5):470–475. [PubMed] [Google Scholar]
- 36.Atkins KA, Arronte N, Darus CJ, Rice LW. The use of p16 in enhancing the histologic classification of uterine smooth muscle tumors. Am J Surg Pathol. 2008;32(1):98–102. doi: 10.1097/PAS.0b013e3181574d1e. [DOI] [PubMed] [Google Scholar]
- 37.O’Neill CJ, McBride HA, Connolly LE, McCluggage WG. Uterine leiomyosarcomas are characterized by high p16, p53 and MIB1 expression in comparison with usual leiomyomas, leiomyoma variants and smooth muscle tumours of uncertain malignant potential. Histopathology. 2007;50(7):851–858. doi: 10.1111/j.1365-2559.2007.02699.x. [DOI] [PubMed] [Google Scholar]
- 38.Chen L, Yang B. Immunohistochemical analysis of p16, p53, and Ki-67 expression in uterine smooth muscle tumors. Int J Gynecol Pathol. 2008;27(3):326–332. doi: 10.1097/PGP.0b013e31815ea7f5. [DOI] [PubMed] [Google Scholar]
- 39.Niemann TH, Raab SS, Lenel JC, Rodgers JR, Robinson RA. p53 protein overexpression in smooth muscle tumors of the uterus. Hum Pathol. 1995;26(4):375–379. doi: 10.1016/0046-8177(95)90136-1. [DOI] [PubMed] [Google Scholar]
- 40.Cornejo K, Shi M, Jiang Z. Oncofetal protein IMP3: a useful diagnostic biomarker for leiomyosarcoma. Hum Pathol. 2012;43(10):1567–1572. doi: 10.1016/j.humpath.2011.12.020. [DOI] [PubMed] [Google Scholar]
- 41.Willems SM, Wiweger M, van Roggen JFG, Hogendoorn PCW. Running GAGs: myxoid matrix in tumor pathology revisited: what’s in it for the pathologist. Virchows Arch. 2010;456(2):181–192. doi: 10.1007/s00428-009-0822-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Clement PB, Young RH, Scully RE. Diffuse, perinodular, and other patterns of hydropic degeneration within and adjacent to uterine leiomyomas. Problems in differential diagnosis. Am J Surg Pathol. 1992;16(1):26–32. doi: 10.1097/00000478-199201000-00004. [DOI] [PubMed] [Google Scholar]
- 43.Busca A, Parra-Herran C. Myxoid mesenchymal tumors of the uterus: an update on classification, definitions, and differential diagnosis. Adv Anat Pathol. 2017;24(6):354–361. doi: 10.1097/PAP.0000000000000164. [DOI] [PubMed] [Google Scholar]
- 44.Wang WL, Soslow R, Hensley M, Asad H, Zannoni GF, de Nictolis M, Branton P, Muzikansky A, Oliva E. Histopathologic prognostic factors in stage I leiomyosarcoma of the uterus: a detailed analysis of 27 cases. Am J Surg Pathol. 2011;35(4):522–529. doi: 10.1097/PAS.0b013e31820ca624. [DOI] [PubMed] [Google Scholar]
- 45.Abeler VM, Røyne O, Thoresen S, Danielsen HE, Nesland JM, Kristensen GB. Uterine sarcomas in Norway. A histopathological and prognostic survey of a total population from 1970 to 2000 including 419 patients. Histopathology. 2009;54(3):355–364. doi: 10.1111/j.1365-2559.2009.03231.x. [DOI] [PubMed] [Google Scholar]
- 46.Mills AM, Longacre TA. Smooth muscle tumors of the female genital tract. Surg Pathol Clin. 2009;2(4):625–677. doi: 10.1016/j.path.2009.08.019. [DOI] [PubMed] [Google Scholar]
- 47.Wang Y, Teng Y, Na S, Yuan Y. Pleomorphic leiomyosarcoma of the adrenal gland in a young woman: a case report and review of the literature. Onco Targets Ther. 2020;13:4705–4713. doi: 10.2147/OTT.S254162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sprogøe-Jakobsen S, Hølund B. Immunohistochemistry (Ki-67 and p53) as a tool in determining malignancy in smooth muscle neoplasms (exemplified by a myxoid leiomyosarcoma of the uterus) APMIS. 1996;104(10):705–708. doi: 10.1111/j.1699-0463.1996.tb04932.x. [DOI] [PubMed] [Google Scholar]
- 49.Lu B, Shi H, Zhang X. Myxoid leiomyosarcoma of the uterus: a clinicopathological and immunohistochemical study of 10 cases. Hum Pathol. 2017;59:139–146. doi: 10.1016/j.humpath.2016.09.014. [DOI] [PubMed] [Google Scholar]
- 50.Laforga JB, Cortés VA. Uterine pleomorphic leiomyosarcoma with osteoclastic giant cells: case report with peritoneal washing cytology and cell block study. Rev Esp Patol. 2020;53(1):61–65. doi: 10.1016/j.patol.2018.06.002. [DOI] [PubMed] [Google Scholar]
- 51.Corscaden JA, Singh BP. Leiomyosarcoma of the uterus. Am J Obstet Gynecol. 1958;75(1):149–153; discussion 153–155. doi: 10.1016/0002-9378(58)90563-5. [DOI] [PubMed] [Google Scholar]
- 52.Montague AC, Swartz DP, Woodruff JD. Sarcoma arising in a leiomyoma of the uterus: factors influencing prognosis. Am J Obstet Gynecol. 1965;92:421–427. doi: 10.1016/0002-9378(65)90395-9. [DOI] [PubMed] [Google Scholar]
- 53.Gallup DG, Corday DR. Leiomyosarcoma of the uterus: case reports and a review. Obstet Gynecol Surv. 1979;34(4):300–312. doi: 10.1097/00006254-197904000-00017. [DOI] [PubMed] [Google Scholar]
- 54.Leibsohn S, d’Ablaing G, Mishell DR, Schlaerth JB. Leiomyosarcoma in a series of hysterectomies performed for presumed uterine leiomyomas. Am J Obstet Gynecol. 1990;162(4):968–974; discussion 974–976. doi: 10.1016/0002-9378(90)91298-q. [DOI] [PubMed] [Google Scholar]
- 55.Ng WK, Lui PCW, Ma L. Peritoneal washing cytology findings of disseminated myxoid leiomyosarcoma of uterus: report of a case with emphasis on possible differential diagnosis. Diagn Cytopathol. 2002;27(1):47–52. doi: 10.1002/dc.10141. [DOI] [PubMed] [Google Scholar]
- 56.Takano Y, Morimura Y, Yamada H, Yanagida K, Sato A, Suzuki O, Suzuki T. Myxoid leiomyosarcoma of the uterus. Fukushima J Med Sci. 2000;46(1–2):41–47. doi: 10.5387/fms.46.41. [DOI] [PubMed] [Google Scholar]
- 57.Schneider D, Halperin R, Segal M, Maymon R, Bukovsky I. Myxoid leiomyosarcoma of the uterus with unusual malignant histologic pattern – a case report. Gynecol Oncol. 1995;59(1):156–158. doi: 10.1006/gyno.1995.1284. [DOI] [PubMed] [Google Scholar]
- 58.Kaleli S, Calay Z, Ceydeli N, Aydýnlý K, Kösebay D. A huge abdominal mass mimicking ovarian cancer: p53-negative but aneuploid myxoid leiomyosarcoma of the uterus. Eur J Obstet Gynecol Reprod Biol. 2001;100(1):96–99. doi: 10.1016/s0301-2115(01)00446-8. [DOI] [PubMed] [Google Scholar]
- 59.Mittal K, Popiolek D, Demopoulos RI. Uterine myxoid leiomyosarcoma within a leiomyoma. Hum Pathol. 2000;31(3):398–400. doi: 10.1016/s0046-8177(00)80258-0. [DOI] [PubMed] [Google Scholar]
- 60.Kunzel KE, Mills NZ, Muderspach LI, d’Ablaing G. Myxoid leiomyosarcoma of the uterus. Gynecol Oncol. 1993;48(2):277–280. doi: 10.1006/gyno.1993.1048. [DOI] [PubMed] [Google Scholar]
- 61.Prat J. FIGO staging for uterine sarcomas. Int J Gynaecol Obstet. 2009;104(3):177–178. doi: 10.1016/j.ijgo.2008.12.008. [DOI] [PubMed] [Google Scholar]
- 62.Toki N, Kashimura M, Hasegawa T, Fukuoka K, Kawagoe T, Sugihara K, Koyama C, Hisaoka M. Myxoid leiomyosarcoma of the uterus. Report of a case with cytologic findings. Acta Cytol. 2000;44(3):415–419. doi: 10.1159/000328489. [DOI] [PubMed] [Google Scholar]
- 63.Brodehl A, Gaertner-Rommel A, Milting H. Molecular insights into cardiomyopathies associated with desmin (DES) mutations. Biophys Rev. 2018;10(4):983–1006. doi: 10.1007/s12551-018-0429-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sequeira V, Nijenkamp LLAM, Regan JA, van der Velden J. The physiological role of cardiac cytoskeleton and its alterations in heart failure. Biochim Biophys Acta. 2014;1838(2):700–722. doi: 10.1016/j.bbamem.2013.07.011. [DOI] [PubMed] [Google Scholar]
- 65.Li Z, Mericskay M, Agbulut O, Butler-Browne G, Carlsson L, Thornell LE, Babinet C, Paulin D. Desmin is essential for the tensile strength and integrity of myofibrils but not for myogenic commitment, differentiation, and fusion of skeletal muscle. J Cell Biol. 1997;139(1):129–144. doi: 10.1083/jcb.139.1.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Edling CE, Hallberg B. c-Kit – a hematopoietic cell essential receptor tyrosine kinase. Int J Biochem Cell Biol. 2007;39(11):1995–1998. doi: 10.1016/j.biocel.2006.12.005. [DOI] [PubMed] [Google Scholar]
- 67.Leong AS, Cooper K, Leong FJ. Manual of diagnostic cytology. 2. Cambridge, UK: Greenwich Medical Media, Ltd; 2003. pp. 149–151.https://www.scirp.org/(S(351jmbntvnsjt1aadkposzje))/reference/ReferencesPapers.aspx?ReferenceID=508105 [Google Scholar]
- 68.Bingle L, Brown NJ, Lewis CE. The role of tumour-associated macrophages in tumour progression: implications for new anticancer therapies. J Pathol. 2002;196(3):254–265. doi: 10.1002/path.1027. [DOI] [PubMed] [Google Scholar]
- 69.Condeelis J, Pollard JW. Macrophages: obligate partners for tumor cell migration, invasion, and metastasis. Cell. 2006;124(2):263–266. doi: 10.1016/j.cell.2006.01.007. [DOI] [PubMed] [Google Scholar]
- 70.Dirkx AEM, Oude Egbrink MGA, Wagstaff J, Griffioen AW. Monocyte/macrophage infiltration in tumors: modulators of angiogenesis. J Leukoc Biol. 2006;80(6):1183–1196. doi: 10.1189/jlb.0905495. [DOI] [PubMed] [Google Scholar]
- 71.Lewis CE, Pollard JW. Distinct role of macrophages in different tumor microenvironments. Cancer Res. 2006;66(2):605–612. doi: 10.1158/0008-5472.CAN-05-4005. [DOI] [PubMed] [Google Scholar]
- 72.Pollard JW. Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer. 2004;4(1):71–78. doi: 10.1038/nrc1256. [DOI] [PubMed] [Google Scholar]
- 73.Ohno S, Suzuki N, Ohno Y, Inagawa H, Soma GI, Inoue M. Tumor-associated macrophages: foe or accomplice of tumors. Anticancer Res. 2003;23(6a):4395–4409. [PubMed] [Google Scholar]
- 74.Hamada I, Kato M, Yamasaki T, Iwabuchi K, Watanabe T, Yamada T, Itoyama S, Ito H, Okada K. Clinical effects of tumor-associated macrophages and dendritic cells on renal cell carcinoma. Anticancer Res. 2002;22(6C):4281–4284. [PubMed] [Google Scholar]
- 75.Hanada T, Nakagawa M, Emoto A, Nomura T, Nasu N, Nomura Y. Prognostic value of tumor-associated macrophage count in human bladder cancer. Int J Urol. 2000;7(7):263–269. doi: 10.1046/j.1442-2042.2000.00190.x. [DOI] [PubMed] [Google Scholar]
- 76.Koide N, Nishio A, Sato T, Sugiyama A, Miyagawa S. Significance of macrophage chemoattractant protein-1 expression and macrophage infiltration in squamous cell carcinoma of the esophagus. Am J Gastroenterol. 2004;99(9):1667–1674. doi: 10.1111/j.1572-0241.2004.30733.x. [DOI] [PubMed] [Google Scholar]
- 77.Leek RD, Landers RJ, Harris AL, Lewis CE. Necrosis correlates with high vascular density and focal macrophage infiltration in invasive carcinoma of the breast. Br J Cancer. 1999;79(5–6):991–995. doi: 10.1038/sj.bjc.6690158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Lissbrant IF, Stattin P, Wikstrom P, Damber JE, Egevad L, Bergh A. Tumor associated macrophages in human prostate cancer: relation to clinicopathological variables and survival. Int J Oncol. 2000;17(3):445–451. doi: 10.3892/ijo.17.3.445. [DOI] [PubMed] [Google Scholar]
- 79.Ohno S, Ohno Y, Suzuki N, Kamei T, Koike K, Inagawa H, Kohchi C, Soma GI, Inoue M. Correlation of histological localization of tumor-associated macrophages with clinicopathological features in endometrial cancer. Anticancer Res. 2004;24(5C):3335–3342. [PubMed] [Google Scholar]
- 80.Istrate Ofiteru AM, Berceanu S, Paitici S, Rosu GC, Iovan L, Voicu NL, Pirici D, Mogoanta L, Vladareanu R, Mehedintu C, Bratilă E, Bratu O, Berceanu C. Endometriosis of the abdominal wall – clinical, histopathological and immunohistochemical aspects. Rev Chim (Bucharest) 2019;70(8):2860–2865. https://revistadechimie.ro/Articles.asp?ID=7444 [Google Scholar]
- 81.Istrate-Ofiţeru AM, Pîrvan IC, Pirici D, Roşu GC, Niculescu M, Berceanu S, Manolea MM, Comănescu MV, Voicu NL, Iovan L, Vasile MM, Căpitănescu RG, Diţescu D, Mogoantă L, Berceanu C. Triple immunohistochemistry for assessing the inflammatory, vascular and progression of adenomyosis. Rom J Morphol Embryol. 2019;60(2):419–428. [PubMed] [Google Scholar]
- 82.Istrate-Ofiţeru AM, Pirici D, Niculescu M, Berceanu C, Berceanu S, Voicu NL, Piringă GD, Roşu GC, Iovan L, Căpitănescu RG, Diţescu D, Sava A, Mogoantă L, Neacşu A. Clinical, morphological and immunohistochemical survey in different types of endometriosis. Rom J Morphol Embryol. 2018;59(4):1133–1153. [PubMed] [Google Scholar]
- 83.Leong AS, Cooper K, Leong FJW. Manual of diagnostic cytology. 2. Cambridge, UK: Greenwich Medical Media, Ltd; 2003. pp. 173–173.https://www.scirp.org/(S(351jmbntvnsjt1aadkposzje))/reference/ReferencesPapers.aspx?ReferenceID=508105 [Google Scholar]
- 84.***. Cytokeratin cocktail AE1 & AE3 (AE1/AE3), MMab. Bio SB – Bioscience for the World, 2016. https://www.biosb.com/biosb-products/cytokeratin-cocktail-ae1-ae3-antibody-mmab-ae1-ae3/
- 85.Ramgolam K, Lauriol J, Lalou C, Lauden L, Michel L, de la, Khatib AM, Aoudjit F, Charron D, Alcaide-Loridan C, Al-Daccak R. Melanoma spheroids grown under neural crest cell conditions are highly plastic migratory/invasive tumor cells endowed with immunomodulator function. PLoS One. 2011;6(4):e18784–e18784. doi: 10.1371/journal.pone.0018784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Mahmood MN, Lee MW, Linden MD, Nathanson SD, Hornyak TJ, Zarbo RJ. Diagnostic value of HMB-45 and anti-Melan A staining of sentinel lymph nodes with isolated positive cells. Mod Pathol. 2002;15(12):1288–1293. doi: 10.1097/01.MP.0000037313.33138.DF. [DOI] [PubMed] [Google Scholar]
- 87.Withers DA, Harvey RC, Faust JB, Melnyk O, Carey K, Meeker TC. Characterization of a candidate bcl-1 gene. Mol Cell Biol. 1991;11(10):4846–4853. doi: 10.1128/mcb.11.10.4846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Inaba T, Matsushime H, Valentine M, Roussel MF, Sherr CJ, Look AT. Genomic organization, chromosomal localization, and independent expression of human cyclin D genes. Genomics. 1992;13(3):565–574. doi: 10.1016/0888-7543(92)90126-d. [DOI] [PubMed] [Google Scholar]
- 89.He Y, Liu Z, Qiao C, Xu M, Yu J, Li G. Expression and significance of Wnt signaling components and their target genes in breast carcinoma. Mol Med Rep. 2014;9(1):137–143. doi: 10.3892/mmr.2013.1774. [DOI] [PubMed] [Google Scholar]
- 90.Diehl JA. Cycling to cancer with cyclin D1. Cancer Biol Ther. 2002;1(3):226–231. doi: 10.4161/cbt.72. [DOI] [PubMed] [Google Scholar]
- 91.Shintani M, Okazaki A, Masuda T, Kawada M, Ishizuka M, Doki Y, Weinstein IB, Imoto M. Overexpression of cyclin D1 contributes to malignant properties of esophageal tumor cells by increasing VEGF production and decreasing Fas expression. Anticancer Res. 2002;22(2A):639–647. [PubMed] [Google Scholar]
- 92.Jares P, Colomer D, Campo E. Genetic and molecular pathogenesis of mantle cell lymphoma: perspectives for new targeted therapeutics. Nat Rev Cancer. 2007;7(10):750–762. doi: 10.1038/nrc2230. [DOI] [PubMed] [Google Scholar]
- 93.Leader M, Collins M, Patel J, Henry K. Vimentin: an evaluation of its role as a tumour marker. Histopathology. 1987;11(1):63–72. doi: 10.1111/j.1365-2559.1987.tb02609.x. [DOI] [PubMed] [Google Scholar]
- 94.Nobori T, Miura K, Wu DJ, Lois A, Takabayashi K, Carson DA. Deletions of the cyclin-dependent kinase-4 inhibitor gene in multiple human cancers. Nature. 1994;368(6473):753–756. doi: 10.1038/368753a0. [DOI] [PubMed] [Google Scholar]
- 95.Stone S, Jiang P, Dayananth P, Tavtigian SV, Katcher H, Parry D, Peters G, Kamb A. Complex structure and regulation of the P16 (MTS1) locus. Cancer Res. 1995;55(14):2988–2994. [PubMed] [Google Scholar]
- 96.Rayess H, Wang MB, Srivatsan ES. Cellular senescence and tumor suppressor gene p16. Int J Cancer. 2012;130(8):1715–1725. doi: 10.1002/ijc.27316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Cioffi-Lavina M, Chapman-Fredricks J, Gomez-Fernandez C, Ganjei-Azar P, Manoharan M, Jorda M. P16 expression in squamous cell carcinomas of cervix and bladder. Appl Immunohistochem Mol Morphol. 2010;18(4):344–347. doi: 10.1097/PAI.0b013e3181d2bbd7. [DOI] [PubMed] [Google Scholar]