Abstract
Desaturation of inert aliphatic C−H bonds in alkanes to form the corresponding alkenes is challenging. In this communication, a new and practical strategy for remote site‐selective desaturation of amides via radical chemistry is reported. The readily installed N‐allylsulfonylamide moiety serves as an N radical precursor. Intramolecular 1,5‐hydrogen atom transfer from an inert C−H bond to the N‐radical generates a translocated C‐radical which is subsequently oxidized and deprotonated to give the corresponding alkene. The commercially available methanesulfonyl chloride is used as reagent and a Cu/Ag‐couple as oxidant. The remote desaturation is realized on different types of unactivated sp3‐C−H bonds. The potential synthetic utility of this method is further demonstrated by the dehydrogenation of natural product derivatives and drugs.
Keywords: alkenes, desaturation, oxidation, radical, reaction mechanism
A copper‐catalyzed/silver‐promoted desaturation method is described which allows the regioselective conversion of various unactivated N‐alkylamides at remote sites to the corresponding N‐alkenylamides via N‐centred radical mediated 1,5 hydrogen atom transfer, metal‐mediated oxidation and deprotonation. The transformation shows good functional group tolerance and can be used for late‐stage desaturation of drug derivatives and biologically more relevant compounds.
Alkenes are key chemical stocks that are also widely found as natural products and as versatile building blocks in organic synthesis. [1] Great efforts towards the synthesis of these privileged motifs have been made. Among them, the removal of two vicinal hydrogen atoms from an alkane to directly give an alkene represents a highly efficient and straightforward approach for the formation of an alkene. [2] In this context, the enzymatic desaturation of fatty acid derivatives [3] and dehydrogenation of activated aliphatic C−H bonds [4] constitute the most general processes for such a purpose. However, the site‐selective desaturation of unactivated C−H bonds has been rarely achieved and is highly challenging for synthetic chemists due to the inherent difficulty for site‐selective activation of such inert σ‐bonds. The biomimetic dehydrogenation of steroid frameworks was first described by Breslow and co‐workers. [5] In their seminal study, intramolecular hydrogen atom transfer (HAT) was used for C−H activation and the potential of such a strategy for late‐stage desaturation of biologically active compounds was convincingly documented. Utilizing the HAT‐strategy, Čeković and co‐workers developed a Fe(II)/Cu(II) induced desaturation of alkyl hydroperoxides via oxygen centered radicals [6] (Scheme 1a). Following these pioneering works, a breakthrough was achieved by the group of Baran who introduced a TEMPO‐mediated dehydrogenation with aryl triazenes as aryl radical precursors, where remote C‐radical generation was achieved by HAT to reactive aryl radicals. [7] Recently, Gevorgyan and co‐workers developed an elegant strategy that used 1,5‐HAT to reactive C‐radicals for radical translocation and subsequent desaturation is achieved via Pd‐catalyzed β‐H elimination (Scheme 1b). [8]
Scheme 1.
Different methods for radical desaturation of unactivated sites.
Surprisingly, remote desaturation mediated by N‐centered radicals is not well investigated, despite the fact, that the Hofmann‐Löffler‐Freytag (HLF) reaction and variants thereof reliably allow for remote site‐selective functionalization through the generation of C‐radicals at unactivated sites via 1,5‐HAT.[ 9 , 10 ] Desaturation via an HLF‐type process could be indeed achieved, but in these cases, the intermediately formed alkenes directly engaged in follow‐up reactions under the applied conditions.[ 11 , 12 ] To the best of our knowledge, N radical‐mediated desaturation at remote unactivated C−H sites remains unknown at present. [13] The big challenge lies in the fact that the desaturated targeted product is far more reactive than the starting saturated substrate! Therefore, the development of a general desaturation method in amides at unactivated positions would be highly valuable.
Applying a HLF‐type strategy, Pratt et al. and our group have recently achieved remote C−H functionalization of unactivated C−H bonds in amides with various sulfones and in situ generated deuterated thiol. [14] The readily installed N‐allylsulfonyl moiety [15] served as the N‐radical precursor in these transformations. Inspired by these results, we questioned whether N radical mediated remote desaturation of amides could be realized by using a mild external oxidant that allows the translocated C‐radical to be cleanly converted to the corresponding alkene. Herein, we describe our results on a copper/silver‐promoted HLF‐type desaturation at unactivated sites in various amides (Scheme 1c).
Our study began with sulfonamide 1 a as the model substrate (Table 1). The reaction of 1 a with Togni's reagent 2 a in the presence of CuCl catalyst and Cs2CO3 in ethyl acetate (EA) provided the desired product 3 a, albeit in only 7 % yield (entry 1). The product was obtained as a 1.2 : 1 mixture of regioisomers with the internal alkene formed as the major compound (rr=regioisomer ratio; dashed line in the structure indicates the double bond position in the minor isomer). A better result was achieved with BPO (2 b) as both radical source and oxidant in the absence of any Cu‐catalyst (32 %, entry 2). Guided by our previous work, [14] we next selected various sulfonyl radical precursors as reagents to mediate the remote desaturation of 1 a. To our delight, in the presence of CuCl as catalyst (10 mol%) and Ag2CO3 as additive, the desired remotely desaturated product 3 a was obtained in 52 % yield and 1.7 : 1 regioselectivity by using commercially available methanesulfonyl chloride as the reagent (entry 3). Reaction optimization was continued by varying the catalyst (entries 4–8) and copper(I) iodide was identified as the ideal choice for this transformation to give the alkene 3 a in 63 % isolated yield with 2 : 1 selectivity (entry 8). Screening of the sulfonyl radical source revealed a similar result with 4‐chlorobenzenesulfonyl chloride, whereas with tosyl chloride or triflyl chloride significantly lower yields were obtained (entries 9–11). No further improvement of the yield was achieved upon additive and solvent screening (entries 12–14 and Supporting Information).
Table 1.
Reaction optimization.
|
| ||||
|---|---|---|---|---|
|
Entry |
2 |
Catalyst |
Additive |
3 a (rr) |
|
1 |
2 a |
CuCl |
Cs2CO3 |
7 % (1.2 : 1)[a] |
|
2 |
2 b |
– |
– |
32 % (2.4 : 1)[b] |
|
3 |
2 c |
CuCl |
Ag2CO3 |
52 % (1.7 : 1) |
|
4 |
2 c |
NiCl2 |
Ag2CO3 |
5 % (2.2 : 1)[c] |
|
5 |
2 c |
CuOAc |
Ag2CO3 |
48 % (2.8 : 1) |
|
6 |
2 c |
Cu(TC) |
Ag2CO3 |
45 % (1.5 : 1) |
|
7 |
2 c |
CuBr |
Ag2CO3 |
44 % (2 : 1) |
|
8 |
2 c |
CuI |
Ag2CO3 |
63 % (2 : 1)[d] |
|
9 |
2 d |
CuI |
Ag2CO3 |
8 % (2.8 : 1) |
|
10 |
2 e |
CuI |
Ag2CO3 |
63 % (2 : 1) |
|
11 |
2 f |
CuI |
Ag2CO3 |
18 % (1.8 : 1) |
|
12 |
2 c |
CuI |
Cs2CO3 |
20 % (1.3 : 1) |
|
13 |
2 c |
CuI |
AgOAc |
<5 % |
|
14 |
2 c |
CuI |
Ag2CO3 [e] |
<5 % |
Conditions: 1 (0.1 mmol), 2 (2 a–b: 0.1 mmol; 2 c–f: 0.15 mmol), catalyst (10 mol%) and additive (0.075 mmol, 0.75 equiv) in EA (0.5 mL) were stirred at 120 °C for 4 h. Yields and regioselectivity determined by GC analysis with n‐tetradecane as the internal standard. [a] 2.0 Equiv of Cs2CO3 was used. Conducted at 80 °C for 1 h. [b] Reaction time 1 h. [c] 10 mol% of 4,4′,5,5′‐tetrahydro‐2,2′‐bioxazole added. [d] Yield of isolated product and regioselectivity determined by 1H NMR spectroscopy. [e] 10 mol% of Ag2CO3 used. Cu(TC)=copper(I) thiophene‐2‐carboxylate.
With the optimized reaction conditions in hand, the scope was explored (Scheme 2). The N‐protecting group was varied first on amines having a δ‐methine hydrogen atom. We found that benzoyl groups bearing electron‐withdrawing para‐substituents such as bromide, fluoride and trifluoromethyl group showed higher yields and higher regioselectivity (3 b–3 d). Following this trend, a lower yield and higher regioselectivity were noted for the methoxy‐congener (3 e). Good yield and high regioselectivity were obtained for the heterocyclic benzofuran‐2‐carbonyl‐protected sulfonamide 1 f (73 %, 7.3 : 1 rr). Benzyloxycarbonyl‐ (Cbz) and 2,2,2‐trichloroethoxycarbonyl (Troc)‐protected amines provided the desaturated amides 3 g and 3 h in 57–59 % yield and lower regioselectivity. We then switched to α‐ and β‐substituted amines that could also be desaturated, as shown by the successful preparation of 3 i and 3 j. Desaturation worked also well for cyclic congeners where the methine C−H is part of the ring. For example, cyclohexenes 3 k–3 n were isolated in 55–81 % yield and good to excellent regioselectivity (5.2 : 1‐>19 : 1). As expected, regioselectivity was complete for desaturation at a benzylic methine site (see 3 p, 73 %). Reaction worked also for the desaturation of a cyclopentane ring as shown by the synthesis of 3 o (69 %, 8.8 : 1 rr). Considering the regiochemistry, it was found that in general deprotonation preferably occurred to give the thermodynamically more stable alkene and reaction of acyclic congeners preferentially led to γ‐/δ‐desaturated products, whereas δ‐/ϵ‐desaturation was noted for the cyclic substrates. Slight decrease of yield was observed when the reaction was performed on larger scale (3 a).
Scheme 2.
Remote desaturation of various amine derivatives. Reaction conditions: 1 (0.2‐0.4 mmol), MeSO2Cl (0.3‐0.6 mmol, 1.50 equiv), CuI (10 mol%) and Ag2CO3 (0.15‐0.30 mmol, 0.75 equiv) in EA (1.0‐2.0 mL) were stirred at 120 °C for 4–16 h. Yields given correspond to isolated yields. Regioselectivity was determined by 1H NMR spectroscopy. [a] Performed on 1.0 mmol scale. [b] 2‐Methylallylsulfonyl‐substituted benzamide was used. Dashed line indicates the double bond position in the minor isomer.
We were very pleased to find that dehydrogenation in cyclic systems could also be achieved at less activated methylene sites. Thus, the cyclohexylethylamine derivative 1 q, the cyclohexylmethylamine derivative 1 r and the cyclooctylamine derived sulfonamide 1 s engaged in the desaturation and 3 q–3 s were isolated in good yields (53‐60 %), albeit with low regioselectivity (1.1 : 1–1.9 : 1 rr). Unfortunately, the challenging desaturation at methylenic sites in non‐cyclic systems could not be achieved. For such substrates a complex product mixture was obtained (see the Supporting Information). Reductive amide product (desulfonylation) was noted for attempted desaturation at primary alkyl C(sp3)‐H bonds.
Functional group tolerance and late‐stage modification of drug derivatives including biologically more relevant compounds were studied next. Amides 1 t containing a piperidine moiety could be desaturated to give unsaturated 3 t in 88 % yield with 5.2 : 1 regioselectivity. Dehydrogenation at the δ‐/ϵ‐positions of amines derived from L‐homocyclohexyl alanine (3 u) and pregabalin (3 v) was achieved in 31–54 % yield and good regioselectivity (4 : 1–4.9 : 1 rr). Lower regioselectivity was obtained for the (S)‐(+)‐leucinol derived sulfonamide 1 w (see 3 w). Notably, the dehydroabietyl amine derivative 1 x, bearing two reactive benzylic sites and two δ‐C−H bonds at the positions C3 and C10, was regioselectively activated at C10 and desaturation provided 3 x in 87 % with 5.4 : 1 regioselectivity. Notably, the same amine substrate showed different reactivity with Baran's TEMPO/TfOH protocol, [7] underlining the complementarity of these two approaches.
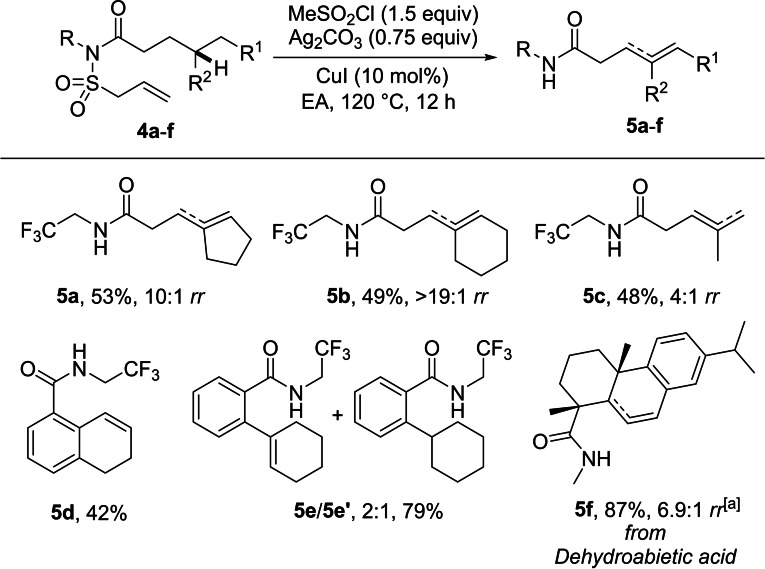
We next investigated whether the remote desaturation also works on carboxylic acid derivatives (Scheme 3). To this end, the amides 4 a–4 f were readily prepared by N‐acylation of N‐2,2,2‐trifluoroethyl‐ or N‐methyl‐allylsulfonamide (see the Supporting Information). To our delight, methine C−H activation and subsequent desaturation worked efficiently for simple carboxylic acid derivatives to give unsaturated amides 5 a–5 c in 48–53 % yield with 4 : 1‐>19 : 1 regioselectivity. The benzylic C−H bonds in benzoic acid derivatives 4 d and 4 e could be oxidized to give styrene type compounds in moderate yield. In the latter case, the inseparable by‐product 5 e′ formed by reduction of the intermediate amidyl radical was obtained in a significant amount (5 e:5 e′: 2 : 1). The dehydroabietic acid derived amide 4 f was also selectively desaturated to give 5 f in 87 % and good regioselectivity (6.9 : 1 rr), further documenting the synthetic value of the developed method.
Scheme 3.
Desaturation of various carboxylic acid derivatives. Reaction conditions: 1 (0.2–0.4 mmol), MeSO2Cl (0.3‐0.6 mmol, 1.50 equiv), CuI (10 mol%) and Ag2CO3 (0.15–0.3 mmol, 0.75 equiv) in EA (1.0–2.0 mL) were stirred at 120 °C for 12 h. Yields given correspond to isolated yields. Regioselectivity was determined by 1H NMR. [a] 1.2 Equiv. of 2 e was used instead of 2 c. Dashed line indicates the double bond position in the minor isomer.
To gain insights into the mechanism, several control experiments were conducted. In the presence of silver carbonate, the δ‐chlorinated amide 6 was quantitatively converted to the alkene 3 a which was formed with a 2 : 1 regioselectivity. Importantly, the regioselectivity is the same with that we obtained in the desaturation reaction of 1 a, indicating that both processes likely proceed through the same intermediate (Scheme 4a, Equation a). Moreover, the Ag‐mediated HCl‐elimination in 6 likely occurs via the corresponding tertiary cation. Both copper(I) iodide and silver carbonate were critical to this transformation, as significantly reduced yields were noted in the absence of either CuI or Ag2CO3 even upon extending reaction time to 16 h in both cases (Scheme 4a, Equation b). Moreover, in the absence of the silver salt, the δ‐chlorinated amide 6 was not detected by HRMS (Supporting Information), revealing that chlorine atom transfer from a Cu(II)Cl complex to the translocated tertiary C‐radical is unlikely. Hence, a cascade with such a Cu‐catalyzed Cl‐transfer followed by Ag‐mediated HCl elimination is not likely. In addition, this experiment also showed that the Cu(II)X species is not able to efficiently oxidize the translocated tertiary C‐radical, since alkene 3 a was identified in only 6 % yield. The generation of a cationic intermediate was further supported by the experiment run with the N‐allylsulfonamide 1 y, in which the pyrrolidine 7 was formed as the major product (Scheme 4a, Equation c).
Scheme 4.
Mechanistic investigations and proposed reaction mechanism.
On the basis of these experiments and previous reports,[ 14 , 15 , 16 , 17 ] a plausible mechanism for the radical remote desaturation of amides is proposed in Scheme 4b. Initially, a single electron transfer (SET) reduction (or chlorine abstraction) of MeSO2Cl with CuI leads to the methanesulfonyl radical along with a Cu(II)Cl complex.[ 16b , 16c , 16d ] Chloride anion exchange between Cu(II)Cl and Ag2CO3 leads to Cu(II)X and AgCl that precipitates, as experimentally observed after completion of the reaction. Subsequently, the methanesulfonyl radical could attack the allylsulfonyl moiety in 1 a to generate, after the fragmentation of A, the amidosulfonyl radical B. SO2 fragmentation and 1,5‐HAT leads to the translocated C‐centered radical D, which then gets oxidized by Ag2CO3 to the corresponding carbocation E along with an Ag(0) species. The alkene product 3 a is eventually generated by deprotonation of E that may be assisted by the N‐protecting group (Scheme 4b). The Cu‐catalysis cycle gets closed by the reduction of Cu(II)X with the Ag(0) species. In principle, the process should also be catalytic in Ag‐salt. However, the optimization studies revealed that 0.75 equivalents of Ag2CO3 are required in order to get a high conversion. This can be understood by the formation of AgCl‐precipitate which leads to consumption of the Ag‐salt.
In conclusion, we have developed a remote site‐controlled desaturation of unactivated C(sp3)‐H bonds in various amides derived from amines and carboxylic acids. Commercially available methanesulfonyl chloride was used as the reagent to mediate the radical cascade. Mechanistic studies revealed that both copper(I) iodide and silver carbonate are essential for this transformation. Silver carbonate not only acts as a chloride scavenger via the formation of insoluble AgCl in organic solution but also oxidizes the C‐centered radical to the corresponding carbocation. With our developed method, various desaturated homoallylic amine derivatives could be prepared in selected cases with excellent regioselectivity. Applications to the site‐selective desaturation at unactivated positions in drug derivatives and biologically more relevant compounds further document the synthetic value of this approach.
Conflict of interest
The authors declare no conflict of interest.
Supporting information
As a service to our authors and readers, this journal provides supporting information supplied by the authors. Such materials are peer reviewed and may be re‐organized for online delivery, but are not copy‐edited or typeset. Technical support issues arising from supporting information (other than missing files) should be addressed to the authors.
Supporting Information
Acknowledgements
This work was supported by the European Research Council (ERC Advanced Grant agreement No.692640). Open Access funding enabled and organized by Projekt DEAL.
Y. Xia, K. Jana, A. Studer, Chem. Eur. J. 2021, 27, 16621.
References
- 1. Zimmermann H., Walzl R., Ullman's Encyclopedia of Industrial Chemistry, Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2009. [Google Scholar]
- 2.
- 2a. Linstead R. P., Braude E. A., Mitchell P. W. D., Wooldridge K. R. H., Jackman L. M., Nature 1952, 169, 100–103; [Google Scholar]
- 2b. Pavlik J. W., Tantayanon S., J. Am. Chem. Soc. 1981, 103, 6755–6757; [Google Scholar]
- 2c. Shono T., Matsumura Y., Tsubata K., Sugihara Y., Yamane S., Kanazawa T., Aoki T., J. Am. Chem. Soc. 1982, 104, 6697–6703; [Google Scholar]
- 2d. Diao T., Stahl S. S., J. Am. Chem. Soc. 2011, 133, 14566–14569; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2e. West J. G., Huang D., Sorensen E. J., Nat. Commun. 2015, 6, 10093; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2f. Jie X., Shang Y., Zhang X., Su W., J. Am. Chem. Soc. 2016, 138, 5623–5633; [DOI] [PubMed] [Google Scholar]
- 2g. Golub T., Becker J. Y., Electrochim. Acta 2016, 205, 207–214; [Google Scholar]
- 2h. Shang Y., Jie X., Jonnada K., Zafar S. N., Su W., Nat. Commun. 2017, 8, 2273; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2i. West J. G., Sorensen E. J., Isr. J. Chem. 2017, 57, 259–269; [Google Scholar]
- 2j. Wang Z., He Z., Zhang L., Huang Y., J. Am. Chem. Soc. 2018, 140, 735–740; [DOI] [PubMed] [Google Scholar]
- 2k. Chen M., Rago A. J., Dong G., Angew. Chem. Int. Ed. 2018, 57, 16205–16209; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2018, 130, 16437–16441; [Google Scholar]
- 2l. Spieß P., Berger M., Kaiser D., Maulide N., J. Am. Chem. Soc. 2021, 143, 10524–10529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.
- 3a. Bloomfield D. K., Bloch K., J. Biol. Chem. 1960, 235, 337–345; [PubMed] [Google Scholar]
- 3b. Solomon E. I., Brunold T. C., Davis M. I., Kemsley J. N., Lee S.-K., Lehnert N., Neese F., Skulan A. J., Yang Y.-S., Zhou J., Chem. Rev. 2000, 100, 235–350. [DOI] [PubMed] [Google Scholar]
- 4.
- 4a. Buckle D. R., Pinto I. L., Comprehensive Organic Synthesis, Vol. 7 Eds.: Trost B. M., Fleming I., Pergamon, Oxford, 1991, 119; [Google Scholar]
- 4b. Muzart J., Eur. J. Org. Chem. 2010, 3779–3790; [Google Scholar]
- 4c. Iosub A. V., Stahl S. S., ACS Catal. 2016, 6, 8201–8213; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4d. Gnaim S., Vantourout J. C., Serpier F., Echeverria P.-G., Baran P. S., ACS Catal. 2021, 11, 883–892. [Google Scholar]
- 5. Breslow R., Baldwin S., Flechtner T., Kalicky P., Liu S., Washburn W., J. Am. Chem. Soc. 1973, 95, 3251–3262. [DOI] [PubMed] [Google Scholar]
- 6.
- 6a. Kochi J. K., J. Am. Chem. Soc. 1963, 85, 1958–1968; [Google Scholar]
- 6b. Acott B., Beckwith A. L. J., Aust. J. Chem. 1964, 17, 1342–1353; [Google Scholar]
- 6c. Čeković Ž., Dimitrijević L., Djokić G., Srnić T., Tetrahedron 1979, 35, 2021–2026. [Google Scholar]
- 7. Voica A.-F., Mendoza A., Gutekunst W. R., Fraga J. O., Baran P. S., Nat. Chem. 2012, 4, 629–635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.
- 8a. Parasram M., Chuentragool P., Sarkar D., Gevorgyan V., J. Am. Chem. Soc. 2016, 138, 6340–6343; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8b. Parasram M., Chuentragool P., Wang Y., Shi Y., Gevorgyan V., J. Am. Chem. Soc. 2017, 139, 14857–14860; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8c. Chuentragool P., Parasram M., Shi Y., Gevorgyan V., J. Am. Chem. Soc. 2018, 140, 2465–2468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.
- 9a. Hofmann A. W., Ber. Dtsch. Chem. Ges. 1883, 16, 558–560; [Google Scholar]
- 9b. Löffler K., Freytag C., Ber. Dtsch. Chem. Ges. 1909, 42, 3427; [Google Scholar]
- 9c. Wolff M. E., Chem. Rev. 1963, 63, 55–64; [Google Scholar]
- 9d. Zard S. Z., Chem. Soc. Rev. 2008, 37, 1603–1618; [DOI] [PubMed] [Google Scholar]
- 9e. Chiba S., Chen H., Org. Biomol. Chem. 2014, 12, 4051–4060; [DOI] [PubMed] [Google Scholar]
- 9f. Stateman L. M., Nakafuku K. M., Nagib D. A., Synthesis 2018, 50, 1569–1586; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9g. Jiang H., Studer A., CCS Chem. 2019, 1, 38–49; [Google Scholar]
- 9h. Kumar G., Pradhan S., Chatterjee I., Chem. Asian J. 2020, 15, 651–672; [DOI] [PubMed] [Google Scholar]
- 9i. Chen H., Yu S., Org. Biomol. Chem. 2020, 18, 4519–4532; [DOI] [PubMed] [Google Scholar]
- 9j. Wu X., Zhu C., CCS Chem. 2020, 2, 813–828; [Google Scholar]
- 9k. Nobile E., Castanheiro T., Besset T., Angew. Chem. Int. Ed. 2021, 60, 12170–12191; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2021, 133, 12278–12299; [Google Scholar]
- 9l. Guo W., Wang Q., Zhu J., Chem. Soc. Rev. 2021, 50, 7359–7377. [DOI] [PubMed] [Google Scholar]
- 10.For selected examples of recently reported 1,5-HAT to N-radicals, see:
- 10a. Choi G. J., Zhu Q., Miller D. C., Gu C. J., Knowles R. R., Nature 2016, 539, 268–271; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10b. Chu J. C. K., Rovis T., Nature 2016, 539, 272–275; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10c. Richers J., Heilmann M., Drees M., Tiefenbacher K., Org. Lett. 2016, 18, 6472–6475; [DOI] [PubMed] [Google Scholar]
- 10d. Wappes E. A., Fosu S. C., Chopko T. C., Nagib D. A., Angew. Chem. Int. Ed. 2016, 55, 9974–9978; [DOI] [PMC free article] [PubMed] [Google Scholar]; Angew. Chem. 2016, 128, 10128–10132; [Google Scholar]
- 10e. Becker P., Duhamel T., Stein J. C., Reiher M., Mũniz K., Angew. Chem. Int. Ed. 2017, 56, 8004–8008; [DOI] [PMC free article] [PubMed] [Google Scholar]; Angew. Chem. 2017, 129, 8117–8121; [Google Scholar]
- 10f. Yuan W., Zhou Z., Gong L., Meggers E., Chem. Commun. 2017, 53, 8964–8967; [DOI] [PubMed] [Google Scholar]
- 10g. Morcillo S. P., Dauncey E. M., Kim J. H., Douglas J. J., Sheikh N. S., Leonori D., Angew. Chem. Int. Ed. 2018, 57, 12945–12949; [DOI] [PMC free article] [PubMed] [Google Scholar]; Angew. Chem. 2018, 130, 13127–13131; [Google Scholar]
- 10h. Na C. G., Alexanian E. J., Angew. Chem. Int. Ed. 2018, 57, 13106–13109; [DOI] [PMC free article] [PubMed] [Google Scholar]; Angew. Chem. 2018, 130, 13290–13293; [Google Scholar]
- 10i. Hu X., Zhang G., Bu F., Nie L., Lei A., ACS Catal. 2018, 8, 9370–9375; [Google Scholar]
- 10j. Li Z., Wang Q., Zhu J., Angew. Chem. Int. Ed. 2018, 57, 13288–13292; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2018, 130, 13472–13476; [Google Scholar]
- 10k. Liu Z., Xiao H., Zhang B., Shen H., Zhu L., Li C., Angew. Chem. Int. Ed. 2019, 58, 2510–2513; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2019, 131, 2532–2535; [Google Scholar]
- 10l. Modak A., Pinter E. N., Cook S. P., J. Am. Chem. Soc. 2019, 141, 18405–18410; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10m. Tang N., Wu X., Zhu C., Chem. Sci. 2019, 10, 6915–6919; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10n. Zhang Z.-H., Dong X.-Y., Du X.-Y., Gu Q.-S., Li Z.-L., Liu X.-Y., Nat. Commun. 2019, 10, 5689; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10o. Zeng X., Yan W., Paeth M., Zacate S. B., Hong P.-H., Wang Y., Yang D., Yang K., Yan T., Song C., Cao Z., Cheng M.-J., Liu W., J. Am. Chem. Soc. 2019, 141, 19941–19949; [DOI] [PubMed] [Google Scholar]
- 10p. Qin Y., Han Y., Tang Y., Wei J., Yang M., Chem. Sci. 2020, 11, 1276–1282; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10q. Wang Q., Tian P., Cao Z.-Y., Zhang H., Jiang C., Adv. Synth. Catal. 2020, 362, 3851–3856; [Google Scholar]
- 10r. Guo Q., Peng Q., Chai H., Huo Y., Wang S., Xu Z., Nat. Commun. 2020, 11, 1463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.
- 11a. Liu T., Mei T.-S., Yu J.-Q., J. Am. Chem. Soc. 2015, 137, 5871–5878; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11b. Bao X., Wang Q., Zhu J., Nat. Commun. 2019, 10, 769; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11c. Prusinowski A. F., Twumasi R. K., Wappes E. A., Nagib D. A., J. Am. Chem. Soc. 2020, 142, 5429–5438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alternative routes not proceeding via N-radicals:
- 12a. Barluenga J., González-Bobes F., González J. M., Angew. Chem. Int. Ed. 2002, 41, 2556–2558; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2002, 114, 2668–2670; [Google Scholar]
- 12b. Yu P., Zheng S.-C., Yang N.-Y., Tan B., Liu X.-Y., Angew. Chem. Int. Ed. 2015, 54, 4041–4045; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2015, 127, 4113–4117. [Google Scholar]
- 13.During the submission, an elegant remote desaturation proceeding via N-radicals was reported, see: Jin W., Yu S., Org. Lett. 2021, 23, 6931–6935. [DOI] [PubMed] [Google Scholar]
- 14.
- 14a. Xia Y., Wang L., Studer A., Angew. Chem. Int. Ed. 2018, 57, 12940–12944; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2018, 130, 13122–13126; [Google Scholar]
- 14b. Wang L., Xia Y., Bergander K., Org. Lett. 2018, 20, 5817–5820; [DOI] [PubMed] [Google Scholar]
- 14c. Wu Z., Pratt D. A., Angew. Chem. Int. Ed. 2021, 60, 15598–15605; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2021, 133, 15726–15733; [Google Scholar]
- 14d. Wang L., Xia Y., Derdau V., Studer A., Angew. Chem. Int. Ed. 2021, 60, 18645–18650; [DOI] [PMC free article] [PubMed] [Google Scholar]; Angew. Chem. 2021, 133, 18793–18798. [Google Scholar]
- 15. Mourtrille C., Zard S. Z., Chem. Commun. 2004, 1848–1849. [DOI] [PubMed] [Google Scholar]
- 16.
- 16a. Zeng X., Ilies L., Nakamura E., J. Am. Chem. Soc. 2011, 133, 17638–17640; [DOI] [PubMed] [Google Scholar]
- 16b. Zhang Z., Tang X., Thomoson C. S., W. R. Dolbier, Jr. Org. Lett. 2015, 17, 3528–3531; [DOI] [PubMed] [Google Scholar]
- 16c. Lin J.-S., Wang F.-L., Dong X.-Y., He W.-W., Yuan Y., Chen S., Liu X.-Y., Nat. Commun. 2017, 8, 14841; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16d. Lia X.-F., Lin J.-S., Liu X.-Y., Synthesis 2017, 49, 4213–4220; [Google Scholar]
- 16e. dos P. Gomes G., Wimmer A., Smith J. M., König B., Alabugin I. V., J. Org. Chem. 2019, 84, 6232–6243. [DOI] [PubMed] [Google Scholar]
- 17.
- 17a. Oh S. H., Malpani Y. R., Ha N., Jung Y.-S., Han S. B., Org. Lett. 2014, 16, 1310–1313; [DOI] [PubMed] [Google Scholar]
- 17b. Bagal D. B., Kachkovskyi G., Knorn M., Rawner T., Bhanage B. M., Reiser O., Angew. Chem. Int. Ed. 2015, 54, 6999–7002; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2015, 127, 7105–7108; [Google Scholar]
- 17c. Hossain A., Bhattacharyya A., Reiser O., Science 2019, 364, eaav9713; [DOI] [PubMed] [Google Scholar]
- 17d. Engl S., Reiser O., Eur. J. Org. Chem. 2020, 1523–1533. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
As a service to our authors and readers, this journal provides supporting information supplied by the authors. Such materials are peer reviewed and may be re‐organized for online delivery, but are not copy‐edited or typeset. Technical support issues arising from supporting information (other than missing files) should be addressed to the authors.
Supporting Information