Abstract
Brain abscesses represent a pathology with significant morbidity and mortality. An underlying predisposing condition may not be apparent or identifiable in some instances. We present a patient with cerebral abscess who was found to have, previously undiagnosed, pulmonary arteriovenous malformations (PAVMs). PAVMs are rare pulmonary vascular anomalies resulting in intrapulmonary right to left shunt. These have been implicated in the development of brain abscesses. Conventional cultures from the lesion were non-revealing; hence, sample was sent for next-generation sequencing (NGS) which revealed multiple organisms, with predominance of Mycoplasma faucium, a bacterium initially felt to be a commensal of the oropharynx, but recently implicated as a human pathogen. This case, along with other documented associations between brain abscesses and pulmonary AVMs, highlights that brain abscess could be an initial clinical presentation in asymptomatic PAVMs. Additionally, novel testing such as NGS should be utilized in select settings where microbiological diagnosis can be elusive. This will help institute pathogen-directed specific antimicrobial therapy for favorable clinical outcomes.
Keywords: Mycoplasma faucium, Brain abscess, Pulmonary AV malformation
Introduction
The causative pathogens implicated in brain abscesses depend on the underlying predisposing condition. Most commonly isolated bacteria are Streptococcus and Staphylococcus species. Newer modalities of microbial identification may help delineate unusual causes of brain abscesses, while advanced imaging technologies may unfold an occult predisposing condition. We present a case of brain abscess associated with Mycoplasma faucium a patient with previously unknown pulmonary arteriovenous malformations (PAVMs).
Case presentation
A 43-year-old male with past medical history of hypertension presented to the Emergency Department with a severe headache that woke him from sleep. The headache subsided on its own, but the patient reported that he had never had a headache like that. He also reported associated nausea and emesis, but denied any other neurologic complaints or constitutional symptoms. He did report of intermittent episodes of epistaxis for many years, but denied history of hemoptysis. On physical examination, he was hemodynamically stable. Notably, he had poor dentition, but otherwise remainder of the exam was unremarkable. Initial labs were remarkable for a white cell count of 14,000/μL; rest of the cell counts, renal and liver function tests were within normal limits. A non-contrast CT scan of the head showed a left parieto-occipital hypodensity. The patient was admitted under the Neurosurgical team for further workup and management.
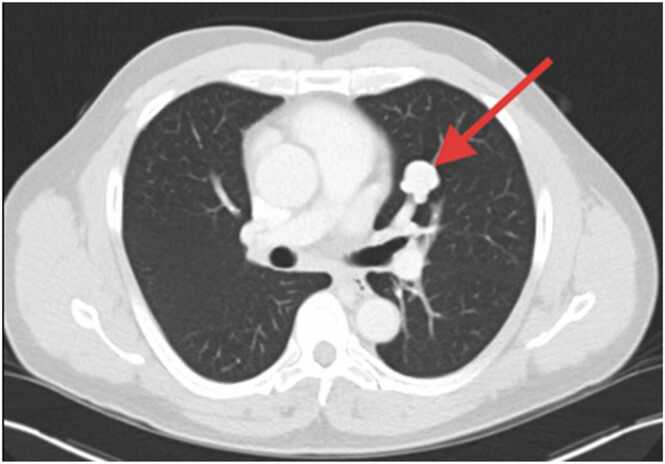
Once admitted, an MRI brain, with and without contrast was done, which was concerning for possible intracranial abscess that had decompressed into the left occipital horn with ventriculitis (Fig. 1). Empiric antimicrobial therapy with intravenous (IV) vancomycin and cefepime was commenced, a CT with intravenous contrast of the chest, abdomen, and pelvis was obtained, and Infectious Disease was consulted. The CT scan was remarkable for two pulmonary arteriovenous malformations (PAVMs) in the left upper lobe (Fig. 2). Antimicrobials were modified to IV ceftriaxone and metronidazole for suspected brain abscess. HIV screening was negative. Blood cultures remained negative. Given poor dentition, the source of infection was felt to be odontogenic. Conservative management with a planned 6-week course of empiric antimicrobials and interval imaging was adopted.
Fig. 1.
A peripherally enhancing lesion measuring approximately 1.2 by 1 cm by 1 cm with surrounding vasogenic edema.
Fig. 2.
A left upper lobe hypervasculature structure, measuring 1.8 by 1.8 cm, suggestive of an AV malformation.
Approximately one week after discharge, the patient had a repeat CT head which unfortunately showed that the abscess had increased in size. He was re-admitted and stereotactic biopsy of the lesion was performed. Pathology was consistent with chronic inflammation and an organizing abscess. Gram, fungal, AFB stain and Fite stains were negative. Bacterial, fungal, and AFB cultures remained negative. A fluid sample collected from his abscess was then sent for next-generation sequencing (NGS).
PAVMs were also considered, in addition to dental issues, as contributory factor for the development of brain abscess. To address this concern, coil embolization of the two AVMs by Interventional Radiology was performed.
NGS results returned, showing major abundance of Mycoplasma faucium. Additionally, in moderate abundance, were Fusobacterium nucleatum, Tannerella forsythia, Parvimonas species related to Parvimonas micra, Treponema species, Parvimonas micra, Porphyromonas gingivalis, Peptostreptococcus stomatis, and Campylobacter rectus. Following these results, the antimicrobial regimen was modified; doxycycline 100 mg twice daily was added to the regimen in order to target M. faucium.
After being on an antibiotic regimen including ceftriaxone, metronidazole, and doxycycline for close to 6 weeks, the patient underwent a repeat brain MRI. Imaging showed near complete resolution of the abscess with minimal residual enhancement (Fig. 3). At this point, antimicrobial therapy was stopped. Additionally, the patient sort care with dental surgery and his infected teeth were subsequently extracted. He was followed up 2 months after completion of antimicrobial therapy when he reported no complaints.
Fig. 3.
Follow up MRI imaging revealing resolution of the brain abscess.
Discussion
Generally, patients with brain abscess have an underlying condition implicated in the pathogenesis [1], [2]. Such predisposing factors typically include infection of a site adjacent to the brain (e.g., paranasal sinusitis, mastoiditis, chronic otitis media, dental infection), or hematogenous spread from a metastatic focus of infection (e.g., endocarditis, bacteremia) [1], [2]. A wide range of organisms have been found to cause brain abscesses. The causative pathogens implicated depend on the underlying predisposing condition. Streptococcus species and Staphylococcus species are overall most common bacterial causes of brain abscess and are often identified culprits when the infection develops via hematogenous spread. Dental and sinus infections tend to be polymicrobial, including anaerobes from the oral flora [1], [2]. Gram negative bacteria are often isolated following neurosurgical interventions and immunocompromised hosts are additionally vulnerable to infections by fungi [1], [2].
Pulmonary AVM is a condition in which pathologic right-to-left shunt is created by a structurally abnormal communication between the pulmonary artery and the pulmonary vein [3]. This condition can either be congenital or sporadic. The vast majority of congenital cases are secondary to hereditary hemorrhagic telangiectasia (HHT) [3]. Sporadic causes are much less common, and are typically secondary to causes such as trauma, prior chest surgery, malignancy, chronic infection (e.g., schistosomiasis, actinomycosis, tuberculosis), and mitral stenosis [3]. Many patients with pulmonary AVMs are asymptomatic and do not suffer from any complications; however, if left untreated, they can cause many life-threatening conditions such as embolic stroke, myocardial infarction, and, as seen in this case, brain abscess [4]. The hypothesis for increased risk for cerebral abscess is that with the right-to-left shunt created by pulmonary AVMs, bacteria in blood stream bypass the filtering effect of the pulmonary capillaries [4]. Normally, they would be removed by marginated neutrophils in the capillary circulation where the concentration of these cells is multifold compared to that in large blood vessels [4], [5]. In our patient with poor dental hygiene, the bacteria from oral flora that made their way into the blood stream were likely not efficiently cleared by the protective neutrophilic function.
The association between PAVMs and infections has been well reported in the literature. Cerebral infections are the most significant and well described in case reports and patient cohorts [6], [7], [8], [9]. In 2016, Nam et al. presented two very similar cases to ours in which the patients, each with an unremarkable past medical history, were admitted to the hospital for brain abscesses and were then found to have pulmonary AVMs on CT imaging [7]. In 2011, Mathis et al. reviewed 26 patients from 5 French medical centers with a concomitant diagnosis of HHT and cerebral abscess [10]. All of them were found to have pulmonary AVMs. It has also been reported that patients with these vascular anomalies are particularly at risk for recurrent brain abscesses if their AVMs are not treated [7], [11]. It is because of this association that it is imperative to treat the AVMs (typically with embolization) in patients found to have brain abscesses.
Interestingly, our case utilized NGS after the initial stains and cultures were negative. Results from the NGS not only lead to identification of a bacterium to target, but also suggested that our empiric antimicrobial therapy would likely not have been sufficient in treating the most predominant organism identified by this modality. With the use of NGS, we were able to isolate multiple organisms, suggesting polymicrobial infection, as has been described in patients with dental infections [1], [2]. Mycoplasma faucium was the most predominant bacterium isolated in our case, in addition to others including oral anaerobes, suggesting odontogenic focus of infection. M. faucium is a bacterium that is found in the human oropharynx, and has been isolated from as many as 25 % of human oral cavities [12]. It is a difficult organism to culture, and generally requires molecular sequencing to detect. While initially thought to be merely a commensal, M. faucium has been detected in brain abscesses of multiple patients, specifically in cases associated with polymicrobial abscesses and those associated with sinusitis or dental defects [13]. As Mycoplasma spp. lack cell wall, they are inherently resistant to beta-lactam antibiotics, which work by inhibiting cell wall biosynthesis. Instead, alternative antibiotics, such as tetracyclines, should be used. Our empiric choice of antimicrobials included ceftriaxone and metronidazole; these would have targeted the other bacteria identified by NGS, but ineffective against Mycoplasma. Hence, doxycycline was added to the antimicrobial regimen. The combination antimicrobial therapy resulted in a favorable response.
Overall, this case sheds light on the importance of recognizing pulmonary AVMs as a risk factor for cryptogenic brain abscesses. Once identified, pulmonary AVMs should be treated in order to address the underlying predisposing factor. In the current case, both dental care (including extraction) and embolization of AV malformations were done to minimize the risk of reinfection.
Sources of funding
No funding applicable to this article.
CRediT authorship contribution statement
All the authors have contributed to the writing of this manuscript.
Ethical approval
Not applicable. This is not a study on a patient or a volunteer.
Consent
Not applicable. We have ensured to not report any potential identifying information in the manuscript.
Conflict of interest
None of the authors have any potential conflict of interest to disclose.
Acknowledgements
We thank Karen Kasarda and Karen Ingham with assistance in obtaining NGS testing.
References
- 1.Brouwer M.C., Tunkel A.R., McKhann G.M., van de Beek D. Brain abscess. N Engl J Med. 2014;371:447–456. doi: 10.1056/nejmra1301635. [DOI] [PubMed] [Google Scholar]
- 2.Brouwer M.C., Coutinho J.M., van de Beek D. Clinical characteristics and outcome of brain abscess: systematic review and meta-analysis. Neurology. 2014;82:806–813. doi: 10.1212/wnl.0000000000000172. [DOI] [PubMed] [Google Scholar]
- 3.Gossage J.R., Kanj G. Pulmonary arteriovenous malformations. A state of the art review. Am J Respir Crit Care Med. 1998;158:643–661. doi: 10.1164/ajrccm.158.2.9711041. [DOI] [PubMed] [Google Scholar]
- 4.Meier N.M., Foster M.L., Battaile J.T. Hereditary hemorrhagic telangiectasia and pulmonary arteriovenous malformations: clinical aspects. Cardiovasc Diag Ther. 2018;8:316–324. doi: 10.21037/cdt.2017.12.07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hogg J.C., Doerschuk C.M. Leukocyte traffic in the lung. Annu Rev Physiol. 1995;57:97–114. doi: 10.1146/annurev.ph.57.030195.000525. [DOI] [PubMed] [Google Scholar]
- 6.Dupuis-Girod S., Giraud S., Decullier E., Lesca G., Cottin V., Faure F., et al. Hemorrhagic hereditary telangiectasia (Rendu-Osler disease) and infectious diseases: an underestimated association. Clin Infect Dis. 2007;44:841–845. doi: 10.1086/511645. [DOI] [PubMed] [Google Scholar]
- 7.Nam T.-K., Park Y.-sook, Kwon J.-taik. Brain abscesses associated with asymptomatic pulmonary arteriovenous fistulas. J Korean Neurosurg Soc. 2017;60:118–124. doi: 10.3340/jkns.2015.0707.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li C., Hao S., Wang J., Gao Z., Ji N. Severe cerebral abscess associated with pulmonary arteriovenous fistula: case report and literature review. Chin Neurosurg J. 2018;4:30. doi: 10.1186/s41016-018-0137-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Moradi M., Adeli M. Brain abscess as the first manifestation of pulmonary arteriovenous malformation: a case report. Adv Biomed Res. 2014;3:28. doi: 10.4103/2277-9175.124677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mathis S., Dupuis-Girod S., Plauchu H., et al. Cerebral abscesses in hereditary haemorrhagic telangiectasia: a clinical and microbiological evaluation. Clin Neurol Neurosurg. 2012;114:235–240. doi: 10.1016/j.clineuro.2011.10.036. [DOI] [PubMed] [Google Scholar]
- 11.Kaido T., Moriyama Y., Ueda K., et al. Recurrent brain abscess induced by pulmonary arteriovenous fistula. J Infect Chemother. 2011;17:552–554. doi: 10.1007/s10156-011-0210-9. [DOI] [PubMed] [Google Scholar]
- 12.Edouard S., Courtois G.D., Gautret P., et al. High prevalence of Mycoplasma faucium DNA in the human oropharynx. J Clin Microbiol. 2016;54:194–196. doi: 10.1128/jcm.02068-15. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 13.Al Masalma M., Lonjon M., Richet H., et al. Metagenomic analysis of brain abscesses identifies specific bacterial associations. Clin Infect Dis. 2011;54:202–210. doi: 10.1093/cid/cir797. [DOI] [PubMed] [Google Scholar]