ABSTRACT.
It is currently not clear whether humoral immunity to Zika virus (ZIKV) elicited upon natural ZIKV infection is long-lasting. In addition, cross-reactivity of anti-ZIKV antibodies with antigenically related dengue viruses (DENV) may have biological implications in nonnaive individuals who subsequently acquire a heterotypic infection. Cross-reactive humoral immunity between ZIKV and DENV also complicates the interpretation of serological tests to evaluate previous exposure to either virus. Here, we have measured the 2-year decay of ZIKV neutralizing antibodies in people living in a ZIKV/DENV endemic area in Brazil who were identified as having an acute infection (group 1) or past (but recent) infection (group 2) with ZIKV in 2015–16. The titers of neutralizing antibodies to ZIKV decreased 9.1 and 2.3 times in groups 1 and 2, respectively. We also show that the plaque reduction neutralization assay (PRNT) is a reliable method to measure past exposure to ZIKV in coendemic areas.
The durability of humoral immunity to Zika virus (ZIKV) is of special interest in regions where the virus continuously circulates and antigenically related dengue viruses (DENV) cocirculate. Anti-ZIKV antibody type (e.g., binding, neutralizing) and titer may be associated with limited or absent Zika disease upon subsequent infections with ZIKV and with protection or enhancement of future heterotypic infections with DENV.1 Recent works have indicated that antibody titers to ZIKV wane more rapidly than the expected,2–5 putting into question the role of anti-ZIKV humoral immunity in future infections with ZIKV and DENV and the validity of ZIKV serological assays to evaluate previous exposure to the virus.
In 2015–16, after the introduction of ZIKV in the Americas, we recruited 263 people presenting with acute symptoms compatible with arboviral infection in Pernambuco State (PE), northeast Brazil, during an ongoing ZIKV epidemic in the region.6 Blood samples were collected at enrollment and at 14 days (mean; min-max 10–37) after the onset of symptoms (“early convalescent sample”) and tested by molecular and serological tests. The latter included plaque reduction neutralization assays resulting in 50% of plaque reduction (PRNT50) to measure ZIKV neutralizing antibodies (nAbs). Based on the assays results and the fact that ZIKV had been introduced in the region in the previous few months,7 42 participants were identified as having an acute infection with ZIKV (positive ZIKV qRTPCR at enrollment or positive ZIKV IgM at enrollment combined with a PRNT50 convalescent/acute titer ratio > 4), and 58 as having a past/recent infection with the virus (ZIKV nAb PRNT50 titers ≥ 100 in both acute and early convalescent samples and a PRNT50 convalescent/acute titer ratio < 2.5). In 2017, 174 of the original participants were enrolled in a second study, along with their household members, to compare the seroprevalence to ZIKV and chikungunya virus (CHIKV) within households to better understand the risk of sexual ZIKV transmission8; in this latter work, a third blood sample was also collected from the participants (“late sample”). Here, we titrated ZIKV nAbs as described previously8 from the early convalescent sample taken in 2015–16 and the late sample taken in 2017 to evaluate changes in PRNT50 titers over the course of 2 years (mean: 2.0; interquartile range: 1.8–2.2). According to official reports from the Ministry of Health of Brazil and relevant to this work, ZIKV circulation in PE, Brazil, was extremely low after the 2015–16 epidemic9,10; thus, the probability that the participants were exposed to the virus after the 2015–16 recruitment and until the 2017 study was low. The circulation of DENV was also unexpectedly low during this time period.9,10
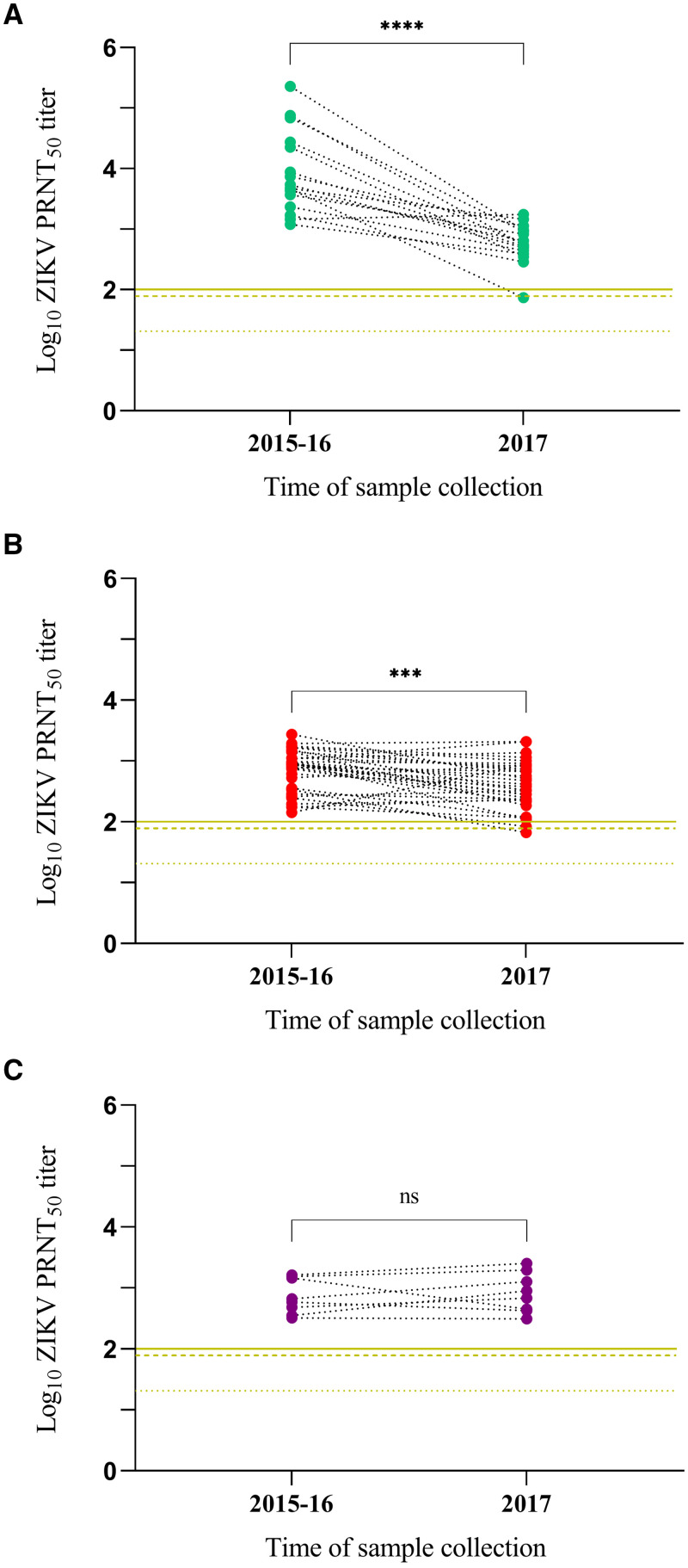
Sixteen of the 42 ZIKV acutely infected people at enrollment in 2015–16 were successfully re-recruited in 2017 (all were adults ≥ 18 years in 2015–16). In this group, the mean ZIKV PRNT50 titers decreased 9.1-fold (from 5032.5 to 553.3; paired t test P < 0.0001) over the time period (Figure 1A). Among participants with a past/recent ZIKV infection at enrollment in 2015–16, 33 of 58 people were successfully re-recruited in 2017. The 2-year decrease in the mean ZIKV PRNT50 titers in this group was 2.3-fold (from 870.4 to 377.3; paired t test’s P = 0.0004) (Figure 1B). It has been shown that the peak of ZIKV nAbs occurs at early convalescence,11 thus the sharper 2-year decrease of nAb titers observed in the first group relative to the second group may be explained by the fact that the early convalescent sample from these participants most likely captured the peak phase of ZIKV nAbs after infection. In addition, these participants had symptomatic Zika, which might also have influenced nAb production, whereas participants in the second group, while presenting with arboviral disease symptoms, could only be determined to have been infected with ZIKV before their enrollment (i.e., the acute infection they were presenting with at enrollment was likely not ZIKV).
Figure 1.
Zika virus (ZIKV) neutralizing antibody titers in 2015–16 and 2017 from acutely infected participants or those with a past/recent ZIKV infection in 2015–16. Titers are shown as the reciprocal dilution of serum (log10-transformed) inhibiting 50% of ZIKV plaque formation (PRNT50). (A) Titers from adults (≥ 18 years of age) acutely infected with ZIKV in 2015–16. (B) Titers from adults who had a past/recent ZIKV infection in 2015–16. (C) Titers from adolescents/children (≤ 16 years of age) who had a past/recent ZIKV infection in 2015–16. Lines going through the Y axis represent three cutoff values (≥ 2 log10 or ≥ 100 [solid], ≥ 1.8 log10 or ≥ 80 [dashed] and ≥ 1.3 log10 or ≥ 20 [dotted]). Asterisks represent the significance (P value) of PRNT50 titer difference between the two time points, calculated through paired t tests in GraphPad Prism 9.3.1. ****P < 0.0001. ***P = 0.0004. ns = nonsignificant or > 0.05.
A third group included eight people who were ≤ 16 years of age and had a past/recent infection with ZIKV at enrollment in 2015–16; their mean ZIKV PRNT50 titers increased 1.3-fold (from 625.8 to 789.2; paired t test’s P > 0.05) over the 2 years (Figure 1C). Although there were few of these participants and so we must be cautious in our interpretations, this observation is in accordance with the data reported by Henderson et al.3 in Fiji, where there were no changes in ZIKV nAb titers over 2 years in people ≤ 16 years of age, contrasting with the titer decrease observed in the adult population.
It has been hypothesized that upon infection with ZIKV, previous exposure to DENV would lead to a less specific, and thus less durable, anti-ZIKV immunity.3,4 If this turns out to be correct, the insignificant change in ZIKV nAbs among the youth could in part be due to their lower chance of having been exposed to DENV before being infected with ZIKV relative to adults. Although the number of participants in our study does not allow us to perform in-depth, age-stratified analysis on the association of previous exposure to DENV and the decrease level of ZIKV nAbs over 2 years, we went back to the DENV PRNTs performed previously with the acute and early convalescent samples collected in 2015–166 to evaluate any specific profile of DENV immunity among participants. We saw that most participants (50/57; 87.7%) had relatively high titers to at least one DENV serotype (Table 1). We also noticed that five of seven (71.4%) participants naive for DENV nAbs in 2015–16 had increased ZIKV nAb titers in 2017 (Table 1); however, nine of 14 (64.3%) of those with increased ZIKV nAbs in 2017 had nAbs to one or more DENV serotypes in 2015–16. Thus, at this point, no conclusions can be drawn about the effect of previous DENV exposure on ZIKV nAb decay, but we believe further studies on the matter are warranted.
Table 1.
Fold decrease of participant’s neutralizing antibodies against Zika virus (ZIKV) over 2 years and their initial titers of neutralizing antibodies against the four dengue viruses (DENV1-4)
| ZIKV infection status in 2015–16 according to PRNT50 results | Fold-difference of ZIKV PRNT50 titers from 2015–16 to 2017*† | DENV1 PRNT50‡ | DENV2 PRNT50 | DENV3 PRNT50 | DENV4 PRNT50 |
|---|---|---|---|---|---|
| Acute ZIKV infection§ | −0.69 | 66.98 | 44.94 | 1,519.00 | 7,580.00 |
| −0.91 | 26.81 | 1,451.00 | 1,516.00 | ||
| −0.85 | – | – | – | – | |
| −0.99 | – | – | 1,710.00 | 812.60 | |
| −0.98 | 613.90 | 128.10 | 1,395.00 | 965.30 | |
| −0.94 | – | – | 140.50 | 1,321.00 | |
| −0.78 | – | 54.78 | 215.20 | 653.10 | |
| −0.83 | – | – | 1,455.00 | 698.10 | |
| −0.98 | – | – | 272.10 | 1,369.00 | |
| −1.00 | – | – | 272.10 | 1,369.00 | |
| −0.99 | 64.25 | – | 217.40 | 2,944.00 | |
| +0.20 | – | – | – | – | |
| −0.98 | – | – | 21,330.00 | 493.40 | |
| −0.67 | – | – | 653.80 | 1,520.00 | |
| −0.81 | – | 92.04 | 1,004.00 | 3,251.00 | |
| −0.85 | – | – | 1,327.00 | 858.10 | |
| Past/recent ZIKV infection‖ | +0.23 | – | – | 100,436.00 | 1,906.00 |
| −0.71 | – | – | 792.30 | 1,016.00 | |
| −0.89 | – | – | 1,968.00 | 1,046.00 | |
| +0.16 | – | – | 841.10 | 1,511.00 | |
| −0.66 | – | – | 602.00 | 799.30 | |
| +5.26 | – | – | – | 314.20 | |
| +0.24 | – | – | – | – | |
| −0.66 | – | – | 292.70 | 705.80 | |
| −0.79 | – | – | 1,167.00 | 2,070.00 | |
| −0.50 | – | – | 787.10 | 648.90 | |
| −0.69 | 352.50 | 60.22 | 284.40 | 1,268.00 | |
| +0.35 | – | – | 116.60 | 613.80 | |
| −0.28 | – | – | 709.30 | 167.70 | |
| −0.48 | – | – | 686.60 | 361.80 | |
| −0.68 | – | – | 437.40 | 801.40 | |
| −0.60 | – | – | 530.00 | 341.30 | |
| −0.68 | – | – | 221.60 | 221.70 | |
| −0.49 | 268.70 | 328.20 | 384.20 | 477.20 | |
| −0.64 | – | – | 558.80 | 1,533.00 | |
| −0.33 | 55.02 | 162.20 | 155.10 | ||
| −0.34 | – | – | 1,192.00 | 877.40 | |
| +0.92 | – | – | – | 662.10 | |
| −0.82 | – | – | 922.30 | 673.20 | |
| +0.07 | 4455 | 1409 | 521.30 | 979.60 | |
| −0.35 | – | – | 2,547.00 | 628.20 | |
| −0.80 | – | – | 303.30 | 276.10 | |
| −0.06 | – | – | – | – | |
| −0.87 | – | – | 300.00 | 246.20 | |
| −0.15 | – | – | 528.00 | 580.70 | |
| +0.65 | – | – | 127.40 | 1,550.00 | |
| −0.38 | – | – | 1,053.00 | 312.90 | |
| −0.18 | – | – | 1,457.00 | 1,095.00 | |
| −0.40 | – | – | 428.70 | 334.10 | |
| Past/recent ZIKV infection¶ | +0.27 | – | – | – | – |
| +0.90 | – | – | 846.00 | 337.70 | |
| −0.04 | – | – | 129.30 | 658.30 | |
| +0.41 | – | – | – | 314.90 | |
| +0.55 | – | – | – | – | |
| −0.28 | – | – | – | 127.80 | |
| +1.54 | – | – | – | – | |
| −0.69 | – | – | 1,423.00 | 782.90 |
Bold indicates people who were naive for DENV neutralizing antibodies (nAbs) in 2015–16.
Fold-difference of Zika virus plaque reduction neutralization tests resulting in 50% of plaque formation inhibition (ZIKV PRNT50) in samples from 2017 compared with samples from 2015–16.
Positive values indicate that the titer increased whereas negative values indicates that the titers decreased in 2017 compared with 2015–16.
Dengue virus plaque reduction neutralization tests resulting in 50% of plaque formation inhibition (DENV PRNT50) were performed with the early convalescent sample collected in 2015–16. DENV PRNTs were performed separately for each serotype (DENV1-4).
Mean age in 2015–16 (min–max): §30 (20–53), ‖39 (17–69), and ¶11.5 (9–16).
The conservative ZIKV PRNT50 cutoff used in our previous studies (≥ 100) as well as the choice of using PRNT50 over PRNT90 was based on previous validation experiments using samples from DENV endemic regions in Brazil, such as the one where our work was conducted.12 If we were to use this ZIKV PRNT50 cutoff to evaluate previous exposure to ZIKV in the same population 2 years after, 6.3% (1 of 16) and 4.9% (2 of 41) of the participants with an acute or a past/recent ZIKV infection in 2015–16, respectively, would be considered negative. Using a slightly lower cutoff (≥ 80), the same number of participants would be considered negative 2 years later. Under a much less conservative PRNT50 cutoff (≥ 20) no participant would be considered negative for ZIKV nAbs when tested over the time period (Figures 1A–C). Thus, PRNTs seem to be a valuable assay to evaluate ZIKV seroprevalence a few years after outbreaks in endemic regions; however, cutoff values might need to be adjusted according to the local fold-decrease in ZIKV nAbs.
Langerak et al.4 found that among 49 people with symptomatic disease and a confirmed ZIKV-RTPCR in Suriname, only 59.2% had positive ZIKV nAb titers and approximately 10% of them had seroreverted (had no detectable ZIKV nAbs) after 3.2 years. In Fiji, a significant decrease in ZIKV nAb levels was observed within 2 years among 45 people, with four of them seroreverting. Although the differences among these studies and ours make it difficult to directly compare data, indications that ZIKV nAb titers significantly wane in adults over time are becoming stronger. It would be important to continue following these cohorts, including ours, for further assessments of ZIKV humoral immunity over time and the factors influencing it and also to evaluate the prevalence and pathogenesis of further infections with ZIKV and DENV. We believe it will also be critical to assess anti-ZIKV immunity associated with the infection route in further studies, given that the virus can be transmitted through mosquito bites or through sex13,14 and that each mode of infection may lead to different infection and immunity courses.
ACKNOWLEDGMENTS
We thank the Paulista-PE participants, the field teams, and the technicians from the 2015–16 and 2017 studies.
REFERENCES
- 1. Langerak T Mumtaz N Tolk VI van Gorp ECM Martina BE Rockx B Koopmans MPG , 2019. The possible role of cross-reactive dengue virus antibodies in Zika virus pathogenesis. PLoS Pathog 15: e1007640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Aubry M et al. 2017. Zika virus seroprevalence, French Polynesia, 2014–2015. Emerg Infect Dis 23: 669–672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Henderson AD et al. 2020. Zika seroprevalence declines and neutralizing antibodies wane in adults following outbreaks in French Polynesia and Fiji. eLife 9: e48460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Langerak T et al. 2021. Zika virus antibody titers three years after confirmed infection. Viruses 13: 1345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Moreira-Soto A de Souza Sampaio G Pedroso C Postigo-Hidalgo I Berneck BS Ulbert S Brites C Netto EM Drexler JF , 2020. Rapid decline of Zika virus NS1 antigen-specific antibody responses, northeastern Brazil. Virus Genes 56: 632–637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Magalhaes T et al. 2017. Zika virus displacement by a chikungunya outbreak in Recife, Brazil. PLoS Negl Trop Dis 11: e0006055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Brito CA Brito CC Oliveira AC Rocha M Atanasio C Asfora C Matos JD Lima AS Albuquerque MF , 2016. Zika in Pernambuco: rewriting the first outbreak. Rev Soc Bras Med Trop 49: 553–558. [DOI] [PubMed] [Google Scholar]
- 8. Magalhaes T et al. 2021. Follow-up household serosurvey in northeast Brazil for Zika virus: sexual contacts of index patients have the highest risk for seropositivity. J Infect Dis 223: 673–685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Ministério da Saúde , 2017. Monitoramento dos casos de dengue, febre de chikungunya e febre pelo vírus Zika até a Semana Epidemiológica 52, 2016. Secretaria de Vigilância em Saúde, ed. Brasília: Ministério da Saúde, 11.
- 10. Ministério da Saúde , 2019. Monitoramento dos casos de dengue, febre de chikungunya e doença aguda pelo vírus Zika até a Semana Epidemiológica 50 de 2018. Secretaria de Vigilância em Saúde, ed. Brasília: Ministério da Saúde, 14.
- 11. Montoya M et al. 2018. Longitudinal analysis of antibody cross-neutralization following Zika virus and eengue virus infection in Asia and the Americas. J Infect Dis 218: 536–545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Castanha PM Cordeiro MT Martelli CM Souza WV Marques ET Jr Braga C , 2013. Force of infection of dengue serotypes in a population-based study in the northeast of Brazil. Epidemiol Infect 141: 1080–1088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Blitvich BJ Magalhaes T Laredo-Tiscareno SV Foy BD , 2020. Sexual transmission of arboviruses: a systematic review. Viruses 12: 933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Foy BD Kobylinski KC Chilson Foy JL Blitvich BJ Travassos da Rosa A Haddow AD Lanciotti RS Tesh RB , 2011. Probable non-vector-borne transmission of Zika virus, Colorado, USA. Emerg Infect Dis 17: 880–882. [DOI] [PMC free article] [PubMed] [Google Scholar]