Abstract
Background
There are few reports on kidney complications after severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) messenger RNA (mRNA) vaccination, especially in the pediatric population. We report a pediatric case diagnosed with crescentic glomerulonephritis (CrGN) after the second dose of the SARS-CoV-2 mRNA vaccine.
Case-diagnosis/treatment
A 16-year-old girl was admitted due to dyspnea and headache approximately 6 weeks after receiving the second SARS-CoV-2 mRNA vaccine (Pfizer-BioNTech). She had previously experienced fever, nausea, vomiting, and dyspnea after the first vaccination, which persisted for a week. On admission, her blood pressure was 155/89 mmHg with a 7 kg weight gain in a month. She had microhematuria and proteinuria. Laboratory findings were as follows: blood urea nitrogen/creatinine, 66/9.57 mg/dL; and brain natriuretic peptide, 1,167 pg/mL. Anti-neutrophil cytoplasmic antibody (ANCA), anti-glomerular basement membrane (GBM) antibody, and antinuclear antibody findings were negative. Kidney doppler sonography revealed swelling and increased echogenicity of both kidneys with increased resistive index. Cardiac magnetic resonance imaging results showed early minimal fibrosis of myocarditis. We then started hemodialysis. Kidney biopsy showed diffuse extra capillary proliferative glomerulonephritis with diffuse crescent formation. We treated the patient with methylprednisolone pulse therapy with subsequent oral steroids and mycophenolate mofetil. Although dialysis was terminated, the patient remained in the chronic kidney disease stage.
Conclusions
This is the first case of ANCA-negative CrGN after SARS-CoV-2 mRNA vaccination in the pediatric population. As children are increasingly vaccinated with SARS-CoV-2 mRNA vaccines, monitoring for kidney complications is warranted.
Keywords: Crescentic glomerulonephritis, SARS-CoV-2 mRNA vaccine, mRNA vaccine, SARS-CoV_2, COVID-19 vaccines
Introduction
The World Health Organization (WHO) guidelines approved the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) messenger RNA (mRNA) vaccine for adolescents aged 12 years or older and recommended vaccination of children over the age of 5 with underlying diseases. Considering the worldwide spread of SARS-CoV-2, the age limit of SARS-CoV-2 vaccination has been decreased. Vaccination has reduced the number of complications caused by severe infections. However, concerns remain about the side effects of vaccines, especially among pediatric populations. A few case reports have been published on acute kidney complications following vaccination in adults, including IgA nephropathy and minimal change disease. However, no studies have been conducted on kidney disease in vaccinated children. Here, we describe a pediatric case of crescentic glomerulonephritis (CrGN) presenting as rapidly progressive glomerulonephritis (RPGN) following the second dose of the SARS-CoV-2 mRNA vaccine (Pfizer-BioNTech).
Case report
A 16-year-old girl was hospitalized due to dyspnea and headache. She had no medical disease history, including kidney disease. Her previous reports from annual school screenings showed that routine urine analysis and blood pressure were normal. She received her first dose of the SARS-CoV-2 mRNA vaccine (Pfizer-BioNTech) 9 weeks before hospitalization without severe complications other than a mild fever and pain at the injection site. She received the second dose of the vaccine about 6 weeks prior to hospitalization. She experienced fever, nausea, vomiting, and dyspnea on exertion after the second vaccination, which persisted for a week. A right temporal headache developed 2 weeks after the second vaccination. Afterward, she continued to suffer from dyspnea and headache.
She was transferred to our hospital since abnormal kidney function and increased cardiac enzymes were detected during a diagnostic workup in a local hospital for persistent dyspnea and headache. Her blood pressure on admission was 155/89 mmHg. Her weight had increased by 7 kg in a month due to edema. She had hematuria and proteinuria (urinary protein/creatinine ratio 9.1 mg/mg). The laboratory findings were blood urea nitrogen (BUN)/creatinine (Cr): 66/9.57 mg/dL; creatine kinase (CK)/lactate dehydrogenase (LD): 410/320 IU/L; brain natriuretic peptide (BNP): 1167 pg/mL; troponin I: 38.08 pg/mL; myoglobin: 738 ng/mL; C3: 126.2 mg/dL (reference: 82–170 mg/dL); and C4: 61.5 mg/dl (reference: 12–36 mg/dL). Anti-neutrophil cytoplasmic antibody (ANCA), anti-glomerular basement membrane (GBM) antibodies, anti-PLA2R IgG, and antinuclear antibody were negative. The result of echocardiography was grade I mitral regurgitation with normal cardiac function. Renal Doppler sonography revealed swelling and increased echogenicity of both kidneys with increased resistive index (Rt: 0.75–0.85, Lt: 0.78–0.82). At follow-up, the troponin I levels had increased to 459.12 pg/mL. Cardiac magnetic resonance imaging performed on the thirteenth day of hospital stay resulted in suspected early minimal fibrosis of myocarditis. No cardiac antibody test was performed.
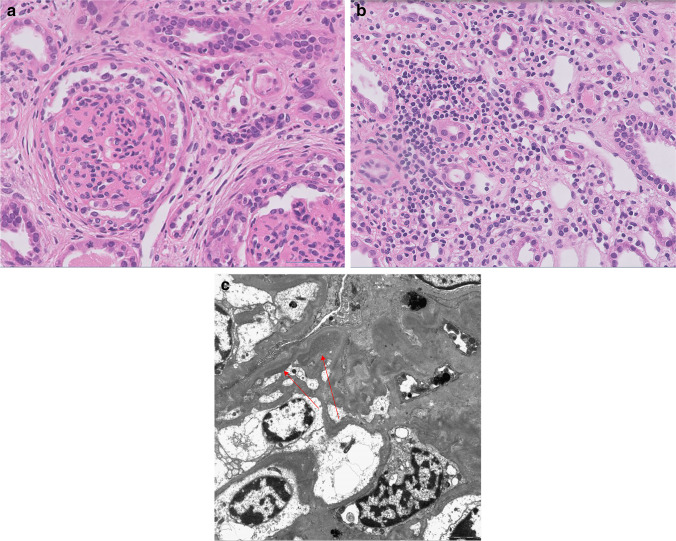
As her kidney failure progressed with increased serum creatinine up to 12.7 mg/dL, we started hemodialysis on the eighth hospital day. We performed a kidney biopsy (Fig. 1), and the results were diffuse extra capillary proliferative glomerulonephritis with diffuse crescent formation favoring pauci-immune glomerulonephritis. Thirteen out of 22 glomeruli (59%) showed diffuse crescent formation (10 fibrocellular, 3 fibrous), and the other 9 were sclerotic (2 focal, 7 global). There was severe interstitial inflammatory cell infiltration (mainly mononuclear cells), edema, and minimal fibrosis. The tubules revealed diffuse dilatation, attenuation, loss of brush border, and detachment of epithelial cells. Immunofluorescence revealed diffuse peripheral IgM (trace), IgA (1 +), C3 (1 +), C4 (trace), and C1q (trace) at glomerulus. Three glomeruli (one globally sclerotic glomerulus and two segmentally sclerotic glomeruli) were examined by electron microscopy, showing a small amount of electron-dense deposits with diffuse foot process effacement.
Fig. 1.
Pathology. a Light microscopy, global sclerosis of glomeruli (H&E, ×400). b Interstitial inflammatory cell infiltration with mild fibrosis (H&E, ×400). c Transmission electron microscopy (arrows point to electron dense deposits)
We treated the patient with methylprednisolone pulse therapy (1 g for 3 days), subsequent oral steroids, mycophenolate mofetil (MMF), and an angiotensin-converting enzyme inhibitor. We terminated dialysis after 2 weeks. Her blood pressure, BUN/Cr, and urine protein/Cr levels were 135/87 mmHg, 61/4.46 mg/dL, and 8.0, respectively, at 3 months of follow-up while maintaining mycophenolate mofetil and slowly tapering oral steroids.
Discussion
To our knowledge, this is the first pediatric case of CrGN after a SARS-CoV-2 mRNA vaccination. Pathologically, CrGN is defined as glomerular crescents greater than 50%. There has been one pediatric case of IgA nephropathy after SARS-CoV-2 mRNA vaccination that showed cellular glomerular crescents, but it is unclear if this report relates to CrGN because it did not describe the proportion of crescentic glomeruli [1]. There have only been a few reports on de novo glomerular disease after the vaccination. Among these, eight cases of CrGN have been reported [2–8].
Five were diagnosed as ANCA-associated glomerulonephritis (GN), two as anti-GBM nephritis, and one as IgA nephropathy. They were all adults, and their age range was 39–81 years. The male-to-female ratio was 3:2 in ANCA-associated GN. The patients in the two cases with anti-GBM nephritis were females, and the one case of IgA nephropathy was male. The patient in our case report was a 16-year-old girl without ANCA or anti-GBM antibodies. In our case, the possibility of diagnosing IgA nephropathy was unclear because the immunofluorescence study on the biopsy specimen was done in sclerosed glomeruli. The diagnosis might be ANCA-negative pauci-immune vasculitis with only kidney involvement. As in our case, all patients in the previous studies manifested symptoms after the second dose of the vaccine. All patients in the earlier studies were diagnosed within 14 days after vaccination, whereas our case was diagnosed 6 weeks after vaccination. Since her uremic symptoms began 2 weeks after the second vaccination dose, kidney disease may already have been present before diagnosis. Patients in the previous reports were initially treated with high-dose steroids and cyclophosphamide. Among these, plasmapheresis was performed in four cases, and rituximab was administered in three. All patients in these studies seemed to improve in their glomerular filtration rate (GFR) after treatment, although achieving normal GFR has not been clearly described in these reports except for one case with anti-GBM nephritis and consistent hemodialysis. Our case also showed a decrease in serum creatinine level after treatment with methyl-prednisolone pulse therapy and MMF. We did not expect a good prognosis in our patient because all the glomeruli were crescentic or sclerotic on biopsy. We chose MMF for treatment instead of cyclophosphamide, considering its toxicity.
The pathophysiology of CrGN development after SARS-CoV-2 vaccination is unclear. One hypothesis proposes that vaccine-induced autoimmunity causes subsequent glomerulonephritis after mRNA vaccination [9]. Antibodies for SARS-CoV-2 spike protein may elicit an immune response to several self-proteins, including kidney components. Heymans et al. suggested a similar immune mechanism of myocarditis after SARS-CoV-2 mRNA vaccination: the SARS-CoV-2 mRNA vaccine induces antibodies against the SARS-CoV-2 spike glycoproteins which may cross-react with the myocardial contractile proteins [10]. In our case, the patient seemed to have preceded SARS-CoV-2 mRNA vaccine-associated myocarditis together with RPGN.
In conclusion, CrGN may be associated with the SARS-CoV-2 mRNA vaccine, especially after the second dose. However, we recommend proceeding with SARS-CoV-2 vaccinations along with other available vaccinations since the incidence of RPGN after SARS-CoV-2 mRNA vaccination is low. Early CrGN diagnosis with a kidney biopsy and treatment with immunosuppressants may be essential to improve kidney outcomes.
Declarations
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Hanna C, Herrera Hernandez LP, Bu L, Kizilbash S, Najera L, Rheault MN, Czyzyk J, Kouri AM. IgA nephropathy presenting as macroscopic hematuria in 2 pediatric patients after receiving the Pfizer COVID-19 vaccine. Kidney Int. 2021;100:705–706. doi: 10.1016/j.kint.2021.06.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sekar A, Campbell R, Tabbara J, Rastogi P. ANCA glomerulonephritis after the Moderna COVID-19 vaccination. Kidney Int. 2021;100:473–474. doi: 10.1016/j.kint.2021.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Anderegg MA, Liu M, Saganas C, Montani M, Vogt B, Huynh-Do U, Fuster DG. De novo vasculitis after mRNA-1273 (Moderna) vaccination. Kidney Int. 2021;100:474–476. doi: 10.1016/j.kint.2021.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Feghali EJ, Zafar M, Abid S, Santoriello D, Mehta S. De-novo antineutrophil cytoplasmic antibody-associated vasculitis following the mRNA-1273 (Moderna) vaccine for COVID-19. Cureus. 2021;13:e19616. doi: 10.7759/cureus.19616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hakroush S, Tampe B. Case report: ANCA-associated vasculitis presenting with rhabdomyolysis and pauci-immune crescentic glomerulonephritis after Pfizer-BioNTech COVID-19 mRNA vaccination. Front Immunol. 2021;12:762006. doi: 10.3389/fimmu.2021.762006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sacker A, Kung V, Andeen N. Anti-GBM nephritis with mesangial IgA deposits after SARS-CoV-2 mRNA vaccination. Kidney Int. 2021;100:471–472. doi: 10.1016/j.kint.2021.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shakoor MT, Birkenbach MP, Lynch M. ANCA-associated vasculitis following Pfizer-BioNTech COVID-19 vaccine. Am J Kidney Dis. 2021;78:611–613. doi: 10.1053/j.ajkd.2021.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tan HZ, Tan RY, Choo JCJ, Lim CC, Tan CS, Loh AHL, Tien CS, Tan PH, Woo KT. Is COVID-19 vaccination unmasking glomerulonephritis? Kidney Int. 2021;100:469–471. doi: 10.1016/j.kint.2021.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Li NL, Coates PT, Rovin BH. COVID-19 vaccination followed by activation of glomerular diseases: does association equal causation? Kidney Int. 2021;100:959–965. doi: 10.1016/j.kint.2021.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Heymans S, Cooper LT. Myocarditis after COVID-19 mRNA vaccination: clinical observations and potential mechanisms. Nat Rev Cardiol. 2022;19:75–77. doi: 10.1038/s41569-021-00662-w. [DOI] [PMC free article] [PubMed] [Google Scholar]