Abstract
Early childhood obesity increases the risk of developing metabolic diseases. We examined the early introduction of exercise in small-litter obese-induced rats (SL) on glucose metabolism in the epididymal adipose tissue (AT) and soleus muscle (SM). On day 3 post-birth, pups were divided into groups of ten or three (SL). On day 22, rats were split into sedentary (S and SLS) and exercise (E and SLE) groups. The rats swam three times/week carrying a load for 30 min. In the first week, they swam without a load; in the 2nd week, they carried a load equivalent to 2% of their body weight; from the 3rd week to the final week, they carried a 5% body load. At 85 days of age, an insulin tolerance test was performed in some rats. At 90 days of age, rats were killed, and blood was harvested for plasma glucose, cholesterol, and triacylglycerol measurements. Mesenteric, epididymal, retroperitoneal, and brown adipose tissues were removed and weighed. SM and AT were incubated in the Krebs-Ringer bicarbonate buffer, 5.5 mM glucose for 1 h with or without 10 mU/mL insulin. Comparison between the groups was performed by 3-way ANOVA followed by the Tukey post-hoc test. Sedentary, overfed rats had greater body mass, more visceral fat, lower lactate production, and insulin resistance. Early introduction of exercise reduced plasma cholesterol and contained the deposition of white adipose tissue and insulin resistance. In conclusion, the early introduction of exercise prevents the effects of obesity on glucose metabolism in adulthood in this rat model.
Keywords: Postnatal overnutrition, Metabolic programming, Exercise, Muscle metabolism, Adipose tissue
Introduction
Early childhood obesity increases the risk of metabolic diseases such as insulin resistance (IR), type 2 diabetes (T2D), cardiovascular disease, and other metabolic disorders (1). In humans, the prevalence of obesity in children has increased markedly (2).
Nutrition during fetal life and soon after birth (metabolic programming) may predispose individuals to metabolic dysfunctions such as obesity (3). This programming may cause disturbances, which might be passed on to future generations (1). Moreover, changes in the environment and energy supply can affect development (4).
Plagemann et al. developed an experimental model of obese rodents by raising rats in small litters (5), which reduces competition for the mother's milk during the lactating period and, therefore, leads to overnourishment because the total calorie intake for each pup is increased. In this model, there are nutritional disturbances during the breastfeeding period that can lead to the development of short- and long-term changes not only in body weight but also in the white adipose tissue (AT) (1). In addition, the rats show an accelerated body weight gain and a long-lasting obese phenotype associated with permanent modulation of hypothalamic circuits that control food intake and energy balance in adulthood (6).
Skeletal muscle (SM), the main site of insulin-dependent glucose disposal, takes up more than 80% of an intravenous glucose load (7). A signal from insulin results in translocation of glucose transporter-4 (GLUT4) to the cell membrane (8). A failure in insulin action leads to insulin resistance (9). Animals that are overnourished during lactation have an epigenetic modification in the insulin-signaling pathway (10). In obese rats, insulin resistance is related to alterations in the expression of insulin receptor substrate-1 (IRS-1) and reduction in the expression of GLUT4, PI3K, and Akt (9,10).
The prevalence of obesity in children has escalated markedly worldwide (11). Inactivity during childhood affects physical well-being in children (12). Exercise is an effective non-pharmacological strategy, promoting well-being and reducing the risk of certain diseases due to the induction of several adaptations at the metabolic level by modulating the expression of specific genes (13). Indeed, in obese animal models and humans, exercise increases insulin sensitivity and augments glucose transport by a non-insulin-dependent pathway (14).
In the present work, we aimed to examine whether early introduction of exercise in 22-day-old small-litter rats prevents in adulthood the effect of obesity on glucose metabolism in vivo and in vitro. For this purpose, we determined several morphometric and biochemical parameters in four groups: sedentary, exercise, small-litter sedentary, and small-litter exercise rats.
Material and Methods
Unless otherwise indicated, chemicals and enzymes were purchased from Sigma Chemical Co. (USA). Radiochemicals were obtained from NEN Life Sciences Products, Inc. (USA). Humulin® R U-100 insulin was obtained from Eli Lilly and Company (USA). Assay kits for measurements of biochemical plasma parameters were purchased from Laborclin (Brazil).
Animals
All animal protocols were approved by the Ethics Committee for Experimental Animals from the Federal University of Paraná (CEUA number 865/2015). Animals were housed under a 12-h light/dark cycle at 23±1°C with free access to water and chow. The animals received a regular chow diet (protein content 230 g/kg, vitamins 660 g/kg, fat 40 g/kg, fiber 60 g/kg, and minerals 10 g/kg; Nuvital®, Brazil).
Food and body weight were monitored every two days from weaning until the age of 90 days.
Induction of obesity by metabolic programming
A total of 21 dams (age of 90 days) were used, of which sixteen were separated for small litter (SL) and five were used as control. After birth, each dam had ten pups. On day three post-birth, one group was kept with three pups per dam, resulting in early overfeeding during lactation (5). The other group was kept with 10 pups per dam. The pups were weaned at 21 days of age (3 male rats per cage). Then, each group was subdivided into two experimental groups forming 4 groups: sedentary (S), exercise (E), small-litter sedentary (SLS), and small-litter exercise (SLE).
Swimming training
After weaning, swimming training was performed as described in our previous work (15). The swimming system consisted of a central compartment and 10 tanks (250 mm diameter and 460 mm high, with 330 mm water level) for individual swimming to avoid the effects of crowding on performance. Briefly, from 22 to 90 days of age, the rats swam three times/week for 30 min. In the first week, they swam without a load; in the 2nd week, they carried a load equivalent to 2% of their body weight; from the 3rd to the last week, they carried a 5% body load attached to their tails. Water temperature was 32±2°C. The use of different load intensities is common in swimming protocols for rats. Slowly increasing the load from 2 to 5% promotes smooth training adaptation while maintaining a low stress level (16). We admit that it is quite difficult to control training intensity when weight is applied to the tail.
Intraperitoneal insulin tolerance test
For the intraperitoneal insulin tolerance test (ITT), we selected some rats from each group at 85 days of age. The ITT was done in 12-h fasting rats 48 h after the last swimming session. Insulin (Humulin® R U-100) 1 U/kg was administered intraperitoneally. Blood samples were collected at 0, 5, 8, 10, 12, 16, 20, and 25 min for glucose determination (LifeScan One Touch, Brazil).
Blood biochemical measurements
Rats not submitted to the ITT were killed at 90 days of age. All experimental protocols were performed 48 h after the last swimming session to avoid any potential acute effect of exercise. After 12 h of fasting, rats were killed by decapitation and total blood was collected in heparinized tubes. Plasma concentrations of glucose, cholesterol, and triacylglycerol were measured by enzymatic procedures using commercial kits.
Adipose tissue harvesting
Fat tissues from mesenteric, epididymal, retroperitoneal, and brown tissues were rapidly removed, and their weight was measured on a digital scale (Denver Instruments Company AA-200, USA).
Incubation of soleus muscle and epididymal fat
Soleus muscles from each leg were rapidly and carefully isolated, split longitudinally into two equal portions (20-30 mg), and attached through the tendons to a small clip to keep the fibers extended. Then, muscles were pre-incubated for 30 min in Krebs-Ringer bicarbonate buffer pre-gassed for 30 min with 95% O2/5% CO2 at 37°C containing 5.6 mM glucose, 1.5% BSA, pH 7.4. Then, the muscles were transferred to flasks that contained identical buffer plus 0.1 µCi/mL D-[U-14C]-glucose in the absence or presence of insulin (Humulin® R U-100, 10 mU/mL). After incubation for 60 min, muscles were removed, and the incubation medium was frozen for lactate measurement (17).
Epididymal fat pads (20-30 mg) were minced in very small pieces using scissors. They were then incubated in Krebs-Ringer bicarbonate buffer at 37°C containing 5.6 mM glucose, 1.5% BSA, pH 7.4, plus 0.1 µCi/mL D-[U-14C]-glucose in the absence or presence of insulin (10 mU/mL). After incubation for 60 min, we added perchloric acid (25%) to stop the reaction. The medium was collected and neutralized with Tris-KOH (2-0.5 M). The medium was harvested for total and radiochemical lactate production measurement.
Lactate production measurement
Total lactate production by isolated incubated soleus muscles and epididymal fat tissues was assayed by measuring absorbance at 340 nm (spectrophotometric Infinite® 200 Pro, USA, TECAN series) as described elsewhere (17). It is important to note that the net rate of lactate formation, which is measured by a spectrophotometric assay, is a measure of the rate of glycolysis from glucose, which can come from either muscle glycogen and/or the incubation medium. Radiochemical lactate was determined as described elsewhere (18).
[3H]-2-deoxy-D-glucose uptake
Sugar transport activity was measured using the non-metabolizable glucose analog [3H]-2-deoxy-D-glucose (19). Briefly, incubation was done as described above but in the presence of [3H]-2-deoxy-D-glucose. After incubation, the muscles were frozen in liquid nitrogen, weighed, and digested for 15 min at 70°C in KOH (1 M). Muscle aliquots were placed in vials with scintillation fluid and glucose was measured in the Beckman LS 6000 IC equipment (USA).
Statistical analyses
Data are reported as means±SE. The data were evaluated for normality (Shapiro-Wilk test) and homoscedasticity (Bartlett test). Ordinary two-way ANOVA was used for fat tissue weight and blood biochemical parameters (factors: litter size and exercise). Two way ANOVA with repeated measures was used for ITT, body weight, and food intake (factors: litter size × exercise × time), followed by Tukey's post hoc test. Three-way ANOVA was used for glucose uptake and lactate production (factors: litter size × exercise × insulin) followed by Sidak's post hoc test. P<0.05 was used to indicate statistical significance. Statistical tests were performed using GraphPad Prism version 8.0 for Windows (GraphPad Software, USA).
Results
At 85 days of age, some rats were subjected to an insulin tolerance test (ITT). In the small-litter sedentary (SLS) group, from 15 min to the end of the ITT, blood glucose concentration was higher compared to the other groups (Figure 1A). Exercise (E) (n=10) increased insulin sensitivity markedly (Figure 1B) by 95.7% compared to the sedentary (S, n=8) group (P<0.0402). SLS (n=10) had higher body weight (Figure 2A and B) and an increased insulin resistance of 52.3% compared to the S group (P<0.0027). On the other hand, the early introduction of exercise in the small-litter exercise group (SLE, n=10) did avoid insulin resistance compared to the SLS group (P<0.0014). No statistical difference was found between exercise groups (P=0.59, E vs SLE).
Figure 1. Insulin tolerance test (1.0 U/kg) at 85 days of age (A) in the sedentary (S), exercise (E), small-litter sedentary (SLS), and small-litter exercise (SLE) groups. B, Glucose disappearance rate (Kitt) of each group. Data are reported as means±SE (n=8-10). *P<0.05; **P<0.005; ***P<0.001 (ANOVA).

Figure 2. Body weight from sedentary (S, n=10), exercise (E, n=11), small-litter sedentary (SLS, n=14), and small-litter exercise (SLE, n=18) rats measured from 22 to 90 days of age (A). Body weight (B) evaluated by area under the curve (AUC). Data are reported as means±SE. #P<0.05, SLS vs SLE; *P<0.05, S vs E and SLS (ANOVA).
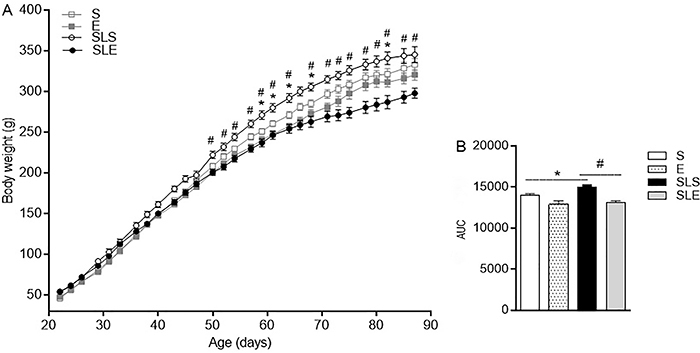
The body weight of all groups increased with age (Figure 2A). The SLS group (n=14) had significantly higher body weight from 50 to 90 days compared to the S (P=0.036), E (P=0.0006), and SLE groups (P=0.0001). The E group (n=11) had a lower body weight but was not different compared to the S group (P>0.05). The early introduction of exercise caused a similar body weight gain in the SLE group (n=18) and E group (P>0.05), which was significantly lower compared to the SLS rats (P<0.05). From 22 to 90 days of age (Figure 2B), the area under the curve (AUC) was 7.4% greater in the SLS rats compared to S rats (P<0.05). On the other hand, in the SLE, the AUC was 12.4% smaller compared to the SLS rats (P<0.05). No significant difference was found between the S and E groups (P>0.05).
From 22 to 90 days of age, no statistical difference in food intake was seen between groups (P>0.05, Figure 3).
Figure 3. Food intake (g) in sedentary (S, n=10), exercise (E, n=11), small-litter sedentary (SLS, n=14), and small-litter exercise (SLE, n=18) rats measured from 22 to 90 days of age. Data are reported as means±SE of at least 10 rats per group. P>0.05 (ANOVA).

In the SLS group, the weight increased by 24% in the mesenteric (Figure 4A), 26% in the epididymal (Figure 4B), and 32% in the retroperitoneal (Figure 4C) fat pads compared to the S group (P<0.05). In both exercise groups (E and SLE), white fat tissues had a lower weight compared with non-exercise groups (S and SLS). In the E and SLE groups, mesenteric fat weight was 22.8 and 24.4%, epididymal fat was 24.4 and 30.7%, and retroperitoneal fat was 32.3 and 30.5% lower, respectively, compared to the S groups (P<0.05). The early introduction of exercise (E and SLE) increased brown fat weight by 118 and 187% compared to non-exercise groups (P<0.05, S and SLS), respectively (Figure 4D). No statistical difference was found in brown adipose tissue weight of the S and SLS groups (P>0.05).
Figure 4. Weight of mesenteric (A), epididymal (B), retroperitoneal (C), and brown (D) fat tissues from sedentary (S), exercise (E), small-litter sedentary (SLS), and small-litter exercise (SLE) groups. Data are reported as means±SE of 10-18 rats per group. *P<0.05 (ANOVA).

Plasma concentrations of glucose, triacylglycerol, and total cholesterol are shown in Table 1. Glycemia and triacylglycerolemia did not differ between groups (P>0.05). Exercise (E and SLE) caused a lower concentration in total cholesterol (6.5%) compared to the sedentary groups (P<0.05).
Table 1. Plasma concentration of glucose, total cholesterol, and triacylglycerol in the sedentary (S), exercise (E), small-litter sedentary (SLS), and small-litter exercise (SLE) groups.
| Blood parameters (mg/dL) | S | E | SLS | SLE |
|---|---|---|---|---|
| Glucose | 76.69±2.08 | 71.63±2.03 | 77.05±1.81 | 77.37±2.51 |
| Cholesterol | 94.52±0.91 | 88.40±1.57# | 93.14 ±0.87 | 89.80±1.22# |
| Triacylglycerol | 66.22±2.53 | 63.10±1.99 | 65.71±2.98 | 63.89±1.96 |
Data are reported as means±SE (n=8-10 rats/group). #P<0.05, vs S and SLS groups (ANOVA).
In basal condition (non-insulin), total lactate production (Figure 5A) did not differ between groups (P>0.05). The presence of insulin increased total lactate production by 1.7-fold in the S (P<0.003), 1.4-fold in the E (P<0.041), 1.8-fold in the SLS (P<0.0005), and 1.5-fold in the SLE (P<0.027 SLE+I vs SLE-I) groups. No statistical significance was found between insulin-stimulated groups (P>0.05).
Figure 5. Total lactate (A) and radiochemical lactate (B) production from glucose in isolated incubated soleus muscles obtained from sedentary (S), exercise (E), small-litter sedentary (SLS), and small-litter exercise (SLE) rats. Muscles were incubated in Krebs-Ringer bicarbonate buffer, containing 5.6 mM glucose, 0.1 uCi/mL D-[U14C] glucose, for 1 h in the absence or presence of 10 mU/mL insulin. Data are reported as means±SE (n=8). *P<0.05 vs non-stimulated (ANOVA).

Radiochemical lactate production (Figure 5B) in the absence of insulin did not differ between groups (P>0.05). The presence of insulin increased radiochemical lactate production by 2.7-fold in the S (P<0.0064), 3.8-fold in the E (P<0.0005), 4.0-fold in the SLS (P<0.0003), and 3.0-fold in the SLE (P<0.0055) groups. No statistical significance was found between insulin-stimulated groups (P>0.05).
Under non-stimulated conditions (Figure 6A), total lactate production by epididymal fat tissue did not differ between groups (P>0.05). In the presence of insulin, total lactate production by epididymal fat tissue was increased in the S and E groups (1.4-fold and 1.6-fold, respectively) compared to non-stimulated conditions (P<0.05). In SLS and SLE groups, insulin increased lactate production by 1.8-fold and 1.4-fold, respectively (P<0.05 vs non-insulin). Regarding [U14C]-lactate production (Figure 6B), no change was seen in the absence of insulin between groups (P>0.05). The presence of insulin increased the [U14C]-lactate production by 2.7-fold and 2.5-fold in the S and E groups, respectively (P<0.05). In the SLS and SLE groups, the increase was by 3.5-fold and 3.1-fold, respectively (P<0.05 vs non-stimulated).
Figure 6. Total lactate (A) and radiochemical lactate (B) production from glucose by epididymal isolated incubated fat tissues obtained from sedentary (S), exercise (E), small-litter sedentary (SLS), and small-litter exercise (SLE) rats. Epididymal fat tissue was incubated in Krebs-Ringer bicarbonate buffer, containing 5.6 mM glucose, 0.1 uCi/mL D-[U14C] glucose, for 1 h in the absence or presence of 10 mU/mL insulin. Data are reported as means±SE (n=8). *P<0.05 vs non-stimulated (ANOVA).

[3H]-2-deoxy-D-glucose (2-DG) uptake by soleus muscle (Figure 7) in basal condition did not differ between groups (P>0.05). In the presence of insulin, the 2DG-glucose uptake by the soleus muscles of all groups increased compared to the basal condition (P<0.05 vs non-stimulated). The 2-DG uptake increased in the S and E groups by 3.9-fold. In the SLS and SLE groups, there was an increase of 1.7-fold and 1.5-fold, respectively.
Figure 7. [3H]-2-deoxy-D-glucose (2DG) uptake by isolated incubated soleus muscles obtained from sedentary (S), exercise (E), small-litter sedentary (SLS), and small-litter exercise (SLE) rats. Muscles were incubated in Krebs-Ringer bicarbonate buffer, containing 5.6 mM glucose, 0.1 μCi/mL 2-DG, for 1 h in the absence or presence of 10 mU/mL insulin. Data are reported as means±SE (n=8). *P<0.05 (ANOVA).

Discussion
Childhood and adolescent obesity is a major health concern and its prevalence has increased worldwide (20,21). Energy intake during lactation can induce alterations in the intermediary metabolism leading to life-long metabolic disturbances (4). Our results corroborate these findings, as 90-day-old rats raised in small litters had obesity. As the rats did not have hyperphagia, obesity was not caused by it. We know that before weaning, the newborn is supplied with nutrients exclusively through the mother's milk (22). An alteration in the quantity and quality of milk content might occur, and this may change the hypothalamic network involved in the energy homeostasis, ultimately leading to obesity (3,5,6).
In the SLS group, the body mass started to increase significantly from 60 days to 90 days of age, accompanied by insulin resistance, and higher mesenteric, epididymal, and retroperitoneal white fat mass without alteration in the brown adipose tissue (BAT) mass. Visceral fat has been associated with insulin resistance in rats submitted to nutritional interventions in gestation and lactation (15,16). Interestingly, despite insulin resistance in vivo, glycemia and triacylglycerolemia (TAG) were not changed in the SLS group nor in the other groups (P>0.05). On the other hand, the introduction of exercise reduced cholesterolemia (P<0.05 vs non-exercise groups). Insulin resistance, both in diabetic and nondiabetic subjects is associated with excess central obesity (23). The diagnosis of T2D is associated with insulin resistance and beta-cell dysfunction (24). It has been reported that nutritional intervention during fetal and early post-natal life affects beta-cell development and glucose-induced insulin secretion (25,26). Therefore, we hypothesize that despite insulin resistance, perhaps at this age, insulin secretion by beta cells is not impaired enough to change glucose and TAG concentrations. This hypothesis remains to be tested.
T2D, dyslipidemia, obesity, and insulin resistance are associated with low-grade chronic inflammation (20). How inflammation is triggered in obesity is not fully understood. One theory is that adipocyte hypertrophy and hyperplasia lead to the expansion of adipose tissue. As a consequence, large adipocytes will not get enough oxygen supply leading to hypoxia, which activates cellular stress pathways. The result is cell autonomous inflammation followed by the release of adipokines (resistin, adiponectin, leptin, among others) that might affect insulin sensitivity in the adipose tissue and skeletal muscle metabolism (27,28). Our approach of early introduction of exercise (starting at 22 days of age) did cause positive effects in rats. SLE rats did not increase body weight compared to the sedentary groups, and the brown fat pad increased significantly, accompanied by non-insulin resistance. During exercise, the tissues demand energy supplied by increased lipolysis and a reduction in the re-esterification of fatty acids followed by an increase in the fatty acid oxidation by peritoneal tissues and particularly by skeletal muscle, leading to a reduction in fat mass (29,30). The improvement in insulin sensitivity caused by fat mass reduction might be related to a change in the immunometabolism of adipose tissue due to exercise (31). It has been reported that exercise reduces the diameter of adipocytes, elevates the secretion of anti-inflammatory adipokines, and changes the phenotype of infiltrated cells in the adipose tissue (32). Exercise increases BAT metabolic activity and expansion (33). Exercise activates the sympathetic nervous system (34), which increases mitochondrial biogenesis and synthesis of uncoupling proteins that will cause an increase in fatty acid oxidation (35). The expansion of BAT might be related to a reduction of circulating lipids. We suggest that exercise affects BAT as an auxiliary mechanism to counterbalance fat mass gain. Pahlavani et al. (36) reported that obese animals have lower visceral adipose mass associated with higher BAT mass when fed with eicosapentaenoic acid. We found a similar result, but we used exercise instead of diet change.
Liver, SM, and AT are the target tissues of insulin (37). In SM, insulin stimulates glucose uptake by translocation of GLUT4 to plasma membrane (8). Therefore, impaired SM insulin signaling results in decreased glucose disposal. Once bound to its receptor, a complex downstream signaling cascade occurs that can be split into two main branches. One is the PI3K-AKT (also known as protein kinase B) pathway, widely responsible for insulin action on glucose uptake (27). The other branch is the Ras-mitogen-activated protein kinase (MAPK) pathway, which besides mediating gene expression also interacts with the PI3K-AKT pathway to control cell growth and differentiation. Both pathways have insulin-receptor-substrate (IRS) activation in common. There is also a non-IRS insulin signaling whose insulin mediator is the heterotrimeric G-protein. In AT, insulin signaling provokes a reduction in the activity of hormone-sensitive lipase, which inhibits free fatty acid (FFA) efflux from adipocytes. This is important because the increased concentration of FFA in the blood results in a reduction of insulin sensitivity and responsiveness in SM due to an elevation of lipid products within the cell, particularly ceramide and fatty acyl-coenzyme A (37). Consequently, these lipids activate the serine-threonine kinase PKC-θ, which, in turn, inhibits the insulin signaling cascade (27).
Obesity modifies carbohydrate (CHO) metabolism by insulin-dependent tissues, particularly skeletal muscle (9). We investigated in vivo and in vitro the CHO metabolism in adipose tissue and skeletal muscle. Obesity causes insulin resistance in vivo. The study of the glycolytic pathway with no interference of systemic factors is possible by the in vitro approach (29). SM and AT are the two main sites for glucose metabolism, and both are insulin-dependent for glucose uptake (7). In the SM and AT, the rate of glycolysis and 2-DG glucose uptake was not modified by obesity, meaning that once inside the cell, glucose is used as an energy source. In both insulin-dependent tissues, the presence of insulin increased total lactate and radiochemical lactate production as well 2-DG glucose uptake. Thus, in vitro, all groups presented a normal insulin-responsiveness. Therefore, the insulin resistance seenin vivowas probably caused by circulating factors. Hotamisligil et al. were the first to report the increased expression and production of TNF-α in adipose tissue of obese subjects and its key role in obesity-induced insulin resistance (38). In the last two decades, several reports corroborated this finding and demonstrated a marked infiltration of immune cells, especially macrophages in adipose tissue of rodents and humans (39,40). Therefore, dyslipidemia and pro-inflammatory cytokines seem to play an important role in insulin resistance.
In summary, our results suggested that the earlier exercise is started, the faster the harmful effects of premature obesity can be reduced or even stopped.
Acknowledgments
We acknowledge funding support from Brazilian Federal Agency for Support and Evaluation of Graduate Education (CAPES) and Brazilian National Council of Technological and Scientific Development (CNPq).
References
- 1.Lecoutre S, Breton C. Maternal nutritional manipulations program adipose tissue dysfunction in offspring. Front Physiol. 2015;6:158. doi: 10.3389/fphys.2015.00158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bradwisch SA, Smith EM, Mooney C, Scaccia D. Obesity in children and adolescents: An overview. Nursing. 2020;50:60–66. doi: 10.1097/01.NURSE.0000718908.20119.01. [DOI] [PubMed] [Google Scholar]
- 3.Bruce KD, Hanson MA. The developmental origins, mechanisms and implications of metabolic syndrome. J Nutr. 2010;140:648–652. doi: 10.3945/jn.109.111179. [DOI] [PubMed] [Google Scholar]
- 4.Kirchner H, Osler ME, Krook A, Zierath JR. Epigenetic flexibility in metabolic regulation: disease cause and prevention? Trends Cell Biol. 2013;23:203–209. doi: 10.1016/j.tcb.2012.11.008. [DOI] [PubMed] [Google Scholar]
- 5.Plagemann A, Harder T, Rake A, Melchior K, Rohde W, Dörner G. Increased number of galanin-neurons in the paraventricular hypothalamic nucleus of neonatally overfed weanling rats. Brain Res. 1999;818:160–163. doi: 10.1016/S0006-8993(98)01264-5. [DOI] [PubMed] [Google Scholar]
- 6.Chen H, Simar D, Lambert K, Mercier J, Morris MJ. Maternal and postnatal overnutrition differentially impact appetite regulators and fuel metabolism. Endocrinology. 2008;149:5348–5356. doi: 10.1210/en.2008-0582. [DOI] [PubMed] [Google Scholar]
- 7.Ferranini E, Lozzo P, Virtanen KA, Honka M-J, Bucci M, Nuutila P. Adipose tissue and skeletal muscle insulin-mediated glucose uptake in insulin resistance: role of blood flow and diabetes. Am J Clin Nutr. 2018;108:749–758. doi: 10.1093/ajcn/nqy162. [DOI] [PubMed] [Google Scholar]
- 8.Jaldin-Fincati JR, Pavarotti M, Frendo-Cumbo S, Bilan PJ, Klip A. Update of GLUT4 vesicle traffic: a cornerstone of insulin action. Trends Endocrinol Metab. 2017;28:597–611. doi: 10.1016/j.tem.2017.05.002. [DOI] [PubMed] [Google Scholar]
- 9.Liu HW, Mahmood S, Srinivasan M, Smiraglia DJ, Patel MS. Developmental programming in skeletal muscle in response to overnourishment in the immediate postnatal life in rats. J Nutr Biochem. 2013;24:1859–1869. doi: 10.1016/j.jnutbio.2013.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rodrigues AL, De Souza EPG, Da Silva SV, Rodrigues DSB, Nascimento AB, Barja-Fidalgo C, et al. Low expression of insulin signaling molecules impairs glucose uptake in adipocytes after early overnutrition. J Endocrinol. 2007;195:485–494. doi: 10.1677/JOE-07-0046. [DOI] [PubMed] [Google Scholar]
- 11.Wang Y, Lim H. The global childhood obesity epidemic and the association between socio-economic status and childhood obesity. Int Rev Psychiatr. 2012;24:176–188. doi: 10.3109/09540261.2012.688195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Janssen I, LeBlanc AG. Systematic review of the health benefits of physical activity and fitness in school-aged children and youth. Int J Behav Nutr Phys Act. 2010;7:40. doi: 10.1186/1479-5868-7-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pareja-Galeano H, Sanchis-Gomar F, Garcia-Giménez JL. Physical exercise and epigenetic modulation: elucidating intricate mechanisms. Sports Med. 2014;44:429–436. doi: 10.1007/s40279-013-0138-6. [DOI] [PubMed] [Google Scholar]
- 14.Flores-Opazo M, McGee SL, Hargreaves M. Exercise and GLUT4. Exerc Sport Sci Rev. 2020;48:110–118. doi: 10.1249/JES.0000000000000224. [DOI] [PubMed] [Google Scholar]
- 15.Fischer SV, Cancian CRC, Montes EG, Leite NC, Grassioli S. Swimming training prevents metabolic imprinting induced by hypernutrition during lactation. Clin Nutr ESPEN. 2015;10:e13–e20. doi: 10.1016/j.clnme.2014.10.003. [DOI] [PubMed] [Google Scholar]
- 16.Gobatto CA, de Mello MA, Sibuya CY, de Azevedo JR, dos Santos LA, Kokubun E. Maximal lactate steady state in rats submitted to swimming exercise. Comp Biochem Physiol A Mol Integr Physiol. 2001;130:21–27. doi: 10.1016/S1095-6433(01)00362-2. [DOI] [PubMed] [Google Scholar]
- 17.Engel PC, Jones JB. Causes and elimination of erratic blanks in enzymatic metabolite assays involving the use of NAD+ in alkaline hydrazine buffers: improved conditions for the assay of L-glutamate, L-lactate, and other metabolites. Anal Biochem. 1978;88:475–484. doi: 10.1016/0003-2697(78)90447-5. [DOI] [PubMed] [Google Scholar]
- 18.Leighton B, Cooper GJ, Da Costa C, Foot EA. Peroxovanadates have full insulin-like effects on glycogen synthesis in normal and insulin-resistant skeletal muscle. Biochem J. 1991;276:289–292. doi: 10.1042/bj2760289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Virkamäki A, Rissanen E, Utriainen T, Yki-Järvinen H. Incorporation of [3-3H]glucose and 2-[1-14C]deoxyglucose into glycogen in heart and skeletal muscle in vivo. Implications for the quantification of tissue glucose uptake. Diabetes. 1997;46:1106–1110. doi: 10.2337/diab.46.7.1106. [DOI] [PubMed] [Google Scholar]
- 20.Cohen E, Margalit I, Shochat T, Goldberg E, Krause I. Markers of chronic inflammation in overweight and obese individuals and the role of gender: a cross-sectional study of a large cohort. J Inflamm Res. 2021;25:567–573. doi: 10.2147/JIR.S294368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ramon-Krauel M, Leal-Witt MJ, Osorio-Conles O, Amat-Bou M, Lerin C, Selva DM. Relationship between adiponectin, TNFα, and SHBG in prepubertal children with obesity. Mol Cell Pediatr. 2021;8:3. doi: 10.1186/s40348-021-00113-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mozeš S, Sefčíková Z, Raček L. Long-term effect of altered nutrition induced by litter size manipulation and cross-fostering in suckling male rats on development of obesity risk and health complications. Eur J Nutr. 2014;53:1273–1280. doi: 10.1007/s00394-013-0630-7. [DOI] [PubMed] [Google Scholar]
- 23.Yudin JS. Adipose tissue, insulin action and vascular disease: inflammatory signals. Int J Obes Relat Metab Disord. 2003;27:S25–S28. doi: 10.1038/sj.ijo.0802496. [DOI] [PubMed] [Google Scholar]
- 24.Khodanbadeloo H, Gorgani-Firuzjaee S, Panahi S, Meshkani R. Molecular and cellular mechanisms linking inflammation to insulin resistance and beta cell dysfunction. Transl Res. 2016;167:228–256. doi: 10.1016/j.trsl.2015.08.011. [DOI] [PubMed] [Google Scholar]
- 25.Waterland RA, Garza C. Early postnatal nutrition determines adult pancreatic glucose-responsive insulin secretion and islet gene expression in rats. J Nutr. 2002;132:357–364. doi: 10.1093/jn/132.3.357. [DOI] [PubMed] [Google Scholar]
- 26.Srinivasan M, Aalinkeel R, Song F, Patel MS. Programming of islet functions in the progeny of hyperinsulinemic/obese rats. Diabetes. 2003;52:984–990. doi: 10.2337/diabetes.52.4.984. [DOI] [PubMed] [Google Scholar]
- 27.de Luca C, Olefsky JM. Inflammation and insulin resistance. FEBS Lett. 2008;582:97–105. doi: 10.1016/j.febslet.2007.11.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kunz EH, Hart CR, Gries KJ, Parvisi M, Laurenti M, Man CD, et al. Adipose tissue macrophage populations and inflammation are associated with systemic inflammation and insulin resistance in obesity. Am J Physiol Endocrinol Metab. 2021;321:E105–E121. doi: 10.1152/ajpendo.00070.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Horowitz JF. Exercise-induced alterations in muscle lipid metabolism improve insulin sensitivity. Exerc Sport Sci Rev. 2007;35:192–196. doi: 10.1097/jes.0b013e318156e084. [DOI] [PubMed] [Google Scholar]
- 30.Egan B, Zierath JR. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metab. 2013;17:162–184. doi: 10.1016/j.cmet.2012.12.012. [DOI] [PubMed] [Google Scholar]
- 31.Vieira-Potter VJ, Zidon TM, Padilla J. Exercise and estrogen make fat cells “fit”. Exerc Sport Sci Rev. 2015;43:172–178. doi: 10.1249/JES.0000000000000046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Goh J, Goh KP, Abbasi A. Exercise and adipose tissue macrophages: new frontiers in obesity research? Front Endocrinol (Lausanne) 2016;7:65. doi: 10.3389/fendo.2016.00065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Stanford KI, Middelbeek RJW, Goodyear LJ. Exercises effects on white adipose tissue: Beiging and metabolic adaptations. Diabetes. 2015;64:2361–2368. doi: 10.2337/db15-0227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Katayama K, Saito M. Muscle sympathetic nerve activity during exercise. J Physiol Sci. 2019;69:589–598. doi: 10.1007/s12576-019-00669-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sanchez-Delgado G, Martinez-Tellez B, Olza J, Aguilera CM, Gil A, Ruiz JR. Role of exercise in the activation of brown adipose tissue. Ann Nutr Metab. 2015;67:21–32. doi: 10.1159/000437173. [DOI] [PubMed] [Google Scholar]
- 36.Pahlavani M, Razafimanjato F, Ramalingam L, Kalupahana NS, Moussa H, Scoggin S, et al. Eicosapentaenoic acid regulates brown adipose tissue metabolism in high-fat-fed mice and in clonal brown adipocytes. J Nutr Biochem. 2017;39:101–109. doi: 10.1016/j.jnutbio.2016.08.012. [DOI] [PubMed] [Google Scholar]
- 37.Posner BI. Insulin signaling: the inside story. Can J Diabetes. 2017;41:101–113. doi: 10.1016/j.jcjd.2016.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hotamisligil GS, Arner P, Caro JF, Atkinson RL, Spiegelman BM. Increased adipose tissue expression of tumor necrosis factor-alpha in human obesity and insulin resistance. J Clin Invest. 1995;95:2409–2415. doi: 10.1172/JCI117936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW., Jr Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112:1796–1808. doi: 10.1172/JCI200319246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.O'Rourke RW, White AE, Metcalf MD, Olivas AS, Mitra P, Larison WG, et al. Hypoxia-induced inflammatory cytokine secretion in human adipose tissue stromovascular cells. Diabetologia. 2011;54:1480–1490. doi: 10.1007/s00125-011-2103-y. [DOI] [PMC free article] [PubMed] [Google Scholar]


