Abstract
Aging is a process in which the various physiological functions of the body gradually deteriorate and eventually lead to death. During this process, the body's resistance to external stresses gradually decreases and the aging-related diseases gradually are increased. Polysaccharides are a group of active substances extracted from living organisms and are widely found in plants, animals, and microorganisms. In the last decade, a variety of natural polysaccharides from functional and medicinal foods have attracted considerable interest for their beneficial effects in the prevention of chronic diseases such as cancers, diabetes, and neurodegenerative diseases. Interestingly, these polysaccharides have also been found to delay aging by reducing oxidative damage, inhibiting telomere shortening, and being anti-inflammatory in different animal models of aging. These reviews summarized the progresses in effects of polysaccharides on antiaging and the potential mechanisms and especially focused on the signaling pathways involved in the antiaging functions. Finally, the applications and prospects of the antiaging effects of polysaccharides are discussed.
1. Introduction
Life expectancy has increased dramatically since the 20th century, but aging is still one of the major problems. Aging is a physiological process in which organs of body undergo an inevitable functional decline with age, a decrease in the ability of the internal environment to stabilize and cope with stress, and a gradual and irreversible deterioration of its structure and components towards death [1]. In addition to direct symptoms such as falls, reduced mobility, frailty, and incontinence, aging could also lead to metabolic-related diseases including diabetes, cardiovascular disease, cancer, and other geriatric syndromes, resulting in reduced quality of life and even death [2, 3]. In 2019, the global population aged 65 and over account for 703 million people, and the number of older people is expected to double to 1.5 billion by 2050, with the proportion of the elderly population rising further to 16% [4]. Population aging has increasingly become a more significant social issue in many countries, including China, Japan, UK, and Germany, and has attracted the attention of the international community in many areas.
Aging is a highly complex process, which also plays an important role in the normal development of the organism, the maintenance of tissue homeostasis, and the inhibition of cancer cell proliferation. It has therefore become crucial to understand how aging works, how it affects organ function, and how it can be prevented and slowed down. Over the past decades, scientific research has started with the analysis of different biological models, comparing genomic, transcriptomic, proteomic, and metabolomic analyses of long- and short-lived species, gradually leading to a partly accepted theory [5–7]. The aging process is divided into two main categories: one is physiological aging, which refers to the corresponding decline in the functions of various tissues and organs as the organism ages, resulting in an increased burden on the organism and affecting its normal life; the other is pathological aging, which refers to the presence of certain diseases that accelerate physiological aging, such as diabetes, cardiovascular disease, and cancer. These two patterns of aging are interrelated and together influence the process of aging [8]. In addition, there are many other factors that influence aging, such as smoking, alcohol consumption, radiation, and unhealthy lifestyles, and some studies suggest that psychological stress is also a major factor in aging [9]. The growing evidences demonstrate that aging is a regulated process, and theories on the mechanisms of aging include telomere shortening, accumulation of DNA damage, mitochondrial dysfunction, and epigenetic changes [10, 11].
Natural polysaccharides are carbohydrate polymeric molecules, usually linear or highly branched biomolecular compounds consisting of more than 10 homogenous or multiple monosaccharides linked by glycosidic bonds. Depending on the source, polysaccharides are classified as plant-based polysaccharides, animal-based polysaccharides, and fungal polysaccharides [12]. Studies indicate that many polysaccharides can exert various biological functions; in particular, the function of antiaging is getting more and more attention [13, 14].
2. Sources, Structures, and Biological Functions of Polysaccharides
Polysaccharides are found in nature from a wide range of sources and can be obtained from plants, fungi, algae, bacteria, etc. Their structures range from being linear to highly branched and include storage polysaccharides (e.g., starch and glycogen) and structural polysaccharides (e.g., cellulose and chitin) [15]. The main structures of polysaccharides in natural products are very complex and diverse, but the basic structure of their main chains usually includes a glucan, fructan, xylan, mannan, galactan, etc., or a polymer of two or more monosaccharides (e.g., galactomannan and pectin). They have a wide variety of branched structures, representing the diversity of polysaccharide structures.
Polysaccharides can be divided into two types depending on their function in living organisms. One is the nutritional polysaccharides that can store energy and nutrients, mostly branched-chain sugar-type molecules. They can be hydrolyzed by enzymes to release monosaccharides, such as starch and liver glycogen, which are soluble in hot water to form colloids. The others are insoluble in water and are structural polysaccharides, mostly straight-chain sugar-type molecules. They mainly form the supporting tissues of plants and animals, such as cellulose in plants and chitin in crustaceans. The structural classification of polysaccharides follows the analysis of proteins and nucleic acids and can be divided into primary, secondary, tertiary, and quaternary structures. As with other biological macromolecules, the advanced structure of the sugar chain is based on the primary structure, which is more complex. It is related to the relative molecular mass, the type and proportion of monosaccharides and glyoxylates, the type of monosaccharide residues, and the order of attachment between the individual monosaccharide residues [16].
Polysaccharides have a variety of biological functions. For instance, polysaccharide fraction isolated from a Cordyceps sinensis (CS) could stimulate iNOS expression and release NO in Raw264.7 cells [17]. Marine microalgae polysaccharides could inhibit DNA topoisomerases I and II and cancer cell proliferation [18]. Astragalus polysaccharide (APS) could improve insulin sensitivity through activation of the AMPK signal pathway in 3T3-L1 adipocytes; combined use of APS and berberine could attenuate insulin resistance in IR-HepG2 cells through modulating the gluconeogenesis signal pathway [19–21]. In this review, we summarized the efficacy and mechanisms of action of certain polysaccharides from medicinal plants, fungi, fruits, vegetables, and medicinal foods in the field of antiaging.
3. Antiaging Effects of Polysaccharides
3.1. The Theory of Aging
Telomere theory is one of the important aging theories. Telomeres are a class of DNA⁃protein complex structures that have the function of closing the ends of chromosome arms and maintaining genomic stability, which are organized into a ring-like structure called a T-loop and are associated with specific proteins, including those that make up the shelterin complex [22]. In all mammals, telomeres are formed by highly conserved hexameric (TTAGGG) tandem repeat DNA sequences [23]. The two main functions of telomeres are to protect the ends of chromosomal arms from inappropriate DNA repair mechanisms preventing the degradation of genes near the ends of chromosome arms due to incomplete DNA replication [24]. Thus, during cell division in humans and other animals, telomeres become progressively shorter in length as DNA continues to be replicated and is continuously depleted to protect chromosome ends. When telomere lengths become very short, the ability to maintain the shelterin complex is lost. In turn, the inhibition of the DNA damage response pathway by the shelterin complex is released and the cell cycle leaves the G1 phase and enters the G0 phase [25]. Upon leaving the cell cycle, the cell enters senescence or apoptosis.
Harman [26] proposed the free radical theory of aging, which states that intracellular reactive oxygen species (ROS) may be one of the most important causes of aging, and aging is caused by an increase in the production of ROS and their accumulation in the body. Under this theory, the level of ROS in the body is in a dynamic equilibrium, and the levels of oxidative stress and antioxidant defenses are in relative balance. However, with age, the secretion level of antioxidant enzymes in the body decreases and the dynamic equilibrium is imbalanced resulting in a large accumulation of ROS in the cells, which leads to the destruction of the biofilm system and impairment of normal cellular functions, thus accelerating the onset of aging. In addition, as the main site of cellular energy metabolism, oxidative phosphorylation of mitochondria is the most important pathway for ROS production. Harman further developed the theory of mitochondrial free radical senescence based on the free radical senescence theory, pointing out that mitochondrial oxidative damage and its dysfunction are the root cause of senescence [27]. Therefore, reducing the level of ROS in the body has become an effective intervention in the research on delaying aging.
The molecular mechanisms of aging and cellular senescence are an extremely complex cascade of reactions involving multiple influences. In addition to the molecular mechanisms described above, there are many other factors that can influence organismal aging. For example, DNA damage accumulation, mitochondrial dysfunction, and epigenetic regulation. In addition, activation of tumor suppressor genes such as p53 and Rb, lamin A/C (LMNA) point mutations, endocrine dysfunction, disruption of the immune system and proteostasis, dietary control, and the mammalian target of rapamycin (mTOR) signal pathway are all closely associated with the onset of aging [28]. In recent years, scientists have continued to study the mechanisms of aging in depth and have proposed various novel theoretical ideas. These theoretical perspectives have made meaningful advances in understanding the mechanisms of senescence; for example, FasL/Fas signaling could promote oocyte senescence [29], and the p53 signaling pathway mediates the human umbilical cord vascular endothelial cell senescence [30]. These theories, which explore the molecular mechanisms and signal pathways of aging from different perspectives, suggest that they are not antagonistic but complementary.
3.2. Animal Investigations of Antiaging Effect
Drosophila is a widely used invertebrate model to investigate the pathology of a variety of diseases and to examine the effects of various active ingredients, including polysaccharides. Studies on the antiaging effects of polysaccharides date back as far as 2008. A study analysed the effects of different molecular weight polysaccharides isolated from porphyran on the longevity and viability of Drosophila melanogaster and showed that large molecular weight (49 kDa) polysaccharides significantly prolonged the life span of Drosophila melanogaster. Compared to controls, porphyran polysaccharides also enhanced the mating ability of middle-aged fruit flies, while small molecular weight (8.2 kDa) polysaccharides increased survival time in heat stress tests [31].
Interestingly, the antiaging activity of polysaccharides from Cordyceps cicadae (CP) also appears to be correlated with molecular weight. Zhu et al. [32] evaluated the in vitro antioxidant and in vivo antiaging activities of CP extracted at different ethanol concentrations (30%-80%) and showed that CP70 (Cordyceps polysaccharides extracted at 70% ethanol concentration) significantly prolonged the life span of Drosophila. This effect was attributed to the upregulation of the expression levels of the antioxidant-related genes catalase (CAT), superoxide dismutase (SOD) 1, and MTH in Drosophila. Another study also confirmed the antiaging effect of polysaccharides contributing to their antioxidant capacity; for example, polysaccharides from Chlorella pyrenoidosa (PCPPs) are also a potential life-extending natural antioxidant [33]. PCPPs increased the total activities of endogenous antioxidant enzymes such as SOD, glutathione peroxidase (GSH-PX), and CAT in Drosophila melanogaster, and the average life span of male and female Drosophila melanogaster treated with PCPPs increased by 11.5% and 10.6%, respectively. Zhang et al. [34] found that Sargassum fusiforme polysaccharides (SFPS), a heteropolysaccharide extracted from the brown alga Sargassum fusiforme, elevated SOD, CAT, and GSH-Px activities and promote longevity by enhancing the Nrf2-mediated antioxidant pathway during the aging process in Drosophila melanogaster. Premna microphylla Turcz (pPMTLs) is a traditional Chinese medicine mainly found in the mountains of Southeastern China, and its roots, stems, and leaves can be used as medicine to detoxify, relieve swelling, and stop bleeding. A recent study found that polysaccharides from pPMTLs could extend the life span of Drosophila through an anti-inflammatory mechanism [35].
Caenorhabditis elegans (C. elegans) have also been used in several instances to test the antiaging activity of polysaccharides. Yuan et al. [36] suggest that two fractions of neutral polysaccharides from Rehmannia glutinosa (NPRRP and NPRR) could increase SOD and CAT activities in C. elegans, scavenging excess ROS and reducing lipofuscin expression in nematodes to prolong life span. Gu et al. [37] investigated the effects of polysaccharides from Auricularia auricula (AAPs-H) on locomotor behaviour, longevity, antioxidant-related enzyme activities, and antioxidant levels in C. elegans. AAPs-H could improve the antioxidant defense system of C. elegans and upregulate oxidative stress-related genes to prevent stress damage in C. elegans and extend its life span. Moreover, Sophora moorcroftiana (S. moorcroftiana) is a dwarf shrub of the legume family endemic to Tibet, China. A soup of its seeds has been used in Chinese folk medicine for anti-inflammation, detoxification, and infectious diseases. Zhang et al. [38] discovered that polysaccharides from S. moorcroftiana seeds significantly improved the survival rate of C. elegans in acute heat stress, prolonged the life span of C. elegans, and improved its reproduction.
In a rat model, Zheng [39] found that injection of Polygonatum sibiricum (PS) polysaccharides significantly increased the SOD and GSH-Px activities of renal tissues in the D-galactosamine- (D-gal-) induced aging rats. The decrease in malondialdehyde (MDA) content and β-galactosidase activity indicated the antiaging effect of PS polysaccharide. Cysteinyl aspartate-specific proteinase-3 (caspase-3) is the most critical apoptosis-executing protease in the apoptotic process, which cleaves Bcl-2 protein and triggers apoptosis upon activation. Moreover, the activity of Bax and caspase-3 in the cells of senescent mice was increased. Jing et al. [40] found that the expression level of Bcl-2 was increased and the expression levels of Bax and caspase-3 were decreased in the cells of senescent mice gavaged with polysaccharides from Athyrium multidentatum (Doll.) Ching (AMC) rhizome, indicating that Athyrium multidentatum polysaccharide could inhibit cell apoptosis and resist senescence. In addition, oral administration of polysaccharides of Fructus corni (PFC) significantly restored the protein levels of Bcl-2 and reduced the expression of Bax and caspase-3 in ovarian granulosa cells of senescent mice. It suggests that PFC can prevent the apoptosis of granulosa cells in the ovary of aging mice, thus reducing the aging rate of the cells [41]. Dendrobium is one of the most important herbal foods for the treatment of geriatric diseases, and Dendrobium officinale polysaccharides (DOP) could reduce cognitive impairment in accelerated aging mouse models by activating microglia [42].
3.3. Cell Investigations of Antiaging Effect
In terms of cellular studies, Tong et al. [43] investigated the effects of sulfated Bupleurum chinense polysaccharides (BCPs) on hydrogen peroxide-induced free radical scavenging activity in mouse lung endothelial cells and their effects on oxidative stress-induced senescence. The results showed that these polysaccharides play an important role in free radical scavenging and are resistant to oxidative damage, and they may be considered novel pharmaceutical products with potential antioxidant and antiaging effects. Angelica sinensis polysaccharides (ASP) could inhibit vascular endothelial cell senescence by enhancing protein kinase B (AKT)/hTERT phosphorylation and inhibiting oxidative stress [44]. They can also delay senescence in response to X-ray-induced hematopoietic stem cells in mice by inhibiting oxidative damage and downregulating p16 mRNA expression [45]. Furthermore, Lycium barbarum polysaccharides (LBP) protect against oxidative stress-induced apoptosis and senescence in human lens epithelial cells [46]. They could suppress zebrafish embryonic cell apoptosis and senescence through a p53-mediated pathway [47] and delay human umbilical vein endothelial cell senescence by downregulating the expression of p53 and p15 [48].
3.4. Clinical Investigations of Antiaging Effect
In terms of clinical research, LBP has pioneered antiaging clinical studies. Amagase and Nance [49] conducted a randomized, double-blind, placebo-controlled clinical study using standardized LBP juice (containing 13.6 mg/mL LBP) given to healthy adults for 14 days. The results demonstrated that daily administration of LBP for 14 days increased subjective perceptions of overall well-being and improved neurological/psychological performance and gastrointestinal function in the subjects. Amagase et al. [50] further conducted a randomized, double-blind, placebo-controlled clinical study in which 50 healthy Chinese adults aged 55-72 years were recruited and treated with LBP juice containing 13.6 mg/mL LBP at a dose of 120 mL/day or placebo (n = 25 per group). The results showed that LBP juice significantly increased serum SOD levels by 8.4%, GPx levels by 9.9%, and MDA by 8.7% in the subjects. These data suggest that LBP is well tolerated in humans and can promote antioxidant capacity in humans through upregulation of antioxidant enzymes.
Overall, the antiaging activity of polysaccharides warrants further exploration through a summary of cellular, animal, and clinical trials. The antiaging effects of natural product polysaccharides are listed in Table 1.
Table 1.
Antiaging effects of natural product polysaccharides.
| Pharmacological action | Polysaccharides | Experiment model | Functions and mechanisms | Reference |
|---|---|---|---|---|
| Enhance vitality | Porphyra yezoensis polysaccharide (0.2%, 1% diets) | Drosophila | Delayed aging process, increased mating capacity | [31] |
| Antioxidation | Polysaccharide from Cordyceps cicadae (0.1%, 0.5%, and 1% diets) | Drosophila | Increased activities of CAT and GSH-Px, inhibited MDA formation, and upregulated the expression level of antioxidant-related genes: CAT, SOD1, and MTH | [32] |
| Antioxidation | Partially purified Chlorella pyrenoidosa polysaccharides (0.25%, 0.5%, and 1% diets) | Drosophila | Scavenged hydroxyl, 1,1-diphenyl-2-picrylhydrazyl, superoxide, and hydroxyl radicals. Increased the activity of endogenous antioxidant enzymes, superoxide dismutase, glutathione peroxidase, and catalase | [33] |
| Antioxidation | Sargassum fusiforme fucoidan (0.4 g/L, 0.8 g/L, and 1.6 g/L) | Drosophila | Improved activities of the antioxidant enzymes: SOD, CAT, and GSH-Px, and reduced the MDA and GSSG contents in older flies | [34] |
| Anti-inflammatory | Premna microphylla Turcz polysaccharide (0.25%, 0.5%, and 1% diets) | Drosophila | Reduced microvillus rupture in the midgut, restored the nuclear structure, and improved the expression levels of immune-related genes in inflamed Drosophila—especially defensin | [35] |
| Antioxidation | Rehmannia glutinosa polysaccharide (5 mg/mL) | Caenorhabditis elegans | Activated the antioxidant enzyme system under oxidative stress by promoting nuclear localization of DAF-16. Improved the activities of SOD and CAT, removed excess ROS, and reduced the expression of lipofuscin | [36] |
| Antioxidation | Auricularia auricula polysaccharide (0.1, 0.2, 0.4, and 0.8 mg/mL) | Caenorhabditis elegans | Improved the antioxidant defense system and upregulated the oxidative stress-related genes for stress damage prevention | [37] |
| Antioxidation | Polysaccharide from Sophora moorcroftiana seeds (2 mg/mL, 4 mg/mL) | Caenorhabditis elegans | Extended the life span and reproduction, increased the oxidative stress resistance and antimicrobial capacity | [19] |
| Antioxidation | Polygonatum sibiricum polysaccharide (100 mg/kg) | Rat | Increased the activities of SOD and GSH PX in kidney tissue, reduced the MDA content and β-galactosidase activity, regulated the Klotho-FGF23 endocrine axis, alleviated oxidative stress, and balanced calcium and phosphorus metabolisms | [39] |
| Antioxidation | Polysaccharide from Athyrium multidentatum (Doll.) Ching (100, 200, and 300 mg/kg) | Mouse | Attenuated D-galactose-induced oxidative stress and cell apoptosis by activating the PI3K/AKT pathway | [40] |
| Inhibition of granulosa cell apoptosis | Fructus corni polysaccharides (1.11 g/kg) | Mouse | Restored the estradiol and progesterone level, reduced the luteinizing hormone and follicle-stimulating hormone levels, increased Bcl-2, and reduced Bax and cleaved caspase-3 | [112] |
| Antioxidation | B. chinense polysaccharides (0.8 and 1.6 mg/mL) | Mouse lung endothelial cells | Scavenged and resisted the H2O2-induced senescence | [43] |
| Antioxidation | Angelica sinensis (Oliv.) Diels (Apiaceae) polysaccharides (20 μg/mL, 100 μg/mL) | Endothelial progenitor cells (EPCs) | Augmented the Akt/hTERT phosphorylation and inhibited the oxidative stress | [44] |
| Inhibition of oxidative damage | Angelica sinensis polysaccharides (200 mg/kg) | Mouse hematopoietic stem cells | Decreased the percentage of SA-beta-Gal-positive cells, the ratio of G1 stages, and the production of ROS. Downregulated the expression levels of p16 mRNA | [45] |
| Antiapoptosis | Lycium barbarum polysaccharides (400 mg/L) | Human lens epithelial cells | Reduced the H2O2-induced cell apoptosis, ROS generation, mitochondrial membrane potential loss, and MDA levels and inhibited the H2O2-induced downregulation of Bcl-2. Upregulated Bax proteins and increased the SOD and GSH enzyme activities levels. Attenuated H2O2-induced cellular senescence | [46] |
| Antiapoptosis | Lycium barbarum polysaccharides (1.0, 2.0, 3.0, and 4.0 mg/mL) | Zebrafish embryos | Decreased the expression of aging-related genes such as p53, p21, and Bax. Increased Mdm2 and TERT genes | [47] |
| Improve cell viability | Lycium barbarum polysaccharides (100 mg/L) | HUVECs | Increased cell viability and decreased p53 and p16 expression levels | [48] |
| Increase the subjective feelings of general well-being | Lycium barbarum polysaccharides (120 mL of GoChi/day) | Healthy adults | Reduced fatigue and stress, improved the neurologic/psychologic performance and gastrointestinal functions | [49] |
| Antioxidation | Lycium barbarum polysaccharides (120 mL of GoChi/day) | Healthy adults | Increased the antioxidant efficacies by stimulating endogenous factors | [50] |
Notes: CAT: catalase; FGF23: fibroblast growth factor 23; GSH-PX: glutathione peroxidase; GSSG: glutathione disulfide; MDA: malondialdehyde; MTH: methuselah; MDM2: mouse double minute 2 homolog; PI3K: phosphatidyl inositol 3-kinase; AKT: protein kinase B; ROS: reactive oxygen species; SOD: superoxide dismutase; hTERT: telomerase reverse transcriptase; GSSG: glutathione disulfide.
4. Antiaging Mechanisms of Polysaccharides
4.1. Regulation of Telomeres and Telomerase
In recent years, an increasing number of studies have shown that polysaccharides could exert unique antiaging effects by increasing telomerase activity or inhibiting telomere shortening. For example, Cynomorium songaricum polysaccharide (CSP) could exert antiaging effects by increasing telomere length in aging mice [51]. Turmeric polysaccharides could upregulate the function of telomerase reverse transcriptase, which promoted cellular immune response, tissue repair, and longevity [52]. ASPs were able to increase telomere length and enhance telomerase activity in hematopoietic stem cells of mice in a senescence model [53, 54]. However, paradoxically, ASPs have also been reported to damage telomeric regions or inhibit telomerase activity to exert anticancer effects [53]. Interestingly, reducing high telomerase activity in cancer cells may actually help to delay organism aging and related diseases [55]. In clinical studies, dietary supplementation with the polysaccharide extract of Astragalus enhanced telomere length in lymphocytes from healthy volunteers [56]. In recent years, the medical community has devoted significant attention to telomeres and telomerase. With the rapid development of molecular biology, more and more phytochemicals with antiaging functions by controlling telomere length and telomerase activity will continue to be discovered and fully explored.
4.2. Free Radical Scavenging
Oxidative stress caused by elevated levels of intracellular ROS is a major trigger for the aging process and age-related pathogenesis, yet intracellular ROS levels and oxidative stress could be reduced by plant or fungal polysaccharides [57]. Polysaccharides from Angelica sinensis not only protect neuronal cells from hydrogen peroxide-induced cytotoxicity but also reduce apoptosis and intracellular reactive oxygen levels. In a rat model of localized cerebral ischemia, ASP could enhance the antioxidant activity of cortical neurons, increase the number of microvessels, and improve postischemic blood flow [58]. In addition, ASP could protect mice from neural stem cell senescence by increasing the activity of SOD and T-AOC (total antioxidant capacity) and reducing the content of MDA, providing a theoretical basis for the treatment of brain aging-related diseases with polysaccharides [59]. Polysaccharide from Trichosanthes peel could increase SOD, CAT, and GSH-PX activities and reduce MDA levels in the liver, brain, kidney, and serum in D-galactose-induced senescent mice [60]. Besides, Auricularia polysaccharides scavenged free radicals and upregulated SOD levels in Cryptorchidium hidradenum. Dictyophora polysaccharides also have antioxidant and neuroprotective functions in Cryptorchis spp. Heat stress is an endogenous oxidative stress in cells, which can promote the production of large amounts of free radicals causing oxidative damage in the body. Polysaccharides from AMC rhizomes could reduce D-galactose-induced oxidative stress and apoptosis by activating the phosphatidyl inositol 3-kinase (PI3K)/AKT pathway, which may partially contribute to its antiaging activity [40]. Zhao et al. [31] extracted polysaccharides from Porphyra haitanensis and added them to the diet of Drosophila melanogaster, which not only increased their resistance to heat stress by 12.37% but also significantly extended the life span of male and female Drosophila under stress-free conditions by 8.6% and 9.34%, respectively. Taken together, polysaccharides play important roles in inhibiting the development of aging-related diseases through antioxidative stress mitigation.
4.3. Enhancement of Immunity
Polysaccharides play an important role in immune regulation and can regulate the immune response by modulating the expression levels of cytokines, thus achieving antiaging effects [61]. Studies showed that LBP could enhance the response of T helper 1 cells (Th1) and T helper 2 cells (Th2) in dendritic cells, which could improve immunity of the body [62]. LBP could significantly increase the lysozyme content in silkworm, which eliminated pathogenic microorganisms by forming soluble glycopeptides in the cell walls of bacteria, thus improving the body's resistance to pathogenic microorganisms and enhancing immunity [63]. Millettia pulchra polysaccharides (MPP) could reduce the production of proinflammatory factors such as interleukin-2 (IL-2) and interleukin-6 (IL-6) in the serum of aging mice, increased the spleen and kidney index of mice, and enhanced their immunity, thereby achieving antiaging effects [64].
4.4. Others
In addition, plant polysaccharides inhibit neurotoxicity and prolong the healthy life span of organisms by regulating proteostasis and alleviating or removing misfolded and abnormally aggregated proteins such as β-amyloid (Aβ), polyQ, or α-synuclein associated with neurodegenerative diseases [65]. For example, PS polysaccharides protect Alzheimer's disease neuronal cells by inhibiting neurotoxicity, and APS block polyQ40 expression, slowing down the abnormal accumulation of polyQ40 causing neurotoxicity and prolonging the life span of C. elegans. Fungal polysaccharides could also regulate some signal pathways closely related to life span, such as IIS, mitogen-activated protein kinases (MAPK), and mTOR pathways, to delay aging. Ganoderma lucidum polysaccharides (GLPs) could activate the expression of DAF-16, a life span-associated transcription factor in the IIS pathway, through the MAPK pathway to prolong the life span of E. hidradiata [66]. Moreover, plant or fungal polysaccharides could extend the life span of model animals or have better effects in reducing the onset of diseases associated with aging (Drosophila, Hidradenia, and rats), but their mechanisms of action are unclear. Therefore, it is an urgent scientific issue to explain how polysaccharides regulate life span extension.
5. Polysaccharides and Aging-Related Signal Pathways
5.1. Regulation of Sirt-1
Sirtuin (Sirt) was firstly discovered in nature in the 1970s. Sirts are an evolutionarily conserved family of class III histone deacetylases that depend on nicotinamide adenine dinucleotides. In mammals, there are seven species of Sirts (Sirt1 to Sirt7), which are involved in maintaining genomic stability, regulating energy metabolism, and modulating cancer stem cells [67]. Despite the fact that functions of the seven Sirts are different, they all share a conserved region for NAD+ binding and catalysis and have two structural domains, a large one consisting mainly of Rossmann folds and a small one consisting of a zinc band structure and a helix. They targeted histones and transcription factors and regulated cellular phenotypes via epigenetic modification. Sirt3, Sirt4, and Sirt5 are found in the mitochondria and could moderate oxidative stress in the mitochondria, while Sirt2 could shuttle between the nucleus and the cytoplasm [68]. Sirt1 is currently the most popular and widely studied protein in aging, and a series of studies have shown that Sirt1 could slow down cellular aging by inhibiting apoptosis, regulating metabolism, and suppressing inflammation [69, 70]. It is also involved in many age-related processes and disorders, such as neurodegenerative and cardiovascular diseases [71].
Polysaccharides could exert unique antiaging effects by activating Sirt1. Probiotics and prebiotics used to prevent and alleviate degenerative changes associated with aging have received much attention. Wang et al. [72] used Lactobacillus plantarum 69-2 in combination with galacto-oligosaccharide (GOS) in a mouse model of aging to assess the effects on aging. The results showed that Lactobacillus plantarum 69-2 and GOS could restore hepatic antioxidant activity and alleviate aging by activating the hepatic adenosine monophosphate-activated protein kinase (AMPK)/Sirt1 signal pathway. For instance, bitter melon polysaccharide could activate Sirt1-mediated β-linked protein deacetylation to exert neuroprotective effects [73, 74]. Lentinan (LEN) has various biological properties such as anticancer, antibacterial, antiviral, and antioxidant effects and could protect cardiomyocytes from hypoxic damage by regulating microRNA-22/Sirt1 [75]. LBP could inhibit cell death by activating Sirt1 [76, 77], and they also could exert protective effects by increasing cell proliferation, inhibiting apoptosis, and regulating Sirt1/hypoxia-inducible factor 1α (HIF-1α) pathway [78]. Tremella polysaccharides could activate Sirt1 and inhibit P. aeruginosa lipopolysaccharide-induced ROS production, apoptosis, and autophagy [79]. APS ameliorates mitochondrial dysfunction through the Sirt1 pathway and has a restorative effect on mitochondria [80]. It also attenuates endoplasmic reticulum stress and apoptosis by regulating the Sirt1-PPARγ coactivator 1α (PGC-1α)/peroxisome proliferator-activated receptor (PPAR) α-FGF21 signaling pathway in the rat liver and the miR-204/Sirt1 axis in retinal pigment epithelial cells [81, 82]. Moreover, polysaccharides from okra (Abelmoschus esculentus (L.) Moench) could inhibit apoptosis and oxidative stress by activating the Sirt1 axis [83], while Tremella fuciformis polysaccharide (TFPs) could inhibit hydrogen peroxide-induced damage through upregulation of Sirt1 in human skin fibroblasts [84]. The tubers of Apios americana Medikus are of high nutritional value and have long been used as food in many countries. However, a study by Chu et al. [85] showed that tuber polysaccharides of Apios americana Medikus with an average molecular weight of 12.16 kDa significantly inhibited the lipopolysaccharide- (LPS-) induced release of nitric oxide (NO) and inflammatory cytokines from Raw264.7 cells by modulating Sirt1, as well as oxidative stress and mitochondrial dysfunction.
5.2. Regulation of mTOR
The mTOR pathway is a class of intracellular serine/threonine protein kinases that act as key regulatory molecules in cellular physiopathological processes, receiving, integrating, and then responding to various stimuli (hormones, growth factors, nutrition, energy, hypoxia, and stress) from both inside and outside the cell. Its mediated signaling pathways play an extremely important role in life processes [86]. Two different complexes of intracellular mTOR are mTORC1 and mTORC2. mTORC1 responds to amino acids, stress, oxygen, energy, and growth factors and is sensitive to rapamycin [87]. It promotes cell growth by inducing and inhibiting anabolic and catabolic processes and also promotes cell cycle progression. mTORC2 responds to growth factors, regulates cell survival and metabolism and the cytoskeleton, and is not sensitive to rapamycin [88]. There has been widespread interest in recent years in the study of the effects of mTORC1 on senescence. It inhibits cellular autophagy and promotes protein synthesis through growth factor and nutrient activation. Over time, this may promote cellular stress (protein aggregation, organelle dysfunction, and oxidative stress) and may lead to the accumulation of damage and reduced cellular function, thereby contributing to the development of aging-related diseases [89, 90]. Studies have also shown that inhibition of the mTOR signal pathway through gene knockout, treatment with rapamycin, or dietary restriction can delay aging in various biological models such as yeast, worms, fruit flies, and mice [91]. Dietary restriction and modulation of the cellular autophagic response are beneficial in extending life span, in which the mTOR signal pathway plays an important role [92].
GLPs are bioactive substances with antioxidant, anticancer, and neuroprotective effects [93, 94]. Liang et al. [95] showed that GLPS exerted protective effects against palmitic acid-induced apoptosis and autophagy by regulating the mTOR signaling pathway. Besides, APS enhanced autophagy by inhibiting the PI3K/AKT/mTOR pathway [96], and a sulfated dextran from cinnamon could reduce the viability of lung cancer cells by inhibiting mTOR activity [97]. Pectic bee pollen polysaccharide (RBPP-P) was isolated from Rosa rosea to reduce hepatic steatosis and insulin resistance by promoting autophagy through the AMPK/mTOR-mediated signaling pathway [98]. Fucoidan, a polysaccharide derived from brown seaweed, has long been used as an ingredient in some dietary supplement products, and it inhibited the PI3K-AKT-mTOR pathway [99]. In addition, Pleurotus ostreatus polysaccharides (POPs) inhibit the AMPK/PI3K/mTOR pathway [100]. Chitosan, an animal polysaccharide derived from the shell of crustaceans, has also been reported to inhibit the mTOR pathway [101].
5.3. Regulation of AMPK
AMPK is a highly conserved regulator of cellular energy metabolism. When the intracellular AMP/ATP ratio is increased, AMPK phosphorylates and activates a large number of downstream target molecules so that ATP utilization is reduced and ATP production is increased, thereby increasing cellular catabolism. When the AMP/ATP ratio is decreased, AMPK activity is inhibited, resulting in an increase in cellular anabolism [102]. AMPK is a heterotrimeric complex composed of catalytic subunits (α1, α2, β1, and β2) and regulatory subunits (γ1, γ2, and γ3). Expressed in various metabolically relevant organs, the different isomers of α, β, and γ could form various possible combinations [103]. The aging process of life is closely related to the metabolic rate, and the aging process of organisms is accompanied by a gradual downregulation of AMPK activity, which tends to be higher in young animals than in older ones. Decreased AMPK activity may be associated with numerous aging-related diseases such as cardiovascular disorders and metabolic syndrome, and activation of AMPK could extend the life span of organisms [104].
APS, an extract of Astragalus membranaceus, is an important bioactive component, and APS improves insulin sensitivity in mice by activating AMPK [28]. In a rat model, APS increased hepatic glycogen synthesis via activation of AMPK in a T2DM rat model [105]. Moreover, in cellular studies, APS significantly enhanced the AMPK/Sirt1 pathway in porcine alveolar macrophages to alleviate apoptosis and proinflammatory cytokine expression [106]. Palmitate-treated Raw264.7 cells produced an inflammatory response, while APS showed anti-inflammatory effects by activating AMPK that effectively ameliorated the palmitate-induced proinflammatory response [107]. Okra is an annual herb of the mallow family, and okra polysaccharides could inhibit apoptosis and oxidative stress by activating the AMPK-Sirt1-PGC-1α signaling axis [83]. Epiphyllum is the roots of a buttercup plant Aconitum carmichaelii Debx, and Epiphyllum polysaccharides protect H9c2 cells from starvation-induced cytotoxicity by activating the AMPK pathway [108]. Schisandra chinensis is a liver-protective herb that has been used in China for centuries. Polysaccharides are one of its main active ingredients and have been reported to improve alcohol-induced liver injury [109]. Moreover, the protective activity of Schisandra chinensis polysaccharides (SCPs) in mice with acute liver injury may be related to the antioxidant, anti-inflammatory, and antiapoptotic properties resulting from activation of the AMPK pathway [110]. Tan et al. studied the antiobesity effects of the polysaccharide-rich red alga Gelidium amansii, a hot water extract of which contained 68.54% water-soluble indigestible carbohydrate polymers. In addition, pectin is a complex polysaccharide found in plant cell walls and consists of four main types of polysaccharides: homogalacturonan, xylogalacturonan, and rhamnogalacturonans I and II, all of which could be degraded into various pectic oligosaccharides (POS). POS are considered to be new potential functional foods with a variety of health-promoting properties, and POS are thought to have a role in regulating AMPK [111]. The aging process is accompanied by a decrease in physical endurance, and polysaccharides from Irpex lacteus, a white-rot tamarin fungus, could increase the antifatigue activity of Kunming mice, whose ability to improve endurance is mainly achieved by activating AMPK [112]. Low molecular weight rockweed polysaccharide (LMWF), a sulphated polysaccharide derived from brown seaweed with strong anti-inflammatory and antioxidant activities, prevented nonalcoholic fatty liver disease (NAFLD) in db/db mice by activating the Sirt1/AMPK/PGC1α signaling pathway [113], while fucoidan polysaccharides ameliorate renal cell senescence by inhibiting autophagy-related activation of the AMPK-ULK1 signaling pathway through VE-like effects in a D-gal-induced senescence model of human proximal renal tubular epithelial cells. Wu et al. [114] extracted chicory polysaccharides (CPs) from chicory roots and fractionated it to isolate two novel polysaccharide fractions, CPs-1 and CPs-2, of which CPs-1 is a heteropolysaccharide composed mainly of sorbitan, glucose, fructose, and glucitol. CPs-1 significantly attenuated high-fat diet-induced nonalcoholic fatty liver disease in rats through AMPK activation [114].
5.4. Regulation of p53
The p53 signal pathway plays an important role in cellular senescence. The cellular senescence pathway can be triggered by inflammatory factors, leading to increased expression of the cellular oncogene p53, which in turn leads to the expression of its downstream target protein p21. The activation of p21 protein could block the progression of cells from G1 to the S phase, causing a transient cell cycle arrest and ultimately cellular senescence [115]. It was found that oral administration of DOPs reduced p53 expression and increased Bcl-2 expression in the ovaries of aging mice, thereby treating premature ovarian failure due to natural aging [116]. Injections of ASPs reduced the expression of p53 and p21 proteins in mice [59], and gavage of MPP reduced the expression of p21 and p53 proteins in the liver and brain of aging mice, thereby delaying cellular aging [64]. The above experiments suggest that polysaccharides regulate the p53 signal pathway to slow down aging. The relationship between natural product polysaccharides and aging-related signal pathways is listed in Table 2, and for the antiaging mechanisms of polysaccharides, see Figure 1.
Table 2.
Natural product polysaccharides and aging-related signal pathways.
| Signal pathway | Polysaccharides | Experiment model | Functions and mechanisms | Reference |
|---|---|---|---|---|
| Sirt-1 regulation | Lactobacillus plantarum 69-2 combined with galacto-oligosaccharide (500 mg/kg) | Aging mouse model | Improved gut microbiota regulation, increased short-chain fatty acid levels, and activated the hepatic AMPK/SIRT1 regulatory pathway | [72] |
| Momordica charantia polysaccharide (200 mg/kg) | Neural stem cells | The neural stem cell neuronal differentiation promoted because of the deacetylated β-catenin by SIRT1 | ([73]; [74]) | |
| Lycium barbarum polysaccharide (100, 200, and 400 mg/L) | Human lens epithelial cell line SRA01/04 cells | Upregulated Sirt1 and Bcl-2, suppressed cell death related genes | [76] | |
| Lycium barbarum polysaccharide (100 mg/kg) | Diabetic rats | Increased cell proliferation, inhibited cell apoptosis, and regulated SIRT1/HIF-1α expression | [78] | |
| Tremella polysaccharides (10 μg/mL) | Human epithelial A549 lung cancer cells | Activated SIRT1 and inhibited the LPS-induced ROS production, apoptosis, and autophagy | [79] | |
| Astragalus polysaccharide (100 mg/kg) | BALB/c male mice | Restored imbalance between mitochondrial fusion-fission processes, activated mitophagy, decreased PGC-1α expression, and ameliorated mitochondrial dysfunction | [80] | |
| Astragalus polysaccharide (700 mg/kg) | Male Sprague-Dawley rats | Suppressed abnormal glycolipid metabolism and insulin resistance by improving hepatic SIRT1-PPARα-FGF21 intracellular signaling. Reduced chronic inflammation by attenuating hepatic steatosis | [81] | |
| Astragalus polysaccharide (2.5, 25, and 50 μg/ml) | Retinal pigment epithelial cells | Inhibited ER stress and subsequent apoptosis via regulating miR-204/SIRT1 axis | [82] | |
| Okra polysaccharides (200 or 400 mg/kg) | Diabetic mice | Suppressed apoptosis and oxidative stress through activating the AMPK-Sirt1-PGC-1α signaling axis | [83] | |
| Tremella fuciformis polysaccharide (100, 200, and 300 μg/mL) | Human skin fibroblasts | Alleviated hydrogen peroxide-induced oxidative stress and apoptosis | [84] | |
| Apios americana Medikus tuber polysaccharide (30 mg/mL) | RAW 264.7 cells | Suppressed NO release, inflammatory cytokines, oxidative stress, and mitochondrial dysfunction | [85] | |
| mTOR regulation | Ganoderma lucidum polysaccharide (1.2 mg/mL) | Intestinal porcine epithelial cell line | Inhibited cell apoptosis and autophagy through the promotion of Akt phosphorylation and mammalian target of rapamycin (mTOR) | [95] |
| Astragalus polysaccharide (50, 100, and 200 μg/mL) | Fibroblast-like synoviocytes | Inhibited cell growth and proinflammatory response by enhancement of autophagy via PI3K/AKT/mTOR inhibition | [96] | |
| A sulfated glucan from Antrodia cinnamomea (650 μg/mL) | Lung cancer cells | Reduced lung cancer cell viability via inhibition of the EGFR and mTOR activities | [97] | |
| Pectic bee pollen polysaccharide from Rosa rugosa (0.1 mg/mL) | Obese mice | Alleviated diet-induced hepatic steatosis and insulin resistance via AMPK/mTOR-mediated autophagy | [98] | |
| Fucoidan from seaweed Fucus vesiculosus (50, 100, and 200 μg/mL) | A549 lung cancer cells | Exhibited antimetastatic effect on A549 lung cancer cells via the downregulation of ERK1/2 and Akt-mTOR as well as NF-κB signaling pathways | [99] | |
| Pleurotus nebrodensis polysaccharide (200 μg/mL) | A549 tumor-bearing mice | Activated AMPK phosphorylation, inhibited PI3K/AKT phosphorylation, suppressed the activation of the mTOR signaling pathway, and decreased the expression of the translation-related protein P70S6K | [100] | |
| Chitosan oligosaccharide (500 mg/kg) | Mouse model of colitis-associated colorectal cancer | Suppressed tumor progression through AMPK activation and suppression of NF-kappaB and mTOR signaling | [101] | |
| AMPK regulation | Astragalus polysaccharide (10 μg/mL) | Mouse 3T3-L1 preadipocytes | Improved insulin sensitivity via AMPK activation | [19] |
| Astragalus polysaccharide (700 mg/kg) | Type 2 diabetes mellitus rat model | Alleviated glucose toxicity by increasing liver glycogen synthesis and skeletal muscle glucose translocation via AMPK activation | [105] | |
| Astragalus polysaccharide (200 mg/kg) | Porcine alveolar macrophages | Attenuated ochratoxin A-induced immune stress by activating the AMPK/SIRT-1 signaling pathway | [106] | |
| Astragalus polysaccharide (400 μg/mL) | RAW264.7 cells | Ameliorated palmitate-induced proinflammatory responses through AMPK activation | [107] | |
| Polysaccharide from Fuzi (200 mg/kg) | H9c2 cells | Increased autophagy through AMPK/mTOR pathway activation | [108] | |
| Schisandra chinensis acidic polysaccharide (10, 20, and 40 mg/kg) | Mouse model of acute liver injury | Diminished MDA levels, GSH, and cleaved caspase-3 expression, elevated the expression of p-AMPK, p-Akt, and p-glycogen synthase kinase 3β, and partially reversed acetaminophen-induced liver injury | [110] | |
| Irpex lacteus polysaccharide-enriched extract (0.04, 0.2, and 1.0 g/kg) | Mouse | Enhanced the endurance capacity of mouse by elevating antioxidant associated with the AMPK pathway | [41] | |
| Low molecular weight fucoidan (40 and 80 mg/kg) | Obese diabetic db/db mice | Prevented NAFLD by activating the SIRT1/AMPK/PGC1α signaling pathway | [113] | |
| Chicory polysaccharides (100 and 200 mg/kg) | High-fat diet rats | Attenuated high-fat diet induced nonalcoholic fatty liver disease via AMPK activation | [114] | |
| p53 regulation | Polysaccharides from Dendrobium officinal (70 mg/kg) | Female mice | Alleviated damage caused by aging through the inhibition of the nuclear NF-κB and p53/Bcl-2-mediated signaling pathways | [116] |
| Angelica polysaccharide (140 mg/kg) | Nestin-GFP transgenic mouse brain tissues and neural stem cells | Delayed aging speed by protecting neural stem cells and upregulating the p53/p21 signaling pathway | [59] | |
| Yulangsan polysaccharide (0.6 g/kg) | D-Galactose-treated mice | Suppressed the aging process by decreasing p21 and p53 gene expressions in the liver and brain | [64] |
Notes: AMPK: adenosine monophosphate-activated protein kinase; ERK: extracellular signal-regulated kinase; FGF21: fibroblast growth factor 21; PPAR: peroxisome proliferator-activated receptor; PI3K: phosphatidyl inositol 3-kinase; AKT: protein kinase B; ROS: reactive oxygen species; SIRT: sirtuin; mTOR: the mammalian target of rapamycin; NAFLD: nonalcoholic fatty liver disease; LPS: lipopolysaccharides; NO: nitric oxide; AMPK: adenosine monophosphate-activated protein kinase; PGC: primordial germ cell; MDA: malondialdehyde; GSH-PX: glutathione peroxidase; HIF: hypoxia-inducible factor.
Figure 1.
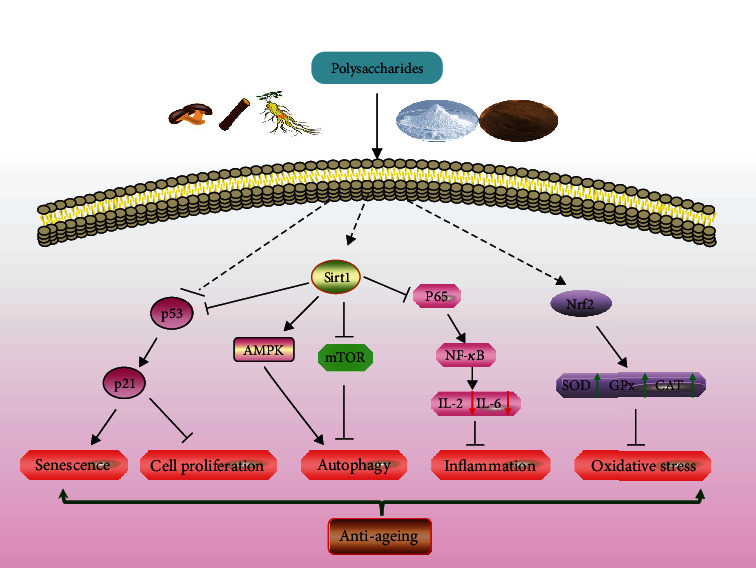
Antiaging mechanisms of polysaccharides and related signal pathways. AMPK: adenosine 5′-monophosphate-activated protein kinase; SOD: superoxide dismutase; GPx: glutathione peroxidase; CAT: catalase enzymes; mTOR: mammalian target of rapamycin; NF-κB: nuclear factor kappa-B; Nrf2: nuclear factor E2-related factor 2.
6. Conclusions and Perspectives
Aging is a complex physiological process influenced by multiple factors, particularly environmental and genetic factors. The carbohydrate component of the human diet is derived almost exclusively from plant sources, while they also have other implications for diet and health. Recent studies indicate that polysaccharides play an important role in antiaging. More and more polysaccharides are used to develop antiaging functional foods. From the viewpoint of aging theory, antioxidant reagents usually have antiaging function and many polysaccharides also have strong antioxidant ability. Besides this, recent investigations show that many polysaccharides could target antiaging key molecules such as mTOR, AMPK, and p53, and the antiaging mechanism seems to be resolved at the gene and molecule levels. Up to now, whether dietary polysaccharides could be absorbed directly into the blood in the digestive tract is still controversial, and many researchers believe that polysaccharides cannot be absorbed directly into the blood, but there is also evidence that dietary polysaccharides could be detected in the blood, suggesting that at least some of them are directly absorbed by digestive gut, but the amount that could be detected in the blood is still very small; this suggests that it may not be the most important way for polysaccharides to play an antiaging role. With the development of investigations on intestinal microflora, more and more evidences show that polysaccharide could change intestinal microflora, and the change of intestinal microflora will inevitably lead to the change of its metabolic products, and these products could enter the blood and regulate AMPK, mTOR, and other aging-related gene expression in different cells. Therefore, it will be an important research direction to study the antiaging effect of polysaccharides through the change of intestinal microflora.
Acknowledgments
This work was supported by the Program for Science & Technology Innovation Platform of Hunan Province (2019TP1029) and the Graduate Innovative Research Project of Hunan Province and Central South University of Forestry and Technology under Grant numbers CX20200699 and CX20201018.
Abbreviations
- AMPK:
Adenosine monophosphate-activated protein kinase
- ASP:
Angelica sinensis polysaccharides
- APS:
Astragalus polysaccharide
- AMC:
Athyrium multidentatum (Doll.) Ching
- BCPs:
Bupleurum chinense polysaccharides
- C. elegans:
Caenorhabditis elegans
- CAT:
Catalase
- CPs:
Chicory polysaccharides
- PCPPs:
Chlorella pyrenoidosa
- CP:
Cordyceps cicadae
- CS:
Cordyceps sinensis
- CSP:
Cynomorium songaricum polysaccharide
- DOP:
Dendrobium officinale polysaccharides
- ERK:
Extracellular signal-regulated kinase
- FGF21:
Fibroblast growth factor 21
- PFC:
Fructus corni
- PGC:
Primordial germ cell
- GOS:
Galacto-oligosaccharide
- GLPs:
Ganoderma lucidum polysaccharides
- GSH-PX:
Glutathione peroxidase
- GSSG:
Glutathione disulfide
- eGFR:
Estimated glomerular filtration rate
- HIF:
Hypoxia-inducible factor
- LEN:
Lentinan
- LMWF:
Low molecular weight rockweed polysaccharide
- LBP:
Lycium barbarum polysaccharides
- MDA:
Malondialdehyde
- MPP:
Millettia pulchra polysaccharide
- MTH:
Methuselah
- MAPK:
Mitogen-activated protein kinases
- MDM2:
Mouse double minute 2 homolog
- NO:
Nitric oxide
- NAFLD:
Nonalcoholic fatty liver disease
- RBPP-P:
Pectic bee pollen polysaccharide
- POS:
Pectic oligosaccharides
- PPAR:
Peroxisome proliferator-activated receptor
- PI3K:
Phosphatidyl inositol 3-kinase
- POPs:
Pleurotus ostreatus polysaccharides
- PS:
Polygonatum sibiricum
- pPMTLs:
Premna microphylla Turcz
- AKT:
Protein kinase B
- ROS:
Reactive oxygen species
- SFPS:
Sargassum fusiforme polysaccharides
- SCPs:
Schisandra chinensis polysaccharides
- Sirt:
Sirtuin
- SOD:
Superoxide dismutase
- hTERT:
Telomerase reverse transcriptase
- T-AOC:
Total antioxidant capacity
- mTOR:
The mammalian target of rapamycin
- TFPs:
Tremella fuciformis polysaccharides.
Conflicts of Interest
No potential conflict of interest was reported by the authors.
References
- 1.López-Otín C., Blasco M. A., Partridge L., Serrano M., Kroemer G. The hallmarks of aging. Cell . 2013;153(6):1194–1217. doi: 10.1016/j.cell.2013.05.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Flacker J. M. What is a geriatric syndrome anyway? Journal of the American Geriatrics Society . 2003;51(4):574–576. doi: 10.1046/j.1532-5415.2003.51174.x. [DOI] [PubMed] [Google Scholar]
- 3.Niccoli T., Partridge L. Ageing as a risk factor for disease. Current Biology . 2012;22(17):R741–R752. doi: 10.1016/j.cub.2012.07.024. [DOI] [PubMed] [Google Scholar]
- 4.Arnold M., Rajagukguk Y. V., Gramza-Michalowska A. Functional food for elderly high in antioxidant and chicken eggshell calcium to reduce the risk of osteoporosis-a narrative review. Foods . 2021;10(3, article 656) doi: 10.3390/foods10030656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Keane M., Semeiks J., Webb A. E., et al. Insights into the evolution of longevity from the bowhead whale genome. Cell Reports . 2015;10(1):112–122. doi: 10.1016/j.celrep.2014.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Seim I., Ma S., Zhou X., et al. The transcriptome of the bowhead whale Balaena mysticetus reveals adaptations of the longest-lived mammal. Aging (Albany NY) . 2014;6(10):879–899. doi: 10.18632/aging.100699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Semeiks J., Grishin N. V. A method to find longevity-selected positions in the mammalian proteome. PLoS One . 2012;7(6, article e38595) doi: 10.1371/journal.pone.0038595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kirkwood T. B., Holliday R. The evolution of ageing and longevity. Proceedings of the London Mathematical Society . 1979;205(1161):531–546. doi: 10.1098/rspb.1979.0083. [DOI] [PubMed] [Google Scholar]
- 9.Gems D. What is an anti-aging treatment? Experimental Gerontology . 2014;58:14–18. doi: 10.1016/j.exger.2014.07.003. [DOI] [PubMed] [Google Scholar]
- 10.Franzke B., Neubauer O., Wagner K. H. Super DNAging--new insights into DNA integrity, genome stability and telomeres in the oldest old. Mutation Research-DNA Repair . 2015;766:48–57. doi: 10.1016/j.mrrev.2015.08.001. [DOI] [PubMed] [Google Scholar]
- 11.Gluck S., Ablasser A. Innate immunosensing of DNA in cellular senescence. Current Opinion in Immunology . 2019;56:31–36. doi: 10.1016/j.coi.2018.09.013. [DOI] [PubMed] [Google Scholar]
- 12.Yu Y., Shen M., Song Q., Xie J. Biological activities and pharmaceutical applications of polysaccharide from natural resources: a review. Carbohydrate Polymers . 2018;183:91–101. doi: 10.1016/j.carbpol.2017.12.009. [DOI] [PubMed] [Google Scholar]
- 13.Chen L., Ge M. D., Zhu Y. J., et al. Structure, bioactivity and applications of natural hyperbranched polysaccharides. Carbohydrate Polymers . 2019;223, article 115076 doi: 10.1016/j.carbpol.2019.115076. [DOI] [PubMed] [Google Scholar]
- 14.Ullah S., Khalil A. A., Shaukat F., Song Y. Extraction and biomedical properties of polysaccharides. Foods . 2019;8(8, article 304) doi: 10.3390/foods8080304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lyu F., Xu X., Zhang L. Natural polysaccharides with different conformations: extraction, structure and anti-tumor activity. Journal of Materials Chemistry B . 2020;8(42):9652–9667. doi: 10.1039/D0TB01713B. [DOI] [PubMed] [Google Scholar]
- 16.Ren Y., Bai Y., Zhang Z., Cai W., del Rio Flores A. The preparation and structure analysis methods of natural polysaccharides of plants and fungi: a review of recent development. Molecules . 2019;24(17):p. 3122. doi: 10.3390/molecules24173122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen W., Zhang W., Shen W., Wang K. Effects of the acid polysaccharide fraction isolated from a cultivated Cordyceps sinensis on macrophages in vitro. Cell Immunology . 2010;262(1):69–74. doi: 10.1016/j.cellimm.2010.01.001. [DOI] [PubMed] [Google Scholar]
- 18.Umemura K., Yanase K., Suzuki M., Okutani K., Yamori T., Andoh T. Inhibition of DNA topoisomerases I and II, and growth inhibition of human cancer cell lines by a marine microalgal polysaccharide. Biochemistry Pharmacology . 2003;66(3):481–487. doi: 10.1016/S0006-2952(03)00281-8. [DOI] [PubMed] [Google Scholar]
- 19.Zhang R., Qin X., Zhang T., Li Q., Zhang J., Zhao J. Astragalus polysaccharide improves insulin sensitivity via AMPK activation in 3T3-L1 adipocytes. Molecules . 2018;23(10):p. 2711. doi: 10.3390/molecules23102711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mao Z. J., Lin M., Zhang X., Qin L. P. Combined use of Astragalus polysaccharide and berberine attenuates insulin resistance in IR-HepG2 cells via regulation of the gluconeogenesis signaling pathway. Frontier in Pharmacology . 2019;10, article 1508 doi: 10.3389/fphar.2019.01508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Liu M., Wu K., Mao X., Wu Y., Ouyang J. Astragalus polysaccharide improves insulin sensitivity in KKAy mice: regulation of PKB/GLUT4 signaling in skeletal muscle. Journal of Ethnopharmacology . 2010;127(1):32–37. doi: 10.1016/j.jep.2009.09.055. [DOI] [PubMed] [Google Scholar]
- 22.de Lange T. Shelterin-mediated telomere protection. Annual Review of Genetics . 2018;52(1):223–247. doi: 10.1146/annurev-genet-032918-021921. [DOI] [PubMed] [Google Scholar]
- 23.Giardini M. A., Segatto M., Da Silva M. S., Nunes V. S., Cano M. I. N. Telomere and telomerase biology. Progress in Molecular Biology and Translational Science . 2014;125:1–40. doi: 10.1016/B978-0-12-397898-1.00001-3. [DOI] [PubMed] [Google Scholar]
- 24.Turner K. J., Vasu V., Griffin D. K. Telomere biology and human phenotype. Cells . 2019;8, article 73 doi: 10.3390/cells8010073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jiang H., Ju Z., Rudolph K. L. Telomere shortening and ageing. Zeitschrift für Gerontologie und Geriatrie . 2007;40(5):314–324. doi: 10.1007/s00391-007-0480-0. [DOI] [PubMed] [Google Scholar]
- 26.Harman D. Aging: a theory based on free radical and radiation chemistry. Journals Of Gerontology Series A-biological Sciences And Medical Sciences . 1956;11(3):298–300. doi: 10.1093/geronj/11.3.298. [DOI] [PubMed] [Google Scholar]
- 27.Barja G. The mitochondrial free radical theory of aging. Progress in Molecular Biology and Translational Science . 2014;127:1–27. doi: 10.1016/B978-0-12-394625-6.00001-5. [DOI] [PubMed] [Google Scholar]
- 28.Aunan J. R., Watson M. M., Hagland H. R., Søreide K. Molecular and biological hallmarks of ageing. British Journal of Surgery . 2016;103(2):e29–e46. doi: 10.1002/bjs.10053. [DOI] [PubMed] [Google Scholar]
- 29.Zhu J., Lin F. H., Zhang J., et al. The signaling pathways by which the Fas/FasL system accelerates oocyte aging. Aging (Albany NY) . 2016;8(2):291–303. doi: 10.18632/aging.100893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Song S., Wu S., Wang Y., et al. 17β-Estradiol inhibits human umbilical vascular endothelial cell senescence by regulating autophagy via p53. Experimental Gerontology . 2018;114:57–66. doi: 10.1016/j.exger.2018.10.021. [DOI] [PubMed] [Google Scholar]
- 31.Zhao T., Zhang Q., Qi H., Liu X., Li Z. Extension of life span and improvement of vitality of Drosophila melanogaster by long-term supplementation with different molecular weight polysaccharides from Porphyra haitanensis. Pharmacology Reserch . 2008;57(1):67–72. doi: 10.1016/j.phrs.2007.12.001. [DOI] [PubMed] [Google Scholar]
- 32.Zhu Y., Yu X., Ge Q., et al. Antioxidant and anti-aging activities of polysaccharides from Cordyceps cicadae. International Journal of Biological Macromolecules . 2020;157:394–400. doi: 10.1016/j.ijbiomac.2020.04.163. [DOI] [PubMed] [Google Scholar]
- 33.Chen Y., Liu X., Wu L., et al. Physicochemical characterization of polysaccharides from Chlorella pyrenoidosa and its anti-ageing effects in Drosophila melanogaster. Carbohydrate Polymers . 2018;185:120–126. doi: 10.1016/j.carbpol.2017.12.077. [DOI] [PubMed] [Google Scholar]
- 34.Zhang Y., Xu M., Hu C., et al. Sargassum fusiforme fucoidan SP2 extends the lifespan of Drosophila melanogaster by upregulating the Nrf2-mediated antioxidant signaling pathway. Oxidation Medicine Cell Longevity . 2019;2019, article 8918914:15. doi: 10.1155/2019/8918914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Song G., Chen F., Chen S., Ye S. Polysaccharides from Premna microphylla turcz ameliorate inflammation via the enhancement of intestinal resistance in host. Journal of Ethnopharmacology . 2021;276, article 114208 doi: 10.1016/j.jep.2021.114208. [DOI] [PubMed] [Google Scholar]
- 36.Yuan Y., Kang N., Li Q., Zhang Y., Liu Y., Tan P. Study of the effect of neutral polysaccharides from Rehmannia glutinosa on lifespan of Caenorhabditis elegans. Molecules . 2019;24(24):p. 4592. doi: 10.3390/molecules24244592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gu J., Li Q., Liu J., et al. Ultrasonic-assisted extraction of polysaccharides from Auricularia auricula and effects of its acid hydrolysate on the biological function of Caenorhabditis elegans. International Journal of Biological Macromolecules . 2021;167:423–433. doi: 10.1016/j.ijbiomac.2020.11.160. [DOI] [PubMed] [Google Scholar]
- 38.Zhang Y., Dan-Yang M. I., Jin W. A. N. G., et al. Constituent and effects of polysaccharides isolated from Sophora moorcroftiana seeds on lifespan, reproduction, stress resistance, and antimicrobial capacity in Caenorhabditis elegans. Chinese Journal of Natural Medicines . 2018;16(4):252–260. doi: 10.1016/S1875-5364(18)30055-4. [DOI] [PubMed] [Google Scholar]
- 39.Zheng S. Protective effect of Polygonatum sibiricum polysaccharide on D-galactose-induced aging rats model. Science Reports . 2020;10(1):p. 2246. doi: 10.1038/s41598-020-59055-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jing L., Jiang J. R., Liu D. M., et al. Structural characterization and antioxidant activity of polysaccharides from Athyrium multidentatum (Doll.) Ching in d-galactose-induced aging mice via PI3K/AKT pathway. Molecules . 2019;24(18):p. 3364. doi: 10.3390/molecules24183364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang Y., Wu J. Z., Li Y., Qi X. Polysaccharides of fructus corni improve ovarian function in mice with aging-associated perimenopause symptoms. Evidence-based Complementary and Alternative Medicine . 2019;2019:8. doi: 10.1155/2019/2089586.2089586 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Feng C. Z., Cao L., Luo D., et al. Dendrobium polysaccharides attenuate cognitive impairment in senescence-accelerated mouse prone 8 mice via modulation of microglial activation. Brain Reserch . 2019;1704:1–10. doi: 10.1016/j.brainres.2018.09.030. [DOI] [PubMed] [Google Scholar]
- 43.Tong H., Zheng X., Song J., et al. Radical scavenging activity of sulfated Bupleurum chinense polysaccharides and their effects against oxidative stress-induced senescence. Carbohydrate Polymers . 2018;192:143–149. doi: 10.1016/j.carbpol.2018.03.061. [DOI] [PubMed] [Google Scholar]
- 44.Lai P., Liu Y. Angelica sinensis polysaccharides inhibit endothelial progenitor cell senescence through the reduction of oxidative stress and activation of the Akt/hTERT pathway. Pharmaceutical Biology . 2015;53(12):1842–1849. doi: 10.3109/13880209.2015.1027779. [DOI] [PubMed] [Google Scholar]
- 45.Zhang X. P., Wang Q. X., Chen B., et al. Angelica sinensis polysaccharides delay aging of hematopoietic stem cells through inhibitting oxidative damge. Zhongguo Zhong Yao Za Zhi . 2013;38(3):407–412. [PubMed] [Google Scholar]
- 46.Qi B., Ji Q., Wen Y., et al. Lycium barbarum polysaccharides protect human lens epithelial cells against oxidative stress-induced apoptosis and senescence. PLoS One . 2014;9, article e110275 doi: 10.1371/journal.pone.0110275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Xia G., Xin N., Liu W., Yao H., Hou Y., Qi J. Inhibitory effect of Lycium barbarum polysaccharides on cell apoptosis and senescence is potentially mediated by the p53 signaling pathway. Molecular Medicine Reports . 2014;9(4):1237–1241. doi: 10.3892/mmr.2014.1964. [DOI] [PubMed] [Google Scholar]
- 48.Liu L., Wang X. N., Liu Z., et al. Effect of lycium bararum polysaccharides on angiotensin II-induced senescence of human umbilical vein endothelial cells and expressions of p53 and p16. Nan Fang Yi Ke Da Xue Xue Bao . 2011;31(7):1212–1215. [PubMed] [Google Scholar]
- 49.Amagase H., Nance D. M. A randomized, double-blind, placebo-controlled, clinical study of the general effects of a standardized Lycium barbarum (goji) juice, GoChi. Journal of Alternative and Complementary Medicine . 2008;14(4):403–412. doi: 10.1089/acm.2008.0004. [DOI] [PubMed] [Google Scholar]
- 50.Amagase H., Sun B., Borek C. Lycium barbarum (goji) juice improves in vivo antioxidant biomarkers in serum of healthy adults. Nutrition Reserch . 2009;29(1):19–25. doi: 10.1016/j.nutres.2008.11.005. [DOI] [PubMed] [Google Scholar]
- 51.Ma L., Chen G., Nie L., Ai M. Effect of Cynomorium songaricum polysaccharide on telomere length in blood and brain of D-galactose-induced senescence mice. Zhongguo Zhong Yao Za Zhi . 2009;34(10):1257–1260. [PubMed] [Google Scholar]
- 52.Pan M. H., Wu J. C., Ho C. T., Badmaev V. Effects of water extract of Curcuma longa (L.) roots on immunity and telomerase function. Journal of Complementary Medicine . 2017;14(3) doi: 10.1515/jcim-2015-0107. [DOI] [PubMed] [Google Scholar]
- 53.Liu J., Xu C. Y., Cai S. Z., et al. Senescence effects of Angelica sinensis polysaccharides on human acute myelogenous leukemia stem and progenitor cells. Asian Pacific Journal of Cancer Prevention . 2014;14(11):6549–6556. doi: 10.7314/apjcp.2013.14.11.6549. [DOI] [PubMed] [Google Scholar]
- 54.Zhang X. P., Liu J., Xu C. Y., et al. Effect of Angelica sinensis polysaccharide on expression of telomere, telomerase and P53 in mice aging hematopoietic stem cells. Zhongguo Zhong Yao Za Zhi . 2013;38(14):2354–2358. [PubMed] [Google Scholar]
- 55.Djojosubroto M. W., Choi Y. S., Lee H. W., Rudolph K. L. Telomeres and telomerase in aging, regeneration and cancer. Molecules and Cells . 2003;15(2):164–175. [PubMed] [Google Scholar]
- 56.M'kacher R., Breton L., Colicchio B., et al. Benefit of an association of an antioxidative substrate and a traditional chinese medicine on telomere elongation. Cell Molecular Biology . 2020;65:54–58. [PubMed] [Google Scholar]
- 57.Huang C., Cao X., Chen X., et al. A pectic polysaccharide from Ligusticum chuanxiong promotes intestine antioxidant defense in aged mice. Carbohydrate Polymers . 2017;174:915–922. doi: 10.1016/j.carbpol.2017.06.122. [DOI] [PubMed] [Google Scholar]
- 58.Lei T., Li H., Fang Z., et al. Polysaccharides from Angelica sinensis alleviate neuronal cell injury caused by oxidative stress. Neural Regeneration Research . 2014;9(3):260–267. doi: 10.4103/1673-5374.128218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Cheng X., Yao H., Xiang Y., et al. Effect of Angelica polysaccharide on brain senescence of nestin-GFP mice induced by D-galactose. Neurochemistry International . 2019;122:149–156. doi: 10.1016/j.neuint.2018.09.003. [DOI] [PubMed] [Google Scholar]
- 60.Zhang M., Su N., Huang Q., et al. Phosphorylation and antiaging activity of polysaccharide from Trichosanthes peel. Journal of Food and Drug Analysis . 2017;25(4):976–983. doi: 10.1016/j.jfda.2016.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gan L., Zhang S. H., Liu Q., Xu H. B. A polysaccharide-protein complex from Lycium barbarum upregulates cytokine expression in human peripheral blood mononuclear cells. European Journal of Clinical Pharmacology . 2003;471(3):217–222. doi: 10.1016/S0014-2999(03)01827-2. [DOI] [PubMed] [Google Scholar]
- 62.Chen Z., Lu J., Srinivasan N., Tan B. K. H., Chan S. H. Polysaccharide-protein complex from Lycium barbarum L. is a novel stimulus of dendritic cell immunogenicity. Journal of Immunology . 2009;182(6):3503–3509. doi: 10.4049/jimmunol.0802567. [DOI] [PubMed] [Google Scholar]
- 63.Song J., Chen M., Li Z., et al. Astragalus polysaccharide extends lifespan via mitigating endoplasmic reticulum stress in the silkworm, Bombyx mori. Aging Disease . 2019;10(6):1187–1198. doi: 10.14336/AD.2019.0515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Doan V. M., Chen C., Lin X., et al. Yulangsan polysaccharide improves redox homeostasis and immune impairment in D-galactose-induced mimetic aging. Food & Function . 2015;6(5):1712–1718. doi: 10.1039/C5FO00238A. [DOI] [PubMed] [Google Scholar]
- 65.Li H., Ma F., Hu M., et al. Polysaccharides from medicinal herbs as potential therapeutics for aging and age-related neurodegeneration. Rejuvenation Reserch . 2014;17(2):201–204. doi: 10.1089/rej.2013.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Chuang M. H., Chiou S. H., Huang C. H., Yang W. B., Wong C. H. The lifespan-promoting effect of acetic acid and Reishi polysaccharide. Bioorganic and Medicinal Chemistry . 2009;17(22):7831–7840. doi: 10.1016/j.bmc.2009.09.002. [DOI] [PubMed] [Google Scholar]
- 67.Chang H. C., Guarente L. SIRT1 and other sirtuins in metabolism. Trends Endocrinology Metabolism . 2014;25(3):138–145. doi: 10.1016/j.tem.2013.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Dali-Youcef N., Lagouge M., Froelich S., Koehl C., Schoonjans K., Auwerx J. Sirtuins: the 'magnificent seven', function, metabolism and longevity. Annals of Medicine . 2007;39(5):335–345. doi: 10.1080/07853890701408194. [DOI] [PubMed] [Google Scholar]
- 69.Guarente L. Sirtuins in aging and disease. Cold Spring Harbor Symposia on Quantitative Biology . 2007;72(1):483–488. doi: 10.1101/sqb.2007.72.024. [DOI] [PubMed] [Google Scholar]
- 70.Wątroba M., Dudek I., Skoda M., Stangret A., Rzodkiewicz P., Szukiewicz D. Sirtuins, epigenetics and longevity. Ageing Research Reviews . 2017;40:11–19. doi: 10.1016/j.arr.2017.08.001. [DOI] [PubMed] [Google Scholar]
- 71.Zhao L., Cao J., Hu K., et al. Sirtuins and their biological relevance in aging and age-related diseases. Aging Disease . 2020;11(4):927–945. doi: 10.14336/AD.2019.0820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wang W., Liu F., Xu C., et al. Lactobacillus plantarum 69-2 combined with galacto-oligosaccharides alleviates d-galactose-induced aging by regulating the AMPK/SIRT1 signaling pathway and gut microbiota in mice. Journal of Agricultural and Food Chemistry . 2021;69(9):2745–2757. doi: 10.1021/acs.jafc.0c06730. [DOI] [PubMed] [Google Scholar]
- 73.Hu Z., Li F., Zhou X., et al. Momordica charantia polysaccharides modulate the differentiation of neural stem cells via SIRT1/Β-catenin axis in cerebral ischemia/reperfusion. Stem Cell Research and Therapy . 2020;11(1):p. 485. doi: 10.1186/s13287-020-02000-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ma J., Fan H., Cai H., et al. Promotion of Momordica charantia polysaccharides on neural stem cell proliferation by increasing SIRT1 activity after cerebral ischemia/reperfusion in rats. Brain Research Bulletin . 2021;170:254–263. doi: 10.1016/j.brainresbull.2021.02.016. [DOI] [PubMed] [Google Scholar]
- 75.Zhang S., Zhao Y. Lentinan protects cardiomyocytes against hypoxia-induced injury by regulation of microRNA-22/Sirt1. Artificial Cells Nanomed Biotechnology . 2019;47(1):3938–3946. doi: 10.1080/21691401.2019.1666863. [DOI] [PubMed] [Google Scholar]
- 76.Yao Q., Zhou Y., Yang Y., et al. Activation of Sirtuin1 by Lyceum barbarum polysaccharides in protection against diabetic cataract. Journal of Ethnopharmacology . 2020;261, article 113165 doi: 10.1016/j.jep.2020.113165. [DOI] [PubMed] [Google Scholar]
- 77.Zhu Y., Sun Y., Guan W., et al. Lycium barbarum polysaccharides enhances SIRT1 expression and decreases MMP-9 and HIF-1α expressions in hypoxic pulmonary vascular smooth muscle cells. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi . 2016;32(7):906–910. [PubMed] [Google Scholar]
- 78.Lei X., Huo P., Wang Y., et al. Lycium barbarum polysaccharides improve testicular spermatogenic function in streptozotocin-induced diabetic rats. Frontiers in Endocrinology . 2020;11, article 164 doi: 10.3389/fendo.2020.00164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Shi X., Wei W., Wang N. Tremella polysaccharides inhibit cellular apoptosis and autophagy induced by Pseudomonas aeruginosa lipopolysaccharide in A549 cells through sirtuin 1 activation. Oncology Letters . 2018;15(6):9609–9616. doi: 10.3892/ol.2018.8554. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 80.Huang Y. F., Lu L., Zhu D. J., et al. Effects of Astragalus polysaccharides on dysfunction of mitochondrial dynamics induced by oxidative stress. Oxidative Medicine and Cellular Longevity . 2016;2016:13. doi: 10.1155/2016/9573291.9573291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Gu C., Zeng Y., Tang Z., et al. Astragalus polysaccharides affect insulin resistance by regulating the hepatic SIRT1-PGC-1α/PPARα-FGF21 signaling pathway in male Sprague Dawley rats undergoing catch-up growth. Molecular Medicine Reports . 2015;12(5):6451–6460. doi: 10.3892/mmr.2015.4245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Peng Q. H., Tong P., Gu L. M., Li W. J. Astragalus polysaccharide attenuates metabolic memory-triggered ER stress and apoptosis via regulation of miR-204/SIRT1 axis in retinal pigment epithelial cells. Bioscience Reports . 2020;40(1) doi: 10.1042/BSR20192121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Liao Z., Zhang J., Liu B., et al. Polysaccharide from okra (Abelmoschus esculentus (L.) Moench) improves antioxidant capacity via PI3K/AKT pathways and Nrf2 translocation in a type 2 diabetes model. Molecules . 2019;24(10):p. 1906. doi: 10.3390/molecules24101906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Shen T., Duan C., Chen B., et al. Tremella fuciformis polysaccharide suppresses hydrogen peroxide-triggered injury of human skin fibroblasts via upregulation of SIRT1. M olecular Medicine Reports . 2017;16(2):1340–1346. doi: 10.3892/mmr.2017.6754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Chu Q., Zhang S., Yu L., et al. Apios americana Medikus tuber polysaccharide exerts anti-inflammatory effects by activating autophagy. International Journal of Biological Macromolecules . 2019;130:892–902. doi: 10.1016/j.ijbiomac.2019.03.012. [DOI] [PubMed] [Google Scholar]
- 86.Huang K., Fingar D. C. Growing knowledge of the mTOR signaling network. Seminars in Cell & Developmental Biology . 2014;36:79–90. doi: 10.1016/j.semcdb.2014.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Laplante M., Sabatini D. M. mTOR signaling in growth control and disease. Cell . 2012;149(2):274–293. doi: 10.1016/j.cell.2012.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Chiang G. G., Abraham R. T. Targeting the mTOR signaling network in cancer. Trends in Molecular Medicine . 2007;13(10):433–442. doi: 10.1016/j.molmed.2007.08.001. [DOI] [PubMed] [Google Scholar]
- 89.Sengupta S., Peterson T. R., Laplante M., Oh S., Sabatini D. M. mTORC1 controls fasting-induced ketogenesis and its modulation by ageing. Nature . 2010;468(7327):1100–1104. doi: 10.1038/nature09584. [DOI] [PubMed] [Google Scholar]
- 90.Zoncu R., Efeyan A., Sabatini D. M. mTOR: from growth signal integration to cancer, diabetes and ageing. Nature Reviews Molecular Cell Biology . 2011;12(1):21–35. doi: 10.1038/nrm3025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Lamming D. W., Ye L., Sabatini D. M., Baur J. A. Rapalogs and mTOR inhibitors as anti-aging therapeutics. Journal of Clinical Investigation . 2013;123(3):980–989. doi: 10.1172/JCI64099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Kaeberlein M., Kennedy B. K. Hot topics in aging research: protein translation and TOR signaling, 2010. Aging Cell . 2011;10(2):185–190. doi: 10.1111/j.1474-9726.2010.00665.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Cor D., Knez Z., Knez Hrncic M. Antitumour, antimicrobial, antioxidant and antiacetylcholinesterase effect of Ganoderma lucidum terpenoids and polysaccharides: a review. Molecules . 2018;23(3, article 649) doi: 10.3390/molecules23030649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Sohretoglu D., Huang S. Ganoderma lucidum polysaccharides as an anti-cancer agent. Anti-Cancer Agents in Medicinal Chemistry . 2018;18(5):667–674. doi: 10.2174/1871520617666171113121246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Liang Z., Yuan Z., Guo J., et al. Ganoderma lucidum polysaccharides prevent palmitic acid-evoked apoptosis and autophagy in intestinal porcine epithelial cell line via restoration of mitochondrial function and regulation of MAPK and AMPK/Akt/mTOR signaling pathway. International Journal of Molecular Science . 2019;20(3, article 478) doi: 10.3390/ijms20030478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Meng Q., Du X., Wang H., Gu H., Zhan J., Zhou Z. Astragalus polysaccharides inhibits cell growth and pro-inflammatory response in IL-1β-stimulated fibroblast-like synoviocytes by enhancement of autophagy via PI3K/AKT/mTOR inhibition. Apoptosis . 2017;22(9):1138–1146. doi: 10.1007/s10495-017-1387-x. [DOI] [PubMed] [Google Scholar]
- 97.Lu M. K., Lin T. Y., Chang C. C. Chemical identification of a sulfated glucan from Antrodia cinnamomea and its anti-cancer functions via inhibition of EGFR and mTOR activity. Carbohydrate Polymers . 2018;202:536–544. doi: 10.1016/j.carbpol.2018.09.009. [DOI] [PubMed] [Google Scholar]
- 98.Li X., Gong H., Yang S., Yang L., Fan Y., Zhou Y. Pectic bee pollen polysaccharide from Rosa rugosa alleviates diet-induced hepatic steatosis and insulin resistance via induction of AMPK/mTOR-mediated autophagy. Molecules . 2017;22(5):p. 699. doi: 10.3390/molecules22050699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Lee H., Kim J. S., Kim E. Fucoidan from seaweed Fucus vesiculosus inhibits migration and invasion of human lung cancer cell via PI3K-Akt-mTOR pathways. PLoS One . 2012;7(11, article e50624) doi: 10.1371/journal.pone.0050624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Cui H., Wu S., Shang Y., et al. Pleurotus nebrodensis polysaccharide (PN50G) evokes A549 cell apoptosis by the ROS/AMPK/PI3K/AKT/mTOR pathway to suppress tumor growth. Food & Function . 2016;7(3):1616–1627. doi: 10.1039/C6FO00027D. [DOI] [PubMed] [Google Scholar]
- 101.Mattaveewong T., Wongkrasant P., Chanchai S., Pichyangkura R., Chatsudthipong V., Muanprasat C. Chitosan oligosaccharide suppresses tumor progression in a mouse model of colitis-associated colorectal cancer through AMPK activation and suppression of NF-κB and mTOR signaling. Carbohydrate Polymers . 2016;145:30–36. doi: 10.1016/j.carbpol.2016.02.077. [DOI] [PubMed] [Google Scholar]
- 102.Carling D. AMPK signalling in health and disease. Current Opinion in Cell Biology . 2017;45:31–37. doi: 10.1016/j.ceb.2017.01.005. [DOI] [PubMed] [Google Scholar]
- 103.Herzig S., Shaw R. J. AMPK: guardian of metabolism and mitochondrial homeostasis. Nature Reviews Molecular Cell Biology . 2018;19(2):121–135. doi: 10.1038/nrm.2017.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Burkewitz K., Zhang Y., Mair W. B. AMPK at the nexus of energetics and aging. Cell Metabolism . 2014;20(1):10–25. doi: 10.1016/j.cmet.2014.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Zou F., Mao X. Q., Wang N., Liu J., Ou-Yang J. P. Astragalus polysaccharides alleviates glucose toxicity and restores glucose homeostasis in diabetic states via activation of AMPK. Acta Pharmaceutica Sinica B . 2009;30(12):1607–1615. doi: 10.1038/aps.2009.168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Liu D., Su J., Lin J., et al. Activation of AMPK-dependent SIRT-1 by astragalus polysaccharide protects against ochratoxin A-induced immune stress in vitro and in vivo. International Journal of Biological Macromolecules . 2018;120:683–692. doi: 10.1016/j.ijbiomac.2018.08.156. [DOI] [PubMed] [Google Scholar]
- 107.Lu J., Chen X., Zhang Y., et al. Astragalus polysaccharide induces anti-inflammatory effects dependent on AMPK activity in palmitate-treated RAW264.7 cells. International Journal of Molecular Medicine . 2013;31(6):1463–1470. doi: 10.3892/ijmm.2013.1335. [DOI] [PubMed] [Google Scholar]
- 108.Liao L. Z., Chen Y. L., Lu L. H., Zhao Y. H., Guo H. L., Wu W. K. Polysaccharide from Fuzi likely protects against starvation-induced cytotoxicity in H9c2 cells by increasing autophagy through activation of the AMPK/mTOR pathway. American Journal of Chinese Medicine . 2013;41(2):353–367. doi: 10.1142/S0192415X13500262. [DOI] [PubMed] [Google Scholar]
- 109.Yuan R., Tao X., Liang S., et al. Protective effect of acidic polysaccharide from Schisandra chinensis on acute ethanol-induced liver injury through reducing CYP2E1-dependent oxidative stress. Biomedicine & Pharmacotherapy . 2018;99:537–542. doi: 10.1016/j.biopha.2018.01.079. [DOI] [PubMed] [Google Scholar]
- 110.Che J., Yang S., Qiao Z., et al. Schisandra chinensis acidic polysaccharide partialy reverses acetaminophen-induced liver injury in mice. Journal of Pharmaceutical Sciences . 2019;140(3):248–254. doi: 10.1016/j.jphs.2019.07.008. [DOI] [PubMed] [Google Scholar]
- 111.Tan H., Chen W., Liu Q., Yang G., Li K. Pectin oligosaccharides ameliorate colon cancer by regulating oxidative stress- and inflammation-activated signaling pathways. Frontiers in Immunology . 2018;9:p. 1504. doi: 10.3389/fimmu.2018.01504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wang J., Li C., Hu W., et al. Studies on the anti-fatigue activities of Irpex lacteus polysaccharide-enriched extract in mouse model. Pakistan Journal of Pharmaceutical Sciences . 2019;32(3):1011–1018. [PubMed] [Google Scholar]
- 113.Zheng Y., Liu T., Wang Z., Xu Y., Zhang Q., Luo D. Low molecular weight fucoidan attenuates liver injury via SIRT1/AMPK/PGC1α axis in db/db mice. International Journal of Biological Macromolecules . 2018;112:929–936. doi: 10.1016/j.ijbiomac.2018.02.072. [DOI] [PubMed] [Google Scholar]
- 114.Wu Y., Zhou F., Jiang H., Wang Z., Hua C., Zhang Y. Chicory (Cichorium intybus L.) polysaccharides attenuate high-fat diet induced non-alcoholic fatty liver disease via AMPK activation. International Journal of Biological Macromolecules . 2018;118:886–895. doi: 10.1016/j.ijbiomac.2018.06.140. [DOI] [PubMed] [Google Scholar]
- 115.Bourgeois B., Madl T. Regulation of cellular senescence via the FOXO4-p53 axis. FEBS Letters . 2018;592(12):2083–2097. doi: 10.1002/1873-3468.13057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Wu Y. Y., Liang C. Y., Liu T. T., et al. Protective roles and mechanisms of polysaccharides from Dendrobium officinal on natural aging-induced premature ovarian failure. Biomedicine & Pharmacotherapy . 2018;101:953–960. doi: 10.1016/j.biopha.2018.03.030. [DOI] [PubMed] [Google Scholar]


