Abstract
The reaction of propargyl esters with alkynylsilanes under gold catalysis provides vinylallene derivatives through consecutive [1,2]‐acyloxy/[1,2]‐silyl rearrangements. Good yields, full atom‐economy, broad substrate scope, easy scale‐up and low catalyst loadings are salient features of this novel transformation. Density Functional Theory (DFT) calculations suggest a reaction mechanism involving initial [1,2]‐acyloxy rearrangement to generate a gold vinylcarbene intermediate which upon regioselective attack of the alkynylsilane affords a vinyl cation which undergoes a type II‐dyotropic rearrangement involving the silyl group and the metal fragment. Preliminary results on the enantioselective version of this transformation are also disclosed.
Keywords: alkynylsilanes, allenes, carbenes, gold, rearrangements
An efficient synthesis of structurally complex allenylsilanes through gold‐catalyzed coupling of propargyl esters with alkynylsilanes is described. DFT calculations support a stepwise pathway consisting of two 1,2‐rearrangements (acyloxy and silyl). A preliminary study on the enantioselective version would demonstrate its feasibility (er up to 95:5).
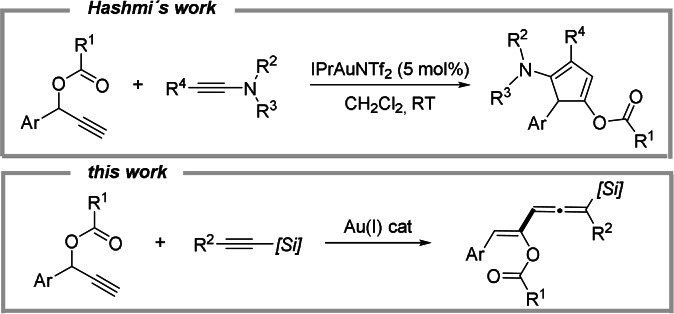
Metal‐carbenes have reached an indisputable position as intermediates in organic synthesis and catalysis. This is due not only to the ample spectrum of reactivity exhibited by these species but also to the wide variety of available precursors for their catalytic generation. [1] Among these carbenoid precursors, propargylic esters have found widespread uses, particularly in combination with gold‐based catalysts. [2] In these gold‐catalyzed transformations of propargylic esters, the generation of the carbene intermediate is proposed to proceed through initial coordination of the triple bond to the carbophilic catalyst followed by [1,2]‐acyloxy rearrangement. Although the actual structure of vinyl gold carbene intermediates was a debatable topic, nowadays it is well‐accepted that their structure depends largely on the carbene substitution and the ancillary ligands coordinated to the transition metal ranging from gold‐stabilized carbenes to allyl gold carbocations. [3] Leaving aside structural considerations, the reactivity of these intermediates has been extensively investigated providing access to a variety of products in most cases with high efficiency and selectivity. In this regard, whereas a diverse range of transformations has been developed employing alkenes [4] and dienes[ 4d , 5 ] as trapping reagents, methods based on the use of alkynes as counterparts remain, however, largely underdeveloped. In fact, although some intramolecular processes have been reported, the intermolecular trapping by alkynes of gold carbene intermediates generated from propargyl esters remains challenging.[ 6 , 7 ] To the best of our knowledge, the regioselective synthesis of functionalized cyclopentadienes by gold‐catalyzed cyclization of ynamides and propargyl esters reported by Hashmi and co‐workers in 2013 represents the only example of intermolecular interception of gold carbene intermediates arising from propargyl esters with a carbon‐carbon triple bond (Scheme 1, top). [8]
Scheme 1.
Intermolecular trapping of gold carbene intermediates arising from propargyl esters with alkynes.
Inspired by Hashmi's elegant work and as part of our on‐going interest in the reactivity of gold vinylcarbene precursors toward unsaturated organosilicon reagents, [9] we became interested in exploring the behavior of propargyl esters toward alkynylsilanes under gold catalysis. We hypothesized that the bulkiness of the silyl group in the alkynylsilane, which might hinder a cyclization reaction, together with the ability of the C‐Si σ‐bond to stabilize an adjacent carbocation (the so‐called β‐silicon effect), [10] could provide a favorable scenario for alternative pathways. We report herein that this transformation does not lead to [3+2] cycloadducts but to vinylallene derivatives (Scheme 1, bottom), which are not only common structural motifs in several natural products [11] but also valuable building blocks. [12] A detailed mechanism for this unusual double 1,2‐rearrangement, [13] supported by DFT calculations, is also proposed.
At the outset, we first studied the reaction of 1‐phenylprop‐2‐yn‐1‐yl acetate (1 a) and 1‐phenyl‐2‐trimethylsilylacetylene (2 a, 2 equiv) in the presence of a variety of AuIII and AuI catalysts (see Table S1 in the Supporting Information for a summary of the screening). To our delight, we found that commercially available IPrAuNTf2 (1 mol %) in DCE at 50 °C produced vinylallene derivative 3 a in 78 % isolated yield (Scheme 2). In agreement with previous gold‐catalyzed transformations of propargyl esters, compound 3 a was isolated as a single (Z)‐isomer.
Scheme 2.
Gold‐catalyzed reaction of propargyl ester 1 a and alkynylsilane 1 b: initial finding.
Interestingly, although the gold‐catalyzed cycloisomerization of vinylallenes to cyclopentadienes has been reported, [14] no cyclic isomers of vinylallene 3 a were observed. Also of note, attempted reaction of propargyl ester 1 a with 1‐phenyl‐1‐propyne failed to provide any product, thus revealing that the trimethylsilyl (TMS) group plays a crucial role in the reaction outcome.
Having established effective reaction conditions for the synthesis of 3 a, we explored then the scope of this transformation (Table 1). First, 1‐phenylprop‐2‐yn‐1‐yl pivalate (1 b, R2= t Bu) was also found to be amenable to the transformation providing the corresponding allene 3 b in good yield (69 %) when reacted with 2 a. On the other hand, propargyl esters with substituted aryl groups also engaged in this transformation as demonstrated by the synthesis of vinylallene 3 c in moderate yield.
Table 1.
Gold‐catalyzed reaction of propargyl esters 1 and alkynylsilanes 2: Substrate scope.[a,b]

[a] Reaction conditions: 1 (0.2 mmol), 2 (0.4 mmol), IPrAuNTf2 (1.0 mol %), DCE (1 mL), 50 °C. [b] Yield of isolated products. [c] 5 mol % of the catalyst was used.
Regarding the alkynylsilane component, we first demonstrated that the reaction also worked well when the TMS group was replaced with a tert‐butyldimethylsilyl (TBS) group, as illustrated the formation of allene derivatives 3 d–3 g in good to excellent yields (65–91 %) when tert‐butyldimethyl(phenylethynyl)silane (2 b) was reacted with a variety of differently substituted propargyl esters. Regarding the substitution on the aryl ring of the alkynylsilane component, we found that electron‐rich substrates such as those featuring p‐methyl‐ and p‐methoxyphenyl groups are particularly well‐suited to the process providing the corresponding products 3 h–3 m in good to excellent yields (69–98 %). p‐Halophenyl substituted alkynylsilanes also proved to be effective substrates for this transformation affording the expected products 3 n–3 s in moderate to good yields (58–88 %). Aryl‐substituted alkynylsilanes with electron‐withdrawing p‐methoxycarbonyl and p‐trifluoromethyl groups were also compatible with the present transformation delivering the corresponding allenylsilanes 3 t and 3 u in moderate yields (58 and 33 %, respectively). Conversely, the reactions with alkynylsilanes featuring p‐acetyl‐ and p‐nitrophenyl groups failed to deliver the expected products. Substituents at the meta position were well‐tolerated under the reaction conditions as demonstrated by the formation of vinylallene 3 v in good yield (74 %). In contrast, the use of an o‐methylphenyl group resulted in essentially no reaction. Pleasingly, a 1‐naphthyl‐substituted alkynylsilane was also an amenable reagent, providing the corresponding allenylsilane 3 w in 91 % yield. Interestingly, an alkynylsilane containing a ferrocenyl group proved to be a suitable substrate, delivering the expected product 3 x (41 %).
Interestingly, the reaction of propargyl esters 1 with alkynylsilanes 2 is not limited to the use of aryl‐substituted alkynylsilanes. For example, a representative alkenyl‐substituted alkynylsilane, namely (cyclohex‐1‐en‐1‐ylethynyl)trimethylsilane, was also a viable substrate in this transformation providing the respective bis‐vinylallene 3 y in a decent yield (58 %). Under the developed conditions, cycloalkyl‐substituted alkynylsilanes were also transformed into the corresponding allenylsilane derivatives 3 z and 3 aa, albeit in low isolated yields. In these cases, we found that increasing the catalyst loading to 5.0 mol % had a positive effect on the reaction outcome allowing the isolation of the desired products in good yields (73 % for 3 z and 66 % for 3 aa). Under otherwise similar conditions, the reaction of (oct‐1‐yn‐1‐yl)silane, proceeded sluggishly to afford the corresponding product 3 ab in lower isolated yield (36 %).
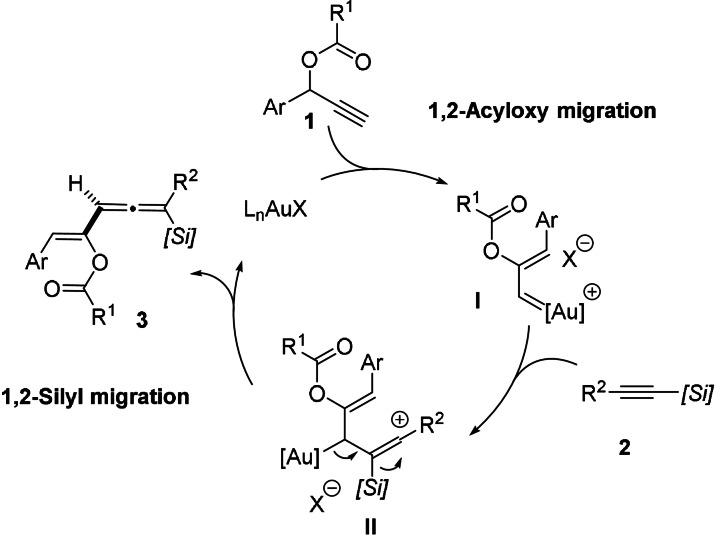
A possible pathway for the formation of allene derivatives 3 is depicted in Scheme 3. After the initial generation of gold carbene intermediate I through 1,2‐acyloxy shift, regioselective attack of the alkynylsilane to the electrophilic carbene carbon atom would generate cationic intermediate II, stabilized by the β‐silicon effect. Then, elimination of the [Au]+ fragment with concurrent 1,2‐silyl shift would account for the formation of the final product.
Scheme 3.
Proposed reaction mechanism for the gold(I)‐catalyzed reaction of propargylic esters 1 and alkynylsilanes 2.
To gain more insight into the reaction mechanism of this novel reaction, DFT calculations were carried out (see computational details in the Supporting Information). To this end, the transformation involving substrate 1 a and alkynylsilane 2 a leading to 3 a (see Scheme 2) catalyzed by a model cationic active AuI‐catalyst (where the IPr ligand was replaced by the 1,3‐bis‐phenyl‐imidazol‐2‐ylidene ligand) was computationally explored. The results are shown in Figure 1, which gathers the corresponding relative free energies (ΔG 298, at 298 K) in DCE as solvent (PCM‐B3LYP‐D3/def2‐TZVPP//PCM‐B3LYP‐D3/def2‐SVP level).
Figure 1.
Computed reaction profile for the gold‐catalyzed reaction of propargyl ester 1 a and alkynylsilane 2 a. Relative free energies (ΔG, at 298 K) and bond distances are given in kcal mol−1 and angstroms, respectively. All data have been computed at the PCM‐B3LYP‐D3/def2‐TZVPP//PCM‐B3LYP‐D3/def2‐SVP level.
As previously proposed, the process begins with the exergonic coordination of the C≡C triple bond of 1 a to the [Au]+ catalyst. The resulting initial intermediate INT0 undergoes a standard stepwise 1,2‐acycloxy shift via the five‐membered intermediate INT1 to produce INT2 in an exergonic transformation (ΔG R=−4.4 kcal mol−1, from INT0). The computed exergonicity and the low barriers involved in this initial rearrangement, which are similar to those reported for related systems, [15] are compatible with the mild reaction conditions used experimentally (see Table S1 in the Supporting Information). Once the gold(I)‐vinylcarbene INT2 is formed, the nucleophilic addition of the alkynylsilane 2 a takes place. Two possible additions can be envisaged, namely that involving the carbon atom attached to the TMS group (via TS3) and the analogous process involving the carbon atom attached to the phenyl group (via TS3′). Our calculations clearly indicate that the former process is kinetically favored over the latter (ΔΔG ≠=5.8 kcal mol−1), which is fully consistent with the complete regioselectivity observed experimentally. This regioselectivity is mainly due to the β‐effect of silicon which greatly stabilizes the benzylic carbocationic intermediate INT3. This species finally evolves into INT4 through the transition state TS4 with a very low barrier (ΔG ≠=3.0 kcal mol−1) and in a strongly exergonic transformation (ΔG R=−28.3 kcal mol−1). As depicted in Figure 1, the latter saddle point is associated with the 1,2‐shift of the TMS group and the concomitant migration of the transition‐metal fragment to the newly formed C=C double bond. Therefore, this step can be viewed as a type II‐dyotropic rearrangement, defined as the simultaneous and intramolecular migration of two atoms/groups without positional interchange. [16] Final decoordination releases the observed allene 3 a and the active [Au]+ catalyst, which can reenter into a new catalytic cycle.
According to our calculations, an alternative mechanism involving the initial formation of a silyl‐substituted cyclopropene intermediate and subsequent rearrangement to the allenylsilane seems unlikely. [17]
We next turned our attention to assessing the scalability of our transformation. Pleasingly, we found that the reaction of propargyl ester 1 b and alkynylsilane 2 e proceeded uneventfully on a 10 mmol‐scale, even when the catalyst loading was reduced to 0.4 mol %, delivering 3.93 g (85 %) of the corresponding silyl‐substituted vinylallene 3 m (Scheme 4 A).
Scheme 4.
A) Gram‐Scale Reaction. B) Product derivatization. C) Implementation to alkynylgermanes.
As an initial reactivity test, compound 3 m underwent easy protodesilylation to the corresponding allene 4 (89 %) when subjected to tetrabutylammonium fluoride (TBAF) in chloroform at room temperature (Scheme 4 B).
Then, we were intrigued as to whether this twofold 1,2‐rearrangement sequence could be extended to alkynylgermanes. Pleasingly, stirring propargyl ester 1 b with 1‐(triethylgermyl)‐2‐phenylethyne (5) in DCE at 50 °C in the presence of IPrAuNTf2 (5 mol %) resulted in the formation of the corresponding vinylallene derivative 6 in 40 % yield after chromatographic purification (Scheme 4 C). Although compound 6 was isolated in moderate yield, this result further demonstrates the ability of the germyl group to participate in a 1,2‐migration, a rather unusual process. [18]
Finally, we evaluated the viability of an enantioselective variant of our protocol. Surprisingly, despite the significant recent progress in enantioselective gold catalysis, [19] enantiocatalytic approaches to axially chiral compounds are scarce and limited to axially chiral biaryls resulting from intramolecular hydroarylation processes. [20] To the best of our knowledge, the gold‐catalyzed enantioselective synthesis of allene derivatives remains elusive, [21] perhaps because of issues associated with gold‐catalyzed allene racemization. [22]
A preliminary evaluation of chiral gold(I) complexes led us to identify the phosphoramidite‐gold complex depicted in Table 2 as a promising catalyst (see the Supporting Information for screening details). Thus, using AgNTf2 as halide abstractor in DCE as solvent at 0 °C, the desired vinylallenes 3 were obtained in moderate to good yields (51–85 %) and with enantiomeric ratios (er) up to 95:5.
Table 2.
Preliminary study on the development of an enantioselective version.[a]

|
R1; R2; R3 |
[Si] |
3 |
t [h] |
Yield [%][b] |
er[c] |
|---|---|---|---|---|---|
|
Ph; Me; Ph |
TMS |
3 a |
24 |
62 |
52:48 |
|
Ph; Me; Ph |
TBS |
3 d |
4 |
51 |
82:18 |
|
Ph; t Bu; Ph |
TBS |
3 e |
24 |
62 |
77:23 |
|
Ph; t Bu; p‐Tolyl |
TBS |
3 j |
24 |
57 |
84:16 |
|
Ph; Me; p‐MeOC6H4 |
TBS |
3 k |
4 |
75 |
85:15[d] |
|
p‐Tolyl; Me; p‐MeOC6H4 |
TBS |
3 l |
15 |
67 |
95:5[d] |
|
Ph; t Bu; p‐MeOC6H4 |
TBS |
3 m |
20 |
85 |
92:8 |
[a] (Ar=6‐Methoxynaphthalen‐2‐yl). [b] Yield of isolated product. [c] Determined by chiral HPLC analysis. [d] 99:1 er after crystallization from pentane.
These preliminary results seem to suggest that the structure of the alkynylsilane plays an important role in the enantioselectivity of the reaction. Specifically, TBS‐substituted alkynylsilanes provided much better er values than those featuring a TMS group. Although further studies are required to elucidate the actual structural factors governing the enantioselectivity of this process, this exploratory study would demonstrate the feasibility of accessing axially chiral compounds in enantioenriched form, a challenging process in asymmetric gold catalysis.
In summary, we have reported an efficient and perfectly atom‐economical approach to functionalized vinylallene derivatives from readily available propargyl esters and alkynylsilanes. This novel transformation represents an unusual example of intermolecular trapping of a gold carbene species with alkynes. DFT calculations are consistent with a stepwise mechanism involving the regioselective attack of the alkynylsilane to the gold carbene intermediate and subsequent dyotropic rearrangement of the resulting cationic intermediate as key steps. Because of its scalable character and the promising results achieved in the enantioselective version, the reported protocol could serve as a convenient platform for accessing enantiomerically enriched compounds by exploiting the multiple potential reactive sites present in the available products. Further investigations toward this objective are currently in progress in our group.
Conflict of interest
The authors declare no conflict of interest.
Supporting information
As a service to our authors and readers, this journal provides supporting information supplied by the authors. Such materials are peer reviewed and may be re‐organized for online delivery, but are not copy‐edited or typeset. Technical support issues arising from supporting information (other than missing files) should be addressed to the authors.
Supporting Information
Acknowledgements
Financial support from Ministerio de Ciencia e Innovación, Agencia Estatal de Investigación (AEI) and Fondo Europeo de Desarrollo Regional (FEDER) (Grants PID2019‐107469RB‐I00, PID2019‐106184GB‐I00 and RED2018‐102387‐T) is gratefully acknowledged. We would also like to acknowledge the technical support provided by Servicios Científico‐Técnicos de la Universidad de Oviedo. We also thank Prof. J. M. González for helpful discussions.
O. Bernardo, S. González-Pelayo, I. Fernández, L. A. López, Angew. Chem. Int. Ed. 2021, 60, 25258.
Dedicated to Professor Gerhard Erker on occasion of his 75th birthday
Contributor Information
Prof. Israel Fernández, Email: israel@quim.ucm.es.
Prof. Luis A. López, Email: lalg@uniovi.es.
References
- 1.Reviews on general aspects of the chemistry of carbenes:
- 1a. Moss R. A., Doyle M. P., Contemporary Carbene Chemistry, Wiley, New York, 2013; [Google Scholar]
- 1b. Jia M., Ma S., Angew. Chem. Int. Ed. 2016, 55, 9134–9166; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2016, 128, 9280–9313. [Google Scholar]
- 2.Selected reviews on metal-catalyzed rearrangements of propargyl esters:
- 2a. Kazem Shiroodi R., Gervorgian V., Chem. Soc. Rev. 2013, 42, 4991–5001; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2b. Marion N., Nolan S. P., Angew. Chem. Int. Ed. 2007, 46, 2750–2752; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2007, 119, 2806–2809; [Google Scholar]
- 2c. Fürstner A., Davies P. W., Angew. Chem. Int. Ed. 2007, 46, 3410–3449; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2007, 119, 3478–3519. [Google Scholar]
- 3.For a seminal contribution on the structure of gold carbene intermediates, see: Benitez D., Shapiro N. D., Tkatchouk E., Wang Y., W. A. Goddard III , Toste F. D., Nat. Chem. 2009, 1, 482–486. For an interesting discussion about the structure of gold carbene intermediates, see: [DOI] [PMC free article] [PubMed] [Google Scholar]; Wang Y., Muratore M. E., Echavarren A. M., Chem. Eur. J. 2015, 21, 7332–7339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.
- 4a. Johansson M. J., Gorin D. J., Staben S. T., Toste F. D., J. Am. Chem. Soc. 2005, 127, 18002–18003; [DOI] [PubMed] [Google Scholar]
- 4b. Gorin D. J., Watson I. D. G., Toste F. D., J. Am. Chem. Soc. 2008, 130, 3736–3737; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4c. Watson I. D. G., Ritter S., Toste F. D., J. Am. Chem. Soc. 2009, 131, 2056–2057; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4d. Garayalde D., Krüger K., Nevado C., Angew. Chem. Int. Ed. 2011, 50, 911–915; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2011, 123, 941–945; [Google Scholar]
- 4e. Wagh S. B., Liu R.-S., Chem. Commun. 2015, 51, 15462–15464. [DOI] [PubMed] [Google Scholar]
- 5.For the use of 1-azadienes as trapping reagents, see:
- 5a. Shapiro N. D., Toste F. D., J. Am. Chem. Soc. 2008, 130, 9244–9245; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5b. Liu H., Li X., Chen Z., Hu W.-X., J. Org. Chem. 2012, 77, 5184–5190. [DOI] [PubMed] [Google Scholar]
- 6.The general reluctance of alkynes to participate in intermolecular gold-catalyzed transformations has been documented: Muratore M. E., Homs A., Obradors C., Echavarren A. M., Chem. Asian J. 2014, 9, 3066–3082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.For a recent review on the reactivity of gold carbene intermediates toward alkynes, see: Sancheti S. P., Patil N. T., Eur. J. Org. Chem. 2021, 1321–1330. [Google Scholar]
- 8. Rettenmeier E., Schuster A. M., Rudolph M., Rominger F., Gade C. A., Hashmi A. S. K., Angew. Chem. Int. Ed. 2013, 52, 5880–5884; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2013, 125, 5993–5997. [Google Scholar]
- 9.Recently, we have reported the synthesis of skipped dienes and enynes by reaction of vinyldiazo compounds and alkenyl- and alkynylsilanes under gold catalysis: Bernardo O., Yamamoto K., Fernández I., López L. A., Org. Lett. 2021, 23, 4452–4456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Lambert J. B., Zhao Y., Emblidge R. W., Salvador L. A., Liu X., So J.-H., Chelius E. C., Acc. Chem. Res. 1999, 32, 183–190. [Google Scholar]
- 11.Review: Hoffmann-Röder A., Krause N., Angew. Chem. Int. Ed. 2004, 43, 1196–1216; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2004, 116, 1216–1236. [Google Scholar]
- 12.For a recent review on the synthesis and reactivity of vinylallenes, see: Afonso M. M., Palenzuela J. A., Curr. Org. Chem. 2019, 23, 3004–3026. [Google Scholar]
- 13.For a recent review on 1,2-silyl shift, see: Beļaunieks R., Puriņš M., Turks M., Synthesis 2020, 52, 2147–2161. [Google Scholar]
- 14.Seminal work: Lee J. H., Toste F. D., Angew. Chem. Int. Ed. 2007, 46, 912–914; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2007, 119, 930–932; For a recent example, see: [Google Scholar]; Chen C.-N., Liu R.-S., Angew. Chem. Int. Ed. 2019, 58, 9831–9835; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2019, 131, 9936–9940. [Google Scholar]
- 15.See, for instance:
- 15a. Correa A., Marion N., Fensterbank L., Malacria M., Nolan S. P., Cavallo L., Angew. Chem. Int. Ed. 2008, 47, 718–721; [DOI] [PubMed] [Google Scholar]; Angew. Chem. 2008, 120, 730–733; [Google Scholar]
- 15b. Jiang J., Liu Y., Hou C., Li Y., Luan Z., Zhao C., Ke Z., Org. Biomol. Chem. 2016, 14, 3558–3563. [DOI] [PubMed] [Google Scholar]
- 16.
- 16a. Reetz M. T., Angew. Chem. Int. Ed. Engl. 1972, 11, 130–131; [Google Scholar]; Angew. Chem. 1972, 84, 163–163; [Google Scholar]
- 16b. Fernández I., Cossío F. P., Sierra M. A., Chem. Rev. 2009, 109, 6687–6711; [DOI] [PubMed] [Google Scholar]
- 16c. Croisant M. F., Hoveln R. V., Schomaker J. M., Eur. J. Org. Chem. 2015, 5897–5907. [Google Scholar]
- 17.For the PtCl2-catalyzed rearrangement of silylated cyclopropenes to silylallenes, see: Li J., Sun C., Demerzhan S., Lee D., J. Am. Chem. Soc. 2011, 133, 12964–12967. [DOI] [PubMed] [Google Scholar]
- 18.There have been isolated reports of 1,2-migrations of a germyl group:
- 18a. Seregin I. V., Gevorgyan V., J. Am. Chem. Soc. 2006, 128, 12050–12051; [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18b. Seregin I. V., Schammel A. W., Gevorgyan V., Tetrahedron 2008, 64, 6876–6883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.For selected reviews on asymmetric gold catalysis, see:
- 19a. Zi W., Toste F. D., Chem. Soc. Rev. 2016, 45, 4567–4589; [DOI] [PubMed] [Google Scholar]
- 19b. Li Y., Li W., Zhang J., Chem. Eur. J. 2017, 23, 467–512; [DOI] [PubMed] [Google Scholar]
- 19c. Jiang J.-J., Wong M.-K., Chem. Asian J. 2021, 16, 364–377; [DOI] [PubMed] [Google Scholar]
- 19d. Zuccarello G., Escofet I., Caniparoli U., Echavarren A. M., ChemPlusChem 2021, 86, 1283–1296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Selected examples:
- 20a. Zhang J., Simon M., Golz C., Alcarazo M., Angew. Chem. Int. Ed. 2020, 59, 5647–5650; [DOI] [PMC free article] [PubMed] [Google Scholar]; Angew. Chem. 2020, 132, 5696–5699; [Google Scholar]
- 20b. Satoh M., Shibata Y., Kimura Y., Tanaka K., Eur. J. Org. Chem. 2016, 4465–4469; [Google Scholar]
- 20c. Shibuya T., Nakamura K., Tanaka K., Beilstein J. Org. Chem. 2011, 7, 944–950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.The asymmetric isomerization of alkyne to allene and subsequent intramolecular trapping has been reported: Cheng X., Wang Z., Quintanilla C. D., Zhang L., J. Am. Chem. Soc. 2019, 141, 3787–3791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.For a mechanistic study on the gold-catalyzed racemization of axially chiral 1,3-disubstituted allene derivatives, see: Li H., Harris R. J., Nakafuku K., Widenhoefer R. A., Organometallics 2016, 35, 2242–2248. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
As a service to our authors and readers, this journal provides supporting information supplied by the authors. Such materials are peer reviewed and may be re‐organized for online delivery, but are not copy‐edited or typeset. Technical support issues arising from supporting information (other than missing files) should be addressed to the authors.
Supporting Information