Abstract
The vasculature plays an essential role in the development and maintenance of blood-tissue interface homeostasis. Knowledge on the morphological and functional nature of the blood vessels in every single tissue is, however, very poor, but it is becoming clear that each organ is characterized by the presence of endothelial barriers with different properties fundamental for the maintenance of tissue resident immune homeostasis and for the recruitment of blood-trafficking immune cells. The tissue specificity of the vascular unit is dependent on the presence of differentiated endothelial cells that form continues, fenestrated, or sinusoidal vessels with different grades of permeability and different immune receptors, according to how that particular tissue needs to be protected. The gut-brain axis highlights the prominent role that the vasculature plays in allowing a direct and prompt exchange of molecules between the gut, across the gut vascular barrier (GVB), and the brain. Recently, we identified a new choroid plexus vascular barrier (PVB) which receives and integrates information coming from the gut and is fundamental in the modulation of the gut-brain axis. Several pathologies are linked to functional dysregulation of either the gut or the choroid plexus vascular barriers. In this review, we unveil the structural and functional analogies between the GVB and PVB, comparing their peculiar features and highlighting the functional role of pitcher and catcher of the gut-brain axis, including their role in the establishment of immune homeostasis and response upon systemic stimuli. We propose that when the gut vascular barrier—the main protecting system of the body from the external world—is compromised, the choroid plexus gatekeeper becomes a second barrier that protects the central nervous system from systemic inflammation.
Keywords: Gut vascular barrier, Choroid plexus vascular barrier, Gut-brain axis, Endothelium, Inflammatory bowel disease
Introduction
The gut-brain axis is characterized by molecular and functional connections between the gut and the brain. In particular, the best documented network is formed by the signaling occurring between the central nervous system (CNS), the enteric nervous systems, and the hypothalamic–pituitary–adrenal axis [1]. The enteric nervous system, residing in the intestinal wall, can communicate with the CNS through the neuroimmune and neuroendocrine signaling mediated by the vagus nerve [1]. However, the CNS has also to maintain tissue homeostatic signaling and nutrient exchange and, at the same time, protect itself from infectious agents, toxins, and inflammation, thus requiring to rapidly sense changes of milieu in the bloodstream. This evidence is changing the paradigm in which the CNS is described as an “immune-privileged” site. Nowadays, a number of studies highlight the dynamic process that intervenes between immune sentinels within the CNS and the periphery [2]. Indeed, the brain continuously receives signals from the periphery [3–12] presumably also from the microbiota, as germ-free animals have impaired brain functions [13]. In agreement, a hyper-ramified morphology of microglia, the major antigen-presenting cells in the brain, is detected in germ-free mice [14].
During enteric microbial dysbiosis and alteration of mucosal barrier permeability, the translocation of bacterial metabolites, microbes, inflammatory molecules, toxins, and immune cells across the gut vascular barrier (GVB) into the blood circulation may lead to neuroinflammation. Indeed, we recently reported that alterations of the GVB lead to leakage of bioactive systemic molecules modulating choroid plexus vascular barrier (PVB) accessibility and causing an impairment of brain immune homeostasis, thus leading to structural and functional alterations in the CNS [5].
When immunosurveillance of the brain is compromised, pathology may arise [15–19]. Thus, an organ often thought to be isolated from the rest of the organism receives and sends signals to the periphery. The nature of these signals is under intense investigation, and there is mounting evidence on a deregulation of the gut-brain-microbiota-immune-mediated network in the development of gastrointestinal disorders [20–24] and systemic [25–28] and neurological [10, 24, 29, 30] pathologies. Understanding the blood circulatory signaling which connects the gut and the brain will offer an insight into novel therapeutic approaches that may improve the outcome of different pathologies such as intestinal bowel diseases (IBDs) [31], Parkinson’s and Alzheimer’s diseases [31–34], multiple sclerosis [35], autism spectrum disorders [30, 36], and brain metastatic processes [37].
In this review, we will discuss the gut and choroid plexus vascular barriers and their communications through the systemic circulation as gatekeepers in maintaining immune cell homeostasis and surveillance, and cell trafficking into the CNS.
The gut vascular barrier
The exposure to trillions of microorganisms in the gut poses a tremendous pressure to our organism to maintain immune homeostasis. The major challenge is to preserve sealing of the intestinal barrier to avoid bacterial translocation [38]. This is achieved via three physical structures, which have different grades of accessibility to bacteria and their metabolites, food antigens, and toxins [39].
The first element is the mucus layer, which generates a size exclusion matrix containing antibacterial molecules such as IgA, defensins, and lysozyme [40, 41] and separates the microbiota from the second element composed of epithelial cells, which sealed one to the other by tight junctions [42]. We described the existence of a third element just below the epithelium, the gut vascular barrier [43–45], which is organized in a vascular unit resembling that of the blood–brain barrier (BBB). The tissue-specific structural morphology of the blood vessels is due to the presence of differentiated endothelial cells [46] and other specialized cells surrounding the endothelium such as pericytes and epithelial cells. The complex network established between different structural tissue layers match with specific needs of signaling and protection peculiar for each blood-tissue interface. In particular, the gut vascular barrier is a more permissive barrier allowing the passage of molecules as large as 4 kDa while the BBB allows molecules smaller than 500 Da [47–49]. Thus, most of the interaction with the microbiota is not physical but is dependent on microbial metabolites (postbiotics) or products, including short- and long-chain fatty acids, small peptides, neurotransmitters (e.g., acetylcholine, serotonin, dopamine, gamma-aminobutyric acid), and vitamins [50–53]. These microbial metabolites are important regulators of the immune system [21], and some of them can cross the BBB [54], while others may translocate via the newly identified choroid plexus vascular barrier [5].
The gut vascular barrier is composed of a layer of endothelial cells surrounded by pericytes and enteric glial cells, the equivalent of astrocytes in the brain. Endothelial cells are sealed one to the other by tight junctions and are fenestrated; however, the fenestrae are closed by the endothelial cell–specific plasmalemma vesicle–associated protein (PV-1) [43–45, 55–58]. PV-1 is a crucial molecule in several membrane structures for the development of diaphragms, including caveolae, trans-endothelial channels, and fenestrae [59–61]. PV-1 is detected by the MECA-32 and PAL-E antibodies, for murine and human antigen detection, respectively [62, 63]. An increase in PV-1 detection highlights an enhancement of vascular permeability, probably because the epitope of the molecule becomes more accessible to the antibody through a conformational change [64].
Furthermore, PV-1 is described as a crucial protein involved in the trans-endothelial migration of leukocytes from the blood circulation to the tissue [65, 66]. Thus, the blockade of PV-1 reduces the transcellular migration of leukocytes through the endothelium [65].
The impairment of the gut vascular barrier, linked to the upregulation of PV-1, triggers the upregulation of the expression of adhesion molecules on the endothelium, stimulating the recruitment of blood-trafficking leukocytes and the activation of the intestinal immune response [67].
The gut immune barrier
The gut is constantly exposed to a plethora of different antigens coming from commensal bacteria, pathogens, and foods. To maintain intestinal homeostasis, the defense layers of mucus, epithelium, and endothelium control antigen exposure to the immune system [68]. Human intestinal resident immune cells are heterogeneously distributed for frequency and localization along the intestinal tract and are changing their dynamics during life [69].
The epithelial layer is populated by intraepithelial lymphocytes, mostly gamma delta (γδ) and unconventional T cells, while the lamina propria is home to innate myeloid and lymphoid cells [69, 70]. Intraepithelial T cells mainly consist of CD8+ T cells expressing the CD103 ligand for E-cadherin, which facilitate the adhesion of these cells to the epithelium. CD8+ intraepithelial lymphocytes are involved in the preservation of intestinal epithelial barrier integrity by executing effector functions against pathogens and cooperate to tissue renewal [71, 72]. However, the major fraction of intestinal resident T cells is localized in the intestinal lamina propria and includes CD4+ T regulatory cells, which act as important regulators of the immune response to food and microbial antigens and mediators of tissue homeostasis via the secretion of IL-10 and TGF-β and the inhibition of Th1/Th2/Th17 immune cells [73].
To maintain the dynamic equilibrium between the establishment of oral tolerance and active response against microbial pathogens, gut resident immune cells, also defined as intestinal barrier–associated immune cells, exploit their role of instructors and sentinels.
Within the resident immune cell population, dendritic cells (DCs) are fundamental for the establishment of both oral tolerance [74] and activation of immune response against pathogens [75, 76].
Subsets of functional distinct dendritic cells contribute to the maintenance of the divalent regulatory and effector functions of the gut immune system by extending dendrites through microfold cells and sampling intestinal antigens. CD103+ DCs are involved in the establishment of tolerance and in the generation of primed gut regulatory T cells [74, 77, 78]. To induce the differentiation of regulatory T cells, CD103+ migratory DCs can receive luminal antigens from CX3CR1+ mononuclear phagocytes [79] and migrate to mesenteric lymph node [80].
CX3CR1+ resident macrophages represent the most abundant immune resident population in the lamina propria. Subepithelial and perivascular resident macrophages are phagocytic cells which express TJ proteins to maintain barrier integrity while sampling of luminal antigens [79] and perform a quick clearance of invading microbes. Once activated, CX3CR1+ cells can induce Th17-mediated responses against pathogens [76, 81]. However, they display a reduced capacity to mount the inflammatory response [82, 83]. Regulatory macrophages are constantly monitoring T cell differentiation through IL-10- and stat3-mediated mechanisms in response to microbial load [84].
At the apical surface of the epithelial layer, adhesion molecules are expressed as intercellular adhesion molecule-1 (ICAM-1). Upon inflammation, epithelial expression of ICAM-1 is increased and mediates the recruitment and retention of neutrophils at the luminal side of the colon. The signaling activated by ICAM-1 is necessary for neutrophil activity and for the activation of epithelial reparative response [85].
Thus, epithelial cells localized at the frontline between microbial luminal side and lamina propria are key players in the regulation of mucosal immune responses. The expression of different receptors allows the cooperation between the activation of pathogen-associated molecular patterns (PAMPs) and the regulatory and effective response of immune cells [86].
The function of the gut vascular barrier
The gut vascular barrier controls the entry of dietary antigens, the microbiota, and its products, into the portal circulation and, subsequently, the liver [43–45]. If a microbe accidentally or purposely crosses the epithelial barrier, it will not reach the systemic circulation unless it can dismantle the gut vascular barrier. For instance, the enteric pathogen Salmonella Typhimurium has evolved strategies to elude the gut vascular barrier by interfering with the Wnt/β-catenin signaling pathway [43, 44]. In addition, the GVB is also disrupted in some pathological non-infectious conditions such as in celiac disease [43], non-alcoholic steatohepatitis (NASH) [45], experimental cirrhosis [55], metastatic colorectal cancer [57], and ankylosing spondylitis [58]. For NASH and colorectal cancer, the alteration of the gut vascular barrier is dependent on a dysbiotic microbiota [57]. We demonstrated that a change in microbiota composition can dismantle the GVB, resulting in the translocation of bacteria or bacterial endotoxins into the portal circulation, which results in chronic inflammation of distal organs. Indeed, using a mouse model in which we stabilized the endothelium by maintaining β-catenin in an activated state only in endothelial cells (β-cateninlox(ex3)/lox(ex3) CRE+ mice) or by pharmacological intervention with obeticholic acid, a bile acid analogue, we did not allow the derangement of the gut vascular barrier [43–45], the translocation of bacterial products, and the establishment of NASH and cirrhosis [45, 55]. Hence, the vascular permeability of the GVB regulates the gut-liver axis and it can control systemic inflammation. As recently published, the gut vascular barrier seems to be crucial also in the maintenance of the gut-brain axis. Indeed, an intestinal inflammation could lead to the impairment of the choroid plexus vascular barrier and the modulation of brain permeability [5].
Immunomodulatory functions of intestinal endothelial cells
The endothelium-forming blood vessels do not only represent the first barrier that peripheral immune cells have to overcome before accessing tissues and organs, but it can serve as an activator of the immune response. Single cell–based analysis on endothelial cells reported a tissue-specific transcriptional signature that reveals a differential expression of cytokines and chemokines, suggesting a strict connection with their immunomodulatory functions [87, 88].
In physiological conditions, the endothelial vascular barrier preserves immune homeostasis by regulating its permeability, trafficking of leukocytes, and maintaining immune cells in a silenced state in the colon. In pathological conditions, vascular endothelial cells of the gut can sense invading pathogens and can activate PAMP-mediated innate immune responses, inducing the intestinal recruitment of leukocytes and macrophages [89]. In the preclinical model of DSS-mediated ulcerative colitis, gut vascular barrier impairment, highlighted by the enhancement of PV-1 detection, is correlated to the recruitment of colonic lamina propria monocytes, neutrophils, and macrophages [5]. In IBD patients, endothelial cells are described to upregulate cell adhesion molecules and release cytokines and chemokines followed by the recruitment of neutrophils, monocytes, and lymphocytes into the lamina propria [90, 91].
Endothelial cells constitutively express innate immune Toll-like receptors (TLRs) which favor the adhesion of immune cells [92] and the expression of pro-inflammatory cytokines [93]. In particular, LPS and flagellin–mediated activation of endothelial cells leads to the upregulation of ICAM-1 which binds leukocyte β2-integrins, promoting the leukocyte adhesion to endothelium and their extravasation through the trans-endothelial route [94–96]. Thus, as reported in IBD patients, there is a correlation between the upregulation of the endothelial ICAM-1 expression and the amount of infiltrating lymphocytes that occurs near colonic crypt abscesses and ulcerated sites [97]. However, as endothelial cells express both MHC class I and II molecules, they could interact directly with cytotoxic CD8+ and helper CD4+ T lymphocytes, protecting the tissue from viral and bacterial infections but also potentially contributing to T cell–mediated autoimmunity [98, 99]. All these factors can contribute to the migration of immune cells within the intestinal lamina propria. Deeper analyses on the crosstalk between PV-1, ICAM-1, and the expression of MHCI/II by gut endothelial cells could bring forward on processes involved in the regulation of intestinal leukocyte recruitment and activation.
The blood-cerebrospinal fluid barrier
The homeostasis of the central nervous system is maintained by the BBB, the meningeal barrier, and the blood-cerebrospinal fluid barrier (BCSFB). Meninges, BBB, and BCSFB control the exchange of nutrients, metabolic solutes, hormones, metabolites, infectious agents, toxins, and waste, from and to the brain interstitium, the cerebrospinal fluid, and the bloodstream [100, 101]. The intrinsic nature of cellular composition and permeability of the endothelium differs drastically between these three barriers, reflecting the different degrees of freedom necessary to balance the exchange and the protection needed to maintain the milieu of the CNS [102]. Here, we will describe only the blood-cerebrospinal fluid barrier which is the barrier that seems to play a role in both immune surveillance and protection of the brain [18].
The vascular unit of the choroid plexus and its associated vascular barrier resides in the deepest layer of the pia mater and is formed by a complex network of fenestrated capillaries, which allow the selective passage of bioactive molecules coming from the systemic blood circulation [103–105]. The choroid plexus represents the interface between the blood and the cerebrospinal fluid (CSF), and it can act as a gatekeeper in the nutritional, metabolic, and hormonal balance, as well as in immune surveillance and trafficking [106]. The effectiveness of the blood-cerebrospinal fluid barrier is essential to protect against microbes and toxins [107], and its disruption is involved in neuroinflammation and brain injury [108–110].
Till now, choroid plexus vascular units, matching with the high detection of PV-1, were considered as an “always permissive” endothelial layer from which the exchange of blood-derived molecules occurs. In the structure of the choroid plexus, the epithelial layer surrounding the vessels was described as the unique selective interface area between the choroid plexus and the CSF. Thus, the epithelium of the choroid plexus was described as the unique defense layer, constituting the BCSFB. Nevertheless, we recently described a different role of the choroid plexus vascular unit, indicating that during an intestinal inflammation, the endothelial cells decrease PV-1 detection mirroring the closure of fenestrated vessels and becoming a selective and effective barrier which modulates CSF composition and immune cell translocation. These data shed light on a new anatomical and functional feature of the choroid plexus vascular barrier which can modify its permeability affecting the homeostasis of the gut-brain interaction [5].
The complex structure of the blood-cerebrospinal fluid barrier makes the PVB the principal barrier involved in the interface between the blood and the brain [106].
The choroid plexus vascular barrier
Conversely to the BBB and the gut vascular barrier, the choroid plexus microvasculature is fenestrated and permeable. The fenestrae are connected by diaphragms permissive to 70 kDa molecules [5], solutes, and water. The access of blood-derived molecules could happen through diffusion across endothelial fenestrae or by vesicular transport [111]. Choroid plexus blood vessels are positive for PV-1 detection at steady state; however, the vascular accessibility can be modified during systemic inflammation [5].
Our recent report demonstrates that, upon intestinal inflammation and systemic infection triggered by lipopolysaccharide injection, the endothelium of the choroid plexus acts properly as a barrier by modulating its accessibility. In particular, we showed that during intestinal inflammation, the gut vascular barrier is disrupted allowing the passage of unwanted molecules from the gut into the systemic circulation and this leads first to a strong decrease in PV-1 detection paralleled by a reduction of 70 kDa access and accumulation in the stroma of the choroid plexus. Consistently, inflammatory monocytes, macrophages, and neutrophils seed the brain quickly after intestinal inflammation and gut vascular barrier impairment [5].
The immunoregulatory function of the choroid plexus
The anatomical position of the choroid plexus as a frontline with the blood circulation makes this site a gateway for blood molecules and circulating immune cells trafficking from the periphery, and it is essential for the maintenance of CNS immune homeostasis [16–19, 112–116]. Choroid plexus endothelial cells and resident immune cells are readily positioned to defend against systemic microbial antigens and to sense pro-inflammatory molecules [117, 118].
Within the choroid plexus, resident immune cells, macrophages, and dendritic cells are the most abundant and they can act as immune sentinels performing immune surveillance and activation through the release of pro- and anti-inflammatory cytokines and chemokines [119]. However, by single cell RNA sequencing across all ages (embryonic, adult, and aged mice), the presence of resident neutrophils, plasma cells, and lymphoid B and T cells was also shown [120].
Resident macrophages of the choroid plexus (CP) are defined as either stromal macrophages or CNS border-associated macrophages (BAMs), based on distinct localization, transcriptional signature, and functions [120, 121]. Stromal macrophages are highly associated with the choroid plexus vascular barrier and perform sampling from the blood. BAMs localized on the apical surface of the epithelial layer of the choroid plexus present with immunomodulatory activity in response to peripheral stimuli and migrate along its surface to present antigens to T cells [121]. The crosstalk of BAMs and DCs with lymphocytes is fundamental for the protection, maintenance, and repair of the CNS [19, 121].
Different from the CSF which is characterize by CD4+ central memory T cells [122, 123], the choroid plexus contains CD4+ effector memory T cells localized within the epithelium near to MHCII cells and with a pattern of receptors (TCR) peculiar and specific to CNS antigens [19, 124].
In physiological conditions, the CNS-specific CD4+ effector memory T cells are regulated by the secretion of IL-10 from resident choroid plexus DCs. During inflammation, resident DCs and BAMs present antigens and activate CD4+ effector memory T cells [125]. The activation of T cells leads to their migration through the ependymal layer into the CSF [126]. This process is mediated by the adhesion of T cells to the apical surface of the choroid plexus epithelium by IFN-γ signaling and through the upregulation of the adhesion molecule ICAM-1 [113]. As in the gut, the epithelial layer of the CP homes γδ T cells that are described as contributors to the first phases of immune regulation and response early in life [127]. These data suggests that choroid plexus could be an essential site of priming, activation, and check of CNS T cells.
[127]. An important role for the immunomodulatory functions of the choroid plexus is carried out by a plethora of adhesion molecules on the luminal side of the epithelium [128]. ICAM-1 and vascular cell adhesion molecule-1 (VCAM-1) are constitutively expressed by the epithelial cells lining the choroid plexus and are upregulated upon immune stimulation in different onset patterns of CNS pathologies [17, 113], including EAE [129] and spinal cord injury [114]. The localization of ICAM-1 and VCAM-1 on the apical side of the epithelial cells of the choroid plexus suggests their involvement in the bidirectional recruitment of immune cells from the stroma to the CSF and back from the CSF to the systemic blood torrent. In both cases, the role of the choroid plexus seems to be fundamental in shaping leukocyte trafficking and immune regulation of the CNS. The choroid plexus epithelial localization of these two adhesion molecules could explain why ICAM-1 and VCAM-1 are found in the CSF of patients with bacterial meningitis, viral encephalitis, multiple sclerosis [130, 131], basal ganglia hemorrhages [132], schizophrenia spectrum disorders [124], and depression [133, 134].
In a previous work, ICAM-1 and VCAM-1 were described to be expressed exclusively in epithelial cells of the choroid plexus [129]. However, recent single cell analysis reported the expression of ICAM-1 and VCAM-1 transcripts in subclustered choroid plexus endothelial cells [120, 135]. The presence of ICAM-1 and VCAM-1 adhesion molecules in the endothelium of the choroid plexus suggests a new role of the PVB in the immune modulation of the CNS. Corroborating this hypothesis, multiple sclerosis and viral encephalitis patients show increased expression of endothelial VCAM-1 in concomitance with increased numbers of memory/effector T and B cells in the stroma of the choroid plexus [123, 136, 137] and increased numbers of antibody-producing CD138+ plasma cells in the CSF [138].
These data, together with our finding of the dynamic nature of choroid plexus permeability, support the hypothesis that this site plays a major role in leukocyte trafficking in and out of the brain.
Structural, functional, and immunological analogies between the gut and the choroid plexus vascular barriers
The central nervous system is a sensitive and effective complex network in constant communication with itself and the distal organs via the afferent and efferent nerves.
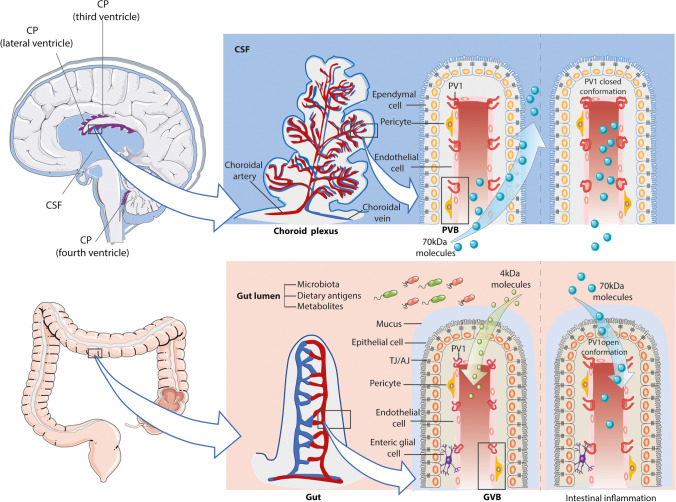
We are here adding an additional way of the immune communication between the CNS and the rest of the body which occurs via the vascular network, and that is controlled by a series of modifiable vascular barriers. In particular, we made a parallelism between the primary defense vascular barrier in the gut, which is the GVB, and the secondary barrier in the choroid plexus, which is the PVB. The first acts as a major determinant of body homeostasis as it controls what enters into the systemic circulation. When the gut vascular barrier is functional, the choroid plexus vascular barrier is open and allows the maximal exchange of molecules and immune cells between the brain and the rest of the body as whatever is in the circulation is considered as “safe.” During intestinal inflammation, the GVB is disrupted and cannot control any longer the intruders (Fig. 1). Thus, the PVB is called into question and is asked to close down to avoid unwanted molecules and inflammatory cells to reach the brain. It is like having two gates for protecting the house: the central door of the building and the doors of the apartments. When the central door works properly, the doors of the apartments can also remain open as the environment is considered as harmless.
Fig. 1.
Schematic representation of the structural and functional analogies between the gut vascular barrier (GVB) and the choroid plexus vascular barrier (PVB)
The GVB-PVB axis represents a key tissue-blood gatekeeper in the maintenance of signaling between the intestine and the brain and to preserve immune homeostasis (Fig. 1).
The gut and choroid plexus vascular barriers share important morphological similarities even though they show different degrees of permeability. Structurally, they are both surrounded by a monolayer of epithelial cells; however, these face the external world in the GVB while they face the internal part of the brain in the choroid plexus. The epithelial barrier in the gut is highly impermeable to bacteria and large molecules, while it allows the passage of 70 kDa molecules in the choroid plexus. They are also “protected” by a series of resident immune cells which are anchored to endothelial or epithelial cells and are ready to respond to an insult, thus contributing to regulating the opening and closure of the vascular barriers in order to protect from propagation of inflammation. Resident immune cells in the gut and the choroid plexus are mainly composed of macrophages, DCs, and CD4+ T cells [16, 72, 73, 79, 83, 119–121]. The most abundant population is represented in both cases by macrophages which are localized in two different spatial niches: (1) stromal macrophages strictly in contact with vessels and (2) subepithelial luminal macrophages. The differential localization suggests the dual role of macrophages in preserving either the endothelial layer which is the frontline with the systemic circulation or the epithelial layer which is a filtrating system with different size exclusions in the two organs to serve tissue-specific functions (Fig. 2).
Fig. 2.
Schematic representation of GVB and PVB immune resident and circulating immune cells
As we and others described, macrophages and DCs in the gut actively sample the intestinal lumen content [79, 139, 140], suggesting that there may be a parallelism in antigen sampling in the choroid plexus (Fig. 2). However, more studies are needed to evaluate the role of these cells in the maintenance of a tolerogenic environment and in the development of CNS immune response. More is known about gut tissue resident CD4+ T cells, but the finding that also choroid plexus resident T cells have effector memory phenotype could highlights a similar “sentinel-like” function of these cells in the choroid plexus (Fig. 2).
The protective structures of the gut are not always infallible. We found that Salmonella enterica serovar Typhimurium through the activation of type III secretion system (TTSS) could invade the intestinal outer mucus and interfere with the Wnt/β-catenin signaling pathway in endothelial cells, thus increasing vessel permeability. The interference of enteric pathogens and some members of the microbiota which are enriched in certain conditions, such as a high-fat diet regimen, intestinal inflammation, or the presence of tumors, with the GVB increases the detection of PV-1 and modifies the intestinal endothelial cutoff of permeability from 4 to 70 kDa molecules, allowing the passage of microbes and microbially derived molecules into the blood circulation [5, 43, 45]. In contrast, in the PVB, the Wnt/β-catenin signaling pathway is only marginally activated at steady state but is upregulated early during inflammation to return to basal levels when the inflammatory stimulus is removed.
In a preclinical model of ulcerative colitis, we showed that paralleling gut vascular barrier impairment, during the early phases of the disease, the blood milieu becomes pro-inflammatory, increasing the local recruitment of immune cells and circulating levels of IL-6 and lipopolysaccharides. By contrast, the choroid plexus vascular barrier first undergoes a slight increase of permeability by downregulation of the Wnt/β-catenin signaling pathway which is followed by a drastic shutdown of permeability and increased Wnt/β-catenin signaling pathway with modulation of choroid plexus immune cells.
The exclusion of dangerous molecules coming from the external environment is elicited by the gut where the GVB, together with the intestinal epithelial and immune barrier, guarantees the physical exclusion of gut microbiota and unwanted microbial molecules from the systemic circulation. Based on its function, the GVB can allow the diffusion from the lumen to the blood of molecules as large as 4 kDa, pitching immune and metabolic products into the blood (Fig. 1). The brain can catch molecules of maximum 0.5 kDa across the BBB and of at least 70 kDa within the PVB, ensuring the maintenance of CNS homeostasis. When the gut defense is impaired, the brain needs to take over at the level of the blood cerebrospinal fluid barrier, the largest gatekeeper of the brain, to protect itself. Here, the PVB massively shuts down when needed (Fig. 1), suggesting a possible modulation of the composition of tissue resident and recruited immune cells (Fig. 2), initiating a defensive program which can affect the BBB and CNS functions.
Conclusions and prospective
In summary, we reviewed two semipermeable endothelial layers that compose the gut and the choroid plexus vascular barriers. We show that these two barriers are connected and cooperate together with resident immune cells to control organ homeostasis. We suggest that infectious agents, toxins, immune, and metabolic alterations could alter the physiological permeability of the two barriers in opposite ways, thus ensuring that when the primary structure is disrupted, the second can take over. The analogies between the GVB and the PVB and their role in immune tissue resident composition and immunomodulatory functions of trafficking cells lead to intriguing future perspectives on their role in intestinal, neurological, and oncological diseases. Targeting the PVB-GVB axis may provide new insight for the simultaneous reconstitution of the two barriers, limiting the IBD-linked neurological comorbidities and opening new possibilities for drug delivery and immune surveillance within the CNS. The description of the brain-CSF barrier as a main route of viral infections in CNS invasion was recently reported. In particular, the Zika virus (ZIKV) and SARS-CoV-2 pathogens were found in the choroid plexus and CSF of patients before invading the brain parenchyma. These data suggest that CNS microorganism invasion and immune recruitment could be mediated by the blood-CSF barrier rather than by the BBB [141–144] and that the choroid plexus vascular barrier could play an important role in mediating CNS invasion and inflammation. Thus, several disorders, including the metabolic syndrome and neurological degeneration [145, 146], may be related to an impairment of the GVB-PVB axis which could become a new target for treatment and prevention. It will be particularly important to switch the interest from the affected organ to the gut in order to reestablish barrier properties.
Important outstanding questions need to be addressed: How are resident immune cells in the CP involved in protecting from invasion of infectious agents? Is the PVB involved in the recruitment in pro- and anti-inflammatory cells in the CNS? Which molecular targets are mediating the selectivity of immune cell trafficking? Can resident dendritic cells within the choroid plexus establish immune tolerance in the brain?
Further analysis integrating transcriptional profile and functional aspects of CP-homing immune cells and trafficking of blood leucocytes is needed to shed light on the emerging role of similarities and connections between the gut and choroid plexus–mediated brain immune homeostasis.
The structures of the intestinal and choroid plexus barriers are similar. The intestinal barrier is composed of the mucus layer, epithelial cells sealed by tight junction and adherens junction (TJ/AJ), and the innermost GVB. The GVB consists of endothelial cells, surrounded by pericytes and enteric glial cells. Similarly, the brain-CSF barrier is composed of specialized ciliated epithelial cells named ependymal cells, sealed by the TJ and AJ, and the inner PVB formed by fenestrated endothelial cells stabilized by pericytes. The structural difference between the GVB and the PVB is due to their anatomical position. The GVB regulates the connection between the external/gut environment and the bloodstream; the PVB instead represents the interface between the blood and the CNS.
In physiological conditions, the GVB allows the exchange of molecules and metabolites from the gut lumen to the bloodstream (4 kDa) and prevents the dissemination of infectious agents. In these conditions, the PVB is highly positive for PV-1 and permissive to 70 kDa molecules, supporting the molecular signaling between the brain and other organs.
During intestinal inflammation, the GVB is impaired, and it becomes positive for PV-1 detection and allows the transfer of 70 kDa molecules, including bacteria, into the bloodstream. The pro-inflammatory environment is sensed by the PVB, which, in turn, decreases the PV-1 detection and shuts down its permeability. Indeed, the PVB blocks the exchange of 70 kDa molecules with the CSF and this event can modulate the CSF milieu and set up the CNS protective program.
The epithelial layer of the gut is inhabited by tissue resident intraepithelial CD103+ dendritic cells (DCs) involved in the establishment of tolerance and in the priming of regulatory T cells. The gut presents also tissue resident lymphocytes as gamma delta (γδ) and unconventional T cells (UNC), CD8+ T cells expressing the CD103 ligand for E-cadherin which promote the adhesion to the epithelium. CD8+ intraepithelial lymphocytes cooperate to tissue repair. Intestinal lamina propria is home to resident innate myeloid cells (MC), lymphoid cells (LC), regulatory macrophages (MAC), and CD4+ T regulatory cells (Treg). MAC and Treg mediate the tolerogenic intestinal activity through the secretion of IL-10 and TGF-β and the inhibition of Th1/Th2/Th17 immune cells. At the apical surface of the epithelial layer, adhesion molecules are expressed as intercellular adhesion molecule-1 (ICAM-1). During infection or dysbiosis, CX3CR1+ mononuclear phagocytes maintain barrier integrity while sampling of luminal antigens and can induce Th17 responses. Upon inflammation, epithelial expression of ICAM-1 is increased and mediates the recruitment of neutrophils (Nφ) at the luminal side of the colon. The bacterial and viral infections are sensed also by the endothelial cells of the GVB, increasing the expression of adhesion molecules, including ICAM-1 and MCHI/II and establishing the CD8T and CD4T–mediated immune response and the recruitment of neutrophils, monocytes (MONO), and macrophages.
The CSF is characterized by CD4+ central memory T cells (CD4 Tcm). Within the CP, resident immune cells are described as stromal MAC, border macrophages (BAM), DCs, Nφ, and lymphoid B and T cells, including γδ T cells. BAMs and DCs activate the immunomodulatory activity in response to peripheral stimuli and migrate along the CP surface to present antigens to T cells. In physiological conditions, DCs regulate CD4+ effector memory T cells (CD4 Tem) by the secretion of IL-10. During inflammation, resident DCs and BAMs present the antigen and activate CD4 Tem cells by IFN-γ signaling and through the upregulation of the adhesion molecule ICAM-1, allowing the migration of CD Tem through the ependymal layer into the CSF. A soluble fraction of ICAM-1 and VCAM-1 was found in the CSF in patients with bacterial meningitis, viral encephalitis, multiple sclerosis, basal ganglia hemorrhage, schizophrenia spectrum disorders, and depression. During an inflammatory status of the CNS, increased numbers of antibody-producing CD138+ plasma cells in the CSF were reported. Immunomodulatory functions of the CP are carried out also by adhesion molecules on the luminal side of the epithelium including ICAM-1 and VCAM-1 which are constitutively expressed by the epithelial and endothelial cells of the CP and are upregulated upon immune stimulation.
Abbreviations
- BBB
Blood-brain barrier
- BCSFB
Brain cerebrospinal fluid barrier
- CNS
Central nervous system
- CSF
Cerebrospinal fluid
- GVB
Gut vascular barrier
- IBD
Intestinal bowel disease
- ICAM-1
Intercellular adhesion molecule-1
- PVB
Choroid plexus vascular barrier
- VCAM-1
Vascular cell adhesion molecule-1
Funding
These researches were supported by AIRC 5 × 1000 2018 ID.21147 and PRIN (Ministero dell’Istruzione dell’Università e della Ricerca) grant no. 2017J3E2W2 to Maria Rescigno.
Declarations
Competing interests
The authors declare no competing interests.
Footnotes
This article is a contribution to the special issue on Heterogeneity of tissue-resident immunity across organs and in health and disease - Guest Editors: Federica Sallusto and Petra Arck
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Sara Carloni, Email: sara.carloni@hunimed.eu.
Maria Rescigno, Email: maria.rescigno@hunimed.eu.
References
- 1.Collins SM, Bercik P. The relationship between intestinal microbiota and the central nervous system in normal gastrointestinal function and disease. Gastroenterology. 2009;136(6):2003–2014. doi: 10.1053/J.GASTRO.2009.01.075. [DOI] [PubMed] [Google Scholar]
- 2.Negi N, Das BK. CNS: not an immunoprivilaged site anymore but a virtual secondary lymphoid organ. Int Rev Immunol. 2017;37(1):57–68. doi: 10.1080/08830185.2017.1357719. [DOI] [PubMed] [Google Scholar]
- 3.Vijay N, Morse BL, Morris ME. A novel monocarboxylate transporter inhibitor as a potential treatment strategy for γ-hydroxybutyric acid overdose. Pharm Res. 2015;32(6):1894. doi: 10.1007/S11095-014-1583-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kekuda R, Manoharan P, Baseler W, Sundaram U. Monocarboxylate 4 mediated butyrate transport in a rat intestinal epithelial cell line. Dig Dis Sci. 2013;58(3):660–667. doi: 10.1007/S10620-012-2407-X. [DOI] [PubMed] [Google Scholar]
- 5.Carloni S, et al. Identification of a choroid plexus vascular barrier closing during intestinal inflammation. Science. 2021;1979:439–448. doi: 10.1126/science.abc6108. [DOI] [PubMed] [Google Scholar]
- 6.O’Connor KM, Lucking EF, Cryan JF, O’Halloran KD. Bugs, breathing and blood pressure: microbiota–gut–brain axis signalling in cardiorespiratory control in health and disease. J Physiol. 2020;598(19):4159–4179. doi: 10.1113/JP280279. [DOI] [PubMed] [Google Scholar]
- 7.Durgan DJ, Lee J, McCullough LD, Bryan RM. Examining the role of the microbiota-gut-brain axis in stroke. Stroke. 2019;50(8):2270–2277. doi: 10.1161/STROKEAHA.119.025140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sherman MP, Zaghouani H, Niklas V. Gut microbiota, the immune system, and diet influence the neonatal gut-brain axis. Pediatr Res. 2015;77(1–2):127–135. doi: 10.1038/PR.2014.161. [DOI] [PubMed] [Google Scholar]
- 9.Dalile B, van Oudenhove L, Vervliet B, Verbeke K. The role of short-chain fatty acids in microbiota-gut-brain communication. Nat Rev Gastroenterol Hepatol. 2019;16(8):461–478. doi: 10.1038/S41575-019-0157-3. [DOI] [PubMed] [Google Scholar]
- 10.Fung TC, Olson CA, Hsiao EY. Interactions between the microbiota, immune and nervous systems in health and disease. Nat Neurosci. 2017;20(2):145–155. doi: 10.1038/NN.4476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Borre YE, O’Keeffe GW, Clarke G, Stanton C, Dinan TG, Cryan JF. Microbiota and neurodevelopmental windows: implications for brain disorders. Trends Mol Med. 2014;20(9):509–518. doi: 10.1016/J.MOLMED.2014.05.002. [DOI] [PubMed] [Google Scholar]
- 12.Silva YP, Bernardi A, Frozza RL. The role of short-chain fatty acids from gut microbiota in gut-brain communication. Front Endocrinol. 2020;11:25. doi: 10.3389/FENDO.2020.00025/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Aswendt M et al (2021) The gut microbiota modulates brain network connectivity under physiological conditions and after acute brain ischemia. iScience 24(10). 10.1016/j.isci.2021.103095 [DOI] [PMC free article] [PubMed]
- 14.Abdel-Haq R, Schlachetzki JCM, Glass CK, Mazmanian SK. Microbiome–microglia connections via the gut–brain axis. J Exp Med. 2019;216(1):41–59. doi: 10.1084/JEM.20180794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Baruch K, et al. Aging-induced type I interferon response at the choroid plexus negatively affects brain function. Science (1979) 2014;346(6205):89–93. doi: 10.1126/science.1252945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Croese T, Castellani G, Schwartz M. Immune cell compartmentalization for brain surveillance and protection. Nat Immunol. 2021;22:1083–1092. doi: 10.1038/s41590-021-00994-2. [DOI] [PubMed] [Google Scholar]
- 17.Kunis G, et al. IFN-γ-dependent activation of the brain’s choroid plexus for CNS immune surveillance and repair. Brain. 2013;136(11):3427–3440. doi: 10.1093/BRAIN/AWT259. [DOI] [PubMed] [Google Scholar]
- 18.Schwartz M, Baruch K. The resolution of neuroinflammation in neurodegeneration: leukocyte recruitment via the choroid plexus. EMBO J. 2014;33(1):7. doi: 10.1002/EMBJ.201386609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Baruch K, et al. CNS-specific immunity at the choroid plexus shifts toward destructive Th2 inflammation in brain aging. Proc Natl Acad Sci U S A. 2013;110(6):2264–2269. doi: 10.1073/PNAS.1211270110/SUPPL_FILE/PNAS.201211270SI.PDF. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Clarke G, Quigley EMM, Cryan JF, Dinan TG. Irritable bowel syndrome: towards biomarker identification. Trends Mol Med. 2009;15(10):478–489. doi: 10.1016/J.MOLMED.2009.08.001. [DOI] [PubMed] [Google Scholar]
- 21.Carding S, Verbeke K, Vipond DT, Corfe BM, Owen LJ (2015) Dysbiosis of the gut microbiota in disease. Microb Ecol Health Dis 26(0). 10.3402/MEHD.V26.26191 [DOI] [PMC free article] [PubMed]
- 22.Martin CR, Osadchiy V, Kalani A, Mayer EA. The brain-gut-microbiome axis. Cell Mol Gastroenterol Hepatol. 2018;6(2):133–148. doi: 10.1016/J.JCMGH.2018.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rhee SH, Pothoulakis C, Mayer EA. Principles and clinical implications of the brain-gut-enteric microbiota axis. Nat Rev Gastroenterol Hepatol. 2009;6(5):306–314. doi: 10.1038/NRGASTRO.2009.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mayer EA. Gut feelings: the emerging biology of gut-brain communication. Nat Rev Neurosci. 2011;12(8):453–466. doi: 10.1038/NRN3071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yang T, et al. Gut dysbiosis is linked to hypertension. Hypertension. 2015;65(6):1331–1340. doi: 10.1161/HYPERTENSIONAHA.115.05315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chow J, Lee SM, Shen Y, Khosravi A, Mazmanian SK. Host-bacterial symbiosis in health and disease. Adv Immunol. 2010;107(C):243–274. doi: 10.1016/B978-0-12-381300-8.00008-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yang T, Zubcevic J (2017) Gut-brain axis in regulation of blood pressure. Front Physiol 8(OCT). 10.3389/FPHYS.2017.00845/FULL [DOI] [PMC free article] [PubMed]
- 28.Banks WA. The blood-brain barrier as a regulatory interface in the gut-brain axes. Physiol Behav. 2006;89(4):472–476. doi: 10.1016/J.PHYSBEH.2006.07.004. [DOI] [PubMed] [Google Scholar]
- 29.Wekerle H. Brain autoimmunity and intestinal microbiota: 100 trillion game changers. Trends Immunol. 2017;38(7):483–497. doi: 10.1016/J.IT.2017.03.008. [DOI] [PubMed] [Google Scholar]
- 30.Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. 2012;13(10):701–712. doi: 10.1038/NRN3346. [DOI] [PubMed] [Google Scholar]
- 31.Günther C, Rothhammer V, Karow M, Neurath M, Winner B. The gut-brain axis in inflammatory bowel disease—current and future perspectives. Int J Mol Sci. 2021;22(16):8870. doi: 10.3390/IJMS22168870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sampson TR et al (2016) Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s disease. Cell 167(6). 10.1016/J.CELL.2016.11.018 [DOI] [PMC free article] [PubMed]
- 33.Wu SC, Cao ZS, Chang KM, Juang JL. Intestinal microbial dysbiosis aggravates the progression of Alzheimer’s disease in Drosophila. Nat Commun. 2017;8(1):1–9. doi: 10.1038/s41467-017-00040-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rothhammer V, et al. Microglial control of astrocytes in response to microbial metabolites. Nature. 2018;557(7707):724–728. doi: 10.1038/S41586-018-0119-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Berer K, et al. Commensal microbiota and myelin autoantigen cooperate to trigger autoimmune demyelination. Nature. 2011;479(7374):538–541. doi: 10.1038/NATURE10554. [DOI] [PubMed] [Google Scholar]
- 36.Vuong HE, Hsiao EY. Emerging roles for the gut microbiome in autism spectrum disorder. Biol Psychiatry. 2017;81(5):411–423. doi: 10.1016/J.BIOPSYCH.2016.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mehrian-Shai R, Reichardt JKV, Harris CC, Toren A. The gut-brain axis, paving the way to brain cancer. Trends Cancer. 2019;5(4):200–207. doi: 10.1016/J.TRECAN.2019.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bhattarai Y. Microbiota-gut-brain axis: interaction of gut microbes and their metabolites with host epithelial barriers. Neurogastroenterol Motil. 2018;30(6):e13366. doi: 10.1111/NMO.13366. [DOI] [PubMed] [Google Scholar]
- 39.Rescigno M. The microbiota revolution: excitement and caution. Eur J Immunol. 2017;47(9):1406–1413. doi: 10.1002/EJI.201646576. [DOI] [PubMed] [Google Scholar]
- 40.Johansson MEV, Hansson GC. Immunological aspects of intestinal mucus and mucins. Nat Rev Immunol. 2016;16(10):639–649. doi: 10.1038/NRI.2016.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Schroeder BO. Fight them or feed them: how the intestinal mucus layer manages the gut microbiota. Gastroenterol Rep (Oxf) 2019;7(1):3–12. doi: 10.1093/GASTRO/GOY052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Buckley A, Turner JR. Cell biology of tight junction barrier regulation and mucosal disease. Cold Spring Harb Perspect Biol. 2018;10(1):a029314. doi: 10.1101/CSHPERSPECT.A029314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Spadoni I, et al. A gut-vascular barrier controls the systemic dissemination of bacteria. Science. 2015;1979(350):830–834. doi: 10.1126/science.aad0135. [DOI] [PubMed] [Google Scholar]
- 44.Spadoni I, Fornasa G, Rescigno M. Organ-specific protection mediated by cooperation between vascular and epithelial barriers. Nat Rev Immunol. 2017;17(12):761–773. doi: 10.1038/nri.2017.100. [DOI] [PubMed] [Google Scholar]
- 45.Mouries J, et al. Microbiota-driven gut vascular barrier disruption is a prerequisite for non-alcoholic steatohepatitis development. J Hepatol. 2019;71(6):1216–1228. doi: 10.1016/J.JHEP.2019.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Augustin HG, Koh GY (2017) Organotypic vasculature: From descriptive heterogeneity to functional pathophysiology. Science 357(6353):eaal2379. 10.1126/science.aal2379 [DOI] [PubMed]
- 47.Haddad-Tóvolli R, Dragano NRV, Ramalho AFS, Velloso LA. Development and function of the blood-brain barrier in the context of metabolic control. Front Neurosci. 2017;11(APR):224. doi: 10.3389/FNINS.2017.00224/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Weiss N, Miller F, Cazaubon S, Couraud PO. The blood-brain barrier in brain homeostasis and neurological diseases. Biochim Biophys Acta. 2009;1788(4):842–857. doi: 10.1016/J.BBAMEM.2008.10.022. [DOI] [PubMed] [Google Scholar]
- 49.Neuwelt EA, et al. Engaging neuroscience to advance translational research in brain barrier biology. Nat Rev Neurosci. 2011;12(3):169–182. doi: 10.1038/nrn2995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Łuc M, et al. Gut microbiota in dementia. Critical review of novel findings and their potential application. Prog Neuro-Psychopharmacol Biol Psychiatry. 2021;104:110039. doi: 10.1016/J.PNPBP.2020.110039. [DOI] [PubMed] [Google Scholar]
- 51.Tsilingiri K, Rescigno M. Postbiotics: what else? Benefic Microbes. 2012;4(1):101–107. doi: 10.3920/BM2012.0046. [DOI] [PubMed] [Google Scholar]
- 52.Tsilingiri K, et al. Probiotic and postbiotic activity in health and disease: comparison on a novel polarised ex-vivo organ culture model. Gut. 2012;61(7):1007–1015. doi: 10.1136/GUTJNL-2011-300971. [DOI] [PubMed] [Google Scholar]
- 53.Reynés B, Palou M, Rodríguez AM, Palou A (2019) Regulation of adaptive thermogenesis and browning by prebiotics and postbiotics. Front Physiol 9(JAN). 10.3389/FPHYS.2018.01908 [DOI] [PMC free article] [PubMed]
- 54.Logsdon AF, Erickson MA, Rhea EM, Salameh TS, Banks WA. Gut reactions: how the blood–brain barrier connects the microbiome and the brain. Exp Biol Med. 2018;243(2):159–165. doi: 10.1177/1535370217743766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sorribas M, et al. FXR modulates the gut-vascular barrier by regulating the entry sites for bacterial translocation in experimental cirrhosis. J Hepatol. 2019;71(6):1126–1140. doi: 10.1016/J.JHEP.2019.06.017. [DOI] [PubMed] [Google Scholar]
- 56.Li Y, et al. Berberine reduces gut-vascular barrier permeability via modulation of ApoM/S1P pathway in a model of polymicrobial sepsis. Life Sci. 2020;261:118460. doi: 10.1016/J.LFS.2020.118460. [DOI] [PubMed] [Google Scholar]
- 57.Bertocchi A, et al. Gut vascular barrier impairment leads to intestinal bacteria dissemination and colorectal cancer metastasis to liver. Cancer Cell. 2021;39(5):708–724.e11. doi: 10.1016/J.CCELL.2021.03.004. [DOI] [PubMed] [Google Scholar]
- 58.Ciccia F, et al. Dysbiosis and zonulin upregulation alter gut epithelial and vascular barriers in patients with ankylosing spondylitis. Ann Rheum Dis. 2017;76(6):1123–1132. doi: 10.1136/ANNRHEUMDIS-2016-210000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hamilton BJN, et al. Phorbol esters induce PLVAP expression via VEGF and additional secreted molecules in MEK1-dependent and p38, JNK and PI3K/Akt-independent manner. J Cell Mol Med. 2019;23(2):920–933. doi: 10.1111/jcmm.13993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Stan RV, et al. The diaphragms of fenestrated endothelia: gatekeepers of vascular permeability and blood composition. Dev Cell. 2012;23(6):1203–1218. doi: 10.1016/j.devcel.2012.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Auvinen K, et al. Fenestral diaphragms and PLVAP associations in liver sinusoidal endothelial cells are developmentally regulated. Sci Rep. 2019 doi: 10.1038/s41598-019-52068-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Leppink DM, et al. Inducible expression of an endothelial cell antigen on murine myocardial vasculature in association with interstitial cellular infiltration. Transplantation. 1989;48(5):874–877. doi: 10.1097/00007890-198911000-00032. [DOI] [PubMed] [Google Scholar]
- 63.Ruiter DJ, Schlingemann RO, Rietveld FJR, de Waal RMW. Monoclonal antibody-defined human endothelial antigens as vascular markers. J Invest Dermatol. 1989;93(2 Suppl):25S–32S. doi: 10.1111/1523-1747.EP12580902. [DOI] [PubMed] [Google Scholar]
- 64.Hallmann R, Mayer DN, Berg EL, Broermann R, Butcher EC. Novel mouse endothelial cell surface marker is suppressed during differentiation of the blood brain barrier. Dev Dyn. 1995;202(4):325–332. doi: 10.1002/AJA.1002020402. [DOI] [PubMed] [Google Scholar]
- 65.Keuschnigg J, Henttinen T, Auvinen K, Karikoski M, Salmi M, Jalkanen S. The prototype endothelial marker PAL-E is a leukocyte trafficking molecule. Blood. 2009;114(2):478–484. doi: 10.1182/BLOOD-2008-11-188763. [DOI] [PubMed] [Google Scholar]
- 66.Keuschnigg J, et al. PV-1 is recognized by the PAL-E antibody and forms complexes with NRP-1. Blood. 2012;120(1):232–235. doi: 10.1182/BLOOD-2012-01-406876. [DOI] [PubMed] [Google Scholar]
- 67.Rodrigues SF, Granger DN (2015) Blood cells and endothelial barrier function. Tissue Barriers 3(1). 10.4161/21688370.2014.978720 [DOI] [PMC free article] [PubMed]
- 68.Pelaseyed T, et al. The mucus and mucins of the goblet cells and enterocytes provide the first defense line of the gastrointestinal tract and interact with the immune system. Immunol Rev. 2014;260(1):8–20. doi: 10.1111/IMR.12182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Elmentaite R, et al. Cells of the human intestinal tract mapped across space and time. Nature. 2021;597(7875):250–255. doi: 10.1038/s41586-021-03852-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Rampoldi F, Prinz I. Three layers of intestinal γδ T cells talk different languages with the microbiota. Front Immunol. 2022;0:1225. doi: 10.3389/FIMMU.2022.849954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Farache J, et al. Luminal bacteria recruit CD103+ dendritic cells into the intestinal epithelium to sample bacterial antigens for presentation. Immunity. 2013;38(3):581–595. doi: 10.1016/j.immuni.2013.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hoytema van Konijnenburg DP, Reis BS, Pedicord VA, Farache J, Victora GD, Mucida D. Intestinal epithelial and intraepithelial T cell crosstalk mediates a dynamic response to infection. Cell. 2017;171(4):783–794.e13. doi: 10.1016/j.cell.2017.08.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Weiner HL, da Cunha AP, Quintana F, Wu H. Oral tolerance. Immunol Rev. 2011;241(1):241–259. doi: 10.1111/J.1600-065X.2011.01017.X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Esterházy D, Loschko J, London M, Jove V, Oliveira TY, Mucida D. Classical dendritic cells are required for dietary antigen–mediated induction of peripheral Treg cells and tolerance. Nat Immunol. 2016;17(5):545–555. doi: 10.1038/ni.3408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sun T, Rojas OL, Li C, Ward LA, Philpott DJ, Gommerman JL. Intestinal Batf3-dependent dendritic cells are required for optimal antiviral T-cell responses in adult and neonatal mice. Mucosal Immunol. 2016;10(3):775–788. doi: 10.1038/mi.2016.79. [DOI] [PubMed] [Google Scholar]
- 76.Mayer JU, Demiri M, Agace WW, MacDonald AS, Svensson-Frej M, Milling SW. Different populations of CD11b+ dendritic cells drive Th2 responses in the small intestine and colon. Nat Commun. 2017;8(1):1–12. doi: 10.1038/ncomms15820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Nutsch K, et al. Rapid and efficient generation of regulatory T cells to commensal antigens in the periphery. Cell Rep. 2016;17(1):206–220. doi: 10.1016/J.CELREP.2016.08.092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Loschko J, et al. Absence of MHC class II on cDCs results in microbial-dependent intestinal inflammation. J Exp Med. 2016;213(4):517–534. doi: 10.1084/JEM.20160062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Rescigno M, et al. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nat Immunol. 2001;2(4):361–367. doi: 10.1038/86373. [DOI] [PubMed] [Google Scholar]
- 80.Fransen F, et al. BALB/c and C57BL/6 mice differ in polyreactive IgA abundance, which impacts the generation of antigen-specific IgA and microbiota Diversity. Immunity. 2015;43(3):527–540. doi: 10.1016/J.IMMUNI.2015.08.011. [DOI] [PubMed] [Google Scholar]
- 81.Atarashi K, et al. ATP drives lamina propria T(H)17 cell differentiation. Nature. 2008;455(7214):808–812. doi: 10.1038/NATURE07240. [DOI] [PubMed] [Google Scholar]
- 82.Bernardo D, et al. Human intestinal pro-inflammatory CD11chighCCR2+CX3CR1+ macrophages, but not their tolerogenic CD11c−CCR2−CX3CR1− counterparts, are expanded in inflammatory bowel disease. Mucosal Immunol. 2018;11(4):1114–1126. doi: 10.1038/s41385-018-0030-7. [DOI] [PubMed] [Google Scholar]
- 83.Bujko A, et al. Transcriptional and functional profiling defines human small intestinal macrophage subsets. J Exp Med. 2018;215(2):441–458. doi: 10.1084/JEM.20170057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hutchins AP, et al. Distinct transcriptional regulatory modules underlie STAT3’s cell type-independent and cell type-specific functions. Nucleic Acids Res. 2013;41(4):2155–2170. doi: 10.1093/NAR/GKS1300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Sumagin R, et al. Neutrophil interactions with epithelial-expressed ICAM-1 enhances intestinal mucosal wound healing. Mucosal Immunol. 2016;9(5):1151–1162. doi: 10.1038/mi.2015.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pardo-Camacho C, González-Castro AM, Rodiño-Janeiro BK, Pigrau M, Vicario M. Epithelial immunity: priming defensive responses in the intestinal mucosa. Am J Physiol-Gastrointest Liver Physiol. 2018;314(2):G247–G255. doi: 10.1152/ajpgi.00215.2016. [DOI] [PubMed] [Google Scholar]
- 87.Amersfoort J, Eelen G, Carmeliet P. Immunomodulation by endothelial cells — partnering up with the immune system? Nat Rev Immunol. 2022;2022:1–13. doi: 10.1038/s41577-022-00694-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Krausgruber T, et al. Structural cells are key regulators of organ-specific immune responses. Nature. 2020;583(7815):296–302. doi: 10.1038/s41586-020-2424-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Danese S, Dejana E, Fiocchi C. Immune regulation by microvascular endothelial cells: directing innate and adaptive immunity, coagulation, and inflammation. J Immunol. 2007;178(10):6017–6022. doi: 10.4049/JIMMUNOL.178.10.6017. [DOI] [PubMed] [Google Scholar]
- 90.Cromer WE, Mathis JM, Granger DN, Chaitanya GV, Alexander JS. Role of the endothelium in inflammatory bowel diseases. World J Gastroenterol. 2011;17(5):578–593. doi: 10.3748/wjg.v17.i5.578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Gravina AG et al (2018) Vascular endothelial dysfunction in inflammatory bowel diseases: pharmacological and nonpharmacological targets. Oxidative Med Cell Longev 2018. 10.1155/2018/2568569 [DOI] [PMC free article] [PubMed]
- 92.el Kebir D, József L, Pan W, Wang L, Filep JG. Bacterial DNA activates endothelial cells and promotes neutrophil adherence through TLR9 signaling. J Immunol. 2009;182(7):4386–4394. doi: 10.4049/JIMMUNOL.0803044. [DOI] [PubMed] [Google Scholar]
- 93.Salvador B, et al. Modulation of endothelial function by Toll like receptors. Pharmacol Res. 2016;108:46–56. doi: 10.1016/J.PHRS.2016.03.038. [DOI] [PubMed] [Google Scholar]
- 94.Etienne-Manneville S, Manneville J-B, Adamson P, Wilbourn B, Greenwood J, Couraud P-O. ICAM-1cCoupled cytoskeletal rearrangements and transendothelial lymphocyte migration involve intracellular calcium signaling in brain endothelial cell lines. J Immunol. 2000;165(6):3375–3383. doi: 10.4049/JIMMUNOL.165.6.3375. [DOI] [PubMed] [Google Scholar]
- 95.Yang L, Froio RM, Sciuto TE, Dvorak AM, Alon R, Luscinskas FW. ICAM-1 regulates neutrophil adhesion and transcellular migration of TNF-α-activated vascular endothelium under flow. Blood. 2005;106(2):584–592. doi: 10.1182/BLOOD-2004-12-4942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Sumagin R, Sarelius IH. A role for ICAM-1 in maintenance of leukocyte-endothelial cell rolling interactions in inflamed arterioles. Am J Physiol - Heart Circ Physiol. 2007;293(5):2786–2798. doi: 10.1152/ajpheart.00720.2007. [DOI] [PubMed] [Google Scholar]
- 97.Vainer B, Horn T, Nielsen OH. Colonic epithelial cell expression of ICAM-1 relates to loss of surface continuity: a comparative study of inflammatory bowel disease and colonic neoplasms. Scand J Gastroenterol. 2009;41(3):318–325. doi: 10.1080/00365520510024241. [DOI] [PubMed] [Google Scholar]
- 98.Milam AV, Allen PM. Functional heterogeneity in CD4(+) T cell responses against a bacterial pathogen. Front Immunol. 2015;6(DEC):621. doi: 10.3389/FIMMU.2015.00621/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Koutsakos M, et al. Human CD8+ T cell cross-reactivity across influenza A, B and C viruses. Nat Immunol. 2019;20(5):613–625. doi: 10.1038/s41590-019-0320-6. [DOI] [PubMed] [Google Scholar]
- 100.Braun M, Iliff JJ. The impact of neurovascular, blood-brain barrier, and glymphatic dysfunction in neurodegenerative and metabolic diseases. Int Rev Neurobiol. 2020;154:413–436. doi: 10.1016/BS.IRN.2020.02.006. [DOI] [PubMed] [Google Scholar]
- 101.Engelhardt B, Vajkoczy P, Weller RO. The movers and shapers in immune privilege of the CNS. Nat Immunol. 2017;18(2):123–131. doi: 10.1038/ni.3666. [DOI] [PubMed] [Google Scholar]
- 102.Mapunda JA, Tibar H, Regragui W, Engelhardt B. How does the immune system enter the brain? Front Immunol. 2022;13:563. doi: 10.3389/FIMMU.2022.805657/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Bill B, Korzh V (2014) Choroid plexus in developmental and evolutionary perspective. Front Neurosci 8. 10.3389/FNINS.2014.00363 [DOI] [PMC free article] [PubMed]
- 104.Damkier HH, Brown PD, Praetorius J. Cerebrospinal fluid secretion by the choroid plexus. Physiol Rev. 2013;93(4):1847–1892. doi: 10.1152/PHYSREV.00004.2013. [DOI] [PubMed] [Google Scholar]
- 105.Liddelow SA (2015) Development of the choroid plexus and blood-CSF barrier. Front Neurosci 9(MAR). 10.3389/FNINS.2015.00032 [DOI] [PMC free article] [PubMed]
- 106.Spector R, Keep RF, Robert Snodgrass S, Smith QR, Johanson CE. A balanced view of choroid plexus structure and function: focus on adult humans. Exp Neurol. 2015;267:78–86. doi: 10.1016/J.EXPNEUROL.2015.02.032. [DOI] [PubMed] [Google Scholar]
- 107.Javed K, Reddy V, Lui F (2021) Neuroanatomy, choroid plexus. StatPearls. Accessed: Jan. 10, 2022. [Online]. Available: https://www.ncbi.nlm.nih.gov/books/NBK538156/
- 108.Lopes Pinheiro MA, et al. Immune cell trafficking across the barriers of the central nervous system in multiple sclerosis and stroke. Biochim Biophys Acta. 2016;1862(3):461–471. doi: 10.1016/J.BBADIS.2015.10.018. [DOI] [PubMed] [Google Scholar]
- 109.Wilson EH, Weninger W, Hunter CA. Trafficking of immune cells in the central nervous system. J Clin Invest. 2010;120(5):1368–1379. doi: 10.1172/JCI41911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Parab S, Quick RE, Matsuoka RL. Endothelial cell-type-specific molecular requirements for angiogenesis drive fenestrated vessel development in the brain. Elife. 2021;10:1–33. doi: 10.7554/ELIFE.64295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Redzic ZB, Segal MB. The structure of the choroid plexus and the physiology of the choroid plexus epithelium. Adv Drug Deliv Rev. 2004;56(12):1695–1716. doi: 10.1016/J.ADDR.2004.07.005. [DOI] [PubMed] [Google Scholar]
- 112.Baruch K, Kertser A, Porat Z, Schwartz M. Cerebral nitric oxide represses choroid plexus NFκB-dependent gateway activity for leukocyte trafficking. EMBO J. 2015;34(13):1816–1828. doi: 10.15252/EMBJ.201591468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Strominger I, et al. The choroid plexus functions as a niche for T-cell stimulation within the central nervous system. Front Immunol. 2018;9(MAY):1066. doi: 10.3389/FIMMU.2018.01066/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Shechter R, et al. Recruitment of beneficial M2 macrophages to injured spinal cord is orchestrated by remote brain choroid plexus. Immunity. 2013;38(3):555–569. doi: 10.1016/J.IMMUNI.2013.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Marques F, et al. Kinetic profile of the transcriptome changes induced in the choroid plexus by peripheral inflammation. J Cereb Blood Flow Metab. 2009;29(5):921–932. doi: 10.1038/JCBFM.2009.15. [DOI] [PubMed] [Google Scholar]
- 116.Marques F, et al. The choroid plexus response to a repeated peripheral inflammatory stimulus. BMC Neurosci. 2009;10(1):1–10. doi: 10.1186/1471-2202-10-135/FIGURES/3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Pollak TA, Drndarski S, Stone JM, David AS, McGuire P, Abbott NJ. The blood–brain barrier in psychosis. Lancet Psychiatry. 2018;5(1):79–92. doi: 10.1016/S2215-0366(17)30293-6. [DOI] [PubMed] [Google Scholar]
- 118.Yang AC, et al. Dysregulation of brain and choroid plexus cell types in severe COVID-19. Nature. 2021;595(7868):565–571. doi: 10.1038/s41586-021-03710-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Meeker RB, Williams K, Killebrew DA, Hudson LC. Cell trafficking through the choroid plexus. Cell Adh Migr. 2012;6(5):390. doi: 10.4161/CAM.21054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Dani N, et al. A cellular and spatial map of the choroid plexus across brain ventricles and ages. Cell. 2021;184(11):3056–3074.e21. doi: 10.1016/J.CELL.2021.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Cui J, Xu H, Lehtinen MK. Macrophages on the margin: choroid plexus immune responses. Trends Neurosci. 2021;44(11):864–875. doi: 10.1016/J.TINS.2021.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.de Graaf MT, et al. Central memory CD4(+) T cells dominate the normal cerebrospinal fluid. Cytometry B Clin Cytom. 2011;80(1):43–50. doi: 10.1002/CYTO.B.20542. [DOI] [PubMed] [Google Scholar]
- 123.Kivisäkk P, et al. Human cerebrospinal fluid central memory CD4+ T cells: evidence for trafficking through choroid plexus and meninges via P-selectin. Proc Natl Acad Sci U S A. 2003;100(14):8389–8394. doi: 10.1073/PNAS.1433000100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Meixensberger S, et al. Upregulation of sICAM-1 and sVCAM-1 levels in the cerebrospinal fluid of patients with schizophrenia spectrum disorders. Diagnostics. 2021;11(7):1134. doi: 10.3390/DIAGNOSTICS11071134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Anandasabapathy N, et al. Flt3L controls the development of radiosensitive dendritic cells in the meninges and choroid plexus of the steady-state mouse brain. J Exp Med. 2011;208(8):1695–1705. doi: 10.1084/JEM.20102657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Louveau A, et al. Structural and functional features of central nervous system lymphatics. Nature. 2015;523(7560):337. doi: 10.1038/NATURE14432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Albertsson AM, et al. γδ T cells contribute to injury in the developing brain. Am J Pathol. 2018;188(3):757. doi: 10.1016/J.AJPATH.2017.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Kleine TO, Benes L. Immune surveillance of the human central nervous system (CNS): different migration pathways of immune cells through the blood-brain barrier and blood-cerebrospinal fluid barrier in healthy persons. Cytometry A. 2006;69(3):147–151. doi: 10.1002/CYTO.A.20225. [DOI] [PubMed] [Google Scholar]
- 129.Steffen BJ, Breier G, Butcher EC, Schulz M, Engelhardt B (1996) ICAM-1, VCAM-1, and MAdCAM-1 are expressed on choroid plexus epithelium but not endothelium and mediate binding of lymphocytes in vitro. Am J Pathol 148(6):1819. Accessed: May 15, 2022. [Online]. Available: /pmc/articles/PMC1861637/?report=abstract [PMC free article] [PubMed]
- 130.Rieckmann P, et al. Soluble intercellular adhesion molecule-1 in cerebrospinal fluid: an indicator for the inflammatory impairment of the blood-cerebrospinal fluid barrier. J Neuroimmunol. 1993;47(2):133–140. doi: 10.1016/0165-5728(93)90023-R. [DOI] [PubMed] [Google Scholar]
- 131.V. G. Sasseville et al. Elevated vascular cell adhesion molecule-1 in AIDS encephalitis induced by simian immunodeficiency virus. The American Journal of Pathology, vol. 141, no. 5, p. 1021, 1992, Accessed: May 15, 2022. [Online]. Available: /pmc/articles/PMC1886675/?report=abstract [PMC free article] [PubMed]
- 132.Kraus J, Gerriets T, Leis S, Stolz E, Oschmann P, Heckmann JG. Time course of VCAM-1 and ICAM-1 in CSF in patients with basal ganglia haemorrhage. Acta Neurol Scand. 2007;116(1):49–55. doi: 10.1111/J.1600-0404.2006.00790.X. [DOI] [PubMed] [Google Scholar]
- 133.Ghersi-Egea JF, Strazielle N, Catala M, Silva-Vargas V, Doetsch F, Engelhardt B. Molecular anatomy and functions of the choroidal blood-cerebrospinal fluid barrier in health and disease. Acta Neuropathologica. 2018;135(3):337–361. doi: 10.1007/S00401-018-1807-1. [DOI] [PubMed] [Google Scholar]
- 134.Müller N. The role of intercellular adhesion molecule-1 in the pathogenesis of psychiatric disorders. Front Pharmacol. 2019;10:1251. doi: 10.3389/FPHAR.2019.01251/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Figueiredo CA, et al. Immune response and pathogen invasion at the choroid plexus in the onset of cerebral toxoplasmosis. J Neuroinflammation. 2022;19(1):1–18. doi: 10.1186/S12974-021-02370-1/FIGURES/6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Vercellino M, et al. Involvement of the choroid plexus in multiple sclerosis autoimmune inflammation: a neuropathological study. J Neuroimmunol. 2008;199(1–2):133–141. doi: 10.1016/J.JNEUROIM.2008.04.035. [DOI] [PubMed] [Google Scholar]
- 137.Giunti D, et al. Phenotypic and functional analysis of T cells homing into the CSF of subjects with inflammatory diseases of the CNS. J Leukoc Biol. 2003;73(5):584–590. doi: 10.1189/JLB.1202598. [DOI] [PubMed] [Google Scholar]
- 138.Corcione A, et al. Recapitulation of B cell differentiation in the central nervous system of patients with multiple sclerosis. Proc Natl Acad Sci U S A. 2004;101(30):11064–11069. doi: 10.1073/PNAS.0402455101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Chieppa M, Rescigno M, Huang AYC, Germain RN. Dynamic imaging of dendritic cell extension into the small bowel lumen in response to epithelial cell TLR engagement. J Exp Med. 2006;203(13):2841–2852. doi: 10.1084/JEM.20061884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Niess JH, et al. CX3CR1-mediated dendritic cell access to the intestinal lumen and bacterial clearance. Science. 2005;307(5707):254–258. doi: 10.1126/SCIENCE.1102901. [DOI] [PubMed] [Google Scholar]
- 141.Kim J et al (2020) Zika virus infects pericytes in the choroid plexus and enters the central nervous system through the blood-cerebrospinal fluid barrier. PLoS Pathog 16(5). 10.1371/JOURNAL.PPAT.1008204 [DOI] [PMC free article] [PubMed]
- 142.Mcquaid C, Brady M, Deane R. SARS-CoV-2: is there neuroinvasion? Fluids Barriers CNS. 2021;18:32. doi: 10.1186/s12987-021-00267-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Brady M, et al. Spike protein multiorgan tropism suppressed by antibodies targeting SARS-CoV-2. Commun Biol. 2021 doi: 10.1038/s42003-021-02856-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Gomes I, et al. SARS-CoV-2 infection of the central nervous system in a 14-month-old child: a case report of a complete autopsy. Lancet Reg Health - Am. 2021;2:100046. doi: 10.1016/J.LANA.2021.100046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Fandriks L (2017) Roles of the gut in the metabolic syndrome: an overview. J Intern Med 319–336. 10.1111/joim.12584 [DOI] [PubMed]
- 146.Parodi B, KerlerodeRosbo N. The gut-brain axis in multiple sclerosis. Is its dysfunction a pathological trigger or a consequence of the disease? Front Immunol. 2021;12:3911. doi: 10.3389/FIMMU.2021.718220/BIBTEX. [DOI] [PMC free article] [PubMed] [Google Scholar]