Abstract
Objective
Weight change is a dynamic function of whole‐body energy balance resulting from the interplay between energy intake and energy expenditure (EE). Recent reports have provided evidence for the existence of a causal effect of EE on energy intake, suggesting that increased EE may drive overeating, thereby promoting future weight gain. This study investigated the relationships between ad libitum energy intake and 24‐hour EE (24‐h EE) in sedentary conditions versus long‐term, free‐living weight change using a mediation analysis framework.
Methods
Native American individuals (n = 61, body fat by dual‐energy x‐ray absorptiometry: 39.7% [SD 9.5%]) were admitted to the clinical inpatient unit and had baseline measurements as follows: 1) 24‐h EE accurately measured in a whole‐room indirect calorimeter during energy balance and weight stability; and 2) ad libitum energy intake objectively assessed for 3 days using computerized vending machines. Free‐living weight change was assessed after a median follow‐up time of 1.7 years (interquartile range: 1.2‐2.9).
Results
The total effect of 24‐h EE on weight change (−0.23 kg per 100‐kcal/d difference in EE at baseline) could be partitioned into the following two independent and counterbalanced effects: higher EE protective against weight gain (−0.46 kg per 100‐kcal/d difference in EE at baseline) and an orexigenic effect promoting overeating, thereby favoring weight gain (+0.23 kg per 100‐kcal/d difference in EE at baseline).
Conclusions
The overall impact of EE on body weight regulation should be evaluated by also considering its collateral effect on energy intake. Any weight loss intervention aimed to induce energy deficits by increasing EE should take into account any potential orexigenic effects that promote compensatory overeating, thereby limiting the efficacy of these obesity therapies.
Study Importance.
What is already known?
-
►
The balance between energy intake and energy expenditure (EE) determines weight change.
-
►
Recent studies have shown a positive association between energy intake and EE.
What does this study add?
-
►
The impact of EE on weight change is the net result of two counterbalanced effects.
-
►
The energy‐consuming effect protects against weight gain by increasing EE.
-
►
The orexigenic effect promotes overeating and favors weight gain.
How might these results change the direction of research or the focus of clinical practice?
-
►
Any effect due to increased EE could be counteracted by increased drive to overeat.
-
►
Physical activity level may modulate the magnitude of the orexigenic effect.
INTRODUCTION
Weight change is a dynamic function of whole‐body energy balance, i.e., energy intake and energy expenditure (EE). A straightforward view of energy homeostasis in relation to body weight regulation assumes that these two factors independently affect energy balance. However, recent research has suggested that that these two factors interact in contributing to daily energy balance, thereby impacting weight change (1, 2). The causal effect of energy intake on EE (i.e., diet‐induced thermogenesis) has indeed been extensively studied in humans (3, 4, 5, 6, 7, 8, 9, 10, 11). However, the causal effect of EE on energy intake has not been fully explored, to our knowledge, despite seminal research on this topic more than 50 years ago (12, 13, 14, 15). This is because of technical difficulties in the precise assessment of energy intake in humans (16). In the past 10 years, research in this field has resurged, with multiple independent reports (17) showing that higher EE is associated with greater energy intake independently from and mediating the effects of body composition on energy intake (18, 19). This might reflect body energy‐sensing mechanisms attempting to achieve energy balance by centrally regulating energy intake on the basis of the rate of energy expended by the organism (1, 2, 17, 20). Although matching energy intake to expenditure in humans would seem to require a tightly regulated system, our research group showed that interindividual variability in the positive relationship between EE and ad libitum energy intake predicts future weight gain (21). Specifically, participants defined by overeating compared with their 24‐hour EE (24‐h EE; i.e., oversensing their daily energy needs) (1) are more prone to gain weight over time (21).
In order to further elucidate the role of EE on weight change in light of its positive influence on energy intake, we performed a mediation analysis between 24‐h EE (as accurately measured in sedentary, non‐exercise conditions inside a whole‐room calorimeter during energy balance) and objective measures of ad libitum energy intake at baseline versus long‐term, free‐living weight change using our previously published data from a longitudinal natural history study (21).
METHODS
This analysis was performed using data from an ongoing clinical trial (ClinicalTrials.gov identifier: NCT00342732) investigating the clinical determinants of free‐living weight change. From 2008 to 2015, 61 healthy individuals aged 18 to 45 years completed the baseline inpatient admission and had available measurements as follows: 1) 24‐h EE as accurately measured in a whole‐room indirect calorimeter during energy balance and weight stability; and 2) ad libitum energy intake objectively assessed for 3 days using computerized vending machines. The characteristics of the study group, including demographics, EE, body composition at baseline, energy intake measurements over 3 days at baseline, and weight change at the follow‐up visit at least 1 year after the baseline visit, are reported in Table 1.
TABLE 1.
Baseline and follow‐up measurements of study cohort (n = 61)
| Age (y) | 34.0 ± 7.9 |
| Body weight (kg) | 94.3 ± 26.1 |
| BMI (kg/m²) | 33.6 ± 8.5 |
| Body fat (%) | 39.7 ± 9.5 |
| Fat mass (kg) | 38.5 ± 16.8 |
| Fat‐free mass (kg) | 55.8 ± 13.7 |
| 24‐h EE (kcal/d) | 2,419 ± 410 |
| 24‐h spontaneous physical activity (%) | 7.8 ± 7.0 |
| Sleeping EE (kcal/d) | 1,726 ± 297 |
| Weight‐maintaining energy intake (kcal/d) | 2,790 ± 281 |
| Ad libitum energy intake (kcal/d) | 4,386 ± 1,144 |
| Ad libitum energy intake (% of weight‐maintaining energy needs) | 154 ± 43 |
| Food weight (g) | 3,024 ± 796 |
| Energy density (kcal/g) | 1.46 ± 0.26 |
| Follow‐up visit | |
| Weight change (kg) | 3.4 ± 7.5 |
| Follow‐up time (y) | 1.7 (1.2‐1.9) |
Data are presented as mean ± SD, except for follow‐up time, which is reported as median with interquartile range in brackets.
Abbreviation: EE, energy expenditure.
Upon admission to the clinical research unit, a weight‐maintaining diet (WMD; 50% carbohydrate, 30% fat, and 20% protein) was provided daily to the participants, and its caloric intake was calculated using unit‐specific equations based on gender and weight. Body weight was recorded daily in the morning after an overnight fast and was maintained within 1% of the admission weight by adjusting the WMD by ±200 kcal/d if needed. On the second day of admission, dual‐energy x‐ray absorptiometry (DXA; DPX‐1 or DPX‐L; Lunar Radiation) was performed to assess body composition with fat mass and fat‐free mass calculated from percentage of body fat. An internally validated regression equation was used to make DXA measurements comparable between different DXA machines.
A 3‐hour oral glucose tolerance test was performed after 3 days on the WMD, and all participants were determined to be free from diabetes based on American Diabetes Association (ADA) criteria (22). All participants provided written informed consent prior to beginning the study. The study was approved by the Institutional Review Board of the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK).
EE measurement
After at least 5 days on the WMD, 24‐h EE was continuously measured in a large, open‐circuit, whole‐room indirect calorimeter (respiratory chamber), as previously described (23, 24, 25, 26, 27, 28). Briefly, participants entered the respiratory chamber after an overnight fast. Four meals were then provided to volunteers inside the chamber via an airlock at 8:00 am, 11:00 am, 4:00 pm, and 7:00 pm. The total energy content of the four meals given was calculated using previously described equations to achieve energy balance in the confined setting of the respiratory chamber. All unconsumed food was returned and weighed in the metabolic kitchen to determine the actual energy intake during each 24‐hour session inside the chamber. While residing in the respiratory chamber, participants were instructed to remain sedentary and not to exercise. Spontaneous physical activity was measured by radar sensors and expressed as the percentage of time over 24 hours when activity was detected (29). Volunteers resided in the calorimeter for 23.25 hours, during which both carbon dioxide (CO2) production and oxygen (O2) consumption were recorded and calculated every 15 minutes, averaged, and extrapolated to 24 hours. The 24‐hour respiratory quotient was calculated as the ratio of 24‐hour CO2 production to 24‐hour O2 consumption, and 24‐h EE was calculated using the Lusk’s equation (30). Sleeping EE was derived as the average EE of all 15‐minute nightly periods between 11:30 pm and 5:00 am during which spontaneous physical activity was <1.5%.
Ad libitum energy intake assessment
Following the 24‐h EE measurement inside the respiratory chamber, ad libitum energy intake was measured over 3 days using automated vending machines as previously described (31, 32, 33). This objective method has a high intraindividual reproducibility (intraclass correlation coefficient = 0.9) for the measurement total energy intake in repeated visits (34). Before accessing the vending machines, participants completed the Geiselman Food Preference Questionnaire (35), which was used to determine food preferences for stocking of vending machines. Specifically, participants were asked to rate their preference for a variety of foods on a nine‐point Likert scale, and 40 different foods, which were given an intermediate rating, were used to stock the vending machines for the 3‐day ad libitum period. These 40 foods, which were weighed before being loaded into the vending machine by the metabolic kitchen staff, were available each of the 3 days, along with condiments. Participants were given free access to the machines for 23.5 hours and were instructed to eat only in the vending room, to eat whatever they wished whenever they desired, and to return the unconsumed food portions to the metabolic kitchen for calculation of actual calories consumed. Television viewing during food consumption was prohibited. In order to access food, each participant entered a unique code into the computer operated vending machine, which recorded the time and date that each food item was dispensed. Ingredients and weight data for each item were imported into Food Processor SQL Edition (version 10.0.0, ESHA Research), which provided calories and macronutrient (fats, proteins, and carbohydrates) content of the foods. The individual energy intake was calculated as the 3‐day average of daily total energy intake and expressed as kilocalories per day.
Free‐living weight change
Upon completion of the inpatient admission, participants were not provided with any lifestyle counseling and were advised to return to their usual habits. Following discharge, all participants had a follow‐up outpatient visit for the measurement of weight change after a median follow‐up time of 1.7 years (interquartile range: 1.2‐1.9 years).
Statistical analysis
Mediation analysis based on hierarchical multiple regression models (36) was performed using the causal model framework shown in Figure 1 to partition the total effect of 24‐EE (independent variable) on weight change (dependent variable) into the direct, “energy‐consuming” effect of 24‐h EE per se and the indirect, “orexigenic” effect via ad libitum energy intake (mediator). Specifically, the indirect effect of 24‐h EE on weight change was calculated as the product of the two path coefficients between 24‐h EE → ad libitum energy intake and ad libitum energy intake → weight change, in which the following applies: 1) the path coefficient between 24‐h EE and ad libitum energy intake was calculated as the regression coefficient of 24‐h EE with ad libitum energy intake as dependent variable; and 2) the path coefficient between ad libitum energy intake and weight change was calculated as the regression coefficient of ad libitum energy intake from the multivariate regression model including 24‐h EE as covariate and weight change as dependent variable. Finally, the direct effect of 24‐h EE on weight change was calculated as the regression coefficient of 24‐h EE with weight change as a dependent variable. Path coefficients are shown as mean (SE).
FIGURE 1.
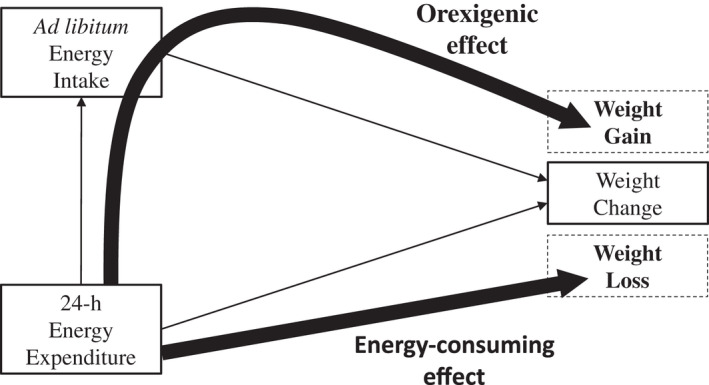
Dissecting the dual effects of 24‐h EE on weight change by considering its influence on ad libitum energy intake. Mediation analysis was performed using the causal model framework described previously (18) to partition the total effect of 24‐h EE (independent variable) on weight change (dependent variable, median follow‐up time: 1.7 years) into the direct, “energy‐consuming” effect of 24‐h EE per se and the indirect, “orexigenic” effect via ad libitum energy intake (mediator). At the baseline inpatient visit, participants had accurate measurements of 24‐h EE in a whole‐room indirect calorimeter during eucaloric conditions and objective assessments of ad libitum energy intake over 3 days using a highly reproducible computerized vending machine paradigm. Following discharge, all individuals had a follow‐up outpatient visit when body weight was recorded. EE, energy expenditure; 24‐h EE, 24‐hour EE
RESULTS
The current reanalysis showed that the total effect of 24‐h EE on weight change can be partitioned into the following two independent and counterbalanced effects (Figure 1): 1) the expected, energy‐consuming effect that is protective of future weight gain; and 2) the orexigenic effect that promotes overeating, thereby favoring future weight gain. As shown (Figure 2), the protective calorie‐burning effect of 24‐h EE per se on future weight gain (−0.46 kg per 100‐kcal/d difference in 24‐h EE at baseline) is counterbalanced by the concomitant positive influence of 24‐h EE on energy intake (e.g., greater degree of overeating), which is ultimately promoting weight gain (+0.23 kg per 100‐kcal/d difference in 24‐h EE). Although protective overall against weight gain, the total (observed) effect of 24‐h EE on weight change (−0.23 kg per 100‐kcal/d difference in 24‐h EE), which is the net sum of the two aforementioned effects estimated by the mediation analysis, is therefore lower than that expected by 24‐h EE per se, as it is reduced by its orexigenic effect that increases energy intake, thereby promoting weight gain. Specifically, in absolute terms, the magnitude of this orexigenic effect attributable to 24‐h EE (0.23 kg per 100‐kcal/d difference in 24‐h EE) lowers the energy‐consuming protective effect (0.46 kg per 100‐kcal/d difference in 24‐h EE) of 24‐h EE against weight gain by half.
FIGURE 2.
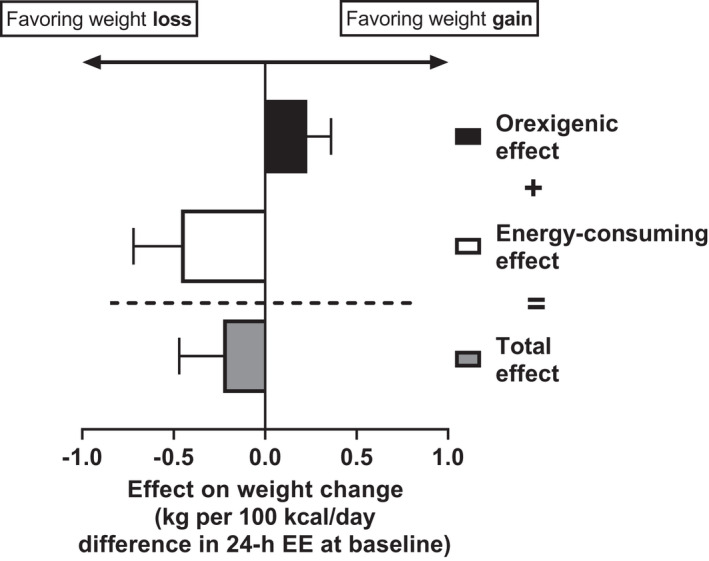
Counterbalancing effects of 24‐h EE on weight change. Effects are shown as mean (SE). The total effect of 24‐h EE on weight change (−0.23 kg per 100‐kcal/d difference in 24‐h EE) is the net sum of the direct, “energy‐consuming” effect of 24‐h EE per se (−0.46 kg per 100 kcal/d) and the indirect, “orexigenic” effect via ad libitum energy intake (+0.23 kg per 100 kcal/d). The indirect effect of 24‐h EE on weight change was calculated as the product of the two path coefficients between 24‐h EE → ad libitum energy intake and ad libitum energy intake → weight change, as shown in the mediation analysis scheme of Figure 1, in which the following apply: 1) the path coefficient between 24‐h EE and ad libitum energy intake was calculated as the β coefficient estimate of 24‐h EE from the respective linear regression model; and 2) the path coefficient between ad libitum energy intake and weight change was calculated as the β coefficient estimate of ad libitum energy intake from the multivariate regression model including 24‐h EE as covariate. EE, energy expenditure; 24‐h EE, 24‐hour EE
DISCUSSION
The magnitude of each counterbalancing effect of EE on weight change may depend on factors such as ethnicity, body habitus, and physical activity level. Our measurements of 24‐h EE obtained in a whole‐room indirect calorimeter represent the individual’s total EE in sedentary (non‐exercise) conditions during controlled dietary condition of energy balance, i.e., during a low level of daily energy turnover. When levels of daily energy turnover are lower, control of appetite is weaker (37) (i.e., ad libitum energy intake is greater than 24‐h EE, thereby leading to positive energy balance), suggesting that the presence and magnitude of the orexigenic effect might be greater in sedentary conditions, as in our study. Conversely, the orexigenic effect may be negligible or absent in settings of higher physical activity levels, which has been more commonly observed in free‐living conditions, as it has been recently demonstrated that overeating and positive energy balance are less likely in conditions of higher energy turnover levels (37).
Furthermore, the reduced level of physical activity in the artificial environment of the metabolic ward in concert with the extent of overeating observed during the vending period (i.e., ad libitum energy intake was, on average, 54% higher than the weight‐maintaining energy needs; Table 1) may not reflect that of free‐living conditions, thus limiting the generalization of our current findings. However, the energy intake assessment by this objective method provides highly reproducible measures within an individual (intraclass correlation coefficient, ICC = 0.9, p < 0.0001) (34), representing one of the most precise and consistent methods to objectively measure energy intake in a clinical setting as opposed to food frequency questionnaires (38) and self‐reported diet diaries (39) used to measure energy intake in real‐life settings, which, in turn, are notoriously inaccurate, as individuals tend to miscalculate and underreport their daily intake.
The existence of an orexigenic effect attributable to EE might partly explain the varying results in the relationship between EE and weight change in multiple studies conducted in different ethnic groups showing direct, inverse, or no relationships (1). These variable results may be due to differing magnitude of the two counterbalancing effects attributable to EE (energy‐consuming vs. orexigenic) on weight change. For instance, the relationship between lower EE and weight gain consistently found in Native American individuals of the Southwestern United States (40, 41, 42, 43) suggests that the magnitude of the orexigenic effect on weight change may be smaller than that of the energy‐consuming effect. However, this orexigenic effect is present and it presumably counterbalances (or blunts) the energy‐consuming effect on weight gain in this ethnic group, resulting in an attenuation, but not in a sign change, of the directionality of the EE‐weight change relationship as in our current study (i.e., from −0.46 to −0.23 kg per 100‐kcal/d difference in 24‐h EE, as shown in Figure 2). Conversely, the reported relationship between higher (instead of lower) EE and weight gain found in Black Nigerian individuals (44) may imply that the magnitude of the orexigenic effect could be larger compared with that in Native Americans and could be responsible for a greater energy intake that may offset EE, thereby leading to weight gain. In summary, the existence of the orexigenic effect may greatly influence the effect of total EE on weight change depending on the setting (e.g., low‐ or high‐energy turnover level, sedentary vs. active conditions, low‐ vs. high‐energy density of food consumed, different exercise paradigms to acutely increase EE, ethnic and environmental differences, etc.) and may be mediated by thermogenic hormones acting centrally to modulating energy intake, such as fibroblast growth factor 21 (FGF21) (33, 45, 46).
In conclusion, the overall effect of EE on daily energy balance and weight change should be evaluated not only by considering its calorie‐burning effect but also in light of its collateral orexigenic effect on energy intake. These results suggest that clinical interventions aimed to induce energy deficits by increasing EE should take into account any potential orexigenic side effects that might reduce (or even prevent) the extent of weight loss by promoting overeating.O
CONFLICT OF INTEREST
The authors declared no conflict of interest.
CLINICAL TRIAL REGISTRATION
ClinicalTrials.gov identifier NCT00342732.
ACKNOWLEDGMENTS
The authors would like to thank the study participants and the nursing, clinical, and dietary staffs, as well as the laboratory technicians of the clinical research center for their valuable assistance and care of the volunteers. Data described in the manuscript will be made available upon request pending application and approval by the Institutional Review Board of the NIDDK. Open Access Funding provided by Universita degli Studi di Pisa within the CRUI‐CARE Agreement. [Correction added on 24 May 2022, after first online publication: CRUI funding statement has been added.].
Piaggi P, Basolo A, Martin CK, Redman LM, Votruba SB, Krakoff J. The counterbalancing effects of energy expenditure on body weight regulation: Orexigenic versus energy‐consuming mechanisms. Obesity (Silver Spring). 2022;30:639–644. doi: 10.1002/oby.23332
Funding information
This research was supported by the Intramural Research Program of the NIH National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). PP was supported by the program “Rita Levi Montalcini for young researchers” from the Italian Minister of Education and Research (Ministry of Education, University and Research)
REFERENCES
- 1. Piaggi P. Metabolic determinants of weight gain in humans. Obesity (Silver Spring). 2019;27:691‐699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Piaggi P, Vinales KL, Basolo A, Santini F, Krakoff J. Energy expenditure in the etiology of human obesity: spendthrift and thrifty metabolic phenotypes and energy‐sensing mechanisms. J Endocrinol Invest. 2018;41:83‐89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Secor SM. Specific dynamic action: a review of the postprandial metabolic response. J Comp Physiol B. 2009;179:1‐56. [DOI] [PubMed] [Google Scholar]
- 4. Tataranni PA, Larson DE, Snitker S, Ravussin E. Thermic effect of food in humans: methods and results from use of a respiratory chamber. Am J Clin Nutr. 1995;61:1013‐1019. [DOI] [PubMed] [Google Scholar]
- 5. Reed GW, Hill JO. Measuring the thermic effect of food. Am J Clin Nutr. 1996;63:164‐169. [DOI] [PubMed] [Google Scholar]
- 6. de Jonge L, Bray GA. The thermic effect of food and obesity: a critical review. Obes Res. 1997;5:622‐631. [DOI] [PubMed] [Google Scholar]
- 7. Thorne A. Diet‐induced thermogenesis. An experimental study in healthy and obese individuals. Acta Chir Scand Suppl. 1990;558:6‐59. [PubMed] [Google Scholar]
- 8. Schlögl M, Piaggi P, Thiyyagura P et al. Overfeeding over 24 hours does not activate brown adipose tissue in humans. J Clin Endocrinol Metab. 2013;98:E1956‐E1960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Hollstein T, Ando T, Basolo A, Krakoff J, Votruba SB, Piaggi P. Metabolic response to fasting predicts weight gain during low‐protein overfeeding in lean men: further evidence for spendthrift and thrifty metabolic phenotypes. Am J Clin Nutr. 2019;110:593‐604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Hollstein T, Basolo A, Ando T, Krakoff J, Piaggi P. Reduced adaptive thermogenesis during acute protein‐imbalanced overfeeding is a metabolic hallmark of the human thrifty phenotype. Am J Clin Nutr. 2021;114:1396‐1407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Begaye B, Vinales KL, Hollstein T et al. Impaired metabolic flexibility to high‐fat overfeeding predicts future weight gain in healthy adults. Diabetes. 2020;69:181‐192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Edholm OG, Fletcher JG, Widdowson EM, McCance RA. The energy expenditure and food intake of individual men. Br J Nutr. 1955;9:286‐300. [DOI] [PubMed] [Google Scholar]
- 13. Mayer J, Roy P, Mitra KP. Relation between caloric intake, body weight, and physical work: studies in an industrial male population in West Bengal. Am J Clin Nutr. 1956;4:169‐175. [DOI] [PubMed] [Google Scholar]
- 14. Edholm OG, Adam JM, Healy MJ, Wolff HS, Goldsmith R, Best TW. Food intake and energy expenditure of army recruits. Br J Nutr. 1970;24:1091‐1107. [DOI] [PubMed] [Google Scholar]
- 15. Edholm OG. Energy balance in man studies carried out by the Division of Human Physiology, National Institute for Medical Research. J Hum Nutr. 1977;31:413‐431. [PubMed] [Google Scholar]
- 16. Lam YY, Ravussin E. Analysis of energy metabolism in humans: a review of methodologies. Mol Metab. 2016;5:1057‐1071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Blundell JE, Gibbons C, Beaulieu K et al. The drive to eat in homo sapiens: energy expenditure drives energy intake. Physiol Behav. 2020;219:112846. doi: 10.1016/j.physbeh.2020.112846 [DOI] [PubMed] [Google Scholar]
- 18. Piaggi P, Thearle MS, Krakoff J, Votruba SB. Higher daily energy expenditure and respiratory quotient, rather than fat‐free mass, independently determine greater ad libitum overeating. J Clin Endocrinol Metab. 2015;100:3011‐3020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Hopkins M, Finlayson G, Duarte C et al. Modelling the associations between fat‐free mass, resting metabolic rate and energy intake in the context of total energy balance. Int J Obes (Lond). 2005;2016:312‐318. [DOI] [PubMed] [Google Scholar]
- 20. Dulloo AG, Jacquet J, Miles‐Chan JL, Schutz Y. Passive and active roles of fat‐free mass in the control of energy intake and body composition regulation. Eur J Clin Nutr. 2017;71:353‐357. [DOI] [PubMed] [Google Scholar]
- 21. Basolo A, Votruba SB, Heinitz S, Krakoff J, Piaggi P. Deviations in energy sensing predict long‐term weight change in overweight Native Americans. Metabolism. 2018;82:65‐71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Expert Committee on the Diagnosis and Classification of Diabetes Mellitus . Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care. 2003;26(suppl 1):S5‐S20. [DOI] [PubMed] [Google Scholar]
- 23. Ravussin E, Lillioja S, Anderson TE, Christin L, Bogardus C. Determinants of 24‐hour energy expenditure in man. Methods and results using a respiratory chamber. J Clin Invest. 1986;78:1568‐1578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Piaggi P, Thearle MS, Bogardus C, Krakoff J. Fasting hyperglycemia predicts lower rates of weight gain by increased energy expenditure and fat oxidation rate. J Clin Endocrinol Metab. 2015;100:1078‐1087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Stinson EJ, Graham AL, Thearle MS, Gluck ME, Krakoff J, Piaggi P. Cognitive dietary restraint, disinhibition, and hunger are associated with 24‐h energy expenditure. Int J Obes (Lond). 2019;43:1456‐1465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Piaggi P, Masindova I, Muller YL et al. A genome‐wide association study using a custom genotyping array identifies variants in GPR158 associated with reduced energy expenditure in American Indians. Diabetes. 2017;66:2284‐2295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Heinitz S, Hollstein T, Ando T et al. Early adaptive thermogenesis is a determinant of weight loss after six weeks of caloric restriction in overweight subjects. Metabolism. 2020;110:154303. doi: 10.1016/j.metabol.2020.154303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Piaggi P, Köroğlu Ç, Nair AK et al. Exome sequencing identifies a nonsense variant in DAO associated with reduced energy expenditure in American Indians. J Clin Endocrinol Metab. 2020;105:e3989‐e4000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Piaggi P, Krakoff J, Bogardus C, Thearle MS. Lower “awake and fed thermogenesis” predicts future weight gain in subjects with abdominal adiposity. Diabetes. 2013;62:4043‐4051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Lusk G. Animal calorimetry: analysis of oxidation of mixtures of carbohydrates and fat. J Biol Chem. 1924;59:41‐42. [Google Scholar]
- 31. Rising R, Alger S, Boyce V et al. Food intake measured by an automated food‐selection system: relationship to energy expenditure. Am J Clin Nutr. 1992;55:343‐349. [DOI] [PubMed] [Google Scholar]
- 32. Basolo A, Ando T, Hollstein T, Votruba SB, Krakoff J, Piaggi P. Higher urinary dopamine concentration is associated with greater ad libitum energy intake in humans. Obesity (Silver Spring). 2020;28:953‐961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Basolo A, Hollstein T, Shah MH et al. Higher fasting plasma FGF21 concentration is associated with lower ad libitum soda consumption in humans. Am J Clin Nutr. 2021;114:1518‐1522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Venti CA, Votruba SB, Franks PW, Krakoff J, Salbe AD. Reproducibility of ad libitum energy intake with the use of a computerized vending machine system. Am J Clin Nutr. 2010;91:343‐348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Geiselman PJ, Anderson AM, Dowdy ML, West DB, Redmann SM, Smith SR. Reliability and validity of a macronutrient self‐selection paradigm and a food preference questionnaire. Physiol Behav. 1998;63:919‐928. [DOI] [PubMed] [Google Scholar]
- 36. MacKinnon DP. Introduction to Statistical Mediation Analysis. Routledge; 2008. [Google Scholar]
- 37. Hägele FA, Büsing F, Nas A et al. Appetite control is improved by acute increases in energy turnover at different levels of energy balance. J Clin Endocrinol Metab. 2019;104:4481‐4491. [DOI] [PubMed] [Google Scholar]
- 38. Weber JL, Reid PM, Greaves KA et al. Validity of self‐reported energy intake in lean and obese young women, using two nutrient databases, compared with total energy expenditure assessed by doubly labeled water. Eur J Clin Nutr. 2001;55:940‐950. [DOI] [PubMed] [Google Scholar]
- 39. Day N, McKeown N, Wong M, Welch A, Bingham S. Epidemiological assessment of diet: a comparison of a 7‐day diary with a food frequency questionnaire using urinary markers of nitrogen, potassium and sodium. Int J Epidemiol. 2001;30:309‐317. [DOI] [PubMed] [Google Scholar]
- 40. Ravussin E, Lillioja S, Knowler WC et al. Reduced rate of energy expenditure as a risk factor for body‐weight gain. New Engl J Med. 1988;318:467‐472. [DOI] [PubMed] [Google Scholar]
- 41. Tataranni PA, Harper IT, Snitker S et al. Body weight gain in free‐living Pima Indians: effect of energy intake vs expenditure. Int J Obes Relat Metab Disord. 2003;27:1578‐1583. [DOI] [PubMed] [Google Scholar]
- 42. Piaggi P, Thearle MS, Bogardus C, Krakoff J. Lower energy expenditure predicts long‐term increases in weight and fat mass. J Clin Endocrinol Metab. 2013;98:E703‐E707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Zurlo F, Lillioja S, Esposito‐Del Puente A et al. Low ratio of fat to carbohydrate oxidation as predictor of weight gain: study of 24‐h RQ. Am J Physiol. 1990;259(5 Pt 1):E650‐E657. [DOI] [PubMed] [Google Scholar]
- 44. Luke A, Durazo‐Arvizu R, Cao G, Adeyemo A, Tayo B, Cooper R. Positive association between resting energy expenditure and weight gain in a lean adult population. Am J Clin Nutr. 2006;83:1076‐1081. [DOI] [PubMed] [Google Scholar]
- 45. Hollstein T, Basolo A, Ando T et al. Recharacterizing the metabolic state of energy balance in thrifty and spendthrift phenotypes. J Clin Endocrinol Metab. 2020;105:1375‐1392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Vinales KL, Begaye B, Bogardus C, Walter M, Krakoff J, Piaggi P. FGF21 is a hormonal mediator of the human “thrifty” metabolic phenotype. Diabetes. 2019;68:318‐323. [DOI] [PMC free article] [PubMed] [Google Scholar]


