Abstract
Purpose
The optimal ventilatory settings in patients after cardiac arrest and their association with outcome remain unclear. The aim of this study was to describe the ventilatory settings applied in the first 72 h of mechanical ventilation in patients after out-of-hospital cardiac arrest and their association with 6-month outcomes.
Methods
Preplanned sub-analysis of the Target Temperature Management-2 trial. Clinical outcomes were mortality and functional status (assessed by the Modified Rankin Scale) 6 months after randomization.
Results
A total of 1848 patients were included (mean age 64 [Standard Deviation, SD = 14] years). At 6 months, 950 (51%) patients were alive and 898 (49%) were dead. Median tidal volume (VT) was 7 (Interquartile range, IQR = 6.2–8.5) mL per Predicted Body Weight (PBW), positive end expiratory pressure (PEEP) was 7 (IQR = 5–9) cmH20, plateau pressure was 20 cmH20 (IQR = 17–23), driving pressure was 12 cmH20 (IQR = 10–15), mechanical power 16.2 J/min (IQR = 12.1–21.8), ventilatory ratio was 1.27 (IQR = 1.04–1.6), and respiratory rate was 17 breaths/minute (IQR = 14–20). Median partial pressure of oxygen was 87 mmHg (IQR = 75–105), and partial pressure of carbon dioxide was 40.5 mmHg (IQR = 36–45.7). Respiratory rate, driving pressure, and mechanical power were independently associated with 6-month mortality (omnibus p-values for their non-linear trajectories: p < 0.0001, p = 0.026, and p = 0.029, respectively). Respiratory rate and driving pressure were also independently associated with poor neurological outcome (odds ratio, OR = 1.035, 95% confidence interval, CI = 1.003–1.068, p = 0.030, and OR = 1.005, 95% CI = 1.001–1.036, p = 0.048). A composite formula calculated as [(4*driving pressure) + respiratory rate] was independently associated with mortality and poor neurological outcome.
Conclusions
Protective ventilation strategies are commonly applied in patients after cardiac arrest. Ventilator settings in the first 72 h after hospital admission, in particular driving pressure and respiratory rate, may influence 6-month outcomes.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00134-022-06756-4.
Keywords: Mechanical ventilation, Cardiac arrest, Outcome, Mechanical power, Driving pressure, Ventilator settings
Take-home message
| Protective ventilation strategies are more commonly applied in patients after cardiac arrest. Ventilatory settings in the first 72 h after hospital admission may influence the 6-month outcomes. |
Introduction
Post cardiac arrest syndrome is characterized by high mortality and morbidity rates, and several strategies have been implemented with the aim to improve survival and neurological outcome [1]. Among these, research has focused on the optimization of respiratory function and the prevention of pulmonary complications, which are common in this population [1, 2].
Mechanical ventilation has the aim to provide appropriate gas exchange (arterial partial pressure of oxygen, PaO2 and arterial partial pressure of carbon dioxide, PaCO2), which can have important effects on the development of secondary brain damage, cerebral blood flow, and cerebrovascular dynamics, and patient’s survival rate [2]. The pathophysiology of cardiac arrest and its systemic effects, as well as the relationship between ventilatory settings and cerebral hemodynamics after resuscitation is complex and not completely elucidated [2]. The literature on the acute respiratory distress syndrome (ARDS) [3] and non-ARDS [4] population has highlighted the importance of the use of lung protective strategies (i.e. low tidal volume, low plateau pressure) to optimize patients’ outcome [4]. Only few and mostly small studies [5] have focused on the effect of mechanical ventilator settings on outcome after cardiac arrest, with no definitive conclusions [6]. Also, the role of parameters such as driving pressure (DP) and mechanical power (MP), which have shown to be potentially associated with ventilator-induced lung injury and worsened outcomes in the non-ARDS and ARDS population, has not been investigated so far in patients after cardiac arrest [7].
We performed a pre-planned secondary analysis of the Target Temperature Management-2 (TTM2) trial. The primary aim of this study was to describe the ventilator settings applied in a homogeneous population of adults after out of hospital cardiac arrest (OHCA) admitted to the intensive care unit (ICU). The secondary aim was to assess the association between ventilator settings and 6-month mortality and neurological outcome [8]. We hypothesized that patients after OHCA are ventilated using lung protective strategies and that some mechanical ventilator settings, in particular tidal volume, respiratory rate, plateau pressure, positive end expiratory pressure, driving pressure, mechanical power, and ventilatory ratio, would be associated with patients’ outcomes (mortality and neurological outcome).
Methods
The TTM2 trial (registered at clinicaltrials.gov NCT02908308) is an international trial randomizing 1861 mechanically ventilated post-cardiac arrest patients with 6-month follow-up. According to the TTM2 protocol, at ICU admission, patients were randomized to normothermia (931 patients, with the aim to maintain a temperature of 37.5 °C or less), and hypothermia (930 patients, target temperature 33 °C until 28 h after randomization, followed by rewarming to 37 °C in hourly increments in one third of a degree) [9, 10]. The Ethic Committees approved the TTM2 study in all participating centres and informed consent was obtained according to local regulations. No further ethical approval was necessary for this subanalysis. We performed a pre-planned analysis focusing on the mechanical ventilation strategies used in the first 72 h in these patients. This sub-study was approved on the 23rd of February 2017 by the TTM2 steering committee (https://ttm2trial.org/substudy-proposals) and the study proposal was then published [8–10]. This report was performed according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines [11] (Electronic Supplemental Material, ESM 1).
Inclusion and exclusion criteria
The TTM2 trial included adult patients (18 years of age or older) admitted to the hospital after out-of-hospital cardiac arrest of presumed cardiac or unknown cause with a return of spontaneous circulation. Eligible patients experienced sustained return of spontaneous circulation (ROSC), were unconscious after ROSC, and required ICU admission and mechanical ventilation. Main exclusion criteria were an interval from ROSC to screening of more than 180 min, unwitnessed cardiac arrest with an initial rhythm of asystole, temperature on admission < 30 °C, obvious or suspected pregnancy, intracranial bleeding at admission. Details regarding the inclusion and exclusion criteria are provided in the main manuscript and protocol [8–10]. We restricted this analysis to TTM-2 trial participants who had data pertaining to mechanical ventilation settings available from at least the first 24 h after hospital admission.
Objectives
The primary objective of this study was to describe the ventilatory settings/ parameters used in mechanically ventilated patients included in the TTM2 trial. Among these, we focused on basic settings–tidal volume (VT), positive end expiratory pressure (PEEP), Plateau pressure (Pplat), respiratory rate (RR)—and composite settings- driving pressure (DP), mechanical power (MP), and ventilatory ratio (VR). Secondary objectives were to evaluate the association of these parameters with patients’ 6-month mortality and neurological outcome.
Clinical outcome measures
Six months mortality and patients’ neurological status, assessed by the Modified Rankin Scale (mRS), were defined as clinical outcome measures. Binary 6 months mRS was used to define poor outcome (mRS = 4–6) and good outcome (mRS = 1–3), respectively. Additional clinical outcomes were ICU mortality, hospital mortality, hospital length of stay, duration of mechanical ventilation, ventilator free days at ICU discharge, and at 30 days. Further details on the study procedure and patients’ clinical management have been previously published [8, 10].
Study procedures and data collection
Data were collected at the time of enrollment, at hospital admission, during the ICU-stay, at ICU-discharge, at hospital-discharge, and at follow-up. Clinical, laboratory, and background data were collected from hospital records, relatives, and ambulance services. Data of the TTM2 trial used for this secondary analysis included patients’ demographic characteristics, pre-injury co-morbidities (including Charlson comorbidity index [12]), and in particular cardiological issues, timing, type and management of cardiac arrest, clinical presentation, data regarding daily ventilator settings/parameters and respiratory mechanics (VT, PEEP, RR, MP, DP, VR, Pplat, static respiratory system compliance (Crs)), arterial blood gases values (pHa, PaO2, PaCO2, base excess) and outcomes.
For 6-month follow up, all responses were obtained by study personnel from patients or from a proxy (where impaired cognitive capacity prevented patient interview), during a face-to-face visit, by telephone interview, or by postal questionnaire. General Intensive Care Unit Care including ventilatory management were according to local care plans at the discretion of the treating physicians.
Ventilatory settings were collected from randomization every 4 h for the first 32 h, and then every 8 h until day 3 (72 h). Crs was calculated as VT (ml)/ .
Mechanical power was estimated according to previously published evidence [13]. Ventilatory ratio was calculated according to the following formula [14]:
According to Costa et al. [7] we also tested the following formula as potential determinant of mortality and poor neurological outcome: [(4* Driving Pressure) + RR].
Statistical analysis
Being a secondary analysis of a randomized trial and having a broad spectrum of exposures (all ventilatory settings), a formal sample size calculation could not be performed a priori for the present study. However, the achieved sample included 898 death events, allowing us to keep the ratio between events and covariates well above the conventional threshold of 10:1. Patient and ventilator characteristics, and arterial blood gases values were described by means ± standard deviation (SD), or medians (interquartile range, IQR) when appropriate. Discrete variables were summarized as percentages. At baseline, the comparisons of means, medians, and frequencies among 6-month survival status were carried out using t-test, Wilcoxon–Mann–Whitney test, and chi-square test, respectively.
Six-month mortality.
The association between baseline ventilator settings and arterial blood gases with mortality was determined with Cox regression analysis. Overall, all regression models were built with variables chosen based on previous knowledge and aims of the study. Essentially, we built five models: (1) basic ventilatory markers, (2) a model for driving pressure; (3) a model for mechanical power; (4) a model for ventilatory ratio; and (5) a model for respiratory system compliance. The basic ventilatory markers model was adjusted by the TTM2 randomization group (from the original randomized controlled trial), arterial blood gases values (pH, PaO2/FiO2, PaCO2, base excess), the basic ventilatory markers (VT, PEEP, RR, and Pplat), patients’ clinical characteristics (age, gender, Body Mass Index (BMI), height, Charlson as a comorbidity score, in state of shock at admission, and ST-elevation myocardial infarction (STEMI) diagnosis on admission), and onsite-related cardio-pulmonary resuscitation (CPR) variables (time of ROSC, bystander performed CPR, cardiac arrest physical location, initial cardiac rhythm, witnesses of cardiac arrest). Composite ventilatory settings derived from other ventilatory constituents (DP, VR, MP, and Crs) were not included in this model due to a high correlation with their constituents, leading to biased estimates driven by multicollinearity. Thus, each of these composite markers was modeled individually, although using a similar set of adjusting covariates. In all models, we ensured that the exposure of interest (ventilator settings) met the linearity assumption through a transformation with the appropriate fractional polynomials [15]. For un-transformed variables, risk estimates were expressed as hazard ratios (HRs) with 95% confidence intervals (CIs). Those transformed by fractional polynomials the association with mortality was depicted through a graph where the HR on the y-scale was plotted against the continuum of the marker. In tables of regression estimates, the interpretation of the HR (95% CI) of the variables transformed with polynomials is per change in 1 unit of the transformed variable, with no-direct clinical interpretation. The p-value associated with each non-linear trajectory (omnibus p-value) indicated that at least 1 point if the continuum of the variable reached the level of significance. This issue of non-linearity association precluded summarizing the results as single HR. Additional analyses were performed to ensure that our regression estimates do not differed significantly with those that included center as a cluster variable; an stratified analysis assessing the effect on mortality for driving pressure, mechanical power and respiratory rate according to elastance values; and for testing the formula proposed by Costa et al. [7] [(4*Driving Pressure) + RR] and its comparison with mechanical power setting. Using relative distribution analysis [16], we searched for the best cut point along the continuum of the marker that separated those who died versus those alive at the end of the follow-up. Linear mixed regression was used to compare the longitudinal trajectories of these hourly measured markers among survival status (dead vs alive). To account for the longitudinal nature of the data (interdependence among repeated measures on the same subject), these models included a random effect (intercept) on subject ID.
Neurological status at 6-month.
For this binary endpoint, poor outcome in the mRS scoring system, a logistic regression analysis was used. A similar set of models were built (as for the mortality endpoint), and all of them used a similar set of adjusting covariates. All continuous variables were modeled in the original scale. Risk estimates were expressed as odds ratios (ORs) with 95% CIs. A 2-sided p value of < 0.05 was the threshold used for significance in all analyses. Stata 16.1 was used for data preparation and statistical analysis.
Results
Baseline, pre-injury characteristics of the overall population
From 1861 patients included in the TTM2 trial, a total of 1848 patients were included in the analysis of this substudy (ESM, Fig. S1). Thirteen patients (0.7%) were excluded because no data on mechanical ventilation settings were available in the first 24 h. Patients’ characteristics, pre-hospital factors, ventilator settings at admission and in the study period and outcomes are summarized in Table 1, ESM Table S1-S5. The mean age of the patients was 64 (Standard Deviation, SD = 14) years, and 379 (20%) were female. Mean BMI was 27.5 (SD = 5.7) kg/m2. During a median follow-up of 169 days (IQR = 155–187), 898 patients (49%) died. Most cases of death occurred in the first 2 weeks following ICU admission (ESM, Figure S2). At 6-month follow-up, neurological outcome (mRS) was evaluated in 1747 patients. Among survivors, 967 (55%) had poor neurological outcome.
Table 1.
Baseline patients’ characteristics, comorbidities, pre-hospital settings/interventions of the overall population and stratified according to 6 months survival status
| Total n = 1848 |
Survivors n = 950 (51%) |
Non-survivors n = 898 (49%) |
|
|---|---|---|---|
| Baseline patients characteristics | |||
| Age, years, mean (SD) | 64 (14) | 59 (14) | 68 (12) |
| Gender (female), n (%) | 379 (20) | 153 (16) | 226 (25) |
| Height, cm, mean (SD) | 174 (9) | 176 (9) | 173 (9) |
| Weight, Kg, mean (SD) | 83 (17) | 85 (16) | 82 (19) |
| BMI, Kg/m2, mean (SD) | 27.5 (5.7) | 27.3 (5.4) | 27.7 (6.1) |
| Comorbidities | |||
| Hypertension, n (%) | 640 (35) | 289 (30) | 351 (39) |
| Diabetes, n (%) | 336 (18) | 138 (14) | 198 (22) |
| Myocardial infarction, n (%) | 291 (16) | 127 (13) | 164 (18) |
| Previous percutaneous coronary intervention, n (%) | 267 (14) | 121 (13) | 146 (16) |
| Coronary artery bypass graft, n (%) | 147 (8) | 62 (6) | 85 (10) |
| Heart failure, n (%) | 181 (10) | 54 (6) | 127 (14) |
| Charlson comorbidity index, median (IQR) | 3 (1; 4) | 2 (1; 3) | 4 (2; 5) |
| Pre-hospital settings/interventions | |||
| Location of cardiac arrest, n (%) | |||
| Home | 971 (52) | 410 (43) | 561 (63) |
| Public place | 653 (35) | 402 (42) | 251 (28) |
| Other | 224 (12) | 138 (15) | 86 (10) |
| Witnessed cardiac arrest, n (%) | 1689 (91) | 881 (93) | 808 (90) |
| CPR performed bystander, n (%) | 1480 (80) | 806 (85) | 674 (75) |
| Type of rhythm, n (%) | |||
| Not shockable | 486 (26) | 105 (11) | 381 (42) |
| Shockable | 1362 (74) | 845 (89) | 517 (58) |
| Time to return of spontaneous circulation (ROSC), min, median (IQR) | 25(17; 40) | 20 (14; 30) | 31 (21; 46) |
| TTM2: randomization treatment, n (%) | |||
| Normothermia | 923 (50) | 485 (51) | 438 (49) |
| Hypothermia | 925 (50) | 465 (49) | 460 (51) |
| Shock diagnosis at hospital admission, n (%) | 529 (29) | 193 (20) | 336 (37) |
| STEMI diagnosis at hospital admission, n (%) | 742 (40) | 429 (45) | 313 (35) |
| Ventilatory parameters at admission | |||
| Positive end expiratory pressure, cmH2O, median (IQR) | 6.90 (2.51) | 6.64 (2.31) | 7.18 (2.67) |
| Respiratory rate, breaths/min, median (IQR) | 17(14; 20) | 16 (14;19) | 18(15;20) |
| Plateau pressure, cmH2O, median (IQR) | 20 (17; 24) | 20(16; 23) | 21(17; 25) |
| Tidal volume, mL, median (IQR) | 499 (441; 555) | 500 (450; 570) | 485 (429; 545) |
| Tidal volume, mL/kg per PBW, median (IQR) | 7.1 (6.3; 8.2) | 7.1 (6.3; 8.1) | 7.2 (6.4; 8.3) |
| Driving pressure, cmH20, median (IQR) | 13 (10; 16) | 13(10; 16) | 14(10; 17) |
| (4*Driving Pressure) + respiratory rate, median (IQR) | 69(54;84) | 68(54;83) | 74(55;88) |
| Compliance of respiratory system, mL/cmH20, median (IQR) | 40 (31; 50) | 42 (33; 51) | 37 (28; 48) |
| Mechanical power, J/min, median (IQR) | 16.2 (12.5; 21.6) | 15.5 (12.6; 20.5) | 17.4 (12.5; 22.9) |
| FiO2, %, median (IQR) | 60 (50; 90) | 60 (44; 80) | 60 (50; 98) |
| PaO2/FiO2 ratio, mmHg, median (IQR) | 173 (110; 256) | 192 (127; 282) | 151 (94; 230) |
Data are expressed as mean and standard deviation (SD) or median and interquartile range (IQR), numbers (n) and percentages (%) when not otherwise specified
BMI body mass index; CPR cardiopulmonary resuscitation; ROSC return of spontaneous circulation; TTM2 Target Temperature Management; STEMI ST-elevation myocardial infarction
Ventilator settings and parameters
Ventilatory settings/parameters and arterial blood gases values at admission are described in ESM Table S1-2, and median values during the mechanical ventilation period in ESM Table S3. During the whole mechanical ventilation period the median tidal volume (VT) was 7 (IQR = 6.2–8.5) mL per Predicted Body Weight (PBW), PEEP was 7 (IQR = 5–9) cmH20, plateau pressure was 20 cmH20 (IQR = 17–23), driving pressure was 12 cmH20 (IQR = 10–15), compliance was 41 mL/cmH20 (IQR = 32.5–50.9), MP was 16.2 J/min (IQR = 12.1–21.8), VR was 1.27 (IQR = 1.04–1.6), and RR was 17 breaths/min (IQR = 14–20). Median PaO2 was 87 mmHg (IQR = 75–105), and PaCO2 was 40.5 mmHg (IQR = 36–45.7). Additional data on ventilator settings and arterial blood gases values in the whole population and according to survival status and to neurological outcome (good vs poor neurological outcome) are presented in ESM Table S1–5.
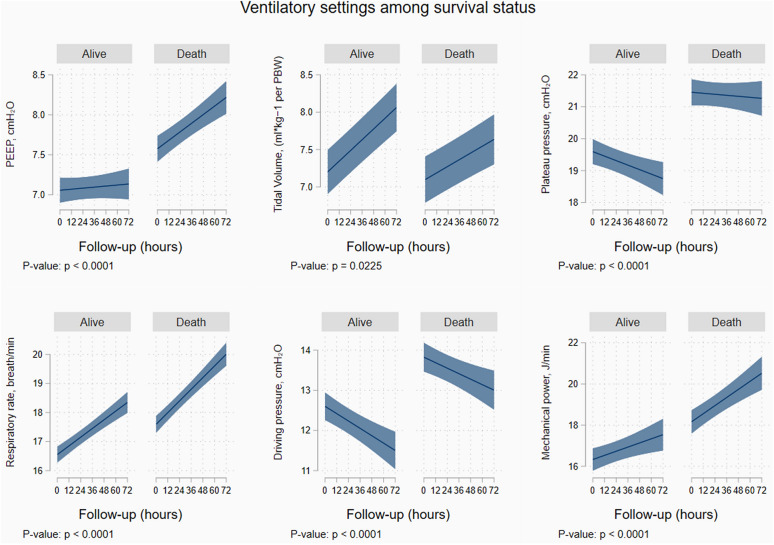
Figure 1 and ESM Figure S3 show the longitudinal trajectories of the different parameters within the first 72 h in survivors and non-survivors.
Fig. 1.
Hourly trajectories of different ventilator settings/parameters according to survival status. Predicted values from a mixed regression analysis with random intercept. PEEP positive end-expiratory pressure; PBW predicted body weight; FiO2 fraction of inspired oxygen
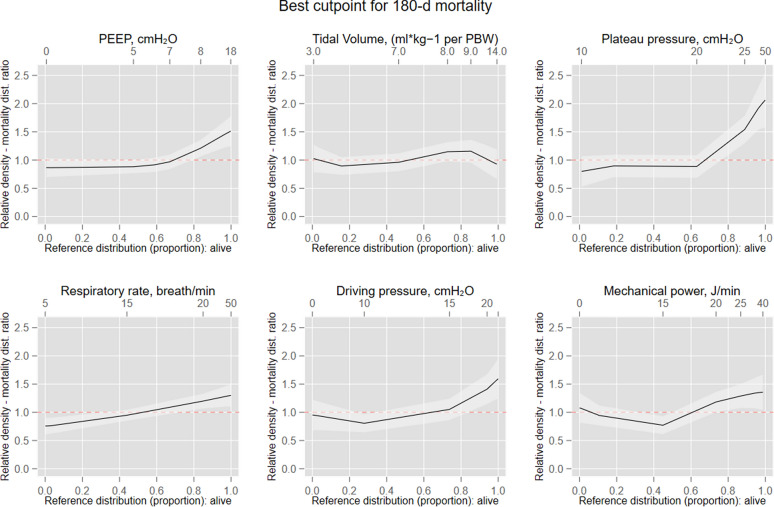
All the ventilator strategies trajectories differed significantly over the 72 h studied according to survival status. Figure 2 and ESM Figure S4 present the relative distribution analysis assessing the best cut off point for mortality for each variable. Using PEEP as example, having a PEEP of 8 cmH20 (corresponding to a quantile 0.85 of the PEEP distribution in those alive) had a corresponding ratio of 1.3 (on the y-axis), thus suggesting that PEEP of 8 included 1.3 times the proportion of patients who died as compared with those who remained alive.
Fig. 2.
Relative distribution analysis for the definition of the best cut-off associated with mortality for each parameter. Best cut-off point along the continuum of the marker that separated survivors versus non survivors at the end of the follow-up. In this analysis, the quantile (or proportion) distribution of the marker survivors (plotted on the x-axis plus the corresponding marker values at the top) is plotted against the proportion ratio of the marker distribution for non survivors. PEEP positive end-expiratory pressure; PBW predicted body weight; FiO2
Association of ventilator parameters with 6 months mortality and neurological outcome
Among respiratory parameters, RR, driving pressure, MP, and VR were independently associated with mortality (omnibus p-values for the non-linear trajectories: p < 0.0001, p = 0.026, p = 0.029, and p = 0.0003, respectively) (Fig. 3a, b, ESM Table S6). Stratified analysis assessing the effect on mortality for driving pressure, MP, and RR according to elastance values are presented in ESM Figure S5. The Omnibus p-value for the three ventilator settings showed no difference according to elastance categories (p = 0.1703, p = 0.3508, p = 0.1887, respectively). Driving pressure and RR were also associated with poor neurological outcome (OR = 0.001, 95% CI = 0.001–0.036, p = 0.048 and OR = 1.035, 95% CI = 1.003–1.068, p = 0.030, respectively), Table 2, ESM Table S7-8. Considering the formula [(4*Driving Pressure) + RR] from Costa et al. [7] we found a significant association with mortality (HR = 1.152, 95% CI = 1.040–1.276, p = 0.006) and poor neurological outcome (OR = 1.244, 95% CI = 1.015–1.525, p = 0.036) with a better performance compared to MP (ESM, Fig. S6, Table 2). Considering ventilator targets, PaCO2 values were not associated with 6 months mortality (HR = 1.089, 95% CI = 0.993–1.195, p = 0.069); or poor neurological outcome (OR = 1.018, 95% CI = 0.910–1.140, p = 0.751) (ESM Table S6; Table 2). PaO2 was independently associated with mortality (HR = 1.105, 95% CI = 1.014–1.205, p = 0.024), but not with neurological outcome (OR = 1.009, 95% CI = 0.993–1.024, p = 0.273) Additional results on arterial blood gases values are presented in ESM Table S7, 8. ESM Table S9 shows the Cox regression estimates of the basic ventilatory markers with stratification according to study center, and eliminating centers including less than ten patients. These results prove that our analysis is robust enough by no including the center effect in the model.
Fig. 3.
a, b Ventilatory markers and 6-month mortality. This regression model was adjusted by (1) clinical variables: TTM2 randomization group, age (years), Charlson comorbidity index, cardiac arrest witnessed, ROSC (min), bystander performed CPR, shockable rhythm, cardiac arrest location (home, public place, other), shock diagnosis on admission, and STEMI diagnosis on admission; (2) arterial blood gas values: arterial partial pressure of oxygen (PaO2) (mmHg)/Fraction of inspired oxygen (FiO2) ratio, arterial partial pressure of carbon dioxide (PaCO2) (mmHg), pH, and Base excess (mEq/L); and (3) by the above markers among them. PEEP, positive end-expiratory pressure.
Table 2.
Regression estimates from the multivariable models for poor neurological outcome
| Ventilator settings | OR | 95% Confidence Interval | p value |
|---|---|---|---|
| Model for basic ventilator markers | |||
| Respiratory rate, per 10 breaths/min | 1.035 | (1.003–1.068) | 0.030 |
| Plateau pressure, cmH2O | 1.016 | (0.964–1.068) | 0.251 |
| Tidal volume, ml*kg−1 per PBW | 0.971 | (0.898–1.051) | 0.473 |
| PEEP, cmH2O | 1.023 | (0.968–1.081) | 0.420 |
| PaCO2, mmHg | 1,018 | (0.910–1.140) | 0.751 |
| PaO2, mmHg | 1.009 | (0.993–1.024) | 0.273 |
| Models for composite ventilator markers | |||
| [4*Driving pressure] + RR | 1.244 | (1.015–1.525) | 0.036 |
| Driving pressure FP2[-1], cmH2O | 1.005 | (1.001–1.036) | 0.048 |
| Mechanical power, J/min | 1.012 | (0.990–1.034) | 0.297 |
| Compliance of respiratory system, mL/cmH2O | 0.984 | (0.981–1.007) | 0.597 |
| Ventilatory ratio | 0.867 | (0.640–1.174) | 0.356 |
All regression models were adjusted by 1) Clinical variables: TTM2 randomization group, age (years), Charlson comorbidity index, cardiac arrest witnessed, return to spontaneous circulation, ROSC (min), bystander performed cardiopulmonary resuscitation, CPR, shockable rhythm, cardiac arrest location (home, public place, other), shock diagnosis on admission, STEMI diagnosis on admission, and arterial blood gases, ABG: PaO2 (mmHg)/FiO2, PaCO2 (mmHg), pH, and Base excess (mEq/L)
OR odds ratio; PEEP positive end-expiratory pressure; TTM2 Target Temperature Management; ROSC return of spontaneous circulation; CRP cardiopulmonary resuscitation; STEMI ST elevation myocardial infarction; ABG arterial blood gases; PaO2 arterial partial pressure of oxygen; FiO2 fraction of inspired oxygen; PaCO2 arterial partial pressure of carbon dioxide; RR respiratory rate
Discussion
In this pre-planned sub-study of the TTM2-trial, including 1848 patients after out of hospital cardiac arrest, we describe ventilation practice and the association of different mechanical ventilation settings with 6 months mortality and functional neurological outcome. Our results can be summarized as follows: (1) protective ventilation strategies are commonly used in this population during the first 72 h; (2) respiratory rate, driving pressure, mechanical power and ventilatory ratio were independently associated with 6-month mortality; (3) respiratory rate and driving pressure were also associated with 6-month functional outcome; (4) the formula [(4*Driving Pressure) + RR] [7] demonstrated to be significantly associated with mortality and poor neurological outcome.
To our knowledge, this is the largest study describing ventilatory settings applied in out of hospital cardiac arrest survivors and their association with 6-month mortality risk as well as functional outcome. In particular, this is the largest investigation to date on the potential effects on mortality and neurological outcome of ventilation settings, in particular driving pressure and respiratory rate, in a non-ARDS population.
The characterization of ventilator settings is fundamental in this group of patients, as mortality and poor outcome are still very high despite several medical interventions have been applied and implemented [2, 17–24]. The recent guidelines of the European Resuscitation Council and European Society of Intensive Care Medicine on post-resuscitation care do not provide specific recommendations on the optimal ventilator settings to be applied after cardiac arrest. It is just suggested to aim at a tidal volume of 6–8 mL/kg ideal body weight [25]. This is consequent to the availability of a limited number of studies for this specific population, and especially on the lack of data in literature of more advanced and specific parameters (such as driving pressure and mechanical power) [14], which have shown in other groups of patients to have a potential effect on outcome, but not in cardiac arrest [26–32]. Although recent literature has highlighted the importance of protective ventilation in ARDS and non-ARDS patients [33–35], some strategies may be potentially detrimental in patients after cardiac arrest; high PEEP might further aggravate cerebral edema by increasing intrathoracic pressure while reducing jugular outflow, and low tidal volume and consequent permissive hypercapnia can cause cerebral vasodilation [36, 37]. The appropriate target of PaCO2 needs to be better determined in this population [21, 38]; early cerebral hypoperfusion and impaired cerebrovascular autoregulation may make normal PaCO2 insufficient to achieve adequate cerebral perfusion and, consequently, cerebral oxygenation, and mild hypercapnia has been suggested by some authors to optimize cerebral blood flow [39]. The ongoing TAME study (ClinicalTrials.gov: NCT03114033) is evaluating the effect of mild hypercapnia on patients’ outcome in this population.
Our results suggest that in a homogeneous population of patients after cardiac arrest, lung protective standards are often applied, similarly to the results of the PRoVENT study [4], an observational study focusing on mechanical ventilation practices in a heterogeneous population of patients without ARDS, but not specifically including cardiac arrest patients.
Evidence have progressively demonstrated that low VT is associate with favorable outcome after cardiac arrest [37], and very low PEEP or zero PEEP (ZEEP) can aggravate the risk of atelectasis and lung damage [40, 41]. As consequence of this, over time, physicians are increasingly applying lower tidal volume and higher PEEP even in cardiac arrest patients [5, 42], and as well as in brain injured patients at risk of intracranial hypertension [43]. In a secondary analysis from a multicenter study in ICU patients receiving mechanical ventilation [44], Sutherasan et al. showed that in 1998 the mean tidal volume used in cardiac arrest patients was 8.9 mL/Kg, and mean PEEP was 3.5 cmH20 [5]. A sub-analysis of the TTM trial published in 2018 [6] demonstrated a median tidal volume = 7.7 mL/Kg PBW, and PEEP 6 cmH20, and driving pressure of 14.7 cmH20. In our cohort, we found even higher median values of PEEP, and lower median values of tidal volume and driving pressure, thus suggesting an increasing application of protective strategies in this population over years.
We found that mechanical power, driving pressure, and ventilatory ratio but not PEEP, plateau pressure of the respiratory system or tidal volume alone were independently associated with 6 months mortality. This suggests the importance of the titration of different settings taking in account intrathoracic pressure and ventilation to avoid ventilator lung injury rather than the application of only one single protective mechanical ventilation strategy. In particular, the fact that tidal volume “per se” is not associated with outcome, further supports the hypothesis that the most relevant parameter to be controlled is the tidal volume standardized for the expected lung volume (i.e., estimated by respiratory compliance), which in practical terms is identified by the driving pressure (i.e., a bedside parameter measuring the “dynamic” strain equal to tidal volume divided the amount of aerated lung). Driving pressure, which is a function of plateau pressure and PEEP, represents the real stress applied to the respiratory system (of the lung and chest wall combined) from end-inspiration to end-expiration [45]. Similarly, mechanical power is the mechanical energy which is transferred to the respiratory system in every respiratory cycle, multiplied with each respiratory rate, and it is therefore considered as a determinant of ventilator-induced lung injury (VILI) [13, 46]. A recent experimental study showed that pulmonary neutrophilic inflammation importantly correlates with mechanical power [47]; similarly, mechanical power has demonstrated to be related to radiological signs of lung edema, and histological features of lung injury [46]. In the clinical settings, mechanical power was found to be associated with mortality in retrospective studies on critically ill patients [7, 26]. For the first time, our results demonstrated in a prospectively enrolled population of critically ill patients after cardiac arrest, that mechanical power is independently associated with 6 months mortality, with a threshold similar to other critically ill populations (Fig. 2) [7, 26]. We also found that the only basic ventilator setting associated with mortality is the respiratory rate, thus suggesting that this could be the major determinant of lung injury. This latter point is of extreme importance in cardiac arrest patients, as often high respiratory rates are used to precisely titrate PaCO2 to modulate cerebral blood flow and vascular tone and avoid hypercapnia, cerebrovascular vasodilation and increase cerebral edema and may have a major role in non-ARDS patients as outcome determinant [6, 48, 49]. Consistently, this might also explain the relationship between ventilatory ratio, which depends on minute ventilation, and mortality. Indeed, ventilatory ratio is a relatively new bedside index able to detect impaired ventilation in ARDS and correlates well with pulmonary dead space fraction [14, 50]. However, stratified analysis assessing the effect on mortality for driving pressure, mechanical power, and respiratory rate according to classes of elastance, showed no difference. This is in contrast with a previous study from Goligher et al. [51], which showed that the mortality benefit in ARDS is greater in patients with high elastance and comparatively lower in patients with low elastance. However, it is important to highlight this study included only ARDS population, with importantly impaired respiratory mechanics, whereas in our study we included a homogeneous population of cardiac arrest patients with relatively healthy lungs, and therefore the effect of elastance may be less clear. Interestingly, we demonstrated that using the formula [(4*Driving Pressure) + RR] [7] previously applied only in patients with ARDS, the combination of driving pressure and respiratory rate has significant association with mortality and poor neurological outcome, and can be even more informative than mechanical power. This is the first study where this formula was applied in non-ARDS patients, and we believe that this is an unique result for clinicians, as this may guide ventilatory settings application: in particular, as this formula represents lung stress/strain and stress rate, lowering VT is beneficial only whether this yields a reduction in driving pressure of 1 cmH2O with increases in respiratory rate not greater than 4 breaths/minute. Therefore, in the attempt to optimize ventilatory settings, first we should minimize the driving pressure, and secondly decrease the respiratory rate. This is of fundamental importance especially in the aim of optimize PaCO2 and pH, through respiratory rate manipulations. Finally, we found an association between respiratory rate, driving pressure and neurological outcome. This suggests that these two parameters are fundamental not only to minimize patients’ mortality but also to reduce secondary brain damage, by modulating carbon dioxide values and ventilator-induced lung injury. However, as shown in our analysis, the effect observed on mRS is mainly related to the inclusion of mortality in this scale (mRS = 6), and this result should be taken with caution.
Limitations
This study presents several limitations. Firstly, this is an observational study, which therefore precludes to draw any causality conclusions from our results. Indeed, observational data do not fully describe if the underlying severity of patients explains the ventilator settings observed in patients with higher mortality, and results should be taken with caution. A randomized controlled trial would be needed to jump to strong conclusions. However, we used statistically robust models, which can provide information about the association between ventilator parameters and outcome and pave the way for the development of prospective randomized controlled trials to confirm these findings. Secondly, although this was a preplanned study, some information is lacking in eCRF (such as the Pittsburgh cardiac arrest score) and there are some missing data in the variables. Third, as per study protocol, paralysis was not routinely applied; this may have altered spontaneous breathing efforts respiratory rate and other ventilation data.
Conclusions
Cardiac arrest patients often and increasingly receive protective ventilation. Optimization of ventilator settings and limiting exposure to modifiable factors of mechanical ventilation and in particular to high respiratory rate, and driving pressure may improve patient’s outcome after cardiac arrest.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The authors would like to thank Eduardo Nunez for the support with the statistical analysis. The members of the TTM2 Trial Collaborators are listed here: Steering Group: Niklas Nielsen, Lund University, Helsingborg Hospital, Department of Clinical Sciences Lund, Anesthesiology and Intensive care, Lund, Sweden (Chair and Chief Investigator); Jan Bělohlávek, 2nd Department of Medicine, First Faculty of Medicine, Charles University in Prague and General University Hospital, Prague, Czech Republic (NI); Clifton Callaway, Department of Emergency Medicine, University of Pittsburgh, Pittsburgh, PA, USA (NI); Alain Cariou, Descaretes University of Paris and Cochin University Hospital, Paris, France (NI); Tobias Cronberg, Lund University, Skåne University Hospital Lund, Department of Clinical Sciences, Neurology, Lund, Sweden (Senior Investigator); Josef Dankiewicz, Lund University, Skåne University Hospital Lund, Department of Clinical Sciences, Cardiology, Lund, Sweden (Coordinating Investigator); Glenn Eastwood, The Australian and New Zealand Intensive Care Research Centre, Monash University, Melbourne, Australia; David Erlinge, Lund University, Skåne University Hospital Lund, Department of Clinical Sciences, Cardiology, Lund, Sweden; Hans Friberg, Lund University, Skåne University Hospital Malmö, Department of Clinical Sciences, Anesthesia & Intensive care, Lund, Sweden (Senior Investigator); Jan Hovdenes, Department of Anesthesiology and Intensive Care, Oslo University Hospital, Rikshospitalet, Oslo, Norway (NI); Janus Christian Jakobsen, Copenhagen Trial Unit, Capital Region, Copenhagen, Denmark; Department of Regional Health Research, The Faculty of Health sciences, University of Southern Denmark, Denmark (Trialist); Michael Joannidis, Division of Intensive and Emergency Medicine, Department of Internal Medicine, Medical University Innsbruck, Innsbruck, Austria (NI); Hans Kirkegaard, Research Center for Emergency Medicine, Department of Clinical Medicine, Aarhus University Hospital and Aarhus University, Aarhus N, Denmark (NI); Helena Levin, Lund University, Skåne University Hospital Lund, Department of Clinical Sciences, Anesthesiology and Intensive care, Lund, Sweden (Clinical Trial Manager); Gisela Lilja, Lund University, Skåne University Hospital Lund, Department of Clinical Sciences, Neurology, Lund, Sweden (Follow-up Coordinator); Matt P. G. Morgan, Adult Critical Care, University Hospital of Wales, Cardiff, United Kingdom; Alistair D. Nichol, University College Dublin- Clinical Research Centre at St Vincent's University Hospital, Dublin, Ireland. Per Nordberg, Department of Medicine, Center for Resuscitation Science, Karolinska Institute, Solna, Sweden; Mauro Oddo, Neuroscience Critical Care Group, Adult Intensive Care Medicine Service, CHUV-Lausanne University Hospital, University of Lausanne, Lausanne, Switzerland (NI); Paolo Pelosi, Anesthesiology and Critical Care, San Martino Policlinico Hospital, IRCCS for Oncology and Neurosciences, Department of Surgical Sciences and Integrated Diagnostics, University of Genoa, Genoa, Italy (NI); Christian Rylander, Department of Anesthesiology and Intensive Care Medicine, Institute of Clinical Sciences, Sahlgrenska Academy, University of Gothenburg, Gothenburg, Sweden (NI); Manoj Saxena, Division of Critical Care and Trauma, George Institute for Global Health. Bankstown-Lidcombe Hospital, South Western Sydney Local Health District, Sydney, Australia (NI); Christian Storm, Department of Nephrology and Medical Intensive Care, Charité—Universitätsmedizin Berlin, Germany (NI); Fabio S. Taccone, Department of Intensive Care, Erasme University Hospital, Université Libre de Bruxelles (ULB), Brussels, Belgium (NI); Susann Ullén, Clinical Studies Sweden – Forum South, Skåne University Hospital, Lund, Sweden (Chief Statistician); Matt P. Wise, Adult Critical Care, University Hospital of Wales, Cardiff, United Kingdom (NI); Paul J. Young, Medical Research Institute of New Zealand, Intensive Care Unit, Wellington Hospital, Wellington, New Zealand (NI). NI-National Coordinating Investigator. Independent Data Monitoring and Safety Committee: Kathy Rowan, Intensive Care National Audit & Research Centre, UK (Chair); David Harrison, Intensive Care National Audit & Research Centre, UK; Paul Mouncey, Intensive Care National Audit & Research Centre, UK; Manu Shankar-Hari, Guy’s and St Thomas’s NHS Foundation Trust, London, UK; Duncan Young, Nuffield Department of Clinical Neurosciences, University of Oxford, UK. Statisticians: Susann Ullén, Clinical Studies Sweden – Forum South, Skåne University Hospital, Lund, Sweden (Chief Statistician); Theis Lange, Department of Biostatistics, University of Copenhagen, Copenhagen, Denmark (Independent Statistician); Karolina Palmér, Department of Medical Statistics and Epidemiology, Region Skåne, Malmö, Sweden Independent statistician). Coordinating Organizations and Trial Management: Region Skåne, Helsingborg Hospital, Helsingborg, Sweden (Sponsor). Lund University, Lund, Sweden. Core management group: Niklas Nielsen (Chair and Chief Investigator), Josef Dankiewicz (Coordinating Investigator), Tobias Cronberg (Senior Investigator, Neurology), Hans Friberg (Senior Investigator, Intensive Care), Gisela Lilja (Follow-up Coordinator), Helena Levin. (Clinical Trial Manager), Janus Christian Jakobsen (Trialist), Susann Ullén (Chief Statistician). Trial financial management: Helsingborg Hospital: Ulla-Britt Karlsson; Lund University: Simon Heissler. Australia: The George Institute for Global Health, Sydney (Local Sponsor): Manoj Saxena, Frances Bass, Naomi Hammond, John Myburgh, Colman Taylor. France: Clinical Research Unit, Paris Descartes Necker Cochin, Paris (Local Representative): Alain Cariou, Adele Bellino. Trial Coordinators And Monitors: Australia: The George Institute for Global Health, Sydney: Marwa Abel-all, Ben Finfer, Carolyn Koch, Yang Li, Anne O’Connor, Julia Pilowsky, Tina Schneider, Anna Tippett; Monash University, Melbourne: Bridget Ady, Tessa Broadley, Amanda Brown, Liz Melgaard, Mimi Morgan, Vanessa Singh, Rebecca Symons. Austria: Medical UniversityInnsbruck, Innsbruck: Kathrin Becker. Belgium: NVS Consulting, Brussels: Nathalie Van Sante. Czech Republic: Aixial, Brno: Vendula Saleova, Silvie Zerzanova. Denmark: Lund University, Lund, Sweden: Helena Levin. France: Clinical Research Unit, Paris Descartes Necker Cochin, Paris: Samia Sefir-Kribel. Germany: Charité Universitätsmedizin, Berlin: Ute Lübeck. Italy: Mario Negri Institute for Pharmacological Research, Milan: Martina Carrara. New Zealand: Medical Research Institute of New Zealand (MRINZ), Wellington: Kathryn Fernando, Diane Mackle, Leanlove Navarra, Judith Riley. Norway: Oslo University Hospital, Oslo: Elin Westerheim; Haukeland University Hospital, Bergen: Marianne Flatebø. Sweden: Helsingborg Hospital, Helsingborg: Ameldina Ceric, Zana Haxhija, Lovisa Terling; Skåne University Hospital, Lund: Lena Bossmar, Liz Jergle, Helén Holm Månsson. Switzerland: Lausanne University Hospital (CHUV), Lausanne: Samia Abed Maillard, Andreja Vujicic Zagar; Cantonal Hospital St. Gallen, St. Gallen: Christina Jodlauk. United Kingdom: University Hospital of Wales, Cardiff: Helen Hill; Niche Science & Technology, Richmond: Jennifer Scrivens; The HRB Irish Critical Care- Clinical Trials Network (ICC-CTN), Dublin, Ireland: Kate Ainscough, Ciara Fahey. Sites, Principal Investigators, and Site Personnel: Australia: Austin Hospital, Melbourne: Rinaldo Bellomo (PI), Glenn Eastwood, Leah Peck, Helen Young; Concord Repatriation General Hospital, Sydney: Winston Cheung (PI), Rosalba Cross, Michael Hayes, Nitin Jain, Mark Kol, Asim Shah, Atul Wagh, Helen Wong; John Hunter Hospital, Newcastle: F. Eduardo Martinez (PI), Gail Brinkerhoff, Dustin Bush; Liverpool Hospital, Sydney: Antony Stewart (PI), Anders Aneman, Lien Lombardo, Peter McCanny, James Penketh; Nepean Hospital, Sydney: Ian Seppelt (PI), Rebecca Gresham, Julie Lowrey, Kristy Masters, Christina Whitehead; Princess Alexandra Hospital, Brisbane: James Walsham (PI), Meg Harward, Josephine Mackay, Jason Meyer, Emma Saylor, Ellen Venz, Krista Wetzig; Royal North Shore Hospital, Sydney: Wade Stedman (PI), Angela Ashelford, Frances Bass, Naomi Hammond, Sharon Mar, Julia Pilowsky, Miyuki Tokumitsu, Elizabeth Yarad; St Vincent's Hospital, Sydney: Hergen Buscher (PI), Claire Reynolds; The Alfred Hospital, Melbourne: Andrew Udy (PI), Aidan Burrell, Jasmin Collins, Dashiell Gantner, Victoria Emma-Leah Martin, Phoebe Mccracken, Vinodh Nanjayya, AlistairNichol, Alexander Sacha Richardson, Meredith Young; The Northern Hospital, Melbourne: Angaj Ghosh (PI), Simone Said. Austria: Medical University Innsbruck, Innsbruck: Michael Joannidis (PI), Ronny Beer, Frank Hartig, Raimund Helbok, Sebastian Klein, Andreas Peer. Belgium: Erasme University Hospital, Brussels: Fabio S. Taccone (PI), Jacques Creteur, Dominique Durand; Ziekenhuis Oost-Limburg, Genk: Matthias Dupont (PI), Sigrid Christiaens, Carola Claes, Sebastiaan Deckx, Bert Ferdinande, Sanne Lenaerts, Wilifred Mullens, Sarah Stroobants, Evi Theunissen, David Verhaert. Czech Republic: General University Hospital, Prague: Ondřej Šmíd (PI), Marek Flaksa, David Kemlink, Jan Malík, Michal Otáhal, Jan Rulíšek, Michal Šíranec, Zdeněk Stach, Anna Valeriánová, Petra Zavadilova; University Hospital Hradec Králové, Hradec Králové: Miroslav Solař (PI), Róber Bánszky, Jana Červená, Renata Černá Pařízková, Libor Šimůnek, Filip Varhaník; Regional Hospital Liberec, Liberec: Jiří Karásek (PI), Matěj Strýček. Denmark: Aarhus University Hospital, Aarhus: Anders Grejs (PI), Steffen Christensen, Peter Juhl-Olsen, Ida Katrine Thomsen, Lisa Gregersen Østergaard. France: Cochin University Hospital (APHP), Paris: Alain Cariou (PI), Albert Cao, Pierre Dupland, Ariane Gavaud, Paul Jaubert, Mathieu Jozwiak, Nathalie Marin, Guillaume Savary; Lariboisiere University Hospital (APHP), Paris: Nicolas Deye (PI), Bruno Megarbane, Pierre Mora, Laetitia Sutterlin; Centre Hospitalier de Versailles, Le Chesnay: Stephane Legriel (PI), Hugo Bellut, Alexis Ferre, Guillaume Lacave, Marine Paul; CHU de Nantes, Nantes: Jean-Baptiste Lascarrou (PI), Emmanuel Canet, Charlotte Garret, Arnaud Felix Miaihle, Jean Reignier; Dupuytren Teaching Hospital, Limoges: Philippe Vignon (PI), Thomas Daix, Arnaud Desachy, Bruno Evrard, Bruno Francois, Anne-Laure Fedou, Marine Goudelin. Germany: Charité Universitätsmedizin, Berlin: Christian Storm (PI), Gabriele Kress, Christoph Leithner, Jens Nee, Kaspar Josche Streitberger. Italy: San Martino Policlinico Hospital, Genoa: Iole Brunetti (PI), Lorenzo Ball, Denise Battaglini, Giulia Bonatti, Iacopo Firpo, Paolo Frisoni, Arianna Iachi, Simona Maiani, Maura Mandelli, Chiara Robba, Fabio Tarantino; Civil Hospital, Baggiovara, Modena: Alberto Barbieri (PI), Elisabetta Bertellini, Enrico Giuliani, Gabriele Melegari; University of Trieste, Trieste: Erik Roman-Pognuz (PI), Giorgio Berlot, Umberto Lucangelo, Elisabetta Macchini. Norway: Oslo University Hospital, Rikshospitalet, Oslo: Jan Hovdenes (PI), Vibeke Aune, Tomas Drægni, Simon Jacobsen, Søren Pieschke, Åse Rasmussen, Gro Ringstad Akselsen; St. Olav’s University Hospital, Trondheim: Halvor Langeland (PI), Daniel Bergum, Therese M. Erbe, Pål Klepstad, Helle M. Næss; Sorlandet Hospital, Arendal: Roy Bjørkholt Olsen (PI), Lena Eriksen Skjelnes, Marius Holen, Joakim Iver Post; Haukeland University Hospital, Bergen: Rune Fanebust (PI), Linda Hårteig Sørensen, Ken Åge Kårstad, Carsten Fredrik Wickman. New Zealand: Wellington Regional Hospital, Wellington: Paul Young (PI), Colin Barnes, Ben Barry, Nina Beehre, Dick Dinsdale, Sam Edney, Anna Hunt, Harriet Judd, Charlotte Latimer-Bell, Cassie Lawrence, James Moore, Shaanti Olatunji, Alex Psirides, Chelsea Robinson, Kate Tietjens, Jason Wright; Christchurch Hospital, Christchurch: David Knight (PI), Brandon. Birker, David Bowie, Tara Burke, David Closey, Rosalind Crombie, Neil Davidson, Seton Henderson, Louise Hitchings, James McKay, Jan Mehrtens, Emmeline Minto, Stacey Morgan, Anna Morris, Jay Ritzemar-Carter, Jessica Roberts, Geoffrey Shaw, Katherine Townend, Kymbalee Vander Heyden. Sweden: Sahlgrenska University Hospital, Gothenburg: Christian Rylander (PI), Marita Ahlqvist, Roman Desta Lindgren, Ingrid Eiving, Andreas Lundin, Patrik Martner, Elisabeth Myhrman, Birgitta Ryding; Skåne University Hospital, Malmö: Joachim Düring (PI), Mattias Bergström, Mattias Bohm, Ingrid Didriksson, Petrea Frid, Katarina Heimburg, Marina Larsson, Oscar Lundberg, Stefan Olsson Hau, Simon Schmidbauer; Skåne University Hospital, Lund: Ola Borgquist (PI), Anne Adolfsson, Anna Bjärnroos, Erik Blennow-Nordström, Irina Dragancea, Thomas Kander, Anna Lybeck, Gustav Mattiasson, Olof Persson, Malin Rundgren, Susann Schrey, Erik Westhall; Helsingborg Hospital, Helsingborg: Martin Annborn (PI), Sara Andertun, Florian Ebner, Nerida Gustavsson, Lisa Hassel, Jesper Johnsson, Marie Nelderup, Heléne Petersson, Jörgen Petersson, Frideriki Stafilidou; Hallands Hospital, Halmstad: Johan Undén (PI), Frida Antonsson, Git Bergman, Jörgen Gamroth, Maria Meirik, Katarina Rudolfsson, Helena Sandberg, Martin Thorsson; Karlstad Central Hospital, Karlstad: Kristin Savolainen (PI), Maria Hansbo, Malin Helliksson, Björne Nödtveidt, Johan Sanner, Victoria Sem, Camilla Sund Lindquist; Södersjukhuset, Karolinska Institute, Stockholm: Per Nordberg (PI), Akil Awad, Anna-Sofia Börjesson, Malin Hedberg, Mia Henning, Jacob Hollenberg; Northern Älvsborg County Hospital, Trollhättan: Per Petersen (PI), Emelia Dahlberg, Johan Forshammar, Veronica Svensson; Capio S:t Görans Hospital, Stockholm: Michael Wanecek (PI), Håkan Eskilsson; Skaraborg Hospital, Skövde: Daniel Rodriguez-Santos (PI), Åsa Appelqvist, Henrietta Jidbratt, Elisabeth Johansson, Lars Kiszakiewicz, Åsa Nilsson, Sinnika Olsson, Anders Paulsson, Urszula Stempel, Andreas Thoren; Örebro University Hospital, Örebro: Stefan Persson (PI), Ida Berglund, Eric Bergström, Cathrine Törnqvist, Ingela Östman; Uppsala University Hospital, Uppsala: Sten Rubertsson (PI), Ing-Marie Larsson, Elin Söderman, Ewa Wallin, Joanna Wessbergh; Linköping University Hospital, Linköping: Thomas Halliday (PI), Filippa Engvall. Switzerland: Lausanne University Hospital (CHUV), Lausanne: Mauro Oddo (PI), Nawfel Ben-Hamouda, Adriano Bernini, Pierre-Nicolas Carron, Philippe Eckert, Eva Favre, John-Paul Miroz, Paola Morelli, Olivier Muller, Jan Novi, Andrea Rosseti, Madeleine Schnorf; Bern University Hospital, Bern: Matthias Haenggi (PI), Anja Levis, Sandra Nansoz, Marianne Roth & Team, Nicole Söll; Cantonal Hospital St. Gallen, St.Gallen: Claudia Schrag (PI), Mensur Alicajic, Philipp Baier, Joel Dütschler, Dominique Flügel, Edith Fässler, Ruth Gamio-Veis, Marc Güpfert, Yvonne Hilpertshauser, Stefan Hägele-Link, Gian-Reto Kleger, Peter Krähenmann, Maria Elisabeth Mair, Nadja Schai, Christoph Strohmaier, Peter Tangl, Dominik Zieglgänsberger; University Hospital Zurich, Zurich: Marco Maggiorini (PI), Gabriele Claus, Gabi Consani-Vogel, Lukas Imbach, Samira Kaiser, Eva-Maria Kleinert, Pedro David Wendel Garcia; Cardiocentro Ticino, Lugano: Tiziano Cassina (PI), Pamela Agazzi, Bruno Capelli, Gabriele Casso, Martino Regazzi, Hervé Schlotterbeck, Gabriele Via, Michele Villa. United Kingdom: University Hospital of Wales, Cardiff: Matt P. Wise (PI), Jenny Brooks, Eve Cocks, Jade Cole, Jacqueline Curtin, Michelle Davies, Rhys Davies, Stephen Fernandez, Julie Highfield, Helen Hill, Matt P. G. Morgan, Lydia Pennant, Sofia Rose, Emma Thomas, Angharad Williams; Royal Victoria Hospital, Belfast: Peter McGuigan (PI), Stephen Haffey, Aisling O'Neill, Kathryn Ward; Bristol Royal Infirmary, Bristol: Matthew Thomas (PI), Jeremy Bewley, Anna Chillingworth, Julie Cloake, Libby Cole, Hilary Galvin, Zoe Garland, Lisa Grimmer, Bethany Gumbrill, Lucy Howie, Rebekah Johnson, Chloe Searles, Agnieszka Skorko, Katie Sweet, Victoria Taylor, Denise Webster; Essex Cardiothoracic Centre, Basildon: Thomas Keeble (PI), Gill Adams, Rajesh K Aggarwal, Jo-Anne Cartwright, Steven Church, Gerald J Clesham, John R Davies, Kelly Farrell, Reto Gamma, Jane Harding, Rohan Jagathesan, Alamgir Kabir, Paul A Kelly, Lauren Kittridge, Maria Maccaroni, Gracie Maloney, Marco Mion, Naveen Nain, Raghunath Nalgirkar, Gyanesh Namjoshi, Stacey Pepper, Emily Redman, Nicholas M Robinson, Jeremy Sayer, Amanda Solesbury, Kare H Tang, Sali Urovi, Kunal Waghmare, Noel Watson, Teresa Webber; University Hospitals Birmingham NHS Foundation Trust, Birmingham: Peter Isherwood (PI), Conor Bentley, Colin Bergin, Ronald Carrera, Amy Clark, Lauren Cooper, Liesl Despy, Natalie Dooley, Karen Ellis, Emma Fellows, Stephanie Goundry, Samantha Harkett, Christopher McGhee, Aoife Neal, Hazel Smith, Catherine Snelson, Elaine Spruce, Tony Whitehouse, Kamal Yakoub; Royal Berkshire Hospital, Reading: Andrew Walden (PI), Shauna Bartley, Parminder Bhuie, Matthew Frise, Nicola Jacques, Liza Keating; Queen Alexandra Hospital, Portsmouth: David Pogson (PI), Zoe Daly, Steve Rose; Manchester Royal Infirmary, Manchester: Jonathan Bannard-Smith (PI), Rachael Quayle; Royal Bournemouth Hospital, Bournemouth: Nigel Chee (PI), Nina Barratt, Katie Bowman, Debbie Branney, Elizabeth Howe, Maria Letts, Sally Pitts, Luke Vamplew. USA: University of Pittsburgh, Pittsburgh PA: Clifton W. Callaway (PI), Sara Difiore Sprouse, Ankur A. Doshi; Mayo Clinic, Rochester MN: Jennifer Fugate (PI), Amy M. Headlee, Eelco F.M.Wijdicks. PI – Principal Investigator. Related Organizations: Copenhagen Trial Unit, Copenhagen, Denmark; Clinical Studies Sweden – Forum South, Skåne University Hospital, Lund, Sweden; The George Institute for Global Health, Sydney, Australia; Australian and New Zealand Intensive Care Research Centre, Monash University, Melbourne, Australia; Medical Research Institute of New Zealand—(MRINZ), Wellington, New Zealand; Clinical Research Unit, Paris Descartes Necker Cochin, Paris, France; Scandinavian Critical Care Trials Group (SCCTG); The HRB Irish Critical Care- Clinical Trials Network (ICC-CTN); The ANZICS Clinical Trials Group (CTG); Spiral Software, Wellington, New Zealand; Allianz Global Corporate & Specialty SE, Copenhagen, Denmark; IBBL (Integrated BioBank of Luxembourg), Dudelange, Luxembourg.
Abbreviations
- ABG
Arterial blood gas
- ARDS
Acute respiratory distress syndrome
- AUC
Area under curve
- BMI
Body mass index
- CI
Confidence interval
- CO2
Carbon dioxide
- COPD
Chronic obstructive pulmonary disorder
- Crs
Respiratory system compliance
- DP
Driving pressure
- eCRF
Electronic case record form
- ESM
Electronic supplemental material
- GCS
Glasgow coma scale
- HR
Hazard ratio
- ICU
Intensive care unit
- MP
Mechanical power
- mRS
Modified Rankin Scale
- OHCA
Out of hospital cardiac arrest
- OR
Odds ratio
- PaCO2
Arterial partial pressure of CO2
- PaO2
Arterial partial pressure of oxygen
- PBW
Predicted body weight
- PEEP
Positive end-expiratory pressure
- pHa
Arterial pH
- PI
Principal investigator
- Ppeak
Peak pressure
- Pplat
Plateau pressure
- RCT
Randomized controlled trial
- ROSC
Return of spontaneous circulation
- RR
Respiratory rate
- STEMI
ST elevation myocardial infarction
- STROBE
Strengthening the Reporting of Observational Studies in Epidemiology
- TTM2
Target Temperature Management 2 Trial
- VR
Ventilator ratio
- VT
Tidal volume
- ZEEP
Zero PEEP
Author contribution
CR: conception of the work, participation in data analysis and interpretation, drafting the manuscript, critical revision of the manuscript, final approval of the version to be published. All the authors: conception of the work, critical revision of the manuscript, final approval of the version to be published. NN, PP: conception of the work, participation in data analysis and interpretation, critical revision of the manuscript, final approval of the version to be published. PP and NN equally contributed.
Funding
Open access funding provided by Università degli Studi di Genova within the CRUI-CARE Agreement. The TTM2 trial was supported by independent research grants from nonprofit or governmental agencies (the Swedish Research Council [Veten-skapsrådet], Swedish Heart–Lung Foundation, Stig and Ragna Gorthon Foundation, Knutsson Foundation, Laerdal Foundation, Hans-Gabriel and Alice Trolle-Wachtmeister Foundation for Medical Research, and Regional Research Support in Region Skåne) and by governmental funding of clinical research within the Swedish National Health Service. No further fundings were requested for this subanalysis.
Declarations
Conflicts of interest
MS, receiving consulting fees from Bard Medical; PJY, receiving lecture fees from Bard Medical; FST, receiving grant support from Bard Medical and ZOLL Medical; AN, receiving grant support, paid to University College Dublin, from AM Pharma and grant sup-port, paid to Monash University, from Baxter Healthcare; MSC, receiving lecture fees from Edwards Lifesciences; HF, receiving fees for academic advising from TEQCool; and NN, receiving lecture fees from Bard Medical and consulting fees from BrainCool. RB is supported by INCLIVA. No other potential conflict of interest relevant to this article was reported.
Ethical approval and informed consent
The Ethic Committees approved the TTM2 study in all participating centres and informed consent was obtained according to local regulations.
Footnotes
The members of the TTM2 Trial Collaborators are mentioned in “Acknowledgement” section.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Niklas Nielsen and Paolo Pelosi equally contributed as senior authors.
Contributor Information
Chiara Robba, Email: kiarobba@gmail.com.
TTM2 Trial Collaborators:
Niklas Nielsen, Jan Bělohlávek, Clifton Callaway, Alain Cariou, Tobias Cronberg, Josef Dankiewicz, Glenn Eastwood, David Erlinge, Hans Friberg, Jan Hovdenes, Janus Christian Jakobsen, Michael Joannidis, Hans Kirkegaard, Helena Levin, Gisela Lilja, Matt P. G. Morgan, Alistair D. Nichol, Per Nordberg, Mauro Oddo, Paolo Pelosi, Christian Rylander, Manoj Saxena, Christian Storm, Fabio S. Taccone, Susann Ullén, Matt P. Wise, Paul J. Young, Kathy Rowan, David Harrison, Paul Mouncey, Manu Shankar-Hari, Duncan Young, Susann Ullén, Theis Lange, Karolina Palmér, Ulla-Britt Karlsson, Simon Heissler, Frances Bass, Naomi Hammond, John Myburgh, Colman Taylor, Adele Bellino, Marwa Abel-all, Ben Finfer, Carolyn Koch, Yang Li, Anne O’Connor, Julia Pilowsky, Tina Schneider, Anna Tippett, Bridget Ady, Tessa Broadley, Amanda Brown, Liz Melgaard, Mimi Morgan, Vanessa Singh, Rebecca Symons, Kathrin Becker, Nathalie Van Sante, Vendula Saleova, Silvie Zerzanova, Helena Levin, Samia Sefir-Kribel, Ute Lübeck, Mario Negri, Martina Carrara, Kathryn Fernando, Diane Mackle, Leanlove Navarra, Judith Riley, Elin Westerheim, Marianne Flatebø, Ameldina Ceric, Zana Haxhija, Lovisa Terling, Lena Bossmar, Liz Jergle, Helén Holm Månsson, Samia Abed Maillard, Andreja Vujicic Zagar, Christina Jodlauk, Helen Hill, Jennifer Scrivens, Kate Ainscough, Ciara Fahey, Rinaldo Bellomo, Leah Peck, Helen Young, Winston Cheung, Rosalba Cross, Michael Hayes, Nitin Jain, Mark Kol, Asim Shah, Atul Wagh, Helen Wong, F. Eduardo Martinez, Gail Brinkerhoff, Dustin Bush, Antony Stewart, Anders Aneman, Lien Lombardo, Peter McCanny, James Penketh, Ian Seppelt, Rebecca Gresham, Julie Lowrey, Kristy Masters, Christina Whitehead, James Walsham, Meg Harward, Josephine Mackay, Jason Meyer, Emma Saylor, Ellen Venz, Krista Wetzig, Wade Stedman, Angela Ashelford, Sharon Mar, Julia Pilowsky, Miyuki Tokumitsu, Elizabeth Yarad, Hergen Buscher, Claire Reynolds, Andrew Udy, Aidan Burrell, Jasmin Collins, Dashiell Gantner, Victoria Emma-Leah Martin, Phoebe Mccracken, Vinodh Nanjayya, Alistair Nichol, Alexander Sacha Richardson, Meredith Young, Angaj Ghosh, Simone Said, Michael Joannidis, Ronny Beer, Frank Hartig, Raimund Helbok, Sebastian Klein, Andreas Peer, Fabio S. Taccone, Jacques Creteur, Dominique Durand, Matthias Dupont, Sigrid Christiaens, Carola Claes, Sebastiaan Deckx, Bert Ferdinande, Sanne Lenaerts, Wilifred Mullens, Sarah Stroobants, Evi Theunissen, David Verhaert, Ondřej Šmíd, Marek Flaksa, David Kemlink, Jan Malík, Michal Otáhal, Jan Rulíšek, Michal Šíranec, Zdeněk Stach, Anna Valeriánová, Petra Zavadilova, Miroslav Solař, Róber Bánszky, Jana Červená, Renata Černá Pařízková, Libor Šimůnek, Filip Varhaník, Jiří Karásek, Matěj Strýček, Anders Grejs, Steffen Christensen, Peter Juhl-Olsen, Ida Katrine Thomsen, Lisa Gregersen Østergaard, Alain Cariou, Albert Cao, Pierre Dupland, Ariane Gavaud, Paul Jaubert, Mathieu Jozwiak, Nathalie Marin, Guillaume Savary, Nicolas Deye, Bruno Megarbane, Pierre Mora, Laetitia Sutterlin, Stephane Legriel, Hugo Bellut, Alexis Ferre, Guillaume Lacave, Marine Paul Jean-Baptiste Lascarrou, Emmanuel Canet, Charlotte Garret, Arnaud Felix Miaihle, Jean Reignier, Philippe Vignon, Thomas Daix, Arnaud Desachy, Bruno Evrard, Bruno Francois, Anne-Laure Fedou, Marine Goudelin, Christian Storm, Gabriele Kress, Christoph Leithner, Jens Nee, Kaspar Josche Streitberger, Iole Brunetti, Lorenzo Ball, Denise Battaglini, Giulia Bonatti, Iacopo Firpo, Paolo Frisoni, Arianna Iachi, Simona Maiani, Maura Mandelli, Chiara Robba, Fabio Tarantino, Alberto Barbieri, Elisabetta Bertellini, Enrico Giuliani, Gabriele Melegari, Erik Roman-Pognuz, Giorgio Berlot, Umberto Lucangelo, Elisabetta Macchini, Jan Hovdenes, Vibeke Aune, Tomas Drægni, Simon Jacobsen, Søren Pieschke, Åse Rasmussen, Gro Ringstad Akselsen, Halvor Langeland, Daniel Bergum, Therese M. Erbe, Pål Klepstad, Helle M. Næss, Roy Bjørkholt Olsen, Lena Eriksen Skjelnes, Marius Holen, Joakim Iver Post, Rune Fanebust, Linda Hårteig Sørensen, Ken Åge Kårstad, Carsten Fredrik Wickman, Paul Young, Colin Barnes, Ben Barry, Nina Beehre, Dick Dinsdale, Sam Edney, Anna Hunt, Harriet Judd, Charlotte Latimer-Bell, Cassie Lawrence, James Moore, Shaanti Olatunji, Alex Psirides, Chelsea Robinson, Kate Tietjens, Jason Wright, David Knight, Brandon Birker, David Bowie, Tara Burke, David Closey, Rosalind Crombie, Neil Davidson, Seton Henderson, Louise Hitchings, James McKay, Jan Mehrtens, Emmeline Minto, Stacey Morgan, Anna Morris, Jay Ritzemar-Carter, Jessica Roberts, Geoffrey Shaw, Katherine Townend, Kymbalee Vander Heyden, Christian Rylander, Marita Ahlqvist, Roman Desta Lindgren, Ingrid Eiving, Andreas Lundin, Patrik Martner, Elisabeth Myhrman, Birgitta Ryding, Joachim Düring, Mattias Bergström, Mattias Bohm, Ingrid Didriksson, Petrea Frid, Katarina Heimburg, Marina Larsson, Oscar Lundberg, Stefan Olsson Hau, Simon Schmidbauer, Ola Borgquist, Anne Adolfsson, Anna Bjärnroos, Erik Blennow-Nordström, Irina Dragancea, Thomas Kander, Anna Lybeck, Gustav Mattiasson, Olof Persson, Malin Rundgren, Susann Schrey, Erik Westhall, Martin Annborn, Sara Andertun, Florian Ebner, Nerida Gustavsson, Lisa Hassel, Jesper Johnsson, Marie Nelderup, Heléne Petersson, Jörgen Petersson, Frideriki Stafilidou, Johan Undén, Frida Antonsson, Git Bergman, Jörgen Gamroth, Maria Meirik, Katarina Rudolfsson, Helena Sandberg, Martin Thorsson, Kristin Savolainen, Maria Hansbo, Malin Helliksson, Björne Nödtveidt, Johan Sanner, Victoria Sem, Camilla Sund Lindquist, Akil Awad, Anna-Sofia Börjesson, Malin Hedberg, Mia Henning, Jacob Hollenberg, Per Petersen, Emelia Dahlberg, Johan Forshammar, Veronica Svensson, Michael Wanecek, Håkan Eskilsson, Daniel Rodriguez-Santos, Åsa Appelqvist, Henrietta Jidbratt, Elisabeth Johansson, Lars Kiszakiewicz, Åsa Nilsson, Sinnika Olsson, Anders Paulsson, Urszula Stempel, Andreas Thoren, Stefan Persson, Ida Berglund, Eric Bergström, Cathrine Törnqvist, Ingela Östman, Sten Rubertsson, Ing-Marie Larsson, Elin Söderman, Ewa Wallin, Joanna Wessbergh, Thomas Halliday, Filippa Engvall, Mauro Oddo, Nawfel Ben-Hamouda, Adriano Bernini, Pierre-Nicolas Carron, Philippe Eckert, Eva Favre, John-Paul Miroz, Paola Morelli, Olivier Muller, Jan Novi, Andrea Rosseti, Madeleine Schnorf, Matthias Haenggi, Anja Levis, Sandra Nansoz, Nicole Söll, Claudia Schrag, Mensur Alicajic, Philipp Baier, Joel Dütschler, Dominique Flügel, Edith Fässler, Ruth Gamio-Veis, Marc Güpfert, Yvonne Hilpertshauser, Stefan Hägele-Link, Gian-Reto Kleger, Peter Krähenmann, Maria Elisabeth Mair, Nadja Schai, Christoph Strohmaier, Peter Tangl, Dominik Zieglgänsberger, Marco Maggiorini, Gabriele Claus, Gabi Consani-Vogel, Lukas Imbach, Samira Kaiser, Eva-Maria Kleinert, Pedro David Wendel Garcia, Tiziano Cassina, Pamela Agazzi, Bruno Capelli, Gabriele Casso, Martino Regazzi, Hervé Schlotterbeck, Gabriele Via, Michele Villa, Matt P. Wise, Jenny Brooks, Eve Cocks, Jade Cole, Jacqueline Curtin, Michelle Davies, Rhys Davies, Stephen Fernandez, Julie Highfield, Helen Hill, Matt P. G. Morgan, Lydia Pennant, Sofia Rose, Emma Thomas, Angharad Williams, Peter McGuigan, Stephen Haffey, Aisling O’Neill, Kathryn Ward, Matthew Thomas, Jeremy Bewley, Anna Chillingworth, Julie Cloake, Libby Cole, Hilary Galvin, Zoe Garland, Lisa Grimmer, Bethany Gumbrill, Lucy Howie, Rebekah Johnson, Chloe Searles, Agnieszka Skorko, Katie Sweet, Victoria Taylor, Denise Webster, Thomas Keeble, Gill Adams, Rajesh K. Aggarwal, Jo-Anne Cartwright, Steven Church, Gerald J. Clesham, John R. Davies, Kelly Farrell, Reto Gamma, Jane Harding, Rohan Jagathesan, Alamgir Kabir, Paul A. Kelly, Lauren Kittridge, Maria Maccaroni, Gracie Maloney, Marco Mion, Naveen Nain, Raghunath Nalgirkar, Gyanesh Namjoshi, Stacey Pepper, Emily Redman, Nicholas M. Robinson, Jeremy Sayer, Amanda Solesbury, Kare H. Tang, Sali Urovi, Kunal Waghmare, Noel Watson, Teresa Webber, Peter Isherwood, Conor Bentley, Colin Bergin, Ronald Carrera, Amy Clark, Lauren Cooper, Liesl Despy, Natalie Dooley, Karen Ellis, Emma Fellows, Stephanie Goundry, Samantha Harkett, Christopher McGhee, Aoife Neal, Hazel Smith, Catherine Snelson, Elaine Spruce, Tony Whitehouse, Kamal Yakoub, Andrew Walden, Shauna Bartley, Parminder Bhuie, Matthew Frise, Nicola Jacques, Liza Keating, David Pogson, Zoe Daly, Steve Rose, Jonathan Bannard-Smith, Rachael Quayle, Nigel Chee, Nina Barratt, Katie Bowman, Debbie Branney, Elizabeth Howe, Maria Letts, Sally Pitts, Luke Vamplew, Clifton W. Callaway, Sara Difiore Sprouse, Ankur A. Doshi, Jennifer Fugate, Amy M. Headlee, and Eelco F. M. Wijdicks
References
- 1.Kim Y-M, Yim H-W, Jeong S-H, Klem ML, Callaway CW. Does therapeutic hypothermia benefit adult cardiac arrest patients presenting with non-shockable initial rhythms? A systematic review and meta-analysis of randomized and non-randomized studies. Resuscitation. 2012;83:188–196. doi: 10.1016/j.resuscitation.2011.07.031. [DOI] [PubMed] [Google Scholar]
- 2.Robba C, Siwicka-Gieroba D, Sikter A, Battaglini D, Dąbrowski W, Schultz MJ, et al. Pathophysiology and clinical consequences of arterial blood gases and pH after cardiac arrest. Intensive Care Med Exp. 2020;8:19. doi: 10.1186/s40635-020-00307-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bellani G, Laffey JG, Pham T, Fan E, Brochard L, Esteban A, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA. 2016;315:788. doi: 10.1001/jama.2016.0291. [DOI] [PubMed] [Google Scholar]
- 4.Neto AS, Barbas CSV, Simonis FD, Artigas-Raventós A, Canet J, Determann RM, et al. Epidemiological characteristics, practice of ventilation, and clinical outcome in patients at risk of acute respiratory distress syndrome in intensive care units from 16 countries (PRoVENT): an international, multicentre, prospective study. Lancet Respir Med. 2016;4:882–893. doi: 10.1016/S2213-2600(16)30305-8. [DOI] [PubMed] [Google Scholar]
- 5.Sutherasan Y, Peñuelas O, Muriel A, Vargas M, Frutos-Vivar F, Brunetti I, et al. Management and outcome of mechanically ventilated patients after cardiac arrest. Crit Care. 2015;19:215. doi: 10.1186/s13054-015-0922-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Harmon MBA, van Meenen DMP, van der Veen ALIP, Binnekade JM, Dankiewicz J, Ebner F, et al. Practice of mechanical ventilation in cardiac arrest patients and effects of targeted temperature management: a substudy of the targeted temperature management trial. Resuscitation. 2018;129:29–36. doi: 10.1016/j.resuscitation.2018.04.040. [DOI] [PubMed] [Google Scholar]
- 7.EL Costa V, Slutsky AS, Brochard LJ, Brower R, Serpa-Neto A, Cavalcanti AB, et al. Ventilatory variables and mechanical power in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 2021;204:303–311. doi: 10.1164/rccm.202009-3467OC. [DOI] [PubMed] [Google Scholar]
- 8.Robba C, Nielsen N, Dankiewicz J, Badenes R, Battaglini D, Ball L, et al. Ventilation management and outcomes in out-of-hospital cardiac arrest: a protocol for a preplanned secondary analysis of the TTM2 trial. BMJ Open. 2022;12:e058001. doi: 10.1136/bmjopen-2021-058001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dankiewicz J, Cronberg T, Lilja G, Jakobsen JC, Bělohlávek J, Callaway C, et al. Targeted hypothermia versus targeted Normothermia after out-of-hospital cardiac arrest (TTM2): a randomized clinical trial—rationale and design. Am Heart J. 2019;217:23–31. doi: 10.1016/j.ahj.2019.06.012. [DOI] [PubMed] [Google Scholar]
- 10.Dankiewicz J, Cronberg T, Lilja G, Jakobsen JC, Levin H, Ullén S, et al. Hypothermia versus normothermia after out-of-hospital cardiac arrest. N Engl J Med. 2021;384:2283–2294. doi: 10.1056/NEJMoa2100591. [DOI] [PubMed] [Google Scholar]
- 11.von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370:1453–1457. doi: 10.1016/S0140-6736(07)61602-X. [DOI] [PubMed] [Google Scholar]
- 12.Charlson M, Szatrowski TP, Peterson J, Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47:1245–1251. doi: 10.1016/0895-4356(94)90129-5. [DOI] [PubMed] [Google Scholar]
- 13.Gattinoni L, Tonetti T, Cressoni M, Cadringher P, Herrmann P, Moerer O, et al. Ventilator-related causes of lung injury: the mechanical power. Intensive Care Med. 2016;42:1567–1575. doi: 10.1007/s00134-016-4505-2. [DOI] [PubMed] [Google Scholar]
- 14.Sinha P, Calfee CS, Beitler JR, Soni N, Ho K, Matthay MA, et al. Physiologic analysis and clinical performance of the ventilatory ratio in acute respiratory distress syndrome. Am J Respir Crit Care Med. 2019;199:333–341. doi: 10.1164/rccm.201804-0692OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Royston P, Saurbrei W (2008) Multivariable model-building: a pragmatic approach to regression anaylsis based on fractional polynomials for modelling continuous variables.
- 16.Jann B. Relative distribution analysis in Stata. Stata J [Internet] 2020;21:885–951. [Google Scholar]
- 17.Young PJ, Bailey M, Bellomo R, Bernard S, Bray J, Jakkula P, et al. Conservative or liberal oxygen therapy in adults after cardiac arrest. Resuscitation. 2020;157:15–22. doi: 10.1016/j.resuscitation.2020.09.036. [DOI] [PubMed] [Google Scholar]
- 18.Roberts BW, Kilgannon JH, Chansky ME, Mittal N, Wooden J, Trzeciak S. Association between postresuscitation partial pressure of arterial carbon dioxide and neurological outcome in patients with post–cardiac arrest syndrome. Circulation. 2013;127:2107–2113. doi: 10.1161/CIRCULATIONAHA.112.000168. [DOI] [PubMed] [Google Scholar]
- 19.Palmer E, Post B, Klapaukh R, Marra G, MacCallum NS, Brealey D, et al. The association between supraphysiologic arterial oxygen levels and mortality in critically ill patients. A multicenter observational cohort study. Am J Respir Crit Care Med. 2019;200:1373–1380. doi: 10.1164/rccm.201904-0849OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pilcher J, Weatherall M, Shirtcliffe P, Bellomo R, Young P, Beasley R. The effect of hyperoxia following cardiac arrest: a systematic review and meta-analysis of animal trials. Resuscitation. 2012;83:417–422. doi: 10.1016/j.resuscitation.2011.12.021. [DOI] [PubMed] [Google Scholar]
- 21.Roberts BW, Kilgannon J, Chansky ME, Trzeciak S. Association between initial prescribed minute ventilation and post-resuscitation partial pressure of arterial carbon dioxide in patients with post-cardiac arrest syndrome. Ann Intensive Care. 2014;4:9. doi: 10.1186/2110-5820-4-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tejerina E, Pelosi P, Muriel A, Peñuelas O, Sutherasan Y, Frutos-Vivar F, et al. Association between ventilatory settings and development of acute respiratory distress syndrome in mechanically ventilated patients due to brain injury. J Crit Care. 2017;38:341–345. doi: 10.1016/j.jcrc.2016.11.010. [DOI] [PubMed] [Google Scholar]
- 23.Nolan JP, Sandroni C, Böttiger BW, Cariou A, Cronberg T, Friberg H, et al. European Resuscitation Council and European Society of Intensive Care Medicine guidelines 2021: post-resuscitation care. Intensive Care Med. 2021;47:369–421. doi: 10.1007/s00134-021-06368-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lundbye JB, Rai M, Ramu B, Hosseini-Khalili A, Li D, Slim HB, et al. Therapeutic hypothermia is associated with improved neurologic outcome and survival in cardiac arrest survivors of non-shockable rhythms. Resuscitation. 2012;83:202–207. doi: 10.1016/j.resuscitation.2011.08.005. [DOI] [PubMed] [Google Scholar]
- 25.Nolan JP, Sandroni C, Böttiger BW, Cariou A, Cronberg T, Friberg H, et al. European Resuscitation Council and European Society of Intensive Care Medicine Guidelines 2021: post-resuscitation care. Resuscitation. 2021;161:220–269. doi: 10.1016/j.resuscitation.2021.02.012. [DOI] [PubMed] [Google Scholar]
- 26.Serpa Neto A, Deliberato RO, Johnson AEW, Bos LD, Amorim P, Pereira SM, et al. Mechanical power of ventilation is associated with mortality in critically ill patients: an analysis of patients in two observational cohorts. Intensive Care Med. 2018;44:1914–1922. doi: 10.1007/s00134-018-5375-6. [DOI] [PubMed] [Google Scholar]
- 27.Coppola S, Caccioppola A, Froio S, Formenti P, De Giorgis V, Galanti V, et al. Effect of mechanical power on intensive care mortality in ARDS patients. Crit Care. 2020;24:246. doi: 10.1186/s13054-020-02963-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bellani G, Grassi A, Sosio S, Gatti S, Kavanagh BP, Pesenti A, et al. Driving pressure is associated with outcome during assisted ventilation in acute respiratory distress syndrome. Anesthesiology. 2019;131:594–604. doi: 10.1097/ALN.0000000000002846. [DOI] [PubMed] [Google Scholar]
- 29.Toufen Junior C, De Santis Santiago RR, Hirota AS, Carvalho ARS, Gomes S, Amato MBP, et al. Driving pressure and long-term outcomes in moderate/severe acute respiratory distress syndrome. Ann Intensive Care. 2018;8:119. doi: 10.1186/s13613-018-0469-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Guo L, Xie J, Huang Y, Pan C, Yang Y, Qiu H, et al. Higher PEEP improves outcomes in ARDS patients with clinically objective positive oxygenation response to PEEP: a systematic review and meta-analysis. BMC Anesthesiol. 2018;18:172. doi: 10.1186/s12871-018-0631-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Serpa Neto A, Filho RR, Cherpanath T, Determann R, Dongelmans DA, Paulus F, et al. Associations between positive end-expiratory pressure and outcome of patients without ARDS at onset of ventilation: a systematic review and meta-analysis of randomized controlled trials. Ann Intensive Care. 2016;6:109. doi: 10.1186/s13613-016-0208-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Torres A, Motos A, Riera J, Fernández-Barat L, Ceccato A, Pérez-Arnal R, et al. The evolution of the ventilatory ratio is a prognostic factor in mechanically ventilated COVID-19 ARDS patients. Crit Care. 2021;25:331. doi: 10.1186/s13054-021-03727-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Amato MBP, Barbas CSV, Medeiros DM, Magaldi RB, Schettino GP, Lorenzi-Filho G, et al. Effect of a protective-ventilation strategy on mortality in the acute respiratory distress syndrome. N Engl J Med. 1998;338:347–354. doi: 10.1056/NEJM199802053380602. [DOI] [PubMed] [Google Scholar]
- 34.Brower R, Matthay M, Morris A, Schoenfeld D, Thompson B, Wheeler A. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342:1301–1308. doi: 10.1056/NEJM200005043421801. [DOI] [PubMed] [Google Scholar]
- 35.Simonis FD, Serpa Neto A, Binnekade JM, Braber A, Bruin KCM, Determann RM, et al. Effect of a low vs intermediate tidal volume strategy on ventilator-free days in intensive care unit patients without ARDS. JAMA. 2018;320:1872. doi: 10.1001/jama.2018.14280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tiruvoipati R, Pilcher D, Botha J, Buscher H, Simister R, Bailey M. Association of hypercapnia and hypercapnic acidosis with clinical outcomes in mechanically ventilated patients with cerebral injury. JAMA Neurol. 2018;75:818–826. doi: 10.1001/jamaneurol.2018.0123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Beitler JR, Ghafouri TB, Jinadasa SP, Mueller A, Hsu L, Anderson RJ, et al. Favorable neurocognitive outcome with low tidal volume ventilation after cardiac arrest. Am J Respir Crit Care Med. 2017;195:1198–1206. doi: 10.1164/rccm.201609-1771OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Eastwood GM, Nichol A. Optimal ventilator settings after return of spontaneous circulation. Curr Opin Crit Care. 2020;26:251–258. doi: 10.1097/MCC.0000000000000720. [DOI] [PubMed] [Google Scholar]
- 39.Eastwood GM, Young PJ, Bellomo R. The impact of oxygen and carbon dioxide management on outcome after cardiac arrest. Curr Opin Crit Care. 2014;20:266–272. doi: 10.1097/MCC.0000000000000084. [DOI] [PubMed] [Google Scholar]
- 40.Farias LL, Faffe DS, Xisto DG, Santana MCE, Lassance R, Prota LFM, et al. Positive end-expiratory pressure prevents lung mechanical stress caused by recruitment/derecruitment. J Appl Physiol. 2005;98:53–61. doi: 10.1152/japplphysiol.00118.2004. [DOI] [PubMed] [Google Scholar]
- 41.Ricard J-D, Dreyfuss D, Saumon G. Ventilator-induced lung injury. Eur Respir J. 2003;22:2s–9s. doi: 10.1183/09031936.03.00420103. [DOI] [PubMed] [Google Scholar]
- 42.Schaefer MS, Serpa Neto A, Pelosi P, de Gama AM, Kienbaum P, Schultz MJ, et al. Temporal changes in ventilator settings in patients with uninjured lungs. Anesth Analg. 2019;129:129–140. doi: 10.1213/ANE.0000000000003758. [DOI] [PubMed] [Google Scholar]
- 43.Tejerina EE, Pelosi P, Robba C, Peñuelas O, Muriel A, Barrios D, et al. Evolution over time of ventilatory management and outcome of patients with neurologic disease. Crit Care Med. 2021;49:1095–1106. doi: 10.1097/CCM.0000000000004921. [DOI] [PubMed] [Google Scholar]
- 44.Esteban A. Characteristics and outcomes in adult patients receiving mechanical ventilation: a 28-day international study. JAMA. 2002;287:345. doi: 10.1001/jama.287.3.345. [DOI] [PubMed] [Google Scholar]
- 45.Sahetya SK, Fan E. Driving pressure: the road ahead. Respir Care. 2019;64:1017–1020. doi: 10.4187/respcare.07226. [DOI] [PubMed] [Google Scholar]
- 46.Cressoni M, Gotti M, Chiurazzi C, Massari D, Algieri I, Amini M, et al. Mechanical power and development of ventilator-induced lung injury. Anesthesiology. 2016;124:1100–1108. doi: 10.1097/ALN.0000000000001056. [DOI] [PubMed] [Google Scholar]
- 47.Scharffenberg M, Wittenstein J, Ran X, Zhang Y, Braune A, Theilen R, et al. Mechanical power correlates with lung inflammation assessed by positron-emission tomography in experimental acute lung injury in pigs. Front Physiol. 2021;12:717266. doi: 10.3389/fphys.2021.717266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Malik AB, Krasney JA, Royce GJ. Respiratory influence on the total and regional cerebral blood flow responses to intracranial hypertension. Stroke. 1977;8:243–249. doi: 10.1161/01.str.8.2.243. [DOI] [PubMed] [Google Scholar]
- 49.Heffner JE. Controlled hyperventilation in patients with intracranial hypertension. Arch Intern Med. 1983;143:765. [PubMed] [Google Scholar]
- 50.Nuckton TJ, Alonso JA, Kallet RH, Daniel BM, Pittet J-F, Eisner MD, et al. Pulmonary dead-space fraction as a risk factor for death in the acute respiratory distress syndrome. N Engl J Med. 2002;346:1281–1286. doi: 10.1056/NEJMoa012835. [DOI] [PubMed] [Google Scholar]
- 51.Goligher EC, EL Costa V, Yarnell CJ, Brochard LJ, Stewart TE, Tomlinson G, et al. Effect of lowering Vt on mortality in acute respiratory distress syndrome varies with respiratory system elastance. Am J Respir Crit Care Med. 2021;203:1378–1385. doi: 10.1164/rccm.202009-3536OC. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.